Carestream Health RVG00002 WiFi and RFID Control Box User Manual SM737 RegulatoryGd
Carestream Health, Inc. WiFi and RFID Control Box SM737 RegulatoryGd
Contents
- 1. Manual - Installation
- 2. Manual - Regulatory
Manual - Regulatory

KODAK RVG 6500 System and
KODAK RVG 6500 IPS System
Safety, Regulatory & Technical
Specification User Guide
Notice
The Regulatory Information & Technical Specifications User Guide for the KODAK
RVG 6500 System and KODAK RVG 6500 IPS System includes information on the
safety instructions, regulatory information and the technical specifications of the
device. We recommend that you thoroughly familiarize yourself with this Guide in
order to make the most effective use of your system.
The information contained in this Guide may be subject to modification without
notice, justification or notification to the persons concerned.
No part of this Guide may be reproduced without the express permission of
Carestream Health, Inc.
U.S. Federal law restricts this device to sale by or on the order of a dentist or
physician.
This document is originally written in English.
Manual Name: KODAK RVG 6500 System and KODAK RVG 6500 IPS System
Safety, Regulatory and Technical Specifications User Guide
Part Number: SM737
Revision Number: 02
Print Date: 06/2010
The Brand names and logos reproduced in this Guide are copyright
KODAK is a trademark of KODAK used under Licence.
KODAK RVG 6500 System, complies with Directive 93/42/CEE relating to medical
equipment.
Manufacturer
Authorized Representative in the European Community
TROPHY
4, Rue F. Pelloutier, Croissy-Beaubourg
77435 Marne la Vallée Cedex 2, France
0086
Carestream Health, Inc.
150 Verona Street
Rochester NY 14 608
EC REP

KODAK RVG 6500 System_Regulatory User Guide (SM737)_Ed02 iii
Contents
Chapter 1
Safety Information
Indications for Use . . . . . . . . 1
Conventions in this Guide . . . . . 1
Warning and Safety Instructions . . 2
Hygiene and Disinfection . . . . . 5
Marking and Labeling Symbols . . 6
Label Locations . . . . . . . . . . 7
KODAK RVG 6500 With or Without
IPS System Battery Specification . 8
Chapter 2
Regulatory
Information
General Regulatory Information . . 9
Compliance with International
Regulations . . . . . . . . . . . .16
FCC Requirements for USA . . . .16
IC Requirements for Canada. . . .17
Declaration of Conformity with the
R-TTE Directives . . . . . . . . .18
Chapter 3
Technic al
Specifications
Factory . . . . . . . . . . . . . .19
Manufacturer: . . . . . . . . . . .19
Model. . . . . . . . . . . . . . .19
KODAK RVG 6500 With or Without
IPS System Technical
Specifications . . . . . . . . . . .19
Minimum Computer System
Requirement . . . . . . . . . . .21
RFID Technical Specification . . .22
WiFi Network Technical
Specification . . . . . . . . . . .22
KODAK RVG 6500 With or Without
IPS System Environmental
Requirements . . . . . . . . . . .23

iv

KODAK RVG 6500 System_Regulatory User Guide (SM737)_Ed02 1
1 Safety Information
Indications for Use
The KODAK RVG 6500 System, wireless digital intra-oral X-ray system, is
intended to produce an image of the dental area at the direction of health care
professionals of dento- maxillo-facial region of the human anatomy.
The KODAK RVG 6500 IPS System, in addition, provides the Intelligent
Positioning System to enable the dentist prior to acquisition to correctly align
the X-ray beam to the RVG sensor.
Conventions in this Guide
The KODAK RVG 6500 Systems is composed of the KODAK RVG 6500
System and the KODAK RVG 6500 Intelligent Positioning System (IPS)
System.
The following special messages emphasize information or indicate potential risk
to personnel or equipment.
WARNING: Warns you to avoid injury to
yourself or others by following the safety
instructions precisely.
Important: Alerts you to a condition that might
cause problems.
Tip: Provides extra information and hints.

2 Chapter 1 Safety Information
Warning and Safety Instructions
WARNINGS
Device:
•Read and understand this Safety Information before using the
KODAK RVG 6500 with or without IPS System.
•This device complies with FCC and Industry Canada RF radiation
exposure limits set forth for general population (uncontrolled
exposure). This device must be installed to provide a separation
distance of at least 20cm from all persons and must not be
collocated or operating in conjunction with any other antenna or
transmitter.
•You are responsible for the operation and maintenance of this
device. Only legally qualified persons can operate this device.
When necessary, have a trained authorized service technician
carry out inspection and maintenance operations.
•Install this device in an X-ray room that complies with current
installation standards. From this location, you must be able to
maintain visual or audio communication with the patient and be
able to access the Acquisition interface module during exposure.
•DO NOT operate the device if there is the threat of an
earthquake. Following an earthquake, ensure that the device is
operating satisfactorily before using it again. Failure to observe
this precaution may expose patients to hazards.
•X-ray equipment is hazardous to patients and the operator if you
do not observe the exposure safety factors and operating
instructions.
•DO NOT place objects within the field of operation of the device.
•Connect this equipment ONLY to a mains power supply with
protective ground to avoid any risk of electric shock.
•To dispose of the device or its components, contact a service
technician.
•No modification of this equipment is allowed.
Note: Emphasizes important information.

KODAK RVG 6500 System_Regulatory User Guide (SM737)_Ed02 3
•DO NOT use this device in conjunction with oxygen-rich
environments. This device is not intended for use with flammable
anesthetics or flammable agents.
•Using accessories other than those specified in this document
with the exception of those sold by Carestream Health may
result in a lower level of security for the entire system.

4 Chapter 1 Safety Information
Computer:
•DO NOT place the computer and the peripheral equipment
connected to it in the immediate vicinity of the patient in the unit.
Leave at least 1.5 m distance between the patient and the unit.
The computer and the peripheral equipment must conform to
the IEC60950 standard.
•See your computer installation guide for details of the data
processing system and screen. Leave a sufficient amount of
clear space around the CPU to ensure that it is properly
ventilated.
•To obtain maximum image quality and visual comfort, position
the screen to avoid direct light reflections from internal or
external lighting.
•The computer and its screen should ideally be situated in or
close to the operating area, in the visual field of the practitioner
when he is with the patient. The visual access of the acquired
image for the patient encourages communication.
•The KODAK RVG 6500 systems must run on a computer with a
keyboard.
•The KODAK RVG 6500 systems must run on a computer with a
mouse equipped with a right and left button and a mouse wheel.
Control Box and IPS Battery:
•To assure proper operation, use only the KODAK RVG 6500
System battery.
•Keep the battery out of the reach of children.
•Do not leave the battery without surveillance it could be
swallowed by a child or animal. In this case call urgently the
doctor.
•Damaged or oxidized battery can burn the skin if in contact with
the skin.
•Do not heat nor dispose of the battery in the fire. It can burst or
release toxic chemicals.
•Do not short circuit the battery.
•Do not disassemble, apply excessive pressure or deform the
battery.
•Avoid placing the battery in reverse polarity.
•Battery disposable method must be in accordance with local and
state regulations.
•Remove the batteries if not used for a long period of time.

KODAK RVG 6500 System_Regulatory User Guide (SM737)_Ed02 5
Hygiene and Disinfection
WARNINGS
•Never place the sensor and/or control box in an autoclave as this
could result in serious damage to the sensor.
•Never immerse the RVG sensor control box in any solution.
•Disinfect the sensor head after each patient.
•Do not use chemical autoclave for the toothbrush holders and
avoid direct contact with the metallic part of the autoclave.
•To prevent cross-contamination, use a new hygienic barrier for
each new patient.
6 Chapter 1 Safety Information
Marking and Labeling Symbols
Type BF device symbol complying with the
IEC 60601-1 standard
In the EEC, this symbol indicates: DO
NOT discard this product in a trash
receptacle; use an appropriate recovery and
recycling facility.
Contact your local sales representative for
additional information on the collection
and recovery programs available for this
product
Warning: General warning sign
The ON/OFF button
General mandatory action sign
Follow operating instructions sign
Non-ionizing radiation
Manufactured Date
Manufacturer’s address
KODAK RVG 6500 System_Regulatory User Guide (SM737)_Ed02 7
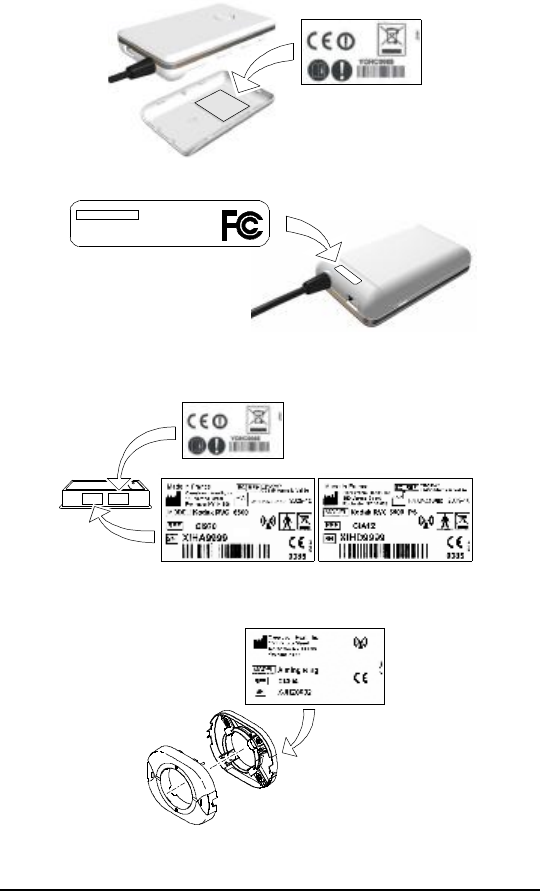
Label Locations
The following Figures illustrate the label locations of the KODAK RVG 6500
Systems.
Figure 1 KODAK RVG 6500 Control Box Label
Figure 2 KODAK RVG 6500 Labels
Figure 3 KODAK RVG 6500 IPS Aiming Ring Label
Model Number: Kodak RVG6500
FCC ID : U72RVG00002
IC : 7027A-RVG00002
**IPS

8 Chapter 1 Safety Information
KODAK RVG 6500 With or Without IPS System Battery
Specification
The KODAK RVG 6500 with or without IPS System provides a medical battery
charge. You can purchase additional battery separately (for battery replacement,
see Chapter “Maintenance” section “Replacing the RVG Battery”).
•A battery is required for wireless use.
•Minimum battery life is 500 charge and discharge cycles where the
cell capacity remains above 80% of initial capacity.
•New battery provides approximately 90 image acquisitions (3.0 hrs
heavy usage).
•A battery charge state is indicated on the Acquisition Interface. The
detector determines if the installed battery is not properly charging,
and provides this battery status to the Acquisition Interface.
Important: To ass ure pro pe r operation, use only
the KODAK RVG 6500 with or without IPS System
battery.

KODAK RVG 6500 System_Regulatory User Guide (SM737)_Ed02 9
2 Regulatory
Information
General Regulatory Information
Compliance with European and International Standards
EN 60601-1 / IEC
60601-1
Medical Electrical Equipment, Part 1 : General
requirements for basic safety and essential
performance
EN 60601-1-2 / IEC
60601-1-2
Medical Electrical Equipment, Part 1-2 :General
requirements for basic safety and essential
performance - Collateral Standard : Electromagnetic
Compatibility
EN 60601-1-3 / IEC
60601-1-3
Medical Electrical Equipment, Part 1-3 : General
requirements for basic safety and essential
performance - Collateral Standard : Radiation
protection in diagnostic X-ray equipment
EN 60601-1-6 / IEC
60601-1-6
Medical Electrical Equipment, Part 1-6 : General
requirements for basic safety and essential
performance - Collateral Standard :Usability
EN ISO 14971 Medical devices - Application of risk management to
medical devices
EN 980 Symbols for use in the labeling of medical devices
EN 1041 Information supplied by the manufacturer of medical
devices
EN 62304/IEC 62304 Medical device software - Software life cycle
processes
EN 10993-1 Biological evaluation of medical devices - Part 1 :
Evaluation and testing
EN 300 328 Electromagnetic Compatibility and Radio Spectrum
Matters

10 Chapter 2 Regulatory Information
EN 302 291-2
Electromagnetic compatibility and Radio spectrum
Matters (ERM); Short Range Devices (SRD); Close
Range Inductive Data Communication equipment
operating at 13,56 MHz; Part 2: Harmonized EN
under article 3.2 of the R&TTE Directive
EN 301 489-1
Electromagnetic compatibility and Radio spectrum
Matters (ERM); ElectroMagnetic Compatibility (EMC)
standard for radio equipment and services; Part 1:
Common technical requirement
EN 301 489-3
Electromagnetic compatibility and Radio spectrum
Matters (ERM); ElectroMagnetic Compatibility (EMC)
standard for radio equipment and services; Part 3:
Specific conditions for Short-Range Devices (SRD)
operating on frequencies between 9kHz to 40GHz
EN 301 489-17
ElectroMagnetic Compatibility and Radio Spectrum
Matters (ERM); ElectroMagnetic Compatibility (EMC)
Standard for Radio Equipment and Services; Part 17 :
Specific Conditions for 2,4 GHz Wide band
Transmission Systems and 5 GHz High Performance
RLAN Equipment
EN 50364
Limitation of human exposure to electromagnetic fields
from devices operating in the frequency range 0Hz to
300GHz, used in electronic article surveillance (EAS),
radio frequency identification (RFID) and similar
applications
EN 50371
Generic Standard to demonstrate the compliance of
low power electronic and electrical apparatus with the
basic restrictions related to human exposure to
electromagnetic fields (10 MHz - 300 MHz). General
Public
Classification in Accordance with EN/IEC 60601-1
Type of protection against
electric shock Class 1 equipment
Degree of protection
against electric shock Typ e B F
Compliance with European and International Standards

KODAK RVG 6500 System_Regulatory User Guide (SM737)_Ed02
11
•Medical electrical equipment requires special precautions regarding
electromagnetic compatibility (EMC).
•KODAK RVG 6500 with or without IPS System must be installed and
put into service according to the EMC information provided in this
document.
•Communication Equipment: Portable and mobile Radio Frequency
(RF) communications equipment can affect the Electromagnetic
Compatibility of KODAK RVG 6500 with or without IPS System.
•KODAK RVG 6500 with or without IPS System may be interfered
with other equipment even if that other equipment complies with
CISPR emission requirements.
Protection against harmful
ingress of water Ordinary equipment
Operation mode Continuous operation
Flammable anesthetics Not suitable for use in presence of flammable
anesthetics or a mixture of flammable anesthetics
with air or oxygen or nitrous oxide
Conformity with EN/IEC 60601-1-3
Attenuation equivalent for the
enclosure of the image receiver
unit (at 100 kV)
0.2 mm eq. AI
KODAK RVG 6500 With or Without IPS System Components
RVG sensor and the control box
Medical power supply Model: MW172KB0500F02
Mains outlet cable: 2.50 meter
WiFi access point WGR614
IPS aiming ring
Classification in Accordance with EN/IEC 60601-1
12 Chapter 2 Regulatory Information
•Use limitation: the use of accessories, cables, or transducers other
than those specified in this document with the exception of cables,
accessories or transducers sold by Carestrem health as replacement
parts of internal components, may result in increased emissions or
decreased immunity of the KODAK RVG 6500 with or without IPS
System.
•The KODAK RVG 6500 with or without IPS System should not be
used adjacent to or stacked with other equipment. If adjacent or
stacked use is necessary, the KODAK RVG 6500 with or without IPS
System should be observed to verify normal operation in the
configuration in which it will be used.
Guidance and Manufacturer’s Declaration - Electromagnetic Emissions
(IEC 60601-1-2)
The KODAK RVG 6500 with or without IPS System is intended for use in the
electromagnetic environment specified below. The customer or the user of the KODAK
RVG 6500 with or without IPS System should assure that it is used in such an
environment.
Emissions Test Compli-a
nce Electromagnetic Environment - Guidance
RF emissions
CISPR 11 Group 1
The KODAK RVG 6500 with or without IPS System uses RF
energy only for its internal function. Therefore, its RF
emissions are very low and are not likely to cause any
interference in nearby electronic equipment.
RF emissions
CISPR 11 Class B
The KODAK RVG 6500 with or without IPS System is
suitable for use in all establishments, including domestic
establishments and those directly connected to the public
low-voltage power supply network that supplies buildings
used for domestic purposes.
Harmonic
emissions
IEC 61000-3-2
Class A
Voltage
fluctuations/
flicker emissions
IEC 61000-3-3
Complies

KODAK RVG 6500 System_Regulatory User Guide (SM737)_Ed02
13
Guidance and Manufacturer’s Declaration - Electromagnetic Immunity
The KODAK RVG 6500 with or without IPS System is intended for use in the
electromagnetic environment specified below. The customer or the user of the KODAK
RVG 6500 with or without IPS System should assure that it is used in such an
environment.
Immunity Test IEC 60601
Test Level Compliance Electromagnetic Environment -
Guidance
Electrostatic
discharge (ESD)
IEC 61000-4-2
±6 kV contact
±8 kV air
±6 kV contact
±8 kV air
Floors should be wood, concrete or
ceramic tile. If floors are covered with
synthetic material, the relative humidity
should be at least 30%.
Electrical fast
transient/burst
IEC 61000-4-4
±2 kV for
power supply
lines
±1 kV for
input/output
lines
±2 kV for
power supply
lines
±1 kV for
input/output
lines
Mains power quality should be that of a
typical commercial or hospital
environment.
Surge
IEC 61000-4-5
±1 kV line(s) to
line(s)
±2 kV line(s) to
earth
±1 kV line(s)
to line(s)
Not applicable
Mains power quality should be that of a
typical commercial or hospital
environment.
Voltage dips,
short
interruptions and
voltage
variations on
power supply
input lines
IEC 61000-4-11
<5% UT
(>95% dip in
UT)
for 0,5 cycle
40% UT
(60% dip in UT)
for 5 cycles
70% UT
(30% dip in UT)
for 25 cycles
<5% UT
(>95% dip in
UT)
for 5 sec
<5% UT
(>95% dip in
UT)
for 0,5 cycle
40% UT
(60% dip in
UT)
for 5 cycles
70% UT
(30% dip in
UT)
for 25 cycles
<5% UT
(>95% dip in
UT)
for 5 sec
Mains power quality should be that of a
typical commercial or hospital
environment. If the user of the KODAK
RVG 6500 with or without IPS System
requires continued operation during
power mains interruptions, it is
recommended that the KODAK RVG
6500 with or without IPS System be
powered from an uninterruptible power
supply or a battery.
Power frequency
(50/60 Hz)
magnetic field
IEC 61000-4-8
3 A/m 3 A/m
Power frequency magnetic fields
should be at levels characteristic of a
typical location in a typical commercial
or hospital environment
NOTE: UT is the a.c. mains voltage prior to application of the test level.
14 Chapter 2 Regulatory Information
Guidance and Manufacturer’s Declaration - Electromagnetic Immunity
(IEC 60601-1-2)
The KODAK RVG 6500 with or without IPS System is intended for use in the
electromagnetic environment specified below. The customer or the user of the KODAK
RVG 6500 with or without IPS System should assure that it is used in such an
environment.
Immunity
Test
IEC 60601
Test Level
Compli
ance
Level
Electromagnetic Environment - Guidance
Conducte
d RF
IEC
61000-4-6
Radiated
RF
IEC
61000-4-3
3 Vrms
150 kHz to
80 MHz
3 V/m
80 MHz to
2,5 GHz
[V1]= 3
V
[E1]= 3
V/m
Portable and mobile RF communications equipment
should be used no closer to any part of the KODAK
RVG 6500 with or without IPS System, including
cables, than the recommended separation distance
calculated from the equation applicable to the
frequency of the transmitter.
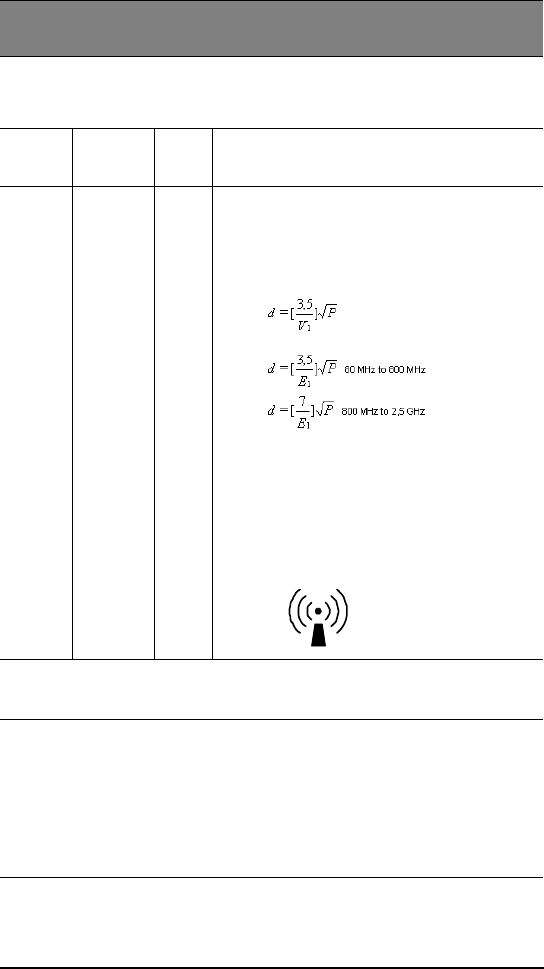
Recommended separation distance
where P is the maximum output power rating of the
transmitter in watts (W) according to the transmitter
manufacturer and d is the recommended separation
distance in metres (m).
Field strengths from fixed RF transmitters, as
determined by an electromagnetic site survey should
be less than the compliance level in each frequency
range.b
Interference may occur in the vicinity of equipment
marked with the following symbol:
NOTE 1: At 80 MHz and 800 MHz, the higher frequency range applies.
NOTE 2: These guidelines may not apply in all situations. Electromagnetic propagation is
affected by absorption and reflection from structures, objects and people.
a Field strengths from fixed transmitters, such as base stations for radio (cellular/cordless)
telephones and land mobile radios, amateur radio, AM and FM radio broadcast and TV
broadcast cannot be predicted theoretically with accuracy. To assess the electromagnetic
environment due to fixed RF transmitters, an electromagnetic site survey should be
considered. If the measured field strength in the location in which the KODAK RVG 6500
with or without IPS System is used exceeds the applicable RF compliance level above, the
KODAK RVG 6500 with or without IPS System should be observed to verify normal
operation. If abnormal performance is observed, additional measures may be necessary,
such as re-orienting or relocating the KODAK RVG 6500 with or without IPS System.
b Over the frequency range 150 kHz to 80 MHz, field strengths should be less than 3 V/m.

KODAK RVG 6500 System_Regulatory User Guide (SM737)_Ed02
15
Recommended Separation Distances between Portable and Mobile RF
Communications Equipment and the KODAK RVG 6500 System (IEC
60601-1-2)
The KODAK RVG 6500 with or without IPS System is intended for use in an
electromagnetic environment in which radiated RF disturbances are controlled. The
customer or the user of the KODAK RVG 6500 with or without IPS System can help
prevent electromagnetic interference by maintaining a minimum distance between portable
and mobile RF communications equipment (transmitters) and the KODAK RVG 6500 with
or without IPS System as recommended below, according to the maximum output power of
the communications equipment.
Rated Maximum Output
Power of Transmitter W
Separation Distance According to Frequency of Transmitter
150 kHz to 80
MHz 80 MHz to 800 MHz 800 MHz to 2,5
GHz
0.01 0.12 0.12 0.23
0.1 0.38 0.38 0.73
1 1.2 1.2 2.3
10 3.8 3.8 7.3
100 12 12 23
For transmitters rated at a maximum output power not listed above, the recommended
separation distance d in metres (m) can be estimated using the equation applicable to the
frequency of the transmitter, where P is the maximum output power rating of the transmitter
in watts (W) according to the transmitter manufacturer.
NOTE 1: At 80 MHz and 800 MHz, the separation distance for the higher frequency range
applies.
NOTE 2: These guidelines may not apply in all situations. Electromagnetic propagation is
affected by absorption and reflection from structures, objects and people.
Note: The communication without breaking has
been determined to be essential performance with
regard to electromagnetic compatibility

16 Chapter 2 Regulatory Information
Compliance with International Regulations
•The KODAK RVG 6500 with or without IPS System is an active
device specifically intended for recording of X-ray diagnostic image.
Medical Device directives 93/42/ European Economic Community
(EEC), Class IIa follow the rule 16.
•Radio and Telecommunications Terminal Equipment directives
1999/5/EEC
•FCC rules part 15
•Medical Devices Regulations (Canada)
FCC Requirements for USA
KODAK RVG 6500 System and KODAK RVG 6500 IPS System WARNING
TO USERS IN THE UNITED STATES
Federal Communication Commission Interference Statement 47 CFR
Section 15.105(b)
This equipment has been tested and found compliant with the limits for a Class
B digital device, pursuant to Part 15 of the FCC Rules. These limits are
designed to provide reasonable protection against harmful interference in a
residential installation. This equipment generates, uses and can radiate radio
frequency energy and if not installed and used in accordance with the
instructions, may cause harmful interference to radio communications. However,
there is no guarantee that the interference will not occur in a particular
installation. If this equipment does cause harmful interference to radio or
television reception, which can be determined by turning the equipment off and
on, the user is encouraged to try to correct the interference by one or more of
the following measures:
•Reorient or relocate the receiving antenna.
•Increase the separation between the equipment and receiver.
•Connect the equipment into an outlet on a circuit different from that
to which the receiver is connected.
•Consult the dealer or an experienced radio/TV technician for help.
No Unauthorized Modifications
47 CFR Section 15.21
CAUTION: This equipment may not be modified, altered, or changed in any
way without signed written permission from Carestream. Unauthorized
modification may void the equipment authorization from the FCC and will
void the Carestream warranty.
Antenna Requirement
47 CFR Section 15.203
This device KODAK RVG 6500 System and KODAK RVG 6500 IPS System
complies with Part 15 of the FCC Rules. Operation is subject to the following 2
conditions: (1) This device may not cause harmful interference, and (2) this
device must accept any interference received, including interference that may
cause undesired operation.

KODAK RVG 6500 System_Regulatory User Guide (SM737)_Ed02
17
IC Requirements for Canada
Operation is subject to the following 2 conditions: (1) This device may not cause
interference, and (2) this device must accept any interference, including
interference that may cause undesired operation of the device.
To reduce potential radio interference to other users, the antenna type and its
gain should be so chosen that the equivalent isotropically radiated power
(e.i.r.p.) is not more than that permitted for successful communication.
L'utilisation de ce dispositif est autorisée seulement aux deux conditions
suivantes : (1) il ne doit pas produire de brouillage, et (2) l'utilisateur du
dispositif doit être prêt à accepter tout brouillage radioélectrique reçu, même si
ce brouillage est susceptible de compromettre le fonctionnement du dispositif.
Afin de réduire le risque d'interférence aux autres utilisateurs, il faut choisir le
type d'antenne et son gain de façon à ce que la puissance isotrope rayonnée
équivalente (p.i.r.e.) ne soit pas supérieure au niveau requis pour l'obtention
d'une communication satisfaisante. Cet appareil numérique de la classe B
respecte toutes les exigences du règlement sur le matériel brouilleur du Canada.

18 Chapter 2 Regulatory Information
Declaration of Conformity with the R-TTE Directives

KODAK RVG 6500 System_Regulatory User Guide (SM737)_Ed02
3 Technic al
Specifications
Factory
TROPHY
4, Rue F. Pelloutier, Croissy-Beaubourg
77435 Marne la Vallée Cedex 2, France
Manufacturer:
Carestream health, Inc.
150 Verona Street
Rochester,
New York - USA 14608
Model
KODAK RVG 6500 System
KODAK RVG 6500 IPS System
KODAK RVG 6500 With or Without IPS System
Technical Specifications
Components KODAK RVG 6500
System
KODAK RVG 6500 IPS
System
RVG Sensor
Sensor
Technol ogy CMOS CMOS
Sensor matrix
〈Size 0: 900 x 1200
pixels
〈Size 1:1200 x 1600
pixels
〈Size 2: 1440 x 1920
pixels
〈Size 1:1200 x 1600
pixels
〈Size 2: 1440 x 1920
pixels
20 Chapter 3 Technical Specifications
Sensor active
surface
dimensions
〈Size 0: 16.6 x 22.2 mm
〈Size 1: 22.2 x 29.6 mm
〈Size 2: 26.6 x 35.5 mm
〈Size 1: 22.2 x 29.6 mm
〈Size 2: 26.6 x 35.5 mm
Gray scale 4096 bits 4096 bits
RVG Control Box
Dimension 83mm (H) x 47mm (W) x
16mm (D)
83mm (H) x 47mm (W) x
21mm (D)
Weight 65g 90g
Battery type Lithium Lithium
Battery
charging time 4hrs 4hrs
RVG Control Box Charger
Mains power
supply Input
voltage
〈230/240 V - 50/60 Hz
〈100/110/130 V - 50/60
Hz
〈230/240 V - 50/60 Hz
〈100/110/130 V -
50/60 Hz
WiFi Access Point
Reference
Model WGR614 WGR614
Dimension 28 x 175 x 119 mm 28 x 175 x 119 mm
Weight 260 g 260 g
Input voltage 100/240 V - 50/60 Hz 100/240 V - 50/60 Hz
IPS aiming ring
Technology NA Electromagnetic signals
Dimensions NA 150 x 115 x 40 mm
Weight NA 235 g
Battery type NA 4 x AAAA
(Continued)
Components KODAK RVG 6500
System
KODAK RVG 6500 IPS
System

KODAK RVG 6500 System_Regulatory User Guide (SM737)_Ed0221
Minimum Computer System Requirement
The computer and the peripheral equipment must conform to the
IEC60950 standard.
Item Viewing and Acquisition Comments
CPU 2 GHz Intel Duo Core
RAM 2 GB
RAM has a major
impact on system
performance.
Hard disk drive
•1.2 GB for software
installation
•80 GB free space to use
the software
Graphic board Graphic board supporting Open
GL 1.2 with 256 MB of video
RAM (Recommended Nvidia / ATI)
The video RAM has
major impact on
system performance.
Monitor
•1 monitor
•17” or larger
•1024 x 768 minimum
screen resolution - 32 bits
color mode
Your monitor is a
vital component in
displaying quality
images. Low-quality
screens will prevent
you from proper
diagnoses and
treatment.
Operating system
•Windows XP Home / Pro
edition SP3 (32 bits)
•Windows Vista (32 bits)
(Recommended SP1)
•Windows 7 (32 or 64 bits)
The operating
system
specifications are
valid as of KODAK
Dental Imaging
Software v6.12.
Ethernet interface 100 Mbits for LAN
USB 2.0 2 ports
Wireless adapter Wireless 802.11g Adapter (USB,
PCMCIA or PCI)
CD/DVD drive DVD-ROM drive is required to
install the product.
Backup media Removable/portable, external hard
disk drive.
We strongly
recommend a daily
backup of x-ray
images and patient
records.
22 Chapter 3 Technical Specifications
RFID Technical Specification
WiFi Network Technical Specification
Item Specification
Frequency band 13.56MHz
Standard classification ISO 14443
Transmission range <2cm
Item Specification
Network Protocol TCP/IP
Network Type Private Wireless LAN (WLAN)
Network Mode Infrastructure mode
Wireless Protocol 802.11g
Frequency band 2.4 GHz ISM radio band
Modulation OFDM
Data rated (measured) Up to 10Mbps
Number of channels
•11 for USA & Canada
•13 for Europe
•14 for Japan
Maximum Power 20 mW
IP Address
The KODAK RVG 6500 / KODAK RVG
6500 IPS client acts as a DHCP. Client
DHCP server is embedded within the
Access Point.
Data Size (maximum) One 5.4 MByte file
Security
WEP 128-bit encryption Factory loaded key
Private Patient Identification Data No patient ID data transmitted

KODAK RVG 6500 System_Regulatory User Guide (SM737)_Ed0223
KODAK RVG 6500 With or Without IPS System
Environmental Requirements
Environmental Specifications
Operating temperature 0° to 40° C
Range Up to 10m
Ambient Operating Conditions
Tem pe ra tures 0 ~ 40°C
Relative humidity 45% ~ 85%
Atmospheric pressure 700 ~ 1060 hpa
Storage Conditions
Tem pe ra tures -20 ~ 60 °C
Relative humidity 45 ~ 85%
Atmospheric pressure 700 ~ 1060 hpa
Transport Conditions
Tem pe ra tures -20 ~ 60 °C
Relative humidity 45 ~ 85%
Atmospheric pressure 700 ~ 1060 hpa

24 Chapter 3 Technical Specifications