1 89224 141942
User Manual: 89224
Open the PDF directly: View PDF ![]() .
.
Page Count: 4
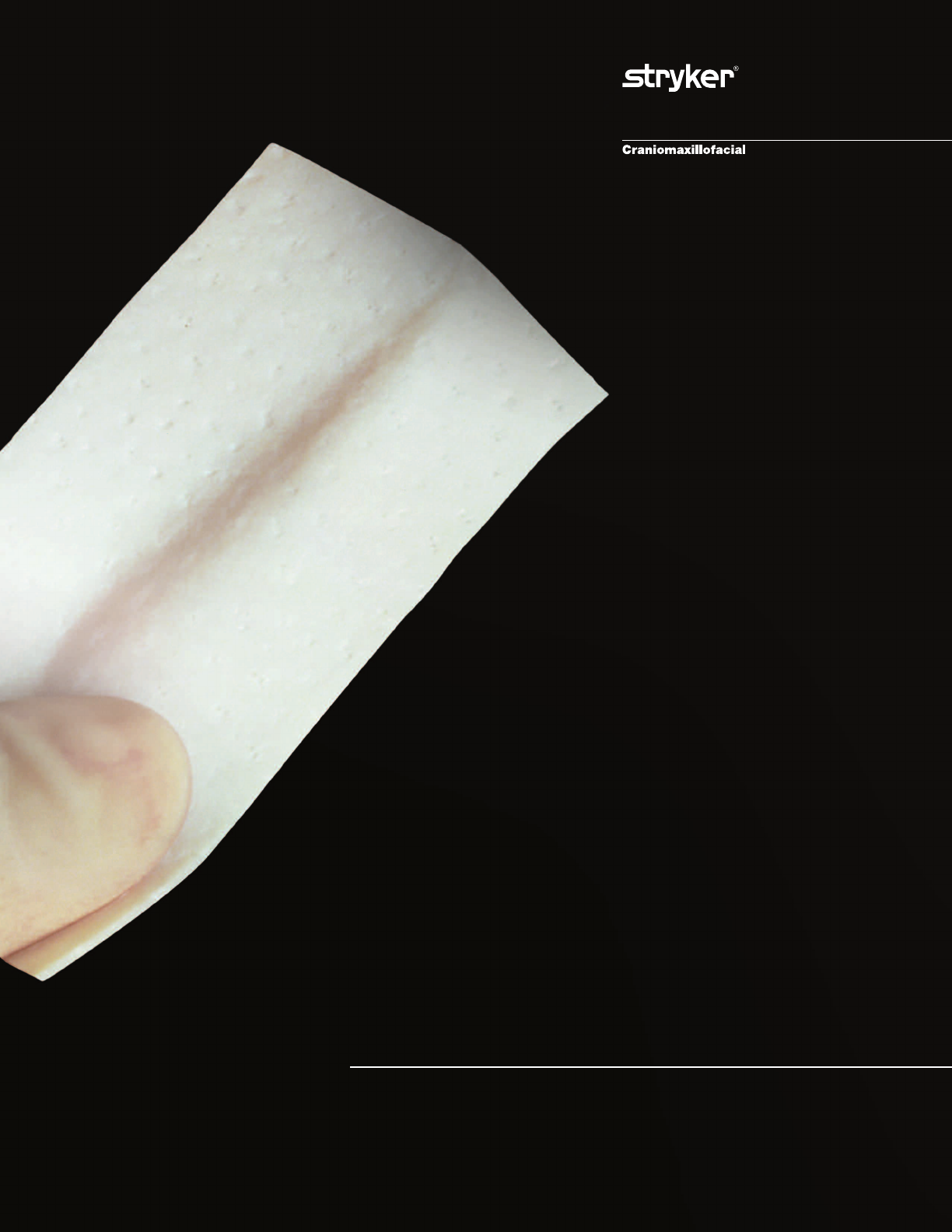
ENDURAGen
Collagen Implant designed for soft tissue
augmentation, reinforcement and repair

Excellent Biomaterial for Excellent Results
The
ENDURAGen
Collagen Implant is specifically designed for soft tissue
augmentation, reinforcement and repair in plastic and reconstructive surgery
of the head and face.
The
ENDURAGen
Biomaterial is a sterile, off-white, moist, tough but flexible
flat sheet of acellular cross-linked porcine dermal collagen with its constituent
elastin fibers. The flexible material readily conforms to anatomical shapes
and may provide surgeons with the flexibility to meet their patient’s
individual requirements.
This patented collagen matrix has a structural architecture similar to human
tissue, which offers a natural scaffold for fibroblast infiltration and vascularization.
The enzymatic digestion and cross-linking manufacturing process makes
ENDURAGen
Implants resistant to breakdown and absorption, thus allowing
the surgeon to effect a durable repair or reconstruction for soft tissue contouring
and/or reinforcement procedures.
ENDURAGen
Implants are supplied Sterile
in double wrapped, heat sealed packets. Requires no hydration prior to use.
ENDURAGen Collagen Implants may be:
• Cut
• Shaped
• Sutured
SEM of
ENDURAGen
Collagen Matrix
ENDURAGen Collagen Implant
Strong, readily accepts sutures
Flexible, conforms to anatomical shapes,
consistent thickness throughout the implan
t
Cut to desired shape

Blood vessels formed on ENDURAGen two
weeks after implantation in animal model1
CAT # A (cm) B (cm) Thickness (mm)
89221 2 5 0.5
89223 1 4 1.0
89224 2 5 1.0
89225 3 8 1.0
(Actual Size)
A
1cm x 4cm x 1.0mm
2cm x 5cm x 1.0mm
2cm x 5cm x 0.5mm
3cm x 8cm x 1.0mm
B
Stryker Craniomaxillofacial
Kalamazoo, MI 49002 USA
t: 269 324 5346
toll free: 800 962 6558
f: 877 648 7114
www.stryker.com
Stryker Corporation or its divisions or other corporate
affiliated entities own, use or have applied for the following
trademarks or service marks: Stryker. All other trademarks
are trademarks of their respective owners or holders.
ENDURAGen is a registered trademark of
Tissue Science Laboratories Plc.
Literature Number: 9410-400-221 Rev. A
UnDe/P.S.
Copyright © 2011 Stryker
Printed in USA
1. In-vivo cellular infiltration and multi-layering of ENDURAGen, Findings from a
GLP conducted study at Northwick Park Institute of Medical Research, London.
A surgeon must always rely on his or her own professional clinical judgment when
deciding whether to use a particular product when treating a particular patient.
Stryker does not dispense medical advice and recommends that surgeons be trained
in the use of any particular product before using it in surgery.
The information presented is intended to demonstrate the breadth of Stryker product
offerings. A surgeon must always refer to the package insert, product label and/or
instructions for use before using any Stryker product. Products may not be available
in all markets because product availability is subject to the regulatory and/or medical
practices in individual markets. Please contact your Stryker representative if you have
questions about the availability of Stryker products in your area.