Otsuka America Pharmaceutical 06120 Proteus Patch, Model SPC-0800 User Manual2
Proteus Digital Health, Inc. Proteus Patch, Model SPC-0800 Users Manual2
Contents
- 1. Users Manual1
- 2. Users Manual2
Users Manual2
LBL‐0176,Rev1|UserSupplementalInformation,
Proteus®Patch
Page1of1Effective:19DEC2013
THEPROTEUSPATCH
SPC‐0800
USERSUPPLEMENTALINFORMATION
TableofContents
1.TECHNICALINFORMATION................................................................................................................2
1.1–Classification....................................................................................................................................2
1.2–EnvironmentalConditions..........................................................................................................2
1.3–MinimizingSkinIrritation............................................................................................................2
1.4–ProtectionagainstIngressofSolidsandLiquids.................................................................2
1.5–AvoidingUnsafeUseConditions..............................................................................................2
1.6–InformationonElectromagneticandOtherInterferences...............................................3
1.7–InformationontheRadioSubsystem......................................................................................3
1.8–EuropeanR&TTEDeclarationofConformity.........................................................................7
1.9–CISPRInterferenceStatement....................................................................................................7
1.10–FCCInterferenceStatement.....................................................................................................8
1.11–FCCWirelessNotice.....................................................................................................................8
1.12–FCCIdentifier.................................................................................................................................8
2–DISPOSALOFWASTEPRODUCTS..................................................................................................8
3–MANUFACTURERCONTACTINFORMATION.............................................................................9
LBL‐0176,Rev1|UserSupplementalInformation,
Proteus®Patch
Page2of2Effective:19DEC2013
1.TECHNICALINFORMATION
1.1–Classification
Caution:Federal(U.S.A)lawrestrictsthisdevicetosalebyorontheorderofaphysician.
TheProteusPatchiscategorizedasClassII(inUS)andIIa(inEU).
TheUser(patient)istheintendedOperatoroftheProteusPatch.
1.2–EnvironmentalConditions
TheProteus®Patchisintendedforstorageandoperationinaroom‐temperatureenvironment.
ConditionTemperatureHumidityPressure(Altitude)
Operating20C–28C 15%‐93%700hPa–1060hPa
Storage20C–28C15%‐93%700hPa–1060hPa
Transport2C–38C15%‐85%700hPa–1060hPa
1.3–MinimizingSkinIrritation
TheProteusPatchhasbeendesignedtominimizethepossibilityofskinirritation.Observingthese
cautionswillreducethelikelihoodofskinirritationorbruisingunderthePatch:
DONOTcontinueuseuntilfurtherinstructionbyaphysicianifyourskinisirritatedor
inflamedaroundthepatch.
DONOTplaceinlocationswhereyourskinisscraped,cracked,inflamed,orirritated.
DONOTplaceinalocationthatoverlapstheareaofthemostrecentlyremovedPatch.
DONOTuseifyouareallergictoadhesivetape.
DONOTwearthesamePatchformorethanoneweek.
DONOTdroporbumpwithexcessiveforce.
1.4–ProtectionagainstIngressofSolidsandLiquids
TheProteusPatchhasanIngressProtectionratingofIP27.Thismeansthattheenclosurehasno
penetrationsandithasbeenratedforimmersioninliquidupto1mdepth.Forcontinuedsafety,
shouldtheenclosurebecomepenetratedortorn,removethePatchimmediatelyandreplaceitwith
anewone.
1.5–AvoidingUnsafeUseConditions
TheProteusPatchisnotadiagnosticdevice.DONOTattempttouseittodiagnoseheart‐related
conditions,anincorrectdiagnosismayresult.
LBL‐0176,Rev1|UserSupplementalInformation,
Proteus®Patch
Page3of3Effective:19DEC2013
TheProteusPatchhasnotbeentestedorapprovedassafeforoperationduringairtravel.DONOT
usethePatchduringairtravel;itmayinterferewiththeaircraftnavigationalinstruments.
TheProteusPatchhasnotbeentestedorapprovedforuseinthepresenceofstrongmagneticor
electricfields.DONOTwearthePatchduringmagneticresonanceimaging(MRI),cautery,and
externaldefibrillationprocedures.DamagetothePatch,yourskin,oranunexpectedmagnetic
attractionmayresult.PleaseinformyourhealthcareprofessionalthatthePatchmustberemoved
priortoengaginginoneoftheseprocedures.
WARNING:Nomodificationofthisequipmentisallowed.ModifyingtheProteusPatchmaycausea
safetyhazardfortheuser.
1.6–InformationonElectromagneticandOtherInterferences
TheProteusPatchhasbeenevaluatedanddeemedcompliantwiththerequirementsinEN60601‐1‐
2ClassBforElectromagneticCompatibility(EMC).MedicalElectricalEquipmentneedsspecial
precautionsregardingEMCandneedstobeinstalledandputintoserviceaccordingtotheEMC
informationprovidedinthisUserManual.PortableandmobileRFcommunicationsequipmentcan
affectMedicalElectricalEquipment.TheProteusPatchshouldnotbeusedadjacenttoorstacked
withotherelectromagneticequipment.Ifadjacentorstackedusewithotherelectromagnetic
equipmentisnecessary,verifythattheProteusPatchoperationisnormalintheconfiguration(s)in
whichitwillbeused.
1.7–InformationontheRadioSubsystem
TheProteusPatchincorporatesaBluetoothTmradiosubsystemwhichiscompliantwiththe
Bluetoothstandard.ThefollowinginformationisprovidedtosatisfytherequirementsofEN/IEC
60601‐1‐2:
TheBluetoothradiotransmitsandreceiveson40frequencybandswhichareequallyspacedat
2MHzintervalsbetween2402MHzand2480MHz.
Theeffectivereceivebandwidthis1.25MHz.
Thetransmitmodulationisfrequency‐hoppingusingGFSK(GaussianFrequencyShiftKeying)witha
bandwidth‐bitperiodproductBT=0.5.TheModulationindexisbetween0.28and0.35.
Theeffectiveradiatedpoweris‐15dBm(P=0.032mW)
Guidanceandmanufacturer’sdeclaration–electromagneticemissions
TheProteusPatchisintendedforuseintheelectromagneticenvironmentspecifiedbelow.The
customerortheuserofProteusPatchshouldassurethatitisusedinsuchanenvironment.
EmissionstestComplianceElectromagneticenvironment–guidance
RFemissions
CISPR11
Group1
ThePatchusesRFenergyonlyforitsinternalfunction.
Therefore,itsRFemissionsareverylowandarenotlikely
LBL‐0176,Rev1|UserSupplementalInformation,
Proteus®Patch
Page4of4Effective:19DEC2013
tocauseanyinterferenceinnearbyelectronicequipment.
RFemissions
CISPR11
ClassBThePatchissuitableforuseinallestablishments,
includingdomesticestablishmentsandthosedirectly
connectedtothepubliclowvoltagepowersupply
networkthatsuppliesbuildingsusedfordomestic
purposes.
Harmonic
emissions
IEC61000‐3‐2
Notapplicable
Voltage
fluctuations/
flickeremissions
IEC61000‐3‐3
Notapplicable

LBL‐0176,Rev1|UserSupplementalInformation,
Proteus®Patch
Page5of5Effective:19DEC2013
Guidanceandmanufacturer’sdeclaration–electromagneticimmunity
TheProteusPatchisintendedforuseintheelectromagneticenvironmentspecifiedbelow.The
customerortheuseroftheProteusPatchshouldassurethatitisusedinsuchanenvironment.
ImmunitytestIEC60601
testlevel
Compliance
level
Electromagneticenvironment–
guidance
PortableandmobileRF
communicationsequipmentshouldbe
Guidanceandmanufacturer’sdeclaration–electromagneticimmunity
TheProteusPatchisintendedforuseintheelectromagneticenvironmentspecifiedbelow.The
customerortheuserofthePatchshouldassurethatitisusedinsuchanenvironment.
ImmunitytestIEC60601testlevelCompliance
level
Electromagneticenvironment–
guidance
Electrostatic
discharge(ESD)IEC
61000‐4‐2
+/‐6kVcontact
+/‐8kVair
+/‐6kV
contact
+/‐8kVair
Floorsshouldbewood,concrete
orceramictile.Iffloorsare
coveredwithsyntheticmaterial,
therelativehumidityshouldbeat
least30%.
Electricalfast
transient/burst
IEC61000‐4‐4
+/‐2kVforpower
supplylines
+/‐1kVfor
input/output
lines
Notapplicable
SurgeIEC61000‐4‐
5
+/‐1kVline(s)to
line(s)
+/‐2kVline(s)toearth
Notapplicable
Voltagedips,short
interruptionsand
voltagevariations
onpowersupply
inputlines
IEC61000‐4‐11
<5%UT(>95%dipin
UT)
for0,5cycle
40%UT(60%dipinUT)
for5cycles
70%UT(30%dipinUT)
for25cycles
<5%UT(>95%dipin
UT)
for5s
Notapplicable
Powerfrequency
(50/60Hz)
magneticfield
IEC61000‐4‐8
3A/m3A/mPowerfrequencymagneticfields
shouldbeatlevelscharacteristic
ofatypicallocationinatypical
commercialorhospital
environment.
NOTEUTisthea.c.mainsvoltagepriortoapplicationofthetestlevel.

LBL‐0176,Rev1|UserSupplementalInformation,
Proteus®Patch
Page6of6Effective:19DEC2013
ConductedRF
IEC61000‐4‐6
RadiatedRF
IEC61000‐4‐3
3Vrms
150kHzto80
MHz
3V/m
80MHzto2,5GHz
Not
Applicable
3V/m
usednoclosertoanypartofthe
ProteusPatch,includingcables,than
therecommendedseparation
distancecalculatedfromtheequation
applicabletothefrequencyofthe
transmitter.
Recommendedseparationdistance
NotApplicable
d=1,17√P80MHzto800MHz
d=2,33√P800MHzto2,5GHz
wherePisthemaximumoutput
powerratingofthetransmitterin
watts(W)accordingtothetransmitter
manufactureranddisthe
recommendedseparationdistancein
metres(m).
FieldstrengthsfromfixedRF
transmitters,asdeterminedbyan
electromagneticsitesurvey,
a
should
belessthanthecompliancelevelin
eachfrequencyrange.
b
Interferencemayoccurinthevicinity
ofequipmentmarkedwiththe
followingsymbol:
NOTE1At80MHzand800MHz,thehigherfrequencyrangeapplies.
NOTE2Theseguidelinesmaynotapplyinallsituations.Electromagneticpropagationisaffectedby
absorptionandreflectionfromstructures,objectsandpeople.
a
Fieldstrengthsfromfixedtransmitters,suchasbasestationsforradio(cellular/cordless)
telephonesandlandmobileradios,amateurradio,AMandFMradiobroadcastandTVbroadcast
cannotbepredictedtheoreticallywithaccuracy.Toassesstheelectromagneticenvironmentdueto
fixedRFtransmitters,anelectromagneticsitesurveyshouldbeconsidered.Ifthemeasuredfield
strengthinthelocationinwhichtheProteusPatchisusedexceedstheapplicableRFcompliance
levelabove,theProteusPatchshouldbeobservedtoverifynormaloperation.Ifabnormal
performanceisobserved,additionalmeasuresmaybenecessary,suchasre‐orientingorrelocating
theProteusPatch.
b
Overthefrequencyrange150kHzto80MHz,fieldstrengthsshouldbelessthan3V/m.
LBL‐0176,Rev1|UserSupplementalInformation,
Proteus®Patch
Page7of7Effective:19DEC2013
Recommendedseparationdistancesbetweenportableandmobile
RFcommunicationsequipmentandtheProteusPatch
TheProteusPatchisintendedforuseinanelectromagneticenvironmentinwhichradiatedRF
disturbancesarecontrolled.ThecustomerortheuserofthePatchcanhelpprevent
electromagneticinterferencebymaintainingaminimumdistancebetweenportableandmobileRF
communicationsequipment(transmitters)andtheProteusPatchasrecommendedbelow,
accordingtothemaximumoutputpowerofthecommunicationsequipment.
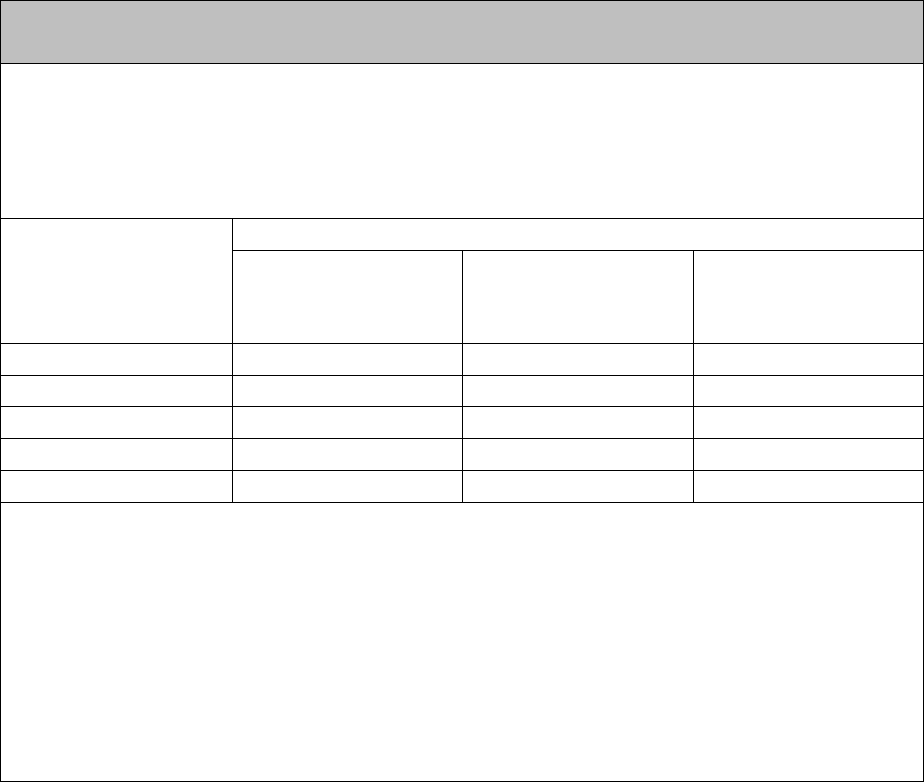
Ratedmaximum
outputpowerof
transmitter
W
Separationdistanceaccordingtofrequencyoftransmitter
150kHzto80MHz
d=1,2√P
80MHzto800MHz
d=1,2√P
800MHzto2.5GHz
d=2,3√P
0.01Notapplicable0.10.23
0.1Notapplicable0.40.74
1Notapplicable1.22.3
10Notapplicable3.77.4
100Notapplicable1223
Fortransmittersratedatamaximumoutputpowernotlistedabove,therecommendedseparation
distancedinmeters(m)canbeestimatedusingtheequationapplicabletothefrequencyofthe
transmitter,wherePisthemaximumoutputpowerratingofthetransmitterinwatts(W)according
tothetransmittermanufacturer.
NOTE1At80MHzand800MHz,theseparationdistanceforthehigherfrequencyrangeapplies.
NOTE2Theseguidelinesmaynotapplyinallsituations.Electromagneticpropagationisaffectedby
absorptionandreflectionfromstructures,objectsandpeople.
1.8–EuropeanR&TTEDeclarationofConformity
Hereby,Proteus®DigitalHealth,Inc.,declaresthattheProteusPatchisincompliancewiththe
essentialrequirementsandotherrelevantprovisionsofR&TTE(RadioandTelecommunications
TerminalEquipment)Directive1999/5/EC.Theproductiscompliantwiththefollowingstandards
and/orothernormativedocuments:
Safety(art.3.1a):EN60601‐1,3rdEd,IEC60601‐1‐11
EMC(art.3.1b):EN301489‐17v2.2.1
Spectrum(art.3.2):EN300328v1.8.1
Other:EN60601‐1‐2(2007)
TheProteusPatchcanbeusedincountriesintheEuropeanUnion.
1.9–CISPRInterferenceStatement
MEDICALELECTRICALEQUIPMENTneedsspecialprecautionsregardingEMCandneedstobe
installedandputintoserviceaccordingtotheEMCinformationprovidedinthissectionofthe
manual.PortableandmobileRFcommunicationsequipmentcanaffectMEDICALELECTRICAL
LBL‐0176,Rev1|UserSupplementalInformation,
Proteus®Patch
Page8of8Effective:19DEC2013
EQUIPMENT.TheProteusPatchmaybeinterferedwithbyotherequipment,evenifthatequipment
complieswithCISPRemissionrequirements.
1.10–FCCInterferenceStatement
ThisdevicecomplieswithPart15oftheFCCRules.Operationissubjecttothefollowingtwo
conditions:(1)thisdevicemaynotcauseharmfulinterference,and(2)thisdevicemustacceptany
interferencereceived,includinginterferencethatmaycauseundesiredoperation.Changesor
modificationsnotexpresslyapprovedbyProteusDigitalHealthcouldvoidyourauthoritytooperate
theequipment.
1.11–FCCWirelessNotice
Thisproductemitsradiofrequencyenergy,buttheradiatedoutputpowerofthisdeviceisfarbelow
theFCCradiofrequencyexposurelimits.
Note:
ThisequipmenthasbeentestedandfoundtocomplywiththelimitsforaClassBdigitaldevice,
pursuanttopart15oftheFCCRules.Theselimitsaredesignedtoprovidereasonableprotection
againstharmfulinterferenceinaresidentialinstallation.Thisequipmentgenerates,usesandcan
radiateradiofrequencyenergyand,ifnotinstalledandusedinaccordancewiththeinstructions,
maycauseharmfulinterferencetoradiocommunications.However,thereisnoguaranteethat
interferencewillnotoccurinaparticularinstallation.Ifthisequipmentdoescauseharmful
interferencetoradioortelevisionreception,whichcanbedeterminedbyturningtheequipmentoff
andon,theuserisencouragedtotrytocorrecttheinterferencebyoneormoreofthefollowing
measures:
— Reorientorrelocatethereceivingantenna.
— Increasetheseparationbetweentheequipmentandreceiver.
— Connecttheequipmentintoanoutletonacircuitdifferentfromthattowhichthereceiveris
connected.
— Consultthedealeroranexperiencedradio/TVtechnicianforhelp.
1.12–FCCIdentifier
FCCID:X7906120
2–DISPOSALOFWASTEPRODUCTS
Productionofthisequipmentrequiredtheextractionanduseofnaturalresources.Theequipment
maycontainsubstancesthatcouldbeharmfultotheenvironmentorhumanhealthifimproperly
handledattheproduct’sendoflife.Inordertoavoidreleaseofsuchsubstancesintothe
environmentandtoreducetheuseofnaturalresources,alldevices,bothusedandunused,should
notbedisposedwithhouseholdwaste.Returntoarecyclingpointforelectricandelectronic
devices.

LBL‐0176,Rev1|UserSupplementalInformation,
Proteus®Patch
Page9of9Effective:19DEC2013
3–MANUFACTURERCONTACTINFORMATION
Torequesttechnicalinformationortoreportunexpectedevents,pleasecontactthemanufacturer
atoneoftheselocations.
UnitedStates
ProteusDigitalHealth,Inc
2600BridgeParkway,Suite101
RedwoodCity,CA94065
PhoneNumber:650‐632‐4031
FaxNumber:650‐632‐4071
UnitedKingdom
ProteusDigitalHealthUKLtd
6
th
Floor,41‐44GreatQueenSt.
LondonWC2B5AD