Process Manual MR
Process%20Manual-MR
User Manual:
Open the PDF directly: View PDF ![]() .
.
Page Count: 16

EPATHUSA
PROCESS MANUAL - MR
Issue No: 01 Date: September 15, 2017
Revision/Version: 1.0 Date: September 15, 2017
Approval Details
Prepared By
Issued & Controlled By
Approved By
Designation
Process expert team
Hari
Anitha
Copy Holders / File Access Authority
Copy No
Designation
1
Management Representative (Master Copy)
2
Director
3
Solution Delivery Head
4
Network & System Administrator
5
Manager – HR
6
Manager – Purchase
Process Manual - MR
Page 2 of 16 Rev.:00,Dt.:
CONTENTS
Sl.
No
Procedure Name
DOC No
Version Status
0
1
2
3
4
5
QUALITY SYSTEM PROCEDURES
1
QSP for Control of Documents
QSP/EPATHUSA/MR/
01
•
2
QSP for Control of Records
QSP/EPATHUSA/MR0
2
•
3
QSP for Management Review
QSP/EPATHUSA/MR/
03
•
4
QSP for Internal Audit
QSP/EPATHUSA/MR/
04
•
5
QSP for Corrective & Preventive
Action
QSP/EPATHUSA/MR/
05
•
DOCUMENTS
6
Master List of Documents -Internal
DOC/EPATHUSA/MR/
01
•
7
Master List Of Documents – External
DOC/EPATHUSA/MR/
02
•
8
Master List Of Records
DOC/EPATHUSA/MR/
03
•
FORMATS
9
Document Change Request
QF/EPATHUSA/MR/0
1
•
10
Document Issue Register
QF/EPATHUSA/MR/0
2
•
11
File Label
NA
•
12
File Index
NA
•
13
Management Review Meeting
Minutes
QF/EPATHUSA/MR/0
3
•
14
Internal Audit Plan
QF/EPATHUSA/MR/0
4
•
15
Internal Audit Schedule
QF/EPATHUSA/MR/0
5
•
16
Internal Audit Check List
QF/EPATHUSA/MR/0
6
•
17
Audit Non-Conformity Report
QF/EPATHUSA/MR/0
7
•
18
Internal Audit Summary Report
QF/EPATHUSA/MR/0
8
•
19
Corrective and Preventive Action
Report
QF/EPATHUSA/MR/0
9
•
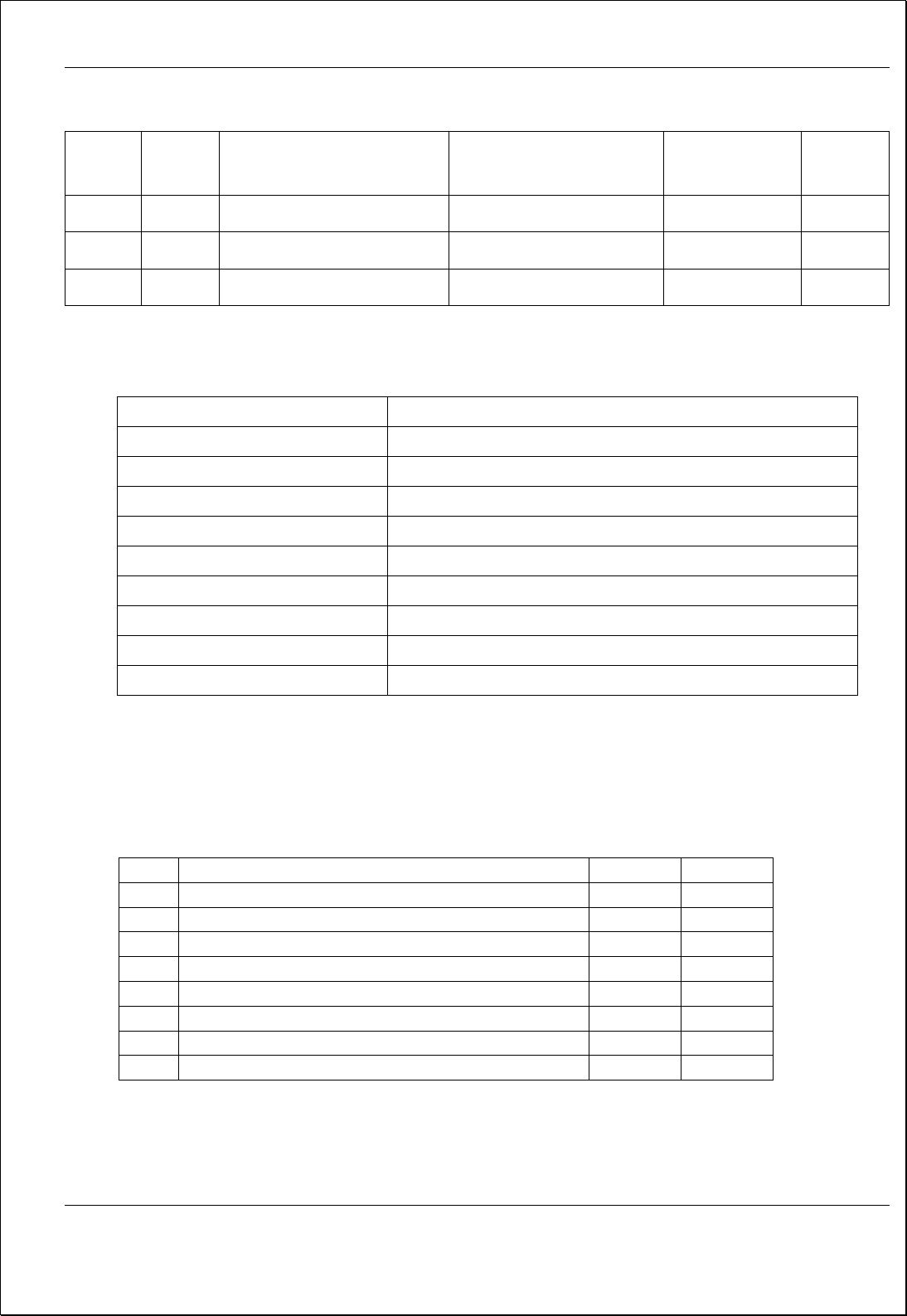
Process Manual - MR
Page 3 of 16 Rev.:00,Dt.:
REVISION HISTORY
DCR
No
Date
Nature of Change
Brief Reason for Change
Page / Section
Where
Changes Made
New
Revision
No
❖ Definitions and Acronyms
ACRONYM/ TERM
DEFINITION/ DESCRIPTION
MR
Management Representative
MD
Director
QSM
Quality System Manual
QSP
Quality System Procedure
QF
Quality Format
IA
Internal Audit
MRM
Management Review Meeting
NCR
Non Conformity Report
CAPA
Corrective Action & Preventive Action
❖ Activity / Responsibility Matrix
NO
ACTIVITY
MD
MR
01
Document Changes
I
R
02
Internal Audit Plan & Scheduling
R
03
Conducting MRM
R
I
04
Closing of NCRs
R
R – Responsible; I - Involved

Page 4 of 16 Rev.:00,Dt.:
QSP for Control of Documents
❖ Purpose
To control all documents and data to ensure their availability of pertinent issues, at removal of obsolete
documents, from all locations
❖ Scope
This procedure covers all internal & external documents.
Internal Documents
1st Level: Quality System Manual (QSM)
2nd Level: Process Manual
3rd Level: Records
External Documents
a) Information Technology Act
b) STPI Guidelines
c) Requirements from Clients
d) Client Information or Data
e) Reference Documents or User Manuals used in project execution
f) ISO 9001:2008
❖ Responsibility
Management Representative (MR) is responsible to ensure that this procedure is implemented & maintained
❖ Action & Method
➢ Identification
Document Name
Identification
Version Indicator
Issue No
Quality System
Manual &Process
Manual depts.
By Unique No in
(QSM/EPATHUSA/SL NO)
format
On each page
after 15 Revisions
Issue no is
incremented and
Revision is set to
ZERO
On cover page of the
manual, after 15
revisions in any one
page of manual issue
no is incremented by
one number
Quality System
Procedures
By Unique No in
(QSP/EPATHUSA/DEPT/SL
NO) format
On Each page
Not Applicable
Documents/ Work
Instructions / Guide
lines
By Unique No in
(DOC/EPATHUSA/DEPT/SL
NO/ Rev no) format
On Each page
Not Applicable
Forms / Templates
By Unique No in
(QF/EPATHUSA/DEPT/SL
NO/Rev No) format
By Effective Date
given in Master List of
Records
Not Applicable
External
Documents
Identified by document owner
given no/ name
Identified with number
given by authority and
organization ensures
latest revision is under
usage
NA

Page 5 of 16 Rev.:00,Dt.:
Approval & Issue
Document Name
Approved By
Issued & Controlled By
Quality System Manual
Director
Management Representative
Process Manual
Director
Management Representative
Project Documents
Director
Management Representative
Forms / Templates
Director
Management Representative
*** Besides above rules, respective approving authority can authorize in writing any position / executive of
the organization to approve any document
➢ Control and Issue of Documents
Documents are maintained and controlled in 2 ways i.e. one is by Soft copy and second is Hard copy
(Optional)
Soft Copy Control
• All approved documents are placed in a server with a folder name EPATHUSA QMS.
• As per given list of copyholders on cover page of each document, the system admin personals
are configured the folder for access.
• Except MR, all the copyholders are allowed to access the document in read only format
• Documents available in EPATHUSA QMS folder are only valid and this procedure is
applicable.
Hard Copy Control
• All approved documents shall be stamped as “MASTER COPY’ (normally in Blue Color) on
the back side of each page.
• Copied are allowed only from Master Copy and the same copies are issued as per distribution
list given in manuals or as per requirement and stamped as “CONTROLLED COPY” in Red on
the front side each page
• Document Issue Register is maintained for Hard copy issues
➢ Document and Data Change
Any Department Head can request for a change in Document through Document Change Request (DCR) –
QF/EPATHUSA/MR/01 and it can be forwarded to MR by email or hard copy
Once DCR is received, MR reviews the request and ensures that changes are not affecting integrity of
quality management system.
Take approval from approving authority of respective document (Refer Approval & Issue table)
Once DCR is approved, Change the respective document as requested and update version number &
effective date of respective document.
Method of Version number increment
Quality System Manual
Version number is changed only of respective page in which changes are made
Documents in Process Manual
Version number of all pages in entire document will be changed even if changes are made in one page
Also update the version status and effective date of document in other relevant documents where the
modified document is referred. Ex.: Master List of Documents, Master List of Records etc.
For Soft Copies, latest version documents shall be placed in the QMSDOC folder and old version
documents are moved to Obsolete Documents folder.

Page 6 of 16 Rev.:00,Dt.:
For Hard Copies, Issue the modified document to all copy holders through Document Issue Register
(QF/EPATHUSA/MR/01) and take back the old copies and file them in obsolete documents file after
stamping as “OBSOLETE DOCUMENT” in Red
➢ External Documents
MR identifies specific documents of external origin and maintains a list in Master List of External
Documents (DOC/EPATHUSA/MR/02) identifying version/release or issue date/period information and
issue records if more than one copy of the same document is in circulation.
Solution Delivery Head (SDH) is the custodian of External documents
MR controls the distribution and applicable version information to ensure that updates when available can
be circulated to all points of use.
➢ Obsolete Documents
To prevent the unintended use of obsolete documents, MR / Approving Authority collects the obsolete
documents from copyholders and dispose them as per disposal action given in table below.
Document Name
Disposal Action for Controlled Copy
Retention Time of
Master Copy
Quality System Manual
Destroy by Tear off or by fire
One Year
Quality System Procedures
Destroy by Tear off or by fire
Or can retain as SUPERCEEDED COPY
One Year
Work Instructions / Guide
lines/ Specifications
Destroy by Tear off or by fire Or can
retain as SUPERCEEDED COPY
One Year
Forms / Templates
Destroy by Tear off or by Burning
One Year
❖ References
Nil
❖ Documents
- Master List of Documents - Internal – DOC/EPATHUSA/MR/01
- Master List of Documents - External– DOC/EPATHUSA/MR/02
❖ Formats and Records
- Document Change Request – QF/EPATHUSA/MR/01
- Document Issue Register – QF/EPATHUSA/MR/02

Page 7 of 16 Rev.:00,Dt.:
QSP for Control of Records
❖ Purpose
To control quality records in order to demonstrate conformance to specified requirement and effective
operation of quality management system
❖ Scope
This procedure covers the records relating to quality management system.
❖ Responsibility
Management Representative and all HoDs are responsible to ensure that this procedure is implemented &
maintained
❖ Action & Method
Quality records essential for the operation of quality system have been identified and listed in various
procedures. The record formats are given unique identification code called form number
All Quality records are controlled in two ways, i.e. soft copy and hard copy (if any record maintained in
hard copy mode then soft copy of that record holds no value)
Procedure for Soft copy control
Identification, Storage, Protection and Retrieval
Directory structure along with naming conventions is the methodology used for identification of
individual components/ files.
The directory folder names and organisation of folders is left to the discretion of the Function Heads
with the condition that any file can be retrieved at any point of time without delay. The directory
structure should specify RW, RO permissions and to whom it is made access.
Access is restricted in the directory where such records are maintained.
Completeness of Records
All the sections of the records are to be completed in all respects and legible. In case of a section not
applicable, the same is to be indicated as “Not applicable”.
Procedure for Hard copy control
All files (except floppies/CDs’s) have a label in prescribed format, which shows identification (by name
and file number), retention period and storage location. The identification as on the labels can be
checked with the file List Register
Storage & Protection
The files are stored in closets after use (filing cabinets, steel/wooden almirhas, side racks, table
drawers’ etc.) The closets are located in dry place and preserved from damaging influences such as
termites, dust or excessive heat. This is usually verified during internal audits.
Retrieval
Access to hard file location is through file list, which identifies location. From storage location the file
can be retrieved quickly through a system of storage location numbering. The ease of access is
practically verified during audits.

Page 8 of 16 Rev.:00,Dt.:
Retention Period & Disposition
The period from the “time of creation” to moving a document/ quality record to “time of archival” status is
known as “retention period till Archival”. All the documents/ Quality records are to be maintained till the
Project closure and subsequently will be archived.
The retention period of the records (soft copy or hard copy) vary and depends mostly on the type, period of
the project, legal requirements (if any) and any contractual requirements with the customer or internal
policy requirements. The suggested minimum retention period after archival for all the documents/ records
is for a minimum period of 6 months. The archived documents/ records are retained for the specified
minimum retention period and later Functional heads may suitably decides either to retain them or dispose
/ (destroy) them depending on the need/contractual obligation.
Each project documents the retention period is maintained as mentioned in the respective Master List or
List of Records. (DOC/EPATHUSA/MR/03)
Disposition
After the retention period is over, MD / SDH / HoD take decision for disposal of the record files. Pending
such decisions the files are stored in archives.
❖ References
Nil
❖ Documents
- Master List of Records – DOC/EPATHUSA/MR/03
❖ Formats and Records
- File Labels
- Files List in each department

Page 9 of 16 Rev.:00,Dt.:
QSP for Management Review
❖ Purpose
To Review the implementation and effectiveness of QMS in regular intervals by top management
❖ Scope
This procedure covers all the activities covered under quality management system
❖ Responsibility
MR is responsible to plan and organize the management review meeting and maintain the minutes
❖ Action Method
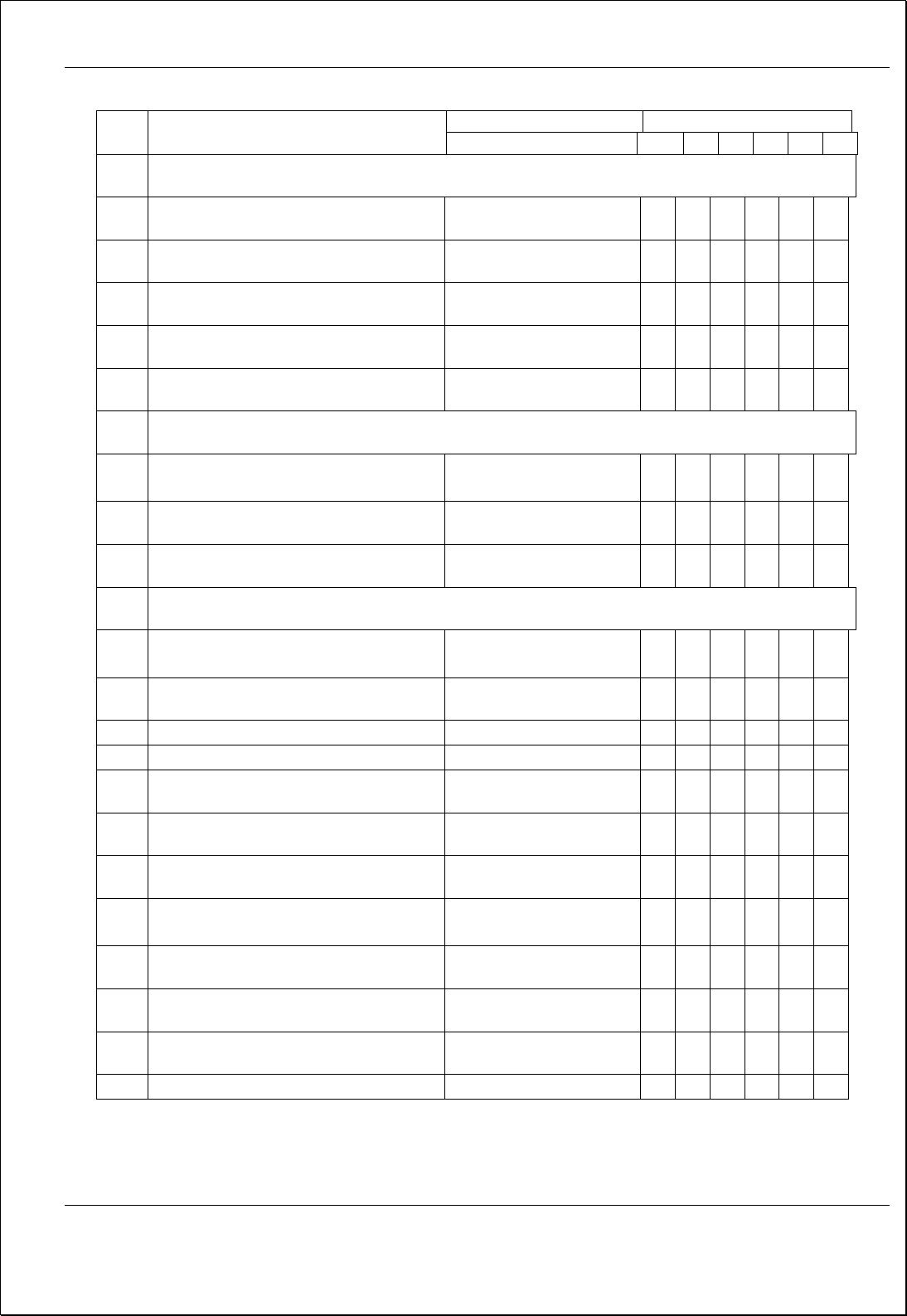
Process Flow Diagram
Input
Output
Quality System
Implementation &
Maintenance
o Suggested corrective / preventive action points
o Management Decisions
o Resource Allocations
o New / Renewed Quality Objectives
o MRM Minutes
Monitor
MRM minutes
Process / Sub Process
MR arranges MRM once in 6 months, and MD conducts meeting in
accordance with 5.6.2 of QSM and review all agenda points, resource
needs, quality policy suitability and current data trend analysis
submitted by HODs
• Agenda Points as per Section 5.6.2 of QSM
(QSM/ ePATHUSA/01)
• Resource Needs
• Suitability of Quality Policy
• Quality Objectives Analysis
• Process Trend Analysis
Corrective & Preventive
Action
• All Agenda Points Covered
• Resource Needs Reviewed
• Action Plan Prepared and Responsibilities
allotted

Page 10 of 16 Rev.:00,Dt.:
❖ Process Description
➢ Frequency
Management review meeting will be carryout once in 6 months (normally after each round of
internal audit) or more frequently as required by the Director
➢ Intimation
MR communicates to all the department heads about meeting date and time through Inter Office
Memo or by internal mail along with agenda points at least 3-4 days before the planned date.
➢ Conducting Meeting
Director chairs the meeting and conduct meeting as per input / agenda given below.
➢ Participants
• MR
• Manager Business Development
• Solution Delivery Head
• System Administrator
• Manager - HR
• Manager – Admin
• Other Team As required by MD or Functional heads
➢ Agenda / Input for MRM
1. Follow-up actions from previous management reviews.
Review the status of Points discussed and planned corrective actions in last MRM
2. Results of internal/external audits. Data to be submitted by MR
Non-conformities (NCs) raised in last internal audit and also external audit held before this
MRM to be reviewed as per data submitted by MR. During review comparison of current NCs
with last audit NCs are recommended
3. Customer feedback (including grievances/complaints)
Customer complaints/ queries received against projects developed during last 6 months are
reviewed to minimize or eliminate the complaints in future.
Customer Satisfaction / Feedback data collected through customer satisfaction survey is
reviewed
Other issues related to customers observed during interaction by organization personal are
also reviewed
4. Project Status
Status of projects under developed / completed during last 3 months shall be reviewed as per
data submitted by SDH .
5. Process performance and product conformity
All HoDs should submit the data of their individual departmental objectives (Current status) in
graphical or tabular format for review.
The points regarding process such as project compliance, technical issues, resource problems
shall be discussed
6. Status of corrective and preventive actions
Status of following shall be reviewed
a) Corrective / Preventive actions initiated against internal audit / External audit NCRs and
based on MRM discussions and should be reviewed.
b) Bug Report of each project and its corrective action shall be reviewed
7. Changes that could affect the quality management system
Any changes which affects the QMS documentation or implementation flow like change of
departmental heads, adding new products / services are to be discussed here and decisions are
taken.

Page 11 of 16 Rev.:00,Dt.:
8. Recommendations for improvement.
Any recommendations by committee to improve the effectiveness of QMS implementation shall
be discussed here
9. Any other agenda with the consent of the Director / Participants
Apart from QMS, any points related to organization activities can be discussed here, like
expansion planning’s, participation in exhibitions or events etc.
10. Resource Needs
Resource needs like staff requirements or any infrastructure requirements are to be discussed
here.
11. Quality Policy Suitability
Quality policy of the company should be reviewed for its awareness among the staff and its
suitability to the organization’s activity.
12. Quality Objective Analysis
MR submits the current stand of quality objectives for review, the committees reviews the data
and take decision on goals whether to continue with same goals or modify them
13. Trainings Records
Manager HR submits the trainings conducted during last 3 months and their effectiveness on
the participants. New trainings shall also be identified here.
➢ Recording Minutes
MR records review meeting minutes in Management review meeting minutes. Minutes are recorded
agenda point wise discussions and suggestions accepted by committee. The responsibility and
target date to implement action plan also recorded in minutes.
➢ Follow up for Action points
MR will follow up with departmental heads (who ever assigned responsibility to implement the
action points) for action initiated and status. Once point is implemented, MRM minutes are updated
with action taken date and close the status of respective document.
❖ References
- Quality System Manual – QSM/EPATHUSA/01
- QSP for Corrective and preventive Action – QSP/EPATHUSA/MR/05
❖ Documents
- Nil
❖ Formats and Records
- Inter Office Memo / Internal Mails
- Management Review Meeting Minutes – (QF/EPATHUSA/MR/03)
Page 12 of 16 Rev.:00,Dt.:
QSP for Internal Audit
❖ Purpose
To carry out audit periodically, to verify whether quality activities and related results comply with
planned arrangement and to determine effectiveness of the quality management system.
❖ Scope
This procedure covers all functions in comprising the quality management system.
❖ Responsibility
MR is Responsible is to ensure that this procedure is implemented and maintained.
❖ Action & Method
Process Flow Diagram
INPUT
• ISO 9001:2015 Requirements
• Quality System Requirements
• Customer Feedback and complaints
• Statutory & regulatory Requirements
• Company’s Quality Objectives
• Documentation of QMS
• Previous Audit Results
• Data Analysis Trends
MR Prepares Audit Plan and Schedule and selects audit
team (Ref. Procedure for Internal Audit)
Auditors Conduct Audits in following manner
• Prepares Audit Check List
• Compare documentation against requirements
• Compare practice against documentation
• Record Non-compliances with objective
evidence
Non-Compliance
Found?
No
Yes
Corrective & Preventive
Action
Record Audit Findings
Finalize report
MR Reviews Audit Report
OK?
Follow up to close the
NC by MR / Auditors
Output
Yes
No
o Assessment
Summery Matrix
o NC Reports
Management
Review
o Audit covered all
areas
o CA plan against NC’S
o Sign of auditee &
auditors

Page 13 of 16 Rev.:00,Dt.:
❖ Process Description
➢ Audit Plan
MR Prepares Internal audit plan (QF/EPATHUSA/MR/04) for every calendar year, Internal Audit
Schedule (QF/EPATHUSA/MR/05) is issued before each round of audit. Each function is audited
at least once in 6 months. While scheduling, MR considers last audit findings, corrective action
status and importance of activities of respective department. Apart from scheduled audits, certain
functions are selected for more frequent auditing, depending on their status, importance and past
compliance history
➢ Audit Team
Personal assigned to carry out audits are independent of those having direct responsibility for the
audited department. Audit activity can be out sourced to proven competent people. On such audits
company people also participate.
➢ Preparing for Audit
Auditors prepare for an audit by fully familiarizing themselves with the ISO 9001 standard,
refreshing their knowledge of the quality system manual and relevant procedures, reviewing
nonconformity reports of last audit and corrective action files and preparing questions/check lists.
➢ Conducting and Reporting the Audit
MR is responsible for the distribution of prepared audit scheduled to auditee at least one week / or
in advance. The acknowledgement from auditee is also taken.
The audit is conducted against the requirements of ISO-9001: 2008 as addressed by the company
along with all associated documents including customer and statutory requirements where
applicable.
Audit is conducted on a sampling basis by following internal audit check list
(QF/EPATHUSA/MR/06) and evidence of implementation is checked by assessment of objective
evidence.
The audits findings are recorded in Audit Non-conformity Report (QF/EPATHUSA/MR/07) The
Auditee write the proposed corrective actions (in consultation with the internal auditor to avoid
subsequent conflict of understanding).
MR reviews audit reports and inform the results of internal audits through internal audit summary
report (QF/EPATHUSA/MR/08) for management review.
➢ Corrective Action and Follow Up
Once nonconformity is identified and documented, the responsible auditee investigates the cause of
the nonconformity, proposes a corrective action to be taken and indicates the date by which the
corrective action will be fully implemented. The auditor reviews and approves the proposed action.
On or immediately after the due date for the implementation of corrective action, the auditor follow
up with an inquiry or an audit to determine if the corrective action has been implemented and if it is
effective, the nonconformity report is closed. If more work needed to fully implement the action, a
new follow-up date is agreed upon and the same is recorded on report

Page 14 of 16 Rev.:00,Dt.:
❖ References
- Quality System Documentation
- ISO 9001 : 2015 Standard
- ISO 19001 – Standard for Internal Audit
❖ Documents
- List of Qualified Internal Auditors – DOC/EPATHUSA/MR/04
❖ Formats and Records
- Internal Audit Plan (QF/EPATHUSA/MR/04)
- Internal Audit Schedule (QF/EPATHUSA/MR/05)
- Internal Audit Check List (QF/EPATHUSA/MR/06)
- Audit Non-Conformity Report (QF/EPATHUSA/MR/07)
- Internal Audit Summary Report (QF/EPATHUSA/MR08)
Page 15 of 16 Rev.:00,Dt.:
QSP for Corrective & Preventive Action
❖ Purpose
To carry out corrective action for eliminating the causes of actual and potential non-conformities and
prevent occurrence / reoccurrence of non-conformities
❖ Scope
This procedure covers system and product related non-conformities found in entire process and quality
system.
❖ Responsibility
All HoDs, and MR are responsible to ensure that this procedure is implemented and maintained.
❖ Action & Method
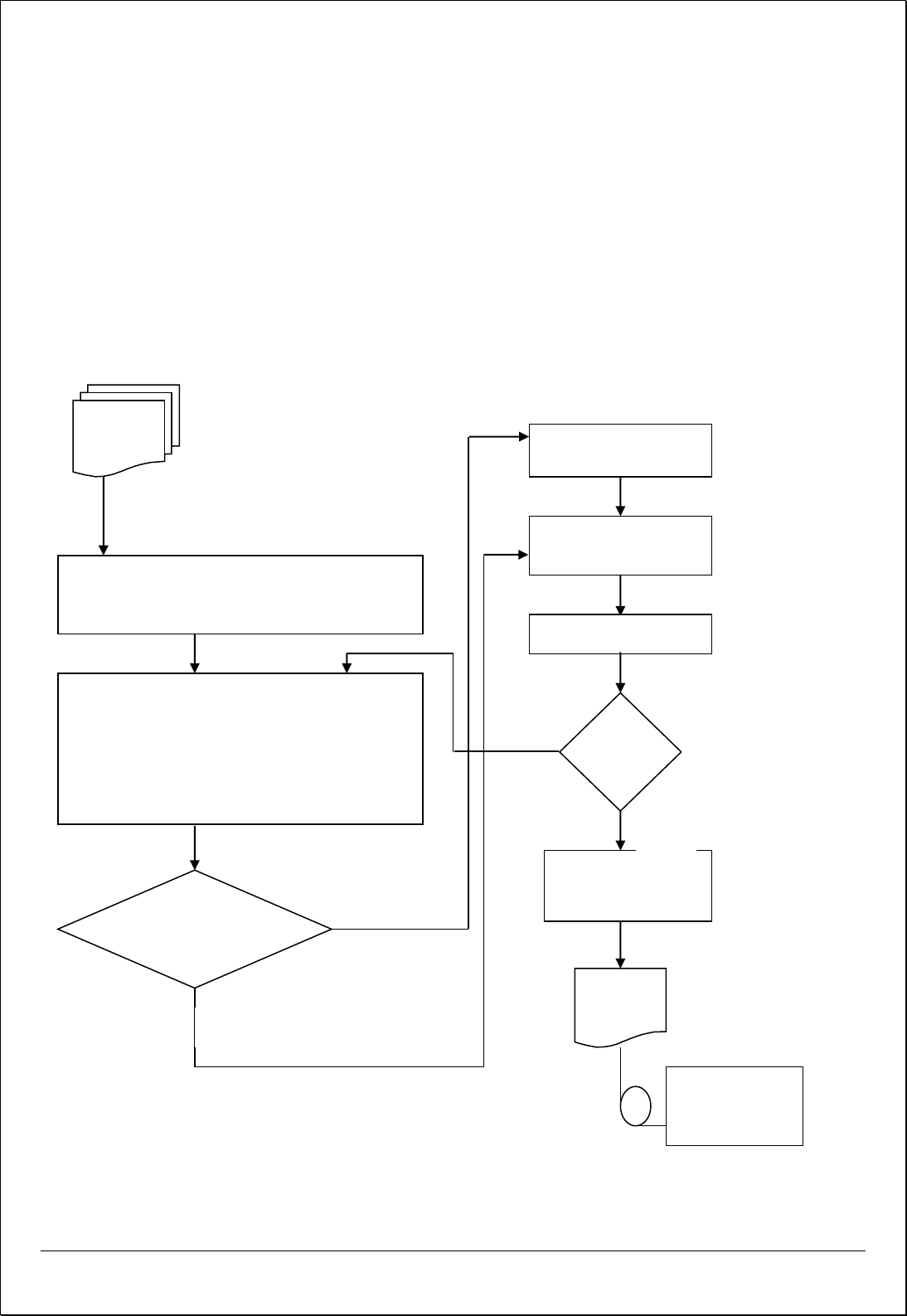
Process Flow for Corrective Action
INPUT
Reviewing & Determining Causes for Non-
Conformities
Evaluating the need for Corrective action to ensure that
nonconformities do not recur
Determining resources requirement and
appointing a person to implement Action
Review Action
Taken
• Effectiveness of Corrective Action
taken
• Further Actions Needed to close the
issue
OUTPUT
• Corrective and Preventive Action Report –
QF/Puyenpa/MR/09
• Customer Feedback
• Customer Complaints
• Internal & External Audit Results
• Bugs during project testing
• Process Performance Indicators
• Quality Objective Analysis
• Project Analysis

Page 16 of 16 Rev.:00,Dt.:
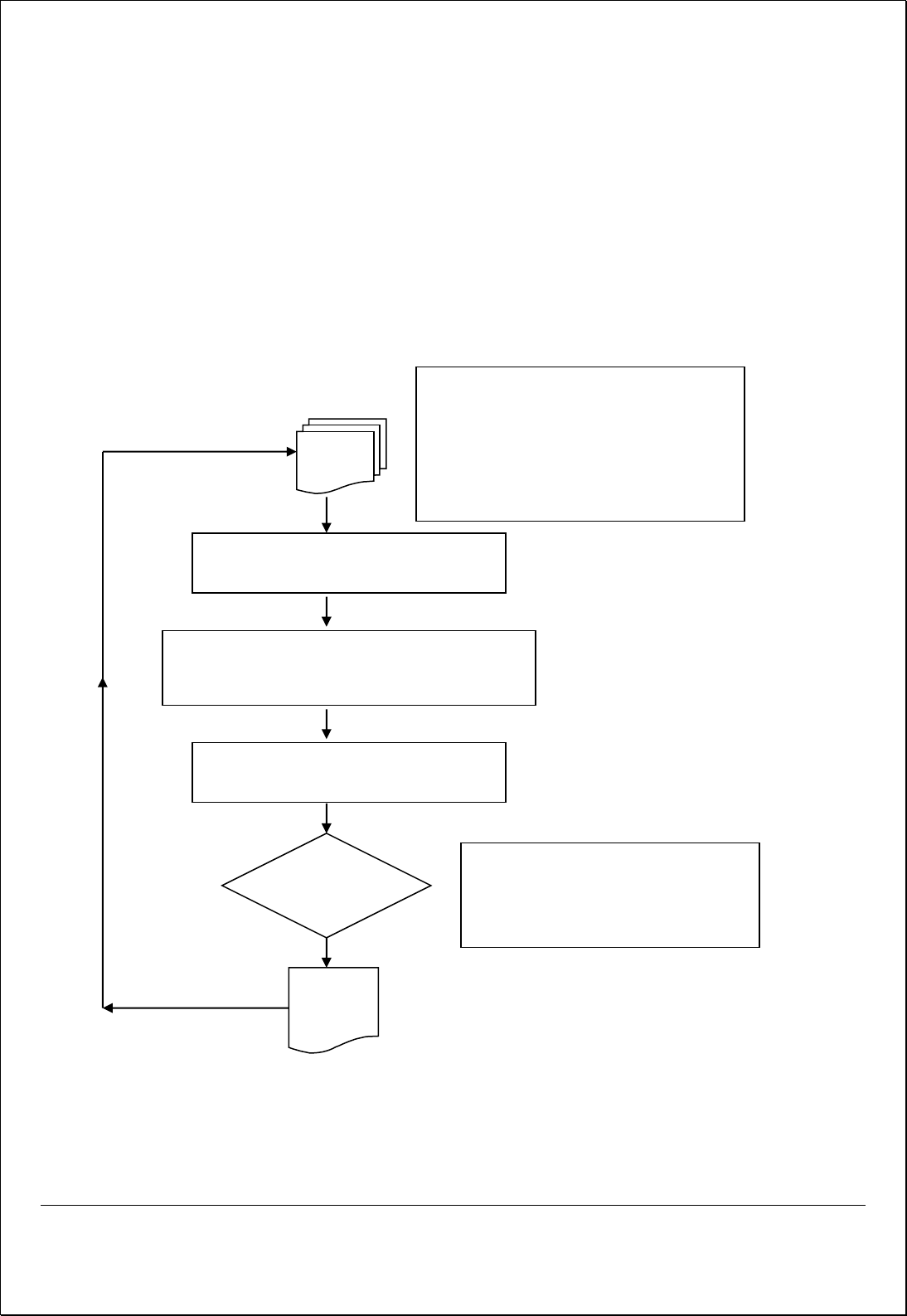
Process Flow for Preventive Action
❖ References
- Nil
❖ Documents
- Nil
❖ Formats and Records
- Corrective and Preventive Action Report – QF/EPATHUSA/MR/09
INPUT
Identifying and Reviewing Causes for Potential
Non-Conformities
Evaluating the need for Preventive action to ensure that
nonconformities do not occur
Determining resources requirement and
appointing a person to implement Action
Review Action
Taken
• Time of Completion
• Effectiveness of Preventive Action
taken
• Further Actions Needed to close the
issue
• Customer Feedback & Complaints
• Internal & External Audit Results
• Trend Analysis
• Quality Objective Analysis
• Bug report by QA Team
• Project Analysis
OUTPUT
• Corrective and Preventive Action Report –
QF/ISPL/MR/09