iHealth Labs BP7S Wireless Blood Pressure Wrist Monitor User Manual 15 BP7S UserMan
iHealth Lab Inc. Wireless Blood Pressure Wrist Monitor 15 BP7S UserMan
15_BP7S UserMan
iHealth®View
Wireless Blood Pressure Wrist Monitor (BP7S)
USER GUIDE
Table of Contents
INTRODUCTION...............................................................................................................................2
PACKAGE CONTENTS.................................................................................................................... 2
INTENDED USE................................................................................................................................ 2
CONTRAINDICATION..................................................................................................................... 2
PARTS AND DISPLAY INDICATORS............................................................................................. 2
SET UP REQUIREMENTS................................................................................................................2
SET UP PROCEDURES.....................................................................................................................3
MEASUREMENT PROCEDURES................................................................................................ 3
SPECIFICATIONS............................................................................................................................. 4
GENERAL SAFETY AND PRECAUTIONS....................................................................................5
BATTERY HANDLING AND USAGE............................................................................................. 6
TROUBLESHOOTING......................................................................................................................6
CARE AND MAINTENANCE.......................................................................................................... 7
WARRANTY INFORMATION......................................................................................................... 8
EXPLANATION OF SYMBOLS.......................................................................................................8
CONTACT AND CUSTOMER SERVICE........................................................................................ 8
IMPORTANT INFORMATION REQUIRED BY THE FCC............................................................ 9
OTHER STANDARDS AND COMPLIANCES................................................................................9
ELECTROMAGNETIC COMPATIBILITY INFORMATION......................................................... 9
INTRODUCTION
Thank you for selecting the iHealth View Wireless Blood Pressure Wrist Monitor. The iHealth Wireless
Blood Pressure Wrist Monitor is a fully automatic wrist cuff blood pressure monitor that uses the
oscillometric principle to measure your blood pressure and pulse rate. The monitor works with
your mobile devices to track and share vital blood pressure data.
PACKAGE CONTENTS
* 1 iHealth View Wireless Blood Pressure Wrist Monitor
*1UserGuide
* 1 Quick Start Guide
* 1 Charging Cable
* 1 Travel Case
INTENDED USE
The iHealth View Wireless Blood Pressure Wrist Monitor (Electronic Sphygmomanometer) is intended for use
in a professional setting or at home and is a non-invasive blood pressure measurement system. It
is designed to measure the systolic and diastolic blood pressures and pulse rate of an adult
individual by using a technique in which an inflatable cuff is wrapped around the wrist. The
measurement range of the standard cuff circumference is 5.3” to 8.7”(13.5cm-22cm).
Note: Consult your physician for proper interpretation of blood pressure results.
CONTRAINDICATION
It is not recommended for people with serious arrhythmia to use this Wireless Blood Pressure
Monitor.
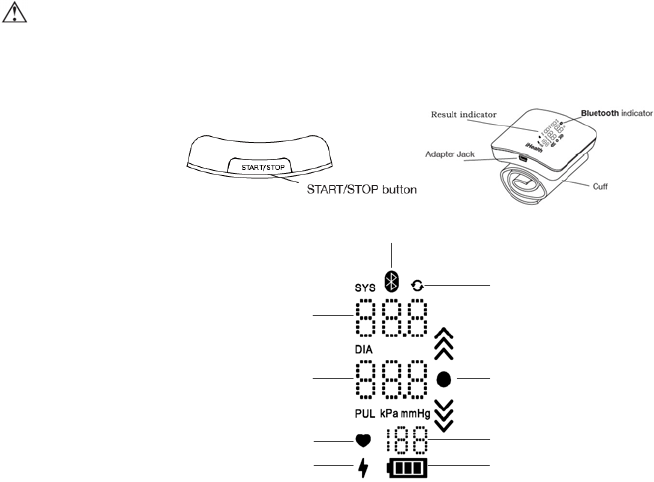
PARTS AND DISPLAY INDICATORS
Systolic
Diastolic
Pulse
Battery
Sync indicator
Charging indicator
Position detect indicator
Bluetooth indicator
Pulse indicator
SET UP REQUIREMENTS
The iHealth View Wireless Blood Pressure Wrist Monitor is designed to be used with the following iPod touch,
iPhone and iPad models:
iPhone4s+
iPad Air+
iPad mini+
iPad 3+
iPod Touch5
Please note that the compatible devices are subject to change. For the latest compatibility list(include Android
devices), visit www.ihealthlabs.com/support
SET UP PROCEDURES
Download the Free iHealth MyVitals App
Prior to first use, download and install “iHealth MyVitals”from the App Store or Google Play Store. Follow the
on-screen instructions to register and set up your personal account.
Access the iHealth Cloud Account
Your iHealth account also gives you access to the free and secure iHealth cloud service. Go to
www.ihealthlabs.com and sign in with the same account.
Charge Battery before First Use
Connect the monitor to a USB port using the charging cable provided until indicator steady.
BODY POSTURE DURING MEASUREMENT
Blood pressure can be affected by the position of the cuff and your physiologic condition. It i
s
very important that the cuff is positioned at your heart level during blood pressur
e
measurements.
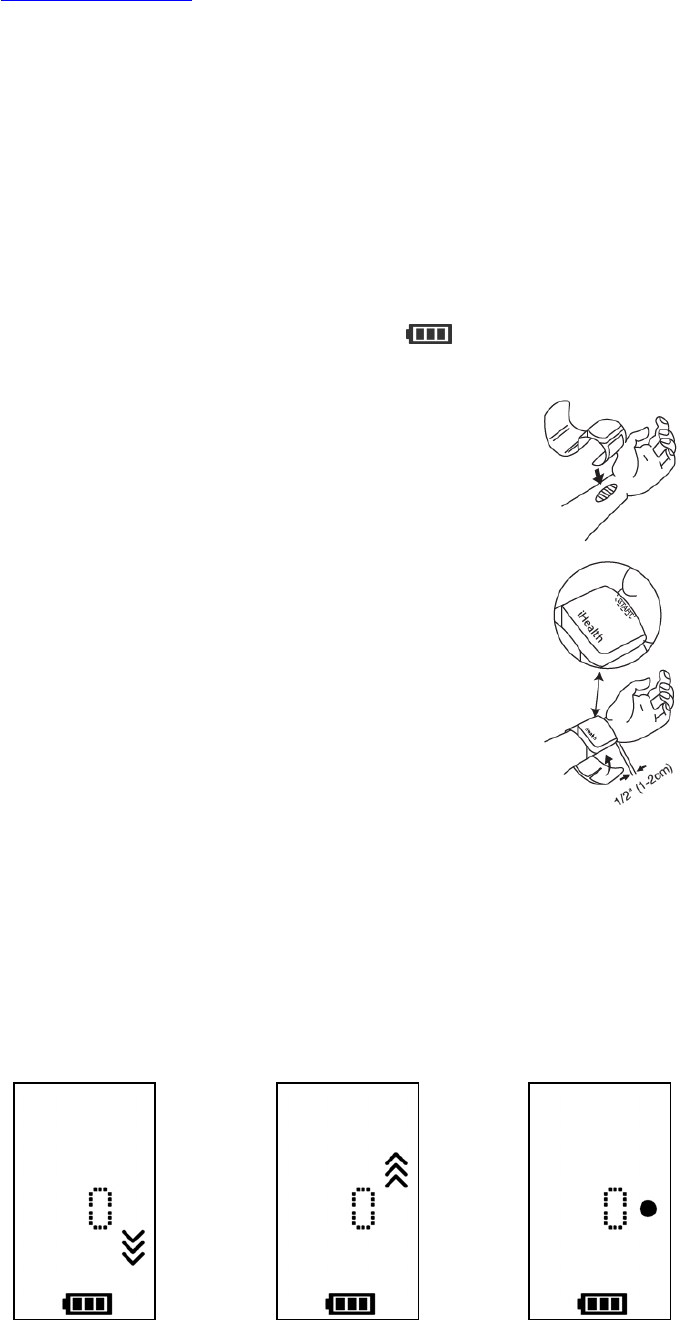
1. Sit comfortably with your feet flat on the floor without
crossing your legs. Stay still during measurement. Do not
move your wrist, body, or the monitor.
2. Place your hand palm-side up in front of you and leave 1-2cm
between the monitor and the bottom of your palm. If the
monitor is correctly placed, iHealth logo will be facing
upright.
3. The center of the cuff should be at your heart level.
4. It is advised to place the monitor’s travel case under your
arm for support and to keep your arm at optimal height for
measurement.
TAKING YOUR BLOOD PRESSURE READING
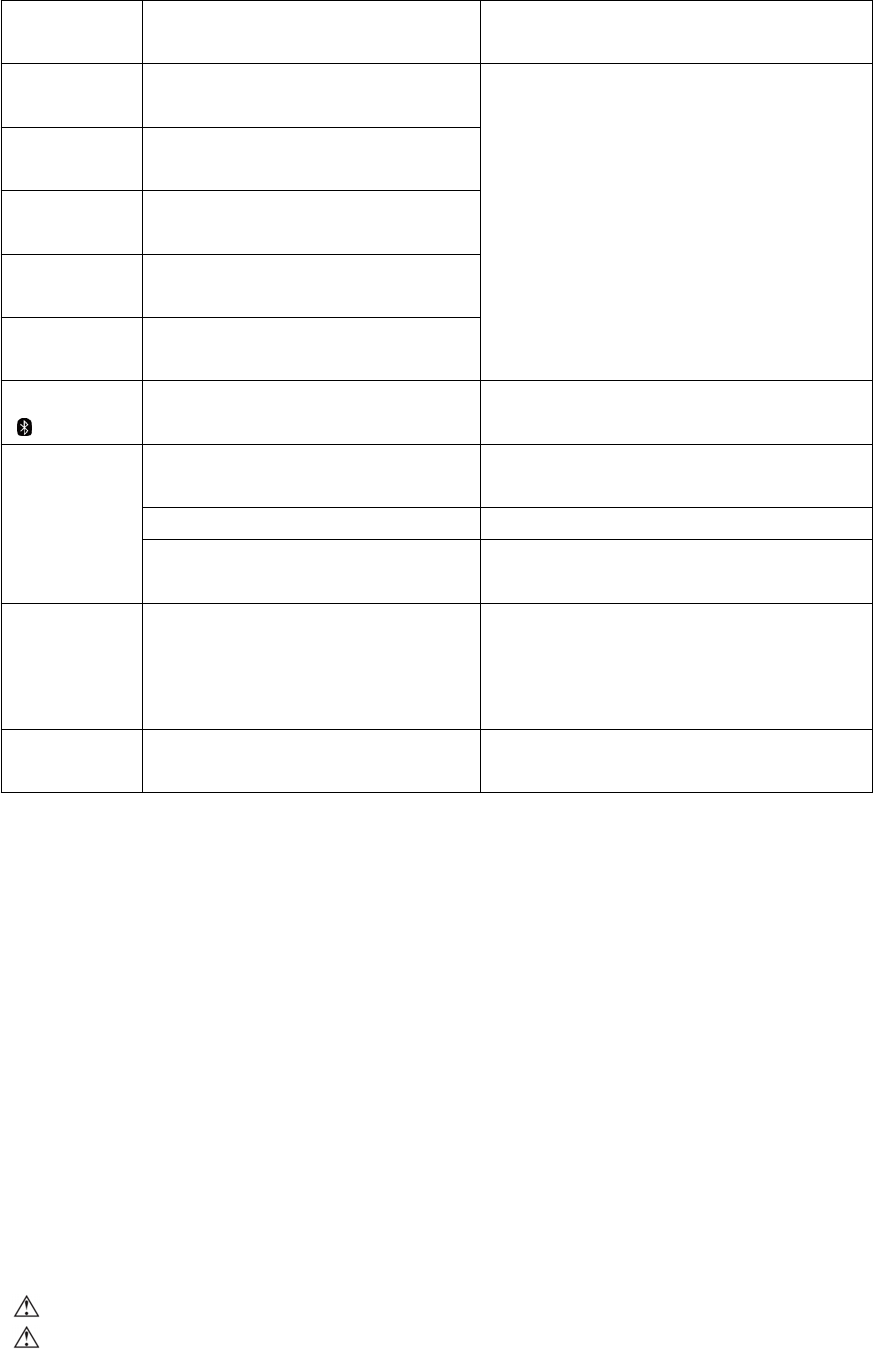
1. Apply the cuff or press the START/STOP button, the monitor will activate and begin to detect your wrist position.
2. Adjust the height of your wrist, the monitor will detect your wrist position and the measurement will start ONLY when the
correct position is detected. When the position is correct, press "START/STOP" button again to start measurement.
Wrist position too low Wrist position too high Wrist position good

3. Then the cuff will be slowly inflated. The blood pressure and pulse will be measured during inflatio
n
Inflation will stop as soon as the blood pressure and pulse rate have been calculated and displayed o
n
the screen. The result will be automatically stored in the memory, and all results will be uploaded to th
e
App automatically upon the next successful Bluetooth connection.
SYNC TIME AND RESULTS VIA BLUETOOTH
Connect to iOS Device via Bluetooth
• Press the START/STOP button for 3 seconds, the Bluetooth indicator will begin flashing.
• Enable the Bluetooth on your iOS device(Setting->General->Bluetooth->On) .
• Launch the "iHealth MyVitals" app to initiate the connection, the Bluetooth indicator will light up and stop flashing when a
successful connection is established.
• When sync is processing, the sync indicator flash and , when sync is finished, the sync indicator lights up .
Connect to Android Device via Bluetooth
• Press the START/STOP button for 3 seconds, the Bluetooth indicator will begin flashing.
• Enable the Bluetooth on your Android Device.
• When using the monitor for the first time, you should pair the monitor to the Android device. Go into your device’s setting to
pair it with your Android device. Check the Bluetooth menu for the model name of your monitor (BP7SXXX) to appear, and then
select it to pair. This may take up to 30 seconds and your monitor’s Bluetooth indicator will light up when a connection has been
established.
• Launch the “iHealth MyVitals” app to sync with your monitor.
• When sync is processing, the sync indicator flash and , when sync is finished, the sync indicator lights up .
• Please repeat these steps when you switch to another Android device with the monitor.
Remember to:
1. Make sure your wrist size is within cuff circumference; see the range in “SPECIFICATIONS”
2. Measure on the same wrist each time.
3. Stay still and calm for one to one and half minutes before taking a blood pressure measurement. Prolonged over-inflation of
the bladder may cause bruises of your wrist.
4. Keep the cuff clean. Cleaning the cuff after every 200 times of usage is recommended. If the cuff becomes dirty, clean it with
a moistened cloth. Do not rinse the monitor or cuff with running water.
5. Press the “START/STOP” button at any time to interrupt a measurement.
Note: Physical activity, eating, drinking, smoking, excitement, stress, and many other factors influence blood pressure results.
SPECIFICATIONS
1. Product name: iHealth View Wireless Blood Pressure Wrist Monitor
2. Model: BP7S
3. Classification: Internally powered; Type BF applied part; IP22, No AP or APG; Continuous operation
4. Machine size: approx. 2.8"×2.9"×0.7" (72mm×74mm×17.6mm)
5. Cuff circumference: 5.3"- 8.7" (13.5cm-22cm)
6. Weight: approx. 4.2oz(120g)(including cuff)
7. Power: DC:5.0V 1.0A, Battery: 1*3.7V Li-ion 400mAh
8. Measurement range:
Cuff pressure: 0-300 mmHg
Systolic: 60-260 mmHg
Diastolic: 40-199 mmHg
Pulse rate: 40-180 beats/minute
9. Accuracy:
Pressure: ±3 mmHg
Pulse rate: ±5%
10. Environmental temperature for operation: 10°C-40°C(50°F -104°F)
11. Environmental humidity for operation: ≤85%RH
12. Environmental temperature for storage and transport:-20°C-55°C(-4°F-131°F)
13. Environmental humidity for storage and transport: ≤85%RH
14. Environmental pressure: 80kPa-105kPa
15. Battery life: more than 80 measurements on a full charge
16. The blood pressure measurement system includes accessories: pump, valve, cuff, and sensor.
Note: These specifications are subject to change without notice.
GENERAL SAFETY AND PRECAUTIONS
1. Read all of the information in the User Guide and other provided instructions before operating the unit.
2. Consult your physician for any of the following situations:
a) The application of the cuff over a wound or inflamed area.
b) The application of the cuff on any limb with intravascular access or therapy, or an arteriovenous (A-V) shunt.
c) The application of the cuff on the arm on the side of a mastectomy.
d) Simultaneous use with other medical monitoring equipment on the same limb.
e) The blood circulation of the user needs to be checked.
3. Do not use this product in a moving vehicle as this may result in inaccurate measurements.
4. Blood pressure measurements determined by this product are equivalent to those obtained by professional healthcare
practitioners using the cuff/stethoscope auscultation method within the limits prescribed by the American National Standard,
Electronic or Automated Sphygmomanometer. This device is also clinically validated according to the 2010 Protocol of the
European Society of Hypertension (ESH 2010).
6. If an Irregular Heartbeat (IHB) is detected during the measurement procedure, the IHB symbol will be displayed in the
“iHealth MyVitals” APP. Under this condition, the Wireless Blood Pressure Wrist Monitor can keep functioning, but the results
may be inaccurate. Please consult your physician for accurate assessment.
The IHB symbol will be displayed under 2 conditions:
1) The coefficient of variation (CV) of pulse period >25%.
2) The difference of adjacent pulse period is ≥0.14s and more than 53 percent of the total number of pulses readings falls within
this definition.
7. Please do not use any cuff other than that supplied by the manufacturer as this may result in inaccurate measurements.
8. For information regarding potential electromagnetic or other interference between the blood pressure monitor and other
devices together with advice regarding avoidance of such interference, please see ELECTROMAGNETIC COMPATIBILITY
INFORMATION. It is suggested that the blood pressure monitor should be operated at least 10 meters away from electric or
wireless devices (e.g. routers, microwave oven, etc.)
9. If the blood pressure measurement (systolic or diastolic) is outside the rated range specified in part SPECIFICATIONS, the
monitor will immediately display a technical alarm on the screen. In this case, repeat the measurement ensuring that the proper
measurement procedures are followed and/or consult with your medical professional. The technical alarm is preset in the factory

and cannot be adjusted or inactivated. This technical alarm is assigned as low priority according to IEC 60601-1-8. The technical
alarm does not need to be reset.
10. This device requires a medical AC adapter with an output of DC 5.0V that complies with IEC 60601-1/UL 60601-1 and IEC
60601-1-2/EN 60601-1-2 such as ASP5-05010002JU (input: 100-240V, 50/60Hz, 200mA; output: DC 5V, 1.0A). Please note that
the monitor jack size is USB mini B. The USB jack should be used for charging only.
This Monitor is designed for adults and should never be used on infants, young children, pregnant or pre-eclamptic patients.
Consult your physician before use on children.
This product might not meet its performance specifications if stored or used outside the specified temperature and humidity
ranges.
Please do not share the cuff with any infectious person to avoid cross-infection.
BATTERY HANDLING AND USAGE
•The battery charge will be displayed on the LED screen when the monitor is active. And when the monitor is connected to the
“iHealth MyVitals” APP, the battery charge will be displayed in the APP. If the power is less than 25%, please charge the battery.
The monitor will not work until the battery has enough power.
• When the monitor needs charging, please connect the monitor to a power source. • You should charge the battery when the
battery is less than 25% charged. Overcharging the battery may reduce its lifetime.
• When in charging mode, the charging status will be displayed on the LED screen. See the table below for details.
Monitor Status Status Indicator
Charging rolling
Fully charged symbol steady
Low battery symbol flashing (for a few seconds)
Do not change the battery. If the battery can no longer be charged, please contact Customer Service.
Overcharging the battery may reduce its lifetime.
Lithium battery replacement by inadequately trained personnel could result in a hazard such as a fire or explosion.
Do not plug or unplug the power cord into the electrical outlet with wet hands. If the AC adapter is abnormal, please
change the adapter.
Do not pull out the adapter when you are using the monitor.
Do not use any other type of AC adapter as it may harm the monitor.
The monitor, cable, battery and cuff must be disposed of according to local regulations at
the end of their usage.
Note: Battery life and charge cycles vary by use and settings.
TROUBLESHOOTING
PROBLEM POSSIBLE CAUSE SOLUTION
Low Battery Battery do not have enough power Charge the battery
LED display reads
“Er0”
Pressure system is unstable before measurement
Retest, make sure not to move your arm or the monitor
LED display reads
“Er1”
Fail to detect systolic pressure
LED display reads
“Er2”
Fail to detect diastolic pressure
LED display reads
“Er3”
Pneumatic system blocked or cuff is too tight during inflation Apply the cuff correctly and try again
LED display reads
“Er4”
Pneumatic system leakage or cuff is too loose during inflation
LED display reads
“Er5”
Cuff pressure above 300mmHg
Measure again after five minutes. If the monitor is still abnormal, please
contact the local distributor or the factory.
LED display reads
“Er6”
More than 160 seconds with cuff pressure above 15 mmHg
LED display reads
“Er7”
memory accessing error
LED display reads
“Er8”
Device parameter checking error
LED display reads
“ErA”
Pressure sensor parameter error
LED display reads
“Er”
Bluetooth communicate error
Connect the mobile device correctly and try again, If the monitor is still
abnormal, please contact the local distributor or the factory.
LED display reads
an abnormal
result
The cuff position was not correct or it was not
properly tightened
Review the cuff application instructions and retest
Body posture was not correct during testing Review body posture instructions and retest
Speaking, moving arm or body, being angry,
excited or nervous during test
Retest when calm; avoid speaking or movement during the test
Bluetooth
connection
unstable
Bluetooth connection unsuccessful, monitor is
abnormal, or strong electromagnetic interference
is present
Reset iOS/Android device. Reset monitor by pressing the START/STOP
button and holding for about 10 seconds. Make sure the monitor and
iOS/Android device are away from other electrical equipment. Please see
GENERAL SAFETY AND PRECAUTIONS
No response when you
press button
Incorrect operation or strong electromagnetic interference
Press the START/STOP button and hold for about 10 seconds to reset the
device.
CARE AND MAINTENANCE
1. If this monitor is stored near freezing temperatures, allow it to return to room temperature before use.
2. If the monitor is not used for a long time, please be sure to fully charge it every month.
3. No monitor component needs to be maintained by the user. The circuit diagrams, component part lists, descriptions, calibration
instructions, or other information which will assist the user’s appropriately qualified technical personnel to repair those parts of
the equipment which are designated for repair can be supplied by the iHealth technical department.
4. Clean the monitor with a dry, soft cloth or a moistened and well wrung soft cloth using water, diluted disinfectant alcohol, or
diluted detergent.
5. The monitor can maintain the safety and performance characteristics for a minimum of 10,000 measurements or three years of
usage, and the cuff integrity is maintained after 1,000 open close cycles.
6. The battery can maintain the performance characteristics for a minimum of 300 charge cycles.
7. It is recommended that if the cuff is used in a hospital or a clinic, it be disinfected twice a week. Wipe the inner side (the side
that contacts skin) of the cuff with a soft cloth lightly moistened with Ethyl alcohol (75-90%). Then air dry the cuff.
8. It is recommended that product performance be checked every 2 years or after each repair. Please contact the
iHealth Customer Service Center to do so.
Do not drop this monitor or subject it to strong impact.
Avoid high temperature and direct sunlight. Do not immerse the monitor in water as this will result in damage to the
monitor.
Do not attempt to disassemble this monitor.
Battery replacement should only be performed by a qualified iHealth technician. To do otherwise will void your warranty
and possibly damage your unit.
Cuff replacement should only be performed by a qualified iHealth technician. To do otherwise
will possibly damage your unit.
WARRANTY INFORMATION
The iHealth View Wireless Blood Pressure Wrist Monitor is warranted to be free from defects in materials and workmanship
within one year from the date of purchase when used in accordance with the provided instructions. The warranty extends only to
the end user. We will, at our option, repair or replace without charge the iHealth View Wireless Blood Pressure Wrist Monitor
covered by the warranty. Repair or replacement is our only responsibility and your only remedy under the warranty.
EXPLANATION OF SYMBOLS
Symbol for "TYPE BF APPLIED PARTS" (cuff only)
Symbol for "THE OPERATION GUIDE MUST BE READ"
The sign background color: blue The sign graphical symbol: white
Symbol for "ENVIRONMENT PROTECTION – Waste electrical products should not be
disposed of with household waste. Please recycle where facilities exist. Check with your
local authority or retailer for recycling advice".
Symbol for "KEEP DRY"
Symbol for "WARNING"
Symbol for "MANUFACTURER"
Symbol for "SERIAL NUMBER"
Symbol for "EUROPEAN REPRESENTATIVE"
Symbol for "COMPILES WITH MDD93/42/EEC REQUIREMENTS"
iHealth is a trademark of iHealth Lab Inc.
"Made for iPod", "Made for iPhone", and "Made for iPad" mean that an electronic accessory has been designed to connect
specifically to iPod, iPhone, or iPad, respectively, and has been certified by the developer to meet Apple performance standards.
Apple is not responsible for the operation of this device or its compliance with safety and regulatory standards. Please note that
the use of this accessory with iPod, iPhone, or iPad may affect wireless performance. iPad, iPhone, and iPod touch are
trademarks of Apple Inc., registered in the U.S. and other countries.
CONTACT AND CUSTOMER SERVICE
Manufactured for iHealth Lab Inc.
719N . Shoreline Blvd.,Mountain View, CA 94043, USA
Tel:+1-855-816-7705 www.ihealthlabs.com

iHealthlabs Europe
3 Rue Tronchet,75008,Paris,France
support@ihealthlabs.eu www.ihealthlabs.eu
ANDON HEALTH CO., LTD.
N
o. 3 Jinping Street, YaAn Road, Nankai District, Tianjin 300190, China.
Tel: 86-22-60526161
IMPORTANT INFORMATION REQUIRED BY THE FCC
This device complies with Part 15 of the FCC Rules. Its operation is subject to the following two conditions:
(1) This device may not cause harmful interference, and
(2) this device must accept any interference received, including interference that may cause undesired operation.
Changes or modifications not expressly approved by iHealth Lab Inc. would void the user’s authority to operate the product.
Note: This product has been tested and found to comply with the limits for a Class B digital device, pursuant to Part 15 of the
FCC Rules. These limits are designed to provide reasonable protection against harmful interference in a residential installation.
This product generates, uses, and can radiate radio frequency energy and, if not installed and used in accordance with the
instructions, may cause harmful interference to radio communications. However, there is no guarantee that interference will not
occur in a particular installation. If this product does cause harmful interference to radio or television reception, which can be
determined by turning the equipment off and on, the user is encouraged to try to correct the interference by one or more of the
following measures:
—Reorient or relocate the receiving antenna.
—Increase the separation between the equipment and receiver.
—Connect the equipment into an outlet on a circuit different from that to which the receiver is connected.
—Consult the dealer or an experienced radio/TV technician for help.
This product complies with Industry Canada. IC: RSS-210
IC NOTICE
This device complies with Industry Canada licence-exempt RSS standard(s). Operation is subject to the following two
conditions:
(1) this device may not cause interference, and
(2) this device must accept any interference, including interference that may cause undesired operation of the device.
This product is approved in accordance to R&TTE directive transmitter.
OTHER STANDARDS AND COMPLIANCES
The Wireless Blood Pressure Wrist Monitor corresponds to the following standards:
IEC 60601-1:2005 corr.1(2006)+corr.2(2007)/EN 60601-1:2006/A11: 2011(Medical electrical equipment – Part 1: General
requirements for safety);
IEC 60601-1-2:2007/EN 60601-1-2:2007 /AC:2010 (Medical electrical equipment – Part 1: General requirements for safety;
Collateral Standard-Electromagnetic compatibility - Requirements and tests);
EN 1060-1: 1995 + A1: 2002 + A2: 2009 (Non-invasive sphygmomanometers - Part 1: General requirements);
EN 1060-3: 1997 + A1: 2005 + A2: 2009 (Non-invasive sphygmomanometers - Part 3: Supplementary requirements for
electro-mechanical blood pressure measuring systems);
AAMI/ANSI 80601-2-30:2009/IEC 80601-2-30:2009+Cor.2010/EN 80601-2-30:2010(Medical electrical equipment –Part 2-30:
Particular requirements for the basic safety and essential performance of automated non-invasive sphygmomanometers).
ELECTROMAGNETIC COMPATIBILITY INFORMATION
Table 1
For all ME EQUIPMENT and ME SYSTEMS
Guidance and manufacture’s declaration - electromagnetic emissions
BP7S is intended for use in the electromagnetic environment specified below. The user of BP7S should ensure that it is used in such an
environment.
Emissions test Compliance Electromagnetic environment - guidance
RF emissions
CISPR 11 Group 1
BP7S uses RF energy only for its internal function. Therefore, its RF
emissions are very low and are not likely to cause any interference in nearby
electronic equipment.
RF emissions
CISPR 11 Class B BP7S is suitable for use in all establishments, including domestic
establishments and those directly connected to the public low-voltage power
supply network that supplies buildings used for domestic purposes.
Harmonic emissions
IEC 61000-3-2 Class A
Voltage fluctuations/
flicker emissions
IEC 61000-3-3
Complies
Table 2
For all ME EQUIPMENT and ME SYSTEMS
Guidance and manufacturer’s declaration - electromagnetic immunity
BP7S is intended for use in the electromagnetic environment specified below. The user of BP7S should ensure that it is used in such an
environment.
IMMUNITY test IEC 60601test level Compliance level Electromagnetic environment - guidance
Electrostatic discharge
(ESD)
IEC 61000-4-2
± 6 kV contact
±8kVair
± 6 kV contact
±8kVair
Floors should be wood, concrete or ceramic tile.
If floors are covered with synthetic material, the
relative humidity should be at least 30 %.
Electrical fast
transient/burst
IEC 61000-4-4
± 2 kV for power supply
lines
± 2 kV for power supply
lines
Main power quality should be that of a typical
commercial or hospital environment.
Surge
IEC 61000-4-5
± 1 kV line(s) to line(s)
± 2 kV line(s) to earth
± 1 kV line(s) to line(s)
± 2 kV line(s) to earth
Main power quality should be that of a typical
commercial or hospital environment.
Voltage dips, short
interruptions and
voltage variations
on power supply
input lines
IEC 61000-4-11
<5 % UT
(>95 % dip in UT)
for 0.5 cycle
40 % UT
(60 % dip in UT)
for 5 cycles
70 % UT
(30 % dip in UT)
for 25 cycles
<5 % UT
(>95 % dip in UT)
for 5 s
<5 % UT
(>95 % dip in UT)
for 0.5 cycle
40 % UT
(60%dipinU
T)
for 5 cycles
70 % UT
(30%dipinU
T)
for 25 cycles
<5 % UT
(>95 % dip in UT)
for 5 s
Main power quality should be that of a typical
commercial or hospital environment. If the user of
BP7S requires continued operation during power
main interruptions, it is recommended that BP7S
be powered from an uninterruptible power supply
or a battery.
Power frequency
(50/60 Hz)
magnetic field
IEC 61000-4-8
3 A/m 3 A/m Power frequency magnetic fields should be at
levels characteristic of a typical location in a
typical commercial or hospital environment.
NOTE: UTis the a.c. main voltage prior to application of the test level.
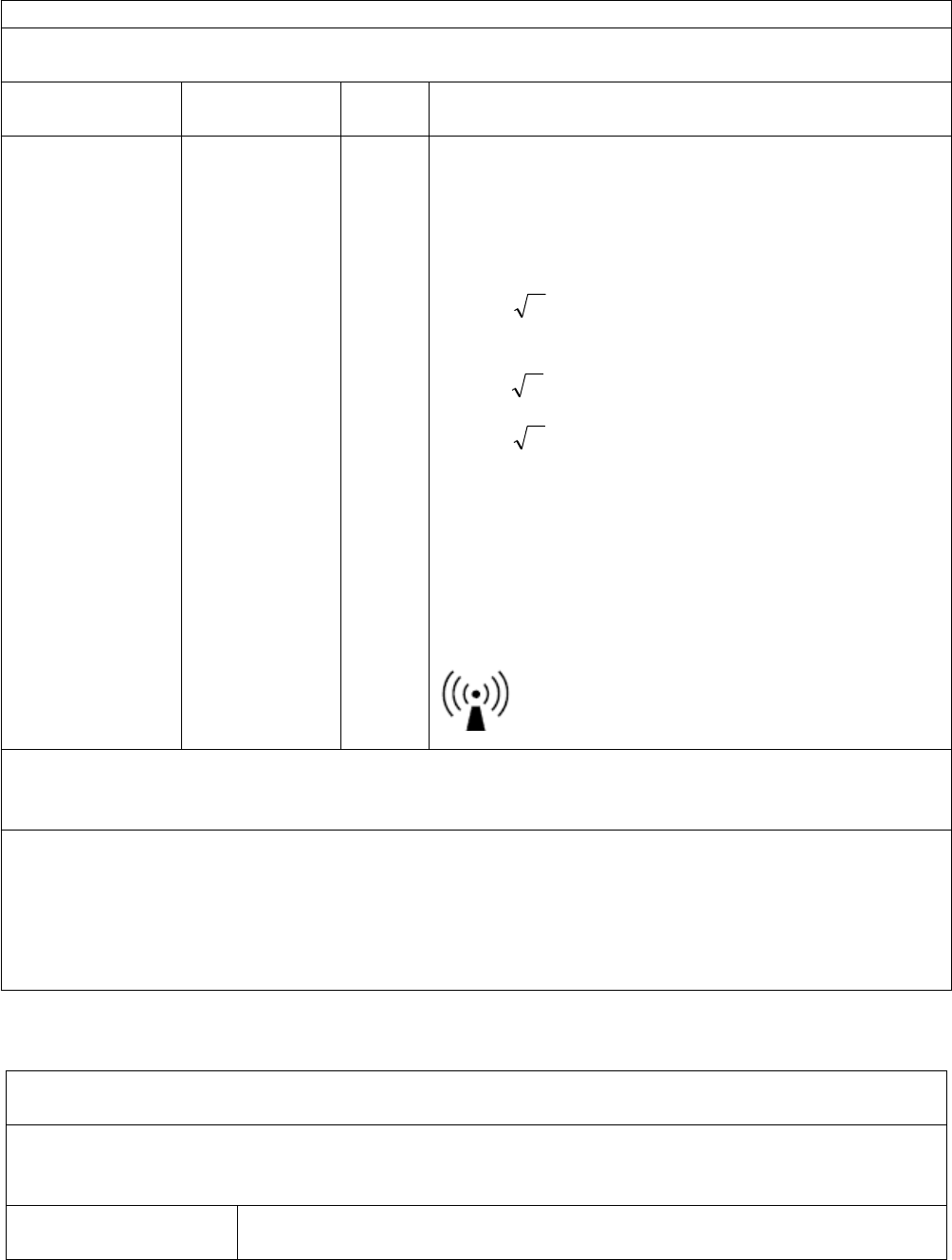
Table 3
For ME EQUIPMENT and ME SYSTEMS that are not LIFE-SUPPORTING
Guidance and manufacturer’s declaration - electromagnetic immunity
BP7S is intended for use in the electromagnetic environment specified below. The user of BP7S should ensure that it is used in such an
environment.
IMMUNITY test IEC 60601test level Complian
ce level Electromagnetic environment - guidance
Conducted RF
IEC 61000-4-6
Radiated RF
IEC 61000-4-3
3 Vrms 150 kHz to
80 MHz
3V/m80MHzto2.5
GHz
3V
3V/m
Portable and mobile RF communications equipment should be used no
closer to any part of BP7S, including cables, than the recommended
separation distance calculated from the equation applicable to the
frequency of the transmitter.
Recommended separation distance:
80 MHz to 800 MHz
800 MHz to 2.5 GHz
Where P is the maximum output power rating of the transmitter in watts (W)
according to the transmitter manufacturer and d is the recommended
separation distance in meters (m).
Field strengths from fixed RF transmitters, as determined by an
electromagnetic site survey,ashould be less than the compliance level in
each frequency range.b
Interference may occur in the vicinity of equipment
marked with the following symbol:
Pd 2.1=
Pd 2.1=
Pd 3.2=
NOTE 1 At 80 MHz and 800 MHz, the higher frequency range applies.
NOTE 2 These guidelines may not apply in all situations. Electromagnetic propagation is affected by absorption and reflection from
structures, objects and people.
a) Field strengths from fixed transmitters, such as base stations for radio (cellular/cordless) telephones and land mobile radios, amateur
radio, AM and FM radio broadcast and TV broadcast cannot be predicted theoretically with accuracy. To assess the electromagnetic
environment due to fixed RF transmitters, an electromagnetic site survey should be considered. If the measured field strength in the
location in which BP7S is used exceeds the applicable RF compliance level above, BP7S should be observed to verify normal operation. If
abnormal performance is observed, additional measures may be necessary, such as re-orienting or relocating BP7S.
b) Over the frequency range 150 kHz to 80 MHz, field strengths should be less than 3 V/m.
Table 4
For ME EQUIPMENT and ME SYSTEMS that are not LIFE-SUPPORTING
Recommended separation distances between
portable and mobile RF communications equipment and the Wireless Blood Pressure Wrist Monitor
BP7S is intended for use in an electromagnetic environment in which radiated RF disturbances are controlled. The user of BP7S can help
prevent electromagnetic interference by maintaining a minimum distance between portable and mobile RF communications equipment
(transmitters) and BP7S as recommended below, according to the maximum output power of the communication equipment.
Rated maximum output
power of transmitter
Separation distance according to frequency of transmitter
m
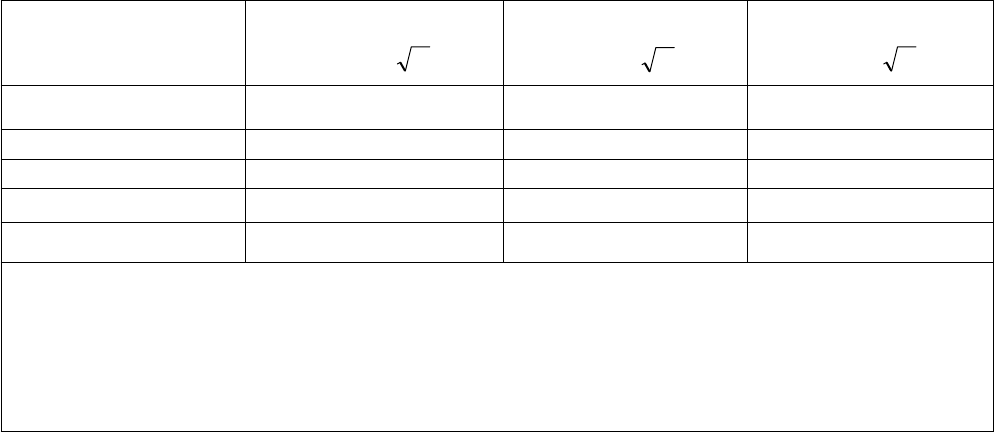
W 150 kHz to 80 MHz
Pd 2.1=
80 MHz to 800 MHz
Pd 2.1=
800 MHz to 2,5 GHz
Pd 3.2=
0.01 0.12 0.12 0.23
0.1 0.38 0.38 0.73
1 1.2 1.2 2.3
10 3.8 3.8 7.3
100 12 12 23
For transmitters rated at a maximum output power not listed above, the recommended separation distance d in meters (m) can be
determined using the equation applicable to the frequency of the transmitter, where P is the maximum output power rating of the
transmitter in watts (W) according to the transmitter manufacturer.
NOTE 1 At 80 MHz and 800 MHz, the separation distance for the higher frequency range applies.
NOTE 2 These guidelines may not apply in all situations. Electromagnetic propagation is affected by absorption and reflection from
structures, objects and people.
BP7S-SMSY01 V1.0