Yuasa Yumicron Yua00Acc02 Users Manual
2015-02-03
: Yuasa Yumicron-Yua00Acc02-Users-Manual yuasa-yumicron-yua00acc02-users-manual-474212 yuasa pdf
Open the PDF directly: View PDF ![]() .
.
Page Count: 44


Part No. YUA1201501
Yuasa’s Automatic 12V 1.5 Amp battery charger incorporates superior 5 stage
charging technology. This high power unit provides error proof operation to fully
charge sealed maintenance free and conventional lead acid batteries
•Provides up to 16 volts to enable recovery of sulfated batteries during
pre-qualification stage
•28 day Charge Recycle – re-evaluates battery status for long term storage
•Designed to prevent overcharging
•AC power LED indicator
•Charging and float mode LED display
•Reverse polarity protection
•Spark-free operation
•Battery accessory leads and fused ring connectors included
•Steel construction for durability
•3 year limited warranty
Part No. YUA1200901
Yuasa’s Automatic 900mA 12V battery charger is the industry’s most powerful
plug-in wall charger. Combining the latest technology and a host of user-friendly
features, this compact, high-power unit provides optimum amperage return
and is designed to fully charge and maintain conventional and sealed
maintenance free batteries.
•Fully automatic three level charge cycle for effective charging
•Reaches 14.4 volts peak, then automatically switches to float
•Designed to prevent overcharging
•AC power LED indicator
•Charging and float mode LED display
•Reverse polarity protection
•Spark-free operation
•Durable construction for longer life
•Battery accessory leads and fused
ring connectors included
•12 ft. output cord
•3 year limited warranty
Yuasa SmartShot 900 Automatic 12V 900mA Battery Charger
Yuasa SmartShot Automatic 12V 1.5 Amp 5 Stage Battery Charger
BATTERY CHARGERS
UPGRADE
COMING
SOON

PREFACE:
A Practical Understanding of Lead Acid Batteries. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2
SECTION ONE: Battery Basics and Safety
About this book . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 3
About YUASA . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 3
Features and Benefits. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 4
The Lead Acid Battery . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 5-6
The Yuasa Battery Line. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 7
Yuasa’s Conventional Battery . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 7
YuMicron Battery . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 7
YuMicron CX Battery . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 8
Sealed (MF) VRLA and High Performance Batteries . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 8
Battery Safety . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 9
SECTION TWO: Selecting and Installing a New Battery
Selecting the Proper Battery . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 10
About Sensors . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 11
Battery Activation for Conventional and YuMicron Types . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 12
Activating Standard Batteries . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 12
SECTION THREE: Inspecting, Testing and Charging
The Discharge – Charge Cycle . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 13
Reasons for Self-discharge . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 14-15
Ampere-Hour and Cold Cranking Amps . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 16
Inspecting a Battery . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 16-17
Battery Testing Devices. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 17
Battery Testing. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 18
Chargers and Charging . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 19-20
Charging a New Standard Battery . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 20-21
Quick Charges . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 21
SECTION FOUR: Maintenance and Storage
Monthly Maintenance for Conventional and YuMicron Types . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 22
Storage . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 22
Sulfation and Freezing . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 22-23
SECTION FIVE: Sealed VRLA Batteries
New Generation Battery Technology. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 24
Sealed VRLA Battery Construction . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 24
Features . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 24-25
How a Sealed VRLA Battery Differs from Conventional Types . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 25
Activation and Installation . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 26
To Activate a Sealed VRLA Battery. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 26-27
Measuring Voltage . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 28
Discharge Characteristics. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 29
Self-discharge. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 30
Choosing a Charger . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 30
Charging a Newly Activated Sealed VRLA Battery. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 31
Routine Charging . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 32
Charging Instructions for Sealed VRLA Batteries with Voltage of 11.5 or Less . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 33
Routine Maintenance for Sealed VRLA Batteries. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 33
GLOSSARY
. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 34-40
CONTENTS
TABLE OF

Lead acid batteries are relatively simple in design.
Dissimilar metal plates are immersed in an electrolyte
solution consisting of sulfuric acid and water. These
are then insulated from each other with a permeable,
non-conductive material, which allows the transfer of
ions. The transfer of ions occurs during the discharge
and recharge of the battery. Also occurring is the change
in specific gravity or density of the electrolyte. During
the discharge period, sulfuric acid is drawn from the
electrolyte into the pores of the plates. This reduces
the specific gravity of the electrolyte and increases the
concentration of water. During the recharge, this action
is reversed and the sulfuric acid is driven from the plates,
back into the electrolyte, increasing the specific gravity.
During the discharge, lead sulfate is being formed on the
battery plates. Although this is the normal activity within
the battery during discharge, a timely recharge is
required to drive out the sulfuric acid into the electrolyte.
Without this recharge, the lead sulfate will continue to
develop and become difficult if not impossible to
breakdown during recharge. Once this advanced
sulfation develops, permanent capacity loss or total
failure of the battery is likely. Besides the sulfation
concerns, many other detrimental actions are taking
place inside the battery while in a discharged condition.
The corrosive effect on the lead plates and connections
within the battery is greatly increased due to the reduced
specific gravity of the electrolyte. The corrosion of the
plates will typically result in a gradual reduction in
performance followed by battery failure. The corrosion
associated with the inter cell connectors and the connecting
welds will in many instances result in a sudden battery
failure. The corroded connector may have sufficient
integrity to support low drain accessories such as lights
and instruments, but lack the necessary strength to
provide the high discharge current required to start
the vehicle. This corrosive effect can also dissolve the
lead into solution, which in turn may compromise the plate
insulators and result in micro shorts. Another condition
that frequently occurs in a discharged battery is freezing.
In a deeply discharged battery, the electrolyte has a
reduced specific gravity and becomes a higher percentage
of water than sulfuric acid. During this condition, the
battery may freeze at temperatures as high as 32°F.
The electrolyte in a fully charged battery will not freeze
in temperatures down to -65°F.
Deep discharge can be created by a multitude of
conditions, but the predominant reason is neglect.
During long periods of storage, the battery state of
charge must be checked and maintained per the battery
manufacturers recommendations. Other conditions that can
drain the battery are inoperative or inadequate charging
systems on vehicles, parasitic or key off drains, loose or
dirty terminal connections, etc. Although many of these
conditions can be corrected, often the problems you
cannot correct may be overcome by a periodic charging
schedule. You can establish a routine by which you check
and charge your battery or choose to permanently attach
a Yuasa Automatic Charger while the vehicle is not in use.
When charging your battery, always refer to the
instructions on both the battery and the charger. While
maintaining your battery at a full state of charge will
insure optimum life, overcharging may significantly
reduce it. With a conventional type battery that offers
access to the cell compartments, the periodic addition of
distilled water may be required. Water loss is normal in
these batteries through the process of electrolysis and
evaporation. Low electrolyte levels that expose the lead
plates to the air will result in permanent damage to the
battery. Maintain the electrolyte levels above the
minimum fill lines on the battery and at or below the
maximum line. A sealed VRLA (Valve Regulated Lead
Acid) battery should be maintained with the same care
as a conventional type battery with the exception of the
addition of distilled water. Sealed VRLA batteries
have a predetermined quantity of electrolyte added at
the factory or in the field using the acid bottle specified
for the battery. Once activated, the battery is permanently
sealed and must never be opened.
A little bit of care and understanding of how your battery
operates and is maintained will insure maximum service life.
A Practical Understanding
of Lead Acid Batteries
PREFACE
2

About this book
If you’re looking for more than everyday information about batteries, read on.
Maybe you’re a retailer, the expert whose battery knowledge and recommendations guide
customers every day. Perhaps you are a service technician or dealer – the person vehicle
owners turn to with questions. Or maybe you’re an enthusiast set on “knowing everything”
about your bike and how to keep it running.
Whatever your reason for wanting to boost your battery IQ, YUASA is pleased to provide
this copy of the ultimate battery book.
It’s filled with in-depth information: how batteries work, maintenance and installation tips and how
to get maximum power and life from your battery. We’ll talk about chargers and testers. Of
course, we’ll also fill you in on the complete line of YUASA batteries, chargers and accessories.
About YUASA
The first thing you need to know about batteries is YUASA. You might say that when it comes
to powersports vehicle batteries, we wrote the book! We’re the largest manufacturer and distributor
of small engine starting batteries in North America.
If you purchased a motorcycle, snowmobile, personal watercraft, ATV, riding mower or garden
tractor manufactured in the U.S., chances are the battery that starts it was made by Yuasa. In
fact, our batteries are original equipment in just about every major make of powersports vehicles.
If you’ve bought a replacement battery for your powersports vehicle, most likely it was made by
Yuasa. Altogether, we manufacture approximately three million batteries a year for small
engine starting applications at our Reading, Pennsylvania plant.
3
Battery Basics and Safety
1
SECTION

Features and Benefits
4
1
SECTION
Battery Basics and Safety
Component Features Benefits
Cast Grid Special Grid Design Severe vibration resistance & improved
conductivity
Active Material Proprietary Formulation Reduced shedding, maximum utilization, &
long service life
Pasted Plates Specified Thickness Ensures vibration resistance through
& Weight precise compression & proper service life
through specified active material balance
Dried-Charged Tank Formed & Properly dried plates in a sealed battery
Plates Dried Plates have a virtually limitless shelf-life, while
still retaining 70-80% state of charge when
filled with acid
Top Lead Through-Partition-Construction, Shorter current path for increased
Connections Large Straps electrical conductivity & higher starting
currents, heavier parts for
maximum durability
Case-Cover Thermal Bonded Plastic Air tight seal to prevent air ingress
Seal & acid seepage
Terminal- Cover Patented Poly-Seal Eliminates acid seepage, reduces
Seal Terminal corrosion, & extends battery life
Case & Cover Polypropylene Superior resistance to gasoline & oil,
impact resistant in extreme
weather conditions
Additive Sulfate Stop Sulfate stop added to reduce the solubility
(Some models) of lead into the acid, which then reduces
battery sulfation potential
YuMicron
Separator Leaf Separator Polyethylene leaf with glass mat sheet
bonded for compression to withstand
severe vibration conditions
YTX
Separator Absorbed Glass Mat Absorbs acid while creating cell compression
for extreme vibration resistance
Top Lead Alloy Calcium Alloy Increased weld strength & severe
vibration resistance
Vent System Valve & Flame Arrestor Valve relieves excess pressure to prevent
bursting, flame arrestor frit prevents
internal combustion
High Performance Maintenance Free
Grid System Computer-Designed Increased conductivity for higher starting
Radial Grids wattage, increased vibration resistance
Separator Puncture Resistant High puncture resistant separator for increased
AGM Material service in high vibration applications

Let’s look first at battery basics: what a
battery is and how it works.
Lead acid batteries are used as a power source for vehicles
that demand a constant and uninterruptible source of
energy. Just about every vehicle today does. For example,
street motorcycles need lights that operate when the
engine isn’t running. They get it from the battery.
Accessories such as clocks and alarms are battery-driven.
The Lead Acid Battery
Starting your vehicle depends on a battery.
Technically speaking, the battery is an electrochemi-
cal device that converts chemical energy to electrical
energy. The first thing you notice inside a battery is
the cells. Each cell has about two volts (actually,
2.12 to 2.2 volts, measured on a DC scale). A 6-
volt battery will have three cells. A 12-volt battery,
six cells.
HEAT SEALED CASE TO COVER
protects against seepage and
corrosion – bonded unit gives extra strength.
THRU-PARTITION CONSTRUCTION
provides shorter current path with less resistance than
“over the partition” construction – you get more
cranking power when you need it!
SPECIAL
ACTIVE
MATERIAL
is compounded to
withstand vibration,
prolong battery life
and dependability.
PATENTED
SEALED POST
prevents acid
seepage, reduces
corrosion – extends
battery life.
POLYPROPYLENE COVER
AND CONTAINER
gives greater resistance to gas and oil –
and impact in extreme weather conditions!
SPECIAL SEPARATOR
provides high cranking power.
HEAVY DUTY GLASS MAT
resists shedding of active material even
under severe vibration.
SPECIAL GRID
DESIGN
withstands severe vibration,
assures maximum conductivity.
Standard Features
5
1
SECTION
Battery Basics and Safety

What’s different? First there’s cranking power: YuMicron
has more because YuMicron batteries boost plate surface
area with thin, high-tech separators that make room for
two extra plates in each cell. YuMicron also has a special
intercell connector that minimizes resistance to further
maximize power. It has a special glass mat that resists
vibration damage.
Just for the record, let’s state how the Conventional and
YuMicron batteries aren’t different: they’re both lead-anti-
mony batteries, for openers (other batteries in YUASA’s
line, including the YuMicron CX, use lead-calcium tech-
nology). They have certain things in common that we
think should be part of every battery: like sealed posts to
resist corrosion, tough polypropylene covers and contain-
ers, and heat sealed construction for a strong, bonded
unit. And both share certain design features, like special
separators and through-partition construction.
Now, does all this mean YuMicron is automatically a bet-
ter choice than the conventional battery? Of course not.
It all depends on what you need to do. Some of the
YuMicron features might not be a big deal to a lawn trac-
tor owner, but a feature like our unique cover design
that minimizes electrolyte spillage is going to be really
important to the guy on a watersport vehicle or ATV.
Each YUASA line of batteries has its unique features
that account for differences in price and differences in
performance – and that’s what makes it the right battery
for a particular vehicle. Buy what you need. Don’t
pay for what you don’t need.
The cells consist of lead plates that are positive and
negative charged. Inside the cell they’re stacked alter-
nately – negative, positive, negative. Insulators or sep-
arators – usually fiberglass or treated paper – are
placed between the plates to prevent contact. Cranking
current increases as the plate surface area in the bat-
tery increases – the more plates in a cell, or the larger
the plates, the greater the current capacity (or flow of
electricity). Typically, capacity increases as the amount
of active material increases in the battery.
The alternate plates in each cell are connected at the
top into two groups, one positive and one negative.
Each cell’s groups of plates are then connected in series
– positive to negative – to those in the next cell.
Basically, that’s the internal hardware. Next, a solution
of sulfuric acid and distilled water – the electrolyte – is
added. And the action starts. A reaction between the
lead plates and the electrolyte sets off a chemical
change. This in turn creates the electrical charge in a
battery.
That’s the process, in a nutshell, that makes every battery
work. So, are all batteries the same?
Obviously not. Actually, there can be a number of differ-
ences, and they go far beyond things like box size or
terminal location. That‘s true for different brands, as well
as for different lines produced by the same manufacturer.
Take two types of YUASA batteries, for example: our
Conventional and YuMicron batteries.
POINTS TO REMEMBER
❐A battery converts chemical energy to electrical energy.
❐Each cell has approximately 2 volts: 3 cells for a 6-volt battery, 6 cells
for a 12-volt battery.
❐Inside each cell are electrically charged positive and negative lead
plates, isolated from each other by separators.
❐Chemical action between plates and electrolyte creates an
electrical charge.
❐Current is the flow of electricity.
6
1
SECTION
Battery Basics and Safety

The industry standard for motorcycles, snowmobiles and riding
mowers, our Conventional Battery is anything but conventional.
This workhorse is engineered to protect against seepage and corro-
sion... withstand vibration... and deliver high cranking power, even
when the weather’s dealing its worst. It’s the rugged, reliable and
dependable battery that customers are looking for!
These features are built into our conventional manifold vented battery...
and every battery in the YUASA line:
●Patented separators provide high cranking power
●Through-partition construction delivers
maximum power
●Unique sealed posts resist corrosion – for longer
battery life
●Polypropylene cover and container resist damage
from gas, oil, impact
●Heat-sealed, bonded unit construction protects against
seepage and corrosion
Personal watercraft, snowmobiles and ATVs make special demands –
and YUASA’s YuMicron Battery meets them head-on. The high-tech,
power-boosting design also makes YuMicron ideal for accessory-laden
touring bikes and modified machines.
●Heavy duty glass mat resists vibration damage
●Special thin YuMicron Separator packs in extra plates,
delivers up to 30% more cranking power than conventional types
●Through-the-wall intercell connector minimizes internal
resistance, maximizes power
●Sulfate Stop curbs plate sulfation – and provides longer life
The right battery for the right job – that’s where it all starts. YUASA’s comprehensive line has the right small engine
starting battery... whatever the vehicle, however tough the application.
YUASA’s Conventional Battery
The YUASA battery line...
7
YuMicron
1
SECTION
Battery Basics and Safety

Sealed (MF) VRLA and High Performance
Sealed (MF) VRLA (Valve Regulated Lead Acid) means a battery
that’s perfect for people who have better things to do than battery
maintenance! Our permanently sealed VRLA battery never needs
refilling; however, it still needs periodic charging. Ideal for motorcycles,
scooters, ATVs, riding mowers and personal watercraft.
●Spill-proof design means virtually no possibility of leaks
●Advanced lead-calcium technology pumps up
starting power
●Sulfation retardant dramatically reduces battery-killing
plate sulfation
●And, sealed VRLA batteries hold voltage
longer and need less charging in standby or storage mode
●High Performance version provides greater cranking
power with 12 extra plates per battery
For top power, less maintenance and longer life, YuMicron CX is the
battery of choice. The first motorcycle battery built on lead-calcium
technology, YuMicron CX is specially designed for today’s big, complex
machines, where higher cranking power is a must. It delivers all the
features of the standard YuMicron – plus...
●Unique CX design for higher cold cranking amps
●Lead-calcium technology reduces water loss – and
servicing – by 66% compared to lead antimony
●And, CX substantially reduces self-discharge – for
longer time between charges
YuMicron CX
1
SECTION
Battery Basics and Safety
8

As with anything, with batteries you have to know what
you’re doing. Batteries can be dangerous. But they don’t
have to be if some simple safety precautions are followed.
Basically, working with batteries poses two hazards:
potentially explosive gases that are given off during
charging, and sulfuric acid, which is very corrosive.
Here’s an 8-point list that’ll help keep those
hazards under control:
1. ABSOLUTELY NO SMOKING, SPARKS OR OPEN
FLAMES AROUND BATTERIES. Batteries can produce
hydrogen and oxygen; if they ignite the battery can rupture.
2. On conventional batteries, loosen vent caps when
charging and ventilate the entire charging area. A
build-up of hydrogen and oxygen levels in the battery
– or in the room where it’s being charged – can create
a hazard.
3. If a battery feels hot to the touch during charging,
stop charging and allow it to cool before resuming.
Heat damages the plates, and a battery that’s too hot
can rupture.
4. Never put the red sealing cap back on the
battery once you take it off. If you do,
gases trapped inside can explode. Make
sure the vent tube isn’t kinked or blocked,
for the same reason.
5. Properly connect charger to battery: positive to
positive, negative to negative. Unplug the charger or
turn it off before you disconnect the leads; that cuts
down on the chance of sparks.
6. Always wear eye protection, protective gloves and
protective clothing.
7. Clean up acid spills immediately, using a water
and baking soda solution to neutralize (1 lb. baking
soda in 1 gal. water).
8. Make sure acid container is clearly marked and
the work area is well lighted.
If sulfuric acid is swallowed or splashed in
the eyes, take immediate action. While the
diluted sulfuric acid used as electrolyte can burn the
skin, this type of injury is generally less serious.
Sulfuric acid in the eyes can cause blindness. Serious
internal injuries or death can result from ingesting sul-
furic acid.
Antidotes:
External – flush with water.
Internal – drink large quantities of milk or water,
followed by milk of magnesia, vegetable oil or
beaten eggs. Call a poison control center or
doctor immediately.
Eyes – flush for several minutes with water, get
immediate medical attention.
Battery Safety
POINTS TO REMEMBER
❐Ventilate battery charging area.
❐Charging gives off gases – no smoking, sparks or flames.
❐Safety glasses or face shields protect against eye damage.
❐Acid swallowed or in the eyes requires immediate antidotes and
medical care.
❐All safety considerations are important... review them frequently.
9
1
SECTION
Battery Basics and Safety

Selecting the Proper Battery
POINTS TO REMEMBER
❐Check current Applications Book or microfiche for right replacement battery.
❐There may be two or more “right” batteries – choose by performance needs.
❐Double check numbers before activation and installation.
High Maintenance
cc Model Year Performance Free YuMicron Conventional
Maintenance VRLA
Free
KAWASAKI
1000 KZ1000-P Police '02-'03 YTX20HL-BS YTX20L-BS - -
KZ1000-P Police '82-'01 YTX20HL-BS *YTX20L-BS *YB18L-A -
KZ1000-C Police '80-'81 YTX20HL-BS *YTX20L-BS *YB16L-B -
KZ1000-C Police, Z1-R '78-'79 YTX14AHL-BS - YB14L-A2 -
KZ1000, LTD '77-'80 YTX14AHL-BS - YB14L-A2 -
KZ1000-E ST, Shaft '79-'80 YTX20HL-BS *YTX20L-BS *YB16L-B -
KZ1000-G Classic '80 YTX14AHL-BS - YB14L-A2 -
KZ1000, LTD, CSR '81-'83 YTX20HL-BS *YTX20L-BS *YB18L-A -
KZ1000-R Replica '82-'83 YTX20HL-BS *YTX20L-BS *YB18L-A -
Z1000 '03-'04 - YTX9-BS - -
MOTORCYCLE
10
cranking power. And if the added benefit of never
adding water again appeals to you, go with the High
Performance Maintenance Free or the Maintenance Free
VRLA style batteries.
A few words of advice: always double-check that you
have the right battery for your application before you
charge and install it. If you have any questions, check
out our website at www.yuasabatteries.com or
contact us toll free at 1-866-431-4784.
Warning: In the event you want to
upgrade to a sealed VRLA battery,
please ensure you have the proper
charging voltage. Always refer to your
service manual.
Selecting the right battery is an important decision.
You’d be amazed how often the “problem” with a battery
is that it’s the wrong one for the application.
To make doubly sure you’re on track, you’ll need one of
two things – either the latest YUASA Battery
Specifications and Applications book, or the original
equipment (OE) microfiche. Of course, you can always
go to the old battery you’re replacing. The trick, though,
is to make sure it’s the original. Otherwise, you may be
simply repeating the same problem that caused the bat-
tery to need replacing.
OK, let’s say you’re replacing the battery on an '81
Kawasaki – a KZ1000-C Police, 1000cc.
Referring to the YUASA Battery Specifications and
Applications book, you first look under the Kawasaki
listing. Then find the right engine size – 1000cc, where
you find the KZ1000-C Police. You’re looking for an '81,
so the place to be is '80 to '81. The chart on this page
shows what it looks like.
If this were a sensor-equipped battery – which it isn’t –
the applications book would mark it with a footnote (†).
That tells you to order it with a sensor.
What’s the right battery? You’ll see there are three of them:
a High Performance Maintenance Free YTX20HL-BS, a
Maintenance Free VRLA YTX20L-BS, and a YuMicron
YB16L-B battery. Any of these will do fine. If your
machine has increased compression modifications to the
engine, for example, you might want the additional
2
SECTION
Selecting and Installing a
New Battery
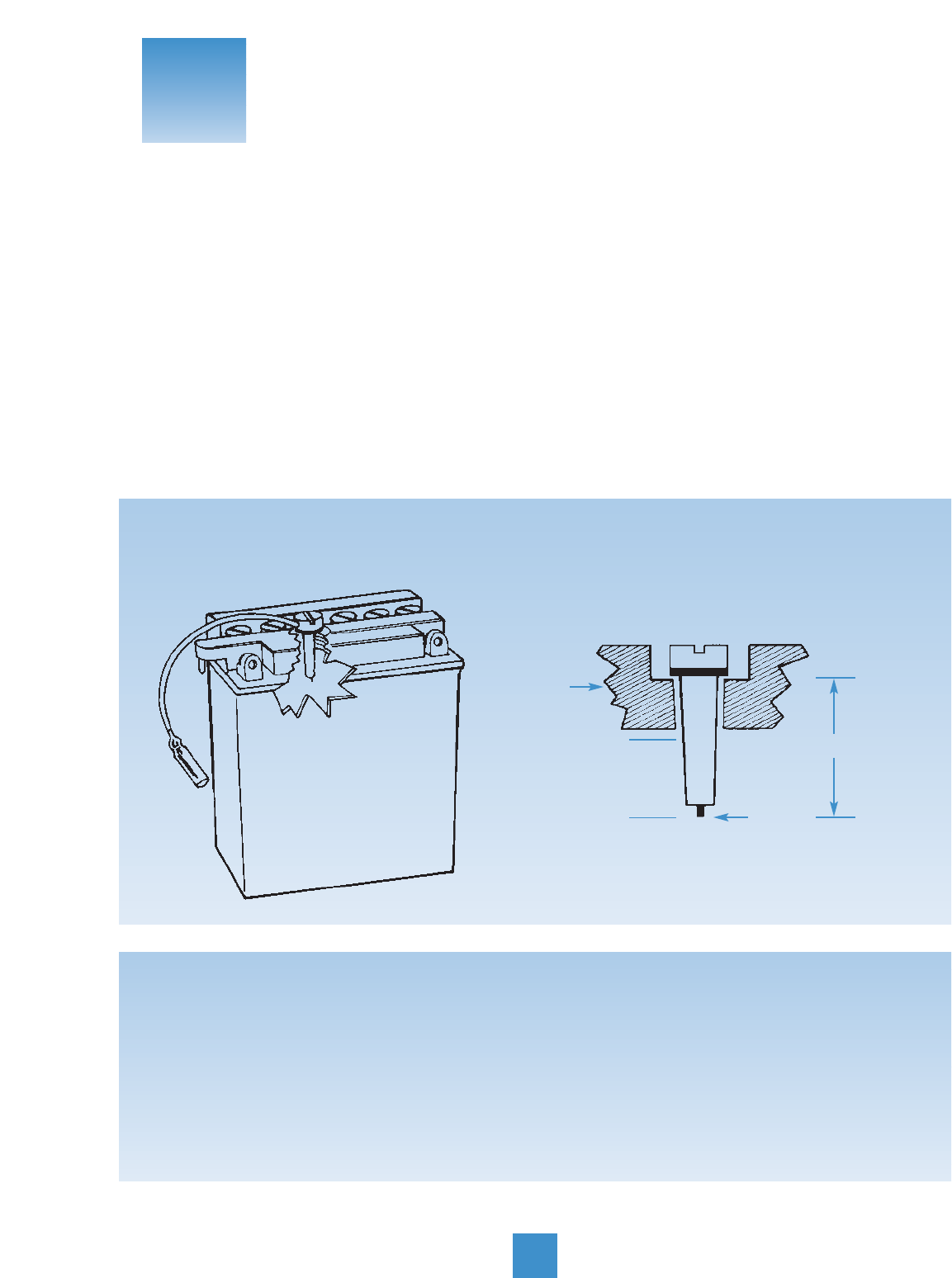
About Sensors
Many of today’s motorcycles use batteries equipped
with sensors. They’re either built in the battery, or
packed with it.
A sensor is a “low fuel” warning light. It tells you when
you’re getting low – in this case, on electrolyte. The
sensor causes a warning light to flash, signaling that
it’s time to add water to the battery. The cutaway views
below show what a sensor looks like.
Sensors are sort of particular: they don’t go with just
any battery. Which means it’s important to replace the
old battery and sensor with the correct YUASA sensor
battery listed in the applications book. So, sensor rule
one is this: replace both battery and sensor at the
same time.
“But the sensor’s original equipment,” you say. Doesn’t
matter. Being OE doesn’t mean it ‘s OK in another man-
ufacturer’s battery. In fact, OE sensor plugs vary consid-
erably in length, size and diameter. A plug that’s too
long can short out a battery and mess up the electrical
system. If the plug’s short, the warning light will flash
way too early.
Note, too, that even YUASA’s sensor batteries are not
interchangeable; they have different vent locations, sen-
sor wire lengths and diameter of cylinder connectors.
POINTS TO REMEMBER
❐Replace battery and sensor at same time.
❐Original equipment sensor isn’t “OK” for a new battery.
❐Sensor batteries and sensors are not interchangeable – check
Applications Book.
COVER
MAX
MIN
ACID LEVEL
LEAD
CUT-AWAY VIEW OF SENSOR
LENGTH OF SENSOR
11
2
SECTION
Selecting and Installing a
New Battery
3. Let battery stand for at least 30 minutes.
Move or gently tap the battery so that any air bubbles
between the plates will be expelled. If acid level has
fallen, refill with acid to upper level.
4. A battery must be completely charged before
installation. Charge for three to five hours at the current
equivalent of 1/10 of its rated capacity found in the
Yuasa Applications Book.
5. During charging, batteries can spit electrolyte out
the open vent. Take care to loosely refit vent caps.
6. Check during
initial charging to see
if electrolyte level has
fallen, and if so, fill
with acid to the
UPPER LEVEL. After
adding, charge for
another hour at same
rate as above to mix
water and acid. Note: this is the last time electrolyte
should be added, but distilled water should be added
as required.
7. When charging’s done, replace plugs
firmly. Do not apply excessive pressure.
Finger tighten only. Do not over-tighten.
8. Wash off spilled acid with water and baking soda
solution, paying particular attention that any acid is
washed off the terminals. Dry the battery case.
Battery Activation for Conventional and YuMicron Types
Sealed at the factory, a new YUASA battery has an
indefinite shelf life as long as it remains sealed, with
the red cap in place, and is stored at room tempera-
ture. Once it’s unsealed, a battery should be activated,
charged and installed. The plates of an unsealed,
uncharged battery begin to oxidize. That makes it
more difficult to charge later. (We talk more about
sulfation later in this book.) And if it’s charged and sits
around, it starts to discharge and sulfate; how fast
depends on temperature.
Here are the steps for activating most batteries,
including YUASA’s Conventional, YuMicron, YuMicron
CX. (Sealed VRLA – YT or YI – batteries are activated
differently, see page 26.)
Activating Standard Batteries
1. Right before adding electrolyte, remove
filling plugs. Also remove the sealing tube – the red
cap – and throw it away. (Putting this cap back on after
the battery’s filled with acid can cause an explosion.)
2. Place battery on a
level surface. Fill battery
with electrolyte (a sulfuric
acid dilution with a specific
gravity of 1.265). Do not
use water or any other liquid
to activate.
Electrolyte should be between 60°F and 86°F before
filling. Fill to UPPER LEVEL as indicated on battery.
NOTE: Never activate a battery on the vehicle.
Electrolyte spillage can cause damage.
YB2.5L-C 2.5 3 3/16 2 3/4 4 1/8
YB2.5L-C-1 2.5 3 3/16 2 3/4 4 1/8
YB3L-A 3 3 7/8 2 1/4 4 3/8
YB3L-B 3 3 7/8 2 1/4 4 3/8
YB4L-A 4 4 3/4 2 3/4 3 5/8
YB4L-B 4 4 3/4 2 3/4 3 5/8
YB5L-B 5 4 3/4 2 3/8 5 1/8
CAPACITY DIMENSIONS – INCHES
BATTERY AH (± 1/16 IN)
TYPE (10H-R)
LWH
YUMICRON Battery Specifications
Rated
Capacity
12
2
SECTION
Selecting and Installing a
New Battery

As we mentioned earlier, an electrochemical action
within the battery produces electricity. To understand
it, let’s look inside a battery again: you’ll see cells
made up of lead plates. Some plates are positive
charged. Others are negative charged. There’s also
the electrolyte – a sulfuric acid solution that conducts
the current. It sets off the chemical process that takes
place in the battery.
So what goes on when a battery discharges?
The electrolyte reacts chemically with the lead plates –
and it’s not exactly a match made in heaven: it turns
them into lead sulfate. If sulfate reminds you of sulfa-
tion, you’re right on target: this build-up of sulfate
crystals is exactly what battery-killing sulfation is.
In the process, the electrolyte – which contains hydrogen,
sulfur and oxygen – gives up its sulfur and some of its
oxygen. The electrolyte turns to water. Now you
know why a discharged battery – filled with water
instead of electrolyte – can turn into an ice block when
the temperature drops even into the 20s. On the other
hand, a properly-charged battery won’t freeze until the
mercury gets way down in the minus range.
The chemical process causes free electrons to slowly
gather on the negative plates. They just hang there
until a load is placed on the battery – a light or starter’s
switched on – which causes a swarm of electrons to rush
to the positive plates.
If the chemical process just went on and on,
unchecked, the lead plates would soon turn totally to
lead sulfate, the electrolyte would become pure water,
and the chemical and electrical activity inside the bat-
tery would come to a standstill. It’s bad for a battery.
So is allowing a battery to remain discharged for a
prolonged period. Recharging becomes hard or
impossible.
The good news: except in extreme cases, the process of
discharging can be reversed. You work that magic by
putting a larger voltage on the battery – for example,
14v on a 12v battery. That’s charging.
Here’s what goes on when a battery charges:
The electrical charge flowing back in causes the lead
sulfates to send their sulfate back into the electrolyte.
As a result, both the electrolyte and the plates return to
their original composition.
You’ll notice bubbles in an actively-charging battery.
That’s called gassing. It occurs because hydrogen and
oxygen gases are liberated as the charging current
breaks down the water.
Several things are actually happening here. The
process breaks down water into hydrogen and oxygen
vapor, which escapes out the vent tube. You have to
replace that loss. Add distilled water to each cell after
charging. Then give the battery a “mixing charge” for
another hour. The hydrogen and oxygen gases that
are given off can also build up pressure in the battery –
which is why batteries are vented, and why the vent
tubes can’t be bent or blocked. Very importantly,
hydrogen and oxygen are very explosive. It bears
repeating that sparks, flames and cigarettes around
charging batteries can be a one-way ticket to trouble.
POINTS TO REMEMBER
❐Deep discharge or prolonged discharge leads to harmful sulfation.
❐A discharged battery freezes much faster than a charged battery.
❐Charging can reverse discharging.
❐Charging gives off hydrogen and oxygen, which are explosive.
The Discharge – Charge Cycle
13
3
SECTION
Inspecting, Testing
and Charging

Batteries have a natural tendency to discharge. There
are a number of reasons why: self-discharge, high
temperatures, drain from electrical accessories on a
vehicle, and short trips that aren’t enough to recharge
the battery.
Self-Discharge: Self-discharge goes on all the time.
It’s a battery fact of life that they get weaker from “just sit-
ting.” How rapidly batteries self-discharge depends, first
of all, on battery type. Lead-calcium batteries, such as
YUASA’s CX, YT, YI, discharge more slowly than conven-
tional batteries. At room temperature lead-calcium dis-
charges at 1/300 volt per day. Conventional lead-anti-
mony batteries discharge at 1/100 volt per day.
Temperature: Outside temperature plays a big part,
too. As the mercury goes up, batteries discharge faster.
Particularly in hot climates, that can mean trouble: every
18°F doubles the discharge rate, so a battery at 95°F
discharges twice as fast as one at 77°F. And temperatures
Keep in mind that at the same time accessories are draw-
ing on the battery, the battery’s self-discharging, too. The
charts below show how fast a battery is discharged by
self-discharge and by current drain:
Motorcycle
Battery Multimeter
Checking Current Drain
Self-discharge
Approx. Number of Days From100%
Charged to 100% Discharged
Temperature Lead-Antimony Lead-Calcium
Battery Battery
104
°
F 100 Days 300 Days
77
°
F 200 Days 600 Days
32
°
F 550 Days 950 Days
104°F
77°F
32°F
0 100 200 300 400 500 600 700 800 900 1000
Days Stored
From 100% Charged to 100% Discharged
Temperature
Self-discharge – Temperature Comparison
Lead-Antimony Lead-Calcium
Reasons for Self-discharge
14
3
SECTION
Inspecting, Testing
and Charging
Negative
Battery
Cable
20
40
60
80
100
2 4 6 8 10 12 14 16 18 20
Months on Storage Shelf
Amount of Self-discharge (%)
Lead-Antimony
Lead-Calcium
Self-discharge Rate Characteristics at 77OF
0
of 130°F are battery-killing. Been in a closed-up garage
or storage building on a hot summer day recently? In
many parts of the country, it’s no trick for inside tempera-
tures to reach that.
Accessories: Electrical accessories on some of today’s
newer and bigger bikes – clocks and computer memory,
for example – will discharge the battery continuously,
even when the ignition’s off. The drain can be consider-
able. You can find out the drain, in milli-amperes, by
disconnecting the negative terminal and putting a multimeter
in line. It should look like this:
Current Drain (Y50-N18L-A)
Days From Days From
Discharging 100% Charged to 100% Charged to
Ampere 50% Discharged 100% Discharged
7 mA 60 Days 119 Days
10 mA 42 Days 83 Days
15 mA 28 Days 56 Days
20 mA 21 Days 42 Days
30 mA 14 Days 28 Days

Let’s see what happens as the two work together to
discharge a battery:
The battery, starting out 100% charged, has a 30mA
discharge rate from electrical accessories on the
motorcycle.
At an average outdoor temperature of 77°F a lead-
antimony battery loses about half its capacity in only
12 days due to the combination of self-discharge and
current drain. In another 12 days, it’s completely
dead. In other words, it doesn’t take long for the dou-
ble whammy of self-discharge/accessory drain to
knock out a battery for good.
If current drain is measurable when the motorcycle is
turned off, you can do one of two things: disconnect
the battery when the vehicle is in storage, or charge the
battery every two weeks to a full charge. However,
cycling – or continually recharging the battery – will
shorten its life. Check the battery’s condition with
either a hydrometer or voltmeter (or a multimeter). The
section on Testing a Battery has details.
Short Trips: What if you use the vehicle now and then –
a couple of times a week for errands, or even daily for
a short trip to work?
You can’t assume that occasional use or
short trips (under 15 or 20 miles) will keep
the battery charged. In fact, they’re probably going
to add to drain, because the bike’s charging system
doesn’t have enough time to make up for losses from
normal starting and self-discharge. You’re going to
have to charge the battery more often. Maybe every
month or so, depending on temperature.
Does the surface the vehicle’s parked on, or a battery’s
sitting on, contribute to how well it holds a charge?
You sometimes hear “experts” say parking on concrete
will accelerate discharge. Bet them it’s not so. Then
collect. Concrete, macadam, wood, dirt, stones, sand
– makes no difference. A battery discharges at the
same rate, no matter what surface it’s on.
And here’s a hint: if a battery suddenly dies and there’s
no apparent reason for it, check the electrical system
before you buy a new battery. One of life’s little let-
downs is to shell out dollars for a new battery when
you didn’t need to – and then still have the problem.
POINTS TO REMEMBER
❐Conventional lead-antimony batteries discharge @ 1/100 volt per day.
❐Lead-calcium batteries discharge more slowly @ 1/300 volt per day.
❐Higher temperatures mean faster discharge.
❐Temperatures over 130°F kill batteries.
❐Self-discharge and short trips cause drain.
❐The more electrical accessories you add to a bike, the greater the
current drain.
15
3
SECTION
Inspecting, Testing
and Charging
Current Drain
12 Days x 0.030 Amps x 24 Hours = 8.64 AH
8.64 AH
÷
20 AH (Y50-N18L-A) = 43.0% Available Capacity
PLUS (+)
Self-discharge
1
2 Days
÷
200 Days to 100% Loss = 6.0% Available Capacity
Total Loss
Current Drain
43.0% Available Capacity
Self-discharge +
6.0% Available Capacity
Loss in 12 Days
49.0% Available Capacity

Ampere-Hour and Cold Cranking Amps
There are two battery ratings you need to know:
capacity, or ampere-hour rating, and cold cranking
amps, or cold start rating.
Ampere-hour rating (in the YUASA Applications book
it’s abbreviated as AH) is a battery’s ability to deliver
current for an extended period of time. Because low
temperatures slow down the chemical reaction inside a
battery, a battery will have a lower ampere-hour rating
in cold temperatures than in warm ones.
Most small engine batteries are rated at 10 hours. That
says they have to last at a given discharge rate that
long. A 14 ampere-hour battery, for example,
discharges at a rate of 1.4 ampere-hours for
10 hours. At this point, cell voltage has dropped to
1.75v per cell (10.5v for a 12v battery, or 5.25v for a
6v battery). Usually, the larger the plates, the greater
the ampere-hour rating.
Inspecting a Battery
It’s good policy to always inspect a battery before you
test it. Here’s how:
1. Make sure the battery
top is clean and dry. That’s
not just because of looking
pretty: a dirty battery actually
discharges across the grime on
top of the case. Use a soft
brush and any grease-cutting
soap or baking soda solution.
Make sure plugs are finger tight
so cleaning materials don’t get into cells and neutralize
the acid.
2. Inspect battery terminals, screws, clamps
and cables for problems: breakage, corrosion or
loose connections. Clean the terminals and clamps with a
wire brush and coat terminals with no ox grease.
3. Inspect case for obvious damages such as cracks
or for leaks; look for discoloration, warping or raised
top, which may indicate that battery has overheated or
been overcharged.
POINTS TO REMEMBER
❐Capacity or ampere-hour rating: a battery’s ability to discharge current
over time.
❐Cold cranking amps measure battery high rate performance in
cold weather.
16
Cold start rating – the high rate or the cold cranking
amps, abbreviated C.C.A. in the YUASA applications
book – tells how well a battery can be expected to
stand up to low temperatures. This rating depends on
the number of plates and their surface area. The rat-
ing’s arrived at by discharging a cold (0°F) battery at a
high rate – for example, 150 amperes – while dis-
charge is monitored with a voltmeter.
Generally, as displacement per cylinder increases, so
does the cranking current – but since starting systems
differ by model and manufacturer, the best advice is to
check the application book for OE replacement. If a
special application demands higher cranking power,
select an appropriate alternate unit from the YUASA
battery line. Once again, match battery features to
needs. Cold start rating is important in a snowmobile.
A lawn tractor owner probably doesn’t care... unless
he plows snow, too.
3
SECTION
Inspecting, Testing
and Charging
4. Check electrolyte level
and add distilled water if
necessary. Don’t add acid –
only water. Before any tests,
charge the battery so the water
and electrolyte mix.
5. Check the vent tube. Make sure it’s not kinked,
pinched or otherwise obstructed. On a motorcycle, it
should exit away from the drive chain and from below
the swing arm. Small cuts in the tube near the battery
vent are OK; they’re an “emergency escape” for gas in
case the tube becomes obstructed.
POINTS TO REMEMBER
❐Inspect before you test.
❐Dirt on top of case causes discharge.
❐Look for obvious damage to battery and connectors.
❐Add water if electrolyte is low.
❐Make sure vent tube is clear.
Battery Testing Devices
How much of a charge does a battery have? There are
two easy and reliable ways to find out:
1) a hydrometer, which comes in floating ball and
calibrated float types, or
2) a voltmeter (or multimeter, which gives DC voltage
readings).
Which is best?
If you’re choosing between two hydrometers,
opt for the calibrated float type. It gives you an
exact specific gravity reading (that is, the density of the
electrolyte compared to water); that’s much more accurate
than floating balls. For readings on calibrated float and
floating ball hydrometers, see “Methods of Checking
Battery Condition” chart on the next page.
A voltmeter or multimeter can be used where a hydrometer
can’t. Most sealed VRLA or low maintenance batteries
have to be tested with a voltmeter.
Battery testing requires a voltmeter that can
measure DC voltage. Remember to always connect
a voltmeter parallel to the circuit being tested, observing
polarity; otherwise, the pointer will travel in the wrong
direction. It’s a good idea to periodically check a
voltmeter against another one of known accuracy.
Hydrometer
Yuasa
Multimeter
17
3
SECTION
Inspecting, Testing
and Charging
Battery Testing
Methods of Checking Battery Condition
State of Syringe Digital 5-Ball
Charge Hydrometer Voltmeter Hydrometer
100% Charged
w/Sulfate Stop 1.280 12.80v 5 Balls Floating
100% Charged 1.265 12.60v 4 Balls Floating
75% Charged 1.210 12.40v 3 Balls Floating
50% Charged 1.160 12.10v 2 Balls Floating
25% Charged 1.120 11.90v 1 Balls Floating
0% Charged less than 1.100 less than 11.80v 0 Balls Floating
POINTS TO REMEMBER
❐Use a voltmeter or hydrometer to test state of charge.
❐In extreme cold or heat, you’ll have to adjust hydrometer readings.
❐Battery can be tested with or without electrical load applied.
❐Unloaded testing is simplest.
❐Applying a load and reading voltage at battery is more accurate.
18
A battery’s specific gravity changes with temperature.
Ideally, readings should be taken at 77°F. Is it really
going to matter if you’re off a couple of degrees one way
or another? Probably not. If you’re working somewhere
that’s uncomfortably hot or cold, it’s time to use the old
conversion factors: add .001 to the specific gravity read-
ing for each 3°F above 77°F or subtract .001 from the
specific gravity reading for each 3°F below 77°F. Cell
voltage can be found by adding .84 to the specific gravity.
Note, too, that YUASA’s “Sulfate Stop,” a chemical addi-
tive that increases battery life by drastically reducing sul-
fate buildup, changes the specific gravity readings; they’ll
be higher than with ordinary batteries.
Test sealed VRLA types with a voltmeter or
multimeter. If the stabilized open circuit voltage is
below 12.5v, the battery needs charging. For a stabilized
open circuit reading, first allow the battery to remain in
an open circuit condition for at least 1 - 2 hours.
LOADED TESTING: There are two types of loaded tests
for motorcycle batteries. You’ll need a voltmeter or multi-
meter.
Low-load test: Basically, this means turning on the
bike’s lights and taking a voltage reading at the battery.
Remember, hook positive (+) to positive (+), negative (-) to
negative (-). The battery in a 12v system should have at
least 11.5v DC with the lights on. A 6v system should
have at least 5.75v DC. If voltage drops below these lev-
els, it’s time to charge.
High-rate discharge test: This is the best test of bat-
tery condition under a starting load. Use a load testing
device that has an adjustable load. Apply a load of three
times the ampere-hour rating.
At 14 seconds into the test, check battery voltage: a
good 12v battery will have at least 10.5v, and a good 6v
battery, at least 5.25v. If the reading’s low, charge.
There are two types of battery tests: unloaded and
loaded. An unloaded test is made on a battery without
discharging current. It’s simplest and most commonly
used. If you need a precise reading, loaded testing is the
answer. It’s more accurate.
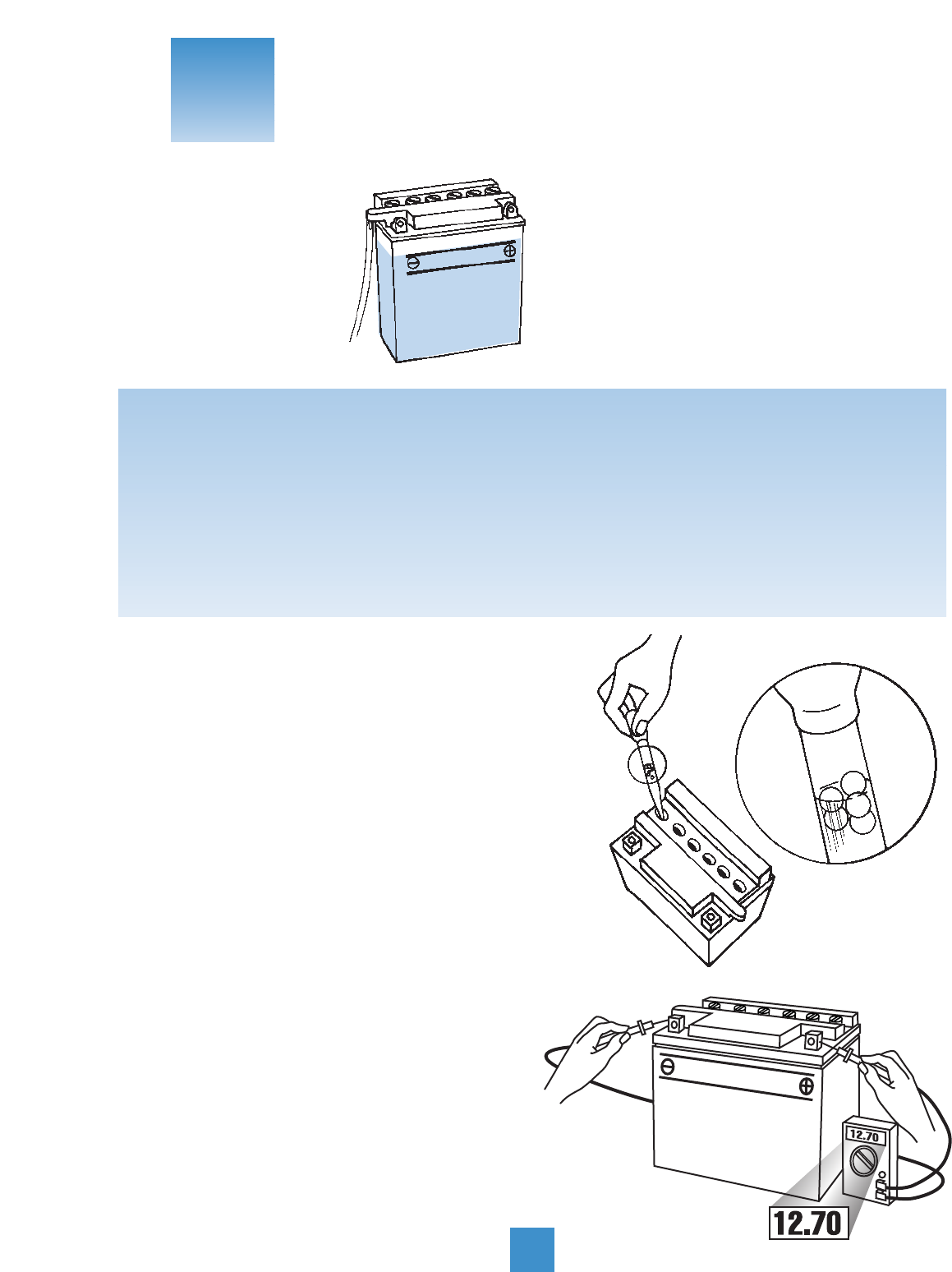
UNLOADED TESTING: Check charge condition using
either a hydrometer or voltmeter. With a voltmeter,
voltage readings appear instantly to show the state of
charge. Remember to hook the positive lead to the
battery’s positive terminal, and the negative lead to the
negative terminal.
A hydrometer measures the specific gravity of each cell.
The specific gravity tells the degree of charge;
generally, a specific gravity of about 1.265 to
1.280 indicates a full charge. A reading of 1.230
to 1.260 indicates the battery should be charged before
testing. The chart below shows the charge level as
measured by syringe float hydrometer, digital voltmeter
and five-ball hydrometer.
3
SECTION
Inspecting, Testing
and Charging

Chargers and Charging
There’s a simple rule of thumb about batteries, and if
you’re a dealer or a mechanic, you know that people
ignore it all the time: for a battery to operate the way it’s
supposed to, it has to be fully charged before it’s used...
and kept fully charged throughout its life.
A charger basically brings a new battery, or a battery
that has been discharged, to full capacity. Plugged into a
wall socket, it sends direct current, flowing in the opposite
direction of the discharge, into the battery.
Charging actually reverses the destructive chemical
process that goes on as a battery discharges: the lead
plates and electrolyte, which were being transformed into
lead sulfate and water, are restored to their original
composition. If a battery has been damaged – for
example, it’s badly sulfated, or the plates have been
damaged from overheating or freezing – it may not
accept a charge.
TYPES OF CHARGERS: There are five basic types of
battery chargers. With all of them, hook the positive
charger lead to the positive battery terminal, and the
negative to the negative. Some chargers on the market
deliver a low charging voltage that can’t fully charge the
battery; avoid them if you’re buying a charger. A 12
volt, 900 mA charger will meet most needs.
Of course, too much of a charge can be a problem, too –
it can “cook” a battery. For small engine starting batter-
ies, don’t use a charge greater than 2 to 2.5 amps for
maintenance purposes. A badly discharged battery with
very high internal resistance may never accept a charge
from a standard charger. It would then require special
charging equipment.
ALWAYS OBSERVE PROPER SAFETY PRECAUTIONS
WHEN CHARGING BATTERIES
TRICKLE CHARGER: This is the charger a consumer –
as opposed to a battery retailer or garage – will usually
have. It charges the battery at a fixed rate. Different
ampere-hour batteries have different charge rates. For
most motorcycle and other small engine start-
ing batteries, charge them at 1/10 of the
rated ampere-hour values in the Yuasa
Applications Book, see example on page 12 for ratings.
Battery voltage increases with the amount of
charge. Find charging time for a completely discharged
battery by multiplying the ampere-hour rating by 1.3
when charging with standard current. The chart on page
21 shows the approximate time needed to fully charge
lead-antimony batteries using a trickle or taper charger.
The chart on page 32 shows the approximate time needed
to fully charge sealed VRLA batteries.
Test the battery during charging, and continue charg-
ing until all cells are gassing. Use either a voltmeter
(or multimeter) or hydrometer. The specific gravity of
the electrolyte in all cells in a fully-charged battery
should come to at least 1.265 in a conventional bat-
tery and 1.280 in a YuMicron and sealed VRLA battery
with Sulfate Stop.
19
Specific Gravity Reading Using a Hydrometer
CX & CX &
YuMicron Conventional YuMicron Conventional
100% 1.27/ 1.26/ 1.28/ 1.27/
1.28 1.27 1.29 1.28
75% 1.22/ 1.21/ 1.23/ 1.22/
1.23 1.22 1.24 1.23
50% 1.17/ 1.16/ 1.18/ 1.17/
1.18 1.17 1.19 1.18
25% 1.13/ 1.12/ 1.14/ 1.13/
1.14 1.13 1.15 1.14
0% 1.11/ 1.10/ 1.12/ 1.11/
or less or less or less or less
Electrolyte Temperature
80
O
F40
O
F
State
of
Charge
During charging, check the electrolyte level periodically
and add water – preferably distilled – to keep the
electrolyte level up to the line. If the battery
becomes hot to the touch, stop charging. Resume after
it has cooled.
Note that permanently sealed batteries – YUASA’s
sealed VRLA battery, for example – generally can be test-
ed only with a voltmeter or multimeter. These batteries
are fully charged when the voltage peaks and then
begins to fall.
Unless using an automatic
charger, do not hook a
battery to a trickle charg-
er and leave it unchecked
for longer than overnight.
After about eight hours maxi-
mum, careful monitoring is
required.
Caps need to be replaced finger
tightened after charging’s done.
3
SECTION
Inspecting, Testing
and Charging
TAPER CHARGER: Similar to the trickle charger, the
automatic taper charger charges at a fixed voltage. As
the battery’s voltage increases with the amount of charge,
the current drops accordingly.
A drawback of both the automatic taper and trickle
chargers is speed... they don’t have it. As the chart on
page 21 shows, it can take days to bring a discharged
battery up to 100%. Here, too, check batteries for over-
heating as they charge.
CONSTANT CURRENT CHARGER: A professional-
quality charger, the constant current makes charging
simple. It maintains a constant supply of current to the
battery at all levels of charging. You select the charging
current. As the battery voltage increases with the amount
of charge, this charger automatically increases the charging
voltage to maintain the current output.
CHARGER/MAINTAINER: This type of charger
monitors the voltage constantly during charging and
Battery Voltage Reading Using a Voltmeter
State of Sealed CX &
Charge VRLA YuMicron Conventional
100% 13.0v 12.7v 12.6v
75% 12.8v 12.5v 12.4v
50% 12.5v 12.2v 12.1v
25% 12.2v 12.0v 11.9v
0% 12.0v or less 11.9v or less 11.8v or less
POINTS TO REMEMBER
❐Fully charge battery when new and keep it fully charged.
❐Test charging batteries as necessary for overheating, water and state
of charge.
❐Trickle and taper chargers are generally slow.
❐Constant current and pulse chargers are professional quality.
❐High rate charger can cause battery damage.
Charging a New Standard Battery
The most important thing to remember about charging a
new battery is do it!
A battery out of the box with only adding electrolyte is
approximately 80% charged. Our recommendation is to
initial charge, bringing the battery to 100% before use.
This completes the electrochemical process. However, a
long ride with a regulated charging system may also bring
the battery’s capacity to a higher level.
Note: See “Section 5” for charging sealed VRLA
batteries.
20
standby modes. When battery voltage reaches a speci-
fied low level, the charger/maintainer then delivers a full
charge. Then when the battery gets to the specified volt-
age, it automatically drops to a float charge.
HIGH RATE CHARGER: Not for use with small engine
starting batteries. They force a high current into the bat-
tery, which can lead to overheating and plate damage.
3
SECTION
Inspecting, Testing
and Charging

don’t. We don’t recommend it, and here’s
why: only the surface area of the battery plates
can be quick charged. A lower current charges
the battery more uniformly. That means better
performance. Also, excessive charging rates
increase the chance of overheating, which can
mean battery damage.
The rule of thumb is to charge a new battery for three to five
hours at a rate equal to 1/10 of its rated capacity. But
there are a lot of exceptions to that rule, as this table shows:
Quick Charges
What about quick charging? The quick answer is
Approximate Charge Times (Hours) Using a “Trickle” (0.25 Amp) Charger
75% 13 15 18 23 24 25
50% 25 30 35 45 48 50
25% 38 45 53 68 71 75
0% 50 60 70 90 95 100
Approximate Charge Times (Hours) Using a 1 Amp Taper Charger
75% 6 7 8 10 10 11
50% 11 13 14 18 19 20
25% 15 18 20 26 27 28
0% 20 23 27 34 35 37
Approximate Charge Times (Hours) Using a 1 Amp Constant Current Charger
75% 3 4 4 6 6 6
50% 6 8 9 11 12 13
25%91113171819
0% 13 15 18 23 24 25
State Battery Types
of 12N10 12N12 12N14 YB18 YB16 Y50
Charge
Charging Times
21
POINTS TO REMEMBER
❐A new battery after activation is approximately 80% charged.
❐Initial charging is always recommended. NEVER quick charge.
❐Charge a new battery at a rate equal to 1/10 of its rated capacity.
3
SECTION
Inspecting, Testing
and Charging

22
Batteries don’t demand a lot of attention. But not giving
your battery the attention it needs can leave you
stranded... or poorer by the cost of a new battery.
How often should you maintain a battery? About
monthly under usual conditions. However, recharging
is necessary when lights get dim, when the starter
sounds weak, or when the battery isn’t used for more
than two weeks.
Important as it is, there’s really not much to battery
maintenance. Basically, just follow the procedure out-
lined in the section “Inspecting a Battery.”
That means:
●Check electrolyte level.
●Keep the top free of grime.
●Check cables, clamps and case for obvious damage
or loose connections.
●Clean terminals and connectors as necessary.
●Make sure exhaust tube is free of kinks or clogs.
●Replace caps, finger tighten only.
Then, test the battery with either a hydrometer or volt-
meter. Keep it charged to 100%.
Monthly Maintenance for Conventional and YuMicron Types
For extended storage, remove the battery from the vehicle
and charge to 100%. Charge the battery every month if
stored at temperatures below 60°F. Charge every two
weeks if stored in a warm area (above 60°F). Make sure
batteries are stored out of reach of children.
Sulfation and Freezing
Two of the biggest battery killers – sulfation and freez-
ing – aren’t a problem if the battery is properly main-
tained and water level is kept where it should be.
Sulfation: This happens because of 1) continuous dis-
charging, or 2) low electrolyte levels.
Let’s back up just a minute: we said earlier that dis-
charge turns the lead plates into lead sulfate. This lead
sulfate is actually a crystal. If the discharge continues
uninterrupted, the sulfate crystals grow and blossom
into sulfation – and a problem. The section titled
“Reasons for Self-discharge” has the gory details.
Much the same happens if the fluid level is too low,
which exposes the plates to air. Then the active lead
material oxidizes and sulfates, and it doesn’t take long
before it won’t hold a charge. (Low electrolyte levels
cause another problem, too: acid in the electrolyte
becomes more concentrated, causing material to cor-
rode and fall to the bottom. In sufficient quantity, it will
short out the battery.)
Keeping a battery charged, disconnecting the
battery cable during storage, and keeping
electrolyte levels up eliminate the problem.
For added protection, YUASA’s YuMicron, YuMicron CX
and Sealed VRLA batteries are treated with a special
chemical formula called “Sulfate Stop.” This dramatically
reduces sulfate crystal buildup on plates. The result: longer
battery life.
How good is Sulfate Stop?
We simulated a constant discharge condition on two
batteries with a 10-watt bulb.
Even after being totally drained for a week, the battery
with Sulfate Stop made a 90% recovery.
The untreated battery: useless.
Storage
If the vehicle is in storage or used infrequently,
disconnect the battery cable. That eliminates
drain from electrical equipment. Check battery every two
weeks and charge as necessary.
4
SECTION
Maintenance
and Storage

POINTS TO REMEMBER
❐Monthly maintenance and testing are a must.
❐Most important: make sure battery is charged and fluid level is correct.
❐Disconnect cables or pull battery for storage.
❐Keep fully charged to prevent sulfation and freezing.
Freezing: It shouldn’t bother you – unless a battery is
inadequately charged. Looking one more time at the
discharge process, remember that electrolyte acid becomes
water as discharge occurs. Now, it takes Arctic temperatures
to freeze acid. But water... as we all know, freezing starts
at 32°F. A sign of this is mossing – little red lines on the
plates. Freezing can also crack the case and buckle the
plates, which means the battery is permanently damaged.
A fully-charged battery can be stored at sub-
freezing temperatures with no damage. As the
chart at right shows, it takes -75°F to freeze electrolyte in
a charged battery. But at just a couple degrees below
freezing – at +27°F – a discharged battery’s electrolyte
turns to ice. That’s a difference of more than 100°F
between the low temperatures a charged and discharged
battery can stand.
Electrolyte Freezing Points
Specific Gravity
of Electrolyte Freezing Point
1.265 -75°F
1.225 -35°F
1.200 -17°F
1.150 + 5°F
1.100 +18°F
1.050 +27°F
23
At temperatures such as these, incidentally, the self-
discharge rate of a battery is so low that a recharge
usually isn’t needed for months. But to stay on the safe
side, test.
4
SECTION
Maintenance
and Storage

SEALED VRLA BATTERY
CONSTRUCTION
New Generation Battery Technology
VRLA batteries… and what makes them special.
Features
The sealed VRLA battery is the battery for vehicles that
may be stored for long periods (riding mowers, personal
watercraft, or scooters or cycles during the off-season,
for example), or where spills could be a problem
(ATVs or personal watercraft). YUASA’s sealed VRLA
batteries deliver:
●No topping – Fill it just once, to activate. No need
to check electrolyte level or add water ever again.
●Reduced self-discharge – Grids manufactured
from a special lead-calcium alloy hold the charge
longer – a real plus with storage or occasional use.
●Easy, instant activation – The “one-push”
electrolyte container makes filling a snap.
●Enhanced safety – A safety valve vents gases
produced by overcharging. In case of fire, the flame
arrestor disk minimizes explosion risk.
24
Yuasa’s innovative sealed VRLA (Valve Regulated Lead
Acid) batteries are a new generation made possible by
advanced gas recombinant technology. These include
the YT and YI series batteries. We refer to them as
“sealed VRLA batteries” here.
Sealed VRLA batteries are easy to activate and maintain.
But, keep these points in mind:
1. There are important differences in activating a
sealed VRLA battery. Be sure to follow the instructions
in this section.
2. While Yuasa sealed VRLA batteries dramatically
reduce the need for maintenance, they do need periodic
charging. It’s important to remember this and to know
how to go about it.
3. When considering upgrading to a sealed VRLA
battery that did not come OE in your vehicle, check
to make sure your charging system has a
regulated output between 14.0 - 14.8v.
Let’s take a closer look at Yuasa’s innovative sealed
5
SECTION
Sealed VRLA
Batteries
HEAT SEALED
CASE TO COVER
protects against seepage
and corrosion – bonded
unit gives extra strength.
THRU-PARTITION
CONSTRUCTION
provides shorter current path with less
resistance than “over the partition”
construction – you get more cranking
power when you need it!
SPECIAL
ACTIVE MATERIAL
is compounded to withstand
vibration, prolong battery life
and dependability.
PATENTED
SEALED POST
prevents acid
seepage, reduces
corrosion – extends
battery life.
POLYPROPYLENE
COVER AND CONTAINER
assures reserve electrolyte capacity for
cooler operating temperatures; gives
greater resistance to gas and oil – and
impact in extreme conditions!
SPECIAL SEPARATOR
makes the battery spill-proof.
Valve regulated design eliminates
water loss and the need to refill
with acid.
SAFETY VALVE/
FLAME ARRESTOR
relieves excess pressure.
SPECIAL GRID
DESIGN
withstands severe vibration,
assures maximum conductivity.

POINTS TO REMEMBER
❐Gas Recombinant technology produces a more compact battery.
❐After activation, no need to check electrolyte or add water.
❐Sealing strip is permanently inserted — it’s never removed.
❐Gas is recombined in the battery; there’s no vented gas and vent tube.
25
The basic discharge-charge cycle is still going on… that’s
what makes any battery tick. But to understand what’s
different, let’s do a little review:
A battery is basically a box containing lead plates. Some
plates have a positive (+) charge, some negative (-).
They’re immersed in a current-conducting electrolyte solu-
tion that sets off the electrochemical process that produces
electricity. Think of a battery as a machine that produces
electricity through a continuous process of charging and
discharging.
During discharge, sulfuric acid electrolyte solution reacts
with the lead plates, turning them into lead sulfate. The
electrolyte – sulfuric acid solution made up of hydrogen,
sulfur and oxygen – gives up its sulfur and some of its
oxygen and turns to water.
PbO2+ Pb + 2 H2SO4→2 PbSO4+ 2 H2O
The process reverses with charging. Electrolytes and
plates return to their original composition. The charging
current breaks down water into its component gases:
hydrogen (from the negatively charged plate) and oxy-
gen (from the positive plate). Gases escape out the vent
tube. With a conventional battery, water is added to
replace that loss.
Here’s the real secret of a sealed VRLA battery: the nega-
tive plate never becomes fully charged… so, no hydrogen
gas. The positive plate still makes oxygen, but instead of
being forced out the vent tube, it reacts with the charged
active material to become water again. That’s “gas
recombinant technology.” That’s the magic of YUASA’s
non-spillable, sealed VRLA battery.
●Compact design – High efficiency in a small
package, with no vulnerable exhaust tubes or other
protrusions to break or kink.
How a Sealed VRLA Battery
Differs from Conventional Types
Some of a sealed VRLA battery’s differences are obvious.
It’s noticeably more compact. The reason: no free elec-
trolyte, making it more “volume efficient” – a fancy way
of saying it can be smaller. Since the battery is filled with
electrolyte just once (at activation), then sealed, you won’t
find a row of filling plugs. Instead, a single sealing plug
strip permanently covers filler ports. Also, there are no
vent tubes – after all, these batteries are sealed!
But as with any magic, there’s more than meets the eye.
The heart of a sealed VRLA battery is what you don’t see.
1. The plates are comprised of special lead-calcium
alloy grids and charged active material. Lead-calcium
reduces self-discharge – the battery holds its charge
longer. The construction of the sealed VRLA battery
causes freed gas to recombine inside the battery instead
of being vented… allowing the battery to be sealed.
2. Separators are made of a special fiber with superior
heat and acid resistance. These special separators make
the battery non-spillable.
3. Sealed VRLA batteries have an internal safety valve.
If battery pressure soars due to accidental overcharging, it
opens to release excess gas, preventing a battery rupture.
The unique design affects what happens inside the battery.
5
SECTION
Sealed VRLA
Batteries
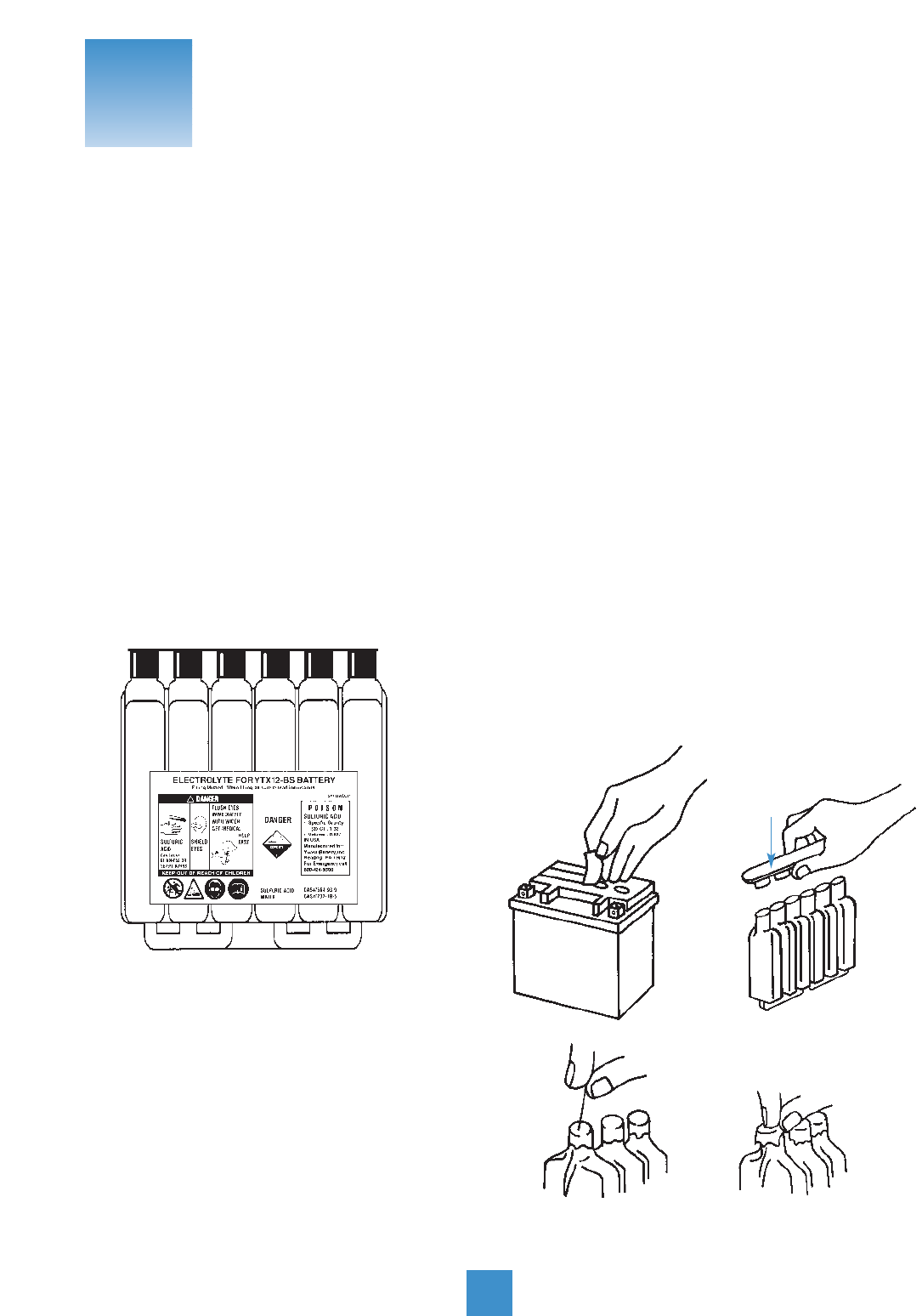
26
●Use only the electrolyte container that comes
with the battery. Sealed VRLA battery electrolyte
is a higher concentration of sulfuric acid. All sealed
VRLA battery electrolyte containers aren’t the same.
Each contains the proper amount of electrolyte for its
specific battery.
●Always wear plastic gloves and protective
eyewear. No Smoking, see page 9 for full safety
instructions.Of course, don’t forget safety precautions
when storing or handling electrolyte solution.
Activating sealed VRLA batteries is easy, although a little
different from conventional activation. For problem-free
start-up and operation, follow the procedure outlined
here. A few things to keep in mind before you get rolling:
●Store the battery in a cool, dry place out of
direct sunlight.
●Do not remove the foil sheet covering the
filler port until activation.
●After removing the electrolyte container cap
strip, do not peel, pierce or otherwise open
the sealed electrolyte receptacles. Don’t
separate the individual cells.
●Read electrolyte handling instruction and
precautions on the label.
Activation and Installation
✘✘
Save
this
strip
5
SECTION
Sealed VRLA
Batteries
To Activate a Sealed VRLA
Battery
1. Place the battery on a level surface. Battery must be
out of the vehicle.
2. Remove electrolyte container from vinyl bag. Remove
the strip of caps. Put the strip aside – you’ll use
this later as the battery sealing plug. Use only
the dedicated container that comes with the battery. It con-
tains the proper amount of electrolyte for your specific
model – important to service life and operation. Do not
pierce or otherwise open the sealed cells of the electrolyte
container. Do not attempt to separate individual cells.
3. Place electrolyte container, sealed top of the cells down,
into the filler ports of the battery. Hold the container level,
push down to break the seals. You’ll see air bubbles as the
ports fill. Do not tilt the electrolyte container.
Warning: Improper activation or excessive
overchargering (possibly by equipment
failure) could cause damage to the battery
or vehicle by forcing acid out of the
safety vent.
See next page.
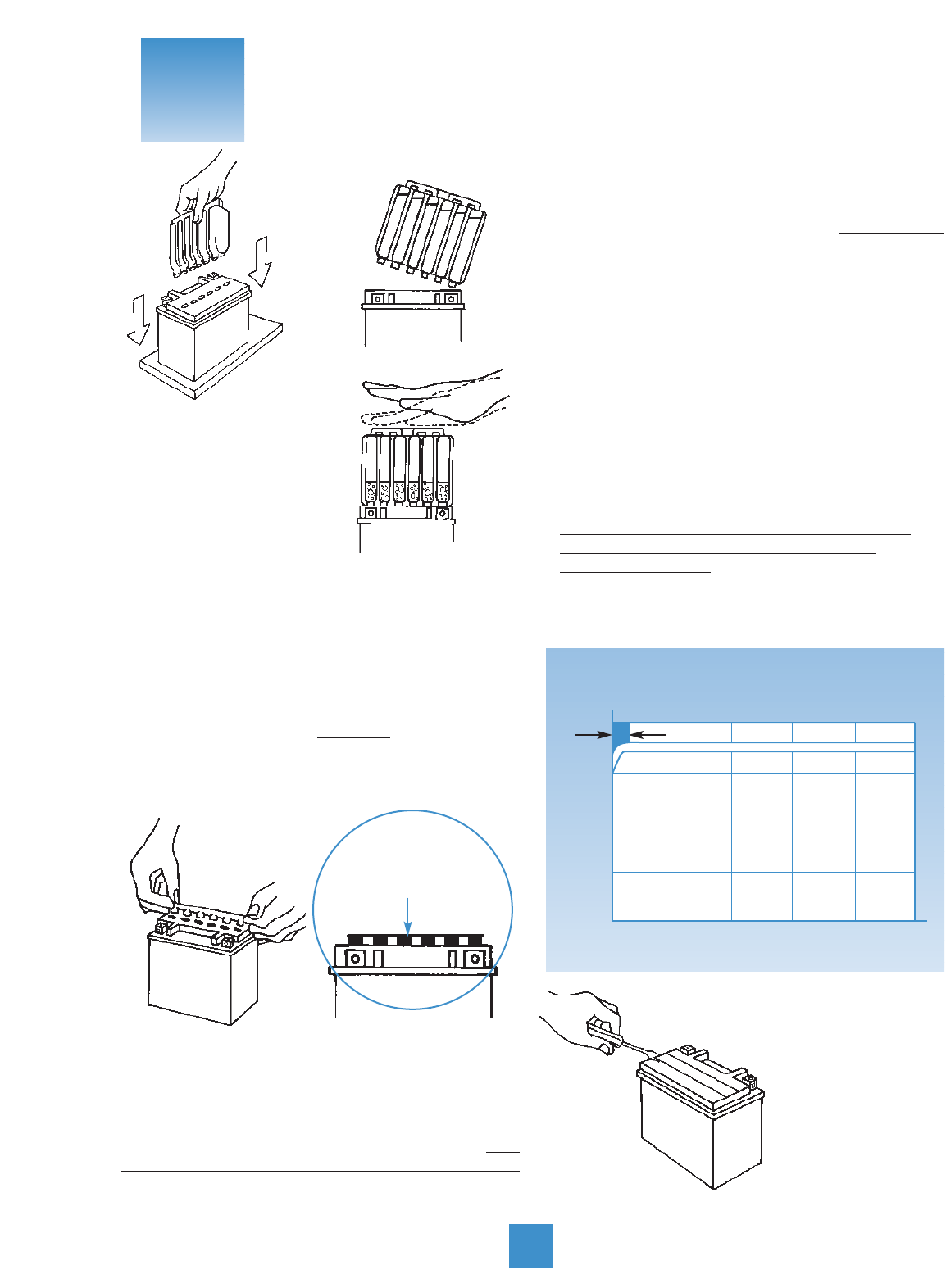
Remember: unlike
a conventional
battery, the sealed
VRLA battery won’t
be topped off
during its life.
Never pry off
sealing caps: it’s
dangerous and
damaging.
✘
4. Check the electrolyte flow.
Keep the container in
place for 20 minutes or
longer until it empties
completely. If no air bubbles
are coming up from the filler
ports, or if container cells
haven’t emptied completely,
tap the container a few times. Don’t remove the contain-
er from the battery until it’s empty. The battery requires
all the electrolyte from the container for proper operation.
Make sure the electrolyte container empties completely.
5. Remove the container. For batteries 3 - 12 AH,
let stand for at least 30 minutes. For batteries
greater than 12 AH, allow the battery to
stand a minimum of 1 HOUR.This allows the
electrolyte to permeate into the plates for optimum
performance. Yuasa sealed VRLA batteries have the amp
hours printed right on the front of the battery.
6. Newly activated sealed VRLA batteries
require an initial charge. After adding electrolyte,
a new battery is approximately 80% charged. Place cap
strip loosely over the filling holes as shown in drawing
above. Immediately charge your battery after the “stand”
period, to bring it to a full state of charge. See
“Charging a Newly Activated Sealed VRLA
Battery” on page 31.
✘
27
Open Circuit Voltage Characteristics
Immediately after Electrolyte Filling
246810
Standing Time (minutes)
Open Circuit Voltage (v)
About 30 Seconds
13
12
11
10
0
Place
cap strip loosely
over filling holes
5
SECTION
Sealed VRLA
Batteries
After charging is completed, press down firmly
with both hands to seat the caps (don’t pound
or hammer).
The battery is sealed. There is no need to remove the strip
of caps or add electrolyte for the life of the battery.
7. The graph below shows an open circuit voltage
characteristic of a sealed VRLA battery just after the elec-
trolyte is filled.
If the battery is only filled with electrolyte, but not being
given a supplementary charge, the open circuit voltage
will be somewhere around 12.5 to 12.6v, as shown in
the graph below. The reasons for the voltage being
low are that:
●The capacity reached by filling with
electrolyte is about 80% of the fully
charged capacity.
●The electrolyte around the plates gets its
concentration lowered temporarily.
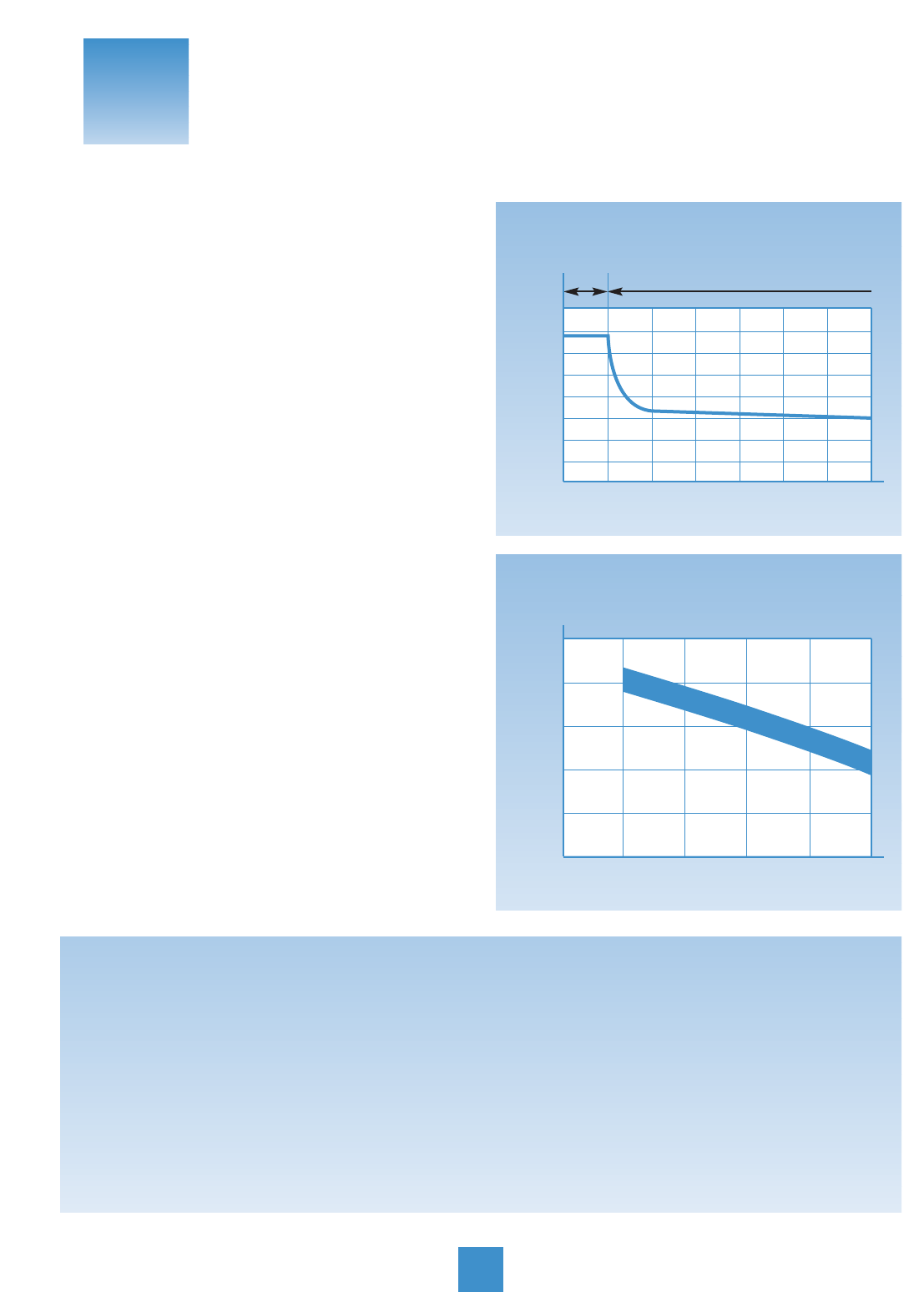
How healthy is your sealed VRLA battery? Since a sealed
VRLA battery is sealed – and the sealing caps are never
removed – you won’t be able to check the state of the
charge by the old hydrometer-and-specific-gravity test.
Rather, use a voltmeter or multimeter to measure DC volt-
age. It should be of class 1 accuracy or better. Some
basics to keep in mind:
●Check voltage using a voltmeter. Readings for a
charged, newly-activated battery should be 12.8v
or higher after the battery is charged and sits for
at least 1 - 2 hours. If less, it needs an
additional charge.
●The graph top right shows open circuit characteristics
of the sealed VRLA battery after end of charging
using a constant current charger set to the standard
current of the specific battery. As shown, the open
circuit voltage is stabilizing 30 minutes after end
of charge.
Therefore, to determine the state of charge and the
health of the battery, measure the open circuit
voltage 1 hour after end of charge.
●For a battery that has been in use, refer to the graph
bottom right to determine state of charge from
open circuit voltage.
Measuring Voltage
Relation between State of Charge
and Open Circuit Voltage
14
13
12
11
10
100 75 50 25 0
State of Charge (%)
Open Circuit Voltage (v)
Ambient Temperature
32-104°F (0~40°C)
28
Open Circuit Voltage Characteristics
after End of Charge
18
17
16
15
14
13
12
11
10 0 102035405060
Elapsed Time (minutes)
Terminal Voltage (v)
Ambient Temperature
77°F (25°C)
Charge Open Circuit
POINTS TO REMEMBER
❐Use a voltmeter to determine state of charge. Because sealing caps are
never removed, you can’t test specific gravity.
❐Don’t use a quick charge for initial activation.
❐A battery that has just been activated or charged needs to stabilize
1 hour for an accurate voltage reading.
5
SECTION
Sealed VRLA
Batteries
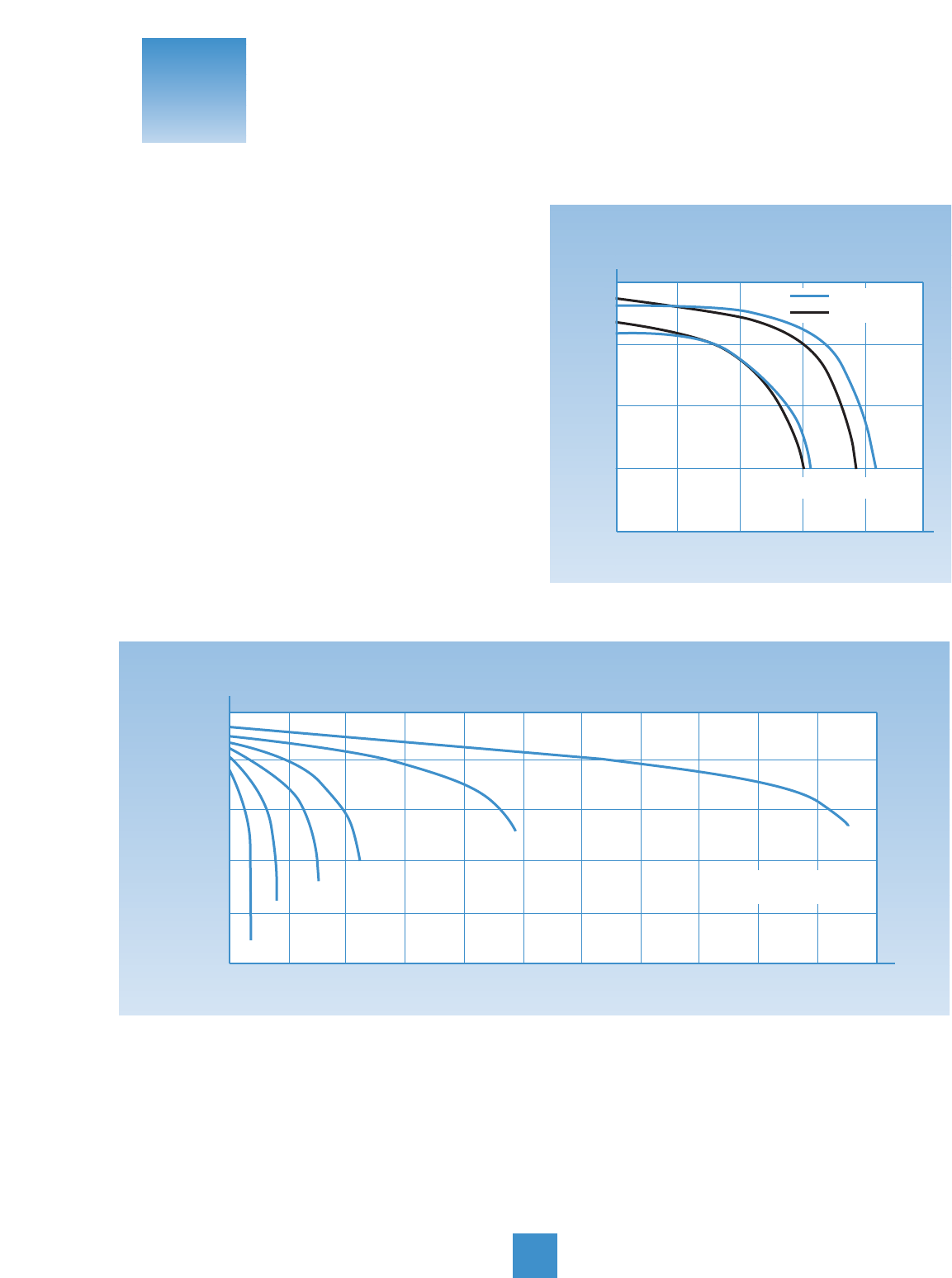
(10 hour rate discharge
characteristics)
Terminal Voltage (v)
2C
1C
0.6C
0.4C
0.2C 0.1C
29
Think about what types of vehicles a sealed VRLA battery
goes into: most aren’t like the family car, driven day-in,
day-out. They’re probably used once in a while, or
maybe even stored for weeks or months at a time.
That demands a special kind of battery – one with
extra power to reliably start that engine, every time. In
YUASA’s sealed VRLA batteries, the plate groups are spe-
cially designed to deliver that. The graph to the right
shows the increase in discharge time of a sealed VRLA
battery compared to a conventional Yumicron battery
at both cold and room temperatures. The graph below
shows the cold temperature performance of the sealed
VRLA battery as the load increases. “C” equals the
Amp Hour Capacity Rating of the battery.
Discharge Characteristics
Starting the engine is a big part of the battery’s job, but
not all. Electrical accessories and safety systems – lights
and horn – need a stable supply of electricity. Now
you’re concerned with the battery’s “low rate discharge
characteristics.” This steady, low rate discharge is
measured in “10-hour rate discharge.” The graph
above shows the discharge characteristics of YUASA’s
YT or YI sealed VRLA batteries at different current rates.
Note that battery capacity is a function of the current
being used (or discharge current) x time.
13
12
10
8
6
01234567891011
Discharge Duration (hour)
Discharge Characteristics of a
Sealed VRLA Battery by Capacity (C = 10 hour rate capacity)
5
SECTION
Sealed VRLA
Batteries
Terminal Voltage (v)
Sealed VRLA
Conventional
01 23 4
Discharge Duration (minute)
12
10
8
6
High Rate Discharge Characteristics of a
Sealed VRLA 4 AH Battery
20A Discharge Characteristics
77°F (25°C)
14°F (-10°C)

30
Lead-calcium technology definitely slows self-discharge,
but a combination of heat and idleness will still drain a
sealed VRLA battery, like the conventional one. You’ll find
step-by-step charging instructions later in this section.
Constant self-discharge is a fact of life for all batteries.
They lose strength as they sit there doing nothing. The
good news is that lead-calcium technology in a sealed
VRLA battery slows down the self-discharge process
substantially. Conventional lead-antimony batteries
discharge at about 1/100 volt a day… the lead-calci-
um sealed VRLA battery, 1/300 volt per day. Looking
at it another way, a conventional battery fully charged
and stored for a month will lose roughly a third of its
charge; the sealed VRLA battery handled the same
way would lose about 10%.
Remember, too, ambient temperature affects battery dis-
charge. Higher the temperature, quicker the discharge –
for all batteries.
So, the sealed garage or storage shed with the sun beat-
ing down on it isn’t doing any favors to the battery in
your vehicle. Excessive heat will prematurely shorten the
life of the battery.
Some people figure sealed VRLA batteries are so
good, there’s no need to worry about routine charges.
Flattering, but wrong. Forgetting routine charging
can mean a one-way ticket to the battery graveyard.
Self-discharge
Self-discharge Characteristics
Relation between Storage Period (months)
and Remaining Capacity (%)
100
90
80
70
60
50
40 01234567
Storage Period (month)
Remaining Capacity (%)
104°F
(40°C) 104°F
(40°C)
77°F
(25°C)
77°F
(25°C)
32°F
(0°C)
32°F
(0°C)
Sealed VRLA Standard
Match your sealed VRLA battery to the right charger. The wrong
one can cause permanent damage and poor performance.
Yuasa offers a complete line of chargers to activate and
maintain your battery to factory specifications.
●Do not use a larger than recommended amp charger to
reduce charging time. That permanently damages the
battery and voids the warranty.
●
To find recommended current output in amps,
divide battery amp hour capacity rating by 10.
Example: 14 AH ÷ 10 = 1.4 amp current.
Choosing a Charger
POINTS TO REMEMBER
❐“High rate discharge” sealed VRLA batteries deliver extra starting power.
❐Lead-calcium technology substantially slows self-discharge.
❐Routine charging is required to maintain a full charge.
5
SECTION
Sealed VRLA
Batteries
Yuasa 1.5 Amp
Automatic Charger
Yuasa 900 mA
Automatic Charger
Yuasa sealed VRLA batteries have the amp hours printed
right on the front of the battery. If you’re not sure, refer to
application manuals at the dealer. Select the charger that
comes closest to the value of that figure.

CHARGING INSTRUCTIONS FOR NEWLY ACTIVATED
SEALED VRLA BATTERIES
31
Charging a Newly Activated Sealed VRLA Battery
5
SECTION
Sealed VRLA
Batteries
Sealed VRLA batteries require an initial charge. If you are
using a constant current charger, refer to the standard
(STD.) charging method printed on the battery. If you are
using an automatic type taper charger, check to make sure
that the charger current (amps) is equal to or greater than
the standard (STD.) charging method listed on the battery.
Yuasa Automatic Chargers and Accessories are the safest and most convenient
method for error proof charging and battery maintenance.
These batteries are a sealed VRLA construction – which means:
NEVER REMOVE THE SEALING STRIP AFTER CHARGING IS COMPLETED!
If the battery gets very hot to the touch, cease charging and allow battery to cool down.
Check voltage using a voltmeter. Readings for a charged, newly-activated battery should
be 12.8v or higher after the battery is charged and sits for at least 1 - 2 hours.
If less, it needs an additional charge.
YTX20L-BS
CHARGING
METHOD
•
THIS IS A SEALED BATTERY; THE SEALING CAPS SHOULD BE CONSIDERED PERMANENT.
•
DO NOT REMOVE THE CAPS TO ADD WATER OR TO CHARGE THE BATTERY; CHARGE AT
12V AT THE AMPERAGE AND HOURS STATED BELOW.
STD.: 1.8A
X
5~10h or QUICK:9.0A
X
1.0h

The single most important thing to maintaining a sealed
VRLA battery is don’t let it sit discharged:
keep it fully charged. A sealed VRLA motorcycle
battery should be kept to near fully charged for peak
performance. In fact, it can need charging more often
than a car battery because it’s probably not used routinely
and, therefore, not “automatically” charged.
Use the following guidelines for boost charge. Always
verify battery condition before charging, and 30 minutes
after charging.
●A fully charged battery should read 12.8v or higher after
battery has been off the charger 1 - 2 hours.
Routine Charging
OVERCHARGING CAN HARM YOUR BATTERY BEYOND
RECOVERY.
A word on overcharging: don’t. Because of the charac-
teristics of a sealed VRLA battery, too much of a boost
charge will decrease the volume of electrolyte. The
longer the overcharge time, the greater the drop in elec-
trolyte – and starting power.
Water can’t be added to the sealed VRLA battery to make up
the difference. Overcharging can warp plates, making
future charging difficult or impossible. Watch charging times
carefully, or ideally, use a Yuasa Automatic Charger. Always
stop charging if the battery becomes really warm to the
touch. Let it cool down 6 - 12 hours and resume charging.
32
Caution:
Always wear safety glasses and charge in a ventilated area. If battery gets really warm to the touch, discontinue
charging and allow battery to cool down. No sparks, flames or smoking when charging.
State Charge Time
*
of Voltage Action (Using a constant
Charge current charger
@ std. amps specified
on the battery)
100% 12.8v - 13.0v None
Check at 3 months None Required
from date of
manufacture
75% -100% 12.5v - 12.8v May need slight
charge, if no 3 - 6 hours
charge given,
check in 3 months
50% - 75% 12.0v - 12.5v Needs charge 5 - 11 hours
25% - 50% 11.5v - 12.0v Needs charge At least 13 hours
verify state
of charge
0% - 25% 11.5v or less Needs charge 20 hours
(see special instructions
on page 33)
* Charging times can vary depending on type of charger. Follow the charger’s instructions.
5
SECTION
Sealed VRLA
Batteries

33
Batteries with voltage below 11.5v may require special
equipment and procedures to recharge.
In charging an overdischarged battery having a terminal
voltage of 11.5v or lower, its internal resistance may be
too high to charge at a normal charge voltage.
Therefore, it may be necessary to raise the voltage of
the battery initially (25v as a maximum), and charge
for approximately 5 minutes. If the ammeter shows
no change in current after 5 minutes, you need a new
battery.
Current flowing into the battery at high voltage can
become excessive. Monitor amperage and adjust voltage
as necessary to keep current at the battery’s standard
amp rating. Charge for approximately 20 hours.
Charging Instructions for Sealed VRLA Batteries
with Voltage of 11.5 or Less
How to determine battery condition
after boost charge.
Determine the condition of a sealed VRLA battery at
least 1 - 2 hours after the charge by measuring the
terminal voltage according to the table below.
Criteria Judgement
12.8v or higher Good
12.0 -12.8v or lower Charge insufficient → Recharge
12.0v or lower Unserviceable → Replace
Adjust voltage so that current will be at
standard amps after 5 minutes.
Check voltage periodically using a
voltmeter.
●Recommended every 3 months from date of activation,
or 3 months from date of manufacture for batteries
activated at the factory. Keep in mind, higher storage
temperatures cause faster self-discharge and require
checking more often.
●If you plan to store your vehicle for an extended time,
make sure your battery is fully charged.
●Fully charged should read 12.8v - 13.0v after standing
1 - 2 hours.
●When a battery is in storage, check and charge it if
the voltage drops below 12.5v for YTX batteries.
Beyond that, maintenance is the same as for any battery,
except you don’t have to worry about electrolyte:
●Keep the battery top free of grime.
●Check cables, clamps and case for obvious damage
or loose connections.
●Clean terminals and connectors as necessary.
●For storage, pull battery or disconnect battery cable.
Routine Maintenance for
Sealed VRLA Batteries
5
SECTION
Sealed VRLA
Batteries

Acid Sulfuric acid, used to describe
the electrolyte or liquid in a cell.
Active Materials Materials in a
battery that react chemically to pro-
duce electrical energy: they are lead
peroxide (positive) and sponge lead
(negative).
Activation Making a dry cell func-
tional by adding electrolyte.
AGM Absorbed glass mat.
Air Oxidized A charged negative
plate that has been removed from
electrolyte and permitted to discharge
in an air atmosphere. Plates must then
be recharged before they are capable
of producing useful electrical energy.
Alloy A combination of two or more
metals. See Antimonial Lead
Alloy and Calcium Lead Alloy.
Ambient Temperature The sur-
rounding temperature, usually refers to
room temperature.
Alternating Current A pulsating
electric current in which direction of
flow is rapidly changed, so the termi-
nal becomes in rapid succession posi-
tive, then negative. Abbreviated AC.
Ammeter An instrument for measur-
ing electrical current.
Ampacity Current carrying capacity
in amperes.
Ampere The unit of electrical current
equal to the steady state current pro-
duced by one volt applied across a
resistance of one ohm.
Ampere-Hour A measure of the
volume of electricity, being one
ampere for one hour. It is used to
express battery capacity, and is regis-
tered by an ampere-hour meter; it can
be obtained by multiplying the current
in amperes by length of time that the
current is maintained.
Ampere-Hour Capacity The
number of ampere-hours that can be
delivered by a storage battery under
specified conditions as to temperature,
rate of discharge and final voltage.
Ampere-Hour Efficiency The
electrochemical efficiency of a storage
battery expressed as the ratio of
ampere-hours output to the ampere-
hours input required for recharge.
Ampere-Hour Meter An instru-
ment that registers the quantity of elec-
tricity in ampere-hours.
Anode An electrode through which
current enters any non-metallic con-
ductor. Specifically, an electrolytic
anode is an electrode at which nega-
tive ions are discharged, positive ions
are formed, or at which other oxidiz-
ing reactions occur.
Antimonial Lead Alloy A
commonly used alloy in battery
castings. The percentage of antimo-
ny varies from 1/2% to 12%. Other
substances are present in small quan-
tities, either as inescapable impurities
or by design to improve the proper-
ties of the cast part.
Antimony A hard, brittle, silver-
white metal with a high luster from the
arsenic family.
Assembly 1. Combining various
parts into a finished battery. 2. Any
particular arrangement of cells, con-
nectors and terminals to form a bat-
tery.
Automotive Battery SLI battery of
3 or 6 cells used for starting, lighting
and ignition of cars, trucks, buses, etc.
Average Voltage A storage bat-
tery’s average value of voltage during
a period of charge or discharge.
Battery (Storage) A connected
group of two or more storage cells.
Common usage applies this term to a
single cell used independently.
Bridge The ribbed supporting struc-
ture in the bottom of a battery contain-
er that provides sediment space under
the elements, thereby preventing short
circuits.
Burning Welding together two or
more lead or lead alloy parts such as
plates, straps, connectors.
Burning Center The center-to-cen-
ter distance between adjacent plates of
the same polarity.
Burning Stick A lead or lead alloy
stick used as a supply of joining mater-
ial in lead burning.
Cadmium A metallic element highly
resistant to corrosion, used as a pro-
tective plating on certain parts and fit-
tings.
Cadmium Electrode A third elec-
trode for separate measurements of
the electrode potential of positive and
negative plate groups.
Calcium Lead Alloy A lead base
alloy that is sometimes used for battery
parts in place of antimonial lead
alloys.
Capacity See Ampere Hour
Capacity.
Capacity Test A test that dis-
charges the battery at constant current
at room temperature to a cutoff volt-
age of usually 1.75 volts/cell.
Cast Forming a molten substance
into a shape by introducing the mate-
rial into a mold and allowing it to
solidify.
Casting A metallic item, such as one
or more grids, straps or connectors,
formed by pouring a molten substance
into a mold and allowing it to solidify.
34
GLOSSARY

Cast-On Strap A multiple connec-
tor that had been cast onto the plates
directly in a combination mold/burn-
ing jig; contrasts with burning of plates
and prefabricated straps.
Cathode An electrode through
which current leaves any non-metallic
conductor. Specifically, an electrolytic
cathode is an electrode at which posi-
tive ions are discharged, or negative
ions are formed, or at which other
reducing actions occur.
Cell (Primary) A cell designed to
produce electric current through an
electrochemical reaction that is not effi-
ciently reversible and hence the cell,
when discharged, cannot be efficiently
recharged by an electric current.
Cell (Storage) An electrolytic
cell for generation of electric ener-
gy, in which the cell after discharge
may be restored to a charged con-
dition by an electric current flowing
in a direction opposite to the flow
of current when the cell discharges.
Charged A storage cell at maxi-
mum ability to deliver current. The
positive plates contain a maximum
of lead oxide and a minimum of
lead sulfate, and the negative
plates contain a maximum of
sponge lead and a minimum of sul-
fate, and the electrolyte is at maxi-
mum specific gravity.
Charged and Dry A battery
assembled with dry,charged plates
and no electrolyte.
Charged and Wet A fully-
charged battery containing elec-
trolyte and ready to deliver current.
Charging The process of con-
verting electrical energy to stored
chemical energy. In the lead acid
battery, it converts lead sulfate in
the plates to lead peroxide (posi-
tive) or lead (negative).
Charging Rate The current,
expressed in amperes, at which a bat-
tery is charged.
Circuit A system of electrical compo-
nents through which an electric current
is intended to flow. The continuous
path of an electric current.
Cold Crank Test A test that applies
a high rate of discharge to a battery at
0°F, and the 30 second cell voltage
must be above 7.2v.
Constant Current Charge A
charge that maintains the current at a
constant value. For some types of
batteries this may involve two rates,
called a starting and a finishing rate.
Constant Potential Charge or
Constant Voltage Charge A
charge that holds the voltage at the
terminals at a constant value.
Container Housing for one or more
cells, commonly called a “jar.”
Cover The lid of an enclosed cell,
generally made of the same material
as the container and through which
the posts and vent plug extend.
Cover Inserts Lead or lead alloy
rings molded or sealed into the cell
cover, and that the element posts are
burned to, thereby creating an effec-
tive acid creep-resistant seal.
Creepage Travel of electrolyte up
the surface of electrodes of other parts
of the cell above the level of the main
body of the electrolyte.
Curing Chemical conversion process
that changes lead oxides and sulfuric
acid to mixtures of basic lead sulfates,
basic lead carbonates, etc., which
consequently forms the desired
structures of lead or lead sulfate on
negative and positive plates during
formation.
Current The time rate of flow of
electricity, normally expressed as
amperes, like the flow of a stream of
water.
Cut-Off Voltage See Final
Voltage.
Cutting (of acid) Dilution of solu-
tion of sulfuric acid to a lower concen-
tration.
Cycle A discharge and its subse-
quent recharge.
Cycle Service Battery operation
that continuously subjects a battery to
successive cycles of charge and dis-
charge, e.g., motive power service.
Deep Discharge Removal of up to
80% of the rated capacity of a cell or
battery.
Dielectric Test An electric test per-
formed on jars, containers and other
insulating materials to determine their
dielectric breakdown strength.
Diffusion The intermingling or distri-
bution of particles or molecules of a
liquid.
Direct Current A one-direction cur-
rent. Abbreviated DC.
Discharge Conversion of a battery’s
chemical energy into electrical energy.
Discharged A storage cell when, as
a result of delivering current, the plates
are sulfated, the electrolyte is exhaust-
ed, and there is little or no potential
difference between the terminals.
Discharge Rate Any specified
amperage rate at which a battery
is discharged.
35
GLOSSARY

Dry Charged Battery plates that
have been subjected to the dry charg-
ing process.
Dry Charging Manufacturing
process in which tank-formed battery
plates are washed free of acid and
then dried.
Efficiency The ratio of the output of
a cell or battery to the input required
to restore the initial state of charge
under specified conditions of tempera-
ture, current rate and final voltage.
Electrode A conductor through
which current passes in or out of a cell.
Electrode (Electrolyte) Potential
The difference in potential between the
electrode and the immediately adja-
cent electrolyte, expressed in terms of
some standard electrode potential dif-
ference.
Electrolysis Electrochemical reac-
tion that causes the decomposition of a
compound.
Electrolyte Any substance that dis-
associates into two or more ions when
dissolved in water. Solutions of elec-
trolyte conduct electricity and are
decomposed by it. For batteries, elec-
trolyte implies a dilute solution of sulfu-
ric acid.
Electromotive Force (EMF)
Electrical pressure or potential,
expressed in volts.
Element An assembly of a positive
plate group, negative plate group and
separators.
End Gravity The specific gravity of
a cell at the end of a prescribed dis-
charge.
Energy Density Ratio of battery
energy content in watt hours to battery
weight in volume.
Envelope A separator folded and
wrapped around a battery plate dur-
ing assembly.
Equalizing Charge An extended
charge given to a storage battery to
ensure complete restoration of active
materials in all the plates of the cells.
Expander An ingredient in the neg-
ative paste that delays shrinking and
solidifying of the sponge lead of the
finished plate, thereby enhancing neg-
ative plate capacity.
Ferroresonant Charger A con-
stant volt power supply containing a
special transformer-capacitor combi-
nation that changes operating charac-
teristics as the draw is varied, ensuring
that voltage output remains constant.
Filling Gravity The specific gravity
of acid used to fill batteries.
Final Voltage The cut-off voltage of
a battery. The prescribed voltage
reached when the discharge is consid-
ered complete.
Finishing Rate The rate of charge,
in amperes, to which charging current
is reduced near the end of the charge
for some types of batteries to prevent
gassing and temperature rise.
Fixed Resistance Discharge
Discharge of a cell or battery through
a fixed resistive load, the current being
allowed to fall off as the terminal volt-
age decreases.
Float Plate A pasted plate.
Float Charging A recharge at a
very low rate, accomplished by con-
nection to a buss whose voltage is
slightly higher than the open circuit
voltage of the battery.
Foot Projections from the grid at the
bottom edge, used to support the plate
group.
Formation or Forming Charge
An initial charging process that elec-
trochemically converts the raw paste of
the plates into charged active material,
lead peroxide in the positive plates
and sponge lead in the negative
plates.
Formed Plates that have undergone
formation.
Freshening Charge A charge
given batteries in storage to replace
the standing loss and ensure that every
plate is periodically brought up to full
charge.
Full Charge Gravity Specific
gravity of the electrolyte when cells are
fully charged and properly leveled.
Gang Vent Vents for usually six
adjacent cells that are connected to a
common manifold.
Gassing Bubbles from gases being
released at one or more of the elec-
trodes during electrolysis.
Glass Mat Fabric made from glass
fibers with a polymeric binder such as
styrene or acrylic which is used to help
retain positive active material.
Gravity Specific gravity.
Gravity Drop The number of points
reduction or drop of specific gravity of
the electrolyte from cell discharge.
Grid A metallic framework used in a
battery for conducting electric current
and supporting the active material.
Group One or more plates of one
type – positive or negative – burned to
a post or strap.
H2SO4Sulfuric Acid.
High Rate On charge, any rate
higher than the normal finishing rate.
Hydration (Lead) Reaction
between water and lead or lead com-
pounds. Gravities lower than those
found in discharged cells are apt to
produce hydration, which appears as
a white coating on plate groups and
separators in a cell.
36
GLOSSARY

Hydrometer A device used to mea-
sure density or specific gravity of elec-
trolyte solutions.
Hydroset Curing process for plates
that oxidizes the lead paste, reducing
free lead to a few percent of total.
Initial Voltage The closed circuit
voltage at the beginning of a dis-
charge. It is usually measured after
current has flowed for a period suffi-
cient for the voltage rate of change to
become practically constant.
Insert A bushing of lead or lead alloy
molded or sealed into cell covers, and
to which the post is burned to create a
creep-resistant, cover-to-post seal.
Intercell Connector Conductor of
lead or lead alloy used to connect two
battery cells.
Internal Resistance Resistance
within a cell or battery to the flow of
electric current, measured by the ratio
of the change in voltage to a specified
change in current for a short period of
time.
Jar Housing, or container, for one or
more cells.
Jar Formation Forming of plates in
the cell jar.
Jumper A short length of conductor
used to connect or cut out part of an
electrical circuit.
Kilovolt One thousand volts.
Kilowatt One thousand watts.
Kilowatt Hours A measure of ener-
gy or work accomplished, being 1000
watt hours.
Lead (Pb) Chemical element used in
lead acid batteries.
Lead Burning Welding of lead or
alloy parts.
Lead Hydrate A white lead com-
pound formed by reaction of very
dilute electrolyte or water and metallic
lead or lead alloys.
Lead Oxide A general term for any
of the lead oxides used to produce bat-
teries.
Lead Peroxide A brown lead oxide
which is the positive material in a fully
formed positive plate.
Lead Plated Part Hardware that
has a thin protective layer of lead elec-
trode deposited on the surface.
Lead Sponge The chief component
of the active material of a fully-charged
negative plate.
Lead Sulfate A compound that
results from the chemical action of sul-
furic acid on oxides of lead or on
lead metal.
Level Lines Horizontal lines molded
or painted near tops of battery contain-
ers indicating maximum and minimum
electrolyte levels.
Litharge A yellow-red oxide of lead
sometimes used in making active
material.
Local Action A battery’s loss of oth-
erwise usable chemical energy by cur-
rents that flow within the cell of a bat-
tery regardless of its connection to an
external circuit.
Loss of Charge Capacity loss in a
cell or battery standing on open circuit
as a result of local action.
Lug A portion of the grid used for sup-
port of the plate group, usually a hang-
ing lug on the top edge of the grid.
Also, a tab on the grid used for connec-
tion of plate to strap and other plates.
Machine Casting A fully or semi-
automatic grid or small parts casting
operation.
MF (Maintenance Free Battery) A
VRLA sealed absorbed glass mat
(AGM) battery.
Manual Discharge Capacity test
in which the operator disconnects the
battery from the test load after all cells
have reached the prescribed final volt-
age. With fixed resistance loads,
boost cells are used to keep the dis-
charge rate fairly constant as the test
cell voltages drop rapidly near the
final voltage. Electronic load manual
discharges generally do not require
boost cells.
Microporous Separator A
veneer or grooved-type separator
made of any material that has many
microscopically small pores.
Milliampere One thousandth of
an ampere.
Millivolt One thousandth of a volt.
Modified Constant Voltage
Charge A charge in which charging
current voltage is held substantially
constant while a fixed resistance is
inserted in the battery circuit, produc-
ing a rising voltage characteristic at
the battery terminals as the charge
progresses.
Mold A cast iron or steel form used
to produce a casting of definite shape
or outline.
Mold Coat A spray applied to
metal molds that acts as a release
agent and an insulator against rapid
heat transfer.
Moss Lead crystals that can grow at
high current density areas of negative
plates— along edges, at feet or at
plate lugs— and cause short circuiting.
Negative Plate The grid and active
material that current flows to from the
external circuit when a battery is dis-
charging.
37
GLOSSARY

Negative Terminal The terminal
from which current flows through the
external circuit to the positive terminal
when the cell discharges.
OHM A unit of electrical resistance.
Oil of Vitriol Concentrated commer-
cial sulfuric acid, abbreviated OV or
O.V.
Open Circuit The state of a battery
when not connected to either a charg-
ing source or a load circuit.
Open Circuit Voltage The voltage
at a battery terminal when no appre-
ciable current is flowing.
Oxide (of lead) A compound of
lead and oxygen in one of several pro-
portions used to prepare battery paste.
Panel A casting consisting of two or
more grids made simultaneously in a
single mold.
Pb Chemical symbol for lead.
PbO Chemical symbol for litharge.
PbO2Chemical symbol for lead per-
oxide.
Pellet The portion of pasted material
contained in a grid section framed by
adjacent horizontal and vertical mem-
bers exclusive of forming bars.
Peroxide See Lead Peroxide.
Pig A cast bar of lead or lead alloy.
Pig Lead A grade of highly refined,
unalloyed lead.
Plate A pasted grid.
Plate Centers Distance between
center lines of adjoining plates of
opposite polarity in a cell. One half
the size of a strap center upon which
the plates of like polarity are burned.
Polarization Change in voltage at
terminals when a specified current is
flowing; equal to the difference
between the actual and the equilibrium
(constant open circuit condition) poten-
tials of the plates, exclusive of the inter-
nal resistance drop.
Porosity The ratio of open spaces or
voids in a material to the volume of its
mass.
Positive Plates The grid and active
materials of a storage battery from
which current flows to the external cir-
cuit when the battery is discharging.
Positive Terminal The terminal that
current flows toward in the external cir-
cuit from the negative terminal.
Post Terminal or other conductor that
connects the plate group strap to the
outside of the cell.
Pure Lead Pig Lead.
Rated Capacity Ampere hours of
discharge that can be removed from a
fully charged cell or battery, at a specif-
ic constant discharge rate at a specified
temperature and at a specified cut-off
voltage.
Rate of Charge See Starting
Rate and Finishing Rate.
Raw Plate An unformed plate.
Rectifier A device that converts alter-
nating (ac) current into unidirectional
(dc) current because of a characteristic
that permits appreciable flow of current
in only one direction.
Red Lead A red oxide of lead used
in making active material.
Reference Electrode Electrode
used to measure acid concentration or
plate state of charge.
Resistance The opposition of a con-
ductor to the passage of an electrical
current, usually expressed in ohms.
Resistor A device used to introduce
resistance into an electrical circuit.
Retainer A sheet of glass mat, per-
forated or slotted rubber, plastic or
some other material installed on each
face of the positive plates in certain
types of cells, to deter loss of active
material.
Reversal A change in the normal
polarity of a cell or battery.
Rib A vertical or nearly vertical ridge
of a grooved separator or spacer.
Secondary Lead Reclaimed as
opposed to virgin lead.
Sediment The sludge or active
material shed from plates that drops
to the bottom of cells.
Sediment Space The portion of a
container beneath the element; sedi-
ment from the wearing of the plates
collects here without short-circuiting.
Self-discharge Loss of charge due
to local action.
Separator A device in a storage
battery that prevents metallic contact
between plates of opposite polarity in
a cell.
Series Cells All cells in a battery
other than pilot cells. They are so
called because the cells are usually
connected in series.
Series Parallel Connection Cells
arranged in a battery so two or more
strings of series connected cells, each
containing the same number of cells,
are connected in parallel; this increas-
es battery capacity.
38
GLOSSARY

Short Circuit Current The current
that flows when the two terminals of a
cell or battery are inadvertently con-
nected to each other.
Side Terminal SLI battery design
with two through-the-container current
connections on one side instead of two
posts on top.
SLI Battery A battery for automotive
use in starting, lighting and ignition.
Sliver, Slyver Extremely fine paral-
lel glass fibers used in retainers next to
positive plates to retard shedding.
Smelting The primary process for
recovering lead and antimony from
scrapped batteries and scrap from bat-
tery manufacture.
Soaking A manufacturing process
following pasting that soaks certain
types of lead plates in sulfuric acid.
This provides a protective surface and
also sulfate helpful in container and
tank formation.
Soda Ash Sodium Carbonate
(Na2CO3) used in neutralizing sulfuric
acid in spills or effluents.
Spalling Shedding of active material,
usually from positives, during formation
due to incomplete or improper plate
curing.
Sponge Lead (Pb) A porous mass
of lead crystals and the chief material
of a full-charged negative plate.
Stacking A cell assembly operation,
alternately piling plates and separators
in a burning box prior to attachment of
straps and posts.
Standard Battery Any of
Conventional, YuMicron or YuMicron CX
batteries consisting of flooded elec-
trolyte and cell accessible construction.
Standing Loss Loss of charge by an
idle cell or battery, resulting from local
action.
Starting Rate A beginning charg-
ing rate that does not produce gassing
or temperatures in excess of 110°F.
State of Charge The amount of
electrochemical energy left in a cell or
battery.
Strap Precast or cast-on piece of
lead or lead alloy used to connect
plates into groups and to connect
groups to the post.
Strap Center Spacing between cen-
ters of adjacent plates in a group.
Stratification Layering of high spe-
cific gravity electrolyte in lower por-
tions of a cell, where it does not circu-
late normally and is of no use.
Sulfated A plate or cell whose
active materials contain an apprecia-
ble amount of lead sulfate.
Sulfation Formation of lead sulfate
on a plate or cell as a result of dis-
charge, self-discharge or pickling.
Sulfuric Acid (H2SO4) The princi-
pal acid compound of sulfur, sulfuric
acid in dilute and highly pure form is
the electrolyte of lead acid storage
cells.
Tack Burn A shallow burn used to
tack together two lead parts.
Tank Formation Electrolytic pro-
cessing of plates prior to assembly in
large tanks of acid.
Temperature Correction In stor-
age cells, specific gravity and charg-
ing voltage vary inversely with temper-
ature, while the open circuit voltage
varies directly though slightly with tem-
perature.
Terminals The points on a battery to
which the external circuit is connected.
Terminal Cable A length of insulat-
ed cable, one end connected to the
battery terminal post, and the other fit-
ted with a plug, receptacle, lug or
other device for connection to an
external circuit.
Top Pour A method of casting in
which molten metal is poured, usual-
ly by hand, into a top gated mold.
Treeing Growth of a lead dendrite or
filament through a crack or hole of a
separator, short-circuiting the cell.
Trickle Charge A low-rate contin-
uous charge approximately equal to
a battery’s internal losses and capa-
ble of maintaining the battery in a
fully-charged state.
TVR A temperature compensating
voltage relay used in charging
equipment.
Unformed A plate that has not
been electrolytically formed.
Useful Acid The acid above the
lower edges of the plates that takes
part in the discharge reactions that
occur within a cell.
Vent An opening that permits the
escape of gas from a cell or mold.
Vent Plug The seal for the vent
and filling well of a cell cover, con-
taining a small hole for escape of
gas.
Vent Well The hole or holes in a
cell cover that allow fluids to be
checked, electrolyte to be added,
and gas to escape. The vent plug fits
into the vent well.
Verticals The members in a plate
grid.
Volt The unit of measurement of
electromotive force, being the force
needed to send a current or one
ampere through a conductor with a
resistance of one ohm.
39
GLOSSARY

Volt Efficiency The ratio of the
average voltage of a cell or battery
during discharge to the average volt-
age during subsequent recharge.
Voltage The difference in electrical
potential that exists between the ter-
minals of a cell or battery or any two
points of an electrical circuit.
Voltage Range The difference
between maximum and minimum cell
voltages within a battery or string of
cells when all cells are charging and
discharging.
Voltmeter An instrument for mea-
suring voltage.
VRLA (Valve Regulated Lead
Acid) Sealed batteries which fea-
ture a safety valve venting system
designed to release excessive internal
pressure, while maintaining sufficient
pressure for recombination of oxygen
and hydrogen into water.
Watering Adding water to battery
electrolyte to replace loss from elec-
trolysis and evaporation.
Watt A unit of electric power, equal
to a current of one ampere under
one volt of pressure.
Watthour A unit of electrical ener-
gy or work, equal to one watt acting
for one hour.
Watthour Capacity The num-
ber of watthours a storage battery
can deliver under specific conditions
of temperature, rate of discharge
and final voltage.
Watthour Efficiency A storage
battery’s energy efficiency expressed
as ratio of watthour output to the
watthours of the recharge.
Watthour Meter An electric
motor that measures and registers
electrical energy in watthours.
Wet Shelf Life The time a wet sec-
ondary cell can be stored before its
capacity falls to the point that the cell
cannot be easily recharged.
40
GLOSSARY

Part No. YUA00ACC02
Syringe Hydrometer
Part No. YUAMHYDRO
•Shows exact specific gravity
reading on conventional
battery’s condition
Battery Analyzer
Part No. YUA00ACC06
•Easy installation
•Shows battery condition at the push of a button
•Offers the confidence of knowing when your battery is
fully charged
Digital Multimeter
Part No. YUA00ACC05
•Simple to use
•Easy and accurately measures
current,voltage and resistance
•Eliminates the guesswork when
your battery needs recharging
•A must for checking maintenance
free batteries as well as
conventional batteries
•90 day limited warranty
Battery Accessory Lead
and Fused Ring Connectors
•Simple, quick connection
•Can be used with Yuasa's 1.5 Amp and 900 mA chargers
for applications ranging from motorcycles, automobiles and
personal watercraft to ATVs, snowmobiles and riding mowers
BATTERY ACCESSORIES
Part No. YUA00ACC04
Battery Tester
Part No. YUA00BTY01
•Developed exclusively for the
Powersports industry
•Determines accurate battery status
in seconds
•Single load, dynamic resistance
technology minimizes battery
drain during test
•Tests both charged and dis
charged batteries accurately
•1 year limited warranty
Jumper Cables
Part No. YUA00ACC07
•Easy to use, easy to store
•Convenient storage bag
•Heavy duty, 8 ft., 8-gauge
cables won’t stiffen or freeze
•Tangle resistant, encased
rubber grips for safer use

FORM NO. YUA-069 (Rev. 4/04) PG – 10,000 PRINTED IN U.S.A.
P.O. Box 14715
Reading, PA 19612-4715
Telephone: 1-866-431-4784 • Fax: 610-929-1295
yuasabatteries.com
TM
Tank Sports Inc.
10925 Schmidt Rd
El Monte, CA 91733
www.tank-sports.com
1-866-969-8265 Orders
1-626-350-4039 Info
1-626-602-8410 FAX