iXensor POC-X01 PixoTest POCT Analyzer User Manual
iXensor Co. Ltd. PixoTest POCT Analyzer Users Manual
iXensor >
Users Manual

文件名稱 Instruction for Use Doc.No.文件編碼 CDM‐C01‐C3‐IFU‐POCX1‐ALL‐01
表單編號:FT‐73‐01‐0008‐0007A2of26
InstructionsforUse
PixoTestPointofCareTest
(POCT)System
文件名稱 Instruction for Use Doc.No.文件編碼 CDM‐C01‐C3‐IFU‐POCX1‐ALL‐01
表單編號:FT‐73‐01‐0008‐0007A3of26
TheInstructionforUseofPixoTestPOCTSystemonPixoTestA1c&Lipidteststrip............1
1.Indicationforuse................................................................................................................4
2.Testprinciple.......................................................................................................................4
3.Beforetest..........................................................................................................................4
4.ThePixoTestPOCTSystemincludes................................................................................5
5.Teststrip.............................................................................................................................5
6.Settings...............................................................................................................................6
7.Preparingformeasurement...............................................................................................6
8.Runningtest‐A1c.................................................................................................................7
9.Runningtest‐lipid................................................................................................................9
10.Understandingresult..........................................................................................................9
11.Record...............................................................................................................................10
12.Calibration.........................................................................................................................11
13.Controlsolutiontest.........................................................................................................11
14.CStestforA1c...................................................................................................................12
15.CStestforLipid.................................................................................................................13
16.ControlSolutionrangeandmeaning................................................................................13
17.Shutdownprocedure........................................................................................................13
18.Maintenanceandtroubleshooting...................................................................................14
19.Cleananddisinfection......................................................................................................14
20.ErrorandWarningmessage.............................................................................................18
21.Disposalinformation.........................................................................................................20
22.Productinformation.........................................................................................................20
23.Limitation..........................................................................................................................22
24.Manufacturer....................................................................................................................22
25.Symbol..............................................................................................................................22
26.Servicing............................................................................................................................23
27.LimitedWarranty..............................................................................................................23

文件名稱 Instruction for Use Doc.No.文件編碼 CDM‐C01‐C3‐IFU‐POCX1‐ALL‐01
表單編號:FT‐73‐01‐0008‐0007A4of26
Appendix1:Reference……………………………………………………………………………………………………………………………………..24
1. Indicationforuse
TheiXensorPixoTestPOCTSystemiscomprisedofPixoTestA1c/LipidTestStrip,PixoTestPOCT
Analyzer,PixoTestPOCTCalibrationCard,andthePixoHealthPOCT‐A1c/LipidAPPasthedisplay
componentofthesystem.TheiXensorPixoTestPOCTSystemisintendedtobeusedforthequantitative
measurementofHbA1candLipids(includingTotalciholesterol,Triglycerides,andHigh‐DensityLipoprotein
(HDL)cholesterol)inwholebloodfrombothcapillaryandvenous.EstimatedvaluesforLow‐Density
Lipoprotein(LDL)cholesterolarecalculatedbythePoxoHealthPOCT‐LipidAPP.
ThePixoTestPOCTSystemisaprofessionaluseonlyproductandintendedforinvitrodiagnosticuseandis
notintendedforuseonneonates. Untrainedpersonnelcouldperformitwell
2. Testprinciple
TheiXensorPixoTestPOCTSystemcombinesenzymaticmethodologyandreflectancephotometryto
measureHbA1candLipids.Samplesfrombothcapillaryandvenouswholebloodcanbeused.Thiswhole
bloodsampleisappliedtoPixoTestA1c/LipidTestStrip,andareactionoccurstoproduceacolorthatis
readbyusingreflectancephotometryofthereagentarea.Thelevelofcolorproducedisproportionaltothe
concentration.
PixoTestPOCTSystem‐A1cusesananti‐HbA1c(%)antibodywhichisspecificforthefirstfewaminoacid
residuesoftheglycatedN‐terminusoftheß‐chainofhemoglobinA0.PixoTestA1cTestKitcontainsthe
PixoTestA1cTestStrip(nitrocellulosemembrane),Spoitwithlatex‐tablet(bluedyedlatexmicroparticles
conjugatedtospecificantibodies)andBuffersolutiontube(hemolysisreagent).Whenwholebloodisadded
tothebuffersolutionandismixedwiththelatex‐tablet,theerythrocytesareinstantlylysedtoreleasethe
glycatedhemoglobin(hereafter,HbA1c).Whenthesamplemixtureisloadedontothesampleportofthe
testpanel,themixturefluidmigratesalongthemembraneofthetestpanelbycapillaryaction,andthe
HbA1cisthenimmobilizedontotheanti‐HbA1cantibodycoatedline.Theamountoftheblueconjugateson
theanti‐HbA1clinereflectstheamountofHbA1cinthesample,theintensityofhemoglobincolor
measuredfromthedesiredareaonthemembraneoftestpanelismeasured.Chemicalandimmune
reactionthatoccursontheteststripismeasuredbytheopticalsysteminPixoTestPOCTSystem.This
systemmeasuresbothfractionsandanalgorithmwhichconvertstheresultintothepercentageHbA1cin
thesample.
PixoTestPOCTSystem‐Lipidscombinesenzymaticmethodologyandreflectancephotometrytomeasure
Totalcholesterol,Triglycerides,andHDLcholesterol.WhenwholebloodsampleisappliedtoPixoTestLipid
TestStrip,thebloodreactstoproducecolorthatisreadbyusingreflectancephotometryofthereagent
area.Thelevelofcolorproducedisproportionaltotheconcentration.Alllipidenzymaticreactionscanrefer
toLipidteststripinsert.
3. Beforetest
BeforeusingthisproducttotestyourA1candLipids,carefullyreadthisInstructionforUse.Priorto
launchingthePixoHealthPOCTA1c/LipidsApp,usethePixoTestPOCTCalibrationCardtoverifythatthe
PixoTestPOCTSystemisworkingproperly.TheA1c/LipidsControlSolutionisusedtoverifythatthe
PixoTestPOCTSystemandPixoTestA1c/LipidTestStripareworkingproperly.
NOTE
Thebatterylifeneedstobeover20%beforeusingthePixoTestPOCTSystem.
DonotchargeyourPixoTestPOCTSystemwhenitisinuse.
Ifthebatteryisbelow20%orcharging,anerrorwillappearandthetestcannotbestarted.
文件名稱 Instruction for Use Doc.No.文件編碼 CDM‐C01‐C3‐IFU‐POCX1‐ALL‐01
表單編號:FT‐73‐01‐0008‐0007A5of26
4. ThePixoTestPOCTSystemincludes
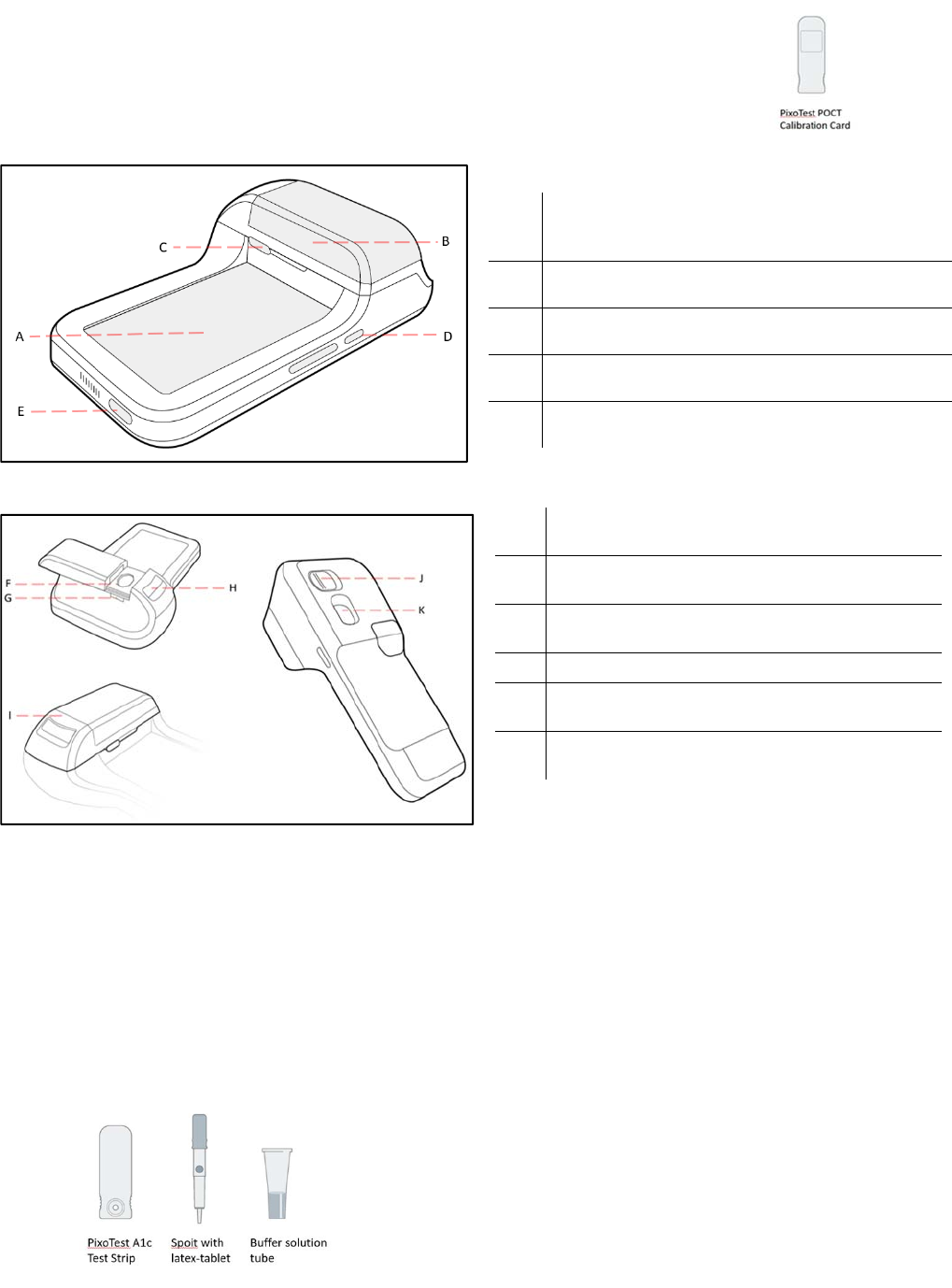
ThePixoTestPOCTAnalyzer:
*PixoHealthPOCTA1c/LipidApp*USBTypeCChargeCable
*USBCharger*Test StripInsert
*IndicationsForUse *PixoTestPOCTCalibrationCard
Fig.1FrontsightofPixoTestPOCTAnalyzer(POC‐X01)
Fig.2BacksightofPixoTestPOCTAnalyzer(POC‐X01)
5. Teststrip
PixoTestPOCTSystemincludesPixoTestPOCTA1cTestkitandPixoTestPOCTLipidTestStrip(bothsold
separately).Thefollowingcontentsareincluded:
PixoTestPOCTA1cTestkit(usewithPixoTestPOCTAnalyzer):
• 20PixoTestA1cTestStrips(fordifferentpackage)
• 20Spoitwithlatex‐tablet
• 20Buffersolutiontube
• 1packageinsert
ADisplay
Showsatestresult,messages,andthetestresults
storedinmemory.
BSlidingDoor
Opentoapplysample.
CItemSelectionKey
Selectmeasurementmode.
D PowerKey
PresstoturnanalyzerONorOFF.
EUSBChargePort
Chargetheanalyzer.
FProtectionCover
Removetoclean.
GLipidTestStripSlot
Insertalipidteststriphere.
HA1cTestStripSlot
InsertA1cteststriphere.
IStripModule
JStripModuleLockSwitch
Unlocktochangethestripmodule.
KCameraModule
ScanQRcode

文件名稱 Instruction for Use Doc.No.文件編碼 CDM‐C01‐C3‐IFU‐POCX1‐ALL‐01
表單編號:FT‐73‐01‐0008‐0007A6of26
PixoTestPOCTLipidTestStrip(usewithPixoTestPOCTAnalyzer):
• 25PixoTestLipidTestStrips(fordifferentpackage)
• 1packageinsert
6. Settings
WhenusingthePixoTestPOCTSystemforthefirsttime,performthefollowingsteps:
Step1.PressthePowerKeyfor2secondstoturnontheAnalyzer.
Step2.SlideupthescreentounlocktheAnalyzer.
Step3.Select“Language”option.Theselectedlanguageisshownonthe2ndlineitem.
Step4.TurnontheWificonnection,typinginthepasswordifnecessary.Connectionstatusisshownatthe
2ndlineitem.
Step5.Click“VersionCheck”toverifythatthemostup‐to‐dateAPPversionisinstalledorneedstobe
updated.
Step6.Select“TimeZone”optiontosetthe24‐hourFormat,andmanuallyselectthecorrecttimezone.
Step7.Press“Next”ontheSystemActivationscreentoscantheproductkeywhichisprovidedbythelocal
distributorortheservicecentertoactivatetheanalyzer.Onceitisactivated,press“Finish”buttonto
completeallsettings.
OnHomescreen,themainscreenofthePixoTestPOCTsystem,pressthe“Settings”buttonforthechange
ofthesystemsettings,includesLanguage,WifiandTimeZone.
NOTE
Aftercompletingallsettings,acalibrationisrequiredbeforeoperating,andmustbedonedaily.Referto
Chapter13forthecalibrationprocess.
7. Preparingformeasurement
TheiXensorPixoTestPOCTSystem
• PixoTestA1c/LipidTestStrip
• PixoTestPOCTAnalyzer:POC‐X01
• PixoHealthPOCT‐A1c/LipidAPP
• PixoTestPOCTCalibrationCard
• A1c/LipidControlSolution
• Spoitwithlatex‐tabletandBuffersolutiontubeforA1ctest
• 35µltubeforLipidtest*
• Instructionsforuse
• Additionalrequiredmaterials:
• Alcoholswabstocleanpuncturesite
• TheEPA‐registereddisinfectantwipestodisinfectthesurfaceofPOCTsystem
• Lancetsforcapillaryorvenousbloodcollectionsupplies(vacuumcollectiontubes,needlesandtube
holders)
• Gloves
• Biohazardwastecontainers
NOTE
*:35µltubearenotincludedandcanbepurchasedfromalocaldistributor;collectsamplewithasuitable
toolfor35µL(ex:micropipetteorquantitativedropper.)

文件名稱 Instruction for Use Doc.No.文件編碼 CDM‐C01‐C3‐IFU‐POCX1‐ALL‐01
表單編號:FT‐73‐01‐0008‐0007A7of26
POC‐X01
PixoTestPOCTSystemisintendedtomonitorA1candLipidsusingPOC‐X01(Fig.1)toconductthetest.
Itemselectionkey:TotestA1C,slidetheItemSelectionkeytothe“II”indication.TotestLipids,slidethe
itemselectionkeytothe“I”indication.
Slidingdoor:WhentestingLipids,opentheslidingdoortoinsertthePixoTestlipidteststripintotheLipid
TestStripSlot.
A1cTestStripSlot:ToinsertthePixoTestA1cteststripintotheA1cteststripslot(Fig.2).
LipidTestStripSlot:ToinsertthePixoTestlipidteststripintothelipidteststripslot(Fig.2)
CAUTION
• KeepPixoTestLipidTestStripstoredinadryplacebetween36–90°F(2–32°C),andavoiddirectsun
exposure.
• KeepA1cTestStripstoredinadryplacebetween34–86°F(1–30°C),andavoiddirectsunexposure.
• DoNotopentheteststrippackageuntilreadytoperformthetest.Useteststripimmediatelyafter
removalfromthepackage.
• DoNotreturntheusedteststriptotheoriginalboxafterperformingthetest;disposeofusedteststrip.
• DoNotre‐useateststripthathadbloodorcontrolsolutionappliedtoit.Thestripisforsingleuseonly.
• DoNotmodifytheteststripinanyway.
• DoNotuseyourPixoTestA1c/LipidTestStripifthepackageisdamagedorleftopentoair.
• DoNotusethedevicenearsourcesofelectromagneticradiationsuchasmicrowaves.
• DoNotcarryoutthetestindirectsunlight.Themeasurementshouldbeperformedinanappropriate
lightenvironment.
• DoNotuseteststripaftertheexpirationdate(printedonthepackage)oryourresultsmaybe
inaccurate.
• AlwayskeepPOC‐X01clean.
• Confirmthatthecodenumberisthesameasthecodeonthefoilpouchpackage.
• Toconservepower,theAnalyzerturnsitselfoffafter10minutesunlessapowerbuttonispressed.When
theAnalyzerturnsitselfoff,alltestresultsobtainedsofarremaininthememory.
• Whenthelowbatterywarning(lessthan20%)appears,atestcannotbeperformed.ChargetheAnalyzer
immediately.
• Measurementresults,includingtherelatedmeasurementdateandtimeaswellasallotherAnalyzer
settings,remainstoredevenwhenthereisnopower.
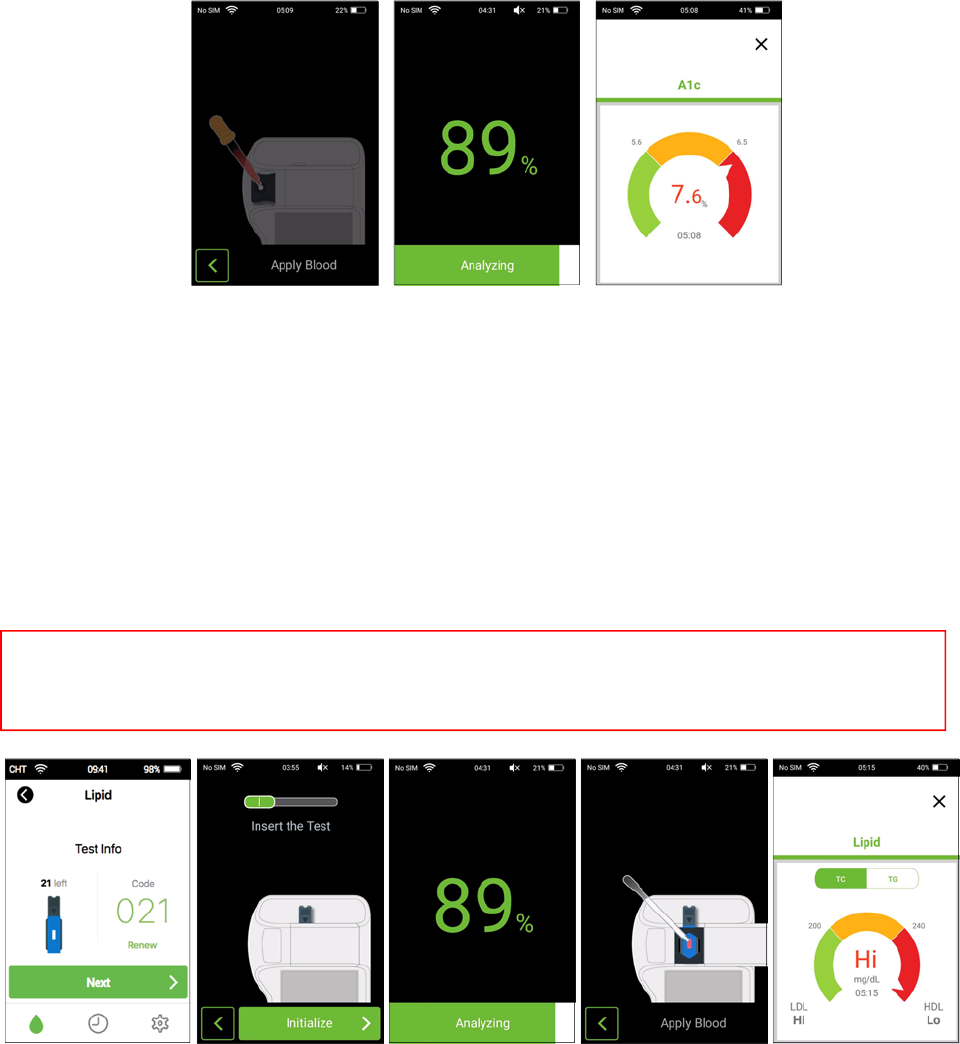
8. Runningtest‐A1c
Step1.Beforetesting,washyourhandswithwarm,soapywater.Rinseanddrycompletely.
Step2.Tap“Settings”buttonontheHomescreenofPixoTestPOCTSystemandtap“A1c” buttontoopen
thePixoHealthPOCT‐A1cApp.
Step3.Tap“Renew”toscantheQRcode,pastedontheteststripbox.
Step4.Tap“Next”buttontostartA1cmeasurement.
Step5.Pleaseslidetheitemselectionkeytothe“II”indication.Checkthestripcodenumberandinsertthe
teststripintheslot.Press“initialize”buttonforinitialization.
NOTE
1. PleasemakesurethePOC‐X01isplacedonaflatsurface.
2. Thesystemshowsthenumberoftheremainingtest.PleaseopenanewstripboxandscanitsQRcode
toresetthecountingwhentheremainingtestsreacheszero.

文件名稱 Instruction for Use Doc.No.文件編碼 CDM‐C01‐C3‐IFU‐POCX1‐ALL‐01
表單編號:FT‐73‐01‐0008‐0007A8of26
Step6.Obtainabloodsampleusingthelancetandlancingdevice.(Ifyouusethevenousblood,checkthe
collectiondateandtheanticoagulant.)
Step7.Collectthe5µlbloodsampleandputitinthebuffersolutiontube.
Step8.Mixthebuffersolutionandbloodsamplebygentlysqueezingtherubbertopofthespot6‐8times.
NOTE
1.Blocktheholeonthestopofthespotwhilemixing.
2.Avoidbubbleswhilemixing.
12
Step9.Collectallthereactionmixturefromthebuffersolutiontube.Oncethe“ApplyBlood”animationon
thescreen,thebloodsamplecanbeappliedontheteststrip.TheA1ctestresultwillappearafter3
minutes.
文件名稱 Instruction for Use Doc.No.文件編碼 CDM‐C01‐C3‐IFU‐POCX1‐ALL‐01
表單編號:FT‐73‐01‐0008‐0007A9of26
9. Runningtest‐lipid
Step1.Beforetesting,washyourhandswithwarm,soapywater.Rinseanddrycompletely.
Step2.Tap“Settings”buttononHomescreenofPixoTestPOCTSystemandtap“Lipid”buttontoopenthe
PixoHealthPOCT‐LipidApp.
Step3.Tap“Renew”toscantheQRcodepastedontheteststripbox.
Step4.Tap“Next”buttontostartthelipidmeasurement.
Step5.Pleaseslidetheitemselectionkeytothe“I”indication.Checkthestripcodenumberandinsertthe
teststripintheslot.Press“initialize”buttonforinitialization.
Step6.Obtainadropofbloodsampleusingthelancetandlancingdevice.
Step7.Collecta35µlbloodsampleusingthetube.Placethebloodsampleintotheapplicationholeofthe
teststrip.Then,closetheslidingdoor.
Step8.Lipidtestresultwillappearafter3minutes.
NOTE
Thesystemshowsthenumberoftheremainingtest.PleaseopenanewstripboxandscanitsQRcodeto
resetthecountingwhentheremainingtestsreacheszero.
10. Understandingresult
MeasuringRange
Teststripscanmeasurenumericalresultsinthefollowingranges:
• HbA1c:4‐15%(68.1–383.8mg/dL,3.77–21.26mmol/L)
• TotalCholesterol:100–450mg/dL(2.59–11.64mmol/L)
• Triglycerides:45–650mg/dL(0.51–7.34mmol/L)
• HDLcholesterol:25–95mg/dL(0.65–2.46mmol/L)
• LDLcalculatedrange:66‐225mg/dL(1.71~5.82mmol/L)
文件名稱 Instruction for Use Doc.No.文件編碼 CDM‐C01‐C3‐IFU‐POCX1‐ALL‐01
表單編號:FT‐73‐01‐0008‐0007A10of26
Iftheresultisoutsidethemeasuringrange,thesystemwilldisplay“Hi”whentheresultsareabovethe
measuringrange,anddisplay“Lo”whentheresultsarebelowthemeasuringrange.
CAUTION
Ifthereisa“Lo/Hi”oranunexpectedresultforanytest,testagainwithanewteststrip.
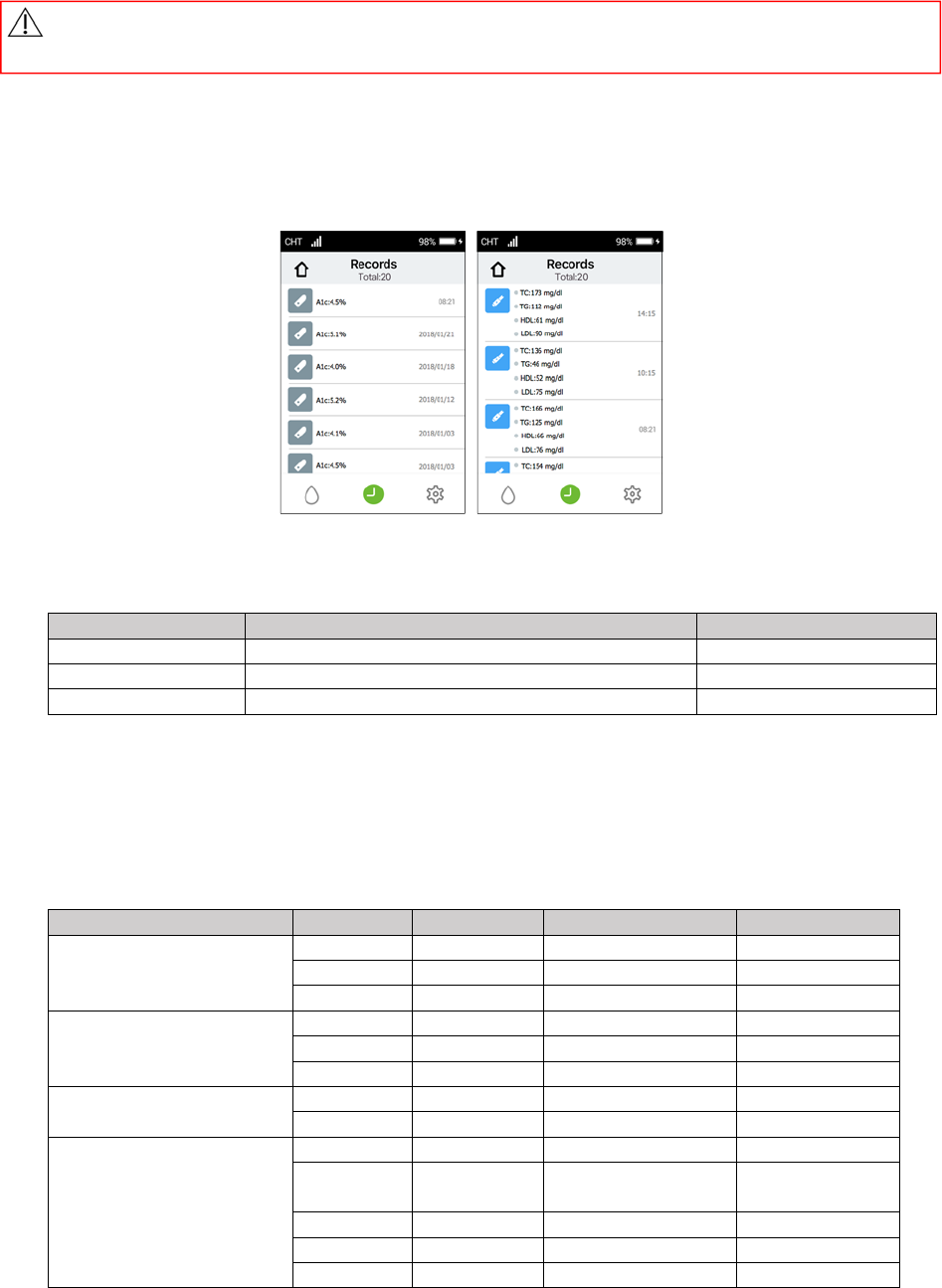
11. Record
ReviewingPastResults
OpenupthePixoHealthPOCT‐A1c/LipidApp.Threefunctionbuttonswillappearonthescreen.Tapthe
“Records”icontorevieweachpastresultbydateandtime.
A1cRecordsLipidRecords
ExpectedValues
A1c:
%HbA1cInterpretationofresults Displaycolor
<=5.6%NonDiabeticRange Green
5.6%‐6.5%ADA(AmericanDiabetesAssociation)Target Yellow
>=6.5%AboveTargetRed
Lipid:
ThenationalHeart,LungandBloodInstituteissuedtheThirdReportofNationalCholesterolEducation
Program(NCEP)ExpertPanelonDetection,Evaluation,andTreatmentofHighBloodCholesterolinAdults
(AdultTreatmentPanelIII)inMay2001.TheATPIIIreportpresentedtheNCEP’supdatedclinicalguidelines
forcholesteroltestingandmanagementanddescribedthefollowingclassificationsforcholesteroland
triglyceridetesting.
mg/dLmmol/L Classification Displaycolor
TC(TotalCholesterol)
<200<5.18 Desirable Green
200‐239 5.18‐6.19 Borderlinehigh Yellow
>=240>=6.22 High Red
TG(Triglycerides)
<150<1.69 Normal Green
150‐199 1.69‐2.25 Borderlinehigh Yellow
>=200>=2.26 High Red
HDL<40<1.03 Low Black
>=60>=1.55 High Black
LDL
<100<2.59 Optimal Black
100‐129 2.59‐3.34 Nearoptimal/
aboveoptimal
Black
130‐159 3.36‐4.11 Borderlinehigh Black
160‐189 4.14‐4.89 High Black
>=190>=4.91 Veryhigh Black
文件名稱 Instruction for Use Doc.No.文件編碼 CDM‐C01‐C3‐IFU‐POCX1‐ALL‐01
表單編號:FT‐73‐01‐0008‐0007A11of26
TheATPIIIidentifiedHDLlevelsbelow40mg/dL(1.03mmol/L)asassociatedwithincreasedriskofcoronary
heartdisease(CHD)inmenandwomen.AhighHDLlevelgreaterthanorequalto60mg/dL(1.55mmol/L)
isprotectiveanddecreasesCHDrisk.
LDL
LDLcanbecalculatedusingtheequationbelow.CalculatedLDLisanestimationofLDL,definedbythe
specificationofTC,TG,andHDL.
• LDL(calculated)=TotalCholesterol‐HDL‐(Triglyceride/5)(mg/dL)
• LDL(calculated)=TotalCholesterol‐HDL‐(Triglyceride/2.17)(mmol/L)
12. Calibration
ThePixotTestPOCTsystemwillautomaticallyalerttheUsertocompleteacalibration.Theusermustto
completethecalibrationwithPixoTestPOCTCalibrationCard.
PixoTestPOCTCalibrationCardisusedtocalibratethePixoTestPOCTsystemandtoconfirmthefunction
ofthefrontcameraandPixoHealthPOCT‐A1c/LipidApp.
ThePixoTest
POCTSystemmustbecalibrateddaily.Ifthishasnotbeencompleted,thePixoHealthPOCT
A1c/LipidAppwillnotbeabletoperformtestsuntilcalibrationhascompleted.
WhentodoaCalibration:
• WhenusinganewboxofA1c/LipidTestStrip.
• Whenthereisanunexpectedtestresult.
WheretoperformaCalibration:
• Thesystemshows“CALIBRATION”ontheHomescreenautomaticallyatthefirsttimeusingtheAnalyzer
everyday.Tapthe“Calibration”buttontostartthecalibrationprocess.
• Toperformacalibrationanytime,tap“Settings”buttononHomescreenofPixoTestPOCTsystemand
tap“Calibration”buttontostartthecalibrationprocess.
• Acalibrationtestcanbeinitializedfromthe“Settings”ofPixoHealthPOCT‐A1c/LipidAppanytime.
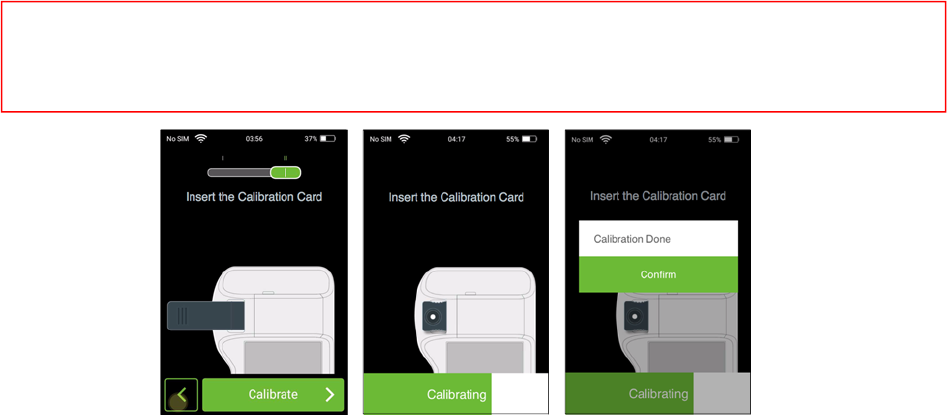
HowtoCalibrate:
Step1:Pleaseslidetheitemselectionkeytothe“II”indication.InserttheCalibrationcardintotheA1cslot
andpress“Calibrate”button.
Step2:TheAPPwillautomaticallystartthecalibration.Waitforafewsecondstocompletethecalibration.
Step3:Whenthecalibrationiscomplete,tap“Confirm”tocontinuethetestingmode.
NOTE
1.Avoidlongexposurestolight.
2.Avoidtouchingfinishedsurfaceoncolorblockofcalibrationcard.
3.Keepcalibrationcardinblackprotectivesheathwhencompletedcalibration.
13. Controlsolutiontest
iXensorCo.Ltd.doesnotprovidecontrolsolutions,Bio‐RadcontrolsolutionofA1candLipidare
文件名稱 Instruction for Use Doc.No.文件編碼 CDM‐C01‐C3‐IFU‐POCX1‐ALL‐01
表單編號:FT‐73‐01‐0008‐0007A12of26
recommendedforPixoTestPOCTSystem:
A1c:Bio‐RadLiquicheckDiabetesControl(level1,2),Cat#171,172
Lipid:Bio‐RadLiquicheckLipidsControl(level1,2),Cat#641,642
TheControlSolutionisusedtoverifythatthePixoTestPOCTSystemisworkingproperly(A1c/LipidControl
Solutionaresoldseparately).Topurchasethecontrolsolution,pleasecontactiXensorCo.Ltd.tollfreeat1‐
800‐218‐0929,Mon‐Fri9AM‐5PM(PST).
WhentodoaA1c/LipidControlSolutionTest
• Ifthestrip,PixoHealthPOCT‐A1c/LipidApporPOCT‐X01seemstonotbeworkingproperly.
• Ifthetestingresultsareunusualorunexpected.
• IftheAnalyzerhasbeendroppedordamaged.
CAUTION
• UseonlytheBioRadA1c/LipidcontrolsolutiontoverifythatthePixoTestPOCTSystemisworking
properly.Donotuseanyotherbrandofcontrolsolution.
• A1c/LipidControlSolutionisforinvitrodiagnosticuseonly.Thismeansthatisonlyusedfortesting
outsideofthebody.
• DoNotswalloworingestA1c/Lipidcontrolsolution.
• DoNotuseA1c/Lipidcontrolsolutionaftertheexpirationdate(printedoncontrolsolutionbottle,shelf
life:3yearat‐20℃to‐70℃)orthediscarddate(14daysfromthedatethecontrolsolutionvialwas
opened),whichevercomesfirst.Usingthesolutionaftertheexpirationordiscarddatewillcauseyour
resultstobeinaccurate.
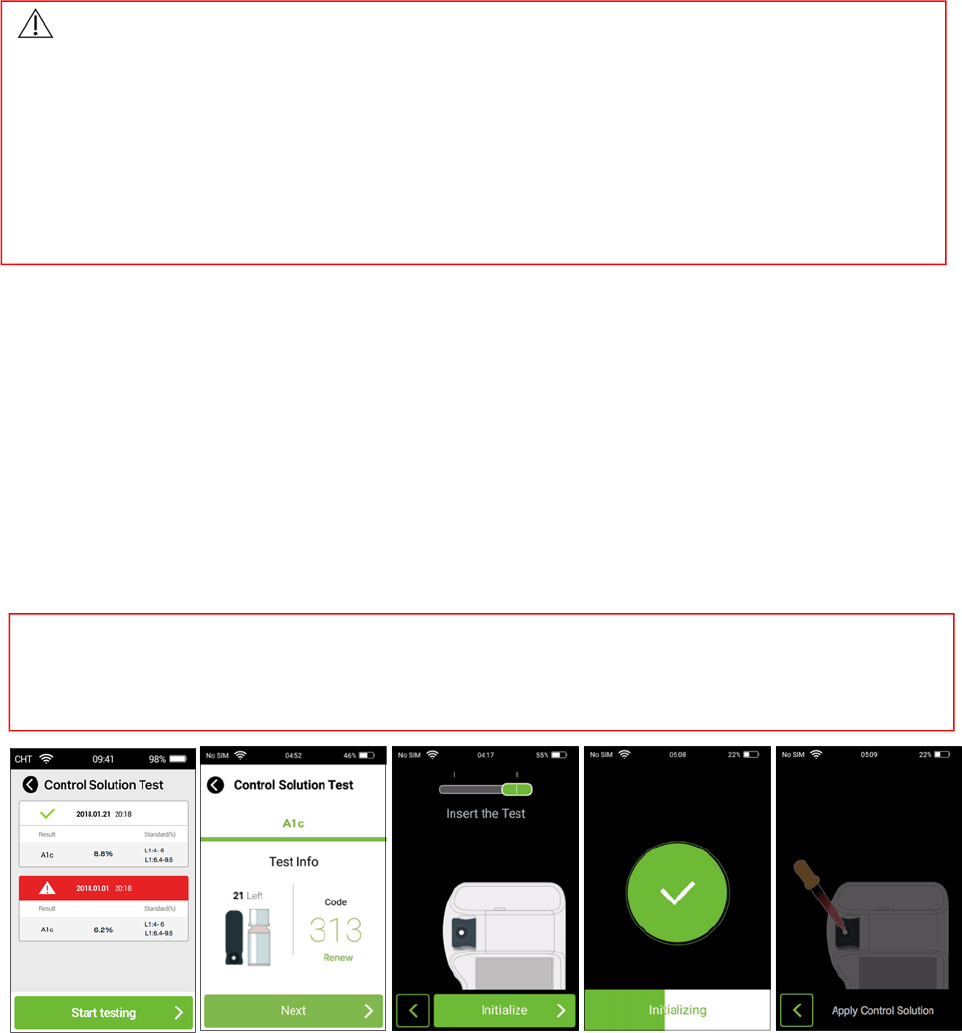
14. CStestforA1c
Step1:Beforetesting,washyourhandswithwarm,soapywater.Rinseanddrycompletely.
Step2:CleananddisinfecttheAnalyzersurfaceasinstructed.Startacontrolsolutiontestbytappingtheicon
“Settings”andselect“ControlSolutionTest ” optioninthePixoHealthPOCT‐A1cApp.
Step3:ThemainscreenofControlSolutionTest listsalltestresults,tapthe“Starttesting”buttontoenter
theTest Infoscreen.
Step4:Tap“Renew”toscanthecodeontheteststripboxifthecodehasn’tbeenscanned.Andpress“Next”
buttontostartthetest.
Step5:Pleaseslidetheitemselectionkeytothe“II”indication.Checkthestripcodenumberandinsertthe
teststripintheslot.Press“initialize”buttonforinitialization.
Step6:Oncethecontrolsolution‐applyinganimationisdisplayedonthescreen,obtainacontrolsolution
sampleofLevel1orLevel2andapplythecontrolsolutionontheteststrip.Thetestresultwillappear
after3minutes.
NOTE
1. PleasemakesuretheAnalyzerisplacedonaflatsurface.
2. Thesystemshowsthenumberofremainingtests.PleaseopenanewstripboxandscanitsQRcodeto
resetthecountingwhentheremainingtestsreacheszero.
文件名稱 Instruction for Use Doc.No.文件編碼 CDM‐C01‐C3‐IFU‐POCX1‐ALL‐01
表單編號:FT‐73‐01‐0008‐0007A13of26
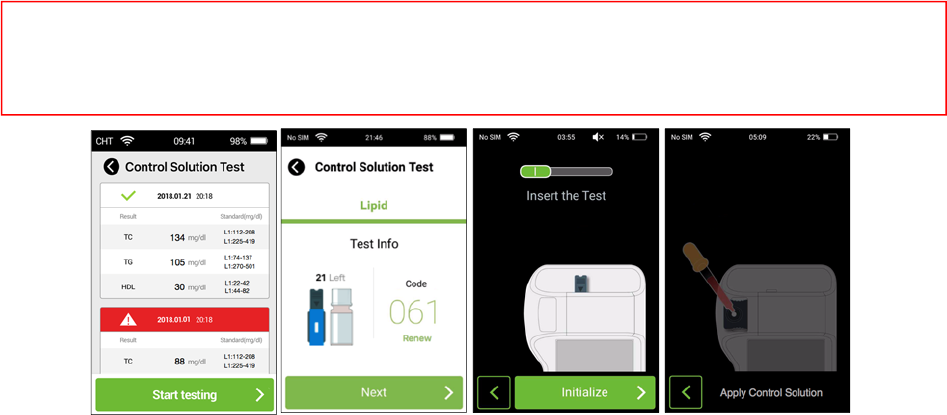
15. CStestforLipid
Step1:Beforetesting,washyourhandswithwarm,soapywater.Rinseanddrycompletely.
Step2:CleananddisinfecttheAnalyzersurfaceasinstructed.Startacontrolsolutiontestbytappingthe
icon“Settings”andselect“ControlSolutionTest ”inthePixoHealthPOCT‐LipidApp.
Step3:ThemainscreenofControlSolutionTest listsalltestresults,tapthe“Starttesting”buttontoenter
theTest Infoscreen.
Step4:Tap“Renew”toscanthecodeontheteststripboxifthecodehasn’tbeenscanned.Andpress
“Next”buttontostartthetest.
Step5:Pleaseslidetheitemselectionkeytothe“I”indication.Checkthestripcodenumberandinsertthe
teststripintheslot.Press“initialize”buttonforinitialization.
Step6:Obtainacontrolsolutionsampleusingthe35µltube.
Step7:Collectthe35µlcontrolsolutionusingthetubeandplacethecontrolsolutionsampletothe
applicationholeoftheteststrip,thenclosetheslidingdoor.
Step8:Lipidcontrolsolutiontestresultwillappearafter3minutes.
NOTE
1. Thesystemshowsthenumberoftheremainingtests.PleaseopenanewstripboxandscanitsQRcode
toresetthecountingwhentheremainingtestsreacheszero.
2. Removeanddiscardtheusedteststrip.Donotaddmorebloodtoausedteststrip.
16. ControlSolutionrangeandmeaning
OutofRangeResults
Aftercontrolsolutiontest,checktheresultofthecontrolsolutionvalueandconfirmitiswithinrange.If
testresultsfalloutsideoftheexpectedrange,pleaserepeatthetest.DonotcontinuewithyourA1c/Lipid
testifthecontrolsolutiontestresultsarecontinuouslyoutofrange.Resultsthatfalloutsideofthe
expectedrangemayindicate:
• Testisnotperformedatroomtemperature.
• Controlsolutionhasexpired,isbeyonditsdiscarddate,orcontaminated.
• AtechnicalproblemwithyourPixoTestPOCTSystem.
• Controlsolutionthatistoowarmortoocold.
• AproblemwiththeTestStrip(expiredordamaged).
• Anerrorcallsduringthetest.
17. Shutdownprocedure
WhenfinishusingthePixoTestPOCTSystem,followthestepstoturntheAnalyzeroff:
Step1.PressthePowerKeyfor2seconds.
Step2.Tap“Poweroff”toturnofftheAnalyzer.
文件名稱 Instruction for Use Doc.No.文件編碼 CDM‐C01‐C3‐IFU‐POCX1‐ALL‐01
表單編號:FT‐73‐01‐0008‐0007A14of26
18. Maintenanceandtroubleshooting
CaringforthePixoTestPOCTSystemiseasy.Simplykeepitfreeofdust.Ifthesystemneedscleaning,follow
theseguidelinescarefullytoobtainthebestperformance.TopreventmalfunctionoftheAnalyzer,keepthe
teststripslotfreeofblood,dirt,ordust.Usealint‐freeclothdampenedwithwatertocleantheAnalyzer.
Ensuretheclothisdamp,andnotwet.Donotuseanabrasiveclothorantisepticsolution,asthesemay
damagethedisplayscreen.
Acleanopticalmeasuringsystemisabasicprerequisitefordeterminingprecisemeasuredvalues.Therefore,
cleantheAnalyzerregularlyandimmediatelyafteritbecomesdirty.AlwaysswitchofftheAnalyzerbefore
cleaningit.
Useonlythefollowingitemsforcleaning:
• Ordinarylint‐freecottonbuds
• Ordinarylint‐freetissues
• Ordinarydisinfectingtissues
• Donotuseanydisinfectantspraysortissuesorcottonbudswhicharedrippingwetastheliquidmay
entertheAnalyzeranddamageit.
CleaningtheouterAnalyzercomponents
1. WipetheoutsideoftheAnalyzerwithalightlymoistened,lint‐freecottoncloth.
2. Removetheprotectioncover.
3. Incaseofsignificantdirt,youcanrinsetheprotectioncover(separatelyfromtheAnalyzer)underwarm
runningwater.
4. Drythemeasurementchambercoverwithafreshcloth.
Cleaningtheopticalmeasuringsystem
1. Removethestripmodule.
2. Cleantheeasilyaccessibleareasoftheopticalmeasuringsystemwithalint‐freepadoramoistened
cottonswab.MakesurethatnoliquidenterstheAnalyzer.
3. AllowtheAnalyzertodrythoroughly.
4. DonotfitthestripmoduleintotheAnalyzeruntilitiscompletelydry.
5. FitthestripmoduleintoAnalyzer.
6. TheAnalyzerisnowreadyforoperationagain
Maintenance,TestingandTransportationAnalyzer
1. Keeptheteststripslotsfreeofdust.
2. ProtecttheinternalAnalyzerfromhumidity.
3. Therearenoeffectsonproductqualityinenvironmentaltemperaturesof‐20–50°C(‐4–122°F)
during8hoursandhumidityofupto93%RH,non‐condensingduring8hours.
19. Cleananddisinfection
Duetotheriskofspreadingbloodborninfections,cleananddisinfectthePOC‐X01aftereverybloodtest.
CleanthePOC‐X01toremovevisibledirt,bloodorothermaterialspriortodisinfecting.Followwitha
secondwipetodisinfectthemeasurementmodule.
文件名稱 Instruction for Use Doc.No.文件編碼 CDM‐C01‐C3‐IFU‐POCX1‐ALL‐01
表單編號:FT‐73‐01‐0008‐0007A15of26
Cleananddisinfectthemeasurementmoduleatleastonceperweekandwhenbloodispresentonthe
surfaceofthePOC‐X01.
FordisinfectingyourPOC‐X01,onlyuseCloroxHealthcareBleachGermicidalWipes(EPA*reg.no.
67619–12),containingSodiumHypochlorite0.55%.[CanbepurchasedfromAmazon.com,
Officedepot.comandWalmart.com].
Donotuseanyothercleaningordisinfectingsolutions.
UseofdisinfectingproductsotherthanCloroxHealthcareBleachGermicidalWipeshavenotbeen
validatedforthePixoTestPOCTSystemhavenotbeenvalidated.AlwaysuseCloroxHealthcareBleach
GermicidalWipes.
Testprocess–Lipid
Usetwowipes.
Followthecleaninginstructionforthefirstwipeandthedisinfectioninstructionforthesecond.
Cleaning:
1. Followthecleaninginstructionfirst.
2. Putoncleangloves
3. Takeonewipeandsqueezeoutexcessmoisturefromthewipebeforeuse.
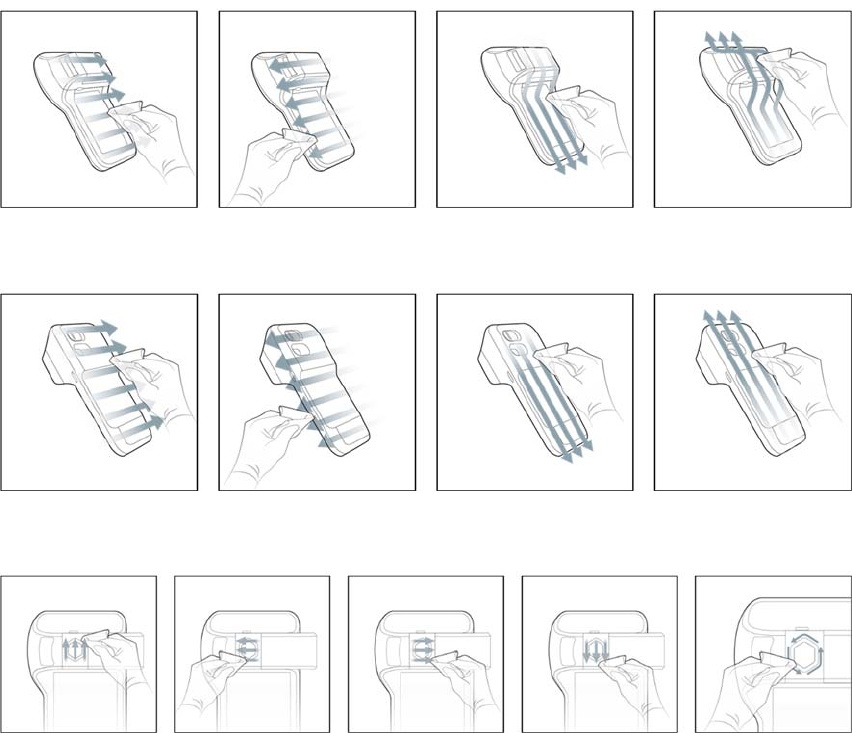
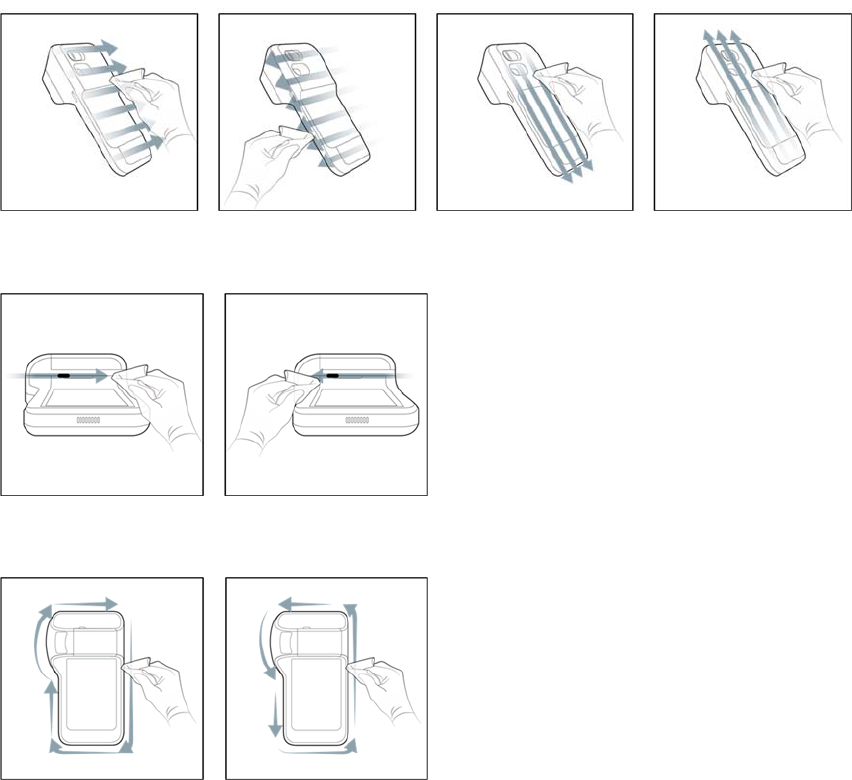
4. FollowtheFigure3towipethemeterfrontandmeterscreenrespectively.
Figure3.Wipethemeterfrontandmeterscreenrespectively.
5. FollowtheFigure4towipethemeterback.
Figure4.Wipethemeterback
6. FollowtheFigure5towipetheinsertionareaofstrip.
Figure5.Wipetheinsertionareaofstrip

文件名稱 Instruction for Use Doc.No.文件編碼 CDM‐C01‐C3‐IFU‐POCX1‐ALL‐01
表單編號:FT‐73‐01‐0008‐0007A16of26
7. FollowtheFigure6towipetheitemselectionkey.
Figure6.Wipetheitemselectionkey.
8. FollowtheFigure7towipethemeterframe.
Figure7.Wipethemeterframe
9. Makesureallthedirt,dust,bloodandotherbodyfluidsarecleanedfrommeterusingthefirstwipe.
Disinfection:
1. Aftercleaning,useasecondwipeandthoroughlywetthesurfacesofmeter.
2. Makesurethetreatedarearemainswetforaminutetoeffectivelykillblood‐bornepathogens.
3. Ensurethetreatedareaarethoroughlydry.
4. Discardthewipesandachieveacleaning/disinfectioncycle.
Testprocess–A1c
Cleaning:
1. Usetwowipes.
2. Followthecleaninginstructionforthefirstwipeandthedisinfectioninstructionforthesecond.
3. Putoncleangloves
4. Takeonewipeandsqueezeoutexcessmoisturefromthewipebeforeuse.
5. FollowtheFigure8towipethemeterfrontandmeterscreenrespectively.
Figure8.Wipethemeterfrontandmeterscreenrespectively.
文件名稱 Instruction for Use Doc.No.文件編碼 CDM‐C01‐C3‐IFU‐POCX1‐ALL‐01
表單編號:FT‐73‐01‐0008‐0007A17of26
6. FollowtheFigure9towipethemeterback.
Figure9.Wipethemeterback
7. FollowtheFigure10towipetheitemselectionkey.
Figure10.Wipetheitemselectionkey.
8. FollowtheFigure11towipethemeterframe.
Figure11.Wipethemeterframe
9. Makesureallthedirt,dust,bloodandotherbodyfluidsarecleanedfrommeterusingthefirstwipe.
Disinfection:
1. Aftercleaning,useasecondwipeandthoroughlywetthesurfacesofmeter.
2. Makesurethetreatedarearemainswetforaminutetoeffectivelykillblood‐bornepathogens.
3. Ensurethetreatedareaarethoroughlydry.
4. Discardthewipesandachieveacleaning/disinfectioncycle.

文件名稱 Instruction for Use Doc.No.文件編碼 CDM‐C01‐C3‐IFU‐POCX1‐ALL‐01
表單編號:FT‐73‐01‐0008‐0007A18of26
20. ErrorandWarningmessage
Errormessagesandtrouble‐shootingtipswillbedisplayedonthescreen.Pleasepayattentiontotheerror
messagesandinformationtomakesurethePixoTestPOCTSystemisperformingaccurately.
Thefollowingtablelistserrormessageandtroubleshootingtips,whichmayappearonthescreenduring
performingthesystem.
Errormessage–A1c
ErrorNumberErrorMessageProblemCauseWhattoDo
Note01
Measurement
Failure
Themeasurementisinterrupted
duetoimproperoperation.
Pleasekeeptheteststrip
stabilizedanddon’tmovethe
teststripduringmeasurement.
Ifthiserrorstillhappenson
bloodsampleswithnormal
rangeoftotalhemoglobin,
pleasecontactyoursystem
provider.
Note02SystemTilt
Itisdetectedthatthetiltsduring
measurement.
PleasekeepthePOC‐X01flat
andstabilizedonatableduring
measurement.
Note03
Out‐of‐range
Temperat u re
Theambienttemperatureisoutside
therecommendedoperating
temperaturerange.
Performatestinan
environmentwiththe
temperaturebetween15‐32°C
(59‐89.6°F).Donotheator
coolyourPOC‐X01.
Note04
BloodSample
Error
Nosampleorinsufficientsample
volumeappliedontheteststripwas
detectedafterthesystemindicates
toapplythebloodsample.
Discardthisteststripandre‐
testwithanewtestkit.
Note05TestStripError
Invalidteststriporimproper
installationoftheteststripwas
detectedduringmeasurement.
Reinstallanewtestkitproperly
andtestagain.
Note06CalibrationError
Incompleteorincorrectcalibration
processwasdetected.
InsertonlytheCalibrationCard
providedbythemanufacturer
andswitchtheItemselection
keytotheA1cmodeforthe
calibration.DoNotmovethe
CalibrationCardduring
calibration.
Note07SystemError
Abnormalsignalscomingfrom
hardwareproblemsweredetected
Dothecalibrationagainandif
theerrorstillpersists,please

文件名稱 Instruction for Use Doc.No.文件編碼 CDM‐C01‐C3‐IFU‐POCX1‐ALL‐01
表單編號:FT‐73‐01‐0008‐0007A19of26
duringcalibrationprocess.contactyoursystemprovider.
Errormessage–Lipid
ErrorNumberErrorMessageProblemCauseWhattoDo
Note01
Measurement
Failure
Themeasurementisinterrupted
duetoimproperoperation.
Pleasekeeptheteststrip
stabilizedanddon’tmovethe
teststripduringmeasurement.
Note02
LightSignal
Error
Themeasurementisinterrupted
duetothestrongstraylightinthe
stripmodule.
Pleasemakesurethatthestrip
moduleissecurelyattached
andtheslidingdoorisclosed
completely.
Note03
Out‐of‐range
Temperat u re
Theambienttemperatureisoutside
therecommendedoperating
temperaturerange.
Performatestinan
environmentwiththe
temperaturebetween18‐32°C
(64.4‐89.6°F).Donotheator
coolyourPOC‐X01.
Note04
BloodSample
Error
Nosampleorinsufficientsample
volumeappliedontheteststripwas
detectedafterthesystemindicates
toapplythebloodsample.
Discardthisteststripandre‐
testwithanewtestkit.
Note05TestStripError
Invalidteststriporimproper
installationoftheteststripwas
detectedduringmeasurement.
Reinstallanewtestkitproperly
andtestagain.
Note06CalibrationError
Incompleteorincorrectcalibration
processwasdetected.
InsertonlytheCalibrationCard
providedbythemanufacturer
andswitchtheItemselection
keytotheA1cmodeforthe
calibration.DoNotmovethe
CalibrationCardduring
calibration.
Note07SystemError
Abnormalsignalscomingfrom
hardwareproblemsweredetected
duringcalibrationprocess.
Dothecalibrationagainandif
theerrorstillpersists,please
contactyoursystemprovider.
21.Disposalinformation
Discardtheusedteststripscarefully.Usedteststripsmaybeconsideredasbiohazardouswasteinyourarea.
Handleanddisposeofallmaterialscomingincontactwithbloodaccordingtouniversalprecautionsand
guidelines.Washyourhandswithsoapandwaterafterhandlingtheteststrip.

文件名稱 Instruction for Use Doc.No.文件編碼 CDM‐C01‐C3‐IFU‐POCX1‐ALL‐01
表單編號:FT‐73‐01‐0008‐0007A20of26
22.Productinformation
Specification
POC‐X01Analyzer
Dimension181mm(L)x111mm(W)x53mm(H)
Weight131.3g
Power5000mAhbattery,non‐removable
storage10000sets
Operating
Temperat u re10℃~40℃(50℉~104℉)
Humidity10%~90%RH
Operationatmosphericpressurerange
760mmHg~526mmHg
Storage&Transportationcondition Temperature1℃~40℃(34℉~104℉)
Humidity10%~90%RH
CalibrationCardStoredinthepackagepouchandwith1yearsshelflife
others• Indooruse
• OvervoltagecategoryII
• Pollutiondegrees2
Charger
USversionManufacture:DONGGUANAOHAIPOWERTECHNOLOGY
CO.,LTD.
Model:A18A‐050100U‐US2
Input:100‐240V~50/60HzMax0.2A
Output:5V1.0A
ENversionManufacture:DONGGUANAOHAIPOWERTECHNOLOGY
CO.,LTD.
Model:A824A‐120150U‐EU1
Input:100‐240V~50/60Hz0.5A
Output:5V2.0A/9V2A/12V1.5A
UKversionManufacture:DONGGUANAOHAIPOWERTECHNOLOGY
CO.,LTD.
Model:A806‐050100U‐UK1
Input:100‐240V~50/60Hz0.2A
Output:5V1.0A
文件名稱 Instruction for Use Doc.No.文件編碼 CDM‐C01‐C3‐IFU‐POCX1‐ALL‐01
表單編號:FT‐73‐01‐0008‐0007A21of26
Test stripforLipid
MeasuringRangeTC:100
–
450mg/dL,(2.59
–
11.64mmol/L)
TG:45–650mg/dL,(0.51–7.34mmol/L)
HDL:25–95mg/dL,(0.65–2.46mmol/L)
SampleFreshcapillaryorvenouswholeblood
SampleSize35µL
Test Time3minutes
HematocritTC/TG:30
–
55%;HDL:30
–
52%
OperatingTemp eratureRange18
–
32°C(64
–
90°F)
Test StripStorageTemperat ure 2
–
32°C(36
–
90°F)
Shelf‐life18months
*:Test stripmaybestoredinarefrigeratorat2–8°C(36‐46°F),butmustbebroughttoroomtemperature
beforeuse.
Test stripforA1c
MeasuringRange4.0
–
15.0%
SampleFreshcapillaryorvenouswholeblood
SampleSize5µL
Test Time3minutes
Hematocrit25
–
65%
OperatingTemp eratureRange15
–
32°C(59‐90°F)
Test StripStorageTemperat ure *1
–
30°C(34to86°F)
Shelf‐life18months
*:Test stripmaybestoredinarefrigeratorat2–8°C(36‐46°F),butmustbebroughttoroomtemperature
beforeuse.
23.Limitations
PixoTestA1cTestStrip
1.Thebatterylifeneedstobeover20%beforeusingthePixoTestPOCTSystem.
2.DonotchargeyoursmartphonewhenyouusethePixoTextPOCTSystem.
3.PixoTestPOCTSystemcanperformsinthehemoglobinarangeof7‐23g/dLandahematocritrangeof25to
65%.
4.PerformanceofPixoTestPOCTSystemhasnotbeentestedonsamplesfromnewborns.
5.Cosmeticssuchashandcreamsorlotionsoftencontainglycerol.Useoftheseproductsmaycauseinaccurate
results.
6.Forinvitrodiagnosticuseonly.
7.PixoTestA1cTestStripisforsingleuse.
8.Donotcarryoutthetestindirectsunlight.Themeasurementshouldbeperformedinanappropriatelight
environment.
9.Strongelectromagneticfields(e.g.microwaveovens)mayaffectperformance.
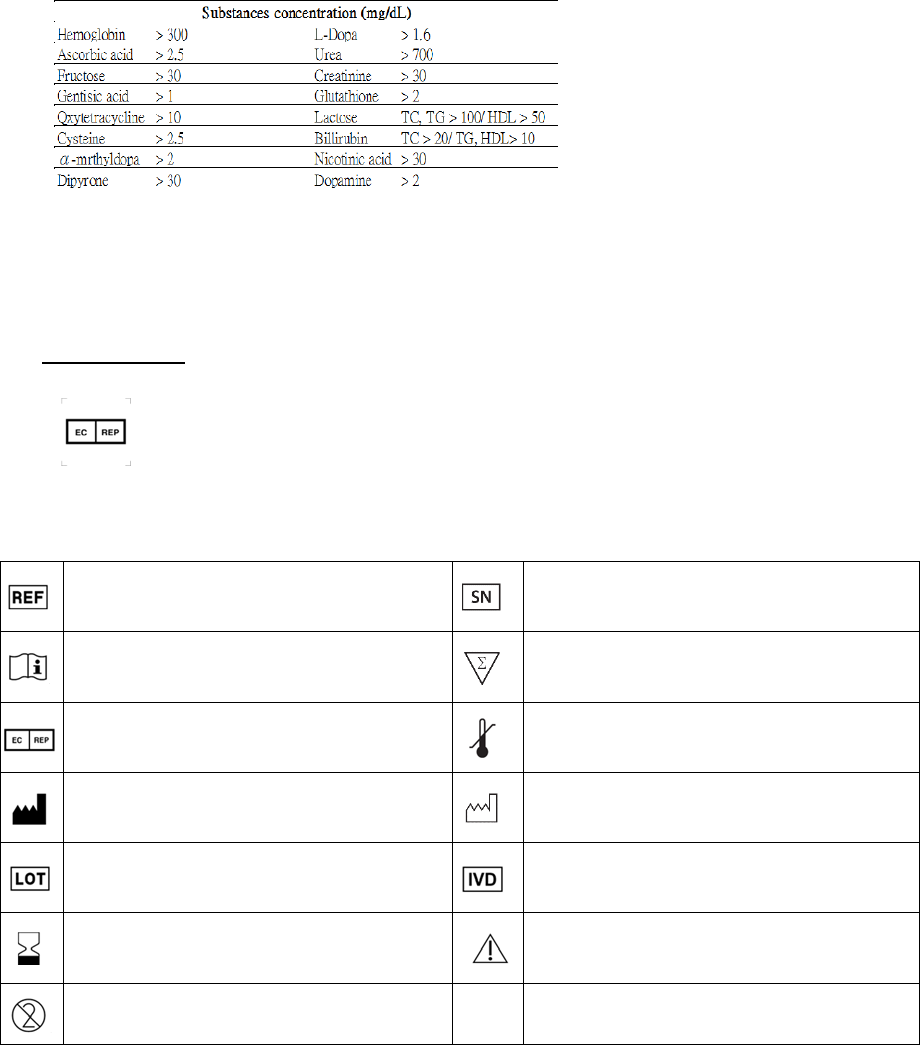
10.Interferences:Thefollowingsubstancesmaycausefalseresultsatlevelsofthebelow.
Acetylsalicylic acid > 30 mg/dL
Ascorbic acid > 10 mg/dL
Acetaminophenol > 30 mg/dL
Billirubin > 20 mg/dL
Caffeine > 30 mg/dL
Hydroxyzine Dihydrocholoride > 30 mg/dL
Triglyceride > 900 mg/dL
Glyburide > 20 mg/dL
Ibuprofen > 20 mg/dL
Dopamine > 2 mg/dL
Substances concentration (mg/dL)
文件名稱 Instruction for Use Doc.No.文件編碼 CDM‐C01‐C3‐IFU‐POCX1‐ALL‐01
表單編號:FT‐73‐01‐0008‐0007A22of26
PixoTestLipidTestStrip
1.Thebatterylifeneedstobeover20%beforeusingthePixoTestPOCTSystem.
2.DonotchargePOC‐X01whenyouusethePixoTextPOCTSystem.
3.Hematocritsof30to55%(TC/TG),30to52%(HDL)forLipidTestStripdonotaffectresults.
4.PerformanceofPixoTestPOCTSystemhasnotbeentestedonsamplesfromnewborns.
5.Cosmeticssuchashandcreamsorlotionsoftencontainglycerol.Useoftheseproductsmaycauseinaccurate
results.
6.Forinvitrodiagnosticuseonly.
7.Test stripisforsingleuse.
8.Donotcarryoutthetestindirectsunlight.Themeasurementshouldbeperformedinanappropriatelight
environment.
9.Strongelectromagneticfields(e.g.microwaveovens)mayaffectperformance.
10.Interferences:Thefollowingsubstancesmaycausefalseresultsatlevelsofthebelow.
24.Manufacturer
iXensorCo.,Ltd.
6F.,No.9,Aly.2,Ln.35,JihuRd.,NeihuDist.,TaipeiCity,11492,Taiwan
www.ixensor.com
Authorizedrepresentative
MDSSGmbH
Schiffgraben4130175Hannover,Germany
25.Symbol
CataloguenumberSerialnumberoftheanalyzer
ConsultinstructionsforuseContainsSufficientfor
<n>Test s
Authorizedrepresentative
intheEuropeanCommunity
Temperat u relimitation
ManufacturerDateofManufacture
BatchCodeIn‐vitrodiagnosticUse
UsebydateCaution,
Consultaccompanyingdocuments
Donotreuse

文件名稱 Instruction for Use Doc.No.文件編碼 CDM‐C01‐C3‐IFU‐POCX1‐ALL‐01
表單編號:FT‐73‐01‐0008‐0007A23of26
26.Servicing
ThePixoTestPOCTSystemsisintendedtoprovidesafeandreliableoperationaccordingto
theinstructionsprovidedbyiXensorCo.,Ltd.iXensorCo.,LtdrecommendsthatthePixoTest
POCTsystemcanbeinspectedandservicedbyauthorizedtechniciansifthereisanyconcern
withdevicefunction.Otherwise,serviceandinspectionofthedevicesgenerallyshouldnot
berequiredifusedbyfollowingtheinstructionforuse.
27.LimitedWarranty
iXensorCo.,Ltd(hereafter‘iXensor’)givesthecustomeralimitedmanufacturerwarranty
onneworiginaliXensorproductsandrelativeaccessoriesinaccordancewiththewarranty
conditionsapplicabletotheiteminquestionandinaccordancewiththewarrantyperiods
frompurchasedate,pleaserefertothelistasbelow.Thiswarrantydoesnotcoverdamage
causedbyoperatorhimself,accident,misuse,abuse,alterationandotherdefectsnot
relatedtomaterialorworkmanship.Toexerciseyourrightsunderthiswarranty,please
contactyourlocal,authorizeddistributorsorservicecenter.
ItemWarrantyperiod
PixoTestPOCTAnalyzer1year
USBCharger1year
PixoTestPOCTCalibrationCard1year
USBTypeCChargeCableNone
OtherdisposablepartsNone
CustomerName:
_______________________________________________________________________
CustomerAddress:
____________________________________________________________________
Telephone:____________________________
E‐mailaddress:_________________________________
Dateofpurchase:_____________________________________________________
Distributorinformation:
文件名稱 I
n
struction for Use Doc.No.文件編碼 CDM‐C01‐C3‐IFU‐POCX1‐ALL‐01
表單編號:FT‐73‐01‐0008‐0007A 24of26
Appendix1:Reference
1. AmericanDiabetesAssociation,ClinicalPracticeRecommendationGuidelines2003,Diabetescare,Vol.26.
Supplement1.p.22
2. Stedman,TL.Stedman’sMedicalDictionary,27thEdition,1999,p.2082.
3. EllenT.Chen,JamesH.Nichols,Show‐HongDuh,GlenHortin,MD:DiabetesTechnology&Therapeutics,
PerformanceEvaluationofBloodGlucoseMonitoringDevices,Oct2003,Vol.5,No.5:749‐768
4. SiedelJ,HageleEO,ZiegenhormJ,WahlefeldAW.Reagentfortheenzymaticdeterminationofserumtotal
cholesterolwithimprovedlipolyticefficiency.ClinChem1983;29:1075‐80.
5. ExpertPanelonDetection,Evaluation,andTreatmentofHighCholesterolinAdults.Executivesummaryof
theThirdReportoftheNationalCholesterolEducationProgram(NCEP)ExpertPanelonDetection,Evaluation,
andTreatmentofHighCholesterolinAdults(AdultTreatmentPanelIII).JAMA2001;285:2486‐97.
6. IDFClinicalGuidelinesTaskForce.GlobalguidelineforType2diabetes.Brussels;InternationalDiabetes
Federation,2005.
7. Definitionanddiagnosisofdiabetesmellitusandintermediatehyperglycemia:reportofaWHO/IDF
consultation.WHO,Geneva2006(ISBN9241594934,ISBN9789841597936).
8. D’Orazioetal.:“ApprovedIFCCRecommendationofReportingResultsforBloodGlucose(Abbreviated);”
ClinicalChemistry51:91573‐1576‐(2005).
9. AmericanDiabetesAssociation,PositionStatement,DiagnosisandClassificationofDiabetesMellitus,
DiabetesCare35:S4‐S10,2012.
10. AmericanDiabetesAssociation:ClinicalPracticeRecommendations(2013)DiabetesCare,Vol36,
Supplement1,p.S1‐S100
FCC Caution: To assure continued compliance, any changes or modifications not expressly approved by the party
responsible for compliance could void the user's authority to operate this equipment. (Example - use only shielded
interface cables when connecting to computer or peripheral devices).
FCC Radiation Exposure Statement
This equipment complies with FCC RF radiation exposure limits set forth for an uncontrolled environment.
This transmitter must not be co-located or operating in conjunction with any other antenna or transmitter.
This device complies with Part 15, 22, 24 of the FCC Rules. Operation is subject to the following two conditions:
(1) This device may not cause harmful interference, and
(2) This device must accept any interference received, including interference that may cause undesired operation.
SAR: 0.73 W/Kg