Siemens Medical Solutions Department MED EH and S PERU098 Physiological Pulse Unit User Manual MAGNETOM Skyrafit Operator Manual MR System
Siemens Medical Solutions, Department MED EH&S; Physiological Pulse Unit MAGNETOM Skyrafit Operator Manual MR System
Contents
- 1. user manual
- 2. user manual II
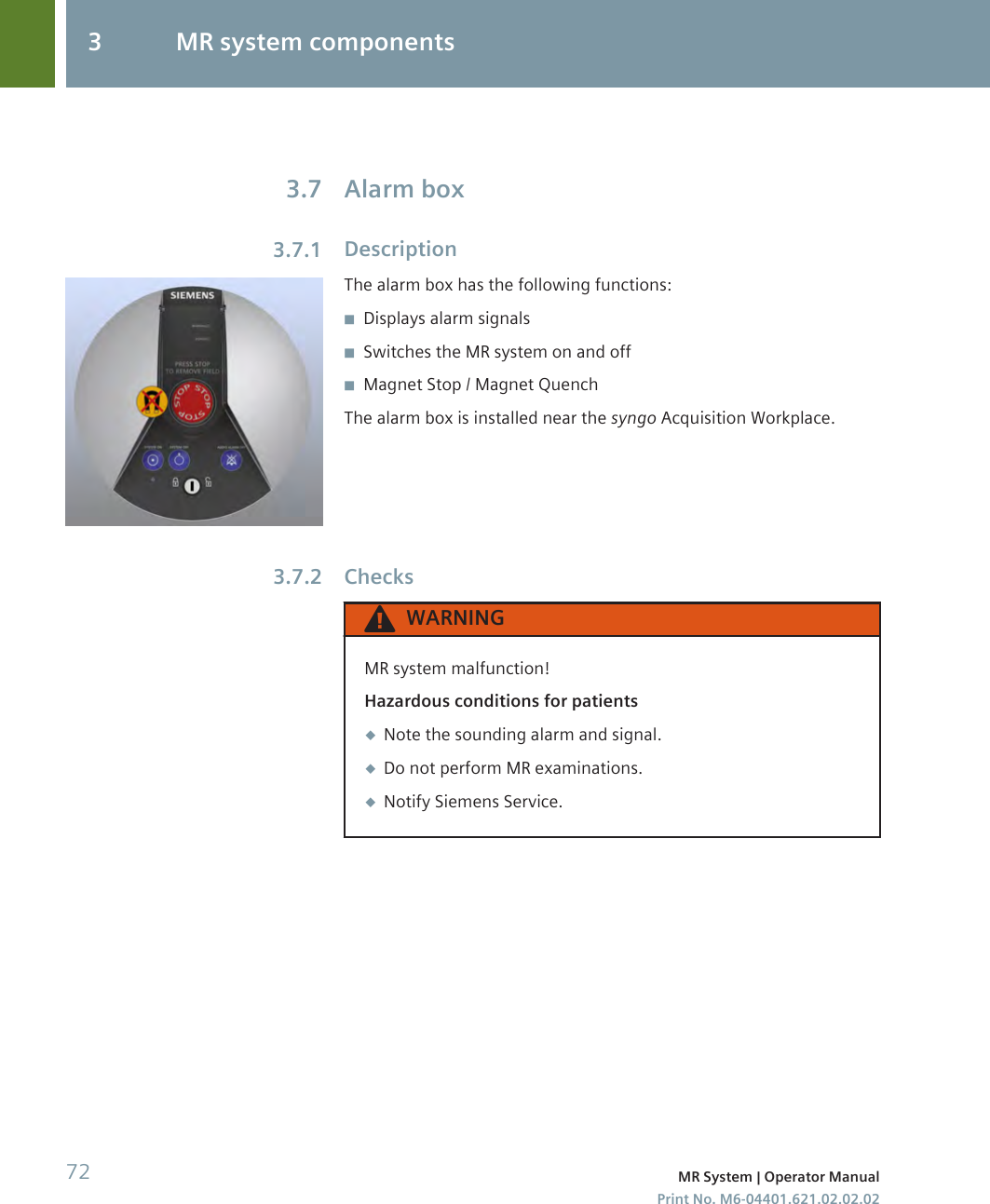
user manual















































![Make sure that no liquids such as contrast medium, blood, orcleaning agents get into the table connections.The following connections are located at the foot end of the patienttable:(1) Headphones(2) Vacuum cushion(3) Squeeze bulb(4) Placeholder [Intended for future use]The paper roll holder can be mounted at the foot end of the tablebelow the handle. It includes a holder to reposit the headphones andcan be removed if required. To replace the paper roll (disposable),you need to detach the headphone holder. If you remove the stick onthe paper roll, you can pull out the right mounting bracket. Then youcan replace the roll and let the bracket again lock into place.Operating the patient tableThe patient table can be controlled via the movement buttons andthe jogwheel on the larger control unit. ( Page 54 Control units)Please note that this section only describes the operation of thepatient table. For details on positioning the patient, see: OperatorManual CoilsConnectionsPaper roll holder3.4.2MR system components 3MAGNETOM Skyrafit | syngo MR E11 59Print No. M6-04401.621.02.02.02](https://usermanual.wiki/Siemens-Medical-Solutions-Department-MED-EH-and-S/PERU098.user-manual/User-Guide-3289130-Page-59.png)



























































![PPU 86Pulse triggeringDescription 95Performing 95QQuality AssuranceCoils 41RRepairSafety information 38Rescuing the patient in anemergency 62Respiratory triggeringDescription 96Performing 96ResponsibilitySafety guidelines 15Return and disposal 120RF and gradient fieldsSafety instructions 20RF coilsDescription 49SSafety guidelinesMR safety 15Responsibility 15Safety informationContraindications 23Emergency switches 41Maintenance/repair 38Preface 15SARLimit values; Safetyinformation 113Limits, high-frequencyfields 113Monitoring 113Signs and symbolsWarning and prohibitionsigns 38Squeeze bulbConnection 106StandbyStarting/ending Standby 105Static magnetic fieldDevice malfunctions 20Safety instructions 18Super-conducting magnetDescription 47syngo Acquisition WorkplaceDescription 49Host processor 50Keyboard 51Monitor 50Mouse 52syngo MR WPDescription 53Shutting down 104Starting up 102System controlControl units 54Description 53TTable StopSafety information 44Temperature controlSafety guidelines 113Tool barDisplay 56Triggering methodsOverview 83UUndockingDockable patient table] 66VVideo displayCleaning 117WWarming of body tissueRadio frequency fields 112Warning labelsWarning and prohibitionsigns 38MAGNETOM Skyrafit | syngo MR E11 127Print No. M6-04401.621.02.02.02](https://usermanual.wiki/Siemens-Medical-Solutions-Department-MED-EH-and-S/PERU098.user-manual/User-Guide-3289130-Page-127.png)



