Belter Health Measurement and Analysis Technology EPA46B sphygmomanometer User Manual
Shenzhen Belter Health Measurement and Analysis Technology Co., Ltd. sphygmomanometer
User Manual

Shenzhen Belter Health Measurement and Analysis Technology Co.,Ltd
ePA-46B
sphygmomanometer
user m anua l
shenzhen belter health measurement and analysis technology co.,ltd

Shenzhen Belter Health Measurement and Analysis Technology Co.,Ltd
Tha nk you for purchasing t he BELTER e lect ronic sphygm om anom et e r
This product is a sphygm om anom et er w hich uses noninva sive m et hod t o
m e asure hum a n blood pre ssure .Plea se be sure to re ad t he inst ruct ions
befor e use.Aft er rea ding, please safe keeping for inspect ion a t any tim e,
reference.
Scope of applicat ion: used for blood pressure and pulse m ea sur em ent of
hum an body.
The sphygm om a nom ete r is a ble t o store up t o 1 0 0 m ea sur em ents per
user ( m a xim um 2 user s) an d equipped w ith the Blue toot h Sm a rt
t echnology t o achieve r eliable t ransm ission link age w it h a m obile device
or t ablet PC tha t is w it h t h e logo and corr esponding a pplica tion. Fr om
now on, you ca n ea sily t ra ck your he alth on a daily basis.
Referent Standards and Documents
No Standard Name Reference No.
1 Standard Name Reference No.
2 Medical Device Quality System for the regulation
requirement EN ISO13485: 2012
3 Medical device risk management to medical devices
application EN ISO14971: 2012
4 Symbol for the label of medical devices EN 980: 2008
5 Term, symbol and information of medical device—
information of medical device manufacturer offering
EN 1041: 2008
6 Medical electrical equipment – Part 1: General
requirements for safety
EN60601-1: 2006/AC:2010
7 Medical electrical equipment – Part 1-2: General
requirements for safety – Collateral standard:
Electromagnetic compatibility – Requirements and
tests
EN 60601-1-2: 2007
8 Clinical investigation of medical devices for human
subjects. General requirements
EN ISO 14155-1:2009
9 Clinical investigation of medical devices for human
subjects. Clinical investigation plans
EN ISO 14155-2:2009
10 Medical device software – Software life-cycle
processes
EN 62304: 2006/AC: 2008
11 Medical electrical equipment – Part 1-6: General
requirements for basic safety and essential
performance – Collateral standard: Usability
EN 60601-1-6: 2010
12 Medical electrical equipment. Part 1-11:General
requirements for basic safety and essential
performance. Collateral standard:Requirements for
medical electrical equipment and medical electrical
EN 60601-1-11
Shenzhen Belter Health Measurement and Analysis Technology Co.,Ltd
systems used in the home healthcare environment
13 Medical devices – Application of usability engineering
to medical devices
EN62366: 2008
14 Council Directive 93/42/EECof 14 June 1993
concerning medical devices
OJ L 169 of 12 July 1993 (including Directive
2007/47/EC )
MDD 93/42/EEC
15 Non- invasive sphygm om anom et ers —Part 1:
General requirem ents
EN 1060-1
16 Non- invasive sphygm om anom et ers.Part
3: Supplem entary requirem ents for
elect ro- m echanical blood pressure m easuring
syst em s
EN 1060-3
17 Manual, elect ronic, or autom at ed
sphygm om anom et ers
SP10
18 User Manual Version:1.0
I m port ant safe t y not e s
SI GN S AND SYMBOLS
The following signs appear in this m anual. READ THE SAFETY SECTI ON AND ALL
I NSTRUCTI ONS AND WARNI NGS COMPLETELY AND CAREFULLY BEFORE USI NG
THI S PRODUCT. FOLLOW ALL SAFETY I NSTRUCTI ONS AND WARNI NGS TO AVOI D
HAZARDOUS SI TUATI ONS AND TO MAKE CORRECT USE OF THI S PRODUCT.
sym bol Description
WARNING WARNING indicates a hazardous situation which, if not avoided,
could Result in death or serious injury
CAUTION
CAUTION indicates a hazardous situation which, if not avoided,
may result in minor or moderate injury
NOTICE addresses practices not related to personal injury, such
as product and/or property damage
Type BF
Bluetooth function
Do not dispose this product as unsorted municipal waste.
Collection of such waste separately for special treatment is
DI SPOSAL
NOTICE

Shenzhen Belter Health Measurement and Analysis Technology Co.,Ltd
necessary
This item is compliant with Medical Device Directive 93/42/EEC of
June 14,1993, a directive of the European Economic community
Refer to instruction manual / booklet
IP22
Degree of protection against harmful ingress of particulate
matter and water
Manufacturer
Authorized representative in the European Community
Date of manufacture
Serial number
SYS systolic
DIA diastolic
PUL pulse
w a rning
Do not use t h is product in the m ixed gas of flam m a ble gas w it h air or
w ih oxygen or w it h nit rous oxide conditions.
Do not dispose of ele ct rical appliance s as unsorte d m unicipal w aste ,
use separ a t e collect ion fa cilit ie s. Cont act you loca l governm ent for
infor m at ion regarding the colle ct ion syst em s available. I f ele ct rica l
appliance s a re dispose d of in landfills or dum ps, hazar dous subst a nces ca n
lea k in t o the groundw at er a nd get int o t he food chain , dam a ging your
he alt h a nd w e ll- being.
The device is not suit able for use in the presence of flam m able
ane sthe tic m ix ture s w it h air or w ith ox ygen or nitrous oxide.
The device requires no calibra tion.
The de vice is not repair able and cont ains no user servicea ble part s.
N o m odificat ion of this e quipm e nt is allow e d.
Rem ove batt er ies if e quipm ent is not lik ely to be used for som e tim e .

Shenzhen Belter Health Measurement and Analysis Technology Co.,Ltd
The user m u st check t hat t he equipm e nt funct ions safely and see that it
is in pr oper w or king condit ion be fore being use d.
The m a nufa ct urer does not require such pre ventive inspections by
oth er pe rsons.
. I t is dangerous t o car ry on self j u dgm e nt a nd self t rea t m ent by t he
sufferer him se lf,so please follow doctor’s inst ruct ion. Self j udgm ent m a y
lea d to exa cerbat ion of disease
. Get a serious blood circulat ion obst acle, blood paroxysm sufferer, please use
under the doct or's inst ruct ion, while m easuring because the arm is squeezed m ay
cause im patient internal bleeding
. I nfant and person not be able to express him self,please do not use
. Please control t he add pressue to t he arm band in the range of 300 m m Hgs( 40
kpas) , otherwise the arm m ay appear blood clot, benumbed sym pt om
. This product is only used for hum an body blood pressure m easurem ent and used
out side t he purpose of m easuring hum an body blood pressure, m ay take place
accident
. Please don't use a cell phone in the neighborhood of this product and have the
danger t o m ake product m ist ake act ion
. Please don't dism ant le to unload or fix by yourself, reform ation of t he blood
pressure account of essence or arm band, ot herwise can not m easure correct ly
. Please use AA alkaline batt ery, don't use other bat teries, ot herwise m ay arouse
breakdown
. I f the elect rolyte in t he bat tery get s into your eye, please im m ediately flush with a
great deal of clear water, will result into to blindness or other harm ful danger, need
to im m ediately arrive hospit al nearby t o cure
. I f the elect rolyt e in the bat tery glues on t he skin or the clothes, please
im m ediat ely flush wit h a great deal of clear wat er, otherwise m ay hurt skin
Please use special arm band, ot herwise m ay not be able t o m easure correct ly.
I f t he abnorm al sit uation of adding pressure cannot st op or abnorm al air exhaustion
occurs while m easuring,please rem ove t he arm band or pull out t he air
pipe,ot herwise m ay suffer peripheral nerve disorder.
CAUTION
.Please be sure t o use the appropriate cuffs band, ot herwise you can't get correct
m easurem ent
.Please don't pack a plus or m inus pole of battery to pack anti-. After t he bat tery is
used up, please replace it with new batt ery at t he sam e tim e, long tim e no need t o
use ,please t ake out t he batt ery
. The m et hod to abandon t he bat t ery aft er use please deal with it according t o cit y
environm ental protect ion regulat ions.
Please do not m ix use new and old bat t eries,otherwise m ay cause leakage, fever,
fract ure of the bat tery to dam age the product .
Please use according t o the st orage t em perature and hum idit y st andard recored in
the user m anual.
Shenzhen Belter Health Measurement and Analysis Technology Co.,Ltd
When arrhythm ia situat ion occurs,the m easurem ent value m ay be incorrect or can
not be m easured.
.Please don't m ake an effort to curve arm band and arm band windpipe
.While dism antling an air t ube, please take t he forepart of t he air tube plug t o pull out
.Please don't bum p shot or drop t he product , this m ay lead t o t he m easurem ent
value of t he product incorrect.
Please do not add pressue before t he arm band wraps around t he upper arm ,or you
can not get correct m easurem ent value.
When the plug or wire of the power adapter is dam aged,please im m ediately drawn it
out of the socket,ot herwise m ay cause the danger of fire or elect ric shock.
Please do not use wet hand t o pull the power adapter from t he socket ,ot herwise m ay
cause elect ric shock or fire burn.
Operating instructions:
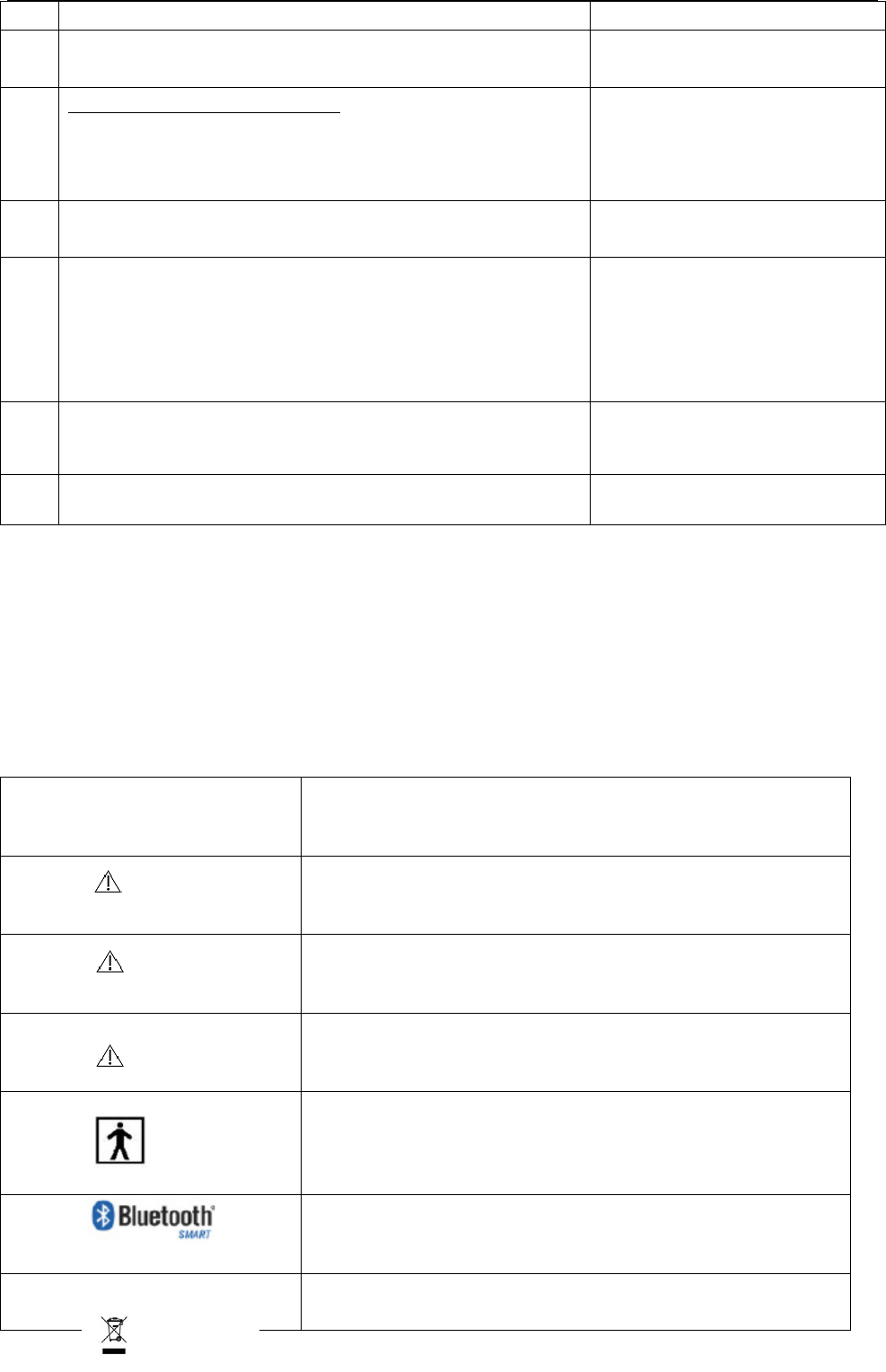
How t o inst all and replace t he bat t e ry
1. Downward push the bat tery cover, rem ove the bat tery cover from ontology
2. I nst all or replace the 4 AA bat teries,Make t he bat tery positive ( + ) and negat ive ( - )
ext rem ely consist ent wit h the m arks in t he battery box,and then close t he batt ery
cover.
Please replace t he bat tery in the st at e of the body cut off power.
I f you rem ove t he batt ery in the boot state, then count t im e to st op.
When the low bat tery icon () appears on the screen please replace 4 batt eries.
Recom m end alkaline bat tery wit h longer service life.
When 3 m ont h or longer tim e wit hout t he use of the blood pressure please t ake out
the batt ery.
After replacing t he bat tery, you m ay need t o reset t he date / tim e.
The m ethod to abandon the bat t ery after use please deal with it according to city
NOTICE
NOTICE
Shenzhen Belter Health Measurement and Analysis Technology Co.,Ltd
environm ental protect ion regulat ions.
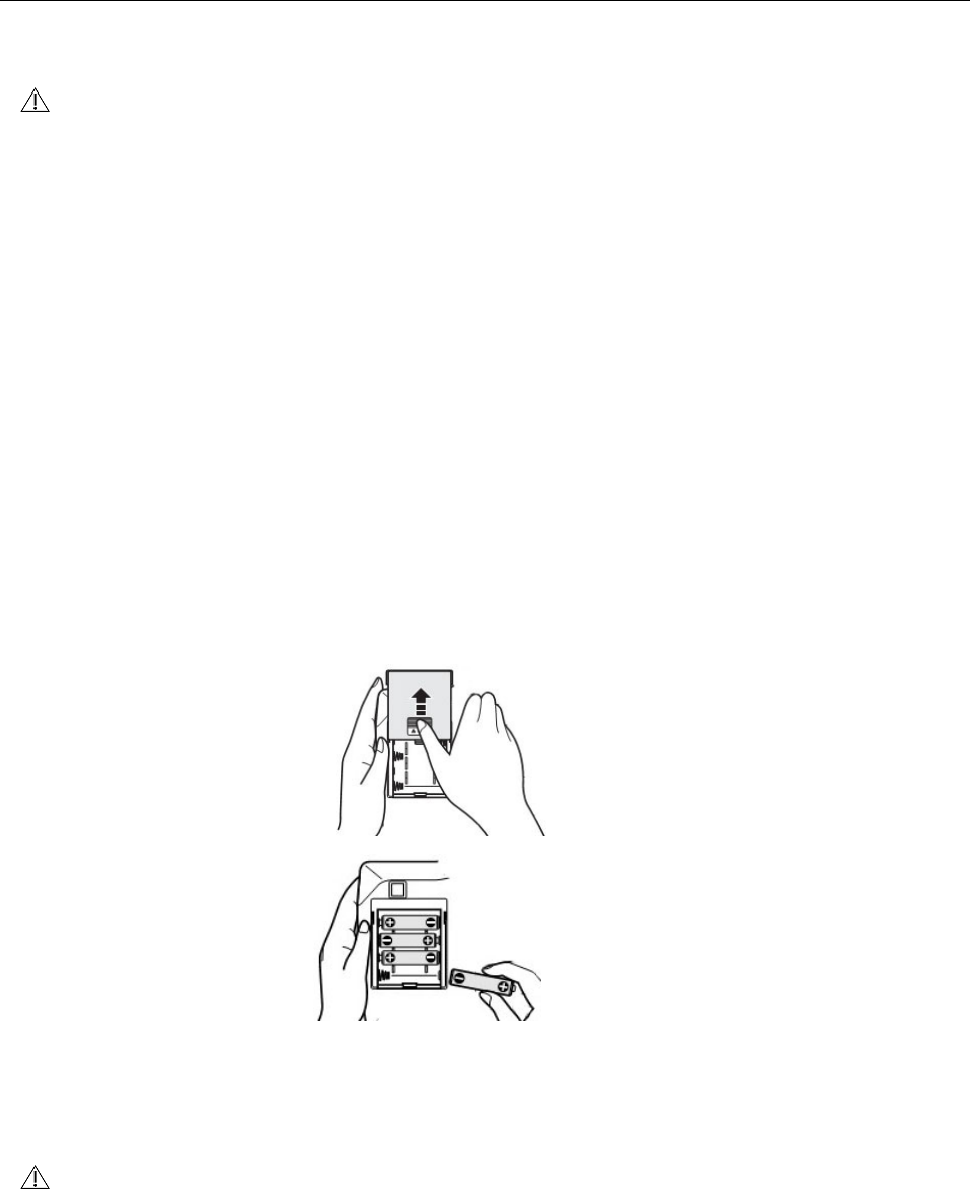
How to sele ct th e adapt er a nd use
Please choose t he output volt age is DC6V, out put current is 1A dedicated to m edical
treat m ent power supply adapter.
Please insert t he plug of the power adapt er into t he power adapter int erface of the
product .
Please insert t he power adapt er into t he socket.
Please do not use nondedicated power adapt er.
Even if you use the power adapter, please st all batt eries into t he product .
The installat ion of bat teries is t o protect t he tim e sett ings.
I f you do not inst all bat t eries, unplug the power adapter from the socket ,t he m achine
will autom at ically st op count ing t im e, t he hist ory dat a will be delet ed.
Se t custom er , dat e and t im e
Please carry on enact m ent according to t he sequence of cust om er, age, m ont h and
date, hour and m inute
Shut down st at us bot tom , double-click SET butt on to get into a cust om er to
choose m ode.
Show a cust om er at t his t im e 1, press MEM button to chose cust om er 2:
Press SET butt on, setage value
Press one tim e[ m em ory] butt on, prom ot e 1.
I t can quickly change dat a while cont inuously pressing down a but ton
NOTICE
power
plug

Shenzhen Belter Health Measurement and Analysis Technology Co.,Ltd
The enactm ent scope of year is 2000-2040.I f the year at tains 2040, it will ret urn
2000
Press SET button, set the m onth be worth
Press one tim e[ MEM] butt on, prom ot e 1.
I t can quickly change dat a while cont inuously pressing down a but ton
Press SETbut ton, set dat e
Press one tim e[ MEM] butt on, prom ot e 1.
I t can quickly change dat a while cont inuously pressing down a butt on
Press SET butt on, sethour& m inute
Press one tim e[ MEM] butt on, prom ot e 1.
I t can quickly change dat a while cont inuously pressing down a but ton
Set unit
At shut down status, press SET but ton, then press MEM butt on t o carry on
the conversion of t he blood pressure unit "m m Hg" and"kPa" .
NOTICE
Shenzhen Belter Health Measurement and Analysis Technology Co.,Ltd
The use of m e m ory funct ion
Press MEM butt on, t he device shows user's group.Press SETbutt on again, it
shows the latest diagraph result .(Press SETbutton up to change user)
During the process of reading m easuring m em ory,
When long press MEM but ton, all the m em ory value will be delet ed.
I f show "-- -", m eans delet e finished.
This blood pressure is autom at ically save t est value and pulse wit h m easurem ent
date and tim e, 60 sets of diagraphs are kept. I f saved dat a is over than 60 sets, t he
oldest diagraph result will be delet ed, newest one will be saved.
Using st eps
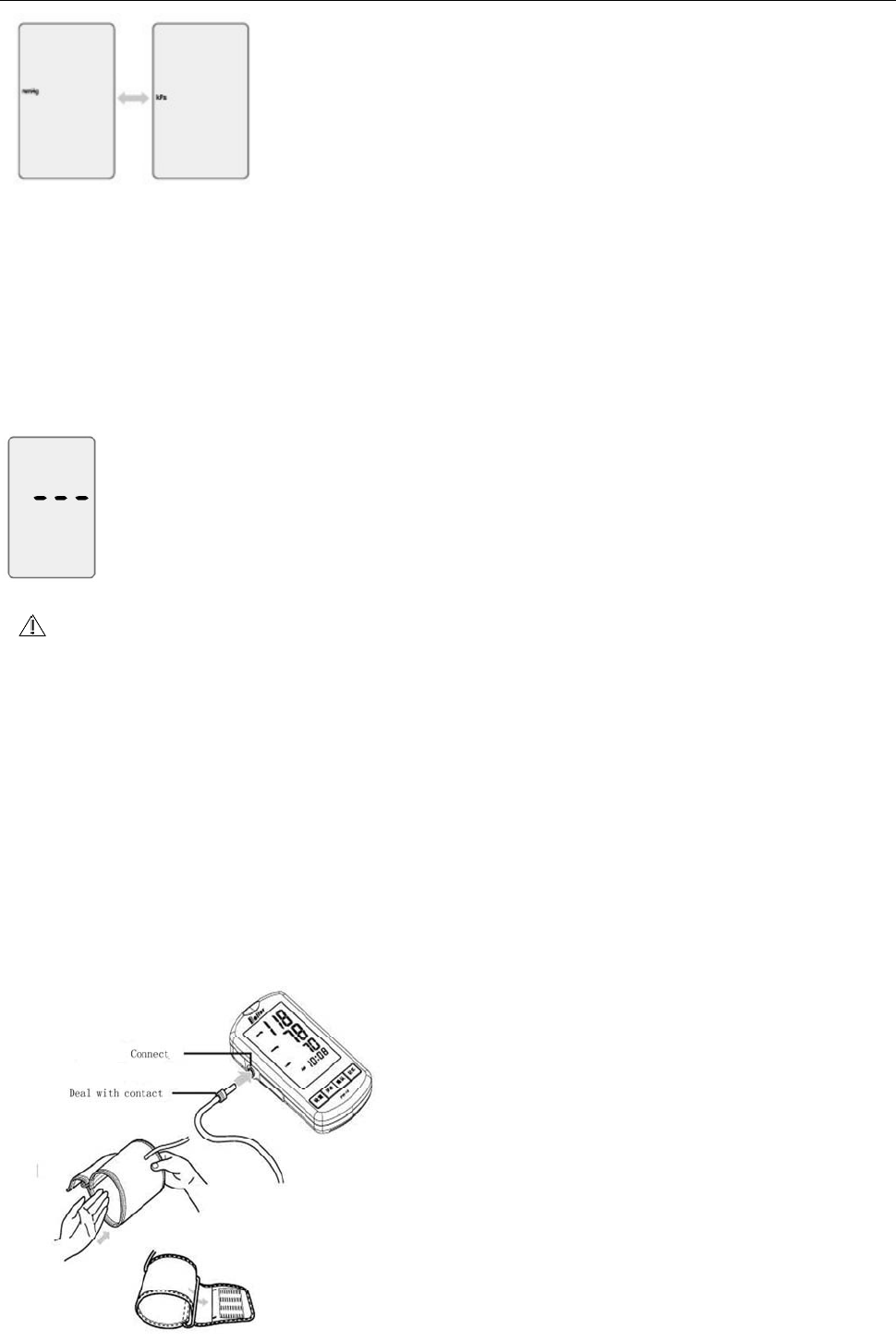
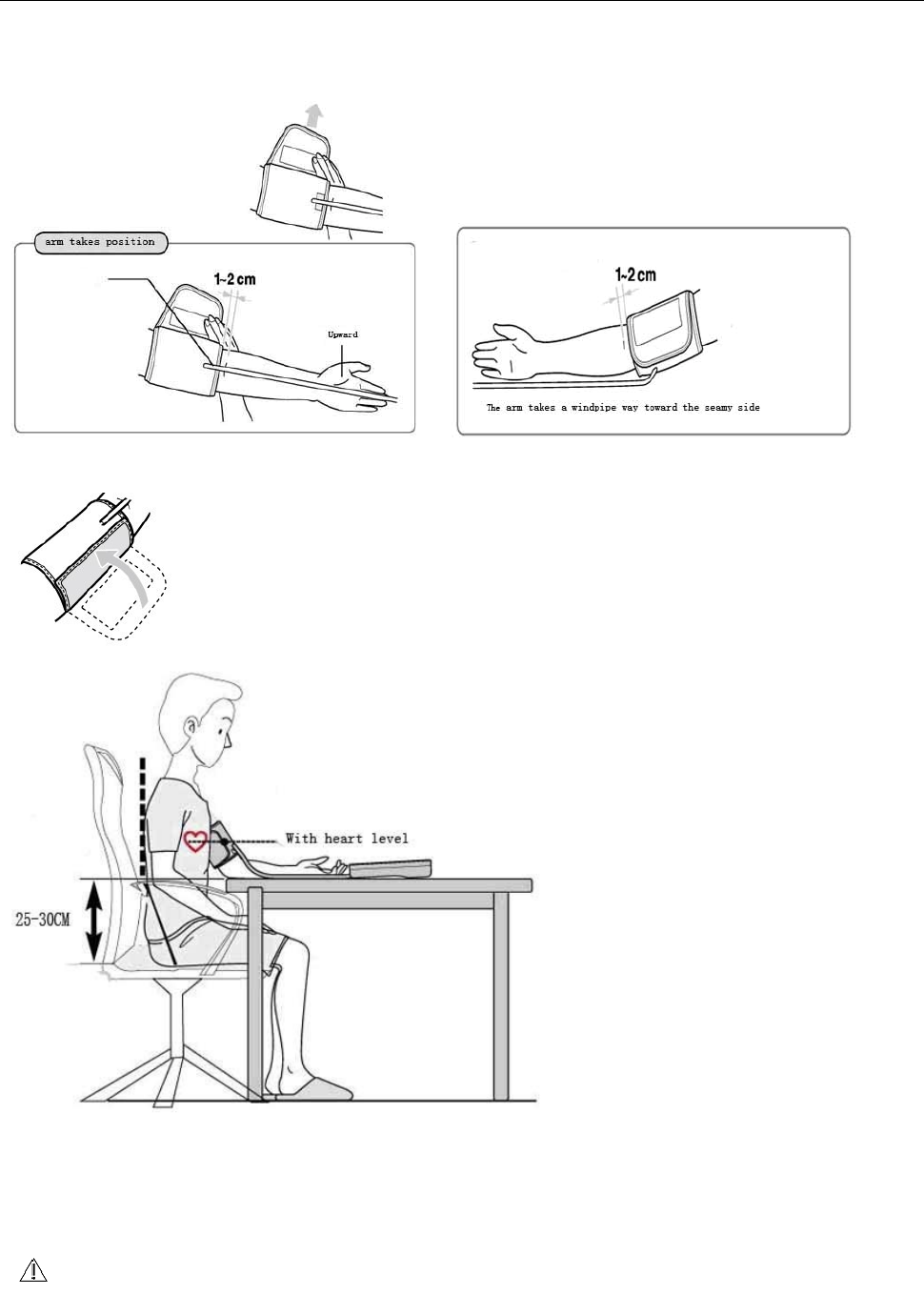
1、 Tie up arm t o t ake
A、plug the air tube of t he arm band into the joint of the product.
B、open the arm band,put arm band air t ube toward the palm inside.(as below
pict ures)
NOTICE
Shenzhen Belter Health Measurement and Analysis Technology Co.,Ltd
C, fix the armband in position, as position shown in below pictures .
C、fix t he arm band t ight ly.
Correct diagraph post ure
I f you use it out of the indicated t em perat ure and hum idit y range,it m ay not be able
t o achieve claim ed perform ance requirem ent.
While m easuring you m ust ensure t he correct post ure t o obtain accurat e results.
NOTICE
Shenzhen Belter Health Measurement and Analysis Technology Co.,Ltd
Even forget to t urn off t he power ,after about 2 m inut es it will shut down
aut om at ically .
Please m easure and control blood pressure at the sam e tim e every day.
Measurem ent should be taken in quiet places ,the m easured person should be in a
relaxed sit ting posit ion.
You'd bet ter have a rest of 15 m inutes before m easurem ent.
dat a transfe r
The dat a uploads t o cellular phone or com puter via blue t oot h.
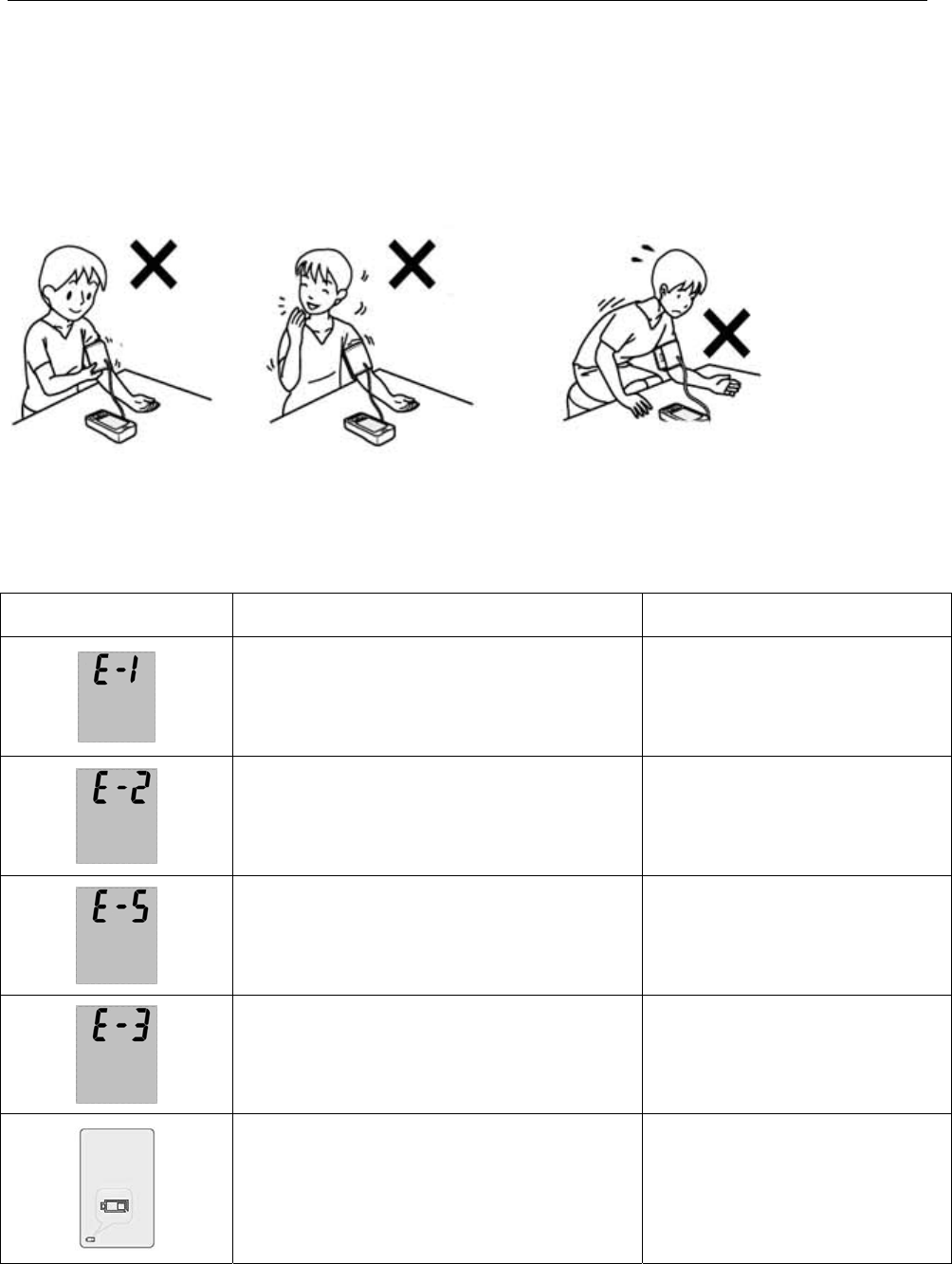
Key of sym bols
signal W ar ning- see use r m a nual Tr e at m ent
The arm band fix t oo loose Check whet her arm band
is loose t o m ove, re-
fast en t ight again for
m easuring.
Speaking during m easurem ent or
m ove the arm
Keep body and arm st able
and m easure again
There's interference or work not
correct ly during m easurem ent .
Shut down, re- m easure
once.
The product leaks air or arm 's
taking don't put good enough.
Please check arm t o t ake
whether already put like.
Bat tery energy shortage Please replace bat tery
Ex cr esce nt check
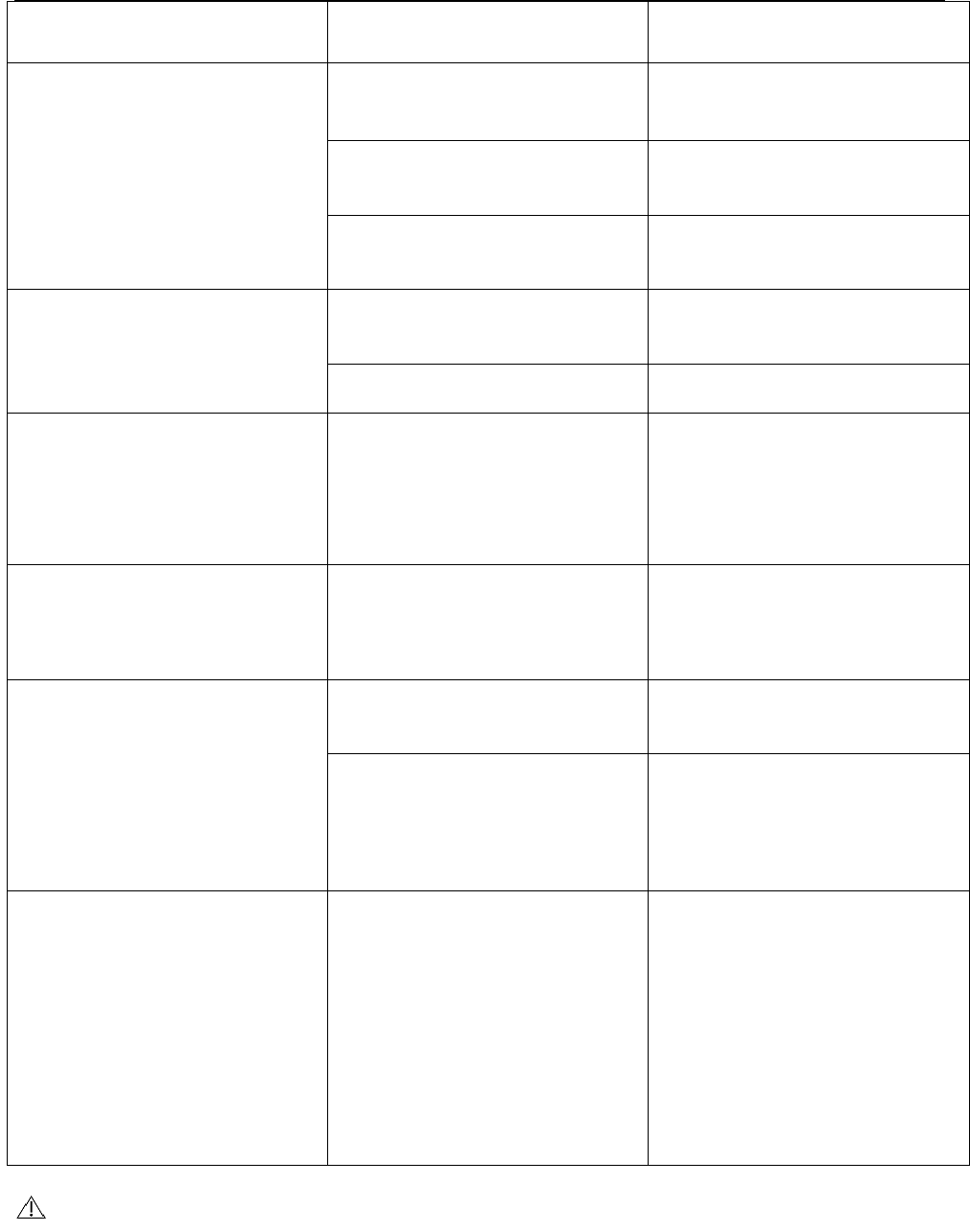
Shenzhen Belter Health Measurement and Analysis Technology Co.,Ltd
Ex cre sce nt
cir cu m st a nce
Rea son
Tr e at m ent
Measured result is big
deviation from norm al
Arm band doesn't t ake
correct ly
The exact itude t ies up good
arm to t ake
t alks or m oves arm during
the m easuring Keep quiet and m easure
The clot hes of curling up
oppressed arm
take off t he clot hes and
m easure again
Don't add to press
Air plug do not plug in the
ontology of air tube correct Connect t he t ube correct ly
The arm t akes to leak air Purchase new arm to take.
The arm t akes in t he in a
short t im e to carry on
exhaust ion
The arm takes t o t ie up too
loosely
Please t ie up t ight arm to
take
Add t o ran over dist ance in
suddenly t he power failure
reset clock
Bat tery energy exhaust ion
Replace new batt ery
After pressing butt on,
don't show
Bat tery energy exhaust ion
Replace new batt ery
The power get s in touch
with bad or pack anti-
The exact it ude put s into
batt ery
Ot her phenom ena
Show the appearance
trem bles
Can not m easure
Please press ON/ OFF
button, re-operat e 1 t im e.
Please replace bat tery t o
try.
I n addition st ill can not
m easure, please consult
room contact wit h BELTER
healt hily
This product has ca r ried on ch e ck in t he fa ctor y,please ex ecut e strict ly in
accordance w it h t he in struct ion m a nual.
funct ion spe cification
NOTICE

Shenzhen Belter Health Measurement and Analysis Technology Co.,Ltd
Nam e Sphygmomanometer
Model num ber ePA- 4 6B
Display m ode Display m ode
Bluetoot h Support ed Bluet oot h V2.0
Measurem ent scope The pressure m easurem ent scope: 0-280
m m Hg(0-37.3 kPa)
Pulse scope: 40- 200 Tim e/ m inut e
Accuracy Pressure: ± 3 m m Hg
Pulse accuracy : ± 5%
Power DC6v/ ” AA* 4”
Weight 196g
Shape size W74 m m * L152 m m * H33 m m
Arm band size: W154 m m * L 486 m m ,air t ube to grow
600 m m Apply arm t he scope22cm - 36cm
operating environment
a) temperature : +5℃-+40℃
a) relative humidity:15%-80%
b) atmospheric pressure :700hpa-1060hpa
Transportation and storage environment a)temperature :-20℃-+50℃
b)relative humidity:15%-90%
c)atmospheric pressure :500hpa-1060hpa
Protection against electric shock internal electric source,.BF application
part
clinical allow maximum error margin This product pass clinical verification, the clinical
maximum allowable error is ≤±5mmHg/±0.67kPa
Bat tery service life 300times(alkaline dry bat t ery can use )
product expects life span 2years
CLASSIFICATION
1. Internally powered equipment;
2. Type BF applied part: cuff;
3. IP22;
4. Not category AP / APG equipment;
5. Mode of operation: continuous operation.
Cleaning, maintenance and Storage
Please always clean sphygmomanometer.
Please use clean soft cloth ,gently wipe the LCD and shell, do not use solvents, and do
not be soak it in water or other liquids.
Please do not wash the arm band of the product.
If the product is especially dirty you can use a soft cloth with water or neutral detergent,
fully wring dry to wipe the product.
Please do not use gasoline, thinner or similar solvent to clean arm band.
When you do not use, please storage the sphygmomanometer and accessories in the
collection bag.

Shenzhen Belter Health Measurement and Analysis Technology Co.,Ltd
Please do not place the sphygmomanometer and arm band in high temperature, damp,
full of water vapor or direct sunlight conditions.
Please do not let the blood pressure cuff suffer strong shock or vibration (e.g., fell on the
ground).
Please do not put the blood pressure at easy splashing places.
Please don't put this product where there is chemical or corrosive gas.
Please do not dismantle the blood pressure.
Please storage the product in requied environment.
Calibration and repair
This product has been calibrated and tested when delivered from the factory.
If you have doubt about the measurement result of the product ,
please contact us and give the product to specialized person to repair
Warranty:
If you use the device according to the manual, replace it within 3 months and guarantee
for free a year.
The following situations are not included in warranty time:
1 problem caused by self dismounting
2 problem caused by shocking during use or delivery.
3 problem caused by incorrect maintenance
4 problem caused by incorrect operating, problems caused by unprofessional repair.
Product Composition
A piece of the product
Cuff band and air pipe linemodel:BEARM-011PCS
Probation dry cell4 AA batteries
Collect soft band 1 piece
A copy of instruction for use
Adverse events :non
ShenZhen Belter Health Measurement and Analysis Technology Co.,ltd
702/704, Block C, Tsinghua Unis Science Park, No.13 Langshan Rd, Hi-Tech Industrial
Park(north), Nanshan District, 518057 Shenzhen, People’s Republic of China

Shenzhen Belter Health Measurement and Analysis Technology Co.,Ltd
2012-09
FCC Statement
This equipment has been tested and found to comply with the limits for a Class B digital device,
pursuant to part 15 of FCC Rules. These limits are designed to provide reasonable protection
against harmful interference in a residential installation. This equipment generates and can radiate
radio frequency energy and, if not installed and used in accordance with the instructions, may cause
harmful interference to radio communications. However, there is no guarantee that interference will
not occur in a particular installation. If this equipment does cause harmful interference to radio or
television reception, which can be determined by turning the equipment off and on, the user is
encouraged to try to correct
The interference by one or more of the following measures:
Reorient or relocate the receiving antenna.
Increase the separation between the equipment and receiver.
Connect the equipment into an outlet on a circuit different from that to which the receiver is
connected.
Consult the dealer or an experienced radio/TV technician for help.
This device complies with Part 15 of FCC Rules.
Operation is subject to the following two conditions:
This device may not cause harmful interference, and This device must accept any interference
received, including interference that may cause undesired operation.
Note: The manufacturer is not responsible for any radio or TV interference caused by unauthorized
modifications to this equipment. such modifications could void the user’s authority to operate this
equipment.