MergedFile 45e42cae 5c8e 41d3 B758 99bc1f335d37
2018-03-05
: Pdf 45E42Cae-5C8E-41D3-B758-99Bc1F335D37 45e42cae-5c8e-41d3-b758-99bc1f335d37 3 2018 pdf
Open the PDF directly: View PDF ![]() .
.
Page Count: 34

iFCG regimen in IGHV mutated CLL
1
Chronic Lymphocytic
Leukemia
Relapsed CLL Treatment
Nitin Jain, MD
Department of Leukemia
MD Anderson Cancer Center
Houston, TX
March 2018
Financial Disclosures
Research Funding
Pharmacyclics, Abbvie, Genentech, Infinity, BMS,
Pfizer, ADC Therapeutics, Seattle Genetics, Incyte,
Celgene, AstraZeneca, Servier, Verastem, Cellectis,
Adaptive Biotechnologies
Advisory Board
Pharmacyclics, Novartis, ADC Therapeutics, Pfizer,
Servier, Novimmune, Abbvie, Verastem, Adaptive
Biotechnologies, Janssen
Meets IWCLL criteria for treatment
‘Age’, Comorbidities, FISH status
Watch and Wait
>65-70 yr old,
major comorbidities del(17p)
FCR Ibrutinib
NO YES
Treatment-naïve Relapsed/Refractory
<65-70 yr and no
major comorbidities
1. Ibrutinib
2. Venetoclax
3. Idelalisib + R
Ibrutinib
Jain N, O'Brien S. Blood Rev. 2016;233-44.
Current Standard Rx of CLL

iFCG regimen in IGHV mutated CLL
2
•Approved therapies in R/R CLL
–BTK inhibitor Ibrutinib
–PI3K inhibitor Idelalisib + Rituximab
–BCL2 inhibitor Venetoclax
•Combination therapies in R/R CLL
•CART therapy in CLL
Presentation Outline
Targeting Key Signaling
Pathways in CLL
Friedman DR, Weinberg JB. The Hematologist. 2013.
CD20
571300080995
Obinutuzumab
Ibrutinib
ACP-196
BGB-3111 Idelalisib
IPI-145
TGR-1202
Fostamatinib
GS-9973
Membrane-bound immunoglobulin
B-cell receptor
Btk
Syk
NF-κB
pathway MAPK
pathway
PI3K/Akt
pathway
Normal B-cell activation and proliferation
Malignant B-cell initiation and progression
Lyn
Bcl-2
Venetoclax Cell survival
CD20
IBRUTINIB
BTK Inhibitor

iFCG regimen in IGHV mutated CLL
3
Ibrutinib (PCI-32765)
A Selective Inhibitor of BTK
•Forms a specific bond with cysteine-
481 in BTK
•Highly potent BTK inhibition at
IC50 = 0.5 nM
•Orally administered with once daily
dosing resulting in 24-hr target
inhibition
•In chronic lymphocytic leukemia
(CLL) cells promotes apoptosis and
inhibits CLL cell migration and
adhesion
N
NN
N
N
O
NH2
O
Advani, R. et al, J Clin Oncol. 2012;42:7906.
Honigberg LA et al, Proc Natl Acad Sci U S A.2010;107:13075.
Herman SEM et al, Blood.2011;117: 6287-6296.
Ponader, et al, ASH Meeting Abstracts. 2010; 116:45.
5-Year Experience With Ibrutinib Monotherapy
PCYC-1102/1103 Phase 2 Study Design
Patients with CLL/SLL
treated with
oral, once-daily ibrutinib
(420 or 840 mg/day)
Long-Term
Follow-Up
≥SD
*R/R includes patients with high-risk CLL/SLL,
defined as progression of disease <24 months after
initiation of a chemoimmunotherapy regimen or
failure to respond
Relapsed/Refractory*
(R/R)
n=101
Treatment Naïve (TN)
≥65 years
n=31
Phase 2 (PCYC-1102)
N=132
Extension Study
(PCYC-1103)
O’Brien et al. ASH 2016, Abstract 233
3% 3% 3%
55%
76% 71%
29%
10% 14%
0%
20%
40%
60%
80%
100%
5-Year Experience With Ibrutinib Monotherapy
Best Response
9
87% 89% 89%
Median DOR, months
(range) NR (0.0+ to 65.5+) 56.8 (0.0+ to 65.5+) NR (0.0+ to 65.5+)
Median follow-up,
months (range) 62 (1–67) 49 (1+–67) 56 (1+–67)
CR
PR
PR-L
TN (n=31) R/R (n=101) Total (N=132)
O’Brien et al. ASH 2016, Abstract 233

iFCG regimen in IGHV mutated CLL
4
5-Year Experience With Ibrutinib Monotherapy
Survival Outcomes: Overall Population
Median PFS 5-year PFS
TN (n=31) NR 92%
R/R (n=101) 52 mo 43%
Progression-Free Survival Overall Survival
Median OS 5-year OS
TN (n=31) NR 92%
R/R (n=101) NR 57%
O’Brien et al. ASH 2016, Abstract 233
5-Year Experience With Ibrutinib Monotherapy
Survival by FISH in R/R Patients*
**No del17p, del11q, del13q, or trisomy 12; in hierarchical order for del17p, and then del11q.
Progression-Free Survival Overall Survival
*Only 2 patients in the TN group showed PD or death. Subgroup analyses, therefore, focused on the R/R population.
Median OS 5-year OS
Del17p (n=34) 57 mo 32%
Del11q (n=28) NR 61%
Trisomy 12 (n=5) NR 80%
Del13q (n=13) NR 91%
No abnormality** (n=16) NR 83%
Median PFS 5-year PFS
Del17p (n=34) 26 mo 19%
Del11q (n=28) 55 mo 33%
Trisomy 12 (n=5) NR 80%
Del13q (n=13) NR 91%
No abnormality** (n=16) NR 66%
O’Brien et al. ASH 2016, Abstract 233
▪Primary Endpoint: PFS
▪Stratification according to:
–Disease refractory to purine analog chemoimmunotherapy (no response or <12 months)
–Presence or absence of 17p13.1 (17p del)
▪At time of interim analysis, median time on study was 9.4 months
RESONATE™ Phase 3 Study Design
R
A
N
D
O
M
I
Z
E
Oral ibrutinib 420 mg once
daily until PD or
unacceptable toxicity
n=195
IV ofatumumab initial dose
of 300 mg followed by 2000
mg x 11 doses over 24
weeks
n=196
1:1
Patients with
previously
treated
CLL/SLL Cross over to ibrutinib
420 mg once daily after
IRC confirmed PD (n=57)
Byrd J et al. NEJM 2014
iFCG regimen in IGHV mutated CLL
5
Progression-Free Survival
0 3 6 9 12
195 183 116 38 7
196 161 83 15 1 0
15
0
10
20
30
40
50
60
70
80
90
100
Progression-Free Survival (%)
No. at risk
Ibrutinib:
Ofatumumab:
Months
Ibrutinib
Ofatumumab
Ofatumumab
Ibrutinib
Median
time (
mo
)
8.08 NR
Hazard
ratio
0.215
(95% CI)
(0.146-0.317)
Log
-
rank
P value
< 0.0001
NR, not reached
Byrd J et al. NEJM 2014
Overall Survival
Ofatumumab
Ibrutinib
Median time (mo)
NR NR
Hazard ratio
0.434
(95% CI)
(0.238-0.789)
Log
-rank P value < 0.0049
Ibrutinib (n=195, 16 events)
Ofatumumab (n=196, 33 events)
NR, not reached
First patient cross over
Byrd J et al. NEJM 2014
Pattern of Response:
Blood Lymphocytes vs. Lymph Nodes
Lymph
node
burden
0 1 2 3 4 5 6 7 8 9 10 11
550
450
350
250
150
50
-50
ALC
Month Month
-100
-75
-50
-25
0
25
0 1
2
3 4 5 6 7 8 9 10 11

iFCG regimen in IGHV mutated CLL
6
IDELALISIB
PI3K-δInhibitor
•Targeted, highly selective, oral inhibitor of
PI3K-delta (δ)
•Inhibits proliferation and induces apoptosis
in CLL cells
•Inhibits homing and retention of CLL cells in
lymphoid tissues reducing cell survival
Expression Ubiquitous Ubiquitous Leukocytes Leukocytes
EC50 (nM) >10,000 1,419 2,500 9
a b gd
Class I PI3K
Isoform
Idelalisib, A Novel Small Molecule Inhibitor
Coutre et al. EHA 2014:S704
Phase 3 Trial of Idelalisib + Rituximab in Relapsed CLL:
Subgroup Analysis of High-Risk Groups
Arm A
N=110
Arm B
N=110
Rituximab (6 mo)
Rituximab (6 mo)
Randomization/
Stratification
Idelalisib (150 mg BID)
Placebo (BID)
Screen
Primary Study 116 Extension Study 117
Double-Blind
Initial Therapy
Double-Blind
Continuous
Therapy
Blinded
Dose Open-Label
Interim
Analyses and
Unblinding
Idelalisib (300 mg BID)
Idelalisib (150 mg BID)
Blinded,
Independent
Review
Independent
Review
Median Follow-up, months
IDELA + R PBO + R
1
st Interim Analysis 4 4
DMC
halted trial (Furman NEJM 2014)
2
nd Interim Analysis 6 5
Blind ended (
Coutre ASCO 2013)
•Arm A continues
•Arm B crosses over
Update
13 11
PFS, OS by subgroup analysis
Disease
Progression
Sharman et al., ASH 2014, Abstract 330

iFCG regimen in IGHV mutated CLL
7
0 2 4 6 8 10 1 2 14 1 6 18 2 0 2 2 2 4 2 6
0
2 0
4 0
6 0
8 0
100
T im e (m o nth s)
P ro gre ss io n -fre e S u rv iv al (% )
PFS, Including Extension Study*
Idelalisib + R vs Placebo + R
All Patients
*Placebo + R includes those patients who received open-label idelalisib after unblinding without prior progression (n=42).
Idelalisib + R (n=110) Placebo + R (n=110)
N at risk
IDELA
+ R
110
102
95 92 83 64 43 26 19 12 7 1 1 0
PBO + R
110
86 66 58 51 33 15 5 1 0 - - - -
Median PFS (95% CI) HR (95% CI) p-value
IDELA
+ R 19.4 mo (16.6, ‒) 0.25 (0.16, 0.39) <0.0001
PBO + R
7.3 mo (5.5, 8.5)
Sharman et al., ASH 2014, Abstract 330
Idelalisib + rituximab approved for
-R/R CLL
SAE, n (%) IDELA + R (N=110) Placebo + R (N=108)
Patients with any SAE 54 (49) 41 (38)
Pneumonia 10 (9) 11 (10)
Pyrexia 10 (9) 3 (3)
Febrile neutropenia 5 (5) 5 (5)
Sepsis 5 (5) 3 (3)
Pneumonitis 4 (4) 1 (1)
Pneum. jirov. pneumonia 3 (3) 1 (1)
Diarrhea 3 (3) 0
Hypercalcemia 2 (2) 2 (2)
Abdominal pain 2 (2) 1 (1)
Hypoxia 2 (2) 1 (1)
Colitis 2 (2) 0
Deep vein thrombosis 2 (2) 0
Sepsis syndrome 2 (2) 0
Neutropenia 2 (2) 0
Neutropenic sepsis 2 (2) 0
Lung infection 2 (2) 0
Transient ischemic attack 2 (2) 0
Serious Adverse Events In ≥2 Patients On IDELA + R
Colitis with PI3K-δInhibition
Okkenhaug et al. Science 2002.
p110δD910A/D910A
-IBD like picture
-Mucosal hyperplasia
-Crypt abscess
Clinical Management
-Late event (>6mos)
-Drug hold
-Oral/IV steroids
WT p110δD910A/D910A

iFCG regimen in IGHV mutated CLL
8
Idelalisib Summary
▪Approved for patients with relapsed CLL
in combination with rituximab
▪Immune-mediated colitis, transaminitis,
pneumonitis
▪EMA/FDA Advisory (March 2016)
–Increased deaths due to infections in Phase
III trials of idelalisib vs. placebo
–All patients PCP prophylaxis
–CMV monitoring
VENETOCLAX
(ABT-199)
Bcl-2 Inhibitor
Bcl-2 in CLL
▪Bcl-2 expression is uniformly high in CLL
▪ABT-199 is a selective, potent, orally bioavailable
Bcl-2 inhibitor
▪ABT-199 binds Bcl-2 with high affinity and with
substantially lower affinity to Bcl-xL, Bcl-w and
MCL-1
24
Souers et al. Nature Med. 2013
ABT-199
O NH
O
O2
S
H
N
N
N
Cl
NO2
O
NN
H

iFCG regimen in IGHV mutated CLL
9
CRR and ORR Rates by Subgroups
Variable
No. ORR %CR %
All
patients 116 79 20
17p deletion
31 71 16
Unmutated IGHV
46 76 17
Flu
- refractory 70 79 16
Prior Rx
≥ 4 56 73 16
Age
≥ 70 34 71 21
Nodes > 5
cm 67 78 8
Roberts et al N. Engl J Med 2016; 374; 311-22
Venetoclax
26
*coincides with first protocol specified CT scan at 6 weeks.
26
Data represents patients with lymphocyte count >5 x 109/L at
baseline
% Change from Baseline
-100
-75
-50
-25
0
25
200 800600 400 Safety Expansion150 400300 1200
Cohort Dose (mg)
Cohort Dose (mg)
Best Percent Change from Baseline in Blood
Lymphocyte Count and Nodal Mass by CT Scan
Nodal Mass by CT Scan (n= 93)
•The median time to 50% reduction 1.4 months, range [0.65 –
13.7]*
•78 (84%) evaluable patients had at least a 50% reduction in sum
of the product of diameters (SPD) of nodal masses
Blood Lymphocytes (n=60)
•Median Time to 50% reduction: 14 days, range [1 –49]
Seymour et al. EHA 2014, Abstract S702
27
•Median time to 50% reduction: 5.5 months, range [1.9 –17.4]*
•46/51 (90%) evaluable patients have had at least a 50% reduction.
Anti-tumor activity of ABT-199 was observed in all tumor compartments.
# Patient had 70% infiltrate at baseline and at Week 24
^Patient did not have CLL infiltrate at baseline.
Best Percent Change from Baseline in Bone
Marrow Infiltrate (n=51)

iFCG regimen in IGHV mutated CLL
10
Relapsed CLL
Combination Therapy: Future?
BCRi
+/-
BCL2i
+/-
CD20 mAb
Combined Venetoclax and Ibrutinib
for Patients with Previously
Untreated High-Risk CLL, and
Relapsed/Refractory CLL
A Phase II Trial
Nitin Jain, Philip Thompson, Alessandra Ferrajoli, Jan Burger, Gautam
Borthakur, Koichi Takahashi, Prithviraj Bose, Zeev Estrov, Elias Jabbour,
Marina Konopleva, Yesid Alvarado, Tapan Kadia, Musa Yilmaz, Courtney
DiNardo, Maro Ohanian, Jorge Cortes, Rashmi Kanagal-Shamanna, Keyur
Patel, Naveen Garg, Xuemei Wang, Nina Fru, Nichole Cruz, Varsha Gandhi,
William Plunkett, Hagop Kantarjian, Michael Keating, William Wierda
Department of Leukemia, MDACC
ASH 2017, Abstract 429
BCR vs. BCL2 Inhibitors
BCR Inhibitor
(Ibrutinib
)
BCL2 Inhibitor
(Venetoclax)
Response
Blood
++
LN +++
Marrow +
Blood
+++
LN ++
Marrow +++
Lymphocytosis
+++
-
CR in R/R CLL
10%
20
-25%
AE
profile
Atrial fibrillation,
neutropenia, bleeding
TLS, neutropenia
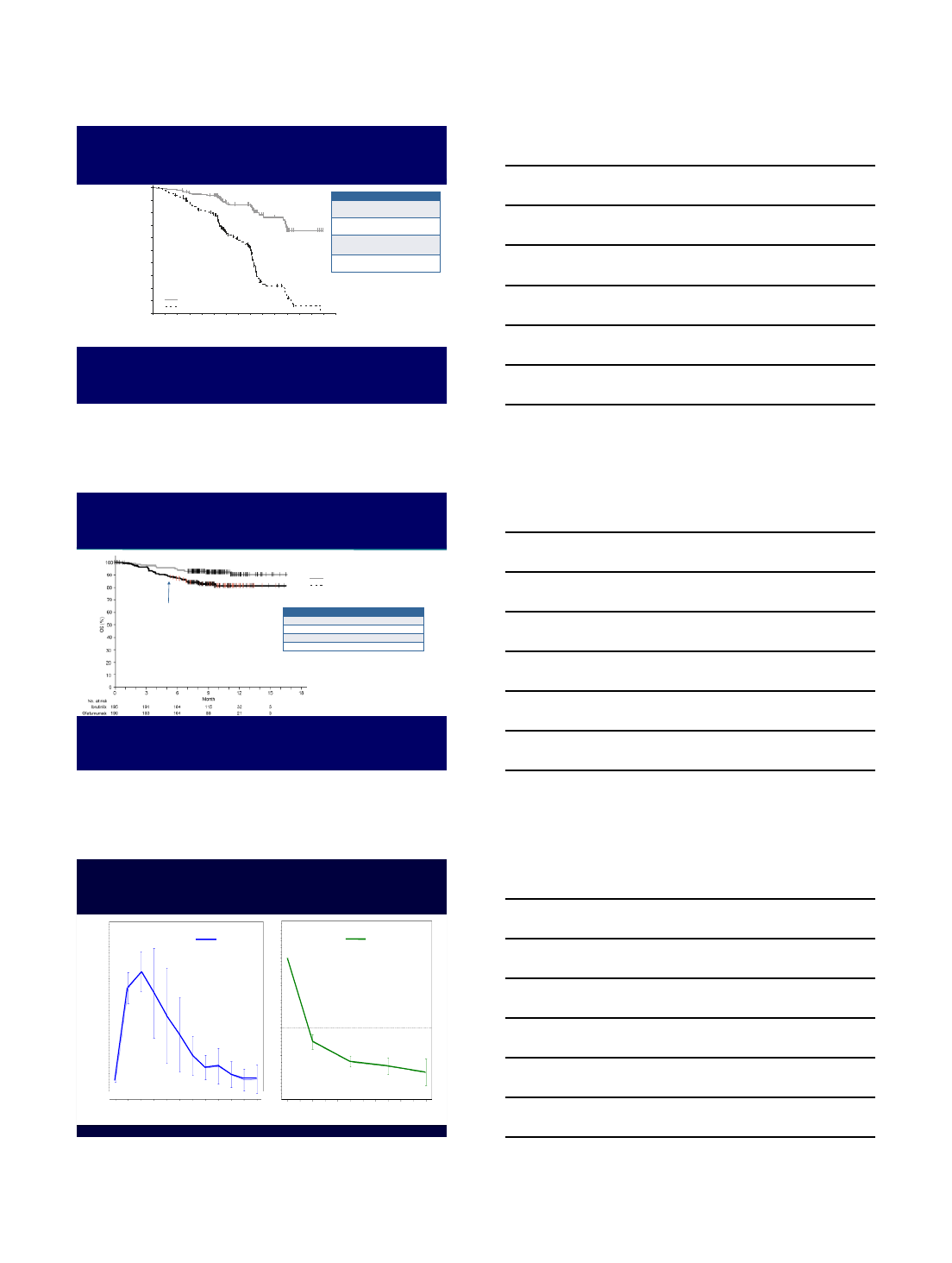
VEN + IBR in CLL, ASH 2017

iFCG regimen in IGHV mutated CLL
11
•Investigator-initiated phase II trial
•Patients with a diagnosis of CLL/SLL
•Cohort 1: relapsed/refractory CLL
•Cohort 2: untreated with at least one
high-risk feature
•del(17p) or mutated TP53
•del(11q)
•unmutated IGHV
•≥65 yrs
Ibrutinib + Venetoclax Clinical Trial
VEN + IBR in CLL, ASH 2017
Response: R/R Cohort
n=34 n=26 n=16 n=13 n=5
VEN + IBR in CLL, ASH 2017
91
58
31 23
3
42
69
77 80
0813 15
40
0
10
20
30
40
50
60
70
80
90
100
PR% CR/CRi % BM MRD neg %
3 mo IBR 3 mo VEN+IBR 6 mo VEN+IBR 9 mo VEN+IBR 12 mo VEN + IBR
20
Seymour, JD et al. ASH 2017, Abstract LBA-2

iFCG regimen in IGHV mutated CLL
12
iFCG regimen in IGHV mutated CLL
13
JCAR014 CART in High-risk R/R CLL
•24 pts with R/R CLL
•No of prior therapies 5
•Ibrutinib refractory 79%
•Venetoclax refractory 25%
•FDG-avid PET 93%
•Documented RT/PLL 33%
•Response at 4 weeks (JCAR014 + Cy/Flu)
•ORR 74%, CR 21%
•PET+ disease CR 64%
•Bone marrow disease 88% MRDneg
Turtle C et al. ASH 2016

iFCG regimen in IGHV mutated CLL
14
Conclusions
•Targeted therapies –great future in CLL
–Ibrutinib –Approved for CLL
–Idelalisib –Approved for R/R CLL
–Venetoclax –Approved for R/R del(17p) CLL
•Combination therapies are the future
Thank you!
njain@mdanderson.org

2/28/2018
1
1
Alexey V. Danilov MD, PhD
Associate Professor of Medicine
Oregon Health & Science University
Danilov@ohsu.edu
Ibrutinib intolerance and resistance
in CLL
2
Disclosures for Danilov
▪Research funding:
▪Leukemia & Lymphoma Society
▪Lymphoma Research Foundation
▪NCI/SWOG
▪Takeda Oncology
▪Gilead Sciences
▪Genentech
▪Consultancy/Honoraria:
▪Genentech
▪Verastem
▪TG Therapeutics
▪Astra Zeneca
▪Juno Therapeutics
▪Gilead Sciences
3
Post-ibrutinib world
▪Ibrutinib intolerance
–Role of other BTK inhibitors (acalabrutinib)
–Alternative pathway medications
–Factors predicting poor ibrutinib outcomes
▪Ibrutinib resistance
–Mechanisms (in CLL)
–What to do?

2/28/2018
2
4
Disposition
TN
(n=31)
R/R
(n=101)
Median time on study, months (range) 62
(1–67)
49
(1–67)
Patients remaining on ibrutinib therapy, n (%) 20 (65%) 30 (30%)
Primary reason for discontinuation, n (%)
Progressive disease
Adverse event
Consent withdrawal
Investigator decision
Lost to follow-up
1 (3%)
6 (19%)
3 (10%)
0
1 (3%)
33 (33%)
21 (21%)
5 (5%)
11 (11%)
1 (1%)
Ibrutinib clinical trials experience
O’Brien, Furman et al, ASH 2016
5
Ibrutinib ‘real world’ experience
Mato AR et al, ASH 2016
d/c rate = 42% after median f/u of 17 months
Toxicity: 50%
Disease progression: 10-20%
6
Reasons for Discontinuation
Front Line (%)
Relapsed (%)
arthralgias
(42)
atrial fibrillation (12.3)
Afib
(25)
Infection (11)
Rash (16)
Pneumonitis (10)
Bleeding (9)
Diarrhea (7)
Bleeding
8 months
Diarrhea
7.5 months
Atrial fibrillation
7 months
infection
6 months
arthralgia
5 months
pneumonitis
4.5 months
rash
3.5 months
Median Time to Discontinuation
Mato AR et al, ASH 2016

2/28/2018
3
7
Outcomes following ibrutinib failure
Mato et al, 2017
8
Outcomes following ibrutinib failures
Woyach et al, 2017
OS 4 months (Richters)
OS 22 months (progression)
Richters syndrome on novel agents:
The median OS for the entire cohort was 3.3 months
With a median follow-up 10.6 months, none of the 7 patients who achieved CR had died
Presented by: Matthew S. Davids, MD, MMSc

2/28/2018
4
Medical comorbidities assessed by CIRS negatively
impact survival in the era
of targeted therapies in CLL: a multicenter
retrospective analysis
Max J. Gordon MD1, Stephen M. Amrock MD SM1, Xavier Rivera3, Spencer James MD MPH2, Sudhir Manda
MD FACP2, Stephen E. Spurgeon MD1, Daniel Persky 3,Alexey V. Danilov MD PhD1
1Oregon Health & Science University, Portland, OR 2Geisel School of Medicine at Dartmouth, Hanover, NH
3University of Arizona, Tucson, AZ
Tolerance of ibrutinib
0
20
40
60
80
CIRS<7
CIRS≥7
≥1
0
0
20
40
60
CIRS<7
CIRS≥7
Yes
No
Relative risk P=
CIRS ≥7
11.9 <0.001
CIRS 3
-4 4.6 0.01
Dose reduction (n=21) D/C therapy (n=24)
Relative risk P=
CIRS ≥7
2.5 0.02
CIRS 3
-4 3.8 0.005
Outcomes with ibrutinib
➢Median 37 vs 23 months ➢OS at 24 months 100 vs 79%

2/28/2018
5
Ibrutinib is a CYP3A4 substrate
De Jong et al, 2015
Black dots –ibrutinib alone
White dots –ibrutinib PLUS
Acalabrutinib Monotherapy in Patients With
Ibrutinib Intolerance: Results From the Phase
1/2 ACE-CL-001 Clinical Study
14
Farrukh T. Awan,1Anna Schuh,2Jennifer R. Brown,3Richard R. Furman,4John M. Pagel,5Peter Hillmen,6
Deborah M. Stephens,7Ahmed Hamdy,8Raquel Izumi,8Priti Patel,8Min Hui Wang,8John C. Byrd1
1The Ohio State University Comprehensive Cancer Center, Columbus, OH; 2University of Oxford, Oxford, UK;
3Dana-Farber Cancer Institute, Boston, MA; 4Weill Cornell Medical College, New York Presbyterian Hospital,
New York, NY;5Swedish Medical Center, Seattle, WA; 6St. James’s University Hospital, Leeds, UK; 7University
of Utah Huntsman Cancer Institute, Salt Lake City, UT; 8Acerta Pharma, Redwood City, CA
Acalabrutinib (ACP-196)
•Acalabrutinib is a highly selective, potent BTK inhibitor
•Minimal off-target effects on TEC, EGFR, or ITK signaling in vitro
15
Awan F, et al. ASH 2016
Kinase Selectivity Profiling at 1 mol/L1
Kinase
Acalabrutinib
Ibrutinib
BTK 5.1 1.5
TEC 93 7.0
BMX 46 0.8
TXK 368 2.0
ERBB2 ~1000 6.4
EGFR >1000 5.3
ITK >1000 4.9
JAK3 >1000 32
BLK >1000 0.1
Kinase Inhibition IC50 (nmol/L)1
Larger red circles represent stronger inhibition
Ibrutinib
Acalabrutinib
1Covey AACR 2015. Abstract 2596.

2/28/2018
6
ACE-CL-001: Acalabrutinib Monotherapy in CLL
16
Awan F, et al. ASH 2016
–ACE-CL-001 is an ongoing, multinational, phase 1/2 study designed to evaluate
acalabrutinib monotherapy in patients with CLL/SLL.
–Previously reported ORR with acalabrutinib monotherapy:
•Relapsed/refractory: 95% (85% PR, 10% PRL; n = 60).1
•Treatment-naïve: 97% (87.5% PR; 10% PRL; n = 72).2
–Data are presented for 33 patients in the ibrutinib-intolerant cohort with data cut on
01 September 2016.
1Byrd JC, et al. N Engl J Med. 2016;374(4):323-332. 2Byrd JC, et al. ASCO 2016 [poster presentation].
Patient Disposition
17
Awan F, et al. ASH 2016
Disposition, n (%)
Treated
33 (100)
Discontinued treatment
9 (27)
Progressive disease 3 (9)
Adverse eventa3 (9)
Physician decisionb1 (3)
Otherc2 (6)
On treatment
24 (73)
aStroke (hemorrhagic)and fungal infection led to death (n = 1 patient each); metastatic endometrial
cancer (n = 1).
bConcurrent hemophilia.
c
Increase in BTK C481S mutation frequency in peripheral blood and central nervous system involvement
(n = 1 patient each).
•Median time on treatment: 12.2 months (range, 0.2-23.6 months)
Recurrence of Prior Ibrutinib-Related AEs
18
Awan F, et al. ASH 2016
Grade Change in Severity on Acalabrutinib vs on Ibrutinib
Adverse Event
Increased Decreased Unchanged
Arthralgia (n
= 1) 2 → 1
Atrial fibrillation (n = 1)
2 → 2
Contusion (n
= 1) 1 → 2a
Diarrhea (n = 2)
2 → 1
3 → 1
Ecchymosis (n = 1)
2 → 1a
Fatigue (n = 3)
1 → 2a2 → 1 1 → 1
Muscle spasms (n = 1)
1 → 1
Myalgia (n = 1)
1 → 1
Peripheral edema (n = 1)
1 → 1
Panniculitis (n = 1)
3 → 2a
Rash (n = 3)
3 → 1* 1 → 1
1 → 1a
Multiple occurrences of the same AE for a given patient were counted once for each Preferred Term.
aDetermined by investigator as related to acalabrutinib.

2/28/2018
7
Key Findings
•Acalabrutinib was well tolerated in ibrutinib-intolerant patients.
–A total of 12 of 33 (36%) patients experienced AE recurrence, most of which were
decreased or the same severity.
•No patients discontinued because of a recurrent AE.
•Acalabrutinib has promising activity in ibrutinib-intolerant patients.
–ORR: 79%
–81% of responding patients have a duration of response (PRL or better) ≥12 months.
–Median PFS has not been reached.
•Acalabrutinib efficacy in ibrutinib-intolerant patients is being evaluated in an
ongoing phase 2 trial (NCT02717611).
19
Awan P, et al. ASH 2016
20
Ibrutinib RESISTANCE
Woyach et al, 2014
Burger J et al, 2016
21
Mutations in BTK/PLCγprecede ibrutinib
RESISTANCE
Woyach et al, 2017

2/28/2018
8
22
After ibrutinib – what’s next?
▪Acalabrutinib (ibrutinib-intolerant only)
▪Alternative BTK inhibitors which do not bind C481S:
–GDC-0853
–SNS-062
–ARQ-531
▪Venetoclax
▪Idelalisib (Duvelisib, Umbralisib)
▪Other: PD-1; CART; CDK inhibitors (dinaciclib, voruciclib)
Venetoclax: Selective BCL-2 Inhibitor
23
Venetoclax Binds to and
Inhibits Overexpressed BCL-2
Venetoclax
BH3-only
BAK BCL-2 BCL-2
Mitochondria
An Increase in BCL-2
Expression Allows the
Cancer Cell to Survive
Mitochondria
Pro-apoptotic
Proteins
(BAX, BAK)
Anti-apoptotic
Proteins
(BCL-2)
2
1Apoptosis is Initiated
Apoptosome
APAF-1
Cytochrome c
Active
Caspase
Procaspase
Mitochondria
3
BAX
▪Venetoclax is a potent, orally bioavailable agent with a BCR-independent
mechanism of action and substantial activity in heavily pre-treated CLL
(Roberts AW et al, NEJM 2015)
Venetoclax Dosing Schedule
24
▪All patients
–had tumor burden assessment by imaging for nodal size and absolute
lymphocyte count at enrollment
–received prophylaxis for tumor lysis syndrome (TLS) with uric acid reducers
and hydration
▪Patients with high tumor burden were hospitalized prior to dosing to facilitate TLS
prophylaxis
▪Laboratory values were monitored for evidence of tumor lysis for at least 24 hours
after the first dose at each dose level
100 mg 200 mg
50 mg
20 mg
Week 5
Week 4
Week 3
Week 2
Week 1 400 mg venetoclax
400 mg
Until
progression
or
unacceptable
toxicity

2/28/2018
9
25
Venetoclax in IBRUTINIB resistance
Jones J et al, 2018
26
Venetoclax after ibrutinib therapy
Jones J et al, 2018
27
Venetoclax after ibrutinib therapy
Jones J et al, 2018
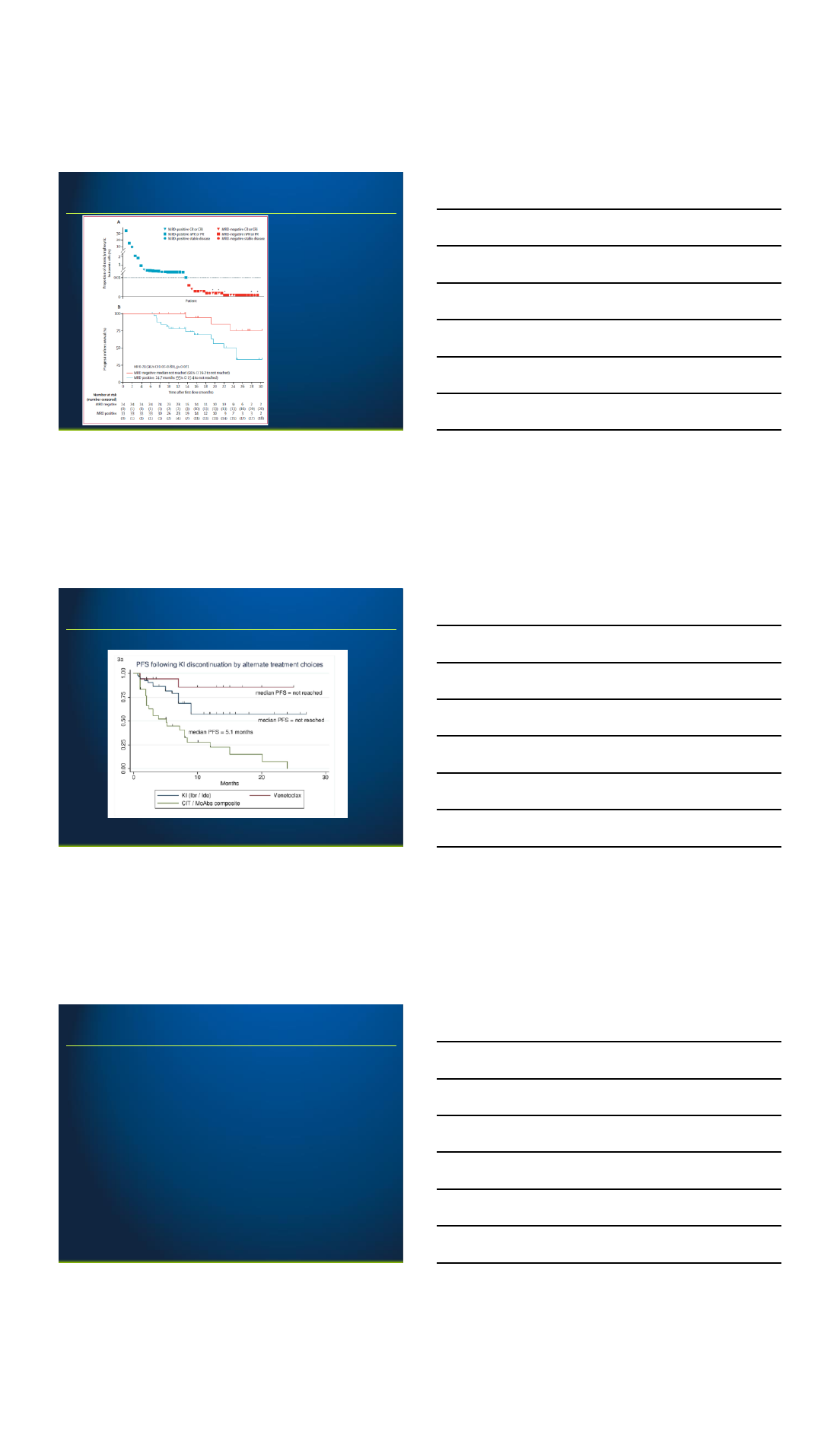
2/28/2018
10
28
Venetoclax after ibrutinib therapy
Jones J et al, 2018
42% blood MRD-negative;
5/13 bone marrow MRD-negative
29
Failed ibrutinib –what to do?
Mato et al, 2017
30
Summary
▪Ibrutinib intolerance and resistance is becoming a
common problem
▪Ibrutinib does not ‘protect’ from Richter’s
transformation
▪Second-generation BTK inhibitors may be a good option
for patients intolerant of ibrutinib
▪Venetoclax > PI3K in patients resistant to ibrutinib

3/1/2018
1
An update on Richter
Transformation
Dr. Philip Thompson
The University of Texas M.D. Anderson Cancer Center,
Department of Leukemia
Disclosures
•Pharmacyclics: Research support, Consultancy
•AbbVie: Research support, Consultancy
•Genentech: Consultancy
•Amgen: Research support, Consultancy
Background
•Richter Transformation (RT) is a transformation of
CLL to an aggressive lymphoma, most commonly
DLBCL and less commonly classical Hodgkin
Lymphoma.1
•Rare cases of transformation to plasmablastic
lymphoma, histiocytic sarcoma and other
uncommon lymphomas2
•Occurs in 2-10% of patients with CLL (0.5-1% per
year).
1Rossi, Blood 2011 2Jain, P, Oncology 2012.

3/1/2018
2
Risk factors for transformation
•Tends to be associated with high-risk CLL biology:
1. Unmutated IGHV1
2. Del(17p), del(11q), trisomy 122
3. Mutations in NOTCH1, which disrupt the PEST
domain3
•Possible association with fludarabine-based
chemotherapy.
1Rossi Blood 2011 2Strati CLML 2015 3Rossi Blood 2012.
Clinical features of transformation
•Non-specific.
•Fevers, drenching night sweats, weight loss.
•Rapidly enlarging lymph nodes.
•Elevated LDH. Sometimes hypercalcemia.
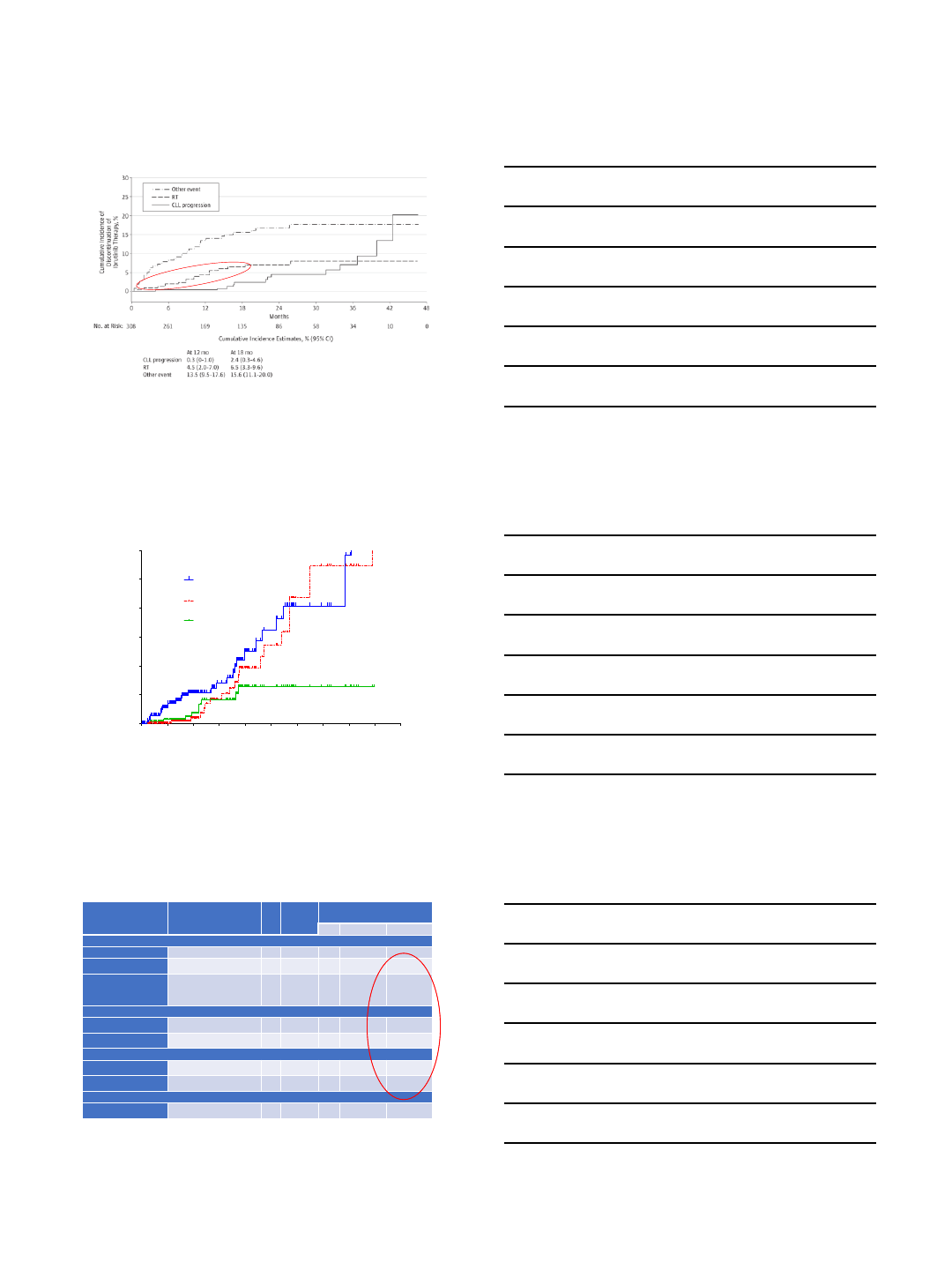
PET/CT as a screening tool

3/1/2018
3
PET/CT as a screening/prognostic tool
•332 MDA patients had PET/CT and subsequent biopsy.
Biopsy results classified as RT, histologically-aggressive CLL
(HAC) and histologically-indolent CLL (HIC).1
•SUVmax strongly correlated with histology: median 17.6 vs
6.8 vs 3.7 in RT vs HAC vs HIC, respectively.
•SUVmax ≥5 had NPV of 92% but only 38% PPV for RT. PPV
improved by cut-off of ≥10 SUVmax ≥10 had the optimal
discriminatory power for survival by ROC analysis. Median
OS was 56.7mo if SUVmax <10 and 6.9mo if ≥10.
•Interestingly, the negative prognostic impact of SUVmax ≥10
was identical, regardless of the histology.
•PET/CT also very useful in identifying optimal biopsy site
1Falchi, Blood 2014
Why biopsy?
•Not all that is hot on PET/CT is RT.
•Multiple other entities may mimic RT:
i) “Accelerated CLL” Important to distinguish as
treatment is different1
ii) EBV-driven lymphomas (eg. after treatment of CLL
with alemtuzumab, post-alloSCT. In contrast, RT is
usually EBV negative.2
iii) Herpetic lymphadenitits3, CMV, EBV.
iv) Nocardiosis, fungal infection (esp. histoplasmosis).
v) Granulomatous diseases (eg. TB).
1Gine Haematologica 2010 2Asano Blood 2009 3Joseph AJH 2001
From: Warnke RA, Weiss LM, Chan JK, et al. Tumors of the lymph nodes and spleen. Atlas of
tumor pathology (electronic fascicle), Third series, fascicle 14, 1995, Washington, DC. Armed
Forces Institute of Pathology.
RT DLBCL Histology

3/1/2018
4
Genomic features of RT
•Clonal relationship to underlying CLL: 80% of
DLBCL-RT arise from underlying CLL clone1,2
through acquisition of additional mutations.
•Genomically less complex than de novo DLBCL and
molecularly distinct:
1. RT frequently associated with TP53 mutations,
inactivating mutations in CDKN2A/B, MYC
overexpression, mutations in the PEST domain of
NOTCH1, stereotyped B-cell receptor.
2. De novo DLBCL frequently associated with BCL2
and BCL6 translocations (not seen in RT).
1. Rossi, Blood 2011. 2. Chigrinova, Blood 2013
Major genomic aberrations in RT
Chigrinova Blood
2013
Patients with ECOG >1 and TP53 disruption have poor outcomes.
Davide Rossi et al. Blood 2011;117:3391-3401
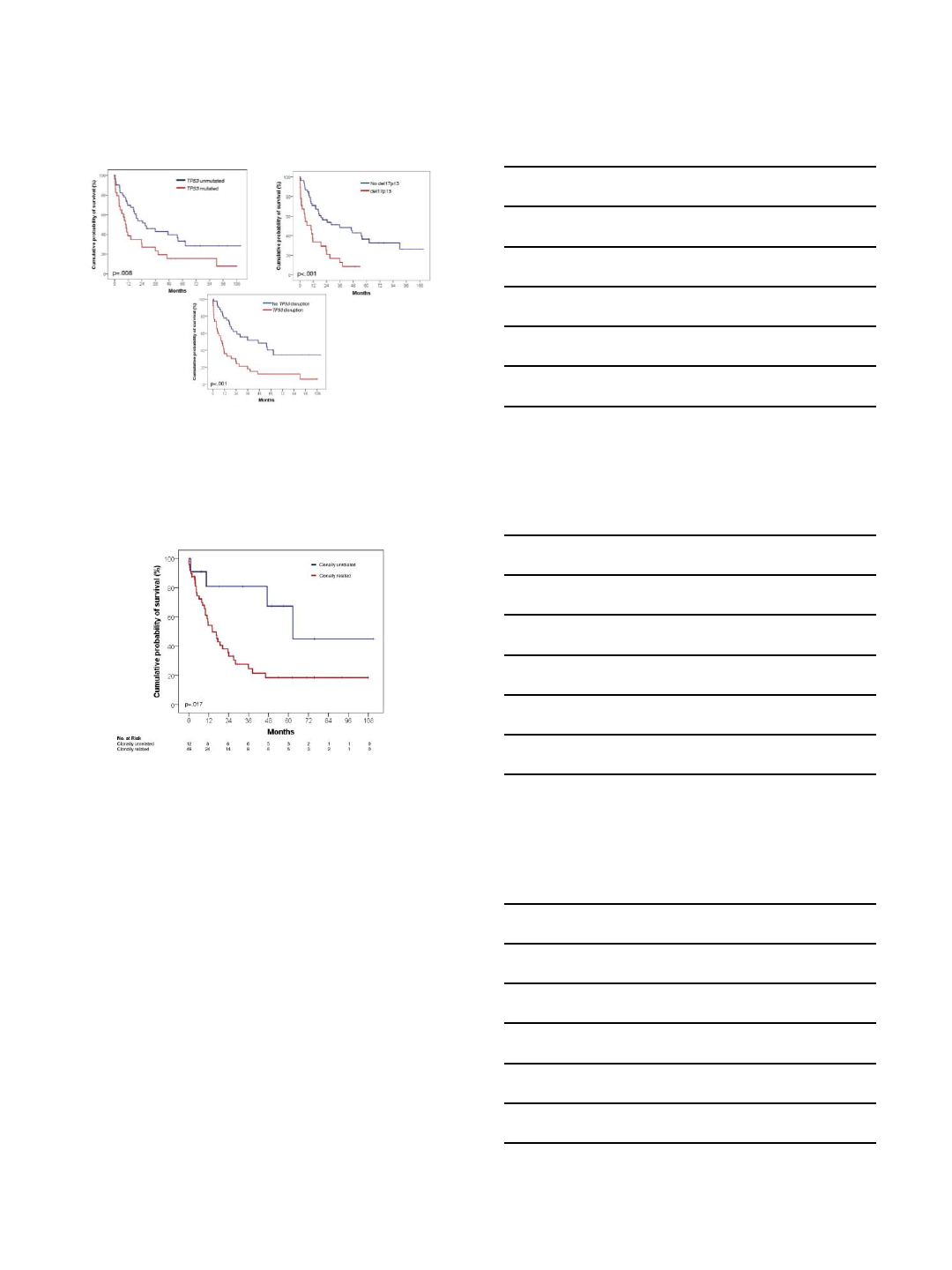
3/1/2018
5
Loss of functional p53 is a key prognostic marker,
regardless of the mechanism
Davide Rossi et al. Blood 2011
Clonally-unrelated “RT” should be thought
of as a different disease
Davide Rossi et al. Blood 2011;117:3391-3401
RT is still a frequent event during
targeted therapies for R/R CLL
•Ibrutinib ~5%.1,2
•Venetoclax up to 16%.3
•Transformation in these studies was an early event.
•Likely driven by disease biology rather than
treatment per se. These early phase studies were
enriched for patients with biologically high-risk
disease.
1Maddocks, JAMA Oncol 2015; 2Jain, Blood 2015; 3Roberts NEJM 2016
3/1/2018
6
Ibrutinib discontinuations over time
Maddocks, K et al. JAMA Oncology. 2015;1:80
MDA Data on ibrutinib discontinuation
0 6 12 18 24 30 36 42 48 54 60
0
5
10
15
20
25
30
Time (Months)
Cumulative incidence of discontinuation
Toxicity
Progression
Richters
Jain, P Cancer 2017
Prognosis of RT with chemoimmunotherapy
Study
Regimen
n
Median
age
(years)
Results
ORR
CRR
Median OS
Anthracycline
-containing Regimens
Jenke
et al, 2011
R-
CHOP
15
69 (N/A)
67%
7%
27 months
Dabaja
et al, 2001
HyperCVAD
29
61 (36
-
75)
41%
38%
10 months
Tsimberidou et al, 2003
Rituximab and GM
-
CSF with
alternating
hyperCVAD &
MTX/
cytarabine
30
59 (27
-
79)
43%
18%
8.5 months
Platinum
-containing Regimens
Tsimberidou et al, 2008
OFAR1
20
59 (34
-
77)
50%
20%
8 months
Tsimberidou et al, 2013
OFAR2
35
63 (40
-
81)
43%
8.6%
6.6 months
Fludarabine
-containing Regimens
Giles et al,
1996
PFA or CFA
12
59 (49
-
74)
45%
N/A
17 months
Tsimberidou et al, 2002
FACPGM
15
62 (42
-
74)
5%
5%
2.2 months
Radio
-Immunotherapy
Tsimberidou
et al,
2004
90
Y-ibritumomab
7
56 (44
-
70)
0%
0%
N/A

3/1/2018
7
Hematopoetic stem cell
transplantation
•Durable responses achieved in patients who have
achieved an objective response after
chemoimmunotherapy (CIT).
•3y OS 75% compared to 27% without transplant.1
•Prolonged survival also achieved after autoSCT.2
•Consolidative transplant should be offered to
patients who respond to CIT, with the exception of
patients who haven’t received treatment for CLL
and achieve CR with CIT.
1Tsimberidou, JCO 2006 2Cwynarski JCO 2012.
Treatment of Hodgkin variant
•Generally treated similarly to de novo HL (eg. ABVD
or BEACOPP).
•Outcome better than for DLBCL-RT, but worse than
for de novo HL.1,2
1Tsimberidou, Cancer 2006 2Brecher Am J Clin Pathol 1990
Novel therapeutic
strategies for RT

3/1/2018
8
Acalabrutinib
•2nd generation BTK inhibitor.
•29 patients with RT.
•Median 4 prior therapies.
•21% del(17p).
•ORR 38% (14% CR). Median duration of response
5.7mo.
•Median PFS of 3mo.
Hillmen, P. ASH 2016.
Pembrolizumab
•A phase II trial of pembrolizumab in relapsed and
transformed CLL included 9 patients with RT; ORR
in these patients was 44% (CR 11%, PR 33%).1
•For RT patients, the median progression-free and
overall survival was 5.4 months and 10.7 months
respectively.
•0/16 patients with CLL responded.
•PD-L1 expression on RT cells: potentially useful
biomarker for response.
1Ding Blood 2017
Novel therapeutics –ibrutinib + nivolumab
•Encouraging activity.
•Approximately 40% response rate in the first 15
patients1
•Rapid responses in RT.
1Jain, N. ASH 2017.
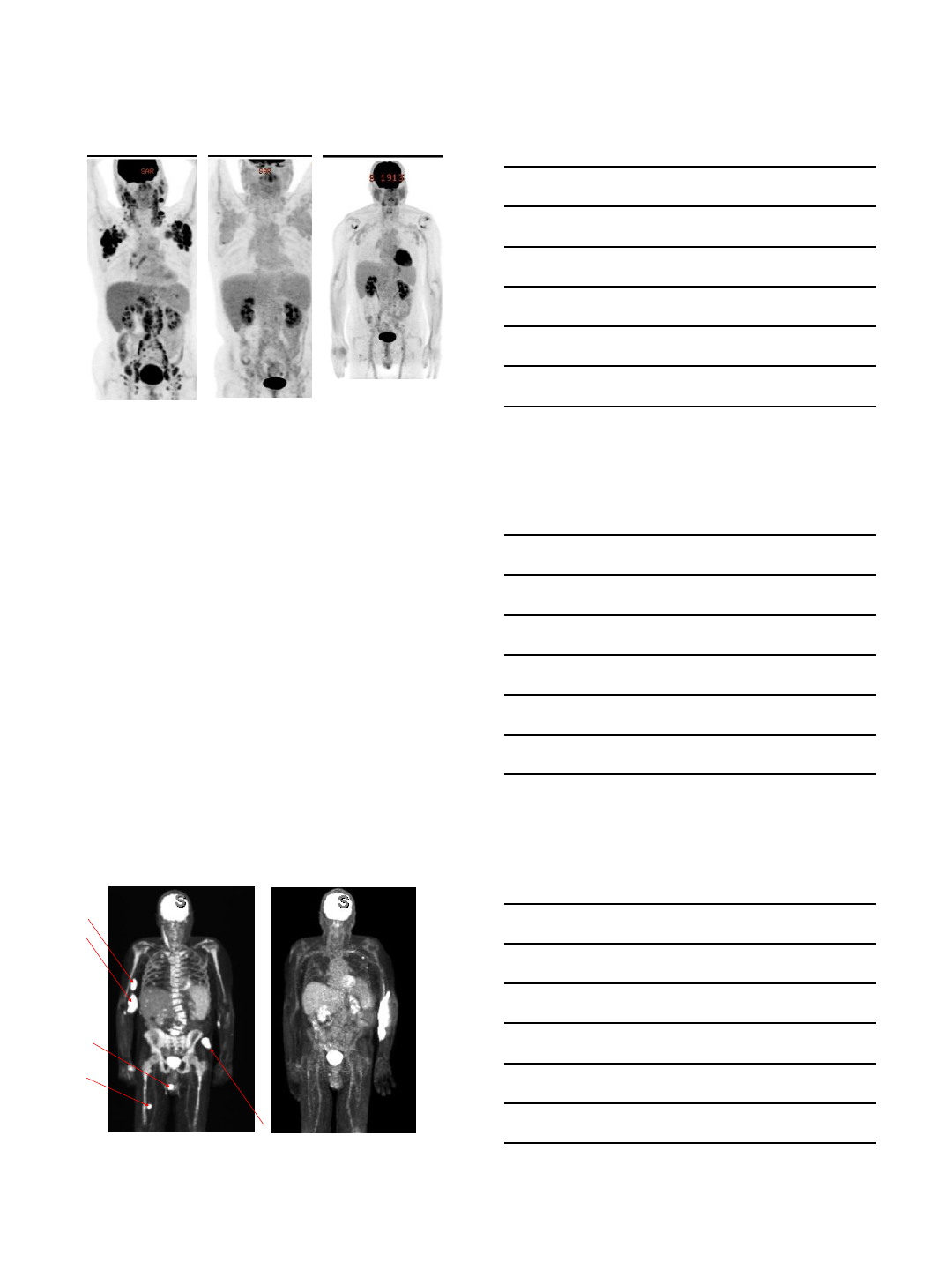
3/1/2018
9
Pre-treatment Post-1m nivolumab Post-3m nivolumab/2m
ibrutinib
Blinatumomab
•CD3x19 bispecific antibody, approved in ALL.
•In R/R DLBCL, overall response rate of 43% (CRR
19%), using high-dose therapy (max 112mcg/d).
•1 of first 3 patients achieved CR.
75M, developed RT on ibrutinib. Failed CIT, obinutuzumab and ibrutinib + nivolumab

3/1/2018
10
EPOCH-R + venetoclax.
•Multi-center study (DFCI, OSU, UCSD, us) of 20
patients.
•Addition of venetoclax to R-EPOCH.
•Some single-agent activity with venetoclax, but of
limited duration. Combination of bcl-2 inhibitor
with chemoimmunotherapy may prevent BCL2-
mediated resistance to chemotherapy-induced
apoptosis.
CAR T-cells
•KiTE CD19 CAR-T highly active in DLBCL. With in the
group of DLBCL patients in ZUMA-1 study, ORR was 79%
and CRR 52%. 3 patients had therapy-related death.
•Major toxicity. Grade 3 CRS in 13 cases. 28%
neurotoxicity (all reversible). 3 treatment-related
deaths.
•Despite the toxicity, this represents a major advance.
•Potential to be tested in RT.
•Turtle et al. reported 24 patients with CLL treated in
Seattle. Mostly ibrutinib-refractory. Many also
venetoclax refractory. ORR 74% (21% CR).
•Possibility to combine with checkpoint inhibition.
Khan and Thompson, Annals of Oncology 2017