GTBR13_cover M_spine20_5 91355 9789241564656 Eng
User Manual: 91355
Open the PDF directly: View PDF ![]() .
.
Page Count: 306 [warning: Documents this large are best viewed by clicking the View PDF Link!]

Warning: This report is out-of-date. In particular, entire
time-series of TB disease burden estimates are updated
every year. For the latest data and analysis, please see
the most recent edition of the global TB report.

Global
tuberculosis
report
2013

© World Health Organization 2013
All rights reserved. Publications of the World Health Organization are available on the WHO web site (www.who.int) or can be purchased from
WHO Press, World Health Organization, 20 Avenue Appia, 1211 Geneva 27, Switzerland (tel.: +41 22 791 3264; fax: +41 22 791 4857; e-mail:
bookorders@who.int). Requests for permission to reproduce or translate WHO publications – whether for sale or for non-commercial distribution
– should be addressed to WHO Press through the WHO web site (www.who.int/about/licensing/copyright_form/en/index.html).
e designations employed and the presentation of the material in this publication do not imply the expression of any opinion whatsoever on
the part of the World Health Organization concerning the legal status of any country, territory, city or area or of its authorities, or concerning
the delimitation of its frontiers or boundaries. Dotted lines on maps represent approximate border lines for which there may not yet be full
agreement.
e mention of specic companies or of certain manufacturers’ products does not imply that they are endorsed or recommended by the World
Health Organization in preference to others of a similar nature that are not mentioned. Errors and omissions excepted, the names of proprietary
products are distinguished by initial capital letters.
All reasonable precautions have been taken by the World Health Organization to verify the information contained in this publication. However,
the published material is being distributed without warranty of any kind, either expressed or implied. e responsibility for the interpretation
and use of the material lies with the reader. In no event shall the World Health Organization be liable for damages arising from its use.
Cover design by Tom Hiatt, Western Pacic Regional Oce and Irwin Law, WHO headquarters. e front cover illustrates the latest status of
global progress for ve indicators that are part of the Millennium Development Goals framework. ese are the incidence rate of tuberculosis
disease per 100 000 population per year, the prevalence of tuberculosis disease per 100 000 population, the tuberculosis mortality rate per 100
000 population per year, the case detection rate (the number of cases detected and reported to national tuberculosis programmes divided by
the estimated incidence) and the treatment success rate for new TB patients started on treatment. Each pair of shapes represents both the most
recent level of the indicator and a baseline year against which progress is measured. For incidence (green and dark orange), prevalence (grey and
pink) and mortality (light orange and light blue), the top of the combined height of each pair of shapes shows the level in 1990. e lower of the
two shapes in each pair shows the level in 2012. For the case detection rate, the combined height of each pair of shapes (dark blue and brown)
shows the level in 2012 and the lower of the two shapes (dark blue) illustrates the level in 1995. For the treatment success rate (red and yellow),
the combined height of each pair shows the level in 2011 and the lower of the two shapes (red) shows the level in 1995. More information about
these indicators and progress towards global targets are provided in Chapter 2 and Chapter 3 of the Global Tuberculosis Report 2013.
Designed by minimum graphics
Printed in France
WHO/HTM/TB/2013.11
WHO Library Cataloguing-in-Publication Data
Global tuberculosis report 2013.
1.Tuberculosis – epidemiology. 2.Tuberculosis, Pulmonary – prevention and control. 3.Tuberculosis – economics.
4.Tuberculosis, Multidrug-Resistant. 5.Annual reports. I.World Health Organization.
ISBN 978 92 4 156465 6 (NLM classication: WF 300)

iiiGLOBAL TUBERCULOSIS REPORT 2013
Contents
Abbreviations iv
Acknowledgements v
Executive summary ix
Chapter 1. Introduction 1
Chapter 2. The burden of disease caused by TB 6
%JCRVGT 6$ECUGPQVKßECVKQPUCPFVTGCVOGPVQWVEQOGU
Chapter 4. Drug-resistant TB 45
Chapter 5. Diagnostics and laboratory strengthening 59
%JCRVGT #FFTGUUKPIVJGEQGRKFGOKEUQH6$CPF*+8
Chapter 7. Financing 75
%JCRVGT 4GUGCTEJCPFFGXGNQROGPV
Annexes
1. Methods used to estimate the global burden of disease caused by TB 99
%QWPVT[RTQßNGU
4GIKQPCNRTQßNGU
4. Key indicators for the world, WHO regions and individual countries 145

iv GLOBAL TUBERCULOSIS REPORT 2013
Abbreviations
ACSM Advocacy, Communication and Social
Mobilization
ACTG AIDS Clinical Trials Group
ADR adverse drug reactions
AFB acid-fast bacilli
AIDS acquired immunodeciency syndrome
ARI annual risk of infection
ART antiretroviral therapy
BCG Bacille-Calmette-Guérin
BRICS Brazil, Russian Federation, India, China,
South Africa
CDR case detection rate
CEM cohort event monitoring
CFR case fatality rate
CFU colony-forming units
CPT co-trimoxazole preventive therapy
CBC community-based care
DOTS the basic package that underpins the Stop
TB Strategy
DR-TB drug-resistant tuberculosis
DRS drug resistance surveillance
DST drug susceptibility testing
DS-TB drug-susceptible tuberculosis
DTLC District TB and Leprosy Coordinator
EBA early bactericidal activity
ECDC European Centre for Disease Prevention and
Control
ERR electronic recording and reporting
EU European Union
FDA Food and Drug Administration
FIND Foundation for Innovative New Diagnostics
GDP gross domestic product
GLC Green Light Committee
GLI Global Laboratory Initiative
GNI gross national income
HBC high-burden country
HIV human immunodeciency virus
HR Hazard ratio
ICD-10 International Classication of Diseases
(10th revision)
IDRI Infectious Disease Research Institute
IGRA interferon-gamma release assay
IPAQT Initiative for Promoting Aordable, Quality
TB Tests
IPT isoniazid preventive therapy
IRR incidence rate ratio
LED light-emitting diode
LPA line-probe assay
LTBI latent TB infection
MDG Millennium Development Goal
MDR-TB multidrug-resistant tuberculosis
MNCH maternal, newborn and child health
NAAT nucleic acid amplication test
NAP national AIDS programme
NFM new funding model
NTP national tuberculosis [control] programme
OECD Organisation for Economic Co-operation and
Development
OR Operational research
PAL Practical Approach to Lung health
PCR polymerase chain reaction
PDA personal digital assistant
PEPFAR US President’s Emergency Plan for AIDS Relief
POC point of care
PPM public–private mix
QMS quality management system
rGLC Regional Green Light Committee
RNTCP Revised National TB Control Programme
[India]
rRNA ribosomal ribonucleic acid
RR relative risk
RR-TB rifampicin-resistant tuberculosis
SD standard deviation
SITT Integrated Tuberculosis Information System
SRL supranational reference laboratory
STAG-TB Strategy and Technical Advisory Group for TB
TAG Treatment Action Group
TB tuberculosis
TB-MAC TB Modelling and Analysis Consortium
TB-TEAM Tuberculosis Technical Assistance Mechanism
TBVI Tuberculosis Vaccine Initiative
TFM transitional funding mechanism
TST tuberculin skin test
UHC universal health coverage
UN United Nations
UNAIDS Joint United Nations Programme on HIV/AIDS
UNITAID international facility for the purchase of
diagnostics and drugs for diagnosis and
treatment of HIV/AIDS, malaria and TB
USAID United States Agency for International
Development
UNPD United Nations Population Division
VR vital registration
WHO World Health Organization
XDR-TB extensively drug-resistant tuberculosis
ZN Ziehl Neelsen

vGLOBAL TUBERCULOSIS REPORT 2013
Acknowledgements
is global tuberculosis (TB) report was produced by a core
team of 15 people: Annabel Baddeley, Anna Dean, Han-
nah Monica Dias, Dennis Falzon, Katherine Floyd, Inés
Garcia, Philippe Glaziou, Tom Hiatt, Irwin Law, Christian
Lienhardt, Linh Nguyen, Charalambos Sismanidis, Hazim
Timimi, Wayne van Gemert and Matteo Zignol. e team
was led by Katherine Floyd. Overall guidance was provided
by the Director of the Global TB Programme, Mario Ravi-
glione.
e data collection forms (long and short versions)
were developed by Philippe Glaziou and Hazim Timimi,
with input from sta throughout the WHO Global TB
Programme. Hazim Timimi led and organized all aspects
of data management. Inés Garcia and Andrea Pantoja
conducted all review and follow-up of nancial data. e
review and follow-up of all other data was done by a team
of reviewers. is included Annabel Baddeley, Annemieke
Brands, Andrea Braza, Katsura Danno, Anna Dean, Han-
nah Monica Dias, Dennis Falzon, Wayne van Gemert, Soleil
Labelle, Knut Lönnroth, Linh Nguyen, Salah Ottmani,
Hazim Timimi, Fraser Wares and Matteo Zignol at WHO
headquarters; Amal Bassili from the Eastern Mediterra-
nean Regional Oce; and Suman Jain, Sai Pothapregada,
Nino Mdivani, Eliud Wandwalo and Mohammed Yassin
from the Global Fund. Data for the European Region were
collected and validated jointly by the WHO Regional Oce
for Europe and the European Centre for Disease Preven-
tion and Control (ECDC); we thank in particular Encarna
Gimenez, Vahur Hollo and Csaba Ködmön from ECDC for
providing validated data les and Andrei Dadu from the
WHO Regional Oce for Europe for his substantial con-
tribution to follow-up and validation of data for all Euro-
pean countries. Review of TB/HIV data was undertaken in
collaboration with Michel Beusenberg, Chika Hayashi, Lisa
Nelson and Michelle Williams from the WHO HIV depart-
ment. Victoria Bendaud, Josephine Dy, and Taavi Erkkola
from UNAIDS managed the process of data collection from
national AIDS programmes, provided a TB/HIV dataset
and worked closely with WHO sta to review and validate
TB/HIV data.
Philippe Glaziou and Charalambos Sismanidis prepared
estimates of TB disease burden and associated gures and
tables (Chapter 2), with support from Tom Hiatt. Particular
thanks are due to Carel Pretorius (Futures Institute), who
worked closely with Philippe Glaziou on analyses and relat-
ed estimates of TB mortality among HIV-positive people,
as well as to Dennis Falzon for coordinating a systematic
review that was used to produce estimates of mortality
related to multidrug-resistant TB (MDR-TB) and to Harish
Nair and Luciana Brondi from the University of Edinburgh
for conducting this review. Tom Hiatt prepared all gures
and tables on TB notication and treatment outcome data
(Chapter 3). Anna Dean, Dennis Falzon and Matteo Zignol
analysed data and prepared the gures and tables related to
drug-resistant TB (Chapter 4), with input from Charalam-
bos Sismanidis. Tom Hiatt and Wayne van Gemert prepared
gures and tables on laboratory strengthening and the roll-
out of new diagnostics (Chapter 5). Annabel Baddeley, Kat-
sura Danno, Tom Hiatt and Linh Nguyen analysed TB/HIV
programmatic data and prepared the associated gures and
tables (Chapter 6). Inés Garcia and Andrew Siroka analysed
nancial data, and prepared the associated gures and
tables (Chapter 7). Christian Lienhardt, Christopher Gilpin
and Karin Weyer prepared the gures on the pipelines for
new TB drugs, diagnostics and vaccines (Chapter 8), with
input from the respective Working Groups of the Stop TB
Partnership. Tom Hiatt coordinated the nalization of all
gures and tables and was the focal point for communica-
tions with the graphic designer.
e writing of the main part of the report was led by
Katherine Floyd, with contributions from Dennis Falzon,
Philippe Glaziou, Irwin Law, Ikushi Onozaki, and Charalam-
bos Sismanidis (Chapter 2); Hannah Monica Dias, Wayne
van Gemert, Haileyesus Getahun, omas Joseph, Mukund
Uplekar and Lana Tomaskovic (Chapter 3); and Inés Gar-
cia and Christian Gunneberg (Chapter 7). Chapter 4, on
drug-resistant TB, was prepared by Anna Dean, Dennis Fal-
zon and Matteo Zignol, with input from Katherine Floyd,
Philippe Glaziou and Charalambos Sismanidis. Chapter 5,
on diagnostics and laboratory strengthening, was prepared
by Wayne van Gemert, with input from Christopher Gilpin,
Fuad Mirzayev and Karin Weyer. Chapter 6 was prepared by
Annabel Baddeley, Haileyesus Getahun, Linh Nguyen and
Katherine Floyd. Chapter 8, on research and development,
was led by Christian Lienhardt, with inputs from Chris-
topher Gilpin, Karin Weyer and Katherine Floyd. Chapter
8 was carefully reviewed by the chairs and secretariats of
the Working Groups of the Stop TB Partnership. Particular
thanks are due to Michael Brennan, Uli Fruth and Jennifer
Woolley (new vaccines); Daniela Cirillo (new diagnostics);
and Barbara Laughon and Mel Spigelman (new TB drugs).
e report team is also grateful to Emily Bloss (US Centers
for Disease Control and Prevention) and Hillary Kipru-
to (WHO Country Oce, Kenya) for their contributions
to content related to strengthening of TB surveillance in
Chapter 2, including a case study of the introduction of

vi GLOBAL TUBERCULOSIS REPORT 2013
electronic recording and reporting in Kenya; to Rajendra
Yadav and Masami Fujita (WHO Country Oce, Cambodia)
for their contribution to an analysis of the integration of
TB, HIV and mother and child health services in Cambodia
(Chapter 6); and to various internal and external reviewers
for useful comments and suggestions on advanced drafts
of chapter text.
e special supplement on the “Countdown to 2015” that
accompanies the global report was prepared by Anna Dean,
Hannah Monica Dias, Katherine Floyd, Irwin Law, Mario
Raviglione, Diana Weil and Karin Weyer, with valuable
inputs from many people at global, regional and country
levels. We thank in particular Sai Pothapregada and Eliud
Wandwalo from the Global Fund, who facilitated discus-
sions with and inputs from many Fund Portfolio Managers.
Annex 1, which explains methods used to produce esti-
mates of the burden of disease caused by TB, was written
by Philippe Glaziou and Charalambos Sismanidis with
very helpful input from Carel Pretorius. We thank Colin
Mathers of the WHO Mortality and Burden of Disease team
for his careful review. e country proles that appear in
Annex 2 and the regional proles that appear in Annex 3
were prepared by Hazim Timimi. Annex 4, which contains
a wealth of global, regional and country-specic data from
the global TB database, was prepared by Tom Hiatt and
Hazim Timimi.
We thank Pamela Baillie in the Global TB Programme’s
monitoring and evaluation team for impeccable admin-
istrative support, Doris Ma Fat from the WHO Mortality
and Burden of Disease team for providing TB mortality
data extracted from the WHO Mortality Database, and
Peter Ghys, Mary Mahy and Karen Stanecki (UNAIDS) for
providing epidemiological data that were used to estimate
HIV-associated TB mortality.
e entire report was edited by Tim France (Inis Com-
munication). We thank him for his excellent work. We also
thank, as usual, Sue Hobbs for her excellent work on the
design and layout of this report. Her contribution, as in
previous years, was greatly appreciated.
e principal source of nancial support for WHO work
on global TB monitoring and evaluation is the United States
Agency for International Development (USAID), without
which it would be impossible to produce the Global Tuber-
culosis Report. Production of the report was also supported
by the governments of Japan and the Republic of Korea. We
acknowledge with gratitude their support.
In addition to the core report team and those mentioned
above, the report beneted from the input of many sta
working in WHO regional and country oces and hundreds
of people working for national TB programmes or within
national surveillance systems who contributed to the
reporting of data and to the review of report material prior
to publication. ese people are listed below, organized by
WHO region. We thank them all for their invaluable contri-
bution and collaboration, without which this report could
not have been produced.
Among the WHO sta not already mentioned above, we
thank in particular Khurshid Alam Hyder, Daniel Kibuga,
Rafael López Olarte, André Ndongosieme, Wilfred Nkhoma
and Henriette Wembanyama for their major contribution
to facilitation of data collection, validation and review.
9*1staHHinregionalandcoWntr[oHßces
WHO African Region
Harura Adamu, Boubacar Ould Abdel Aziz, Esther Aceng,
Inacio Alvarenga, Balde Amadou, Ayodele Awe, Sanni Baba-
tunde, Bazie Babou, Nayé Bah, Marie Barouan, Abera Bekele,
Norbert Bidounga, Gaël Claquin, Augusto da Cruz Claudi-
na, Peter Clement, Noel Djemadji, Ismael Hassen Endris,
Amos Omoniyi Fadare, Louisa Ganda, Boingotlo Gasen-
nelwe, Patrick Hazangwe, Joseph Imoko, Michael Jose,
Joel Kangangi, Katherine Lao, Nzuzi Katondi, Bah Keita,
Daniel Kibuga, Hillary Kipruto, Désiré Aristide Komangoya
Nzonzo, Sharmila Lareef-Jah, Frank Lule, Mwendaweli
Maboshe, Mbemba Leonard, Richard Mbumba, Julie Mug-
abekazi, André Ndongosieme, Denise Nkezimana, Wilfred
Nkhoma, Nicolas Nkiere, Ghislaine Nkone Asseko, Ishmael
Nyasulu, Laurence Nyiramasarabwe, Samuel Ogiri, Daniel
Olusoti, Amos Omoniyi, Chijioke Osakwe, Felicia Owu-
su-Antwi, Philips Patrobas, Kalpeshsinh Rahevar, Bacary
Sambou, Kefas Samson, Neema Simkoko, Desta Tiruneh,
Alexis Tougordi, Henriette Wembanyama.
WHO Region of the Americas
Monica Alonso Gonzalez, Angel Manuel Alvarez, Luis
Gerardo Castellanos, Gerardo de Cossio, Rachel Eersel,
Marcos Espinal, Ingrid García, Mirtha Del Granado, Rosa-
linda Hernández, Vidalia Lesmo, Rafael López Olarte,
Wilmer Marquiño, ais dos Santos, Alfonso Tenorio, Jorge
Victoria, Anna Volz.
WHO Eastern Mediterranean Region
Mohamed Abdel Aziz, Ali Akbar, Samiha Baghdadi, Amal
Bassili, Najwa El Emam, Hamida Khattabi, Aayid Munim,
Ghulam Nabi Kazi, Ali Reza Aloudel, Gabriele Riedner,
Karam Shah, Sindani Ireneaus Sebit, Bashir Suleiman,
Rahim Taghizadeh.
WHO European Region
Martin van den Boom, Brenda van den Bergh, Andreea
Cassandra Butu, Silvu Ciobanu, Pierpaolo de Colombani,
Andrei Dadu, Irina Danilova, Masoud Dara, Jamshid
Gadoev, Gayane Ghukasyan, Sayohat Hasanova, Arax Hov-
hannesyan, Saliya Karymbaeva, Mehmet Kontas, Kristin
Kremer, Dmitriy Pashkevich, Valiantsin Rusovich, Bog-
dana Shcherbak-Verlan, Javahir Suleymanova, Szabolcs
Szigeti, Melita Vujnovic.
WHO South-East Asia Region
Mohammad Akhtar, Vikarunnesa Begum, Erwin Coore-
man, Deki, Khurshid Alam Hyder, Navaratnasingam
Janakan, Kim Tong Hyok, La Win Maung, Jorge Luna, Par-
tha Mandal, Amaya Maw-Naing, Giampaolo Mezzabotta,

viiGLOBAL TUBERCULOSIS REPORT 2013
Bo Myint, Ye Myint, Eva Nathanson, Rajesh Pandav, Razia
Pendse, Sri Prihatini, K Rezwan, Rim Kwang Il, Hwang Kum
Ryong, Mukta Sharma, Aminath Shenalin, Achuthan Nair
Sreenivas, Chawalit Tantinimitkul, Wangchuk Lungten.
9*19GUVGTP2CEKßE4GIKQP
Shalala Ahmadova, Niño Dayanghirang, Asaua Faasino,
Salu Failauga, Ogtay Gozalov, Cornelia Hennig, Tom Hiatt,
Tauhid Islam, Narantuya Jadambaa, Ridha Jebeniani, Sung
Hye Kim, Miwako Kobayashi, Woo-Jin Lew, Katsunori
Osuga, Khanh Pham, Fabio Scano, Jacques Sebert, Cathari-
na van Weezenbeek, Rajendra Yadav, Dongbao Yu.
National respondents who contributed to
reporting and verißcation oH data
WHO African Region
Abdou-Salam Abderemane, Ouédraogo Adama, Abdelra-
him Barka Abderramane, Jean Louis Abena Foe, Soane
Alihalassa, Arlindo Amaral, Kouamé Amenan, Séverin
Anagonou, Younoussa Assoumani, Georges Bakaswa, Ada-
ma Marie Bangoura, Jorge Noel Barreto, Ballé Boubakar,
Victor Bonkoungou, Frank Adae Bonsu, Miguel Camara,
Evangelista Chisakaitwa, Ernest Cholopray, Nkemdilim
Chukwueme, Catherine Cooper, Swasilanne da Silva, B. de
Sousa Bandeira, Isaias Dambe, Davi Kokou Mawulé, Serge
Diagbouga, Aicha Diakité, Awa Helene Diop, Sicelo Dlami-
ni, emba Dlamini, addée Ndikumana, Oumou Fofana,
Susan Gacheri, Evariste Gasana, Michel Gasana, Sandile
Ginindza, Martin Gninafon, Nii Nortey Hanson-Nortey,
Adama Jallow, Saa Kamara, Madou Kane, Henry Kanyer-
ere, Nathan Kapata, Biruck Kebede, Kerram Aziza, Deogra-
tias Kibambazi, Patrick Konwuloh, Jacquemin Kouakou,
Popaul Kulonga, Rossin Lebeke, Lillian Ishengoma, Llang
Bridget Maama-Maime, Marcel Lougue, Maxime Lunga,
Ghislaine Mabeluanga Tshitenge, Jocelyn Mahoumbou,
Angelo Makpenon, David Mametja, Ivan Manhiça, Tseliso
Marata, Farai Mavhunga, Mba Bekolo Frenk José Mathieu,
Salem Salem Mohameden, Louine Morel, Youwaoga Isidore
Moyenga, James Mpunga, Frank Mugabe, Kenneth Mugi-
sha, Cliord Munyandi, Lindiwe Mvusi, Aboubacar Mzem-
baba, Ronald Ncube, Fulgence Ndayikengurukiye, Yvon
Martial Ngana, Antoine Ngoulou, Lourenço Nhocuana,
Blasdus Franz Njako, Emmanuel Nkiligi, M Nkou, Josh-
ua Obasanya, Davidson Ogunade, Hermann Ongouo,
Abdelhadi Oumar, Issoufou Ousmane, Maria Conceição
Palma, Victor Pereira, ato Raleting, Sahondra Jeanine
Randriambeloson, Rujeedawa Mohammed Fezul, Samey
Agbenyegan, Charles Sandy, Kebba D Sanneh, Marie Sarr
Diouf, Mineab Sebhatu, Mamie Shoma, Angele Shoma
Matota, René Simalo, Joseph Sitienei, Nicholas Siziba,
Philippe Takongo, Celstino Francisco Teixeira, Mohamed
Abdallahi Traoré, Nassiama Traoré, Kassim Traoré, Alie
Wurie, Eucher Dieudonné Yazipo, Ranivomahefa Myrienne
Bakoliarisoa Zanajohary, Abbas Zezai, Eric Ismaël Zoun-
grana.
WHO Region of the Americas
Christian Acosta, Shalauddin Ahmed, Valentina Antonie-
ta Alarcon Guizado, Xochil Alemán de Cruz, Kiran kumar
Alla, Valeria Almanza Torrez, Mirian Alvarez, Raúl Álva-
rez, Aisha Andrewin, A. Alister Antoine, Chris Archibald,
Carlos Alberto Marcos Ayala Luna, Wiedjaiprekash Bale-
sar, Draurio Barreira, Patricia Bartholomay, Soledad Bel-
trame, María del Carmen Bermúdez, Lynrod Brooks, Marta
Calona de Abrego, Martín Castellanos Joya, Jorge Castillo
Carbajal, Kenneth Castro, Judith Cazares, Gemma Chery,
Carlos Cuadra, Ofelia Cuevas, D’Auvergne Cleophas, Jose
Davy, Cecilia de Arango, Eva de Weever-Lista, Camille Dele-
veaux, Dy-Juan De Roza, Roger Duncan, España Cedeño
Mercedes, Manuel Salvador España Rueda, Fernandez
Hugo, Cecilia Figueroa Benites, Victor Gallant, Julio Garay
Ramos, Sarita Aguirre García, Izzy Gerstenbluth, Margar-
ita Godoy, Roscio Gómez, Ilse Maria Góngora Rivas, Sil-
vino González, Yaskara Halabi, Kevin Harvey, Dorothea
Hazel, Maria Henry, Tania Herrera, Carla Jeries, Dihad-
enys Lemus Molina, Athelene Linton, Maria Josefa Llanes
Cordero, Marvin Andres Maldonado Rivera, Maldonado
Saavedra Andrea, Marcelino Belkys, Eva Martìnez, María
de Lourdes Martínez Olivares, Zeidy Mata Azofeifa, Joan
McLeod-Simon, Timothy McLaughlin-Munroe, Roque
Miramontes, Leilawatie Mohammed, Jeetendra Mohanlall,
Ernesto Moreno, Francis Morey, Willy Morose, Michael
Owen, Cheryl Peek-Ball, Janelle Pickering, Tomasa Por-
tillo, Irad Potter, Manohar Singh Rajamanickam, Dottin
Ramoutar, Anna Esther Reyes Godoy, Paul Ricketts, Jorge
Rodriguez De Marco, Myrian Román, Nilda de Romero,
Carolyn Russell, Wilmer Salazar, Deborah Stijnberg, Sutton
Jackurlyn, Torres Clarita, Maribelle Tromp, William Turn-
er, Melissa Valdez, Daniel Vázquez, Nestor Vera, Michael
Williams, David Yost, Oritta Zachariah.
WHO Eastern Mediterranean Region
Fadhil Abbas, Mohammad S Abouzeid, Khaled Abu Ruh-
man, Nadia Abu Sabra, Ahmadi Shahnaz, Mohamed Redha
Al Lawati, Al Saidi Fatmah, Samia Ali Alagab, Abdelbary
Abdullah Ahmed Al-Hammadi, Abdullatif Al-Khal, Saeed
Al Saar, Kifah Alshaqeldi, Bahnasy Samir, Bennani Ken-
za, Kinaz Cheikh, Walid Daoud, Mohamed Furjani, Amal
Galal, Dhikrayet Gamara, Assia Haissama Mohamed, Hiba
Kamal Hamad Elneel, Kaalthoom Hassan, Hawa Hassan
Guessod, Lou Joseph, Onwar Otien Jwodh Chol, Basharat
Khan, Joseph Lasu, Sayed Daoud Mahmoodi, Khadi-
ga Adam Mohammed, Mokhtar Alaa, Mulham Mustafa,
Nasehi Mahshid, Ejaz Qadeer, Mohammad Khalid Seddiq,
Sghiar Mohammed, Mohemmed Tabena, Tamara Tayeb,
Najib Abdul aziz Abdullah abit, Seddik Walha, Yaacoub
Hiam.
WHO European Region
Abildaev Tleukhan Shildebaevich, Mokhonim Abdulloeva,
Ibrahim Abubakar, Rag Abuzarov, Nurhan Albayrak,
Natavan Alikhanova, Avtandil Alisherov, Ewa Augustynowicz-

viii GLOBAL TUBERCULOSIS REPORT 2013
Kopeć, Ekkehardt Altpeter, Laura Anderson, Delphine
Antoine, Trude Margrete Arnesen, Rusudan Aspindzelash-
vili, Andrei Astrovko, Elizabeta Bachiyska, Anna Ivanovna
Barbova, Yana Besstraschnova, Venera Lazarevna Bismil-
da, Oktam Ikramovich Bobokhojaev, Olivera Bojovic, Eric
C. Böttger, Bonita Brodhun, Noa Cedar, Daniel Chemtob,
Domnica Ioana Chiotan, Ana Ciobanu, Nico Cioran, Andra
Cirule, ierry Comolet, Radmila Curcic, Manfred Danilo-
vitš, Edita Davidaviciene, Hayk Davtyan, Pava Dimitrijevic,
António Diniz, Francis Drobniewski, Raquel Duarte, Mlad-
en Duronjic, Connie Erkens, Jennifer Fernandez Garcia,
Lyalya Gabbasova, Viktor Gasimov, Lárus Jón Guðmunds-
son, Gennady Gurevich, Walter Haas, Hasan Hazi, Evg-
eny Hanyukov, Armen Hayrapetyan, Peter Helbling, Sven
Honer, Daniela Homorodean, Jahongir Jurakhonovich
Ismoilov, Mamuka Japaridze, Vincent Jarlier, Soledad
Jiménez Pajares, Jerker Jonsson, Abdullat Kadyrov, Gul-
mira Kalmambetova, Dmitry Klymuk, Maria Korzeniewska-
Kosela, Ainura Koshoeva, Košnik Mitja, Gabor Kovacs,
Tiina Kummik, Nino Lomtadze, Stevan Lučić, Jasminka
Maglajllic, Turid Mannsåker, Mathys Vanessa, Rafail Meh-
diyev, Rukije Mehmeti, Donika Mema, Vladimir Milanov,
Alvard Mirzoyan, Gjyle Mulliqi, Gulnora Murmusaeva,
Seher Musaonbasioglu, Ucha Nanava, Zdenka Novakova,
Joan O’Donnell, Analita Pace Asciak, Clara Palma Jordana,
Elena Pavlenko, Olga Pavlova, Monique Perrin, Edita Pim-
kina, Monika Polanova, Georgeta Gilda Popescu, Gordana
Radosavljevic Asic, Bozidarka Rakocevic, omas Rendal,
Vija Riekstina, Jerome Robert, Elena Rodríguez Valín, Tom
Rogers, Elena Romancenco, Kazimierz Roszkowski-Sliz,
Sabine Rüsch-Gerdes, Branislava Savic, Gérard Scheiden,
Hasia Kaidar Shwartz, Anabela Silva, Girts Skenders, Cath-
rine Slorbak, Erika Slump, Hanna Soini, Ivan Solovic, Dick
van Soolingen, Flemming Stenz, Sergey Sterlikov, Jana
Svecova, Svetina Šorli Petra, Silva Tafaj, Talevski Stefan,
Odorina Tello Anchuela, Mirzagaleb Tillyashaykhov, Aida
Ustamujic, Gulnoz Uzakova, Tonka Varleva, Piret Viiklepp,
Cveta Vragoterova, Gerard de Vries, Jiri Wallenfels, Wan-
lin Maryse, Pierre Weicherding, Aysegul Yildirim, Zakoska
Maja, Oksana Zalutskaya, Ilona Zemanová, Manca Žolnir
Dovč, Hasan Zutic.
WHO South-East Asia Region
Shina Ahmed, Aminath Aroosha, Choe Kum Song,
Emdadul Hoque, RS Gupta, Sirinapha Jittimanee, Suksont
Jittimanee, Niraj Kulshrestha, Constantino Lopes, an-
dar Lwin, Dyah Erti Mustikawati, Tin Zar Naing, Chawet-
san Namwat, Md Nuruzzaman Haque, Nirupa Pallewatta,
Rajendra Prasad Pant, Kiran Rade, Dyah Armi Riana, Che-
wang Rinzin, Sudath Samaraweera, Gamini Senevirathne,
Janaka ilakarathne, Sabino Viegas, Bimal Kumar Yadav.
9*19GUVGTP2CEKßE4GIKQP
Paul Aia, Cecilia Teresa T. Arciaga, Nemia Bainivalu, Chris-
tina Bareja, Risa J. Bukbuk, Cheng Shiming, Phonenaly
Chittamany, Chou Kuok Hei, Nese Ituaso Conway, Du
Xin, Mayleen J. Ekiek, Fanai Saen, Rangiau Fariu, Ludovic
Floury, Louise Fonua, Jiloris Frederick Dony, Anna Marie
Celina Garn, Go Un-Yeong, Shakti Gounder, Anie Hary-
ani Hj Abdul Rahman, Noel Itogo, Tom Jack, Seiya Kato,
Khin Mar Kyi Win, Lamar Daniel, Leo Lim, Liza Lopez,
Sakiusa Mainawalala, Henri-Pierre Mallet, Tan Eang Mao,
Markleen Tagaro, Sera Moa, Suzana Mohd Hashim, Nguy-
en Binh Hoa, Nguyen Viet Nhung, Nou Chanly, Ochirbat
Batbayar, Connie Bieb Olikong, Park Yoon-Sung, Nukutau
Pokura, Waimanu Pulu, Purev Nasanjargal, Rabauliman
Marcelina, Bereka Reiher, Bernard Rouchon, Temilo Seono,
Tokuaki Shobayashi, Vita A. Skilling, Grant Storey, Phan-
nasinh Sylavanh, Kenneth Reuee Tabutoa, Tam Cheuk
Ming, Kyaw u, Tieng Sivanna, Tong Ka Io, Rosalind Vian-
zon, Wang Yee Tang, Wang Lixia.

ixGLOBAL TUBERCULOSIS REPORT 2013
Executive summary
Tuberculosis (TB) remains a major global health problem.
In 2012, an estimated 8.6 million people developed TB and
1.3 million died from the disease (including 320000 deaths
among HIV-positive people).1 e number of TB deaths is
unacceptably large given that most are preventable.
Nearly 20 years after the WHO declaration of TB as a
global public health emergency, major progress has been
made towards 2015 global targets set within the context
of the Millennium Development Goals (MDGs). Two years
ahead of the deadline, the Global Tuberculosis Report 2013
and accompanying supplement Countdown to 2015 assess
progress towards the 2015 targets and the top priority
actions needed to achieve and/or move beyond them.
C17N6&19N 61 key ßndings
On track:
e rate of new TB cases has been falling worldwide for
about a decade, achieving the MDG global target. TB
incidence rates are also falling in all six WHO regions.
e rate of decline (2% per year) remains slow.
Globally by 2012, the TB mortality rate had been reduced
by 45% since 1990. e target to reduce deaths by 50%
by 2015 is within reach.
Two WHO regions have already achieved the 2015 tar-
gets for reduced incidence, prevalence and mortality: the
Region of the Americas and the Western Pacic Region.
Of the 22 high TB burden countries (HBCs) that account
for about 80% of the world’s TB cases,2 seven have met
all 2015 targets for reductions in TB incidence, preva-
lence and mortality. Four more HBCs are on track to do
so by 2015.
Off track:
By 2012, the level of active TB disease in the community
(prevalence) had fallen by 37% globally since 1990. e
target of a 50% reduction by 2015 is not expected to be
achieved.
e African and European regions are currently not on
track to achieve the mortality and prevalence targets.
Among the 22 HBCs, 11 are not on track to reduce inci-
dence, prevalence and mortality in line with targets.
Reasons include resource constraints, conict and insta-
bility, and generalized HIV epidemics.
Progress towards targets for diagnosis and treatment
of multidrug-resistant TB (MDR-TB) is far o-track.
Worldwide and in most countries with a high burden of
MDR-TB, less than 25% of the people estimated to have
MDR-TB were detected in 2012.
Many countries have made considerable progress to
address the TB/HIV co-epidemic. However, global-
level targets for HIV testing among TB patients and
provision of antiretroviral therapy (ART) to those who
are HIV-positive have not been reached.
Five priority actions required to accelerate progress
towards 2015 targets:
1. Reach the missed cases. About 3 million people who
developed TB in 2012 were missed by national notica-
tion systems. Key actions needed to detect people with
the illness and ensure that that they get the right treat-
ment and care include: expanded services (including
rapid tests) throughout health systems bolstered by the
support of nongovernmental organizations, community
workers and volunteers to diagnosis and report cases;
intensied collaboration with public hospitals and pri-
vate health facilities who are treating patients but not
reporting; instituting mandatory notication of cases in
more countries; and better data compilation.
2. Address MDR-TB as a public health crisis. In high
MDR-TB burden countries, increased capacity to diagnose
MDR-TB must be matched with supplies of quality drugs
and scaled-up country capacity to deliver eective treat-
ment and care. is will require high-level political will
and leadership and more collaboration among partners,
including drug regulatory authorities, donor and techni-
cal agencies, civil society and the pharmaceutical industry.
3. Accelerate the response to TB/HIV. e top prior-
ity is to increase coverage of ART for HIV-positive TB
patients towards the 100% target. Expanded coverage of
TB preventive treatment among people living with HIV
is the second priority.
4. Increase nancing to close all resource gaps. An
estimated US$7–8 billion per year is required for a full
response to the TB epidemic in low- and middle-income
countries in 2014 and 2015 (excluding research and
development for new TB diagnostics, drugs and vac-
cines). Funding in 2013 is about US$6 billion. Increas-
es in both domestic and donor nancing are needed to
close the gap of up to US$ 2 billion per year, including
via the full replenishment of the Global Fund in 2013.
Progress remains fragile and could be reversed without
adequate funding.
5. Ensure rapid uptake of innovations. e fast uptake
of new tools and strategies for better diagnosis, treat-
ment and prevention of all forms of TB can be accelerated
by country-specic operational research and translation
of ndings into policy and practice.

xGLOBAL TUBERCULOSIS REPORT 2013
ADDITIONAL FINDINGS
e report is based primarily on data provided by WHO’s
Member States. In 2013, data were reported by 178 Mem-
ber States and a total of 197 countries and territories that
collectively have more than 99% of the world’s TB cases.
Burden of disease
e current global picture of TB shows continued
progress, but not fast enough.
An estimated 1.1 million (13%) of the 8.6 million peo-
ple who developed TB in 2012 were HIV-positive. About
75% of these cases were in the African Region.
Globally in 2012, an estimated 450 000 people devel-
oped MDR-TB and there were an estimated 170 000
deaths from MDR-TB .
Most TB cases and deaths occur among men, but TB
remains among the top three killers of women world-
wide. ere were an estimated 410 000 TB deaths among
women in 2012, including 160 000 among HIV-positive
women. Half of the HIV-positive people who died from
TB in 2012 were women. Of the estimated 8.6 million new
TB cases worldwide in 2012, 2.9 million were women.
ere were an estimated 530 000 TB cases among
children (under 15 years of age) and 74000 TB deaths
(among HIV-negative children) in 2012 (6% and 8% of
the global totals, respectively).
e majority of cases worldwide in 2012 were in the
South-East Asia (29%), African (27%) and Western Pacif-
ic (19%) regions. India and China alone accounted for
26% and 12% of total cases, respectively.
e TB incidence rate at country level ranges substan-
tially, with around 1000 or more cases per 100 000 peo-
ple in South Africa and Swaziland, and fewer than 10
per 100000 population in parts of the Americas, several
countries in western Europe, Japan, Australia and New
Zealand.
TB detection and treatment outcomes
Millions of people access eective TB care each year
but “missed cases” hold back gains.
Between 1995 and 2012, 56 million people were success-
fully treated for TB in countries that had adopted WHO’s
global TB strategy, saving 22 million lives.
In 2012, 6.1 million cases of TB were notied to national
TB programmes (NTPs). Of these, 5.7 million were peo-
ple newly diagnosed in 2012 and 0.4 million were previ-
ously diagnosed TB patients whose treatment regimen
was changed.
In 2011, the treatment success rate continued to be high
at 87% among all new TB cases.
Notications of TB cases have stabilized globally. In
2012, about 66% (5.7 million) of the estimated 8.6 mil-
lion people who developed TB were notied as newly
diagnosed cases.
About 75% of the estimated 2.9 million missed cases
– people who were either not diagnosed or diagnosed but
not reported to NTPs – were in 12 countries. In order of
total numbers, these were India (31% of the global total),
South Africa, Bangladesh, Pakistan, Indonesia, China,
Democratic Republic of the Congo, Mozambique, Nige-
ria, Ethiopia, the Philippines and Myanmar.
Xpert® MTB/RIF, a rapid molecular diagnostic test,
is being rapidly adopted by countries to detect TB and
rifampicin-resistant TB. By end June 2013, 1402 testing
machines and 3.2 million test cartridges had been pro-
cured by 88 of the 145 countries eligible for concessional
prices.
Treatment success rates for TB remain lowest in the
European Region, where in 2011 only 72% of new cases
were successfully treated.
MDR-TB and XDR-TB detection and treatment outcomes
Undetected cases and treatment coverage gaps con-
stitute a public health crisis.
Globally in 2012, data from drug resistance surveys and
continuous surveillance among notied TB cases sug-
gest that 3.6% of newly diagnosed TB cases and 20% of
those previously treated for TB had MDR-TB. e high-
est levels of MDR-TB are found in eastern Europe and
central Asia, where in some countries more than 20%
of new TB cases and more than 50% of those previously
treated for TB have MDR-TB.
A total of 94000 TB patients eligible for MDR-TB treat-
ment were detected in 2012: 84000 people with con-
rmed MDR-TB (i.e. resistance to both rifampicin, the
most powerful TB drug, and isoniazid), plus 10 000
with rifampicin resistance detected using Xpert MTB/
RIF. is was a 42% increase in detected cases eligible
for treatment compared with 2011. e largest increases
between 2011 and 2012 were in India, South Africa and
Ukraine.
Just over 77000 people with MDR-TB were started on
second-line treatment in 2012, equivalent to 82% of
the 94 000 newly detected cases that were eligible for
treatment globally. Treatment coverage gaps for detect-
ed cases were much larger in some countries, especially
in the African Region (51% enrolled in treatment), and
widened in China, Pakistan and South Africa.
At least one case of extensively drug-resistant TB (XDR-
TB) had been reported by 92 countries by the end of
2012. On average, an estimated 9.6% of MDR-TB cases
have XDR-TB.
Globally, only 48% of MDR-TB patients in the 2010
cohort of detected cases were successfully treated,
reecting high mortality rates and loss to follow-up. A
treatment success rate of 75% or more for patients with
MDR-TB was achieved in 34 of 107 countries.

xiGLOBAL TUBERCULOSIS REPORT 2013
Addressing TB-HIV
TB-HIV collaborative services are expanding, but
global targets are not yet in sight.
e main interventions to reduce the burden of HIV in
TB patients are HIV testing and provision of ART and
cotrimoxazole preventive therapy (CPT) to those found
to be HIV-positive. e main interventions to reduce TB
among people living with HIV are regular screening for
TB among people in HIV care and provision of isoniazid
preventive therapy (IPT) to those without active TB who
meet eligibility criteria (estimated at 50% of those newly
enrolled in HIV care).
Progress in the implementation of TB/HIV interven-
tions was further consolidated in 2012. Globally, 46%
of TB patients knew their HIV status (up from 40% in
2011). In the African Region that has the highest TB/
HIV burden, 74% of TB patients knew their HIV status
(up from 69% in 2011). Among the 41 countries with the
highest TB/HIV burden, more than 85% of TB patients
knew their HIV status in 15 countries, and in 7 of these
countries over 90% of patients knew their HIV status.
e coverage of ART among TB patients who were
known to be HIV-positive reached 57% in 2012, up from
49% in 2011. As in the past few years, about 80% of HIV-
positive TB patients were treated with CPT.
In 2012, 4.1 million people enrolled in HIV care were
reported to have been screened for TB, up from 3.5 mil-
lion in 2011. Of the reported 1.6 million people newly
enrolled in HIV care in 2012, 0.5 million (31%) were
provided with IPT.
6$ßPCPEKPI
International donor funding and more domestic
investments are essential.
Of the US$ 7‒8 billion per year required in low and
middle-income countries in 2014 and 2015, about two
thirds is needed for the detection and treatment of drug-
susceptible TB, 20% for treatment of MDR-TB, 10%
for rapid diagnostic tests and associated laboratory
strengthening, and 5% for collaborative TB/HIV activ-
ities.
Growth in domestic and international donor funding
has been clearly documented since 2002. ere is capac-
ity to further increase domestic funding, especially in
BRICS (Brazil, the Russian Federation, India, China and
South Africa) that have almost 50% of global TB cases.
International donor funding reported by NTPs amount-
ed to US$ 0.8 billion in 2013, about three-quarters of
which was from the Global Fund. To close resource gaps,
at least US$1.6 billion is needed in both 2014 and 2015.
International donor funding is crucial in many coun-
tries, accounting for more than 50% of total funding in
the group of 17 HBCs excluding BRICS, and in all low-
income countries. e proportion is even higher in some
individual countries.
Research and development
New TB diagnostics, medicines and vaccines are cru-
cial to end the global TB epidemic.
More than 50 companies are involved in development of
new diagnostic tests.
10 new or repurposed TB drugs are in late phases of clin-
ical development. In late 2012, bedaquiline became the
rst novel TB drug approved in 40 years. In June 2013,
WHO issued interim guidance for its use in treatment
of MDR-TB.
ere are 10 vaccines for TB prevention and two immu-
notherapeutic vaccines in the pipeline. In early 2013,
results from a Phase IIb proof-of-concept study of one of
the preventive vaccine candidates were published. While
ecacy was not superior to the Bacille-Calmette-Guérin
(BCG) vaccine alone, the study demonstrated that a
trial of a novel TB vaccine is feasible in a high TB burden
setting.
Short, eective and well-tolerated treatments for latent
TB infection, a point-of-care diagnostic test, and an
eective post-exposure vaccine are needed to help end
the global TB epidemic.
1 e estimated number of TB deaths among HIV-positive people
in 2011 was 336000. Estimates of TB deaths among HIV-positive
people for the entire period 1990‒2012 were updated in 2013 using
the Spectrum software, which has been used for more than a decade
to produce estimates of the burden of disease caused by HIV. In
2013, a TB module in Spectrum was available for the rst time for
use in the country consultations on HIV burden estimates that are
organized by UNAIDS every two years. Estimation of the number
of TB cases living with HIV, and of the number of TB deaths among
HIV-positive people, was integrated into this process.
2 e 22 HBCs are Afghanistan, Bangladesh, Brazil, Cambodia,
China, the Democratic Republic of the Congo, Ethiopia, India,
Indonesia, Kenya, Mozambique, Myanmar, Nigeria, Pakistan, the
Philippines, the Russian Federation, South Africa, ailand, Ugan-
da, the United Republic of Tanzania, Viet Nam and Zimbabwe.
1GLOBAL TUBERCULOSIS REPORT 2013
%*#26'4
Introduction
Tuberculosis (TB) remains a major global health problem.
It causes ill-health among millions of people each year and
ranks as the second leading cause of death from an infec-
tious disease worldwide, after the human immunode-
ciency virus (HIV). e latest estimates included in this
report are that there were 8.6 million new TB cases in 2012
and 1.3 million TB deaths (just under 1.0 million among
HIV-negative people and 0.3 million HIV-associated TB
deaths). Most of these TB cases and deaths occur among
men, but the burden of disease among women is also high.
In 2012, there were an estimated 2.9 million cases and
410 000 TB deaths among women, as well as an estimat-
ed 530 000 cases and 74 000 deaths among children.1 e
number of TB deaths is unacceptably large given that most
are preventable if people can access health care for a diag-
nosis and the right treatment is provided. Short-course reg-
imens of rst-line drugs that can cure around 90% of cases
have been available for decades.
ese large numbers of cases and deaths notwithstand-
ing, 20 years on from the 1993 World Health Organization
(WHO) declaration of TB as a global public health emergen-
cy, major progress has been made. Globally, the TB mortal-
ity rate (deaths per 100 000 population per year) has fallen
by 45% since 1990 and TB incidence rates (new cases per
100 000 population per year) are falling in most parts of
the world. In the 18 years since the launch of a new inter-
national strategy for TB care and control by WHO in the
mid-1990s (the DOTS strategy) and the subsequent global
rollout of DOTS and its successor (the Stop TB Strategy,2
Box 1.2), a cumulative total of 56 million people were suc-
cessfully treated for TB between 1995 and 2012, saving
approximately 22 million lives.
e overarching goal of the Stop TB Strategy is to achieve
2015 global targets (shown in Box 1.2) for reductions in
the burden of disease caused by TB. e target set within
the United Nations (UN) Millennium Development Goals
(MDGs) is that TB incidence should be falling by 2015
(MDG Target 6.c). Besides incidence, four other TB indi-
cators are included in the MDG monitoring framework:
the prevalence rate, the mortality rate, the case detection
rate (the number of notied cases divided by the estimated
number of incident cases in the same year, expressed as a
percentage), and the treatment success rate (the percentage
BOX 1.1
Basic facts about TB
TB is an infectious disease caused by the bacillus
Mycobacterium tuberculosis. It typically affects the
lungs (pulmonary TB) but can affect other sites as well
(extrapulmonary TB). The disease is spread in the air when
people who are sick with pulmonary TB expel bacteria,
for example by coughing. In general, a relatively small
proportion of people infected with M. tuberculosis will
develop TB disease; however, the probability of developing
TB is much higher among people infected with HIV. TB is
also more common among men than women, and affects
mostly adults in the economically productive age groups.
The most common method for diagnosing TB worldwide
is sputum smear microscopy (developed more than 100
years ago), in which bacteria are observed in sputum
samples examined under a microscope. Following recent
breakthroughs in TB diagnostics, the use of rapid molecular
tests for the diagnosis of TB and drug-resistant TB is
increasing, as highlighted in Chapter 5 and Chapter 8
of this report. In countries with more developed laboratory
capacity, cases of TB are also diagnosed via culture methods
(the current reference standard).
Without treatment, TB mortality rates are high. In studies
of the natural history of the disease among sputum smear-
positive/HIV-negative cases of pulmonary TB, around 70%
died within 10 years; among culture-positive (but smear-
negative) cases, 20% died within 10 years.a
'HHGEVKXGFTWIVTGCVOGPVUYGTGßTUVFGXGNQRGFKPVJG
U6JGOQUVGHHGEVKXGßTUVNKPGCPVK6$FTWITKHCORKEKP
became available in the 1960s. The currently recommended
treatment for new cases of drug-susceptible TB is a six-
OQPVJTGIKOGPQHHQWTßTUVNKPGFTWIUKUQPKC\KFTKHCORKEKP
GVJCODWVQNCPFR[TC\KPCOKFG6TGCVOGPVUWEEGUUTCVGUQH
QTOQTGHQTPGYECUGUCTGTGIWNCTN[TGRQTVGFVQ9*1
by Member States (Chapter 3). Treatment for multidrug-
TGUKUVCPV6$/&46$FGßPGFCUTGUKUVCPEGVQKUQPKC\KFCPF
rifampicin (the two most powerful anti-TB drugs) is longer,
and requires more expensive and more toxic drugs. For most
RCVKGPVUYKVJ/&46$VJGEWTTGPVTGIKOGPUTGEQOOGPFGF
by WHO last 20 months, and treatment success rates are
much lower (Chapter 4(QTVJGßTUVVKOGKPHQWTFGECFGU
new TB drugs are starting to emerge from the pipeline and
combination regimens that include new compounds are
being tested in clinical trials, as discussed in Chapter 8.
There are several TB vaccines in Phase I or Phase II trials
(Chapter 8). For the time being, however, a vaccine that is
effective in preventing TB in adults remains elusive.
a 6KGOGTUOC'9GVCN0CVWTCNJKUVQT[QHVWDGTEWNQUKUFWTCVKQPCPF
HCVCNKV[QHWPVTGCVGFRWNOQPCT[VWDGTEWNQUKUKP*+8PGICVKXGRCVKGPVU
A systematic review. PLoS ONEG
1 e estimated number of deaths among children excludes TB
deaths in HIV-positive children, for which estimates are not yet
available. Further details are provided in Chapter 2.
2 Raviglione M, Uplekar M. WHO’s new Stop TB strategy. e Lancet,
2006, 367: 952–5.

2GLOBAL TUBERCULOSIS REPORT 2013
BOX 1.2
The Stop TB Strategy at a glance
THE STOP TB STRATEGY
VISION A TB-free world
GOAL To dramatically reduce the global burden of TB by 2015 in line with the Millennium Development Goals
(MDGs) and the Stop TB Partnership targets
OBJECTIVES ■ Achieve universal access to high-quality care for all people with TB
■ 4GFWEGVJGJWOCPUWHHGTKPICPFUQEKQGEQPQOKEDWTFGPCUUQEKCVGFYKVJ6$
■ Protect vulnerable populations from TB, TB/HIV and drug-resistant TB
■ Support development of new tools and enable their timely and effective use
■ Protect and promote human rights in TB prevention, care and control
TARGETS ■ /&)6CTIGVE*CNVCPFDGIKPVQTGXGTUGVJGKPEKFGPEGQH6$D[
■ 6CTIGVUNKPMGFVQVJG/&)UCPFGPFQTUGFD[VJG5VQR6$2CTVPGTUJKR
¿TGFWEGRTGXCNGPEGQHCPFFGCVJUFWGVQ6$D[EQORCTGFYKVJCDCUGNKPGQH
¿GNKOKPCVG6$CUCRWDNKEJGCNVJRTQDNGOFGßPGFCUECUGRGTOKNNKQPRQRWNCVKQPRGT[GCT
COMPONENTS
1. Pursue high-quality DOTS expansion and enhancement
C 5GEWTGRQNKVKECNEQOOKVOGPVYKVJCFGSWCVGCPFUWUVCKPGFßPCPEKPI
b. Ensure early case detection, and diagnosis through quality-assured bacteriology
E 2TQXKFGUVCPFCTFK\GFVTGCVOGPVYKVJUWRGTXKUKQPCPFRCVKGPVUWRRQTV
d. Ensure effective drug supply and management
e. Monitor and evaluate performance and impact
2. Address TB/HIV, MDR-TB, and the needs of poor and vulnerable populations
a. Scale up collaborative TB/HIV activities
D 5ECNGWRRTGXGPVKQPCPFOCPCIGOGPVQH/&46$
c. Address the needs of TB contacts, and of poor and vulnerable populations
3. Contribute to health system strengthening based on primary health care
C *GNRKORTQXGJGCNVJRQNKEKGUJWOCPTGUQWTEGFGXGNQROGPVßPCPEKPIUWRRNKGUUGTXKEGFGNKXGT[CPFKPHQTOCVKQP
b. Strengthen infection control in health services, other congregate settings and households
c. Upgrade laboratory networks, and implement the Practical Approach to Lung Health
F #FCRVUWEEGUUHWNCRRTQCEJGUHTQOQVJGTßGNFUCPFUGEVQTUCPFHQUVGTCEVKQPQPVJGUQEKCNFGVGTOKPCPVUQHJGCNVJ
4. Engage all care providers
a. Involve all public, voluntary, corporate and private providers through public–private mix approaches
b. Promote use of the International Standards for Tuberculosis Care
5. Empower people with TB, and communities through partnership
C 2WTUWGCFXQECE[EQOOWPKECVKQPCPFUQEKCNOQDKNK\CVKQP
b. Foster community participation in TB care, prevention and health promotion
c. Promote use of the Patients’ Charter for Tuberculosis Care
6. Enable and promote research
a. Conduct programme-based operational research
b. Advocate for and participate in research to develop new diagnostics, drugs and vaccines

3
GLOBAL TUBERCULOSIS REPORT 2013
of TB patients who are successfully treated). e Stop TB
Partnership adopted the MDG target and in addition set
global targets to halve TB prevalence and death rates by
2015 compared with their levels in 1990. e scale at which
interventions included in the Stop TB Strategy need to be
implemented to achieve the 2015 targets for reductions in
disease burden, and the associated funding requirements,
have been described in Global Plans developed by the Stop
TB Partnership. e latest plan covers the period 2011–
2015 and has a price tag of US$47 billion.1
As the MDG target year of 2015 approaches, work on a
post-2015 development framework is assuming increasing
prominence. In June 2013, a high-level panel established
by the UN Secretary General to provide recommendations
about the content of a post-2015 development framework,
including possible goals and targets, submitted its report.2
One of the twelve proposed goals for 2030 is to “Ensure
healthy lives”, under which a suggested target is to “Reduce
the burden of disease from HIV/AIDS, TB, malaria, neglect-
ed tropical diseases and priority noncommunicable diseas-
es”. Important themes within the report are building on
the MDGs and equity, and for health speci cally the impor-
tance of steady progress towards universal health coverage
(UHC) is highlighted.
In line with the development of a post-2015 develop-
ment framework and in response to a request from Member
States, WHO began the process of developing a post-2015
global TB strategy in 2012. Following a series of consulta-
tions between June 2012 and July 2013, the draft strategy
includes the goal of ending the global TB epidemic by 2035,
with corresponding global targets for major reductions
in TB cases and deaths by 2035 and milestones for 2020,
2025 and 2030. Achieving the proposed targets is based on
three strategic pillars: integrated, patient-centred TB care
1 The Global Plan to Stop TB, 2011–2015. Geneva, World Health
Organization, 2010 (WHO/HTM/STB/2010.2). Available at
http://www.stoptb.org/assets/documents/global/plan/TB_
GlobalPlanToStopTB2011-2015.pdf
2 http://www.un.org/sg/management/beyond2015.shtml
and prevention; bold policies and supportive systems; and
intensi ed research and innovation. It is anticipated that
the strategy will be reviewed by the WHO Executive Board
in January 2014 and discussed at the World Health Assem-
bly in May 2014.
In the context of global TB strategies and targets, WHO
has published a global TB report every year since 1997
(Figure 1.1). e main aim of the report is to provide a
comprehensive and up-to-date assessment of the TB epi-
demic and progress in prevention, diagnosis and treatment
of the disease at global, regional and country levels, based
primarily on data that are reported by countries and terri-
tories to WHO in annual rounds of global TB data collec-
tion (Box 1.3). is 2013 global TB report is the eighteenth
in the series of annual reports, and uses data reported by a
total of 197 countries and territories including 178 Member
States that account for over 99% of the world’s estimated
cases of TB (Table 1.1). With just over two years remaining
before the end of 2015, a special feature of this 2013 global
report is that it is accompanied by a supplement focused on
the ‘Countdown to 2015’ (Box 1.4).
e main part of the report contains seven major chap-
ters. Each chapter is intended to stand alone, but links to
other chapters are highlighted where appropriate.
Chapter 2 contains the latest estimates of the burden of
disease caused by TB and assessment of progress towards
the 2015 targets at global, regional and country levels. Esti-
mates for women and children speci cally are given par-
ticular attention. Following new analytical and modelling
work in 2013, the chapter also contains new estimates of
the number of cases of and deaths from MDR-TB and of
HIV-related TB mortality. e latest status of e orts to
improve measurement of TB cases and deaths at country
level, with guidance and support from the WHO Global
Task Force on TB Impact Measurement, is described.
Chapter 3 presents data on the numbers of cases noti-
ed to NTPs and reported to WHO and their treatment
outcomes, including breakdowns of TB cases by type, sex
and age. Recent progress in increasing the reporting of
cases by private sector providers through engagement of
FIGURE 1.1
Seventeen annual WHO global TB reports, 1997–2012
1997: First report:
epidemiology and
surveillance
2002: Added fi nancing and
strategy for 22 high-burden
countries (HBCs)
July 2009: Online data collection introduced
December 2009: Short update to 2009 report in transition
to earlier reporting of data and report publication
2003: Financing
and strategy
(all countries)
1310_0237_PM_003.indd 31310_0237_PM_003.indd 3 28/10/13 13:1528/10/13 13:15
4GLOBAL TUBERCULOSIS REPORT 2013
large hospitals in ve countries, the contribution of com-
munity health workers and volunteers to the referral of TB
cases and treatment support in 13 countries, and strikingly
high notication rates in prisons in parts of the European
Region, are highlighted.
Chapter 4 focuses on drug-resistant TB. e rst part
of the chapter covers progress in drug resistance surveil-
lance and associated estimates of the absolute number and
proportion of TB patients that have MDR-TB and exten-
sively drug-resistant TB (XDR-TB). e second part of the
chapter presents and discusses the latest data on the pro-
BOX 1.3
Data collected in the 2013 round of global TB data collection
&CVCYGTGTGSWGUVGFQPVJGHQNNQYKPIVQRKEU6$ECUGPQVKßECVKQPUCPFVTGCVOGPVQWVEQOGUKPENWFKPIDTGCMFQYPUD[6$ECUG
type, age, sex and HIV status; an overview of services for the diagnosis and treatment of TB; laboratory diagnostic services;
drug management; monitoring and evaluation; surveillance and surveys of drug-resistant TB; management of drug-resistant TB;
collaborative TB/HIV activities; TB infection control; engagement of all care providers in TB control; the budgets of national TB
EQPVTQNRTQITCOOGU062UKPCPFWVKNK\CVKQPQHIGPGTCNJGCNVJUGTXKEGUJQURKVCNK\CVKQPCPFQWVRCVKGPVXKUKVUFWTKPI
treatment; and NTP expenditures in 2012. A shortened version of the online questionnaire was used for high-income countries (that
KUEQWPVTKGUYKVJCITQUUPCVKQPCNKPEQOGRGTECRKVCQHÜ75aaKPCUFGßPGFD[VJG9QTNF$CPMa and/or low-incidence
EQWPVTKGUFGßPGFCUEQWPVTKGUYKVJCPKPEKFGPEGTCVGQHECUGURGTaRQRWNCVKQPQTECUGUKPVQVCN
Countries reported data using an online web-based system (www.stoptb.org/tme). The system was opened for reporting on 14
/CTEJYKVJCFGCFNKPGQH/C[HQTCNN9*1TGIKQPUGZEGRVVJG4GIKQPQHVJG#OGTKECU/C[CPFVJG'WTQRGCP4GIKQP
/C[%QWPVTKGUKPVJG'WTQRGCP7PKQPUWDOKVPQVKßECVKQPFCVCVQCU[UVGOOCPCIGFD[VJG'WTQRGCP%GPVTGHQT&KUGCUG2TGXGPVKQP
and Control (ECDC). Data from the ECDC system were uploaded into the WHO online system.
Data were reviewed, and followed up with countries where appropriate, by a team of reviewers from WHO (headquarters and
TGIKQPCNQHßEGUCPFVJG)NQDCN(WPFVQ(KIJV#+&56WDGTEWNQUKUCPF/CNCTKCVJG)NQDCN(WPF8CNKFCVKQPQHFCVCD[TGURQPFGPVU
was also encouraged via a series of in-built, real-time checks of submitted data as well as a summary report of apparent
inconsistencies or inaccuracies (this report can be generated at any time within the online system). Following corrections and
updates by countries, the data used for the main part of this report were the data available in July 2013. Annex 4 was produced on
1 October, by which time additional data had been reported by a few European countries.b
Besides the data reported through the standard TB questionnaire, data about screening for TB among people living with HIV and
RTQXKUKQPQHKUQPKC\KFRTGXGPVKXGVJGTCR[+26VQVJQUGYKVJQWVCEVKXG6$YGTGEQNNGEVGFD[VJG*+8FGRCTVOGPVKP9*1CPFVJG,QKPV
United Nations Programme on HIV/AIDS (UNAIDS). The data were jointly validated and imported into the global TB database.
a. JVVRFCVCYQTNFDCPMQTICDQWVEQWPVT[ENCUUKßECVKQPU
b. For this reason, there may be slight discrepancies between the main part of the report and Annex 4.
TABLE 1.1
Reporting of data in the 2013 round of global TB data collection
9*14')+10145'61(%17064+'5
%17064+'5#0&6'44+614+'5 /'/$'456#6'5
07/$'4 07/$'46*#64'2146'&# 07/$'4 07/$'46*#64'2146'&#
#HTKECP4GIKQP 46 45 46 45
'CUVGTP/GFKVGTTCPGCP4GIKQP 23 23 22 22
'WTQRGCP4GIKQPa54 42 53 41
4GIKQPQHVJG#OGTKECU 46 46 35 35
5QWVJ'CUV#UKC4GIKQP 11 11 11 11
9GUVGTP2CEKßE4GIKQP 36 30 27 24
High-burden countries (HBCs)b22 22 22 22
World 216 197 194 178
a Countries that did not report by the deadlines were mostly low-incidence countries in Western Europe.
b 6JG*$%UCTG#HIJCPKUVCP$CPINCFGUJ$TC\KN%CODQFKC%JKPCVJG&GOQETCVKE4GRWDNKEQHVJG%QPIQ'VJKQRKC+PFKC+PFQPGUKC-GP[C/Q\CODKSWG/[CPOCT0KIGTKC
2CMKUVCPVJG2JKNKRRKPGUVJG4WUUKCP(GFGTCVKQP5QWVJ#HTKEC6JCKNCPF7ICPFCVJG7PKVGF4GRWDNKEQH6CP\CPKC8KGV0COCPF<KODCDYG
grammatic response to MDR-TB, including the coverage of
testing for drug resistance among new and previously treat-
ed TB patients, the number of cases detected with MDR-
TB and enrolled on treatment, and treatment outcomes.
Chapter 5, on TB diagnostics and laboratory strength-
ening, covers three topics. ese are policy developments
between mid-2012 and mid-2013, the status of laboratory
capacity and incorporation of WHO guidance into national
policy in 2012, and recent progress in strengthening labo-
ratories and associated diagnostic capacity. e latest data
on the roll out of the rapid molecular test Xpert MTB/RIF
5GLOBAL TUBERCULOSIS REPORT 2013
BOX 1.4
Special supplement on the Countdown to 2015
The MDGs were established by the UN at the turn of the
21st century, with targets set for 2015 (www.un.org/
millenniumgoals). Designed to drive progress worldwide
and endorsed by all countries, the targets have been the
focus of international and national development efforts
for more than a decade. TB was included as part of MDG
6. In addition to TB targets and indicators that are part of
the MDG framework, targets for the response needed to
CFFTGUUVJGURGEKßEEJCNNGPIGUQH/&46$CPFVJG6$*+8
co-epidemic have been set for 2015 in the Global Plan to
Stop TB 2011–2015.
With just over two years remaining before the target dead-
line of the end of 2015, this 2013 global TB report is accom-
panied by a special supplement called Countdown to 2015.
The supplement provides an overview of progress towards
the 2015 targets set within the MDG framework and for the
TGURQPUGVQ6$*+8CPF/&46$URGEKßECNN[CPFVJGVQR
priority actions needed to either move beyond or accelerate
towards these targets. Snapshots are provided globally,
TGIKQPCNN[CPFHQTVJG*$%UVJCVJCXGCDQWVQHVJG
world’s TB cases and that have received greatest attention at
the global level since 2000. The snapshots are based on the
data presented in the main chapters of the report and the
annexes, complemented by recommendations from recent
programme reviews, published literature, and discussions
with experts at global, regional and national levels.
since it was recommended in 2010 and two multinational
projects (EXPAND-TB and TBXpert) are included.
Chapter 6 contains the most recent data on progress
in implementing collaborative TB/HIV activities to joint-
ly address the epidemics of TB and HIV. ese include HIV
testing for TB patients, provision of antiretroviral therapy
(ART) to HIV-positive TB patients, intensied screening
for TB among people living with HIV and treatment for
those without active TB with IPT.
Chapter 7 assesses nancing for TB care and control.
Funding requirements for a full response to the global TB
epidemic up to 2015, which were updated in early 2013 as
part of preparatory work undertaken to inform the replen-
ishment of the Global Fund, are presented rst. Key nd-
ings from a study of long-term trends (2002–2011) using
data compiled in the WHO annual rounds of data collection
and recently published in e Lancet Global Health are then
summarized, followed by a detailed analysis of new data
reported in 2013.
Chapter 8 discusses research and development for new
TB diagnostics, drugs and vaccines. After years of stagna-
tion, considerable progress has occurred in the past decade
and the development pipelines as of mid-2013 are described
and discussed.
e report also has four annexes. Annex 1 explains the
methods used to produce estimates of the burden of disease
caused by TB. Annex 2 contains country proles for the 22
HBCs that collectively account for about 80% of the world’s
TB cases (proles for all countries are available online1).
Annex 3 contains regional proles. Annex 4 consists of
summary tables that provide data on key indicators for the
world, the six WHO regions and individual countries.
1 www.who.int/tb/data

6GLOBAL TUBERCULOSIS REPORT 2013
%*#26'4
The burden of disease caused by TB
e burden of disease caused by TB can be mea-
sured in terms of incidence (dened as the num-
ber of new and relapse cases of TB arising in a
given time period, usually one year), prevalence
(dened as the number of cases of TB at a giv-
en point in time) and mortality (dened as the
number of deaths caused by TB in a given time
period, usually one year).
is chapter presents estimates of TB inci-
dence, prevalence and mortality (absolute num-
bers and rates) between 1990 and 2012 and (for
prevalence and mortality) forecasts up to 2015
(in sections 2.1–2.3). ese data are used to
assess progress towards achieving the global
targets for reductions in TB disease burden set
for 2015: that incidence should be falling (MDG
Target 6.c) and that prevalence and death rates
should be halved by 2015 compared with 1990
(Box 1.2 in Chapter 1). Key aspects of the meth-
ods used to produce the estimates are provided
at the beginning of each section.1 Estimates of
the number of incident TB cases among people
living with HIV, the number of incident cases
of MDR-TB, mortality due to MDR-TB and TB
deaths disaggregated by HIV status are includ-
ed in the relevant sections. Estimates are pre-
sented globally, for the six WHO Regions, and
at country level with particular focus on the 22
HBCs. In response to increasing demand and
global attention, special consideration is given
to estimates of TB disease burden among wom-
en and children. Updates to data sources and
methods used to produce estimates of TB dis-
ease burden compared with those published in
2012 are highlighted in Box 2.1.
ere is uncertainty in all estimates of the
burden of disease caused by TB. Section 2.4
proles eorts to improve measurement of this
burden under the umbrella of the WHO Glob-
al Task Force on TB Impact Measurement. e
recent and unprecedented progress in imple-
menting national TB prevalence surveys is
summarized and expanding eorts to strength-
en surveillance of cases and deaths via noti-
cation and vital registration (VR) systems are
described.
KEY FACTS AND MESSAGES
■ The global burden of TB remains enormous. In 2012, there were an
GUVKOCVGFOKNNKQPKPEKFGPVECUGUQH6$CPFOKNNKQPRGQRNGFKGF
HTQOVJGFKUGCUGaFGCVJUCOQPIRGQRNGYJQYGTG*+8PGICVKXG
CPFaCOQPIRGQRNGYJQYGTG*+8RQUKVKXG#OQPIVJGUGFGCVJU
VJGTGYGTGCPGUVKOCVGFaHTQO/&46$CTGNCVKXGN[JKIJVQVCN
EQORCTGFYKVJaKPEKFGPVECUGUQH/&46$
■ Although the number of TB cases and deaths remains unnecessarily
large for a mostly curable disease, there has been major progress towards
global targets for reductions in the burden of disease. The 2015 MDG
target of halting and reversing TB incidence has been achieved, with
TB incidence falling globally for several years (2% per year in 2012).
Globally, the TB mortality rate has fallen by 45% since 1990 and the
Stop TB Partnership target of a 50% reduction by 2015 is within reach.
Mortality and incidence rates are falling in all six WHO regions and in
OQUVQHVJG*$%UVJCVCEEQWPVHQTQXGTQHVJGYQTNFÂU6$ECUGU
■ 6JKUKUVJGßTUV[GCTKPYJKEJGUVKOCVGUQH6$FGCVJUCOQPI*+8
positive people were produced using the UNAIDS Spectrum model,
leading to revisions to previously published estimates for the period
1990–2011. The estimated percentage of TB cases living with HIV
remains unchanged, at 13% globally in 2012.
■ Although most TB cases and deaths occur among men, the burden of
FKUGCUGKUJKIJCOQPIYQOGP+PCPGUVKOCVGFaYQOGP
FKGFHTQO6$aCOQPI*+8PGICVKXGYQOGPCPFaCOQPI
*+8RQUKVKXGYQOGP6JGTGYGTGCNUQCPGUVKOCVGFa6$FGCVJU
among HIV-negative children (estimates of HIV-associated mortality are
not yet available).
■ 6JG5QWVJ'CUV#UKCCPF9GUVGTP2CEKßE4GIKQPUEQNNGEVKXGN[
CEEQWPVGFHQTQHVJGYQTNFÂU6$ECUGUKP6JG#HTKECP4GIKQP
had approximately one quarter of the world’s cases, and the highest
rates of cases and deaths relative to population (255 incident cases per
aQPCXGTCIGOQTGVJCPFQWDNGVJGINQDCNCXGTCIGQH+PFKC
and China had the largest number of cases (26% and 12% of the global
VQVCNTGURGEVKXGN[5QWVJ#HTKECCPF5YC\KNCPFJCFVJGJKIJGUVKPEKFGPEG
rate per capita (about 1 new case for every 100 people each year).
■ The quality and coverage of data available to estimate TB disease
burden continues to improve. In 2012, data from vital registration
systems were used to estimate TB mortality in 121 countries (up from
EQWPVTKGUKPVJGTGJCUDGGPWPRTGEGFGPVGFRTQITGUUKPVJG
KORNGOGPVCVKQPQHPCVKQPCN6$RTGXCNGPEGUWTXG[UUKPEGCPFGHHQTVU
to improve the monitoring of TB incidence by strengthening routine
health information systems and implementing inventory studies to
measure under-reporting of diagnosed cases are expanding.
■ Five national TB prevalence surveys were implemented in 2012 (in
VJG)CODKC0KIGTKC4YCPFCVJG7PKVGF4GRWDNKEQH6CP\CPKCCPF
6JCKNCPFCPFCHWTVJGTßXGYKNNUVCTVQTDGEQORNGVGFKPKP)JCPC
+PFQPGUKC/CNCYK5WFCPCPF<CODKC6JGUGUWTXG[URTQXKFGCFKTGEV
OGCUWTGQHFKUGCUGDWTFGPQHVGPHQTVJGßTUVVKOGCPFYKNNDGWUGFVQ
WRFCVGGUVKOCVGUQHFKUGCUGDWTFGPQPEGTGUWNVUCTGßPCNK\GF6JG[CNUQ
provide rich data to inform programme policy and strategy.
1 A detailed description is provided in Annex 1.

7GLOBAL TUBERCULOSIS REPORT 2013
BOX 2.1
Updates to estimates of TB disease burden in this report and updates that are anticipated in the near future
Each year, new data become available for the estimation of TB disease burden. Periodically, new approaches to the use of
CXCKNCDNGFCVCCTGFGXGNQRGF6JKUDQZRTQXKFGUCUWOOCT[QHWRFCVGUVJCVYGTGOCFGKP7RFCVGUHQTURGEKßEEQWPVTKGU
VJCVCTGGZRGEVGFKPVJGPGCTHWVWTGRGPFKPIVJGßPCNK\CVKQPQHCPCN[UGUQHFCVCHTQOTGEGPVN[EQORNGVGFRTGXCNGPEG
surveys, are also highlighted.
UPDATES IN THIS REPORT
1. TB/HIV burden estimates
+PCPFHQTVJGßTUVVKOGGUVKOCVGU
of TB incidence among people living
with HIV and TB mortality among HIV-
positive people were generated using
the Spectrum software programme.a
Spectrum has been used for more than
a decade to produce estimates of the
burden of disease caused by HIV, to build
projections about the future course of the
HIV epidemic and to assess the potential
impact of interventions. A TB module was
developed in 2012 and 2013 through
a collaboration between the Futures
Institute, the TB Modelling and Analysis
Consortium (TB-MAC), UNAIDS and WHO.
It was initially tested in two regional
workshops held in Johannesburg, South
Africa (in March 2013) and subsequently in
a workshop for countries in western Africa.
The mathematical methods implemented
in Spectrum as well as the input data are
described in Annex 1. It is anticipated
that the TB module will be extended to
include projections of the future course of
the TB epidemic and the potential impact
of selected interventions, building on
existing estimates of TB disease burden
generated by WHO.
The updated estimates of TB
incidence among people living with HIV
published in this report are generally
very consistent with previously published
estimates, especially for countries with a
IGPGTCNK\GF*+8GRKFGOKECPFUVTQPI6$
HIV surveillance systems. The updated
time series of mortality estimates at global
NGXGNCPFHQTVJG#HTKECP4GIKQPKPFKECVG
a lower level of TB mortality among HIV-
positive people compared with estimates
published in 2012. As a result of the use of
5RGEVTWOEQWPVT[URGEKßEGUVKOCVGUQH6$
mortality among HIV-positive people that
are fully consistent with overall estimates
QH*+8OQTVCNKV[CTGCXCKNCDNGHQTVJGßTUV
time. These are shown in Annex 2 and in
QPNKPGEQWPVT[RTQßNGU
2. MDR-TB mortality and incidence
'UVKOCVGUQH/&46$OQTVCNKV[CPF
KPEKFGPEGYGTGNCUVRTQFWEGFKPCPF
published in a 2010 WHO report on the
/&46$GRKFGOKE#U[UVGOCVKENKVGTCVWTG
review of evidence about mortality
CUUQEKCVGFYKVJ/&46$YCUEQOOKUUKQPGF
by WHO in 2013. The results have been
used to produce global estimates of
/&46$KPEKFGPEGCPFOQTVCNKV[KP
6JGGUVKOCVGQHOQTVCNKV[FWGVQ/&46$
is slightly higher than before, but the
uncertainty interval greatly overlaps the
RTGXKQWUQPG6JGGUVKOCVGQH/&4
TB incidence is similar to the previous
estimate.
3. Newly reported data
There are relatively small changes to
estimates of TB incidence, mortality
and prevalence for many countries that
TGàGEVXKVCNTGIKUVTCVKQPFCVCTGRQTVGF
to WHO between mid-2012 and mid-
2013, updated WHO estimates of the
overall number of deaths (that provide
overall mortality envelopes), updates to
estimates of the burden of HIV-associated
6$CPFPGY6$PQVKßECVKQPFCVCKPENWFKPI
corrections made to historical data. In
most instances, changes are well within
the uncertainty intervals of previously
published estimates of TB burden and time
trends are generally consistent. Newly-
reported data are the reason for small
changes to estimates of the number of TB
deaths among women and children.
4. In-depth epidemiological reviews
In January 2013, estimates of TB burden
for Viet Nam were updated in close
consultation with the NTP and other
stakeholders. These resulted in changes
to estimates of the level of and trends in
TB incidence, prevalence and mortality
compared with those published in the
2012 global TB report. Updates drew on
new analyses from prevalence survey data,
GXKFGPEGCDQWVVJGKPàWGPEGQPVTGPFUKP
ECUGPQVKßECVKQPUQHKPETGCUGFTGRQTVKPIVQ
the NTP of cases diagnosed in the private
sector and prisons and new analyses of
DTQCFGTKPàWGPEGUQP6$FKUGCUGDWTFGP
such as economic growth, health system
performance and the coverage of health
insurance.
5. Inclusion of newly reported cases
without documented treatment
history in incidence estimates
+PRTGXKQWU[GCTUPQVKßGF6$RCVKGPVU
without any reported treatment history
were not considered as incident cases
(incident cases were the sum of new and
TGNCRUGECUGU+PVJKUTGRQTVPQVKßGFECUGU
for which the treatment history is unknown
are considered to be incident cases.
6JKUEJCPIGKULWUVKßGFHQTVYQTGCUQPU
ßTUVKPEQWPVTKGUHCEKPIRTQDNGOUYKVJ
incomplete documentation of treatment
history, the vast majority of such cases are
ßTUVGRKUQFGUQTTGNCRUGGRKUQFGUUGEQPF
WHO received several requests from NTPs
(or equivalent) to include all patients with
no documented treatment history in the
count of new and relapse episodes to avoid
understating the true burden of TB. This
change affects relatively few countries,
most of which are in western Europe.
UPDATES ANTICIPATED IN
THE NEAR FUTURE
Updates to estimates of disease burden
are expected in several countries that
have recently completed or will soon
complete national TB prevalence surveys.
6JGUGKPENWFGßXG*$%U+PFQPGUKC
Nigeria, Pakistan, Thailand and the United
4GRWDNKEQH6CP\CPKC#FFKVKQPCNEQWPVTKGU
KPENWFGVJG)CODKCCPF4YCPFCDQVJ
of which completed surveys in 2012,
and Ghana where a survey began in
March 2013. In addition to a prevalence
survey, an inventory study to estimate TB
underreporting was completed in Pakistan
in 2012 (see also section 2.4) and an
in-depth epidemiological review was
conducted in Thailand in August 2013.
A workshop for the six countries that had
completed surveys by July 2013
(i.e. the Gambia, Nigeria, Pakistan,
4YCPFC6JCKNCPFCPF7PKVGF4GRWDNKE
QH6CP\CPKCCUYGNNCUVJGKTVGEJPKECN
partners will be held at WHO headquarters
in November 2013, to conduct and
complete analyses of survey data.
Following this workshop, updates to
estimates of TB disease burden will be
possible. These updates will be made
CXCKNCDNGKPQPNKPGEQWPVT[RTQßNGUCPF
associated data sets.
In 2014, a thorough review of the
current epidemiological and modelling
methods used to estimate TB disease
burden will be conducted by the
WHO Global Task Force on TB Impact
Measurement. The recommendations may
result in some further updates in the 2014
global TB report.
a JVVRYYYHWVWTGUKPUVKVWVGQTIURGEVTWOCURZ
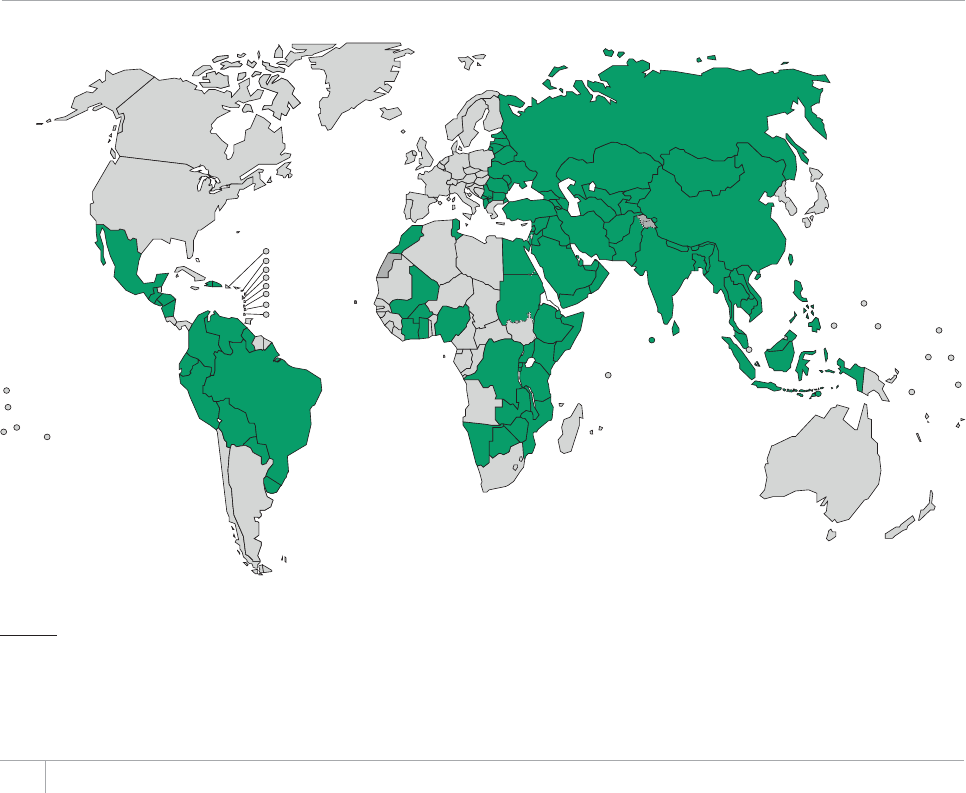
GLOBAL TUBERCULOSIS REPORT 2013
2.1 TB incidence
TB incidence has never been measured at national level
because this would require long-term studies among large
cohorts of people (hundreds of thousands) at high cost and
with challenging logistics. Notications of TB cases pro-
vide a good proxy indication of TB incidence in countries
that have both high-performance surveillance systems
(for example, there is little underreporting of diagnosed
cases) and where the quality of and access to health care
means that few cases (or a negligible number) are not diag-
nosed. In the large number of countries where these crite-
ria are not yet met, TB incidence can be estimated using
an inventory study (in which the level of underreporting
is assessed) combined with capture–recapture analysis to
estimate under-diagnosis, provided that certain assump-
tions are satised.1 To date, such studies have been under-
taken in only a few countries: examples include Egypt,
Iraq and Yemen (see section 2.4). e ultimate goal is to
directly measure TB incidence from TB notications in all
countries. is requires a combination of strengthened sur-
veillance, better quantication of underreporting (i.e. the
number of cases that are missed by surveillance systems)
and universal access to health care. A TB surveillance check-
list developed by the WHO Global Task Force on TB Impact
Measurement denes the standards that need to be met
for notication data to provide a direct measure of TB inci-
dence (further details in section 2.4).
For most countries, incidence estimates are currently
based on notication data combined with country consul-
tations in which in-depth analyses of the available surveil-
lance, survey and programmatic data are undertaken, and
expert opinion about the fraction of cases diagnosed but
not reported, or not diagnosed at all, is elicited and docu-
mented. e 96 countries (with 89% of estimated TB cases)
covered by such consultations since 2008 are shown in Fig-
ure 2.1. For remaining countries not covered in workshops
and in which notications do not provide a good proxy indi-
cation of TB incidence, estimates are based on extending
previously published time series, mortality data from VR
systems combined with evidence about the case fatality
rate, or ecological modelling (see Annex 1 for details).
In 2012, there were an estimated 8.6 million incident
cases of TB (range, 8.3million–9.0million) globally, equiv-
alent to 122 cases per 100000 population (Table 2.1, Table
2.2). e absolute number of incident cases is falling, albeit
slowly (Figure 2.2).
Most of the estimated number of cases in 2012 occurred
in Asia (58%) and the African Region (27%);2 smaller pro-
portions of cases occurred in the Eastern Mediterranean
Region (8%), the European Region (4%) and the Region of
FIGURE 2.1
Coverage of country consultations on estimates of TB disease burden, 2008–2013
1 An inventory study can be used to measure the number of cases that are diagnosed but not reported, but using results to estimate the total
number of incident cases using capture–recapture methods requires that certain conditions are met. ese are explained in a guide on inven-
tory studies recently published by WHO, which is available at: www.who.int/tb/publications/inventory_studies/en/index.html
2 Asia refers to the WHO Regions of South-East Asia and the Western Pacic.
9GLOBAL TUBERCULOSIS REPORT 2013
TABLE 2.1
Estimated epidemiological burden of TB, 2012. Numbers in thousands.a
POPULATION
/146#.+6;bHIV-POSITIVE
6$/146#.+6; 24'8#.'0%' INCIDENCE HIV-POSITIVE
INCIDENT TB CASES
BESTcLOW HIGH BEST LOW HIGH BEST LOW HIGH BEST LOW HIGH BEST LOW HIGH
Afghanistan 11 4.6 20 0.3 110 54 56 47 67 0.3 0.2 0.5
Bangladeshd154 695 70 29 130 0.1 670 340 1 100 350 290 410 0.2 0.2 0.3
$TC\KN 4.9 4.6 5.2 2.5 2.2 3 120 51 210 92 76 110 16 13 19
Cambodia 9.3 4.3 16 0.6 0.4 0.7 110 96 130 61 52 70 2.7 2.3 3.1
China 1 377 065 44 43 46 1.2 0.9 1.5 1 400 1 200 1 600 1 000 1 100 7.3 6.4
&4%QPIQ 65 705 36 16 64 6.3 5.5 200 620 210 190 250 16 14 19
Ethiopia 91 729 16 12 21 5.6 4.6 7.3 210 170 250 230 170 290 23 17 30
Indiae 270 170 390 42 37 1 900 3 900 2 200 2 000 2 400 130 120 140
Indonesia 67 30 120 2.1 37303501 200460 540 7.5 5.6 9.7
Kenya 9.5 5.4 15 7.7 6.6 130 71 210 120 110 120 45 44 47
/Q\CODKSWG 25 203 13 1 41 45 35 53 140 340 140 96 190 110
Myanmar 52 797 25 12 44 4.6 5.3 260 200 320 200 170 230 19 16 21
Nigeria 27 1.6 19 11 25 270 43 710 310 46 21
Pakistan 179 160 62 27 110 1.2 1.3 670 320 1 100 410 340 490 3.1 4.6
Philippines 96 707 23 22 25 0.1 0.1 450 390 500 260 210 310 0.5 0.4 0.6
4WUUKCP
Federation 143 170 19 20 1.5 2.2 170 73 320 130 110 150 9.3 7.9 11
South Africa 31 3.7 75 100 450 160 530 430 630 330 270 390
Thailand 9.2 17 2.2 1.9 110 47 190 66 95 12 10 14
Uganda 36 346 4.7 12 9.2 12 64 24 120 65 53 79 35 42
746CP\CPKC 6.1 3.2 9.9 7 45 140 79 74 32 30 34
Viet Nam 90 796 12 25 2.1 2.7 200 370 130 99 170 9.3 6.9 12
<KODCDYG 13 724 4.6 0.2 16 15 20 59 13 140 77 60 97 55 42 69
High-burden
countries 4 432 959 780 630 940 270 250 280 9 600 8 200 11 000 7 000 6 700 7 400 880 810 960
#(4 230 160 310 250 230 270 2 700 2 100 3 300 2 300 2 100 2 500 760 910
#/4 961 103 19 16 21 6.4 5.6 7.2 390 300 490 260 300 31 34
'/4 616 591 100 63 150 4.2 4.7 1 100 730 1 600 670 590 750 11 10 12
'74 904 540 36 35 36 3.9 3.4 4.4 510 650 360 340 390 19 17 21
5'#4 450 330 590 51 46 56 3 700 6 100 3 400 3 200 3 700 170 160
924 110 96 120 4.2 5.4 2 400 2 100 2 600 1 600 1 500 24 21 27
Global 7 053 684 940 790 1 100 320 300 340 12 000 11 000 13 000 8 600 8 300 9 000 1 100 1 000 1 200
a 0WODGTUHQTOQTVCNKV[RTGXCNGPEGCPFKPEKFGPEGUJQYPVQVYQUKIPKßECPVßIWTGU6QVCNU*$%UTGIKQPCNCPFINQDCNCTGEQORWVGFRTKQTVQTQWPFKPI
b /QTVCNKV[GZENWFGUFGCVJUCOQPI*+8RQUKVKXG6$ECUGU&GCVJUCOQPI*+8RQUKVKXG6$ECUGUCTGENCUUKßGFCU*+8FGCVJUCEEQTFKPIVQ+%&CPFCTGUJQYPUGRCTCVGN[KPVJKU
table.
c Best, low and high indicate the point estimate and lower and upper bounds of the 95% uncertainty interval.
d Estimates of TB disease burden have not been approved by the NTP in Bangladesh and a joint reassessment (by the NTP and WHO) will be undertaken following completion of
the national TB prevalence survey scheduled for 2014.
e 'UVKOCVGUHQT+PFKCJCXGPQV[GVDGGPQHßEKCNN[CRRTQXGFD[VJG/KPKUVT[QH*GCNVJ(COKN[9GNHCTG)QXGTPOGPVQH+PFKCCPFUJQWNFVJGTGHQTGDGEQPUKFGTGFRTQXKUKQPCN
10 GLOBAL TUBERCULOSIS REPORT 2013
TABLE 2.2
Estimated epidemiological burden of TB, 2012. 4CVGURGTRQRWNCVKQPGZEGRVYJGTGKPFKECVGFa
POPULATION
(THOUSANDS)
/146#.+6;b*+8215+6+8'6$/146#.+6; 24'8#.'0%' INCIDENCE *+824'8#.'0%'+0
INCIDENT TB CASES (%)
BEST LOW HIGH BEST LOW HIGH BEST LOW HIGH BEST LOW HIGH BEST LOW HIGH
Afghanistan 37 15 0.3 1.1 595 156 226 0.55 0.41
Bangladeshc154 695 45 19 434 721 225
$TC\KN 2.5 2.3 2.6 1.3 1.1 1.5 59 25 107 46 55 17.3 17.1 17.4
Cambodia 63 29 110 2.7 4.7 764 645 411 353 474 4.34 4.21 4.44
China 1 377 065 3.2 3.1 3.3 0.1 99 113 73 64 0.73 0.73 0.73
&4%QPIQ 65 705 54 24 97 9.7 12 576 301 327 375 7.66 7.65 7.66
Ethiopia 91 729 13 23 6.1 5 224 272 247 321 10.2 10.1 10.2
Indiad 22 14 32 3.4 3 3.9 230 155 319 176 159 193 5.95 5.93 5.97
Indonesia 27 12 0.9 0.7 1.2 297 144 506 153 220 1.65 1.65 2.33
Kenya 22 13 34 15 21 299 164 475 272 261
/Q\CODKSWG 25 203 53 3.9 163 177 209 553 111 1 340 552 753 59.7 59.6
Myanmar 52 797 23 7.3 10 377 616 377 322 435 9.33 9.32 9.33
Nigeria 16 0.9 51 11 6.7 15 161 25 420 50 25.2 25.7
Pakistan 179 160 34 15 61 0.7 0.5 376 641 231 190 276 0.92 0.96
Philippines 96 707 24 22 26 0.1 0.1 461 405 520 265 219 316
4WUUKCP
Federation 143 170 13 13 14 1.2 1 1.5 121 51 221 91 77 106 7.14 7.03 7.25
South Africa 59 7 164 144 192 305 1 000 1 190 63.0 62.9 63.0
Thailand 14 25 3.3 2.9 4.2 159 71 119 142 15.2 15.2 15.3
Uganda 36 346 13 2.3 33 25 22 32 175 67 334 179 145 216 53.2 52.9 53.3
746CP\CPKC 13 21 15 12 17 176 95 165 154 175 41.2 41.2 41.3
Viet Nam 90 796 20 13 27 2.4 2 2.9 410 147 109 192 6.97 6.94 6.99
<KODCDYG 13 724 33 1.2 117 132 111 147 433 92 1 030 562 434 706 70.9 70.7 71.4
High-burden
countries 4 432 959 18 14 21 6 5.6 6.4 216 186 248 159 151 166 7.37 7.35 7.40
#(4 26 35 26 30 303 239 373 255 235 275 36.6 34.7
#/4 961 103 1.9 1.7 2.2 0.7 0.6 0.7 40 31 51 29 27 31 11.4 14.4
'/4 616 591 16 10 24 0.7 0.6 256 109 96 122 1.34 2.52
'74 904 540 3.9 3.9 4 0.4 0.4 0.5 56 42 72 40 43 5.26 6.93
5'#4 25 32 2.5 3.1 264 203 333 174 200 4.94 4.31 5.62
924 5.2 6.4 0.3 0.2 0.3 115 142 95 1.49 0.92
Global 7 053 684 13 11 16 4.6 4.3 4.8 169 149 190 122 117 127 12.8 11.6 14.0
a Best, low and high indicate the point estimate and lower and upper bounds of the 95% uncertainty interval.
b /QTVCNKV[GZENWFGUFGCVJUCOQPI*+8RQUKVKXG6$ECUGU&GCVJUCOQPI*+8RQUKVKXG6$ECUGUCTGENCUUKßGFCU*+8FGCVJUCEEQTFKPIVQ+%&CPFCTGUJQYPUGRCTCVGN[KPVJKU
table.
c Estimates of TB disease burden have not been approved by the NTP in Bangladesh and a joint reassessment (by the NTP and WHO) will be undertaken following completion of
the national TB prevalence survey scheduled for 2014.
d 'UVKOCVGUHQT+PFKCJCXGPQV[GVDGGPQHßEKCNN[CRRTQXGFD[VJG/KPKUVT[QH*GCNVJ(COKN[9GNHCTG)QXGTPOGPVQH+PFKCCPFUJQWNFVJGTGHQTGDGEQPUKFGTGFRTQXKUKQPCN
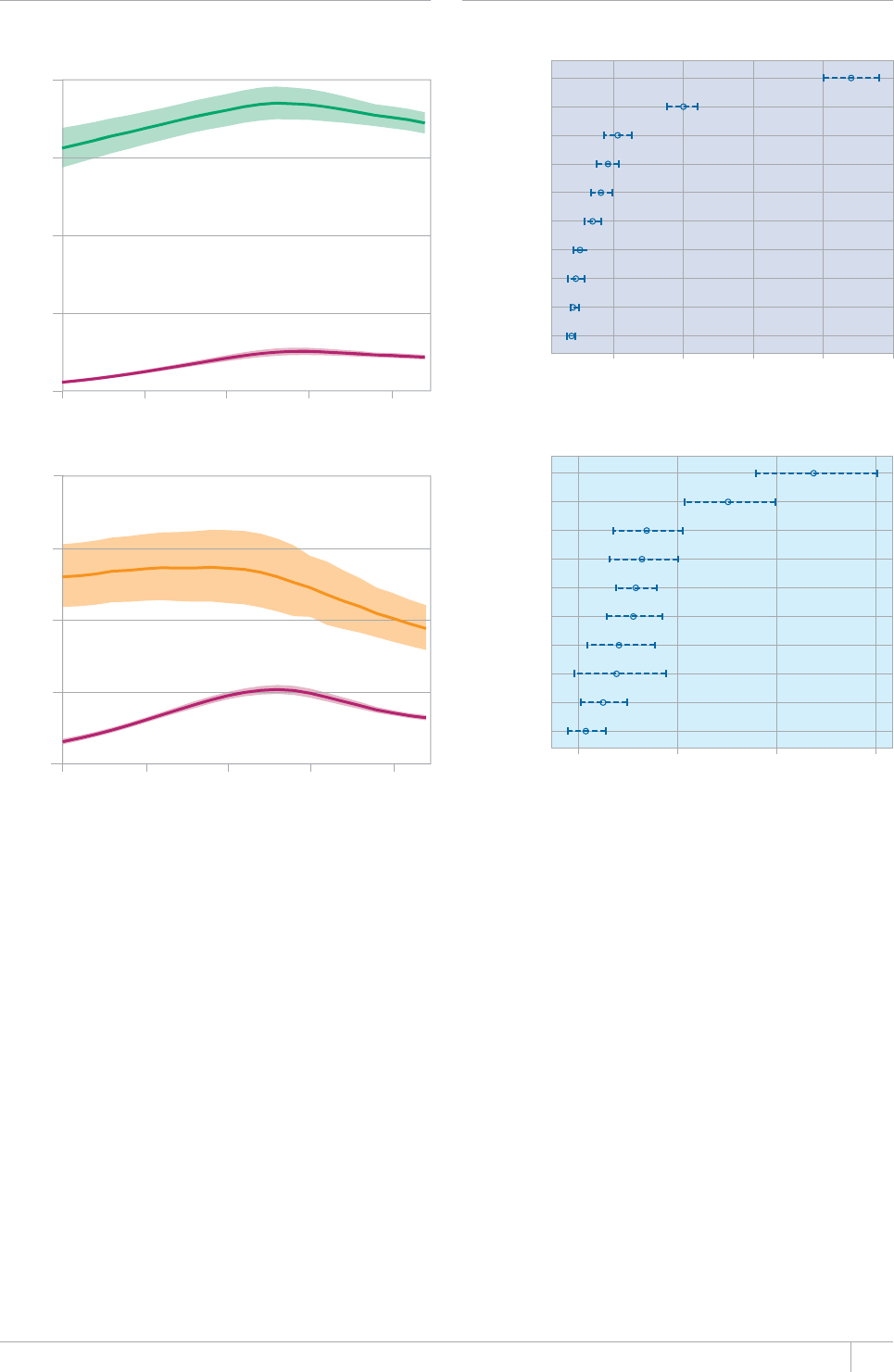
11GLOBAL TUBERCULOSIS REPORT 2013
FIGURE 2.2
Estimated absolute numbers of TB cases and deaths
(in millions), 1990–2012
a *+8CUUQEKCVGF6$FGCVJUCTGENCUUKßGFCU*+8FGCVJUCEEQTFKPIVQ+%&
0
2.5
5.0
7.5
10.0
1990 1995 2000 2005 2012
Millions
All TB cases
HIV-positive TB cases
TB incidence
0
0.5
1.0
1.5
2.0
1990 1995 2000 2005 2012
Millions
TB deaths among HIV-negative people
TB deaths among HIV-positive peoplea
TB deaths
the Americas (3%). e 22 HBCs that have been given high-
est priority at the global level since 2000 (listed in Table
2.1 and Table 2.2) accounted for 81% of all estimated inci-
dent cases worldwide. e ve countries with the largest
number of incident cases in 2012 were India (2.0 million–
2.4 million), China (0.9 million–1.1 million), South Africa
(0.4 million–0.6 million), Indonesia (0.4 million–0.5 mil-
lion) and Pakistan (0.3 million–0.5 million); these and the
other ve countries that make up the top ten in terms of
numbers of cases are highlighted in Figure 2.3. India and
China alone accounted for 26% and 12% of global cases,
respectively. Of the 8.6 million incident cases, an estimat-
ed 0.5 million were children and 2.9 million (range, 2.7–
3.1 million) occurred among women (Box 2.2).
e 8.6 million incident TB cases in 2012 included
1.0 million–1.2 million (12–14%) among people living with
HIV, with a best estimate of 1.1 million (13%) (Table 2.1,
Table 2.2). e proportion of TB cases co-infected with HIV
was highest in countries in the African Region (Figure 2.4).
Overall, 37% of TB cases were estimated to be co-infect-
ed with HIV in this region, which accounted for 75% of TB
cases among people living with HIV worldwide. In parts of
southern Africa, more than 50% of TB cases were co-infect-
ed with HIV (Figure 2.4).
Following a systematic review of evidence about mor-
tality caused by MDR-TB (
Box 2.3), global estimates of
the burden of MDR-TB were updated in 2013 (Box 2.1).
e best estimate is that there were 450000 (range,
300 000‒600 000) new cases of MDR-TB worldwide in
2012. is total includes cases of primary and acquired
MDR-TB.
e number of incident TB cases relative to population
(the incidence rate) varies widely among countries (Fig-
ure 2.5). e lowest rates are found predominantly in
high-income countries including most countries in west-
ern Europe, Canada, the United States of America, Japan,
Australia and New Zealand. In these countries, the inci-
dence rate per 100000 population is less than 10 cases per
FIGURE 2.3
Estimated TB incidence: top-ten countries, 2012
●
●
●
●
●
●
●
●
●
●
Myanmar
DR Congo
Ethiopia
Philippines
Bangladesh
Pakistan
Indonesia
South Africa
China
India
a. Incidence: absolute numbers
Millions
0.5 1.0 1.5 2.0 2.5
●
●
●
●
●
●
●
●
●
●
Rate per 100 000 population per year
Gabon
Timor-Leste
Mozambique
Zimbabwe
Djibouti
Lesotho
Namibia
Sierra Leone
South Africa
Swaziland
400 800 1200 1600
b. Incidence: rate per 100 000 population
12 GLOBAL TUBERCULOSIS REPORT 2013
BOX 2.2
The burden of TB disease among women and children
6JGDWTFGPQH6$OQTDKFKV[CPFOQTVCNKV[COQPIYQOGPFGßPGF
CUHGOCNGUCIGFÜ[GCTUCPFEJKNFTGPFGßPGFCURGQRNG
CIGF[GCTUKUNCTIGTVJCPQHVGPTGCNKUGF6JKUKUVJGUGEQPF
consecutive year in which the WHO global TB report highlights the
DWTFGPQHFKUGCUGCOQPIEJKNFTGPCPFHQTVJGßTUVVKOGKPENWFGU
estimates of the burden among women disaggregated by region
and HIV status.
There were an estimated 2.9 million new cases of TB and
aFGCVJUHTQOVJGFKUGCUGCOQPIYQOGPKP#OQPI
EJKNFTGPVJGTGYGTGCPGUVKOCVGFaPGYECUGUKPCPF
aFGCVJUCOQPIEJKNFTGPYJQYGTG*+8PGICVKXG/GVJQFU
used to produce these estimates and further details about results
are provided below. The estimates of TB morbidity and mortality
among children are slightly higher than those published in the
INQDCN6$TGRQTVTGàGEVKPIPGYUWTXGKNNCPEGFCVCVJCVUJQY
OQTG6$ECUGUDGKPIPQVKßGFCOQPIEJKNFTGPINQDCNN[CPFPGY84
data.
The burden of TB in women: estimates of TB incidence
and mortality, 2012
Incidence
4GIKQPCNGUVKOCVGUQHVJGYQOGPOGPTCVKQHQTPGYCNNECUGV[RGU
6$ECUGPQVKßECVKQPUKPYGTGIGPGTCVGFCPFCUUWOGFVQDG
the same as the ratio among incident TB cases in 2012 (see Annex
1 for further details). The resulting global and regional estimates
of incidence are shown in Table B2.2.1. Women account for
QHVJGVQVCNQHOKNNKQPKPEKFGPVECUGUKP6JG#HTKECP
CPF5QWVJ'CUV#UKCTGIKQPUCEEQWPVHQTQHVJGECUGUCOQPI
women.
TABLE B2.2.1
6QVCNPWODGTQHPGY6$PQVKßECVKQPUCNNECUGV[RGU
and estimated incident cases among women in 2012,
disaggregated by WHO region
WHO
REGION
NUMBER OF TB CASE
NOTIFICATIONS
ESTIMATED TB INCIDENCE
BEST ESTIMATE UNCERTAINTY INTERVAL
#(4 361 645 ¿
#/4 63 626 100 000 91 000–110 000
'/4 101 910 240 000–330 000
'74 79 279 120 000 110 000–130 000
5'#4 431 470 1 100 000 990 000–1 200 000
924 392 030 510 000 460 000–550 000
Global 1 429 960 2 900 000 2 700 000–3 100 000
Mortality
In total, there were an estimated 410 000 TB deaths among
women in 2012. This includes 250 000 (range, 210 000–290 000)
TB deaths among HIV-negative women (29% of all TB deaths
among HIV-negative adults) and 160 000 (range, 150 000–
170 000) HIV-associated TB deaths (50% of all HIV-associated
TB deaths). Newly reported data and a decrease in the overall TB
mortality envelope explain the decrease in the estimated number
QH6$FGCVJUCOQPIYQOGPEQORCTGFYKVJßIWTGUTGRQTVGFKP
previous years (see also Box 2.1).
/QTVCNKV[FCVCFKUCIITGICVGFD[CIGCPFUGZHTQO84U[UVGOU
were used to produce estimates of TB deaths among HIV-negative
CFWNVUHQTEQWPVTKGU84FCVCYGTGCXCKNCDNGHQTEQWPVTKGU
but for China, age and sex-disaggregated data were not available).
TB deaths were calculated for women and men, after adjustment
HQTKPEQORNGVGEQXGTCIGCPFKNNFGßPGFECWUGUUGGAnnex 1
HQTHWTVJGTFGVCKNU(QTEQWPVTKGUYKVJQWV84FCVCCPGEQNQIKECN
statistical model was used to predict the ratio of male to female
TB mortality. The model included a set of risk factors known to be
associated with TB mortality (GDP per capita, the percentage of
PGYECUGUYKVJ/&46$*+8RTGXCNGPEGKPVJGIGPGTCNRQRWNCVKQP
and the treatment success rate). Globally, there were 2.55 (range,
¿OCNGFGCVJUCOQPI*+8PGICVKXGCFWNVUHQTGXGT[
female death (Figure B2.2.14GIKQPCNFKHHGTGPEGUCTGGXKFGPV
(Table B2.2.2), with the African and South-East Asia regions
accounting for 69% of total deaths. The main limitation in the
OGVJQFUWUGFKUVJCVVJGEQWPVTKGUTGRQTVKPIWUCDNG84FCVC
were all middle- or high-income countries. Predictions for low-
income countries had to be extrapolated from these countries.
TB deaths among HIV-positive people were disaggregated
by sex using the assumption that the male to female sex ratio
is similar to the sex ratio of AIDS deaths estimated by UNAIDS.
Globally, the numbers of HIV-associated TB deaths were similar
among men and women (Figure B2.2.2). However, there were
striking regional variations (Table B2.2.2+PVJG#HTKECP4GIKQP
more deaths occurred among women than men, while in other
regions more deaths were estimated to have occurred among men.
TABLE B2.2.2
Estimated number of TB deaths among women in 2012,
disaggregated by WHO region
HIV-NEGATIVE HIV-POSITIVE
BEST
ESTIMATE
UNCERTAINTY
INTERVAL
BEST
ESTIMATE
UNCERTAINTY
INTERVAL
#(4 53 000–110 000 140 000 130 000–150 000
#/4 5 900 5 000–6 700 2 000 1 900–2 200
'/4 32 000 ¿ 1 400 1 300–1 600
'74 10 000 9 700–10 000 1 200 1 000–1 300
5'#4 93 000 65 000–120 000 16 000–20 000
924 26 000 24 000–29 000 1 200 1 000–1 300
Global 250 000 210 000–290 000 160 000 150 000–170 000
The burden of TB in children: estimates of TB
notißcations, incidence and mortality (among those HIV-
negative), 2012
6$PQVKßECVKQPUCPFKPEKFGPEG
6JGINQDCNPWODGTQHPGY6$ECUGPQVKßECVKQPUCOQPIEJKNFTGP
CIGF[GCTUKUGUVKOCVGFCVaKPTable
B2.2.3). This includes cases reported among children and an
estimate of the number of cases among children in countries that
FKFPQVTGRQTVPQVKßECVKQPUFKUCIITGICVGFD[CIG(QTEQWPVTKGU
that did not report age-disaggregated data (Figure B2.2.3), it
YCUCUUWOGFVJCVVJGTCVKQQHEJKNFVQCFWNVPQVKßGFECUGUYCUVJG
same (for each case type) as in those countries that did report
PQVKßECVKQPUFKUCIITGICVGFD[CIGCPCNVGTPCVKXGOGVJQFWUKPI
VJGCUUWORVKQPVJCVVJGTCVKQQHEJKNFJQQFVQCFWNVPQVKßECVKQP
rates was the same gave similar results). WHO does not request
age-disaggregated data for relapse cases or those reported as of
unknown case type, and the number of children in these categories
YCUCUUWOGFVQDG\GTQ
To estimate TB incidence among children, it was assumed that
the case detection rate for all ages at the global level in 2012 (best

13GLOBAL TUBERCULOSIS REPORT 2013
estimate 66%, range 64%–69%)
was the same for adults and
children. On this basis, TB incidence
among children is estimated at
530 000 (range, 510 000–550 000)
in 2012, equivalent to about 6%
QHVJGVQVCNPWODGTQHOKNNKQP
incident cases.
Limitations of the methods used
KPENWFG
The assumption that the case
detection rate is the same for
adults and children, in the
absence of any data on levels
of underreporting of diagnosed
cases for children and adults
separately.
The assumption that reported
cases were true cases of TB. Misdiagnosis is possible, especially
IKXGPVJGFKHßEWNVKGUQHFKCIPQUKPI6$KPEJKNFTGP
The proportion of cases among children may be different in
countries for which age-disaggregated data were not available.
However, reporting of cases disaggregated by age has been
improving and the number of countries not reporting age-
disaggregated data was relatively low in 2012.
Mortality among HIV-negative children
/QTVCNKV[FCVCTGRQTVGFVQ9*1HTQO84U[UVGOUVJCVYGTG
disaggregated by age were available for 120 countries. These data
were used to calculate TB death rates per 100 000 population
for children and adults, after adjustment for incomplete coverage
CPFKNNFGßPGFECWUGUUGGAnnex 1 for further details). For
EQWPVTKGUYKVJQWV84FCVCCPGEQNQIKECNUVCVKUVKECNOQFGNYCUWUGF
to predict the ratio of childhood to adult TB mortality rates. The
total number of deaths from TB among HIV-negative children was
estimated to be 74 000 (range, 59 000–90 000), equivalent to
CDQWVQHVJGVQVCNPWODGTQHa6$FGCVJUCOQPI*+8
negative people in 2012.
An estimate of TB mortality among HIV-positive children
KUPQVKPENWFGFKPVJKUTGRQTVFWGVQVJGFKHßEWNVKGUCTKUKPI
from the miscoding of HIV deaths as TB deaths. However, age
disaggregation of HIV-associated TB mortality will be one of the
future outcomes of the TB component of Spectrum (see Box 2.1).
Steps to improve estimation of
6$ECUGUCOQPIEJKNFTGPKPENWFG
a global consultation to further
develop analytical methods and
VQFGßPGCPFRTKQTKVK\GCEVKQPU
needed to obtain new data in
September 2013;
promotion of case-based
electronic recording and
reporting systems that facilitate
compilation and analysis of age-
disaggregated data;
nationwide inventory surveys
to measure underreporting of
childhood TB;
more contact-tracing studies and
the integration of TB activities
in maternal, newborn and child
JGCNVJUGTXKEGUVQßPFEJKNFJQQF
cases that might otherwise not
be diagnosed.
●
●
●
●
●
●
●
Global
WPR
SEAR
EUR
EMR
AMR
AFR
1234
Sex Ratio (M:F)
FIGURE B2.2.1
The male:female ratio for HIV-negative
6$FGCVJUCOQPICFWNVUCIGFÜ
[GCTUINQDCNN[CPFHQT9*1TGIKQPU
●
●
●
●
●
●
●
Global
WPR
SEAR
EUR
EMR
AMR
AFR
123
Sex Ratio (M:F)
FIGURE B2.2.2
The male:female ratio for HIV-
associated TB deaths among adults
CIGFÜ[GCTUINQDCNN[CPFHQT
WHO regions
Age disaggregation
All case types disaggregated
Only smear-positive cases disaggregated
No age disaggregation
No data reported
FIGURE B2.2.3
4GRQTVKPIQHPQVKßECVKQPFCVCFKUCIITGICVGFD[CIG
TABLE B2.2.3
New TB case notißcations in 2012, by case type and
age disaggregation
SMEAR-
POSITIVE
SMEAR-
NEGATIVEa
EXTRA-
PULMONARY
Total notißcations 1 935 971
Countries disaggregating by age 2 551 136 1 597 530
Countries not disaggregating
by age 17 653
VQVCNPQVKßECVKQPU
disaggregated) (99%)
Number of countries that report-
ed notißcations disaIIreIated by
aIe number of *BCsb
204 (22)
Total childhood notißcations from
countries disaIIreIatinI by aIe 163 477
Total estimated childhood
notißcations amonI all countries 349 000
a This includes reported cases for whom smear results were unknown or not done.
b #PCFFKVKQPCNPKPGEQWPVTKGUTGRQTVGF\GTQ6$ECUGUHQTCPFVJTGGEQWPVTKGU
had not reported data to WHO by July 2013.
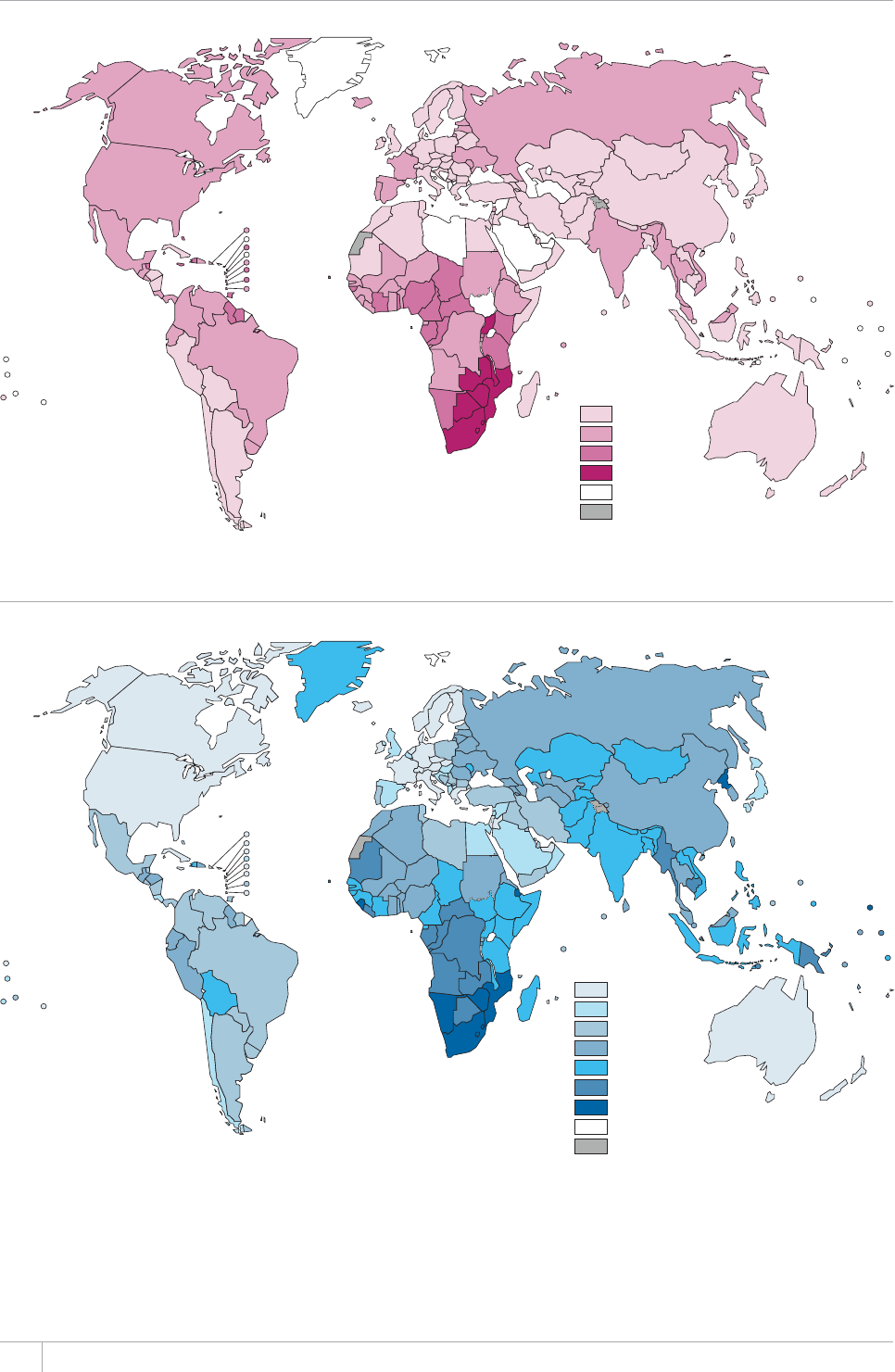
14 GLOBAL TUBERCULOSIS REPORT 2013
FIGURE 2.4
Estimated HIV prevalence in new TB cases, 2012
HIV prevalence
in new TB cases,
all ages (%)
0–4
5–19
20–49
≥50
No data
Not applicable
FIGURE 2.5
Estimated TB incidence rates, 2012
Estimated new TB
cases (all forms) per
100 000 population
per year
0–9.9
10–19
20–49
50–124
125–299
300–499
≥500
No data
Not applicable

15GLOBAL TUBERCULOSIS REPORT 2013
FIGURE 2.6
Global trends in estimated rates of TB incidence, prevalence and mortality. .GHV)NQDCNVTGPFUKPGUVKOCVGF
incidence rate including HIV-positive TB (green) and estimated incidence rate of HIV-positive TB (red%GPVTGCPFTKIJV
Trends in estimated TB prevalence and mortality rates 1990–2012 and forecast TB prevalence and mortality rates
¿6JGJQTK\QPVCNFCUJGFNKPGUTGRTGUGPVVJG5VQR6$2CTVPGTUJKRVCTIGVUQHCTGFWEVKQPKPRTGXCNGPEGCPF
mortality rates by 2015 compared with 1990. Shaded areas represent uncertainty bands. Mortality excludes TB deaths among
HIV-positive people.
Rate per 100 000 population per year
Incidence
1990 1995 2000 2005 2010
0
50
100
150
Prevalence
1990 1995 2000 2005 2010 2015
0
100
200
300
Rate per 100 000 population
Mortality
1990 1995 2000 2005 2010 2015
0
10
20
30
Rate per 100 000 population per year
FIGURE 2.7
Estimated TB incidence rates by WHO region, 1990–2012. 4GIKQPCNVTGPFUKPGUVKOCVGF6$KPEKFGPEGTCVGUgreen)
and estimated incidence rates of HIV-positive TB (red). Shaded areas represent uncertainty bands.
Rate per 100 000 population per year
1990 1995 2000 2005 2012 1990 1995 2000 2005 20121990 1995 2000 2005 2012
Africa The Americas Eastern Mediterranean
Europe South-East Asia Western Pacific
0
100
200
300
400
0
20
40
60
80
0
50
100
150
200
0
25
50
75
100
0
100
200
300
0
50
100
150
200
100000 population. Most countries in the Region of the
Americas have rates below 50 per 100000 population and
this is the region with the lowest burden of TB on average.
Most of the HBCs have rates of around 150‒300 cases per
100000 population (Table 2.2); HBCs with markedly lower
rates are Brazil and China, while rates are above 500 per
100000 population in Mozambique, South Africa and Zim-
babwe. Other countries in the top ten worldwide in terms of
incidence rates are mostly in Africa (Figure 2.3). In South
Africa and Swaziland, the best estimate is that at least 1 in
every 100 people (1000 or more per 100000 population)
develops TB each year.
Globally, the incidence rate was relatively stable from
1990 up to around 2001, and then started to fall (Figure
2.6), achieving the MDG target ahead of the 2015 deadline.
Between 2011 and 2012, the rate of decline was 2%. is
downward trend needs to be sustained to ensure that the
MDG target is met in 2015. Incidence rates are also declin-
16 GLOBAL TUBERCULOSIS REPORT 2013
ing in all of six WHO regions (Figure 2.7), fastest in the
European Region (6.5% per year) and slowest in the Eastern
Mediterranean and South-East Asia Regions (less than 1%
per year and 2% per year, respectively). Incidence rates have
been falling since the mid-1990s in the Eastern Mediterra-
nean Region and since around 2000 in the South-East Asia
Region; they peaked around 1997 in the European Region
and around 2002 in the African region, and have been fall-
ing since 1990 in the Region of the Americas and the West-
ern Pacic Region. e latest assessment for the 22 HBCs
suggests that incidence rates are falling in most countries
(Figure 2.8).
2.2 TB prevalence
In countries with a relatively high burden of TB (around
100 cases per 100000 population or more), the prevalence
of bacteriologically-conrmed pulmonary TB can be direct-
ly measured in nationwide population-based surveys using
sample sizes of around 50000 people. Survey results can be
FIGURE 2.8
Estimated TB incidence rates, 22 high-burden countries, 1990–2012. Trends in estimated TB incidence rates
(green) and estimated incidence rates of HIV-positive TB (red). Shaded areas represent uncertainty bands.
a Estimates of TB disease burden have not been approved by the national TB programme in Bangladesh and a joint reassessment will be undertaken following the completion
of the prevalence survey planned for 2014.
Rate per 100 000 population per year
0
100
200
300
0
100
200
300
400
500
0
250
500
750
1000
1250
0
50
100
150
200
0
100
200
300
400
0
250
500
750
1000
1250
0
250
500
750
1000
1250
0
50
100
150
200
250
0
250
500
750
1000
1250
0
100
200
300
0
200
400
600
0
200
400
600
0
100
200
300
400
0
200
400
600
0
200
400
600
800
0
100
200
300
100
200
300
0
100
200
300
400
0
100
200
300
400
0
50
100
150
0
250
500
750
1000
0
50
100
150
200
1990 1995 2000 2005 2012 1990 1995 2000 2005 2012
1990 1995 2000 2005 2012 1990 1995 2000 2005 2012 1990 1995 2000 2005 2012
Afghanistan BangladeshaBrazil Cambodia China
DR Congo Ethiopia India Indonesia Kenya
Mozambique Myanmar Nigeria Pakistan Philippines
Russian Federation South Africa Thailand Uganda UR Tanzania
Viet Nam Zimbabwe
0
1 TB prevalence surveys: a handbook. Geneva, World Health Organiza-
tion, 2011 (WHO/HTM/TB/2010.17). Available at www.who.int/
tb/advisory_bodies/impact_measurement_taskforce/resources_
documents/thelimebook/
used to produce a national estimate of TB prevalence that
includes all forms of TB. e cost of a survey usually rang-
es from US$1 to4 million, and comprehensive theoretical
and practical guidance on survey design, implementation,
analysis and reporting of results is available.1 Repeat sur-
veys conducted about every 10 years allow trends in disease
burden to be assessed. HBCs that have completed repeat
surveys in the last 10 years include Cambodia, China, the
Philippines and ailand, and repeat surveys are planned
in Myanmar and Viet Nam. Countries in which surveys
have been implemented or are planned in the near future
are shown in Figure 2.9. Between 2008 and 2017, an
unprecedented number of national TB prevalence surveys
have been or will be conducted (see also section 2.4).
In low- and medium-burden countries, sample sizes and
17GLOBAL TUBERCULOSIS REPORT 2013
costs become prohibitively large. If survey data are not
available, prevalence can be indirectly estimated as the
product of incidence and the average duration of disease,
but with considerable uncertainty (Annex 1). TB preva-
lence can be estimated only indirectly for most countries.
ere were an estimated 12 million prevalent cases
(range, 11million–13million) of TB in 2012 (Table 2.1),
equivalent to 169 cases per 100000 population (Table 2.2).
By 2012, the prevalence rate had fallen 37% globally since
1990. Current forecasts suggest that the Stop TB Partner-
ship target of halving TB prevalence by 2015 compared
with a baseline of 1990 will not be met worldwide (Figure
2.6). Regionally, prevalence rates are declining in all six
WHO regions (Figure 2.10). e Region of the Americas
halved the 1990 level of TB prevalence by around 2004,
well in advance of the target year of 2015, and the best
estimate suggests that the Western Pacic Region achieved
the 50% reduction target in 2012. Reaching the 50% reduc-
tion target by 2015 appears feasible in the South-East Asia
Region and also in the European Region with a relatively
small acceleration in the current rate of progress. e target
appears out of reach in the African and Eastern Mediterra-
nean Regions.
FIGURE 2.9
Countries in which national population-based surveys of the prevalence of TB disease have been
implemented using currently recommended screening and diagnostic methodsa since 1990 or are planned
in the near future: status in July 2013
a 5ETGGPKPIOGVJQFUKPENWFGßGNFEJGUV:TC[EWNVWTGKUWUGFVQEQPßTOFKCIPQUKU
b “National survey planned” means that a country has submitted at least a draft survey protocol and a budget to the WHO Global Task Force on TB Impact Measurement.
No national
survey planned
National survey
plannedb
National survey
ongoing
One national
survey completed
Repeat national survey
planned
≥1 repeat national survey
completed
Not applicable
2.3 TB mortality
TB mortality among HIV-negative people can be directly
measured using data from national VR systems, provided
that these systems have high coverage and causes of death
are accurately coded according to the latest revision of the
International classication of diseases (ICD-10). Sample VR
systems covering representative areas of the country (e.g.
as in China) provide an interim solution. Mortality surveys
can also be used to estimate deaths caused by TB. In 2012,
most countries with a high burden of TB lacked national or
sample VR systems and few had conducted mortality sur-
veys. In the absence of VR systems or mortality surveys, TB
mortality can be estimated as the product of TB incidence
and the case fatality rate, or from ecological modelling
based on mortality data from countries with VR systems.
TB mortality among HIV-positive people is hard to measure
even when VR systems are in place because deaths among
HIV-positive people are coded as HIV deaths and contribu-
tory causes (such as TB) are often not reliably recorded. For
this 2013 report, country-specic estimates of TB deaths
among HIV-positive people were produced for the rst time
using the Spectrum software that has been used for HIV
burden estimates for over a decade (Box 2.1).
Until 2008, WHO estimates of TB mortality used
VR data for only three countries. is was substantially
improved to 89 countries in 2009; however most of the
data were from countries in the European Region and the

GLOBAL TUBERCULOSIS REPORT 2013
FIGURE 2.10
Trends in estimated TB prevalence rates 1990–2012 and forecast TB prevalence rates 2013–2015, by WHO
region. 5JCFGFCTGCUTGRTGUGPVWPEGTVCKPV[DCPFU6JGJQTK\QPVCNFCUJGFNKPGUTGRTGUGPVVJG5VQR6$2CTVPGTUJKRVCTIGVQHC
50% reduction in the prevalence rate by 2015 compared with 1990. The other dashed lines show projections up to 2015.
Rate per 100 000 population
Africa The Americas Eastern Mediterranean
Europe South-East Asia Western Pacific
0
200
400
0
50
100
150
0
50
100
0
200
400
0
100
200
300
400
0
100
200
1990 1995 2000 2005 2010 2015 1990 1995 2000 2005 2010 2015 1990 1995 2000 2005 2010 2015
BOX 2.3
MDR-TB mortality – methods used to produce updated estimates
#URCTVQHCINQDCNTGRQTVQPVJG/&46$GRKFGOKECPFVJGINQDCNTGURQPUGKVYCUGUVKOCVGFVJCVVJGTGYGTGaFGCVJU
TCPIGa¿aHTQO/&46$KPa6JKUYCUVJGßTUVVKOG9*1RWDNKUJGFCINQDCNGUVKOCVGQH/&46$OQTVCNKV[CPF
given limitations in the available evidence, annual updates have not been attempted.
+PVJGQT[VJGPWODGTQHFGCVJUHTQO/&46$ECPDGGUVKOCVGFCUVJGRTQFWEVQHVQVCNFGCVJUHTQO6$VJGQXGTCNNRTQRQTVKQP
QH6$ECUGUVJCVJCXG/&46$CPFVJGTGNCVKXGTKUM44QHF[KPICOQPIRGQRNGYKVJ/&46$EQORCTGFYKVJVJQUGYKVJQWV
/&46$*QYGXGTYJKNGGUVKOCVGUQHVQVCN6$OQTVCNKV[CPFVJGRTGXCNGPEGQH/&46$JCXGDGGPCXCKNCDNGHQTUGXGTCN[GCTUHTQO84
FCVCKGHQTVQVCN6$FGCVJUCPFTGRTGUGPVCVKXGUWTXGKNNCPEGQTUWTXG[FCVCHQTVJGRTQRQTVKQPQHECUGUYKVJ/&46$CPGUVKOCVG
QHVJG44YCUPQV
+P9*1EQOOKUUKQPGFCU[UVGOCVKETGXKGYQHVJG44QHF[KPIHTQO/&46$EQORCTGFYKVJ6$6YGPV[ßXGUVWFKGUVJCV
KPENWFGFFCVCCDQWVOQTVCNKV[COQPIRCVKGPVUGPTQNNGFQPVTGCVOGPVHQT/&46$CPF6$YKVJQWV/&46$FWTKPICPFCHVGT
VTGCVOGPVYGTGKFGPVKßGF6JGUGCNNQYGFECNEWNCVKQPQHCINQDCNGUVKOCVGQHVJG44QHF[KPIHTQO/&46$TCPIG¿
6JGUVWFKGUJCFCDTQCFIGQITCRJKECNEQXGTCIGCPFKPENWFGFEQWPVTKGUYKVJDQVJJKIJCPFNQYDWTFGPUQH/&46$CPF*+8DWV
YGTGKPUWHßEKGPVVQGUVKOCVGTGIKQPURGEKßE44U
$CUGFQPVJGTGUWNVUQHVJGOGVCCPCN[UKUKVKUGUVKOCVGFVJCVINQDCNN[KPVJGTGYGTGaFGCVJUTCPIGa¿
aHTQO/&46$
a MultidruI- and eZtensiXely druI-resistant TB M:&4-TB 2010 Ilobal report on surXeillance and response (WHO/HTM/TB/2010.3). Geneva, World Health
1TICPK\CVKQP#XCKNCDNGCVJVVRYYYYJQKPVVDRWDNKECVKQPUGP
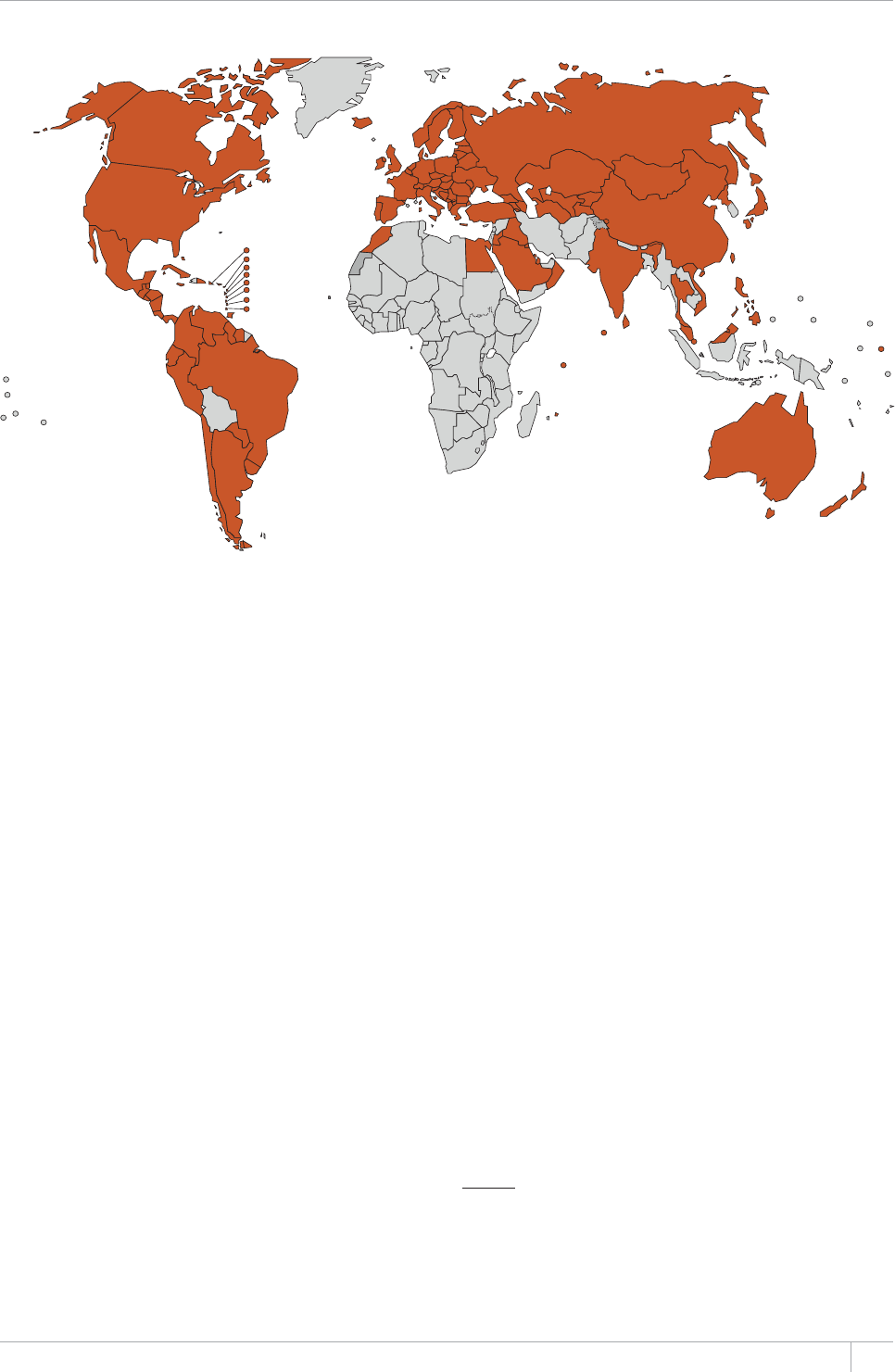
19GLOBAL TUBERCULOSIS REPORT 2013
Region of the Americas, which accounted for less than 10%
of the world’s TB cases. In 2011, the rst uses of sample
VR data from China and survey data from India enabled a
further major improvement to estimates of TB mortality.
For the current report, VR data of sucient coverage and
quality were available for 121 countries. Combined with
survey data from India and Viet Nam, this means that esti-
mates of TB mortality are based on direct measurements
of TB mortality in 123 countries (shown in Figure 2.11).
Collectively, these 123 countries account for 45% of the
estimated number of TB deaths globally. e parts of the
world where there are major gaps in the availability of VR
data are the African Region and parts of the South-East
Asia Region; in the latter, Indonesia is currently building a
sample VR system.
ere were an estimated 1.3 million TB deaths in 2012
(Table 2.1, Figure 2.2): 940000 among HIV-negative peo-
ple and 320 000 among HIV-positive people (TB deaths
among HIV-positive people are classied as HIV deaths in
ICD-10).1 ese deaths included 410000 among women and
74000 among children (Box 2.2). ere were approximate-
ly 170000 deaths from MDR-TB (range, 102000‒242000):
methods used to produce this new global estimate of MDR-
TB mortality are explained in Box 2.3.
Approximately 75% of total TB deaths occurred in the
African and South-East Asia Regions in 2012 (both includ-
ing and excluding TB deaths among HIV-positive people).
India and South Africa accounted for about one-third of
global TB deaths.
e number of TB deaths per 100000 population aver-
aged 13 globally in 2012 (
Table 2.2) and 17.6 when TB
deaths among HIV-positive people are included. ere is
considerable variation among countries (Figure 2.12),
ranging from under 1 TB death per 100 000 population
(examples include most countries in western Europe, Can-
ada, the United States of America, Australia and New Zea-
land) to more than 40 deaths per 100000 population in
much of the African Region as well as three HBCs in Asia
(Bangladesh, Cambodia and Myanmar).
Globally, mortality rates (excluding deaths among
HIV-positive people)2 have fallen by 45% since 1990; the
current forecast suggests that the Stop TB Partnership
target of a 50% reduction in TB mortality by 2015 com-
pared with a baseline of 1990 will be achieved (Figure 2.6).
Mortality rates are declining in all six WHO regions (Fig-
ure 2.13). e 2015 target has already been surpassed in
the Region of the Americas (since 2004) and the Western
Pacic Region (since 2002), and may have been reached in
the Eastern Mediterranean Region. Among the other three
regions, the South-East Asia Region appears best placed
to achieve the target. Mortality rates appear to be falling
in most of the 22 HBCs (Figure 2.14), although there is
considerable uncertainty about the level of and trends in
mortality in some countries, notably Mozambique, Nigeria,
South Africa and Zimbabwe.
FIGURE 2.11
Countries (in orange) for which TB mortality is estimated using measurements from vital registration (n=121)
systems and/or mortality surveys (n=2, India and Viet Nam)
1 International statistical classication of diseases and related health
problems, 10th revision (ICD-10), 2nd ed. Geneva, World Health
Organization, 2007.
2 Trends in TB mortality rates are restricted to TB deaths among
HIV-negative people, given that TB deaths among HIV-positive
people are classied as HIV deaths in ICD-10.
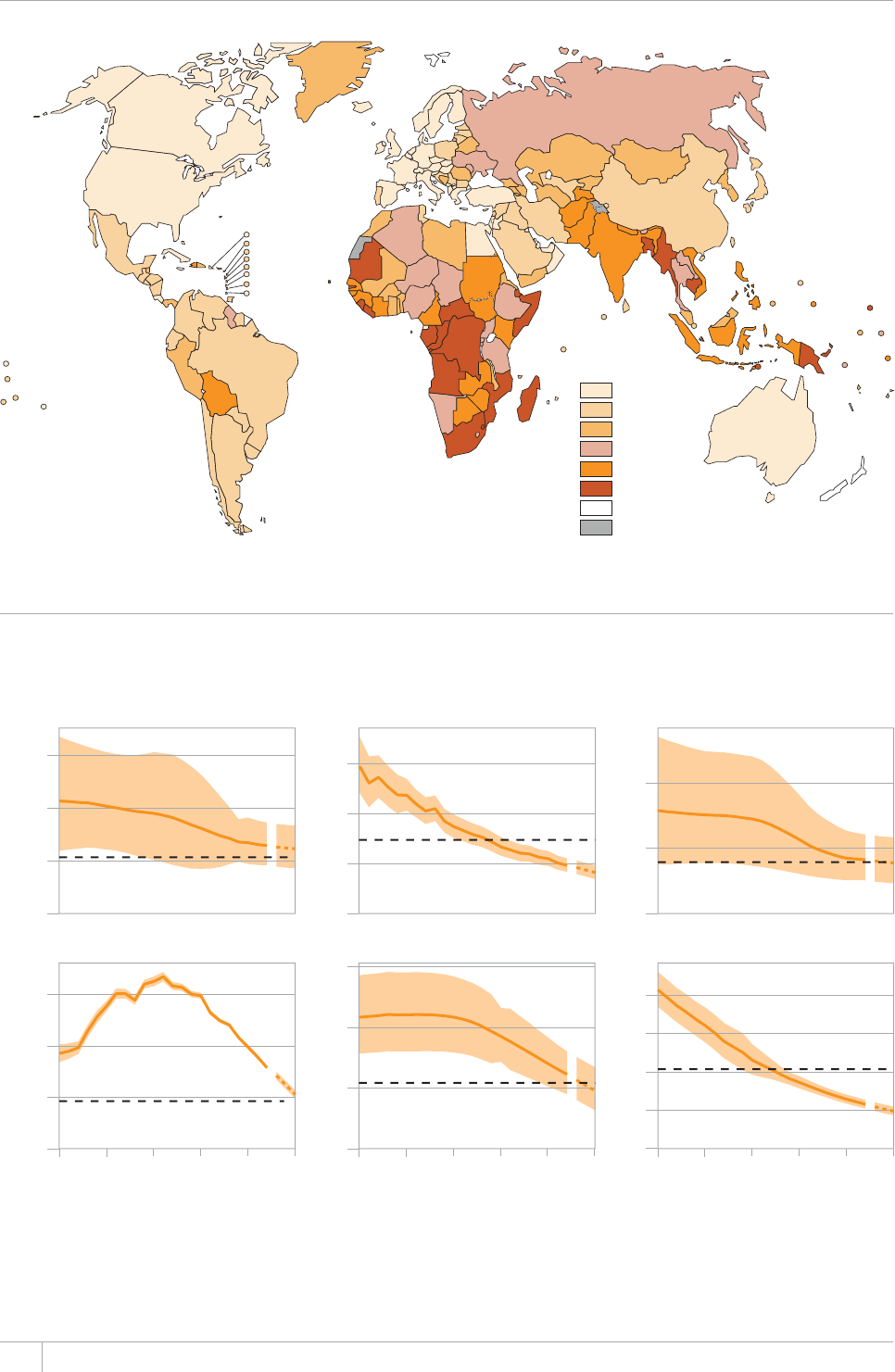
20 GLOBAL TUBERCULOSIS REPORT 2013
FIGURE 2.12
Estimated TB mortality rates excluding TB deaths among HIV-positive people, 2012
Estimated TB
deaths per
100 000 population
0–0.9
1–3.9
4–9.9
10–19
20–39
≥40
No data
Not applicable
FIGURE 2.13
Trends in estimated TB mortality rates 1990–2012 and forecast TB mortality rates 2013–2015, by WHO
region. Estimated TB mortality excludes TB deaths among HIV-positive people. Shaded areas represent uncertainty bands.a
6JGJQTK\QPVCNFCUJGFNKPGUTGRTGUGPVVJG5VQR6$2CTVPGTUJKRVCTIGVQHCTGFWEVKQPKPVJGOQTVCNKV[TCVGD[
compared with 1990. The other dashed lines show projections up to 2015.
a The width of an uncertainty band narrows as the proportion of regional mortality estimated using vital registration data increases or the quality and completeness of the vital
registration data improves.
0
20
40
60
0
2
4
6
0
20
40
0
2.5
5.0
7.5
0
20
40
60
0
5
10
15
20
1990 1995 2000 2005 2010 2015 1990 1995 2000 2005 2010 2015 1990 1995 2000 2005 2010 2015
Rate per 100 000 population per year
Africa The Americas Eastern Mediterranean
Europe South-East Asia Western Pacific
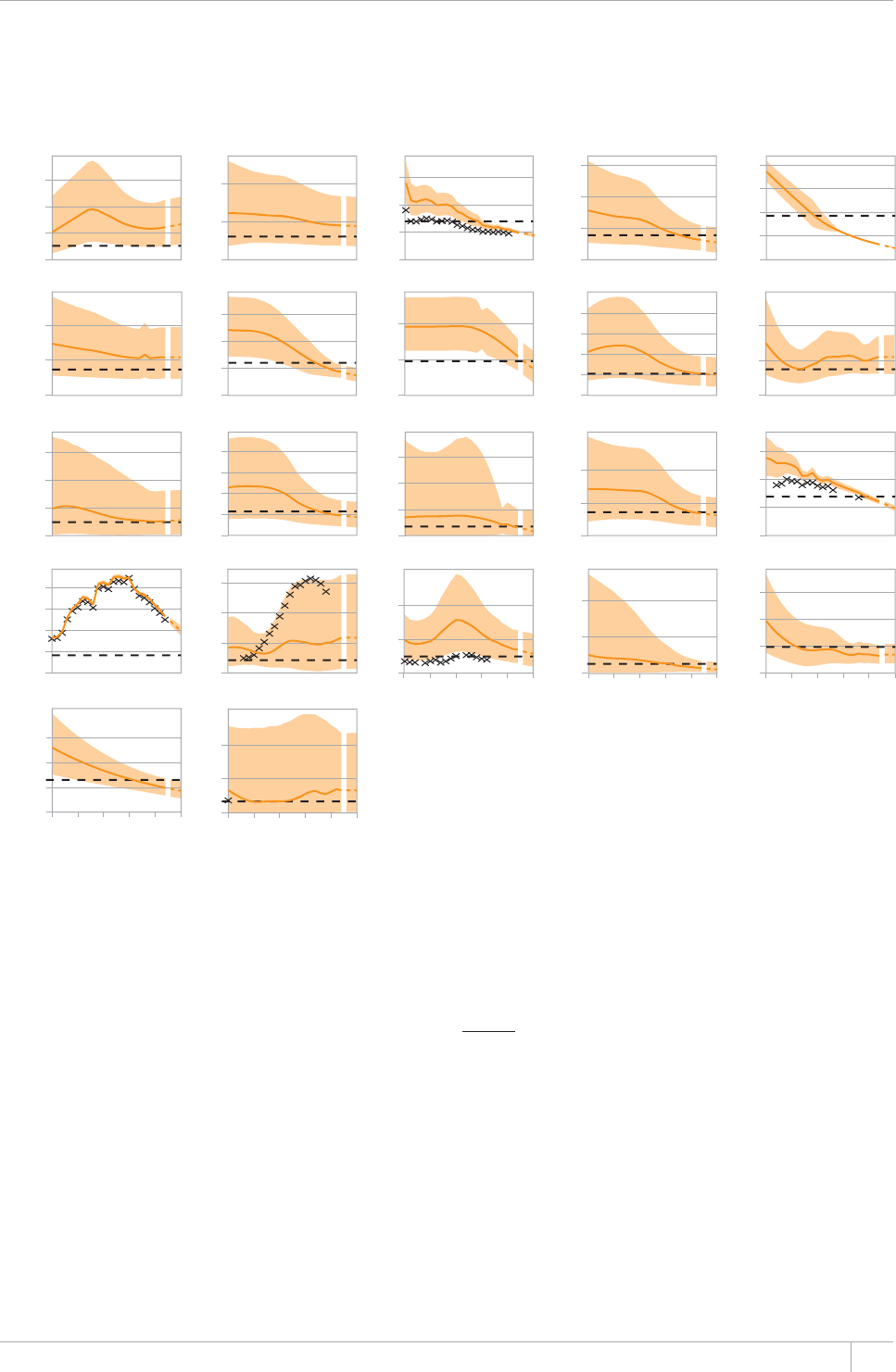
21GLOBAL TUBERCULOSIS REPORT 2013
FIGURE 2.14
Trends in estimated TB mortality rates 1990–2012 and forecast TB mortality rates 2013–2015, 22 high-
burden countries. 'UVKOCVGF6$OQTVCNKV[GZENWFGU6$FGCVJUCOQPI*+8RQUKVKXGRGQRNG6JGJQTK\QPVCNFCUJGFNKPGU
represent the Stop TB Partnership target of a 50% reduction in the mortality rate by 2015 compared with 1990. The
other dashed lines show projections up to 2015.a Uncertainty is due to adjustments made to the mortality data from vital
registration systems that were reported by countries (mortality data from vital registration systems are represented by the “x”
symbol). Further explanation of methods is provided in Annex 1.
a The width of an uncertainty band narrows as the quality and completeness of vital registration
data improves.
b Estimates of TB disease burden have not been approved by the national TB programme in
Bangladesh and a joint reassessment will be undertaken following the completion of the
prevalence survey planned for 2014.
Afghanistan BangladeshbBrazil Cambodia China
DR Congo Ethiopia India Indonesia Kenya
Mozambique Myanmar Nigeria Pakistan Philippines
Russian Federation South Africa Thailand Uganda UR Tanzania
Viet Nam Zimbabwe
Rate per 100 000 population per year
0
5
10
15
20
0
50
100
150
0
20
40
60
0
50
100
1990 1995 2000 2005 2010 2015 1990 1995 2000 2005 2010 2015
1990 1995 2000 2005 2010 2015 1990 1995 2000 2005 2010 2015
0
20
40
60
1990 1995 2000 2005 2010 2015
0
100
200
0
50
100
0
20
40
60
0
5
10
15
20
0
20
40
0
100
200
300
0
25
50
75
100
0
30
60
90
0
50
100
0
2.5
5.0
7.5
0
50
100
0
20
40
60
0
20
40
0
100
200
300
0
50
100
150
200
0
50
100
150
0
20
40
2.4 Strengthening measurement of the
burden of disease caused by TB:
the WHO Global Task Force on TB Impact
Measurement
e estimates of TB incidence, prevalence and mortality
and their trend presented in sections 2.1–2.3 are based
on the best available data and analytical methods. None-
theless, there remains considerable scope to improve mea-
surement of the level of and trends in TB disease burden.
is nal section of the chapter describes the latest status
of eorts to improve measurement of the burden of dis-
ease caused by TB, under the umbrella of the WHO Global
Task Force on TB Impact Measurement. is task force was
established in 2006 and includes representatives from lead-
ing technical and nancial partners and countries with a
high burden of TB.1
At its second meeting in December 2007, the Global Task
Force on TB Impact Measurement dened three strategic
areas of work:2
strengthening surveillance towards the ultimate goal
of direct measurement of incidence and mortality from
notication and VR systems, respectively;
1 Many countries with a high burden of TB are engaged in the work of
the Task Force. Partners that are actively participating in the work
of the Task Force include the Centers for Disease Control and Pre-
vention in the USA, the European Centre for Disease Prevention
and Control, the Global Fund, Public Health England, the KNCV
Tuberculosis Foundation, the London School of Hygiene and Trop-
ical Medicine in the UK, the Research Institute for Tuberculosis in
Japan, the Union and the United States Agency for International
Development (USAID).
2 TB impact measurement: policy and recommendations for how to
assess the epidemiological burden of TB and the impact of TB control.
Geneva, World Health Organization, 2009 (Stop TB policy paper
no. 2; WHO/HTM/TB/2009.416). Available at www.who.int/tb/
publications/2009/impactmeasurementpolicy/

22 GLOBAL TUBERCULOSIS REPORT 2013
conducting surveys of the prevalence of TB disease in a
set of global focus countries that meet epidemiological
and other relevant criteria; and
periodic review and updating of the methods used to
translate surveillance and survey data into estimates of
TB incidence, prevalence and mortality.
In 2008 and 2009, methods were thoroughly reviewed and
updated by an expert group convened by the task force.
Updates were discussed and endorsed by the full task force
in March 2010. Current methods are described in detail in
Annex 1, and an updated review is planned in 2014 (Box
2.1). e following sections focus on the other two strate-
gic areas of work: strengthened surveillance and national
TB prevalence surveys. Further details are available on the
task force’s web site.1
2.4.1 Strengthening surveillance
Reasons for uncertainty in current estimates of TB inci-
dence include use of expert opinion about both the num-
ber of cases that are diagnosed but not reported to national
surveillance systems and the number of cases that are not
diagnosed at all (section 2.1). Major challenges in estimat-
ing TB mortality include the lack of VR systems of su-
cient coverage and quality in many countries, notably in
Africa and parts of Asia (Figure 2.11). e long-term goal
of directly measuring the level of and trends in TB disease
burden from routine surveillance data, using notication
data to measure TB incidence and VR data to measure TB
mortality, requires strengthened surveillance in many
countries. Countries for which more robust estimates of
mortality were available in 2012 are shown in Figure 2.11.
TB surveillance checklist of standards and benchmarks
Strengthening surveillance to move towards the goal of
direct measurement of TB incidence and mortality requires
a clear understanding of what a ‘model’ surveillance system
should look like and a method for assessing the current per-
formance of TB surveillance. Following considerable work
in 2011 and 2012, a TB surveillance checklist that denes
the standards and associated benchmarks that need to be
met for a country’s notication and VR data to be used as
a direct measure of TB incidence and mortality has been
developed (Box 2.4).
Use of the checklist began in January 2013 and it is being
applied in a growing number of countries (Figure 2.15) as
the basis for identifying what standards are already met
and the investments required to close remaining gaps.
is work is being undertaken in close collaboration with
the Global Fund so that use of the checklist is integrated
into the fund’s grant processes and ndings can inform
investments by the fund as well as national governments
and other partners (Box 2.5). With more than 100 low-
and middle-income countries receiving TB grants from the
Global Fund, this approach has great potential to make a
real dierence to TB surveillance worldwide. An initial list
of 25 priority countries has been dened.
Inventory studies to measure or estimate
TB underreporting
One of the standards in the TB surveillance checklist is
that all diagnosed cases of TB are reported to the national
surveillance system. e two benchmarks that must be sat-
ised are that TB reporting is a legal requirement, and that
≥90% of TB cases are reported to national health authori-
ties, as determined by a national-level investigation such as
an inventory study. To date, few countries have implement-
ed an inventory study but as the number doing so increas-
es, estimates of the level of and trend in TB incidence will
improve. Even when underreporting is considerable and
notication data are not a good proxy for TB incidence,
results from inventory studies can be used to quantify the
gap and obtain more precise estimates of disease burden
and provide valuable information about where eorts to
collaborate with public and private sector providers are
needed (see also Chapter 3, section 3.2.1). In 2012, the
Global Task Force on TB Impact Measurement completed a
guide on how to design and implement an inventory study,
and how to analyse and report results.2
In the past 10 years, inventory studies combined with
capture–recapture analysis have been implemented in the
Netherlands, the UK, French Guiana, Egypt, Iraq, Pakistan
and Yemen. Results from the study in Iraq are summarized
in Box 2.6.
Electronic recording and reporting of data
Several of the standards in the TB surveillance checklist
are about data quality. In all of the regional and country
workshops held between 2008 and 2013, it was evident that
it is much easier to assess the quality of TB surveillance
data in countries with case-based electronic recording and
reporting systems. Besides facilitating assessment of data
quality, electronic recording and reporting systems have
other major advantages compared to systems based solely
on paper-based recording and reporting. ese include:
Better programme and resource management, by
encouraging sta to use and act upon live data. is may
help to prevent defaulting from treatment and assist
with management of drug supplies (including avoidance
of stock-outs).
Improved surveillance by making it easier for facilities
not traditionally linked to the NTP, such as hospitals,
prisons and the private sector, to report TB cases, and by
reducing the burden of compiling and submitting data
through paper-based quarterly reports.
Analysis and use of data is facilitated, since data can be
readily imported into statistical packages. Results are
then available to decision-makers more quickly and it is
possible to detect outbreaks promptly.
1 www.who.int/tb/advisory_bodies/impact_measurement_
taskforce
2 Assessing tuberculosis underreporting through inventory studies. Gene-
va, World Health Organization, 2013 (WHO/HTM/TB/2012.12).
Available at: www.who.int/tb/publications/inventory_studies/en/
index.html

23GLOBAL TUBERCULOSIS REPORT 2013
BOX 2.4
The TB surveillance checklist of standards and benchmarks
A major goal of TB surveillance is to
provide an accurate measure of the
number of new TB cases and TB deaths
that occur each year, and to be able
to assess trends over time. In some
countries, TB surveillance already meets
the standards necessary to do this, but
in others there are important gaps. For
example, TB cases that are diagnosed
in the private sector are not reported
in many settings, and in many low- and
middle-income countries some people
with TB may not easily access health
care and therefore not be diagnosed
at all. Furthermore, a large number of
countries lack vital registration systems
with the geographical coverage and
quality required to accurately measure
deaths caused by TB (section 2.3). The
Checklist of standards and benchmarks
for TB surXeillance and Xital reIistration
systems was developed with the
HQNNQYKPIQDLGEVKXGU
To assess a national surveillance
system’s ability to accurately measure
TB cases and deaths.
To identify TB surveillance gaps in
national surveillance systems that
need to be addressed.
The outcomes of the checklist can
be used to identify countries with
surveillance systems that already provide
an accurate measure of the number of
TB cases and deaths that occur each
[GCTCPFVQFGßPGVJGCEVKQPUPGEGUUCT[
to strengthen surveillance in countries
KPYJKEJICRUCTGKFGPVKßGFa Countries
KPVJGHQTOGTECVGIQT[ECPDGEGTVKßGF
as having surveillance data that provide
a direct measure of TB incidence and/or
mortality.
The checklist was developed by a
team of experts in disease surveillance
in conjunction with expert advice
HTQOOGGVKPIUQTICPK\GFD[9*1KP
September 2011 and May 2012. The
checklist underwent two rounds of
ßGNFVGUVKPIKPGNGXGPEQWPVTKGU$TC\KN
China, Egypt, Estonia, Japan, Kenya,
the Netherlands, Thailand, Uganda, the
United Kingdom and the United States
of America.
The checklist is ten pages long
and has two parts. Part A consists of
eighteen questions that are used to
characterise the national TB surveillance
system; these provide the background
for part B, which consists of thirteen
standards and their associated
benchmarks. The standards are general
statements about the characteristics
VJCVFGßPGCJKIJRGTHQTOCPEG6$
surveillance system; nine standards
are related to the measurement of
TB cases and one is related to the
measurement of TB deaths. There are
three supplementary standards that can
be used to assess whether a country’s
surveillance system provides a direct
measure of the number of drug-resistant
TB cases, HIV-positive TB cases, and TB
cases among children. For each of the
VJKTVGGPUVCPFCTFUDGPEJOCTMUFGßPG
(in quantitative terms wherever possible)
the level of performance considered
UWHßEKGPVVQOGGVVJGTGURGEVKXG
standard. An accompanying user guide
explains the rationale for each standard
and associated benchmark(s), and the
methods that should be used to assess
whether the benchmarks and hence the
standard are met. Illustrative examples
are also provided in the user guide.
Based on a completed assessment
using the checklist, countries can
identify key actions needed to address
KFGPVKßGFICRUKPPQVKßECVKQPCPFXKVCN
registration systems. It is anticipated
that an assessment of TB surveillance
using the checklist would take place
GXGT[VJTGGVQßXG[GCTUDWVEQWNFCNUQ
be done more frequently.
Following the 2012
recommendations of the Global Fund’s
6GEJPKECN'XCNWCVKQP4GHGTGPEG)TQWR
and a collaborative agreement between
the fund and WHO, assessments of TB
surveillance using the checklist are
increasingly being integrated within
the fund’s grant mechanisms. As such,
assessments with the checklist should
be timed to coincide with programme
reviews, Global Fund grant renewals
and the development of the concept
notes required to access funding in
the fund’s new funding model (NFM)
NCWPEJGFKP4GUWNVUECPVJGPDG
used to develop or update monitoring
and evaluation investment plans that
can be supported through grants from
the Global Fund as well as by national
budgets and by other partners. This
collaborative effort with the Global
Fund has great potential to help
strengthen TB surveillance in more than
a hundred countries receiving grants
worldwide. Assessments in 15 high-
burden and high-impact countries are
DGKPIRTKQTKVK\GFKPCPF
by August 2013, a total of eleven
countries including eight of the fund’s
high-burden or high-impact countries
had completed the assessment (Figure
2.15).
The checklist and user guide
are available on the website of the
WHO Global Task Force on TB Impact
/GCUWTGOGPV
JVVRYYYYJQKPVVDCFXKUQT[ADQFKGU
KORCEVAOGCUWTGOGPVAVCUMHQTEGGP
a The checklist is not intended to assess a
U[UVGOÂUCDKNKV[VQHWNßNQVJGTRTQITCOOCVKE
requirements, e.g. patient care, delivery of
laboratory results, or drug management.
Higher quality data, since automated data quality checks
can be used and duplicate or misclassied notications
can be identied and removed (which is very dicult or
impossibleto do nationally with paper-based systems).
It is also easier to introduce new data items.
Identication of clusters of cases in space and time,
including clusters of drug-resistant cases, thus allowing
early investigation and containment of epidemics.
Countries that have national electronic case-based data-
bases of TB patients are shown in Figure 2.16. A recent
example of the implementation of a case-based electron-
ic recording and reporting system, in Kenya, is described
in Box 2.7. Recent guidance on electronic recording and
reporting for TB care and control, developed by WHO and
partners in 2011, is available on the task force’s website.1
1 Electronic recording and reporting for TB care and control. Geneva,
World Health Organization, 2013 (WHO/HTM/TB/2011.22).
Available at www.who.int/tb/publications/electronic_recording_
reporting

24 GLOBAL TUBERCULOSIS REPORT 2013
BOX 2.5
The TB surveillance checklist in Indonesia: from implementation to resource mobilization
A national assessment of the TB surveillance system in
Indonesia using the Checklist of standards and benchmarks for
TB surXeillance and Xital reIistration systems (see Box 2.4)
was undertaken in February 2013, linked to a national
programme review.
A thorough analysis of all available national, provincial
CPFFKUVTKEVNGXGNVKOGUGTKGUQH6$PQVKßECVKQPCPFQVJGT
available surveillance data was completed. A desk review of NTP
manuals, guidelines, policy and training documents, annual
reports, reporting forms and registers was conducted. Other
information was collected through interviews with NTP staff,
partners and other stakeholders.
The TB surveillance system is based on quarterly reporting
QHPQVKßGFECUGUHTQOJGCNVJHCEKNKVKGUVQFKUVTKEVUVQRTQXKPEGU
CPFßPCNN[VQVJGPCVKQPCNNGXGN+VKUEWTTGPVN[VTCPUKVKQPKPI
to a web- and case-based electronic recording and reporting
U[UVGO6$ECUGFGßPKVKQPUYGTGEQPUKUVGPVYKVJKPVGTPCVKQPCN
IWKFGNKPGU6JGTGYGTGQWVQHFKUVTKEVUKPVJGEQWPVT[
that submitted all quarterly reports to the national level in
2011. The system produced externally but not internally
consistent data. Since TB reporting is not a legal requirement,
not all TB cases were reported to the NTP, but the level of
underreporting of cases from the private to the public sector
has not been measured nationally. There have been steady
improvements in access to health care, but it is still not at
CNGXGNUWHßEKGPVVQGPUWTGVJCVCNN6$ECUGUJCXGCEEGUUVQ
FKCIPQUKUCPFECTG#PCVKQPCNN[TGRTGUGPVCVKXG84U[UVGOYKVJ
standard coding of causes of death is being developed. Only
provincial level drug resistance surveys have been conducted
so far, and while HIV testing of TB cases was improving the
coverage remains low. Finally, childhood TB was diagnosed in
limited settings.
#EVKXKVKGUVQCFFTGUUVJGICRUVJCVYGTGKFGPVKßGFHTQO
VJGKORNGOGPVCVKQPQHVJGEJGEMNKUVYGTGFGßPGFUGGTable
B2.7.1). One of the top priorities is maintenance of the sample
84U[UVGOYJKEJEQUVU75¿RGTECRKVCKPVJGUCORNGF
CTGCUGSWKXCNGPVVQCDQWV75a¿OKNNKQPRGT[GCTHQTVJG
population of 5 million to be covered). For the other activities
in Table B2.7.1, the total budget requirement was estimated
VQDG75aOKNNKQPCOQPIYJKEJQPGVQRRTKQTKV[KFGPVKßGF
in the key recommendations arising from the 2013 programme
TGXKGYKUKORNGOGPVCVKQPQHCOCPFCVQT[PQVKßECVKQPRQNKE[
Through continuous consultations between the NTP, WHO and
VJG)NQDCN(WPFVJGßPCPEKPITGSWKTGFHQTVJGKPXGUVOGPVRNCP
YCUKFGPVKßGFCPFUGEWTGF
This example shows how the checklist can be used to
EQPFWEVCUVCPFCTFK\GFCUUGUUOGPVQH6$UWTXGKNNCPEGJKIJNKIJV
progress achieved as well as remaining gaps to be addressed,
and to secure funding for an investment plan to close the gaps
with support from the Global Fund.
TABLE B2.7.1
Investment plan for strengthening surveillance in
+PFQPGUKCDCUGFQPICRUKFGPVKßGFVJTQWIJVJG
implementation of the Checklist of standards and
benchmarks for TB surveillance and vital registration
systemsVQVCNDWFIGV75aOKNNKQPGZENWFKPI84
U[UVGOHWPFGFUGRCTCVGN[
Activity
■ 8KVCNTGIKUVTCVKQP84OCKPVCKPKPICPFUECNKPIWRVJGPCVKQPCNN[
TGRTGUGPVCVKXGUCORNG84U[UVGO
■ Inventory study to measure the level of underreporting
■ Capacity building for data management and statistical analysis –
VJTQWIJCVVGPFKPIEQWTUGUCPFGZVTCUVCHßPICVVJGEGPVTCNNGXGN
■ Implementation of the SerXice #Xailability and 4eadiness
#ssessment Tool and health facility data quality assessment
■ Assessment of the Integrated Tuberculosis Information System (SITT)
Phase 2 in 2014
■ +ORNGOGPVKPIOCPFCVQT[PQVKßECVKQPRQNKE[
■ Analysis of available mortality data
■ Drug resistance survey or sentinel surveillance
■ Nationally representative survey of HIV prevalence among TB
patients
■ Corrective actions required to compile all the reports from Papua
1 TB prevalence surveys: a handbook. Geneva, World Health Organiza-
tion, 2011 (WHO/HTM/TB/2010.17). Available at www.who.int/
tb/advisory_bodies/impact_measurement_taskforce/resources_
documents/thelimebook/
2.4.2 National surveys of the prevalence of
TB disease
Before 2007, few countries had implemented nationwide
prevalence surveys. In the 1990s, national surveys were
conned to China, Myanmar, the Philippines and the
Republic of Korea. Before 2009 and with the exception of
Eritrea in 2005, the last national surveys in the African
Region were undertaken between 1957 and 1961. From
2002 to 2008, there was typically one survey per year.
In 2007, WHO’s Global Task Force on TB Impact Mea-
surement identied 53 countries that met epidemiological
and other criteria for implementing a survey. A set of 22
global focus countries were selected to receive particular
support in the years leading up to 2015. e African coun-
tries were: Ethiopia, Ghana, Kenya, Malawi, Mali, Mozam-
bique, Nigeria, Rwanda, Sierra Leone, South Africa, Uganda,
the United Republic of Tanzania and Zambia. Countries in
Asia were: Bangladesh, Cambodia, China, Indonesia, Myan-
mar, Pakistan, the Philippines, ailand and Viet Nam.
Since early 2008, substantial eorts to support countries
to design, implement, analyse and report on surveys have
been made. Examples include development of updated guid-
ance,1 coordination of technical assistance, expert reviews
of protocols, organization of study tours and mid-term sur-
vey reviews, and global and regional workshops to support
survey design and implementation and to share results and
lessons learned among countries. As part of these eorts,
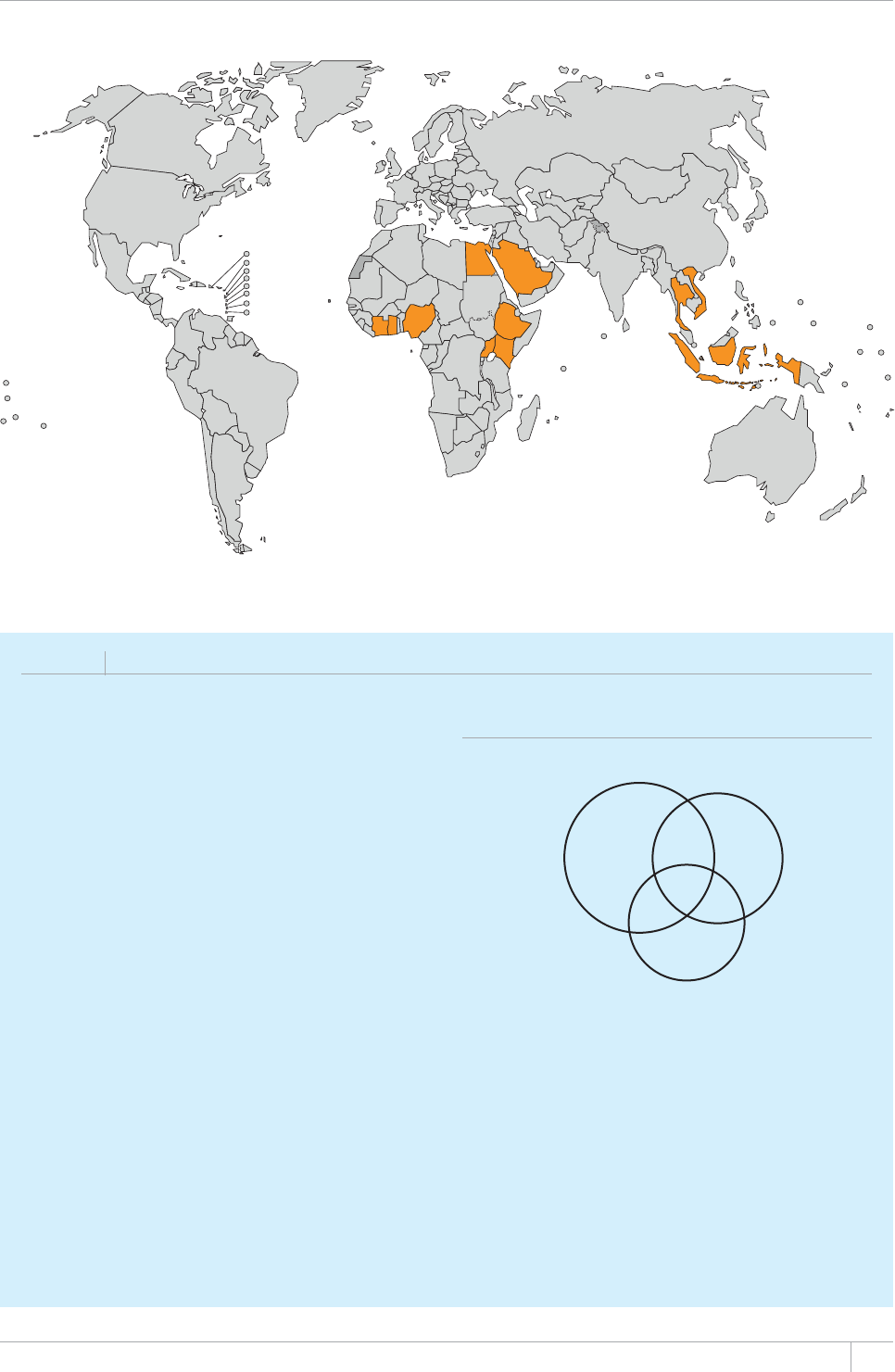
25GLOBAL TUBERCULOSIS REPORT 2013
FIGURE 2.15
Countries (in orange) where the TB surveillance checklist of standards and benchmarks has been used:
status in August 2013
BOX 2.6
Inventory studies to estimate TB underreporting: an example from Iraq
Inventory studies compare the number of TB cases meeting
UVCPFCTFECUGFGßPKVKQPUKPCNNQTKPCUCORNGQHRWDNKECPF
RTKXCVGJGCNVJHCEKNKVKGUYKVJVJGTGEQTFUQH6$ECUGUPQVKßGF
to local and national authorities. This enables the level of
WPFGTTGRQTVKPIQHFKCIPQUGFECUGUVQDGSWCPVKßGF+PEGTVCKP
circumstances, the results from inventory studies can be
combined with a type of modelling called capture–recapture
analysis to estimate TB incidence. A WHO guide on the design
and implementation of inventory studies, and analysis and
reporting of results, was published in 2013.
The results from the survey in Iraq,a which was completed
in 2011, are illustrated below. The number of TB cases that
were detected by three types of health service providers
was studied during a three-month period in eight randomly
UGNGEVGFIQXGTPQTCVGUQWVQHCVQVCNQH6JGVQVCNPWODGT
QHFGVGEVGFECUGUYCU%CUGUVJCVYGTGFGVGEVGFDWVPQV
reported to the NTP accounted for 16% of total detected cases
i.e. the level of underreporting was 16%. Capture–recapture
modelling was used to estimate that an additional 473 cases
EQPßFGPEGKPVGTXCN¿JCFPQVDGGPFGVGEVGFD[
any of the three types of health providers. These results were
WUGFVQGUVKOCVGVJCVVJGTGYGTGCRRTQZKOCVGN[aKPEKFGPV
cases of TB in Iraq in 2011 (a downward revision compared with
previous estimates) and that about 60% of cases were being
detected (an upward revision from the previous best estimate
QH
The value of study results went beyond updates to estimates
QH6$KPEKFGPEG'ZCORNGUKPENWFG
NTP
(n=1673) Private
(n=649)
0 Public
(n=377)
416 199
25
244 9
99
Updated estimates were crucial for the development of a
sound national strategic plan and to assess progress towards
the 2015 MDG target.
The national strategic plan includes interventions designed
VQCFFTGUUVJGECWUGUQHWPFGTTGRQTVKPIVJCVYGTGKFGPVKßGF
during the study.
The mapping of all health facilities delivering care to
chest-symptomatic patients in study areas (that covered
50% of the country) provided a foundation for sustained
engagement of all care providers through PPM initiatives.
a Huseynova S et al. Estimating tuberculosis burden and reporting in
TGUQWTEGNKOKVGFEQWPVTKGUCECRVWTGTGECRVWTGUVWF[KP+TCSInternational
,ournal of Tuberculosis and LunI &iseasea¿
FIGURE B2.6.1
Results from the 2011 inventory study in Iraq
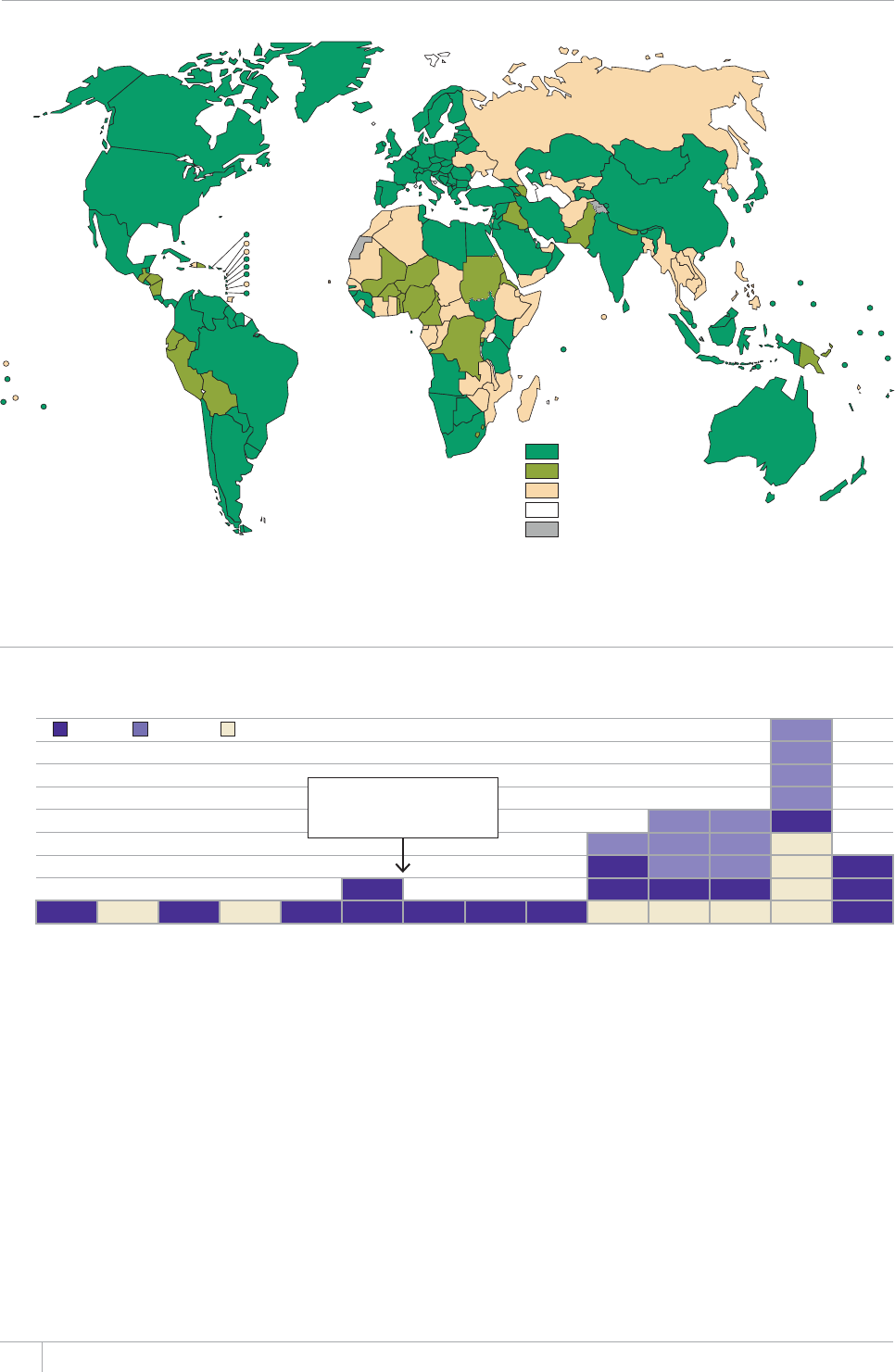
26 GLOBAL TUBERCULOSIS REPORT 2013
FIGURE 2.16
Availability of national electronic case-based databases of TB patients, 2012
All TB patients
MDR-TB patients only
None
No data
Not applicable
FIGURE 2.17
Global progress in implementing national surveys of the prevalence of TB disease, actual (2002–2013)
and expected (2014–2017)
Number of surveys
9
Kenya
/Q\CODKSWG
7
South Africa
6
Uganda
5
Nigeria Ghana Bangladesh
4
Ethiopia Rwanda Malawi DPR Korea
3
Cambodia 746CP\CPKC Zambia Mongolia Myanmar
2
Philippines Pakistan Thailand Indonesia Nepal Philippines
1
Cambodia Malaysia Indonesia Eritrea Thailand Viet Nam Bangladesh Myanmar China Lao PDR Gambia Sudan Zimbabwe Viet Nam
0
2002 2003 2004 2005 2006 2007 2009 2010 2011 2012 2013 2014–
2015
2016–
2017
Global focus countries (GFC)
selected by WHO Global Task Force
on TB Impact Measurement
Asia – GFC Africa – GFC Non – GFC
the concept of Asia–Asia, Asia–Africa and Africa–Africa
(‘AA’) collaboration has been strongly promoted.
Following six years of substantial eort at country,
regional and global levels, unprecedented progress has
been achieved (Figure 2.17). If surveys are implemented
according to schedule, more than 20 surveys will be
implemented between 2011 and 2015. Five national TB
prevalence surveys were implemented in 2012 (Gambia,
Nigeria, Rwanda, the United Republic of Tanzania and
ailand) and a further ve w ill start or be completed in 2013
(Ghana, Indonesia, Malawi, Sudan and Zambia). ese
surveys provide an unbiased estimation of disease burden,
often for the rst time, and will be used to update estimates
of disease burden once results are nalized (Box 2.1).
Surveys are also providing a rich source of data to
inform programme policy and strategy. Although results
remain provisional pending nalization of analyses in late
2013 (Box 2.1), an excellent recent example is provided
by the 2012 survey in Nigeria (Box 2.8). For other recent
surveys, some country-specic reports are already public-
ly available (for example, from China, Cambodia, Ethiopia
and Myanmar) and others are in the pipeline. Papers for
peer-reviewed journals are also in preparation, from these
and other recent surveys.
WHO, together with countries and technical partners,
started preparing or planning global and regional synthe-
27GLOBAL TUBERCULOSIS REPORT 2013
BOX 2.7
Implementation of an innovative electronic surveillance and management system in Kenya
The NTP in Kenya has rolled out an innovative electronic
system to support surveillance and management, called TIBU
(which means cure in Swahili). In addition to running electronic
versions of its standard district TB registers, the TIBU system
makes use of the country’s extensive mobile communications
network and widespread use of mobile phones to make
RC[OGPVUVQ/&46$RCVKGPVUVJCVJGNRVQUWRRQTVVJGKT
VTGCVOGPVVJTQWIJVJGRQRWNCTaM-Pesa mobile payment system.
The system will also be used to manage drug supplies and
laboratory data and consumables.
At TIBU’s core is a national case-based database that stores
details about each individual patient episode of TB (including
ECUGUQH/&46$7UGTUCEEGUUVJGU[UVGOGKVJGTVJTQWIJCYGD
browser or by using an Android ‘app’. The NTP has given each
District TB and Leprosy Coordinator (DTLC) a tablet computer
VJCVTWPUVJG#PFTQKFQRGTCVKPIU[UVGOCPFKUßVVGFYKVJC
SIM card to connect to the internet through mobile telephone
networks. DTLCs can access the system during their regular
visits to all facilities providing TB diagnostic and treatment
services within their district and enter TB patient details into
the TIBU app during their visits to these facilities. Data are
transmitted directly to the national database via the mobile
network. Data remain stored on the tablet if no connection
is available at a facility and are subsequently transmitted to
the national database as soon as a connection is available.
TIBU automatically generates various reports, including
standard quarterly reports, charts and maps for all levels of the
administrative chain.
The NTP and developers work closely with other parts of the
Ministry of Health to ensure that TIBU complies with national
standards, such that it can communicate with the ministry’s
other health information systems.a TIBU uses Kenya’s national
facility coding scheme (JVVRYYYGJGCNVJQTMGHCEKNKVKGU
facilitytypes.aspx) and therefore the developers were able to
DWKNFCUGCONGUUNKPMVQVJGa-enya *ealth Information Systemb
VJCVRTQXKFGUFKUVTKEVRTQXKPEKCNCPFPCVKQPCNJGCNVJQHßEKCNU
YKVJKPFKECVQTUHQTOWNVKRNGJGCNVJCTGCUKPENWFKPI6$a6JKU
allows standard TB indicators to be automatically updated every
quarter in the ministry’s system.
TIBU has been developed in phases. Development was
initiated in 2007 and the original intention was to run the
system on personal digital assistants (PDAs). However, problems
with initial attempts to implement the system combined with
the rapid rise, availability and falling costs of Android-based
mobile devices, as well as the widespread adoption of mobile
phones, led to a decision to switch to the development of an
#PFTQKFaCRRaHQTWUGQPUOCTVRJQPGUCPFVCDNGVEQORWVGTU
Plans for future phases include extending coverage beyond TB
and leprosy to patients with asthma and other lung diseases,
integration with the laboratory management system and
eventually, if resources allow, roll out of tablets to over 4000
health care facilities where TB diagnostic and treatment
services are offered.
a In May 2013 the World Health Assembly adopted resolution WHA66.24 on
promoting such standardization and interoperability of health information
systems (JVVRCRRUYJQKPVIDGDYJCRFHAßNGU9*##A4GPRFHa
b Based on the open source DHIS2 platform JVVRYYYFJKUQTI
BOX 2.8
The 2012 national TB prevalence survey in Nigeria: programmatic implications
+ORNGOGPVCVKQPQHVJGßTUVGXGTPCVKQPCN6$RTGXCNGPEGUWTXG[KP0KIGTKCDGICPKP(GDTWCT[CPFßGNFCEVKXKVKGUEQXGTKPI
70 geographic clusters) were completed in October 2012. Nigeria became the second African country to successfully complete a
PCVKQPCNUWTXG[CEEQTFKPIVQEWTTGPV9*1IWKFGNKPGUHQNNQYKPI'VJKQRKCKP#VQVCNQHaRGQRNGCIGFÜRCTVKEKRCVGF
in the study.
Survey results included that 75% of previously undetected cases found during the survey had sputum smear-positive TB and
classic TB symptoms that met national screening criteria (this proportion was higher than in any other survey implemented since
%QORCTKUQPQHVJGPWODGTQHRTGXCNGPVECUGUQHURWVWOUOGCTRQUKVKXG6$YKVJPQVKßECVKQPFCVCHQTVJGUCOGCIGITQWR
UJQYGFCRTGXCNGPEGPQVKßECVKQPTCVKQQHCNUQJKIJGTVJCPKPCP[QVJGTUWTXG[KORNGOGPVGFUKPEG6JGUWTXG[CNUQ
illustrated that the burden of TB was geographically uneven, ranging from very low to extremely high levels among survey clusters.
An important conclusion already drawn from the survey results is that a top priority is to expand access to and improve the
quality of basic TB diagnostic and treatment services. If high-quality DOTS services were readily available, it would be expected that
DQVJVJGRGTEGPVCIGQHRTGXCNGPVECUGUYKVJV[RKECN6$U[ORVQOUCPFVJGRTGXCNGPEGPQVKßECVKQPTCVKQYQWNFDGOWEJNQYGT
#UGEQPFEQPENWUKQPKUVJCVURGEKßEHQEWUKUPGGFGFQPIGQITCRJKEÁJQVURQVUÂYJGTGVJGFKUGCUGDWTFGPKUJKIJGUV
ses of the main results and lessons learned from national
TB prevalence surveys in 2012. A paper that summarizes
the results and lessons learned from surveys conducted
1990–2012 in Asia is nearing completion and a similar
paper about recent surveys in Africa is planned. An overall
synthesis of the main implications of results from recent
prevalence surveys implemented in Asia and Africa for
post-2015 global TB policy and strategy is also in the pipe-
line. ese global and regional summaries will be widely
disseminated once available, starting in 2014.
GLOBAL TUBERCULOSIS REPORT 2013
%*#26'4
TB case notißcations
and treatment outcomes
Routine recording and reporting of the numbers of TB
cases diagnosed and treated by NTPs and monitoring of
treatment outcomes was one of the ve components of
the global TB strategy (DOTS) launched by WHO in the
mid-1990s and it remains a core element of its successor,
the Stop TB Strategy (Chapter 1). With the standard de-
nitions of cases and treatment outcomes recommended by
WHO and associated recording and reporting framework
as a foundation, global monitoring of trends in case noti-
cations and treatment outcomes has been possible since
1995. e number of people diagnosed and treated for TB
and associated treatment outcomes is routinely monitored
by NTPs in almost all countries, which in turn report these
data to WHO in annual rounds of global TB data collection
(Chapter 1).
is chapter has four parts. Section 3.1 summarizes
the total number of people diagnosed with TB and noti-
ed by NTPs in 2012, including disaggregation by case
type, age and sex. e share of notications accounted for
by the prison sector in the European Region and the high
case notication rates among the prison population are
also highlighted. Section 3.2 presents and discusses the
contribution to total case notications of public–public
and public–private mix (PPM) initiatives in 29 countries
and of community-based care in 13 countries. Section 3.3
presents trends in notications between 1990 and 2012
and compares these with trends in estimated TB incidence.
Estimates of the ratio of notied:incident cases (an indica-
tor known as the case detection rate or CDR) are provided
for selected years. Section 3.4 describes the latest data on
treatment outcomes (for cases registered for treatment in
2011) as well as treatment outcomes achieved in each year
since 1995.
3.1 Case notißcations in 2012 by type
of disease, age and sex
e denitions of TB cases recommended by WHO until the
end of 2012, and that were used in the 2013 round of glob-
al TB data collection, are shown in Box 3.1. Although not
used in the global TB report this year, it should be highlight-
ed that after a two-year consultation process, WHO issued
updated guidance on denitions of cases and treatment
outcomes and an associated reporting framework in March
2013.1 ese updates were necessary to accommodate
KEY FACTS AND MESSAGES
■ +POKNNKQPECUGUQH6$YGTGPQVKßGFD[
062UCPFTGRQTVGFVQ9*1OKNNKQPYGTGKPFKXKFWCNU
newly diagnosed in 2012 and 0.4 million were previously
diagnosed TB patients whose treatment regimen was
EJCPIGF+PFKCCPF%JKPCCEEQWPVGFHQTQHPQVKßGF
cases of TB worldwide in 2012, African countries for 23%
CPFVJG*$%UHQT
■ +PVJGVTGCVOGPVUWEEGUUTCVGYCUCOQPI
CNNPGY6$ECUGUCPFCOQPIPGYECUGUQHURWVWO
smear-positive pulmonary TB (the most infectious cases).
Improvement in treatment outcomes is needed in the
'WTQRGCP4GIKQPYJGTGVJGVTGCVOGPVUWEEGUUTCVGKP
was 72% and 65% for new cases and new smear-positive
cases respectively.
■ The provision of diagnosis and treatment according
to the DOTS/Stop TB StrateIy has resulted in major
achievements in TB care and control. Between 1995 and
2012, 56 million people were successfully treated for TB
in countries that had adopted the DOTS/Stop TB StrateIy,
saving 22 million lives.
■ 0QVKßECVKQPUQH6$ECUGUJCXGUVCDKNK\GFKPTGEGPV
years, and in 2012 represented 66% (range, 64–69%) of
GUVKOCVGFKPEKFGPVECUGU6JGICRDGVYGGPPQVKßECVKQPU
and incident cases can be explained by a mixture of
underreporting of diagnosed TB cases (for example, failure
to notify cases diagnosed in the private sector) and under-
diagnosis due to poor access to health care and/or failure to
detect cases when people visit health care facilities. Major
efforts are needed to ensure that all cases are detected,
PQVKßGFVQPCVKQPCNUWTXGKNNCPEGU[UVGOUCPFVTGCVGF
according to international standards.
■ +POQUVPQVKßGF6$RCVKGPVUYGTG¿[GCTUQH
CIG%JKNFTGPCIGF[GCTUCEEQWPVGFHQTQHPQVKßGF
ECUGU6JGOCNGHGOCNGTCVKQYCUINQDCNN[TCPIKPIHTQO
1.0 to 2.1 among the six WHO regions.
1 Denitions and reporting framework for tuberculosis – 2013 revision
(WHO/HTM/TB/2013.2). Geneva, World Health Organization,
2013. (Available at www.who.int/iris/bitstream/10665/79199/1/
9789241505345_eng.pdf).
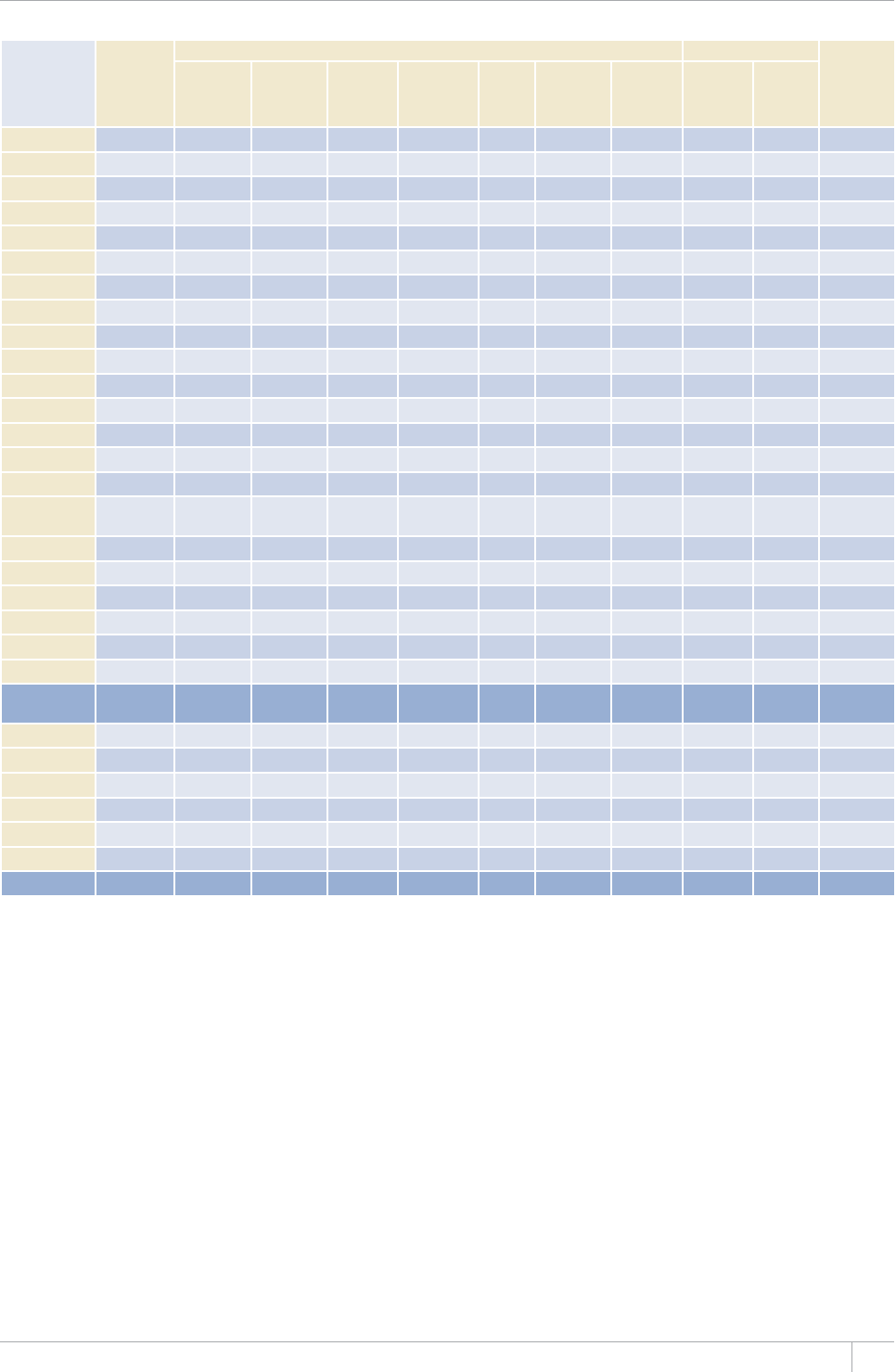
29GLOBAL TUBERCULOSIS REPORT 2013
diagnosis using Xpert MTB/RIF and other WHO-endorsed
molecular tests (Chapter 5), as well as oering an opportu-
nity to improve aspects of the existing (2006) framework,
such as inclusion of more comprehensive reporting of TB
cases among children. e updated denitions, which will
be used in WHO’s 2014 round of global TB data collection,
are summarized in Box 3.2.
In 2012, 6.1 million people with TB were notied to
NTPs and reported to WHO. Of these, 5.7 million had a new
episode of TB (shown as the total of new and relapse cases
in Table 3.1). Of these 5.7 million cases, 5.4 million had
TB for the rst time and 0.3 million were people who had a
recurrent episode of TB after being previously cured of the
disease. Besides a small number of cases whose history of
treatment was not recorded, the remaining 0.4 million had
already been diagnosed with TB but their treatment was
changed to a retreatment regimen.
Among people who were diagnosed with TB for the rst
time (new cases), 2.5 million had sputum smear-positive
pulmonary TB, 1.9 million had sputum smear-negative
pulmonary TB, and 0.8 million had extrapulmonary TB;
the remaining cases did not have a sputum smear done or
their case type was unknown (Table 3.1). India and China
accounted for 39% of the 5.7 million new and relapse cas-
es of TB that were notied in 2012 (23% and 16%, respec-
tively); the South-East Asia and Western Pacic Regions
in which these countries are respectively located together
accounted for 60% of such cases globally. African countries
TABLE 3.1
Case notißcations, 2012
TOTAL NOTIFIED
NEW CASES 4'64'#6/'06%#5'5
NEW AND
4'.#25'a
5/'#4215+6+8' 5/'#40')#6+8'
5/'#4016
DONE
':64#
27./10#4;
%#5'6;2'
UNKNOWN
27./10#4;
CASES
.#$14#614;
%10(+4/'&
2'4%'06#)'1(
27./10#4;
CASES
.#$14#614;
%10(+4/'& 4'.#25'
4'64'#6/'06
':%.4'.#25'
Afghanistan 13 319 4 740 2 665 6 906 702 13 319 62 1 049 197
Bangladesh 173 619 106 790 24 451 0 30 549 0 106 790 3 065 4 936
$TC\KN 40 152 10 297 11 70 7 633 75 122
Cambodia 015 290 0 64 446 73
China 316 332 533 977 2 073 6 479 0 316 332 37 10 033
&4%QPIQ 112 499 71 124 13 214 20 669 71 124 3 977 3 515
Ethiopia 147 592 47 236 47 340 2 073 0 47 236 49 2 269 145 323
India 317 616 234 029 2 139 637 273 67 106 463 177 749
Indonesia 331 424 202 319 15 697 202 319 66 5 942 2 600
Kenya 99 149 36 937 15 934 0 36 937 50 3 419 6 162
/Q\CODKSWG 20 951 19 797 5 542 0 20 951 51 1 451 47 741
Myanmar 42 909 73 042 0 20 661 0 42 909 37 6 979 141 170
Nigeria 52 901 32 972 4 432 52 901 62 2 513 5 035
Pakistan 273 097 110 545 109 425 0 41 410 0 110 545 50 6 095 5 622 267 475
Philippines 115 263 0 3 270 0 45 19 409 216 199
4WUUKCP
Federation 149 921 27 467 59 019 1 039 10 017 0 41 123 47 105 753
South Africa 63 210 71 421 42 467 0 71 323 664
Thailand 17 537 b64 904 60 304
Uganda 47 211 24 916 5 143 0 24 916 65 1 334 44 663
746CP\CPKC 21 393 0 14 595 0 54 1 052 1 714
Viet Nam 103 906 51 033 21 706 3 210 51 033 67 7 259 1 794 102 112
<KODCDYG 12 163 14 354 2 962 4 912 0 12 163 41 1 369 2 960 35 760
High-burden
countries 5 005 111 2 091 141 1 654 670 100 731 582 909 6 062 2 144 779 56 228 309 335 304 4 669 807
#(4 1 412 639 600 355 345 947 100 537 234 539 977 656 272 63 60 497 67 770
#/4 232 695 122 606 35 606 14 564 34 400 1 669 132 070 76 9 949
'/4 173 963 135 346 90 943 702 175 025 55 10 020 420 769
'74 337 167 6 257 39 029 30 112 577 55 67 662 269 505
5'#4 2 331 455 0 3 004 1 027 902 62 131 245 201 335 2 130 120
924 1 345 466 500 171 691 714 9 751 59 294 502 652 42 45 277 34 740 1 310 726
Global 6 090 211 2 541 283 1 913 682 139 632 796 508 9 669 2 606 498 57 283 361 395 389 5 694 822
Blank cells indicate data not reported.
a 0'9#0&4'.#25'KPENWFGUECUGUHQTYJKEJVJGVTGCVOGPVJKUVQT[KUWPMPQYP
b .#$14#614;%10(+4/'&FCVCHQT6JCKNCPFTGHGTVQUOGCTRQUKVKXGECUGUQPN[&CVCQPECUGUVJCVYGTGNCDQTCVQT[EQPßTOGFWUKPIQVJGTOGVJQFUYGTGPQVTGRQTVGF

30 GLOBAL TUBERCULOSIS REPORT 2013
accounted for 24% of new and relapse TB cases globally
(one quarter of these cases were from one country – South
Africa). e WHO Eastern Mediterranean and European
Regions and the Region of the Americas accounted for 16%
of new and relapse TB cases notied in 2012 (7%, 5% and
4%, respectively); combined, the 22 HBCs accounted for
82% of such cases.
Among the 22 HBCs, the percentage of new pulmonary
cases that were bacteriologically conrmed was highest in
Bangladesh (81%) and the Democratic Republic of the Con-
go (84%), and relatively low in China (37%), the Philippines
(45%), the Russian Federation (47%) and Zimbabwe (41%).
Almost all (96%) of the notications of new cases of
smear-positive pulmonary TB were disaggregated by age
and sex (Table 3.2); 88% were aged 15–64 years, 59% were
aged 15–45 years and 2% were children (aged <15 years).
e global male:female sex ratio was 1.9, but among HBCs
this varied from 0.5 in Afghanistan to 3.0 in Viet Nam.
Variation among countries may reect real dierences
in epidemiology as well as dierential access to or use of
health care services linked to the NTP.
Reporting of cases disaggregated by age and sex was
much less complete for new smear-negative pulmonary and
extrapulmonary cases. For example, data disaggregated by
age and sex according to the categories shown in Table 3.2
were not available for 11 HBCs. When the available data
for all new cases were combined, most cases (82%) were
aged 15–64 years, 55% were aged 15–45 years and 6% were
among children (<15 years); the male:female ratio was 1.7,
ranging from 1.0 to 2.1 among the six WHO regions. Fur-
ther eorts are needed to improve reporting of all cases dis-
aggregated by age and sex.
In the European Region, WHO and the European Cen-
tre for Disease Control and Prevention (ECDC) also request
countries to report notications in the civilian and prison
sectors separately. ese data show that notications in
the prison sector can be a considerable share of all cases,
and that case notication rates in the prison population
can be strikingly high. A summary of the latest data in the
European Region and selected countries, and an example of
success in reducing notication rates in the Russian Feder-
ation, are provided in Box 3.3.
BOX 3.1
WHO deßnitions of TB cases used until the end of 2012 (and in this global TB report)a
Deßnite case of TB A patient with Mycobacterium
tuberculosisEQORNGZKFGPVKßGFHTQOCENKPKECNURGEKOGP
either by culture or by a newer method such as molecular line
probe assay (LPA). In countries lacking laboratory capacity
to routinely identify M. tuberculosis, a pulmonary case with
one or more initial sputum specimens positive for acid-fast
DCEKNNK#($KUCNUQEQPUKFGTGFVQDGCÁFGßPKVGÂECUGRTQXKFGF
that there is functional external quality assurance with blind
rechecking.
Case of TB #FGßPKVGECUGQH6$FGßPGFCDQXGQTQPGKP
which a health worker (clinician or other medical practitioner)
has diagnosed TB and decided to treat the patient with a full
course of anti-TB treatment.
Case of pulmonary TB A patient with TB disease involving
the lung parenchyma.
Smear-positive pulmonary case of TB A patient with
one or more initial sputum smear examinations (direct smear
microscopy) AFB-positive; or one sputum examination AFB-
positive plus radiographic abnormalities consistent with active
pulmonary TB as determined by a clinician. Smear-positive
cases are the most infectious and thus of the highest priority
from a public health perspective.
Smear-negative pulmonary case of TB A patient with
pulmonary TB who does not meet the above criteria for smear-
RQUKVKXGFKUGCUG&KCIPQUVKEETKVGTKCUJQWNFKPENWFGCVNGCUV
two AFB-negative sputum smear examinations; radiographic
abnormalities consistent with active pulmonary TB; no response
to a course of broad-spectrum antibiotics (except in a patient
HQTYJQOVJGTGKUNCDQTCVQT[EQPßTOCVKQPQTUVTQPIENKPKECN
evidence of HIV infection); and a decision by a clinician to
treat with a full course of anti-TB chemotherapy. A patient with
positive culture but negative AFB sputum examinations is also a
smear-negative case of pulmonary TB.
Extrapulmonary case of TB A patient with TB of organs
other than the lungs (e.g. pleura, lymph nodes, abdomen,
genitourinary tract, skin, joints and bones, meninges).
Diagnosis should be based on one culture-positive specimen,
or histological or strong clinical evidence consistent with active
extrapulmonary disease, followed by a decision by a clinician
to treat with a full course of anti-TB chemotherapy. A patient
in whom both pulmonary and extrapulmonary TB has been
FKCIPQUGFUJQWNFDGENCUUKßGFCUCRWNOQPCT[ECUG
New case of TB A patient who has never had treatment for TB
or who has taken anti-TB drugs for less than one month.
Retreatment case of TB There are three types of retreatment
ECUGKCRCVKGPVRTGXKQWUN[VTGCVGFHQT6$YJQKUUVCTVGFQP
a retreatment regimen after previous treatment has failed
(treatment after failure); (ii) a patient previously treated for
TB who returns to treatment having previously defaulted; and
(iii) a patient who was previously declared cured or treatment
completed and is diagnosed with bacteriologically-positive
(sputum smear or culture) TB (relapse).
Case of multidrug-resistant TB (MDR-TB) TB that is
TGUKUVCPVVQVYQßTUVNKPGFTWIUKUQPKC\KFCPFTKHCORKEKP(QT
OQUVRCVKGPVUFKCIPQUGFYKVJ/&46$9*1TGEQOOGPFU
treatment for 20 months with a regimen that includes second-
line anti-TB drugs.
a See Treatment of tuberculosis Iuidelines, 4th ed. Geneva, World Health
1TICPK\CVKQP9*1*6/56$#XCKNCDNGCVJVVRYJSNKDFQE
YJQKPVRWDNKECVKQPUAGPIRFH

31GLOBAL TUBERCULOSIS REPORT 2013
BOX 3.2
WHO deßnitions of TB cases recommended for use starting in 2013 and that will be used in the 2014
global TB reporta
Bacteriologically conßrmed case of TB A patient from
whom a biological specimen is positive by smear microscopy,
EWNVWTGQT9*1CRRTQXGFTCRKFFKCIPQUVKEVGUVUWEJCU:RGTV
/6$4+(#NNUWEJECUGUUJQWNFDGPQVKßGFTGICTFNGUUQH
whether TB treatment is started.
Clinically diagnosed case of TB A patient who does not
HWNßNVJGETKVGTKCHQTDCEVGTKQNQIKECNN[EQPßTOGF6$DWVJCU
been diagnosed with active TB by a clinician or other medical
practitioner who has decided to give the patient a full course
QH6$VTGCVOGPV6JKUFGßPKVKQPKPENWFGUECUGUFKCIPQUGF
QPVJGDCUKUQH:TC[CDPQTOCNKVKGUQTUWIIGUVKXGJKUVQNQI[
CPFGZVTCRWNOQPCT[ECUGUYKVJQWVNCDQTCVQT[EQPßTOCVKQP
Clinically diagnosed cases subsequently found to be
bacteriologically positive (before or after starting treatment)
UJQWNFDGTGENCUUKßGFCUDCEVGTKQNQIKECNN[EQPßTOGF
Case of pulmonary TB #P[DCEVGTKQNQIKECNN[EQPßTOGFQT
clinically diagnosed case of TB involving the lung parenchyma
QTVJGVTCEJGQDTQPEJKCNVTGG/KNKCT[6$KUENCUUKßGFCU
pulmonary TB because there are lesions in the lungs.
Tuberculous intra-thoracic lymphadenopathy (mediastinal and/
or hilar) or tuberculous pleural effusion, without radiographic
abnormalities in the lungs, constitute a case of extrapulmonary
TB. A patient with both pulmonary and extrapulmonary TB
UJQWNFDGENCUUKßGFCUCECUGQHRWNOQPCT[6$
Case of extrapulmonary TB #P[DCEVGTKQNQIKECNN[EQPßTOGF
or clinically diagnosed case of TB involving organs other than
the lungs, e.g. pleura, lymph nodes, abdomen, genitourinary
tract, skin, joints and bones, meninges.
New case of TB A patient who has never been treated for TB
or has taken anti-TB drugs for less than 1 month.
Retreatment case of TB A patient who has been treated for
OQPVJQTOQTGYKVJCPVK6$FTWIUKPVJGRCUV4GVTGCVOGPV
ECUGUCTGHWTVJGTENCUUKßGFD[VJGQWVEQOGQHVJGKTOQUVTGEGPV
course of treatment into four categories.
1. 4elapse patients have previously been treated for TB, were
declared cured or treatment completed at the end of their
most recent course of treatment, and are now diagnosed
with a recurrent episode of TB (either a true relapse or a new
episode of TB caused by reinfection).
2. Treatment after failure patients have previously been
treated for TB and their most recent course of treatment
failed.
3. Treatment after loss to follow-up patients have previously
been treated for TB and were declared ‘lost to follow-up’
at the end of their most recent course of treatment (this
ECVGIQT[EQTTGURQPFUVQVJGÁFGHCWNVGFÂECVGIQT[FGßPGFKP
Box 3.1).
4. Other preXiously treated patients are those who have
previously been treated for TB but whose outcome after
their most recent course of treatment is unknown or
undocumented.
Case of multidrug-resistant TB (MDR-TB) #UFGßPGFKP
Box 3.1
Case of rifampicin-resistant TB (RR-TB) A patient with
TB that is resistant to rifampicin detected using phenotypic
or genotypic methods, with or without resistance to other
anti-TB drugs. It includes any resistance to rifampicin, whether
mono-resistance, multidrug resistance, polydrug resistance or
extensive drug resistance.
a &GßPKVKQPUCPFTGRQTVKPIHTCOGYQTMHQTVWDGTEWNQUKU¿TGXKUKQP9*1
*6/6$)GPGXC9QTNF*GCNVJ1TICPK\CVKQP#XCKNCDNGCV
YYYYJQKPVKTKUDKVUVTGCOAGPIRFH
3.2 Contribution of public–public and
public–private mix initiatives
and community-based TB activities to
TB case notißcations in 2012
2WDNKE¿RWDNKECPFRWDNKE¿RTKXCVGOKZ22/
Ensuring proper diagnosis, standardized treatment and
prompt notication of all TB cases to NTPs requires collab-
oration with the full range of health care providers. Engag-
ing all care providers in TB care and control is component 4
of the Stop TB Strategy (Chapter 1). Its two subcomponents
are:
involving all public, voluntary, corporate and private
providers through PPM approaches; and
promoting the International Standards for Tuberculosis
Care.1
Many countries have scaled up PPM initiatives. Demon-
strating progress in terms of the contribution of non-NTP
providers to total case notications requires systematic
recording of the source of referral and place of TB treat-
ment locally, and reporting and analysis of aggregated data
nationally.2 In 2013, 73 countries reported summary data
to WHO, and data for 29 of these countries (including 14
HBCs) are shown in Table 3.3. In most of these countries,
PPM initiatives contributed about 10% to 40% of total noti-
cations.
Considering that the private medical sector in Africa is
much smaller compared with that in Asia, the contribution
of private-for-prot and not-for-prot providers in Ethio-
pia, Kenya, Nigeria and the United Republic of Tanzania is
noteworthy. Progress in parts of Asia is also noticeable –
almost every fourth case in Indonesia and the Philippines
was notied by non-NTP care providers in 2012. Large pub-
lic sector hospitals have contributed sizeable proportions of
cases in China and Indonesia as well as in the Philippines,
and engagement of large hospitals is one of the major strat-
egies required to improve detection and notication of TB
1 http://www.istcweb.org/ISTC_Documents.html
2 WHO recommends that the source of referral and the place of treat-
ment should be routinely recorded and reported.
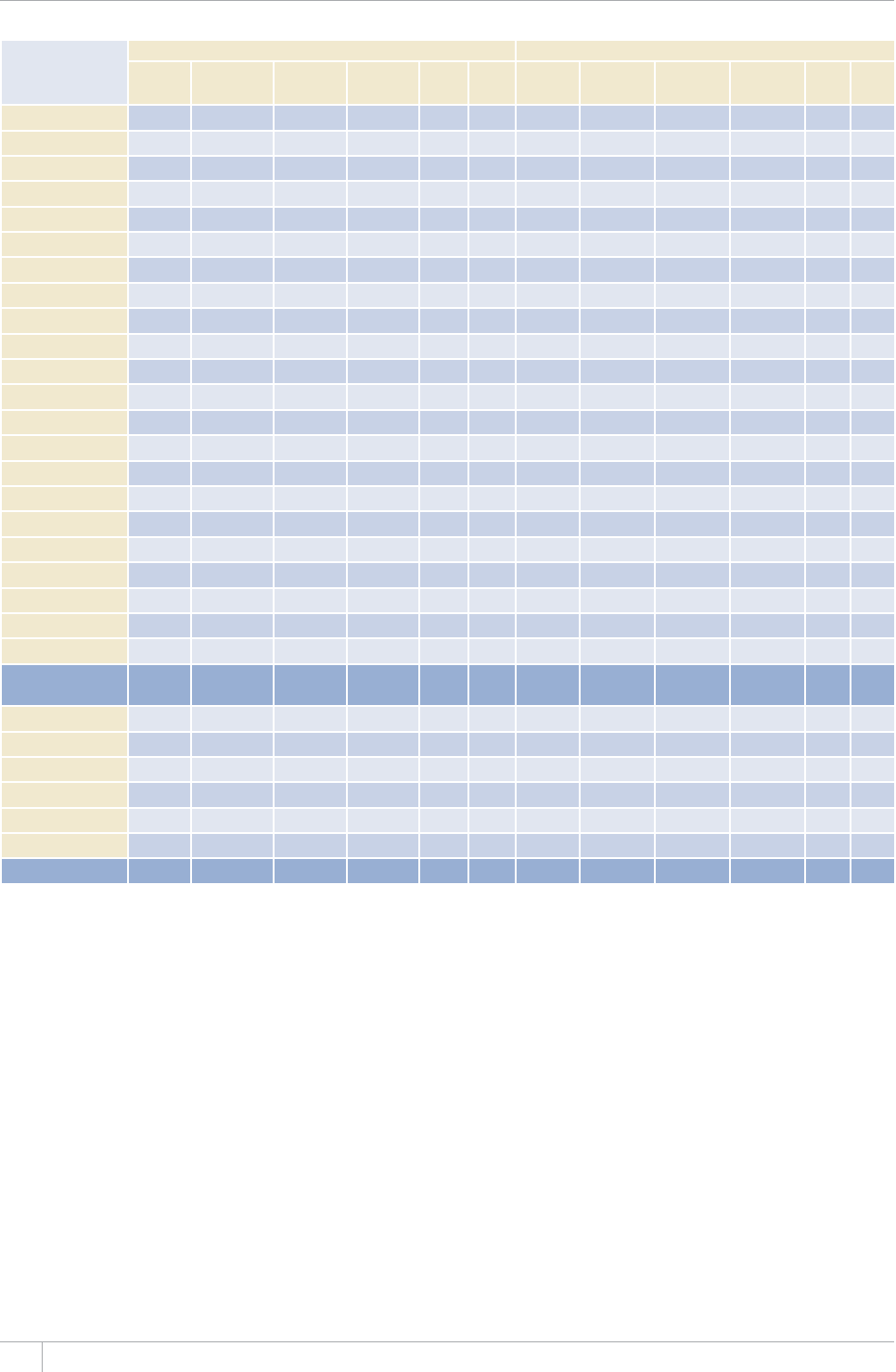
32 GLOBAL TUBERCULOSIS REPORT 2013
TABLE 3.2
TB case notißcations by age and sex, 2012
0'95/'#4215+6+8'%#5'5 ALL NEW CASESa
¿;'#45 ¿;'#45 ¿;'#45 Ü;'#45
% AGED
;'#45
/#.'
FEMALE
4#6+1 ¿;'#45 ¿;'#45 ¿;'#45 Ü;'#45
% AGED
;'#45
/#.'
FEMALE
4#6+1
Afghanistan 3 106 1 156 4 0.5 15 0.6
Bangladesh 966 56 209 34 674 14 941 1.9 20 602 3 1.6
$TC\KN 25 209 11 129 3 190 1 2.3 42 306 20 056 6 322 3 2.0
Cambodia 53 6 000 6 064 2 724 1.2 – –
China 1 091 110 614 65 960 2.5 5 625 397 615 2.2
&4%QPIQ 47 722 17 066 41.3 – –
Ethiopia – – 16 1.2
India 12 957 22.2 7 –
Indonesia 1 703 116 326 70 491 13 799 1.5 27 343 172 706 100 254 22 579 1.4
Kenya 996 29 779 5 114 31.6 3 994 7 1.4
/Q\CODKSWG –– 13–
Myanmar 24 076 14 405 4 090 1.9 – –
Nigeria 10 571 2 553 2 1.6 – –
Pakistan 3 947 66 901 29 149 41.1 10 1.0
Philippines 1 032 49 736 29 617 6 943 1 2.3 – 1.9
4WUUKCP(GFGTCVKQP 17 039 2.7 26 302 5 254 4 2.2
South Africa 2 650 24 964 4 151 2 1.3 13 1.2
Thailand 117 13 525 11 435 5 921 2.4 – –
Uganda 636 4 777 914 3 ––
746CP\CPKC 490 5 257 1 536 2 91.5
Viet Nam 142 23 310 3.0 – –
<KODCDYG 293 432 2 1.3 2 911 23 541 5 954 1.2
High-burden
countries 32 952 1 172 486 604 196 205 733 2 1.9 231 674 1 724 885 894 852 343 231 6 1.7
#(4 14 340 3 1.5 97 629 571 919 167 236 91.3
#/4 2 012 61 956 27 462 2 1.7 9 646 19 974 5 1.6
'/4 5 641 31.2 133 536 53 351 21 545 10 1.0
'74 325 24 440 7 355 2.4 10 042 60 455 20 575 5 1.9
5'#4 17 116 617 926 336 069 94 741 2 2.0 739 149 397 219 112 503 6
924 2 693 230 572 172 377 2.4 13 945 195 965 1 2.1
Global 42 127 1 451 897 710 738 241 395 2 1.9 292 295 2 171 779 1 075 428 409 559 6 1.7
Blank cells indicate data that could not be reported for the age categories shown.
– indicates values that cannot be calculated.
a Numbers in each age category are only shown if data were reported for all four age categories for each category of TB case. For this reason, there are small discrepancies
between numbers presented in this table and in the tables that appear in Box 2.2 of Chapter 2.
cases. Experience from a project that was recently complet-
ed in ve countries is proled in Box 3.4.
Approaches to engage non-NTP care providers vary
according to the local context, but there are some import-
ant cross-cutting elements. One is provision of standard-
ized care by non-NTP providers according to national
guidelines, in return for provision of free anti-TB drugs,
supervision and quality assurance, and nancial or non-
nancial incentives by NTPs. A second is the use of the Inter-
national Standards for Tuberculosis Care, which facilitates the
use of best practices in TB diagnosis and treatment among
all care providers, especially those in the private sector. In
the European Region and the Region of the Americas, con-
tributions to case notications from public sector provid-
ers outside the purview of the Ministry of Health, such as
social security organizations and prison health services, are
relatively large.
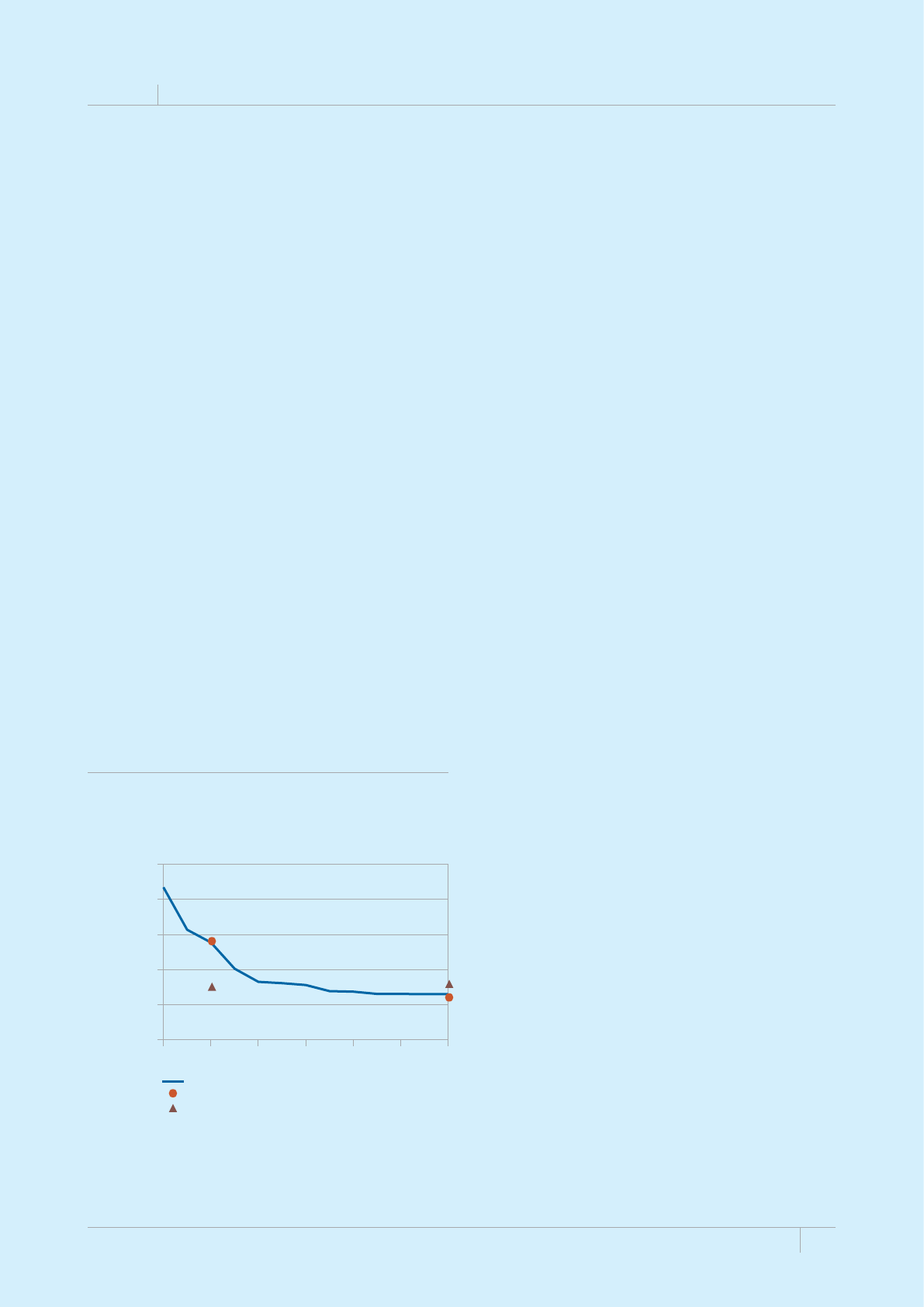
33GLOBAL TUBERCULOSIS REPORT 2013
BOX 3.3
TB and prisons
As an airborne disease, TB thrives in the crowded, poorly-
ventilated environments found in prisons in many parts of the
world. Conditions of prison life including malnutrition and stress
can also contribute to a higher risk of developing TB disease,
and inadequate or inaccessible medical care can lead to poor
treatment outcomes and acquisition of resistance. Prisoners
FKURTQRQTVKQPCVGN[EQOGHTQOOCTIKPCNK\GFUQEKQGEQPQOKE
backgrounds – including substance users, homeless people,
people with mental illness, ethnic minorities, asylum seekers and
immigrants – and therefore often enter the prison system with
an existing high prevalence of TB infection or even with active
disease. Prisons also contribute to overall TB burden in that they
CTGPQVGPVKTGN[ENQUGFU[UVGOU6$ECPDGURTGCFVQRTKUQPUVCHH
and visitors, and at some point most prisoners are released into
the general population.
6JG9*1'WTQRGCP4GIKQPKUEWTTGPVN[VJGQPN[TGIKQPVJCV
systematically collects and analyses data from Member States
on the burden of TB in prisons.a As is likely the situation in
most countries around the world, the burden of TB in prisons in
European countries is disproportionately high and often makes
CEQPUKFGTCDNGEQPVTKDWVKQPVQQXGTCNNEQWPVT[ECUGPQVKßECVKQPU
0QVKßECVKQPTCVGUQHPGY6$ECUGUKPRTKUQPUKPCNNTGRQTVKPI
countries were multiple times higher than the rates found in
VJGIGPGTCNRQRWNCVKQPTGNCVKXGTKUMUTCPIKPIHTQOVQ
CPFYGTGQXGTRGTFGVCKPGGUKP#\GTDCKLCP
)GQTIKCCPF-[TI[\UVCPCPFRGT
detainees, respectively) in 2011. Among reporting countries, case
PQVKßECVKQPUHTQORTKUQPUCEEQWPVGFHQTQXGTQHPCVKQPCN
PQVKßECVKQPUQHPGY6$ECUGUKP)GQTIKCCPFVJG4WUUKCP
Federation (11%) in 2011. Given that some countries in the region
JCXGPQVDGGPCDNGVQTGRQTVFCVCQP6$PQVKßECVKQPUKPRTKUQPU
and that limited data are available on trends, the contribution of
6$PQVKßECVKQPUKPRTKUQPUVQQXGTCNN6$PQVKßECVKQPUKPVJGTGIKQP
is uncertain.
To reduce the burden of TB in prisons, a comprehensive
package of measures is required.b,c These include early diagnosis
using systematic screeningd and rapid diagnostics, proper
infection control, improved living conditions and nutrition,
supervised and complete TB treatment with appropriate drugs,
treatment of comorbidities including HIV, diabetes, hepatitis and
substance use disorders, and continuity of care in the public sector
when a prisoner under treatment is released.
6JG4WUUKCP(GFGTCVKQPYCUUWEEGUUHWNKPKPVTQFWEKPIUGXGTCN
OGCUWTGUVJCVUKIPKßECPVN[TGFWEGFVJGDWTFGPQH6$KPKVU
penitentiary system (Figure B3.3.1).e By reinforcing systematic
screening, improving infection control measures, strengthening
treatment, and building cooperation between the Ministry of
Justice, Ministry of Health institutions, and international partners,
6$PQVKßECVKQPTCVGUFGETGCUGFUJCTRN[HTQOECUGURGT
100 000 detainees in 1999 (i.e., TB was detected in 1 of 25
FGVCKPGGUKPVQECUGURGTFGVCKPGGUKP
6JGFGETGCUGUKPEGJCUDGGPITCFWCNTGàGEVKPIVJG
continuing challenges facing TB control in the penitentiary sector,
including rising rates of TB/HIV coinfection and drug-resistant
TB, as well as the continued concentration of socioeconomically
OCTIKPCNK\GFRGQRNGGPVGTKPIVJGRTKUQPU[UVGO1HPQVGKUVJG
JKIJGTPQVKßECVKQPTCVGHQWPFKPVJGRTGVTKCNFGVGPVKQPEGPVTGU
EQORCTGFYKVJEQTTGEVKQPCNHCEKNKVKGUKPEQORCTGFYKVJ
RGTFGVCKPGGUTGURGEVKXGN[TGàGEVKPIKPRCTVVJG
underlying high prevalence of TB infection and disease among
UQEKQGEQPQOKECNN[OCTIKPCNK\GFRGQRNGYJQGPVGTVJGRTGVTKCN
detention centres from the general population.
In Eastern Europe, drug-resistant TB has been associated with
detention and in many countries prisons have had to deal with
UWDUVCPVKCNECUGNQCFUQH/&46$RCVKGPVUf–h The provision of
GHHGEVKXG/&46$ECTGHQTRTKUQPKPOCVGUKUVJGTGHQTGKORQTVCPV
The possibility of close monitoring of imprisoned patients may
also be conducive to achievement of good treatment outcomes.
(QTGZCORNGFCVCHTQOVJGRGPKVGPVKCT[UGEVQTKP#\GTDCKLCPUJQY
VTGCVOGPVUWEEGUUTCVGUKPVJGTCPIG¿KPVJG¿
patient cohorts treated in accordance with WHO-recommended
standards.i
a Tuberculosis surXeillance and monitorinI in Europe 2012. Stockholm, European
%GPVTGHQT&KUGCUG2TGXGPVKQPCPF%QPVTQN9*14GIKQPCN1HßEGHQT'WTQRG
b See Guidelines for the control of tuberculosis in prisons. Geneva, World Health
1TICPK\CVKQP9*16$
c Dara M, Chadha SS, Melchers NV, van den Hombergh J, Gurbanova E, Al-Darraji H,
van der Meer JBW. Time to act to prevent and control tuberculosis among inmates.
International ,ournal of Tuberculosis and LunI &isease,CP¿
d Systematic screeninI for actiXe tuberculosis principles and recommendations.
)GPGXC9QTNF*GCNVJ1TICPK\CVKQP9*1*6/6$
e Tuberculosis in the 4ussian (ederation 2011 an analytical reXiew of statistical
indicators used in the 4ussian (ederation and in the worldKP4WUUKCP/QUEQY
/KPKUVT[QH*GCNVJQHVJG4WUUKCP(GFGTCVKQPGVCN
f 5MTCJKPC#*WTGXKEJ*<CNWVUMC[C#GVCN/WNVKFTWITGUKUVCPVVWDGTEWNQUKUKP
$GNCTWUVJGUK\GQHVJGRTQDNGOCPFCUUQEKCVGFTKUMHCEVQTUBulletin of the World
*ealth OrIanization¿
g #GTVU#*CDQW\KV//UEJKNCF\G.GVCN2WNOQPCT[VWDGTEWNQUKUKPRTKUQPUQH
VJGGZ7554UVCVGQH)GQTIKCTGUWNVUQHCPCVKQPYKFGRTGXCNGPEGUWTXG[COQPI
sentenced inmates. International ,ournal of Tuberculosis and LunI &isease, 2000
&GE¿
h 5JKP552CUGEJPKMQX#&)GNOCPQXC+;2GTGOKVKP))5VTGNKU#-/KUJWUVKP5
GVCN6TGCVOGPVQWVEQOGUKPCPKPVGITCVGFEKXKNKCPCPFRTKUQP/&46$VTGCVOGPV
RTQITCOKP4WUUKCInternational ,ournal of Tuberculosis and LunI &isease, 2006
#RT¿
i 4eXiew of tuberculosis preXention control and care ina#zerbaiLan. Copenhagen,
9QTNF*GCNVJ1TICPK\CVKQP
TB notification rate per 100 000
detainees per year
1999 2001 2003 2005 2007 2009 2011
0
1000
2000
3000
4000
5000
Combined prison facilities
Correctional facilities
Pre-trial detention centres
FIGURE B3.3.1
6$PQVKßECVKQPTCVGKPVJGRTKUQPHCEKNKVKGUQHVJG
4WUUKCP(GFGTCVKQP¿QXGTCNNCPF
disaggregated by pre-trial detention centres and
correctional facilities
34 GLOBAL TUBERCULOSIS REPORT 2013
TABLE 3.3
Contribution of public-private and public-public mix (PPM) to notißcations of TB cases in 29 countries, 2012
%17064; 6;2'51(%#4'2418+&'45'0)#)'&
07/$'41(6$%#5'5
016+(+'&$;27$.+%010062
%#4'2418+&'45a
07/$'41(6$%#5'5
016+(+'&$;24+8#6'%#4'
2418+&'45b
%1064+$76+1061616#.
NOTIFICATIONS OF TB CASES
IN 2012 (%)
#(4+%#04')+10a
Ethiopia Diverse private providers – 17 133 12
Ghana Diverse non-NTP public and private providers 1 107 13
Kenya Private clinics and hospitals, and prisons 10 364 12
Lesotho Diverse private providers – 1 044 10
Nigeria Public non-NTP and NGO hospitals and private
clinics 14 096 24
5YC\KNCPF Diverse non-NTP public and private providers 33
7PKVGF4GRWDNKEQH6CP\CPKC 2TKXCVGHCEKNKVKGUCPFHCKVJDCUGFQTICPK\CVKQPU – 13 734 22
REGION OF THE AMERICASa
El Salvador Diverse non-NTP public and private providers 761 50 40
Peru 5QEKCNUGEWTKV[QTICPK\CVKQPUCPFQVJGTPQP062
public providers 6 576 – 22
EASTERN MEDITERRANEAN REGIONa
Afghanistan 2TKXCVGENKPKEUJQURKVCNUNCDQTCVQTKGUaCPF
pharmacies 1 362 12
Egypt *GCNVJKPUWTCPEGQTICPK\CVKQPU0)1UCPFQVJGT
public non-NTP providers 1 993 213 26
+TCP+UNCOKE4GRWDNKEQH *GCNVJKPUWTCPEGQTICPK\CVKQPURTKUQPUOKNKVCT[CPF
private care providers 1 205 40
Iraq Diverse non-NTP public and private providers 2 693 65
Pakistan Private clinics and hospitals 925 56 363 21
Sudan Diverse private and non-NTP public providers 450 1 475 10
5[TKCP#TCD4GRWDNKE Diverse private and non-NTP public providers 175 2 400
;GOGP Public hospitals including university, military and
police hospitals, prisons and private hospitals –35
EUROPEAN REGIONa
Georgia Diverse non-NTP public and private providers, and
prisons 673
Tajikistan Diverse non-NTP public providers and prisons 1 549 – 24
SOUTH-EAST ASIA REGION
Bangladesh Diverse private, non-NTP public and NGO providers 2 429 14 934 10
IndiacDiverse private, non-NTP public and NGO providers 13 572 3 533 –
Indonesia Public and private hospitals 77 376 5 432 25
Myanmar Diverse private, non-NTP public and NGO providers 23
Nepal Diverse private providers – 5 366 15
Sri Lanka Diverse non-NTP public and private providers 5 004 445 60
Thailand Diverse non-NTP public and private providers 1 532 1 267 4.6
WESTERN PACIFIC REGION
China General public hospitals –44
Philippines Private clinics and hospitals 36 744 24
Viet Nam Diverse non-NTP public and private providers 3 404 4 724
a Includes all contributions from non-NTP providers, including public hospitals, public medical colleges, prisons/detention centres, military facilities, railways and public health
KPUWTCPEGQTICPK\CVKQPU
b 2TKXCVGUGEVQTRTQXKFGTUKPENWFGRTKXCVGKPFKXKFWCNCPFKPUVKVWVKQPCNRTQXKFGTUEQTRQTCVGDWUKPGUUUGEVQTRTQXKFGTUOKUUKQPJQURKVCNUPQPIQXGTPOGPVCNQTICPK\CVKQPUCPF
HCKVJDCUGFQTICPK\CVKQPU
c Data for India are for smear-positive cases of pulmonary TB in 14 cities where PPM surveillance is in place.

35GLOBAL TUBERCULOSIS REPORT 2013
BOX 3.4
Engaging hospitals to improve TB care and prevention
In 2009, WHO initiated a project to help intensify TB case
FGVGEVKQPKPßXGEQWPVTKGUKP#HTKECCPF#UKC+PVGPUKßGF
hospital engagement, mainly targeting large hospitals in urban
CTGCUYCUVJGOCKPKPVGTXGPVKQPKPCNNßXGEQWPVTKGU6JTQWIJ
a consultative process involving NTPs, departments responsible
for hospitals within the ministries of health, directors of
RCTVKEKRCVKPIJQURKVCNUCPF9*1CVQVCNQHJQURKVCNU
covering a total population of 10 million were involved. This
KPENWFGFJQURKVCNUKP-KPUJCUCKPVJG&GOQETCVKE4GRWDNKEQH
the Congo; 10 in Accra, Ghana; 17 in Manila, The Philippines;
KP5YC\KNCPFCPFVJGNCTIGUVPCVKQPCNIGPGTCNJQURKVCNU
KPVJTGGEKVKGUKP8KGVa0CO*CPQK*Q%JK/KPJ%KV[CPF*WG
The initiative was funded by the Department of Foreign Affairs,
Trade and Development of Canada.
Prior to the project, hospitals provided TB diagnosis
and treatment for many patients without following national
IWKFGNKPGUQTJCXKPIHQTOCNTGHGTTCNCPFPQVKßECVKQPTQWVKPGU
6JGURGEKßEQDLGEVKXGUQHVJGRTQLGEVYGTGVQKORTQXG6$
diagnosis and management of patients presenting to hospitals
through setting up mechanisms for internal coordination,
and to improve external networking to help intensify TB case
FGVGEVKQPCPFPQVKßECVKQP6JGOCKPCEVKXKVKGUYGTGKORTQXKPI
KFGPVKßECVKQPQHRGQRNGYKVJUWURGEVGF6$UVCPFCTFK\CVKQP
of diagnostic routines and introduction of external quality
assurance in hospital laboratories; establishing a ‘hospital
DOTS unit’; a systematic approach to internal referrals so that
cases diagnosed in hospitals would be referred to the hospital
&165WPKVHQTOCNK\CVKQPQHTQWVKPGUHQTGZVGTPCNTGHGTTCNQH
cases to health centres and feedback about referrals from
health centres; ensuring proper treatment and follow up of
patients started on treatment in hospitals; and introduction of
UVCPFCTFK\GFTGEQTFKPICPFTGRQTVKPI
After successful implementation of project activities, total
JQURKVCNPQVKßECVKQPUKPETGCUGFHTQOCDQWVRGT[GCT
CETQUUVJGßXGUKVGUDGHQTGVJGRTQLGEVVQCDQWVRGT[GCT
in 2012. The documented number of people tested for TB with
a bacteriological test increased in all sites, and the average
increase was roughly fourfold (Figure B3.4.1).
Documentation of referrals for treatment and feedback to
EQPßTOVTGCVOGPVKPKVKCVKQPFGOQPUVTCVGFVJCVVJGNQUUGUCHVGT
referral were very large at baseline in the two Asian countries
VJG2JKNKRRKPGUCPF8KGVa0CO6JGUGNQUUGUYGTGUWDUVCPVKCNN[
reduced by the end of the third project year through improved
communication between hospitals and the primary health care
facilities to which they were making referrals. Treatment success
rates among those started on treatment in hospitals were
similar to those reported by the NTP.
This project helped to describe a baseline situation in
which hospitals were not engaged. It then demonstrated that
it is possible to proactively engage hospitals and align their
services to national guidelines and in turn to improve detection
QH6$ECUGUCPFPQVKßECVKQPVQ062U#NNEQWPVTKGUJCXGGKVJGT
developed new national policies for hospital engagement or are
in the process of doing so, based on the project results. Similar
efforts are anticipated and needed in other countries.
FIGURE B3.4.1
Trends in bacteriological testing for TB and TB case
PQVKßECVKQPUKPRTQLGEVUKVGU¿
Number
0
20 000
40 000
60 000
80 000
100 000
2010 2011 2012
Number of bacteriological tests done
Total number of TB cases diagnosed and notified
Number of bacteriologically-positive cases diagnosed
%QOOWPKV[EQPVTKDWVKQPUVQ6$PQVKßECVKQPU
and treatment support
Community-based TB activities can be dened as activ-
ities that are conducted outside the premises of formal
health facilities, within community-based structures (for
example, schools and places of worship) and homesteads.
Such activities can be implemented by community health
workers1 and community volunteers,2 regardless of wheth-
er they are employed and supervised by a government
department or by a nongovernmental organization, and
make an important contribution to health services includ-
ing prevention, diagnosis, improved treatment adherence,
care and support. In the specic context of TB, commu-
nity activities can help to increase case notications and
improve treatment outcomes, especially in settings where
people with TB have poor access to formal health services.
As shown in section 3.3, approximately one third of people
with TB are diagnosed but not reported to national surveil-
lance systems, or not diagnosed at all.
Accurate documentation of the contributions of commu-
nities to TB notications and treatment support has been
challenging. One reason has been the lack of standardiza-
tion of indicators that can be used for routine recording and
1 Community health workers can be dened as people with some
formal education who have been given training to contribute to
community-based health services, including TB prevention and
patient care and support. eir prole, roles and responsibilities
vary greatly among countries, and their time is often compensated
by incentives in kind or in cash.
2 Community volunteers can be dened as community members who
have been systematically sensitized about TB prevention and care,
either through a short, specic training scheme or through repeat-
ed, regular contact sessions with professional health workers.
36 GLOBAL TUBERCULOSIS REPORT 2013
BOX 3.5
The ENGAGE-TB approach
The ENGAGE-TB approacha describes the need for
PQPIQXGTPOGPVCNQTICPK\CVKQPUCPFQVJGTEKXKNUQEKGV[
QTICPK\CVKQPUVQKPVGITCVGEQOOWPKV[DCUGF6$CEVKXKVKGU
KPVQVJGKTGZKUVKPIYQTM2KNQVRTQLGEVUKPßXG#HTKECP
EQWPVTKGUVJG&GOQETCVKE4GRWDNKEQHVJG%QPIQ'VJKQRKC
-GP[C5QWVJ#HTKECCPFVJG7PKVGF4GRWDNKEQH6CP\CPKC
are showing promising results. Selected nongovernmental
QTICPK\CVKQPUKPVJGUGEQWPVTKGUJCXGUVCTVGFKPVGITCVKPI
TB services into community-based programmes for HIV,
maternal, newborn and child health (MNCH), and cancer
UETGGPKPIYKVJßPCPEKCNUWRRQTVHTQOVJG$TKUVQN/[GTU
Squibb Foundation.
NGO %17064; 241,'%6(1%75
Femmeplus Democratic
4GRWDNKEQH
the Congo
Integration of TB services into
community-based HIV activities
in two major cities (Kinshasa and
Kikwit)
AMREF Ethiopia Integration of TB/HIV services
into community-based MNCH
activities in a pastoralist region
CUAMM Ethiopia Integrated community-based TB,
HIV and cancer screening project
Save the
Children
Ethiopia Integration of community-based
TB/HIV services into MNCH
programmes in pastoralist
communities
Centre for
Positive Care
South Africa Strengthening integration of
TB into community-based HIV
activities
2CVJßPFGT 746CP\CPKC Integration of TB services into
community-based HIV services
0)1PQPIQXGTPOGPVCNQTICPK\CVKQP
The challenge is to scale up these experiences and
UKIPKßECPVN[KPETGCUGVJGPWODGTQHEQOOWPKV[DCUGF
workers and volunteers who are providing screening for TB,
referring those who might have TB for diagnosis and then
providing follow-up care and support to those diagnosed
YKVJVJGFKUGCUG9*1KUßPCNK\KPICPKORNGOGPVCVKQP
manual that will help to inform nongovernmental
QTICPK\CVKQPUCPF062UCDQWVJQYVJG[ECPYQTMVQIGVJGTVQ
implement integrated community-based TB activities, with a
RCTVKEWNCTHQEWUQPPQPIQXGTPOGPVCNQTICPK\CVKQPUYQTMKPI
on MNCH, HIV, primary health care, agriculture, livelihood
development and education services.
a ENG#GE-TB – InteIratinI community-based tuberculosis actiXities into
the work of nonIoXernmental orIanizations. Geneva, World Health
1TICPK\CVKQP
reporting. To address this challenge, WHO recently devel-
oped a minimum set of standardized indicators as part of
its ENGAGE-TB approach (Box 3.5). In 2013, these indi-
cators were used to collect standardized, comparable data
from a set of 13 countries in which data were known to be
routinely recorded and reported in at least some geograph-
ical areas.1 Data collection was undertaken separately from
the main round of global TB data collection, since most
countries are not routinely recording and reporting such
data and they are not relevant in all settings.
Among the 13 countries (Table 3.4), notied TB patients
referred from the community as a share of total notica-
tions in the areas covered by reporting ranged from 2% in
Myanmar (in 92/330 districts) to 33% in Ethiopia (in 98
out of 821 districts). It is possible that these gures are
an underestimate, pending optimization of recording and
reporting systems. Nonetheless, the nding that the con-
tribution of communities in referring people with TB was
under 10% in several countries suggests that there may
be opportunities to use untapped community resources in
TB prevention, diagnosis and treatment. In settings where
access to formal health services is limited, more emphasis
in policy and practice on the role of community referrals
of people with presumptive TB as early as possible is war-
ranted.
e share of patients receiving treatment support in the
community was generally high: for example, 50% country-
wide in India and 88% countrywide in Kenya.
Kenya also provides an interesting example of the
untapped potential of communities. While 88% of all TB
patients were reported as having received support for treat-
ment adherence, demonstrating the spread and reach of
community workers and volunteers in the country, only 5%
of TB case notications had been referred by community
members. is suggests that more could be done to increase
community engagement in and contribution to TB screen-
ing and referral.
It is evident that data on community contributions to
referrals and treatment adherence are not collected uni-
formly or systematically, even in the 13 countries shown
in Table 3.4. Only three of the 13 countries reported data
for both indicators that covered all districts in the country
(Burkina Faso, Kenya and Rwanda). e remaining coun-
tries reported data that covered only parts of the country
(sometimes very limited areas) or data were not available
for both indicators. Better understanding of the contri-
bution of communities to TB services will require more
routine collection of data; this is of greatest relevance in
settings where community contributions are considered a
necessary and integral part of TB services.
1 ere was no attempt to compile data about the contribution of
communities to programme design and implementation (including
advocacy activities at local levels). Such data are not routinely avail-
able.
37GLOBAL TUBERCULOSIS REPORT 2013
1 www.who.int/tb/data
2 e CDR is actually a ratio rather than a rate, but the term ‘rate’ has
become standard terminology in the context of this indicator.
3 It is approximate because of uncertainty in the underlying inci-
dence of TB and because notied cases are not necessarily a subset
of incident cases that occurred in the same year; see Chapter 2 for
further discussion.
TABLE 3.4
Community contributions to TB case notißcations and treatment adherence support, 2011–2012
%17064;
NOTIFIED PATIENTS
(41/%1//70+6;4'('44#.5
2#6+'0659*14'%'+8'&64'#6/'065722146+06*'%1//70+6;
%1*14670.'5552'%+(+'&
5*#4'1(616#.016+(+%#6+105+0#4'#5
(149*+%*#9'4'4'2146'& )'1)4#2*+%%18'4#)'1(# 5*#4'1(%1*146+0#4'#5(149*+%*
#9'4'4'2146'& )'1)4#2*+%%18'4#)'1(#
Burkina Faso 9% All districts 33% All districts
Côte d’Ivoire 16% FKUVTKEVU Not available
&4%QPIQ 10% 45/515 districts 3% FKUVTKEVU
Ethiopia 33% FKUVTKEVU 40%aFKUVTKEVU
India 3% 374/662 districts 50% All districts
Kenya 5% All districts All districts
Malawi 20% FKUVTKEVU 91% FKUVTKEVU
Myanmar 2% 92/330 districts 2% 92/330 districts
Nigeria Not available 5% 36/774 districts
4YCPFC All districts 46% All districts
Senegal 6% All districts Not available
Uganda Not available 35% All districts
746CP\CPKC 14% 63/162 districts All districts
a Data are for the 2012 cohort.
FIGURE 3.1
Global trends in case notißcation (black) and
estimated TB incidence (green) rates, 1990–2012.
%CUGPQVKßECVKQPUKPENWFGPGYCPFTGNCRUGECUGUCNNHQTOU
1990 1995 2000 2005 2012
0
50
100
150
200
Rate per 100 000 population per year
3.3 Trends in case notißcations since 1990
and estimates of the case detection rate
Globally, the number of TB cases diagnosed and notied per
100000 population was relatively stable between 1990 and
2000, rose sharply between 2000 and 2008 and has subse-
quently started to fall slowly (Figure 3.1). Globally and in
all WHO regions, a clear gap exists between the numbers of
notied cases and the estimated numbers of incident cases,
although this has narrowed in the past decade globally and
in all six WHO regions (Figure 3.2). Trends in the 22 HBCs
are shown in Figure 3.3, and for other countries are illus-
trated in country proles that are available online.1
e case detection rate (CDR)2 for TB is an indicator
that is included within the MDGs (Chapter 1). For a giv-
en country and year, the CDR is calculated as the number
of new and relapse TB cases (see Box 3.1 for denitions)
that were notied by NTPs (Table 3.1), divided by the esti-
mated number of incident cases of TB that year. e CDR is
expressed as a percentage; it gives an approximate3 indica-
tion of the proportion of all incident TB cases that are actu-
ally diagnosed, reported to NTPs and started on treatment.
e best estimate of the CDR for all forms of TB globally
in 2012 was 66% (range, 64–69%), up from 53–59% in 2005
and 38–43% in 1995 – the year in which the DOTS strategy
began to be introduced and expanded (Table 3.5). e high-
est CDRs in 2012 were estimated to be in the Region of the
Americas (best estimate 79%; range, 74–85%), the Western
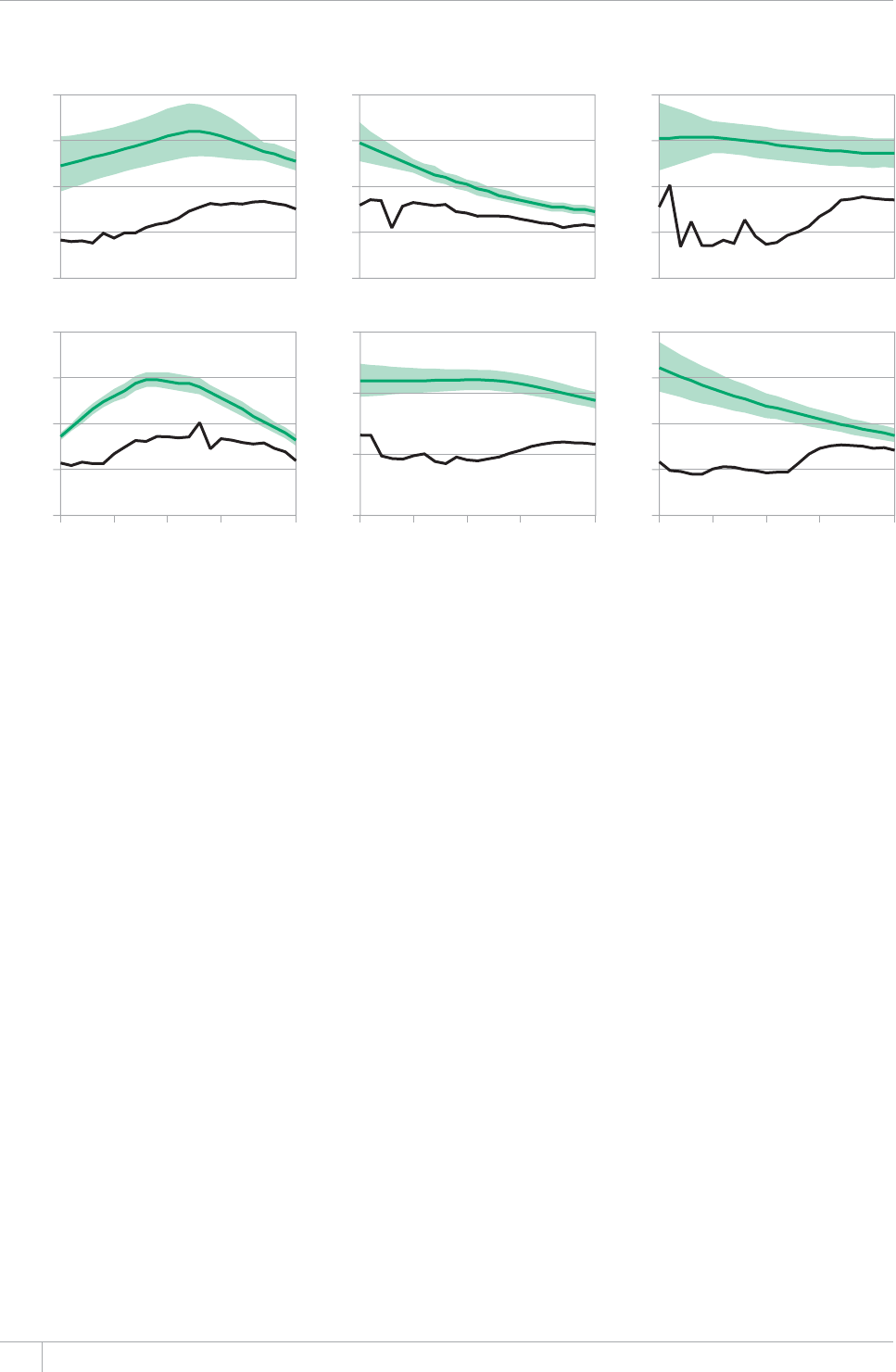
GLOBAL TUBERCULOSIS REPORT 2013
FIGURE 3.2
Case notißcation and estimated TB incidence rates by WHO region, 1990–2012. 4GIKQPCNVTGPFUKPECUG
PQVKßECVKQPTCVGUPGYCPFTGNCRUGECUGUCNNHQTOUblack) and estimated TB incidence rates (green). Shaded areas
represent uncertainty bands.
Rate per 100 000 population per year
0
100
200
300
400
0
25
50
75
100
0
20
40
60
80
0
100
200
300
0
40
80
120
160
0
50
100
150
200
1990 1995 2000 2005 2012 1990 1995 2000 2005 20121990 1995 2000 2005 2012
Africa The Americas Eastern Mediterranean
Europe South-East Asia Western Pacific
Pacic Region (best estimate 81%; range, 75–89%) and the
European Region (best estimate 74%; range, 70–79%). e
other regions had estimated CDRs in the range of 55–71%,
with best estimates of around 60%. All regions have
improved their estimated CDRs since the mid-1990s, with
improvements particularly evident since 2000. Among the
22 HBCs, the highest rates of case detection in 2012 were
estimated to be in Brazil, China, Kenya, the Philippines and
the Russian Federation. e lowest rates, with best esti-
mates of around 50%, were in Afghanistan, Bangladesh,
the Democratic Republic of the Congo, Mozambique, Nige-
ria and Zimbabwe.
e gap between notications to national surveillance
systems and the true number of incident cases can be
explained by two factors. e rst is underreporting of
diagnosed TB cases, for example because private sector pro-
viders fail to notify cases. e second is under-diagnosis of
people with TB for reasons such as poor access to health
care and failure to recognize TB signs and symptoms and
test for TB when people do present to health care facilities.
Achieving the goal of universal health coverage, imple-
menting PPM initiatives such as those described in sec-
tion 3.2, and ensuring that there is an eective regulatory
framework that includes mandatory notication of cases
are essential to reduce underreporting and under-diagnosis.
A point-of-care diagnostics test would also help.
3.4 Treatment outcomes
Denitions of the categories used to report treatment out-
comes in this report are provided in Box 3.6. e updated
denitions that will be used from 2014 are explained in
Box 3.7.
3.4.1 New cases of smear-positive pulmonary TB
Data on treatment outcomes for new sputum smear-
positive cases of pulmonary TB are shown in Table 3.6 and
Figure 3.4. Globally, the rate of treatment success for the
2.6 million new cases of sputum smear-positive pulmonary
TB who were treated in the 2011 cohort was 87%. is was
the fth successive year that the target of 85% (rst set by
the World Health Assembly in 1991) was met or exceeded
globally. It is also impressive that as the size of the global
treatment cohort grew from 1.0 million in 1995 to 2.7 mil-
lion in 2009 and 2010 and 2.6 million in 2011, the treat-
ment success rate progressively improved.
Among the six WHO regions, three met or exceeded the
85% target: the Eastern Mediterranean Region, the South-
East Asia Region and the Western Pacic Region. e treat-
ment success rate was 82% in the African Region (where
there has been steady improvement since 1999), 78% in the
Region of the Americas (similar to the previous seven years)
and 65% in the European Region (where major eorts to
increase treatment success rates are needed).
Of the 22 HBCs, 16 reached or exceeded the 85% target
in 2011, including Ethiopia and Nigeria for the rst time.
Five HBCs reported lower rates of treatment success: Brazil
(76%), the Russian Federation (52%), South Africa (79%),
Uganda (77%) and Zimbabwe (81%). Nonetheless, among
these ve countries all except the Russian Federation sus-
tained their level or made progress compared with 2010.

39GLOBAL TUBERCULOSIS REPORT 2013
FIGURE 3.3
Case notißcation and estimated TB incidence rates, 22 high-burden countries, 1990–2012. Trends in case
PQVKßECVKQPTCVGUPGYCPFTGNCRUGECUGUCNNHQTOUblack) and estimated TB incidence rates (green). Shaded areas
represent uncertainty bands.
Afghanistan Bangladesh Brazil Cambodia China
DR Congo Ethiopia India Indonesia Kenya
Mozambique Myanmar Nigeria Pakistan Philippines
Russian Federation South Africa Thailand Uganda UR Tanzania
Viet Nam Zimbabwe
Rate per 100 000 population per year
0
100
200
300
0
100
200
300
400
500
0
250
500
750
1000
1250
0
50
100
150
200
0
100
200
300
400
0
250
500
750
1000
1250
0
250
500
750
1000
1250
0
50
100
150
200
250
0
250
500
750
1000
1250
0
100
200
300
0
100
200
300
400
500
600
0
200
400
600
0
100
200
300
400
0
200
400
600
0
200
400
600
800
0
100
200
300
100
200
300
0
100
200
300
400
0
100
200
300
400
0
50
100
150
0
250
500
750
1000
0
50
100
150
200
1990 1995 2000 2005 2012 1990 1995 2000 2005 2012
1990 1995 2000 2005 2012 1990 1995 2000 2005 2012 1990 1995 2000 2005 2012
0
FIGURE 3.4
Treatment outcomes by WHO region, 2011 cohorts
Global
WPR
SEAR
EUR
EMR
AMR
AFR
0% 20% 40% 60% 80% 100%
Global
WPR
SEAR
EUR
EMR
AMR
AFR
0% 20% 40% 60% 80% 100%
a. New smear-positive cases b. All new cases
Successfully treated Died Treatment failed Defaulted Not evaluated
40 GLOBAL TUBERCULOSIS REPORT 2013
TABLE 3.5
Estimates of the case detection rate for new and relapse cases (%), 1995–2012a
1995 2000 2005 2010 2012
BESTbLOW HIGH BEST LOW HIGH BEST LOW HIGH BEST LOW HIGH BEST LOW HIGH
Afghanistan ––– 15 22 46 39 57 52 44 63 52 44 63
Bangladesh 21 17 26 25 21 31 32 47 45 55 49 41 59
$TC\KN 79 66 97 74 62 91 72 100 69 99 69 99
Cambodia 23 19 30 27 22 34 52 44 63 64 56 75 66 57 77
China 32 27 39 33 74 65 76 79 100
&4%QPIQ 31 26 40 34 55 47 64 56 49 65 51 44 59
Ethiopia 11 7.2 33 22 55 32 66 49 93 64 49
India 595267494555494455605466595466
Indonesia 7.1 12 20 16 25 57 47 71 66 56 72 61
Kenya 61 56 66 72 67 77 76 79 76
/Q\CODKSWG 23 11 73 23 13 51 30 20 53 33 25 34 25 50
Myanmar 10 13 15 13 19 53 45 63 66 57 77 71 62
Nigeria 2.7 170 12 3.9 170 26 9.6 200 40 23 51 29 110
Pakistan 4.5 3.7 5.5 3.3 4.1 39 32 66 55 65 54
Philippines 40 47 39 57 53 44 65 65 54 79 71 100
4WUUKCP(GFGTCVKQP 60 51 70 75 65 66 56 71 70 96
South Africa 56 47 69 59 49 72 61 50 74 70 59 62 52 75
Thailand 59 50 72 32 27 39 57 69 79 66 95 76 64 92
Uganda 23 14 41 29 20 47 36 65 60 50 75 69 57
746CP\CPKC 59 51 69 60 77 74 69 77 72 79 74
Viet Nam 33 25 47 56 43 63 49 70 54 95 76 59 100
<KODCDYG 55 40 79 56 45 71 50 40 63 53 43 69 46 37 60
High-burden countries 38 36 42 39 37 42 55 51 58 66 63 69 66 63 70
#(4 32 27 39 39 33 52 44 61 60 56 65 59 55 64
#/4 67 63 72 70 65 75 75 71 76 71 79 74
'/4 23 21 26 25 22 43 54 64 57 72 63 56 71
'74 51 49 54 59 55 62 65 61 70 77 72 74 70 79
5'#4 44 40 49 41 44 50 46 53 61 56 65 62 66
924 37 32 42 39 35 44 70 63 77 73 75
Global 40 38 43 41 39 44 56 53 59 66 63 68 66 64 69
– indicates values that cannot be calculated.
a 'UVKOCVGUHQTCNN[GCTUCTGTGECNEWNCVGFCUPGYKPHQTOCVKQPDGEQOGUCXCKNCDNGCPFVGEJPKSWGUCTGTGßPGFUQVJG[OC[FKHHGTHTQOVJQUGRWDNKUJGFRTGXKQWUN[
b $GUVNQYCPFJKIJKPFKECVGDGUVGUVKOCVGUHQNNQYGFD[NQYGTCPFWRRGTDQWPFU6JGNQYGTCPFWRRGTDQWPFUCTGFGßPGFCUVJGVJCPFVJEGPVKNGUQHQWVEQOG
distributions produced in simulations.
Data for Mozambique were not reported to WHO, but the
level in 2010 was 85%. In the Russian Federation, improve-
ment of treatment outcomes has been identied as a high
priority by the Ministry of Health and actions to improve
it have been dened. ese include ensuring earlier detec-
tion of MDR-TB and enrolment of patients on second-line
treatment, and strengthening patient support to improve
adherence to treatment (especially among the most social-
ly and economically disadvantaged patients). It is expected
that the introduction of a patient-based monitoring system
for those with MDR/XDR-TB and patients coinfected with
HIV in the near future will also help to improve the quality
of care and treatment outcomes.
3.4.2 All new cases
Data on treatment outcomes for all new cases of TB are
shown in Table 3.7 and Figure 3.4. Globally, the rate of
treatment success was 87% in 2011. Among the six WHO
regions, the highest rates were in the Eastern Mediterra-
nean Region (88%), the South-East Asia Region (89%) and
Western Pacic Region (93%). e treatment success rate
was 79% in the African Region, a big improvement from
73% in 2010. In the Region of the Americas and the Euro-
pean Region it was 75% and 72%, respectively.
Of the 22 HBCs, 15 reached or exceeded a treatment
success rate of 85% among all new cases in 2011, includ-
ing Ethiopia (following a major improvement from 77% in

41GLOBAL TUBERCULOSIS REPORT 2013
BOX 3.6
Deßnitions of treatment outcomes for drug-
susceptible TB until the end of 2012 and in this
global TB reporta
Cured A patient who was initially sputum smear-positive and
who was sputum smear-negative in the last month of treatment
and on at least one previous occasion.
Completed treatment A patient who completed treatment
DWVFKFPQVOGGVVJGETKVGTKCHQTEWTGQTHCKNWTG6JKUFGßPKVKQP
applies to sputum smear-positive and sputum smear-
negative patients with pulmonary TB and to patients with
extrapulmonary disease.
Died A patient who died from any cause during treatment.
Failed A patient who was initially sputum smear-positive
and who remained sputum smear-positive at month 5 or later
FWTKPIVTGCVOGPV#NUQKPENWFGFKPVJKUFGßPKVKQPCTGRCVKGPVU
found to have a multidrug-resistant strain at any point in time
during treatment, whether they are smear-negative or smear–
positive.
Defaulted A patient whose treatment was interrupted for two
consecutive months or more.
Not evaluated A patient whose treatment outcome is not
known.
Successfully treated A patient who was cured or who
completed treatment.
Cohort A group of patients in whom TB has been diagnosed,
CPFYJQYGTGTGIKUVGTGFHQTVTGCVOGPVFWTKPICURGEKßGFVKOG
period (e.g. the cohort of new sputum smear-positive cases
registered in the calendar year 2010). This group forms the
denominator for calculating treatment outcomes. The sum of
the above treatment outcomes, plus any cases for whom no
outcome is recorded (including those ‘still on treatment’ in the
'WTQRGCP4GIKQPCPFÁVTCPUHGTTGFQWVÂECUGUUJQWNFGSWCNVJG
number of cases registered. Some countries monitor outcomes
COQPIEQJQTVUFGßPGFD[URWVWOUOGCTCPFQTEWNVWTG
CPFFGßPGEWTGCPFHCKNWTGCEEQTFKPIVQVJGDGUVNCDQTCVQT[
evidence available for each patient.
a See Treatment of tuberculosis Iuidelines th ed. Geneva, World Health
1TICPK\CVKQP9*1*6/56$#XCKNCDNGCV
JVVRYJSNKDFQEYJQKPVRWDNKECVKQPUAGPIRFH
BOX 3.7
WHO deßnitions of treatment outcomes for
drug-susceptible TB recommended for use
starting in 2013 and that will be used in the
2014 global TB reporta
Cured A pulmonary TB patient with bacteriologically-
EQPßTOGF6$CVVJGDGIKPPKPIQHVTGCVOGPVYJQYCU
smear- or culture-negative in the last month of treatment
and on at least one previous occasion.
Completed treatment A TB patient who completed
treatment without evidence of failure but with no record
to show that sputum smear or culture results in the
last month of treatment and on at least one previous
occasion were negative, either because tests were not
done or because results are unavailable.
Died A patient who died from any cause during
treatment.
Failed A TB patient whose sputum smear or culture is
positive at month 5 or later during treatment.
Lost to follow-up A TB patient who did not start
treatment or whose treatment was interrupted for two
consecutive months or more.
Not evaluated A TB patient for whom no treatment
outcome is assigned. This includes cases ‘transferred out’
to another treatment unit as well as cases for whom the
treatment outcome is unknown to the reporting unit.
Successfully treated A patient who was cured or who
completed treatment.
Cohort #UFGßPGFKPBox 3.6. In addition, it should
be highlighted that any patient found to haXe druI-
resistant TB and placed on second-line treatment is
remoXed from the druI-susceptible TB outcome cohort.
This means that management of the standard TB register
and of the second-line TB treatment register needs to be
coordinated to ensure proper accounting of the outcomes
of treatment. (See also Box 4.4)
a &eßnitions and reportinI framework for tuberculosis – 201 reXision
9*1*6/6$)GPGXC9QTNF*GCNVJ1TICPK\CVKQP
Available at www.who.int/iris/bitstream/10665/79199/
AGPIRFH
BOX 3.8
Achievements in global TB care and control, 1995–2012
WHO began systematic monitoring of TB control progress in 1995. Data compiled on an annual basis since then allow achievements
in TB care and control to be assessed.
Between 1995 and 2012, 56 million people were successfully treated for TB in countries that had adopted the DOTS/Stop TB
StrateIy. This saved approximately 22 million lives.a
The number of lives saved is based on the estimate that in the absence of treatment, approximately one third of people with TB
would die of the disease. This estimate allows for differences in the mortality rates for smear-positive compared with other types of
TB disease (see Chapter 1), and for differences in mortality rates between HIV-negative and HIV-positive people.
a For estimates of the incremental number of lives saved by improvements in TB care associated with implementation of the DOTS and Stop TB Strategy compared
YKVJRTGUVCPFCTFUQHECTGUGG)NC\KQW2GVCN.KXGUUCXGFD[VWDGTEWNQUKUEQPVTQNCPFRTQURGEVUHQTCEJKGXKPIVJGINQDCNVCTIGVHQTTGFWEKPIVWDGTEWNQUKU
mortality. Bulletin of the World *ealth OrIanization¿

42 GLOBAL TUBERCULOSIS REPORT 2013
TABLE 3.6
Treatment success for new smear-positive cases (%) and cohort size (thousands), 1995–2011
a. Treatment success (%)
1995 1996 1997 1999 2000 2001 2002 2003 2004 2005 2006 2007 2009 2010 2011
Afghanistan – – 45 33 90 90 91
Bangladesh 71 63 73 77 79 90 91 92 92 91 92 92 92
$TC\KN 17 20 27 40 71 55 77 76 76 73 72 71 72 74 76
Cambodia 91 94 91 95 93 91 92 92 93 91 93 93 94 95 95 94 93
China 9394959595939592939494949494959695
&4%QPIQ 74 64 70 69 77 90
Ethiopia 61 71 72 74 74 76 76 70 79 90
India 25 21 27 21 34 54 60 76
Indonesia 91 54 50 90 91 91 91 91 91 90 90
Kenya 75 77 65 77 79 79
/Q\CODKSWG 39 55 65 – 71 75 76 77 79 79 –
Myanmar 67 79
Nigeria 4932737375797979 73 75 76
Pakistan 70 – 67 23 70 74 77 79 91 90 91 91 92
Philippines 60 35 71 91 90
4WUUKCP(GFGTCVKQP 65 57 67 65 67 67 61 60 57 55 53 54
South Africa 61 72 57 63 61 67 69 71 74 74 76 73 79 79
Thailand 64 77 69 75 74 73 74 75 77
Uganda 44 33 40 62 61 63 56 60 70 73 70 75 70 67 71 77
746CP\CPKC 73 76 77 76 90
Viet Nam 92 92 92 93 92 92 93 92 93 92 92 92 92 93
<KODCDYG 53 32 69 70 73 69 71 67 66 54 60 74
High-burden countries 53 50 56 62 60 67 72 75 81 84 86 87 87 87 88 88 88
#(4 60 56 64 70 71 70 73 73 74 76 75
#/4 50 51 67 79 76 69 79 79 76 79 77 76 75
'/4 79 66 73 57 79
'74 67 72 63 75 75 74 74 75 71 72 70 71 70 69 67 65
5'#4 33 31 29 40 34 50 63 79
924 72 91 92 91 90 91 90 91 91 92 92 92 92 93 93 94
Global 57 54 60 64 64 69 73 76 80 83 85 84 86 86 86 87 87
D%QJQTVUK\GVJQWUCPFU
1995 1996 1997 1999 2000 2001 2002 2003 2004 2005 2006 2007 2009 2010 2011
Afghanistan 2.0 2.9 2.0 3.1 6.3 10 10 12 13 13 12 13 14
Bangladesh 11 30 34 41 47 54 63 102 104 106 109 106 99
$TC\KN 46 45 43 30 27 34 41 29 43 42 41 41 42 43
Cambodia 4.4 9.1 12 13 16 15 14 17 19 19 21 19 19 20 17 16
China 131 175 210 214 190 194 267 473 470 466 464 449 430 377
&4%QPIQ 16 25 26 33 35 36 41 45 54 62 65 63 66 66 72 73 71
Ethiopia 5.1 11 12 15 21 30 32 37 40 41 39 37 41 45 47 41
India 265 291 293 345 349 396 420 507 553 592 616 625 630 642
Indonesia 3.0 12 21 40 46 52 54 76 93 129 159 175 161 166 169
Kenya 6.5 13 19 22 27 31 31 34 41 40 39 37 37 36 37
/Q\CODKSWG 11 13 11 12 13 14 15 16 17 19 20 20
Myanmar 7.9 9.7 9.2 10 12 17 21 24 27 31 37 40 43 41 42 42 42
Nigeria 9.5 24 11 13 15 16 17 21 34 35 40 44 46 45 45 47
Pakistan 29 3.0 4.1 6.3 15 20 32 66 100 102 104 106
Philippines 90 126 27 21 37 50 55 59 94
4WUUKCP(GFGTCVKQP 0.05 43 0.7 0.7 1.5 3.6 4.1 5.2 6.3 26 26 31 32 32 32 30 37
South Africa 45 55 37 101 99 114 127 135 140 143 144 139 134 133
Thailand 20 0.1 3.7 14 23 20 27 30 29 30 33 30 31
Uganda 15 15 13 14 14 17 19 20 21 21 20 21 23 23 23 26
746CP\CPKC 20 21 22 24 24 24 24 24 25 26 25 25 25 24 25 24 24
Viet Nam 54 55 53 53 54 57 56 55 56 54 53 51 52 51
<KODCDYG 9.712121313141716141513161110101213
High-burden countries 739 967 879 912 1 044 1 119 1 186 1 260 1 450 1 776 1 965 2 087 2 132 2 181 2 184 2 185 2 140
#(4 233 235 323 365 409 452 491 552 564 566 577 591 606 599 579
#/4 129 134 125 111 110 111 102 105 110 121 119 132 116 109 123 126 127
'/4 46 51 60 66 64 52 76 114 132 156 167 167 170 171
'74 34 94 24 22 41 50 54 60 114 105 99 97
5'#4 360 376 399 473 512 550 604 661 974 1 011 1 022 1 045 1 065
924 296 372 294 313 353 360 346 357 439 575 663 663 661 657 641 622 560
Global 1 001 1 245 1 147 1 195 1 347 1 453 1 510 1 649 1 842 2 206 2 396 2 529 2 591 2 649 2 665 2 662 2 599
Blank cells indicate data not reported.
– indicates values that cannot be calculated.
43GLOBAL TUBERCULOSIS REPORT 2013
TABLE 3.7
Treatment success for all new cases (%) and cohort size (thousands), 1995–2011
a. Treatment success (%)
1995 1996 1997 1999 2000 2001 2002 2003 2004 2005 2006 2007 2009 2010 2011
Afghanistan – – 45 33 90
Bangladesh 71 63 73 77 79 90 90 91 90 91 91 91 91
$TC\KN 17 20 27 40 71 55 77 72 72 69 72 69 70 72 73
Cambodia 91 94 91 95 93 91 92 92 93 91 91 92 93 94 94 94
China 9394959595939592939292929393949595
&4%QPIQ 74 64 70 69 77 60
Ethiopia 61 71 72 74 74 76 76 70 79 77
India 25 21 27 21 34 54 60 76
Indonesia 91 54 50 90 90 90
Kenya 75 77 65 77 79 79 77
/Q\CODKSWG 39 55 65 – 71 75 76 77 79 79 –
Myanmar 67 79
Nigeria 49 32 73 73 75 79 79 79 73 75 76
Pakistan 70 – 67 23 70 74 77 79 90 91 90 92
Philippines 60 35 71 90
4WUUKCP(GFGTCVKQP 65 57 67 65 67 67 61 65 67 69 69 69 66 65
South Africa 61 72 57 63 61 67 65 69 70 71 73 53 77
Thailand 64 77 69 75 74 73 71 71 75
Uganda 44 33 40 62 61 63 56 60 70 73 72 67 64 73
746CP\CPKC 73 76 77 76
Viet Nam 92 92 92 93 92 92 92 92 92 91 92 92 92 93
<KODCDYG 53 32 69 70 73 69 71 67 66 66 67 70 75 76
High-burden countries 53 50 56 62 60 67 72 75 81 82 85 85 87 87 86 86 88
#(4 60 56 64 70 71 70 73 73 70 74 72 77 77 76 73 79
#/4 50 51 67 79 76 69 76 75 73 73 73 73 75
'/4 79 66 73 57 79
'74 67 72 63 75 75 74 74 75 76 77 75 76 76 75 74 72
5'#4 33 31 29 40 34 50 63 79
924 72 91 92 91 90 91 90 91 90 90 91 91 91 92 93
Global 57 54 60 64 64 69 73 76 80 81 84 84 85 85 85 84 87
D%QJQTVUK\GVJQWUCPFU
1995 1996 1997 1999 2000 2001 2002 2003 2004 2005 2006 2007 2009 2010 2011
Afghanistan 2.0 2.9 2.0 3.1 6.3 10 10 12 13 13 12 26 26
Bangladesh 11 30 34 41 47 54 63 119 141 144 106 156 150
$TC\KN 46 45 43 30 27 34 41 29 47 73 75 71
Cambodia 4.4 9.1 12 13 16 15 14 17 19 30 34 34 35 39 40 37
China 131 175 210 214 190 194 267 644 932 923
&4%QPIQ 16 25 26 33 35 36 41 45 54 62 65 92 93 106 109 92
Ethiopia 5.11112152130323740413937 139 139 152 91
India 265 291 293 345 349 396 420 1 066 1 071 1 137 1 199 1 226 1 244 1 229 1 209
Indonesia 3.0 12 21 40 46 52 54 76 93 206 244 266 263 293 296 314
Kenya 6.5 13 19 22 27 31 31 34 97 101 99 99 99 90
/Q\CODKSWG 11 13 11 12 13 14 15 16 17 19 20 20
Myanmar 7.9 9.7 9.2 10 12 17 21 24 27 66 73 90 91 127 135
Nigeria 9.5 24 11 13 15 16 17 21 34 35 40 44 46
Pakistan 29 3.0 4.1 6.3 15 20 117 149 191 206 212 256 255
Philippines 90 126 27 21 37 50 55 59 126 123 136 140 141 162 190
4WUUKCP(GFGTCVKQP 0.05 43 0.7 0.7 1.5 3.6 4.1 5.2 6.3 39 74 97 99 103 101 94
South Africa 45 55 37 101 99 114 243 259 271 247 236 367 292
Thailand 20 0.1 3.7 14 23 20 27 47 49 47 47 54 43 49
Uganda 15 15 13 14 14 17 19 20 21 21 31 37 39 40 43
746CP\CPKC 20 21 22 24 24 24 24 24 25 61 59 25 59 60 59 59
Viet Nam 54 55 53 53 54 57 56 92 55 91 91 91
<KODCDYG 9.7 12 12 13 13 14 17 16 14 54 43 43 39 40 45 46 40
High-burden countries 739 967 879 912 1 044 1 119 1 186 1 260 1 450 3 183 3 430 3 799 3 872 4 134 4 374 4 403 4 252
#(4 233 235 323 365 409 452 491 940 930 1 297 1 215 1 094
#/4 129 134 125 111 110 111 102 105 110 191 197 157 191 200
'/4 46 51 60 66 64 52 76 226 259 307 320 331 391
'74 34 94 24 22 42 50 55 60 221 274 276 279 250 217
5'#4 360 376 399 473 512 550 604 661 1 530 1 639 1 940
924 296 372 294 313 353 360 346 357 439 963 1 030 1 163 1 216 1 261 1 259 1 240 1 213
Global 1 001 1 245 1 147 1 195 1 347 1 453 1 511 1 649 1 843 3 892 4 188 4 592 4 720 4 995 5 267 5 275 5 096
Blank cells indicate data not reported.
– indicates values that cannot be calculated.
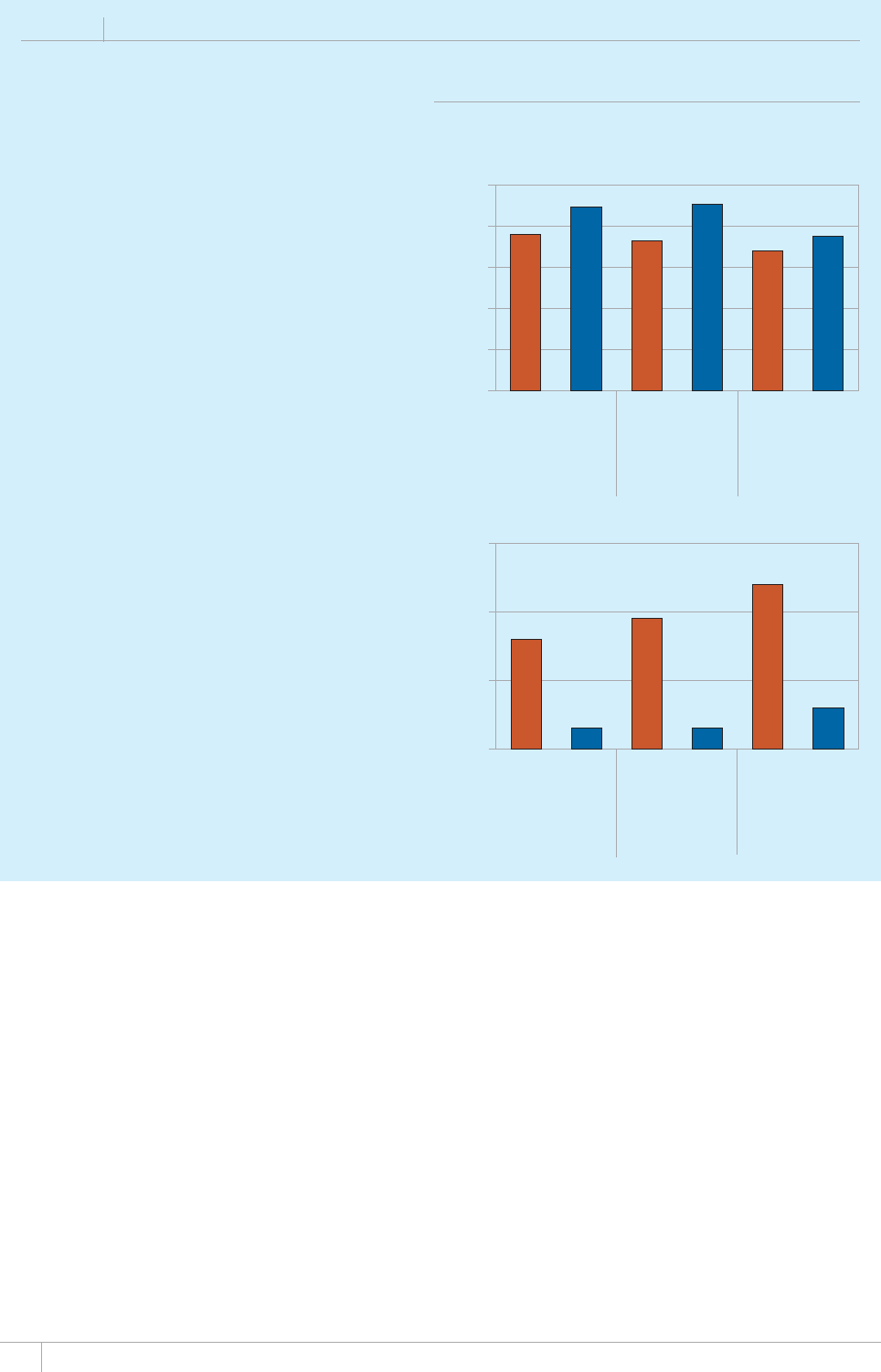
44 GLOBAL TUBERCULOSIS REPORT 2013
BOX 3.9
Outcomes of TB treatment by HIV status
HIV+
(109 137)
HIV-
(925 024)
HIV+
(148 798)
HIV-
(811 586)
HIV+
(73 354)
HIV-
(370 385)
0
10
20
30
HIV+
(117 094)
HIV-
(959 174)
HIV+
(163 300)
HIV-
(845 907)
HIV+
(79 817)
HIV-
(385 748)
New smear-positive
patients (data from
88 countries)
New smear-negative
and extrapulmonary
patients (data from
72 countries)
Retreatment
patients (data from
59 countries)
0
20
40
60
80
100
Percentage of cohort (%)
a. Treatment success, 2011
b. Death rate among evaluated cases, 2011
Percentage (%)
New smear-positive
patients (data from
88 countries)
New smear-negative
and extrapulmonary
patients (data from
72 countries)
Retreatment
patients (data from
59 countries)
FIGURE B3.9.1
6TGCVOGPVUWEEGUUCCPFFGCVJTCVGUDCOQPI*+8
positive and HIV-negative TB patients, 2011
+PEQWPVTKGUYKVJa*+8RQUKVKXG6$
patients reported treatment outcomes for 2011 that were
disaggregated by HIV status. These countries accounted for
QHCNN*+8RQUKVKXG6$RCVKGPVUTGIKUVGTGFKPVJCV[GCT
This was a considerable increase from 2010, when countries
that reported outcomes disaggregated by HIV status
accounted for 25% of TB patients with a documented HIV-
positive test result. Much of the improvement is due to the
TGRQTVKPIQHFCVCFKUCIITGICVGFD[*+8UVCVWUHQTVJGßTUV
time by high TB/HIV burden countries such as South Africa
and Uganda. Of the 41 TB/HIV priority countries (listed in
Table 6.1 of Chapter 6), 19 reported treatment outcomes
FKUCIITGICVGFD[*+8UVCVWU$WTWPFK$WTMKPC(CUQ$TC\KN
Botswana, China, Ghana, Haiti, India, Kenya, Lesotho,
/CNK/[CPOCT0COKDKC0KIGTKC5QWVJ#HTKEC5YC\KNCPF
6JCKNCPFVJG7PKVGF4GRWDNKEQH6CP\CPKCCPF8KGV0CO
Data for 2011 show that treatment outcomes for HIV-
positive TB patients continue to be worse than those of
HIV-negative TB patients. The treatment success rate for all
PGY*+8RQUKVKXG6$RCVKGPVUYCUEQORCTGFYKVJ
among HIV-negative TB patients (Figure B3.9.1). If it is
assumed that HIV-positive TB patients who defaulted from
treatment would have died from TB, the death rate was 19%
among HIV-positive TB patients compared with 3% among
*+8PGICVKXG6$RCVKGPVU5WEJßPFKPIUCTGEQPUKUVGPVYKVJ
two autopsy studies in South Africa, which showed that
undiagnosed TB remains the main cause of death among
HIV-positive people.a,b
a /WVGXGF\K2GVCNEarly mortality followinI initiation of #4T in
rural South #frica the contribution of eZistinI co-morbidities. 20th
%QPHGTGPEGQP4GVTQXKTWUGUCPF1RRQTVWPKUVKE+PHGEVKQPU#VNCPVC
)GQTIKC75#¿/CTEJ2CRGTwww.retroconference.
org/2013b/Abstracts/46910.htm, accessed 3 June 2013).
b Martinson N et al. 7ndiaInosed infectious TB in adult home deaths
South #frica 201VJ%QPHGTGPEGQP4GVTQXKTWUGUCPF1RRQTVWPKUVKE
+PHGEVKQPU#VNCPVC)GQTIKC75#¿/CTEJ2CRGTwww.
TGVTQEQPHGTGPEGQTID#DUVTCEVUJVOCEEGUUGF,WPG).
2010 to 89% in 2011) and Nigeria for the rst time. e
six countries that reported lower rates of treatment suc-
cess were Brazil (73%), the Russian Federation (65%),
South Africa (77%), ailand (82%), Uganda (73%) and
Zimbabwe (80%). Data were not reported for Mozambique.
Treatment outcomes are worse among HIV-positive TB
patients compared with HIV-negative TB patients (Box 3.9).
Further eorts are needed to narrow the gap.

45GLOBAL TUBERCULOSIS REPORT 2013
%*#26'4
Drug-resistant TB
KEY FACTS AND MESSAGES
■ By the end of 2012, data on anti-TB drug resistance were available for 136 countries (70% of 194 WHO Member States), either
HTQOEQPVKPWQWUUWTXGKNNCPEGOQUVN[JKIJKPEQOGEQWPVTKGUCPFQVJGTEQWPVTKGUQHVJG9*1'WTQRGCP4GIKQPQTURGEKCNUWTXG[U
■ 5WTXG[UWPFGTYC[KPKPVJGITQWRQHJKIJ6$CPFQT/&46$DWTFGPEQWPVTKGUCPFHTQOYJKEJTGUWNVUCTGGZRGEVGFKP
KPENWFGVJGßTUVPCVKQPYKFGUWTXG[UKP#\GTDCKLCP+PFKC2CMKUVCP6WTMOGPKUVCPCPF7MTCKPGCPFTGRGCVUWTXG[UKP%JKPC
'VJKQRKC-GP[CVJG2JKNKRRKPGU5QWVJ#HTKEC6JCKNCPFCPF8KGVa0CO
■ )NQDCNN[CPGUVKOCVGF%+¿QHPGYECUGUCPF%+¿QHRTGXKQWUN[VTGCVGFECUGU
JCXG/&46$6JGJKIJGUVNGXGNUCTGKPGCUVGTP'WTQRGCPFEGPVTCN#UKCYJGTGKPUGXGTCNEQWPVTKGUOQTGVJCPQHPGYECUGUCPF
OQTGVJCPQHRTGXKQWUN[VTGCVGFECUGUJCXG/&46$
■ 6JGTGYGTGCPGUVKOCVGFTCPIG¿PGYECUGUQH/&46$YQTNFYKFGKP#OQPIRCVKGPVUYKVJ
RWNOQPCT[6$PQVKßGFKPKGVJGITQWRQHRCVKGPVUMPQYPVQ062UCPFVJCVECPDGVGUVGFHQTFTWITGUKUVCPEGWUKPI9*1
TGEQOOGPFGFFKCIPQUVKEVGUVUVJGTGYGTGCPGUVKOCVGFaTCPIGa¿a/&46$ECUGUKP/QTGVJCPJCNH
QHVJGUGECUGUYGTGKP+PFKC%JKPCCPFVJG4WUUKCP(GFGTCVKQP
■ 'ZVGPUKXGN[FTWITGUKUVCPV6$:&46$JCUDGGPTGRQTVGFD[EQWPVTKGU1PCXGTCIGCPGUVKOCVGF%+¿
QH/&46$ECUGUJCXG:&46$
■ #VQVCNQH6$ECUGUGNKIKDNGHQT/&46$VTGCVOGPVYKVJ/&46$CPFYKVJTKHCORKEKPTGUKUVCPEGFGVGEVGF
WUKPI:RGTV/6$4+(YGTGPQVKßGFINQDCNN[KPOQUVN[D['WTQRGCPEQWPVTKGU+PFKCCPF5QWVJ#HTKEC6JKUTGRTGUGPVGF
RTQITGUUEQORCTGFYKVJYJGP/&46$ECUGUCPFTKHCORKEKPTGUKUVCPV6$ECUGUYGTGFGVGEVGFVJGNCTIGUV
increases between 2011 and 2012 were in India, South Africa and Ukraine. However, worldwide and in most countries with a high
DWTFGPQH/&46$NGUUVJCPQPGVJKTFQHVJG6$RCVKGPVUGUVKOCVGFVQJCXG/&46$YGTGCEVWCNN[FGVGEVGFKP
■ %QWPVTKGUFGVGEVKPIENQUGVQQHVJGPQVKßGF6$RCVKGPVUGUVKOCVGFVQJCXG/&46$KPKPENWFGF'UVQPKC-C\CMJUVCP
.CVXKC.KVJWCPKC5QWVJ#HTKECCPF7MTCKPG6JGNQYGUVßIWTGUYGTGKPVJG5QWVJ'CUV#UKC4GIKQPCPFVJG9GUVGTP2CEKßE
4GIKQPYJKEJEQODKPGFJCXGQHVJGYQTNFÂUECUGUQH/&46$
■ ,WUVQXGTRGQRNGYKVJ/&46$YGTGUVCTVGFQPUGEQPFNKPGVTGCVOGPVKPGSWKXCNGPVVQQHVJGPGYN[
FGVGEVGFECUGUVJCVYGTGGNKIKDNGHQTUWEJVTGCVOGPVINQDCNN[&KCIPQUVKEVTGCVOGPVICRUYGTGOWEJNCTIGTKPUQOGEQWPVTKGU
GURGEKCNN[KPVJG#HTKECP4GIKQPQHFGVGEVGFECUGUGPTQNNGFQPVTGCVOGPVCPFYKFGPGFDGVYGGPCPFKP%JKPC
Pakistan and South Africa.
■ 6JGVTGCVOGPVUWEEGUUVCTIGVQHÜUGVKPVJGGlobal Plan to Stop TB 2011–2015HQT/&46$YCUTGCEJGFD[QH
EQWPVTKGUVJCVTGRQTVGFQWVEQOGFCVCHQTVJGRCVKGPVEQJQTV*QYGXGTQXGTCNNQPN[QHRCVKGPVUYGTGUWEEGUUHWNN[VTGCVGF
■ +PVGPUKßGFINQDCNCPFPCVKQPCNGHHQTVUVQFGVGEVECUGUQH/&46$VQGPTQNVJGOQPVTGCVOGPVCPFVQKORTQXGVTGCVOGPV
outcomes are urgently required.
Drug-resistant TB (DR-TB) threatens global TB control and
is a major public health concern in several countries. is
chapter summarizes the progress made in global surveil-
lance of anti-TB drug resistance, using the most recent data
on MDR-TB and XDR-TB gathered from special surveys and
continuous surveillance systems, and summarizes global
estimates of disease burden associated with MDR-TB based
on these data (section 4.1). It also includes an assessment
of national progress in diagnosing and treating MDR-TB,
using data on diagnostic testing for DR-TB, enrolment on
treatment with second-line drugs for those found to have
MDR-TB, and treatment outcomes (section 4.2).
4.1 Surveillance of drug-resistant TB
4.1.1 Progress in the coverage of drug resistance
surveillance
Since the launch of the Global Project on Anti-tuberculosis
Drug Resistance Surveillance in 1994, data on drug resis-
tance have been systematically collected and analysed from
136 countries worldwide (70% of WHO Member States).
is includes 70 countries that have continuous surveil-
lance systems based on routine diagnostic drug suscepti-
bility testing (DST) of all TB patients and 66 countries that
rely on special epidemiological surveys of representative
samples of patients. e progress towards achieving global
coverage of drug resistance data is shown in Figure 4.1.
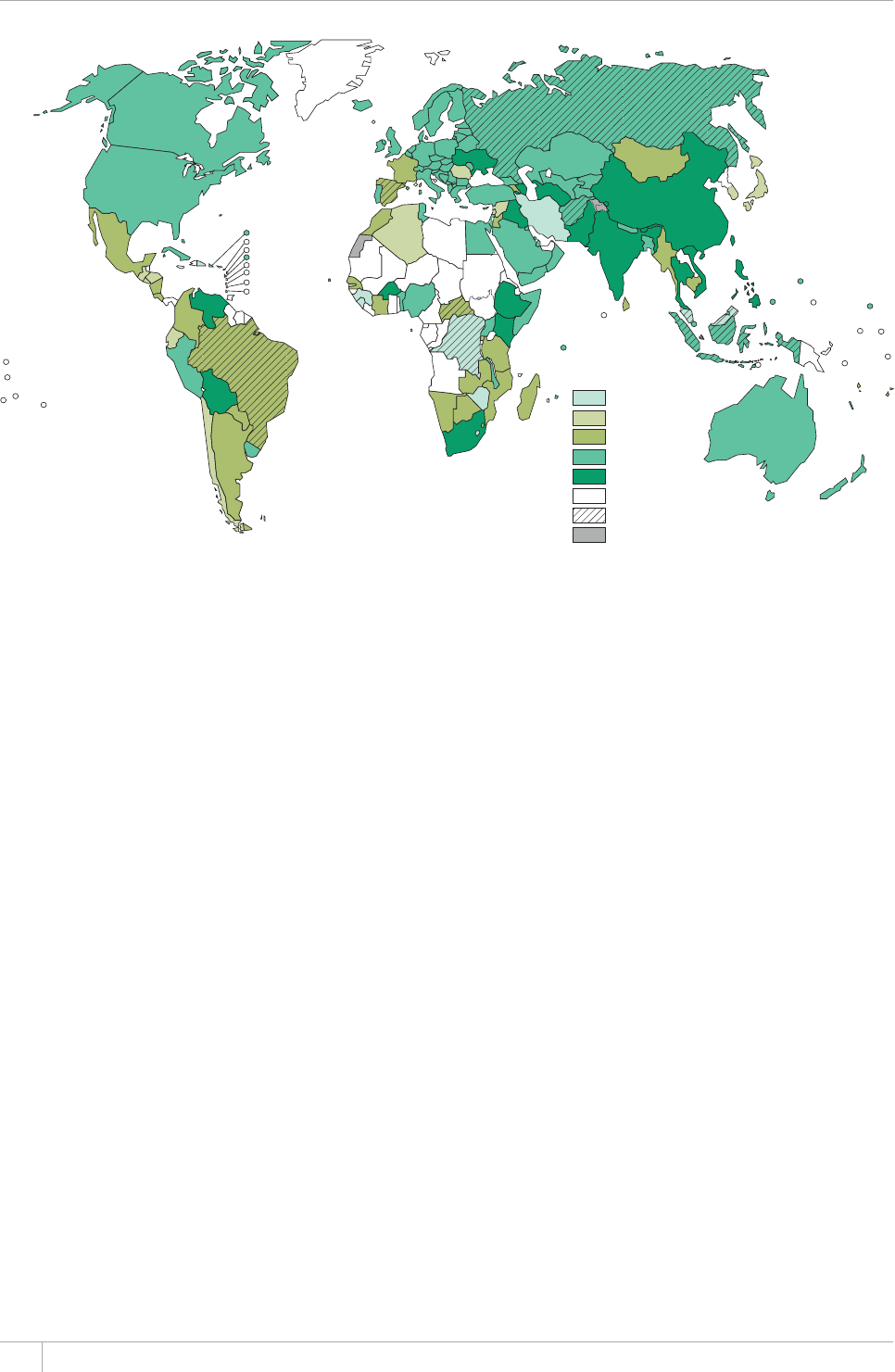
46 GLOBAL TUBERCULOSIS REPORT 2013
Continuous surveillance for MDR-TB, based on routine
DST of TB patients and systematic collection, collation and
analysis of data, is the most eective approach to monitor
trends in drug resistance over time. Additionally, such sys-
tems can detect outbreaks that might otherwise be unde-
tected, even during the course of a survey if the outbreak
site was not among those sites selected for patient enrol-
ment.
e number of countries that can rely on data generat-
ed by continuous surveillance systems is increasing, due to
eorts invested in scaling up the availability of culture and
DST services. Several high MDR-TB burden countries in the
European Region, including Belarus, Georgia, Kazakhstan,
Republic of Moldova, Ukraine and the Baltic States, have
put in place high quality surveillance systems to monitor
drug resistance both in new and previously treated TB cas-
es. A group of countries – Bolivia, Chile, Colombia, Costa
Rica, Ecuador, Egypt, El Salvador, Kyrgyzstan, Lebanon,
Mongolia, Nicaragua, Rwanda, Sri Lanka, Syrian Arab
Republic and Tajikistan – that previously relied on special
surveys to monitor drug resistance, have now established
routine surveillance systems for all previously treated cas-
es. is is the rst step towards achieving routine DST for
all TB patients.
Special surveys still represent the most common
approach to investigating the burden of drug resistance in
resource-limited settings where routine DST is not acces-
sible to all TB patients due to lack of laboratory capacity
or resources. Between 2010 and 2012, drug resistance
surveys were completed for the rst time in 16 countries:
Afghanistan (Central region), Albania, Bangladesh, Belar-
us, Benin, Bulgaria, Kyrgyzstan, Malawi, Nigeria, Saudi
Arabia, Somalia, Tajikistan, Tunisia, Uganda, Uzbekistan
and Yemen. In addition, Egypt, Brazil, Nepal and Zambia
completed a repeat survey.
In mid-2013, drug resistance surveys were ongoing in
12 high TB and MDR-TB burden countries. ese include
the rst nationwide surveys in Azerbaijan, India, Pakistan,
Turkmenistan, Ukraine, and repeat surveys in China, Ethi-
opia, Kenya, the Philippines, South Africa, ailand and
VietNam.
Molecular technologies are increasingly being used in
drug resistance surveys to simplify logistics and reduce
laboratory workload. GenoType® MTBDRplus (Hain Life-
science, Germany) was used in the national survey com-
pleted in 2012 in Nigeria and Xpert® MTB/RIF (Cepheid,
USA) is being used in the surveys underway in Pakistan and
Papua New Guinea. Several more countries are planning to
use Xpert MTB/RIF as a screening tool in drug resistance
surveys. ough not a complete surrogate for MDR-TB,
particularly in settings where levels of drug resistance are
low, rifampicin resistance is the most important indicator
of MDR-TB, with serious clinical implications for aected
patients. In countries where there is not yet the capacity
for culture and DST using conventional methods or where
laboratories cannot cope with the large workload generat-
ed by a drug resistance survey, Xpert MTB/RIF can play
an important role. It can be used to screen specimens for
rifampicin resistance and identify those requiring further
testing to be performed at national or supranational TB ref-
erence laboratories, also reducing the cost of initial screen-
ing by conventional commercial DST systems.
FIGURE 4.1
Progress in global coverage of data on drug resistance, 1994–2013
Year of most
recent data
1995–1999
2000–2004
2005–2009
2010–2012
Ongoing in 2013
No data
Subnational data only
Not applicable
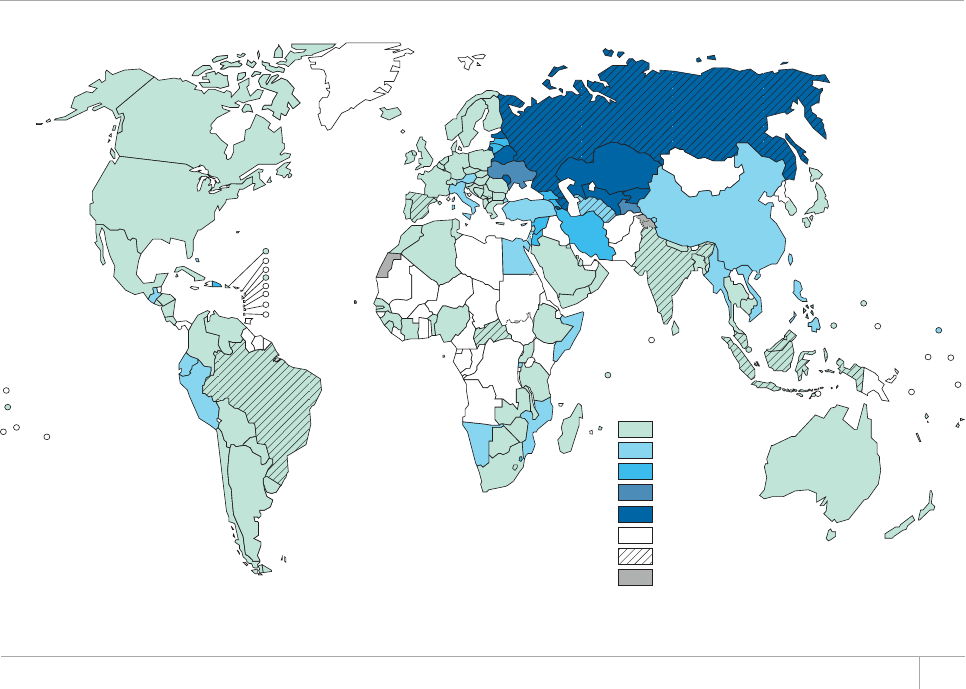
47GLOBAL TUBERCULOSIS REPORT 2013
Five high TB and MDR-TB burden countries (Afghani-
stan, Brazil, Democratic Republic of the Congo, Indonesia
and the Russian Federation) still rely on drug resistance
surveillance data gathered from sub-national areas only.
ese countries should consider conducting nationwide
drug resistance surveys in the short term to better under-
stand the burden of MDR-TB and to guide the planning of
diagnostic and treatment services. A further six countries
(Dominican Republic, Guinea, Iran, Lesotho, Sierra Leone
and Zimbabwe) rely on drug resistance data gathered from
studies conducted in the late 1990s and should consider
implementing repeat surveys. Central and Francophone
Africa remain the parts of the world where drug resistance
surveillance data are most lacking, largely as a result of the
current weak laboratory infrastructure. Eorts should be
made to increase diagnostic and surveillance capacity in
these settings so that a drug resistance survey can be con-
ducted.
Of the 136 countries with surveillance data on drug
resistance, 35% (48 countries) have only one data point and
should consider repeating surveys to assess time trends.
Data on time trends in drug resistance were available
from 88 countries and 10 territories worldwide for a total
of 870 country-year data points. Among the 36 high TB and
high MDR-TB burden countries, 11 countries (Cambodia,
Estonia, Georgia, Latvia, Lithuania, Mozambique, Myan-
mar, Republic of Moldova, the Russian Federation (7 Fed-
eral Subjects), ailand and Viet Nam) have completed at
least two surveys at least ve years apart, allowing trends
over time to be evaluated. However, for ve of these coun-
tries (Cambodia, Mozambique, Myanmar, ailand and
Viet Nam) the most recent data are more than ve years
old. Among the six countries with recent data, in Estonia
and Latvia, surveillance data show that the rates of both
TB and MDR-TB have been declining. ese data suggest
that MDR-TB can indeed be controlled once eective policy
decisions are put into practice, and the necessary preven-
tion and control measures are implemented. In Lithuania,
Georgia, Republic of Moldova and most Federal Subjects of
the Russian Federation, MDR-TB rates appear to be stable
whereas in Ivanovo Oblast and Mary-El Republic MDR-TB
rates are increasing. Extending trend analyses to other
countries requires more data from repeat surveys or contin-
uous surveillance systems. NTPs should plan to repeat drug
resistance surveys regularly, approximately every ve years,
until capacity for continuous surveillance is established.
4.1.2 Percentage of new and previously treated
TB cases that have MDR-TB
Globally, 3.6% (95% CI: 2.1–5.1%) of new TB cases and
20.2% (95%CI: 13.3–27.2%) of previously treated cases are
estimated to have MDR-TB (Table 4.1). ese estimates are
essentially unchanged from 2011.
e proportions of new and previously treated TB cases
with MDR-TB at the country level are shown in Figure 4.2
and Figure 4.3, and for the 27 high MDR-TB burden coun-
tries in Table 4.1. Eastern European and especially central
Asian countries continue to have the highest levels of MDR-
TB. Among new cases, examples include Azerbaijan (22.3%
in 2007), Belarus (34.8% in 2012), Estonia (19.7% in 2012),
FIGURE 4.2
Percentage of new TB cases with MDR-TBa
Percentage
of cases
0–2.9
3–5.9
6–11.9
12–17.9
≥18
No data
Subnational data only
Not applicable
a Figures are based on the most recent year for which data have been reported, which varies among countries.
GLOBAL TUBERCULOSIS REPORT 2013
TABLE 4.1
Estimated proportion of TB cases that have
MDR-TB, globally and for 27 high MDR-TB burden
countries and WHO regions
ESTIMATED
% OF NEW TB
CASES WITH
/&46$a
CONFIDENCE
+06'48#.
ESTIMATED
% OF
4'64'#6/'06
TB CASES WITH
/&46$a
CONFIDENCE
+06'48#.
Armenia 9.4 7.0–12 43 ¿
#\GTDCKLCP 22 19–27 56 50–62
Bangladesh 1.4 0.7–2.5 29 24–34
Belarus 35 33–37 69 66–71
Bulgaria 2.3 ¿ 23 17–31
China 5.7 4.5–7.0 26 22–30
&4%QPIQ 2.5 0.1–5.0 10 .5–1
Estonia 20 14–26 50 35–65
Ethiopia 1.6 ¿ 12 5.6–21
Georgia 9.2 7.9–11 31 27–35
India 2.2 1.9–2.6 15 11–19
Indonesia 1.9 1.4–2.5 12 ¿
-C\CMJUVCP 23 22–24 55 54–56
-[TI[\UVCP 26 23–31 65–72
Latvia 11 ¿ 32 23–42
Lithuania 11 9.5–14 44 39–49
Myanmar 4.2 3.1–5.6 10 6.9–14
Nigeria 2.9 2.1–4.0 14 10–19
Pakistan .5 0.1–12 2.5–5
Philippines 4.0 2.9–5.5 21 14–29
4GRWDNKEQH/QNFQXC 24 21–26 62 59–65
4WUUKCP(GFGTCVKQP 23 21–25 49 45–53
South Africa 1.4–2.3 6.7 ¿
Tajikistan 13 ¿ 56 52–61
Ukraine 14 14–15 32 31–33
7\DGMKUVCP 23 ¿ 62 53–71
Viet Nam 2.7 2.0–3.7 19 14–25
High MDR-TB
burden countries 4.2 2.1–6.2 21 12–30
#(4 2.3 0.2–4.4 11 4.4–17
#/4 2.2 1.4–3.0 14 4.7–22
'/4 3.5 0.1–11 33 12–54
'74 16 10–22 45 39–52
5'#4 2.2 ¿ 16 11–21
924 4.7 3.3–6.1 22 ¿
Global 3.6 2.1–5.1 20 13–27
a Best estimates are for the latest available year. Estimates in italics are based on
regional data.
Kazakhstan (22.9% in 2012), Kyrgyzstan (26.4% in 2011),
the Republic of Moldova (23.7% in 2012), the Russian Fed-
eration (average: 23.1%, with Yamalo-Nenets Autonomous
Area being the highest: 41.9% in 2011) and Uzbekistan
(23.2% in 2011). Among previously treated cases, exam-
ples include Azerbaijan (Baku City: 55.8% in 2007), Belar-
us (68.6% in 2012), Estonia (50.0% in 2012), Kazakhstan
(55.0% in 2012), Kyrgyzstan (68.4% in 2012), the Republic
of Moldova (62.3% in 2012), Tajikistan (56.0% in 2012) and
Uzbekistan (62.0% in 2011). In the Russian Federation,
even though the average proportion of cases with MDR-
TB does not exceed 50%, the proportion is well above 50%
in several Federal Subjects (with Ulyanovsk Oblast at the
highest level: 74.0% in 2011).1
1 Tuberculosis in the Russian Federation 2011: an analytical review of
statistical indicators used in the Russian Federation and in the world (in
Russian). Moscow: Ministry of Health of the Russian Federation et
al., 2013.
BOX 4.1
MDR-TB in children
TB in children poses a diagnostic challenge, as
paucibacillary disease is more likely. Specimens suitable
HQTEWNVWTGCPF&56CTGOQTGFKHßEWNVVQQDVCKPRCTVKEWNCTN[
from the youngest children who cannot expectorate sputum.
%QPUGSWGPVN[NKVVNGKUMPQYPCDQWVVJGDWTFGPQH/&46$KP
children.
6JGTGNCVKQPUJKRDGVYGGP/&46$CPFCIGITQWR
(children aged less than 15 years versus adults aged 15
years or older) was recently assessed using representative
drug resistance surveillance data reported to WHO between
1994 and 2012. Data were analysed for 376 293 TB cases
for whom age and DST data were available. Odds ratios
were derived by logistic regression with robust standard
errors, as described in detail elsewhere.a1HVJGEQWPVTKGU
reporting data from nationwide surveys or surveillance
U[UVGOUTGRQTVGFCVNGCUVQPGRCGFKCVTKE/&46$ECUG
A child with TB was shown to be as likely as an adult
YKVJ6$VQJCXG/&46$+VKUVJGTGHQTGGUUGPVKCNVJCVVJG
KFGPVKßECVKQPQH/&46$KPEJKNFTGPDGUVTGPIVJGPGF
Efforts should be made to systematically conduct household
EQPVCEVKPXGUVKICVKQPQHCNNRCVKGPVUYKVJ/&46$KPENWFKPI
children. Additionally, children must be routinely included
in all drug resistance surveillance activities, including drug
resistance surveys.
a <KIPQNGVCN/WNVKFTWITGUKUVCPVVWDGTEWNQUKUKPEJKNFTGPGXKFGPEGHTQO
global surveillance. European 4espiratory ,ournal¿
More positively, levels of drug resistance among new cas-
es remain low (<3%) in many parts of the world, including
almost all countries in the Region of the Americas, most
African countries where drug resistance surveys have been
conducted, most of the South-East Asia Region, most of
western Europe, and several countries in the Western Pacif-
ic Region (examples include Australia, Cambodia, Japan,
New Zealand and VietNam).
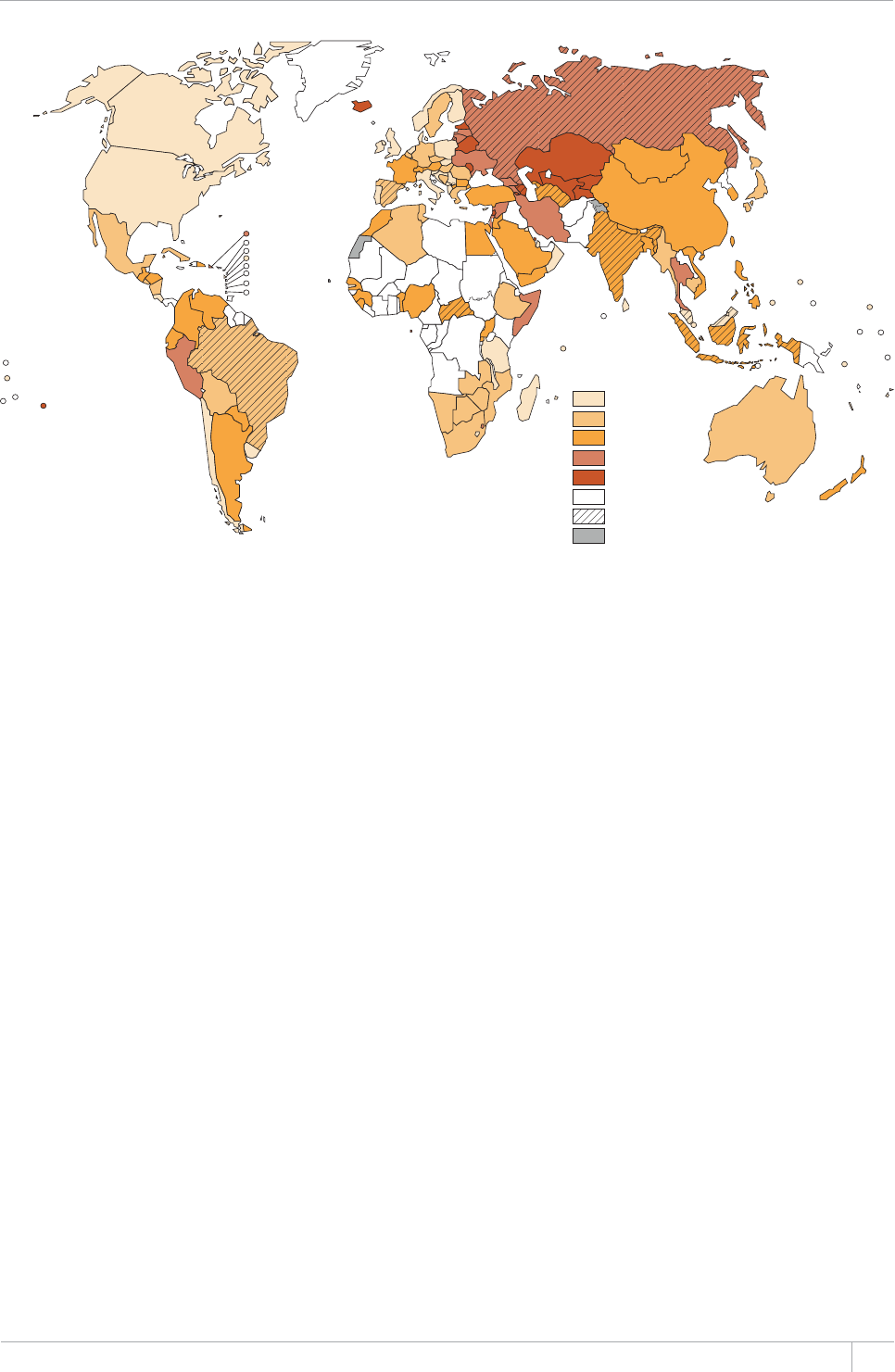
49GLOBAL TUBERCULOSIS REPORT 2013
FIGURE 4.3
Percentage of previously treated TB cases with MDR-TBa
Percentage
of cases
0–5.9
6–11.9
12–29.9
30–49.9
≥50
No data
Subnational data only
Not applicable
a (KIWTGUCTGDCUGFQPVJGOQUVTGEGPV[GCTHQTYJKEJFCVCJCXGDGGPTGRQTVGFYJKEJXCTKGUCOQPIEQWPVTKGU6JGJKIJRGTEGPVCIGUQHRTGXKQWUN[VTGCVGF6$ECUGUYKVJ/&46$
KP$CJTCKP$QPCKTG¿5CKPV'WUVCVKWUCPF5CDC%QQM+UNCPFU+EGNCPF5CQ6QOGCPF2TKPEKRGCPF.GDCPQPTGHGTVQQPN[CUOCNNPWODGTQHPQVKßGFECUGU
4.1.3 Estimated global incidence of MDR-TB and
estimated number of MDR-TB cases among
PQVKßGF6$RCVKGPVUKP
e data compiled from surveillance of drug resistance
among TB patients allow estimation of the total number of
incident cases of MDR-TB worldwide in 2012. e number
of incident cases includes not only cases among notied
TB patients, but also cases among people diagnosed with
TB that were not notied to NTPs (and in whom MDR-TB
may not have been detected) and cases among people not
yet diagnosed with TB. Globally in 2012, there were an
estimated 450 000 (range: 300 000‒600 000) new cases
of MDR-TB. Methods used to produce this estimate are
explained in Annex 1.
Data compiled from surveillance of drug resistance
among TB patients also allow production of global as well as
country-specic estimates of the number of MDR-TB cases
among notied TB patients with pulmonary TB. ese are
the MDR-TB cases that could be found by NTPs if all noti-
ed patients were tested for drug resistance to rifampicin
and isoniazid using WHO-recommended diagnostic tests,
and is a useful indicator for assessing country performance
in detecting cases of MDR-TB and enrolling them on treat-
ment. Globally in 2012, there were an estimated 300000
(range: 220000–380 000) MDR-TB cases among notied
TB patients. Country-specic estimates are discussed in
section 4.2.
4.1.4 Resistance to second-line drugs
Extensively drug-resistant TB (XDR-TB) had been reported
by 92 countries globally by the end of 2012 (Figure 4.4). A
total of 75 countries and 4 territories reported represen-
tative data from continuous surveillance or special sur-
veys regarding the proportion of MDR-TB cases that had
XDR-TB. Combining their data, the average proportion of
MDR-TB cases with XDR-TB was 9.6% (95% CI: 8.1%–11%),
similar to the estimate from 2011 (9.0%). irteen of these
countries reported more than 10 XDR-TB cases in the most
recent year for which data were available. Among those
countries, the proportion of MDR-TB cases with XDR-
TB was highest in Azerbaijan (Baku city: 12.8%), Belarus
(11.9%), Latvia (16.0%), Lithuania (24.8%) and Tajikistan
(Dushanbe city and Rudaki district: 21.0%).
e proportion of MDR-TB cases with resistance to u-
oroquinolones and second-line injectable agents was 16.5%
(95% CI: 12.3–20.7) and 22.7% (15.4%–30.0%), respective-
ly. A total of 32.0% (21.9%–42.1%) of patients with MDR-
TB have resistance to a uoroquinolone, a second-line
injectable agent, or both. ese patients would likely be
eligible to receive bedaquiline, the new bactericidal drug
recently approved for use in patients with MDR-TB when
options to treat using existing drugs have been exhausted
(see Box 8.2 in Chapter 8).
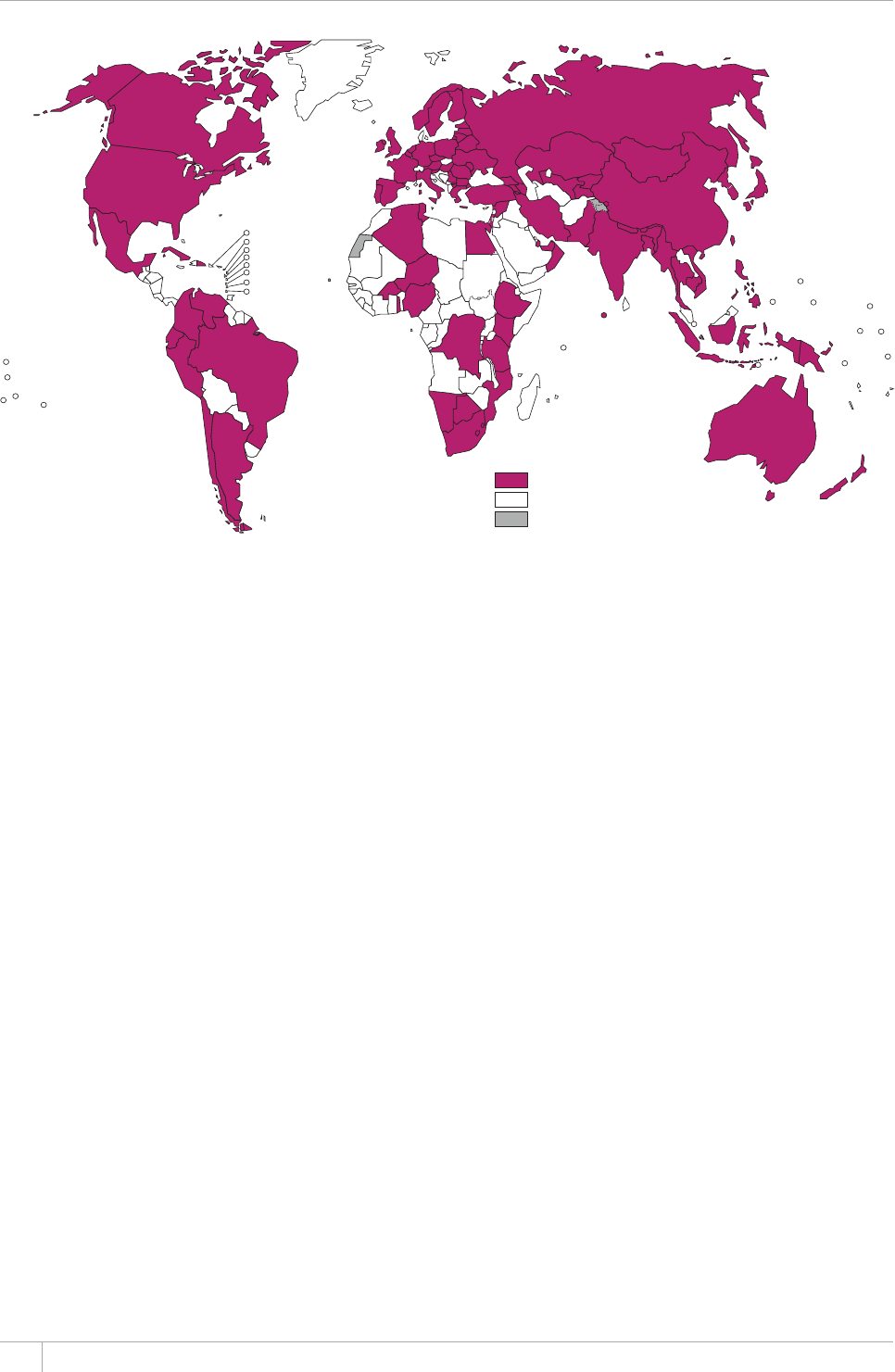
50 GLOBAL TUBERCULOSIS REPORT 2013
FIGURE 4.4
Countries that had notißed at least one case of XDR-TB by the end of 2012
At least one case reported
No cases reported
Not applicable
4.2 Management of drug-resistant TB
%QXGTCIGQHFTWIUWUEGRVKDKNKV[VGUVKPI&56
e diagnosis of DR-TB requires TB patients to be tested
for susceptibility to drugs. Notication data combined with
data from drug resistance surveillance suggest that if all
notied TB patients with pulmonary TB had been tested in
2012, around 300000 cases of MDR-TB would have been
found (section 4.1.3).
Targets included in the Global Plan to Stop TB 2011–2015
are that by 2015 all new cases of TB considered at high risk
of MDR-TB (estimated to be about 20% of all new bacterio-
logically-positive TB cases globally), as well as all previously
treated cases, should undergo DST for at least the rst-line
drugs rifampicin and isoniazid. Similarly, all patients with
MDR-TB should be tested for XDR-TB.
First-line DST results were reported by just over 50%
of countries in 2012 and overall for a small proportion of
cases (Table 4.2). Globally, only 5% of new bacteriological-
ly-conrmed TB cases and 9% of those previously treated
for TB were tested for MDR-TB in 2012. e proportion of
new cases with DST results has increased slightly in recent
years but remains below the target envisaged for 2012
by the Global Plan (Figure 4.5). Coverage was highest in
the European Region, where 72% of new cases and 41% of
previously treated cases were tested for MDR-TB in 2012,
reecting the relatively better access to TB laboratory ser-
vices than elsewhere. Levels of testing were particularly
low in the African and South-East Asia Regions (0.3% and
0.1% of new bacteriologically cases and 3.1% and 0.7% of
previously treated cases, respectively).
Among the 27 high MDR-TB burden countries – which
account for >85% of estimated MDR-TB cases in the world
– the proportion of TB patients who were tested ranged
from 56 to 100% among new cases in 13 of the 14 Euro-
pean countries reporting data (17% in Tajikistan; no data
reported by Azerbaijan), and exceeded 60% among pre-
viously treated cases in nine of these countries. Among
non-European high MDR-TB burden countries, testing for
MDR-TB among new cases was highest in China (3.6%). In
previously treated cases, the coverage of testing was higher
and reached 10% in Indonesia and 12% in China and the
Philippines. In South Africa, 16% of TB cases overall were
tested for MDR-TB although DST data were not available
separately for new and previously treated cases. Five other
countries did not report data, including India, the country
estimated to have the highest number of MDR-TB cases
among notied TB patients (Table 4.2).
Among TB patients who were notied and conrmed to
have MDR-TB in 2012, 23% were reported to have DST per-
formed for both uoroquinolones and second-line injectable
drugs. Second-line DST coverage exceeded 90% in Armenia,
Bulgaria, the Democratic Republic of the Congo, Georgia
and Latvia. South Africa accounted for most of the global
cases for which second-line DST data were reported, as well
as the highest proportion observed in the African Region
(the regional gure drops from 62% to 1% when South Afri-
ca is excluded). Second-line DST reports were available for
53% of MDR-TB cases in the Western Pacic Region, 47% in
the Region of the Americas and 3–8% in the other regions.
Improving the coverage of diagnostic DST is urgently
needed to improve the detection of MDR-TB and XDR-TB.
51GLOBAL TUBERCULOSIS REPORT 2013
TABLE 4.2
DST coverage among TB and MDR-TB cases, globally and for 27 high MDR-TB burden countries and WHO
regions, 2012
0'9$#%6'4+1.1)+%#..;215+6+8'%#5'5 4'64'#6/'06%#5'5 %10(+4/'&/&46$%#5'5
07/$'49+6*
DSTa4'57.65
% OF CASES WITH
&564'57.65
07/$'49+6*
DSTa4'57.65
% OF CASES WITH
&564'57.65
07/$'49+6*
DSTb4'57.65
% OF CASES WITH
&564'57.65
Armenia 64 27 92 100
#\GTDCKLCP –––
Bangladesh 41 557 7.0 142
Belarus 2 164 90 –
Bulgaria 71 142 45 49 100
China 11 472 3.6 12 2 042
&4%QPIQ 12 95 1.3 65 100
Estonia 193 100 46 55
Ethiopia 469 1.0 4.4 –
Georgia 1 931 541 45 341 99
India – – 597 3.6
Indonesia 2 10 43
-C\CMJUVCPc >100 10 443 93 –
-[TI[\UVCP 57 662 61 511 53
Latvia 666 97 100 106 96
Lithuania 1 017 100 350 100 210 77
Myanmar – – 11
Nigeria 11 94 1.2 –
Pakistan 461 0.4 154 1.3 –
Philippines 35 –
4GRWDNKEQH/QNFQXC 1 264 67 933 63 –
4WUUKCP(GFGTCVKQP 32 647 79 12 324 24 –
South Africa – – 11 046 72
Tajikistan 919 17 496 66 345 50
Ukraine 77 5 925 72 –
7\DGMKUVCP 2 703 56 30 356 21
Viet Nam – – –
High MDR-TB burden countries 77 277 3.9 42 851 7.7 16 225 21
#(4 2 216 0.3 3 969 3.1 11 303 62
#/4 22 23 47
'/4 1 990 1.1 1 617 7.6 51 3.2
'74 72 37 774 41 2 523 6.7
5'#4 1 352 0.1 2 292 0.7 1 619
924 3.3 10 2 365 53
Global 136 630 5.1 59 267 8.7 19 245 23
Blank cells indicate data not reported.
– indicates values that cannot be calculated.
a &56KUHQTKUQPKC\KFCPFTKHCORKEKP
b &56KUHQTCàWQTQSWKPQNQPGCPFCUGEQPFNKPGKPLGEVCDNGFTWI
c #RQUUKDNGGZRNCPCVKQPHQTYJ[VJGRGTEGPVCIGHQTPGYECUGUKP-C\CMJUVCPGZEGGFUKUKPCFGSWCVGNKPMCIGUDGVYGGPENKPKECNCPFNCDQTCVQT[TGIKUVGTU
52 GLOBAL TUBERCULOSIS REPORT 2013
FIGURE 4.5
DST coverage among new cases and enrolment on MDR-TB treatment, compared with the targets in the Global
Plan to Stop TB, 2011–2015. Lines indicate the planned targets, blue squares show the situation in 2009–2012 and
orange circles the projected enrolments 2013–2015. Data on projected enrolments in 2015 were incomplete.
Number of patients
b. Enrolment on MDR-TB treatment
2009 2010 2011 2012 2013 2014 2015
0
50 000
100 000
150 000
200 000
250 000
300 000
0
5
10
15
20
25
Percentage of cases
2009 2010 2011 2012 2013 2014 2015
a. DST coverage among new bacteriologically-positive cases
is requires the strengthening of laboratory capacity, the
introduction of new rapid diagnostics and improved report-
ing from diagnostic centres (see Chapter 5). e identi-
cation of XDR-TB cases in countries worldwide (Box 4.2,
Figure 4.4) reects the risk of acquisition of additional sec-
ond-line drug resistance and the transmission of resistant
strains when TB care and prevention (including infection
control) are inadequate.
0QVKßECVKQPQH/&46$ECUGUCPFGPTQNOGPV
on treatment
e low coverage of DST in many countries is one of the
main constraints limiting the detection of MDR-TB among
people diagnosed with TB. Globally, 83 715 cases of MDR-
TB were notied to WHO in 2012, with India, the Russian
Federation and South Africa reporting more than a half of
these cases (Table 4.3). In addition, just over 10 000 rifam-
picin-resistant TB (RR-TB) cases were reported to have
been detected using rapid molecular techniques.1 India,
Kyrgyzstan, the Philippines and Uzbekistan each reported
>500 of such cases.
e 83 715 reported cases of MDR-TB cases represent-
ed 28% of the 300 000 (range, 220 000–380 000) pulmo-
nary TB patients estimated to have MDR-TB in 2012 (Table
4.3), up from 20% in 2011, and 19% of the 450 000 (range:
300 000‒600 000) estimated incident MDR-TB cases in
the world in 2012. Much of the increase between 2011 and
2012 was accounted for by India (4237 to 16 588), South
Africa (10 085 to 15 419)2 and Ukraine (4305 to 6934),
although increases were reported by a total of 17 high
MDR-TB burden countries and all WHO regions with the
exception of the Region of the Americas. In the Democratic
BOX 4.2
XDR-TB in Africa
+PCENWUVGTQH:&46$RCVKGPVUKPTWTCN5QWVJ#HTKEC
made international headlines.a All of the patients from this
cluster who were tested for HIV were found to be infected.
Most of these patients died very quickly. South Africa
TGOCKPUVJGEQWPVT[VJCVTGRQTVUVJGOQUV:&46$ECUGU
KPVJGYQTNFCPFCPPWCNPQVKßECVKQPUJCXGKPETGCUGFHTQO
KPVQKP#DQWVQH/&46$ECUGU
TGRQTVGFKPVJKUEQWPVT[JCXG:&46$
FIGURE B4.2.1
Treatment outcomes for 623 TB patients with XDR-
TB in South Africa, 2010
Completed 6%
Cured 12%
Not evaluated 17%
Lost to follow-up 9%
Treatment failed 8%
Died 49%
By the end of 2012, 15 countries in the African region had
KFGPVKßGFCPFTGRQTVGFCVNGCUVQPGECUGQH:&46$Figure
4.4+PVYQJKIJ/&46$DWTFGPEQWPVTKGUKPVJG
#HTKECP4GIKQP¿VJG&GOQETCVKE4GRWDNKEQHVJG%QPIQ
CPF0KIGTKC¿GCEJTGRQTVGFVJGKTßTUV:&46$ECUG5GXGP
#HTKECPEQWPVTKGUTGRQTVGFUVCTVKPI:&46$RCVKGPVUQP
treatment in 2011 or 2012, most of them in South Africa.
Treatment outcomes reported by South Africa reveal the very
low likelihood of a favourable outcome in such patients and
the high proportion of patients lost to or not evaluated by
the health services (see Figure B4.2.1).
a )CPFJK04/QNN#5VWTO#92CYKPUMK4)QXGPFGT6.CNNQQ7GVCN
Extensively drug-resistant tuberculosis as a cause of death in patients
co-infected with tuberculosis and HIV in a rural area of South Africa. The
Lancet¿
1 ese are in addition to other rifampicin-resistant cases detected
by Xpert MTB/RIF, which were included under MDR-TB notica-
tions following subsequent testing for isoniazid resistance.
2 In South Africa, the number of cases detected was above the esti-
mated number of cases among pulmonary TB patients; this could
reect either that the estimates of the number of MDR-TB cases
among TB patients are too conservative and/or the absence of link-
ages between the clinical and laboratory registers.

53GLOBAL TUBERCULOSIS REPORT 2013
TABLE 4.3
Estimated MDR-TB cases in 2012, notißed cases of MDR-TB and enrolments on MDR-TB treatment
2009–2012, and treatment outcome reporting for 2010 cohort, globally and for 27 high MDR-TB burden
countries and WHO regions
'56+/#6'&/&46$#/10)016+(+'&
27./10#4;6$%#5'5 NOTIFIED CASES %#5'5'041..'&10/&46$64'#6/'06
/&46$%#5'5
4'2146'&9+6*
64'#6/'06176%1/'
#%1*146
BEST LOW HIGH 2009 2010 2011 2012
2012
NOTIFIED /
ESTIMATED
(%)a
2009 2010 2011 2012 N %b
Armenia 250 220 156 177 79 92 37 134 154 101 132 75
#\GTDCKLCP 2 600 3 000 552 596 21 592 406 263
Bangladesh 4 200 3 100 5 200 339 509 513 12 352 339 390 513 329 97
Belarus 2 200 2 100 2 200 1 342 1 576 1 594 1 604 73 200 1 446 1 442 91
Bulgaria 100 130 43 56 55 49 49 43 56 42 36 56 100
China 59 000 52 000 66 000 474 2 792 1 601 3 007 5.1 1 222 1 155 1 906 1 222 44
&4%QPIQ 2 900 670 5 100 91 121 65 2.2 176 191 179 105 121
Estonia 70 56 63 62 63 75 54 64 102
Ethiopia 2 100 1 200 3 000 233 140 212 14 120 199 114
Georgia 630 570 690 369 359 475 346 55 266 737 665 504 140
India 64 000 49 000 79 000 1 660 2 967 4 237 26 1 136 2 967 14 143 74
Indonesia 6 900 5 200 6.2 20 142 260 426 140 77
-C\CMJUVCP 9 000 3 644 3 209 5 705 5 261 7 213 5 777
-[TI[\UVCP 1 600 1 900 566 53 545 566 492 790 441
Latvia 120 100 140 131 105 110 92 124 103 110 101
Lithuania 300 270 330 322 310 296 271 90 322 310 296 271 310 100
Myanmar 6 000 4 600 7 500 192 690 13 64 192 163 442
Nigeria 3 600 2 700 4 500 21 95 107 3.0 0 23 125 23 110
Pakistan 11 000 0 29 000 49 444 344 1 602 15 424 344 1 045 195 44
Philippines 13 000 10 000 16 000 1 073 522 679 5.2 501 2 397 150
4GRWDNKEQH/QNFQXC 1 700 1 600 1 069 1 001 53 334 791 765 –
4WUUKCP(GFGTCVKQP 46 000 43 000 49 000 13 692 13 612 30 13 692 34
South Africa 6 900 9 400 9 070 15 419 >100 4 143 5 402 5 643 6 494 66
Tajikistan 910 1 000 319 333 604 694 76 52 245 535 245 74
Ukraine 6 500 7 000 5 336 4 305 6 934 >100 4 950 7 672 3 902 73
7\DGMKUVCP 4 000 3 700 4 300 654 1 023 43 464 1 491 61
Viet Nam 3 000 4 600 217 101 601 273 7.2 307 101 713 97 96
High MDR-TB
burden countries 270 000 180 000 350 000 40 798 47 772 52 813 75 301 28 24 521 38 942 49 663 69 320 28 793 60
#(4 14 000 62 000 10 741 9 340 5 994 7 209 7 467 9 303 6 166 66
#/4 7 100 4 500 9 600 2 661 3 474 2 967 42 3 153 3 249 3 102 2 374
'/4 0 42 000 496 2 236 12 707 967 756 1 602 676 77
'74 74 000 60 000 33 776 34 199 50 17 169 36 313 42 399 19 496
5'#4 90 000 71 000 110 000 2 560 3 942 6 615 19 202 21 2 040 3 901 4 597 3 113 79
924 74 000 57 000 91 000 2 059 4 295 4 394 4 473 6.0 1 429 2 210 4 946 5 070 2 456 57
Global 300 000 220 000 380 000 46 897 54 887 61 907 83 715 28 30 492 45 872 57 166 77 321 34 281 62
Blank cells indicate data not reported.
– indicates values that cannot be calculated.
a 0QVKßGFECUGUQH/&46$KPCUCRGTEGPVCIGQHVJGDGUVGUVKOCVGQH/&46$ECUGUCOQPICNNECUGUQHRWNOQPCT[6$KPVJGUCOG[GCT6JGRGTEGPVCIGOC[GZEGGF
KHGUVKOCVGUQHVJGPWODGTQH/&46$CTGVQQEQPUGTXCVKXGCPFKHNKPMCIGDGVYGGPVJGENKPKECNCPFNCDQTCVQT[TGIKUVGTUKUKPCFGSWCVG
b 6JGRGTEGPVCIGQH/&46$ECUGUQTKIKPCNN[PQVKßGFKPYKVJQWVEQOGUTGRQTVGF6JGRGTEGPVCIGOC[GZEGGFCUCTGUWNVQHWRFCVGFKPHQTOCVKQPCDQWV/&46$
ECUGUKPKPCFGSWCVGNKPMCIGUDGVYGGPPQVKßECVKQPU[UVGOUHQT6$CPF/&46$CPFVJGKPENWUKQPKPVJGVTGCVOGPVEQJQTVQHECUGUQH/&46$ECUGUHTQOC[GCTRTKQTVQ
2010.
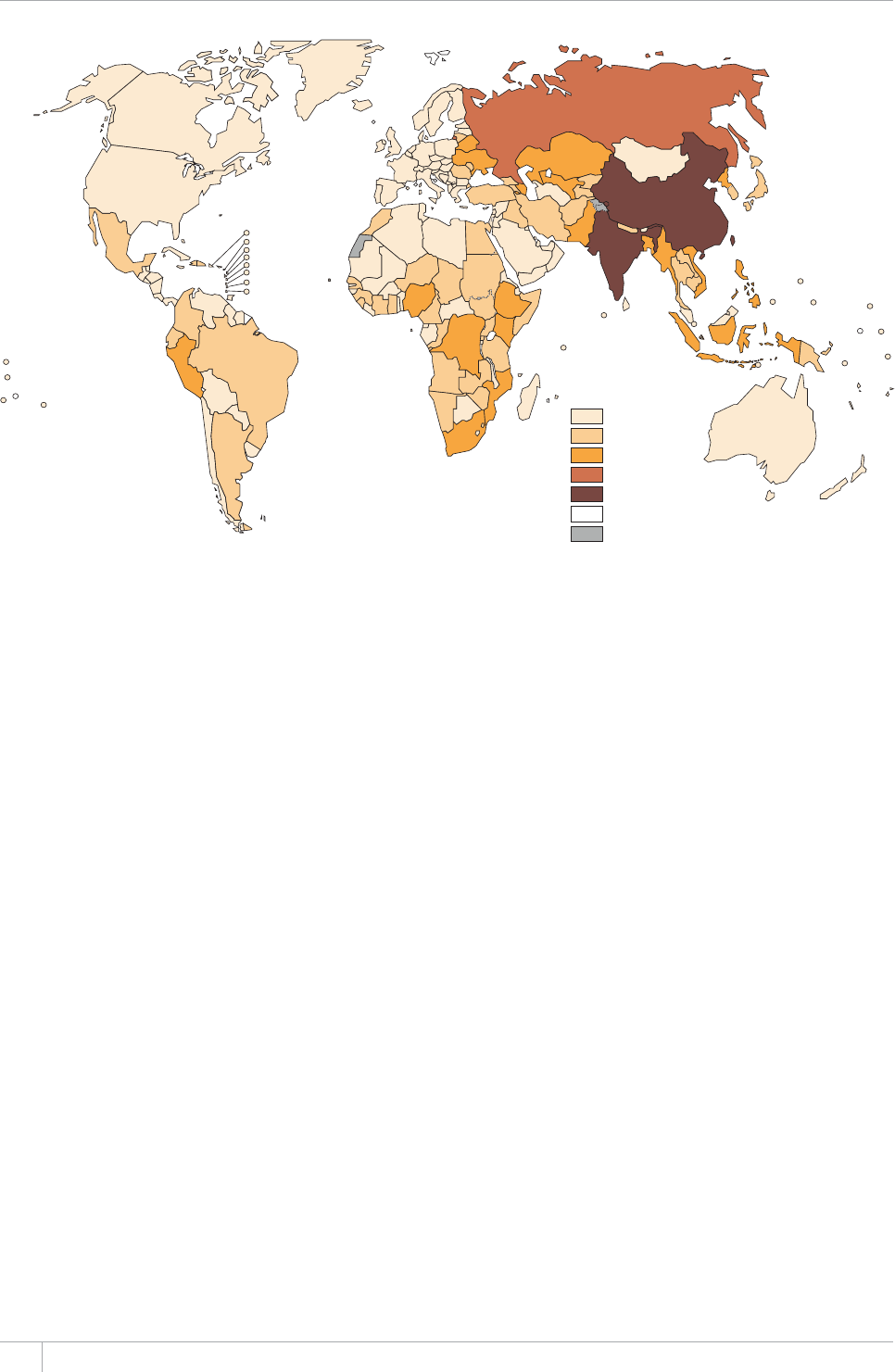
54 GLOBAL TUBERCULOSIS REPORT 2013
Republic of the Congo, the Philippines and Viet Nam, which
detected less than 30% of their estimated burden in 2012,
MDR-TB notications decreased between 2011 and 2012.
Of the MDR-TB cases reported globally in 2012, most (82%)
were detected in either the European Region (36 708), India
(16 588) or South Africa (15 419).
Countries detecting close to 100% of the TB patients esti-
mated to have MDR-TB in 2012 included Estonia, Kazakh-
stan, Latvia, Lithuania, South Africa and Ukraine (Table
4.3). In the African and European Regions and the Region
of the Americas, about 50% of the TB patients estimated
to have MDR-TB were detected in 2012. e lowest gures
were in the two regions with the largest number of cases:
the South-East Asia region (21%) and the Western Pacic
Region (6%). India and China, the two countries estimat-
ed to have the largest numbers of TB patients with MDR-
TB (both over 50000, Figure 4.6), strongly inuence the
overall gures for the South-East Asia and Western Pacic
Regions. China and India, together with the Russian Federa-
tion – which ranks third globally in total cases of MDR-TB –
detected and reported less than one third of the TB patients
estimated to have MDR-TB (5%, 26% and 30% respectively).
e absolute numbers of TB cases started on second-line
treatment for MDR-TB increased from 30492 in 2009 to
77321 in 2012 (+154%). ere was a 40% increase in enrol-
ments between 2011 and 2012 in the 27 high MDR-TB
burden countries, which reected progress in 20 of these
countries and especially in India, Kazakhstan and Ukraine
(Table 4.3). e ratio of the numbers of patients starting
treatment with second-line drug regimens for MDR-TB,
to those notied with MDR-TB in 2012, was 92% globally
FIGURE 4.6
Number of MDR-TB cases estimated to occur among notißed pulmonary TB cases, 2012
MDR-TB cases
0–199
200–1999
2000–19 999
20 000–49 999
≥ 50 000
No data
Not applicable
(82% when RR-TB cases are included), but was lower in the
African (51%) and South-East Asia (83%) Regions (Table
4.3). Waiting lists of people requiring treatment for MDR-
TB are persisting or growing in several countries, particu-
larly when additional RR-TB cases diagnosed using Xpert
MTB/RIF are taken into account. Diagnosis:treatment
gaps of 5% or more were evident in 14 of the high MDR-
TB burden countries in 2012 (Figure 4.7), and the ratio of
MDR-TB cases diagnosed to enrolments on MDR-TB treat-
ment increased by more than 10% between 2011 and 2012
in China, Pakistan and South Africa. e number of XDR-
TB cases reported worldwide increased from 1464 to 2230
between 2011 and 2012. All the WHO regions reported
more XDR-TB cases enrolled on treatment in 2012 than in
2011, reaching 1557 globally in 2012.
Common constraints to treatment scale up include a
critical shortage of trained sta, insucient availability
of second-line medications, inadequate numbers of facili-
ties for treatment and monitoring, incomplete diagnosis
of patients and other weaknesses in the coordination of
activities required for eective programmatic management
of DR-TB. ere is a global shortfall in capacity to place peo-
ple diagnosed with MDR-TB on treatment, and increased
resources for the programmatic management of MDR-TB
are urgently required.
In a few countries, such as Georgia, the Russian Feder-
ation and Ukraine, enrolments have outstripped notica-
tions of MDR-TB in recent years. Possible explanations for
this include frequent empirical treatment of TB patients
considered at risk of having MDR-TB but for whom a labo-
ratory-conrmed diagnosis is missing, incomplete report-
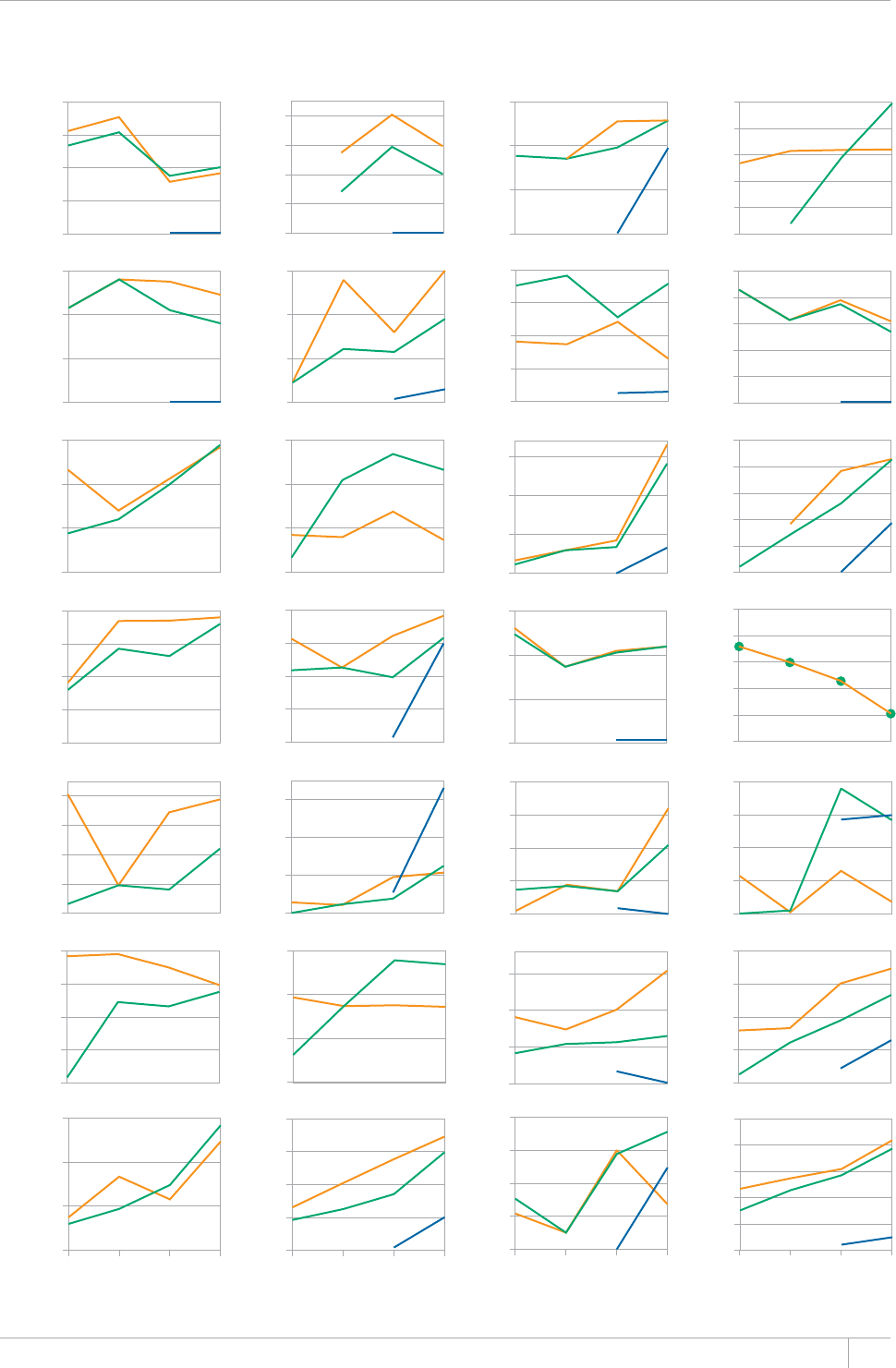
55GLOBAL TUBERCULOSIS REPORT 2013
FIGURE 4.7
MDR-TB cases (orange) and additional rifampicin-resistant TB cases (blue) detected compared with
TB cases enrolled on MDR-TB treatment (green) 2009–2012, globally and in 27 high MDR-TB burden
countries, 2009–2012
Number of cases
0
50
100
150
200
0
200
400
600
800
0
200
400
600
0
20
40
60
0
1000
2000
3000
0
50
100
150
200
0
20
40
60
80
100
Armenia Azerbaijan Bangladesh Belarus
Bulgaria China DR Congo Estonia
0
100
200
300
200
400
600
800
0
5000
10000
15000
0
100
200
300
400
500
Ethiopia Georgia India Indonesia
0
2000
4000
6000
8000
0
250
500
750
1000
0
50
100
150
250
270
290
310
330
350
Kazakhstan Kyrgyzstan Latvia Lithuania
0
200
400
600
800
0
100
200
300
0
500
1000
1500
2000
Myanmar Nigeria Pakistan Philippines
500
1000
1500
2000
2500
300
500
700
900
1100
5000
10000
15000
20000
0
5000
10000
15000
0
200
400
600
800
Republic of Moldova Russian Federation South Africa Tajikistan
2000
4000
6000
8000
0
500
1000
1500
2000
0
200
400
600
800
2009 2010 2011 2012 2009 2010 2011 2012 2009 2010 2011 2012 2009 2010 2011 2012
Ukraine Uzbekistan Viet Nam Global
0
500
1000
1500
2000
2500
0
20 000
40 000
60 000
80 000
100 000
56 GLOBAL TUBERCULOSIS REPORT 2013
ing of laboratory data, or enrolment of ‘backlogs’ or waiting
lists of MDR-TB patients who were detected before 2012.
Among 119 countries reporting sex-disaggregated data
for enrolments, the median male:female ratio was 2. Most
countries that reported data on MDR-TB patient enrol-
ments did not report the inclusion of any children. In the
44 countries that did, the proportion of children ranged
from <1% to 33% of total enrolments.
Many countries envisage increases in the number of
patients enrolled on treatment for MDR-TB between 2013
and 2015. However, global projections remain well below
Global Plan targets, partly as a result of slow rates of
increase as well as incomplete information regarding fore-
casts, notably for China (2015) and the Russian Federation
BOX 4.3
Pharmacovigilance for TB care
2JCTOCEQXKIKNCPEGKUFGßPGFD[
9*1CUÄThe science and actiXities
relatinI to the detection assessment
understandinI and preXention of
adXerse effects or any other druI-
related problem.”
#FXGTUGFTWITGCEVKQPU#&4UECP
lead to a TB patient interrupting
treatment before completion, thus contributing to avoidable
morbidity, drug resistance, treatment failure, reduced
quality of life, or death. It is important to routinely monitor
VJGQEEWTTGPEGQH#&4UKP6$RCVKGPVUQPVTGCVOGPVKP062U
This is particularly relevant in the care of patients with
&46$CPFRCVKGPVUYJQCTG*+8RQUKVKXG
6JTGGCRRTQCEJGUVQRJCTOCEQXKIKNCPEGCTGKPWUG
Spontaneous reporting. This involves the reporting of
#&4U¿GIQVQVQZKEKV[CUUQEKCVGFYKVJCOKPQIN[EQUKFGU
– to the national pharmacovigilance centre.
Targeted spontaneous reporting. This is an extension
of spontaneous reporting that can be focused on the
UWTXGKNNCPEGQHUGTKQWUCFXGTUGGXGPVUKPURGEKßERCVKGPV
ITQWRUUWEJCURCVKGPVUYKVJ/&46$
Cohort event monitoring (CEM). This is an active form of
surveillance, similar in design and management to an
epidemiological cohort study. CEM is particularly well
suited to the post-marketing surveillance of new drugs.
In 2012, WHO produced a handbook on pharmacovigilance
for TB.a WHO offers technical assistance to countries for
the introduction and strengthening of pharmacovigilance
in their programmes. The handbook explains how
pharmacovigilance can be effectively implemented in a TB
programme through key stakeholders, including regulators
and manufacturers, and provides a step-by-step approach
to identifying signals, assessing the relationship between
an event and a drug, determination of causality, acting on
QDUGTXCVKQPUCPFEQOOWPKECVKQPQHßPFKPIU
a # practical handbook on the pharmacoXiIilance of medicines used in
the treatment of tuberculosis enhancinI the safety of the TB patient.
)GPGXC9QTNF*GCNVJ1TICPK\CVKQPwww.who.int/medicines/
RWDNKECVKQPURJCTOCEQXKIKNCPEGAVD).
(2013) (Figure 4.5b). To reach the targets set out in the
Global Plan and advance towards universal access to treat-
ment, a bold and concerted drive is still needed on many
fronts of TB care, particularly in the countries where the
burden is highest. e capacity to address this challenge has
increased in recent years as a result of the intensied tech-
nical assistance provided by international organizations.
With the reform of the Green Light Committee (GLC) struc-
ture in 2011, and the creation of regional level committees
(rGLCs) in all six WHO regions, international support to
national eorts to strengthen programmatic management
of DR-TB is now focused on devolving available resources
and technical assistance closer to countries.
4.2.3 Treatment outcomes for MDR-TB and XDR-TB
Standardized monitoring methods and indicators have
allowed countries to report MDR-TB treatment outcomes
in a comparable manner for several years. In 2013, the de-
nitions for treatment outcomes were simplied and the
reporting requirements changed to allow for the inclusion
of RR-TB cases in the MDR-TB cohort (Box 4.4).
e number of cases reported in annual MDR-TB treat-
ment outcome cohorts has tripled between 2007 and 2010,
reecting increases in all regions (Figure 4.8). All high
MDR-TB burden countries have now reported treatment
outcomes for at least one annual cohort since 2007.
A total of 107 countries reported outcomes for more
than 34000 MDR-TB cases started on treatment in 2010
(Table 4.3). is is equivalent to 62% of the number of
MDR-TB cases notied by countries in the same year. e
low proportion reects weaknesses in reporting systems to
reconcile outcome data with notications. e Global Plan
envisages that by 2015, all countries will report outcomes
for all notied MDR-TB cases. In 2010, only 71 countries
– including 13 high MDR-TB burden countries – reported
outcomes for a cohort whose size exceeded 80% of the orig-
inal number of MDR-TB notications in 2010.
Overall, the proportion of MDR-TB patients in the 2010
cohort who successfully completed treatment was 48%,
while 28% of cases were reported as lost to follow-up or had
no outcome information. Treatment success was highest in
the Eastern Mediterranean Region (56%), as well as in the
Region of the Americas (54%) where this proportion has
increased steadily since 2007 alongside a reduction in the
proportion of patients whose treatment outcome was not
evaluated. In the 2010 cohort, deaths were highest in the
African Region (17%) and the proportion of patients whose
treatment failed was highest in the European Region (11%).
e Global Plan’s target of achieving at least 75% treatment
success in MDR-TB patients by 2015 was only reached by
34/107 countries reporting outcomes for the 2010 cohort,
but included three high MDR-TB burden countries: Bangla-
desh, Ethiopia and VietNam.
Among a subset of 795 XDR-TB patients in 26 countries,
treatment success was 20% overall and 44% of patients
died; excluding South Africa, the gures were 27% and 28%
respectively (Box 4.2).
A PRACTICAL HANDBOOK
ON THE PHARMACOVIGILANCE
OF MEDICINES USED
IN THE TREATMENT OF
TUBERCULOSIS
ENHANCING THE SAFETY OF THE TB PATIENT
57GLOBAL TUBERCULOSIS REPORT 2013
FIGURE 4.8
Treatment outcomes for patients diagnosed with MDR-TB by WHO region, 2007–2010 cohorts.
The total numbers of cases with outcome data are shown beside each bar.
Africa The Americas
Eastern Mediterranean Europe
South-East Asia Western Pacific
Global
2010
2009
2008
2007
2010
2009
2008
2007
2010
2009
2008
2007
2010
2009
2008
2007
2010
2009
2008
2007
2010
2009
2008
2007
2010
2009
2008
2007
0% 20% 40% 60% 80% 100%
0% 20% 40% 60% 80% 100%
6 166
6 143
5 496
4 570
676
511
262
128
34 281
23 250
15 565
10 959
3 113
1 140
413
253
2 374
2 298
1 732
1 458
19 496
12 131
6 904
4 097
2 456
1 027
758
453
Cured Completed Died
Treatment failed Lost to follow-up Not evaluated
Progressing towards the target for treatment success
requires the scale up of treatment programmes globally,
enhancing the eectiveness of drug regimens, support to
patients to avoid treatment interruption and improved
data collection. In particular, countries need to analyse the
poor treatment outcomes observed in MDR-TB cases and
intensify measures to improve adherence and monitoring.
TB programmes need to apply a package of services for
MDR-TB patients that include free TB and ancillary med-
ications, free laboratory testing, enablers and social sup-
port, and the use of short treatment regimens following
current WHO policy in selected patients. e treatment of
XDR-TB patients in particular remains very unsatisfactory
and more eective regimens for this condition are urgently
required.
4.2.4 Other aspects of MDR-TB programme
management
During their illness, patients with MDR-TB may be cared
for as either outpatients or within hospitals, usually sec-
ondary or tertiary facilities. WHO recommends that, where
possible, patients with MDR-TB should be treated using
ambulatory or community-based care rather than models
of care based principally on hospitalization.
National policies and practices dier in the predominant
model of care that is employed. Among the high MDR-TB
burden countries, the lowest level of hospitalization was
reported by the Philippines (5% of MDR-TB patients),
while levels in Eastern European countries ranged between
75 and 100% but were lower in Central Asia (30–50% in
Kazakhstan, Tajikistan and Uzbekistan). In the African
Region, there is wide variation in the extent to which

GLOBAL TUBERCULOSIS REPORT 2013
patients with MDR-TB are hospitalized, ranging from 10%
of patients (Democratic Republic of the Congo) to 100%
(Ethiopia and Nigeria). Globally, the average duration of
hospital stay ranged from 7 to 240 days (median: 84 days).
e number of visits to a health facility after diagnosis of
BOX 4.4
WHO deßnitions of treatment outcomes for
RR-TB, MDR-TB and XDR-TB
Cured Treatment completed as recommended by the
national policy without evidence of failure AND three or
more consecutive cultures taken at least 30 days apart are
negative after the intensive phase.
Treatment completed Treatment completed as
recommended by the national policy without evidence
of failure BUT no record that three or more consecutive
cultures taken at least 30 days apart are negative after the
intensive phase.
Treatment failed Treatment terminated or need for
permanent regimen change of at least two anti-TB drugs
DGECWUGQH
lack of conversion by the end of the intensive phase; or
bacteriological reversion in the continuation phase after
conversion to negative; or
evidence of additional acquired resistance to
àWQTQSWKPQNQPGUQTUGEQPFNKPGKPLGEVCDNGFTWIUQT
adverse drug reactions.
Died A patient who died for any reason during the course
of treatment.
Lost to follow-up A patient whose treatment was
interrupted for two consecutive months or more.
Not evaluated A patient for whom no treatment outcome
is assigned (this includes cases ‘transferred out’ to another
treatment unit and whose treatment outcome is unknown).
Successfully treated The sum of cured and treatment
completed.
Cohort #ITQWRQHRCVKGPVUYJGTG446$JCUDGGP
FKCIPQUGFKPENWFKPI/&46$CPF:&46$CPFYJQYGTG
UVCTVGFQPCHWNNEQWTUGQHCUGEQPFNKPG/&46$FTWI
TGIKOGPFWTKPICURGEKßGFVKOGRGTKQFGIVJGEQJQTVQH
/&46$ECUGUTGIKUVGTGFKPVJGECNGPFCT[GCT6JKU
group forms the denominator for calculating treatment
QWVEQOGU9KVJVJGTGXKUGFFGßPKVKQPUany patient found to
haXe druI-resistant TB and placed on second-line treatment
is remoXed from the druI-susceptible TB outcome cohort.
This means that management of the basic management unit
TB register and of the second-line TB treatment register
needs to be coordinated to ensure proper accounting of the
outcomes of treatment.
/QTGFGVCKNUQPVJGFGßPKVKQPQHEQPXGTUKQPTGXGTUKQPCPF
the end of the intensive phase are provided in the WHO
guidance.a
a &eßnitions and reportinI framework for tuberculosis – 201 reXision
9*1*6/6$)GPGXC9QTNF*GCNVJ1TICPK\CVKQPwww.
YJQKPVKTKUDKVUVTGCOAGPIRFH).
MDR-TB also varies markedly among countries, from 30
or less (Bangladesh, the Democratic Republic of the Con-
go, Estonia, Pakistan, and VietNam) to over 600 (Bulgaria,
Indonesia, Latvia, Tajikistan and Uzbekistan).
Palliative and end-of-life care delivered through home-
based or institutional services is fundamental to allevi-
ate the suering associated with MDR-TB, particularly in
patients with advanced disease that is not responding to
treatment. Only eleven high MDR-TB burden countries –10
in the European region plus South Africa – reported that
they provided such care within the scope of their NTPs.
When considered in the context of the poor outcomes
reported in patients with MDR-TB and especially XDR-TB,
this nding attests to the persistent, huge unmet need for
palliative care services in countries with the largest bur-
dens of drug-resistant TB.
Among 18 high MDR-TB burden countries providing
information on the quality of second-line drugs in the pub-
lic sector in 2012, two countries reported that all of the
drugs that they used conformed only to national regulatory
norms. In the other 16 countries, most reported conformi-
ty to international standards for all supplies of kanamycin
(11), capreomycin (9, with 2 other countries not using it),
levooxacin (10, with 1 other not using it), ethionamide/
prothionamide (12), cycloserine/terizidone (11) and p-ami-
nosalicylic acid (10, with 2 others not using it).
More information is required to adequately monitor TB
patients on MDR-TB treatment than is needed for drug-
susceptible TB. e denitions for monitoring of RR-TB
and MDR-TB and their outcomes were revised in 2013
(see Chapter 3 and Box 4.4). e employment of electron-
ic systems to manage patient data is therefore strongly
encouraged. One of the Global Plan’s targets is that all 27
high MDR-TB countries manage their data on treatment
of MDR-TB patients electronically by 2015. By 2012, 19
reported that national databases were in place for MDR-
TB patients (see Figure 2.16 in Chapter 2). ese systems
dier markedly from one country to another, varying
from individual patient medical records accessible online
to the periodic collation of records from registers across
the country. Before introducing electronic systems to han-
dle patient data, WHO recommends that NTPs undertake
a detailed assessment of their needs and expectations
and then try to match these with the best suited infor-
matics solution. A fragmentary approach with parallel
systems dealing with dierent programme components
(for example, management of data for patients with drug-
susceptible and drug-resistant TB in separate systems)
should be avoided. Guidance on the design and implementa-
tion of electronic systems for recording and reporting data
was produced by WHO and technical partners in 2012.1
4 Electronic recording and reporting for TB care and control. Geneva,
World Health Organization, 2013 (WHO/HTM/TB/2011.22).
59GLOBAL TUBERCULOSIS REPORT 2013
%*#26'4
Diagnostics and laboratory
strengthening
KEY FACTS AND MESSAGES
■ The conventional laboratory tests for the diagnosis of
TB, which have been used for decades, are sputum smear
microscopy and bacterial culture. Diagnosis based on
cultured specimens is the reference standard but results
take weeks to obtain. Drug susceptibility testing (DST) on
EWNVWTGUKUWUGFVQFGVGEVTGUKUVCPEGVQßTUVCPFUGEQPFNKPG
TB drugs.
■ There have been important breakthroughs in TB
FKCIPQUVKEUKPTGEGPV[GCTU+P9*1GPFQTUGFVJGßTUV
rapid molecular test that can be used to simultaneously test
HQTRWNOQPCT[6$CPFTKHCORKEKPTGUKUVCPEG:RGTV® MTB/
4+(6JGUGPUKVKXKV[QHVJGVGUVKUOWEJDGVVGTVJCPUOGCT
microscopy and is comparable to solid culture. In 2013,
a review of the 2010 policy was initiated, to examine the
substantial body of new evidence on the use and positioning
QH:RGTV/6$4+(HQTVJGFKCIPQUKUQHRWNOQPCT[
extrapulmonary and paediatric TB. Updated guidance is
expected in 2014.
■ :RGTV/6$4+(KUDGKPITCRKFN[CFQRVGFD[EQWPVTKGU
$[VJGGPFQH,WPG)GPG:RGTVOCEJKPGUCPF
OKNNKQP:RGTV/6$4+(ECTVTKFIGUJCFDGGPRTQEWTGF
D[QHVJGEQWPVTKGUGNKIKDNGHQTEQPEGUUKQPCNRTKEGU
Almost half (49%) of reporting low- and middle-income
countries and territories indicated that WHO policy
IWKFCPEGQP:RGTV/6$4+(JCFDGGPKPEQTRQTCVGFKPVQ
VJGKTPCVKQPCNIWKFGNKPGU5QWVJ#HTKECKUVJGßTUVEQWPVT[VQ
CFQRV:RGTV/6$4+(CUVJGRTKOCT[FKCIPQUVKEVGUVHQT6$
replacing smear microscopy.
■ Laboratory capacity to conduct high-quality sputum
UOGCTOKETQUEQR[TGSWKTGUUKIPKßECPVUVTGPIVJGPKPI1PN[
14 of the 22 HBCs met the target of having 1 microscopy
EGPVTGRGTaRQRWNCVKQPKPCPFQPN[GKIJV
reported a programme for external quality assessment that
covered at least 95% of all centres in the country.
■ Globally, laboratory capacity to perform DST continues
to be low and is not growing quickly enough to ensure that
6$RCVKGPVUYKVJ/&46$CTGRTQORVN[FKCIPQUGF(TQO
2009 to 2012, the percentage of new and previously treated
TB patients receiving DST increased from 4% to 5% and
HTQOVQTGURGEVKXGN[6JG':2#0&6$RTQLGEV
which started in 2009 and has entered a phase of routine
testing in 25 countries, shows how it is possible to introduce
routine testing for drug resistance and achieve considerable
KPETGCUGUKPVJGPWODGTQH/&46$ECUGUFGVGEVGF
■ The national reference laboratory of Uganda has become
the newest member of the WHO/Global Laboratory Initiative
).+5WRTCPCVKQPCN4GHGTGPEG.CDQTCVQT[54.0GVYQTM
ßNNKPICETKVKECNIGQITCRJKECNICRKP'CUV#HTKEC
e early, rapid and accurate detection of TB and drug resis-
tance relies on a well-managed and equipped laboratory
network. Laboratory conrmation of TB and drug resis-
tance is critical to ensure that people with TB signs and
symptoms are correctly diagnosed and have access to the
correct treatment as soon as possible.
e conventional laboratory tests for the diagnosis of
TB, which have been used for decades, are sputum smear
microscopy and culture. Diagnosis based on culture is the
reference standard but results take weeks to obtain. Drug
susceptibility testing (DST) on cultured specimens is the
conventional method used to detect resistance to rst- and
second-line TB drugs. Following increased investments in
TB research and development in the past decade (Chapter
8), there have been important breakthroughs in TB diag-
nostics. In 2008, rapid molecular tests (line probe assays,
or LPAs) for detection of RR-TB and MDR-TB using posi-
tive sputum specimens or cultures were recommended by
WHO. In 2010, the rst rapid molecular test that can be
used to simultaneously test for TB and rifampicin resis-
tance, Xpert® MTB/RIF (Cepheid, Sunnyvale, CA, USA),
was recommended for diagnosis of pulmonary TB and
rifampicin resistance in adults. e sensitivity of the test
is much better than smear microscopy and similar to solid
culture.1
Although laboratories play a fundamental role in TB care
and control, only 57% of the 4.6 million new pulmonary
TB patients notied globally in 2012 were bacteriologically
conrmed using a WHO-recommended diagnostic meth-
od. Low coverage of laboratory conrmation may result in
people without TB needlessly being enrolled on TB treat-
ment, while true TB cases are being missed. Furthermore,
the 5.7 million incident (new and relapse) TB patients diag-
nosed and notied to NTPs in 2012 represent only 66% of
the estimated 8.6 million incident TB cases globally. e
gap reects both underreporting of diagnosed TB cases and
failure to diagnose cases at all; the latter can be attributed
in part to weak laboratory capacity in many countries.
Detection of TB without investigating for drug resis-
tance can lead to poor treatment outcomes, additional and
unnecessary suering and costs for patients and further
spread of drug-resistant strains. While there was a small
increase between 2011 and 2012, only 5.1% of new cases
and 8.7% of previously treated cases received DST in 2012.
1 Steingart KR et al. Xpert® MTB/RIF assay for pulmonary tubercu-
losis and rifampicin resistance in adults (Review). Cochrane Data-
base of Systematic Reviews 2013, Issue 1. Art. No.: CD009593. 2013.
60 GLOBAL TUBERCULOSIS REPORT 2013
Of the 300 000 cases of MDR-TB estimated to exist among
notied TB patients with pulmonary TB in 2012 (i.e. the
group of patients known to NTPs and that could be tested
for drug resistance using WHO-recommended diagnostic
tests), only 83715 received a laboratory-conrmed diagno-
sis of MDR-TB and were notied in 2012. In addition, just
over 10 000 RR-TB cases were detected using rapid molec-
ular methods, though without results for isoniazid DST at
the time of reporting. Given the large burden of undiag-
nosed DR-TB, strengthening DST capacity is a high priority
for NTPs (see also Chapter 4).
is chapter has three parts. Section 5.1 summarizes
the key developments in WHO guidance on TB diagnostics
and laboratory strengthening during 2012–2013. Section
5.2 provides the status of laboratory capacity globally,
regionally and nationally based on data reported to WHO
by countries in 2013. e focus is on the 36 countries in
the combined list of 22 HBCs and 27 high MDR-TB burden
countries. Innovative public–private mix (PPM) laboratory
initiatives are highlighted as well. Section 5.3 describes
recent achievements in strengthening TB laboratories, cov-
ering incorporation of WHO guidance into policy and prac-
tice at country level and the latest status of progress of two
multinational projects (EXPAND-TB and TBXpert) that are
helping to introduce new diagnostics.
5.1 Developments in WHO policy guidance
on TB diagnostics and laboratory
strengthening, 2012–2013
WHO follows a systematic process for policy development
on TB diagnostics, involving synthesis of the available evi-
dence through systematic reviews and meta-analyses where
possible, assessment of the evidence by an external Expert
Group using the GRADE approach,1 and development of
policy guidance2 for dissemination to Member States and
other stakeholders.3 Policy documents are reviewed every
3–5 years, and revised as necessary when new evidence
becomes available.
e rst WHO policy guidance on the use of Xpert®
MTB/RIF was issued in December 2010. e recommen-
dations were that Xpert MTB/RIF should be used as the
initial diagnostic test in individuals at risk of having MDR-
TB or HIV-associated TB (strong recommendation), and
that Xpert MTB/RIF could be used as a follow-on test to
microscopy in settings where MDR and/or HIV is of lesser
concern, especially in smear-negative specimens (this was
a conditional recommendation, recognizing major resource
implications). e 2010 recommendations applied to the
use of Xpert MTB/RIF in sputum specimens only, as data
on its performance (sensitivity and specicity) for testing
of extrapulmonary specimens at that time were limited.
e recommendations applied to children, but only based
on generalization of data from adults.
Following rapid uptake of Xpert MTB/RIF (see section
5.2), a substantial body of new evidence had been generat-
ed by 2013.4 is included much more data about the test’s
performance characteristics (sensitivity and specicity) in
a wide range of laboratory and epidemiological settings,
additional data on test accuracy in detection of extrapul-
monary and paediatric TB, and more evidence about aord-
ability and cost-eectiveness from early implementers in
a limited number of settings. WHO therefore embarked
on a review of policy guidance in 2013. ree systematic
reviews were commissioned on the sensitivity and specic-
ity of Xpert MTB/RIF for the diagnosis of pulmonary and
extrapulmonary TB and RR-TB, in adults and children. A
review of published studies on the aordability and cost-ef-
fectiveness of Xpert MTB/RIF was also conducted.
An Expert Group convened by WHO met in May 2013
to review the expanded body of evidence, according to
GRADE procedures. Based on the outcomes of the review
and the recommendations of the Expert Group, which were
also supported by WHO’s Strategy and Technical Advisory
Group for TB (STAG-TB) in June 2013, updated WHO pol-
icy guidance was under development at the time that the
current report went to press. Upon nalization, the recom-
mendations are expected to have a major impact on further
country adoption of Xpert MTB/RIF into diagnostic and
clinical algorithms.
Several other new TB diagnostic tests are on the hori-
zon, in various stages of research and development (see
Chapter 8). Once data on their performance are available in
varying epidemiological settings, WHO will be in a position
to evaluate their performance and develop corresponding
policy guidance. A comprehensive list of existing WHO pol-
icy documents, including those on the use of microscopy,
culture, DST and non-commercial and molecular methods,
can be found at: http://www.who.int/tb/laboratory/policy_
statements
In addition to diagnostics, WHO
also develops guidance in other areas
of laboratory strengthening. In 2013,
the WHO Tuberculosis laboratory bio-
safety manual was issued, featuring a
risk-based approach that guides the
essential biosafety measures required
for performing dierent technical
procedures. e manual describes the
combination of good laboratory practices together with
administrative controls, containment principles, safety
equipment and laboratory facilities that are required to
minimize the generation of infectious aerosols and thus
prevent laboratory-acquired infections. e risk-based
approach to laboratory biosafety is framed around a three-
tiered system of ‘low’, ‘moderate’ and ‘high’ TB risk precau-
tions:
1 www.gradeworkinggroup.org
2 WHO handbook for guideline development. Geneva, World Health
Organization, 2012.
3 WHO policies on TB diagnostics are available at: www.who.int/tb/
laboratory/policy_statements
4 Weyer K et al. R apid molecular T B diagnosis: ev idence, polic y-making
and global implementation of Xpert® MTB/RIF. European Respirato-
ry Journal. November 22, 2012, doi: 10.1183/09031936.00157212

61GLOBAL TUBERCULOSIS REPORT 2013
Low TB risk precautions. ese apply to direct acid-fast
bacilli (AFB) microscopy and to Xpert MTB/RIF.
Moderate TB risk precautions. ese apply to the pro-
cessing of sputum specimens for primary culture inoc-
ulation, direct testing (i.e. on sputum smear-positive
samples) using direct non-commercial drug susceptibil-
ity assays and LPAs.
High TB risk precautions in TB containment laborato-
ries. ese apply to procedures used to manipulate cul-
tures (solid and liquid) for identication and DST, and
for indirect testing (i.e. on culture isolates) using LPA
and non-commercial DST.
5.2 Status of laboratory capacity globally,
regionally and nationally
Diagnosis of TB in most low- and middle-income countries
still relies on low-cost sputum smear microscopy, despite its
relatively low sensitivity and inability to detect drug resis-
tance. e Global Plan to Stop TB 2011–2015 includes the tar-
get that countries maintain at least one smear microscopy
centre per 100 000 population. Globally the target has been
met (1.1 centres per 100 000 population in 2012), but con-
siderable disparities remain at regional and country levels
(Table 5.1). Eight of the 22 HBCs did not meet the target in
2012: Bangladesh, China, Myanmar, Nigeria, Pakistan, the
Russian Federation, South Africa and Viet Nam. Overall,
the Western Pacic and Eastern Mediterranean Regions
had less than one centre per 100000 population.
Given the continued critical role of microscopy in
TB detection and monitoring of treatment, ensuring
high-quality performance of smear microscopy is essential.
Of the 153 countries and territories that reported data on
the number of smear microscopy centres in 2012, only 39%
indicated the existence of an external quality assessment
programme that covered all centres in the country. Among
the 22 HBCs, only three reported such a programme that
encompassed all centres in 2012 (Bangladesh, India and
Viet Nam), ve reported a programme that included at least
95% of centres (Cambodia, China, Myanmar, the Russian
Federation and South Africa), and 14 reported a programme
that included at least 80% of centres.
In 2009, WHO recommended the use of the more sensi-
tive uorescent light-emitting diode (LED) microscopy as
a replacement for traditional Ziehl–Neelsen (ZN) micros-
copy. Globally the switch to LED microscopes has been
gradual, and they were reported to be present in only 2%
of microscopy centres in 2012. Overall in 2012, the African
Region was the most advanced in rolling out LED micro-
scopes (6% of microscopy centres), led by South Africa
where 97% of microscopy centres were reported to have
them. Other HBCs in the African Region have shown sig-
nicant increases in uptake from 2011 to 2012, including
the United Republic of Tanzania (3% to 17% of microscopy
centres) and Mozambique (<1% to 9%).
e current target in the Global Plan to Stop TB 2011–
2015 for both culture and DST (to at least rifampicin and
isoniazid) capacity is one laboratory per 5 million popu-
lation. In 2012, 14 of the 27 high MDR-TB burden coun-
tries did not reach the target (Table 5.1; there were two
additional countries that did not report data). Of these 27
countries, 9 reported more than one laboratory per 5 mil-
lion population using LPAs – a high-throughput molecular
tool that can be used at central and regional levels to rapid-
ly detect resistance to rifampicin and, in some cases, isoni-
azid. e nine countries comprise eight European countries
and South Africa.
Of the 147 countries and territories that reported
numbers of laboratories with capacity to perform DST, 22
indicated that such capacity did not exist in 2012. While
countries and territories with small TB patient populations
may nd it more practical to send specimens to neighbour-
ing countries for DST than to establish national capacity,
countries with larger patient populations should aim as
a priority to build sustainable DST capacity in-country
to allow timely diagnosis of drug-resistant strains. Eight
countries reported more than 1000 notied TB cases in
2012 yet reported having no capacity to perform DST:
Afghanistan, Chad, Eritrea, Guinea-Bissau, Liberia, Papua
New Guinea, Sierra Leone and Somalia.
Quality-assured DST is critical to ensure accurate detec-
tion of drug resistance for subsequent treatment decisions
and to avoid false diagnoses. Of the high TB and MDR-TB
burden countries that reported on external quality assess-
ment coverage of DST laboratories (34 of 36), 27 (79%)
reported having a scheme that encompassed all DST lab-
oratories. Of the 117 countries globally that reported on
external quality assessment coverage of DST laboratories,
70% (82 countries) reported such a scheme.
Given its high sensitivity to detect TB and rifampicin
resistance together with its ability to be placed at relative-
ly low levels of laboratory networks, Xpert MTB/RIF has
been rapidly adopted by countries. By the end of June 2013,
3.2 million test cartridges and 1402 GeneXpert machines
(comprising 7553 machine modules) had been procured in
88 of the 145 countries eligible to purchase machines and
cartridges at concessional prices (Figure 5.1).1 e current
price per cartridge is US$9.98, following a novel nancing
agreement reached in August 2012 between the manufac-
turer and the United States Agency for International Devel-
opment (USAID), the United States President’s Emergency
Plan for AIDS Relief (PEPFAR), UNITAID and the Bill &
Melinda Gates Foundation. South Africa alone accounts
for 43% of the modules and 60% of the cartridges procured
globally, and is aiming to position Xpert MTB/RIF as a
replacement for microscopy for the diagnosis of TB. After
South Africa, leading procurers include India, Pakistan,
Zimbabwe and Nigeria.
e complete or partial replacement of microscopy
by Xpert MTB/RIF as the initial diagnostic test and the
increasing number of rifampicin-resistant cases being
detected by Xpert MTB/RIF will require adjustment of
countries’ smear, culture and DST capacities going forward.
1 http://www.who.int/tb/laboratory/mtbrifrollout/
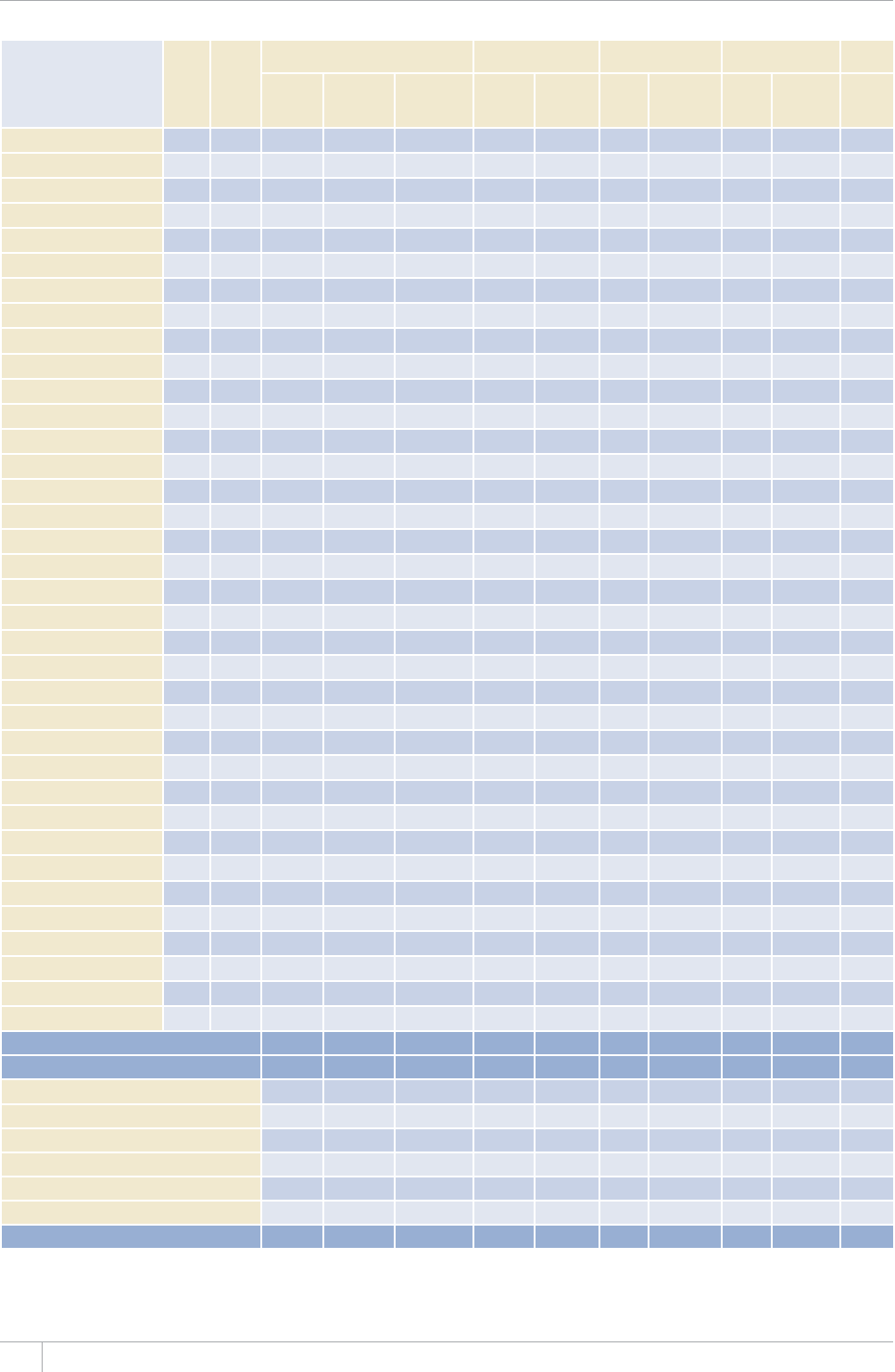
62 GLOBAL TUBERCULOSIS REPORT 2013
TABLE 5.1
Laboratory capacity, 2012a
HIGH
TB
$74&'0
HIGH
/&46$
$74&'0
5/'#4/+%415%12; %7.674' &47)575%'26+$+.+6;
TESTING .+0'241$'#55#; :2'46
/6$4+(
07/$'4
1(.#$14#-
614+'5
.#$14#614+'5
2'4
POPULATION
2'4%'06#)'1(
.#$14#614+'5
USING LED
/+%415%12'5
07/$'41(
.#$14#61-
4+'5
.#$14#-
614+'52'4
5 MILLION
POPULATION
07/$'4
OF
.#$14#-
614+'5
.#$14#614+'5
2'4/+..+10
POPULATION
07/$'4
OF LABO-
4#614+'5
.#$14#614+'5
2'4/+..+10
POPULATION
07/$'4
OF SITES
Afghanistan 603 2.0 2 2 0.3 0 0 0 0 1
Armenia 30 1.0 0 1 1.7 1 1.7 1 1.7 0
#\GTDCKLCP 72 47 31.6 1 0.5 7
Bangladesh
1 070 0.7 2 3 3 1 12
Belarus 196 2.1 2 29 15 4.3 4.3
$TC\KN 4 000 2.0 – 220 5.5 35 0.9 0.2 13
Bulgaria 34 0.5 0 31 21 14 9.6 4 2.7 0
Cambodia 214 1.4 10 3 1.0 1 0.3 0 0 6
China
0.2 2 1 014 3.7 190 0.7 21 16
&4%QPIQ
1 522 2.3 40.3 20.2 1 26
Estonia 5 0.4 10 0 2 7.7 2 7.7 2 7.7 2
Ethiopia
2 531 050.31 50.3 7
Georgia 11 0.3 9 2 2.3 1 1.1 2 2.3 1
India
1.1 2 70 0.3 0.2 33 0.1 32
Indonesia
5 566 2.3 0 46 0.9 5 0.1 2 9
-C\CMJUVCP 466 2.9 0 22 22 11 3.4 4
Kenya 4.2 2 0.2 2 0.2 2 0.2 15
-[TI[\UVCP 122 2.2 0 11 10 3 2.7 2 7
Latvia 16 0 4 9.7 1 2.4 1 2.4 2
Lithuania –– – – –
/Q\CODKSWG 300 1.2 9 3 0.6 2 0.4 0 0 12
Myanmar 0.9 14 2 0.2 2 0.2 2 0.2 3
Nigeria
1 314 250.13 40.132
Pakistan
70.2 40.1 2 15
Philippines
2 565 2.7 13 0.7 3 0.2 1 17
4GRWDNKEQH/QNFQXC –– – – –
4WUUKCP(GFGTCVKQP
1 031 0.7 – 117 4.1 110 –
South Africa 0.4 97 15 1.4 15 1.4 15 1.4 100
Tajikistan 1.1 4 3 1.9 1 0.6 1 0.6 3
Thailand 1.6 6 65 4.9 1.3 12 0.9 14
Uganda 1 152 3.2 4 0.6 4 0.6 4 0.6 25
Ukraine 5 9.4 41 4.5 0 0 15
746CP\CPKC 945 2.0 17 4 0.4 1 0.1 3 0.3 13
7\DGMKUVCP 291 1.0 1 7 1.2 3 0.5 3 0.5 7
Viet Nam 0.9 25 1.4 2 0.1 2 0.1 22
<KODCDYG 1.3 1 2 0.7 2 0.7 0 0 17
High-burden countries – 1.0 2 – 1.8 – 0.5 – 0.1 –
High MDR-TB burden countries – 0.9 2 – 1.9 – 0.6 – 0.2 –
#(4 – 1.5 6 – 0.6 – 0.4 – 0.3 –
#/4 –2.2 –16 – –0.2–
'/4 – – 1.4 – 0.4 – 0.1 –
'74 –0.7 2 – –4.6– –
5'#4 – 1.2 2 – 0.5 – 0.2 – 0.1 –
924 – 0.5 2 – 3.4 – 0.6 – 0.1 –
Global – 1.1 2 – 3.8 – 0.9 – 0.3 –
Blank cells indicate data not reported.
– indicates values that cannot be calculated.
a 6JGTGIKQPCNCPFINQDCNßIWTGUCTGCIITGICVGUQHFCVCTGRQTVGFD[NQYCPFOKFFNGKPEQOGEQWPVTKGUCPFVGTTKVQTKGU&CVCHQTVJGXCTKCDNGUUJQYPKPVJGVCDNGCTGPQV
requested from high-income countries in the WHO data collection form.
;'5 NO
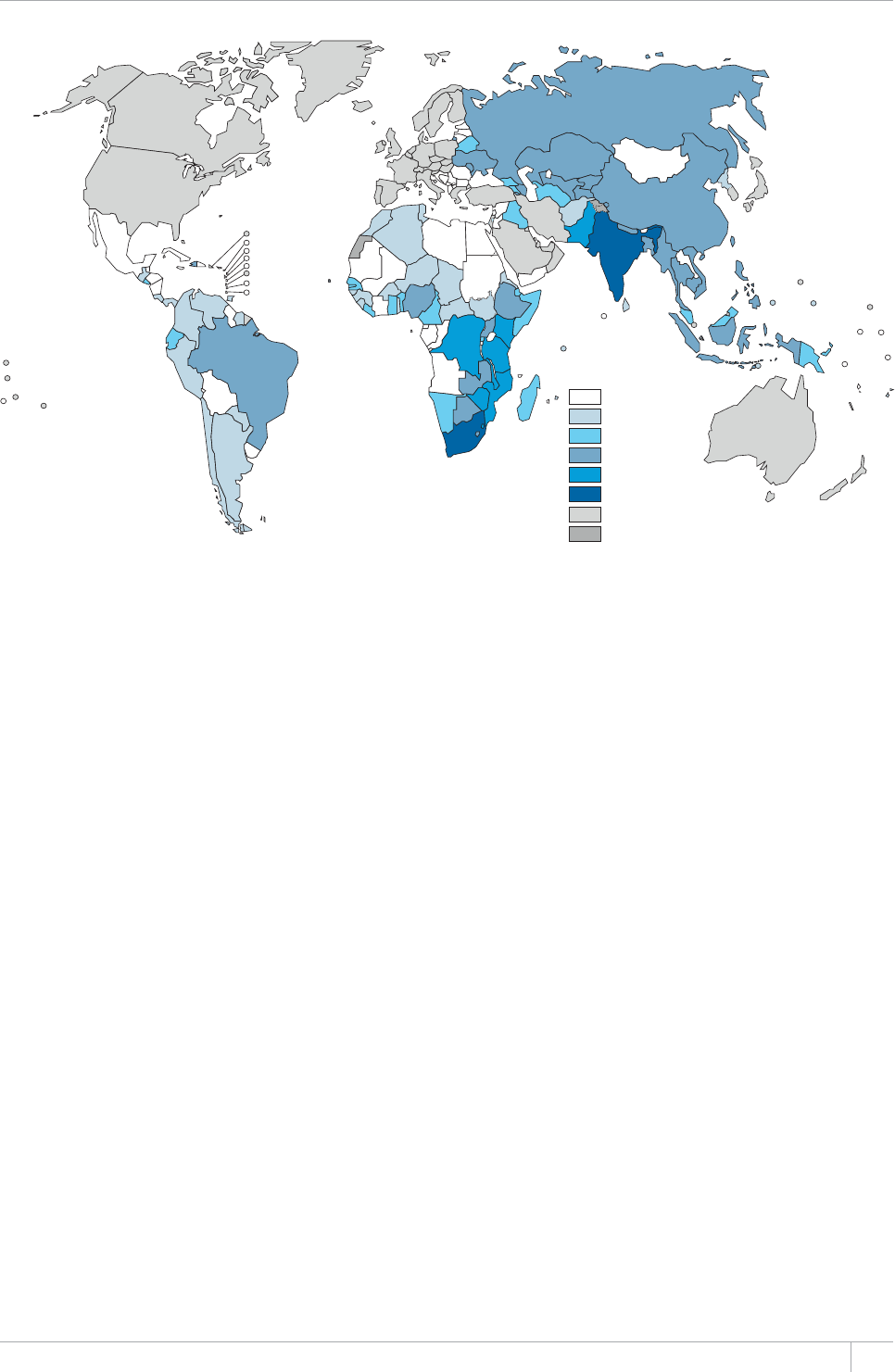
63GLOBAL TUBERCULOSIS REPORT 2013
e introduction of Xpert MTB/RIF reduces the need for
culture as a diagnostic test, yet the growing number of
RR-TB cases will require culture for monitoring of treat-
ment and DST of other anti-TB drugs to guide the design of
treatment regimens. e increasing capacity of countries to
diagnose RR-TB must also be matched by increased capaci-
ty to provide appropriate treatment to the diagnosed cases
(see also Chapter 4).
One of the main reasons for low case detection rates in
many parts of the world (Chapter 3) is the existence of a
signicant private sector, in which care providers frequent-
ly diagnose people with TB but fail to notify these cases to
national authorities. e quality of diagnostic services in
the private sector is highly variable, and some private prac-
titioners continue to use tests that are not recommended by
WHO, including antibody-based serodiagnostics and inter-
feron-gamma release assays (IGRAs) for detection of active
TB. Furthermore in some settings, laboratories in the pub-
lic sector that are not under the auspices of the NTP also
diagnose TB without necessarily following recommended
guidelines and quality assurance procedures. Collaboration
between NTPs and all laboratories oering TB diagnosis is
therefore critical to ensure that national guidelines are fol-
lowed, that appropriate diagnostic tests are used, and that
patients diagnosed with TB are notied to the NTP and
receive proper care. In 2012, 20 of 36 high TB and MDR-
TB burden countries reported some level of collaboration
with laboratories in the private sector, and 25 reported
collaboration with non-NTP laboratories in the public sec-
tor. Additionally, the availability of WHO-recommended
diagnostic tests at concessional prices from manufacturers
under specied conditions has been used as leverage by new
initiatives to form innovative PPM partnerships, increas-
ing access to WHO-recommended diagnostics for people
seeking care in the private sector. Examples are provided
in Box 5.1.
5.3 Strengthening TB laboratories globally,
regionally and nationally
Advances in TB diagnostics in recent years provide an
opportunity to improve laboratory capacity to rapidly and
accurately detect TB and drug resistance. One of the main
prerequisites for eective uptake of new diagnostics is
dynamic policy reform, properly incorporating new tests
and testing methods into diagnostic algorithms. Table
5.2 presents the uptake of selected WHO policy guidance
on TB diagnostics into NTP guidelines at global, region-
al and country levels, focusing on the 36 countries in the
combined list of 22 HBCs and 27 high MDR-TB burden
countries. Overall, high burden countries have been faster
in adopting WHO TB diagnostic guidelines than the glob-
al average. All reporting high MDR-TB burden countries,
95% of HBCs and 84% of reporting countries globally had
reported incorporation of the WHO policy guidance on con-
ventional phenotypic DST into their national guidelines by
2012. ree quarters (74%) of all countries globally had
incorporated guidance on liquid culture and rapid specia-
tion. Countries in the European Region have been particu-
larly fast in adopting these policies, with 97% of countries
reporting having taken up these technologies.
Uptake of WHO policy on use of LPAs for detection of
resistance to rifampicin remains relatively modest, with
FIGURE 5.1
Progress in the roll-out of Xpert MTB/RIF, by July 2013
0
1–1999
2000–9999
10 000–39 999
40 000–99 999
≥ 100 000
Not eligible for preferential pricing
Not applicable
Xpert MTB/RIF
cartridges ordered

64 GLOBAL TUBERCULOSIS REPORT 2013
only 58% of countries globally adopting the policy to date.
Uptake is, however, growing. In the Region of the Ameri-
cas, for example, 61% of countries reported incorporation
of the policy in their national guidelines in 2012 compared
to only 17% in 2011.
Approximately half of low- and middle-income coun-
tries and territories globally (49%) indicated that they had
incorporated WHO guidance on Xpert MTB/RIF into their
diagnostic algorithms for people at risk of HIV-associated
and DR-TB by the end of 2012, highlighting fast uptake
of recommendations rst issued in December 2010. High
MDR-TB burden countries have been particularly quick
to adopt WHO guidance, with 84% of countries reporting
incorporation of the test into their diagnostic algorithms
for people at risk of drug-resistant TB. Funding from sourc-
es including the Global Fund, PEPFAR, USAID, TB REACH
and Médecins Sans Frontières has supported ministries of
health to rapidly establish capacity to use Xpert MTB/RIF.
ese initiatives, together with the TBXpert and EXPAND-
TB projects, will enable further roll out and scale up of the
test in targeted low- and middle-income countries, with
BOX 5.1
Innovative PPM initiatives to increase access to WHO-recommended diagnostics
Some manufacturers of rapid
diagnostics, including Becton, Dickenson
and Company (producer of the BD
MGIT™ 960 automated liquid culture
system), Hain LifeScience (Genotype®
/6$&4RNWUNKPGRTQDGCUUC[CPF
%GRJGKF:RGTV®/6$4+(QHHGTVJGKT
RTQFWEVUVQ062UCPFVJGKTPQVHQTRTQßV
partners in low- and middle-income
countries at concessional prices. Private
HQTRTQßVUGEVQTNCDQTCVQTKGUJCXG
traditionally not been included in such
arrangements, resulting in prices that
are prohibitively high for poor people
seeking care in the private sector and
encouraging use of other diagnostics
that are not recommended by WHO.
4GEGPVN[VYQRWDNKE¿RTKXCVGOKZ22/
initiatives that aim to increase access
to rapid and accurate diagnostics for
vulnerable populations in Asian settings
with vast private sector markets have
been established.
In June 2012, the government of
India took the unprecedented step
of banning the import, manufacture,
distribution and sale of antibody-based
TB serodiagnostic tests, in line with
the WHO recommendation that such
tests should not be used to diagnose
TB. Unfortunately, this ban created a
gap in the private market that allowed
other suboptimal tests to gain market
share, especially since TB diagnostics
recommended by WHO were considered
too expensive and well beyond the
reach of the typical TB patient. To
overcome this market shortcoming, the
Initiative for Promoting Affordable,
Quality TB Tests (IPAQT)a in India was
launched in March 2013. IPAQT is a
consortium of 42 private diagnostic
NCDQTCVQTKGUUWRRQTVGFD[PQVHQTRTQßV
stakeholders (examples include the
Clinton Health Access Initiative and the
McGill International TB Centre). It has
established agreements with Cepheid
Inc, Hain LifeScience, and Becton,
Dickenson and Company that allow
CEEGUUVQEQPEGUUKQPCNRTKEGUHQT:RGTV
/6$4+(ßTUVNKPGNKPGRTQDGCUUC[UCPF
liquid culture in the private sector, which
is normally excluded from negotiated
pricing agreements. Participating
laboratories must abide by several
EQPFKVKQPUVJG[PGGFVQDGCEETGFKVGF
to assure quality; they must report
EQPßTOGFECUGUVQVJG4GXKUGF0CVKQPCN
6$%QPVTQN2TQITCOOG406%2VJG[
must adhere to a ceiling price when
charging patients; and they must
refrain from using any tests that are not
TGEQOOGPFGFD[9*1QTVJG406%2
Together, the laboratories participating
in IPAQT have approximately 3000
franchisee laboratories and over 10 000
specimen collection centres across India,
thus increasing access to rapid, accurate
and affordable diagnostics for patients
seeking care in the country’s extensive
private sector.
As part of the recently launched
70+6#+&HWPFGF6$:RGTVRTQLGEVBox
5.2) and with support from the Stop TB
2CTVPGTUJKR6$4'#%*KPKVKCVKXGHWPFGF
by the Department of Foreign Affairs,
Trade and Development of Canada,
innovative social business models have
been formed in Bangladesh, Indonesia
CPF2CMKUVCPD[+PVGTCEVKXG4GUGCTEJ
and Development in cooperation with
local partners and NTPs. Based in
the megacities of Dhaka, Jakarta and
Karachi and equipped with up to 25
)GPG:RGTVKPUVTWOGPVUGCEJVJGUG
social business models will provide
:RGTV/6$4+(VGUVUTGEGKXGFHTQO
VJG6$:RGTVRTQLGEVHTGGQHEJCTIGVQ
people at high risk of TB who seek care
at private screening centres and other
partnering locations. Free treatment will
be provided to everyone diagnosed with
6$KPEQQRGTCVKQPYKVJ062U4GXGPWG
will be generated from adjunct tests and
services provided to patients, allowing
for sustainability of the businesses
beyond the duration of the three-year
6$:RGTVRTQLGEV
a www.ipaqt.org/
expected increased detection of DR-TB and HIV-associated
TB (Box 5.2).
e WHO/Global Laboratory Initiative (GLI) TB Supra-
national Reference Laboratory (SRL) Network is a driving
force in strengthening national and central level labora-
tories globally, providing long-term technical assistance
to countries under the framework of collaborative agree-
ments. e network comprises 29 laboratories covering all
six WHO regions. e newest addition to the network is the
national TB reference laboratory of Uganda; this lls a criti-
cal geographical gap that had existed in the network in East
Africa. e laboratory has already established collaborative
agreements with Somalia, South Sudan and Zambia for
provision of technical assistance. Additionally, four can-
didate SRLs are under mentorship, including the national
TB reference laboratories of Benin, Denmark and South
Africa, and the Aga Khan University of Pakistan. Pending
completion of successful mentorship and the establishment
of country partners, the new laboratories will help to widen
the geographical reach of the network, in particular in the
African and Eastern Mediterranean Regions.
65GLOBAL TUBERCULOSIS REPORT 2013
TABLE 5.2
Incorporation of WHO guidance for diagnosis of TB into national policy, 2012a
*+)*6$$74&'0
*+)*/&46$
$74&'0
CONVENTIONAL
&47)
575%'26+$+.+6;
TESTING (DST)
.+37+&%7.674'
#0&4#2+&
SPECIATION TEST
.+0'241$'#55#;
(14&'6'%6+0)
4'5+56#0%'61
4+(#/2+%+0
#.)14+6*/(14
THE DIAGNOSIS
OF TB IN PEOPLE
LIVING WITH HIV
:2'46/6$4+((14
DIAGNOSIS OF TB IN
2'45105#64+5-1(
HIV-ASSOCIATED TB
:2'46/6$4+(
(14&+#)015+51(
&47)4'5+56#06
6$+02'45105
#64+5-
Afghanistan
Armenia
#\GTDCKLCP
Bangladesh
Belarus
$TC\KN
Bulgaria
Cambodia
China
&4%QPIQ
Estonia
Ethiopia
Georgia
India
Indonesia
-C\CMJUVCP
Kenya
-[TI[\UVCP
Latvia
Lithuania
/Q\CODKSWG
Myanmar
Nigeria
Pakistan
Philippines
4GRWDNKEQH/QNFQXC
4WUUKCP(GFGTCVKQP
South Africa
Tajikistan
Thailand
Uganda
Ukraine
746CP\CPKC
7\DGMKUVCP
Viet Nam
<KODCDYG
High-burden countries 95% 77% 77% 95% 73% 77%
High MDR-TB burden countries 100% 88% 92% 96% 84% 84%
#(4 67% 54% 74% 60% 62%
#/4 91% 61% 35% 35%
'/4 77% 75% 32% 36%
'74 100% 97% 60% 56%
5'#4 73% 64% 64% 64%
924 61% 56% 39% 33% 33%
Global 84% 74% 58% 78% 49% 49%
Blank cells indicate data not reported.
a 6JGTGIKQPCNCPFINQDCNßIWTGUCTGCIITGICVGUQHFCVCTGRQTVGFD[NQYCPFOKFFNGKPEQOGEQWPVTKGUCPFVGTTKVQTKGU&CVCHQTVJGXCTKCDNGUUJQYPKPVJGVCDNGCTGPQV
requested from high-income countries in the WHO data collection form.
;'5 NO
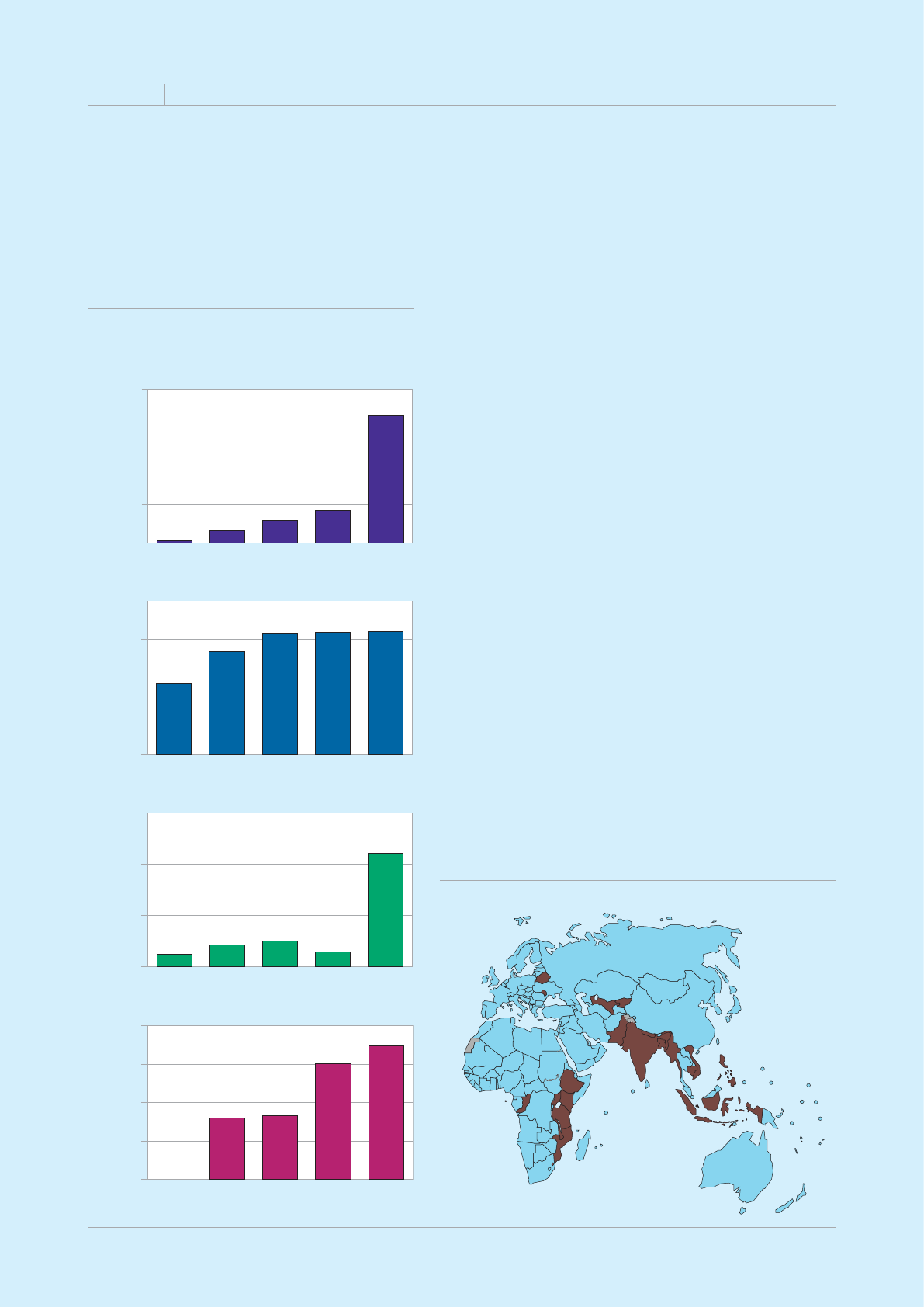
66 GLOBAL TUBERCULOSIS REPORT 2013
BOX 5.2
The EXPAND-TB and TBXpert projects: progress to date
.CWPEJGFKPCPFEQPVKPWKPIWPVKNVJGGPFQHVJG':2#0&6$RTQLGEVCKOUVQCEEGNGTCVGCPFGZRCPFCEEGUUVQFKCIPQUVKEUHQT
RCVKGPVUCVTKUMQH/&46$KPEQWPVTKGU':2#0&6$JCUHWNNQYPGTUJKRD[VJGOKPKUVTKGUQHJGCNVJQHVJGTGEKRKGPVEQWPVTKGUCPFYQTMU
QPCOQFGNQHDGUVRTCEVKEGUNGCTPKPID[FQKPICPFQRVKOK\KPITGUQWTEGUHQTNCDQTCVQT[UVTGPIVJGPKPICVEQWPVT[NGXGN6JGRTQLGEV
is a collaboration between WHO, the Global Laboratory Initiative (GLI), the Foundation for Innovative New Diagnostics (FIND) and the
5VQR6$2CTVPGTUJKR)NQDCN&TWI(CEKNKV[)&(CPFKUHWPFGFD[70+6#+&CPFQVJGTRCTVPGTU':2#0&6$DWKNFUQP75OKNNKQPQH
70+6#+&UWRRQTVVQOCZKOK\GTGUQWTEGUCPFVGEJPKECNCUUKUVCPEGHTQOOWNVKRNGRCTVPGTUHQTNCDQTCVQT[UVTGPIVJGPKPIKPENWFKPIVJG)NQDCN
(WPFVJG9QTNF$CPM2'2(#475#+&VJG#OGTKECP5QEKGV[HQT/KETQDKQNQI[VJG75%GPVGTUHQT&KUGCUG%QPVTQNCPF2TGXGPVKQP,QJPU
Hopkins University, the KfW Development Bank, the KNCV Tuberculosis
Foundation, Partners in Health, Project Hope, PATH, the International
%QOOKVVGGQHVJG4GF%TQUUCPF6JG7PKQP
Overcoming the challenges to establish the necessary infrastructure
for central level laboratories capable of using liquid culture and LPAs, the
':2#0&6$RTQLGEVKUUJQYKPIOCLQTRTQITGUUKPTQWVKPGFGVGEVKQPCPF
TGRQTVKPIQHFTWITGUKUVCPV6$(QTGZCORNG/&46$ECUGUYGTG
diagnosed in supported laboratories in 24 reporting countries in 2012.
6JGEWOWNCVKXGPWODGTQHFKCIPQUGFECUGUQH/&46$TGCEJGFD[
the end of 2012, equivalent to 32% of the overall project target. Several
of the countries participating in the project have reported striking
KPETGCUGUKPVJGPWODGTUQHNCDQTCVQT[EQPßTOGFECUGUQHFTWITGUKUVCPV
TB, especially in 2012 (Figure B5.2.1). The project has recently been
COGPFGFVQCFF:RGTV/6$4+(VQVJGNKUVQHRTQEWTGFFKCIPQUVKEUCNQPI
with liquid culture and LPAs. In October 2012, project partners began
to pilot a strategy for a transition from project-funded to country-
DCUGFßPCPEKPI6JGGZRGTKGPEGHTQOVJGRKNQVYKNNDGWUGFCUCOQFGN
HQTKORNGOGPVCVKQPCETQUUCNN':2#0&6$TGEKRKGPVEQWPVTKGUHQTVJG
remainder of the project, ensuring a smooth transition and sustainability
QHCEJKGXGOGPVUYJGP':2#0&6$GPFU
2TQEWTGOGPVCPFKPUVCNNCVKQPQH)GPG:RGTVKPUVTWOGPVUUVCTVGFKP
OKFHQTVJGPGY6$:RGTVRTQLGEVYJKEJYKNNRTQXKFGCRRTQZKOCVGN[
OKNNKQP:RGTV/6$4+(VGUVECTVTKFIGUCPF)GPG:RGTVOCEJKPGU
to 21 recipient low- and middle-income countries over three years. The
75OKNNKQPRTQLGEVKUHWPFGFD[70+6#+&CPFOCPCIGFD[VJG9*1
Global TB Programme and the Stop TB Partnership. To ensure country
CDUQTRVKXGECRCEKV[CPFGHHGEVKXGWUGQHVJGVGEJPQNQI[VJG6$:RGTV
project links a broad network of partners and existing initiatives for TB
laboratory strengthening and innovative approaches to expand access to
vulnerable populations in both the public and private sector (Box 5.1),
resulting in increased and rapid case detection of TB, HIV-associated TB
CPF446$6$:RGTVRTQLGEVRCTVPGTUKPENWFGVJG).+6$4'#%*VJG)&(
VJG':2#0&6$RTQLGEV+PVGTCEVKXG4GUGCTEJCPF&GXGNQROGPVCPFVJG
African Society for Laboratory Medicine.
MDR-TB cases detected
0
5000
10 000
15 000
20 000
2008 2009 2010 2011 2012
308 1660 2967 4237
16 588
India
MDR-TB cases detected
Belarus
0
500
1000
1500
2000
923
1342
1576 1594 1604
2008 2009 2010 2011 2012
MDR-TB cases detected
2008 2009 2010 2011 2012
24 43 50 30
221
0
100
200
300
Côte d’Ivoire
MDR-TB cases detected
Tajikistan
0
200
400
600
800
319 333
604
694
2008 2009 2010 2011 2012
FIGURE B5.2.1
Increase in cases of MDR-TB reported by selected
EQWPVTKGURCTVKEKRCVKPIKPVJG':2#0&6$RTQLGEV
2009–2012, compared with 2008 baseline
FIGURE B5.2.2
%QWPVTKGUKPbrownRCTVKEKRCVKPIKPVJG6$:RGTVRTQLGEV

67GLOBAL TUBERCULOSIS REPORT 2013
e SRL Network is expanding its membership to
include Centres of Excellence (SRL-CE), a new category that
recognizes laboratories that are performing well in large
low- and middle-income countries and that work primar-
ily to build in-country laboratory capacity. Countries with
laboratories currently eligible to apply for designation as
an SRL-CE include Brazil, China, India, the Russian Fed-
eration and South Africa. To be eligible for this designa-
tion, laboratories need to be nominated by their NTP to the
WHO country oce, establish a collaborative agreement
with an existing SRL, undergo a laboratory assessment
by WHO, and actively implement a quality management
system towards accreditation.

GLOBAL TUBERCULOSIS REPORT 2013
%*#26'4
Addressing the co-epidemics
of TB and HIV
KEY FACTS AND MESSAGES
■ +POKNNKQPQHOKNNKQPRGQRNGYJQ
developed TB worldwide were HIV-positive. The African
4GIKQPCEEQWPVGFHQTQHVJGGUVKOCVGFPWODGTQH*+8
positive incident TB cases.
■ The number of people dying from HIV-associated TB has
been falling since 2003. However, there were still 320 000
deaths from HIV-associated TB in 2012 and further efforts
are needed to reduce this burden.
■ The prevalence of HIV co-infection among TB patients is
JKIJGUVKPVJG#HTKECP4GIKQP1H6$RCVKGPVUYKVJCP*+8
test result, 43% tested positive in 2012, ranging from 9.6%
KP#PIQNCCPF'VJKQRKCVQKP5YC\KNCPF
■ )NQDCNN[VJGRGTEGPVCIGQHPQVKßGF6$RCVKGPVUYKVJC
documented HIV test result was 46% in 2012, up from 40%
in 2011 and 15 times higher than the 2004 level. In the
#HTKECP4GIKQPQHPQVKßGF6$RCVKGPVUJCFCP*+8VGUV
result in 2012, a further improvement compared with 69%
in 2011. Among the 41 countries with the highest TB/HIV
DWTFGPCEJKGXGF*+8VGUVKPINGXGNUQHÜKPENWFKPI
UGXGP-GP[C/CNCYK/Q\CODKSWG4YCPFC5YC\KNCPF
6QIQCPF<CODKCCDQXG
■ 6JGTGYCUCPGPEQWTCIKPIKPETGCUGKP#46EQXGTCIG
among HIV-positive TB patients between 2011 and 2012,
from 49% worldwide in 2011 to 57% in 2012. Nonetheless,
given the WHO recommendation that all HIV-positive TB
RCVKGPVUCTGGNKIKDNGHQT#46VJGEQXGTCIGQH#46HQT*+8
positive TB patients still needs to be greatly improved.
■ +PQH*+8RQUKVKXG6$RCVKGPVUYGTGRTQXKFGF
YKVJEQVTKOQZC\QNGRTGXGPVKXGVJGTCR[%26CNGXGNUKOKNCT
to recent years.
■ In 2012, 4.1 million people enrolled in HIV care were
reported to have been screened for TB, up from 3.5 million
in 2011. Of the reported 1.6 million people newly enrolled
KP*+8ECTGKPCNOQUVaYGTGRTQXKFGFYKVJ
KUQPKC\KFRTGXGPVKXGVJGTCR[+26%QXGTCIGPGGFUVQDG
increased, since about 50% of those newly enrolled in HIV
care and screened for TB are likely to be eligible for IPT.
People living with HIV who are also infected with TB are
much more likely to develop TB disease than those who are
HIV-negative.1 Starting in the 1980s, the HIV epidemic led
to a major upsurge in TB cases and TB mortality in many
countries, especially in southern and eastern Africa (Chap-
ter 2, Chapter 3).
In 2012, 1.1 million (13%) of the 8.6 million people who
developed TB worldwide were HIV-positive (Chapter 2,
Table 2.1); 75% of these HIV-positive TB cases were in
the African Region. Although the number of people dying
from HIV-associated TB has continued to fall globally and
in most regions including the African Region, there were
still 320 000 deaths from HIV-associated TB in 2012, with
approximately equal numbers among men and women (see
Chapter 2). UNAIDS and the Stop TB Partnership have set
a target of halving TB mortality rates among people who
are HIV-positive by 2015 compared with 2004.2
WHO recommendations on the interventions needed to
prevent, diagnose and treat TB in people living with HIV
have been available since 2004,3,4 and are collectively known
as collaborative TB/HIV activities. ey include establishing
and strengthening coordination mechanisms for delivering
integrated TB and HIV services, testing TB patients for HIV,
providing ART and CPT to TB patients living with HIV, pro-
viding HIV prevention services for TB patients, intensifying
TB case-nding among people living with HIV, oering IPT
to people living with HIV who do not have active TB, and
controlling the spread of TB infection in health care and
congregate settings (the latter three activities are referred
to as the ree ‘Is’ for HIV/TB). Since December 2010, the
rapid molecular test Xpert MTB/RIF has been recommend-
ed as the primary diagnostic test for TB among people living
with HIV who have TB signs and symptoms.
WHO began monitoring the implementation and expan-
sion of collaborative TB/HIV activities in 2004. is chap-
1 e probability of developing TB among people living with HIV
divided by the probability of developing TB among HIV-negative
people is the incidence rate ratio (IRR). e estimated global IRR
(all ages) in 2012 was 29.6 (uncertainty interval 27.1–32.1). Fur-
ther details are provided in Annex 1.
2 Getting to zero: 2011–2015 strategy. Geneva, Joint United Nations
Programme on HIV/AIDS, 2010.
3 Interim policy on collaborative TB/HIV activities. Geneva, World
Health Organization, 2004 (WHO/HTM/TB/2004.330; WHO/
HTM/HIV/2004.1). Available at http://whqlibdoc.who.int/hq/
2004/who_htm_tb_2004.330_eng.pdf
4 WHO policy on collaborative TB/HIV activities: guidelines for national
programmes and other stakeholders. Geneva, World Health Organi-
zation, 2012 (WHO/ HTM/TB/2012.1). Available at http://whqlibdoc.
who.int/publications/2012/9789241503006_eng_Annexes.pdf
69GLOBAL TUBERCULOSIS REPORT 2013
FIGURE 6.1
Number of TB patients with known HIV status,
2004Ų2012
FIGURE 6.2
Percentage of TB patients with known HIV status,
2004Ų2012
HIV status
Negative
Positive
TB patients (thousands)
0
1000
2000
3000
2004 2005 2006 2007 2008 2009 2010 2011 2012
Percentage of TB patients
2004 2005 2006 2007 2008 2009 2010 2011 2012
0
20
40
60
80
African region
Global
Regions outside Africa
ter presents the latest status of progress, using data for
2004 up to 2012.
6.1 HIV testing for TB patients
In 2012, the number of notied TB patients who had a doc-
umented HIV test result reached 2.8 million (Figure 6.1),
equivalent to 46% of notied TB cases (Table 6.1, Figure
6.2). is was an increase from 2.5 million and 40% respec-
tively in 2011, and 15 times the level of 3.1% reported in
2004 (Figure 6.2).
e coverage of HIV testing for TB patients was particu-
larly high in the African Region, where 74% of TB patients
had a documented HIV test result in 2012, up from 69%
in 2011 (Figure 6.2). Impressively, in 29 of 46 African
countries, ≥75% of TB patients had a documented HIV test
result in 2012 (Figure 6.3).
Overall, among the 41 countries identied as priorities
for the global TB/HIV response, (listed in Table 6.1), 53%
of TB patients notied had a documented HIV test. Of these
countries, 15 achieved testing levels of ≥85% including
seven (Kenya, Malawi, Mozambique, Rwanda, Swaziland,
Togo and Zambia) that achieved levels above 90%. In addi-
tion, although national data for China show that 34% of
TB patients were tested for HIV in 2012, coverage was 88%
in the 294 high TB/HIV burden counties in which testing
of all notied TB patients is recommended. Globally, there
were 87 countries in which ≥75% of TB patients had a docu-
mented HIV test result.
FIGURE 6.3
Percentage of patients with known HIV status by country, 2012a
Percentage of
notified TB patients
0–14
15–49
50–74
≥75
No data
Not applicable
a +PVJGEQWPVKGUKP%JKPCKFGPVKßGFHQT*+8VGUVKPICOQPIPQVKßGF6$RCVKGPVUQHPQVKßGFECUGUYGTGVGUVGFHQT*+8&CVCHQTVJG4WUUKCP
Federation are for new TB patients only excluding cases in prisons.
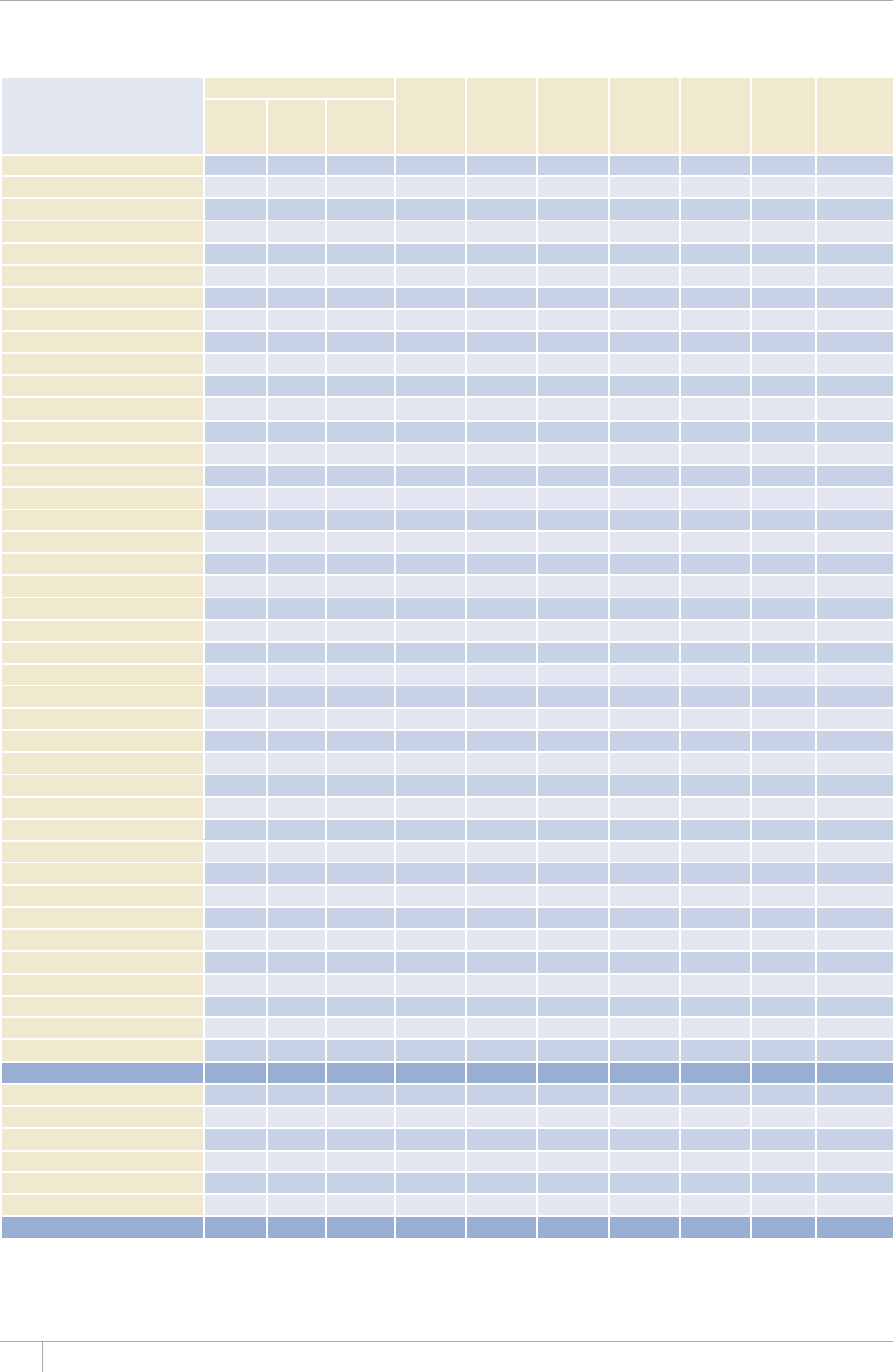
70 GLOBAL TUBERCULOSIS REPORT 2013
TABLE 6.1
HIV testing, treatment for HIV-positive TB patients and prevention of TB among people living with HIV,
globally and for 41 high TB/HIV burden countries and WHO regions, 2012. Numbers in thousands except
where indicated.
ESTIMATED HIV-POSITIVE INCIDENT TB CASES
07/$'41(
TB PATIENTS
WITH KNOWN
HIV STATUS
% OF
NOTIFIED
TB PATIENTS
6'56'&(14
HIV
% OF
TESTED
TB PATIENTS
HIV-POSITIVE
% OF
IDENTIFIED
HIV-POSITIVE
TB PATIENTS
56#46'&10
CPT
% OF
IDENTIFIED
HIV-POSITIVE
TB PATIENTS
56#46'&10
#46
07/$'41(
HIV-
POSITIVE
PEOPLE
5%4''0'&
(146$
07/$'4
OF
HIV-POSITIVE
PEOPLE
2418+&'&
WITH IPT
BEST LOW HIGH
Angola 5.5 4.7 6.5 12 23 9.6 100 100 12 1.1
Botswana 5.1 4.5 5.6 6.0 63 91 66
$TC\KN 16 13 19 46 55 20 0 100
Burkina Faso 1.6 1.3 4.6 15 96 75 7.4
Burundi 2.5 2.2 5.7 19 94 55 0.2
Cambodia 2.7 2.3 3.1 32 4.4 1.1
Cameroon 19 16 23 21 37 55 12
%GPVTCN#HTKECP4GRWDNKE 5.3 4.4 6.4 46 39 20
Chad 4.1 3.4 44 20 65 1.0
China 7.3 6.4 309 34a1.9 59 295
Congo 3.6 2.9 4.3 2.0 17 33 20 23
Côte d’Ivoire 6.9 9.2 21 27 75 44
Djibouti 0.54 0.45 0.64 1.3 36 10 64 0
&4%QPIQ 16 14 19 35 31 16 61 40
Ethiopia 23 17 30 96 65 10 37 272 30
Ghana 2.4 3.1 12 24 72 37
Haiti 4.3 3.5 5.1 14 20 59 46 2.1 15
India 130 120 140 56 5.4 92 59 1 324
Indonesia 7.5 5.6 9.7 2.7 29 23
Kenya 45 44 47 93 94 39 74
Lesotho 9.9 11 10 75 97 53 21 16
Malawi 16 15 17 19 93 59 393 21
Mali 1.2 1.2 1.3 1.5 42 100
/Q\CODKSWG 110 94 55 17
Myanmar 19 16 21 19 13 27
Namibia 7.3 9.9 47 99 72 12 12
Nigeria 46 21 23 56 140 2.3
4WUUKCP(GFGTCVKQP 9.3 7.9 11 76b
4YCPFC 2.9 2.6 3.2 6.1 99 26 99 122
Sierra Leone 3.9 3.2 12 12 26 69 1.1
South Africa 330 270 390 294 65 74 54 950 370
Sudan 4.3 3.5 5.1 3.1 15 7.5 0 17 1.3
5YC\KNCPF 13 11 15 7.4 95 77 66 69 1.9
Thailand 12 10 14 44 72 13 77 62
Togo 1.2 1.4 2.7 91 24 76
Uganda 35 42 41 50 94 49
Ukraine 3.9 5.7 34 14 94 14
746CP\CPKC 32 30 34 52 39 96 54 357
Viet Nam 9.3 6.9 12 66 7.0 73 47 5.7
<CODKC 35 32 39 45 100 54 93 60
<KODCDYG 55 42 69 34 70 26
High TB/HIV burden countries 1 000 960 1 100 2 454 53 21 80 57 4 024 509
#(4 760 910 1 040 74 43 79 55 2 392 473
#/4 31 34 129 56 16 61 76 4.5 19
'/4 11 10 12 14 3.5 69 15 0.2
'74 19 17 21 204 60 6.3 67 74 24
5'#4 170 160 904 39 6.2 61 1 352
924 24 21 27 451 34 3.1 79 56
Global 1 100 1 000 1 200 2 787 46 20 80 57 4 095 519
Blank cells indicate data not reported.
a +PVJGEQWPVKGUKP%JKPCKFGPVKßGFHQT*+8VGUVKPICOQPIPQVKßGF6$RCVKGPVUQHPQVKßGFECUGUYGTGVGUVGFHQT*+8#OQPIVJGUGYGTG
HIV-positive (1.6%).
b &CVCHQTVJG4WUUKCP(GFGTCVKQPGZENWFGTGVTGCVOGPVECUGUCPFECUGUHTQORTKUQPU

71GLOBAL TUBERCULOSIS REPORT 2013
Outside the African Region, in 2012 the percentage of
TB patients who had a documented HIV test result reached
60% in the European Region. It should be noted, how ever,
that the coverage of testing in the Russian Federation is
underestimated since the national data on HIV testing
reported to WHO are for new TB cases in the civilian sector
only (i.e. excluding prisons) while the denominator used
in calculations of coverage is all notied TB cases. e per-
centage of TB patients with a documented HIV test result
in the Region of the Americas was 56% in 2012. Brazil
(where 55% of new TB cases had a documented HIV test
result, very similar to the regional average) accounted for
more than a third of all cases tested in the region, followed
by Mexico (12%) and Haiti (10%). In other regions, where
testing rates have remained consistently low, the percent-
age ranged from 14% in the Eastern Mediterranean Region
to 39% in the South-East Asia Region.
e highest rates of HIV co-infection were reported for
TB patients in the African Region (Table 6.1), where 43%
of those with an HIV test result were positive (compared
with 46% in 2011). e percentage of TB patients found
to be HIV-positive in the 28 African countries in the list
of 41 priority countries ranged from 10% in Ethiopia and
Angola to 77% in Swaziland. In the Region of the Americas,
the percentage of TB patients with a documented HIV test
result who were HIV-positive was 16%. In the Eastern Med-
iterranean, European, South-East Asia and Western Pacic
Regions, less than 10% of TB patients with a documented
HIV test result were HIV-positive. e global average across
all regions was 20%, and 21% among the 41 high TB/HIV
burden countries.
6.2 Antiretroviral therapy and co-trimoxazole
preventive therapy for TB patients living
with HIV
ART is a critical intervention for reducing the risk of TB
morbidity and mortality among people living with HIV. It
reduces the individual risk of TB disease by 65%, irrespec-
tive of CD4 cell count,1 and when combined with IPT it can
have a signicant impact on TB prevention.2 In the latest
WHO guidelines released in July 2013,3 the threshold CD4
count at which starting ART is recommended has been
raised from a CD4 count of ≤350 to ≤500 CD4/mm3. Imple-
mentation of these guidelines on a large scale should sub-
stantially reduce morbidity and mortality resulting from
HIV-associated TB. As in previous guidelines, ART is rec-
ommended for all TB patients living with HIV, irrespective
of their CD4 cell count. CPT also helps to reduce mortality
among HIV-positive TB patients.4
e number of HIV-positive TB patients on ART has
grown from a very low level in 2004 (Figure 6.4) to reach
0.3 million in 2012. Among TB patients notied in 20125
and who had a documented HIV-positive test result, 57%
were on ART globally (Table 6.1, Figure 6.5); this is a
considerable improvement from 49% in 2011. In the Afri-
can Region, 55% of TB patients notied in 2012 who had
a documented HIV-positive test result were on ART (up
from 48% in 2011). Among the 41 high TB/HIV burden
countries, 28 reported enrolling more than 50% of notied
TB patients known to be living with HIV on ART in 2012
(Table 6.1, Figure 6.6). is important progress notwith-
standing, the WHO recommendation that all HIV-positive
TB patients are eligible for ART irrespective of their CD4
cell count also means that the coverage of ART for HIV-pos-
itive TB patients still needs to be greatly improved with the
goal of reaching the 2015 target of 100% set in the Global
Plan to Stop TB 2011–2015.
Early initiation of ART, as soon as possible within eight
weeks after initiation of TB treatment or within two weeks
for profoundly immunosuppressed patients (CD4 count
<50), is recommended. WHO also strongly recommends
the integration of ART and TB treatment services for TB
patients living with HIV either through TB or HIV treat-
ment facilities in settings with a high burden of TB and HIV.
In many settings, facilities providing TB services are more
decentralized than ART services and oer an opportunity
to scale up the delivery of integrated TB and HIV services
through task shifting and task sharing.6 A recent example
of the integration of TB services with those for HIV and
1 Suthar AB et al. Antiretroviral therapy for prevention of tuberculosis
in adults with HIV: a systematic review and meta-analysis. PLoS Med-
icine, 2012, 9(7): e1001270. (doi:10.1371/journal.pmed.1001270).
2 Samandari T et al. 6-month versus 36-month isoniazid preven-
tive treatment for tuberculosis in adults with HIV infection in
Botswana: a randomised, double-blind, placebo-controlled trial.
e Lancet. 2011 May 7;377(9777):1588-98. doi: 10.1016/S0140-
6736(11)60204-3.
3 Consolidated guidelines on the use of antiretroviral drugs for treat-
ing and preventing HIV infection. Geneva, World Health Organi-
zation, 2013. Available at http://apps.who.int/iris/bitstream/
10665/85321/1/9789241505727_eng.pdf
4 Nunn AJ et al. Role of co-trimoxazole prophylaxis in reducing mor-
tality in HIV infected adults being treated for tuberculosis: ran-
domized clinical trial. British Medical Journal. 2008, 337:a257.
5 In the annual WHO TB data collection form, countries are asked to
report the number of TB patients notied in the most recent calen-
dar year who were living with HIV and who “started or continued
on ART”.
6 Global Tuberculosis Report 2012. Geneva, World Health Organiza-
tion, 2012.
FIGURE 6.4
Number of HIV-positive TB patients enrolled
on co-trimoxazole preventive therapy (CPT) and
antiretroviral therapy (ART), 2004–2012
TB patients (thousands)
HIV-positive
CPT
ART
0
100
200
300
400
500
600
2004 2005 2006 2007 2008 2009 2010 2011 2012
72 GLOBAL TUBERCULOSIS REPORT 2013
FIGURE 6.5
Percentage of TB patients with known HIV status who were HIV positive, and percentage of HIV-positive TB
patients enrolled on co-trimoxazole preventive therapy (CPT) and antiretroviral therapy (ART), 2007–2012a
a The solid lines show values for countries that reported data. The shaded areas show upper and lower limits when countries that did not report data are considered.
Percentage of TB patients
2007 2008 2009 2010 2011 2012 2007 2008 2009 2010 2011 2012 2007 2008 2009 2010 2011 2012
0
20
40
60
80
100
0
20
40
60
80
100
0
20
40
60
80
100
% of TB patients with known
HIV status who were HIV-positive
% of HIV-positive patients on CPT % of HIV-positive patients on ART
FIGURE 6.6
Percentage of HIV-positive TB patients enrolled on antiretroviral therapy (ART), 2012
Percentage of
HIV-positive
TB patients
0–24
25–49
50–74
75–100
No data
Not applicable
maternal, new-born and child health (MNCH) services is
provided in Box 6.1.
Globally, 0.4 million TB patients living with HIV were
enrolled on CPT in 2012, up from a negligible number in
2004. e absolute number fell between 2011 and 2012,
which is at least partly explained by the decrease in the
number of HIV-positive TB cases reported between 2011
and 2012 (Figure 6.4). e coverage of CPT among TB
patients with a documented HIV-positive test result was
80% in 2012, similar to the level of 2010 and 2011 (Table
6.1, Figure 6.5). e African, South-East Asia and Western
Pacic Regions achieved particularly high levels of enrol-
ment on CPT: 79%, 89% and 79%, respectively (Table 6.1).
Of the 41 high TB/HIV burden countries, the percentage of
HIV-positive TB patients enrolled on CPT in 2012 exceeded
90% in Angola, Botswana, Burkina Faso, Burundi, Cambo-
dia, India, Kenya, Lesotho, Mozambique, Namibia, Rwan-
da, Swaziland, Uganda, the United Republic of Tanzania
and Zambia.
6.3 Intensifying TB screening and
isoniazid preventive therapy among
people living with HIV
Recording and reporting of TB screening among people liv-
ing with HIV and provision of IPT to those without active
TB is a particular challenge in many countries, and further
eorts are needed to facilitate and improve the tracking of
progress nationally and globally (Box 6.2).

73GLOBAL TUBERCULOSIS REPORT 2013
BOX 6.1
Linkages between TB, HIV and maternal, newborn and child health (MNCH) services in Cambodia
Cambodia has achieved great progress in responding to its HIV epidemic and in reducing TB prevalence and mortality. It has also
made progress in improving services for maternal, newborn and child health (MNCH). Attendance at antenatal services and the
RGTEGPVCIGQHFGNKXGTKGUCVJGCNVJHCEKNKVKGUJCXGKPETGCUGFCPFOCVGTPCNCPFWPFGTßXGOQTVCNKV[JCXGDQVJDGGPTGFWEGF/CLQT
efforts to establish and strengthen service linkages between the TB, HIV and MNCH programmes have also been made.
Linkages between TB and
HIV services
+PQHPQVKßGF6$RCVKGPVU
MPGYVJGKT*+8UVCVWUCPFQH
*+8RQUKVKXG6$RCVKGPVUYGTGQP#46
The number of people living with HIV
given IPT increased by a factor of 22
between 2006 and 2012, following
the introduction of the WHO screening
algorithm to rule out active TB (and
associated removal of the previous
requirement for a positive tuberculin
skin test before initiation of IPT).
Linkages between MNCH and
HIV services
The percentage of pregnant women
tested for HIV increased from 16% in
VQKP6JGEQXGTCIG
QH#46COQPI*+8KPHGEVGFRTGIPCPV
women increased from 11% in 2007 to
65% in 2012. The percentage of infants
born to HIV-positive women who were
RTQXKFGFYKVJ#46VQRTGXGPVOQVJGT
to-child transmission rose from 50% in
2010 to 73% in 2012.
Linkages between MNCH and
TB services
A new MNCH-TB collaborative framework
offering cross-programme referrals
between TB services and clinics
providing antenatal, growth monitoring
CPFKOOWPK\CVKQPUGTXKEGURTQOKUGUVQ
further reduce the burden of TB among
women and children.
Scaling up collaboration among
the three programmes
Collaboration among the three
programmes aims to strengthen
linkages and synergies to achieve better
outcomes. The government, with support
from WHO, has piloted efforts to set
up a collaborative project involving
the three programmes in two districts.
Lessons learned from these and other
pilot sites are helping the country to
OCZKOK\GRQVGPVKCNHQTETQUURTQITCOOG
EQNNCDQTCVKQPCPFVQQRVKOK\GVJGWUGQH
resources.
The three-programme collaborative
CEVKXKVKGUDGKPIRKNQVGFKPENWFG
*CTOQPK\KPIVTCPURQTVCVKQPQHDNQQF
samples required for testing HIV
that are collected at sites providing
services for pregnant women, TB
patients and populations at high risk
of HIV.
*CTOQPK\KPIKPHQTOCVKQPGFWECVKQP
and communication related to MNCH,
HIV and TB at the sites providing
MNCH, HIV and TB services.
3. Expanding cross-programme
laboratory services.
4. Strengthening the system
for referrals between the
three programmes including
UVCPFCTFK\CVKQPQHEQOOWPKECVKQP
and referral procedures.
*CTOQPK\CVKQPQHEQOOWPKV[U[UVGO
strengthening by sharing the costs
and time spent during monthly
meetings of village health support
groups at health centres.
BOX 6.2
Improving the quality of TB/HIV data: challenges and solutions
Major efforts have been made in recent years to improve the quality of TB/HIV data. Indicators used by TB and HIV programmes have
DGGPUVCPFCTFK\GFCPFEQNNCDQTCVKQPDGVYGGP6$CPF*+8RTQITCOOGUJCUDGGPKORTQXGFYKVJENGCTFGßPKVKQPQHTGURQPUKDKNKVKGU
for data collection related to TB/HIV interventions. WHO and UNAIDS have worked intensively with countries to try to ensure
complete and consistent reporting and to reconcile any apparent discrepancies between data reported by TB and HIV programmes.
6JGUGGHHQTVUPQVYKVJUVCPFKPIEJCNNGPIGUTGOCKP
Missing or inaccurate denominators required to calculate the coverage of TB screening and IPT among people
living with HIV. There has been an increase in the number of countries capturing and reporting data on the number of people
living with HIV who are screened for TB and the number without active TB who are provided with IPT. However, many of these
countries are not reporting the corresponding denominators needed to calculate coverage (i.e. people registered in HIV care
CPFRGQRNGPGYN[TGIKUVGTGFKP*+8ECTGHQTUETGGPKPICPF+26TGURGEVKXGN[6JGTGCTGCNUQGZCORNGUQHVJGUCOGßIWTGUDGKPI
reported for both denominators.
Discrepant reporting by NTPs and National AIDS Programmes (NAPs). In some countries, the NTP and NAP report
FKHHGTGPVßIWTGUHQTVJGPWODGTQH*+8RQUKVKXG6$RCVKGPVUYJQCTGQP#46+PEQWPVTKGUVJGPWODGTUTGRQTVGFD[VJG
062CPF0#2YGTGFKHHGTGPVKPDQVJCPF#NVJQWIJUWDUGSWGPVFCVCXGTKßECVKQPCPFJCTOQPK\CVKQPGHHQTVUNGFVQ
consensus on one number in most countries, the different numbers could not be reconciled for either year in Angola, Myanmar
CPFVJG7PKVGF4GRWDNKEQH6CP\CPKC5QNWVKQPUVQCFFTGUUVJKURTQDNGOKPENWFGKORTQXKPIU[UVGOUHQTTGEQTFKPICPFTGRQTVKPI
data and further strengthening of collaboration and communication between the NAP and NTP as well as their partners.
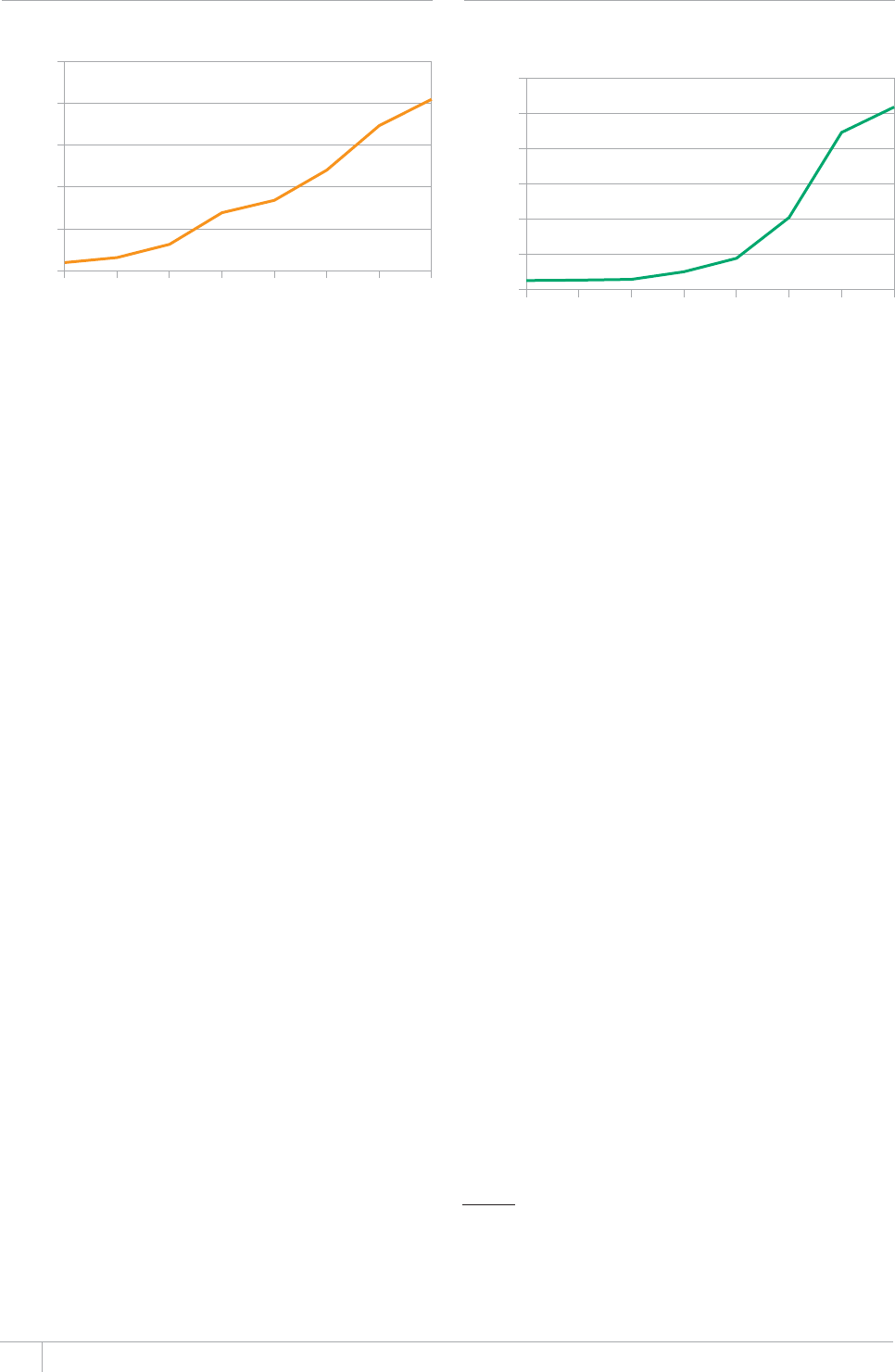
74 GLOBAL TUBERCULOSIS REPORT 2013
In 2012, a total of 4.1 million people who were enrolled in
HIV care were screened for TB in 61 countries, an increase
from 3.5 million in 58 countries in 2011 (Figure 6.7). In
the 49 countries that reported both the number screened
for TB and the number in HIV care, the coverage of screen-
ing was 66% (3.9/5.9 million).
Among 42 countries that reported data, IPT was ini-
tiated among almost 520 000 people newly registered
in HIV care in 2012. is was an increase from less than
450000 people in 2011 (Figure 6.8). One country – South
Africa – accounted for 71% of the global total with 370000
people reported to have been provided with IPT in 2012,
followed by Ethiopia (30 000), Malawi (21000), Mozam-
1 Getahun H, et al. Development of a standardized screening rule for
tuberculosis in people living with HIV in resource-constrained set-
tings: Individual participant data meta-analysis of observational
studies. PLoS Medicine, 2011, 8(1): e1000391. doi:10.1371/journal.
pmed.1000391.
FIGURE 6.7
Intensißed TB case-ßnding among people living
with HIV, 2005–2012
Number of people screened (millions)
2005 2006 2007 2008 2009 2010 2011 2012
0
1
2
3
4
5
FIGURE 6.8
Provision of isoniazid preventive therapy (IPT)
to people living with HIV without active TB,
2005–2012
Number of HIV-positive people without
active TB (thousands)
0
100
200
300
400
500
600
2005 2006 2007 2008 2009 2010 2011 2012
bique (17000), Lesotho (16000), Haiti (15000), Ukraine
(14000), and Namibia (12000).
irty countries reported both the total number of peo-
ple newly enrolled in HIV care (1.6 million) and the num-
ber of people living with HIV who were started on IPT
(0.47 million) in 2012 i.e. 30% of those newly enrolled in
HIV care were initiated on IPT. If the WHO-recommend-
ed four-symptom screening algorithm is used to rule out
active TB in people living with HIV, approximately 50% of
people living with HIV would be expected to be eligible for
IPT.1 Further eorts are needed to reach the Global Plan’s
2015 target of providing IPT to all those eligible.
75GLOBAL TUBERCULOSIS REPORT 2013
%*#26'4
Financing
KEY FACTS AND MESSAGES
■ Funding required for a full response to the global TB
epidemic in low- and middle-income countries is estimated
CV75aDKNNKQPRGT[GCTD[GZENWFKPITGUGCTEJCPF
development for new TB diagnostics, drugs and vaccines).
Of this total, about two thirds is needed for the detection
and treatment of drug-susceptible TB, 20% for treatment
QH/&46$HQTTCRKFFKCIPQUVKEVGUVUCPFCUUQEKCVGF
laboratory strengthening, and 5% for collaborative TB/HIV
activities.
■ A recent long-term study using data reported to WHO
shows that TB funding in low- and middle-income countries
grew substantially between 2002 and 2011, especially in
$TC\KNVJG4WUUKCP(GFGTCVKQP+PFKC%JKPCCPF5QWVJ#HTKEC
$4+%56JGKPETGCUKPIUGNHUWHßEKGPE[QHVJGUGCPFUQOG
other countries is a success story for these countries and the
global TB community.
■ Despite growth in funding for TB, funding gaps persist
CPFCFFKVKQPCNHWPFKPIPGGFUVQDGOQDKNK\GFHTQODQVJ
domestic and international donor sources. There is capacity
to increase funding from domestic sources beyond the
75aDKNNKQPCXCKNCDNGKPGURGEKCNN[KP$4+%5
Funding required from international donor sources is
GUVKOCVGFCV75a¿DKNNKQPRGT[GCT
■ Funding from international donor sources is expected
VQTGCEJ75aDKNNKQPKPOQUVQHVJKUHWPFKPIKU
from the Global Fund and USAID. Donor funding accounts
HQTCNCTIGUJCTGÜQHVQVCNHWPFKPIKPUQOGEQWPVT[
ITQWRUPQVCDN[VJG*$%UGZENWFKPI$4+%5CPFCNNNQY
income countries, and an even higher proportion in some
individual countries. International donors have a crucial
role in sustaining and ensuring further progress in TB
prevention, diagnosis and treatment worldwide.
■ 6JGEQUVRGTRGTUQPUWEEGUUHWNN[VTGCVGFHQT6$YKVJßTUV
NKPGFTWIUKUKPVJGTCPIG75aVQ75aKPCNOQUVCNN
countries with a high burden of TB.
Progress in TB prevention, diagnosis and treatment
requires adequate funding sustained over many years.
WHO began annual monitoring of funding for TB in 2002,
and ndings have subsequently been published in global
TB reports. Particular attention has always been given to
the 22 HBCs that account for about 80% of estimated cases
(Chapter 2). Recent reports have included aggregated anal-
yses of trends since 2006 for approximately 100 countries.
In 2012, WHO conducted a comprehensive analysis of
long-term trends in TB funding in low- and middle-income
countries for the decade 2002–2011, using data reported
by countries between 2002 and 2012. e analysis was able
to include 104 out of a total of 154 countries classied by
the World Bank as low- or middle-income in 2011 (gross
national income (GNI) per capita < US$12,476). ese 104
countries had 94% of the world’s estimated cases of TB and
88% of the world’s estimated cases of MDR-TB in 2011. Lev-
els of funding in 2011 were then analysed in combination
with the most recent estimates of resource requirements
for TB prevention, diagnosis and treatment to assess the
funding that could be mobilized from domestic sources and
the balance required from international donors up to 2015.
Results from these analyses were published in an article in
the August 2013 issue of e Lancet Global Health.1
Given this very recent publication, the scope of this
nancing chapter has been adjusted compared with pre-
vious years to avoid unnecessary duplication. Section 7.1
presents the most up-to-date estimates of nancial resourc-
es required until the end of 2015 in all of the 154 countries
that were classied as low- or middle-income countries in
2011, alongside projections of the funding that could be
mobilized domestically. Section 7.2 provides a summary
of the main ndings from the analysis of trends in fund-
ing between 2002 and 2011 in 104 low- and middle-income
countries. With this background and context, the rest of
the chapter (section 7.3) contains detailed analyses of TB
funding in 2013, using data compiled in the 2013 round of
global TB data collection. Funding levels in 2013 are pre-
sented by WHO region and for other country groupings
based on income level, burden and geography, with break-
downs by source of funding (section 7.3.1) and category
of expenditure (section 7.3.2). Funding gaps reported by
countries are also illustrated and discussed (section 7.3.3).
1 Floyd K, Fitzpatrick C, Pantoja A and Raviglione M. Domestic and
donor nancing for tuberculosis care and control in low-income
and middle-income countries: an analysis of trends, 2002–11, and
requirements to meet 2015 targets. e Lancet Global Health; 1:
e105–15.

76 GLOBAL TUBERCULOSIS REPORT 2013
Further country-specic data can be found in nance pro-
les that are available online.1
7.1 Estimates of funding required up to
2015 for a full response to the global
TB epidemic
e Global Plan to Stop TB 2011–20152 sets out the actions
and funding needed for a full response to the TB epidem-
ic, based on the Stop TB Strategy.3 e overall goal of the
plan is to achieve the 2015 global targets for reductions in
cases of and deaths from TB (i.e. that incidence should be
falling and that prevalence and mortality rates should be
halved compared with their levels in 1990) (Chapter 1). Key
components of the plan include increasing the number of
patients detected and treated according to WHO’s recom-
mended strategy from 5.8 million in 2011 to 6.9 million
by 2015 (which would be equivalent to more than 80% of
projected incident cases in that year); ensuring that all pre-
viously treated patients and all new patients with known
risk factors for MDR-TB are tested for drug resistance by
2015 (including with recently endorsed rapid tests such as
Xpert MTB/RIF that are discussed in Chapter 5); enrol-
ment of all TB patients with conrmed MDR-TB (projected
to be around 300000 in 2015) on second-line treatment;
HIV testing of all patients with TB; and prompt initiation
of ART in all HIV-positive TB patients.
In 2013, the Global Plan datasets were used in combi-
nation with new country-specic planning and budgeting
work with nine high TB or high MDR-TB burden countries
to produce updated estimates of funding needs for TB
prevention, diagnosis and treatment in low- and middle-
income countries.4 e nine countries were Ethiopia, India,
Indonesia, Kazakhstan, Kenya, Nigeria, Pakistan, South
Africa and Ukraine. Analyses were conducted in the context
of estimates of funding needs and funding gaps required
for the Global Fund’s replenishment eorts in 2013.5 WHO
subsequently extended these analyses to cover all low- and
middle-income countries and not only the countries eligi-
ble to apply to the fund.6 Notable countries (in terms of TB
burden and funding requirements) that are not eligible to
apply to the Global Fund are Brazil, China and the Russian
Federation.
During the course of the work done for the rst pre-
replenishment meeting held in April 2013, it should be
highlighted that the Global Fund, WHO, UNAIDS, and
other partners agreed that funding needs for ART for HIV-
positive TB patients should be included in estimates of HIV
resource needs to avoid double-counting. For this reason,
the estimates of resource requirements for TB/HIV inter-
ventions included in the updated estimates of resource
needs for TB are lower than those published in the Global
Plan.
Funding needs were compared with the domestic fund-
ing that could be mobilized in two alternative scenarios.
e rst scenario was that TB funding could increase (from
a 2011 baseline) in line with International Monetary Fund
forecasts for growth in total government expenditures.7
e second scenario had the same assumptions as the rst,
but also assumed that countries that currently under-
perform in domestic nancing relative to their income level
(i.e. their ability to pay) and disease burden reach the lev-
el of the median performer by 2020. ese scenarios were
chosen to be fully consistent with the methods previously
used to assess the potential to mobilize domestic funding
for prevention, treatment and care of HIV.8
e main results from these analyses are summarized
in Figure 7.1. e total funding required in all low- and
middle-income countries reaches about US$ 8 billion in
2015, compared with US$6 billion in 2012 (Figure 7.2).9
Of the total funding required, about two thirds is needed
for the detection and treatment of drug-susceptible TB,
20% for treatment of MDR-TB, 10% for rapid diagnostic
tests and associated laboratory strengthening, and 5% for
collaborative TB/HIV activities (excluding ART). Funding
needed for each of these four categories increases over time.
e largest relative increases are for treatment of MDR-TB
and diagnostics/laboratory strengthening.
ere is potential to mobilize a large share of these fund-
ing needs from domestic resources in some country groups,
notably BRICS and upper middle-income countries (Figure
7.1). Elsewhere, there are relatively large gaps between
the estimated amounts of domestic funding that could be
mobilized and the total funding needed, especially in three
country groups: the 17 HBCs excluding BRICS; low-income
countries; and the African Region excluding South Africa.
In the rst scenario in which domestic funding grows from
2011 levels in line with projected growth in total govern-
ment expenditures, the total gap amounts to US$2.3 bil-
lion per year by 2015. In the second and more optimistic
scenario, the gap would be US$1.6 billion per year by 2015.
1 www.who.int/tb/data
2 e Global Plan to Stop TB, 2011–2015. Geneva, World Health Orga-
nization, 2010 (WHO/HTM/STB/2010.2).
3 Raviglione M, Uplekar M. WHO’s new Stop TB strategy. Lancet
2006; 367: 952–5.
4 Funding required for research and development for new TB diag-
nostics, drugs and vaccines was not considered. In the Global
Plan, it is estimated that about US$2 billion per year is needed for
research and development.
5 e Global Fund to Fight AIDS, Tuberculosis and Malaria fourth replen-
ishment (2014–2016): needs assessment. Geneva, Global Fund to
Fight AIDS, Tuberculosis and Malaria, 2013.
6 Floyd K, Fitzpatrick C, Pantoja A and Raviglione M. Domestic and
donor nancing for tuberculosis care and control in low-income
and middle-income countries: an analysis of trends, 2002–11, and
requirements to meet 2015 targets. e Lancet Global Health; 1:
e105–15.
7 World economic outlook database. Washington, International Mon-
etary Fund, 2012 (www.imf.org/external/pubs/ft/weo/2012/02/
weodata/index.aspx).
8 Schwartlander B, Stover J, Hallett T, et al. Towards an improved
investment approach for an eective response to HIV/AIDS. e
Lancet 2011; 377: 2031–41.
9 In Figure 7.1, country groups are not all mutually exclusive. e
global total can be calculated by adding together the totals in the
panels for BRICS, low-income countries, lower middle-income
countries (excluding China and India) and upper middle-income
countries (excluding Brazil, the Russian Federation and South
Africa).

77GLOBAL TUBERCULOSIS REPORT 2013
FIGURE 7.1
Forecast of funding that could be mobilized from domestic sources compared with total funding needed for
a full response to the global TB epidemic in nine country groups, 2012–2015. The black line shows the total
funding required. The blueDCPFTGRTGUGPVUUEGPCTKQYJKEJUJQYUFQOGUVKEHWPFKPIVJCVEQWNFDGOQDKNK\GFKHFQOGUVKE
funding increases from a 2011 baseline at the same rate of growth as International Monetary Fund forecasts of growth in total
government expenditures. The greenDCPFUJQYUCFFKVKQPCNTGUQWTEGUVJCVEQWNFDGOQDKNK\GFEQORCTGFYKVJUEGPCTKQKH
current underperformers (relative to income level and TB disease burden) improve at a consistent rate to reach the level of the
median performer by 2020. $4+%5$TC\KNVJG4WUUKCP(GFGTCVKQP+PFKC%JKPC5QWVJ#HTKEC
US$ billions
Africa excluding S
US$ billions
BRICS
Low-income countries
17 HBCs excluding BRICS
Lower-middle income countries
excluding I and C
Asia excluding I and C Rest of world excluding B and R
Upper-middle income countries
excluding B, R, S
Low-income countries excluding HBCs
US$ billions
0
1.0
2.0
3.0
4.0
5.0
2012 2013 2014 2015
0
0.5
1.0
1.5
2012 2013 2014 2015
0
0.10
0.20
0.30
2012 2013 2014 2015
0
0.2
0.4
0.6
0.8
1.0
1.2
2012 2013 2014 2015
0.2
0.4
0.6
1.2
1.4
1.6
1.8
0.8
2012 2013 2014 2015
1.0
0
0.2
0.4
0.6
0.8
2012 2013 2014 2015
0
0.2
0.4
0.6
1.0
1.2
1.4
1.6
1.8
0.8
2012 2013 2014 2015
0
0.2
0.4
0.6
0.8
1.0
1.2
2012 2013 2014 20152012 2013 2014 2015
0
0.1
0.2
0.3
0.4
0.5
0.6
0.7
0.8
0.9
0

GLOBAL TUBERCULOSIS REPORT 2013
It should be highlighted that in the second and more
optimistic scenario, it is assumed that countries that cur-
rently underperform in terms of their levels of domestic
nancing relative to their TB burden and income level will
steadily progress to reach the level of the current median
peformer (in terms of domestic funding relative to burden
and income level) by 2020. Two countries in particular –
India and Indonesia – would need to substantially increase
their levels of domestic funding for this scenario to mate-
rialize in practice, since they account for about two-thirds
of the additional funding in Scenario 2 compared with
Scenario 1. Current trends are not in line with Scenario
2. In India, domestic funding reported for 2013 is lower
than the amount available in 2012, while in Indonesia an
increase in domestic funding between 2012 and 2013 was
reported but by a relatively small amount (see Annex 2 for
further details).
7.2 Trends in TB funding, 2002–2011:
a summary
Data reported to WHO between 2002 and 2012 allowed
analysis of trends 2002–2011 in 104 countries. ese 104
countries are shown in Table 7.1 (an additional 21 coun-
tries that could be included in analyses of funding in 2013,
described in section 7.3, are shown in bold). Among the 104
countries, there were ≥6 observations for 83 countries. For
most countries, there were between 7 and 10 observations,
including 14/22 HBCs that had 10 observations each and
5/22 HBCs for which there were 9/10 observations. Values
for country-year combinations for which data were missing
in the 104 countries were imputed using country-specic
linear regression models. Details on the criteria used to
include or exclude countries and the imputation methods
are available in an online technical appendix.1
In the 104 low- and middle-income countries with 94%
of the world’s TB cases and 88% of the world’s MDR-TB cas-
es, total funding for TB (domestic plus international donor
sources) grew in real terms (2011 US$ prices) from US$1.7
billion in 2002 to US$ 4.4 billion in 2011. e increases var-
ied among country groups, from 100% in low-income coun-
tries to 177% in upper middle-income countries. Increases
in funding were accompanied by large increases in the num-
ber of people successfully treated for TB, from 2.8 million
in 2002 to 5.0 million in 2011. A cumulative total of 43 mil-
lion people were treated between 2002 and 2011. e cost
per patient treated was in the range US$100–500 in most
of the countries with the highest burdens of TB. e size
of the patient caseload and gross domestic product (GDP)
per capita explained more than 70% of the variation among
countries in the cost per patient treated.
Domestic funding (national and local budgets, and loans)
in the 104 countries included in trend analyses rose from
US$1.5 billion in 2002 to US$3.9 billion in 2011. Loans
accounted for a small proportion (≤5%) of total domestic
funding each year. Most of the increase in total domestic
funding (US$1.7 out of US$2.4 billion [71%]) was account-
ed for by BRICS (which account for almost half of the world’s
TB cases) and other middle-income countries in Asia, Latin
America and Europe. e magnitude of domestic funding
in these country groups (69–98% of total funding per year)
and BRICS in particular (>95% of total funding per year)
meant that domestic funding dominated total funding for
TB globally (88–92% per year).
International donor funding in the 104 countries includ-
ed in trend analyses grew from US$0.2 billion in 2002 to
US$0.5 billion in 2011. ere was striking variation among
country groups in terms of the share of total funding pro-
vided from international donor sources. By 2011, donor
funding represented 39% of total funding in the 17 HBCs
excluding BRICS, which account for about one third of
the world’s TB cases; 42% of funding in African countries
excluding South Africa; and 67% of total funding in low-
income countries (25 of which are in Africa). e Global
Fund accounted for 64% of all donor funding reported by
countries during the decade 2002–2011.
Most funding was used for the diagnosis and treat-
ment of drug-susceptible TB (over 85% each year). Small
amounts were used for diagnosis and treatment of MDR-
TB, although funding started to increase in BRICS, upper
middle-income countries, and countries in Europe and
Latin America around 2006.
Despite growth in funding from domestic and interna-
tional donor sources, NTPs were not able to mobilize all the
funding that they estimated to be needed. Funding gaps
(i.e. the dierence between assessments by NTPs of funding
needs for TB prevention, diagnosis and treatment and the
actual amount of funds mobilized) persisted, and increased
from US$257 million in 2002 to US$563 million in 2011. It
should be noted that the funding gaps reported by NTPs are
sometimes based on relatively conservative assessments of
funding needs. When national strategic plans with more
FIGURE 7.2
Total funding required for a full response
to the global TB epidemic, by intervention area,
2013–2015
TB/HIV
Laboratory
strengthening/
diagnostics
MDR-TB
Drug-susceptible
TB
US$ billions
0
2
4
6
8
2012 2013 2014 2015
1 Floyd K, Fitzpatrick C, Pantoja A and Raviglione M. Domestic and
donor nancing for tuberculosis care and control in low-income
and middle-income countries: an analysis of trends, 2002–11, and
requirements to meet 2015 targets. e Lancet Global Health; 1:
e105–15.
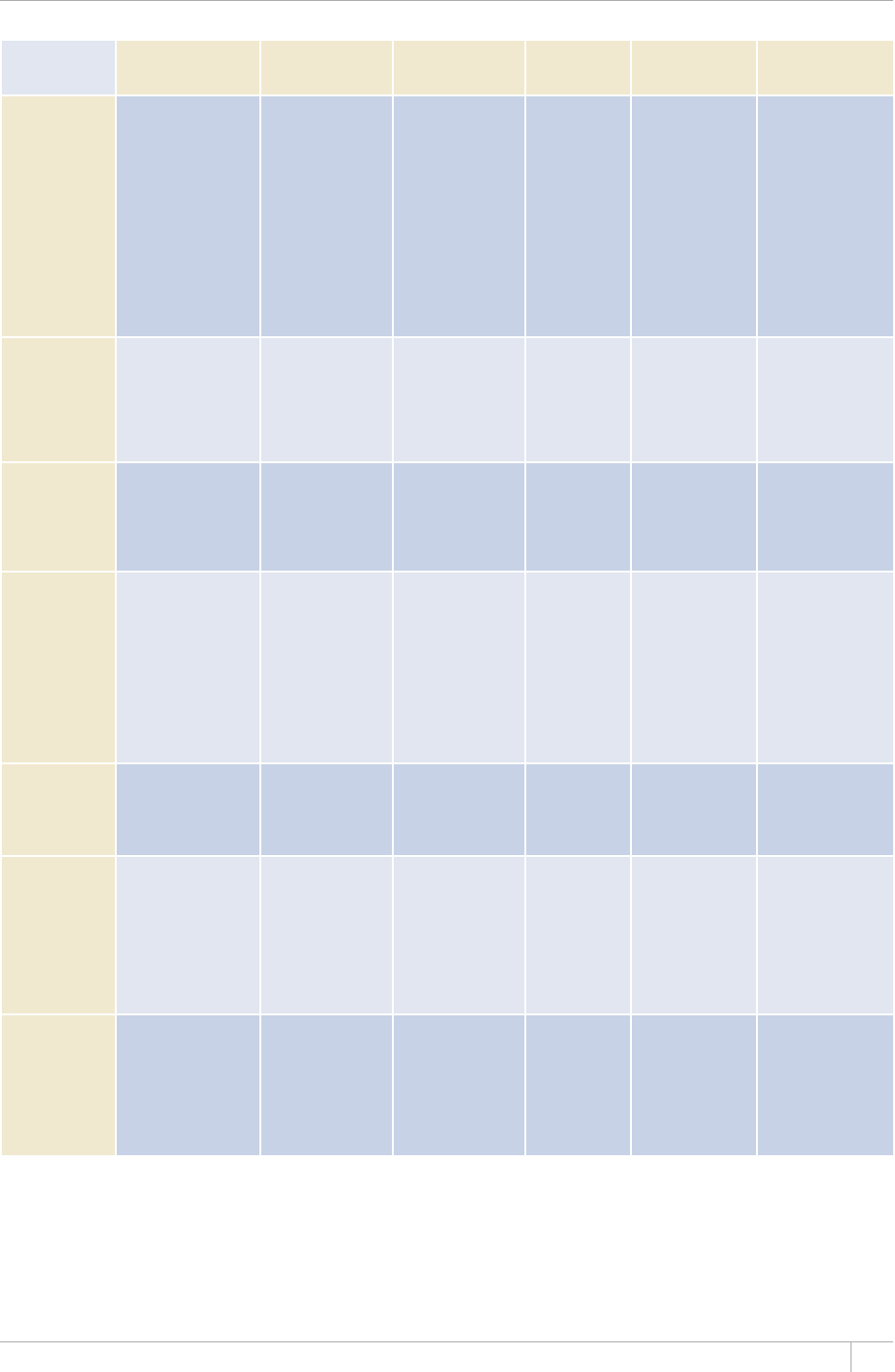
79GLOBAL TUBERCULOSIS REPORT 2013
TABLE 7.1
125 countries included in analyses of TB ßnancing in 2013a,b
LOW-INCOME
QHPQVKßGFECUGUINQDCNN[
.19'4/+&&.'+0%1/'
QHQHPQVKßGFECUGU
globally)
722'4/+&&.'+0%1/'
QHPQVKßGFECUGUINQDCNN[
$4+%5
QHPQVKßGFECUGU
globally)
*+)*$74&'0%17064+'5
':%.7&+0)$4+%5
QHPQVKßGFECUGU
globally)
*+)*/&46$$74&'0
%17064+'5016+06*'.+561(
*+)*$74&'0%17064+'5
QHPQVKßGFECUGUINQDCNN[
African Benin, Burkina Faso,
Burundi, Central
#HTKECP4GRWDNKE
Chad, Comoros,
&4%QPIQ'TKVTGC
Ethiopia, Gambia,
Guinea, Guinea-
Bissau, Kenya, Liberia,
Madagascar, Malawi,
/CNK/Q\CODKSWG
0KIGT4YCPFC
Sierra Leone, Togo,
7ICPFC746CP\CPKC
<KODCDYG
Cameroon, Cape
Verde, Congo, Côte
d’Ivoire, Ghana,
Lesotho, Mauritania,
Nigeria, Sao Tome
and Principe,
5GPGICN5YC\KNCPF
<CODKC
Algeria, Botswana,
Gabon, Namibia,
South Africa
South Africa &4%QPIQ
Ethiopia, Kenya,
/Q\CODKSWG
Nigeria, Uganda,
7PKVGF4GRWDNKE
QH6CP\CPKC
<KODCDYG
Americas Haiti Bolivia, El Salvador,
Guatemala,
Guyana, Honduras,
Nicaragua,
Paraguay
Argentina, Belize,
$TC\KN%QNQODKC
Dominican
4GRWDNKE'EWCFQT
Jamaica, Mexico,
Panama, Suriname,
8GPG\WGNC
$TC\KN
Eastern
Mediterranean
Afghanistan,
South Sudan
Djibouti, Egypt,
Morocco, Pakistan,
Sudan, Syrian
Arab Republic,
9GUV$CPMCPF)C\C
5VTKR;GOGP
Iran, Iraq, Jordan,
Lebanon, Libya,
Tunisia
Afghanistan ,
Pakistan
European Kyrgyzstan,
Tajikistan
Armenia, Georgia,
Moldova, Ukraine,
7\DGMKUVCP
Bosnia and
Herzegovina,
Bulgaria,
Kazakhstan,
Montenegro,
4QOCPKCSerbia,
The Former
Yugoslav
Republic of
Macedonia,
Turkey
4WUUKCP
Federation
Armenia, Bulgaria,
Estonia, Georgia,
-C\CMJUVCP
-[TI[\UVCP.CVXKC
4GRWDNKEQH/QNFQXC
Tajikistan, Ukraine,
7\DGMKUVCP
South-East
Asia
Bangladesh,
Democratic
People’s Republic
of Korea, Myanmar,
Nepal
Bhutan, India,
Indonesia, Sri
Lanka, Timor-Leste
Maldives, Thailand India Bangladesh,
Indonesia,
Myanmar, Thailand
Western
Pacißc
Cambodia Federal States
of Micronesia,
Kiribati, Lao
People’s Democratic
4GRWDNKE/QPIQNKC
Papua New Guinea,
Phillipines, Samoa,
Solomon Islands,
Vanuatu, Viet Nam
American Samoa,
China, Fiji,
Malaysia, Marshall
Islands, Palau,
Tonga, Tuvalu
China Cambodia,
Phillipines, Viet
Nam
Excluded due
to insufßcient
data
Albania, Angola,
#\GTDCKLCP
%QUVC4KEC%WDC
Dominica, Grenada,
Palau, Peru, Saint
Lucia, Saint Vincent
and the Grenadines,
Turkmenistan
#\GTDCKLCP$GNCTWU
Lithuania
a #PCN[UGUHQEWURTKOCTKN[QPNQYCPFOKFFNGKPEQOGEQWPVTKGU6JTGGJKIJKPEQOGEQWPVTKGU'UVQPKC.CVXKCCPFVJG4WUUKCP(GFGTCVKQPYGTGKPENWFGFDGECWUGVJG[CTGKPVJG
NKUVQHJKIJDWTFGPEQWPVTKGUQTVJGNKUVQHJKIJ/&46$DWTFGPEQWPVTKGU
b #FFKVKQPCNEQWPVTKGUKPENWFGFKPCPCN[UGUQH6$ßPCPEKPIKPEQORCTGFYKVJVJQUGKPENWFGFKPCPCN[UGUQHVTGPFU¿CTGUJQYPKPDQNF

GLOBAL TUBERCULOSIS REPORT 2013
1 World malaria report 2012. Geneva, World Health Organization,
2012.
2 World AIDS day report 2012. Geneva, Joint United Nations Pro-
gramme on HIV/AIDS, 2012 (
www.unaids.org/en/resources/
presscentre/pressreleaseandstatementarchive/2012/november/
20121120prresults).
FIGURE 7.3
Available funding for TB care and control in
125 countries reporting 96% of global cases by
source of funding and WHO region, 2013
Global Fund
Grants (excluding
Global Fund)
Government,
general health-
care services
(inpatient and
outpatient care)
Government,
NTP budget
(including loans)
US$ billions (current 2013 US$)
0
0.5
1.0
1.5
2.0
2.5
3.0
EUR AFR WPR SEAR AMR EMR
FIGURE 7.4
Available funding for TB care and control in
125 countries reporting 96% of global cases by
source of funding and income group, 2013
Global Fund
Grants (excluding
Global Fund)
Government,
general health-
care services
(inpatient and
outpatient care)
Government,
NTP budget
(including loans)
US$ billions (current 2013 US$)
Upper-
middle-income
High-
income
Lower-
middle-income
Low-
income
0
0.5
1.0
1.5
2.0
2.5
3.0
FIGURE 7.5
Available funding for TB care and control in BRICS,
17 other HBCs and Africa excluding South Africa,
by source of funding, 2013
Global Fund
Grants (excluding
Global Fund)
Government,
general health-
care services
(inpatient and
outpatient care)
Government,
NTP budget
(including loans)
US$ billions (current 2013 US$)
0
0.5
1.0
1.5
2.0
2.5
3.0
3.5
BRICS Africa without
South Africa
Other HBCs
ambitious targets are developed, as was done for the nine
countries described in section 7.1, funding needs and gaps
invariably increase. e gap between the US$8 billion esti-
mated to be needed for a full response to the TB epidemic in
2015 (section 7.1) and the US$6.1 billion available in 2013
(see section 7.3 below) is US$1.9 billion.
Overall, these ndings show that TB funding increased
substantially between 2002 and 2011, resulting in impres-
sive and cost-eective gains. e increasing self-suciency
of many countries, including BRICS, which account for
almost half of the world’s TB cases, is a success story for
these countries and the global TB community. At the same
time, it is clear that international donor funding remains
crucial in many countries and needs to be continued to sus-
tain and consolidate recent gains.
7.3 TB funding in 2013
Data reported by countries to WHO in the 2013 round of
global TB data collection allowed inclusion of 122 low-
and middle-income countries (GNI per capita less than
US$12616 in 2012) in analyses of TB funding by source
of funding and category of expenditure in 2013 (Table 7.1;
the additional countries compared with those included in
analyses of trends 2002–2011 are shown in bold). An addi-
tional three high-income countries (Estonia, Latvia and
the Russian Federation) were also included in analyses
because they are in the list of 22 HBCs or 27 high MDR-
TB HBCs. Collectively, the 125 countries account for 96%
of the world’s TB cases. Methods used to compile, validate
and analyse these data are summarized in Box 7.1.
e total funding available in the 125 countries in 2013
amounts to US$6.1 billion, and US$3.1 billion excluding
the European Region (Figure 7.3). Approximately US$1.3
billion is available in the African Region, much of which
is accounted for by South Africa. In the other four WHO
regions, funding is in the range US$0.2 to US$0.7 billion.
7.3.1 Funding in 2013 by source of funding
Of the total of US$ 6.1 billion reported for 2013, 87%
(US$ 5.3 billion) is from domestic sources and 13% (US$0.8
billion) is from international donor sources (Figure 7.3,
Figure 7.4). Both fall short of amounts needed for a full
response to the TB epidemic up to 2015 (section 7.1). e
US$5.3 billion from domestic sources represents at most
67% of the total needed by 2015. e US$0.8 billion from
international donor sources is at most 50% of the US$1.6–
2.3 billion per year estimated to be required by 2015, and
remains much less than international donor funding for
malaria (US$ 1.8 billion in 2011)1 and HIV (US$8.2 bil-
lion in 2011).2 Of the international donor funding for
TB in 2013, approximately three-quarters was from the

GLOBAL TUBERCULOSIS REPORT 2013
Global Fund; the remainder was largely from USAID. Tech-
nical assistance to support countries to eectively mobilize
funding from the Global Fund and to implement grants
once approved is provided by the TB Technical Assistance
Mechanism (Box 7.2).
Breakdowns of total funding by source for dierent coun-
try groups are shown in Figure 7.4 and Figure 7.5. Find-
ings strongly reinforce those previously reported for the
decade 2002–2011 (Section 7.2). BRICS are relatively self-
sucient overall (95% of funding from domestic sources),
although India is an exception where only 64% of funding
in 2013 is from domestic sources (and as shown in Annex 1,
BOX 7.1
Methods used to compile, validate and analyse ßnancial data reported by countries to WHO
WHO began monitoring government
CPFKPVGTPCVKQPCNFQPQTßPCPEKPI
for TB in 2002. All data are stored
in the WHO global TB database. The
standard methods used to compile,
review, validate and analyse these
ßPCPEKCNFCVCJCXGDGGPFGUETKDGFKP
detail elsewhere.a,b This box provides a
summary.
Each year, WHO requests data from
low- and middle-income countries
about funding for NTPs by category
of expenditure and source of funding,
and funding gaps by category of
expenditure, in US dollars. Categories
QHGZRGPFKVWTGHQT6$EQORTKUGßTUV
line drugs; NTP staff; programme
management and supervision
activities; laboratory supplies and
equipment; advocacy, communication,
CPFUQEKCNOQDKNK\CVKQPCEVKXKVKGU
community-based care; public–private
mix approaches; collaborative TB/
HIV activities; the Practical Approach
to Lung Health; operational research
including surveys; outpatient visits;
and hospital admissions. Categories of
GZRGPFKVWTGHQT/&46$CTGUGEQPF
NKPGFTWIUQVJGTKVGOUURGEKßECNN[HQT
programmatic management of patients
YKVJ/&46$JQURKVCNCFOKUUKQPUCPF
outpatient visits. Funding sources are
FGßPGFCUPCVKQPCNQTNQECNIQXGTPOGPV
NQCPUDQVJENCUUKßGFCUFQOGUVKE
funding), grants from the Global
Fund, and grants from other donors
DQVJENCUUKßGFCUKPVGTPCVKQPCNFQPQT
HWPFKPI%QWPVTKGUVJCVCTGENCUUKßGF
as high-income are asked to report data
on total funding and total expenditures
(without breakdowns by source of
funding and category of expenditure).
WHO uses methods to review and
validate data that have remained
consistent since 2002. These methods
include routine checks for plausibility
and consistency, including validation
checks that are built into the online
reporting system. Examples of validation
checks are checks for implausibly large
year-to-year changes (for example
in total reported funding by source
and by category of expenditure), or
implausibly high or low values relative
to the number of TB patients (for
GZCORNGßTUVNKPGQTUGEQPFNKPGFTWI
budgets or expenditures per patient
that greatly exceed prices quoted by
the Global TB Drug Facility). Methods
to review and validate data also include
discussions with country respondents to
resolve queries, and triangulation with
other data sources such as the detailed
budgets prepared using the WHO TB
planning and budgeting tool,c economic
evaluations that include detailed
cost data, the Global Fund and the
1TICPK\CVKQPHQT'EQPQOKE%QQRGTCVKQP
and Development (OECD) Creditor
4GRQTVKPI5[UVGO2CTVKEWNCTCVVGPVKQP
has always been given to the 22 HBCs.
In a few countries (China and the
4WUUKCP(GFGTCVKQPCTGRTQOKPGPV
examples), funding for TB reported
by NTPs includes funding for all
staff, infrastructure, and other inputs
necessary for hospital admissions and
outpatient visits during TB treatment,
DGECWUGECTGKURTQXKFGFKP6$URGEKßE
hospitals and clinics that have dedicated
budgets. In most countries, however,
the funding used for inpatient and
outpatient care for TB patients is
not captured in funding reported by
NTPs. Since detailed costing studies
of TB diagnosis and treatment in a
wide range of countries show that
JQURKVCNK\CVKQPCPFQWVRCVKGPVECTGCTG
the most important costs not captured
D[ßPCPEKCNFCVCTGRQTVGFD[062UDQVJ
HQTFTWIUWUEGRVKDNG6$CPF/&46$
VJGGUVKOCVKQPQHßPCPEKCNTGUQWTEGU
used for inpatient and outpatient care
of TB patients has always been given
considerable attention in WHO’s work
QPINQDCNOQPKVQTKPIQH6$ßPCPEKPI
For all countries with the exception of
VJQUGUWEJCU%JKPCCPFVJG4WUUKCP
Federation, the funding used for
inpatient and outpatient care of TB
patients is estimated by multiplying
the number of outpatient visits and
days of inpatient care per patient
(reported by NTPs to WHO each year) by
EQWPVT[URGEKßEGUVKOCVGUQHVJGKTWPKV
cost available from the WHO-CHOICE
database,d and then by the reported
number of TB patients. This is done
UGRCTCVGN[HQTCRCVKGPVUYKVJFTWI
susceptible TB; and b) patients with
/&46$DCUGFQPVJGWVKNK\CVKQPFCVC
that are reported separately for these
two groups of patients on the annual
WHO TB data collection form.
a Floyd K, Pantoja A, Dye C. Financing tuberculosis
EQPVTQNVJGTQNGQHCINQDCNßPCPEKCN
monitoring system. Bulletin of the World *ealth
OrIanization¿
b (NQ[F-(KV\RCVTKEM%2CPVQLC#CPF4CXKINKQPG
/&QOGUVKECPFFQPQTßPCPEKPIHQT
tuberculosis care and control in low-income and
OKFFNGKPEQOGEQWPVTKGUCPCPCN[UKUQHVTGPFU
2002–11, and requirements to meet 2015
targets. The Lancet Global *ealthG¿
c PlanninI and budIetinI for TB control actiXities.
)GPGXC9QTNF*GCNVJ1TICPK\CVKQP
(YYYYJQKPVVDFQVURNCPPKPIADWFIGVKPIAVQQN).
d ChoosinI interXentions that are cost effectiXe
W*O-C*OICE. Geneva, World Health
1TICPK\CVKQPwww.who.int/choice/
EQWPVT[EQWPVT[AURGEKßE).
for the NTP budget specically, 37% is funded from domes-
tic sources in 2013). High-income countries are fully self-
sucient and the group of upper middle-income countries
rely on international donor funding for only a small share
(4%) of their total funding (and most is accounted for by
China). Low- and lower middle-income countries account
for most of the international donor funding (US$0.7 bil-
lion, 88%). In the group of low-income countries, it accounts
for about half of total funding. International donor fund-
ing also has a crucial role in the 17 HBCs excluding BRICS,
and in African countries excluding South Africa (Figure
7.5), where it accounts for 35% and 54% respectively of

GLOBAL TUBERCULOSIS REPORT 2013
BOX 7.2
Technical Assistance for national TB programmes; the role of TB-TEAM
The TB Technical Assistance Mechanism (TB-TEAM) was
established to coordinate and monitor the provision of technical
assistance to NTPs. The secretariat and a dedicated websitea are
hosted by WHO’s Global TB Programme and funded by USAID.
In mid-2013, there were 34 technical partners actively engaged
in TB-TEAM. Each partner shares information about country
missions, including reports that are uploaded to the country-
URGEKßERCIGUQHVJG6$6'#/YGDUKVG
In 2012, TB-TEAM partners reported 706 missions. By
topic area, laboratory strengthening and the programmatic
OCPCIGOGPVCPFUECNGWRQH/&4CPF:&46$CEEQWPVGFHQT
one quarter of all missions. A further 30% of missions were
related to monitoring and evaluation/impact measurement,
national TB programme reviews and management of drugs and
commodities. Most missions were conducted by WHO (40%)
and KNCV Tuberculosis Foundation (20%). A further 24% were
conducted by the US Centers for Disease Control and Prevention
(CDC); the Union, and the Global TB Drug Facility (GDF).
The main focus of TB-TEAM to date has been provision of
technical assistance to support the implementation of grants
from the Global Fund. Within the context of the fund’s new
funding model (NFM) established in 2013, this is now being
extended to support the development of robust national
strategic plans and associated concept notes that are required
HQTVJGOQDKNK\CVKQPQHPGYßPCPEKCNTGUQWTEGUCUQRRQUGF
to implementation of grants that have already been secured)
from the Global Fund. The focus on support to countries that
CTGEWTTGPVQTRQVGPVKCN)NQDCN(WPFTGEKRKGPVUTGàGEVUVJGHCEV
that the fund is the main source of international donor funding
in many countries, especially in low-income countries and
several high-burden, lower-middle income countries (section
7.2). TB-TEAM partners are taking a proactive approach to
providing technical support to countries, giving particular
attention to grants that are not performing well. The TB-TEAM
UGETGVCTKCVOQPKVQTURTQITGUUKPOQDKNK\CVKQPQHHWPFKPICPF
implementation of grants using indicators such as proposal
success rates, funding for TB as a share of total grant approvals,
disbursement rates and grant performance ratings.
5VCVKUVKEUHQTVJGUGKPFKECVQTUKPECPDGUWOOCTK\GFCU
HQNNQYU
Proposal success rates. TB-TEAM helped 21 countries
VQOQDKNK\GTGUQWTEGUXKCVJG)NQDCN(WPFÂUVTCPUKVKQPCN
funding mechanism (TFM). This was put in place during
the transition to the NFM to prevent countries from
experiencing critical funding shortages that would affect
essential services. Among the three diseases supported
by the fund, TB proposals had the best recommendation
TCVGVJG6GEJPKECN4GXKGY2CPGNQHVJG)NQDCN(WPF
TGEQOOGPFGFVJCVQH6$RTQRQUCNUUJQWNFDGCRRTQXGF
compared with 79% for malaria and 62% for HIV).
TB as a share of total funding.+PVJG6(/75a
million was awarded to TB grants, equivalent to 25% of all
approved funding.
Disbursement rates. In total and across all grants in 101
EQWPVTKGU75aOKNNKQPYCUFKUDWTUGFHQT6$KP
GSWKXCNGPVVQQHVQVCNFKUDWTUGOGPVU75aDKNNKQP
D[VJG)NQDCN(WPF1HVJGTGOCKPKPIHWPFKPI75
DKNNKQPYCUFKUDWTUGFVQ*+8ITCPVU75aDKNNKQP
VQOCNCTKCITCPVUCPF75aOKNNKQPVQETQUU
cutting investments.
Grant performance ratings. At the end of 2012, TB
grants were performing relatively well (Figure B7.1.1),
YKVJKPVJGVQRVJTGGECVGIQTKGUQH#GZEGNNGPV#
(meets expectations) and B1 (adequate), compared with
53% for malaria grants and 79% for HIV grants. The other
categories are B2 (adequate but potential demonstrated)
and C (inadequate).
FIGURE B.7.1.1
Latest Global Fund performance rating by disease
HQTCNNCEVKXGITCPVU)NQDCN(WPF&CVCDCUG
CEEGUUGF,CPWCT[
C
B2
B1
A2
A1
Percentage (%)
0
20
40
60
80
100
Malaria
(110 grants)
HIV/AIDS
(183 grants)
TB
(130 grants)
Grand Total
(423 grants)
In 2013 and 2014, Global Fund projections suggest that an
GUVKOCVGF75aDKNNKQPYKNNDGFKUDWTUGFVQ6$ITCPVU6JKU
equates to an amount per year that is approximately double the
level of 2012. TB-TEAM aims to support countries as effectively
as possible to help to ensure that these funds are disbursed and
used well.
a www.stoptb.org/countries/tbteam/

GLOBAL TUBERCULOSIS REPORT 2013
TABLE 7.2
Reported NTP budget, available funding for NTP budget by intervention area and estimated cost
of inpatient and outpatient care for drug-susceptible (DS-TB) and MDR-TB, 36 high TB or high MDR-TB
burden countries, 2013 (current US$ millions)
4'2146'&
NTP BUDGET
AVAILABLE FUNDING
INPATIENT AND
1762#6+'06%#4'
DS-TBb
INPATIENT AND
1762#6+'06%#4'
/&46$b
DS-TB /&46$ TB/HIV
PPM/PAL/ACSM/
%$%145748';5 16*'4
22 HIGH-BURDEN COUNTRIES
Afghanistan 13 6.0 00.71.32.90
Bangladesh 43 4.6 1.6 0 1.9 0.2 5.0 1.4
$TC\KN 60 6.3 2.3 020 1.4
Cambodia 24 5.3 0.6 0.2 2.2 0.7 6.7 0.2
China 359 267 25 0.2 12 0.5 0 0
&GOQETCVKE4GRWDNKEQHVJG%QPIQ 61 1.7 0.3 1.0 4.5 0.2 0
Ethiopia 145 47 6.0 3.1 12 3.6 11 0.6
India 67 0 2.1 32
Indonesia 119 39 1.3 0.6 39 2.0
Kenya 55 19 0.5 0.5 0.5 90.3
/Q\CODKSWG 11 5.6 1.1 0 05.70.1
Myanmar 36 9.1 3.5 1.6 0.4 0 5.6 1.6
Nigeria 154 17 4.6 1.6 3.6 22 6.2 1.3
Pakistan 73 26 34 0.1 1.5 5.0 11
Philippines 149 27 0.4 6.9 2.7 109 3.4
4WUUKCP(GFGTCVKQPc1 592 1 332 129 27 0.4 104 0 0
South Africa 475 217 41 124 19 67 109 232
Thailanda44 31 3.9 0.1 03.30
Uganda 31 6.0 2.2 0.2 3.9 9.3 0.6 0
7PKVGF4GRWDNKEQH6CP\CPKC 14 0.5 2.1 0.9 1.3 1.5 0.1
Viet Nam 66 4.4 4.6 1.2 3.4 49 0.6
<KODCDYG 11 0.1 3.4 0.5 1.4 15 0.1
22 high-burden countries total 3 814 2 241 350 170 111 232 494 279
REMAINING HIGH MDR-TB BURDEN COUNTRIES
Armenia 5.4 4.5 0.6 0 0.1 0.2 7.5 1.2
#\GTDCKLCP ––
Belarusa––
Bulgaria 16 14 0.3 0 0.6 0.3 22 1.5
Estonia 0.1 0.5 0 0 0.2 0.1 0.1
Georgia 10 2.6 00 3.6 3.2
-C\CMJUVCP 242 149 70 1.0 1.7 21 192 57
-[TI[\UVCP 35 11 5.6 0.3 13 4.0 13 3.1
Latvia 3.7 1.1 0 0 0.1 20 7.3
Lithuania ––
4GRWDNKEQH/QNFQXCa35 2.6 00.615113.2
Tajikistan 46 5.0 1.1 0.6 1.1 6.6 1.1
Ukraine 21 15 0.6 0 7.9 66 40
7\DGMKUVCP 76 15 14 0 1.5 46 5.7
27 high MDR-TB burden
countries 4 011 2 312 448 164 108 325 854 400
36 high TB or high MDR-TB
burden countries 4 371 2 471 464 172 130 340 919 402
Blank cells indicate data not reported.
– indicates values that cannot be calculated.
a Based on data reported for 2013 in the 2012 round of data collection. In 2013, Thailand was not able to report funding for the sub-national level.
b 0QCOQWPVKUUJQYPHQT%JKPCCPFVJG4WUUKCP(GFGTCVKQPDGECWUG062DWFIGVKPENWFGUCNNEQUVUHQTKPRCVKGPVCPFQWVRCVKGPVECTG
c 6JGUVCHHCPFKPHTCUVTWEVWTGTGSWKTGFHQT6$ECTGCPFEQPVTQNEQWNFPQVDGFKUCIITGCVGFHQT/&46$CPF&56$UGRCTCVGN[CPFCTGUJQYPWPFGT&56$6JGHWNNCOQWPVHQTUVCHH
and other recurrent costs for TB hospitals is included in the column for DS-TB.
GLOBAL TUBERCULOSIS REPORT 2013
FIGURE 7.6
Available funding for TB care and control in
125 countries reporting 96% of global cases by
intervention area and WHO region, 2013
Other
TB/HIV
Inpatient and
outpatient care:
MDR-TB
MDR-TB
Inpatient and
outpatient care:
DS-TB
DS-TBb
US$ billions (current 2013 US$)
0
0.5
1.0
1.5
2.0
2.5
3.0
EURaAFR WPR SEAR AMR EMR
a (QT'74&56$KPENWFGUCNNQHVJGUVCHHCPFKPHTCUVTWEVWTGTGSWKTGFHQT6$ECTG
CPFEQPVTQNKPVJG4WUUKCP(GFGTCVKQPVJCVEQWNFPQVDGFKUCIITGICVGFHQT/&46$
CPF&56$UGRCTCVGN[6JGCOQWPVQHHWPFKPIUJQYPHQT/&46$KPVJG'WTQRGCP
4GIKQPKUVJWUCPWPFGTGUVKOCVG
b &TWIUWUEGRVKDNG6$&56$KPENWFGUHWPFKPICXCKNCDNGHQTßTUVNKPGFTWIU062
staff, programme management and supervision, and laboratory equipment and
supplies.
FIGURE 7.7
Available funding for TB care and control in BRICS,
17 other HBCs and Africa excluding South Africa,
by intervention area, 2013
Other
TB/HIV
Inpatient and
outpatient care:
MDR-TB
MDR-TB
Inpatient and
outpatient care:
DS-TB
DS-TBa
US$ billions (current 2013 US$)
0
0.5
1.0
1.5
2.0
2.5
3.0
3.5
BRICS Africa without
South Africa
Other HBCs
a (QT$4+%5FTWIUWUEGRVKDNG6$&56$KPENWFGUCNNQHVJGUVCHHCPFKPHTCUVTWEVWTG
TGSWKTGFHQT6$ECTGCPFEQPVTQNKPVJG4WUUKCP(GFGTCVKQPVJCVEQWNFPQVDG
FKUCIITGICVGFHQT/&46$CPF&56$UGRCTCVGN[6JGCOQWPVQHHWPFKPIUJQYPHQT
/&46$KP$4+%5KUVJWUCPWPFGTGUVKOCVG
total funding in 2013. e share is even higher in specic
countries and above 80% in four HBCs: Afghanistan, the
Democratic Republic of the Congo, Pakistan and Uganda
(Annex 2).
7.3.2 Funding in 2013 by budget category
Funding in 2013 by budget category is shown by WHO
region in Figure 7.6, for other country groups in Figure 7.7
and for HBCs and high-MDR-TB burden countries in Table
7.2. It should be highlighted that the amount of funding
shown for MDR-TB in the European Region is an underesti-
mate due to the fact that the budget category ‘Drug-suscep-
tible-TB’ (DS-TB) includes all of the sta and infrastructure
required for TB prevention, diagnosis and treatment in the
Russian Federation that could not be disaggregated for
MDR-TB and DS-TB separately. Among the 122 low- and
middle-income countries for which a breakdown could be
calculated, most of the funding available in 2013 is for
diagnosis and treatment of DS-TB.
e WHO regions in which the shares of funding
for MDR-TB are highest are the African Region (mostly
explained by South Africa), the European Region and the
South-East Asia Region. is is consistent with the distri-
bution of the burden of MDR-TB cases, which are mostly in
BRICS and the European Region, and with the latest data
on numbers of MDR-TB patients detected and enrolled on
treatment (Chapter 4). ese data show that European
countries and South Africa are enrolling the highest pro-
portion of estimated cases of MDR-TB on treatment and
that progress in scaling up treatment in India (in the South-
East Asia Region) is accelerating. e low share of funding
for MDR-TB in the Western Pacic Region, within which
most of the estimated cases of MDR-TB are in China, is
consistent with the small number of cases reported to have
been detected and started on treatment in China in 2012
(just over 3000, equivalent to 5% of the estimated number
of TB patients with MDR-TB). Among the 22 HBCs, 85% of
the available funding for MDR-TB treatment is accounted
for by BRICS (Table 7.2).
Most of the reported funding for collaborative TB/HIV
activities is accounted for by the African Region (77%),
followed by Europe (16%). is is consistent with the dis-
tribution of the burden of TB/HIV: the latest estimates are
that 75% of HIV-positive TB patients are in the African
Region (Chapter 6).
7.3.3 Reported funding gaps in 2013
In 2013, funding gaps reported by NTPs (i.e. the dierence
between assessments by NTPs of funding needs for TB pre-
vention, diagnosis and treatment and the actual amount of
funds mobilized) amount to US$1 billion. is is a consid-
erable increase from gaps in the range US$0.3–0.6 billion
that were reported during the decade 2002–2011 (section
7.2). A possible explanation may be that NTPs are devel-
oping more ambitious plans for implementation and scale
up of interventions with resulting increases in funding
gaps. African countries account for almost half of the total
(Figure 7.8a), followed by Asian countries (37% of the
total). Funding gaps were reported by countries in all
income groups with the exception of high-income countries
(Figure 7.8b), and for multiple elements of TB prevention,
diagnosis and treatment (Figure 7.8c).
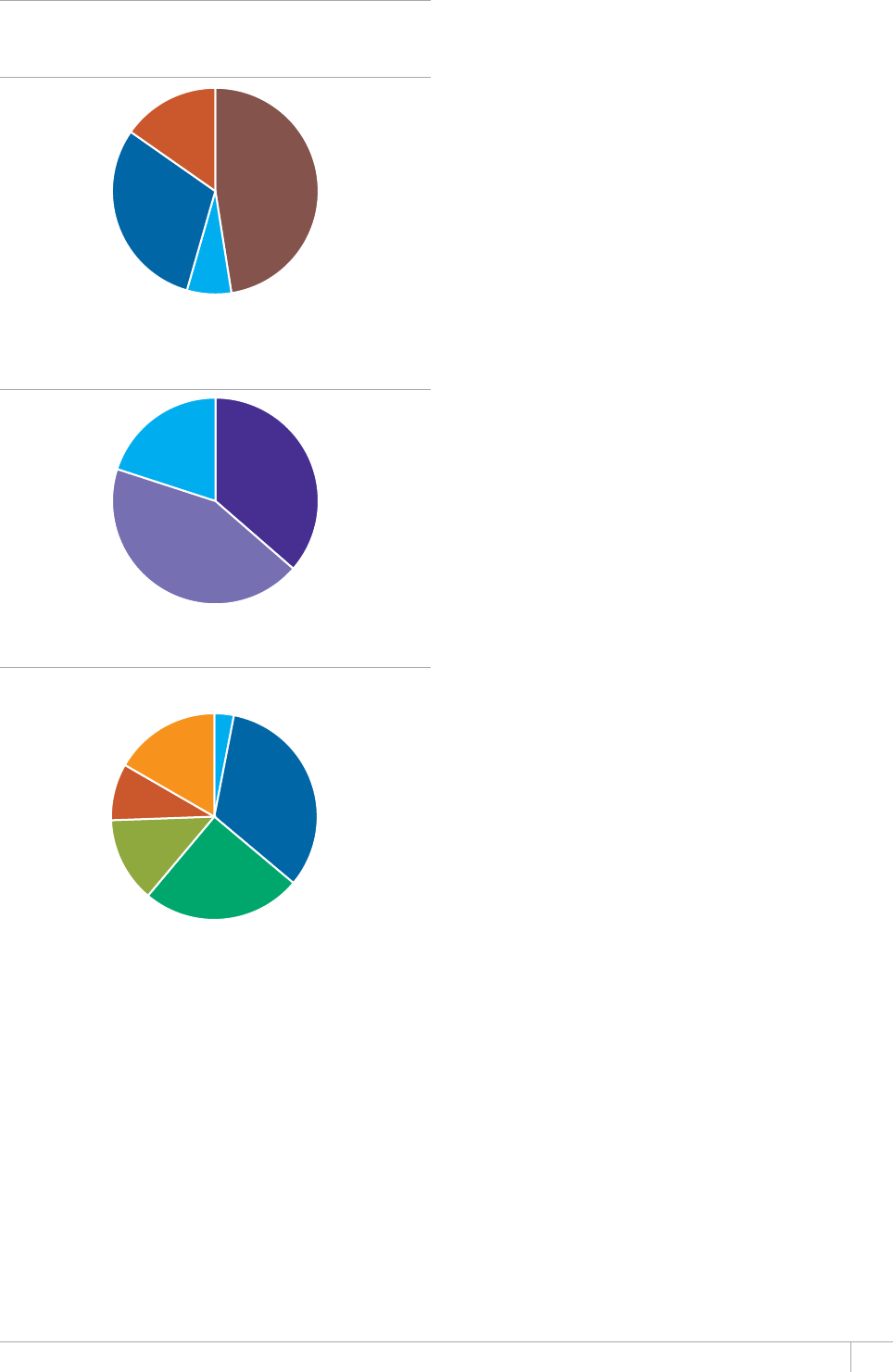
GLOBAL TUBERCULOSIS REPORT 2013
Africa 48%
Asia: Low-income,
high-burden 7%
Asia: Other 30%
Rest of World 15%
FIGURE 7.8
Funding gaps reported by national TB programmes
in 125 countries with 96% of global cases, 2013
a. By region and income group
b. By income group
c. By intervention area
Low-income 36%
Lower-middle-income 44%
Upper-middle-income 20%
DS-TB, first-line
drugs 3%
DS-TB, excluding
first-line drugs 33%
ACSM/CBC/PPM/PAL/OR surveys 25%
MDR-TB 13%
TB/HIV 9%
Other 17%
GLOBAL TUBERCULOSIS REPORT 2013
%*#26'4
Research and development
KEY FACTS AND MESSAGES
■ Efforts to develop new TB diagnostics, drugs and
XCEEKPGUJCXGKPVGPUKßGFFWTKPIVJGRCUVFGECFGCPF
considerable progress has been made.
■ More than 50 companies are involved in the development
of TB diagnostics. Although many new diagnostic
VGEJPQNQIKGUCTGCXCKNCDNGQPVJGOCTMGVCEEGNGTCVGFßGNF
evaluation of diagnostic accuracy and robustness of these
assays is needed.
■ Increased and sustained investment in new TB
diagnostics remains essential for the development of an
accurate, easy-to-use, affordable point-of-care assay for the
rapid and early diagnosis of TB.
■ There are 10 new or repurposed anti-TB drugs currently
in late phases of clinical development. In December 2012,
one of the new compounds, bedaquiline, was approved for
WUGKPVTGCVOGPVQHRCVKGPVUYKVJ/&46$D[VJG75(QQF
and Drug Administration (FDA). Interim guidance about the
WUGQHDGFCSWKNKPGKPVJGVTGCVOGPVQH/&46$YCUKUUWGF
D[9*1KP,WPG$GFCSWKNKPGKUVJGßTUVPGYFTWI
approved for TB treatment in many years.
■ 4GUWNVUHTQOVYQ2JCUG+++VTKCNUQHHQWTOQPVJTGIKOGPU
for the treatment of drug-susceptible TB are expected in late
2013. New combination regimens are also being tested in
a series of early bactericidal activity (EBA) or two-month
sputum-culture conversion Phase II studies.
■ There are 10 vaccine candidates for TB prevention
in Phase I, Phase II or Phase IIb trials and two
immunotherapeutic vaccines in Phase II or Phase III trials.
■ 4GUWNVUHTQOC2JCUG++DRTQQHQHEQPEGRVUVWF[QHVJG
XCEEKPGECPFKFCVG/8##YGTGRWDNKUJGFKP(GDTWCT[
2013. Among infants who received the vaccine as a boost
to the Bacille-Calmette-Guérin (BCG) vaccine, no additional
protection was conferred compared with BCG alone. This
study demonstrated, however, that the vaccine had an
CEEGRVCDNGUCHGV[RTQßNGKPVJKURQRWNCVKQPCPFVJCVCJKIJ
quality trial of a novel TB vaccine can be conducted and
produce robust results in a high TB burden setting.
■ 4GUGCTEJCPFFGXGNQROGPVKUQPGQHVJGVJTGGRKNNCTUQH
the WHO post-2015 global TB strategy, in recognition of its
crucial role in accelerating reductions in TB incidence and
mortality to reach post-2015 global TB targets.
e proposed goal of the post-2015 global TB strategy is
to end the global TB epidemic (Chapter 1). Despite major
progress in TB care and control since the mid-1990s (Chap-
ters 2–7), reaching this goal will require major technolog-
ical breakthroughs from the research and development
pipeline. Short, eective and well-tolerated treatments for
latent TB infection, a point-of-care diagnostic test able to
distinguish latent TB infection from active TB disease, and
an eective post-exposure vaccine are of key importance to
end the global TB epidemic.
is is the third successive year in which a chapter on
research and development has been included in the Global
tuberculosis report. e status of progress in the develop-
ment of new TB diagnostics, drugs and vaccines as of July
2013 is summarized, drawing on information provided by
the secretariats of the relevant Working Groups of the Stop
TB Partnership and recent publications. Particular atten-
tion is given to developments between August 2012 and
July 2013. e nal section of the chapter highlights key
elements of the research and development agenda post-
2015.
8.1 New diagnostics for TB
Sputum smear microscopy remains the most widely used
diagnostic test for TB, despite its relatively low sensitivity
(especially for those with paucibacillary TB such as people
living with HIV and children). e current reference stan-
dard for the bacteriological conrmation of TB is culture
in liquid media. However, culture-based diagnosis is not
widely available in most high TB burden settings because it
requires sophisticated laboratory and biosafety infrastruc-
ture, and test results take up to several weeks to obtain.
Recent breakthroughs include the development of rapid
molecular tests that can be used to diagnose TB and rifam-
picin-resistant TB at decentralized levels of health systems.
ese tests are now being rolled out worldwide (see also
Chapter 5). However, TB remains unique among the major
infectious diseases in lacking accurate and rapid point-of-
care tests, largely due to insucient progress in biomark-
er discovery despite active ongoing research. Indeed, the
most pressing priority in TB diagnostics research today
is the development of a simple, low-cost, instrument-free
rapid test using one or more reliable biomarkers that can
be implemented at the rst point of patient contact with
peripheral health services, or used as a triage test at com-
munity level to rapidly identify people who should be
referred for conrmatory testing.
e status of development and evaluation of new TB

GLOBAL TUBERCULOSIS REPORT 2013
diagnostics in July 2013 is summarized in Figure 8.1,
based on recent documentation produced by UNITAID1
and the Treatment Action Group (TAG).2 In Figure 8.1,
diagnostic tests and methods on the market are grouped
according to whether they have been evaluated by WHO
and, if so, whether they have been endorsed. Given the
rapidly evolving TB diagnostic landscape, WHO has estab-
lished a systematic process for the timely evaluation of
evidence and formulation of policy on new TB diagnostics.
is is described in Box 8.1 and further details are avail-
able elsewhere. It should also be highlighted that the list of
technologies in ‘early development’ is not necessarily com-
plete or exhaustive.3 ose listed are the ones documented
in the UNITAID and TAG reports.
Development of molecular technologies such as nucle-
ic acid amplication tests (NAATs) is most advanced i.e.
either already commercially available or in late-stage devel-
opment. e majority of tests are, however, intended for
use at reference laboratory level only, requiring dedicated
infrastructure and experienced sta. Most NAATs require
manual preparation of samples, which is technically chal-
lenging and prevents their use at more decentralized labo-
ratory levels. Testing in reference laboratories oers higher
throughput of tests and/or improved screening of samples
for drug resistance markers, but is typically relatively
expensive. e next-generation molecular tests that have
emerged since Xpert® MTB/RIF have not yet undergone rig-
orous eld trials in the settings where their use is intend-
ed, and substantial challenges with sample processing and
DNA extraction in peripheral laboratories has been report-
ed for all of them.
Technologies in the early stages of development (rst
part of Figure 8.1) include tests to detect TB, drug resis-
tance, or TB and drug resistance combined. ese include
microarray-based multiplexing diagnostic platforms for
the simultaneous detection of a large number of resistance-
conferring mutations; assays that use novel approaches to
combine nucleic acid testing with phage-based technology
to identify drug resistance in clinical isolates; a rapid colo-
rimetric culture-based method for detection of resistance
to rifampicin, isoniazid and uoroquinolones for use at
the intermediate laboratory level; second-generation Xpert
assays for the detection of resistance to drugs other than
rifampicin; and a cartridge-based point-of-care isothermal
amplication platform. In addition to technologies aimed
a This is not an exhaustive list of technologies in early development. Those listed are
the ones documented in recent (2013) publications by UNITAID and TAG.
b 7RFCVGFRQNKE[IWKFCPEGQP:RGTV/6$4+(KUWPFGTFGXGNQROGPV5GG%JCRVGT
for further details.
FIGURE 8.1
An overview of progress in the development and
evaluation of TB diagnostics, July 2013
Technologies in early developmenta
Volatile organic compounds
$TGCVJ.KPM/GPUUCPC4GUGCTEJ75#
2TQVQV[RGDTGCVJCPCN[\GTFGXKEG0GZV&KOGPUKQPU
Technology, USA
Molecular technologies
Alere Q, Alere, USA
$5/#46.CD%QTR75#
)GPFTKXG/6$4+(+&'RKUVGO7-
.#6'2%4$TCPFGKU7PKXGTUKV[75#
)GPG:RGTV:&4ECTVTKFIG%GRJGKF75#
6TW#TTC[/&46$#MMQPK75#
INFINITIMTB Assay, AutoGenomics, USA
Culture-based technologies
BNP Middlebrook, NanoLogix, USA
/&4:&46$%QNQT6GUV(+0&5YKV\GTNCPF+ORGTKCN
College, UK
64'-5GPUKVKVTG/;%16$/+%RNCVG6TGM&KCIPQUVKE
5[UVGOU6JGTOQ(KUJGT5EKGPVKßE75#
Other technologies
6$4CRKF5ETGGP)NQDCN$KQ&KCIPQUVKEU75#
TBDx, Signature Mapping Medical Sciences, USA
Evaluated by WHO but not yet endorsed
due to insufßcient evidence
Molecular technologies
TB LAMP, Eiken, Japan
)GPQV[RG/6$&4UN*CKP.KHGUEKGPEG)GTOCP[
On the market but evidence for use not yet
submitted to WHO for evaluation
Molecular technologies
iCubate System, iCubate, USA
TB drug resistance array, Capital Bio, China
EasyNAT TB Diagnostic kit, Ustar Biotechnologies, China
Truelab/Truenat MTB, Molbio/bigtec Diagnostics, India
Non-molecular technologies
Alere Determine TB-LAM, Alere, USA
Evaluated by WHO and not recommended
Commercial serodiagnostics (all manufacturers)
Interferon-gamma release assays for the detection of
active TB (all settings)
Technologies endorsed by WHO
Molecular technologies
:RGTV/6$4+(b
Line probe assays (acid-fast bacilli smear-positive
sputum specimens or culture-positive specimens)
Microscopy
<KGJN0GGNUGPCPFàWQTGUEGPEGOKETQUEQR[OGVJQFU
Culture-based technologies
Commercial liquid culture systems and rapid speciation
Non-commercial culture and drug susceptibility testing
methods
1 Tuberculosis: Diagnostics Technology and Market Landscape 2013.
Geneva, UNITAID/World Health Organization, 2013. Available
at: http://www.unitaid.eu/images/marketdynamics/publications/
TB-Dx-Landscape_1-Jul-2013.pdf
2 Clayden P. et al (on behalf of e HIV i-Base/Treatment Action
Group) 2013 Pipeline Report: HIV, Hepatitis C Virus (HCV), and
Tuberculosis (TB) Drugs, Diagnostics, Vaccines, Preventive Technolo-
gies, Research Toward a Cure, and Immune-Based and Gene erapies in
Development. New York, Treatment Action Group, 2013. Available
at: http://www.treatmentactiongroup.org/pipeline-report
3 Weyer K et al. Rapid molecular TB diagnosis: evidence, policy-mak-
ing and global implementation of Xpert® MTB/RIF European
Respiratory Journal erj01572-2012; published ahead of print 2012,
doi:10.1183/09031936.00157212.
GLOBAL TUBERCULOSIS REPORT 2013
at diagnosis of TB and drug-resistant TB, assays for use
in monitoring patients’ response to treatment are needed
as alternatives to culture. Ribosomal RNA (rRNA)-based
amplication assays have potential to be used in this way,
given that rRNA levels per TB bacilli are much higher than
genomic DNA targets and that they are present only in
viable organisms.
Several new diagnostic technologies are on the market,
but evidence to support their use has not been provided
to WHO and thus their performance characteristics have
not been assessed and WHO cannot recommend their use.
As an alternative to real-time polymerase chain reaction
(PCR) assays (e.g. Xpert MTB/RIF) or line-probe assays for
the detection of TB and drug resistance, these technolo-
gies include PCR assays combined with DNA microarrays
(arrays), which allow the detection of a greater number
of resistance conferring alleles and may potentially oer
superior performance for the rapid detection of drug resis-
tance. e technologies include:
iCubate system (iCubate, USA). is is a multiplexed
PCR assay that detects TB, non-tuberculous mycobac-
teria and drug resistance-conferring mutations in a
single reaction. e assay allows multiple targets to be
amplied with array detection technology that simulta-
neously analyses multiple targets. e assay is currently
available for research purposes only.
Capital Bio Corporation (China) has developed a TB drug
resistance detection array kit that can detect 14 of the
most frequently found mutations in three genes asso-
ciated with resistance to rifampicin and isoniazid. e
assay is currently only appropriate for testing at refer-
ence laboratory level given the complexity of performing
the assay.
EasyNAT TB Diagnostic kit, Ustar Biotechnologies,
(China) has developed three isothermal based NAATs
for the detection of TB as well as rifampicin and isoni-
azid resistance-conferring mutations. A clinical trial
conducted in four provinces across central and northern
China showed promising preliminary results for a rapid
and easy-to-use screening tool for the diagnosis of pul-
monary TB.
A micro-PCR system developed by Truelab™ (Molbio,
India) was launched in 2013 in India. e system uses
microchips with TB-specic genetic sequences for the
quantitative detection of TB DNA in sputum samples
in a one hour reaction from sample preparation to nal
reporting of results. Battery powered equipment is used
for the steps of DNA extraction, amplication and detec-
tion. Although promising, only limited evaluation data
are currently available.
Alere Determine LAM, (Alere, USA). is is an assay for
the detection of M. tuberculosis lipoarabinomannan in
urine. e assay seems to be most useful for the diagno-
sis of TB in people living with HIV who have a low CD4
count.
BOX 8.1
Evidence required for WHO review of
new diagnostics
Phase 1: Research and Development
7RUVTGCOTGUGCTEJCPFFGXGNQROGPVVQFGßPGCPF
validate a prototype;
Laboratory validation under international standards that
culminates in a design-locked product;
WHO interacts with developers if requested to discuss
end-user requirements such as biosafety, assay
robustness and intended settings of use.
Phase 2: Evaluation and Demonstration
The performance of the new diagnostic product should
be evaluated in controlled trials at 3–5 trial sites in
high-burden TB and HIV countries;
Product registration with global and/or national
regulatory authorities;
2TQFWEVURGEKßECVKQPUCPFRGTHQTOCPEGUJQWNF
subsequently be validated in uncontrolled trials under
ßGNFEQPFKVKQPUKP¿VTKCNUKVGUKPJKIJDWTFGP6$CPF
HIV countries, and include cost-effectiveness studies.
Phase 3: Evidence Assessment
NEW TECHNOLOGIES
Submission of a dossier with Phase I and Phase II data to
WHO.
FAST-FOLLOWER
/CPWHCEVWTGFWPFGT+51UVCPFCTFU
Equivalent performance demonstrated – Supranational
4GHGTGPEG.CDQTCVQT[EQORCTKUQP
Structured evidence assessment using the Grading
QH4GEQOOGPFCVKQPU#UUGUUOGPV&GXGNQROGPVCPF
'XCNWCVKQP)4#&'CRRTQCEJ
WHO does not recommend technologies for individual
country use.
Phase 4: Phased uptake and collection of evidence
for scale-up
New diagnostic successfully implemented in routine
diagnostic services by early implementers in high-burden
countries;
Systematic assessment of proposed algorithms,
laboratory workload, operational constraints and cost-
effectiveness;
Lessons learnt by early implementers used for country
adaptation.
Phase 5: Scale-up and Policy reßnement
Scale-up of the new diagnostic, with subsequent data
WUGFVQKPHQTOCPFTGßPG9*1RQNKE[IWKFCPEGKPC
dynamic and on-going process.

GLOBAL TUBERCULOSIS REPORT 2013
Two tests are commercially available but have not been
endorsed by WHO after evaluation. e rst is a manual
molecular assay to detect TB DNA in sputum specimens
(TB-LAMP®, Eiken Chemical Co. Ltd., Japan). e evi-
dence-based process followed by WHO concluded that the
data available for the TB-LAMP assay were insucient to
proceed with the development of policy guidance. Addition-
al independent evaluation studies to investigate TB-LAMP
as a replacement test for culture are now underway in 16
countries (17 sites). e second assay evaluated by WHO
but not endorsed is a line probe assay for detecting resis-
tance to second-line anti-TB agents (GenoType® MTBDRsl,
Hain Lifescience, Germany). is cannot be recommended
as a replacement test for conventional phenotypic testing
for drug susceptibility because of suboptimal sensitivity in
detecting resistance to uoroquinolones and second-line
injectable agents. e latter group of drugs also share muta-
tions, which means that even if they are detected by the
line probe assay it is not possible to identify exactly which
drug(s) is linked to the detected mutation(s), and therefore
the test cannot be used to guide the choice of individual
injectable drugs to be used in treatment regimens for MDR-
TB. Conventional phenotypic testing for drug susceptibil-
ity to second-line drugs therefore remains necessary for
all detected strains of MDR-TB and to conrm or exclude
XDR-TB.
Two rapid molecular tests have been evaluated and
endorsed by WHO in recent years (bottom of Figure 8.1).
Line-probe assays that allow the rapid diagnosis of TB
and drug resistance within a day were endorsed in 2008.1
eir use is currently limited to acid-fast bacilli sputum
smear-positive samples or positive cultures. Xpert MTB/
RIF (Cepheid, Sunnyvale, CA, USA) was endorsed by WHO
in 2010 for the rapid diagnosis (i.e. within 2 hours) of pul-
monary TB and rifampicin-resistance in adults.2 In July
2013, the Xpert MTB/RIF assay remained the only fully
automated real-time DNA-based cartridge test that can
detect both TB and resistance to rifampicin, and the only
mature technology representing a new generation of auto-
mated molecular diagnostic platforms.
Since 2010, almost 100 articles on Xpert MTB/RIF have
been published and others are underway.3 In 2013, given
the amount of additional data, WHO commissioned three
systematic reviews of the evidence on sensitivity and
specicity of Xpert MTB/RIF as a test for pulmonary and
extrapulmonary TB, in both adults and children. Findings
were reviewed by an expert group and updated recommen-
dations are anticipated in 2014 (see Chapter 5).
e UNITAID 2013 Report: Tuberculosis: Diagnostic tech-
nology and market landscape4 describes the following four
innovations to the Xpert MTB/RIF technology, which were
made or under development in 2012 and 2013.
Assay improvements. A new prototype assay for MDR-
TB is in development. is uses new dyes and quenchers
that increase the spectral range for detection of targets
using 10 uorophores rather than the six currently used.
Remote calibration. is was made available in late
2012 and is already being used in more than 40 coun-
tries. It allows users to recalibrate the optical system,
verify the functioning of the thermal system and con-
duct a series of system-level tests to ensure full system
functionality within specications. It is anticipated that
over 90% of modules can be successfully calibrated over
the internet.
Enhancements to data management. Real-time
aggregation of geo-positioned test data (from which per-
sonal identiers have been removed) is being evaluated
in South Africa. is oers the potential to substantially
improve monitoring of the TB epidemic and the associ-
ated programmatic response.
HIV cartridges for use with the GeneXpert plat-
form. ese are planned for release in 2014. A separate
cartridge for the qualitative and quantitative detection
of HIV viral load is in development.
With over 50 companies working on TB diagnostics, there
is now considerable industry interest in TB diagnostics.
Nonetheless, a recent survey of more than 25 test devel-
opers identied several critical frequently-asked questions
for which answers are required by industry to invest in TB
diagnostic test development (www.tbfaqs.org). Test devel-
opers are particularly interested in identifying the most
important attributes on which to focus test development
eorts (examples include cost, sensitivity, specicity,
infrastructure requirements, time to result, throughput,
sputum versus other samples, manual versus automated,
point-of-care versus centralized laboratory testing, inte-
grated or reex drug resistance test and which drugs are
critical for DST). In addition, updated market analyses
are urgently needed, given that the TB diagnostics market
landscape has changed signicantly since the last global
assessment of the TB diagnostics market in 2006.5 Updat-
ed market analyses and development of target product
1 Molecular Line Probe Assay for rapid screening of patients at risk
of MDR-TB. Policy Statement. Geneva, World Health Organization,
2008. Available at http://www.who.int/tb/features_archive/policy_
statement.pdf
2 Policy Statement: Automated real-time Nucleic Acid Amplication
Technology for Rapid and Simultaneous Detection of Tuberculosis and
Rifampicin Resistance: Xpert MTB/RIF System. Geneva: World Health
Organization, 2011 (WHO/HTM/TB/2011.4). Available at http://
whqlibdoc.who.int/publications/2011/9789241501545_eng.pdf
3 Weyer K et al. Rapid molecular TB diagnosis: evidence, policy-mak-
ing and global implementation of Xpert® MTB/RIF European
Respiratory Journal erj01572-2012; published ahead of print 2012,
doi:10.1183/09031936.00157212.
4 Tuberculosis: Diagnostics Technology and Market Landscape 2013.
Geneva, UNITAID/World Health Organization, 2013. Available
at: http://www.unitaid.eu/images/marketdynamics/publications/
TB-Dx-Landscape_1-Jul-2013.pdf
5 Diagnostics for tuberculosis. Global Demand and market potential.
Geneva, Special Programme for Research and Training in Tropical
Diseases (TDR) and Foundation for Innovative New Diagnostics
(FIND), 2006. Available at: http://www.who.int/tdr/publications/
documents/tbdi.pdf

90 GLOBAL TUBERCULOSIS REPORT 2013
proles could facilitate greater engagement of test develop-
ers in TB diagnostics.
Despite good progress with the pipeline for new diagnos-
tics, much more eort and investment are needed by both
donors and manufacturers to expedite evaluations of new
technologies in dierent epidemiological settings in order
to determine their diagnostic accuracy and robustness in
the settings of intended use. Substantial additional fund-
ing and innovation for new TB diagnostic development
remain essential to ensure the availability of tests that are
reliable, easy to use, aordable, and accessible to all those
with TB. More than ever, the TB diagnostic pipeline needs
increased and sustained investment.
8.2 New drugs to treat and prevent TB
e anti-TB drugs currently used in rst-line treatments
are around 50 years old. e regimen that is currently rec-
ommended by WHO for new cases of drug-susceptible TB is
h i g h ly e c a c io us , w it h c u re ra te s o f a r ou n d 9 0 % i n H I V- ne g -
ative patients. Nonetheless, it requires six months of treat-
ment with rst-line drugs (a combination of rifampicin,
isoniazid, ethambutol and pyrazinamide for two months,
followed by a four-month continuation phase of rifampicin
and isoniazid). Regimens for treatment of MDR-TB cur-
rently recommended by WHO entail at least 20 months of
treatment with second-line drugs for most patients, and
are associated with multiple (and sometimes serious) side-
eects and lower cure rates (see Chapter 4). ere are also
interactions between anti-TB treatments and antiretro-
viral therapy (ART) for people living with HIV. New drugs
are required to shorten and simplify treatment, to improve
the ecacy and tolerability of treatment for MDR-TB and
to improve the treatment of TB among people living with
HIV.
e status of the pipeline for new anti-TB drugs in July
2013 is shown in Figure 8.2. ere are seven drugs in Phase
II (early bactericidal activity, EBA, and eight-week culture
conversion) trials and four drugs in Phase III (ecacy) tri-
als. In total, there are 10 new or repurposed drugs in Phase
II or Phase III trials; one drug (rifapentine, a rifamycin that
has a longer half-life than rifampicin) is in both Phase II
and Phase III trials, for dierent indications.
8.2.1 Phase III trials
Results from a Phase III trial (the ‘Rifaquin trial’) that eval-
uated the safety and ecacy of two regimens for patients
with drug-susceptible TB, in which moxioxacin was sub-
stituted for isoniazid in the intensive phase of treatment
and rifapentine was used in the continuation phase of
treatment, were presented in March 2013.1 A total of 827
patients with drug-susceptible TB were enrolled in Botswa-
na, South Africa, Zambia and Zimbabwe. Both new reg-
imens were well tolerated. e six-month regimen with a
weekly dose of rifapentine (1200 mg) and moxioxacin in
the continuation phase was not inferior to the currently
recommended regimen. However, the four-month regimen
with twice-weekly doses of rifapentine (900 mg) and moxi-
oxacin in the continuation phase was clearly inferior to
the currently recommended regimen.
Two Phase III trials are evaluating four-month combina-
tion regimens in which a uoroquinolone (gatioxacin in the
case of the OFLOTUB trial and moxioxacin in the case of
the ReMOX trial) is substituted for either ethambutol (in the
FIGURE 8.2
The development pipeline for new TB drugs, July 2013a
Discovery Preclinical development Clinical development
Lead
optimization Preclinical
development
Good
Laboratory
Practice
toxicity
Phase I Phase II Phase III
Cyclopeptides
Diarylquinoline
DprE Inhibitors
InhA Inhibitor
LeuRS Inhibitor
Macrolides
Mycobacterial Gyrase
Inhibitors
Pyrazinamide Analogs
Riminophenazines
Ruthenium (II)
complexes
Spectinamides
Translocase-1
Inhibitors
CPZEN-45
DC-159a
Q203
SQ609
SQ641
TBI-166
#<&
Bedaquiline
(TMC-207)
.KPG\QNKF
Novel Regimensb
2#
4KHCRGPVKPG
SQ-109
5WVG\QNKF207
Delamanid
12%
Gatifloxacin
Moxifloxacin
4KHCRGPVKPG
Chemical classes: àuoroSuinolone, rifamycin, oxazolidinone, nitroimidazole, diarylSuinoline, benzothiazinone
a &GVCKNUHQTRTQLGEVUNKUVGFECPDGHQWPFCVYYYPGYVDFTWIUQTIRKRGNKPGCPFQPIQKPIRTQLGEVUHQTYJKEJCNGCFEQORQWPFJCUPQVDGGPKFGPVKßGFECPDGXKGYGFCV
www.newtbdrugs.org/pipeline-discovery.
b %QODKPCVKQPTGIKOGPU0%,/2C<2JCUG++C0%/2C<2JCUG++D0%%,2C<2JCUG++C2CP#%'#/#/56$*4<'3/2JCUG++D
2$6<
TBA-354
1 Jindani A et al. 2013. A Multicentre Randomized Clinical Trial to Eval-
uate High-dose Rifapentine with a Quinolone for Treatment of Pulmo-
nary TB: e RIFAQUIN Trial. Oral abstract and paper 147LB. 20th
Conference on Retroviruses and Opportunistic Infections (CROI),
March 3–6 2013, Atlanta.

91GLOBAL TUBERCULOSIS REPORT 2013
OFLOTUB trial) or ethambutol or isoniazid (in the ReMOX
trial). e results from both trials are expected in late 2013.
A new compound, delamanid (OPC-67683), is currently
being tested in a Phase III trial as an adjunct to existing
optimized regimens for treatment of MDR-TB.
8.2.2 Phase II trials – individual compounds
e safety, tolerability and antimicrobial activity of an
increased daily dose of rifapentine (at 10, 15 and 20 mg/
kg) in combination with isoniazid, pyrazinamide and
ethambutol during the rst two months of treatment are
being investigated in a Phase IIb trial (TBTC trial 29X).
Early results were reported in May 2013.1 ese showed
that rifapentine-based regimens were well-tolerated, with
no toxicity events specically related to increasing doses
of the drug. Compared with the currently recommended
six-month regimen, a higher proportion of patients who
received the regimens including rifapentine had convert-
ed to culture-negative status (both in solid and liquid
BOX 8.2
WHO interim guidance on the use of bedaquiline to treat MDR-TB
1 Moro et al. Tolerability and safety of escalating Rifapentine (RPT) dos-
es during the rst two months of tuberculosis (TB) treatment. Abstract
A6051. American oracic Society International Conference, Phila-
delphia, May 17–22, 2013
9*1GUVKOCVGUVJCVCDQWVaPGYECUGUQH/&46$QEEWT
worldwide each year (Chapter 2). Current drug regimens
TGEQOOGPFGFD[9*1HQTVTGCVOGPVQH/&46$RTGUGPVOCP[
EJCNNGPIGUVTGCVOGPVNCUVUOQPVJUQTOQTGCPFTGSWKTGU
daily dosages of drugs that are more toxic, less effective, and
far more expensive than those used to treat drug-susceptible
TB.a)NQDCNN[QPN[CDQWVQHRCVKGPVUYJQUVCTV/&46$
therapy are treated successfully (Chapter 4).
(QTVJGßTUVVKOGKPQXGT[GCTUCPGY6$FTWIYKVJCPQXGN
mechanism of action – bedaquiline – has become available
for use. It was approved by the US FDA in December 2012,
following an accelerated approval process. There is considerable
KPVGTGUVKPVJGRQVGPVKCNQHVJKUFTWIVQVTGCV/&46$*QYGXGT
information remains limited, since it has only been evaluated
KPVYQ2JCUG++DVTKCNUHQTUCHGV[CPFGHßECE[(QTVJGUGTGCUQPU
WHO has issued “interim policy guidance”.b
This interim guidance provides advice on the inclusion of
DGFCSWKNKPGKPEQODKPCVKQPVJGTCR[HQT/&46$KPCEEQTFCPEG
with the existing WHO guidelines for the programmatic
management of drug-resistant TB.a The interim guidance lists
ßXGEQPFKVKQPUVJCVOWUVDGHWNßNNGFHQTDGFCSWKNKPGVQDGWUGF
VQVTGCVCFWNVUYKVJ/&46$
1. Effective treatment and monitoring. Treatment must be
closely monitored for effectiveness and safety, using sound
treatment and management protocols approved by relevant
national authorities.
2. Proper patient inclusion. Special caution is required
when bedaquiline is used in people aged 65 and over, and
in adults living with HIV. Its use among pregnant women
and children is not advised.
3. Informed consent. Patients must be fully aware of the
RQVGPVKCNDGPGßVUCPFTKUMUQHVJGPGYFTWICPFIKXG
documented informed consent before embarking on
treatment.
4. Adherence to WHO recommendations. All principles on
YJKEJ9*1TGEQOOGPFGF/&46$VTGCVOGPVTGIKOGPUCTG
based must be followed. In particular, four effective second-
line drugs must be part of the regimen. In line with the
general principles of TB treatment, bedaquiline should not
be introduced into a regimen in which the companion drugs
are failing to show effectiveness.
5. Active pharmacovigilance and management of
adverse events. Active pharmacovigilance measures
must be in place to ensure early detection and proper
management of adverse drug reactions and potential
interactions with other drugs.
WHO strongly recommends the acceleration of Phase III trials
to generate more comprehensive evidence that can inform
HWVWTGRQNKE[IWKFCPEGQPDGFCSWKNKPG6JGQTICPK\CVKQPYKNN
review, revise or update the interim guidance as additional
KPHQTOCVKQPQPGHßECE[CPFUCHGV[DGEQOGUCXCKNCDNG9*1
is also developing a document that will provide operational
guidance on the implementation of bedaquiline and is working
with partners to help ensure rational introduction of the drug.
a Guidelines for the proIrammatic manaIement of druI-resistant tuberculosis
– 2011 update)GPGXC9QTNF*GCNVJ1TICPK\CVKQP9*1*6/6$
2011.6).
b The use of bedaSuiline in the treatment of multidruI-resistant tuberculosis
interim policy Iuidance9QTNF*GCNVJ1TICPK\CVKQP)GPGXC5YKV\GTNCPF
2013 (WHO/HTM/TB/2013.6).
medium) after eight weeks. Among patients receiving the
regimen with the highest dose of rifapentine, 100% were
culture-negative after eight weeks of treatment (compared
with 16 weeks for those receiving the current standard of
care). e trial investigators concluded that the robust anti-
microbial activity alongside the good tolerability and safety
of the compound at increasing doses justied the assess-
ment of daily high-dose rifapentine in regimens of shorter
than six months duration in a Phase III trial.
Among other drugs tested in Phase II trials, the high-
est-prole in the past year is bedaquiline (TMC-207). e
US Food and Drug Administration (FDA) approved the use
of bedaquiline as an adjunct to existing regimens for the
treatment of MDR-TB in December 2012, under an accel-
erated procedure. Bedaquiline became the rst new TB
drug to be approved for use in 40 years. A Phase III trial,
which will investigate the safety and ecacy of bedaquiline
when used in combination with a short MDR-TB regimen,
is scheduled to start before the end of 2013. Following the
release of trial results and the FDA decision, WHO issued
interim guidance about the use of bedaquiline in the treat-
ment of MDR-TB in June 2013 (Box 8.2).
Five other individual compounds are in the Phase II

92 GLOBAL TUBERCULOSIS REPORT 2013
development phase. ese are linezolid, sutezolid, PA-824,
SQ-109 and AZD-5847.
Linezolid (a member of the oxazolidinone antibiotic
class of drugs) was approved in 2000 for the treatment of
drug-resistant, gram-positive bacterial infections. It has
good anti-mycobacterial activity in vitro and is increasingly
used ‘o-label’ for patients who have highly drug-resistant
TB. However, serious adverse events (such as peripheral
and optic neuropathies, anaemia and thrombocytopenia)
have been reported. Results from a prospective, random-
ized trial in which linezolid was used to treat patients with
XDR-TB who had not responded to other available chemo-
therapeutic options were published in late 2012.1 A total
of 41 patients were randomly assigned to linezolid therapy
(600 mg per day), which was either started immediately or
after two months without any change to the background
regimen. After conrmed sputum-smear conversion or
after four months of treatment (whichever came rst),
patients underwent a second randomization to continue
linezolid therapy at a dose of either 600 mg or 300 mg per
day for at least an additional 18 months, with close moni-
toring of toxicity.
e results showed that at four months, 15 of the 19
patients (79%) in the immediate-start group and 7 of the
20 (35%) in the delayed-start group had converted to cul-
ture-negative status (p = 0.001). Most patients (34 of 39
[87%]) had a negative sputum culture within six months
after linezolid had been added to their drug regimen. Of the
38 patients treated with linezolid, 31 (82%) had clinically
signicant adverse events that were possibly or probably
related to linezolid, including three patients who discontin-
ued therapy. Patients who received 300 mg per day after the
second randomization had fewer adverse events than those
who continued taking 600 mg per day. irteen patients
completed therapy and had not relapsed at the end of fol-
low up. Four cases of acquired resistance to linezolid were
observed. Trial investigators concluded that linezolid was
eective at achieving culture conversion among patients
with chronic XDR pulmonary TB, but warned that patients
must be monitored carefully for adverse events. Study lim-
itations include the small number of patients evaluated,
and that 10% of patients acquired resistance to linezolid.
Further data are needed to balance the long-term risks and
benets of linezolid when used as part of a combination
regimen with other eective anti-TB drugs.
Sutezolid (PNU-100480) is an oxazolidinone and an
analogue of linezolid. It has been tested in an EBA study at
doses of either 600 mg twice a day or 1200 mg once a day.
Results were presented in 2012 and showed that sutezolid
led to a signicant reduction in log colony forming units
(CFU) counts compared with the baseline level following 14
days of treatment, using both dosage options.2 e results
suggested a superior response with the 600 mg twice-daily
dose.
PA-824 is a nitroimidazole compound that is being test-
ed as part of several potential combination regimens (see
below).
SQ-109, originally synthesized as a derivative of etham-
butol, is also being tested as part of a combination regimen
(see below).
AZD-5847 is being tested in a Phase II trial.
8.2.3 Phase II trials – new regimens
Besides individual compounds, new combinations of drugs
are or will soon be tested in various Phase II trials. In the
Global tuberculosis report 2012, the results of the EBA study
of a new combination regimen (NC-001) that included moxi-
oxacin, pyrazinamide and the novel drug PA-824 were
summarized.3 ree trials of various combination regimens
are currently underway. e rst of these is NC-002, which
is building on the NC-001 study to test the same regimen in
a two-month trial. e trial is being implemented in South
Africa and the United Republic of Tanzania. e regimen
is being tested in patients with drug-susceptible TB and in
patients who have drug-resistant TB but not resistance to
the drugs included in the new regimen. e NC-002 trial
is a landmark trial, since it is the rst to simultaneously
investigate treatment of both drug-sensitive and drug-
resistant TB with the same regimen. Results are expected
at the end of 2013.4
e second trial, NC-003, is testing the EBA of various
combinations of clofazimine, bedaquiline, PA-824 and pyr-
azinamide in patients with drug-susceptible TB.5
e MAMS-TB-01 trial, conducted by the PanACEA
consortium, is evaluating new three-month combina-
tion regimens using a new adaptive study design.6 e
drugs included in the combination regimens are isoniazid,
rifampicin, pyrazinamide, ethambutol, moxioxacin and
SQ-109. e end-point of the trial is time to culture conver-
sion in liquid media. e trial started in May 2013.7
8.2.4 New developments in the treatment of latent
TB infection
New drugs are being tested for the treatment of latent TB
infection (LTBI) in people without active TB disease.
Rifapentine has been investigated as part of a combined
regimen (TBTC 26, also called PREVENT-TB), and the rst
results were published in December 2011.8 Enrolment and
1 Lee M et al. Linezolid for Treatment of Chronic Extensively
Drug-Resistant Tuberculosis. New England Journal of Medicine
2012;367:1508-18. DOI: 10.1056/NEJMoa1201964
2 Wallis R et al. Safety, tolerability and early bactericidal activity in spu-
tum of PNU-100480 (sutezolid) in patients with pulmonary tuberculo-
sis (Abstract THLBB02). 19th International AIDS Conference 2012,
July 22–27, Washington DC.
3 Diacon A H et a l. 14-day bactericida l activity of PA- 824, bedaquil ine,
pyrazinamide and moxioxacin combinations: a randomised trial.
e Lancet, 2012
4 See: http://clinicaltrials.gov/show/NCT01498419
5 See: http://clinicaltrials.gov/show/NCT01691534
6 Phillips P et al. Innovative trial designs are practical solutions for
improving the treatment of tuberculosis. Journal of Infectious Dis-
eases. 2012;205 Suppl 2:S250–7.
7 See: http://clinicaltrials.gov/show/NCT01785186
8 Sterling T et al. ree Months of Rifapentine and Isoniazid for
Latent Tuberculosis Infection. New England Journal of Medicine
2011; 365;23: 2155–66.

93GLOBAL TUBERCULOSIS REPORT 2013
follow-up for two groups of particular interest (young chil-
dren 2–11 years of age, and people living with HIV) were
extended and are scheduled to end in September 2013.Pre-
liminary results showed that the once-weekly, three month
regimen of rifapentine and isoniazid (3HP) was generally
well-tolerated and oered ‘substantial advantages’ com-
pared with the current standard of nine months of isoniazid
for treatment of LTBI in children.1Study 33, also called iAd-
here, is a follow-up Phase IV study of TBTC 26, investigating
the eectiveness of the 3HP combination (tested in PRE-
VENT-TB), either given by: (1) DOT, (2) self-administered,
or (3) self-administered with text message reminders by cell
phone.is study is expected to be completed in March 2014.
A second study is an AIDS Clinical Trials Group (ACTG)
trial of daily rifapentine and isoniazid for one month to
treat LTBI in people living with HIV. A third study to eval-
uate the eect of single and repeated administration of
rifapentine (given as a daily or weekly regimen) on steady-
state pharmacokinetic parameters of efavirenz, emtric-
itabine and tenofovir given as a xed dose combination
BOX 8.3
Raising the proßle of treatment for latent TB infection
One third of the world’s population is
estimated to be latently infected with
M. tuberculosis. People with latent TB
infection (LTBI) do not have symptoms
of TB and are not infectious, but they
are at risk of developing active disease
and becoming infectious. Studies show
that 5–20% of those infected will
develop active TB at some point in their
lifetime, with the majority developing
TB disease within 2–5 years of the initial
infection. Several factors increase the
risk of progressing from infection to
CEVKXG6$FKUGCUGKOOWPQUWRRTGUUKQP
(for example, related to HIV infection
or immunosuppressive treatment),
malnutrition, diabetes and alcohol
abuse. Preventing active TB by
addressing these risk factors as well as
proper diagnosis and treatment of LTBI
in selected risk groups is thus important
for the individual and public health.
Modelling has shown that diagnosis
and treatment of LTBI could play a
key role in TB elimination. WHO has
recently published guidelines on TB
contact investigation and on systematic
screening of active TB,a,b both of which
QHHGTCPGPVT[RQKPVVQKFGPVKßECVKQP
of risk groups for LTBI diagnosis and
treatment.
+UQPKC\KFRTGXGPVKXGVJGTCR[
(IPT) is the mainstay of current WHO
recommendations on treatment of
LTBI. Treatment is recommended for
VYQURGEKßERQRWNCVKQPITQWRURGQRNG
living with HIV, and children less than
ßXG[GCTUQNFYJQCTGJQWUGJQNFQT
close contacts of TB patients. A recent
Cochrane review showed that rifampicin-
and rifapentine-containing regimens
among HIV negative people have higher
completion rate and fewer adverse events
compared with those based on IPT only.c
Before initiating LTBI treatment, it
is essential that active TB is effectively
ruled out and the diagnosis of LBTI
reliably established. The tuberculin
skin test (TST) and interferon-gamma
TGNGCUGCUUC[U+)4#CTGFGUKIPGFVQ
detect a cellular immune response to
M. tuberculosis, but do not differentiate
between latent infection and active
disease and, if negative, do not allow
TB infection to be ruled out. Most
importantly, they cannot accurately
predict the risk of infected individuals
developing active TB disease, and their
use in routine practice poses operational
and resource challenges.
There are several unanswered
questions related to the detection
and management of LTBI that require
WTIGPVUEKGPVKßECVVGPVKQPCPF
increased research investments. There
is still limited understanding of the
fundamental biology of latency and
there are no truly adequate animal
models to study it. There is also no
diagnosis and treatment for people
who are latently infected with drug-
resistant strains of M. tuberculosis.
Expediting the discovery of robust
tools to effectively diagnose and treat
LTBI is crucially important for global
TB control. Particular emphasis needs
to be given to development of a better
understanding of the basic pathogenesis
of M. tuberculosisCPFVJGKFGPVKßECVKQP
of biomarkers that will enable reliable
diagnosis and shorter and less toxic
treatment for LTBI.
Following recent developments in the
treatment of LTBI, WHO plans to update
its guidelines on the management of
LTBI. This will entail a review of the
existing evidence with a particular focus
on risk groups that have the highest
likelihood of progression to active TB
disease following infection, and due
EQPUKFGTCVKQPVQTKUMDGPGßVCPCN[UKU
and concomitant risk factors.
a 4GEQOOGPFCVKQPUHQTKPXGUVKICVKPIEQPVCEVU
of persons with infectious tuberculosis
in low- and middle-income countries.
Available at JVVRCRRUYJQKPVKTKU
DKVUVTGCOA
eng.pdf
b Systematic screening for active tuberculosis
– Principles and recommendations. Available
at JVVRCRRUYJQKPVKTKUDKVUVTGCO
AGPIRFH
c 5JCTOC5-GVCN4KHCO[EKPUTKHCORKEKP
TKHCDWVKPCPFTKHCRGPVKPGEQORCTGFVQKUQPKC\KF
for preventing tuberculosis in HIV-negative
people at risk of active TB. Cochrane &atabase
of Systematic 4eXiews+UUWG#TV0Q
%&&1+%&
pub2.
(ATRIPLA™) started patient enrolment in September 2012
and recruitment was completed in August 2013. A fourth
study to compare the safety and eectiveness of a four-week
daily regimen of rifapentine and isoniazid with a standard
nine-month regimen of daily isoniazid among people liv-
ing with HIV started patient enrolment in May 2012 and
is expected to complete enrolment in March 2018. A fth
study to determine the safety and tolerability of a four-
month, once daily rifampicin regimen in children is being
conducted by the Canadian Institutes of Health Research
and McGill University and results are expected in 2016.2
1 Villarino et al. Tolerability among children of three months of
once-weekly rifapentine + INH (3HP) vs. 9 months of daily INH (9H)
for treatment of latent tuberculosis infection: e PREVENT TB Study
(TBTC Study 26/ACTG 5259). IDSA Conference 2012.
2 Clayden P et al, on behalf of the HIV i-Base/Treatment Action
Group. 2013 Pipeline Report: HIV, Hepatitis C Virus (HCV), and Tuber-
culosis (TB) Drugs, Diagnostics, Vaccines, Preventive Technologies,
Research Toward a Cure, and Immune-Based and Gene erapies in
Development. Available at: http://www.treatmentactiongroup.org/
pipeline-report

94 GLOBAL TUBERCULOSIS REPORT 2013
8.3 New vaccines to prevent TB
e slow decline in TB incidence globally and the growing
problem of MDR-TB highlight the critical need for new
eective TB vaccines. e BCG vaccine for the prevention
of TB is almost 100 years old, and while the vaccine pro-
tects against severe forms of TB in children (TB meningitis
and miliary TB), its ecacy in preventing pulmonary TB
in adults is highly variable. BCG is also not recommended
for use in infants known to be infected with HIV, due to
the risk of disseminated BCG disease. e development of
techniques for genetic manipulation of mycobacteria, com-
pletion of the genome sequence of M. tuberculosis in the
1990s, and recent advances in immunology provide historic
opportunities for developing a new generation of TB vac-
cines that can achieve dramatically higher levels of impact.
For the past decade, two major strategies have been used
to develop new vaccines for prevention of TB.1 One strategy
has been to develop vaccines that would have a higher e-
cacy than BCG and replace it – such as an improved version
of BCG or a new attenuated live M. tuberculosis vaccine. e
second strategy has been a ‘prime-boost’ strategy in which
BCG continues to be given to neonates (as is done current-
ly, since it prevents TB in infants and children), and a new
vaccine is given as a ‘booster’ dose to increase the ecacy
and extend the duration of protection. Modelling of the
potential public health impact of new TB vaccines in the
WHO South-East Asia Region suggested that a TB vaccine
for infants with 60% ecacy would contribute to a signif-
icant decline in TB incidence by 2050.2 Furthermore, this
modelling also indicated that if a preventive TB vaccine of
similar ecacy among adolescents and adults was deliv-
ered as part of a mass vaccination strategy, the impact on
the TB epidemic would be much larger. More recent mod-
elling of the public health impact of a new vaccine at global
level3 has reinforced this nding, indicating that an adoles-
cent and adult vaccine with 60% ecacy could potentially
avert 30‒50 million new TB cases over a 25 year period.
e much greater potential impact of an adult/adolescent
vaccine has shifted the focus of TB vaccine development
towards a new paradigm that emphasizes the development
of a diverse pipeline of new TB vaccine candidates that tar-
get the prevention of TB in this older population. Scientic
advances have also enabled the pursuit of more sophisticat-
ed approaches to vaccine design, and the global pipeline of
TB vaccine candidates in clinical trials is more robust than
at any previous period in history, now including recombi-
nant BCGs, attenuated M. tuberculosis strains, recombinant
viral-vectored platforms, protein/adjuvants combinations,
and mycobacterial extracts.
e status of the pipeline for new vaccines in July 2013
is shown in Figure 8.3. ere are 12 vaccine candidates in
clinical trials. Most are designed for prevention of TB, either
to prevent infection (pre-exposure) or to prevent primary
progression to disease or reactivation of latent TB (post-
exposure). Two are BCG replacement vaccines and two are
proposed as immunotherapeutic agents, to improve respon-
siveness to chemotherapy or prevent relapse or re-infection.
Two vaccines are in Phase IIb studies.
MVA85A is an attenuated vaccinia-vectored vaccine
candidate expressing Ag85A of M. tuberculosis. It was
designed as a booster vaccine for BCG vaccinated infants
and the rst Phase IIb trial of this vaccine was conducted
in South Africa from 2009 to 2012 with results published
in early 2013 (Box 8.4).4 An additional Phase IIb trial of
MVA85A is currently being conducted in adults living with
HIV in Senegal and South Africa; the trial has been recently
re-designed as a safety trial in which up to 650 participants
will be enrolled.
M72+AS01E is a protein subunit vaccine, formulated
in a novel adjuvant to enhance immunogenicity. It con-
tains a fusion protein of the M. tuberculosis antigens 32A
and 39A in the adjuvant AS01E. Safety and immunogenic-
ity are being tested in three dierent populations: infants
in e Gambia, people living with HIV in India and adults
with TB disease in China (Province of Taiwan) and Esto-
nia. e Phase IIb study will be the largest trial of a novel
TB vaccine in adults, aiming to enrol 4500 HIV-negative
adults in TB-endemic countries in Africa. e primary end-
point will be the protective ecacy of two doses of M72+
AS01E against pulmonary TB disease. Secondary endpoints
include safety and immunogenicity.
ere are six additional vaccines in Phase II trials.
AERAS-402/Crucell Ad35 is an adenovirus-vectored
vaccine candidate expressing three M. tuberculosis antigens:
Ag85A, Ag85B and TB10.4. It is designed as a booster vac-
cine for infants, adolescents and adults. Although started
as a Phase IIb proof-of-concept trial, based on preliminary
data the trial is now being revised to be a smaller Phase
II trial with safety and immunogenicity as primary end-
points. Of note, AERAS-402/Crucell Ad35 and MVA85A
are also being tested in combination, to try to drive a bal-
anced CD4+/CD8+ immune response. One or two doses of
AERAS-402/Crucell Ad35 followed by a dose of MVA85A
are being evaluated in a combined Phase I/Phase II trial
in adults in the United Kingdom for safety and immuno-
genicity.
ree vaccines are protein subunit adjuvanted vac-
cines, initially developed by the Statens Serum Institute in
Copenhagen, Denmark. Hybrid 1 + IC31 contains Ag85B
and ESAT-6 in an adjuvant, IC31. Hybrid 56 + IC31 con-
tains antigens 85B and ESAT6 as well as AgRv2660, which
is expressed during latency. Hybrid 4 + IC31, now being
developed with Sano Pasteur, is a fusion protein candi-
1 Evans TG, Brennan MJ, Barker L and ole J. Preventive vaccines
for tuberculosis. Vaccine. 31S (2013) B223– B226.
2 Abu-Raddad LJ, et al. Epidemiological benets of more-eective
tuberculosis vaccines, drugs, and diagnostics. Proceedings of the
National Academy of Science. 2009. 106:33; 13980–13985.
3 A model of the global public health impact of new TB vaccines was
commissioned by Aeras and developed by Applied Strategies. For-
mal publication of the model and associated results is pending.
4 Tameris MD, et al. Safety and ecacy of MVA85A, a new tuber-
culosis vaccine, in infants previously vaccinated with BCG: a
randomised, placebo-controlled phase 2b trial. e Lancet. 2013.
381:9871; 1021–1028.

95GLOBAL TUBERCULOSIS REPORT 2013
date that expresses Ag85B and TB10.4; the latter antigen is
from the same gene family as ESAT-6. All three vaccines are
being studied in Phase IIa clinical trials in Africa.
VPM 1002 is a live recombinant vaccine, derived from
the Prague strain of BCG into which the listerolysin gene
from Listeria monocytogenes has been cloned and the urease
gene deleted to improve immunogenicity. A Phase IIa trial
of this vaccine has recently been completed in South Africa.
A second Phase II trial will assess the safety and immunoge-
nicity of the vaccine in HIV exposed/unexposed newborns.
RUTI is a non-live vaccine based on fragmented M. tuber-
culosis bacteria. It is in a Phase IIa trial in Spain and is being
developed as an immunotherapeutic vaccine.
In addition to the vaccine candidates described above,
AnHui Longcom, a Chinese pharmaceutical company, is
studying Mycobacterium vaccae, a non-living preparation
from the non-pathogenic bacterium, as an adjunct to stan-
dard antimicrobial therapy. Phase III ecacy studies are
reportedly underway.
ere are three vaccine candidates in Phase I clinical
trials. ese include the rst live attenuated M. tuberculosis
vaccine, MTBVAC, as well as a new fusion protein vaccine,
ID93, formulated with a novel adjuvant GLA-SE.
MTBVAC is being developed by the University of Zara-
gosa, Institut Pasteur, BIOFABRI and the Tuberculosis
Vaccine Initiative (TBVI). It is a live M. tuberculosis strain
attenuated via deletions of the phoP and fadD26 genes. It
is the rst live attenuated M. tuberculosis vaccine to enter a
Phase I clinical trial.
ID93+GLA-SE is a recombinant fusion protein formu-
lated in the novel adjuvant, GLA-SE. It is being developed
by the Infectious Disease Research Institute (IDRI) in col-
laboration with Aeras. It expresses three M. tuberculosis
virulence antigens (Rv2608, Rv3619 and Rv3620) and one
M. tuberculosis latency antigen (Rv1813). It is beginning a
Phase 1b trial in adults in South Africa to assess safety and
immunogenicity in this population.
Ad5 Ag85A is an adenovirus serotype 5 vector express-
ing Ag85A. It has been developed by McMaster University
with support from CanSino, a Chinese biotechnology com-
pany based in Tianjin. e vaccine was recently evaluated in
a Phase I trial that demonstrated no vaccine-related serious
adverse events and showed greater immunogenicity in the
study group primed with BCG.
Research on new TB vaccines is now at a crucial juncture.
Despite the diversity that already exists in the global portfo-
lio of TB vaccine candidates in clinical trials, there is grow-
ing recognition among scientists and researchers in the eld
that there is still too much similarity in the immunological
strategies being pursued.1 In the absence of known immune
correlates for either protective immunity against TB or con-
trol of infection, the portfolio must be further diversied so
that candidates explore a dierent and novel immunological
‘space’. ere is already a robust pipeline of candidates being
evaluated pre-clinically – including nucleic acid-based (DNA
and RNA) vaccines – and these pursuits may help to broaden
the diversity of the clinical portfolio and ll the scientic
gaps that currently exist. To rationalize and streamline the
advancement of TB vaccine candidates, consensus has been
Phase I Phase II Phase III
Phase IIb
VPM 1002
Max Planck, VPM, TBVI,
Serum Institute
P B
H1+IC31
SSI, TBVI, EDCTP,
Intercell
P B PI
476+
Archivel Farma, S.L.
B PI it
*#'4#5+%
SSI, Aeras, Intercell
P B PI
*#'4#5+%
SSI, Sanofi Pasteur,
Aeras, Intercell
B
%TWEGNN#F#'4#5
Crucell, Aeras
B
P Prime B Boost PI Post-infection it Immunotherapy
#F#I#
McMaster, CanSino
P B PI
MTBVAC
TBVI, Zaragoza, Biofabri
P
ID93+GLA-SE
Infectious Disease
Research Institute (IDRI),
Aeras
B
%TWEGNN#F/8##
Crucell, Oxford, Aeras
P B
/8###'4#5
Oxford, Aeras, EDCTP
B PI it
M72+AS01
GSK, Aeras
B PI
M. Vaccae
Anhui Longcom
it
6$8CEEKPG6[RGU 8KTCNXGEVQTGF/8###'4#5#F#I#
2TQVGKPCFLWXCPV/*[DTKF*[XCE*+&
T$%)82/
-KNNGF9%QT'ZVTCEV/Y476+
5QWTEG6WDGTEWNQUKU8CEEKPG%CPFKFCVGU9QTMKPI)TQWRQP0GY8CEEKPGU
FIGURE 8.3
The development pipeline for new TB vaccines, July 2013
1 Evans TG et al. Preventive vaccines for tuberculosis. Vaccine 31S
(2013) B223– B226.

96 GLOBAL TUBERCULOSIS REPORT 2013
developed among key stakeholders on ‘stage-gating’ crite-
ria for new TB vaccines, and increased emphasis is being
placed on global coordination among key stakeholders to
advance a common research agenda.
To supplement these existing eorts, a re-prioritized
focus on early stage research is also underway. In accor-
dance with this shift in emphasis, more energy and resourc-
es will be directed towards the pursuit of novel designs, to
studies focused on immunological mechanisms and bio-
markers, and to a diversication of scientic approaches
and strategies to ensure that a more diverse pipeline of new
TB vaccine candidates moves forward into clinical trials.1
8.4 The post-2015 global TB strategy: the
critical role of research and development
Fundamental science is necessary to drive innovations in
new tools for improved TB care and control. Fundamental
research is required to better characterize M. tuberculosis
and to improve understanding of the interaction between
the bacillus and the human host, as a basis for maintain-
ing the ow of new technologies into the product pipeline.
Researchers are making great strides in redening the
1 Brennan MJ and ole J (editors). Tuberculosis vaccines: A stra-
tegic blueprint for the next decade. Tuberculosis. 2012. 92: Supple-
ment 1; S6–S13.
BOX 8.4
The MVA85A trial in South Africa
/8##KUCRQZXKTWUÄ/QFKßGF
Vaccinia Ankara”, MVA)-vectored vaccine
that expresses the immune-dominant
M. tuberculosisCPVKIGP#+VYCU
originally developed at the University
of Oxford. An infant Phase IIb proof-
of-concept trial was recently completed
in South Africa.a The study population
consisted of 2794 BCG-vaccinated,
HIV-negative infants aged 4–6 months,
with both study arms almost equally
UK\GFKPHCPVUTGEGKXGFQPGFQUG
QH/8##YJKNGKPHCPVUKPVJG
control arm received a placebo (Candin,
a C. albicans-derived skin test antigen).
Follow up lasted 37 months. The primary
objective of the study was to assess the
UCHGV[QH/8##KPVJGUGKPHCPVU6JG
secondary objectives were to evaluate
VJGGHßECE[QHVJGXCEEKPGCICKPUV
(a) the disease and (b) M. tuberculosis
infection, as measured by Quantiferon
conversion (this distinction is important
as infection only leads to active TB
disease in a small minority of immune-
competent individuals). Additional
objectives included the evaluation of
immunogenicity.
Conclusions drawn from the
results of the Phase IIb trial
6JKUYCUVJGßTUVENKPKECNVTKCNVQGXCNWCVG
VJGGHßECE[QHCPGY6$XCEEKPG
candidate for prevention of clinical
TB or M. tuberculosis infection, and
results were therefore of considerable
interest to the vaccine research and
public health communities. In this trial,
/8##CRRGCTGFVQDGUCHGCPFYGNN
VQNGTCVGFEQPßTOKPIUKOKNCTßPFKPIU
from previous Phase I and Phase IIa
clinical trials using this vaccine. None
of the observed serious adverse events
(or deaths) observed in the study arm
were assessed by the investigators to
be related to the vaccine, and only one
serious adverse event involving a brief
JQURKVCNK\CVKQPQEEWTTGFKPVJGRNCEGDQ
ITQWR6JGRTKOCT[GHßECE[CPCN[UKUYCU
based on the number of TB cases among
the vaccinated versus control subjects.
In the vaccine arm, there were 32 cases,
while in the placebo arm there were
39 cases. Based on this, the calculated
XCEEKPGGHßECE[YCU%+
VQHQTVJGRTKOCT[
6$ECUGFGßPKVKQPYJKEJYCUPQV
UVCVKUVKECNN[UKIPKßECPV/QTGQXGTVJGTG
was no evidence of protection against
M. tuberculosisKPHGEVKQPWUKPIVJG
Quantiferon-TB Gold assay as the read-
out. A total of 349 out of 2792 infants
DGECOGKPHGEVGFKPVJGXCEEKPGCTO
and 171 in the placebo arm), giving a
ECNEWNCVGFXCEEKPGGHßECE[QH¿
%+VQYJKEJYCU
CNUQPQVUVCVKUVKECNN[UKIPKßECPV
Implications for future studies
of this and other TB vaccine
candidates
Phase IIb proof–of-concept trials are
designed to allow ‘triage’ of vaccine
candidates and target populations, to
decrease risks before embarking on
hugely complex and resource consuming
Phase III trials. Current regulations
require a Phase IIb proof–of-concept
trial to be corroborated in larger
Phase III trials before a vaccine can be
licensed.
This study demonstrated that the
XCEEKPGJCFCPCEEGRVCDNGUCHGV[RTQßNG
in infants, and that a high quality trial
of a novel TB vaccine can be conducted
and produce robust results in a high
TB burden setting. The vaccine was
given months after all the infants had
received BCG vaccine, and it is possible
that BCG may have provided a plateau
level of protection, with very little, if any
CFFKVKQPCNRTQVGEVKQPCFFGFD[/8##
4CVGUQH6$KP5QWVJ#HTKECCPFVJG
Western Cape province in particular)
are exceptionally high in all age groups,
including young children, and this high
HQTEGQHKPHGEVKQPOC[DGFKHßEWNVVQ
address with any vaccine. It cannot be
assumed that similar results would have
been obtained in other populations. It
is also possible that adults, adolescents
and older children could be a better
VCTIGVRQRWNCVKQPHQTVJKUXCEEKPG
there is some evidence that it induces
a stronger immune response in older
age groups than in infants. Adults and
adolescents are the primary source
of transmission as they more likely to
develop the most infectious forms of the
disease and account for the largest share
of the burden of TB disease worldwide.
The vaccine is currently being evaluated
in HIV-infected adults in Senegal and
South Africa, using a two-dose regimen.
For all these reasons, the results of
the trial should not be considered as
RTQXKFKPICP[FGßPKVKXGCPUYGTVQVJG
question of whether a new TB vaccine
can provide better protection than BCG
alone. Further studies of this and other
vaccines are urgently needed. Several
of the other TB vaccine candidates in
VJGENKPKECNRKRGNKPGFKHHGTHTQO/8##
both in their antigenic composition and
in the way these antigens are delivered.
a 6COGTKU/&GVCN5CHGV[CPFGHßECE[QH
/8##CPGYVWDGTEWNQUKUXCEEKPGKPKPHCPVU
RTGXKQWUN[XCEEKPCVGFYKVJ$%)CTCPFQOK\GF
placebo-controlled phase 2b trial. The Lancet.
¿

97GLOBAL TUBERCULOSIS REPORT 2013
spectrum of TB disease and the transition from latent to
active TB, and developing a better understanding of the
behaviour of M. tuberculosis within the host. is progress
is expected to deliver better knowledge about pathogenesis
and identication of biomarkers and bio-signatures rele-
vant to new TB diagnostics. It is also expected to point to
new targets for anti-TB drugs as well as early indicators of
protective immunity, vaccine ecacy and early response to
treatment. Such developments will facilitate the selection
and testing of new interventions.
To highlight the crucial role of research in ending the
global TB epidemic, the WHO post-2015 global TB strategy
that is currently under development includes “Intensied
Research and Innovation” as one of three strategic pillars
(Chapter 1). e strategy is being developed as a successor
to the Stop TB Strategy, which covers the period 2006–2015.
In wide consultations held during 2012 and 2013, there has
been strong support for this pillar and its two main sub-
components, which are:
1. Discovery, development and rapid uptake of new tools,
interventions and strategies;
2. Research to optimize implementation and impact.
e research pillar will be essential to the success of the two
other pillars of the post-2015 global TB strategy and the
achievement of post-2015 global TB targets.
Biomedical research will need to be integrated as a crit-
ical component of the new post-2015 research strategy.
Creating connections among scientic disciplines that have
historically been inadequate or lacking (for example, bio-
medical research, epidemiology and operational research)
will depend upon close collaboration, consultation and
input from many research and public health stakeholders.
e need for more and expanded operational research
to optimize implementation and adopt innovations will
require extensive work at the country level, for example to
generate essential data on the epidemiology of TB (‘Know
your epidemic’) and universal health coverage, and to allow
adaptation of global recommendations and policies at the
national level.

#00':
Methods used to
estimate the global
burden of disease
caused by TB


101GLOBAL TUBERCULOSIS REPORT 2013
is annex explains the methods that were used to pro-
duce estimates of the global burden of disease caused by TB
(measured in terms of incidence, prevalence and mortality).
It has nine major sections:
General approach. is section provides some back-
ground information about the methods used to produce
estimates of disease burden.
Denitions. is section denes TB incidence, prev-
alence and mortality, the case fatality rate (CFR) and
the case notication rate. It also explains the regions
for which estimates of disease burden are produced and
sources of information on population estimates.
Estimates of TB mortality, 1990–2012. is section
explains the three methods used to estimate TB mor-
tality, and the countries for which they were applied.
Methods for estimating the number of HIV-associated
TB deaths and for disaggregation of TB mortality by age
and sex are also described.
Estimates of TB incidence, 1990–2012. is section
explains the main methods used to estimate TB inci-
dence, and the countries for which they were applied.
Methods to estimate the prevalence of HIV among inci-
dent TB cases are described.
Estimates of TB prevalence, 1990–2012. is sec-
tion explains the two methods used to estimate TB prev-
alence, and the countries for which they were applied.
Estimates of multidrug-resistant TB (MDR-TB)
incidence and mortality. is section explains the
main methods used to estimate MDR-TB mortality and
incidence based on drug resistance surveillance data and
parameters obtained from a recent literature review.
Projections of TB incidence, prevalence and mor-
tality. is section explains how projections from 2013
to 2015 were produced.
Uncertainty framework. is section explains the
general approach to including uncertainty in all esti-
mates.
1. General approach
Estimates of the burden of disease caused by TB (mea-
sured in terms of incidence, prevalence and mortality) are
produced annually by WHO using information gathered
through surveillance systems (case notications and death
registrations), special studies (including surveys of the
prevalence of disease, mortality surveys and in-depth anal-
yses of surveillance data), expert opinion and consultations
with countries. Two recent publications provide up-to-date
guidance about how TB incidence, prevalence and mortality
should be measured,1 based on the work of the WHO Global
Task Force on TB Impact Measurement.2 e methods used
to estimate the burden of disease were updated in 2009 fol-
lowing 18 months of work by an expert group convened by
the Task Force. ese updates were endorsed at a meeting of
the full Task Force in March 2010. Improvements to meth-
ods included systematic documentation of expert opinion
and how this has been used to produce estimates of disease
burden, simplication of models,3 updates to parameter
values based on the results of systematic reviews, much
greater use of mortality data from vital registration (VR)
systems and systematic documentation of uncertainty
(hence the uncertainty intervals shown on all of the esti-
mates of disease burden in this report).
2. Deßnitions
2.1 Incidence, prevalence, mortality, case fatality
TCVGECUGPQVKßECVKQPTCVG
Incidence is dened as the number of new and recurrent
(relapse) episodes of TB (all forms) occurring in a given
year. Recurrent episodes are dened as a new episode of TB
in people who have had TB in the past and for whom there
was bacteriological conrmation of cure and/or documen-
tation that treatment was completed (Box 3.1, Chapter 3).
In the remainder of this Annex, relapse cases are referred
to as recurrent cases because the term is more useful when
explaining the estimation of TB incidence. Recurrent cas-
es may be true relapses or a new episode of TB caused by
reinfection. In current case denitions, both relapse cases
and patients who require a change in treatment are called
‘retreatment cases’. However, people with a continuing epi-
sode of TB that requires a treatment change are prevalent
cases, not incident cases.
Prevalence is dened as the number of TB cases (all forms)
at a given point in time.
Mortality from TB is dened as the number of deaths
caused by TB in HIV-negative people, according to the
latest revision of the International classication of diseases
(ICD-10). TB deaths among HIV-positive people are classi-
ed as HIV deaths in ICD-10. For this reason, estimates of
deaths from TB in HIV-positive people are presented sepa-
rately from those in HIV-negative people.
e case fatality rate is the risk of death from TB among
people with active TB disease.4
e case notication rate refers to new and recurrent
episodes of TB notied to WHO for a given year, expressed
per 100000 population. e case notication rate for new
and recurrent TB is important in the estimation of TB inci-
dence. In some countries, however, information on treat-
ment history may be missing for some cases. When data
on treatment history are not available, recurrent cases
cannot be distinguished from cases whose treatment was
1 TB impact measurement: policy and recommendations for how to assess
the epidemiological burden of TB and the impact of TB control. Gene-
va, World Health Organization, 2009 (Stop TB policy paper, no.
2; WHO/HTM/TB/2009.416). e policy paper is available on the
Task Force web site: www.who.int/tb/advisory_bodies/impact_
measurement_taskforce
2 For further details, see the Task Force web site at: www.who.int/tb/
advisory_bodies/impact_measurement_taskforce
3 For example, some parameter values are now estimated only at glob-
al level or for regions, rather than for each country individually.
4 Straetemans M et al. Assessing tuberculosis case fatality ratio: a
meta-analysis. PLoS One. 2011, 6(6):e20755.

102 GLOBAL TUBERCULOSIS REPORT 2013
changed, since both are registered and reported in the cat-
egory ‘retreatment’. Patients reported in the ‘unknown his-
tory’ category are considered incident TB episodes (new or
relapse). is is a change from previous years in view of past
diculties to estimate with NTPs the proportion of true
new or relapse TB episodes in this category of patients (pre-
viously, patients with unknown treatment history were not
considered new or relapse cases). is change aects rela-
tively few countries, mostly in Western Europe.
2.2 Regions
Regional analyses are generally undertaken for the six
WHO regions (that is, the African Region, the Region of the
Americas, the Eastern Mediterranean Region, the Euro-
pean Region, the South-East Asia Region and the Western
Pacic Region). For analyses related to MDR-TB and for an
ecological model used to estimate TB mortality in some
countries, nine epidemiological regions were dened. ese
were African countries with high HIV prevalence, African
countries with low HIV prevalence, Central Europe, East-
ern Europe, high-income countries,1 Latin America, the
Eastern Mediterranean Region (excluding high-income
countries), the South-East Asia Region (excluding high-
income countries) and the Western Pacic Region (exclud-
ing high-income countries). e countries in these nine
regions are listed in Appendix 1.
2.3 Population estimates
e source of population estimates needed to calculate var-
ious TB indicators was the 2012 revision of the World Pop-
ulation Prospects, which is produced by the United Nations
Population Division (UNPD).2 e UNPD estimates some-
times dier from those made by countries.
3. Estimates of TB mortality, 1990–2012
e best sources of data about deaths from TB (excluding
TB deaths among HIV-positive people) are VR systems
in which causes of death are coded according to ICD-10
(although the older ICD-9 and ICD-8 classication are still
in use in several countries). Deaths from TB in HIV-positive
people are coded under HIV-associated codes.
ree methods were used to estimate TB mortality
among HIV-negative people:
direct measurements of mortality from VR systems or
mortality surveys;
indirect estimates based on an ecological model that
uses data from VR systems;
indirect estimates derived from multiplying estimates
of TB incidence by estimates of the CFR.
Each method is described in more detail below. Details
on the method used for each country are available online
at www.who.int/tb/publications/global_report/gtbr13_
mortality_source.csv.
3.1 Estimating TB mortality among HIV-negative
people from vital registration data and
mortality surveys
Data from VR systems are reported to WHO by Member
States and territories every year. In countries with func-
tioning VR systems in which causes of death are coded
according to the two latest revisions of the International
classication of diseases (underlying cause of death: ICD-10
A15-A19, equivalent to ICD-9: 010-018), VR data are the
best source of information about deaths from TB among
people not infected with HIV. When people with AIDS
die from TB, HIV is registered as the underlying cause of
death and TB is recorded as a contributory cause. Since one
third of countries with VR systems report to WHO only the
underlying causes of death and not contributory causes, VR
data usually cannot be used to estimate the number of TB
deaths in HIV-positive people.
TB mortality data obtained from VR systems are essen-
tial to understanding trends in TB disease burden where
case notications have incomplete coverage or their cov-
erage is not documented through an inventory study. An
updated description of the global coverage and quality of
VR data is available in World Health Statistics 2013.3
As of May 2013, 125 countries had reported mortality
data to WHO (including data from sample VR systems and
mortality surveys), among 217 countries and territories
from which TB data were requested. ese 125 countries
included 9 of the 22 high TB burden countries (HBCs): Bra-
zil, China, India, the Philippines, the Russian Federation,
South Africa, ailand, Viet Nam and Zimbabwe. However,
the VR data on TB deaths from South Africa and Zimbabwe
were not used for this report because large numbers of HIV
deaths were miscoded as TB deaths. Improved empirical
adjustment procedures have recently been published,4 and
options for specic post-hoc adjustments for misclassi-
cation errors in the measurement of TB mortality will be
reviewed extensively by the WHO Global Task Force on TB
Impact Measurement in 2014.
Among the countries for which VR data could be used
(see Figure 2.11 in Chapter 2), there were 2087 coun-
try-year data points 1990–2012. Of these data points, 24
outliers and points obtained from systems with very low
coverage were excluded for analytical purposes. Outliers
were detected visually by plotting country-specic time
series of reported TB mortality rates. As of June 2013, 62
data points were available for 2010, 35 for 2011 and none
for 2012. On average, 16 data points were retained for anal-
ysis per country (standard deviation (SD) of 6.7) from a
total of 2063 usable data points.
1 High-income countries are dened by the World Bank as countries
with a per capita gross national income (GNI) of ≥US$12 616 in
2012.
2 http://esa.un.org/unpd/wpp/ (accessed June 2013).
3 www.who.int/gho/publications/world_health_statistics/2013/en/
(accessed July 2013) (see particularly pages 15–16).
4 Birnbaum JK, Murray CJL, Lozano R. Exposing misclassied HIV/
AIDS deaths in South Africa. Bulletin of the World Health Organiza-
tion, 2011, 89:278–285.
103GLOBAL TUBERCULOSIS REPORT 2013
Reports of TB mortality were adjusted upwards to
account for incomplete coverage (estimated deaths with no
cause documented) and ill-dened causes of death (ICD-9
code B46, ICD-10 codes R00–R99).1
It was assumed that the proportion of TB deaths among
deaths not recorded by the VR system was the same as the
proportion of TB deaths in VR-recorded deaths. For VR-
recorded deaths with ill-dened causes, it was assumed
that the proportion of deaths attributable to TB was the
same as the observed proportion in recorded deaths.
e adjusted number of TB deaths da was obtained from
the VR report d as follows:
d
da =
c(1 – g)
where c denotes coverage (i.e. the number of deaths with
a documented cause divided by the total number of esti-
mated deaths) and g denotes the proportion of ill-dened
causes.
e uncertainty related to the adjustment was estimated
with standard deviation SD = d/4[1/c(1 – g) – 1]. e uncer-
tainty calculation does not account for miscoding, such as
HIV deaths miscoded as deaths due to TB.
Missing data between existing adjusted data points were
interpolated. Trailing missing values were predicted using
exponential smoothing models for time series.2 A penal-
ized likelihood method based on the in-sample t was used
for country-specic model selection. Leading missing val-
ues were similarly predicted backwards to 1990. A total of
799 country-year data points were thus imputed.
Results from mortality surveys were used to estimate
TB mortality in India and VietNam.
In 2012, 45% of global TB mortality (excluding HIV) was
directly measured from VR or survey data (or imputed from
survey or VR data from previous years). e remaining 55%
was estimated using the indirect methods described in
section 3.2 and section 3.3.
3.2 Estimating TB mortality among HIV-negative
people from an ecological model
An out-of-sample, goodness-of-t, stepwise selection
approach was used in 2012 using the series 1990–2011 to
select an ecological model that could predict TB mortality
in countries without VR data. e model was based on the
time series of VR data reported to WHO as described above,
expressed as counts of TB deaths and corrected for ill-
dened causes of deaths and VR coverage.
A population-averaged negative binomial model, with
total population as the oset converting model outputs to
rates, was used to account for the longitudinal structure of
the data as well as the observed over-dispersion of counts
of TB deaths.
Ten variables were investigated for inclusion in the
model. ese were: the infant mortality rate per 1000 live
births; gross domestic product per capita; HIV prevalence
among the general population; the percentage of the total
population aged <15 and ≥65 years; the TB treatment suc-
cess rate; the total number of newly notied TB cases per
year; whether or not a country had a high or low burden
of MDR-TB; whether a country was among the 22 HBCs or
not; and a categorical variable classifying countries in nine
groups with similar TB epidemiology (see Appendix 1).
At the univariate level, all risk factors were associated
with the outcome of TB mortality. e nal multivariate
model included the infant mortality rate per 1000 live
births, HIV prevalence among the general population, gross
domestic product per capita, the percentage of the total
population aged <15 and ≥65 years, whether a country was
in the list of 22 HBCs or not; and the categorical variable
that dened country groups with similar TB epidemiology.
Out of a total 4686 country-year observations in the
time series for 1990–2011, 802 could not be predicted
due to data not being available for any of the ten variables
included in the model.
Estimates of TB mortality predicted by the model were
used for 26 countries3 in which VR or mortality survey data
of sucient quality and coverage were not available and for
which estimates of TB incidence were judged too uncertain.
3.3 Estimating TB mortality among HIV-negative
people from estimates of case-fatality rates and
TB incidence
In 68 countries lacking VR data of the necessary coverage
and quality (in total, 94 countries lacked VR data of su-
cient coverage and quality but among 26 of them, the eco-
logical model described above was used), TB mortality was
estimated as the product of TB incidence (see section 4)
and the CFR using a model developed in 2012.
CFRs were estimated separately for TB cases notied to
NTPs and non-notied cases and, within these two groups,
separate estimates were made for HIV-negative TB cases in
high-income and other countries (Table A1.1).
TABLE A1.1
Estimates of TB case-fatality rates (HIV-negative) by
case type and country
%#5'6;2'#0&%17064;)4172 /'#056#0&&'8+#6+10
0QPPQVKßGFJKIJKPEQOG
countries
0.12 (0.042)
0QPPQVKßGFQVJGTEQWPVTKGU 0.32 (0.13)
0QVKßGFJKIJKPEQOGEQWPVTKGU 0.039 (0.042)
0QVKßGFQVJGTEQWPVTKGU 0.074 (0.03)
For consistency with VR- or survey-based mortality esti-
mates, CFRs were estimated such that they gave the best t
to the directly measured TB death rates (within their uncer-
tainty ranges) in the 123 countries with VR or mortality
1 Mathers CD et al. Counting the dead and what they died from: an
assessment of the global status of cause of death data. Bulletin of the
World Health Organization, 2005, 83:171–177.
2 Hyndman R et al. Forecasting with exponential smoothing: the state
space approach. Springer Series in Statistics, 2008.
3 For the list of the 26 countries, see www.who.int/tb/publications/
global_report/gtbr13_ mortality_source.csv.

104 GLOBAL TUBERCULOSIS REPORT 2013
survey data that were retained for analysis, in conjunction
with WHO estimates of distributions of TB incidence in
those countries. is statistical tting used Bayesian linear
models and was done separately for two groups of countries
(high-income and all other countries), to account for dier-
ences in the ratio of reported TB mortality to TB notica-
tion rates among these two groups (data not shown).
e models used normal errors and Gibbs sampling:
y = (I – N)β1 + Nβ2 + ¡, ¡ ~ N(0,σ2)
where y is TB mortality from VR, I denotes TB incidence
excluding people living with HIV, N denotes TB notica-
tions excluding people living with HIV, and parameters β1
and β2 denote the CFR in non-notied and notied cases
respectively. Semi-conjugate priors were set with an unin-
formative inverse Gamma prior on the conditional error
variance:
b ~ N(bi,Bi-2), σ2 ~ IG(5.10-4,5.10-4)
For low- and middle-income countries, priors b and their
precision B were dened based on literature reviews1 and
the country-year CFR parameters used by WHO for the
years 1999–2008. For high-income countries, non-infor-
mative priors were used. Convergence of Markov Chains
was assessed graphically and using convergence diagnos-
tic tests. Within each case category 1990–2011, mortality
estimates were computed by taking the product of posteri-
or distributions of the CFR, assumed to be time-indepen-
dent (Table A1.1), and country-year specic distributions
of estimated incidence.
3.4 Estimating TB mortality among
HIV-positive people
No nationally representative measurements of HIV-associ-
ated TB mortality were available from VR systems for use
in this report. In the absence of direct measurements, TB
mortality among HIV-positive people was estimated indi-
rectly according to the following methods (also see section
4.5) implemented in the Spectrum software.2
TB mortality is calculated as the product of HIV-positive
TB incidence (see section 4.5) and case fatality ratios:
M = (I-N)Fu + NFn
where I represents incident TB cases among people living
with HIV, N represents HIV-positive cases that are notied,
(I-N) represents HIV-positive TB cases that are not notied
and M represents TB mortality among HIV-positive peo-
ple. Fn and Fu are the case fatality ratios for notied and
non-notied incident cases, respectively.
e case fatality ratios were obtained in collabora-
tion with the TB Modeling and Analysis Consortium
(TB-MAC),3,4,5 and are shown in Table A1.2.
e disaggregation of incident TB into notied and not
notied cases is based on the ratio of the point estimates
for incident and notied cases. A single CFR was used for all
bootstrapped mortality estimates.
Direct measurements of HIV-associated TB mortality
are urgently needed. is is especially the case for coun-
tries such as South Africa and Zimbabwe, where national
VR systems are already in place. In other countries, more
eorts are needed to initiate the implementation of sample
VR systems as an interim measure.
3.5 TB mortality disaggregated by age and sex
For countries with VR data, it was possible to estimate TB
deaths (excluding TB deaths among HIV-positive people)
among children (aged <15 years) and adults (aged ≥ 15 years)
separately. It was also possible to disaggregate TB deaths by
sex. For these countries, male:female and child:adult ratios
of TB deaths (expressed as rates per 100000 population)
were calculated (after correction for ill-dened causes of
deaths and VR coverage). e ecological model described in
section 3.2 was used to predict ratios for countries with no
VR data. Directly measured (i.e. based on VR data for the
latest available year) or predicted country-level ratios were
then used to estimate ratios for WHO regions. ese were
then used to estimate the global ratio which was in turn
applied to the global number of estimated TB deaths among
HIV-negative TB cases to produce age and sex-disaggregat-
ed estimates.
TB deaths among HIV-positive people were disaggregat-
ed by sex using the assumption that the male:female sex
ratio is the same as the sex ratio of AIDS deaths estimated
by UNAIDS. Further details are provided in Box 2.2, Chap-
ter 2. Disaggregation of TB deaths by age and sex will be
one of the future developments of the TB component of the
Spectrum software (also see section 3.4).
TABLE A1.2
Estimates of the case fatality ratio among
HIV-positive TB cases
NON-NOTIFIED NOTIFIED
HIV-
Mode of triangular distribution 0.43 0.03
*+8PQVTGEGKXKPI#46
Mode of triangular distribution 0.09
4GEGKXKPI#46HQTNGUUVJCPQPG[GCT
Mode of triangular distribution 0.62 0.06
4GEGKXKPI#46HQTOQTGVJCPQPG[GCT
Mode of triangular distribution 0.49 0.04
1 Straetemans M et al. Assessing tuberculosis case fatality ratio: a
meta-analysis. PLoS One. 2011, 6(6):e20755.
2 http://www.futuresinstitute.org/spectrum.aspx
3 Tiemersma EW, van der Werf MJ, Borgdor MW, Williams BG,
Nagelkerke NJ (2011) Natural history of tuberculosis: duration
and fatality of untreated pulmonary tuberculosis in HIV negative
patients: a systematic review. PLoS One 6: e17601.
4 Corbett EL, Watt CJ, Walker N, Maher D, Williams BG, et al. (2003)
e growing burden of tuberculosis: global trends and interactions
with the HIV epidemic. Archives of Internal Medicine; 163: 1009–
1021.
5 Mukadi YD, Maher D, Harries A (2001) Tuberculosis case fatality
rates in high HIV prevalence populations in sub-Saharan Africa.
AIDS; 15: 143–152.

105GLOBAL TUBERCULOSIS REPORT 2013
4. Estimates of TB incidence, 1990–2012
No country has ever undertaken a nationwide survey of
TB incidence because of the large sample sizes required
and associated major logistic and nancial challenges. As
a result, there are no direct measurements of the incidence
of TB. eoretically, data from TB surveillance systems
that are linked to health systems of high coverage and per-
formance may capture all (or almost all) incident cases of
TB. e WHO Global Task Force on TB Impact Measure-
ment has developed a set of TB surveillance standards and
benchmarks that, if met, would allow direct measurement
of TB cases and deaths from surveillance data (Chapter 2).
In the absence of direct measurements, estimates of TB
incidence for almost all countries rely on methods described
in sections 4.1–4.3.
It should be emphasized that incidence estimates are
no longer derived from surveys of the prevalence of TB
infection as measured in tuberculin surveys. e WHO
Global Task Force on TB Impact Measurement has agreed
that methods for deriving incidence from the prevalence
of infection are unreliable. e Task Force has also stated
that, with a few exceptions, repeat tuberculin surveys do
not provide a reliable estimate of the trend in TB incidence.1
4.1 Estimating TB incidence from estimates of
the proportion of cases detected
Notication data for new and recurrent cases have been
analysed in combination with evidence about the cover-
age of the TB surveillance system and expert opinion in
six regional workshops and country missions held during
the period 2009–2013, according to methods developed by
the WHO Global Task Force on TB Impact Measurement.
By May 2013, these workshops and country missions had
covered 96 countries (Figure 2.1, Chapter 2), with several
countries re-assessed multiple times.
For the 96 countries covered by these regional work-
shops and country missions, incidence was estimated
according to the following equation:
case notications
incidence =
1 – underreporting
Expert opinion about the proportion of TB cases2 that were
not reported was elicited for three reference years (1997,
2003 and, depending on when the workshop was held,
2008–2012). is was done following in-depth analysis of
notication data (including data from sub-national admin-
istrative levels), programmatic data reecting eorts in TB
care and control (for example, data on infrastructure, sta-
ing, the performance of services and funding) and (where
available) data from inventory studies.3 In addition, data
on access to health care from Demographic and Health Sur-
veys and the overall performance of health systems (using
indicators such as the infant mortality rate) were used to
substantiate opinion on the proportion of cases with no
or very limited access to health care (Table A1.3). Results
from inventory studies combined with capture–recapture
modelling were used to estimate the gap between notied
cases and TB incidence in three countries that participated
in regional workshops: Egypt, Iraq and Yemen.
A full description of the methods used in these work-
shops is available in a report of the workshop held for
countries in the African Region (in Harare, Zimbabwe,
December 2010).4
TABLE A1.3
Sources of information and data on TB incidence
used in regional workshops and country missions
2155+$.'%#6')14+'51(+0%+&'06%#5'5 5174%'51(#
Do not have physical or
ßPCPEKCNCEEGUUVQJGCNVJECTG
Demographic and health
surveys, KABPa surveys Capture–
recapture
modelling
Seek care, but TB not
diagnosed Survey
TB diagnosed, but not
reported ‘Inventory’ survey
4GRQTVGFECUGU TB surveillance
a KABP = knowledge, attitudes, behaviour and practices.
Distributions of the proportion of cases that were not
reported in the three reference years were assumed to fol-
low a Beta distribution (Table A1.4). Reasons for using
Beta distributions include the following:
ey are continuous and dened on the interval (0, 1).
Since the variance of the proportions of cases that were
not reported tend to be large as a result of high uncer-
tainty, random draws of numbers from a normal distri-
bution would yield numbers outside the interval (0, 1).
e use of truncated normal distributions may result in
excess density towards one of the bounds.
ey are not necessarily symmetrical.
ey are dened with two parameters that can be
estimated from available data using the method of
moments.5
e shape and scale parameters necessary to dene the Beta
distribution were computed using the method of moments,
as follows:
First, the variance for the distribution was taken as:
V = ((u – l)/4)2
where l and u are the lower and upper bounds of the plau-
sible range for the proportion of incident cases that were
1 TB impact measurement: policy and recommendations for how to assess
the epidemiological burden of TB and the impact of TB control. Gene-
va, World Health Organization, 2009 (Stop TB policy paper, no. 2;
WHO/HTM/TB/2009.416).
2 Dened as cases of all forms of TB, including sputum smear-pos-
itive pulmonary cases, sputum smear-negative pulmonary cases
and extrapulmonary cases.
3 Measurements from ‘inventory’ studies can be used to quantify the
number of cases that are diagnosed but not reported to national
surveillance systems.
4 See www.who.int/tb/advisory_bodies/impact_measurement_
taskforce
5 Rényi A. Probability theory. New York, Dover Publications Inc.,
2007.

106 GLOBAL TUBERCULOSIS REPORT 2013
reported (also referred to as the case detection rate in
Chapter 3).
Shape 1 (noted α) and 2 (noted β) follow from:
α = sE
β = s(l – E)
where E is the expected value of the distribution.
Time series for the period 1990–2012 were built accord-
ing to the characteristics of the levels of underreporting
and under-diagnosis that were estimated for the three ref-
erence years. A cubic spline extrapolation of V and E, with
knots set at the reference years, was used for countries
with low-level or concentrated HIV epidemics. In countries
with a generalized HIV epidemic, the trajectory of inci-
dence from 1990 to the rst reference year (usually 1997)
was based on the annual rate of change in HIV prevalence.
Incidence trajectories were derived from the series of noti-
ed TB cases using Monte Carlo simulations from which
expected values, 2.5th and 97.5th centiles were extracted.
All computations were conducted in the R statistical envi-
ronment.1
In two countries, incidence rates were estimated to be
similar to those in a neighbouring country because infor-
mation from surveillance systems was insucient: esti-
mates for West Bank and Gaza Strip were extrapolated
from estimates for Jordan and estimates for South Sudan
were extrapolated from estimates for Sudan. e estimates
for West Bank and Gaza Strip and South Sudan should
therefore be considered as preliminary.
Trends in incidence were derived from repeat tuberculin
survey results in Bhutan, India and Yemen and for 40 coun-
tries (including countries in Eastern Europe) from trends
in mortality.
If there were insucient data to determine the factors
leading to time-changes in case notications, incidence was
assumed to follow a horizontal trend going through the
most recent estimate of incidence.
4.2 Estimating TB incidence from data on
case notißcations and expert opinion for
high-income countries
For high-income countries, the level of TB incidence was
assumed to be distributed between the notication rate
for new and recurrent cases combined, including reported
cases with undocumented treatment history as explained
in section 2.1 (lower uncertainty bound, noted l) and 1.3
times the notication rate (upper uncertainty bound, not-
ed u), as informed by expert opinion. e distribution of
incidence was assumed to follow a Beta distribution with
shape and scale parameters computed using the method of
moments, as described above.
In the absence of country-specic data on the quality
and coverage of TB surveillance systems, it was assumed
that TB surveillance systems from countries in the high-in-
come group performed similarly well, although the mod-
el does allow for stochastic uctuations. e exceptions
were the United Kingdom and the Netherlands, where the
underreporting of TB cases has been measured using inven-
tory studies and capture–recapture modelling.2,3 For these
two countries, the results from these studies were used to
measure TB incidence directly.
4.3 Estimating TB incidence from empirical
measurements of disease prevalence
Incidence can be estimated using measurements from
national surveys of the prevalence of TB disease combined
with estimates of the duration of disease. Incidence is esti-
mated as the prevalence of TB divided by the average dura-
tion of disease.
In practice, the duration of disease cannot be directly
measured. For example, measurements of the duration of
symptoms in prevalent TB cases that are detected during a
prevalence survey are systematically biased towards lower
values, since active case-nding truncates the natural his-
tory of undiagnosed disease. Measurements of the duration
of disease in notied cases ignore the duration of disease
among non-notied and untreated cases.
Literature reviews commissioned by the WHO Global
Task Force on TB Impact Measurement have provided esti-
mates of the duration of disease in untreated TB cases from
the pre-chemotherapy era (before the 1950s). e best esti-
mate of the mean duration of disease (for smear-positive
cases and smear-negative cases combined) in HIV-negative
individuals is about three years. However, the proportion
of incident cases that remain untreated is unknown. ere
are few data on the duration of disease in HIV-positive indi-
viduals.
When measurements from two prevalence surveys were
available, trends in TB prevalence were derived by tting a
log-linear model to available measurements. When three or
more prevalence measurements were available, the preva-
lence trajectory was built using cubic spline interpolation.
If only one prevalence survey measurement was available,
time-trends were assessed using in-depth analysis of sur-
veillance data, as described above.
In this report, the prevalence to incidence method was
used for two countries: Ethiopia and the Lao People’s Dem-
ocratic Republic.
4.4 Disaggregation of TB incidence
In this report, TB incidence is disaggregated by HIV-infec-
tion status (see section 4.5) at country level. e estima-
tion of smear-positive TB incidence was discontinued in
E(l – E)
s = – l
V
1 R Development Core Team. R: a language and environment for sta-
tistical computing. Vienna, R Foundation for Statistical Computing,
2009 (www.R-project.org).
2 Tuberculosis in the UK: annual report on tuberculosis surveillance in the UK
2010. London, Health Protection Agency Centre for Infections, 2010
(also available at: www.hpa.org.uk/web/HPAweb&HPAwebStandard/
HPAweb_C/1287143581697; accessed July 2011).
3 van Hest NA et al. Completeness of notication of tuberculosis in
e Netherlands: how reliable is record-linkage and capture–recap-
ture analysis? Epidemiology and Infection, 2007, 135(6):1021–1029.

107GLOBAL TUBERCULOSIS REPORT 2013
2010, for reasons explained in detail in the global report
published in 2010.
Global and WHO regional estimates of sex-disaggregat-
ed incidence were also calculated, based on country-level
female:male ratios of total new (all case types) TB case
notications, under the assumption that they are a proxy
of female:male ratios of incidence. Model-based estimated
WHO regional ratios were applied to global incidence for
the nal sex disaggregation (Chapter 2).
TB incidence was also disaggregated by age, to produce
global estimates among children (aged <15 years) and
adults (aged ≥ 15 years). Details of methods are provided in
Chapter 2, Box 2.2.
4.5 Estimates of HIV prevalence among
incident TB cases, 1990–2012
TB incidence was disaggregated by HIV and CD4 status
using the Spectrum software.1 WHO estimates of TB inci-
dence were used as inputs to the Spectrum HIV model. e
model was tted to WHO estimates of TB incidence, and
then used to produce estimates of TB incidence among
people living with HIV disaggregated by CD4 category.2 A
regression method was used to estimate the relative risk
(RR) for TB incidence according to the CD4 categories used
by Spectrum for national HIV projections. Spectrum data
were based on the national projections prepared for the
UNAIDS Report on the global AIDS epidemic 2012. e model
can also be used to estimate TB mortality among HIV-pos-
itive people, the resource requirements associated with
recently updated guidance on ART3 and the impact of ART
expansion.
A exible and relatively simple way of modelling TB inci-
dence (or any time-dependent function) is to represent it as
k time-dependent m’th order cubic-spline functions:
I(x) = Σi=1 to k βi Bmi(x)
where βi is the i’th spline coecient and Bmi(x) represents
the evaluation of the i-th basis function at time(year) x. e
TABLE A1.4
Parameter estimates used to produce estimates of TB incidence, prevalence and mortality
/1&'.2#4#/'6'4 &+564+$76+10 &+564+$76+102#4#/'6'45b
Incidence, high-income countries Betaa
where ě was set at 1. times the notißcation rate noted N and V is
deßned by
HIV prevalence among incident TB Betaa
Where –
Z is the eZpected Xalue and V is IiXen by
&WTCVKQPQHFKUGCUGPQPPQVKßGF 7PKHQTO NW[GCTU
HIV-negative cases of TB
&WTCVKQPQHFKUGCUGPQPPQVKßGF 7PKHQTO NW[GCTU
HIV-positive cases of TB
&WTCVKQPQHFKUGCUGPQVKßGF 7PKHQTO NW[GCTU
HIV-negative cases of TB
&WTCVKQPQHFKUGCUGPQVKßGF 7PKHQTO NW[GCTU
HIV-positive cases of TB
a 6JGRTQDCDKNKV[FGPUKV[HWPEVKQPQHVJG$GVCFKUVTKDWVKQPKU
b u and l denote upper and lower bounds.
α = –
Z .–
Z (1– –
Z )
V– 1
α = ě .ě (1– ě )
V– 1
β = (1– ě )
.ě (1– ě )
V– 1
β = (1– –
Z ) .–
Z (1– –
Z )
V– 1
4 (Z; _, β) = Z _ –1 (1– x) ` –1
1
0t _ –1 (1– t) ` –1 dt
V = 0.3
4N2
V = u – l
4
2
1 http://www.futuresinstitute.org/spectrum.aspx
2 Stover J, McKinnon R, Winfrey B. Spectrum: a model platform for
linking maternal and child survival interventions with AIDS, fam-
ily planning and demographic projections. International Journal of
Epidemiology 2010; 39 Suppl 1:i7–10.
3 http://www.who.int/hiv/pub/guidelines/arv2013/en/index.html

GLOBAL TUBERCULOSIS REPORT 2013
order of each basis function is m and cubic splines are used,
i.e. m=3. e equation simply states that any time-depen-
dent function, such as incidence, can be represented as a
linear combination of cubic-spline basis functions. e val-
ues of the cubic-spline coecients β were determined by
an optimization routine that minimizes the least squares
error between incidence data (Iobs) and the estimated inci-
dence curve I(x):
Σx=1990:2012 |I(x) - Iobs(x)|2 + λ β T S β
Here |I - Iobs|2 is the sum of squared errors in estimat-
ed incidence and S is a dierence penalty matrix applied
directly to the parameters β to control the level of varia-
tion between adjacent coecients of the cubic-spline, and
thus control (through a choice of λ) the smoothness of the
time-dependent case incidence curve. Another important
purpose of the use of the smoothness penalty matrix S is to
regularize (by creating smoothness dependencies between
adjacent parameters) the ill-conditioned inverse problem
(more unknown parameters than the data can resolve) that
would tend to over t the data when left ill-conditioned.
Cubic5plines and conßdence intervals
e cubic-spline method was then used to t indicators
(incidence, case notications, etc.) to a set of bootstrapped
data, obtained by sampling from the normal error distribu-
tion resulting from tting the ‘point estimate’. is boot-
strap method produces a sample of projected cubic-spline
curves that are practically equivalent to a set that would
be obtained from tting the model to the same number of
repeated measurements (or assessments) of the given indi-
cator. Condence intervals based on the bootstrapped data
are typically narrow in the years where the model has data
to utilize, and ‘spread out’ after that, according to a Gauss-
ian process with an increasing variance.
Projecting TB incidence among people living with HIV
by CD4 category
e disaggregation of TB incidence by CD4 category among
people living with HIV was based on the idea that an
increase in the relative risk for TB incidence is a function of
CD4 decline. Williams et al captured this idea in a model for
the relationship between the RR for TB and CD4 decline.1
ey suggested a 42% (+/- 17%) increase in RR for TB for
each unit of 100μL CD4 decline.
e Spectrum-TB model’s disaggregation method is
based on the Williams et al. model. e model rst esti-
mates incidence among people living with HIV, and then
calculates the ‘risk of TB’ F=I- / P-, where I- is TB incidence
among people living with HIV and P- is the number of peo-
ple living with HIV who are susceptible to TB.
An assumption is made that the risk of TB infection
among people living with HIV with CD4 count > 500 μL is
proportional to F (it was assumed that it was higher by a
factor of 2.52). For each 100 μL CD4 decline in the remain-
ing categories (350–499, 250–349, 200–249, 100–199,
50–99 CD4 cells/μL, and CD4 count less than 50 cells μL),
the risk of infection is represented as:
F(c<500) = F(c>500)∙p(1)∙p(2)dc,
where p(1) is a parameter that is used to recognize that
people living with HIV who have high CD4 counts could
be at higher risk of TB infection relative to those who are
HIV-negative, and p(2) controls the exponential increase in
RR that occurs with CD4 decline. dc is the number of 100μL
CD4 decline associated with the midpoint of each CD4 cate-
gory relative to 500: dc= (3.0, 4.4, 8.6, 12.9, 19.2, 28.6, 37.3)
for the six CD4 categories.
A reduction in RR is applied for those who have been on
ART for more than one year.
Parameter assumptions
To match total TB incidence and estimates of the num-
ber of HIV-positive TB cases from HIV testing data where
available, it was assumed that p(1)=2.5 and p(2) was tted
accordingly.
In the RR-approach, the ‘biological meaning’ that should
be attached to the parameters and a more straightforward
interpretation of these parameters as regression coe-
cients need to be balanced. Both parameters can be tted
or both can be xed. Varying at least p(2) captures the vari-
ation among countries that is expected due to variation
in the baseline (HIV-negative) CD4 count, and it strikes a
balance between the biological and regression mechanisms.
e RR model approach to estimation of TB incidence
was used for people on ART. Although an estimate of TB
incidence among people on ART could be obtained from
surveillance data reported to WHO (such that it is arguably
not necessary to use the RR model), limitations of the ART
data (in particular that some countries appear to report
cumulative totals of people on ART) meant that the RR
approach needed to be used.
Hazard ratios (HR) of 0.35 were assumed for all CD4 at
ART initiation categories. Suthar et al have reported HRs
of 0.16, 0.35 and 0.43 for those on ART with CD4 count
< 200, 200–350 and > 350,3 and these values could in prin-
ciple be used. However, Spectrum tracks only CD4 at initi-
ation, thus limiting the use of CD4-specic HRs for people
on ART.
It was further assumed that the HR of 0.35 applies only
to patients on ART for more than six months. Spectrum’s
ART-mortality estimates, derived mostly from ART cohorts
in Sub-Saharan Africa, suggest that mortality remains very
1 Williams B. e impact of ART for HIV on TB. http://www.who.int/
hiv/topics/artforprevention/williams.pdf (accessed July 2013).
2 Sonnenberg P, et al. How Soon after Infection with HIV Does the
Risk of Tuberculosis Start to Increase? A Retrospective Cohort
Study in South African Gold Miners. Journal of Infectious Diseas-
es.2005 Jan 15;191(2):150–8.
3 Suthar AB, Lawn SD, del Amo J, Getahun H, Dye C, et al. (2012)
Antiretroviral erapy for Prevention of Tuberculosis in Adults
with HIV: A Systematic Review and Meta-Analysis. PLoS Med 9(7):
e1001270. doi:10.1371/journal.pmed.1001270

109GLOBAL TUBERCULOSIS REPORT 2013
high in the rst six months of ART. Since TB is a leading
contributor to mortality among HIV-positive people, it was
judged that the HR for patients on ART for 0‒6 months is
likely to remain high; therefore, a reduction factor due to
ART was not applied for this subset of patients.
Likelihood function
A simple least squares approach was used to t the model to
total TB incidence, and to all available estimates of TB inci-
dence among people living with HIV. ese estimates of TB
incidence among people living with HIV were obtained by
three sampling methods: population surveys of the preva-
lence of HIV among TB cases (least biased, but scarce due
to logistical constraints), sentinel HIV data (biases include
more testing of people with advanced HIV-related disease)
and routine HIV testing of reported TB patients (variable
coverage). To increase the inuence of survey data, replicas
of the survey data were included in the likelihood function.
In other words, for years for which data from HIV testing
were available, identical copies of the HIV-test data were
added to the likelihood function. e estimate of total TB
incidence was based on much more data, evenly spread out
in the estimation period 1990–2015.
Model testing showed that using two replicates of the
HIV survey data (i.e. duplicating the survey data) and
two replicates of the routine testing data with coverage
greater than 90% was the best approach to disaggregating
TB incidence: the t passed close to the survey or high-
coverage routine testing data points that were available.
For each of a) HIV sentinel and b) routine testing with cov-
erage between 50–90%, data were not used.
A prototype Bayesian importance sampling (IMIS)
algorithm was developed to handle complex data weigh-
ing possibilities, but it was based on subjective priors and
likelihood functions and is more time-consuming to run
than simple least squares. For the purposes of producing
estimates for all countries automatically, the least squares
method was used. In future, least squares and IMIS tting
could be made available to the end user.
For countries with no data, a range for p(2) was esti-
mated from countries with survey or testing data, which
suggest that p(2) = 1.96 [1.8–2.1]. e RR-model was then
tted to total TB incidence only. ere is no satisfactory
way to verify results for TB incidence among people living
with HIV when no HIV-testing data are available. However,
comparison of the global estimate for TB incidence among
people living with HIV produced by Spectrum and esti-
mates previously published by WHO (based on a dierent
method using HIV prevalence instead of CD4 distributions
and using HIV-test data in a dierent way) suggests that
the RR-model works reasonably well.
Provider-initiated testing and counselling with at least
50% HIV testing coverage is the most widely available
source of information on the prevalence of HIV in TB
patients. However, this source of data is aected by biases,
particularly when coverage is closer to 50% than to 100%.
In all countries with repeat data from testing, the relation-
ship between the prevalence of HIV in TB patients and the
coverage of HIV testing was examined graphically. In some
countries, the prevalence of HIV in TB patients was found
to decrease with increasing HIV testing coverage while in
others it increased with increasing HIV testing coverage;
in most countries, the prevalence of HIV followed highly
inconsistent patterns (with repeat changes in direction) as
HIV testing coverage increased. erefore, it was not pos-
sible to adjust for the eect of incomplete coverage of HIV
testing on estimates of the prevalence of HIV among TB
patients. e assumption was thus made that TB patients
with an HIV test result were statistically representative
of all TB cases. As coverage of HIV testing continues to
increase globally, biases will decrease.
For the 1003 country-year data points corresponding to
countries for which no surveillance data were available, the
prevalence of HIV was estimated indirectly according to
the following equation:
In this equation, t is HIV prevalence among incident TB
cases, h is HIV prevalence among the general population
(from the latest time-series provided by UNAIDS) and ρ is
the incidence rate ratio (IRR) (dened as the incidence rate
of TB in HIV-positive people divided by the incidence rate
of TB in HIV-negative people). We then let logit(t) be log(t/
(1-t)) and logit(h) be log(h/(1-h)). Using data from countries
where HIV prevalence has been estimated by UNAIDS as an
independent variable, a linear model of logit-transformed
t was tted using logit-transformed h according to the fol-
lowing equation, written in matrix notation:
ˆ
TXβ
where ˆ
T is a vector of predicted logit(t), X is an n x 2 matrix
in which the rst column holds 1s, and the second column
holds logit(h). e vector β holds estimated model parame-
ters. Models were tested with lags set for logit(h) ranging
from no lag to a lag of eight years. e best t was obtained
with a lag of one year.
Models were run using Monte Carlo simulations in
which h was drawn randomly from a Beta distribution with
shape parameters computed as described in Section 4.1,
(low and high uncertainty bounds are provided by UNAIDS
– also see Table A1.5). e model was run 50000 times
TABLE A1.5
Sources of data on HIV prevalence among
incident TB cases
&+4'%6/'#574'/'061(6*'24'8#.'0%'1(*+8+06$2#6+'065 07/$'41(%17064;;'#45
National surveysa124
HIV sentinel surveillance 24
Provider-initiated testing and counselling with at
least 50% coverage of testing 1297
Total, at least one data source available 1322
a the reported survey number is over-stated as a number of country reports confused
survey and routine testing with near 100% coverage
l + h(l – l)
hl
t =

110 GLOBAL TUBERCULOSIS REPORT 2013
using country-specic distributions for H and T (noted in
capital letters to denote vectors or matrices) based on their
uncertainty intervals. e uncertainty bounds for β were
chosen as the 2.5th and 97.5th centiles.
5. Estimates of TB prevalence, 1990–2012
e best way to measure the prevalence of TB is through
national population-based surveys of TB disease.1,2 Data
from such surveys are available for an increasing number
of countries (Chapter 2). It should be noted, however, that
measurements of prevalence are typically conned to the
adult population. Furthermore, prevalence surveys exclude
extrapulmonary cases and do not allow the diagnosis of
cases of culture-negative pulmonary TB.
When there is no direct measurement from a national
survey of the prevalence of TB disease, prevalence is the
most uncertain of the three TB indicators used to measure
disease burden. is is because prevalence is the product of
two uncertain quantities: (i)incidence and (ii) disease dura-
tion. e duration of disease is very dicult to quantify
because it cannot be measured during surveys of the prev-
alence of TB disease (surveys truncate the natural history
of disease). Duration can be assessed in self-presenting
patients, but there is no practical way to measure the dura-
tion of disease in patients who are not notied to NTPs.
Indirect estimates of prevalence were calculated accord-
ing to the following equation:
P = -Ii,jdi,j, iD{1,2}, jD{1,2}
where the index variable i denotes HIV+ and HIV–, the
index variable j denotes notied and non-notied cases, d
denotes the duration of disease in notied cases and I is
total incidence. In the absence of measurements, we did not
allow duration in notied cases to vary among countries.
Given their underlying uncertainty, prevalence estimates
should be used with great caution in the absence of direct
measurements from a prevalence survey. Unless measure-
ments were available from national programmes (for exam-
ple, Turkey), assumptions of the duration of disease were
used as shown in the last four rows of Table A1.3.
6. Estimates of the number of cases
of and deaths from MDR-TB
2TQRQTVKQPQHPQVKßGFECUGUQH6$VJCVJCXG
MDR-TB, 2012
Global and regional estimates of the proportion of new and
retreatment cases of TB that had MDR-TB in 2012 were cal-
culated using country-level information. If countries had
reported data on the proportion of new and retreatment
cases of TB that have MDR-TB from routine surveillance
or a survey of drug resistance the latest available informa-
tion was used. For countries that have not reported such
data, estimates of the proportion of new and retreatment
cases of TB that have MDR-TB were produced using mod-
elling (including multiple imputation) that was based on
data from countries for which data do exist. Estimates for
countries without data were based on countries that were
considered to be similar in terms of TB epidemiology (for
country groups see Appendix 1). e observed and imputed
estimates of the proportion of new and retreatment cases
of TB that have MDR-TB were then pooled to give a global
estimate, with countries weighted according to their share
of global notications of new and retreatment cases.
6.2 MDR-TB mortality, 2012
e VR mortality data reported to WHO by Member States
does not dierentiate between MDR-TB and non-MDR-TB
as a cause of death (there is no specic ICD-9 or ICD-10
codes for MDR-TB, although countries such as South Africa
have allocated two specic codes U51 and U52 to classify
deaths from MDR-TB and XDR-TB respectively).3 ere-
fore, a systematic review and meta-analysis of the pub-
lished literature was undertaken to estimate the relative
risk of dying from MDR-TB compared with non MDR-TB.
e global estimate of MDR-TB deaths (Box 2.3) was then
based on the following formula:
m = M.p.r
Where:
m = global MDR-TB mortality,
M = global TB mortality,
p = overall proportion of MDR-TB among prevalent TB
cases, approximated by the weighted average of the
proportion of new and retreated cases that have MDR-
TB,
r = the relative risk of dying from MDR-TB versus non-
MDR-TB.
6.3 Numbers of incident cases of MDR-TB, 2012
e global estimate of MDR-TB incidence was calculated as
the addition of three groups of MDR-TB incident cases:
1. incident MDR-TB among new pulmonary and extra-pul-
monary incident TB cases, using the proportion of
MDR-TB among new cases from drug resistance surveil-
lance (DRS);
2. incident MDR-TB among relapses, using the proportion
of MDR-TB among new cases from DRS and the esti-
mated relative risk of MDR among relapse versus new
cases; and
3. incident MDR-TB among retreated cases that are not
relapses, which was assumed to follow a uniform distri-
bution with min=0, max=upper limit of the global pro-
portion of MDR-TB among retreated cases estimated
from DRS.
A second method to estimate global MDR-TB incidence was
also explored, in which the global estimate of mortality due
1 Glaziou P et al. Tuberculosis prevalence surveys: rationale and
cost. International Journal of Tuberculosis and Lung Disease, 2008,
12(9):1003–1008.
2 TB prevalence surveys: a handbook. Geneva, World Health Organiza-
tion, 2011 (WHO/HTM/TB/2010.17).
3 Mortality and causes of death in South Africa, 2010: Findings from
death notification. http://www.statssa.gov.za/publications/
p03093/p030932010.pdf

111GLOBAL TUBERCULOSIS REPORT 2013
to MDR-TB was divided by the estimated case fatality ratio
(CFR) among cases of MDR-TB. e CFR was calculated as
a weighted average of the case fatality ratio among patients
that are treated and those that are not, according to the fol-
lowing formula:
4 = pt * 4t + (1-pt)*4un
Where:
pt = proportion treated, approximated by the proportion
of enrolled MDR-TB patients on treatment out of
those estimated to exist among notied TB patients
with pulmonary TB;
4t = case fatality rate among patients treated for MDR-TB,
using treatment outcome data for MDR-TB patient
cohorts;
4un = case fatality rate among people with MDR-TB who are
not treated, which was assumed to follow a uniform
distribution with min=0.4, max=0.6.
Outputs from both methods gave similar best estimates
of MDR-TB incidence with largely overlapping condence
intervals.
6.4 Resistance to second-line drugs among
patients with MDR-TB
Data from 75 countries were used to produce global esti-
mates of the following proportions: (i) patients with MDR-
TB who had XDR-TB; (ii) patients with MDR-TB who had
uoroquinolone resistance; (iii) patients with MDR-TB who
had resistance to second-line injectable drugs and uoro-
quinolones but not XDR-TB. e latest available national
and subnational data from each country were analysed
using logistic regression models with robust standard
errors to account for the clustering eect at the level of the
country or territory. e analysis was limited to countries
in which more than 66% of MDR-TB cases received sec-
ond-line DST.
7. Projections of incidence, prevalence
and mortality up to 2015
Projections of TB incidence, prevalence and mortality
ratesup to 2015enable assessment of whether global tar-
gets set for 2015 are likely to be achieved at global, region-
al and country levels. Projections for the years 2013–2015
weremade using exponential smoothingmodels tted to
data from 2006–2012.
8. Estimation of uncertainty
ere are many potential sources of uncertainty associated
with estimates of TB incidence, prevalence and mortality,
as well as estimates of the burden of HIV-associated TB
and MDR-TB. ese include uncertainties in input data, in
parameter values, in extrapolations used to impute missing
data, and in the models used.
We used xed population values from the UNPD. We did
not account for any uncertainty in these values.
Notication data are of uneven quality. Cases may be
underreported (for example, missing quarterly reports
from remote administrative areas are not uncommon), mis-
classied (in particular, misclassication of recurrent cases
in the category of new cases is common), or overreported
as a result of duplicated entries in TB information systems.
e latter two issues can only be addressed eciently in
countries with case-based nationwide TB databases that
include patient identiers. Sudden changes in notications
over time are often the result of errors or inconsistencies in
reporting, but may sometimes reect abrupt changes in TB
epidemiology (for example, resulting from a rapid inux of
migrants from countries with a high burden of TB, or from
rapid improvement in case-nding eorts).
Missing national aggregates of new and recurrent cas-
es were imputed by interpolation. Notication trajectories
were smoothed using a penalized cubic splines function
with parameters based on the data. Attempts to obtain cor-
rections for historical data are made every year, but only
rarely do countries provide appropriate data corrections.
Mortality estimates incorporated the following sourc-
es of uncertainty: sampling uncertainty in the underlying
measurements of TB mortality rates from data sources,
uncertainty in estimates of incidence rates and rates of HIV
prevalence among both incident and notied TB cases, and
parameter uncertainty in the Bayesian model. Time series
of TB mortality were generated for each country through
Monte Carlo simulations.
Unless otherwise specied, uncertainty bounds and
ranges were dened as the 2.5th and 97.5th centiles of out-
come distributions. roughout this report, ranges with
upper and lower bounds dened by these centiles are pro-
vided for all estimates established with the use of simula-
tions. When uncertainty was established with the use of
observed or other empirical data, 95% condence intervals
are reported.
e model used the following sequence: (1) Overall TB
incidence estimation after review and cleaning of case
notication data; (2) cleaning and adjustment of raw mor-
tality data from VR systems and mortality surveys, fol-
lowed by imputation of missing values in countries with
VR or survey data – in some countries, step 1 was updat-
ed to account for mortality data; (3) cleaning of measure-
ments of HIV prevalence among TB patients followed by
estimating HIV-positive TB incidence using the Spectrum
programme and HIV-positive TB mortality; (4) estimation
of HIV prevalence among incident cases of TB through
modelling in countries with no measurements; (5) estima-
tion of HIV-negative TB mortality in countries with no VR
data followed with an update of step 1 in some countries;
(6) review of prevalence measurements, adjustments for
childhood TB and bacteriologically unconrmed TB, and
estimation of prevalence followed with an update of step 1
in some countries; (7) estimation of incidence and mortali-
ty disaggregated by age and sex and disaggregated by drug
resistance status.
e general approach to uncertainty analyses was to
draw values from specied distributions for every param-

112 GLOBAL TUBERCULOSIS REPORT 2013
eter (except for notications and population values) in
Monte Carlo simulations, with the number of simulation
runs set so that they were sucient to ensure stability
in the outcome distributions. For each country, the same
random generator seed was used for every year, and errors
were assumed to be time-dependent within countries
(thus generating autocorrelation in time series). Regional
parameters were used in some instances (for example, for
CFRs). Summaries of quantities of interest were obtained
by extracting the mean, 2.5th and 97.5th centiles of pos-
terior distributions. Wherever possible, uncertainty was
propagated analytically by approximating the moments of
functions of random variables using Taylor expansions –
such as when taking the product or the ratio of two random
variables – rather than through Monte Carlo simulations,
in order to shorten computing time.
Appendix 1. Epidemiological regions used
for analyses
Africa – countries with high HIV prevalence: Botswa-
na, Burundi, Cameroon, the Central African Republic, the
Congo, Côte d’Ivoire, the Democratic Republic of the Con-
go, Ethiopia, Gabon, Kenya, Lesotho, Malawi, Mozambique,
Namibia, Nigeria, Rwanda, South Africa, South Sudan,
Swaziland, Uganda, the United Republic of Tanzania, Zam-
bia, Zimbabwe.
Africa – countries with low HIV prevalence: Alge-
ria, Angola, Benin, Burkina Faso, Cape Verde, Chad, the
Comoros, Djibouti, Eritrea, the Gambia, Ghana, Guinea,
Guinea-Bissau, Liberia, Madagascar, Mali, Mauritania,
Mauritius, the Niger, Sao Tome and Principe, Senegal, Sey-
chelles, Sierra Leone, Somalia, Sudan, Togo.
Central Europe: Albania, Bosnia and Herzegovina, Mon-
tenegro, Serbia, the former Yugoslav Republic of Macedo-
nia, Turkey.
Eastern Europe: Armenia, Azerbaijan, Belarus, Bulgaria,
Georgia, Kazakhstan, Kyrgyzstan, Latvia, Lithuania, the
Republic of Moldova, Romania, the Russian Federation,
Tajikistan, Turkmenistan, Ukraine, Uzbekistan.
High-income countries: Andorra, Aruba, Australia,
Austria, the Bahamas, Bahrain, Barbados, Belgium, Bermu-
da, Brunei Darussalam, Canada, the Cayman Islands, Chi-
na, Hong Kong SAR, China Macao SAR, Croatia, Cyprus,
the Czech Republic, Denmark, Equatorial Guinea, Esto-
nia, Finland, France, French Polynesia, Germany, Greece,
Greenland, Guam, Hungary, Iceland, Ireland, Israel, Italy,
Japan, Kuwait, Luxembourg, Malta, Monaco, the Nether-
lands, the Netherlands Antilles, New Caledonia, New Zea-
land, Northern Mariana Islands, Norway, Oman, Poland,
Portugal, Puerto Rico, Qatar, the Republic of Korea, Saint
Kitts and Nevis, San Marino, Saudi Arabia, Singapore, Slo-
vakia, Slovenia, Spain, Sweden, Switzerland, Trinidad and
Tobago, the Turks and Caicos Islands, US Virgin Islands,
United Arab Emirates, the United Kingdom, the United
States.
Eastern Mediterranean: Afghanistan, Egypt, Iran (Islamic
Republic of), Iraq, Jordan, Lebanon, Libya, Morocco, Paki-
stan, Syrian Arab Republic, Tunisia, West Bank and the
Gaza Strip, Yemen.
Latin America: Anguilla, Antigua and Barbuda, Argen-
tina, Belize, Bolivia (Plurinational State of), Bonaire,
Saint Eustatius and Saba, Brazil, British Virgin Islands,
Chile, Colombia, Costa Rica, Cuba, Curaçao, Dominica, the
Dominican Republic, Ecuador, El Salvador, Grenada, Gua-
temala, Guyana, Haiti, Honduras, Jamaica, Mexico, Mont-
serrat, Nicaragua, Panama, Paraguay, Peru, Saint Kitts
and Nevis, Saint Lucia, Saint Vincent and the Grenadines,
Sint Maarten (Dutch part), Suriname, Uruguay, Venezuela
(Bolivarian Republic of).
South East Asia: Bangladesh, Bhutan, Democratic Peo-
ple’s Republic of Korea, India, Indonesia, Maldives, Myan-
mar, Nepal, Sri Lanka, ailand, Timor-Leste.
West Pacic: American Samoa, Cambodia, China, Cook
Islands, Fiji, Kiribati, Lao People’s Democratic Republic,
Malaysia, Marshall Islands, Micronesia (Federated State
of), Mongolia, Nauru, Niue, Palau, Papua New Guinea,
the Philippines, Samoa, Solomon Islands, Tokelau, Tonga,
Tuvalu, Vanuatu, Viet Nam, Wallis and Futuna Islands.

#00':
Country proßles

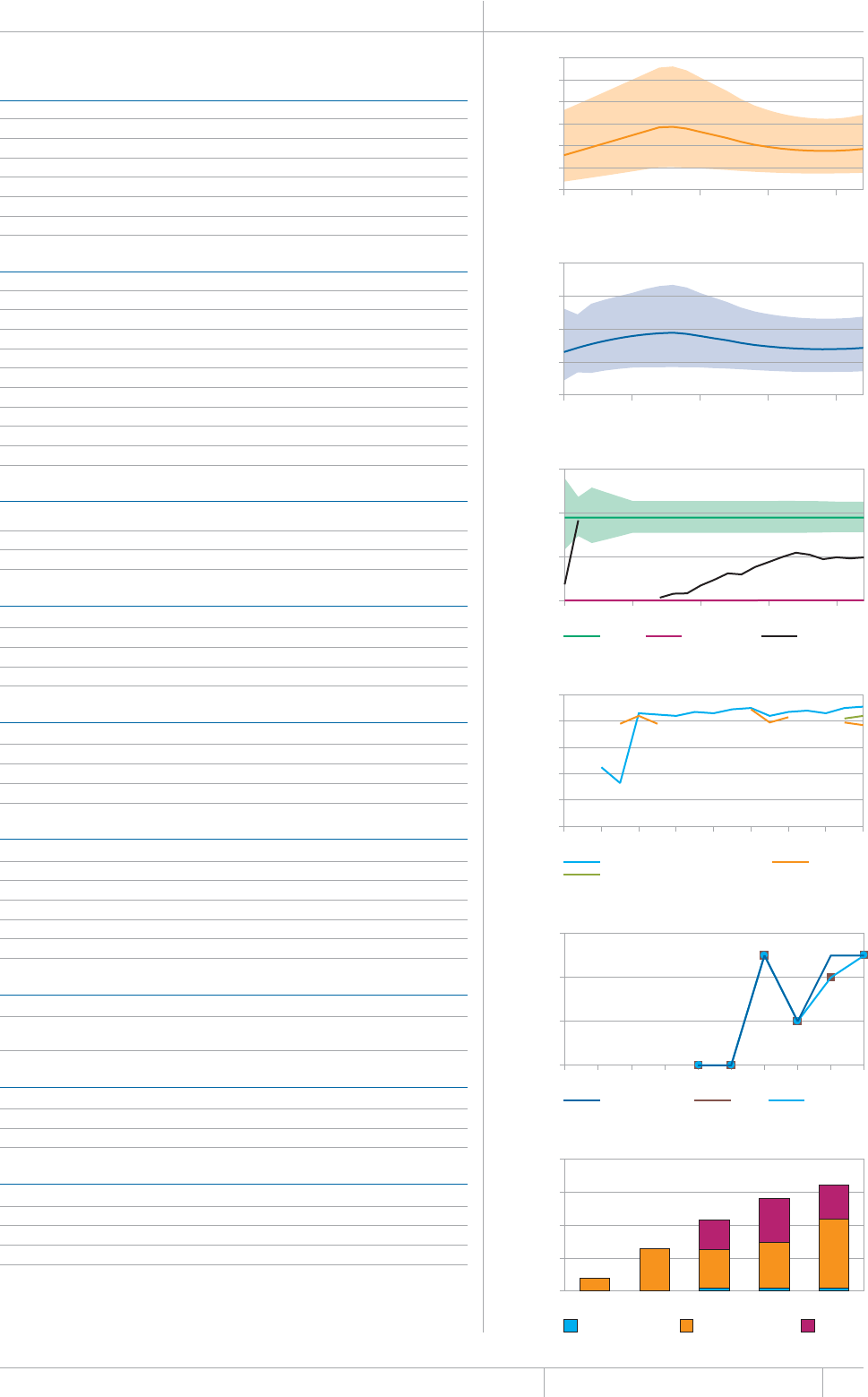
115 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
Data are as reported to WHO. Estimates of TB and MDR-TB burden are produced by WHO in consultation with
countries.
a Ranges represent uncertainty intervals.
HIGH TB BURDEN
Estimates of TB burdena 2012
07/$'4(thousands)4#6' (per 100 000 population)
/QTVCNKV[GZENWFGU*+86$ ¿ ¿
/QTVCNKV[*+86$QPN[ ¿ ¿
2TGXCNGPEGKPENWFGU*+86$ ¿ ¿
+PEKFGPEGKPENWFGU*+86$ ¿ ¿
Incidence (HIV+TB only) 0.31 (0.19–0.46) 1 (0.63–1.5)
Case detection, all forms (%) 52 (44–63)
6$ECUGPQVKßECVKQPU
0'9%#5'5 4'64'#6/'06%#5'5
5OGCTRQUKVKXG 4GNCRUG
Smear-negative 4 740 (17) Treatment after failure 160 (13)
Smear-unknown / not done 2 665 (9) Treatment after default 37 (3)
Extrapulmonary 6 906 (24) Other
Other 702 (2)
Total new 28 332 Total retreatment 1 246
Other (history unknown)
6QVCNPGYCPFTGNCRUG 6QVCNECUGUPQVKßGF
New cases
5/'#40')#6+8'70-0190
5/'#4215+6+8' 016&10' ':64#27./10#4;
/(TCVKQ
#IG
Laboratories 2012
Smear (per 100 000 population) 2.0
Culture (per 5 million population) 0.3
Drug susceptibility testing (per 5 million population) 0
+UUGEQPFNKPGFTWIUWUEGRVKDKNKV[VGUVKPICXCKNCDNG! ;GUQWVUKFGEQWPVT[
6TGCVOGPVUWEEGUUTCVG
New smear-positive and/or culture-positive 91
0GYUOGCTPGICVKXGGZVTCRWNOQPCT[
4GVTGCVOGPV
Is rifampicin used throughout treatment for new patients? No
TB/HIV 2012 07/$'4 (%)
TB patients with known HIV status 7 275 (25)
*+8RQUKVKXG6$RCVKGPVU
*+8RQUKVKXG6$RCVKGPVUQPEQVTKOQZC\QNGRTGXGPVKXGVJGTCR[%26
*+8RQUKVKXG6$RCVKGPVUQPCPVKTGVTQXKTCNVJGTCR[#46
*+8RQUKVKXGRGQRNGUETGGPGFHQT6$
HIV-positive people provided with IPT 25
Estimates of MDR-TB burden 2012a 0'9 4'64'#6/'06
QH6$ECUGUYKVJ/&46$ ¿ ¿
/&46$ECUGUCOQPIPQVKßGF
pulmonary TB cases 750 (21–2 600) 400 (93–700)
Reported cases of MDR-TB 20120'9 4'64'#6/'06 616#.
%CUGUVGUVGFHQT/&46$
.CDQTCVQT[EQPßTOGF/&46$ECUGU
2CVKGPVUUVCTVGFQP/&46$VTGCVOGPV
Financing TB control 2013
0CVKQPCN6$RTQITCOOGDWFIGV75OKNNKQPU
% Funded domestically 3%
% Funded internationally 65%
% Unfunded 32%
AFGHANISTAN Population 2012 30 million
Mortality (excludes HIV+TB) (rate
per 100 000 population per year)
1990 1995 2000 2005 2010
0
20
40
60
80
100
120
Prevalence
(rate per 100 000 population)
1990 1995 2000 2005 2010
0
250
500
750
1000
Incidence (rate per 100 000
population per year)
1990 1995 2000 2005 2010
Incidence Incidence (HIV+TB) Notifications
0
100
200
300
Treatment success rate (%)
1995 1997 1999 2001 2003 2005 2007 2009 2011
0
20
40
60
80
100
New smear-positive (and/or culture-positive) Retreatment
New smear-negative/extrapulmonary
Number of patients
HIV-positive TB patients on CPT on ART
2003 2004 2005 2006 2007 2008 2009 2010 2011 2012
0
2
4
6
Total budget (US$ millions)
Funded domestically Funded internationally Unfunded
2009 2010 2011 2012 2013
0
4
8
12
16
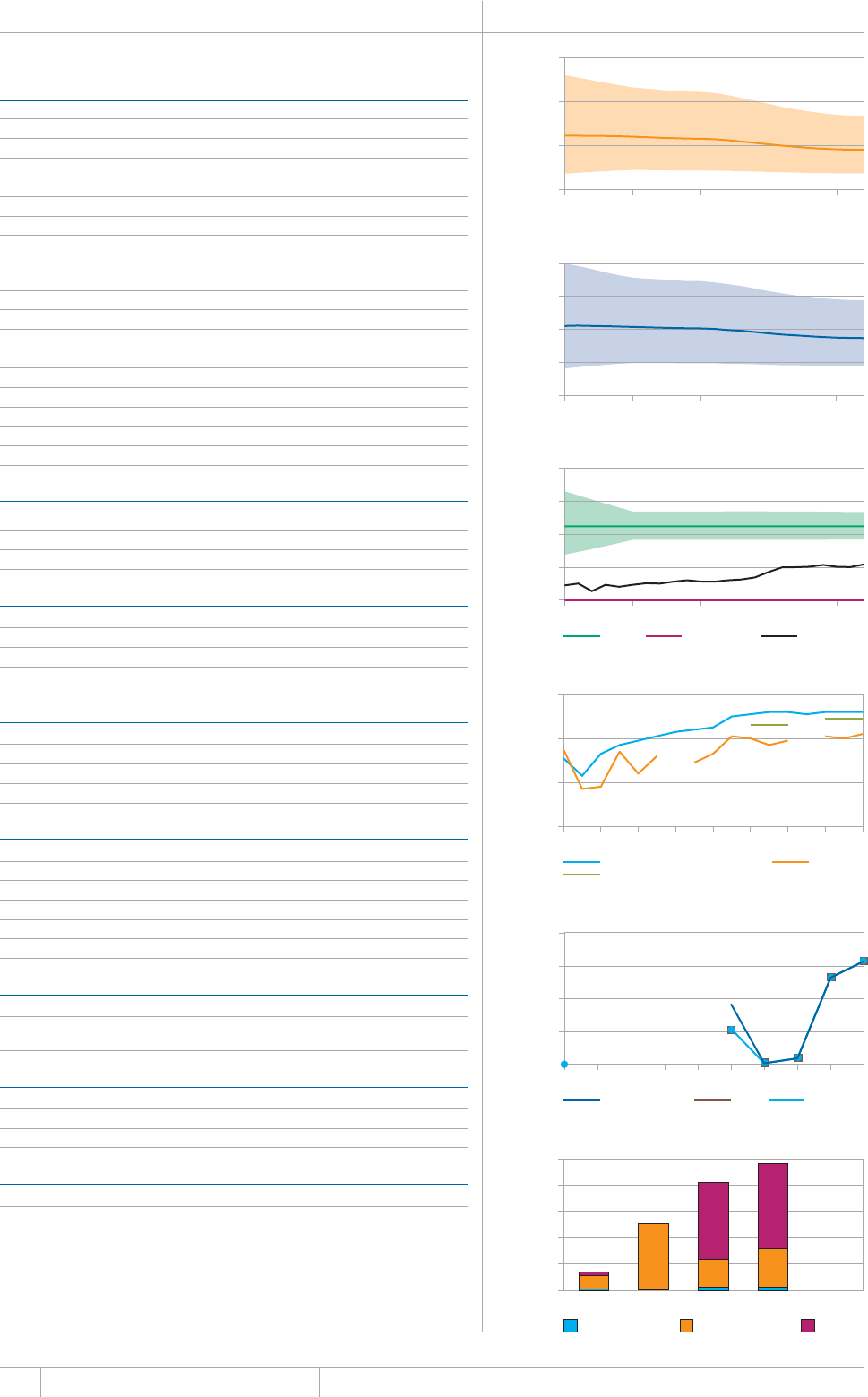
116 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
&CVCCTGCUTGRQTVGFVQ9*1'UVKOCVGUQH6$CPF/&46$DWTFGPCTGRTQFWEGFD[9*1KPEQPUWNVCVKQPYKVJ
countries.
a 4CPIGUTGRTGUGPVWPEGTVCKPV[KPVGTXCNU'UVKOCVGUQH6$FKUGCUGDWTFGPJCXGPQVDGGPCRRTQXGFD[
the national TB programme in Bangladesh and a joint reassessment will be undertaken following the
completion of the prevalence survey planned for 2014.
b Comprehensive data on domestic and international funding in 2013 could not be reported. Funding from
75#+&HQT1EVQDGT¿5GRVGODGTYCU75OKNNKQP
BANGLADESH Population 2012 155 million
HIGH TB BURDEN | HIGH MDR-TB BURDEN
Estimates of TB burdena 2012
07/$'46*175#0&5 4#6'(per 100 000 population)
/QTVCNKV[GZENWFGU*+86$ ¿ ¿
/QTVCNKV[*+86$QPN[ ¿ ¿
2TGXCNGPEGKPENWFGU*+86$ ¿ ¿
+PEKFGPEGKPENWFGU*+86$ ¿ ¿
Incidence (HIV+TB only) 0.24 (0.2–0.29) 0.16 (0.13–0.19)
Case detection, all forms (%) 49 (41–59)
6$ECUGPQVKßECVKQPU
0'9%#5'5 4'64'#6/'06%#5'5
5OGCTRQUKVKXG 4GNCRUG
5OGCTPGICVKXG 6TGCVOGPVCHVGTHCKNWTG
Smear-unknown / not done 0 (0) Treatment after default 257 (3)
'ZVTCRWNOQPCT[ 1VJGT
Other 0 (0)
Total new 161 790 Total retreatment 8 001
1VJGTJKUVQT[WPMPQYP
6QVCNPGYCPFTGNCRUG 6QVCNECUGUPQVKßGF
New cases
5/'#40')#6+8'70-0190
5/'#4215+6+8' 016&10' ':64#27./10#4;
/(TCVKQ
#IG
Laboratories 2012
Smear (per 100 000 population) 0.7
%WNVWTGRGTOKNNKQPRQRWNCVKQP
&TWIUWUEGRVKDKNKV[VGUVKPIRGTOKNNKQPRQRWNCVKQP
+UUGEQPFNKPGFTWIUWUEGRVKDKNKV[VGUVKPICXCKNCDNG! ;GUQWVUKFGEQWPVT[
6TGCVOGPVUWEEGUUTCVG
New smear-positive and/or culture-positive 92
0GYUOGCTPGICVKXGGZVTCRWNOQPCT[
4GVTGCVOGPV
+UTKHCORKEKPWUGFVJTQWIJQWVVTGCVOGPVHQTPGYRCVKGPVU! ;GU
TB/HIV 2012 07/$'4 (%)
6$RCVKGPVUYKVJMPQYP*+8UVCVWU
HIV-positive TB patients 63 (3)
*+8RQUKVKXG6$RCVKGPVUQPEQVTKOQZC\QNGRTGXGPVKXGVJGTCR[%26
*+8RQUKVKXG6$RCVKGPVUQPCPVKTGVTQXKTCNVJGTCR[#46
HIV-positive people screened for TB 429
HIV-positive people provided with IPT 0
Estimates of MDR-TB burden 2012a 0'9 4'64'#6/'06
QH6$ECUGUYKVJ/&46$ ¿ ¿
/&46$ECUGUCOQPIPQVKßGF
pulmonary TB cases 1 900 (920–3 300) 2 300 (1 900–2 700)
Reported cases of MDR-TB 20120'9 4'64'#6/'06 616#.
%CUGUVGUVGFHQT/&46$
.CDQTCVQT[EQPßTOGF/&46$ECUGU
2CVKGPVUUVCTVGFQP/&46$VTGCVOGPV
Financing TB controlb 2013
0CVKQPCN6$RTQITCOOGDWFIGV75OKNNKQPU
Mortality (excludes HIV+TB) (rate
per 100 000 population per year)
1990 1995 2000 2005 2010
0
50
100
150
Prevalence
(rate per 100 000 population)
1990 1995 2000 2005 2010
0
250
500
750
1000
Incidence (rate per 100 000
population per year)
1990 1995 2000 2005 2010
Incidence Incidence (HIV+TB) Notifications
0
100
200
300
400
Treatment success rate (%)
New smear-positive (and/or culture-positive) Retreatment
New smear-negative/extrapulmonary
40
60
80
100
1995 1997 1999 2001 2003 2005 2007 2009 2011
Number of patients
2003 2004 2005 2006 2007 2008 2009 2010 2011 2012
HIV-positive TB patients on CPT on ART
0
20
40
60
80
Total budget (US$ millions)
Funded domestically Funded internationally Unfunded
2009 2010 2011 2012
0
10
20
30
40
50
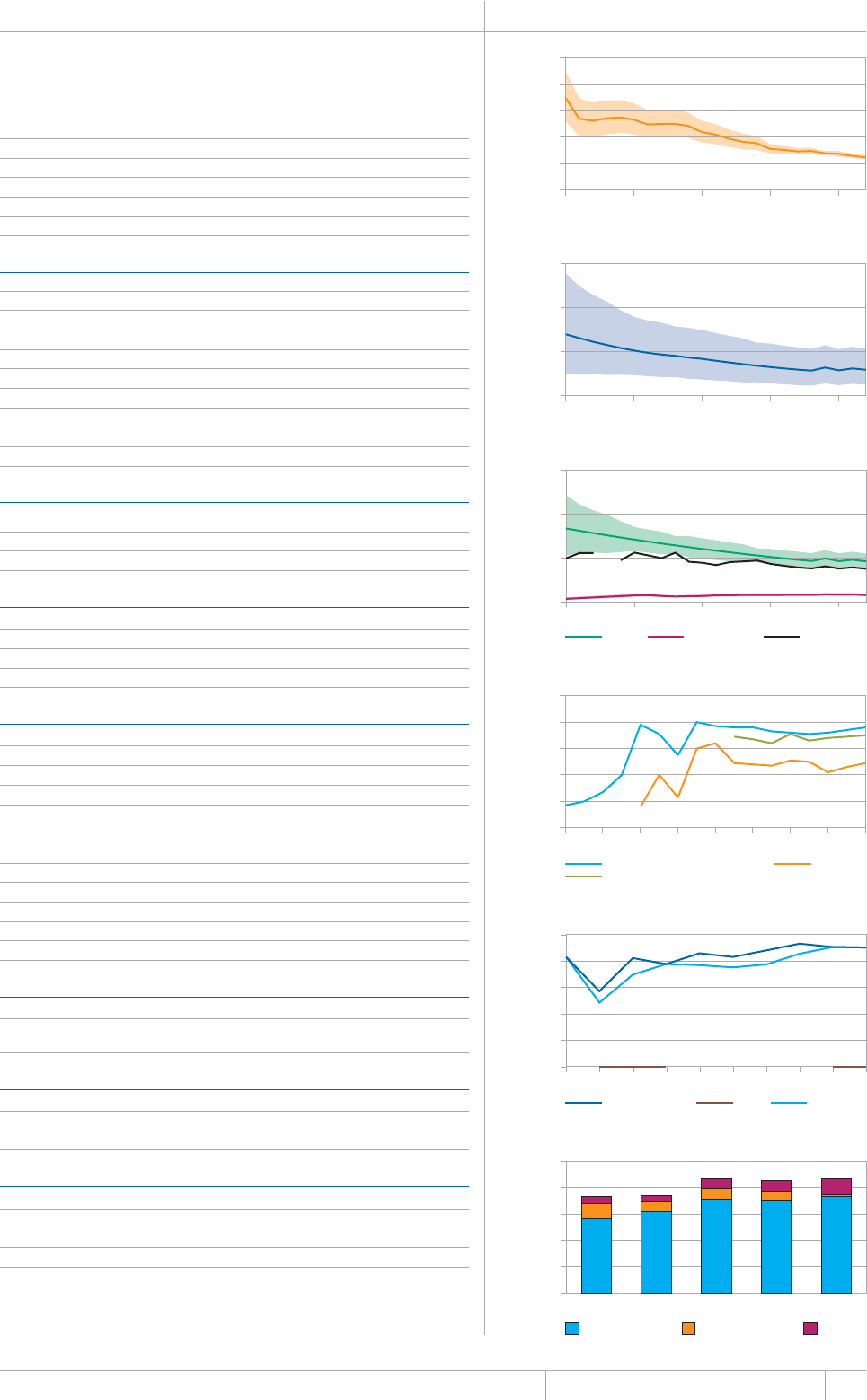
117 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
Data are as reported to WHO. Estimates of TB and MDR-TB burden are produced by WHO in consultation with
countries.
a Ranges represent uncertainty intervals.
BRAZIL Population 2012 199 million
HIGH TB BURDEN | HIGH HIV BURDEN
Estimates of TB burdena 2012
07/$'4(thousands)4#6' (per 100 000 population)
Mortality (excludes HIV+TB) 4.9 (4.6–5.2) 2.5 (2.3–2.6)
Mortality (HIV+TB only) 2.5 (2.2–3) 1.3 (1.1–1.5)
Prevalence (includes HIV+TB) 120 (51–210) 59 (25–107)
+PEKFGPEGKPENWFGU*+86$ ¿ ¿
+PEKFGPEG*+86$QPN[ ¿ ¿
%CUGFGVGEVKQPCNNHQTOU ¿
6$ECUGPQVKßECVKQPU
0'9%#5'5 4'64'#6/'06%#5'5
5OGCTRQUKVKXG 4GNCRUG
5OGCTPGICVKXG 6TGCVOGPVCHVGTHCKNWTG
5OGCTWPMPQYPPQVFQPG 6TGCVOGPVCHVGTFGHCWNV
Extrapulmonary 10 297 (14) Other 4 133 (36)
1VJGT
Total new 71 230 Total retreatment 11 500
Other (history unknown) 25
6QVCNPGYCPFTGNCRUG 6QVCNECUGUPQVKßGF
New cases
5/'#40')#6+8'70-0190
5/'#4215+6+8' 016&10' ':64#27./10#4;
/(TCVKQ
#IG
Laboratories 2012
Smear (per 100 000 population) 2.0
Culture (per 5 million population) 5.5
Drug susceptibility testing (per 5 million population) 0.9
+UUGEQPFNKPGFTWIUWUEGRVKDKNKV[VGUVKPICXCKNCDNG! ;GUKPEQWPVT[
6TGCVOGPVUWEEGUUTCVG
New smear-positive and/or culture-positive 76
New smear-negative/extrapulmonary 70
4GVTGCVOGPV
+UTKHCORKEKPWUGFVJTQWIJQWVVTGCVOGPVHQTPGYRCVKGPVU! ;GU
TB/HIV 2012 07/$'4 (%)
TB patients with known HIV status 45 733 (55)
HIV-positive TB patients 9 049 (20)
*+8RQUKVKXG6$RCVKGPVUQPEQVTKOQZC\QNGRTGXGPVKXGVJGTCR[%26
*+8RQUKVKXG6$RCVKGPVUQPCPVKTGVTQXKTCNVJGTCR[#46
HIV-positive people screened for TB
HIV-positive people provided with IPT
Estimates of MDR-TB burden 2012a 0'9 4'64'#6/'06
QH6$ECUGUYKVJ/&46$ ¿ ¿
/&46$ECUGUCOQPIPQVKßGF
RWNOQPCT[6$ECUGU ¿ ¿
Reported cases of MDR-TB 20120'9 4'64'#6/'06 616#.
%CUGUVGUVGFHQT/&46$
.CDQTCVQT[EQPßTOGF/&46$ECUGU
2CVKGPVUUVCTVGFQP/&46$VTGCVOGPV
Financing TB control 2013
0CVKQPCN6$RTQITCOOGDWFIGV75OKNNKQPU
(WPFGFFQOGUVKECNN[
% Funded internationally 2%
% Unfunded 14%
Mortality (excludes HIV+TB) (rate
per 100 000 population per year)
1990 1995 2000 2005 2010
0
2
4
6
8
10
Prevalence
(rate per 100 000 population)
1990 1995 2000 2005 2010
0
100
200
300
Incidence (rate per 100 000
population per year)
1990 1995 2000 2005 2010
Incidence Incidence (HIV+TB) Notifications
0
50
100
150
Treatment success rate (%)
New smear-positive (and/or culture-positive) Retreatment
New smear-negative/extrapulmonary
1995 1997 1999 2001 2003 2005 2007 2009 2011
0
20
40
60
80
100
Number of patients
2003 2004 2005 2006 2007 2008 2009 2010 2011 2012
HIV-positive TB patients on CPT on ART
0
2000
4000
6000
8000
10 000
Total budget (US$ millions)
Funded domestically Funded internationally Unfunded
2009 2010 2011 2012 2013
0
20
40
60
80
100
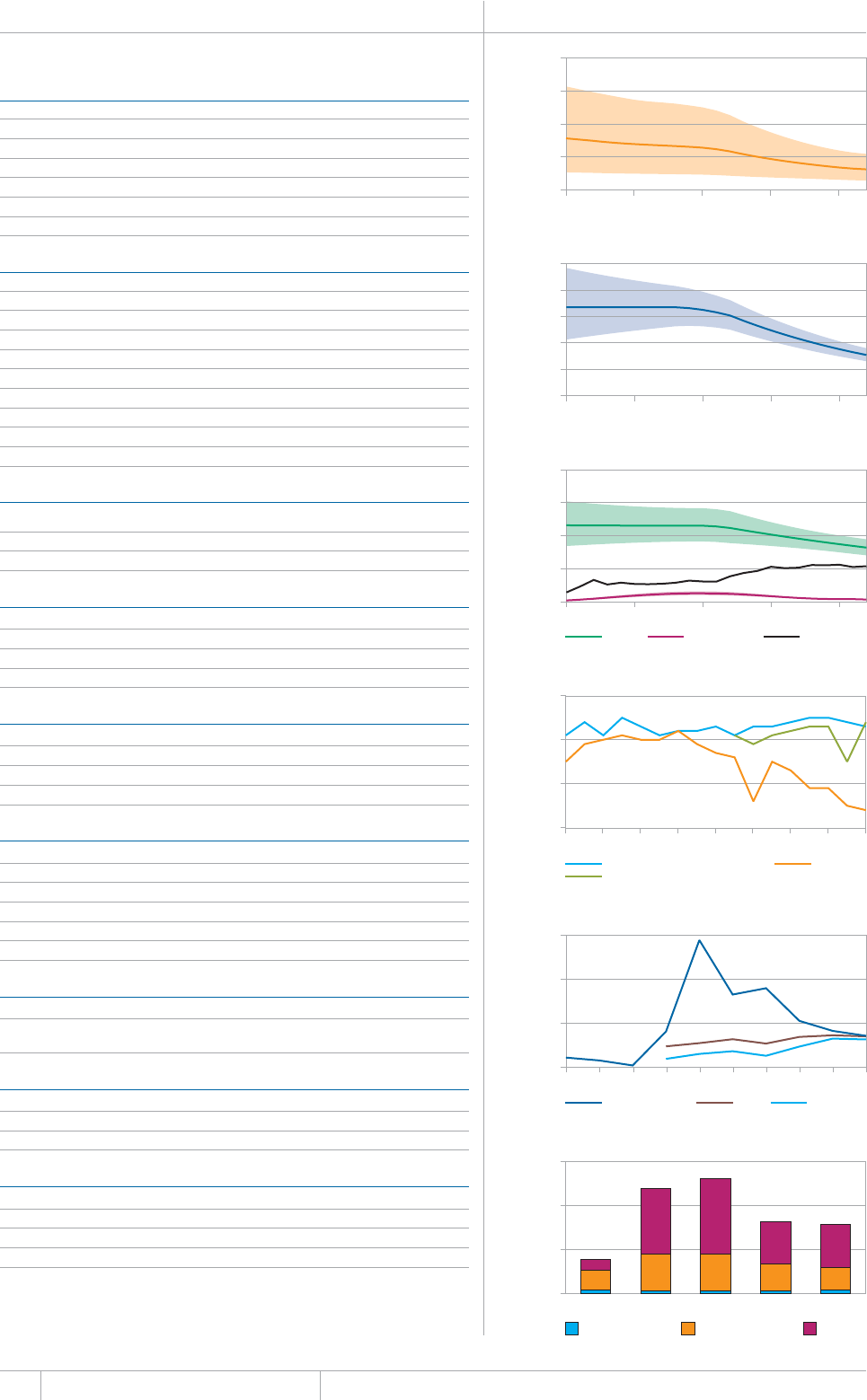
GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
Data are as reported to WHO. Estimates of TB and MDR-TB burden are produced by WHO in consultation with
countries.
a Ranges represent uncertainty intervals.
CAMBODIA Population 2012 15 million
HIGH TB BURDEN | HIGH HIV BURDEN
Estimates of TB burdena 2012
07/$'4(thousands)4#6'(per 100 000 population)
Mortality (excludes HIV+TB) 9.3 (4.3–16) 63 (29–110)
/QTVCNKV[*+86$QPN[ ¿ ¿
2TGXCNGPEGKPENWFGU*+86$ ¿ ¿
Incidence (includes HIV+TB) 61 (52–70) 411 (353–474)
+PEKFGPEG*+86$QPN[ ¿ ¿
Case detection, all forms (%) 66 (57–77)
6$ECUGPQVKßECVKQPU
0'9%#5'5 4'64'#6/'06%#5'5
5OGCTRQUKVKXG 4GNCRUG
5OGCTPGICVKXG 6TGCVOGPVCHVGTHCKNWTG
Smear-unknown / not done 0 (0) Treatment after default 22 (4)
Extrapulmonary 15 290 (40) Other
Other 0 (0)
Total new 38 637 Total retreatment 519
Other (history unknown) 1 102
6QVCNPGYCPFTGNCRUG 6QVCNECUGUPQVKßGF
New cases
5/'#40')#6+8'70-0190
5/'#4215+6+8' 016&10' ':64#27./10#4;
/(TCVKQ
#IG
Laboratories 2012
Smear (per 100 000 population) 1.4
Culture (per 5 million population) 1.0
Drug susceptibility testing (per 5 million population) 0.3
Is second-line drug susceptibility testing available? No
6TGCVOGPVUWEEGUUTCVG
New smear-positive and/or culture-positive 93
New smear-negative/extrapulmonary 94
4GVTGCVOGPV
+UTKHCORKEKPWUGFVJTQWIJQWVVTGCVOGPVHQTPGYRCVKGPVU! ;GU
TB/HIV 2012 07/$'4 (%)
6$RCVKGPVUYKVJMPQYP*+8UVCVWU
HIV-positive TB patients 1 433 (4)
*+8RQUKVKXG6$RCVKGPVUQPEQVTKOQZC\QNGRTGXGPVKXGVJGTCR[%26
*+8RQUKVKXG6$RCVKGPVUQPCPVKTGVTQXKTCNVJGTCR[#46
HIV-positive people screened for TB
HIV-positive people provided with IPT 1 145
Estimates of MDR-TB burden 2012a 0'9 4'64'#6/'06
QH6$ECUGUYKVJ/&46$ ¿ ¿
/&46$ECUGUCOQPIPQVKßGF
pulmonary TB cases 330 (160–590) 56 (21–110)
Reported cases of MDR-TB 20120'9 4'64'#6/'06 616#.
%CUGUVGUVGFHQT/&46$
.CDQTCVQT[EQPßTOGF/&46$ECUGU
2CVKGPVUUVCTVGFQP/&46$VTGCVOGPV
Financing TB control 2013
0CVKQPCN6$RTQITCOOGDWFIGV75OKNNKQPU
% Funded domestically 5%
% Funded internationally 34%
% Unfunded 62%
Mortality (excludes HIV+TB) (rate
per 100 000 population per year)
1990 1995 2000 2005 2010
0
100
200
300
400
Prevalence
(rate per 100 000 population)
1990 1995 2000 2005 2010
0
500
1000
1500
2000
2500
Incidence (rate per 100 000
population per year)
1990 1995 2000 2005 2010
Incidence Incidence (HIV+TB) Notifications
0
250
500
750
1000
Treatment success rate (%)
New smear-positive (and/or culture-positive) Retreatment
New smear-negative/extrapulmonary
1995 1997 1999 2001 2003 2005 2007 2009 2011
70
80
90
100
Number of patients
2003 2004 2005 2006 2007 2008 2009 2010 2011 2012
HIV-positive TB patients on CPT on ART
0
2000
4000
6000
Total budget (US$ millions)
Funded domestically Funded internationally Unfunded
2009 2010 2011 2012 2013
0
15
30
45
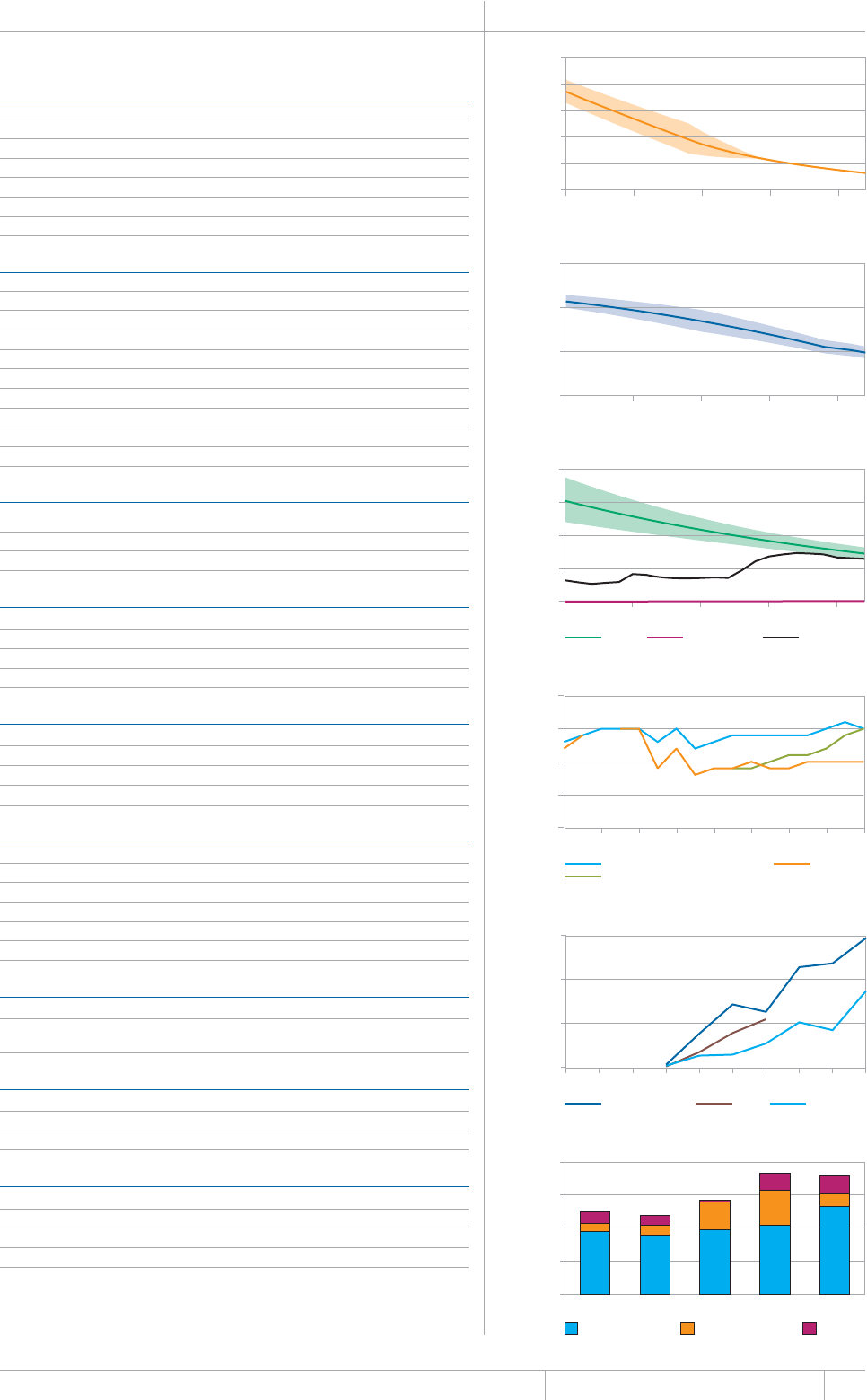
119 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
Data are as reported to WHO. Estimates of TB and MDR-TB burden are produced by WHO in consultation with
countries.
a Ranges represent uncertainty intervals.
CHINA Population 2012 1 377 million
HIGH TB BURDEN | HIGH HIV BURDEN | HIGH MDR-TB BURDEN
Estimates of TB burdena 2012
07/$'4(thousands)4#6' (per 100 000 population)
Mortality (excludes HIV+TB) 44 (43–46) 3.2 (3.1–3.3)
/QTVCNKV[*+86$QPN[ ¿ ¿
2TGXCNGPEGKPENWFGU*+86$ ¿ ¿
+PEKFGPEGKPENWFGU*+86$ ¿ ¿
+PEKFGPEG*+86$QPN[ ¿ ¿
%CUGFGVGEVKQPCNNHQTOU ¿
6$ECUGPQVKßECVKQPU
0'9%#5'5 4'64'#6/'06%#5'5
5OGCTRQUKVKXG 4GNCRUG
5OGCTPGICVKXG 6TGCVOGPVCHVGTHCKNWTG
5OGCTWPMPQYPPQVFQPG 6TGCVOGPVCHVGTFGHCWNV
'ZVTCRWNOQPCT[ 1VJGT
Other 0 (0)
Total new 858 861 Total retreatment 41 817
Other (history unknown) 0
6QVCNPGYCPFTGNCRUG 6QVCNECUGUPQVKßGF
New cases
5/'#40')#6+8'70-0190
5/'#4215+6+8' 016&10' ':64#27./10#4;
/(TCVKQ
#IG
Laboratories 2012
Smear (per 100 000 population) 0.2
Culture (per 5 million population) 3.7
Drug susceptibility testing (per 5 million population) 0.7
+UUGEQPFNKPGFTWIUWUEGRVKDKNKV[VGUVKPICXCKNCDNG! ;GUKPEQWPVT[
6TGCVOGPVUWEEGUUTCVG
New smear-positive and/or culture-positive 95
New smear-negative/extrapulmonary 95
4GVTGCVOGPV
+UTKHCORKEKPWUGFVJTQWIJQWVVTGCVOGPVHQTPGYRCVKGPVU! ;GU
TB/HIV 2012 07/$'4 (%)
TB patients with known HIV statusb
*+8RQUKVKXG6$RCVKGPVU
*+8RQUKVKXG6$RCVKGPVUQPEQVTKOQZC\QNGRTGXGPVKXGVJGTCR[%26
*+8RQUKVKXG6$RCVKGPVUQPCPVKTGVTQXKTCNVJGTCR[#46
HIV-positive people screened for TB 294 795
HIV-positive people provided with IPT
Estimates of MDR-TB burden 2012a 0'9 4'64'#6/'06
QH6$ECUGUYKVJ/&46$ ¿ ¿
/&46$ECUGUCOQPIPQVKßGF
RWNOQPCT[6$ECUGU ¿ ¿
Reported cases of MDR-TB 20120'9 4'64'#6/'06 616#.
%CUGUVGUVGFHQT/&46$
.CDQTCVQT[EQPßTOGF/&46$ECUGU
2CVKGPVUUVCTVGFQP/&46$VTGCVOGPV
Financing TB control 2013
0CVKQPCN6$RTQITCOOGDWFIGV75OKNNKQPU
% Funded domestically 74%
% Funded internationally 11%
% Unfunded 15%
Mortality (excludes HIV+TB) (rate
per 100 000 population per year)
1990 1995 2000 2005 2010
0
5
10
15
20
25
Prevalence
(rate per 100 000 population)
1990 1995 2000 2005 2010
0
100
200
300
Incidence (rate per 100 000
population per year)
1990 1995 2000 2005 2010
Incidence Incidence (HIV+TB) Notifications
0
50
100
150
200
Treatment success rate (%)
New smear-positive (and/or culture-positive) Retreatment
New smear-negative/extrapulmonary
1995 1997 1999 2001 2003 2005 2007 2009 2011
80
85
90
95
100
Number of patients
2003 2004 2005 2006 2007 2008 2009 2010 2011 2012
HIV-positive TB patients on CPT on ART
0
2000
4000
6000
Total budget (US$ millions)
Funded domestically Funded internationally Unfunded
2009 2010 2011 2012 2013
0
100
200
300
400
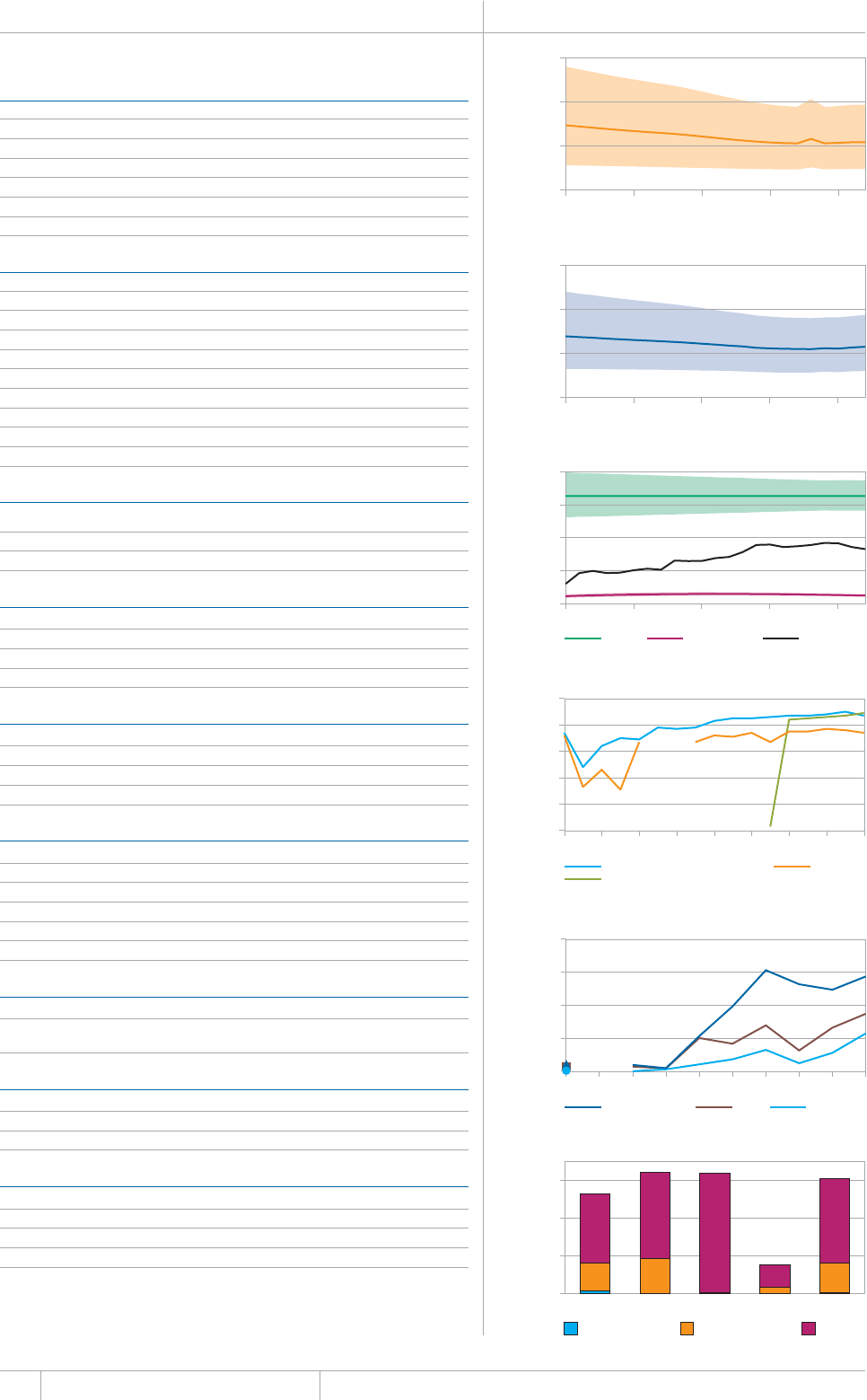
120 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
Data are as reported to WHO. Estimates of TB and MDR-TB burden are produced by WHO in consultation with
countries.
a Ranges represent uncertainty intervals.
DEMOCRATIC REPUBLIC OF THE CONGO Population 2012 66 million
HIGH TB BURDEN | HIGH HIV BURDEN | HIGH MDR-TB BURDEN
Estimates of TB burdena 2012
07/$'4(thousands)4#6'(per 100 000 population)
Mortality (excludes HIV+TB) 36 (16–64) 54 (24–97)
/QTVCNKV[*+86$QPN[ ¿ ¿
2TGXCNGPEGKPENWFGU*+86$ ¿ ¿
+PEKFGPEGKPENWFGU*+86$ ¿ ¿
Incidence (HIV+TB only) 16 (14–19) 25 (22–29)
Case detection, all forms (%) 51 (44–59)
6$ECUGPQVKßECVKQPU
0'9%#5'5 4'64'#6/'06%#5'5
5OGCTRQUKVKXG 4GNCRUG
5OGCTPGICVKXG 6TGCVOGPVCHVGTHCKNWTG
5OGCTWPMPQYPPQVFQPG 6TGCVOGPVCHVGTFGHCWNV
Extrapulmonary 20 669 (20) Other 2 321 (31)
Other
Total new 105 007 Total retreatment 7 492
Other (history unknown)
6QVCNPGYCPFTGNCRUG 6QVCNECUGUPQVKßGF
New cases
5/'#40')#6+8'70-0190
5/'#4215+6+8' 016&10' ':64#27./10#4;
/(TCVKQ
#IG
Laboratories 2012
Smear (per 100 000 population) 2.3
Culture (per 5 million population) 0.3
Drug susceptibility testing (per 5 million population) 0.2
+UUGEQPFNKPGFTWIUWUEGRVKDKNKV[VGUVKPICXCKNCDNG! ;GUKPCPFQWVUKFGEQWPVT[
6TGCVOGPVUWEEGUUTCVG
0GYUOGCTRQUKVKXGCPFQTEWNVWTGRQUKVKXG
0GYUOGCTPGICVKXGGZVTCRWNOQPCT[
4GVTGCVOGPV
+UTKHCORKEKPWUGFVJTQWIJQWVVTGCVOGPVHQTPGYRCVKGPVU! ;GU
TB/HIV 2012 07/$'4 (%)
TB patients with known HIV status 35 097 (31)
*+8RQUKVKXG6$RCVKGPVU
*+8RQUKVKXG6$RCVKGPVUQPEQVTKOQZC\QNGRTGXGPVKXGVJGTCR[%26
*+8RQUKVKXG6$RCVKGPVUQPCPVKTGVTQXKTCNVJGTCR[#46
HIV-positive people screened for TB
HIV-positive people provided with IPT
Estimates of MDR-TB burden 2012a 0'9 4'64'#6/'06
QH6$ECUGUYKVJ/&46$ ¿ ¿
/&46$ECUGUCOQPIPQVKßGF
RWNOQPCT[6$ECUGU ¿ ¿
Reported cases of MDR-TB 20120'9 4'64'#6/'06 616#.
%CUGUVGUVGFHQT/&46$
.CDQTCVQT[EQPßTOGF/&46$ECUGU
2CVKGPVUUVCTVGFQP/&46$VTGCVOGPV
Financing TB control 2013
0CVKQPCN6$RTQITCOOGDWFIGV75OKNNKQPU
% Funded domestically 1%
% Funded internationally 25%
% Unfunded 74%
Mortality (excludes HIV+TB) (rate
per 100 000 population per year)
1990 1995 2000 2005 2010
0
50
100
150
Prevalence
(rate per 100 000 population)
1990 1995 2000 2005 2010
0
500
1000
1500
Incidence (rate per 100 000
population per year)
1990 1995 2000 2005 2010
Incidence Incidence (HIV+TB) Notifications
0
100
200
300
400
Treatment success rate (%)
New smear-positive (and/or culture-positive) Retreatment
New smear-negative/extrapulmonary
1995 1997 1999 2001 2003 2005 2007 2009 2011
0
20
40
60
80
100
Number of patients
2003 2004 2005 2006 2007 2008 2009 2010 2011 2012
HIV-positive TB patients on CPT on ART
0
2000
4000
6000
8000
Total budget (US$ millions)
Funded domestically Funded internationally Unfunded
2009 2010 2011 2012 2013
0
20
40
60
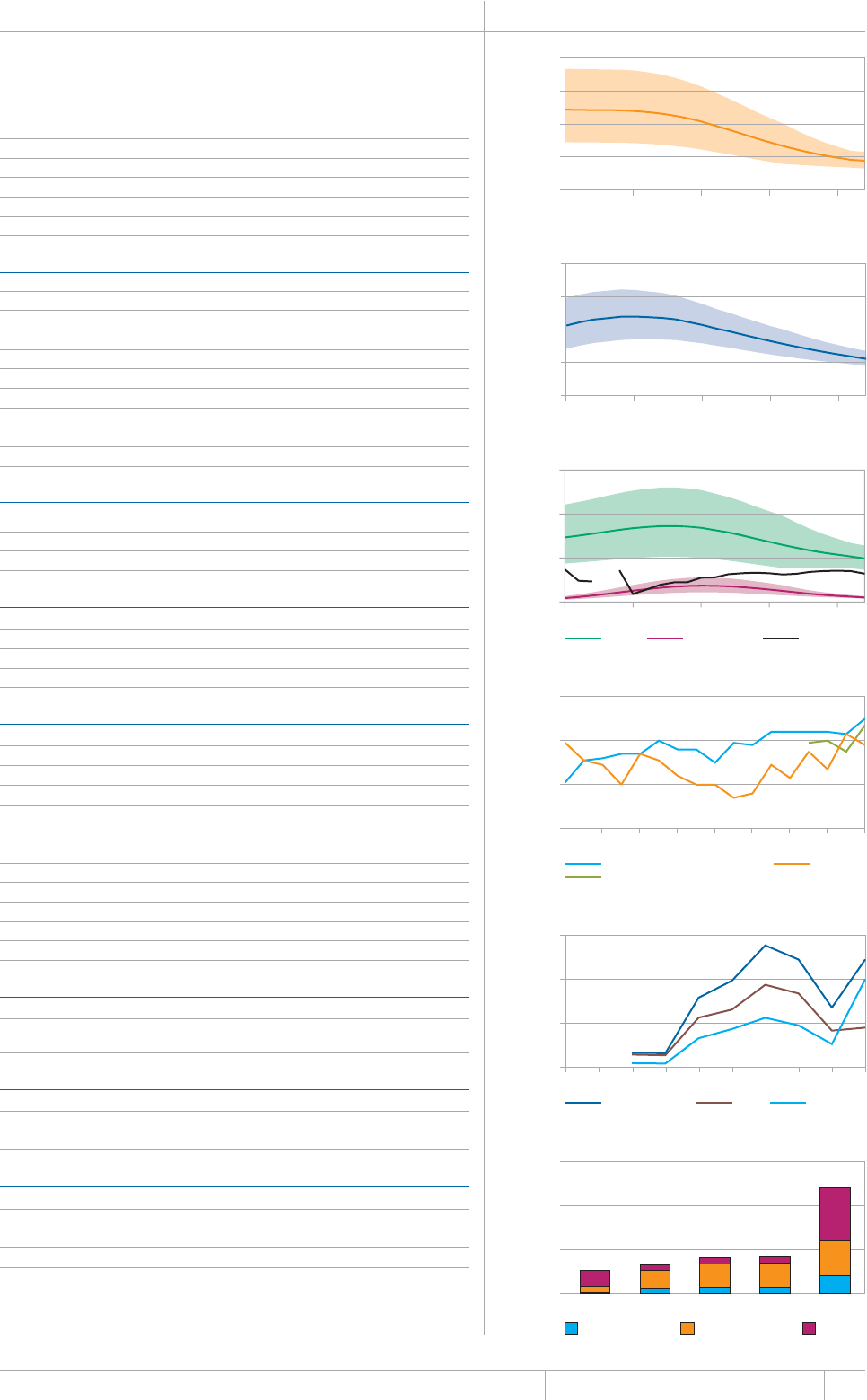
121 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
Data are as reported to WHO. Estimates of TB and MDR-TB burden are produced by WHO in consultation with
countries.
a Ranges represent uncertainty intervals.
ETHIOPIA Population 2012 92 million
HIGH TB BURDEN | HIGH HIV BURDEN | HIGH MDR-TB BURDEN
Estimates of TB burdena 2012
07/$'4(thousands)4#6' (per 100 000 population)
/QTVCNKV[GZENWFGU*+86$ ¿ ¿
/QTVCNKV[*+86$QPN[ ¿ ¿
2TGXCNGPEGKPENWFGU*+86$ ¿ ¿
+PEKFGPEGKPENWFGU*+86$ ¿ ¿
Incidence (HIV+TB only) 23 (17–30) 25 (19–33)
%CUGFGVGEVKQPCNNHQTOU ¿
6$ECUGPQVKßECVKQPU
0'9%#5'5 4'64'#6/'06%#5'5
5OGCTRQUKVKXG 4GNCRUG
5OGCTPGICVKXG 6TGCVOGPVCHVGTHCKNWTG
5OGCTWPMPQYPPQVFQPG 6TGCVOGPVCHVGTFGHCWNV
'ZVTCRWNOQPCT[ 1VJGT
Other 0 (0)
Total new 143 503 Total retreatment 4 089
Other (history unknown) 0
6QVCNPGYCPFTGNCRUG 6QVCNECUGUPQVKßGF
New cases
5/'#40')#6+8'70-0190
5/'#4215+6+8' 016&10' ':64#27./10#4;
/(TCVKQ
#IG
Laboratories 2012
5OGCTRGTRQRWNCVKQP
Culture (per 5 million population) 0.3
&TWIUWUEGRVKDKNKV[VGUVKPIRGTOKNNKQPRQRWNCVKQP
Is second-line drug susceptibility testing available? No
6TGCVOGPVUWEEGUUTCVG
New smear-positive and/or culture-positive 90
0GYUOGCTPGICVKXGGZVTCRWNOQPCT[
4GVTGCVOGPV
+UTKHCORKEKPWUGFVJTQWIJQWVVTGCVOGPVHQTPGYRCVKGPVU! ;GU
TB/HIV 2012 07/$'4 (%)
TB patients with known HIV status 96 245 (65)
*+8RQUKVKXG6$RCVKGPVU
*+8RQUKVKXG6$RCVKGPVUQPEQVTKOQZC\QNGRTGXGPVKXGVJGTCR[%26
*+8RQUKVKXG6$RCVKGPVUQPCPVKTGVTQXKTCNVJGTCR[#46
*+8RQUKVKXGRGQRNGUETGGPGFHQT6$
HIV-positive people provided with IPT 30 395
Estimates of MDR-TB burden 2012a 0'9 4'64'#6/'06
QH6$ECUGUYKVJ/&46$ ¿ ¿
/&46$ECUGUCOQPIPQVKßGF
RWNOQPCT[6$ECUGU ¿ ¿
Reported cases of MDR-TB 20120'9 4'64'#6/'06 616#.
%CUGUVGUVGFHQT/&46$
.CDQTCVQT[EQPßTOGF/&46$ECUGU
2CVKGPVUUVCTVGFQP/&46$VTGCVOGPV
Financing TB control 2013
0CVKQPCN6$RTQITCOOGDWFIGV75OKNNKQPU
% Funded domestically 17%
% Funded internationally 32%
% Unfunded 51%
Mortality (excludes HIV+TB) (rate
per 100 000 population per year)
1990 1995 2000 2005 2010
0
20
40
60
80
Prevalence
(rate per 100 000 population)
1990 1995 2000 2005 2010
0
200
400
600
800
Incidence (rate per 100 000
population per year)
1990 1995 2000 2005 2010
Incidence Incidence (HIV+TB) Notifications
0
250
500
750
Treatment success rate (%)
New smear-positive (and/or culture-positive) Retreatment
New smear-negative/extrapulmonary
1995 1997 1999 2001 2003 2005 2007 2009 2011
40
60
80
100
Number of patients
2003 2004 2005 2006 2007 2008 2009 2010 2011 2012
HIV-positive TB patients on CPT on ART
0
4000
8000
12 000
Total budget (US$ millions)
Funded domestically Funded internationally Unfunded
2009 2010 2011 2012 2013
0
60
120
180
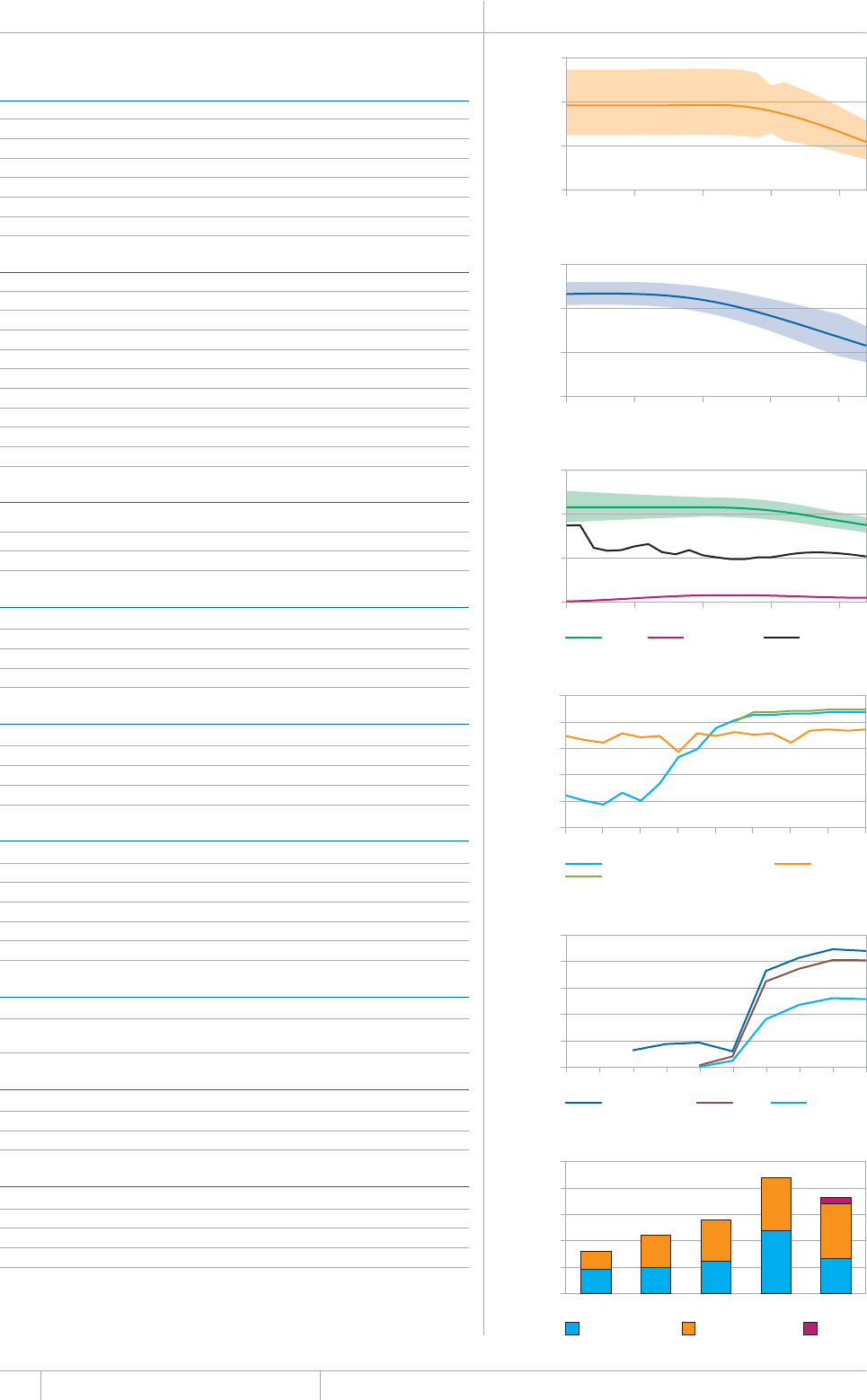
122 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
&CVCCTGCUTGRQTVGFVQ9*1'UVKOCVGUQH6$CPF/&46$DWTFGPCTGRTQFWEGFD[9*1KPEQPUWNVCVKQPYKVJ
countries.
a 4CPIGUTGRTGUGPVWPEGTVCKPV[KPVGTXCNU'UVKOCVGUHQT+PFKCJCXGPQV[GVDGGPQHßEKCNN[CRRTQXGFD[VJG
/KPKUVT[QH*GCNVJ(COKN[9GNHCTG)QXGTPOGPVQH+PFKCCPFUJQWNFVJGTGHQTGDGEQPUKFGTGFRTQXKUKQPCN
INDIA Population 2012 1 237 million
HIGH TB BURDEN | HIGH HIV BURDEN | HIGH MDR-TB BURDEN
Estimates of TB burdena 2012
07/$'4(thousands)4#6'(per 100 000 population)
Mortality (excludes HIV+TB) 270 (170–390) 22 (14–32)
/QTVCNKV[*+86$QPN[ ¿ ¿
2TGXCNGPEGKPENWFGU*+86$ ¿ ¿
Incidence (includes HIV+TB) 2 200 (2 000–2 400) 176 (159–193)
Incidence (HIV+TB only) 130 (120–140) 10 (9.4–12)
Case detection, all forms (%) 59 (54–66)
6$ECUGPQVKßECVKQPU
0'9%#5'5 4'64'#6/'06%#5'5
5OGCTRQUKVKXG 4GNCRUG
Smear-negative 317 616 (27) Treatment after failure 16 400 (6)
5OGCTWPMPQYPPQVFQPG 6TGCVOGPVCHVGTFGHCWNV
Extrapulmonary 234 029 (20) Other 96 567 (34)
1VJGT
Total new 1 183 373 Total retreatment 284 212
Other (history unknown)
6QVCNPGYCPFTGNCRUG 6QVCNECUGUPQVKßGF
New cases
5/'#40')#6+8'70-0190
5/'#4215+6+8' 016&10' ':64#27./10#4;
/(TCVKQ
#IG
Laboratories 2012
Smear (per 100 000 population) 1.1
Culture (per 5 million population) 0.3
Drug susceptibility testing (per 5 million population) 0.2
+UUGEQPFNKPGFTWIUWUEGRVKDKNKV[VGUVKPICXCKNCDNG! ;GUKPEQWPVT[
6TGCVOGPVUWEEGUUTCVG
0GYUOGCTRQUKVKXGCPFQTEWNVWTGRQUKVKXG
New smear-negative/extrapulmonary 90
4GVTGCVOGPV
+UTKHCORKEKPWUGFVJTQWIJQWVVTGCVOGPVHQTPGYRCVKGPVU! ;GU
TB/HIV 2012 07/$'4 (%)
6$RCVKGPVUYKVJMPQYP*+8UVCVWU
HIV-positive TB patients 44 063 (5)
*+8RQUKVKXG6$RCVKGPVUQPEQVTKOQZC\QNGRTGXGPVKXGVJGTCR[%26
*+8RQUKVKXG6$RCVKGPVUQPCPVKTGVTQXKTCNVJGTCR[#46
*+8RQUKVKXGRGQRNGUETGGPGFHQT6$
HIV-positive people provided with IPT
Estimates of MDR-TB burden 2012a 0'9 4'64'#6/'06
QH6$ECUGUYKVJ/&46$ ¿ ¿
/&46$ECUGUCOQPIPQVKßGF
RWNOQPCT[6$ECUGU ¿ ¿
Reported cases of MDR-TB 20120'9 4'64'#6/'06 616#.
%CUGUVGUVGFHQT/&46$
.CDQTCVQT[EQPßTOGF/&46$ECUGU
2CVKGPVUUVCTVGFQP/&46$VTGCVOGPV
Financing TB control 2013
0CVKQPCN6$RTQITCOOGDWFIGV75OKNNKQPU
% Funded domestically 37%
% Funded internationally 57%
% Unfunded 6%
Mortality (excludes HIV+TB) (rate
per 100 000 population per year)
1990 1995 2000 2005 2010
0
20
40
60
Prevalence
(rate per 100 000 population)
1990 1995 2000 2005 2010
0
200
400
600
Incidence (rate per 100 000
population per year)
1990 1995 2000 2005 2010
Incidence Incidence (HIV+TB) Notifications
0
100
200
300
Treatment success rate (%)
New smear-positive (and/or culture-positive) Retreatment
New smear-negative/extrapulmonary
1995 1997 1999 2001 2003 2005 2007 2009 2011
0
20
40
60
80
100
Number of patients
2003 2004 2005 2006 2007 2008 2009 2010 2011 2012
HIV-positive TB patients on CPT on ART
0
10 000
20 000
30 000
40 000
50 000
Total budget (US$ millions)
Funded domestically Funded internationally Unfunded
2009 2010 2011 2012 2013
0
50
100
150
200
250
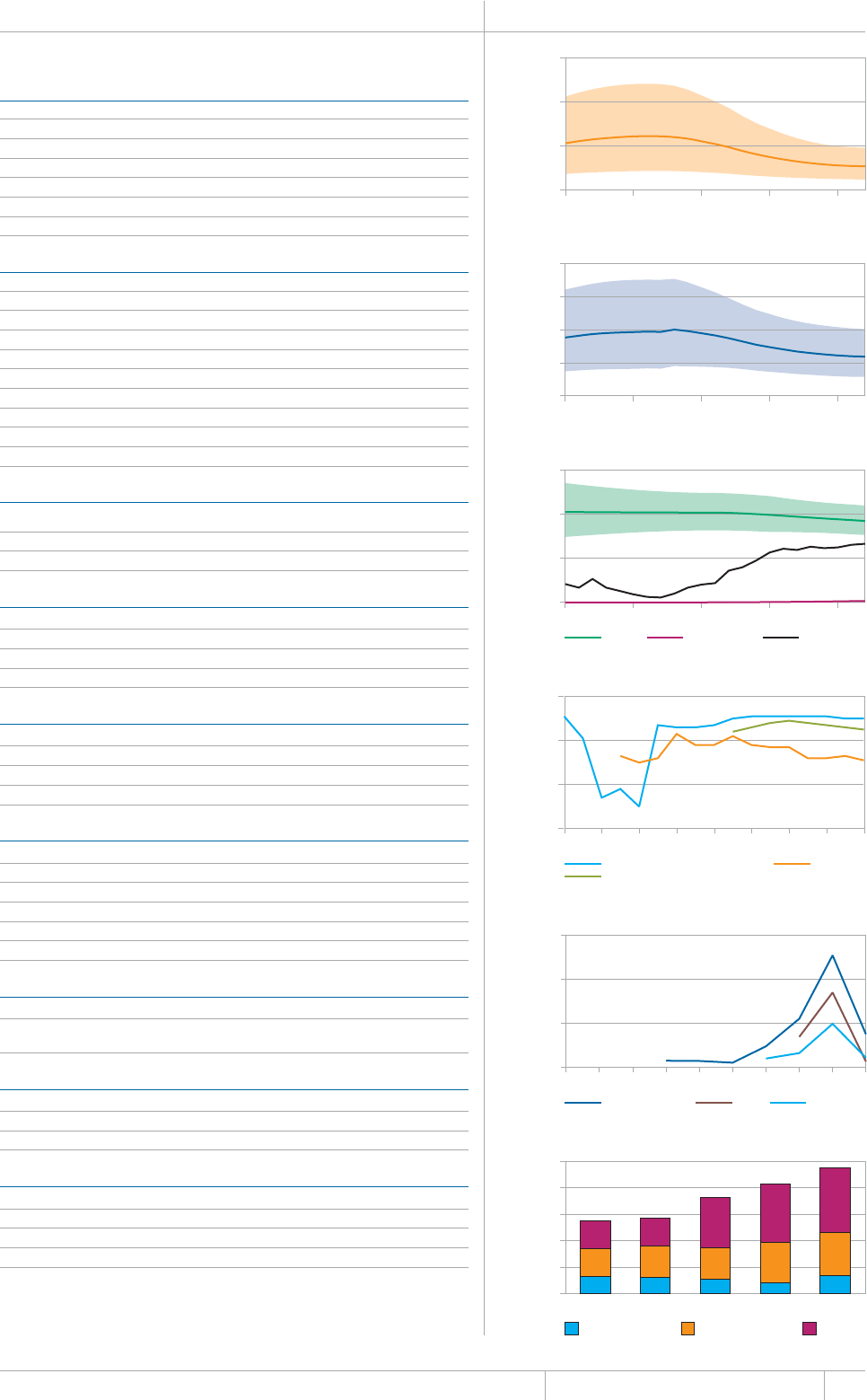
123 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
Data are as reported to WHO. Estimates of TB and MDR-TB burden are produced by WHO in consultation with
countries.
a Ranges represent uncertainty intervals.
INDONESIA Population 2012 247 million
HIGH TB BURDEN | HIGH HIV BURDEN | HIGH MDR-TB BURDEN
Estimates of TB burdena 2012
07/$'4(thousands)4#6'(per 100 000 population)
/QTVCNKV[GZENWFGU*+86$ ¿ ¿
/QTVCNKV[*+86$QPN[ ¿ ¿
Prevalence (includes HIV+TB) 730 (350–1 200) 297 (144–506)
+PEKFGPEGKPENWFGU*+86$ ¿ ¿
Incidence (HIV+TB only) 7.5 (5.6–9.7) 3.1 (2.3–3.9)
%CUGFGVGEVKQPCNNHQTOU ¿
6$ECUGPQVKßECVKQPU
0'9%#5'5 4'64'#6/'06%#5'5
5OGCTRQUKVKXG 4GNCRUG
5OGCTPGICVKXG 6TGCVOGPVCHVGTHCKNWTG
Smear-unknown / not done Treatment after default 954 (11)
Extrapulmonary 15 697 (5) Other 1 179 (14)
Other
Total new 322 882 Total retreatment 8 542
Other (history unknown)
6QVCNPGYCPFTGNCRUG 6QVCNECUGUPQVKßGF
New cases
5/'#40')#6+8'70-0190
5/'#4215+6+8' 016&10' ':64#27./10#4;
/(TCVKQ
#IG
Laboratories 2012
Smear (per 100 000 population) 2.3
Culture (per 5 million population) 0.9
Drug susceptibility testing (per 5 million population) 0.1
+UUGEQPFNKPGFTWIUWUEGRVKDKNKV[VGUVKPICXCKNCDNG! ;GUKPEQWPVT[
6TGCVOGPVUWEEGUUTCVG
New smear-positive and/or culture-positive 90
0GYUOGCTPGICVKXGGZVTCRWNOQPCT[
4GVTGCVOGPV
+UTKHCORKEKPWUGFVJTQWIJQWVVTGCVOGPVHQTPGYRCVKGPVU! ;GU
TB/HIV 2012 07/$'4 (%)
6$RCVKGPVUYKVJMPQYP*+8UVCVWU
*+8RQUKVKXG6$RCVKGPVU
*+8RQUKVKXG6$RCVKGPVUQPEQVTKOQZC\QNGRTGXGPVKXGVJGTCR[%26
*+8RQUKVKXG6$RCVKGPVUQPCPVKTGVTQXKTCNVJGTCR[#46
HIV-positive people screened for TB 22 677
HIV-positive people provided with IPT
Estimates of MDR-TB burden 2012a 0'9 4'64'#6/'06
QH6$ECUGUYKVJ/&46$ ¿ ¿
/&46$ECUGUCOQPIPQVKßGF
RWNOQPCT[6$ECUGU ¿ ¿
Reported cases of MDR-TB 20120'9 4'64'#6/'06 616#.
%CUGUVGUVGFHQT/&46$
.CDQTCVQT[EQPßTOGF/&46$ECUGU
2CVKGPVUUVCTVGFQP/&46$VTGCVOGPV
Financing TB control 2013
0CVKQPCN6$RTQITCOOGDWFIGV75OKNNKQPU
% Funded domestically 14%
% Funded internationally 35%
% Unfunded 51%
Mortality (excludes HIV+TB) (rate
per 100 000 population per year)
1990 1995 2000 2005 2010
0
50
100
150
Prevalence
(rate per 100 000 population)
1990 1995 2000 2005 2010
0
250
500
750
1000
Incidence (rate per 100 000
population per year)
1990 1995 2000 2005 2010
Incidence Incidence (HIV+TB) Notifications
0
100
200
300
Treatment success rate (%)
New smear-positive (and/or culture-positive) Retreatment
New smear-negative/extrapulmonary
1995 1997 1999 2001 2003 2005 2007 2009 2011
40
60
80
100
Number of patients
2003 2004 2005 2006 2007 2008 2009 2010 2011 2012
HIV-positive TB patients on CPT on ART
0
1000
2000
3000
Total budget (US$ millions)
Funded domestically Funded internationally Unfunded
2009 2010 2011 2012 2013
0
25
50
75
100
125
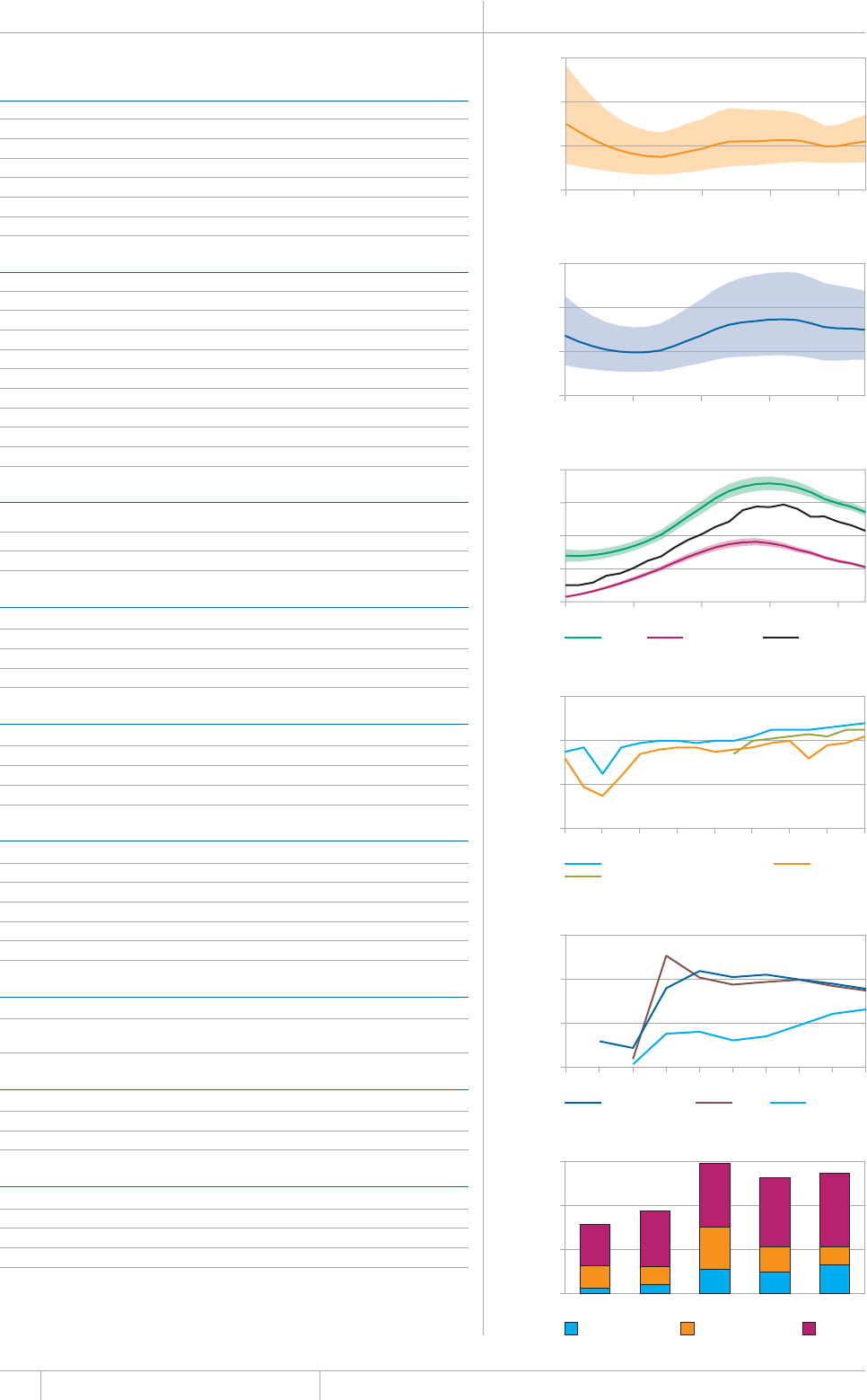
124 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
Data are as reported to WHO. Estimates of TB and MDR-TB burden are produced by WHO in consultation with
countries.
a Ranges represent uncertainty intervals.
KENYA Population 2012 43 million
HIGH TB BURDEN | HIGH HIV BURDEN
Estimates of TB burdena 2012
07/$'4(thousands)4#6'(per 100 000 population)
Mortality (excludes HIV+TB) 9.5 (5.4–15) 22 (13–34)
/QTVCNKV[*+86$QPN[ ¿ ¿
Prevalence (includes HIV+TB) 130 (71–210) 299 (163–475)
+PEKFGPEGKPENWFGU*+86$ ¿ ¿
Incidence (HIV+TB only) 45 (44–47) 105 (101–109)
%CUGFGVGEVKQPCNNHQTOU ¿
6$ECUGPQVKßECVKQPU
0'9%#5'5 4'64'#6/'06%#5'5
5OGCTRQUKVKXG 4GNCRUG
5OGCTPGICVKXG 6TGCVOGPVCHVGTHCKNWTG
5OGCTWPMPQYPPQVFQPG 6TGCVOGPVCHVGTFGHCWNV
'ZVTCRWNOQPCT[ 1VJGT
Other 0 (0)
Total new 89 568 Total retreatment 9 581
Other (history unknown) 0
6QVCNPGYCPFTGNCRUG 6QVCNECUGUPQVKßGF
New cases
5/'#40')#6+8'70-0190
5/'#4215+6+8' 016&10' ':64#27./10#4;
/(TCVKQ
#IG
Laboratories 2012
Smear (per 100 000 population) 4.2
Culture (per 5 million population) 0.2
Drug susceptibility testing (per 5 million population) 0.2
+UUGEQPFNKPGFTWIUWUEGRVKDKNKV[VGUVKPICXCKNCDNG! ;GUKPCPFQWVUKFGEQWPVT[
6TGCVOGPVUWEEGUUTCVG
0GYUOGCTRQUKVKXGCPFQTEWNVWTGRQUKVKXG
0GYUOGCTPGICVKXGGZVTCRWNOQPCT[
4GVTGCVOGPV
+UTKHCORKEKPWUGFVJTQWIJQWVVTGCVOGPVHQTPGYRCVKGPVU! ;GU
TB/HIV 2012 07/$'4 (%)
6$RCVKGPVUYKVJMPQYP*+8UVCVWU
*+8RQUKVKXG6$RCVKGPVU
*+8RQUKVKXG6$RCVKGPVUQPEQVTKOQZC\QNGRTGXGPVKXGVJGTCR[%26
*+8RQUKVKXG6$RCVKGPVUQPCPVKTGVTQXKTCNVJGTCR[#46
HIV-positive people screened for TB
HIV-positive people provided with IPT
Estimates of MDR-TB burden 2012a 0'9 4'64'#6/'06
QH6$ECUGUYKVJ/&46$ ¿ ¿
/&46$ECUGUCOQPIPQVKßGF
RWNOQPCT[6$ECUGU ¿ ¿
Reported cases of MDR-TB 20120'9 4'64'#6/'06 616#.
%CUGUVGUVGFHQT/&46$
.CDQTCVQT[EQPßTOGF/&46$ECUGU
2CVKGPVUUVCTVGFQP/&46$VTGCVOGPV
Financing TB control 2013
0CVKQPCN6$RTQITCOOGDWFIGV75OKNNKQPU
% Funded domestically 24%
% Funded internationally 15%
% Unfunded 61%
Mortality (excludes HIV+TB) (rate
per 100 000 population per year)
1990 1995 2000 2005 2010
0
20
40
60
Prevalence
(rate per 100 000 population)
1990 1995 2000 2005 2010
0
200
400
600
Incidence (rate per 100 000
population per year)
1990 1995 2000 2005 2010
Incidence Incidence (HIV+TB) Notifications
0
100
200
300
400
Treatment success rate (%)
New smear-positive (and/or culture-positive) Retreatment
New smear-negative/extrapulmonary
1995 1997 1999 2001 2003 2005 2007 2009 2011
40
60
80
100
Number of patients
2003 2004 2005 2006 2007 2008 2009 2010 2011 2012
HIV-positive TB patients on CPT on ART
0
20 000
40 000
60 000
Total budget (US$ millions)
Funded domestically Funded internationally Unfunded
2009 2010 2011 2012 2013
0
20
40
60
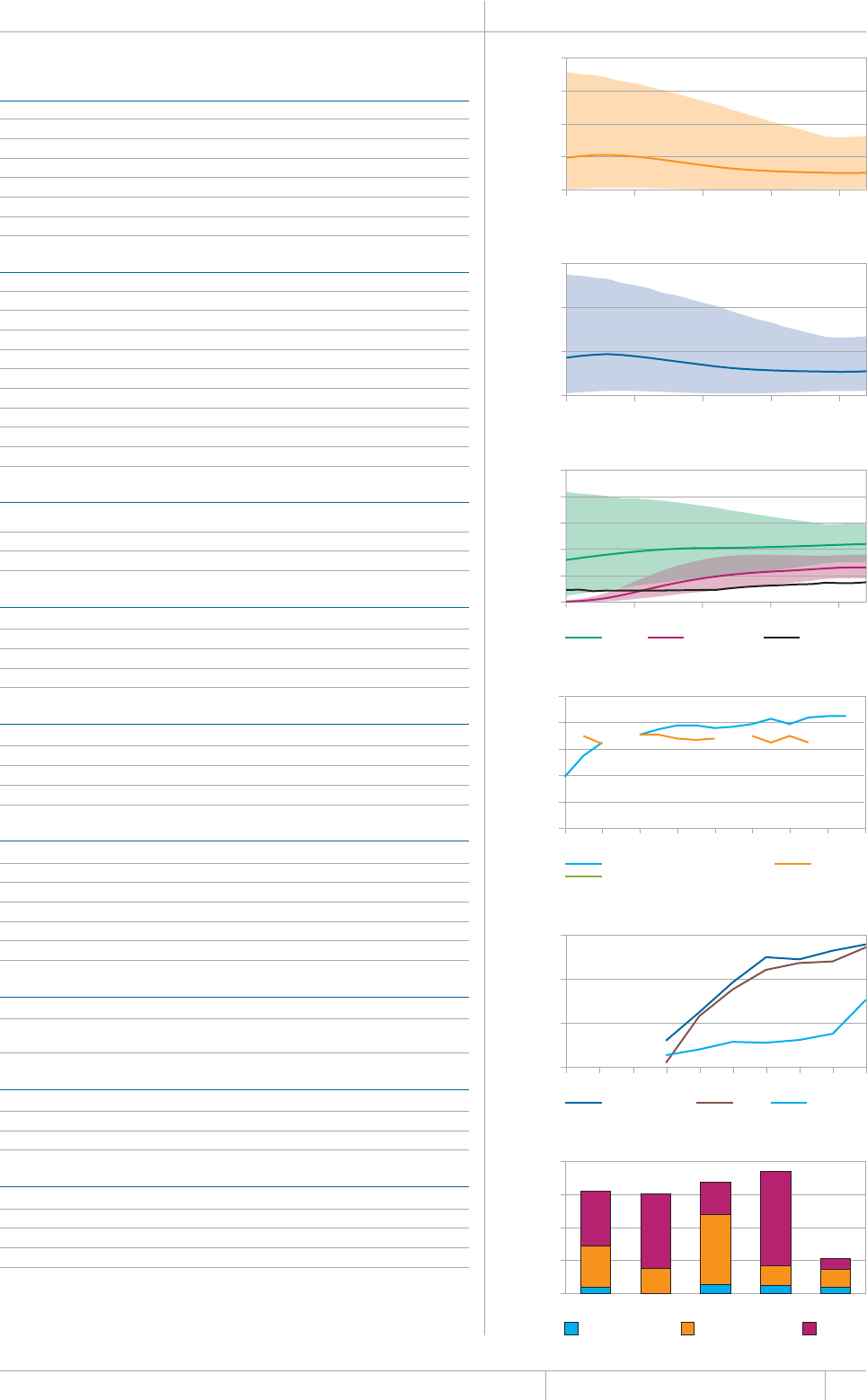
125 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
Data are as reported to WHO. Estimates of TB and MDR-TB burden are produced by WHO in consultation with
countries.
a Ranges represent uncertainty intervals.
MOZAMBIQUE Population 2012 25 million
HIGH TB BURDEN | HIGH HIV BURDEN
Estimates of TB burdena 2012
07/$'4(thousands)4#6'(per 100 000 population)
/QTVCNKV[GZENWFGU*+86$ ¿ ¿
/QTVCNKV[*+86$QPN[ ¿ ¿
2TGXCNGPEGKPENWFGU*+86$ ¿ ¿
+PEKFGPEGKPENWFGU*+86$ ¿ ¿
+PEKFGPEG*+86$QPN[ ¿ ¿
Case detection, all forms (%) 34 (25–50)
6$ECUGPQVKßECVKQPU
0'9%#5'5 4'64'#6/'06%#5'5
5OGCTRQUKVKXG 4GNCRUG
Smear-negative 19 797 (43) Treatment after failure 243 (5)
5OGCTWPMPQYPPQVFQPG 6TGCVOGPVCHVGTFGHCWNV
Extrapulmonary 5 542 (12) Other 2 595 (57)
Other 0 (0)
Total new 46 290 Total retreatment 4 537
Other (history unknown) 0
6QVCNPGYCPFTGNCRUG 6QVCNECUGUPQVKßGF
New cases
5/'#40')#6+8'70-0190
5/'#4215+6+8' 016&10' ':64#27./10#4;
/(TCVKQ
#IG
Laboratories 2012
Smear (per 100 000 population) 1.2
Culture (per 5 million population) 0.6
Drug susceptibility testing (per 5 million population) 0.4
+UUGEQPFNKPGFTWIUWUEGRVKDKNKV[VGUVKPICXCKNCDNG! ;GUQWVUKFGEQWPVT[
6TGCVOGPVUWEEGUUTCVG
New smear-positive and/or culture-positive
New smear-negative/extrapulmonary
4GVTGCVOGPV
+UTKHCORKEKPWUGFVJTQWIJQWVVTGCVOGPVHQTPGYRCVKGPVU! ;GU
TB/HIV 2012 07/$'4 (%)
TB patients with known HIV status 47 960 (94)
*+8RQUKVKXG6$RCVKGPVU
*+8RQUKVKXG6$RCVKGPVUQPEQVTKOQZC\QNGRTGXGPVKXGVJGTCR[%26
*+8RQUKVKXG6$RCVKGPVUQPCPVKTGVTQXKTCNVJGTCR[#46
HIV-positive people screened for TB
HIV-positive people provided with IPT 17 317
Estimates of MDR-TB burden 2012a 0'9 4'64'#6/'06
QH6$ECUGUYKVJ/&46$ ¿ ¿
/&46$ECUGUCOQPIPQVKßGF
pulmonary TB cases 1 400 (900–2 000) 540 (0–1 100)
Reported cases of MDR-TB 20120'9 4'64'#6/'06 616#.
%CUGUVGUVGFHQT/&46$
.CDQTCVQT[EQPßTOGF/&46$ECUGU
2CVKGPVUUVCTVGFQP/&46$VTGCVOGPV
Financing TB control 2013
0CVKQPCN6$RTQITCOOGDWFIGV75OKNNKQPU
% Funded domestically 19%
% Funded internationally 51%
% Unfunded 30%
Mortality (excludes HIV+TB) (rate
per 100 000 population per year)
1990 1995 2000 2005 2010
0
100
200
300
400
Prevalence
(rate per 100 000 population)
1990 1995 2000 2005 2010
0
1000
2000
3000
Incidence (rate per 100 000
population per year)
1990 1995 2000 2005 2010
Incidence Incidence (HIV+TB) Notifications
0
250
500
750
1000
1250
Treatment success rate (%)
New smear-positive (and/or culture-positive) Retreatment
New smear-negative/extrapulmonary
1995 1997 1999 2001 2003 2005 2007 2009 2011
0
20
40
60
80
100
Number of patients
2003 2004 2005 2006 2007 2008 2009 2010 2011 2012
HIV-positive TB patients on CPT on ART
0
10 000
20 000
30 000
Total budget (US$ millions)
Funded domestically Funded internationally Unfunded
2009 2010 2011 2012 2013
0
10
20
30
40
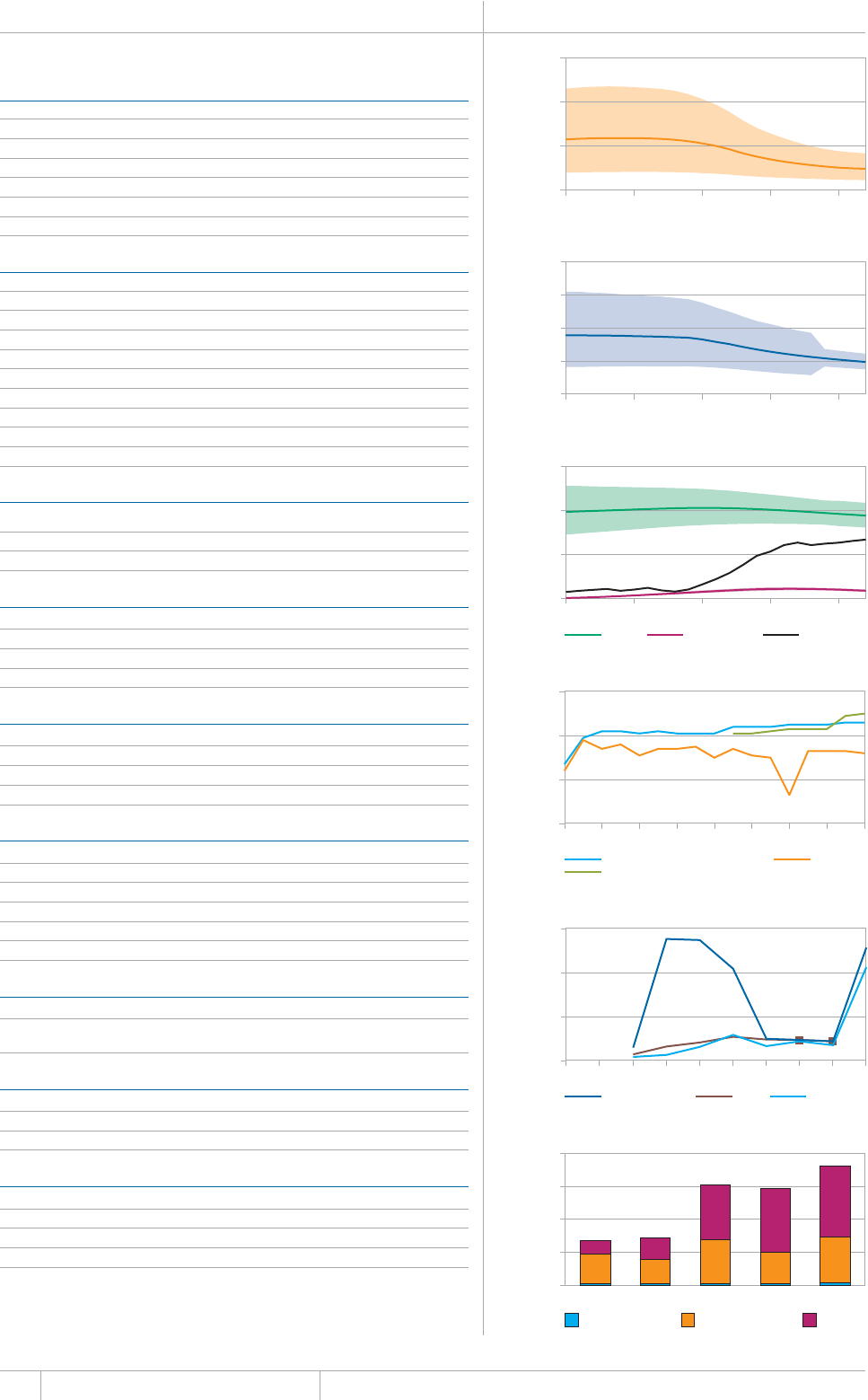
126 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
Data are as reported to WHO. Estimates of TB and MDR-TB burden are produced by WHO in consultation with
countries.
a Ranges represent uncertainty intervals.
MYANMAR Population 2012 53 million
HIGH TB BURDEN | HIGH HIV BURDEN | HIGH MDR-TB BURDEN
Estimates of TB burdena 2012
07/$'4(thousands)4#6'(per 100 000 population)
/QTVCNKV[GZENWFGU*+86$ ¿ ¿
/QTVCNKV[*+86$QPN[ ¿ ¿
2TGXCNGPEGKPENWFGU*+86$ ¿ ¿
Incidence (includes HIV+TB) 200 (170–230) 377 (322–435)
Incidence (HIV+TB only) 19 (16–21) 35 (30–41)
%CUGFGVGEVKQPCNNHQTOU ¿
6$ECUGPQVKßECVKQPU
0'9%#5'5 4'64'#6/'06%#5'5
5OGCTRQUKVKXG 4GNCRUG
Smear-negative 73 042 (53) Treatment after failure 1 671 (14)
Smear-unknown / not done 0 (0) Treatment after default 521 (5)
'ZVTCRWNOQPCT[ 1VJGT
Other 0 (0)
Total new 136 612 Total retreatment 11 537
Other (history unknown) 0
6QVCNPGYCPFTGNCRUG 6QVCNECUGUPQVKßGF
New cases
5/'#40')#6+8'70-0190
5/'#4215+6+8' 016&10' ':64#27./10#4;
/(TCVKQ
#IG
Laboratories 2012
Smear (per 100 000 population) 0.9
Culture (per 5 million population) 0.2
Drug susceptibility testing (per 5 million population) 0.2
+UUGEQPFNKPGFTWIUWUEGRVKDKNKV[VGUVKPICXCKNCDNG! ;GUKPCPFQWVUKFGEQWPVT[
6TGCVOGPVUWEEGUUTCVG
0GYUOGCTRQUKVKXGCPFQTEWNVWTGRQUKVKXG
New smear-negative/extrapulmonary 90
4GVTGCVOGPV
+UTKHCORKEKPWUGFVJTQWIJQWVVTGCVOGPVHQTPGYRCVKGPVU! ;GU
TB/HIV 2012 07/$'4 (%)
TB patients with known HIV status 19 219 (13)
HIV-positive TB patients 5 161 (27)
*+8RQUKVKXG6$RCVKGPVUQPEQVTKOQZC\QNGRTGXGPVKXGVJGTCR[%26
*+8RQUKVKXG6$RCVKGPVUQPCPVKTGVTQXKTCNVJGTCR[#46
HIV-positive people screened for TB
HIV-positive people provided with IPT
Estimates of MDR-TB burden 2012a 0'9 4'64'#6/'06
QH6$ECUGUYKVJ/&46$ ¿ ¿
/&46$ECUGUCOQPIPQVKßGF
pulmonary TB cases 4 900 (3 600–6 500) 1 200 (790–1 600)
Reported cases of MDR-TB 20120'9 4'64'#6/'06 616#.
%CUGUVGUVGFHQT/&46$
.CDQTCVQT[EQPßTOGF/&46$ECUGU
2CVKGPVUUVCTVGFQP/&46$VTGCVOGPV
Financing TB control 2013
0CVKQPCN6$RTQITCOOGDWFIGV75OKNNKQPU
% Funded domestically 2%
% Funded internationally 39%
% Unfunded 60%
Mortality (excludes HIV+TB) (rate
per 100 000 population per year)
1990 1995 2000 2005 2010
0
100
200
300
Prevalence
(rate per 100 000 population)
1990 1995 2000 2005 2010
0
500
1000
1500
2000
Incidence (rate per 100 000
population per year)
1990 1995 2000 2005 2010
Incidence Incidence (HIV+TB) Notifications
0
200
400
600
Treatment success rate (%)
New smear-positive (and/or culture-positive) Retreatment
New smear-negative/extrapulmonary
1995 1997 1999 2001 2003 2005 2007 2009 2011
40
60
80
100
Number of patients
2003 2004 2005 2006 2007 2008 2009 2010 2011 2012
HIV-positive TB patients on CPT on ART
0
2000
4000
6000
Total budget (US$ millions)
Funded domestically Funded internationally Unfunded
2009 2010 2011 2012 2013
0
10
20
30
40
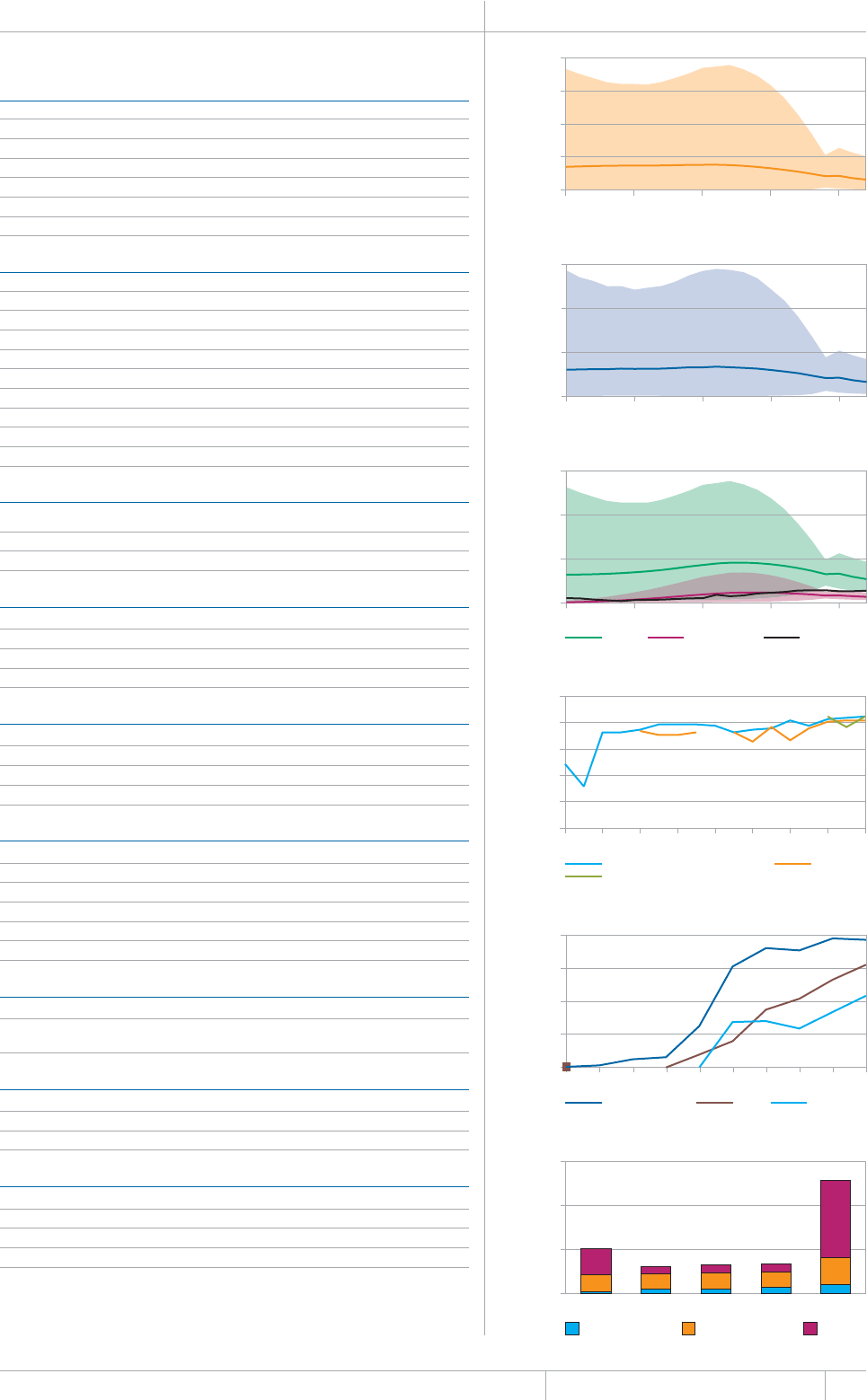
127 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
Data are as reported to WHO. Estimates of TB and MDR-TB burden are produced by WHO in consultation with
countries.
a Ranges represent uncertainty intervals.
NIGERIA Population 2012 169 million
HIGH TB BURDEN | HIGH HIV BURDEN | HIGH MDR-TB BURDEN
Estimates of TB burdena 2012
07/$'4(thousands)4#6'(per 100 000 population)
/QTVCNKV[GZENWFGU*+86$ ¿ ¿
Mortality (HIV+TB only) 19 (11–25) 11 (6.7–15)
Prevalence (includes HIV+TB) 270 (43–710) 161 (25–420)
+PEKFGPEGKPENWFGU*+86$ ¿ ¿
+PEKFGPEG*+86$QPN[ ¿ ¿
Case detection, all forms (%) 51 (29–110)
6$ECUGPQVKßECVKQPU
0'9%#5'5 4'64'#6/'06%#5'5
5OGCTRQUKVKXG 4GNCRUG
5OGCTPGICVKXG 6TGCVOGPVCHVGTHCKNWTG
Smear-unknown / not done Treatment after default 1 174 (16)
Extrapulmonary 4 432 (5) Other 3 249 (43)
Other
Total new 90 305 Total retreatment 7 548
Other (history unknown)
6QVCNPGYCPFTGNCRUG 6QVCNECUGUPQVKßGF
New cases
5/'#40')#6+8'70-0190
5/'#4215+6+8' 016&10' ':64#27./10#4;
/(TCVKQ
#IG
Laboratories 2012
5OGCTRGTRQRWNCVKQP
Culture (per 5 million population) 0.1
&TWIUWUEGRVKDKNKV[VGUVKPIRGTOKNNKQPRQRWNCVKQP
+UUGEQPFNKPGFTWIUWUEGRVKDKNKV[VGUVKPICXCKNCDNG! ;GUQWVUKFGEQWPVT[
6TGCVOGPVUWEEGUUTCVG
0GYUOGCTRQUKVKXGCPFQTEWNVWTGRQUKVKXG
0GYUOGCTPGICVKXGGZVTCRWNOQPCT[
4GVTGCVOGPV
+UTKHCORKEKPWUGFVJTQWIJQWVVTGCVOGPVHQTPGYRCVKGPVU! ;GU
TB/HIV 2012 07/$'4 (%)
6$RCVKGPVUYKVJMPQYP*+8UVCVWU
HIV-positive TB patients 19 342 (23)
*+8RQUKVKXG6$RCVKGPVUQPEQVTKOQZC\QNGRTGXGPVKXGVJGTCR[%26
*+8RQUKVKXG6$RCVKGPVUQPCPVKTGVTQXKTCNVJGTCR[#46
HIV-positive people screened for TB 140 460
HIV-positive people provided with IPT 2 257
Estimates of MDR-TB burden 2012a 0'9 4'64'#6/'06
QH6$ECUGUYKVJ/&46$ ¿ ¿
/&46$ECUGUCOQPIPQVKßGF
RWNOQPCT[6$ECUGU ¿ ¿
Reported cases of MDR-TB 20120'9 4'64'#6/'06 616#.
%CUGUVGUVGFHQT/&46$
.CDQTCVQT[EQPßTOGF/&46$ECUGU
2CVKGPVUUVCTVGFQP/&46$VTGCVOGPV
Financing TB control 2013
0CVKQPCN6$RTQITCOOGDWFIGV75OKNNKQPU
(WPFGFFQOGUVKECNN[
% Funded internationally 24%
7PHWPFGF
Mortality (excludes HIV+TB) (rate
per 100 000 population per year)
1990 1995 2000 2005 2010
0
50
100
150
200
Prevalence
(rate per 100 000 population)
1990 1995 2000 2005 2010
0
500
1000
1500
Incidence (rate per 100 000
population per year)
1990 1995 2000 2005 2010
Incidence Incidence (HIV+TB) Notifications
0
100
200
300
400
500
600
Treatment success rate (%)
New smear-positive (and/or culture-positive) Retreatment
New smear-negative/extrapulmonary
1995 1997 1999 2001 2003 2005 2007 2009 2011
0
20
40
60
80
100
Number of patients
2003 2004 2005 2006 2007 2008 2009 2010 2011 2012
HIV-positive TB patients on CPT on ART
0
5000
10 000
15 000
20 000
Total budget (US$ millions)
Funded domestically Funded internationally Unfunded
2009 2010 2011 2012 2013
0
60
120
180
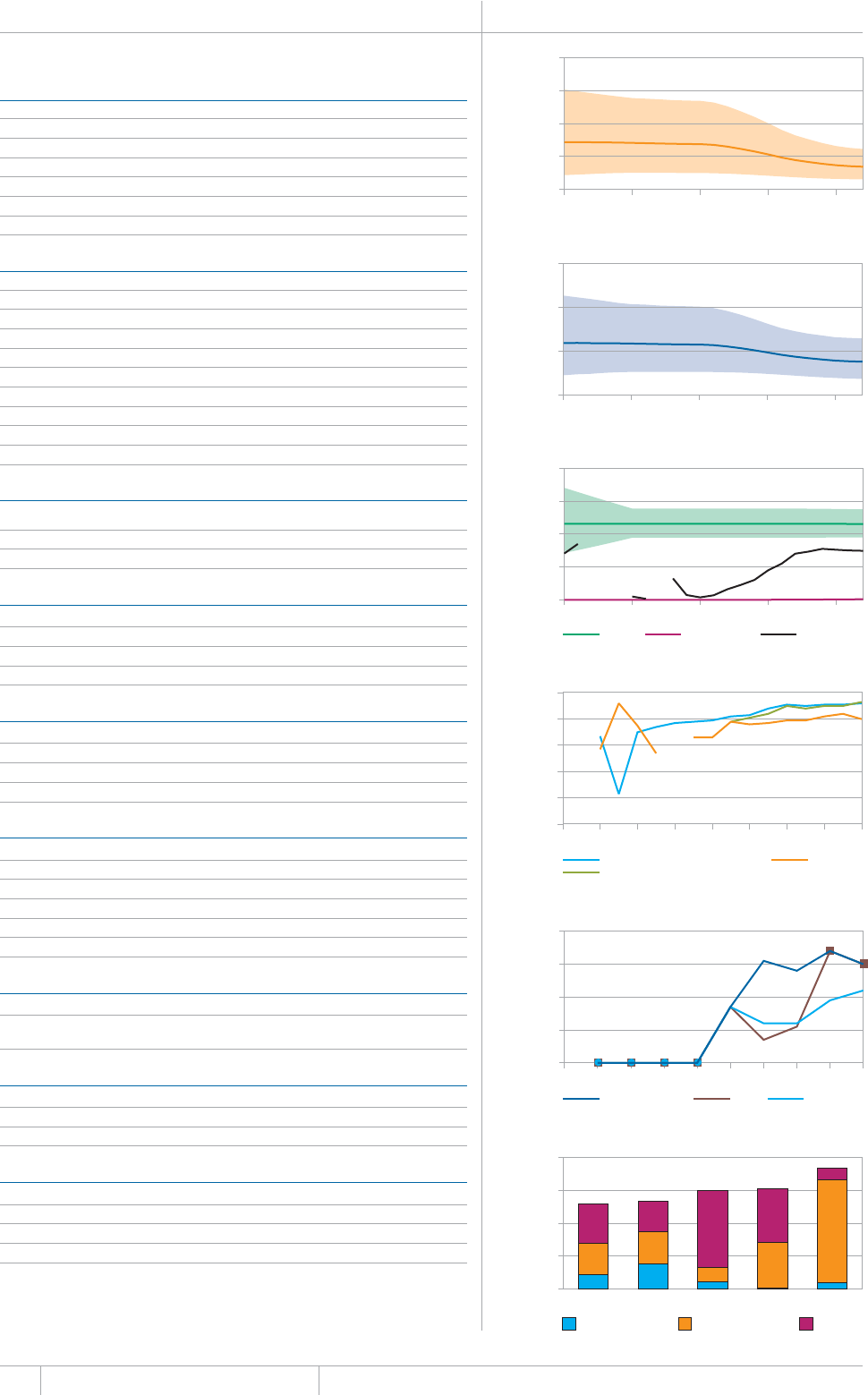
GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
Data are as reported to WHO. Estimates of TB and MDR-TB burden are produced by WHO in consultation with
countries.
a Ranges represent uncertainty intervals.
PAKISTAN Population 2012 179 million
HIGH TB BURDEN | HIGH MDR-TB BURDEN
Estimates of TB burdena 2012
07/$'4(thousands)4#6'(per 100 000 population)
Mortality (excludes HIV+TB) 62 (27–110) 34 (15–61)
/QTVCNKV[*+86$QPN[ ¿ ¿
2TGXCNGPEGKPENWFGU*+86$ ¿ ¿
Incidence (includes HIV+TB) 410 (340–490) 231 (190–276)
+PEKFGPEG*+86$QPN[ ¿ ¿
%CUGFGVGEVKQPCNNHQTOU ¿
6$ECUGPQVKßECVKQPU
0'9%#5'5 4'64'#6/'06%#5'5
5OGCTRQUKVKXG 4GNCRUG
5OGCTPGICVKXG 6TGCVOGPVCHVGTHCKNWTG
Smear-unknown / not done 0 (0) Treatment after default 1 241 (11)
Extrapulmonary 41 410 (16) Other 3 534 (30)
Other 0 (0)
Total new 261 380 Total retreatment 11 717
Other (history unknown) 0
6QVCNPGYCPFTGNCRUG 6QVCNECUGUPQVKßGF
New cases
5/'#40')#6+8'70-0190
5/'#4215+6+8' 016&10' ':64#27./10#4;
/(TCVKQ
#IG
Laboratories 2012
5OGCTRGTRQRWNCVKQP
Culture (per 5 million population) 0.2
Drug susceptibility testing (per 5 million population) 0.1
+UUGEQPFNKPGFTWIUWUEGRVKDKNKV[VGUVKPICXCKNCDNG! ;GUKPEQWPVT[
6TGCVOGPVUWEEGUUTCVG
New smear-positive and/or culture-positive 92
New smear-negative/extrapulmonary 93
4GVTGCVOGPV
+UTKHCORKEKPWUGFVJTQWIJQWVVTGCVOGPVHQTPGYRCVKGPVU! ;GU
TB/HIV 2012 07/$'4 (%)
TB patients with known HIV status 10 419 (4)
*+8RQUKVKXG6$RCVKGPVU
*+8RQUKVKXG6$RCVKGPVUQPEQVTKOQZC\QNGRTGXGPVKXGVJGTCR[%26
*+8RQUKVKXG6$RCVKGPVUQPCPVKTGVTQXKTCNVJGTCR[#46
HIV-positive people screened for TB
HIV-positive people provided with IPT
Estimates of MDR-TB burden 2012a 0'9 4'64'#6/'06
QH6$ECUGUYKVJ/&46$ ¿ ¿
/&46$ECUGUCOQPIPQVKßGF
RWNOQPCT[6$ECUGU ¿ ¿
Reported cases of MDR-TB 20120'9 4'64'#6/'06 616#.
%CUGUVGUVGFHQT/&46$
.CDQTCVQT[EQPßTOGF/&46$ECUGU
2CVKGPVUUVCTVGFQP/&46$VTGCVOGPV
Financing TB control 2013
0CVKQPCN6$RTQITCOOGDWFIGV75OKNNKQPU
% Funded domestically 5%
(WPFGFKPVGTPCVKQPCNN[
% Unfunded 9%
Mortality (excludes HIV+TB) (rate
per 100 000 population per year)
1990 1995 2000 2005 2010
0
50
100
150
200
Prevalence
(rate per 100 000 population)
1990 1995 2000 2005 2010
0
500
1000
1500
Incidence (rate per 100 000
population per year)
1990 1995 2000 2005 2010
Incidence Incidence (HIV+TB) Notifications
0
100
200
300
400
Treatment success rate (%)
New smear-positive (and/or culture-positive) Retreatment
New smear-negative/extrapulmonary
1995 1997 1999 2001 2003 2005 2007 2009 2011
0
20
40
60
80
100
Number of patients
2003 2004 2005 2006 2007 2008 2009 2010 2011 2012
HIV-positive TB patients on CPT on ART
0
10
20
30
40
Total budget (US$ millions)
Funded domestically Funded internationally Unfunded
2009 2010 2011 2012 2013
0
20
40
60
80

129 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
Data are as reported to WHO. Estimates of TB and MDR-TB burden are produced by WHO in consultation with
countries.
a Ranges represent uncertainty intervals.
PHILIPPINES Population 2012 97 million
HIGH TB BURDEN | HIGH MDR-TB BURDEN
Estimates of TB burdena 2012
07/$'4(thousands)4#6'(per 100 000 population)
Mortality (excludes HIV+TB) 23 (22–25) 24 (22–26)
Mortality (HIV+TB only) 0.11 (0.09–0.13) 0.11 (0.09–0.14)
Prevalence (includes HIV+TB) 450 (390–500) 461 (405–520)
Incidence (includes HIV+TB) 260 (210–310) 265 (219–316)
+PEKFGPEG*+86$QPN[ ¿ ¿
%CUGFGVGEVKQPCNNHQTOU ¿
6$ECUGPQVKßECVKQPU
0'9%#5'5 4'64'#6/'06%#5'5
5OGCTRQUKVKXG 4GNCRUG
Smear-negative 115 263 (54) Treatment after failure 591 (3)
Smear-unknown / not done 0 (0) Treatment after default 1 243 (5)
Extrapulmonary 3 270 (2) Other 17 575 (75)
Other 0 (0)
Total new 212 119 Total retreatment 23 489
Other (history unknown) 0
6QVCNPGYCPFTGNCRUG 6QVCNECUGUPQVKßGF
New cases
5/'#40')#6+8'70-0190
5/'#4215+6+8' 016&10' ':64#27./10#4;
/(TCVKQ
#IG
Laboratories 2012
Smear (per 100 000 population) 2.7
Culture (per 5 million population) 0.7
Drug susceptibility testing (per 5 million population) 0.2
+UUGEQPFNKPGFTWIUWUEGRVKDKNKV[VGUVKPICXCKNCDNG! ;GUKPEQWPVT[
6TGCVOGPVUWEEGUUTCVG
New smear-positive and/or culture-positive 90
0GYUOGCTPGICVKXGGZVTCRWNOQPCT[
4GVTGCVOGPV
+UTKHCORKEKPWUGFVJTQWIJQWVVTGCVOGPVHQTPGYRCVKGPVU! ;GU
TB/HIV 2012 07/$'4 (%)
6$RCVKGPVUYKVJMPQYP*+8UVCVWU
*+8RQUKVKXG6$RCVKGPVU
*+8RQUKVKXG6$RCVKGPVUQPEQVTKOQZC\QNGRTGXGPVKXGVJGTCR[%26
*+8RQUKVKXG6$RCVKGPVUQPCPVKTGVTQXKTCNVJGTCR[#46
HIV-positive people screened for TB
HIV-positive people provided with IPT
Estimates of MDR-TB burden 2012a 0'9 4'64'#6/'06
QH6$ECUGUYKVJ/&46$ ¿ ¿
/&46$ECUGUCOQPIPQVKßGF
RWNOQPCT[6$ECUGU ¿ ¿
Reported cases of MDR-TB 20120'9 4'64'#6/'06 616#.
%CUGUVGUVGFHQT/&46$
.CDQTCVQT[EQPßTOGF/&46$ECUGU
2CVKGPVUUVCTVGFQP/&46$VTGCVOGPV
Financing TB control 2013
0CVKQPCN6$RTQITCOOGDWFIGV75OKNNKQPU
% Funded domestically 16%
% Funded internationally 15%
% Unfunded 69%
Mortality (excludes HIV+TB) (rate
per 100 000 population per year)
1990 1995 2000 2005 2010
0
20
40
60
80
Prevalence
(rate per 100 000 population)
1990 1995 2000 2005 2010
0
500
1000
1500
Incidence (rate per 100 000
population per year)
1990 1995 2000 2005 2010
Incidence Incidence (HIV+TB) Notifications
0
200
400
600
Treatment success rate (%)
New smear-positive (and/or culture-positive) Retreatment
New smear-negative/extrapulmonary
1995 1997 1999 2001 2003 2005 2007 2009 2011
0
20
40
60
80
100
Number of patients
2003 2004 2005 2006 2007 2008 2009 2010 2011 2012
HIV-positive TB patients on CPT on ART
0
2
4
6
8
10
Total budget (US$ millions)
Funded domestically Funded internationally Unfunded
2009 2010 2011 2012 2013
0
40
80
120
160
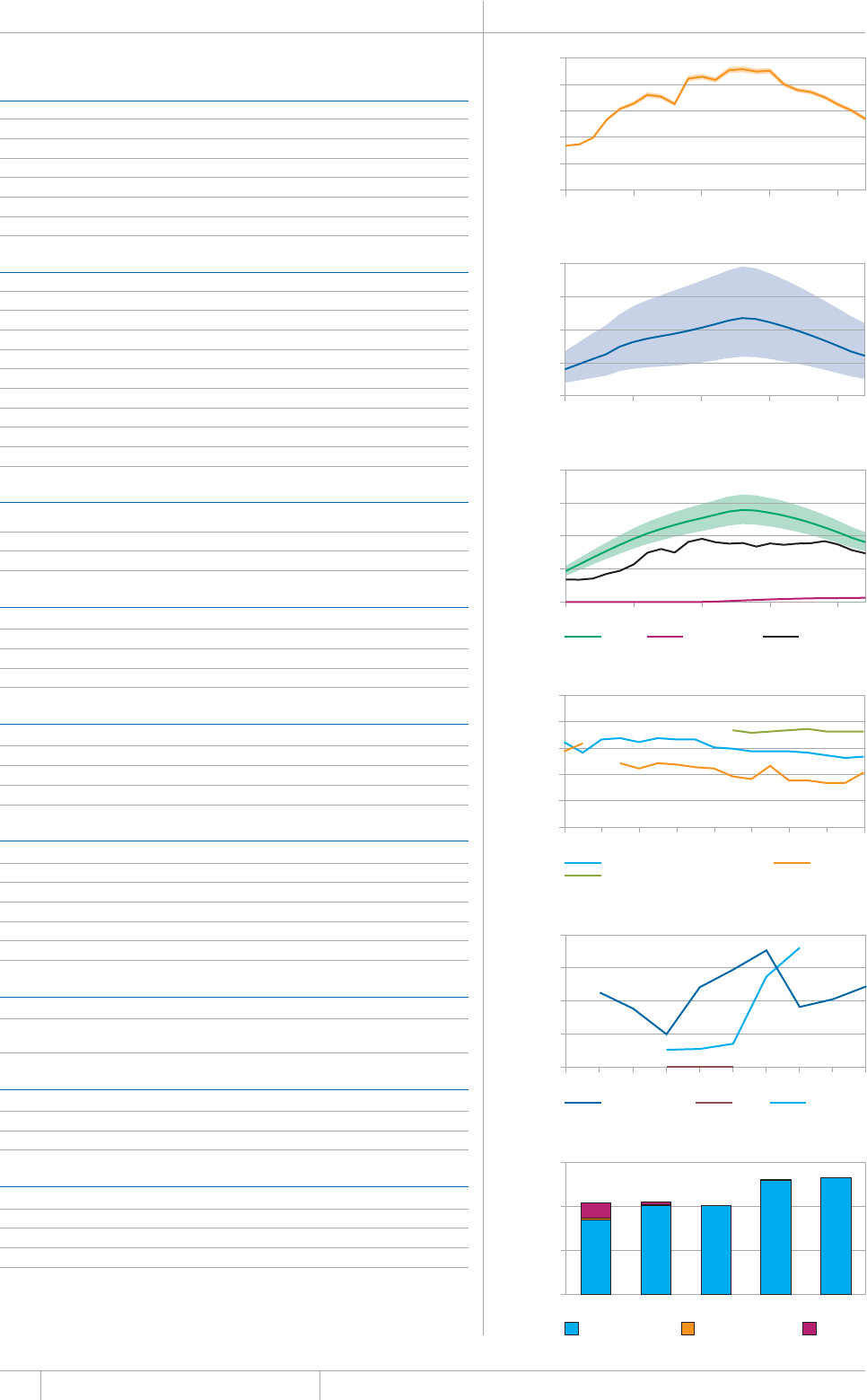
130 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
&CVCCTGCUTGRQTVGFVQ9*1'UVKOCVGUQH6$CPF/&46$DWTFGPCTGRTQFWEGFD[9*1KPEQPUWNVCVKQPYKVJ
countries.
a 4CPIGUTGRTGUGPVWPEGTVCKPV[KPVGTXCNU
b The reported number of TB patients with known HIV status is for new TB patients in the civilian sector only.
It was not possible to calculate the percentage of all TB patients with known HIV status.
RUSSIAN FEDERATION Population 2012 143 million
HIGH TB BURDEN | HIGH HIV BURDEN | HIGH MDR-TB BURDEN
Estimates of TB burdena 2012
07/$'4(thousands)4#6'(per 100 000 population)
/QTVCNKV[GZENWFGU*+86$ ¿ ¿
/QTVCNKV[*+86$QPN[ ¿ ¿
Prevalence (includes HIV+TB) 170 (73–320) 121 (51–221)
Incidence (includes HIV+TB) 130 (110–150) 91 (77–106)
Incidence (HIV+TB only) 9.3 (7.9–11) 6.5 (5.5–7.5)
%CUGFGVGEVKQPCNNHQTOU ¿
6$ECUGPQVKßECVKQPU
0'9%#5'5 4'64'#6/'06%#5'5
5OGCTRQUKVKXG 4GNCRUG
Smear-negative 59 019 (61) Treatment after failure 9 109 (17)
Smear-unknown / not done 1 039 (1) Treatment after default 2 593 (5)
Extrapulmonary 10 017 (10) Other 32 466 (62)
Other 0 (0)
Total new 97 542 Total retreatment 52 379
Other (history unknown) 0
6QVCNPGYCPFTGNCRUG 6QVCNECUGUPQVKßGF
New cases
5/'#40')#6+8'70-0190
5/'#4215+6+8' 016&10' ':64#27./10#4;
/(TCVKQ
#IG
Laboratories 2012
Smear (per 100 000 population) 0.7
Culture (per 5 million population) 4.1
&TWIUWUEGRVKDKNKV[VGUVKPIRGTOKNNKQPRQRWNCVKQP
+UUGEQPFNKPGFTWIUWUEGRVKDKNKV[VGUVKPICXCKNCDNG! ;GUKPEQWPVT[
6TGCVOGPVUWEEGUUTCVG
New smear-positive and/or culture-positive 54
New smear-negative/extrapulmonary 73
4GVTGCVOGPV
+UTKHCORKEKPWUGFVJTQWIJQWVVTGCVOGPVHQTPGYRCVKGPVU! ;GU
TB/HIV 2012 07/$'4 (%)
TB patients with known HIV statusb 75 995
*+8RQUKVKXG6$RCVKGPVU
*+8RQUKVKXG6$RCVKGPVUQPEQVTKOQZC\QNGRTGXGPVKXGVJGTCR[%26
*+8RQUKVKXG6$RCVKGPVUQPCPVKTGVTQXKTCNVJGTCR[#46
HIV-positive people screened for TB
HIV-positive people provided with IPT
Estimates of MDR-TB burden 2012a 0'9 4'64'#6/'06
QH6$ECUGUYKVJ/&46$ ¿ ¿
/&46$ECUGUCOQPIPQVKßGF
RWNOQPCT[6$ECUGU ¿ ¿
Reported cases of MDR-TB 20120'9 4'64'#6/'06 616#.
%CUGUVGUVGFHQT/&46$
.CDQTCVQT[EQPßTOGF/&46$ECUGU
2CVKGPVUUVCTVGFQP/&46$VTGCVOGPV
Financing TB control 2013
0CVKQPCN6$RTQITCOOGDWFIGV75OKNNKQPU
% Funded domestically 100%
(WPFGFKPVGTPCVKQPCNN[
% Unfunded 0%
Mortality (excludes HIV+TB) (rate
per 100 000 population per year)
1990 1995 2000 2005 2010
0
5
10
15
20
25
Prevalence
(rate per 100 000 population)
1990 1995 2000 2005 2010
0
100
200
300
400
Incidence (rate per 100 000
population per year)
1990 1995 2000 2005 2010
Incidence Incidence (HIV+TB) Notifications
0
50
100
150
200
Treatment success rate (%)
New smear-positive (and/or culture-positive) Retreatment
New smear-negative/extrapulmonary
1995 1997 1999 2001 2003 2005 2007 2009 2011
0
20
40
60
80
100
Number of patients
2003 2004 2005 2006 2007 2008 2009 2010 2011 2012
HIV-positive TB patients on CPT on ART
0
2000
4000
6000
8000
Total budget (US$ millions)
Funded domestically Funded internationally Unfunded
2009 2010 2011 2012 2013
0
600
1200
1800
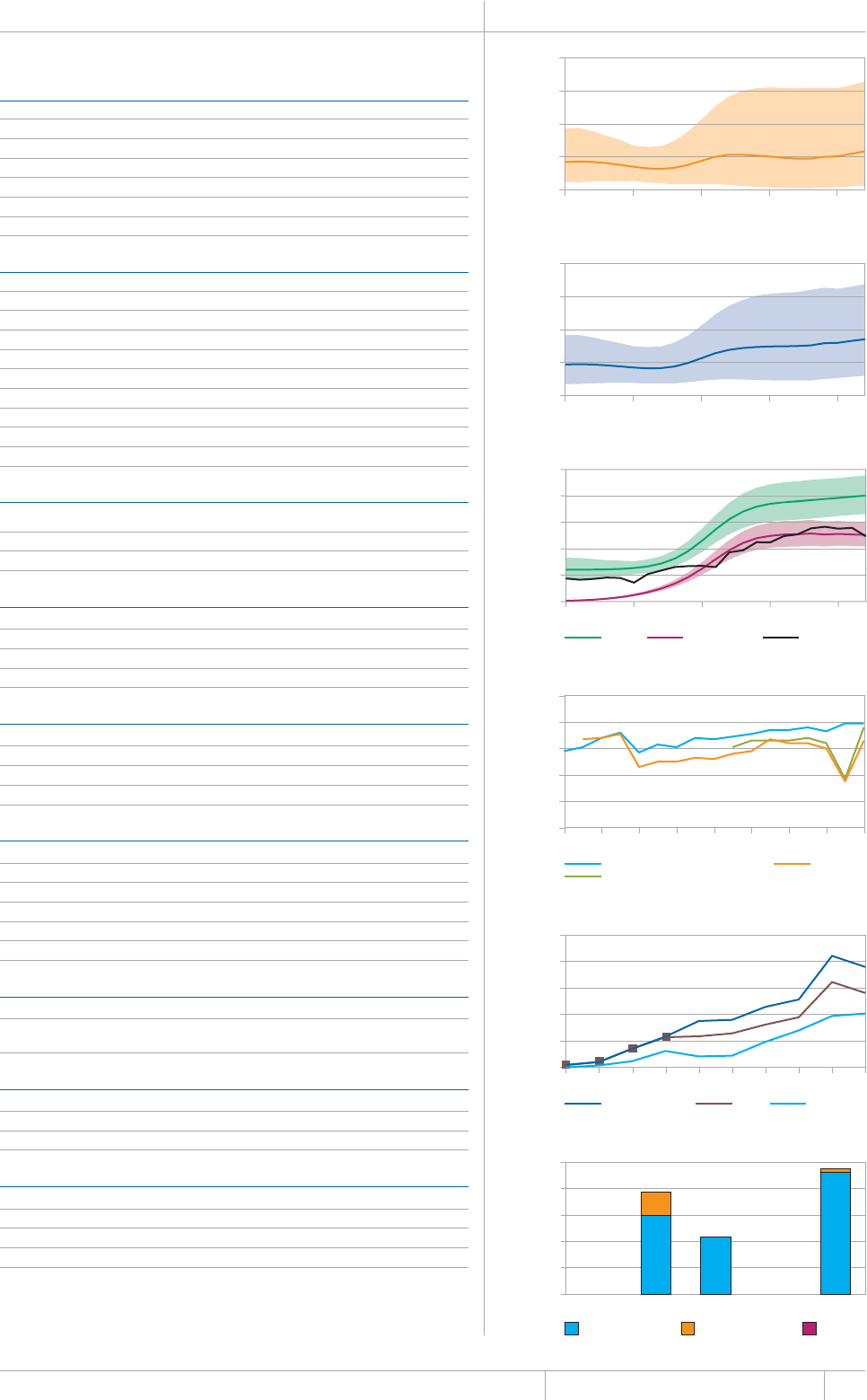
131 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
Data are as reported to WHO. Estimates of TB and MDR-TB burden are produced by WHO in consultation with
countries.
a Ranges represent uncertainty intervals.
SOUTH AFRICA Population 2012 52 million
HIGH TB BURDEN | HIGH HIV BURDEN | HIGH MDR-TB BURDEN
Estimates of TB burdena 2012
07/$'4(thousands)4#6'(per 100 000 population)
/QTVCNKV[GZENWFGU*+86$ ¿ ¿
/QTVCNKV[*+86$QPN[ ¿ ¿
2TGXCNGPEGKPENWFGU*+86$ ¿ ¿
+PEKFGPEGKPENWFGU*+86$ ¿ ¿
Incidence (HIV+TB only) 330 (270–390) 631 (521–752)
Case detection, all forms (%) 62 (52–75)
6$ECUGPQVKßECVKQPU
0'9%#5'5 4'64'#6/'06%#5'5
5OGCTRQUKVKXG 4GNCRUG
Smear-negative 63 210 (21) Treatment after failure 3 123 (6)
5OGCTWPMPQYPPQVFQPG 6TGCVOGPVCHVGTFGHCWNV
Extrapulmonary 42 467 (14) Other 15 007 (29)
Other 0 (0)
Total new 296 996 Total retreatment 52 586
Other (history unknown) 0
6QVCNPGYCPFTGNCRUG 6QVCNECUGUPQVKßGF
New cases
5/'#40')#6+8'70-0190
5/'#4215+6+8' 016&10' ':64#27./10#4;
/(TCVKQ
#IG
Laboratories 2012
Smear (per 100 000 population) 0.4
Culture (per 5 million population) 1.4
Drug susceptibility testing (per 5 million population) 1.4
+UUGEQPFNKPGFTWIUWUEGRVKDKNKV[VGUVKPICXCKNCDNG! ;GUKPEQWPVT[
6TGCVOGPVUWEEGUUTCVG
New smear-positive and/or culture-positive 79
New smear-negative/extrapulmonary 76
4GVTGCVOGPV
+UTKHCORKEKPWUGFVJTQWIJQWVVTGCVOGPVHQTPGYRCVKGPVU! ;GU
TB/HIV 2012 07/$'4 (%)
6$RCVKGPVUYKVJMPQYP*+8UVCVWU
HIV-positive TB patients 190 093 (65)
*+8RQUKVKXG6$RCVKGPVUQPEQVTKOQZC\QNGRTGXGPVKXGVJGTCR[%26
*+8RQUKVKXG6$RCVKGPVUQPCPVKTGVTQXKTCNVJGTCR[#46
*+8RQUKVKXGRGQRNGUETGGPGFHQT6$
HIV-positive people provided with IPT 369 747
Estimates of MDR-TB burden 2012a 0'9 4'64'#6/'06
QH6$ECUGUYKVJ/&46$ ¿ ¿
/&46$ECUGUCOQPIPQVKßGF
RWNOQPCT[6$ECUGU ¿ ¿
Reported cases of MDR-TB 20120'9 4'64'#6/'06 616#.
%CUGUVGUVGFHQT/&46$
.CDQTCVQT[EQPßTOGF/&46$ECUGU
2CVKGPVUUVCTVGFQP/&46$VTGCVOGPV
Financing TB control 2013
0CVKQPCN6$RTQITCOOGDWFIGV75OKNNKQPU
% Funded domestically 97%
% Funded internationally 3%
% Unfunded 0%
Mortality (excludes HIV+TB) (rate
per 100 000 population per year)
1990 1995 2000 2005 2010
0
50
100
150
200
Prevalence
(rate per 100 000 population)
1990 1995 2000 2005 2010
0
500
1000
1500
2000
Incidence (rate per 100 000
population per year)
1990 1995 2000 2005 2010
Incidence Incidence (HIV+TB) Notifications
0
250
500
750
1000
1250
Treatment success rate (%)
New smear-positive (and/or culture-positive) Retreatment
New smear-negative/extrapulmonary
1995 1997 1999 2001 2003 2005 2007 2009 2011
0
20
40
60
80
100
Number of patients
2003 2004 2005 2006 2007 2008 2009 2010 2011 2012
HIV-positive TB patients on CPT on ART
0
50 000
100 000
150 000
200 000
250 000
Total budget (US$ millions)
Funded domestically Funded internationally Unfunded
2009 2010 2011 2012 2013
0
100
200
300
400
500
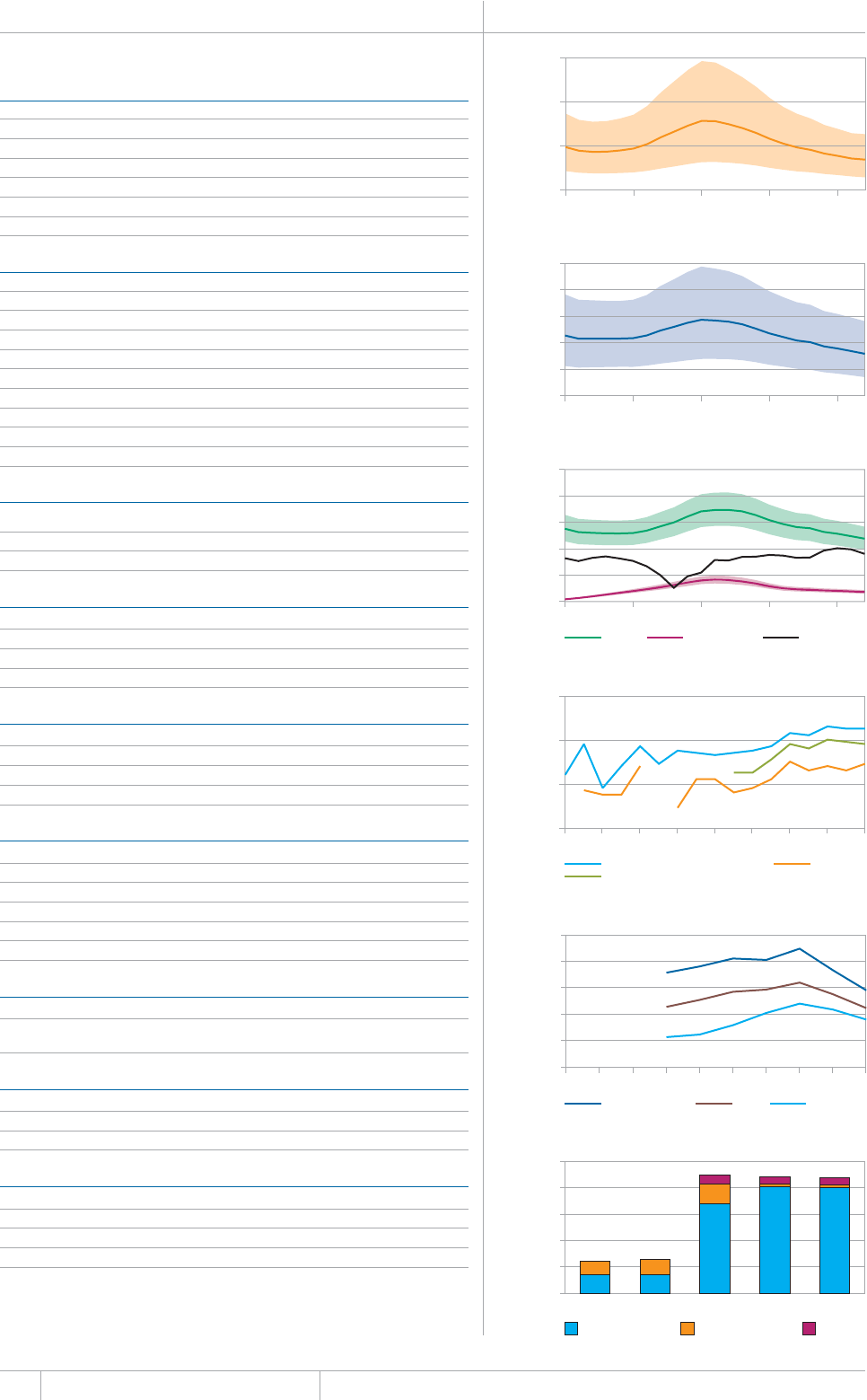
132 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
&CVCCTGCUTGRQTVGFVQ9*1'UVKOCVGUQH6$CPF/&46$DWTFGPCTGRTQFWEGFD[9*1KPEQPUWNVCVKQPYKVJ
countries.
a 4CPIGUTGRTGUGPVWPEGTVCKPV[KPVGTXCNU
b Based on data reported for 2013 in the 2012 round of data collection. In 2013, Thailand was not able to
report funding for the sub-national level.
THAILAND Population 2012 67 million
HIGH TB BURDEN | HIGH HIV BURDEN
Estimates of TB burdena 2012
07/$'4(thousands)4#6'(per 100 000 population)
/QTVCNKV[GZENWFGU*+86$ ¿ ¿
/QTVCNKV[*+86$QPN[ ¿ ¿
2TGXCNGPEGKPENWFGU*+86$ ¿ ¿
+PEKFGPEGKPENWFGU*+86$ ¿ ¿
+PEKFGPEG*+86$QPN[ ¿ ¿
Case detection, all forms (%) 76 (64–92)
6$ECUGPQVKßECVKQPU
0'9%#5'5 4'64'#6/'06%#5'5
5OGCTRQUKVKXG 4GNCRUG
Smear-negative 17 537 (31) Treatment after failure 327 (12)
Smear-unknown / not done Treatment after default 577 (21)
'ZVTCRWNOQPCT[ 1VJGT
Other
Total new 57 387 Total retreatment 2 791
Other (history unknown) 1 030
6QVCNPGYCPFTGNCRUG 6QVCNECUGUPQVKßGF
New cases
5/'#40')#6+8'70-0190
5/'#4215+6+8' 016&10' ':64#27./10#4;
/(TCVKQ
#IG
Laboratories 2012
Smear (per 100 000 population) 1.6
Culture (per 5 million population) 4.9
Drug susceptibility testing (per 5 million population) 1.3
+UUGEQPFNKPGFTWIUWUEGRVKDKNKV[VGUVKPICXCKNCDNG! ;GUKPEQWPVT[
6TGCVOGPVUWEEGUUTCVG
0GYUOGCTRQUKVKXGCPFQTEWNVWTGRQUKVKXG
0GYUOGCTPGICVKXGGZVTCRWNOQPCT[
4GVTGCVOGPV
+UTKHCORKEKPWUGFVJTQWIJQWVVTGCVOGPVHQTPGYRCVKGPVU! ;GU
TB/HIV 2012 07/$'4 (%)
TB patients with known HIV status 44 035 (72)
*+8RQUKVKXG6$RCVKGPVU
*+8RQUKVKXG6$RCVKGPVUQPEQVTKOQZC\QNGRTGXGPVKXGVJGTCR[%26
*+8RQUKVKXG6$RCVKGPVUQPCPVKTGVTQXKTCNVJGTCR[#46
HIV-positive people screened for TB
HIV-positive people provided with IPT
Estimates of MDR-TB burden 2012a 0'9 4'64'#6/'06
QH6$ECUGUYKVJ/&46$ ¿ ¿
/&46$ECUGUCOQPIPQVKßGF
RWNOQPCT[6$ECUGU ¿ ¿
Reported cases of MDR-TB 20120'9 4'64'#6/'06 616#.
%CUGUVGUVGFHQT/&46$
.CDQTCVQT[EQPßTOGF/&46$ECUGU
2CVKGPVUUVCTVGFQP/&46$VTGCVOGPV
Financing TB control 2013
0CVKQPCN6$RTQITCOOGDWFIGV75OKNNKQPU b
% Funded domestically 92%
% Funded internationally 2%
% Unfunded 6%
Mortality (excludes HIV+TB) (rate
per 100 000 population per year)
1990 1995 2000 2005 2010
0
20
40
60
Prevalence
(rate per 100 000 population)
1990 1995 2000 2005 2010
0
100
200
300
400
500
Incidence (rate per 100 000
population per year)
1990 1995 2000 2005 2010
Incidence Incidence (HIV+TB) Notifications
0
50
100
150
200
250
Treatment success rate (%)
New smear-positive (and/or culture-positive) Retreatment
New smear-negative/extrapulmonary
1995 1997 1999 2001 2003 2005 2007 2009 2011
40
60
80
100
Number of patients
2003 2004 2005 2006 2007 2008 2009 2010 2011 2012
HIV-positive TB patients on CPT on ART
0
2000
4000
6000
8000
10 000
Total budget (US$ millions)
Funded domestically Funded internationally Unfunded
2009 2010 2011 2012 2013
0
10
20
30
40
50
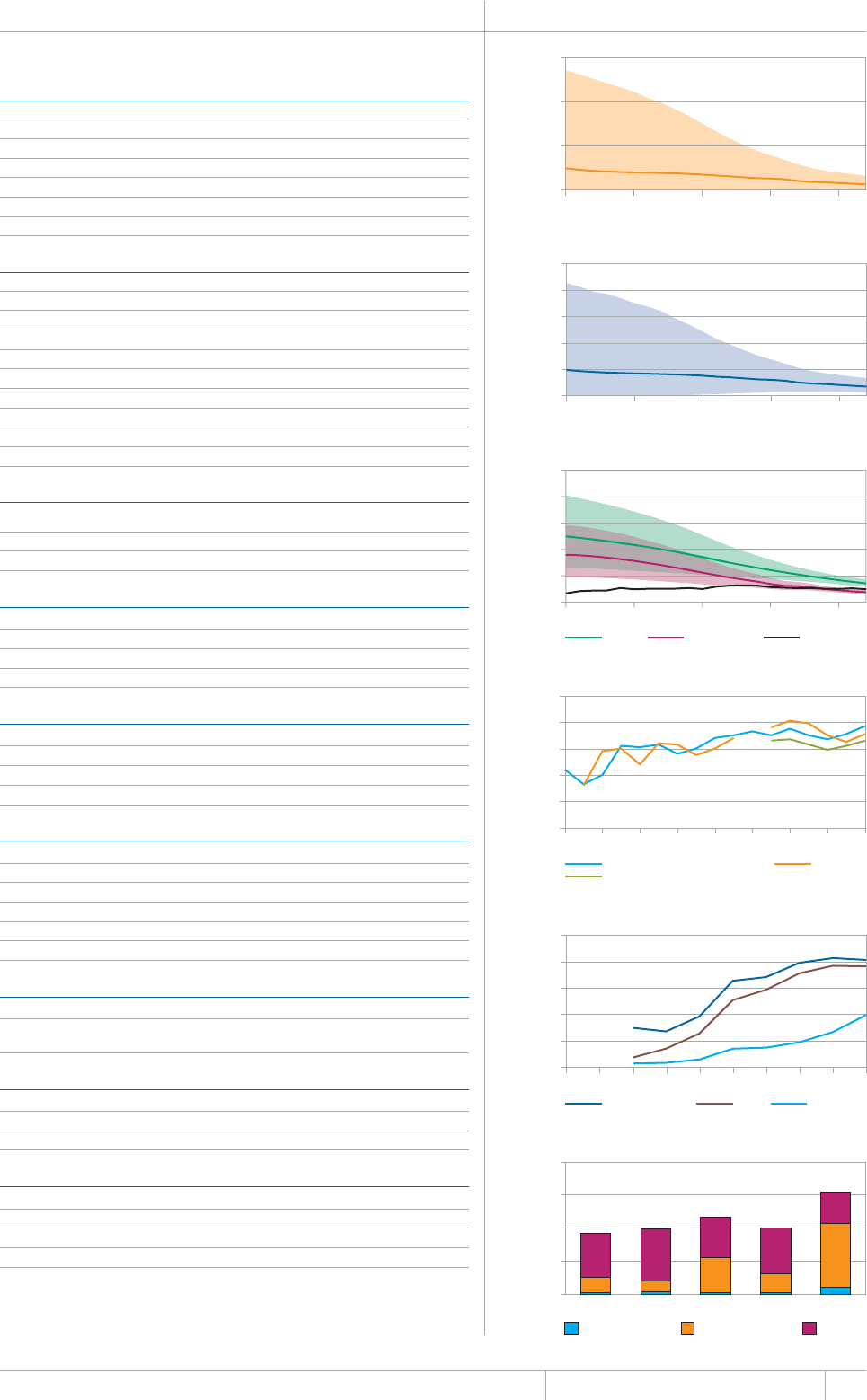
133 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
Data are as reported to WHO. Estimates of TB and MDR-TB burden are produced by WHO in consultation with
countries.
a Ranges represent uncertainty intervals.
UGANDA Population 2012 36 million
HIGH TB BURDEN | HIGH HIV BURDEN
Estimates of TB burdena 2012
07/$'4(thousands)4#6'(per 100 000 population)
/QTVCNKV[GZENWFGU*+86$ ¿ ¿
/QTVCNKV[*+86$QPN[ ¿ ¿
Prevalence (includes HIV+TB) 64 (24–120) 175 (67–334)
Incidence (includes HIV+TB) 65 (53–79) 179 (145–216)
+PEKFGPEG*+86$QPN[ ¿ ¿
%CUGFGVGEVKQPCNNHQTOU ¿
6$ECUGPQVKßECVKQPU
0'9%#5'5 4'64'#6/'06%#5'5
5OGCTRQUKVKXG 4GNCRUG
5OGCTPGICVKXG 6TGCVOGPVCHVGTHCKNWTG
5OGCTWPMPQYPPQVFQPG 6TGCVOGPVCHVGTFGHCWNV
Extrapulmonary 5 143 (12) Other 1 114 (29)
Other 0 (0)
Total new 43 329 Total retreatment 3 882
Other (history unknown) 0
6QVCNPGYCPFTGNCRUG 6QVCNECUGUPQVKßGF
New cases
5/'#40')#6+8'70-0190
5/'#4215+6+8' 016&10' ':64#27./10#4;
/(TCVKQ
#IG
Laboratories 2012
Smear (per 100 000 population) 3.2
Culture (per 5 million population) 0.6
Drug susceptibility testing (per 5 million population) 0.6
+UUGEQPFNKPGFTWIUWUEGRVKDKNKV[VGUVKPICXCKNCDNG! ;GUKPEQWPVT[
6TGCVOGPVUWEEGUUTCVG
New smear-positive and/or culture-positive 77
New smear-negative/extrapulmonary 66
4GVTGCVOGPV
+UTKHCORKEKPWUGFVJTQWIJQWVVTGCVOGPVHQTPGYRCVKGPVU! ;GU
TB/HIV 2012 07/$'4 (%)
6$RCVKGPVUYKVJMPQYP*+8UVCVWU
HIV-positive TB patients 20 376 (50)
*+8RQUKVKXG6$RCVKGPVUQPEQVTKOQZC\QNGRTGXGPVKXGVJGTCR[%26
*+8RQUKVKXG6$RCVKGPVUQPCPVKTGVTQXKTCNVJGTCR[#46
HIV-positive people screened for TB
HIV-positive people provided with IPT
Estimates of MDR-TB burden 2012a 0'9 4'64'#6/'06
QH6$ECUGUYKVJ/&46$ ¿ ¿
/&46$ECUGUCOQPIPQVKßGF
RWNOQPCT[6$ECUGU ¿ ¿
Reported cases of MDR-TB 20120'9 4'64'#6/'06 616#.
%CUGUVGUVGFHQT/&46$
.CDQTCVQT[EQPßTOGF/&46$ECUGU
2CVKGPVUUVCTVGFQP/&46$VTGCVOGPV
Financing TB control 2013
0CVKQPCN6$RTQITCOOGDWFIGV75OKNNKQPU
% Funded domestically 7%
% Funded internationally 62%
% Unfunded 31%
Mortality (excludes HIV+TB) (rate
per 100 000 population per year)
1990 1995 2000 2005 2010
0
100
200
300
Prevalence
(rate per 100 000 population)
1990 1995 2000 2005 2010
0
500
1000
1500
2000
2500
Incidence (rate per 100 000
population per year)
1990 1995 2000 2005 2010
Incidence Incidence (HIV+TB) Notifications
0
250
500
750
1000
1250
Treatment success rate (%)
New smear-positive (and/or culture-positive) Retreatment
New smear-negative/extrapulmonary
1995 1997 1999 2001 2003 2005 2007 2009 2011
0
20
40
60
80
100
Number of patients
2003 2004 2005 2006 2007 2008 2009 2010 2011 2012
HIV-positive TB patients on CPT on ART
0
5000
10 000
15 000
20 000
25 000
Total budget (US$ millions)
Funded domestically Funded internationally Unfunded
2009 2010 2011 2012 2013
0
10
20
30
40
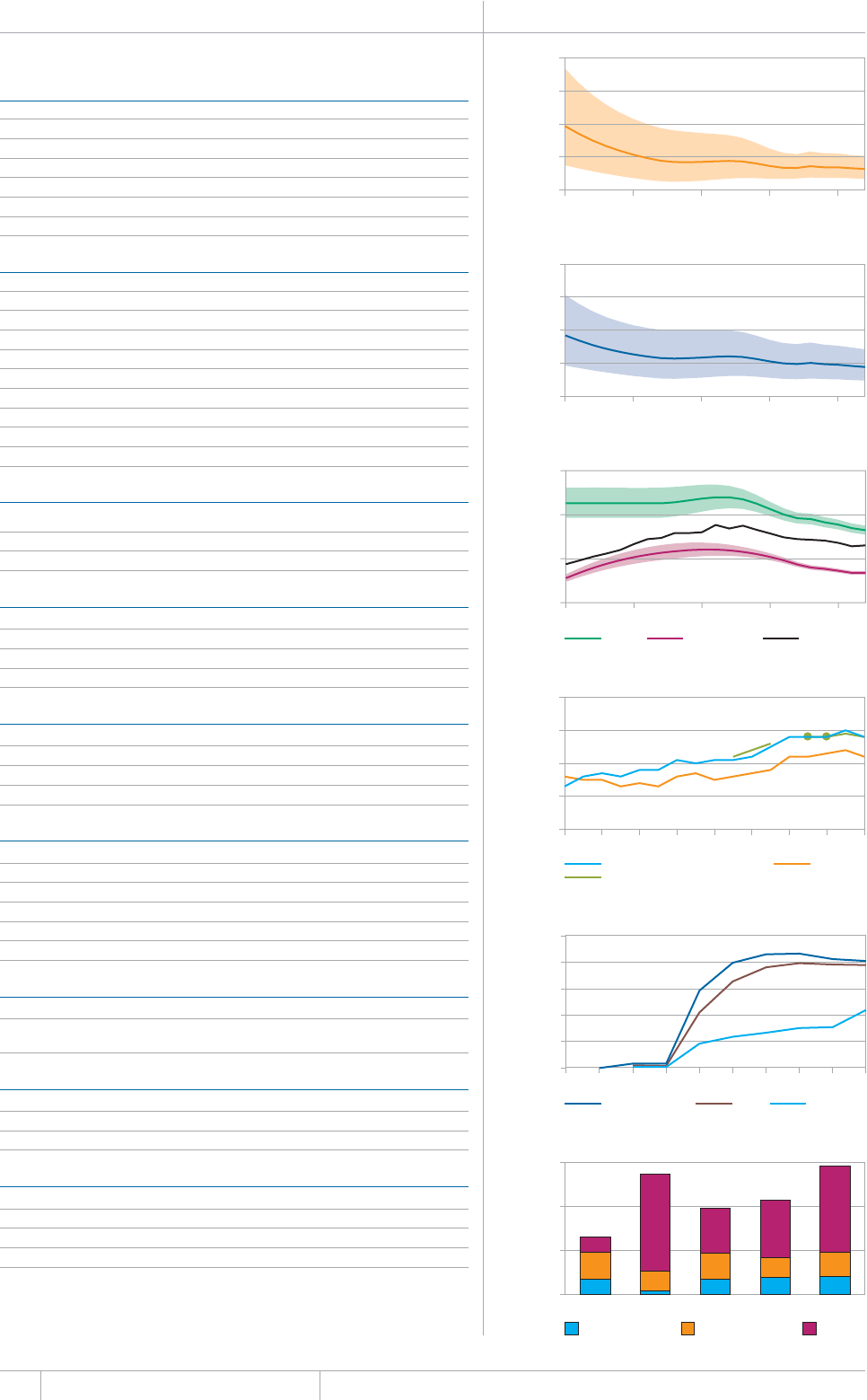
134 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
Data are as reported to WHO. Estimates of TB and MDR-TB burden are produced by WHO in consultation with
countries.
a Ranges represent uncertainty intervals.
UNITED REPUBLIC OF TANZANIA Population 2012 48 million
HIGH TB BURDEN | HIGH HIV BURDEN
Estimates of TB burdena 2012
07/$'4(thousands)4#6'(per 100 000 population)
/QTVCNKV[GZENWFGU*+86$ ¿ ¿
/QTVCNKV[*+86$QPN[ ¿ ¿
2TGXCNGPEGKPENWFGU*+86$ ¿ ¿
+PEKFGPEGKPENWFGU*+86$ ¿ ¿
+PEKFGPEG*+86$QPN[ ¿ ¿
%CUGFGVGEVKQPCNNHQTOU ¿
6$ECUGPQVKßECVKQPU
0'9%#5'5 4'64'#6/'06%#5'5
5OGCTRQUKVKXG 4GNCRUG
Smear-negative 21 393 (35) Treatment after failure 154 (6)
Smear-unknown / not done 0 (0) Treatment after default 201 (7)
Extrapulmonary 14 595 (24) Other 1 359 (49)
Other 0 (0)
Total new 61 126 Total retreatment 2 766
Other (history unknown) 0
6QVCNPGYCPFTGNCRUG 6QVCNECUGUPQVKßGF
New cases
5/'#40')#6+8'70-0190
5/'#4215+6+8' 016&10' ':64#27./10#4;
/(TCVKQ
#IG
Laboratories 2012
Smear (per 100 000 population) 2.0
Culture (per 5 million population) 0.4
Drug susceptibility testing (per 5 million population) 0.1
+UUGEQPFNKPGFTWIUWUEGRVKDKNKV[VGUVKPICXCKNCDNG! ;GUKPEQWPVT[
6TGCVOGPVUWEEGUUTCVG
0GYUOGCTRQUKVKXGCPFQTEWNVWTGRQUKVKXG
0GYUOGCTPGICVKXGGZVTCRWNOQPCT[
4GVTGCVOGPV
+UTKHCORKEKPWUGFVJTQWIJQWVVTGCVOGPVHQTPGYRCVKGPVU! ;GU
TB/HIV 2012 07/$'4 (%)
6$RCVKGPVUYKVJMPQYP*+8UVCVWU
HIV-positive TB patients 20 269 (39)
*+8RQUKVKXG6$RCVKGPVUQPEQVTKOQZC\QNGRTGXGPVKXGVJGTCR[%26
*+8RQUKVKXG6$RCVKGPVUQPCPVKTGVTQXKTCNVJGTCR[#46
HIV-positive people screened for TB 357 400
HIV-positive people provided with IPT
Estimates of MDR-TB burden 2012a 0'9 4'64'#6/'06
QH6$ECUGUYKVJ/&46$ ¿ ¿
/&46$ECUGUCOQPIPQVKßGF
pulmonary TB cases 500 (140–1 300) 0 (0–160)
Reported cases of MDR-TB 20120'9 4'64'#6/'06 616#.
%CUGUVGUVGFHQT/&46$
.CDQTCVQT[EQPßTOGF/&46$ECUGU
2CVKGPVUUVCTVGFQP/&46$VTGCVOGPV
Financing TB control 2013
0CVKQPCN6$RTQITCOOGDWFIGV75OKNNKQPU
% Funded domestically 14%
% Funded internationally 19%
% Unfunded 67%
Mortality (excludes HIV+TB) (rate
per 100 000 population per year)
1990 1995 2000 2005 2010
0
20
40
60
80
Prevalence
(rate per 100 000 population)
1990 1995 2000 2005 2010
0
200
400
600
800
Incidence (rate per 100 000
population per year)
1990 1995 2000 2005 2010
Incidence Incidence (HIV+TB) Notifications
0
100
200
300
Treatment success rate (%)
New smear-positive (and/or culture-positive) Retreatment
New smear-negative/extrapulmonary
60
70
80
90
100
1995 1997 1999 2001 2003 2005 2007 2009 2011
Number of patients
2003 2004 2005 2006 2007 2008 2009 2010 2011 2012
HIV-positive TB patients on CPT on ART
0
5000
10 000
15 000
20 000
25 000
Total budget (US$ millions)
Funded domestically Funded internationally Unfunded
2009 2010 2011 2012 2013
0
20
40
60
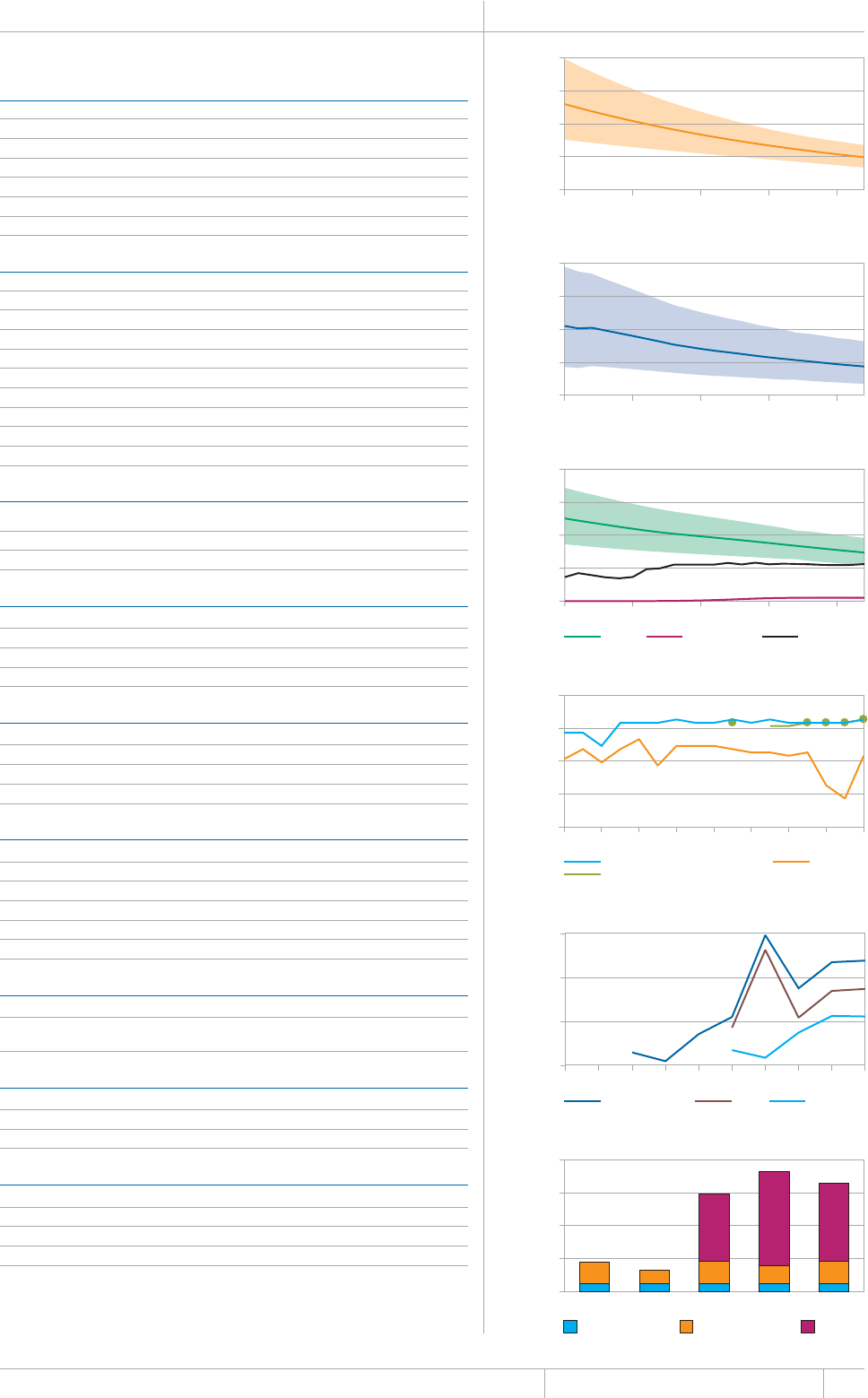
135 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
Data are as reported to WHO. Estimates of TB and MDR-TB burden are produced by WHO in consultation with
countries.
a Ranges represent uncertainty intervals.
VIET NAM Population 2012 91 million
HIGH TB BURDEN | HIGH HIV BURDEN | HIGH MDR-TB BURDEN
Estimates of TB burdena 2012
07/$'4(thousands)4#6'(per 100 000 population)
/QTVCNKV[GZENWFGU*+86$ ¿ ¿
/QTVCNKV[*+86$QPN[ ¿ ¿
2TGXCNGPEGKPENWFGU*+86$ ¿ ¿
Incidence (includes HIV+TB) 130 (99–170) 147 (109–192)
Incidence (HIV+TB only) 9.3 (6.9–12) 10 (7.6–13)
Case detection, all forms (%) 76 (59–100)
6$ECUGPQVKßECVKQPU
0'9%#5'5 4'64'#6/'06%#5'5
5OGCTRQUKVKXG 4GNCRUG
Smear-negative 21 706 (23) Treatment after failure 567 (6)
Smear-unknown / not done Treatment after default 494 (5)
'ZVTCRWNOQPCT[ 1VJGT
Other 3 210 (3)
Total new 94 853 Total retreatment 9 053
Other (history unknown)
6QVCNPGYCPFTGNCRUG 6QVCNECUGUPQVKßGF
New cases
5/'#40')#6+8'70-0190
5/'#4215+6+8' 016&10' ':64#27./10#4;
/(TCVKQ
#IG
Laboratories 2012
Smear (per 100 000 population) 0.9
Culture (per 5 million population) 1.4
Drug susceptibility testing (per 5 million population) 0.1
+UUGEQPFNKPGFTWIUWUEGRVKDKNKV[VGUVKPICXCKNCDNG! ;GUKPEQWPVT[
6TGCVOGPVUWEEGUUTCVG
New smear-positive and/or culture-positive 93
New smear-negative/extrapulmonary 93
4GVTGCVOGPV
Is rifampicin used throughout treatment for new patients? No
TB/HIV 2012 07/$'4 (%)
6$RCVKGPVUYKVJMPQYP*+8UVCVWU
HIV-positive TB patients 4 775 (7)
*+8RQUKVKXG6$RCVKGPVUQPEQVTKOQZC\QNGRTGXGPVKXGVJGTCR[%26
*+8RQUKVKXG6$RCVKGPVUQPCPVKTGVTQXKTCNVJGTCR[#46
HIV-positive people screened for TB
HIV-positive people provided with IPT 5 663
Estimates of MDR-TB burden 2012a 0'9 4'64'#6/'06
QH6$ECUGUYKVJ/&46$ ¿ ¿
/&46$ECUGUCOQPIPQVKßGF
RWNOQPCT[6$ECUGU ¿ ¿
Reported cases of MDR-TB 20120'9 4'64'#6/'06 616#.
%CUGUVGUVGFHQT/&46$
.CDQTCVQT[EQPßTOGF/&46$ECUGU
2CVKGPVUUVCTVGFQP/&46$VTGCVOGPV
Financing TB control 2013
0CVKQPCN6$RTQITCOOGDWFIGV75OKNNKQPU
(WPFGFFQOGUVKECNN[
% Funded internationally 20%
% Unfunded 72%
Mortality (excludes HIV+TB) (rate
per 100 000 population per year)
1990 1995 2000 2005 2010
0
20
40
60
80
Prevalence
(rate per 100 000 population)
1990 1995 2000 2005 2010
0
250
500
750
1000
Incidence (rate per 100 000
population per year)
1990 1995 2000 2005 2010
Incidence Incidence (HIV+TB) Notifications
0
100
200
300
400
Treatment success rate (%)
New smear-positive (and/or culture-positive) Retreatment
New smear-negative/extrapulmonary
60
70
80
90
100
1995 1997 1999 2001 2003 2005 2007 2009 2011
Number of patients
2003 2004 2005 2006 2007 2008 2009 2010 2011 2012
HIV-positive TB patients on CPT on ART
0
2000
4000
6000
Total budget (US$ millions)
Funded domestically Funded internationally Unfunded
2009 2010 2011 2012 2013
0
20
40
60
80
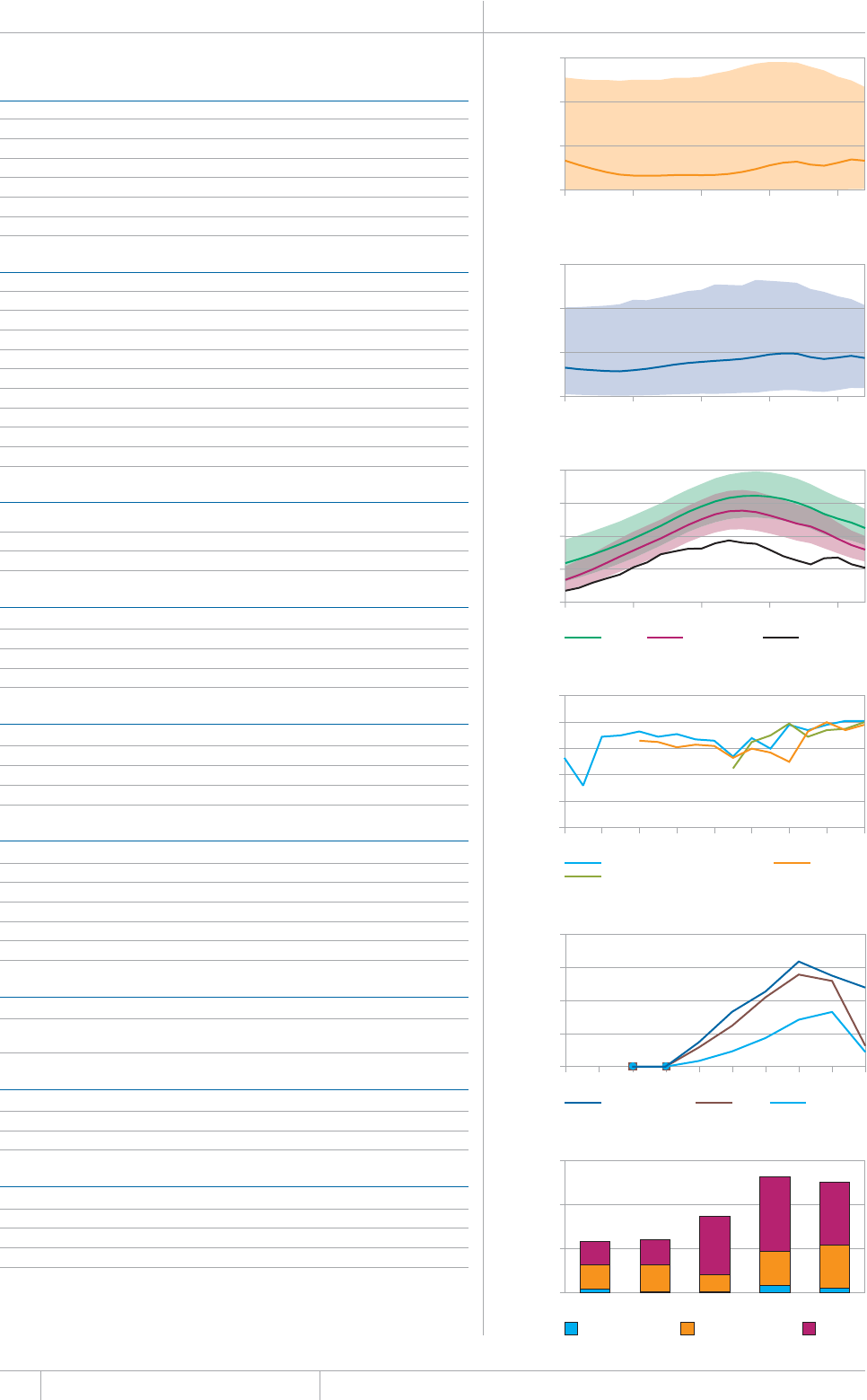
136 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
Data are as reported to WHO. Estimates of TB and MDR-TB burden are produced by WHO in consultation with
countries.
a Ranges represent uncertainty intervals.
ZIMBABWE Population 2012 14 million
HIGH TB BURDEN | HIGH HIV BURDEN
Estimates of TB burdena 2012
07/$'4(thousands)4#6'(per 100 000 population)
Mortality (excludes HIV+TB) 4.6 (0.16–16) 33 (1.2–117)
/QTVCNKV[*+86$QPN[ ¿ ¿
Prevalence (includes HIV+TB) 59 (13–140) 433 (92–1 034)
Incidence (includes HIV+TB) 77 (60–97) 562 (434–706)
+PEKFGPEG*+86$QPN[ ¿ ¿
Case detection, all forms (%) 46 (37–60)
6$ECUGPQVKßECVKQPU
0'9%#5'5 4'64'#6/'06%#5'5
5OGCTRQUKVKXG 4GNCRUG
Smear-negative 14 354 (42) Treatment after failure 200 (5)
Smear-unknown / not done 2 962 (9) Treatment after default 176 (4)
'ZVTCRWNOQPCT[ 1VJGT
Other 0 (0)
Total new 34 391 Total retreatment 4 329
Other (history unknown) 0
6QVCNPGYCPFTGNCRUG 6QVCNECUGUPQVKßGF
New cases
5/'#40')#6+8'70-0190
5/'#4215+6+8' 016&10' ':64#27./10#4;
/(TCVKQ
#IG
Laboratories 2012
Smear (per 100 000 population) 1.3
Culture (per 5 million population) 0.7
Drug susceptibility testing (per 5 million population) 0.7
Is second-line drug susceptibility testing available? No
6TGCVOGPVUWEEGUUTCVG
0GYUOGCTRQUKVKXGCPFQTEWNVWTGRQUKVKXG
0GYUOGCTPGICVKXGGZVTCRWNOQPCT[
4GVTGCVOGPV
+UTKHCORKEKPWUGFVJTQWIJQWVVTGCVOGPVHQTPGYRCVKGPVU! ;GU
TB/HIV 2012 07/$'4 (%)
6$RCVKGPVUYKVJMPQYP*+8UVCVWU
HIV-positive TB patients 23 957 (70)
*+8RQUKVKXG6$RCVKGPVUQPEQVTKOQZC\QNGRTGXGPVKXGVJGTCR[%26
*+8RQUKVKXG6$RCVKGPVUQPCPVKTGVTQXKTCNVJGTCR[#46
HIV-positive people screened for TB
HIV-positive people provided with IPT
Estimates of MDR-TB burden 2012a 0'9 4'64'#6/'06
QH6$ECUGUYKVJ/&46$ ¿ ¿
/&46$ECUGUCOQPIPQVKßGF
pulmonary TB cases 570 (300–960) 360 (76–970)
Reported cases of MDR-TB 20120'9 4'64'#6/'06 616#.
%CUGUVGUVGFHQT/&46$
.CDQTCVQT[EQPßTOGF/&46$ECUGU
2CVKGPVUUVCTVGFQP/&46$VTGCVOGPV
Financing TB control 2013
0CVKQPCN6$RTQITCOOGDWFIGV75OKNNKQPU
% Funded domestically 4%
% Funded internationally 39%
% Unfunded 56%
Mortality (excludes HIV+TB) (rate
per 100 000 population per year)
1990 1995 2000 2005 2010
0
50
100
150
Prevalence
(rate per 100 000 population)
1990 1995 2000 2005 2010
0
500
1000
1500
Incidence (rate per 100 000
population per year)
1990 1995 2000 2005 2010
Incidence Incidence (HIV+TB) Notifications
0
250
500
750
1000
Treatment success rate (%)
New smear-positive (and/or culture-positive) Retreatment
New smear-negative/extrapulmonary
1995 1997 1999 2001 2003 2005 2007 2009 2011
0
20
40
60
80
100
Number of patients
2003 2004 2005 2006 2007 2008 2009 2010 2011 2012
HIV-positive TB patients on CPT on ART
0
10 000
20 000
30 000
40 000
Total budget (US$ millions)
Funded domestically Funded internationally Unfunded
2009 2010 2011 2012 2013
0
15
30
45

#00':
Regional proßles

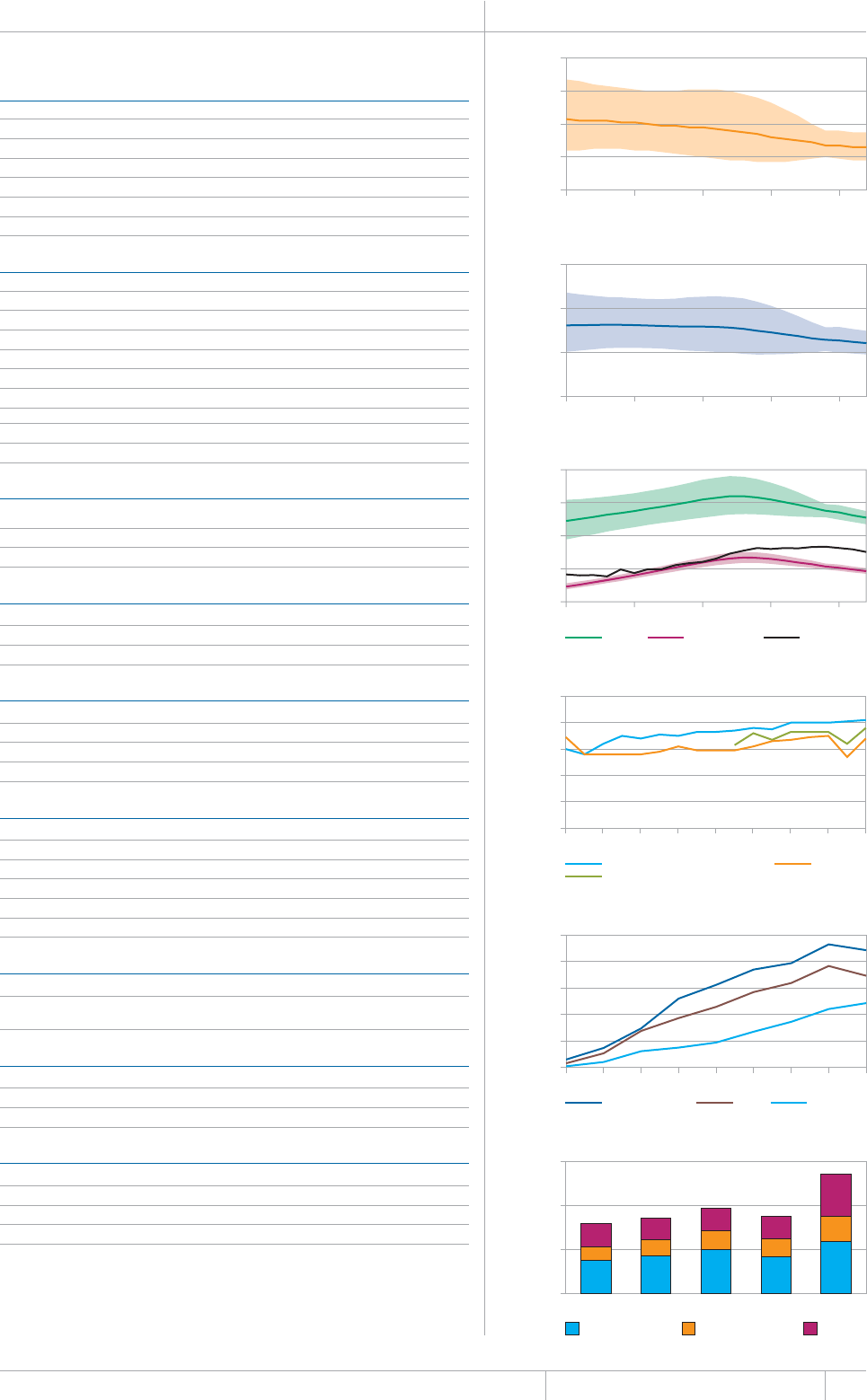
139 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
WHO AFRICAN REGION Population 2012 893 million
WHO MEMBER STATES 46
Estimates of TB burdena 2012
07/$'4thousands 4#6'per 100 000 population)
/QTVCNKV[GZENWFGU*+86$ ¿ ¿
/QTVCNKV[*+86$QPN[ ¿ ¿
Prevalence (includes HIV+TB) 2 700 (2 100–3 300) 303 (239–373)
Incidence (includes HIV+TB) 2 300 (2 100–2 500) 255 (235–275)
+PEKFGPEG*+86$QPN[ ¿ ¿
Case detection, all forms (%) 59 (55–64)
6$ECUGPQVKßECVKQPU
0'9%#5'5 4'64'#6/'06%#5'5
5OGCTRQUKVKXG 4GNCRUG
Smear-negative 345 947 (27) Treatment after failure 9 174 (7.2)
5OGCTWPMPQYPPQVFQPG 6TGCVOGPVCHVGTFGHCWNV
'ZVTCRWNOQPCT[ 1VJGT
1VJGT
Total new 1 282 355 Total retreatment 128 267
Other (history unknown) 2 017
6QVCNPGYCPFTGNCRUG 6QVCNECUGUPQVKßGF
New cases
5/'#40')#6+8'70-0190
5/'#4215+6+8' 016&10' ':64#27./10#4;
/(TCVKQ
#IG
Laboratories 2012 07/$'41(/'/$'456#6'5b
5OGCTRGTRQRWNCVKQPÜ QWVQH
%WNVWTGRGTOKNNKQPRQRWNCVKQPÜ QWVQH
&TWIUWUEGRVKDKNKV[VGUVKPIRGTOKNNKQPRQRWNCVKQPÜ QWVQH
6TGCVOGPVUWEEGUUTCVG
0GYUOGCTRQUKVKXGCPFQTEWNVWTGRQUKVKXG
New smear-negative/extrapulmonary 76
4GVTGCVOGPV
/&46$EQJQTV
TB/HIV 2012 07/$'4 (%)c
TB patients with known HIV status 1 040 292 (74)
*+8RQUKVKXG6$RCVKGPVU
*+8RQUKVKXG6$RCVKGPVUQPEQVTKOQZC\QNGRTGXGPVKXGVJGTCR[%26
*+8RQUKVKXG6$RCVKGPVUQPCPVKTGVTQXKTCNVJGTCR[#46
HIV-positive people screened for TB 2 391 601
HIV-positive people provided with IPT 473 214
Estimates of MDR-TB burden 2012a 0'9 4'64'#6/'06
QH6$ECUGUYKVJ/&46$ ¿ ¿
/&46$ECUGUCOQPIPQVKßGF
pulmonary TB cases 24 000 (2 100–46 000) 14 000 (5 600–22 000)
Reported cases of MDR-TB 20120'9 4'64'#6/'06 616#.
%CUGUVGUVGFHQT/&46$
.CDQTCVQT[EQPßTOGF/&46$ECUGU
2CVKGPVUUVCTVGFQP/&46$VTGCVOGPV
(KPCPEKPI6$EQPVTQNNQYCPFOKFFNGKPEQOGEQWPVTKGUd 2013
0CVKQPCN6$RTQITCOOGDWFIGV75OKNNKQPU
% Funded domestically 44
% Funded internationally 21
% Unfunded 36
&CVCCTGCUTGRQTVGFVQ9*1'UVKOCVGUQH6$CPF/&46$DWTFGPCTGRTQFWEGFD[9*1KPEQPUWNVCVKQPYKVJ
countries.
a 4CPIGUTGRTGUGPVWPEGTVCKPV[KPVGTXCNU
b Data are not collected from all Member States.
c Calculations exclude countries with missing numerators or denominators.
d Financing indicators exclude funding for general healthcare services provided outside NTPs.
Mortality (excludes HIV+TB) (rate
per 100 000 population per year)
1990 1995 2000 2005 2010
0
20
40
60
80
Prevalence
(rate per 100 000 population)
1990 1995 2000 2005 2010
0
250
500
750
Incidence (rate per 100 000
population per year)
1990 1995 2000 2005 2010
Incidence Incidence (HIV+TB) Notifications
0
100
200
300
400
Treatment success rate (%)
New smear-positive (and/or culture-positive) Retreatment
New smear-negative/extrapulmonary
1995 1997 1999 2001 2003 2005 2007 2009 2011
0
20
40
60
80
100
Number of patients
HIV-positive TB patients on CPT on ART
2004 2005 2006 2007 2008 2009 2010 2011 2012
0
100
200
300
400
500
Total budget (US$ millions)
Funded domestically Funded internationally Unfunded
2009 2010 2011 2012 2013
0
500
1000
1500
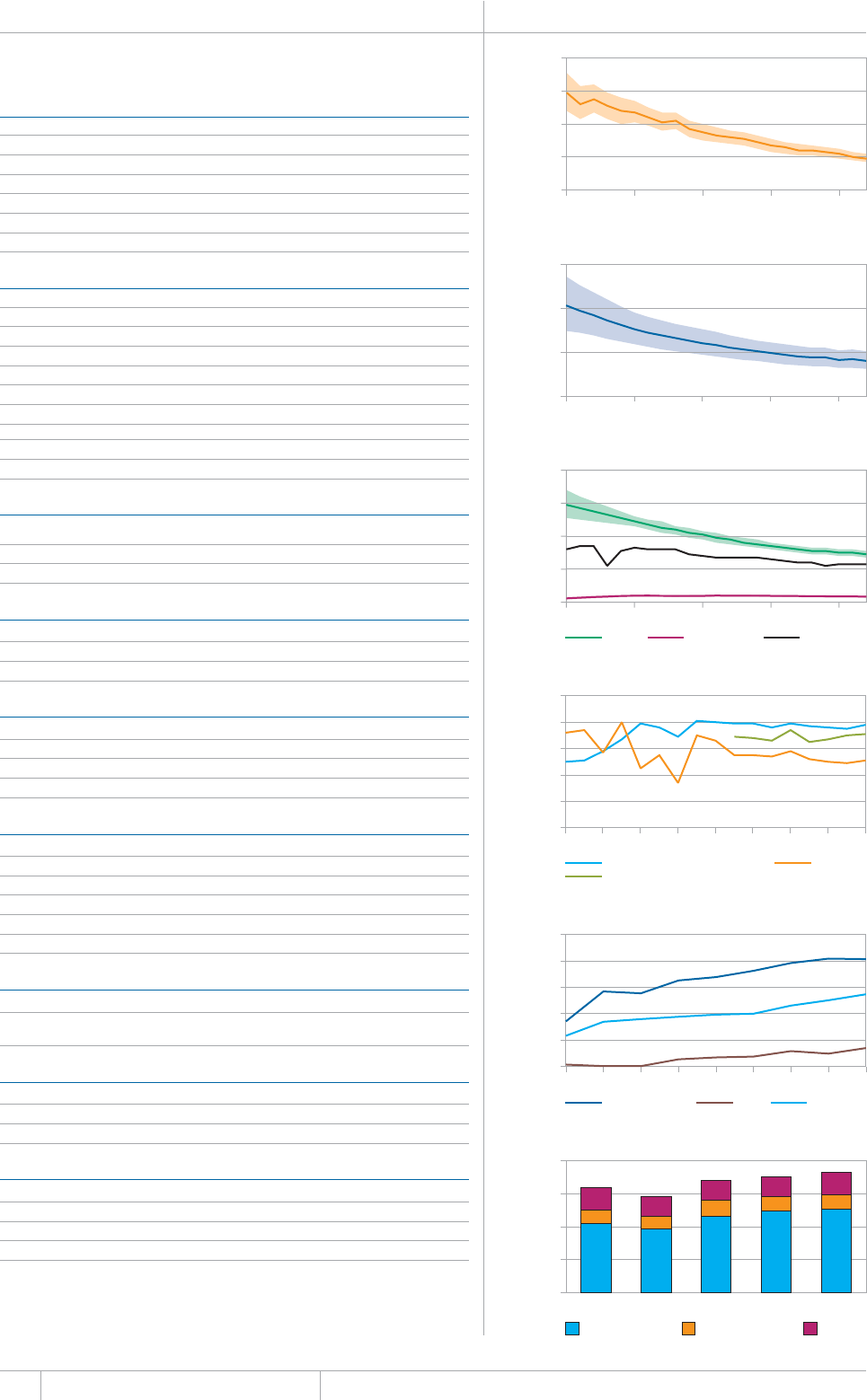
140 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
WHO REGION OF THE AMERICAS Population 2012 961 million
WHO MEMBER STATES 35
OTHER COUNTRIES AND TERRITORIES 11
Estimates of TB burdena 2012
07/$'4thousands 4#6'per 100 000 population)
Mortality (excludes HIV+TB) 19 (16–21) 1.9 (1.7–2.2)
/QTVCNKV[*+86$QPN[ ¿ ¿
Prevalence (includes HIV+TB) 390 (300–490) 40 (31–51)
+PEKFGPEGKPENWFGU*+86$ ¿ ¿
+PEKFGPEG*+86$QPN[ ¿ ¿
%CUGFGVGEVKQPCNNHQTOU ¿
6$ECUGPQVKßECVKQPU
0'9%#5'5 4'64'#6/'06%#5'5
5OGCTRQUKVKXG 4GNCRUG
Smear-negative 35 606 (17) Treatment after failure 1 195 (5.0)
5OGCTWPMPQYPPQVFQPG 6TGCVOGPVCHVGTFGHCWNV
'ZVTCRWNOQPCT[ 1VJGT
1VJGT
Total new 208 845 Total retreatment 23 811
Other (history unknown) 39
6QVCNPGYCPFTGNCRUG 6QVCNECUGUPQVKßGF
New cases
5/'#40')#6+8'70-0190
5/'#4215+6+8' 016&10' ':64#27./10#4;
/(TCVKQ
#IG
Laboratories 2012 07/$'41(/'/$'456#6'5b
5OGCTRGTRQRWNCVKQPÜ QWVQH
%WNVWTGRGTOKNNKQPRQRWNCVKQPÜ QWVQH
&TWIUWUEGRVKDKNKV[VGUVKPIRGTOKNNKQPRQRWNCVKQPÜ QWVQH
6TGCVOGPVUWEEGUUTCVG
0GYUOGCTRQUKVKXGCPFQTEWNVWTGRQUKVKXG
New smear-negative/extrapulmonary 71
4GVTGCVOGPV
/&46$EQJQTV
TB/HIV 2012 07/$'4 (%)c
TB patients with known HIV status 129 174 (56)
HIV-positive TB patients 20 355 (16)
*+8RQUKVKXG6$RCVKGPVUQPEQVTKOQZC\QNGRTGXGPVKXGVJGTCR[%26
*+8RQUKVKXG6$RCVKGPVUQPCPVKTGVTQXKTCNVJGTCR[#46
*+8RQUKVKXGRGQRNGUETGGPGFHQT6$
*+8RQUKVKXGRGQRNGRTQXKFGFYKVJ+26
Estimates of MDR-TB burden 2012a 0'9 4'64'#6/'06
QH6$ECUGUYKVJ/&46$ ¿ ¿
/&46$ECUGUCOQPIPQVKßGF
RWNOQPCT[6$ECUGU ¿ ¿
Reported cases of MDR-TB 20120'9 4'64'#6/'06 616#.
%CUGUVGUVGFHQT/&46$
.CDQTCVQT[EQPßTOGF/&46$ECUGU
2CVKGPVUUVCTVGFQP/&46$VTGCVOGPV
(KPCPEKPI6$EQPVTQNNQYCPFOKFFNGKPEQOGEQWPVTKGUd 2013
0CVKQPCN6$RTQITCOOGDWFIGV75OKNNKQPU
% Funded domestically 69
% Funded internationally 12
% Unfunded 19
Mortality (excludes HIV+TB) (rate
per 100 000 population per year)
1990 1995 2000 2005 2010
0
2
4
6
8
Prevalence
(rate per 100 000 population)
1990 1995 2000 2005 2010
0
50
100
150
Incidence (rate per 100 000
population per year)
1990 1995 2000 2005 2010
Incidence Incidence (HIV+TB) Notifications
0
20
40
60
80
Treatment success rate (%)
New smear-positive (and/or culture-positive) Retreatment
New smear-negative/extrapulmonary
1995 1997 1999 2001 2003 2005 2007 2009 2011
0
20
40
60
80
100
Number of patients
HIV-positive TB patients on CPT on ART
2004 2005 2006 2007 2008 2009 2010 2011 2012
0
5
10
15
20
25
Total budget (US$ millions)
Funded domestically Funded internationally Unfunded
2009 2010 2011 2012 2013
0
50
100
150
200
&CVCCTGCUTGRQTVGFVQ9*1'UVKOCVGUQH6$CPF/&46$DWTFGPCTGRTQFWEGFD[9*1KPEQPUWNVCVKQPYKVJ
countries.
a 4CPIGUTGRTGUGPVWPEGTVCKPV[KPVGTXCNU
b Data are not collected from all Member States.
c Calculations exclude countries with missing numerators or denominators.
d Financing indicators exclude funding for general healthcare services provided outside NTPs.
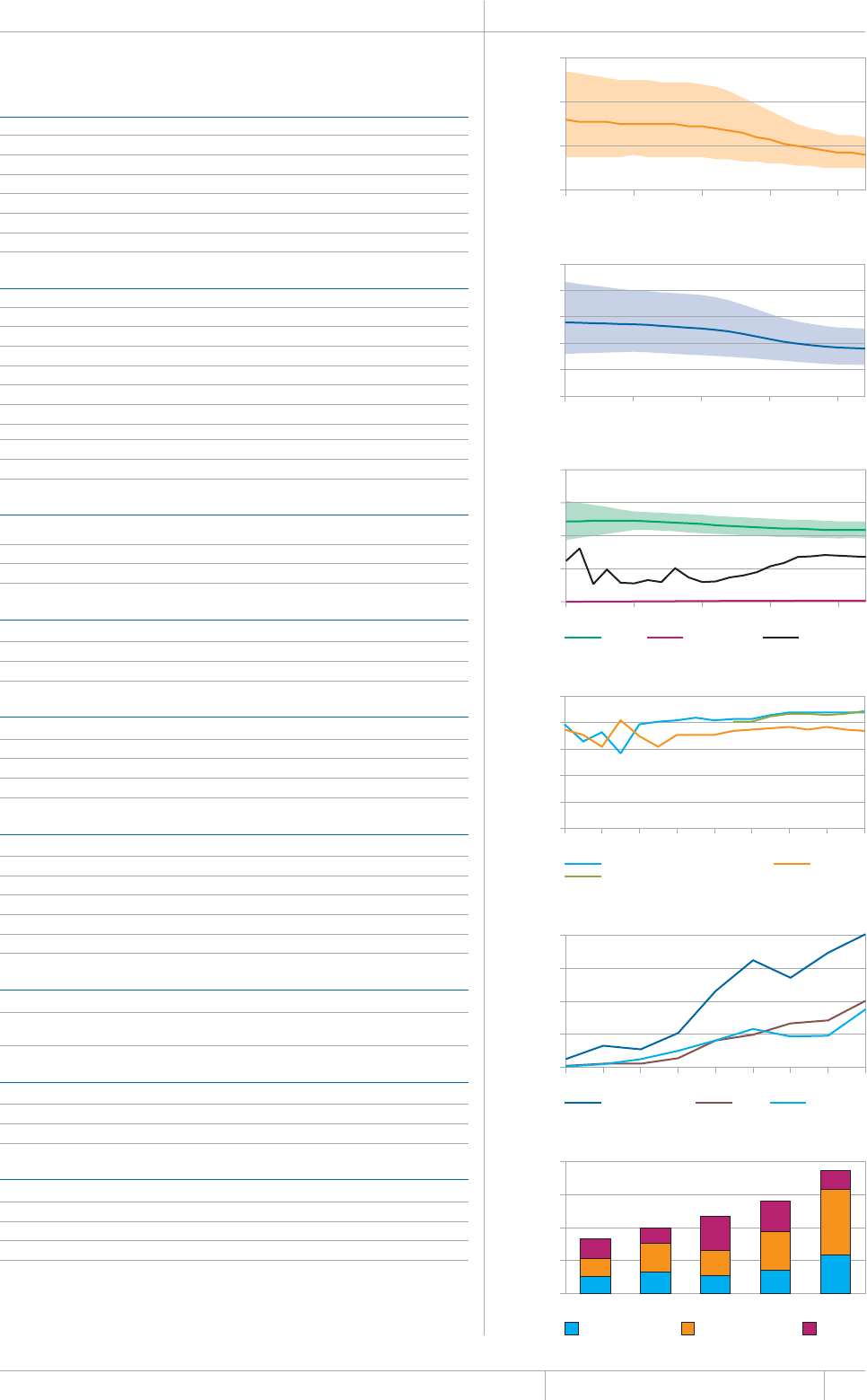
141 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
WHO EASTERN MEDITERRANEAN REGION Population 2012 617 million
WHO MEMBER STATES 22
OTHER COUNTRIES AND TERRITORIES 1
Estimates of TB burdena 2012
07/$'4thousands 4#6'per 100 000 population)
Mortality (excludes HIV+TB) 100 (63–150) 16 (10–24)
/QTVCNKV[*+86$QPN[ ¿ ¿
2TGXCNGPEGKPENWFGU*+86$ ¿ ¿
Incidence (includes HIV+TB) 670 (590–750) 109 (96–122)
+PEKFGPEG*+86$QPN[ ¿ ¿
Case detection, all forms (%) 63 (56–71)
6$ECUGPQVKßECVKQPU
0'9%#5'5 4'64'#6/'06%#5'5
5OGCTRQUKVKXG 4GNCRUG
Smear-negative 135 346 (33) Treatment after failure 2 007 (9.5)
5OGCTWPMPQYPPQVFQPG 6TGCVOGPVCHVGTFGHCWNV
Extrapulmonary 90 943 (22) Other 5 200 (24)
1VJGT
Total new 409 477 Total retreatment 21 228
1VJGTJKUVQT[WPMPQYP
6QVCNPGYCPFTGNCRUG 6QVCNECUGUPQVKßGF
New cases
5/'#40')#6+8'70-0190
5/'#4215+6+8' 016&10' ':64#27./10#4;
/(TCVKQ
#IG
Laboratories 2012 07/$'41(/'/$'456#6'5b
5OGCTRGTRQRWNCVKQPÜ QWVQH
%WNVWTGRGTOKNNKQPRQRWNCVKQPÜ QWVQH
&TWIUWUEGRVKDKNKV[VGUVKPIRGTOKNNKQPRQRWNCVKQPÜ QWVQH
6TGCVOGPVUWEEGUUTCVG
0GYUOGCTRQUKVKXGCPFQTEWNVWTGRQUKVKXG
0GYUOGCTPGICVKXGGZVTCRWNOQPCT[
4GVTGCVOGPV
/&46$EQJQTV
TB/HIV 2012 07/$'4 (%)c
6$RCVKGPVUYKVJMPQYP*+8UVCVWU
HIV-positive TB patients 2 020 (3.5)
*+8RQUKVKXG6$RCVKGPVUQPEQVTKOQZC\QNGRTGXGPVKXGVJGTCR[%26
*+8RQUKVKXG6$RCVKGPVUQPCPVKTGVTQXKTCNVJGTCR[#46
HIV-positive people screened for TB 15 012
HIV-positive people provided with IPT 243
Estimates of MDR-TB burden 2012a 0'9 4'64'#6/'06
QH6$ECUGUYKVJ/&46$ ¿ ¿
/&46$ECUGUCOQPIPQVKßGF
pulmonary TB cases 11 000 (320–36 000) 6 900 (2 400–11 000)
Reported cases of MDR-TB 20120'9 4'64'#6/'06 616#.
%CUGUVGUVGFHQT/&46$
.CDQTCVQT[EQPßTOGF/&46$ECUGU
2CVKGPVUUVCTVGFQP/&46$VTGCVOGPV
(KPCPEKPI6$EQPVTQNNQYCPFOKFFNGKPEQOGEQWPVTKGUd 2013
0CVKQPCN6$RTQITCOOGDWFIGV75OKNNKQPU
% Funded domestically 32
% Funded internationally 53
% Unfunded 16
Mortality (excludes HIV+TB) (rate
per 100 000 population per year)
1990 1995 2000 2005 2010
0
20
40
60
Prevalence
(rate per 100 000 population)
1990 1995 2000 2005 2010
0
100
200
300
400
500
Incidence (rate per 100 000
population per year)
1990 1995 2000 2005 2010
Incidence Incidence (HIV+TB) Notifications
0
50
100
150
200
Treatment success rate (%)
New smear-positive (and/or culture-positive) Retreatment
New smear-negative/extrapulmonary
1995 1997 1999 2001 2003 2005 2007 2009 2011
0
20
40
60
80
100
Number of patients
HIV-positive TB patients on CPT on ART
2004 2005 2006 2007 2008 2009 2010 2011 2012
0
0.5
1.0
1.5
2.0
Total budget (US$ millions)
Funded domestically Funded internationally Unfunded
2009 2010 2011 2012 2013
0
50
100
150
200
&CVCCTGCUTGRQTVGFVQ9*1'UVKOCVGUQH6$CPF/&46$DWTFGPCTGRTQFWEGFD[9*1KPEQPUWNVCVKQPYKVJ
countries.
a 4CPIGUTGRTGUGPVWPEGTVCKPV[KPVGTXCNU
b Data are not collected from all Member States.
c Calculations exclude countries with missing numerators or denominators.
d Financing indicators exclude funding for general healthcare services provided outside NTPs.
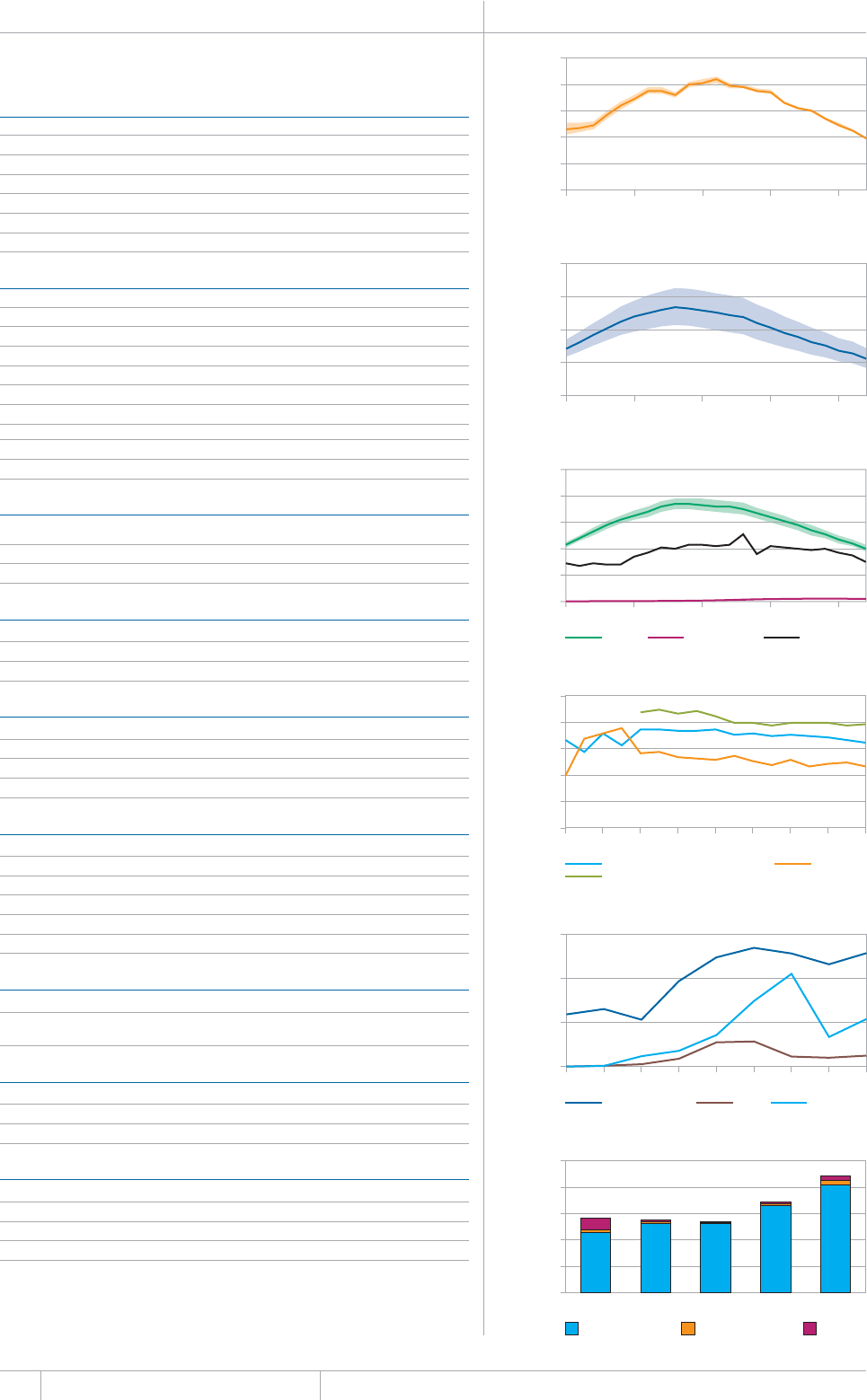
142 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
WHO EUROPEAN REGION Population 2012 905 million
WHO MEMBER STATES 53
OTHER COUNTRIES AND TERRITORIES 1
Estimates of TB burdena 2012
07/$'4thousands 4#6'per 100 000 population)
Mortality (excludes HIV+TB) 36 (35–36) 3.9 (3.9–4)
/QTVCNKV[*+86$QPN[ ¿ ¿
2TGXCNGPEGKPENWFGU*+86$ ¿ ¿
+PEKFGPEGKPENWFGU*+86$ ¿ ¿
Incidence (HIV+TB only) 19 (17–21) 2.1 (1.9–2.3)
Case detection, all forms (%) 74 (70–79)
6$ECUGPQVKßECVKQPU
0'9%#5'5 4'64'#6/'06%#5'5
5OGCTRQUKVKXG 4GNCRUG
5OGCTPGICVKXG 6TGCVOGPVCHVGTHCKNWTG
5OGCTWPMPQYPPQVFQPG 6TGCVOGPVCHVGTFGHCWNV
Extrapulmonary 39 029 (16) Other 51 237 (55)
1VJGT
Total new 242 266 Total retreatment 92 847
Other (history unknown) 2 054
6QVCNPGYCPFTGNCRUG 6QVCNECUGUPQVKßGF
New cases
5/'#40')#6+8'70-0190
5/'#4215+6+8' 016&10' ':64#27./10#4;
/(TCVKQ
#IG
Laboratories 2012 07/$'41(/'/$'456#6'5b
5OGCTRGTRQRWNCVKQPÜ QWVQH
%WNVWTGRGTOKNNKQPRQRWNCVKQPÜ QWVQH
&TWIUWUEGRVKDKNKV[VGUVKPIRGTOKNNKQPRQRWNCVKQPÜ QWVQH
6TGCVOGPVUWEEGUUTCVG
New smear-positive and/or culture-positive 65
New smear-negative/extrapulmonary 79
4GVTGCVOGPV
/&46$EQJQTV
TB/HIV 2012 07/$'4 (%)c
TB patients with known HIV status 203 705 (60)
HIV-positive TB patients 12 900 (6.3)
*+8RQUKVKXG6$RCVKGPVUQPEQVTKOQZC\QNGRTGXGPVKXGVJGTCR[%26
*+8RQUKVKXG6$RCVKGPVUQPCPVKTGVTQXKTCNVJGTCR[#46
HIV-positive people screened for TB 23 567
*+8RQUKVKXGRGQRNGRTQXKFGFYKVJ+26
Estimates of MDR-TB burden 2012a 0'9 4'64'#6/'06
QH6$ECUGUYKVJ/&46$ ¿ ¿
/&46$ECUGUCOQPIPQVKßGF
RWNOQPCT[6$ECUGU ¿ ¿
Reported cases of MDR-TB 20120'9 4'64'#6/'06 616#.
%CUGUVGUVGFHQT/&46$
.CDQTCVQT[EQPßTOGF/&46$ECUGU
2CVKGPVUUVCTVGFQP/&46$VTGCVOGPV
(KPCPEKPI6$EQPVTQNNQYCPFOKFFNGKPEQOGEQWPVTKGUd 2013
0CVKQPCN6$RTQITCOOGDWFIGV75OKNNKQPU
% Funded domestically 92
% Funded internationally 3.7
% Unfunded 4.3
Mortality (excludes HIV+TB) (rate
per 100 000 population per year)
1990 1995 2000 2005 2010
0
2
4
6
8
10
Prevalence
(rate per 100 000 population)
1990 1995 2000 2005 2010
0
50
100
150
200
Incidence (rate per 100 000
population per year)
1990 1995 2000 2005 2010
Incidence Incidence (HIV+TB) Notifications
0
20
40
60
80
100
Treatment success rate (%)
New smear-positive (and/or culture-positive) Retreatment
New smear-negative/extrapulmonary
1995 1997 1999 2001 2003 2005 2007 2009 2011
0
20
40
60
80
100
Number of patients
HIV-positive TB patients on CPT on ART
2004 2005 2006 2007 2008 2009 2010 2011 2012
0
5
10
15
Total budget (US$ millions)
Funded domestically Funded internationally Unfunded
2009 2010 2011 2012 2013
0
500
1000
1500
2000
2500
&CVCCTGCUTGRQTVGFVQ9*1'UVKOCVGUQH6$CPF/&46$DWTFGPCTGRTQFWEGFD[9*1KPEQPUWNVCVKQPYKVJ
countries.
a 4CPIGUTGRTGUGPVWPEGTVCKPV[KPVGTXCNU
b Data are not collected from all Member States.
c Calculations exclude countries with missing numerators or denominators.
d Financing indicators exclude funding for general healthcare services provided outside NTPs.

143 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
WHO SOUTH-EAST ASIA REGION Population 2012 1 833 million
WHO MEMBER STATES 11
Estimates of TB burdena 2012
07/$'4thousands 4#6'per 100 000 population)
/QTVCNKV[GZENWFGU*+86$ ¿ ¿
/QTVCNKV[*+86$QPN[ ¿ ¿
2TGXCNGPEGKPENWFGU*+86$ ¿ ¿
+PEKFGPEGKPENWFGU*+86$ ¿ ¿
+PEKFGPEG*+86$QPN[ ¿ ¿
%CUGFGVGEVKQPCNNHQTOU ¿
6$ECUGPQVKßECVKQPU
0'9%#5'5 4'64'#6/'06%#5'5
5OGCTRQUKVKXG 4GNCRUG
5OGCTPGICVKXG 6TGCVOGPVCHVGTHCKNWTG
Smear-unknown / not done 0 (0) Treatment after default 69 100 (21)
'ZVTCRWNOQPCT[ 1VJGT
1VJGT
Total new 1 993 614 Total retreatment 332 580
Other (history unknown) 5 261
6QVCNPGYCPFTGNCRUG 6QVCNECUGUPQVKßGF
New cases
5/'#40')#6+8'70-0190
5/'#4215+6+8' 016&10' ':64#27./10#4;
/(TCVKQ
#IG
Laboratories 2012 07/$'41(/'/$'456#6'5b
5OGCTRGTRQRWNCVKQPÜ QWVQH
%WNVWTGRGTOKNNKQPRQRWNCVKQPÜ QWVQH
&TWIUWUEGRVKDKNKV[VGUVKPIRGTOKNNKQPRQRWNCVKQPÜ QWVQH
6TGCVOGPVUWEEGUUTCVG
0GYUOGCTRQUKVKXGCPFQTEWNVWTGRQUKVKXG
0GYUOGCTPGICVKXGGZVTCRWNOQPCT[
4GVTGCVOGPV
/&46$EQJQTV
TB/HIV 2012 07/$'4 (%)c
TB patients with known HIV status 904 223 (39)
HIV-positive TB patients 56 093 (6.2)
*+8RQUKVKXG6$RCVKGPVUQPEQVTKOQZC\QNGRTGXGPVKXGVJGTCR[%26
*+8RQUKVKXG6$RCVKGPVUQPCPVKTGVTQXKTCNVJGTCR[#46
*+8RQUKVKXGRGQRNGUETGGPGFHQT6$
*+8RQUKVKXGRGQRNGRTQXKFGFYKVJ+26
Estimates of MDR-TB burden 2012a 0'9 4'64'#6/'06
QH6$ECUGUYKVJ/&46$ ¿ ¿
/&46$ECUGUCOQPIPQVKßGF
pulmonary TB cases 36 000 (26 000–46 000) 54 000 (37 000–70 000)
Reported cases of MDR-TB 20120'9 4'64'#6/'06 616#.
%CUGUVGUVGFHQT/&46$
.CDQTCVQT[EQPßTOGF/&46$ECUGU
2CVKGPVUUVCTVGFQP/&46$VTGCVOGPV
(KPCPEKPI6$EQPVTQNNQYCPFOKFFNGKPEQOGEQWPVTKGUd 2013
0CVKQPCN6$RTQITCOOGDWFIGV75OKNNKQPU
% Funded domestically 30
% Funded internationally 41
% Unfunded 29
Mortality (excludes HIV+TB) (rate
per 100 000 population per year)
1990 1995 2000 2005 2010
0
20
40
60
80
Prevalence
(rate per 100 000 population)
1990 1995 2000 2005 2010
0
200
400
600
Incidence (rate per 100 000
population per year)
1990 1995 2000 2005 2010
Incidence Incidence (HIV+TB) Notifications
0
100
200
300
Treatment success rate (%)
New smear-positive (and/or culture-positive) Retreatment
New smear-negative/extrapulmonary
1995 1997 1999 2001 2003 2005 2007 2009 2011
0
20
40
60
80
100
Number of patients
HIV-positive TB patients on CPT on ART
2004 2005 2006 2007 2008 2009 2010 2011 2012
0
20
40
60
Total budget (US$ millions)
Funded domestically Funded internationally Unfunded
2009 2010 2011 2012 2013
0
200
400
600
&CVCCTGCUTGRQTVGFVQ9*1'UVKOCVGUQH6$CPF/&46$DWTFGPCTGRTQFWEGFD[9*1KPEQPUWNVCVKQPYKVJ
countries.
a 4CPIGUTGRTGUGPVWPEGTVCKPV[KPVGTXCNU
b Data are not collected from all Member States.
c Calculations exclude countries with missing numerators or denominators.
d Financing indicators exclude funding for general healthcare services provided outside NTPs.
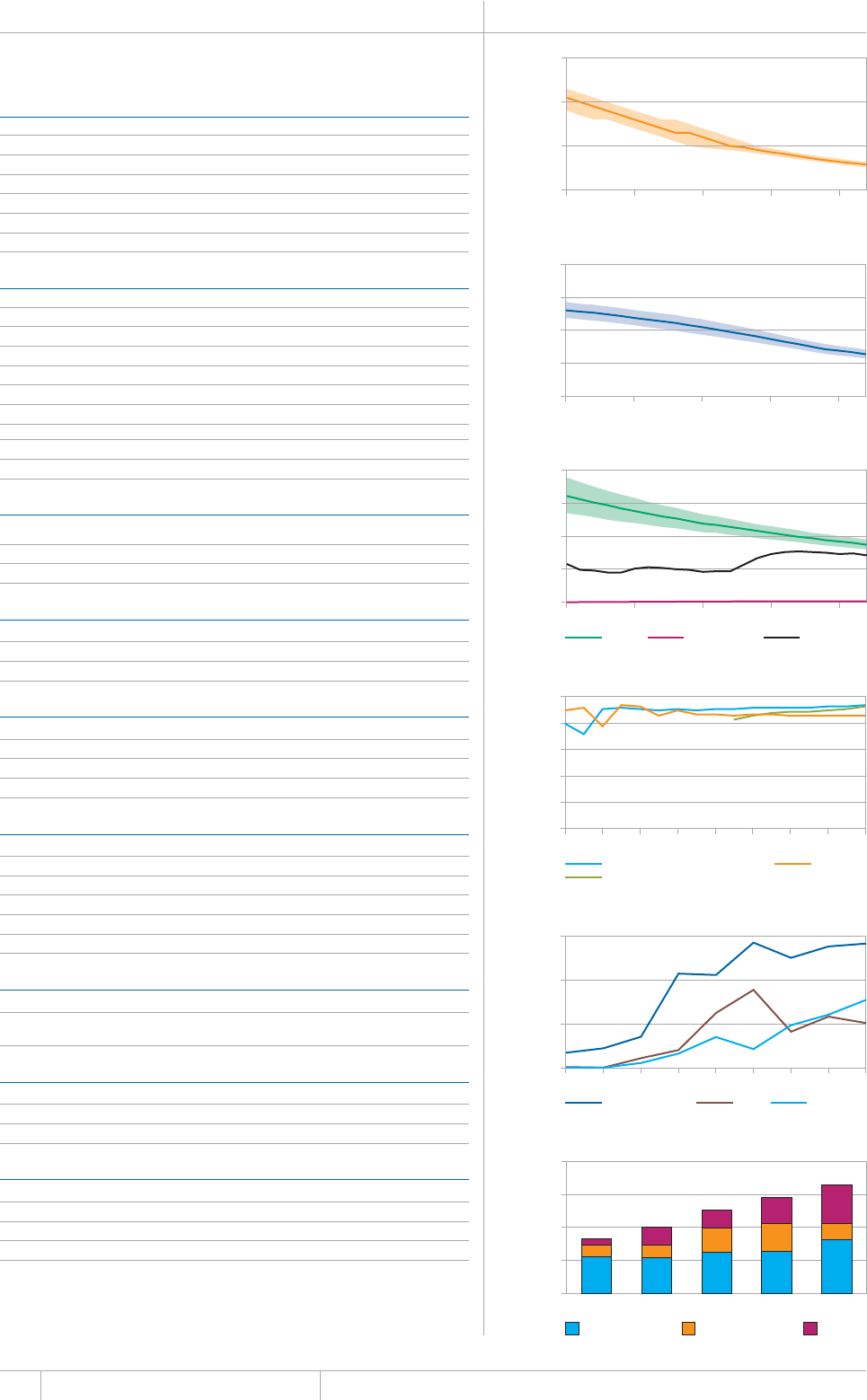
144 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
WHO WESTERN PACIFIC REGION Population 2012 1 846 million
WHO MEMBER STATES 27
OTHER COUNTRIES AND TERRITORIES 9
Estimates of TB burdena 2012
07/$'4thousands 4#6'per 100 000 population)
/QTVCNKV[GZENWFGU*+86$ ¿ ¿
Mortality (HIV+TB only) 5 (4–5) 0.26 (0.23–0.29)
2TGXCNGPEGKPENWFGU*+86$ ¿ ¿
+PEKFGPEGKPENWFGU*+86$ ¿ ¿
Incidence (HIV+TB only) 24 (21–27) 1.3 (1.1–1.5)
%CUGFGVGEVKQPCNNHQTOU ¿
6$ECUGPQVKßECVKQPU
0'9%#5'5 4'64'#6/'06%#5'5
5OGCTRQUKVKXG 4GNCRUG
Smear-negative 691 714 (55) Treatment after failure 3 714 (4.6)
5OGCTWPMPQYPPQVFQPG 6TGCVOGPVCHVGTFGHCWNV
'ZVTCRWNOQPCT[ 1VJGT
1VJGT
Total new 1 264 217 Total retreatment 80 017
Other (history unknown) 1 232
6QVCNPGYCPFTGNCRUG 6QVCNECUGUPQVKßGF
New cases
5/'#40')#6+8'70-0190
5/'#4215+6+8' 016&10' ':64#27./10#4;
/(TCVKQ
#IG
Laboratories 2012 07/$'41(/'/$'456#6'5b
5OGCTRGTRQRWNCVKQPÜ QWVQH
%WNVWTGRGTOKNNKQPRQRWNCVKQPÜ QWVQH
&TWIUWUEGRVKDKNKV[VGUVKPIRGTOKNNKQPRQRWNCVKQPÜ QWVQH
6TGCVOGPVUWEEGUUTCVG
New smear-positive and/or culture-positive 94
New smear-negative/extrapulmonary 93
4GVTGCVOGPV
/&46$EQJQTV
TB/HIV 2012 07/$'4 (%)c
TB patients with known HIV status 451 302 (34)
HIV-positive TB patients 14 119 (3.1)
*+8RQUKVKXG6$RCVKGPVUQPEQVTKOQZC\QNGRTGXGPVKXGVJGTCR[%26
*+8RQUKVKXG6$RCVKGPVUQPCPVKTGVTQXKTCNVJGTCR[#46
*+8RQUKVKXGRGQRNGUETGGPGFHQT6$
*+8RQUKVKXGRGQRNGRTQXKFGFYKVJ+26
Estimates of MDR-TB burden 2012a 0'9 4'64'#6/'06
QH6$ECUGUYKVJ/&46$ ¿ ¿
/&46$ECUGUCOQPIPQVKßGF
RWNOQPCT[6$ECUGU ¿ ¿
Reported cases of MDR-TB 20120'9 4'64'#6/'06 616#.
%CUGUVGUVGFHQT/&46$
.CDQTCVQT[EQPßTOGF/&46$ECUGU
2CVKGPVUUVCTVGFQP/&46$VTGCVOGPV
(KPCPEKPI6$EQPVTQNNQYCPFOKFFNGKPEQOGEQWPVTKGUd 2013
0CVKQPCN6$RTQITCOOGDWFIGV75OKNNKQPU
% Funded domestically 50
% Funded internationally 15
% Unfunded 36
Mortality (excludes HIV+TB) (rate
per 100 000 population per year)
1990 1995 2000 2005 2010
0
10
20
30
Prevalence
(rate per 100 000 population)
1990 1995 2000 2005 2010
0
100
200
300
400
Incidence (rate per 100 000
population per year)
1990 1995 2000 2005 2010
Incidence Incidence (HIV+TB) Notifications
0
50
100
150
200
Treatment success rate (%)
New smear-positive (and/or culture-positive) Retreatment
New smear-negative/extrapulmonary
1995 1997 1999 2001 2003 2005 2007 2009 2011
0
20
40
60
80
100
Number of patients
HIV-positive TB patients on CPT on ART
2004 2005 2006 2007 2008 2009 2010 2011 2012
0
5
10
15
Total budget (US$ millions)
Funded domestically Funded internationally Unfunded
2009 2010 2011 2012 2013
0
200
400
600
800
&CVCCTGCUTGRQTVGFVQ9*1'UVKOCVGUQH6$CPF/&46$DWTFGPCTGRTQFWEGFD[9*1KPEQPUWNVCVKQPYKVJ
countries.
a 4CPIGUTGRTGUGPVWPEGTVCKPV[KPVGTXCNU
b Data are not collected from all Member States.
c Calculations exclude countries with missing numerators or denominators.
d Financing indicators exclude funding for general healthcare services provided outside NTPs.

#00':
Key indicators for the
world, WHO regions
and individual countries
Summary by WHO region 147
#HTKECP4GIKQP
4GIKQPQHVJG#OGTKECU
'CUVGTP/GFKVGTTCPGCP4GIKQP
'WTQRGCP4GIKQP
5QWVJ'CUV#UKC4GIKQP
9GUVGTP2CEKßE4GIKQP


57//#4;$;9*14')+10
Table A4.1 Estimates of the burden of disease caused by TB, 1990–2012 149
6CDNG# +PEKFGPEGPQVKßECVKQPCPFECUGFGVGEVKQPTCVGUCNNHQTOU¿
6CDNG# %CUGPQVKßECVKQPU¿
Table A4.4 Treatment outcomes, new smear-positive cases, 1995–2011 152
Table A4.5 Treatment outcomes, retreatment cases, 1995–2011 152
6CDNG# *+8VGUVKPICPFRTQXKUKQPQH%26#46CPF+26¿
6CDNG# 6GUVKPIHQT/&46$CPFPWODGTQHEQPßTOGFECUGUQH/&46$¿
6CDNG# 0GYUOGCTRQUKVKXGECUGPQVKßECVKQPD[CIGCPFUGZ¿

GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
Estimates of mortality, prevalence and incidence
Estimated values are shown as best estimates followed by lower and upper bounds. e lower and upper bounds are dened
as the 2.5th and 97.5th centiles of outcome distributions produced in simulations. See ANNEX 1 for further details.
Estimated numbers are shown rounded to two signicant gures. Estimated rates are shown rounded to three signicant
gures unless the value is under 100, in which case rates are shown rounded to two signicant gures.
Estimates for all years are recalculated as new information becomes available and techniques are rened, so
they may dier from those published in previous reports in this series. e main updates implemented in this
report are explained in Box 2.1 of Chapter 2. Estimates published in previous global TB control reports should no
longer be used.
Data source
Data shown in this annex are taken from the WHO global TB database on 1 October 2013. Data shown in the main part of
the report were taken from the database in July 2013. As a result, data in this annex may dier slightly from those in the
main part of the report.
Data for all years can be downloaded from www.who.int/tb/data.

149 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
SUMMARY BY WHO REGION
7$%/($(VWLPDWHVRIWKHEXUGHQRIGLVHDVHFDXVHGE\7%±
YEAR POPULATION
(MILLIONS)
NUMBER
(THOUSANDS) RATEaNUMBER
(THOUSANDS) RATEaNUMBER
(THOUSANDS) RATEa
Global 1990 5 298 1 300 (1 100–1 500) 25 (21–29) 15 000 (13 000–16 000) 274 (249–302) 7 800 (7 200–8 500) 147 (136–160)
1995 5 718 1 400 (1 100–1 600) 24 (20–28) 16 000 (14 000–17 000) 275 (251–301) 8 400 (7 900–9 000) 148 (139–157)
2000 6 102 1 400 (1 100–1 600) 22 (18–27) 16 000 (14 000–18 000) 263 (237–290) 9 000 (8 500–9 500) 148 (139–156)
2005 6 489 1 200 (1 000–1 400) 19 (16–22) 15 000 (13 000–16 000) 225 (200–250) 9 200 (8 700–9 700) 142 (134–150)
2010 6 890 1 000 (850–1 200) 15 (12–17) 13 000 (11 000–14 000) 182 (160–205) 8 800 (8 400–9 100) 128 (123–133)
2011 6 972 980 (820–1 100) 14 (12–16) 12 000 (11 000–14 000) 176 (155–198) 8 700 (8 400–9 100) 125 (120–130)
2012 7 054 940 (790–1 100) 13 (11–16) 12 000 (11 000–13 000) 169 (149–190) 8 600 (8 300–9 000) 122 (117–127)
Africa 1990 503 210 (120–340) 43 (24–67) 2 000 (1 300–3 000) 404 (254–590) 1 200 (950–1 600) 245 (189–309)
1995 577 230 (140–350) 41 (24–61) 2 300 (1 600–3 200) 405 (276–558) 1 600 (1 300–1 900) 275 (226–329)
2000 655 250 (130–400) 38 (20–61) 2 600 (1 700–3 700) 397 (257–567) 2 000 (1 700–2 400) 310 (255–370)
2005 744 240 (130–390) 32 (17–53) 2 700 (1 800–3 800) 364 (239–515) 2 300 (2 000–2 700) 310 (263–361)
2010 847 230 (160–310) 27 (19–36) 2 700 (2 100–3 300) 318 (249–395) 2 300 (2 100–2 500) 271 (249–293)
2011 870 230 (160–310) 26 (18–35) 2 700 (2 100–3 300) 310 (244–383) 2 300 (2 100–2 500) 262 (242–284)
2012 893 230 (160–310) 26 (18–35) 2 700 (2 100–3 300) 303 (239–373) 2 300 (2 100–2 500) 255 (235–275)
The Americas 1990 727 43 (35–52) 5.9 (4.8–7.1) 750 (540–990) 103 (74–136) 430 (370–490) 59 (51–68)
1995 783 37 (32–42) 4.7 (4.1–5.4) 600 (470–750) 76 (59–95) 380 (360–410) 49 (46–52)
2000 841 29 (25–33) 3.5 (3.0–4.0) 510 (390–640) 60 (47–76) 340 (320–370) 41 (38–43)
2005 892 24 (21–27) 2.7 (2.3–3.1) 440 (340–550) 49 (38–61) 310 (290–330) 34 (32–36)
2010 942 21 (18–24) 2.2 (1.9–2.5) 390 (300–490) 41 (32–52) 280 (260–300) 30 (28–32)
2011 951 19 (17–22) 2 (1.8–2.3) 400 (300–500) 42 (32–53) 280 (260–300) 30 (28–32)
2012 961 19 (16–21) 1.9 (1.7–2.2) 390 (300–490) 40 (31–51) 280 (260–300) 29 (27–31)
Eastern 1990 378 120 (57–200) 32 (15–54) 1 100 (600–1 600) 279 (159–433) 460 (360–580) 122 (94–153)
Mediterranean 1995 429 130 (67–210) 30 (16–50) 1 200 (720–1 700) 272 (168–401) 530 (470–590) 123 (109–137)
2000 480 140 (70–230) 29 (15–48) 1 200 (740–1 800) 256 (155–383) 560 (500–630) 118 (104–132)
2005 533 120 (65–190) 23 (12–36) 1 200 (740–1 700) 216 (138–312) 600 (530–670) 112 (99–126)
2010 593 100 (61–150) 17 (10–25) 1 100 (710–1 500) 184 (120–260) 650 (570–720) 109 (96–122)
2011 605 100 (62–150) 17 (10–25) 1 100 (720–1 600) 182 (119–258) 660 (580–740) 109 (97–122)
2012 617 100 (63–150) 16 (10–24) 1 100 (730–1 600) 180 (118–256) 670 (590–750) 109 (96–122)
Europe 1990 849 39 (36–43) 4.6 (4.2–5.1) 610 (500–720) 71 (59–85) 370 (350–380) 43 (41–45)
1995 863 60 (58–62) 6.9 (6.7–7.2) 1 000 (840–1 200) 120 (97–144) 560 (530–590) 65 (62–69)
2000 870 71 (69–73) 8.1 (7.9–8.4) 1 100 (890–1 400) 129 (103–159) 640 (600–680) 73 (69–78)
2005 882 66 (64–67) 7.4 (7.3–7.6) 910 (700–1 100) 103 (79–130) 570 (530–600) 64 (60–68)
2010 899 44 (43–46) 4.9 (4.8–5.1) 620 (470–790) 68 (52–87) 420 (400–450) 47 (44–50)
2011 902 40 (39–41) 4.5 (4.4–4.6) 580 (440–740) 64 (49–82) 400 (380–430) 44 (42–47)
2012 905 36 (35–36) 3.9 (3.9–4.0) 510 (380–650) 56 (42–72) 360 (340–390) 40 (38–43)
South-East 1990 1 310 570 (410–750) 43 (31–57) 6 100 (5 200–7 000) 465 (400–535) 2 900 (2 500–3 200) 218 (192–246)
Asia 1995 1 435 640 (460–840) 44 (32–58) 6 700 (5 800–7 700) 469 (404–538) 3 100 (2 800–3 400) 218 (198–239)
2000 1 560 680 (500–890) 43 (32–57) 7 000 (6 000–8 100) 449 (387–516) 3 400 (3 200–3 700) 220 (203–237)
2005 1 682 620 (480–780) 37 (29–47) 6 300 (5 300–7 400) 375 (314–442) 3 600 (3 300–3 900) 213 (197–229)
2010 1 790 500 (370–660) 28 (21–37) 5 200 (4 000–6 600) 293 (224–371) 3 500 (3 200–3 700) 194 (181–208)
2011 1 812 480 (350–620) 26 (19–34) 5 000 (3 900–6 400) 278 (213–352) 3 500 (3 200–3 700) 191 (177–204)
2012 1 833 450 (330–590) 25 (18–32) 4 800 (3 700–6 100) 264 (203–333) 3 400 (3 200–3 700) 187 (174–200)
Western 1990 1 532 320 (280–350) 21 (18–23) 4 000 (3 600–4 400) 261 (238–286) 2 500 (2 100–2 900) 161 (135–189)
Pacific 1995 1 630 260 (230–300) 16 (14–18) 3 900 (3 500–4 300) 238 (216–262) 2 300 (2 000–2 600) 138 (120–158)
2000 1 697 200 (160–230) 12 (9.6–14) 3 600 (3 200–4 000) 210 (187–234) 2 000 (1 800–2 300) 119 (106–133)
2005 1 756 150 (140–170) 8.6 (7.9–9.5) 3 100 (2 700–3 400) 174 (156–193) 1 800 (1 700–2 000) 105 (95–115)
2010 1 820 120 (110–130) 6.4 (5.9–7.1) 2 500 (2 300–2 800) 139 (124–153) 1 700 (1 500–1 800) 92 (84–100)
2011 1 833 110 (100–120) 6.1 (5.5–6.7) 2 500 (2 200–2 700) 134 (120–148) 1 600 (1 500–1 800) 90 (82–98)
2012 1 846 110 (96–120) 5.8 (5.2–6.4) 2 400 (2 100–2 600) 128 (115–142) 1 600 (1 500–1 800) 87 (80–95)
MORTALITY (EXCLUDING HIV) PREVALENCE (INCLUDING HIV) INCIDENCE (INCLUDING HIV)
a Rates are per 100 000 population.

150 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($,QFLGHQFHQRWLILFDWLRQDQGFDVHGHWHFWLRQUDWHVDOOIRUPV±
INCIDENCE (INCLUDING HIV) INCIDENCE HIV-POSITIVE NOTIFIED NEW AND RELAPSE
b
CASE DETECTION
YEAR POPULATION
(MILLIONS)
NUMBER
(THOUSANDS) RATEaNUMBER
(THOUSANDS) RATEaNUMBER RATEaPERCENT
Global 1990 5 298 7 800 (7 200–8 500) 147 (136–160) 280 (230–320) 5.2 (4.4–6.1) 3 740 222 71 48 (44–52)
1995 5 718 8 400 (7 900–9 000) 148 (139–157) 620 (560–680) 11 (9.8–12) 3 400 278 59 40 (38–43)
2000 6 102 9 000 (8 500–9 500) 148 (139–156) 1 100 (960–1 200) 17 (16–19) 3 748 455 61 42 (39–44)
2005 6 489 9 200 (8 700–9 700) 142 (134–150) 1 300 (1 200–1 400) 20 (18–21) 5 148 342 79 56 (53–59)
2010 6 890 8 800 (8 400–9 100) 128 (123–133) 1 100 (1 100–1 200) 17 (15–18) 5 792 075 84 66 (63–69)
2011 6 972 8 700 (8 400–9 100) 125 (120–130) 1 100 (1 000–1 200) 16 (15–17) 5 833 253 84 67 (64–70)
2012 7 054 8 600 (8 300–9 000) 122 (117–127) 1 100 (1 000–1 200) 15 (14–16) 5 776 838 82 67 (64–70)
Africa 1990 503 1 200 (950–1 600) 245 (189–309) 230 (190–280) 46 (38–56) 418 520 83 34 (27–44)
1995 577 1 600 (1 300–1 900) 275 (226–329) 460 (410–520) 80 (71–91) 504 377 87 32 (27–39)
2000 655 2 000 (1 700–2 400) 310 (255–370) 780 (690–880) 119 (105–134) 794 464 121 39 (33–48)
2005 744 2 300 (2 000–2 700) 310 (263–361) 960 (850–1 100) 130 (115–145) 1 188 876 160 52 (44–61)
2010 847 2 300 (2 100–2 500) 271 (249–293) 880 (800–950) 103 (94–113) 1 380 530 163 60 (56–65)
2011 870 2 300 (2 100–2 500) 262 (242–284) 850 (780–930) 98 (89–107) 1 386 327 159 61 (56–66)
2012 893 2 300 (2 100–2 500) 255 (235–275) 830 (760–910) 93 (85–102) 1 344 122 151 59 (55–64)
The Americas 1990 727 430 (370–490) 59 (51–68) 17 (14–20) 2.3 (2.0–2.7) 231 215 32 54 (47–63)
1995 783 380 (360–410) 49 (46–52) 31 (28–33) 3.9 (3.6–4.3) 258 232 33 67 (63–72)
2000 841 340 (320–370) 41 (38–43) 32 (30–35) 3.8 (3.5–4.2) 238 636 28 70 (65–75)
2005 892 310 (290–330) 34 (32–36) 34 (31–37) 3.8 (3.5–4.1) 230 124 26 75 (71–81)
2010 942 280 (260–300) 30 (28–32) 33 (30–36) 3.5 (3.1–3.8) 214 930 23 76 (71–82)
2011 951 280 (260–300) 30 (28–32) 33 (30–36) 3.5 (3.1–3.8) 221 625 23 78 (73–84)
2012 961 280 (260–300) 29 (27–31) 31 (28–34) 3.3 (3.0–3.6) 219 349 23 79 (74–85)
Eastern 1990 378 460 (360–580) 122 (94–153) 0.91 (0.77–1.1) 0.2 (0.20–0.28) 234 620 62 51 (40–66)
Mediterranean 1995 429 530 (470–590) 123 (109–137) 2.8 (2.5–3.2) 0.7 (0.59–0.74) 121 745 28 23 (21–26)
2000 480 560 (500–630) 118 (104–132) 5.9 (5.3–6.6) 1.2 (1.1–1.4) 141 748 30 25 (22–28)
2005 533 600 (530–670) 112 (99–126) 8.6 (7.6–9.6) 1.6 (1.4–1.8) 287 178 54 48 (43–54)
2010 593 650 (570–720) 109 (96–122) 11 (9.9–12) 1.9 (1.7–2.1) 412 913 70 64 (57–72)
2011 605 660 (580–740) 109 (97–122) 11 (9.7–12) 1.8 (1.6–1.9) 415 719 69 63 (56–71)
2012 617 670 (590–750) 109 (96–122) 11 (10–12) 1.8 (1.6–2.0) 420 769 68 63 (56–71)
Europe 1990 849 370 (350–380) 43 (41–45) 1.8 (1.8–1.9) 0.2 (0.21–0.23) 242 429 29 66 (63–69)
1995 863 560 (530–590) 65 (62–69) 3.4 (3.3–3.6) 0.4 (0.38–0.42) 289 874 34 51 (49–54)
2000 870 640 (600–680) 73 (69–78) 6.7 (6.2–7.1) 0.8 (0.71–0.82) 373 094 43 59 (55–62)
2005 882 570 (530–600) 64 (60–68) 17 (15–18) 1.9 (1.7–2.0) 368 624 42 65 (61–70)
2010 899 420 (400–450) 47 (44–50) 20 (18–21) 2.2 (2.0–2.4) 328 254 37 77 (73–83)
2011 902 400 (380–430) 44 (42–47) 19 (18–21) 2.1 (2.0–2.3) 312 588 35 78 (73–83)
2012 905 360 (340–390) 40 (38–43) 19 (17–21) 2.1 (1.9–2.3) 286 765 32 79 (74–84)
South-East 1990 1 310 2 900 (2 500–3 200) 218 (192–246) 22 (19–26) 1.7 (1.5–2.0) 1 719 365 131 60 (53–68)
Asia 1995 1 435 3 100 (2 800–3 400) 218 (198–239) 110 (96–120) 7.5 (6.7–8.4) 1 401 096 98 45 (41–49)
2000 1 560 3 400 (3 200–3 700) 220 (203–237) 210 (190–230) 13 (12–15) 1 414 228 91 41 (38–45)
2005 1 682 3 600 (3 300–3 900) 213 (197–229) 220 (200–240) 13 (12–14) 1 789 388 106 50 (46–54)
2010 1 790 3 500 (3 200–3 700) 194 (181–208) 180 (160–190) 9.9 (9.1–11) 2 124 237 119 61 (57–66)
2011 1 812 3 500 (3 200–3 700) 191 (177–204) 170 (160–180) 9.4 (8.7–10) 2 142 573 118 62 (58–67)
2012 1 833 3 400 (3 200–3 700) 187 (174–200) 170 (160–180) 9.2 (8.5–10) 2 130 120 116 62 (58–67)
Western 1990 1 532 2 500 (2 100–2 900) 161 (135–189) 1.8 (1.5–2.1) 0.1 (0.10–0.14) 894 073 58 36 (31–43)
Pacific 1995 1 630 2 300 (2 000–2 600) 138 (120–158) 8.6 (7.4–9.9) 0.5 (0.45–0.61) 824 954 51 37 (32–42)
2000 1 697 2 000 (1 800–2 300) 119 (106–133) 17 (15–19) 1 (0.90–1.1) 786 285 46 39 (35–44)
2005 1 756 1 800 (1 700–2 000) 105 (95–115) 24 (21–27) 1.4 (1.2–1.5) 1 284 152 73 70 (63–77)
2010 1 820 1 700 (1 500–1 800) 92 (84–100) 24 (22–27) 1.3 (1.2–1.5) 1 331 211 73 80 (73–87)
2011 1 833 1 600 (1 500–1 800) 90 (82–98) 24 (22–27) 1.3 (1.2–1.5) 1 354 421 74 83 (76–90)
2012 1 846 1 600 (1 500–1 800) 87 (80–95) 24 (21–27) 1.3 (1.1–1.5) 1 375 713 75 85 (78–93)
a Rates are per 100 000 population.
b NOTIFIED NEW AND RELAPSE includes cases for which the treatment history is unknown.

151 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
SUMMARY BY WHO REGION
7$%/($&DVHQRWLILFDWLRQV±
NEW CASES
YEAR SMEAR-
POSITIVE
SMEAR-NEGATIVE/
UNKNOWN
EXTRA-
PULMONARY OTHER RELAPSE RE-TREAT EXCL.
RELAPSE
TOTAL
RETREAT
HISTORY
UNKNOWN
Global 1990 3 740 222 30 046 22 393 4 237 0 734 49 783 29 57
1995 3 400 278 1 175 290 1 811 850 262 728 5 59 240 0 59 240 44 39
2000 3 748 455 1 541 607 1 615 263 399 677 37 115 334 236 107 351 441 229 49
2005 5 148 342 2 413 708 1 722 281 686 525 8 111 259 937 406 355 666 292 18 172 58
2010 5 792 075 2 655 557 2 002 463 806 373 12 870 285 966 418 071 704 037 28 846 57
2011 5 833 253 2 630 564 2 037 926 817 668 12 164 284 815 413 363 698 178 50 116 56
• 71 82 • 2012 5 776 838 2 563 744 2 084 246 813 960 9 689 288 119 393 437 681 556 17 080 55
Africa 1990 418 520 24 064 6 137 2 067 0 554 49 603 0 80
1995 504 377 212 910 191 477 72 689 0 15 133 0 15 133 0 53
2000 794 464 368 750 222 230 141 255 0 19 173 68 118 87 291 0 62
2005 1 188 876 550 004 364 785 208 979 2 941 60 092 66 449 126 541 2 075 60
2010 1 380 530 601 149 477 516 247 020 561 53 967 94 506 148 473 317 56
2011 1 386 327 606 085 467 022 240 839 1 073 52 357 74 545 126 902 18 951 56
• 83 151 • 2012 1 344 122 600 355 446 213 234 707 977 60 085 67 960 128 045 1 785 57
The Americas 1990 231 215 1 542 516 723 0 180 0 180 29 75
1995 258 232 138 932 72 312 32 991 5 1 723 0 1 723 44 66
2000 238 636 131 294 60 392 32 037 37 10 834 14 344 25 178 56 68
2005 230 124 124 840 56 056 33 285 3 685 10 152 12 481 22 633 2 106 69
2010 214 930 116 994 52 265 32 240 2 133 10 413 12 133 22 546 885 69
2011 221 625 122 010 51 165 34 048 1 502 10 087 11 856 21 943 2 813 70
• 32 23 • 2012 219 349 122 730 50 338 34 496 1 636 10 100 13 879 23 979 49 71
Eastern 1990 234 620 1 587 12 394 754 0 0 0 0 0 11
Mediterranean 1995 121 745 46 851 51 823 33 382 0 2 407 0 2 407 0 47
2000 141 748 60 959 34 289 40 754 0 5 568 0 5 568 0 64
2005 287 178 113 765 102 274 64 612 12 6 495 5 334 11 829 20 53
2010 412 913 168 627 137 301 92 070 633 11 203 8 713 19 916 3 079 55
2011 415 719 170 748 135 388 93 605 623 11 223 10 102 21 325 4 132 56
• 62 68 • 2012 420 769 173 963 143 869 90 943 702 11 208 10 020 21 228 84 55
Europe 1990 242 429 0 0 0 0 0 0 0 0
1995 289 874 104 444 146 592 29 866 0 7 927 0 7 927 0 42
2000 373 094 94 442 208 147 35 081 0 21 607 19 127 40 734 173 31
2005 368 624 96 121 157 237 49 747 0 22 248 64 831 87 079 3 663 38
2010 328 254 91 324 145 140 40 951 8 008 24 304 60 736 85 040 18 527 39
2011 312 588 85 551 136 456 46 012 3 381 24 628 67 986 92 614 16 560 39
• 29 32 • 2012 286 765 80 453 129 293 43 134 83 25 133 65 121 90 254 8 669 38
South-East 1990 1 719 365 2 769 3 241 656 0 0 0 0 0 46
Asia 1995 1 401 096 357 882 939 945 76 865 0 5 546 0 5 546 0 28
2000 1 414 228 510 053 741 471 120 708 0 27 095 80 444 107 539 0 41
2005 1 789 388 857 371 594 185 242 332 1 439 93 859 158 215 252 074 202 59
2010 2 124 237 1 047 013 615 463 328 421 1 508 130 714 208 542 339 256 1 118 63
2011 2 142 573 1 067 367 598 800 333 993 2 878 135 650 215 554 351 204 3 885 64
• 131 116 • 2012 2 130 120 1 065 852 586 455 338 303 3 004 131 245 201 335 332 580 5 261 65
Western 1990 894 073 84 105 37 0 0 0 0 0 44
Pacific 1995 824 954 314 271 409 701 16 935 0 26 504 0 26 504 0 43
2000 786 285 376 109 348 734 29 842 0 31 057 54 074 85 131 0 52
2005 1 284 152 671 607 447 744 87 570 34 67 091 99 045 166 136 10 106 60
2010 1 331 211 630 450 574 778 65 671 27 55 365 33 441 88 806 4 920 52
2011 1 354 421 578 803 649 095 69 171 2 707 50 870 33 320 84 190 3 775 47
• 58 75 • 2012 1 375 713 520 391 728 078 72 377 3 287 50 348 35 122 85 470 1 232 42
NEW AND RELAPSE
NOTIFICATION RATE
a
1990–2012
NEW AND
RELAPSE
b
% SMEAR-
POS AMONG
NEW PULM
a Rates are per 100 000 population.
b NEW AND RELAPSE includes cases for which the treatment history is unknown.

152 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($7UHDWPHQWRXWFRPHVQHZVPHDUSRVLWLYHFDVHV±
% OF COHORT
TREATMENT SUCCESS (%)
a
1995–2011 YEAR NUMBER
NOTIFIED
SIZE OF
COHORT
COHORT AS
% NOTIFIED CURED COMPLETED DIED FAILED DEFAULTED NOT
EVALUATED
Global 1995 1 175 290 1 000 581 85 40 17 3 1 5 34
2000 1 541 607 1 452 991 94 60 941719
2005 2 413 708 2 396 387 99 7774254
2009 2 662 588 2 664 704 100 8074244
2010 2 655 557 2 661 653 100 80 7 4 2 4 3
• 57 87 • 2011 2 630 564 2 610 821 99 80 7 4244
Africa 1995 212 910 177 567 83 46 14 6 2 12 20
2000 368 750 364 804 99 59 12 7 1 11 10
2005 550 004 563 750 102 62 13 7 1 9 7
2009 607 254 605 932 100 70 10 5 1 6 7
2010 601 149 598 985 100 72 9 5 1 6 6
• 60 82 • 2011 606 085 578 920 96 72 10 5166
The Americas 1995 138 932 128 531 93 37 14 3 1 6 39
2000 131 294 110 642 84 60 17 5 1 8 11
2005 124 840 118 840 95 55 24 5 1 7 9
2009 110 614 122 534 111 53 23 5 1 8 11
2010 116 994 126 450 108 53 22 5 1 8 11
• 50 78 • 2011 122 010 126 859 104 54 23 5279
Eastern 1995 46 851 46 318 99 60 19 2 3 13 4
Mediterranean 2000 60 959 63 749 105 69 12 4 2 8 6
2005 113 765 113 742 100 72 11 3 1 8 5
2009 168 013 167 317 100 74 14 3153
2010 168 627 169 872 101 74 14 2 1 5 3
• 79 88 • 2011 170 748 170 903 100 74 14 2 1 5 4
Europe 1995 104 444 33 823 32 58 10 6 6 4 16
2000 94 442 41 480 44 47 28 5 6 6 7
2005 96 121 81 410 85 59 13 8775
2009 100 493 105 441 105 56 13 8 12 6 5
2010 91 324 98 689 108 54 13 8 12 6 7
• 67 66 • 2011 85 551 106 626 125 51 15 8 8 6 12
South-East 1995 357 882 318 410 89 9 23 1 0 2 64
Asia 2000 510 053 512 286 100 44 6 2 1 7 40
2005 857 371 855 962 100 83 4 4261
2009 1 028 656 1 022 380 99 8534251
2010 1 047 013 1 045 179 100 8544251
• 33 89 • 2011 1 067 367 1 064 879 100 8544251
Western 1995 314 271 295 932 94 67 13 2 1 4 13
Pacific 2000 376 109 360 030 96 85 5 2124
2005 671 607 662 683 99 89 32113
2009 647 558 641 100 99 9032113
2010 630 450 622 478 99 9032113
• 80 94 • 2011 578 803 562 634 97 91 3 2 1 1 2
7$%/($7UHDWPHQWRXWFRPHVUHWUHDWPHQWFDVHV±
% OF COHORT
TREATMENT SUCCESS (%)
a
1995–2011 YEAR NUMBER
NOTIFIED
SIZE OF
COHORT
COHORT AS
% NOTIFIED CURED COMPLETED DIED FAILED DEFAULTED NOT
EVALUATED
Global 1995 59 240 71 395 121 8243334
2000 351 441 188 509 54 60 10 6 4 11 10
2005 666 292 546 182 82 51 19 7 4 12 6
2009 673 854 594 019 88 49 23 7 6 10 5
2010 704 037 613 895 87 47 22 7 5 10 10
• 86 72 • 2011 698 178 601 904 86 48 24 7 5 10 7
Africa 1995 15 133 5 756 38 57 12 9 3 12 6
2000 87 291 44 147 51 47 11 9 3 16 14
2005 126 541 114 838 91 35 27 11 3 13 12
2009 144 320 94 342 65 50 20 9 3 9 10
2010 148 473 113 405 76 41 13 6 3 7 31
• 69 68 • 2011 126 902 85 278 67 53 15 7 3 9 12
The Americas 1995 1 723 1 104 64 61 11 6 4 11 8
2000 25 178 15 302 61 478531225
2005 22 633 18 603 82 38 16 6 2 15 21
2009 21 492 19 158 89 29 22 8 3 19 21
2010 22 546 17 499 78 26 23 7 2 20 21
• 72 51 • 2011 21 943 20 228 92 27 24 8 3 20 18
Eastern 1995 2 407 1 860 77 61 14 3 4 12 5
Mediterranean 2000 5 568 4 217 76 51 11 6 7 15 11
2005 11 829 12 860 109 60 15 5 4 10 6
2009 17 964 16 332 91 56 21 4 3 10 6
2010 19 916 18 326 92 54 21 4 3 10 8
• 75 74 • 2011 21 325 22 191 104 52 22 4 3 10 8
Europe 1995 7 927 480 6 20 20 11 8 32 8
2000 40 734 10 739 26 39 19 9 14 11 8
2005 87 079 39 497 45 32 18 11 13 14 10
2009 67 190 58 966 88 27 22 11 22 11 7
2010 85 040 58 698 69 25 25 11 16 10 13
• 40 47 • 2011 92 614 58 831 64 24 23 10 15 10 18
South-East 1995 5 546 3 271 59 62645158
Asia 2000 107 539 59 337 55 57 14 6 5 15 3
2005 252 074 254 378 101 49 22 7 5 15 2
2009 331 424 332 286 100 48 27 7 4 12 2
2010 339 256 338 748 100 47 28 7 4 12 2
• 68 75 • 2011 351 204 350 251 100 45 30 7 4 11 3
Western 1995 26 504 58 924 222 88 2 3 3 1 3
Pacific 2000 85 131 54 767 64 83 3 2 2 1 9
2005 166 136 106 006 64 8163326
2009 91 464 72 935 80 7973227
2010 88 806 67 219 76 7973227
• 90 86 • 2011 84 190 65 125 77 80 6 3 3 2 6
a TREATMENT SUCCESS = percent cured + percent completed then rounded to the nearest digit.

153 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
7$%/($+,9WHVWLQJDQGSURYLVLRQRI&37$57DQG,37–
% OF TB PATIENTS WITH
KNOWN HIV STATUS
2005–2012
YEAR
% OF TB
PATIENTS WITH
KNOWN HIV
STATUS
NUMBER OF TB
PATIENTS WITH
KNOWN HIV
STATUS
PATIENTS
NOTIFIED
(NEW AND
RETREAT)
NUMBER OF
HIV-POSITIVE
TB PATIENTS
% OF TESTED
TB PATIENTS
HIV-POSITIVE
% OF HIV-
POSITIVE TB
PATIENTS ON
CPT
% OF HIV-
POSITIVE TB
PATIENTS ON
ART
NUMBER OF
HIV-POSITIVE
PEOPLE
PROVIDED IPT
Global 2005 8.3 463 027 5 554 697 103 683 22 76 35 25 938
2010 34 2 080 846 6 210 146 493 186 24 81 46 204 802
2011 40 2 526 072 6 246 616 569 074 23 82 49 446 598
• 8 46 • 2012 46 2 808 221 6 170 275 549 769 20 79 57 518 670
Africa 2005 11 140 713 1 255 325 73 332 52 78 29 22 211
2010 60 888 765 1 475 036 394 332 44 81 44 182 524
2011 69 1 013 342 1 460 872 465 647 46 82 47 438 121
• 11 74 • 2012 74 1 040 262 1 412 082 443 558 43 79 55 473 214
The Americas 2005 35 84 032 242 605 14 232 17 10 81 3 727
2010 53 121 421 227 063 19 615 16 50 63 12 906
2011 56 129 613 233 481 20 497 16 41 69 1 705
• 35 57 • 2012 57 132 943 233 228 20 798 16 63 77 18 710
Eastern 2005 0.88 2 582 292 512 330 13 18 16 0
Mediterranean 2010 11 44 596 421 626 1 360 3 50 44 253
2011 11 48 271 425 821 1 738 3.6 60 31 52
• 1 14 • 2012 14 58 498 430 789 2 036 3.5 69 49 243
Europe 2005 40 171 248 433 455 6 543 2.8 25 16 0
2010 55 212 727 388 990 12 858 5.9 58 61 6 575
2011 57 215 256 380 574 11 790 5.3 63 58 4 565
• 40 60 • 2012 60 212 880 351 886 13 103 6.2 71 62 17 938
South-East 2005 1.6 31 847 1 947 603 7 025 22 50 31 0
Asia 2010 23 546 350 2 332 779 52 519 9.6 86 56 581
2011 33 767 813 2 358 127 55 608 7.2 88 58 368
• 2 39 • 2012 39 909 026 2 331 455 56 093 6.2 89 61 8
Western 2005 2.4 32 605 1 383 197 2 221 6.8 31 33 0
Pacific 2010 20 266 987 1 364 652 12 502 4.6 55 41 1 963
2011 25 351 777 1 387 741 13 794 3.9 71 48 1 787
• 2 32 • 2012 32 454 612 1 410 835 14 181 3.1 79 56 8 557
7$%/($7HVWLQJIRU0'57%DQGQXPEHURIFRQILUPHGFDVHVRI0'57%±
NEW PULMONARY CASES PREVIOUSLY TREATED CASES
YEAR
ESTIMATED CASES
OF MDR-TB AMONG
NOTIFIED
NUMBER OF
BACT+VE
b
TESTED FOR
MDR-TB
% OF
BACT+VE
b
TESTED FOR
MDR-TB
ESTIMATED CASES
OF MDR-TB AMONG
NOTIFIED
NUMBER OF
NOTIFIED
TESTED FOR
MDR-TB
% OF NOT IFIED
TESTED FOR
MDR-TB
Global 2005 11988 72870 2.9 24002 3.6
2010 54887 118835 4 47315 6.7
2011 61907 133064 4.6 48124 6.9
2012 85085 310 000 (230 000–380 000) 170 000 (98 000–240 000) 153626 5.7 140 000 (91 000–190 000) 60589 8.9
Africa 2005 2445 1826 0.32 3922 3.1
2010 9340 2732 0.36 4294 2.9
2011 12384 1311 0.19 3707 2.9
2012 18146 38 000 (14 000–62 000) 24 000 (2 100–46 000) 2565 0.39 14 000 (5 600–22 000) 4118 3.2
The Americas 2005 4427 14568 11 11003 49
2010 2661 11309 8.6 4234 19
2011 3474 13334 10 4234 19
2012 2967 7 100 (4 600–9 600) 3 800 (2 400–5 200) 29869 23 3 200 (1 100–5 300) 5565 23
Eastern 2005 350 1442 1.3 94 0.79
Mediterranean 2010 873 2397 1.4 1257 6.3
2011 841 2264 1.2 1466 6.9
2012 2249 18 000 (0–42 000) 11 000 (320–36 000) 1990 1.1 6 900 (2 400–11 000) 1617 7.6
Europe 2005 4347 34527 27 7024 8.1
2010 33776 89005 68 34212 40
2011 34199 89438 67 31646 34
2012 36772 74 000 (60 000–88 000) 33 000 (20 000–46 000) 92580 76 41 000 (35 000–46 000) 38268 42
South-East 2005 68 661 <0.1 420 0.17
Asia 2010 3942 1073 0.1 1264 0.37
2011 6615 1204 0.1 1935 0.55
2012 19202 90 000 (71 000–110 000) 36 000 (26 000–46 000) 1352 0.13 54 000 (37 000–70 000) 2292 0.69
Western 2005 351 19846 2.9 1539 0.93
Pacific 2010 4295 12319 1.7 2054 2.3
2011 4394 25513 4.2 5136 6.1
2012 5749 78 000 (60 000–95 000) 59 000 (41 000–76 000) 25270 4.6 19 000 (15 000–23 000) 8729 10
TOTAL
CONFIRMED
CASES OF
MDR-TB
a
ESTIMATED CASES
OF MDR-TB AMONG
NOTIFIED
a TOTAL CONFIRMED CASES OF MDR-TB includes cases with unknown previous treatment history (i.e. not included under NEW CASES or PREVIOUSLY TREATED
CASES).
b BACT+VE = bacteriologically positive cases.
SUMMARY BY WHO REGION

154 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($1HZVPHDUSRVLWLYHFDVHQRWLILFDWLRQE\DJHDQGVH[±
MALE FEMALE
YEAR 0–14 15–24 25–34 35–44 45–54 55–64 65+
UN-
KNOWN
0–14 15–24 25–34 35–44 45–54 55–64 65+
UN-
KNOWN
MALE:FEMALE
RATIO
Global 1995 7 491 48 816 76 799 65 678 49 514 41 756 34 776 0 7 730 41 378 50 102 32 741 22 688 17 816 16 686 0 1.7
2000 12 387 115 250 172 896 156 274 121 277 82 844 75 156 0 14 749 94 641 110 306 74 705 49 823 33 696 33 829 0 1.8
2005 18 415 242 356 329 720 312 526 261 233 184 836 166 858 42 26 178 199 700 220 530 153 503 106 029 72 022 65 717 15 1.8
2010 20 239 268 884 345 937 336 981 298 715 227 530 186 815 7 502 28 825 210 729 225 986 163 260 118 565 86 264 75 368 2 601 1.9
2011 19 701 265 503 349 803 333 792 300 666 229 756 183 782 579 28 133 209 821 224 552 162 884 119 644 87 668 74 004 313 1.9
2012 17 046 246 030 330 650 321 408 290 214 225 684 177 736 268 24 834 197 407 210 454 153 967 115 659 86 968 74 189 172 1.9
Africa 1995 2 910 16 754 28 172 20 240 12 017 7 008 4 104 0 3 167 15 873 19 005 11 339 6 643 3 655 1 734 0 1.5
2000 3 625 29 522 47 654 34 435 17 923 8 970 5 751 0 4 315 29 530 35 386 20 037 9 402 4 581 2 578 0 1.4
2005 7 635 54 066 94 388 71 072 40 974 18 931 12 143 0 10 023 57 115 75 056 43 213 22 855 11 047 7 163 0 1.3
2010 8 393 57 146 98 636 78 660 48 543 24 094 14 478 17 10 287 55 537 76 051 47 070 26 299 13 522 8 685 9 1.4
2011 8 551 59 072 105 549 81 247 49 967 24 393 14 732 516 10 632 57 027 76 968 47 873 26 401 13 543 8 843 301 1.4
2012 6 032 51 158 96 915 79 312 46 870 23 665 14 186 31 8 003 48 828 67 255 43 481 23 378 12 683 8 642 37 1.5
The Americas 1995 437 2 888 3 443 3 157 2 448 1 866 2 251 0 431 2 293 2 434 1 654 1 109 912 1 311 0 1.6
2000 3 464 18 564 21 869 19 787 15 138 9 899 9 717 0 3 535 15 305 14 961 10 323 7 294 5 038 5 894 0 1.6
2005 1 520 16 410 16 671 14 369 12 340 7 801 7 951 0 1 718 12 405 11 563 7 891 5 933 3 788 4 751 0 1.6
2010 1 050 11 461 14 267 11 332 10 627 7 433 7 084 59 1 137 8 405 8 496 5 818 4 880 3 467 4 068 22 1.7
2011 1 103 12 436 15 023 11 704 11 234 7 709 7 198 56 1 241 8 517 8 766 5 875 4 973 3 690 4 243 9 1.8
2012 935 12 125 14 784 11 278 10 716 7 596 6 989 67 1 044 8 615 8 561 5 710 5 023 3 760 4 157 30 1.7
Eastern 1995 2 010 6 796 8 673 5 475 3 731 3 732 2 604 0 1 881 5 035 5 797 3 679 3 047 2 742 1 902 0 1.4
Mediterranean 2000 1 339 8 135 9 002 6 525 4 409 2 990 3 036 0 1 711 6 710 5 780 3 922 2 851 2 039 1 893 0 1.4
2005 1 546 13 558 14 609 10 798 8 729 6 581 5 595 0 2 766 13 529 12 098 8 386 6 245 4 383 3 399 0 1.2
2010 2 316 19 526 19 993 14 908 13 086 10 596 9 521 0 4 377 21 108 17 151 12 183 9 776 7 532 7 032 0 1.1
2011 1 924 19 630 20 303 14 984 13 857 11 049 9 871 0 3 839 21 322 17 214 12 380 10 060 7 770 6 432 0 1.2
2012 1 999 20 119 20 411 15 178 14 006 11 333 10 059 160 3 642 22 258 17 341 12 564 10 187 8 082 6 784 20 1.2
Euro
p
e 1995 553 3 588 7 046 10 157 7 625 5 716 4 842 0 548 2 906 3 636 2 594 1 549 1 560 3 289 0 2.5
2000 201 4 636 8 322 9 862 8 065 4 313 3 321 0 290 3 506 4 405 2 945 1 798 1 243 2 490 0 2.3
2005 299 6 170 9 151 9 150 8 704 4 443 4 089 42 422 4 667 5 101 3 161 2 242 1 336 3 176 15 2.1
2010 156 7 319 13 259 12 447 12 270 6 916 4 125 7 423 301 4 958 6 559 4 218 3 051 2 033 3 398 2 567 2.4
2011 164 6 536 13 704 13 498 12 966 7 569 4 329 7 257 4 734 6 767 4 507 3 195 2 292 3 693 3 2.3
2012 138 5 997 13 038 13 394 12 301 7 624 4 113 5 224 4 258 6 336 4 387 2 986 2 125 3 528 3 2.4
South-East 1995 165 3 179 6 467 6 508 5 241 4 682 3 523 0 250 2 187 2 834 2 404 2 003 1 866 1 480 0 2.3
Asia 2000 2 453 30 093 45 720 47 107 38 058 25 080 16 208 0 3 222 21 518 25 653 19 241 13 019 8 142 5 468 0 2.1
2005 5 064 94 638 120 560 122 256 107 228 74 084 45 533 0 8 591 71 923 76 779 54 000 37 709 24 289 12 975 0 2.0
2010 6 737 114 806 136 683 142 080 132 411 101 728 67 131 0 10 923 84 006 84 704 63 272 48 470 34 052 20 004 0 2.0
2011 6 490 114 254 136 142 141 636 135 592 106 420 72 264 0 10 654 85 376 84 383 64 868 50 920 36 755 21 593 0 2.0
2012 6 581 111 501 133 040 140 542 136 569 108 866 72 554 0 10 535 85 726 82 947 64 170 52 118 38 516 22 187 0 2.0
Western 1995 1 416 15 611 22 998 20 141 18 452 18 752 17 452 0 1 453 13 084 16 396 11 071 8 337 7 081 6 970 0 1.8
Pacific 2000 1 305 24 300 40 329 38 558 37 684 31 592 37 123 0 1 676 18 072 24 121 18 237 15 459 12 653 15 506 0 2.0
2005 2 351 57 514 74 341 84 881 83 258 72 996 91 547 0 2 658 40 061 39 933 36 852 31 045 27 179 34 253 0 2.2
2010 1 587 58 626 63 099 77 554 81 778 76 763 84 476 3 1 800 36 715 33 025 30 699 26 089 25 658 32 181 3 2.4
2011 1 469 53 575 59 082 70 723 77 050 72 616 75 388 0 1 510 32 845 30 454 27 381 24 095 23 618 29 200 0 2.4
2012 1 361 45 130 52 462 61 704 69 752 66 600 69 835 5 1 386 27 722 28 014 23 655 21 967 21 802 28 891 82 2.4

#(4+%#04')+10
Table A4.1 Estimates of the burden of disease caused by TB, 1990–2012 157
6CDNG# +PEKFGPEGPQVKßECVKQPCPFECUGFGVGEVKQPTCVGUCNNHQTOU¿
6CDNG# %CUGPQVKßECVKQPU¿
Table A4.4 Treatment outcomes, new smear-positive cases, 1995–2011 166
Table A4.5 Treatment outcomes, retreatment cases, 1995–2011 169
6CDNG# *+8VGUVKPICPFRTQXKUKQPQH%26#46CPF+26¿
6CDNG# 6GUVKPIHQT/&46$CPFPWODGTQHEQPßTOGFECUGUQH/&46$¿
6CDNG# 0GYUOGCTRQUKVKXGECUGPQVKßECVKQPD[CIGCPFUGZ¿
Table A4.9 Laboratories, NTP services, drug management and infection control, 2012 179
6CDNG# /GCUWTGFRGTEGPVCIGQH6$ECUGUYKVJ/&46$OQUVTGEGPV[GCTCXCKNCDNG

156 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
Estimates of mortality, prevalence and incidence
Estimated values are shown as best estimates followed by lower and upper bounds. e lower and upper bounds are dened
as the 2.5th and 97.5th centiles of outcome distributions produced in simulations. See ANNEX 1 for further details.
Estimated numbers are shown rounded to two signicant gures. Estimated rates are shown rounded to three signicant
gures unless the value is under 100, in which case rates are shown rounded to two signicant gures.
Estimates for all years are recalculated as new information becomes available and techniques are rened, so
they may dier from those published in previous reports in this series. e main updates implemented in this
report are explained in Box 2.1 of Chapter 2. Estimates published in previous global TB control reports should no
longer be used.
Data source
Data shown in this annex are taken from the WHO global TB database on 1 October 2013. Data shown in the main part of
the report were taken from the database in July 2013. As a result, data in this annex may dier slightly from those in the
main part of the report.
Data for all years can be downloaded from www.who.int/tb/data.

157 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
AFRICAN REGION
7$%/($(VWLPDWHVRIWKHEXUGHQRIGLVHDVHFDXVHGE\7%±
YEAR POPULATION
(MILLIONS)
NUMBER
(THOUSANDS) RATE
a
NUMBER
(THOUSANDS) RATE
a
NUMBER
(THOUSANDS) RATE
a
Algeria 1990 26 2.8 (0.970–5.5) 11 (3.7–21) 29 (13–53) 112 (49–202) 17 (13–22) 66 (48–86)
1995 29 2.9 (0.980–5.9) 9.9 (3.4–20) 32 (13–59) 110 (46–203) 20 (15–26) 68 (50–89)
2000 32 4.4 (1.5–8.8) 14 (4.9–28) 47 (20–85) 148 (64–267) 28 (20–36) 87 (64–114)
2005 34 5 (1.7–9.9) 15 (5.1–29) 53 (23–95) 156 (67–281) 31 (23–41) 93 (68–121)
2010 37 5.3 (1.8–11) 14 (5.0–28) 56 (24–100) 151 (65–273) 33 (24–44) 90 (66–118)
2011 38 5.4 (1.9–11) 14 (5.0–29) 57 (25–100) 152 (65–274) 34 (25–44) 90 (66–117)
2012 38 5.6 (1.9–11) 15 (5.1–29) 59 (25–110) 152 (66–274) 34 (25–45) 89 (65–117)
Angola 1990 10 4 (1.0–8.9) 39 (9.9–87) 39 (14–76) 378 (137–738) 21 (13–31) 205 (127–303)
1995 12 6.1 (2.3–12) 50 (19–96) 55 (27–93) 458 (225–772) 27 (22–33) 226 (185–272)
2000 14 5.8 (2.4–11) 42 (17–77) 59 (29–99) 421 (207–709) 35 (28–42) 250 (204–300)
2005 17 4.4 (1.7–8.1) 26 (11–49) 57 (22–110) 347 (132–663) 46 (37–54) 276 (227–329)
2010 20 6.6 (3.0–12) 34 (16–60) 80 (35–140) 411 (181–731) 59 (50–69) 304 (256–355)
2011 20 7.6 (3.5–13) 38 (17–66) 90 (42–160) 447 (209–772) 62 (53–73) 310 (261–362)
2012 21 8.7 (3.9–15) 42 (19–73) 99 (48–170) 474 (230–804) 66 (55–77) 316 (266–369)
Benin 1990 5 0.97 (0.390–1.8) 19 (7.9–36) 9.7 (4.7–17) 195 (93–333) 6.4 (5.3–7.6) 128 (106–152)
1995 6 0.95 (0.390–1.7) 16 (6.6–29) 8.9 (4.3–15) 149 (72–253) 6 (4.9–7.1) 100 (82–118)
2000 7 0.95 (0.400–1.7) 14 (5.8–25) 9.3 (4.6–16) 134 (66–225) 6 (4.9–7.1) 86 (71–102)
2005 8 0.87 (0.380–1.6) 11 (4.7–19) 9.2 (4.6–15) 113 (56–189) 6 (5.0–7.2) 74 (61–88)
2010 10 0.88 (0.390–1.6) 9.3 (4.1–16) 10 (5.0–17) 107 (53–179) 6.5 (5.4–7.8) 69 (57–82)
2011 10 0.91 (0.400–1.6) 9.3 (4.1–17) 11 (5.3–18) 109 (54–182) 6.8 (5.6–8.1) 70 (58–83)
2012 10 0.94 (0.420–1.7) 9.4 (4.2–17) 11 (5.6–18) 110 (55–184) 7 (5.8–8.3) 70 (58–83)
Botswana 1990 1 1.3 (0.095–4.2) 97 (6.9–302) 13 (1.9–33) 915 (135–2 410) 7.4 (2.9–14) 533 (212–997)
1995 2 1.3 (0.076–4.4) 85 (4.8–276) 15 (2.6–37) 925 (166–2 310) 14 (8.8–19) 855 (553–1 220)
2000 2 0.88 (0.046–2.9) 50 (2.6–165) 13 (3.4–28) 720 (194–1 580) 16 (13–20) 918 (736–1 120)
2005 2 0.66 (0.190–1.4) 35 (10–76) 11 (5.2–19) 579 (276–991) 14 (13–15) 733 (667–802)
2010 2 0.48 (0.120–1.1) 24 (6.0–56) 8.1 (3.6–14) 411 (185–727) 9.9 (8.8–11) 503 (449–560)
2011 2 0.45 (0.120–1.0) 23 (5.9–51) 7.5 (3.4–13) 380 (172–668) 9 (8.1–10) 455 (406–507)
2012 2 0.42 (0.110–0.920) 21 (5.5–46) 6.9 (3.1–12) 343 (157–600) 8.2 (7.3–9.1) 408 (364–454)
Burkina Faso 1990 9 1.2 (0.470–2.3) 14 (5.3–26) 12 (5.4–20) 132 (61–230) 7.6 (6.5–8.9) 87 (73–101)
1995 10 1.4 (0.540–2.6) 14 (5.3–26) 12 (5.7–21) 121 (57–209) 8.3 (7.0–9.6) 82 (70–95)
2000 12 1.5 (0.580–2.8) 13 (5.0–24) 13 (6.0–22) 108 (52–186) 8.2 (7.0–9.6) 71 (60–83)
2005 13 1.5 (0.610–2.7) 11 (4.5–20) 13 (6.6–22) 97 (49–161) 8.4 (7.1–9.8) 62 (53–73)
2010 16 1.4 (0.630–2.5) 9.2 (4.0–16) 14 (7.1–23) 89 (46–147) 9 (7.6–10) 58 (49–67)
2011 16 1.4 (0.620–2.5) 8.8 (3.9–16) 14 (7.2–23) 88 (45–145) 9.1 (7.7–11) 57 (48–66)
2012 16 1.4 (0.600–2.5) 8.5 (3.7–15) 14 (6.9–22) 82 (42–136) 9 (7.6–10) 54 (46–63)
Burundi 1990 6 1.3 (0.570–2.3) 23 (10–40) 15 (7.8–24) 263 (140–425) 9.1 (8.0–10) 162 (143–183)
1995 6 3 (1.2–5.6) 48 (19–90) 32 (16–53) 510 (251–858) 20 (18–23) 321 (283–362)
2000 7 2.6 (1.1–4.9) 40 (16–73) 27 (14–45) 408 (207–675) 19 (17–22) 288 (254–325)
2005 8 2.2 (0.960–4.0) 28 (12–51) 22 (12–37) 289 (151–471) 15 (14–17) 198 (174–223)
2010 9 1.8 (0.840–3.2) 20 (9.1–35) 20 (11–33) 219 (116–354) 13 (12–15) 144 (127–163)
2011 10 1.8 (0.840–3.2) 19 (8.8–33) 20 (11–33) 214 (114–345) 13 (12–15) 139 (122–157)
2012 10 1.8 (0.790–3.2) 18 (8.0–32) 20 (10–32) 199 (106–322) 13 (11–14) 130 (114–147)
Cameroon 1990 12 2.3 (0.980–4.1) 19 (8.1–34) 24 (12–39) 195 (98–325) 14 (11–16) 112 (92–133)
1995 14 5.8 (2.2–11) 42 (16–80) 56 (26–98) 404 (186–704) 29 (24–34) 206 (170–246)
2000 16 8.1 (3.1–16) 51 (19–98) 80 (36–140) 504 (227–889) 49 (41–59) 310 (255–369)
2005 18 7.3 (3.0–14) 40 (16–75) 78 (36–140) 432 (201–750) 57 (47–68) 312 (258–372)
2010 21 6.6 (2.8–12) 32 (14–58) 76 (36–130) 366 (174–629) 56 (47–67) 274 (226–327)
2011 21 6.3 (2.7–11) 30 (13–54) 68 (32–120) 320 (152–549) 51 (42–61) 243 (200–290)
2012 22 6.4 (2.7–12) 29 (12–54) 69 (33–120) 319 (153–544) 52 (43–61) 238 (197–283)
Cape Verde 1990 < 1 0.13 (0.034–0.290) 37 (9.8–81) 1.2 (0.440–2.3) 340 (125–660) 0.62 (0.380–0.910) 175 (108–259)
1995 < 1 0.14 (0.055–0.270) 36 (14–68) 1.3 (0.640–2.2) 326 (161–549) 0.67 (0.550–0.800) 168 (137–201)
2000 < 1 0.15 (0.057–0.290) 34 (13–65) 1.4 (0.670–2.3) 311 (152–526) 0.71 (0.580–0.850) 160 (131–193)
2005 < 1 0.15 (0.057–0.280) 31 (12–59) 1.4 (0.680–2.3) 288 (142–485) 0.73 (0.600–0.880) 153 (125–184)
2010 < 1 0.13 (0.051–0.240) 26 (11–49) 1.3 (0.630–2.1) 257 (129–427) 0.71 (0.590–0.850) 147 (121–175)
2011 < 1 0.12 (0.049–0.220) 25 (10–46) 1.2 (0.610–2.0) 248 (124–414) 0.71 (0.590–0.850) 145 (120–173)
2012 < 1 0.11 (0.047–0.210) 23 (9.5–42) 1.2 (0.590–2.0) 237 (119–395) 0.71 (0.590–0.850) 144 (119–172)
Central African 1990 3 3.6 (1.3–6.9) 122 (45–236) 40 (17–72) 1 360 (583–2 460) 25 (21–30) 861 (710–1 030)
Republic 1995 3 5.1 (1.9–9.9) 156 (57–303) 55 (23–100) 1 680 (704–3 070) 39 (32–47) 1 200 (988–1 430)
2000 4 5.2 (1.9–10) 143 (53–277) 54 (23–99) 1 500 (631–2 720) 39 (32–46) 1 070 (884–1 280)
2005 4 3.9 (1.5–7.5) 99 (37–190) 40 (17–71) 1 000 (438–1 790) 27 (23–33) 690 (569–822)
2010 4 2.6 (1.0–4.8) 59 (24–110) 28 (13–47) 637 (304–1 090) 19 (16–22) 433 (357–515)
2011 4 2.4 (0.980–4.4) 54 (22–99) 26 (12–44) 579 (279–987) 18 (15–21) 400 (330–477)
2012 5 2.2 (0.840–4.3) 50 (19–95) 24 (11–40) 520 (251–884) 17 (14–20) 367 (302–438)
Chad 1990 6 0.86 (0.370–1.5) 14 (6.2–26) 9.4 (4.7–16) 157 (78–264) 5.6 (4.7–6.7) 95 (78–112)
1995 7 1.5 (0.610–2.9) 22 (8.8–41) 16 (7.7–27) 228 (111–388) 9 (7.4–11) 128 (106–153)
2000 8 2 (0.800–3.7) 24 (9.7–45) 21 (10–36) 252 (122–429) 13 (10–15) 151 (125–180)
2005 10 2.3 (0.940–4.2) 23 (9.4–42) 24 (12–41) 243 (119–412) 15 (12–18) 150 (124–178)
2010 12 2.3 (1.0–4.1) 20 (8.5–35) 28 (14–47) 237 (118–397) 18 (15–21) 151 (125–179)
2011 12 2.2 (0.960–3.9) 18 (8.0–33) 28 (14–46) 229 (114–383) 18 (15–22) 151 (125–179)
2012 12 2.3 (0.980–4.1) 18 (7.9–33) 28 (14–46) 221 (109–372) 19 (16–22) 151 (125–180)
Comoros 1990 < 1 0.043 (0.018–0.077) 10 (4.4–19) 0.38 (0.180–0.660) 93 (45–159) 0.22 (0.180–0.270) 54 (44–64)
1995 < 1 0.038 (0.017–0.069) 8.3 (3.6–15) 0.35 (0.170–0.600) 75 (36–129) 0.21 (0.180–0.250) 46 (38–55)
2000 < 1 0.037 (0.016–0.065) 6.9 (3.0–12) 0.34 (0.160–0.580) 64 (30–111) 0.21 (0.170–0.250) 39 (32–46)
2005 < 1 0.045 (0.019–0.083) 7.6 (3.2–14) 0.4 (0.200–0.680) 67 (33–114) 0.22 (0.180–0.260) 36 (30–43)
2010 < 1 0.045 (0.019–0.082) 6.6 (2.8–12) 0.42 (0.210–0.710) 62 (31–103) 0.23 (0.190–0.270) 33 (28–40)
2011 < 1 0.046 (0.019–0.083) 6.5 (2.8–12) 0.45 (0.230–0.740) 64 (32–105) 0.24 (0.200–0.290) 34 (28–41)
2012 < 1 0.045 (0.019–0.084) 6.3 (2.6–12) 0.44 (0.220–0.740) 62 (31–103) 0.25 (0.200–0.290) 34 (28–41)
Congo 1990 2 0.7 (0.220–1.4) 29 (9.4–61) 7.7 (2.6–16) 323 (108–654) 4 (2.5–5.9) 169 (104–250)
1995 3 0.83 (0.340–1.5) 31 (13–56) 9.6 (4.6–16) 352 (168–601) 6.7 (5.4–8.0) 245 (200–294)
2000 3 1.1 (0.500–2.0) 36 (16–63) 14 (6.9–24) 455 (222–770) 11 (9.3–13) 353 (298–412)
2005 4 1.7 (0.730–3.0) 47 (21–85) 21 (9.8–35) 580 (276–994) 15 (12–18) 425 (347–510)
2010 4 1.8 (0.790–3.3) 44 (19–80) 23 (11–39) 557 (265–955) 16 (13–19) 391 (320–470)
2011 4 1.8 (0.800–3.3) 44 (19–78) 23 (11–40) 548 (262–938) 16 (13–20) 387 (317–465)
2012 4 1.8 (0.780–3.4) 42 (18–77) 23 (11–40) 530 (250–913) 17 (14–20) 381 (311–458)
Côte d'Ivoire 1990 12 4.8 (1.9–9.0) 40 (16–74) 48 (23–80) 394 (193–664) 29 (25–33) 238 (206–272)
1995 14 8.2 (3.2–16) 58 (23–109) 78 (38–130) 551 (265–940) 54 (47–62) 379 (329–433)
2000 16 9 (3.6–17) 56 (22–105) 83 (40–140) 513 (249–870) 60 (52–68) 369 (320–422)
2005 17 6.6 (2.8–12) 38 (16–69) 64 (33–100) 366 (187–604) 46 (40–53) 267 (232–306)
2010 19 4.7 (2.1–8.2) 25 (11–43) 49 (25–80) 258 (133–424) 36 (31–41) 190 (165–217)
2011 19 4.7 (2.2–8.3) 24 (11–43) 51 (26–83) 262 (135–430) 37 (32–42) 191 (165–218)
2012 20 4.4 (1.8–8.0) 22 (9.1–41) 45 (23–75) 228 (115–380) 34 (30–39) 172 (149–198)
Democratic 1990 35 26 (9.9–49) 74 (28–140) 240 (110–420) 695 (327–1 200) 110 (92–140) 327 (262–398)
Republic 1995 42 28 (11–53) 67 (27–126) 270 (130–470) 654 (318–1 110) 140 (110–160) 327 (268–392)
of the Congo 2000 47 29 (12–53) 61 (25–112) 290 (140–480) 611 (308–1 020) 150 (130–180) 327 (273–385)
2005 54 29 (13–52) 54 (24–97) 300 (150–500) 558 (285–920) 180 (150–200) 327 (279–379)
2010 62 33 (15–59) 54 (24–96) 350 (180–560) 555 (288–908) 200 (180–230) 327 (282–375)
2011 64 35 (15–62) 54 (24–97) 360 (190–590) 568 (297–923) 210 (180–240) 327 (282–375)
2012 66 36 (16–64) 54 (24–97) 380 (200–620) 576 (301–938) 210 (190–250) 327 (282–375)
Equatorial 1990 < 1 0 (0–0.063) 0 (0–17) 0.38 (0.150–0.720) 101 (39–193) 0.3 (0.260–0.340) 80 (70–91)
Guinea 1995 < 1 0 (0–0.087) 0 (0–20) 0.42 (0.160–0.820) 96 (35–187) 0.35 (0.310–0.400) 80 (70–90)
2000 < 1 0 (0–0.086) 0 (0–17) 0.67 (0.300–1.2) 130 (58–231) 0.52 (0.460–0.590) 101 (88–114)
2005 < 1 0 (0–0.058) 0 (0–9.7) 0.81 (0.350–1.5) 135 (58–243) 0.66 (0.580–0.750) 110 (96–124)
2010 < 1 0 (0–0.058) 0 (0–8.3) 1.2 (0.520–2.0) 166 (75–294) 0.94 (0.830–1.1) 135 (119–153)
2011 < 1 0 (0–0.053) 0 (0–7.4) 1.2 (0.560–2.2) 174 (79–305) 1 (0.890–1.1) 142 (124–161)
2012 < 1 0 (0–0.054) 0 (0–7.4) 1.2 (0.510–2.2) 164 (69–299) 1 (0.900–1.2) 139 (122–158)
Eritrea 1990 3 0.39 (0.260–0.540) 12 (7.9–17) 16 (6.5–29) 484 (198–894) 8 (5.3–11) 243 (162–341)
1995 3 0.33 (0.220–0.460) 9.7 (6.4–14) 12 (4.1–25) 362 (121–731) 6.7 (4.5–9.5) 198 (132–278)
2000 4 0.3 (0.200–0.420) 7.7 (5.1–11) 7.6 (1.9–17) 194 (49–436) 6.2 (4.1–8.7) 157 (104–220)
2005 5 0.29 (0.190–0.400) 5.9 (3.9–8.3) 9.1 (3.2–18) 187 (67–368) 5.9 (3.9–8.2) 121 (80–169)
2010 6 0.28 (0.190–0.390) 4.9 (3.2–6.9) 9.6 (3.6–18) 167 (63–319) 5.8 (3.8–8.1) 100 (67–140)
2011 6 0.28 (0.190–0.390) 4.7 (3.1–6.6) 9.5 (3.6–18) 160 (60–307) 5.7 (3.8–8.1) 97 (64–136)
2012 6 0.28 (0.190–0.400) 4.6 (3.0–6.5) 9.3 (3.5–18) 152 (56–294) 5.7 (3.8–8.0) 93 (62–131)
MORTALITY (EXCLUDING HIV) PREVALENCE (INCLUDING HIV) INCIDENCE (INCLUDING HIV)
a
Rates are per 100 000 population.

GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($(VWLPDWHVRIWKHEXUGHQRIGLVHDVHFDXVHGE\7%±
YEAR POPULATION
(MILLIONS)
NUMBER
(THOUSANDS) RATE
a
NUMBER
(THOUSANDS) RATE
a
NUMBER
(THOUSANDS) RATE
a
MORTALITY (EXCLUDING HIV) PREVALENCE (INCLUDING HIV) INCIDENCE (INCLUDING HIV)
Ethiopia 1990 48 23 (14–35) 49 (29–73) 200 (140–290) 426 (285–594) 180 (100–270) 367 (218–553)
1995 57 27 (16–41) 48 (28–72) 270 (200–370) 480 (342–642) 240 (140–360) 419 (249–633)
2000 66 27 (16–41) 41 (25–63) 280 (210–370) 429 (318–556) 280 (170–420) 421 (251–636)
2005 76 22 (13–33) 29 (17–44) 250 (190–320) 331 (250–422) 260 (150–390) 342 (203–516)
2010 87 17 (12–23) 20 (14–26) 220 (170–270) 250 (199–307) 230 (170–310) 269 (191–359)
2011 89 16 (12–21) 18 (14–24) 210 (170–260) 237 (191–288) 230 (170–300) 258 (191–335)
2012 92 16 (12–21) 18 (13–23) 210 (170–250) 224 (180–272) 230 (170–290) 247 (183–321)
Gabon 1990 < 1 0.39 (0.160–0.710) 41 (17–74) 4 (2.0–6.6) 419 (210–699) 2.1 (1.7–2.5) 221 (182–263)
1995 1 0.62 (0.250–1.2) 57 (23–107) 6.4 (3.1–11) 592 (289–1 000) 3.4 (2.8–4.1) 315 (260–375)
2000 1 0.99 (0.390–1.9) 81 (32–152) 11 (5.2–19) 898 (426–1 540) 6.5 (5.3–7.7) 527 (435–627)
2005 1 1.1 (0.430–2.0) 78 (32–145) 13 (6.0–21) 908 (434–1 550) 8.1 (6.7–9.6) 586 (484–698)
2010 2 0.8 (0.340–1.4) 51 (22–93) 10 (5.0–17) 663 (323–1 120) 7.4 (6.1–8.8) 475 (392–566)
2011 2 0.73 (0.320–1.3) 46 (20–83) 9.8 (4.7–17) 612 (295–1 040) 7.2 (5.9–8.6) 450 (372–536)
2012 2 0.72 (0.300–1.3) 44 (18–81) 9.2 (4.3–16) 563 (265–971) 7 (5.8–8.3) 428 (354–510)
Gambia 1990 < 1 0.33 (0.087–0.730) 36 (9.4–80) 3.2 (1.2–6.2) 350 (129–679) 1.7 (1.0–2.5) 185 (114–273)
1995 1 0.4 (0.160–0.740) 38 (15–70) 4 (2.0–6.6) 372 (186–622) 2.2 (1.8–2.6) 204 (167–245)
2000 1 0.44 (0.180–0.810) 36 (15–66) 4.6 (2.3–7.7) 373 (184–628) 2.8 (2.3–3.3) 225 (184–271)
2005 1 0.57 (0.230–1.1) 39 (16–74) 5.8 (2.9–9.7) 404 (200–677) 3.6 (2.9–4.3) 248 (203–298)
2010 2 0.78 (0.300–1.5) 46 (18–87) 7.7 (3.8–13) 455 (227–762) 4.6 (3.8–5.5) 273 (226–325)
2011 2 0.84 (0.330–1.6) 49 (19–92) 8.2 (4.1–14) 472 (236–788) 4.8 (4.0–5.7) 279 (230–331)
2012 2 0.91 (0.360–1.7) 51 (20–96) 8.8 (4.4–15) 490 (245–819) 5.1 (4.2–6.0) 284 (234–337)
Ghana 1990 15 5.3 (0.880–14) 36 (6.0–93) 47 (12–110) 320 (81–722) 23 (10–40) 155 (69–275)
1995 17 5.4 (1.1–13) 32 (6.3–79) 50 (15–110) 301 (91–634) 28 (16–44) 167 (93–263)
2000 19 5.1 (1.2–12) 27 (6.3–62) 48 (17–96) 257 (89–510) 29 (18–41) 152 (97–220)
2005 21 4 (1.3–8.3) 19 (5.9–39) 40 (17–72) 188 (81–338) 25 (19–33) 119 (88–154)
2010 24 2.5 (1.2–4.4) 10 (5.0–18) 29 (15–48) 121 (62–199) 21 (18–24) 86 (75–97)
2011 25 2.1 (1.0–3.6) 8.6 (4.2–15) 26 (13–44) 106 (52–179) 20 (17–22) 79 (69–89)
2012 25 1.7 (0.880–2.9) 6.9 (3.5–11) 23 (11–41) 92 (41–162) 18 (16–21) 72 (63–82)
Guinea 1990 6 3.7 (1.4–7.2) 62 (23–119) 33 (15–58) 556 (257–968) 15 (12–18) 248 (204–295)
1995 8 4.2 (1.6–8.1) 54 (21–103) 40 (19–68) 505 (237–873) 20 (16–23) 249 (205–297)
2000 9 3.9 (1.5–7.3) 44 (17–83) 38 (18–64) 429 (205–734) 20 (17–24) 234 (193–279)
2005 10 3.3 (1.4–6.1) 34 (14–63) 33 (16–57) 350 (171–590) 20 (17–24) 211 (173–251)
2010 11 2.8 (1.2–5.0) 25 (11–46) 33 (16–54) 299 (149–500) 20 (17–24) 188 (155–224)
2011 11 2.6 (1.2–4.7) 24 (10–42) 32 (16–54) 287 (143–481) 20 (17–24) 183 (151–219)
2012 11 2.6 (1.1–4.8) 23 (9.8–42) 31 (16–53) 274 (136–459) 20 (17–24) 178 (146–213)
Guinea-Bissau 1990 1 0.21 (0.051–0.480) 21 (5.0–47) 2.4 (0.860–4.8) 237 (84–469) 1.6 (1.1–2.2) 158 (108–217)
1995 1 0.26 (0.100–0.470) 22 (9.2–41) 2.9 (1.4–5.0) 256 (120–442) 2 (1.6–2.4) 174 (142–209)
2000 1 0.36 (0.130–0.690) 28 (11–54) 3.7 (1.8–6.2) 290 (142–490) 2.4 (2.0–2.9) 192 (157–230)
2005 1 0.32 (0.110–0.660) 23 (7.5–46) 3.8 (1.7–6.6) 264 (121–462) 3 (2.5–3.6) 211 (173–254)
2010 2 0.43 (0.150–0.860) 27 (9.4–54) 4.8 (2.3–8.1) 300 (144–513) 3.7 (3.0–4.4) 233 (192–278)
2011 2 0.46 (0.150–0.920) 28 (9.5–57) 5 (2.4–8.5) 306 (145–525) 3.9 (3.2–4.6) 238 (196–283)
2012 2 0.49 (0.160–0.990) 29 (9.8–59) 5.2 (2.5–8.9) 312 (148–537) 4 (3.3–4.8) 242 (200–289)
Kenya 1990 23 7.1 (2.8–13) 30 (12–57) 64 (32–110) 272 (138–452) 33 (28–37) 139 (121–159)
1995 27 4.5 (2.0–7.9) 16 (7.4–29) 54 (29–85) 196 (108–311) 46 (43–50) 169 (155–184)
2000 31 5.9 (2.8–10) 19 (9.0–32) 85 (46–140) 273 (147–437) 89 (84–95) 286 (267–305)
2005 36 8.1 (4.3–13) 23 (12–36) 120 (65–200) 345 (183–559) 130 (120–140) 359 (339–380)
2010 41 8.2 (5.1–12) 20 (12–30) 130 (65–200) 306 (159–500) 120 (120–130) 298 (286–311)
2011 42 8.9 (5.3–14) 21 (13–32) 130 (68–210) 305 (163–491) 120 (120–130) 288 (276–300)
2012 43 9.5 (5.4–15) 22 (13–34) 130 (71–210) 299 (164–475) 120 (110–120) 272 (261–283)
Lesotho 1990 2 0.35 (0.100–0.730) 22 (6.5–46) 4.3 (1.6–8.2) 267 (99–515) 2.9 (2.2–3.8) 184 (135–240)
1995 2 0.34 (0.110–0.680) 19 (6.5–39) 5.7 (2.4–10) 323 (134–593) 5.7 (5.0–6.4) 323 (283–367)
2000 2 0.29 (<0.01–1.1) 16 (0.38–58) 7.2 (2.4–15) 387 (129–784) 10 (9.0–12) 553 (484–626)
2005 2 0.2 (0–1.6) 10 (0–82) 7.9 (1.3–20) 409 (65–1 060) 12 (10–14) 639 (535–752)
2010 2 0.27 (<0.01–1.3) 14 (<0.1–66) 8.5 (2.5–18) 425 (126–903) 13 (11–14) 633 (553–719)
2011 2 0.34 (<0.01–1.4) 17 (0.20–67) 8.9 (2.8–18) 439 (139–905) 13 (11–15) 632 (551–717)
2012 2 0.34 (<0.01–1.4) 17 (0.18–68) 8.7 (2.7–18) 424 (130–888) 13 (11–15) 630 (550–716)
Liberia 1990 2 0.62 (0.130–1.5) 29 (6.0–71) 6.7 (2.2–14) 321 (102–661) 4.2 (2.6–6.2) 199 (123–293)
1995 2 1.1 (0.400–2.0) 51 (19–97) 9.4 (4.6–16) 453 (220–769) 4.6 (3.7–5.5) 219 (179–263)
2000 3 1.6 (0.580–3.0) 54 (20–104) 14 (6.7–24) 482 (231–822) 7 (5.7–8.4) 242 (197–290)
2005 3 1.6 (0.630–3.1) 50 (19–94) 16 (7.7–26) 475 (234–798) 8.7 (7.1–10) 266 (218–320)
2010 4 1.9 (0.790–3.5) 48 (20–88) 20 (9.8–33) 493 (247–822) 12 (9.6–14) 293 (242–349)
2011 4 1.9 (0.810–3.5) 47 (20–86) 20 (10–34) 494 (245–827) 12 (10–15) 299 (247–356)
2012 4 1.9 (0.830–3.5) 46 (20–84) 21 (10–35) 495 (244–832) 13 (11–15) 304 (251–362)
Madagascar 1990 12 13 (4.9–25) 114 (43–220) 110 (50–190) 946 (434–1 650) 45 (37–54) 391 (322–466)
1995 13 11 (4.4–21) 82 (33–154) 98 (48–170) 729 (356–1 230) 45 (37–54) 335 (276–400)
2000 16 11 (4.4–20) 69 (28–127) 96 (47–160) 609 (300–1 020) 46 (38–55) 293 (241–349)
2005 18 10 (4.3–19) 56 (24–103) 95 (48–160) 522 (261–870) 48 (39–57) 262 (216–313)
2010 21 10 (4.3–18) 48 (21–88) 97 (49–160) 461 (233–767) 51 (42–61) 242 (199–288)
2011 22 10 (4.4–18) 47 (20–85) 98 (50–160) 452 (228–749) 52 (43–62) 238 (196–284)
2012 22 10 (4.3–19) 46 (19–84) 99 (50–160) 442 (222–735) 52 (43–62) 234 (193–280)
Malawi 1990 9 3.8 (0.700–9.5) 40 (7.4–101) 39 (12–80) 412 (131–849) 31 (22–41) 326 (230–438)
1995 10 3.7 (0.760–8.9) 37 (7.6–90) 43 (16–81) 427 (165–810) 46 (38–55) 462 (383–548)
2000 11 3.2 (0.360–9.0) 28 (3.2–80) 41 (13–85) 365 (118–749) 53 (44–63) 467 (387–554)
2005 13 2.3 (0.130–7.4) 18 (1.0–57) 34 (10–72) 262 (77–556) 46 (38–54) 354 (292–421)
2010 15 1.8 (0.680–3.5) 12 (4.5–23) 27 (14–45) 182 (95–298) 33 (31–35) 219 (203–236)
2011 15 1.5 (0.560–3.0) 9.9 (3.6–19) 24 (12–40) 156 (80–256) 30 (27–32) 191 (177–206)
2012 16 1.4 (0.570–2.7) 9 (3.6–17) 22 (11–36) 140 (72–229) 26 (24–28) 163 (151–176)
Mali 1990 8 1.2 (0.510–2.1) 15 (6.4–26) 11 (6.0–18) 138 (75–221) 6 (5.8–6.3) 76 (72–80)
1995 9 1.2 (0.540–2.2) 13 (6.0–24) 12 (6.5–18) 131 (73–206) 7.2 (6.9–7.6) 80 (76–84)
2000 10 1.2 (0.560–2.2) 12 (5.5–21) 12 (6.7–19) 117 (65–184) 7.9 (7.6–8.3) 77 (74–81)
2005 12 1.3 (0.600–2.2) 11 (5.0–19) 12 (7.0–20) 105 (58–164) 8.3 (7.9–8.7) 69 (66–73)
2010 14 1.3 (0.620–2.2) 9.3 (4.4–16) 13 (7.3–21) 94 (52–149) 8.8 (8.4–9.2) 63 (60–66)
2011 14 1.3 (0.630–2.2) 9.1 (4.4–16) 13 (7.4–21) 94 (52–148) 8.9 (8.5–9.3) 62 (59–65)
2012 15 1.3 (0.630–2.3) 9 (4.3–15) 14 (7.6–22) 92 (51–146) 9 (8.5–9.4) 60 (57–63)
Mauritania 1990 2 0.38 (0.015–1.3) 19 (0.76–65) 5.7 (1.1–14) 283 (53–703) 4.6 (2.8–6.8) 228 (140–336)
1995 2 0.92 (0.390–1.7) 39 (17–71) 9.7 (4.7–17) 417 (202–707) 5.9 (4.8–7.0) 251 (205–302)
2000 3 1.5 (0.610–2.9) 57 (23–106) 15 (7.3–24) 536 (268–895) 7.5 (6.1–9.0) 277 (226–333)
2005 3 2.3 (0.880–4.4) 73 (28–140) 20 (9.9–35) 651 (315–1 110) 9.6 (7.9–12) 305 (250–367)
2010 4 3.2 (1.2–6.1) 88 (33–170) 27 (13–47) 756 (357–1 300) 12 (10–15) 337 (277–402)
2011 4 3.4 (1.2–6.5) 91 (34–175) 29 (13–50) 775 (364–1 340) 13 (10–15) 344 (283–410)
2012 4 3.5 (1.3–6.9) 93 (34–181) 30 (14–52) 794 (373–1 370) 13 (11–16) 350 (288–418)
Mauritius 1990 1 0.027 (0.026–0.028) 2.5 (2.4–2.6) 0.58 (0.220–1.1) 55 (21–105) 0.29 (0.180–0.430) 28 (17–41)
1995 1 0.013 (0.012–0.013) 1.1 (1.1–1.2) 0.57 (0.290–0.950) 51 (25–85) 0.29 (0.240–0.350) 26 (21–31)
2000 1 <0.01 (<0.01–<0.01) 0.68 (0.67–0.70) 0.54 (0.270–0.910) 46 (23–76) 0.29 (0.240–0.350) 24 (20–29)
2005 1 0.013 (0.013–0.014) 1.1 (1.1–1.1) 0.52 (0.260–0.870) 43 (22–72) 0.28 (0.230–0.330) 23 (19–28)
2010 1 0.012 (0.011–0.012) 0.94 (0.93–0.95) 0.5 (0.250–0.830) 41 (20–68) 0.27 (0.220–0.320) 22 (18–26)
2011 1 0.019 (0.019–0.019) 1.5 (1.5–1.6) 0.49 (0.250–0.820) 40 (20–66) 0.26 (0.220–0.310) 21 (18–25)
2012 1 0.012 (0.012–0.012) 0.97 (0.96–0.98) 0.48 (0.240–0.810) 39 (20–65) 0.26 (0.210–0.310) 21 (17–25)
Mozambique 1990 14 13 (0.360–48) 98 (2.6–357) 120 (7.5–370) 863 (56–2 730) 54 (8.5–140) 401 (62–1 050)
1995 16 16 (1.0–52) 101 (6.4–323) 140 (17–400) 897 (104–2 520) 76 (24–160) 478 (153–985)
2000 18 14 (0.430–49) 75 (2.3–270) 130 (10–390) 701 (56–2 130) 94 (41–170) 513 (227–914)
2005 21 12 (0.360–44) 58 (1.7–208) 120 (12–350) 576 (59–1 660) 110 (63–170) 524 (298–811)
2010 24 12 (0.890–38) 51 (3.7–160) 130 (25–320) 541 (105–1 320) 130 (90–180) 544 (377–741)
2011 25 13 (0.890–40) 51 (3.6–161) 130 (26–330) 544 (106–1 330) 130 (93–180) 548 (380–747)
2012 25 13 (0.980–41) 53 (3.9–163) 140 (28–340) 553 (111–1 340) 140 (96–190) 552 (383–753)
Namibia 1990 1 0.074 (0.058–0.091) 5.2 (4.1–6.4) 11 (5.1–18) 751 (358–1 280) 5.4 (4.3–6.6) 379 (300–468)
1995 2 0.15 (0.110–0.180) 8.8 (6.9–11) 13 (5.7–23) 770 (344–1 360) 9.5 (7.5–12) 575 (456–709)
2000 2 0.46 (0.360–0.580) 24 (19–31) 27 (9.8–53) 1 430 (517–2 790) 27 (21–33) 1 410 (1 110–1 730)
2005 2 0.46 (0.350–0.580) 23 (17–28) 23 (6.5–51) 1 160 (321–2 510) 28 (22–35) 1 390 (1 100–1 720)
2010 2 0.34 (0.270–0.410) 15 (12–19) 18 (6.3–36) 834 (291–1 660) 19 (15–23) 867 (686–1 070)
2011 2 0.32 (0.250–0.390) 14 (11–18) 15 (5.3–31) 699 (241–1 390) 16 (13–20) 723 (573–891)
2012 2 0.32 (0.260–0.400) 14 (11–18) 16 (6.1–29) 688 (271–1 300) 15 (12–18) 655 (524–800)
a
Rates are per 100 000 population.
159 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
AFRICAN REGION
7$%/($(VWLPDWHVRIWKHEXUGHQRIGLVHDVHFDXVHGE\7%±
YEAR POPULATION
(MILLIONS)
NUMBER
(THOUSANDS) RATE
a
NUMBER
(THOUSANDS) RATE
a
NUMBER
(THOUSANDS) RATE
a
MORTALITY (EXCLUDING HIV) PREVALENCE (INCLUDING HIV) INCIDENCE (INCLUDING HIV)
Niger 1990 8 7.7 (2.9–15) 99 (37–191) 65 (30–110) 839 (388–1 460) 28 (23–33) 358 (295–426)
1995 9 6.8 (2.5–13) 74 (28–143) 57 (26–99) 620 (283–1 080) 25 (20–29) 270 (223–321)
2000 11 5 (2.0–9.5) 46 (18–86) 44 (21–75) 396 (189–678) 21 (17–25) 191 (157–227)
2005 13 3.7 (1.5–6.8) 28 (12–51) 34 (17–57) 261 (130–436) 19 (15–22) 142 (118–170)
2010 16 2.9 (1.3–5.2) 18 (8.0–33) 30 (15–50) 187 (94–312) 18 (15–21) 113 (94–135)
2011 17 2.8 (1.2–5.0) 17 (7.4–30) 29 (14–49) 176 (88–294) 18 (15–21) 108 (90–129)
2012 17 2.8 (1.2–5.1) 16 (6.8–30) 28 (14–48) 166 (83–277) 18 (15–21) 104 (86–124)
Nigeria 1990 96 34 (0.019–180) 35 (<0.1–183) 290 (0.550–1 400) 302 (0.58–1 440) 120 (1.3–500) 128 (1.3–526)
1995 108 40 (0.250–170) 37 (0.23–161) 340 (4.9–1 300) 311 (4.5–1 230) 150 (8.0–490) 139 (7.4–456)
2000 123 47 (0.077–230) 38 (<0.1–185) 400 (2.6–1 700) 326 (2.1–1 400) 210 (15–660) 172 (12–536)
2005 140 46 (0.084–220) 33 (<0.1–159) 420 (4.2–1 700) 298 (3.0–1 220) 240 (33–660) 175 (23–476)
2010 160 34 (3.1–100) 22 (2.0–64) 330 (62–830) 210 (39–521) 210 (100–360) 133 (64–225)
2011 164 30 (2.0–93) 18 (1.2–57) 300 (49–760) 181 (30–464) 190 (91–340) 118 (55–204)
2012 169 27 (1.6–86) 16 (0.92–51) 270 (43–710) 161 (25–420) 180 (85–310) 108 (50–186)
Rwanda 1990 7 2.7 (1.1–5.1) 37 (15–70) 26 (13–44) 356 (173–603) 21 (19–23) 290 (259–323)
1995 6 4.4 (1.7–8.4) 78 (30–149) 37 (17–64) 655 (305–1 130) 29 (26–32) 513 (458–571)
2000 8 4.1 (1.6–7.7) 49 (19–91) 35 (17–59) 417 (205–701) 27 (24–30) 325 (290–362)
2005 9 2 (0.880–3.7) 22 (9.3–39) 21 (11–35) 228 (120–370) 17 (15–19) 181 (162–202)
2010 11 1.3 (0.610–2.3) 12 (5.7–21) 15 (7.9–24) 136 (73–219) 11 (10–13) 106 (94–118)
2011 11 1.2 (0.580–2.1) 11 (5.2–19) 14 (7.2–22) 121 (65–196) 11 (9.4–12) 94 (84–105)
2012 11 1.2 (0.530–2.1) 10 (4.6–18) 13 (7.0–21) 114 (61–183) 9.8 (8.8–11) 86 (77–96)
Sao Tome and 1990 < 1 0.031 (<0.01–0.070) 27 (7.1–59) 0.3 (0.110–0.590) 258 (96–499) 0.16 (0.098–0.230) 135 (83–199)
Principe 1995 < 1 0.034 (0.013–0.064) 26 (10–49) 0.32 (0.160–0.530) 244 (122–408) 0.16 (0.130–0.190) 124 (102–149)
2000 < 1 0.018 (<0.01–0.032) 13 (5.3–23) 0.22 (0.093–0.410) 159 (67–291) 0.16 (0.130–0.190) 114 (93–137)
2005 < 1 0.012 (<0.01–0.023) 7.6 (2.8–15) 0.2 (0.070–0.390) 128 (46–253) 0.16 (0.140–0.190) 105 (88–123)
2010 < 1 0.024 (0.010–0.045) 14 (5.7–25) 0.26 (0.130–0.450) 149 (72–252) 0.17 (0.140–0.200) 96 (79–115)
2011 < 1 0.027 (0.011–0.050) 15 (6.1–27) 0.28 (0.140–0.470) 154 (76–258) 0.17 (0.140–0.210) 94 (78–113)
2012 < 1 0.03 (0.012–0.055) 16 (6.4–29) 0.3 (0.150–0.500) 159 (80–264) 0.17 (0.140–0.210) 93 (76–111)
Senegal 1990 8 1.8 (0.800–3.3) 24 (11–44) 19 (9.4–31) 249 (125–414) 10 (8.5–12) 138 (114–164)
1995 9 2.2 (0.980–4.0) 26 (11–46) 23 (12–39) 269 (135–448) 13 (11–16) 153 (126–183)
2000 10 2.6 (1.1–4.7) 26 (12–48) 27 (14–45) 273 (137–453) 15 (13–18) 155 (128–184)
2005 11 2.5 (1.1–4.4) 22 (9.6–39) 26 (13–44) 234 (116–393) 16 (13–19) 142 (117–169)
2010 13 2.5 (1.1–4.5) 20 (8.7–35) 28 (14–47) 217 (106–366) 18 (15–21) 137 (113–163)
2011 13 2.6 (1.2–4.7) 20 (8.7–35) 29 (14–49) 217 (106–366) 18 (15–22) 136 (112–162)
2012 14 2.7 (1.2–4.8) 20 (8.8–35) 30 (15–50) 219 (108–368) 19 (16–22) 137 (113–163)
Seychelles 1990 < 1 <0.01 (<0.01–<0.01) 2 (1.9–2.0) 0.035 (<0.01–0.091) 50 (7.7–131) 0.03 (0.019–0.044) 43 (27–64)
1995 < 1 <0.01 (<0.01–<0.01) 2 (1.9–2.0) 0.059 (0.028–0.100) 79 (37–135) 0.03 (0.025–0.036) 40 (33–48)
2000 < 1 <0.01 (<0.01–<0.01) 2 (1.9–2.0) 0.045 (0.021–0.080) 57 (26–100) 0.029 (0.024–0.035) 37 (30–44)
2005 < 1 <0.01 (<0.01–<0.01) 2.5 (2.4–2.7) 0.053 (0.026–0.091) 61 (29–104) 0.029 (0.024–0.035) 33 (27–40)
2010 < 1 <0.01 (<0.01–<0.01) 1.8 (1.8–1.9) 0.048 (0.023–0.082) 52 (25–90) 0.028 (0.023–0.033) 31 (25–37)
2011 < 1 <0.01 (<0.01–<0.01) 1.8 (1.8–1.9) 0.039 (0.016–0.071) 42 (17–78) 0.028 (0.023–0.033) 30 (25–36)
2012 < 1 <0.01 (<0.01–<0.01) 1.8 (1.8–1.9) 0.036 (0.013–0.072) 39 (14–78) 0.027 (0.023–0.033) 30 (24–35)
Sierra Leone 1990 4 2.4 (0.730–5.2) 61 (18–128) 21 (7.8–39) 507 (194–968) 8.4 (5.2–12) 207 (128–305)
1995 4 2 (0.730–3.9) 51 (18–99) 18 (8.3–31) 454 (211–788) 8.3 (6.4–11) 212 (162–269)
2000 4 2.4 (0.860–4.8) 59 (21–116) 22 (10–39) 537 (245–940) 11 (8.1–14) 264 (196–341)
2005 5 5.9 (2.2–11) 116 (43–223) 53 (25–90) 1 030 (491–1 750) 26 (21–31) 503 (410–605)
2010 6 8.2 (3.1–16) 142 (54–273) 74 (36–130) 1 290 (624–2 180) 38 (31–45) 660 (540–791)
2011 6 8.3 (3.1–16) 142 (53–274) 76 (37–130) 1 290 (625–2 200) 39 (32–47) 668 (542–807)
2012 6 8.5 (3.2–16) 143 (53–275) 78 (37–130) 1 300 (626–2 220) 40 (32–49) 674 (540–821)
South Africa 1990 37 16 (4.2–34) 42 (11–93) 170 (64–340) 475 (173–925) 110 (76–150) 301 (206–413)
1995 41 15 (5.6–28) 35 (14–68) 180 (81–310) 427 (195–747) 130 (110–160) 317 (259–381)
2000 45 20 (4.0–48) 44 (8.9–107) 250 (100–480) 568 (225–1 070) 260 (210–310) 576 (471–691)
2005 48 25 (2.0–75) 51 (4.1–156) 360 (110–750) 748 (234–1 560) 450 (360–540) 925 (756–1 110)
2010 51 26 (2.2–80) 51 (4.3–155) 410 (140–840) 803 (266–1 630) 500 (420–600) 981 (809–1 170)
2011 52 28 (2.9–83) 55 (5.6–159) 430 (150–860) 831 (289–1 650) 520 (430–610) 993 (819–1 180)
2012 52 31 (3.7–86) 59 (7.0–164) 450 (160–880) 857 (305–1 680) 530 (430–630) 1 000 (827–1 190)
Swaziland 1990 < 1 0.31 (0.051–0.800) 36 (5.9–93) 3.4 (1.0–7.3) 397 (116–847) 2.3 (1.4–3.4) 267 (165–394)
1995 < 1 0.27 (0.065–0.620) 28 (6.8–64) 3.4 (1.4–6.3) 357 (149–653) 3.2 (2.7–3.9) 337 (275–405)
2000 1 0.37 (<0.01–1.4) 34 (0.74–129) 6.1 (1.5–14) 573 (145–1 290) 8.5 (7.0–10) 803 (657–964)
2005 1 0.32 (<0.01–2.0) 29 (0–177) 7.4 (1.1–19) 666 (104–1 730) 13 (10–15) 1 150 (938–1 380)
2010 1 0.46 (<0.01–2.3) 39 (<0.1–196) 9 (1.5–23) 751 (130–1 900) 15 (13–18) 1 290 (1 060–1 530)
2011 1 0.67 (0.015–2.5) 56 (1.3–208) 11 (2.6–24) 870 (213–1 970) 16 (13–19) 1 320 (1 090–1 570)
2012 1 0.78 (0.031–2.7) 63 (2.5–219) 11 (2.9–25) 907 (232–2 030) 17 (14–20) 1 350 (1 110–1 610)
Togo 1990 4 0.23 (0.100–0.410) 6.1 (2.7–11) 2.7 (1.3–4.6) 71 (34–122) 1.8 (1.5–2.1) 47 (39–56)
1995 4 0.37 (0.160–0.670) 8.7 (3.8–16) 4 (2.0–6.6) 92 (46–155) 2.5 (2.0–3.0) 58 (48–69)
2000 5 0.59 (0.250–1.1) 12 (5.0–22) 5.6 (2.7–9.4) 114 (56–193) 3.5 (2.9–4.2) 72 (59–86)
2005 6 0.58 (0.250–1.0) 10 (4.5–19) 5.9 (2.9–10) 107 (52–181) 4.2 (3.5–5.1) 77 (63–91)
2010 6 0.54 (0.230–0.970) 8.5 (3.7–15) 6.2 (3.0–11) 99 (47–169) 4.6 (3.8–5.5) 73 (60–87)
2011 6 0.56 (0.240–1.0) 8.6 (3.8–15) 6.6 (3.2–11) 102 (49–174) 4.7 (3.9–5.6) 73 (60–87)
2012 7 0.58 (0.250–1.0) 8.7 (3.8–15) 6.9 (3.4–12) 104 (51–176) 4.9 (4.0–5.8) 73 (60–87)
Uganda 1990 18 8.7 (<0.01–48) 50 (<0.1–273) 86 (0.490–370) 492 (2.8–2 140) 110 (57–180) 624 (328–1 010)
1995 21 8.3 (<0.01–46) 40 (<0.1–223) 89 (0.760–370) 429 (3.6–1 800) 110 (62–180) 542 (297–860)
2000 24 8.5 (0.055–37) 35 (0.23–151) 92 (6.1–290) 380 (25–1 200) 100 (63–150) 427 (259–636)
2005 29 7.6 (0.660–23) 26 (2.3–79) 88 (22–200) 305 (76–689) 87 (63–120) 304 (220–402)
2010 34 5.5 (1.0–14) 16 (3.0–40) 70 (27–130) 207 (80–392) 71 (57–86) 209 (169–253)
2011 35 5.1 (0.960–13) 15 (2.7–36) 68 (26–130) 192 (74–366) 68 (55–82) 193 (156–234)
2012 36 4.7 (0.820–12) 13 (2.3–33) 64 (24–120) 175 (67–334) 65 (53–79) 179 (145–216)
United Republic 1990 25 9.9 (3.8–19) 39 (15–74) 94 (47–160) 368 (185–611) 58 (49–67) 226 (193–261)
of Tanzania 1995 30 6.4 (2.2–13) 21 (7.3–43) 76 (37–130) 254 (123–430) 68 (58–78) 226 (193–261)
2000 34 5.8 (1.9–12) 17 (5.6–35) 80 (38–140) 234 (113–399) 80 (70–91) 236 (207–268)
2005 39 5.7 (2.7–9.8) 15 (6.9–25) 82 (43–130) 211 (111–341) 83 (76–89) 213 (197–229)
2010 45 6.2 (3.3–10) 14 (7.4–22) 86 (46–140) 190 (102–306) 80 (75–85) 177 (166–189)
2011 46 6.1 (3.3–9.8) 13 (7.1–21) 85 (45–140) 183 (97–295) 78 (74–83) 169 (159–180)
2012 48 6.1 (3.2–9.9) 13 (6.8–21) 84 (45–140) 176 (95–283) 79 (74–84) 165 (154–175)
Zambia 1990 8 4.9 (1.5–10) 63 (19–132) 52 (24–91) 665 (308–1 160) 56 (49–63) 710 (624–801)
1995 9 3.9 (1.1–8.3) 44 (13–93) 53 (26–90) 605 (299–1 020) 70 (64–76) 788 (719–861)
2000 10 3.1 (0.940–6.5) 31 (9.4–64) 53 (26–89) 524 (256–885) 72 (67–77) 713 (661–767)
2005 11 2.2 (0.450–5.4) 19 (3.9–47) 47 (20–83) 406 (177–727) 65 (59–71) 566 (519–615)
2010 13 3.2 (0.970–6.8) 24 (7.4–51) 51 (25–87) 387 (186–659) 61 (55–67) 462 (418–509)
2011 14 3.4 (1.1–7.0) 25 (8.0–52) 52 (25–87) 379 (185–640) 61 (55–67) 444 (401–489)
2012 14 3.9 (1.4–7.7) 28 (9.8–55) 55 (28–90) 388 (197–642) 60 (54–66) 427 (385–470)
Zimbabwe 1990 10 3.5 (0.068–13) 33 (0.65–128) 34 (2.4–110) 323 (23–1 000) 31 (17–50) 296 (159–476)
1995 12 1.9 (0–15) 16 (0–125) 34 (0.860–130) 295 (7.4–1 090) 56 (39–77) 483 (335–658)
2000 13 2.1 (0–16) 17 (0–129) 49 (3.9–150) 389 (31–1 180) 91 (72–110) 726 (573–897)
2005 13 3.6 (<0.01–18) 28 (<0.1–146) 60 (7.0–170) 473 (55–1 320) 100 (81–130) 799 (634–984)
2010 13 4 (0.037–17) 31 (0.28–129) 57 (9.1–150) 438 (70–1 140) 83 (64–100) 633 (489–795)
2011 13 4.6 (0.140–17) 35 (1.1–125) 61 (12–150) 458 (93–1 110) 81 (62–100) 603 (466–757)
2012 14 4.6 (0.160–16) 33 (1.2–117) 59 (13–140) 433 (92–1 030) 77 (60–97) 562 (434–706)
a
Rates are per 100 000 population.

160 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($,QFLGHQFHQRWLILFDWLRQDQGFDVHGHWHFWLRQUDWHVDOOIRUPV±
INCIDENCE (INCLUDING HIV) INCIDENCE HIV-POSITIVE NOTIFIED NEW AND RELAPSE
b
CASE DETECTION
YEAR POPULATION
(MILLIONS)
NUMBER
(THOUSANDS) RATE
a
NUMBER
(THOUSANDS) RATE
a
NUMBER RATE
a
PERCENT
Algeria 1990 26 17 (13–22) 66 (48–86) <0.01 (<0.01–<0.01) <0.1 (0–<0.1) 11 607 44 67 (52–92)
1995 29 20 (15–26) 68 (50–89) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 13 507 46 68 (52–92)
2000 32 28 (20–36) 87 (64–114) 0.025 (0.018–0.032) <0.1 (<0.1–0.10) 18 572 59 67 (52–92)
2005 34 31 (23–41) 93 (68–121) 0.057 (0.042–0.074) 0.2 (0.12–0.22) 21 336 63 68 (52–92)
2010 37 33 (24–44) 90 (66–118) 0.08 (0.059–0.10) 0.2 (0.16–0.28) 22 336 60 67 (51–91)
2011 38 34 (25–44) 90 (66–117) 0.085 (0.062–0.11) 0.2 (0.16–0.29) 21 429 57 63 (48–87)
2012 38 34 (25–45) 89 (65–117) 0.086 (0.063–0.11) 0.2 (0.16–0.29) 21 880 57 64 (49–87)
Angola 1990 10 21 (13–31) 205 (127–303) 0.51 (0.31–0.75) 4.9 (3.0–7.2) 10 271 99 48 (33–78)
1995 12 27 (22–33) 226 (185–272) 1.3 (1.1–1.5) 11 (8.7–13) 5 143 42 19 (16–23)
2000 14 35 (28–42) 250 (204–300) 2.5 (2.0–3.0) 18 (14–21) 16 062 115 46 (38–56)
2005 17 46 (37–54) 276 (227–329) 4 (3.3–4.8) 24 (20–29) 37 175 225 82 (68–99)
2010 20 59 (50–69) 304 (256–355) 5.3 (4.5–6.2) 27 (23–32) 44 655 228 75 (64–89)
2011 20 62 (53–73) 310 (261–362) 5.3 (4.5–6.2) 26 (22–31) 47 240 234 76 (65–90)
2012 21 66 (55–77) 316 (266–369) 5.5 (4.7–6.5) 27 (22–31) 51 819 249 79 (67–94)
Benin 1990 5 6.4 (5.3–7.6) 128 (106–152) 2.1 (1.7–2.4) 41 (34–49) 2 074 41 32 (27–39)
1995 6 6 (4.9–7.1) 100 (82–118) 1.9 (1.5–2.2) 31 (26–37) 2 400 40 40 (34–49)
2000 7 6 (4.9–7.1) 86 (71–102) 1.5 (1.3–1.8) 22 (18–26) 2 697 39 45 (38–55)
2005 8 6 (5.0–7.2) 74 (61–88) 1.2 (1.0–1.5) 15 (12–18) 3 270 40 54 (46–66)
2010 10 6.5 (5.4–7.8) 69 (57–82) 0.99 (0.82–1.2) 10 (8.6–12) 3 756 39 57 (48–69)
2011 10 6.8 (5.6–8.1) 70 (58–83) 1 (0.84–1.2) 10 (8.6–12) 4 212 43 62 (52–75)
2012 10 7 (5.8–8.3) 70 (58–83) 1 (0.84–1.2) 10 (8.3–12) 3 966 39 57 (48–68)
Botswana 1990 1 7.4 (2.9–14) 533 (212–997) 1.5 (0.58–2.7) 105 (42–196) 2 938 212 40 (21–100)
1995 2 14 (8.8–19) 855 (553–1 220) 7.2 (4.7–10) 456 (295–651) 5 665 358 42 (29–65)
2000 2 16 (13–20) 918 (736–1 120) 11 (9.0–14) 638 (511–777) 9 292 529 58 (47–72)
2005 2 14 (13–15) 733 (667–802) 9.3 (8.4–10) 494 (449–541) 10 058 536 73 (67–80)
2010 2 9.9 (8.8–11) 503 (449–560) 6.3 (5.6–7.0) 321 (286–357) 7 013 356 71 (64–79)
2011 2 9 (8.1–10) 455 (406–507) 5.7 (5.1–6.3) 285 (254–317) 6 603 332 73 (66–82)
2012 2 8.2 (7.3–9.1) 408 (364–454) 5.1 (4.5–5.6) 253 (226–281) 6 161 307 75 (68–84)
Burkina Faso 1990 9 7.6 (6.5–8.9) 87 (73–101) 2.9 (2.5–3.4) 33 (28–38) 1 497 17 20 (17–23)
1995 10 8.3 (7.0–9.6) 82 (70–95) 3.3 (2.8–3.8) 32 (27–37) 2 572 25 31 (27–37)
2000 12 8.2 (7.0–9.6) 71 (60–83) 2.8 (2.4–3.3) 25 (21–29) 2 331 20 28 (24–33)
2005 13 8.4 (7.1–9.8) 62 (53–73) 2.3 (2.0–2.7) 17 (15–20) 3 478 26 41 (36–49)
2010 16 9 (7.6–10) 58 (49–67) 1.8 (1.5–2.1) 11 (9.7–13) 4 800 31 54 (46–63)
2011 16 9.1 (7.7–11) 57 (48–66) 1.7 (1.4–1.9) 10 (8.9–12) 5 286 33 58 (50–68)
2012 16 9 (7.6–10) 54 (46–63) 1.6 (1.3–1.8) 9.5 (8.0–11) 5 210 32 58 (50–69)
Burundi 1990 6 9.1 (8.0–10) 162 (143–183) 1.6 (1.4–1.8) 28 (25–31) 4 575 82 50 (45–57)
1995 6 20 (18–23) 321 (283–362) 6.7 (5.9–7.5) 107 (94–121) 3 326 54 17 (15–19)
2000 7 19 (17–22) 288 (254–325) 7.4 (6.5–8.3) 110 (97–124) 6 421 96 33 (30–38)
2005 8 15 (14–17) 198 (174–223) 4.8 (4.3–5.5) 62 (55–70) 6 585 85 43 (38–49)
2010 9 13 (12–15) 144 (127–163) 3 (2.7–3.4) 33 (29–37) 7 611 82 57 (51–65)
2011 10 13 (12–15) 139 (122–157) 2.8 (2.4–3.1) 29 (26–33) 6 742 71 51 (45–58)
2012 10 13 (11–14) 130 (114–147) 2.5 (2.2–2.8) 25 (22–28) 6 921 70 54 (48–62)
Cameroon 1990 12 14 (11–16) 112 (92–133) 0.71 (0.58–0.84) 5.8 (4.8–7.0) 5 892 49 44 (37–53)
1995 14 29 (24–34) 206 (170–246) 5.5 (4.6–6.6) 40 (33–47) 3 292 24 11 (9.6–14)
2000 16 49 (41–59) 310 (255–369) 17 (14–20) 105 (87–126) 5 251 33 11 (8.9–13)
2005 18 57 (47–68) 312 (258–372) 22 (18–26) 121 (100–145) 21 499 119 38 (32–46)
2010 21 56 (47–67) 274 (226–327) 21 (18–25) 103 (85–123) 24 073 117 43 (36–52)
2011 21 51 (42–61) 243 (200–290) 19 (16–23) 91 (75–109) 24 533 116 48 (40–58)
2012 22 52 (43–61) 238 (197–283) 19 (16–23) 88 (73–104) 24 802 114 48 (40–58)
Cape Verde 1990 < 1 0.62 (0.380–0.910) 175 (108–259) 0.039 (0.024–0.058) 11 (6.8–16) 221 63 36 (24–58)
1995 < 1 0.67 (0.550–0.800) 168 (137–201) 0.057 (0.046–0.069) 14 (12–17) 303 76 45 (38–55)
2000 < 1 0.71 (0.580–0.850) 160 (131–193) 0.071 (0.057–0.085) 16 (13–19)
2005 < 1 0.73 (0.600–0.880) 153 (125–184) 0.074 (0.060–0.089) 15 (12–19) 292 61 40 (33–49)
2010 < 1 0.71 (0.590–0.850) 147 (121–175) 0.067 (0.055–0.081) 14 (11–17) 356 73 50 (42–60)
2011 < 1 0.71 (0.590–0.850) 145 (120–173) 0.068 (0.055–0.081) 14 (11–17) 380 77 53 (45–65)
2012 < 1 0.71 (0.590–0.850) 144 (119–172) 0.071 (0.058–0.086) 14 (12–17) 420 85 59 (49–72)
Central African 1990 3 25 (21–30) 861 (710–1 030) 9.8 (8.1–12) 336 (277–400) 2 124 73 8.5 (7.1–10)
Republic 1995 3 39 (32–47) 1 200 (988–1 430) 18 (15–21) 549 (453–654) 3 339 102 8.5 (7.1–10)
2000 4 39 (32–46) 1 070 (884–1 280) 18 (15–21) 492 (406–587)
2005 4 27 (23–33) 690 (569–822) 11 (9.4–14) 287 (237–342) 3 210 81 12 (9.9–14)
2010 4 19 (16–22) 433 (357–515) 6.3 (5.2–7.5) 145 (119–172) 6 643 153 35 (30–43)
2011 4 18 (15–21) 400 (330–477) 5.8 (4.8–6.9) 131 (108–156) 5 611 126 32 (27–38)
2012 5 17 (14–20) 367 (302–438) 5.3 (4.4–6.4) 118 (97–141) 8 084 179 49 (41–59)
Chad 1990 6 5.6 (4.7–6.7) 95 (78–112) 0.69 (0.57–0.83) 12 (9.7–14) 2 591 44 46 (39–56)
1995 7 9 (7.4–11) 128 (106–153) 1.8 (1.5–2.2) 26 (22–31) 3 186 46 36 (30–43)
2000 8 13 (10–15) 151 (125–180) 3.1 (2.5–3.6) 37 (31–44)
2005 10 15 (12–18) 150 (124–178) 3.7 (3.1–4.4) 37 (31–44) 6 311 63 42 (35–51)
2010 12 18 (15–21) 151 (125–179) 3.6 (2.9–4.2) 30 (25–36) 9 452 81 53 (45–65)
2011 12 18 (15–22) 151 (125–179) 3.8 (3.2–4.6) 32 (26–38) 10 505 87 58 (48–70)
2012 12 19 (16–22) 151 (125–180) 4.1 (3.4–4.8) 33 (27–39) 10 585 85 56 (47–68)
Comoros 1990 < 1 0.22 (0.180–0.270) 54 (44–64) 140 34 63 (53–76)
1995 < 1 0.21 (0.180–0.250) 46 (38–55) 123 26 58 (48–70)
2000 < 1 0.21 (0.170–0.250) 39 (32–46) 120 23 58 (49–71)
2005 < 1 0.22 (0.180–0.260) 36 (30–43) 111 18 51 (43–62)
2010 < 1 0.23 (0.190–0.270) 33 (28–40) <0.01 (<0.01–<0.01) 0.1 (<0.1–0.13)
2011 < 1 0.24 (0.200–0.290) 34 (28–41) <0.01 (<0.01–<0.01) 0.5 (0.45–0.65) 117 17 49 (41–59)
2012 < 1 0.25 (0.200–0.290) 34 (28–41) 0.01 (<0.01–0.012) 1.4 (1.2–1.7) 120 17 49 (41–59)
Congo 1990 2 4 (2.5–5.9) 169 (104–250) 0.94 (0.58–1.4) 40 (24–58) 591 25 15 (9.9–24)
1995 3 6.7 (5.4–8.0) 245 (200–294) 2.1 (1.7–2.5) 76 (62–91) 3 615 133 54 (45–66)
2000 3 11 (9.3–13) 353 (298–412) 3.2 (2.7–3.7) 102 (86–119) 9 239 296 84 (72–99)
2005 4 15 (12–18) 425 (347–510) 3.8 (3.1–4.6) 108 (88–130) 9 853 278 66 (55–80)
2010 4 16 (13–19) 391 (320–470) 3.4 (2.8–4.1) 83 (68–100) 10 150 247 63 (53–77)
2011 4 16 (13–20) 387 (317–465) 3.5 (2.8–4.2) 82 (67–98) 10 975 260 67 (56–82)
2012 4 17 (14–20) 381 (311–458) 3.6 (2.9–4.3) 83 (68–100) 11 303 261 68 (57–84)
Côte d'Ivoire 1990 12 29 (25–33) 238 (206–272) 8.3 (7.2–9.5) 68 (59–78) 7 841 65 27 (24–31)
1995 14 54 (47–62) 379 (329–433) 21 (19–24) 151 (131–172) 11 988 84 22 (19–26)
2000 16 60 (52–68) 369 (320–422) 25 (22–28) 154 (134–177) 15 094 94 25 (22–29)
2005 17 46 (40–53) 267 (232–306) 17 (15–19) 98 (85–112) 19 681 113 42 (37–49)
2010 19 36 (31–41) 190 (165–217) 9.4 (8.2–11) 50 (43–57) 22 708 120 63 (55–73)
2011 19 37 (32–42) 191 (165–218) 9.5 (8.3–11) 49 (43–56) 22 476 116 61 (53–70)
2012 20 34 (30–39) 172 (149–198) 8.8 (7.6–10) 44 (38–51) 23 762 120 69 (61–81)
Democratic 1990 35 110 (92–140) 327 (262–398) 8.1 (6.5–9.9) 23 (19–28) 21 131 61 19 (15–23)
Republic 1995 42 140 (110–160) 327 (268–392) 12 (9.7–14) 28 (23–34) 42 819 102 31 (26–38)
of the Congo 2000 47 150 (130–180) 327 (273–385) 14 (12–17) 30 (25–35) 61 024 130 40 (34–48)
2005 54 180 (150–200) 327 (279–379) 16 (14–19) 30 (25–34) 97 075 180 55 (47–64)
2010 62 200 (180–230) 327 (282–375) 16 (14–19) 26 (23–30) 114 170 184 56 (49–65)
2011 64 210 (180–240) 327 (282–375) 17 (14–19) 26 (22–30) 110 132 172 53 (46–61)
2012 66 210 (190–250) 327 (282–375) 16 (14–19) 25 (22–29) 108 984 166 51 (44–59)
Equatorial 1990 < 1 0.3 (0.260–0.340) 80 (70–91) 0.014 (0.012–0.016) 3.7 (3.2–4.2) 260 70 87 (77–99)
Guinea 1995 < 1 0.35 (0.310–0.400) 80 (70–90) 0.028 (0.024–0.032) 6.3 (5.5–7.1) 306 69 87 (77–99)
2000 < 1 0.52 (0.460–0.590) 101 (88–114) 0.062 (0.054–0.071) 12 (11–14)
2005 < 1 0.66 (0.580–0.750) 110 (96–124) 0.11 (0.095–0.12) 18 (16–20)
2010 < 1 0.94 (0.830–1.1) 135 (119–153) 0.18 (0.16–0.21) 26 (23–30) 820 118 87 (77–99)
2011 < 1 1 (0.890–1.1) 142 (124–161) 0.2 (0.18–0.23) 29 (25–32) 883 123 87 (77–99)
2012 < 1 1 (0.900–1.2) 139 (122–158) 0.2 (0.17–0.23) 27 (24–31)
Eritrea 1990 3 8 (5.3–11) 243 (162–341) 0.15 (0.098–0.21) 4.5 (3.0–6.3) 3 699 113 46 (33–70)
1995 3 6.7 (4.5–9.5) 198 (132–278) 0.4 (0.27–0.56) 12 (7.8–16) 21 453 630 320 (230–480)
2000 4 6.2 (4.1–8.7) 157 (104–220) 0.97 (0.64–1.4) 25 (16–34) 6 652 169 110 (77–160)
2005 5 5.9 (3.9–8.2) 121 (80–169) 1.1 (0.72–1.5) 22 (15–31) 3 585 74 61 (44–92)
2010 6 5.8 (3.8–8.1) 100 (67–140) 0.8 (0.53–1.1) 14 (9.3–20) 2 870 50 50 (36–75)
2011 6 5.7 (3.8–8.1) 97 (64–136) 0.79 (0.53–1.1) 13 (8.9–19) 3 049 51 53 (38–80)
2012 6 5.7 (3.8–8.0) 93 (62–131) 0.73 (0.49–1.0) 12 (7.9–17) 3 143 51 55 (39–83)
a
Rates are per 100 000 population.
b
NOTIFIED NEW AND RELAPSE includes cases for which the treatment history is unknown.
161 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
AFRICAN REGION
7$%/($,QFLGHQFHQRWLILFDWLRQDQGFDVHGHWHFWLRQUDWHVDOOIRUPV±
INCIDENCE (INCLUDING HIV) INCIDENCE HIV-POSITIVE NOTIFIED NEW AND RELAPSE
b
CASE DETECTION
YEAR POPULATION
(MILLIONS)
NUMBER
(THOUSANDS) RATE
a
NUMBER
(THOUSANDS) RATE
a
NUMBER RATE
a
PERCENT
Ethiopia 1990 48 180 (100–270) 367 (218–553) 11 (6.3–16) 22 (13–33) 88 634 184 50 (33–85)
1995 57 240 (140–360) 419 (249–633) 36 (22–55) 64 (38–97) 26 034 46 11 (7.2–18)
2000 66 280 (170–420) 421 (251–636) 61 (36–92) 93 (55–140) 91 101 138 33 (22–55)
2005 76 260 (150–390) 342 (203–516) 54 (32–81) 71 (42–107) 124 262 163 48 (32–80)
2010 87 230 (170–310) 269 (191–359) 30 (21–40) 35 (25–46) 154 694 178 66 (49–93)
2011 89 230 (170–300) 258 (191–335) 27 (20–35) 30 (22–39) 156 539 175 68 (52–91)
2012 92 230 (170–290) 247 (183–321) 23 (17–30) 25 (19–33) 145 323 158 64 (49–87)
Gabon 1990 < 1 2.1 (1.7–2.5) 221 (182–263) 0.12 (0.096–0.14) 12 (10–15) 917 97 44 (37–53)
1995 1 3.4 (2.8–4.1) 315 (260–375) 0.47 (0.39–0.56) 44 (36–52) 1 115 103 33 (28–40)
2000 1 6.5 (5.3–7.7) 527 (435–627) 1.6 (1.3–1.9) 132 (108–157)
2005 1 8.1 (6.7–9.6) 586 (484–698) 2.5 (2.0–2.9) 178 (146–212) 2 512 182 31 (26–38)
2010 2 7.4 (6.1–8.8) 475 (392–566) 2.1 (1.7–2.5) 136 (112–162) 3 790 244 51 (43–62)
2011 2 7.2 (5.9–8.6) 450 (372–536) 2 (1.6–2.4) 125 (103–149) 4 404 276 61 (52–74)
2012 2 7 (5.8–8.3) 428 (354–510) 1.9 (1.5–2.2) 115 (95–137) 4 929 302 71 (59–85)
Gambia 1990 < 1 1.7 (1.0–2.5) 185 (114–273) 0.023 (0.014–0.033) 2.5 (1.5–3.6)
1995 1 2.2 (1.8–2.6) 204 (167–245) 0.071 (0.058–0.085) 6.7 (5.5–8.0) 1 023 96 47 (39–57)
2000 1 2.8 (2.3–3.3) 225 (184–271) 0.2 (0.16–0.24) 16 (13–19) 1 553 126 56 (47–69)
2005 1 3.6 (2.9–4.3) 248 (203–298) 0.47 (0.38–0.56) 33 (27–39) 2 031 141 57 (47–70)
2010 2 4.6 (3.8–5.5) 273 (226–325) 0.78 (0.65–0.93) 47 (39–56) 1 989 118 43 (36–52)
2011 2 4.8 (4.0–5.7) 279 (230–331) 0.77 (0.64–0.91) 44 (37–53) 2 302 133 48 (40–58)
2012 2 5.1 (4.2–6.0) 284 (234–337) 0.76 (0.63–0.90) 42 (35–50) 2 333 130 46 (39–56)
Ghana 1990 15 23 (10–40) 155 (69–275) 1.7 (0.74–2.9) 11 (5.0–20) 6 407 44 28 (16–63)
1995 17 28 (16–44) 167 (93–263) 4.2 (2.3–6.6) 25 (14–39) 8 636 52 31 (20–55)
2000 19 29 (18–41) 152 (97–220) 5.9 (3.8–8.6) 31 (20–46) 10 933 58 38 (26–60)
2005 21 25 (19–33) 119 (88–154) 5.4 (4.0–7.0) 25 (19–33) 12 124 57 48 (37–64)
2010 24 21 (18–24) 86 (75–97) 3.7 (3.2–4.2) 15 (13–17) 14 607 60 70 (62–80)
2011 25 20 (17–22) 79 (69–89) 3.3 (2.9–3.7) 13 (12–15) 15 389 62 79 (70–90)
2012 25 18 (16–21) 72 (63–82) 2.8 (2.4–3.1) 11 (9.6–12) 14 753 58 81 (71–92)
Guinea 1990 6 15 (12–18) 248 (204–295) 1.1 (0.90–1.3) 18 (15–22) 1 988 33 13 (11–16)
1995 8 20 (16–23) 249 (205–297) 2.8 (2.3–3.3) 35 (29–42) 3 523 45 18 (15–22)
2000 9 20 (17–24) 234 (193–279) 4.1 (3.3–4.8) 46 (38–55) 5 440 62 27 (22–32)
2005 10 20 (17–24) 211 (173–251) 4.5 (3.7–5.4) 47 (39–56) 6 863 72 34 (29–41)
2010 11 20 (17–24) 188 (155–224) 3.8 (3.2–4.6) 35 (29–42) 11 038 101 54 (45–66)
2011 11 20 (17–24) 183 (151–219) 3.7 (3.1–4.5) 33 (28–40) 11 359 102 55 (47–67)
2012 11 20 (17–24) 178 (146–213) 3.7 (3.0–4.4) 32 (27–39) 11 407 100 56 (47–68)
Guinea-Bissau 1990 1 1.6 (1.1–2.2) 158 (108–217) 0.08 (0.054–0.11) 7.8 (5.3–11) 1 163 114 73 (53–110)
1995 1 2 (1.6–2.4) 174 (142–209) 0.23 (0.19–0.27) 20 (16–24) 1 613 142 81 (68–100)
2000 1 2.4 (2.0–2.9) 192 (157–230) 0.55 (0.45–0.66) 43 (35–52) 1 273 100 52 (43–64)
2005 1 3 (2.5–3.6) 211 (173–254) 1 (0.84–1.2) 73 (59–87) 1 774 125 59 (49–72)
2010 2 3.7 (3.0–4.4) 233 (192–278) 1.3 (1.1–1.5) 81 (67–97) 2 183 138 59 (50–72)
2011 2 3.9 (3.2–4.6) 238 (196–283) 1.4 (1.2–1.7) 88 (73–105) 2 063 127 53 (45–65)
2012 2 4 (3.3–4.8) 242 (200–289) 1.6 (1.3–1.9) 94 (77–112) 1 939 117 48 (40–58)
Kenya 1990 23 33 (28–37) 139 (121–159) 3.6 (3.1–4.1) 15 (13–17) 11 788 50 36 (32–41)
1995 27 46 (43–50) 169 (155–184) 19 (18–21) 70 (64–76) 28 142 103 61 (56–66)
2000 31 89 (84–95) 286 (267–305) 47 (44–50) 151 (141–161) 64 159 205 72 (67–77)
2005 36 130 (120–140) 359 (339–380) 64 (60–67) 177 (168–188) 102 680 287 80 (76–85)
2010 41 120 (120–130) 298 (286–311) 51 (49–53) 124 (119–129) 99 272 243 81 (78–85)
2011 42 120 (120–130) 288 (276–300) 49 (47–51) 116 (111–121) 97 320 232 80 (77–84)
2012 43 120 (110–120) 272 (261–283) 45 (44–47) 105 (101–109) 92 987 215 79 (76–83)
Lesotho 1990 2 2.9 (2.2–3.8) 184 (135–240) 0.083 (0.061–0.11) 5.2 (3.8–6.8) 2 525 158 86 (66–120)
1995 2 5.7 (5.0–6.4) 323 (283–367) 2.2 (1.9–2.5) 125 (109–142) 5 181 295 91 (81–100)
2000 2 10 (9.0–12) 553 (484–626) 7.6 (6.6–8.6) 408 (357–462) 9 746 525 95 (84–110)
2005 2 12 (10–14) 639 (535–752) 10 (8.3–12) 517 (433–608) 10 802 561 88 (75–100)
2010 2 13 (11–14) 633 (553–719) 9.7 (8.5–11) 483 (422–549) 11 674 581 92 (81–110)
2011 2 13 (11–15) 632 (551–717) 9.5 (8.3–11) 467 (408–531) 11 561 570 90 (79–100)
2012 2 13 (11–15) 630 (550–716) 9.9 (8.7–11) 485 (423–550) 10 776 525 83 (73–95)
Liberia 1990 2 4.2 (2.6–6.2) 199 (123–293) 0.095 (0.058–0.14) 4.5 (2.8–6.7)
1995 2 4.6 (3.7–5.5) 219 (179–263) 0.31 (0.25–0.38) 15 (12–18) 1 393 67 31 (25–37)
2000 3 7 (5.7–8.4) 242 (197–290) 0.8 (0.65–0.98) 28 (22–34) 1 500 52 21 (18–26)
2005 3 8.7 (7.1–10) 266 (218–320) 1 (0.83–1.3) 32 (25–38) 3 432 105 39 (33–48)
2010 4 12 (9.6–14) 293 (242–349) 0.91 (0.73–1.1) 23 (18–28) 6 597 167 57 (48–69)
2011 4 12 (10–15) 299 (247–356) 0.83 (0.67–1.0) 20 (16–25) 7 906 194 65 (54–79)
2012 4 13 (11–15) 304 (251–362) 0.76 (0.61–0.93) 18 (15–22) 8 093 193 64 (53–77)
Madagascar 1990 12 45 (37–54) 391 (322–466) 0.34 (0.28–0.40) 2.9 (2.4–3.5) 6 261 54 14 (12–17)
1995 13 45 (37–54) 335 (276–400) 0.55 (0.46–0.66) 4.1 (3.4–4.9) 21 616 161 48 (40–58)
2000 16 46 (38–55) 293 (241–349) 0.73 (0.60–0.87) 4.6 (3.8–5.5)
2005 18 48 (39–57) 262 (216–313) 0.77 (0.64–0.92) 4.2 (3.5–5.0) 18 993 104 40 (33–48)
2010 21 51 (42–61) 242 (199–288) 0.7 (0.57–0.83) 3.3 (2.7–3.9) 24 432 116 48 (40–58)
2011 22 52 (43–62) 238 (196–284) 0.67 (0.55–0.80) 3.1 (2.6–3.7) 26 019 120 50 (42–61)
2012 22 52 (43–62) 234 (193–280) 0.64 (0.53–0.77) 2.9 (2.4–3.4) 25 782 116 49 (41–60)
Malawi 1990 9 31 (22–41) 326 (230–438) 13 (9.5–18) 143 (101–192) 12 395 131 40 (30–57)
1995 10 46 (38–55) 462 (383–548) 29 (24–34) 291 (241–345) 19 155 192 42 (35–50)
2000 11 53 (44–63) 467 (387–554) 37 (31–44) 329 (273–391) 23 604 208 45 (38–54)
2005 13 46 (38–54) 354 (292–421) 33 (27–39) 256 (211–305) 25 491 197 56 (47–68)
2010 15 33 (31–35) 219 (203–236) 21 (20–23) 142 (132–153) 21 092 140 64 (59–69)
2011 15 30 (27–32) 191 (177–206) 19 (18–21) 125 (116–135) 19 361 125 66 (61–71)
2012 16 26 (24–28) 163 (151–176) 16 (15–17) 100 (93–108) 20 335 128 78 (73–85)
Mali 1990 8 6 (5.8–6.3) 76 (72–80) 0.59 (0.56–0.62) 7.4 (7.0–7.8) 2 933 37 49 (46–51)
1995 9 7.2 (6.9–7.6) 80 (76–84) 1.2 (1.2–1.3) 14 (13–14) 3 087 34 43 (41–45)
2000 10 7.9 (7.6–8.3) 77 (74–81) 1.6 (1.6–1.7) 16 (15–17) 4 216 41 53 (51–56)
2005 12 8.3 (7.9–8.7) 69 (66–73) 1.6 (1.5–1.7) 13 (13–14) 4 704 39 57 (54–60)
2010 14 8.8 (8.4–9.2) 63 (60–66) 1.3 (1.3–1.4) 9.4 (9.0–9.9) 5 291 38 60 (57–63)
2011 14 8.9 (8.5–9.3) 62 (59–65) 1.3 (1.2–1.4) 8.9 (8.5–9.4) 5 428 38 61 (58–64)
2012 15 9 (8.5–9.4) 60 (57–63) 1.2 (1.2–1.3) 8.2 (7.8–8.6) 5 446 37 61 (58–64)
Mauritania 1990 2 4.6 (2.8–6.8) 228 (140–336) 0.05 (0.031–0.074) 2.5 (1.5–3.7) 5 284 261 110 (78–190)
1995 2 5.9 (4.8–7.0) 251 (205–302) 0.1 (0.083–0.12) 4.4 (3.6–5.2) 3 849 165 66 (55–80)
2000 3 7.5 (6.1–9.0) 277 (226–333) 0.17 (0.14–0.20) 6.3 (5.1–7.5) 3 067 113 41 (34–50)
2005 3 9.6 (7.9–12) 305 (250–367) 0.27 (0.22–0.32) 8.4 (6.9–10) 2 162 69 22 (19–28)
2010 4 12 (10–15) 337 (277–402) 0.44 (0.36–0.53) 12 (10–15) 2 461 68 20 (17–25)
2011 4 13 (10–15) 344 (283–410) 0.5 (0.41–0.60) 14 (11–16) 1 804 49 14 (12–17)
2012 4 13 (11–16) 350 (288–418) 0.57 (0.47–0.68) 15 (12–18) 2 616 69 20 (16–24)
Mauritius 1990 1 0.29 (0.180–0.430) 28 (17–41) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 119 11 41 (28–66)
1995 1 0.29 (0.240–0.350) 26 (21–31) <0.01 (<0.01–<0.01) 0.3 (0.24–0.35) 131 12 45 (37–55)
2000 1 0.29 (0.240–0.350) 24 (20–29) 0.011 (<0.01–0.013) 0.9 (0.74–1.1) 160 14 55 (46–68)
2005 1 0.28 (0.230–0.330) 23 (19–28) 0.017 (0.014–0.021) 1.4 (1.2–1.7) 125 10 45 (37–55)
2010 1 0.27 (0.220–0.320) 22 (18–26) 0.016 (0.013–0.020) 1.3 (1.1–1.6) 122 9.9 46 (39–56)
2011 1 0.26 (0.220–0.310) 21 (18–25) 0.015 (0.013–0.018) 1.2 (1.0–1.5) 114 9.2 43 (36–53)
2012 1 0.26 (0.210–0.310) 21 (17–25) 0.014 (0.011–0.017) 1.1 (0.92–1.3) 128 10 49 (41–60)
Mozambique 1990 14 54 (8.5–140) 401 (62–1 050) 0.84 (0.13–2.2) 6.2 (0.97–16) 15 899 117 29 (11–190)
1995 16 76 (24–160) 478 (153–985) 15 (4.8–31) 94 (30–194) 17 882 112 23 (11–73)
2000 18 94 (41–170) 513 (227–914) 41 (18–74) 227 (100–404) 21 158 116 23 (13–51)
2005 21 110 (63–170) 524 (298–811) 61 (35–94) 290 (165–449) 33 231 158 30 (20–53)
2010 24 130 (90–180) 544 (377–741) 78 (54–110) 327 (227–446) 43 558 182 33 (25–48)
2011 25 130 (93–180) 548 (380–747) 81 (56–110) 331 (229–451) 44 627 182 33 (24–48)
2012 25 140 (96–190) 552 (383–753) 83 (58–110) 330 (228–450) 47 741 189 34 (25–50)
Namibia 1990 1 5.4 (4.3–6.6) 379 (300–468) 0.48 (0.38–0.59) 34 (27–41) 2 671 189 50 (40–63)
1995 2 9.5 (7.5–12) 575 (456–709) 2.8 (2.2–3.4) 168 (133–207) 1 540 93 16 (13–20)
2000 2 27 (21–33) 1 410 (1 110–1 730) 15 (12–19) 798 (631–983) 10 799 569 40 (33–51)
2005 2 28 (22–35) 1 390 (1 100–1 720) 19 (15–23) 932 (738–1 150
)
14 920 736 53 (43–67)
2010 2 19 (15–23) 867 (686–1 070) 11 (8.3–13) 483 (383–596) 11 281 518 60 (48–75)
2011 2 16 (13–20) 723 (573–891) 8.7 (6.9–11) 391 (310–482) 10 806 487 67 (55–85)
2012 2 15 (12–18) 655 (524–800) 7.3 (5.8–8.9) 323 (258–394) 10 003 443 68 (55–84)
a
Rates are per 100 000 population.
b
NOTIFIED NEW AND RELAPSE includes cases for which the treatment history is unknown.
162 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($,QFLGHQFHQRWLILFDWLRQDQGFDVHGHWHFWLRQUDWHVDOOIRUPV±
INCIDENCE (INCLUDING HIV) INCIDENCE HIV-POSITIVE NOTIFIED NEW AND RELAPSE
b
CASE DETECTION
YEAR POPULATION
(MILLIONS)
NUMBER
(THOUSANDS) RATE
a
NUMBER
(THOUSANDS) RATE
a
NUMBER RATE
a
PERCENT
Niger 1990 8 28 (23–33) 358 (295–426) 1.1 (0.91–1.3) 14 (12–17) 5 200 67 19 (16–23)
1995 9 25 (20–29) 270 (223–321) 1.7 (1.4–2.1) 19 (16–22) 1 980 22 8 (6.7–9.7)
2000 11 21 (17–25) 191 (157–227) 2.1 (1.7–2.5) 19 (16–23) 4 701 43 22 (19–27)
2005 13 19 (15–22) 142 (118–170) 2.2 (1.8–2.6) 16 (14–20) 7 873 60 42 (35–51)
2010 16 18 (15–21) 113 (94–135) 1.9 (1.6–2.3) 12 (9.8–14) 10 130 64 56 (47–68)
2011 17 18 (15–21) 108 (90–129) 1.9 (1.6–2.3) 11 (9.5–14) 10 510 64 59 (49–71)
2012 17 18 (15–21) 104 (86–124) 1.9 (1.5–2.2) 11 (8.9–13) 10 989 64 62 (52–75)
Nigeria 1990 96 120 (1.3–500) 128 (1.3–526) 2.2 (0.023–9.1) 2.3 (<0.1–9.5) 20 122 21 16 (4.0–1 600)
1995 108 150 (8.0–490) 139 (7.4–456) 16 (0.84–52) 15 (0.78–48) 13 423 12 8.9 (2.7–170)
2000 123 210 (15–660) 172 (12–536) 46 (3.3–140) 38 (2.7–118) 25 821 21 12 (3.9–170)
2005 140 240 (33–660) 175 (23–476) 64 (8.6–170) 46 (6.2–125) 63 990 46 26 (9.6–200)
2010 160 210 (100–360) 133 (64–225) 53 (26–91) 33 (16–57) 84 121 53 40 (23–82)
2011 164 190 (91–340) 118 (55–204) 49 (23–85) 30 (14–52) 86 778 53 45 (26–95)
2012 169 180 (85–310) 108 (50–186) 46 (21–80) 27 (13–47) 92 818 55 51 (29–110)
Rwanda 1990 7 21 (19–23) 290 (259–323) 11 (9.5–12) 148 (132–165) 6 387 89 30 (27–34)
1995 6 29 (26–32) 513 (458–571) 15 (13–16) 260 (232–290) 3 054 54 11 (9.4–12)
2000 8 27 (24–30) 325 (290–362) 13 (12–15) 157 (140–175) 6 093 73 22 (20–25)
2005 9 17 (15–19) 181 (162–202) 7.5 (6.7–8.3) 79 (71–88) 7 220 77 42 (38–47)
2010 11 11 (10–13) 106 (94–118) 3.7 (3.3–4.1) 34 (31–38) 6 703 62 59 (53–66)
2011 11 11 (9.4–12) 94 (84–105) 3.3 (3.0–3.7) 30 (26–33) 6 623 59 63 (57–71)
2012 11 9.8 (8.8–11) 86 (77–96) 2.9 (2.6–3.2) 25 (22–28) 6 091 53 62 (56–69)
Sao Tome and 1990 < 1 0.16 (0.098–0.230) 135 (83–199) <0.01 (<0.01–<0.01) 2 (1.2–2.9) 17 14 11 (7.3–17)
Principe 1995 < 1 0.16 (0.130–0.190) 124 (102–149) <0.01 (<0.01–<0.01) 3.8 (3.1–4.6)
2000 < 1 0.16 (0.130–0.190) 114 (93–137) <0.01 (<0.01–0.010) 6.2 (5.0–7.4) 97 70 61 (51–75)
2005 < 1 0.16 (0.140–0.190) 105 (88–123) 0.015 (0.012–0.017) 9.6 (8.0–11) 136 88 84 (72–100)
2010 < 1 0.17 (0.140–0.200) 96 (79–115) 0.018 (0.015–0.021) 9.9 (8.1–12) 121 68 71 (59–86)
2011 < 1 0.17 (0.140–0.210) 94 (78–113) 0.018 (0.014–0.021) 9.6 (7.9–11) 136 74 79 (66–96)
2012 < 1 0.17 (0.140–0.210) 93 (76–111) 0.017 (0.014–0.021) 9.2 (7.5–11) 115 61 66 (55–80)
Senegal 1990 8 10 (8.5–12) 138 (114–164) 0.17 (0.14–0.20) 2.2 (1.8–2.6) 4 977 66 48 (40–58)
1995 9 13 (11–16) 153 (126–183) 0.4 (0.33–0.48) 4.6 (3.8–5.5) 7 561 87 57 (47–69)
2000 10 15 (13–18) 155 (128–184) 0.8 (0.66–0.96) 8.1 (6.7–9.7) 8 508 86 56 (47–68)
2005 11 16 (13–19) 142 (117–169) 1.2 (1.0–1.4) 11 (8.9–13) 9 765 87 61 (51–74)
2010 13 18 (15–21) 137 (113–163) 1.5 (1.3–1.8) 12 (9.7–14) 11 061 85 63 (52–76)
2011 13 18 (15–22) 136 (112–162) 1.6 (1.3–1.9) 12 (9.6–14) 11 022 83 61 (51–74)
2012 14 19 (16–22) 137 (113–163) 1.7 (1.4–2.0) 12 (9.9–14) 12 265 89 65 (55–79)
Seychelles 1990 < 1 0.03 (0.019–0.044) 43 (27–64) 41 59 140 (92–220)
1995 < 1 0.03 (0.025–0.036) 40 (33–48) 8 11 27 (22–33)
2000 < 1 0.029 (0.024–0.035) 37 (30–44) 20 25 69 (57–84)
2005 < 1 0.029 (0.024–0.035) 33 (27–40) 14 16 48 (40–59)
2010 < 1 0.028 (0.023–0.033) 31 (25–37) <0.01 (<0.01–<0.01) 1.8 (<0.1–8.0) 17 19 61 (51–74)
2011 < 1 0.028 (0.023–0.033) 30 (25–36) <0.01 (<0.01–0.011) 5.8 (1.6–12) 21 23 76 (64–92)
2012 < 1 0.027 (0.023–0.033) 30 (24–35) <0.01 (<0.01–<0.01) 2.7 (<0.1–10) 20 22 73 (61–88)
Sierra Leone 1990 4 8.4 (5.2–12) 207 (128–305) 0.011 (<0.01–0.016) 0.3 (0.17–0.40) 632 16 7.5 (5.1–12)
1995 4 8.3 (6.4–11) 212 (162–269) 0.087 (0.067–0.11) 2.2 (1.7–2.8) 1 955 50 23 (19–31)
2000 4 11 (8.1–14) 264 (196–341) 0.45 (0.33–0.58) 11 (8.0–14) 3 760 91 34 (27–46)
2005 5 26 (21–31) 503 (410–605) 2.3 (1.9–2.7) 44 (36–53) 6 737 132 26 (22–32)
2010 6 38 (31–45) 660 (540–791) 4.2 (3.4–5.0) 73 (60–87) 12 859 224 34 (28–41)
2011 6 39 (32–47) 668 (542–807) 4.3 (3.5–5.2) 73 (59–88) 12 734 217 32 (27–40)
2012 6 40 (32–49) 674 (540–821) 3.9 (3.2–4.8) 66 (53–81) 13 074 219 32 (27–40)
South Africa 1990 37 110 (76–150) 301 (206–413) 2.5 (1.7–3.4) 6.7 (4.6–9.2) 80 400 219 73 (53–110)
1995 41 130 (110–160) 317 (259–381) 25 (21–30) 61 (50–73) 73 917 178 56 (47–69)
2000 45 260 (210–310) 576 (471–691) 140 (110–170) 311 (254–374) 151 239 337 59 (49–72)
2005 48 450 (360–540) 925 (756–1 110) 300 (250–360) 622 (508–746) 270 178 560 61 (50–74)
2010 51 500 (420–600) 981 (809–1 170) 330 (270–390) 640 (528–763) 354 786 690 70 (59–85)
2011 52 520 (430–610) 993 (819–1 180) 330 (270–390) 635 (524–756) 362 453 698 70 (59–85)
2012 52 530 (430–630) 1 000 (827–1 190) 330 (270–390) 631 (521–752) 323 664 618 62 (52–75)
Swaziland 1990 < 1 2.3 (1.4–3.4) 267 (165–394) 0.38 (0.23–0.56) 44 (27–64)
1995 < 1 3.2 (2.7–3.9) 337 (275–405) 1.6 (1.3–1.9) 161 (132–194) 2 050 213 63 (53–77)
2000 1 8.5 (7.0–10) 803 (657–964) 6.3 (5.2–7.6) 595 (486–714) 5 877 552 69 (57–84)
2005 1 13 (10–15) 1 150 (938–1 380) 11 (8.7–13) 962 (787–1 150
)
8 705 788 69 (57–84)
2010 1 15 (13–18) 1 290 (1 060–1 530) 13 (11–15)
1
070 (882–1 270
)
10 101 847 66 (55–80)
2011 1 16 (13–19) 1 320 (1 090–1 570) 12 (10–15)
1
020 (844–1 220
)
8 337 688 52 (44–63)
2012 1 17 (14–20) 1 350 (1 110–1 610) 13 (11–15)
1
040 (856–1 240
)
7 165 582 43 (36–52)
Togo 1990 4 1.8 (1.5–2.1) 47 (39–56) 0.18 (0.15–0.21) 4.7 (3.8–5.6) 1 324 35 74 (62–90)
1995 4 2.5 (2.0–3.0) 58 (48–69) 0.44 (0.37–0.53) 10 (8.5–12) 1 520 35 61 (51–74)
2000 5 3.5 (2.9–4.2) 72 (59–86) 0.89 (0.73–1.1) 18 (15–22) 1 409 29 40 (34–49)
2005 6 4.2 (3.5–5.1) 77 (63–91) 1.2 (1.0–1.5) 22 (18–26) 2 541 46 60 (50–73)
2010 6 4.6 (3.8–5.5) 73 (60–87) 1.2 (0.99–1.4) 19 (16–23) 2 791 44 61 (51–74)
2011 6 4.7 (3.9–5.6) 73 (60–87) 1.2 (0.97–1.4) 18 (15–22) 2 888 45 61 (51–74)
2012 7 4.9 (4.0–5.8) 73 (60–87) 1.2 (0.98–1.4) 18 (15–21) 2 843 43 59 (49–71)
Uganda 1990 18 110 (57–180) 624 (328–1 010) 79 (41–130) 449 (236–729) 14 740 84 13 (8.3–26)
1995 21 110 (62–180) 542 (297–860) 81 (44–130) 390 (214–619) 25 316 122 23 (14–41)
2000 24 100 (63–150) 427 (259–636) 68 (41–100) 280 (170–417) 30 372 125 29 (20–48)
2005 29 87 (63–120) 304 (220–402) 50 (36–66) 173 (125–228) 41 040 143 47 (36–65)
2010 34 71 (57–86) 209 (169–253) 38 (31–46) 113 (91–137) 42 885 126 60 (50–75)
2011 35 68 (55–82) 193 (156–234) 36 (29–44) 102 (83–124) 46 306 132 68 (56–84)
2012 36 65 (53–79) 179 (145–216) 35 (28–42) 95 (77–115) 44 663 123 69 (57–85)
United Republic 1990 25 58 (49–67) 226 (193–261) 14 (12–16) 56 (47–64) 22 249 87 39 (33–45)
of Tanzania 1995 30 68 (58–78) 226 (193–261) 31 (26–36) 103 (88–119) 39 847 133 59 (51–69)
2000 34 80 (70–91) 236 (207–268) 41 (36–46) 121 (106–137) 54 442 160 68 (60–77)
2005 39 83 (76–89) 213 (197–229) 40 (37–43) 104 (96–112) 61 022 157 74 (69–80)
2010 45 80 (75–85) 177 (166–189) 33 (31–35) 72 (68–77) 61 098 136 77 (72–82)
2011 46 78 (74–83) 169 (159–180) 31 (29–33) 68 (63–72) 59 357 128 76 (71–81)
2012 48 79 (74–84) 165 (154–175) 32 (30–34) 68 (64–72) 62 178 130 79 (74–84)
Zambia 1990 8 56 (49–63) 710 (624–801) 35 (31–40) 449 (395–507) 16 863 215 30 (27–34)
1995 9 70 (64–76) 788 (719–861) 49 (45–54) 559 (510–611) 35 958 407 52 (47–57)
2000 10 72 (67–77) 713 (661–767) 52 (48–56) 513 (475–551) 49 806 493 69 (64–75)
2005 11 65 (59–71) 566 (519–615) 47 (43–51) 409 (375–445) 49 576 432 76 (70–83)
2010 13 61 (55–67) 462 (418–509) 39 (35–43) 292 (264–322) 44 154 334 72 (66–80)
2011 14 61 (55–67) 444 (401–489) 38 (34–42) 277 (250–305) 43 583 320 72 (65–80)
2012 14 60 (54–66) 427 (385–470) 35 (32–39) 251 (226–276) 40 726 289 68 (62–75)
Zimbabwe 1990 10 31 (17–50) 296 (159–476) 18 (9.5–29) 170 (91–273) 9 132 87 29 (18–55)
1995 12 56 (39–77) 483 (335–658) 46 (32–62) 392 (272–535) 30 831 265 55 (40–79)
2000 13 91 (72–110) 726 (573–897) 79 (62–97) 629 (496–777) 50 855 407 56 (45–71)
2005 13 100 (81–130) 799 (634–984) 83 (66–100) 657 (521–809) 50 454 397 50 (40–63)
2010 13 83 (64–100) 633 (489–795) 63 (48–79) 480 (371–603) 44 209 338 53 (43–69)
2011 13 81 (62–100) 603 (466–757) 58 (45–73) 433 (335–543) 38 404 287 48 (38–62)
2012 14 77 (60–97) 562 (434–706) 55 (42–69) 399 (308–501) 35 760 261 46 (37–60)
a
Rates are per 100 000 population.
b
NOTIFIED NEW AND RELAPSE includes cases for which the treatment history is unknown.

163 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
AFRICAN REGION
7$%/($&DVHQRWLILFDWLRQV±
NEW CASES
YEAR SMEAR-
POSITIVE
SMEAR-NEGATIVE/
UNKNOWN
EXTRA-
PULMONARY OTHER RELAPSE RE-TREAT EXCL.
RELAPSE
TOTAL
RETREAT
HISTORY
UNKNOWN
Al
g
eria 1990 11 607
–
1995 13 507 5 735 2 256 5 065 451 451 72
2000 18 572 8 328 2 019 7 758 467 80 547 80
2005 21 336 8 654 1 651 10 216 267 548 165 713 84
2010 22 336 8 299 1 770 11 770 0 497 194 691 0 82
2011 21 429 7 790 1 753 11 444 0 442 168 610 0 82
• 44 57 • 2012 21 880 7 510 1 702 12 294 0 374 202 576 0 82
An
g
ola 1990 10 271
–
1995 5 143 3 804 1 631 266 134 134 70
2000 16 062 9 053 5 367 1 102 540 540 63
2005 37 175 20 410 12 467 2 569 1 729 1 142 2 871 62
2010 44 655 21 146 17 285 3 780 2 444 5 332 7 776 55
2011 47 240 21 703 18 380 4 399 0 2 758 1 686 4 444 0 54
• 99 249 • 2012 51 819 21 124 23 056 4 776 0 2 863 1 607 4 470 48
Benin 1990 2 074 1 410 310 182 172 49 221 82
1995 2 400 1 839 281 212 68 68 87
2000 2 697 2 277 130 199 91 189 280 95
2005 3 270 2 739 96 285 150 187 337 97
2010 3 756 2 973 296 367 0 120 85 205 0 91
2011 4 212 3 331 329 398 0 154 108 262 0 91
• 41 39 • 2012 3 966 3 171 305 316 0 174 109 283 0 91
Botswana 1990 2 938
–
1995 5 665 1 903 2 885 720 147 147 40
2000 9 292 3 091 4 789 1 231 181 1 058 1 239 39
2005 10 058 3 170 5 166 1 220 502 46 548 38
2010 7 013 3 295 2 055 1 210 0 453 619 1 072 0 62
2011 6 603 2 669 1 983 1 213 0 738 130 868 0 57
• 212 307 • 2012 6 161 2 426 2 208 1 151 0 376 62 438 0 52
Burkina Faso 1990 1 497
–
1995 2 572 1 028 195 195 45 45 84
2000 2 331 1 545 196 502 88 90 178 0 89
2005 3 478 2 290 367 571 90 160 167 327 0 86
2010 4 800 3 041 736 729 77 217 335 552 0 81
2011 5 286 3 450 692 742 175 227 257 484 0 83
• 17 32 • 2012 5 210 3 583 662 617 154 194 195 389 0 84
Burundi 1990 4 575
–
1995 3 326 1 121 908 1 116 181 181 55
2000 6 421 3 159 1 489 1 568 0 205 20 225 0 68
2005 6 585 3 262 1 160 2 089 0 74 42 116 0 74
2010 7 611 4 590 963 1 826 8 224 108 332 0 83
2011 6 742 4 060 799 1 649 5 229 86 315 0 84
• 82 70 • 2012 6 921 4 075 746 1 887 3 210 95 305 0 85
Cameroon 1990 5 892
–
1995 3 292 2 896 142 18 236 236 95
2000 5 251 3 960 625 415 251 251 86
2005 21 499 13 001 5 021 2 461 0 1 016 574 1 590 0 72
2010 24 073 14 464 5 437 3 157 0 1 015 479 1 494 0 73
2011 24 533 14 927 4 941 3 597 0 1 068 593 1 661 0 75
• 49 114 • 2012 24 802 15 016 5 204 3 524 0 1 058 558 1 616 0 74
Ca
p
e Verde 1990 221
–
1995 303 111 150 12 30 30 43
2000
–
2005 292 135 93 43 0 21 13 34 0 59
2010 356 186 98 54 18 9 27 65
2011 380 182 127 54 0 17 10 27 0 59
• 63 85 • 2012 420 189 151 66 0 14 5 19 0 56
Central African 1990 2 124
–
Re
p
ublic 1995 3 339 1 794 964 393 188 188 65
2000
–
2005 3 210 2 153 608 286 0 163 128 291 0 78
2010 6 643 3 638 1 598 1 079 24 304 117 421 0 69
2011 5 611 3 479 964 876 60 232 113 345 0 78
• 73 179 • 2012 8 084 4 641 1 752 1 356 335 199 534 73
Chad 1990 2 591
–
1995 3 186 2 002 518 463 203 203 79
2000
–
2005 6 311 2 516 2 419 1 055 321 194 515 51
2010 9 452 3 833 3 746 1 217 193 463 245 708 0 51
2011 10 505 4 434 4 211 1 033 249 578 269 847 0 51
• 44 85 • 2012 10 585 3 849 4 809 1 113 180 634 215 849 0 44
Comoros 1990 140
–
1995 123 103 10 7 7 7 91
2000 120 87 14 15 4 1 5 86
2005 111 79 14 16 0 2 1 3 0 85
2010
–
2011 117 62 13 28 5 9 2 11 0 83
• 34 17 • 2012 120 71 24 23 2 2 4 75
Con
g
o 1990 591
–
1995 3 615 2 013 849 675 78 78 70
2000 9 239 4 218 2 016 2 810 169 650 819 68
2005 9 853 3 640 3 249 2 665 299 108 407 53
2010 10 150 3 568 3 545 2 692 345 171 516 50
2011 10 975 3 716 3 930 2 990 0 339 168 507 0 49
• 25 261 • 2012 11 303 3 984 3 937 3 110 0 272 209 481 0 50
Côte d'Ivoire 1990 7 841
–
1995 11 988 8 254 1 508 1 577 649 649 85
2000 15 094 10 276 1 616 2 756 0 446 447 893 0 86
2005 19 681 12 496 2 315 4 235 0 635 345 980 0 84
2010 22 708 14 131 2 381 5 179 0 1 017 502 1 519 0 86
2011 22 476 14 416 2 316 4 729 0 1 015 444 1 459 0 86
• 65 120 • 2012 23 762 14 660 2 818 5 344 0 940 460 1 400 0 84
Democratic 1990 21 131
–
Re
p
ublic 1995 42 819 20 914 7 953 9 112 2 891 2 891 72
of the Con
g
o 2000 61 024 36 513 8 089 13 785 2 637 2 637 82
2005 97 075 65 040 9 959 18 494 3 582 2 483 6 065 87
2010 114 170 73 653 14 039 22 340 0 4 138 4 466 8 604 0 84
2011 110 132 71 321 13 471 21 579 3 761 4 158 7 919 84
• 61 166 • 2012 108 984 71 124 13 214 20 669 3 977 3 515 7 492 84
E
q
uatorial 1990 260
–
Guinea 1995 306 219 45 41 1 1 83
2000
–
2005
–
2010 820 579 98 109 0 34 33 67 0 86
2011 883 611 118 131 0 23 30 53 0 84
• 70 0 • 2012
–
NEW AND
RELAPSE
b
% SMEAR-
POS AMONG
NEW PULM
NEW AND RELAPSE
NOTIFICATION RATE
a
1990–2012
a
Rates are per 100 000 population.
b
NEW AND RELAPSE includes cases for which the treatment history is unknown.

164 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($&DVHQRWLILFDWLRQV±
NEW CASES
YEAR SMEAR-
POSITIVE
SMEAR-NEGATIVE/
UNKNOWN
EXTRA-
PULMONARY OTHER RELAPSE RE-TREAT EXCL.
RELAPSE
TOTAL
RETREAT
HISTORY
UNKNOWN
NEW AND
RELAPSE
b
% SMEAR-
POS AMONG
NEW PULM
NEW AND RELAPSE
NOTIFICATION RATE
a
1990–2012
Eritrea 1990 3 699
–
1995 21 453 18 205 3 248
–
2000 6 652 590 5 332 683 47 20 67 10
2005 3 585 687 1 764 1 001 97 27 124 36 28
2010 2 870 832 1 115 836 0 87 121 208 0 43
2011 3 049 835 1 163 888 60 103 44 147 0 42
• 113 51 • 2012 3 143 779 1 154 1 093 0 117 111 228 0 40
Ethio
p
ia 1990 88 634
–
1995 26 034 9 040 8 888 7 763 343 343 50
2000 91 101 30 510 30 565 28 907 1 119 1 658 2 777 50
2005 124 262 38 525 39 816 43 675 2 246 873 3 119 49
2010 154 694 46 634 54 979 50 417 0 2 664 2 234 4 898 46
2011 156 539 49 594 55 497 49 305 0 2 143 2 478 4 621 0 47
• 184 158 • 2012 145 323 47 236 49 413 46 854 0 1 820 2 269 4 089 0 49
Gabon 1990 917
–
1995 1 115 486 517 68 44 44 48
2000
–
2005 2 512 1 042 1 071 241 158 99 257 49
2010 3 790 1 560 1 366 379 0 168 390 558 317 53
2011 4 404 1 740 1 959 384 0 321 512 833 0 47
• 97 302 • 2012 4 929 1 745 2 353 414 0 175 486 661 242 43
Gambia 1990
–
1995 1 023 778 171 68 6 6 82
2000 1 553 919 515 99 20 33 53 64
2005 2 031 1 127 749 78 0 77 89 166 0 60
2010 1 989 1 344 462 143 0 40 41 81 0 74
2011 2 302 1 375 673 199 4 51 31 82 0 67
• 0 130 • 2012 2 333 1 429 643 169 0 92 54 146 69
Ghana 1990 6 407
–
1995 8 636 2 638 1 225 109 159 159 68
2000 10 933 7 316 2 500 615 502 502 75
2005 12 124 7 505 3 068 1 019 532 532 71
2010 14 607 7 656 5 068 1 400 0 483 538 1 021 0 60
2011 15 389 7 616 5 875 1 471 427 451 878 56
• 44 58 • 2012 14 753 7 097 5 979 1 301 376 454 830 54
Guinea 1990 1 988
–
1995 3 523 2 263 527 620 55 55 81
2000 5 440 3 920 430 938 152 294 446 90
2005 6 863 5 479 524 629 231 227 458 91
2010 11 038 7 041 1 472 2 077 86 362 286 648 0 83
2011 11 359 6 934 1 446 2 284 273 422 247 669 83
• 33 100 • 2012 11 407 6 653 1 510 2 434 321 489 234 723 0 82
Guinea-Bissau 1990 1 163
–
1995 1 613 956 714 19 59 59 57
2000 1 273 526 600 57 90 90 47
2005 1 774 1 132 522 24 0 96 42 138 0 68
2010 2 183 1 409 636 22 0 116 76 192 0 69
2011 2 063 1 230 644 63 0 126 7 133 0 66
• 114 117 • 2012 1 939 1 324 521 43 0 51 11 62 0 72
Ken
y
a 1990 11 788 6 800 100
1995 28 142 13 934 9 676 3 468 1 064 1 064 59
2000 64 159 28 773 24 143 9 118 1 773 704 2 477 54
2005 102 680 40 389 43 772 15 265 3 254 5 721 8 975 48
2010 99 272 36 260 41 962 17 382 0 3 668 6 811 10 479 0 46
2011 97 320 37 085 39 810 17 069 0 3 356 6 661 10 017 0 48
• 50 215 • 2012 92 987 36 937 36 697 15 934 0 3 419 6 162 9 581 0 50
Lesotho 1990 2 525
–
1995 5 181 1 361 2 685 653 147 147 34
2000 9 746 3 041 2 838 2 520 385 1 096 1 481 52
2005 10 802 4 280 4 063 2 020 439 602 1 041 51
2010 11 674 3 600 5 331 2 222 521 1 464 1 985 40
2011 11 561 3 666 5 296 2 095 504 1 224 1 728 41
• 158 525 • 2012 10 776 3 298 5 142 1 877 459 1 195 1 654 39
Liberia 1990
–
1995 1 393 1 154 119 120 91
2000 1 500 1 021 285 187 7 25 32 78
2005 3 432 2 167 575 657 33 24 57 79
2010 6 597 3 750 1 385 1 363 0 99 71 170 0 73
2011 7 906 4 261 1 967 1 612 0 66 59 125 0 68
• 0 193 • 2012 8 093 4 342 1 946 1 749 0 56 39 95 0 69
Mada
g
ascar 1990 6 261
–
1995 21 616 8 026 987 2 219 596 596 89
2000
–
2005 18 993 13 056 1 287 3 634 0 1 016 482 1 498 0 91
2010 24 432 16 795 1 657 4 545 0 1 435 674 2 109 0 91
2011 26 019 17 927 1 726 4 851 1 515 703 2 218 91
• 54 116 • 2012 25 782 17 206 1 804 4 964 0 1 519 427 1 946 289 91
Malawi 1990 12 395 4 301 5 827 1 885 382 382 42
1995 19 155 6 285 7 054 5 257 551 551 47
2000 23 604 8 260 8 846 5 734 764 0 764 48
2005 25 491 8 443 10 132 5 823 1 093 2 119 3 212 45
2010 21 092 7 240 8 245 4 857 0 750 1 444 2 194 47
2011 19 361 7 003 6 612 5 076 670 1 493 2 163 51
• 131 128 • 2012 20 335 6 951 6 550 4 886 0 694 128 822 1 254 51
Mali 1990 2 933
–
1995 3 087 1 866 609 459 153 153 75
2000 4 216 2 527 797 653 239 239 76
2005 4 704 3 530 482 492 200 180 380 0 88
2010 5 291 3 686 481 926 0 198 157 355 0 88
2011 5 428 3 777 491 984 176 145 321 88
• 37 37 • 2012 5 446 3 724 487 1 081 0 154 156 310 0 88
Mauritania 1990 5 284
–
1995 3 849 2 074 800 455 520 520 72
2000 3 067 1 583 687 580 580 358 938 70
2005 2 162 1 155 454 403 0 150 56 206 0 72
2010 2 461 1 422 390 524 0 125 28 153 78
2011 1 804 1 009 222 458 0 87 16 103 28 82
• 261 69 • 2012 2 616 1 522 354 628 0 112 20 132 0 81
Mauritius 1990 119
–
1995 131 113 8 12 2 2 93
2000 160 115 14 23 8 4 12 89
2005 125 110 4 8 3 2 5 96
2010 122 105 5 6 0 6 1 7 0 95
2011 114 100 3 8 0 3 2 5 0 97
• 11 10 • 2012 128 118 3 5 0 2 2 4 0 98
a
Rates are per 100 000 population.
b
NEW AND RELAPSE includes cases for which the treatment history is unknown.

165 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
AFRICAN REGION
7$%/($&DVHQRWLILFDWLRQV±
NEW CASES
YEAR SMEAR-
POSITIVE
SMEAR-NEGATIVE/
UNKNOWN
EXTRA-
PULMONARY OTHER RELAPSE RE-TREAT EXCL.
RELAPSE
TOTAL
RETREAT
HISTORY
UNKNOWN
NEW AND
RELAPSE
b
% SMEAR-
POS AMONG
NEW PULM
NEW AND RELAPSE
NOTIFICATION RATE
a
1990–2012
Mozambi
q
ue 1990 15 899
–
1995 17 882 10 566 5 054 1 363 899 899 68
2000 21 158 13 257 4 037 2 262 917 546 1 463 77
2005 33 231 17 877 9 184 4 771 1 399 487 1 886 66
2010 43 558 20 097 16 408 5 621 0 1 432 2 616 4 048 0 55
2011 44 627 19 537 18 159 5 504 0 1 427 2 825 4 252 0 52
• 117 189 • 2012 47 741 20 951 19 797 5 542 0 1 451 3 086 4 537 0 51
Namibia 1990 2 671
–
1995 1 540 697 507 248 88 88 58
2000 10 799 4 012 4 724 1 459 604 930 1 534 46
2005 14 920 5 222 4 455 1 907 2 487 849 974 1 823 54
2010 11 281 4 464 3 309 2 330 1 178 1 344 2 522 57
2011 10 806 4 503 3 034 2 039 0 1 230 1 132 2 362 0 60
• 189 443 • 2012 10 003 4 333 2 473 2 063 1 134 1 142 2 276 64
Ni
g
er 1990 5 200
–
1995 1 980 1 492 116 372 93
2000 4 701 3 045 699 702 255 255 81
2005 7 873 5 050 1 193 1 227 403 351 754 0 81
2010 10 130 6 283 1 730 1 492 173 452 215 667 0 78
2011 10 510 6 604 1 856 1 489 376 204 580 185 78
• 67 64 • 2012 10 989 6 848 1 989 1 689 116 347 218 565 0 77
Ni
g
eria 1990 20 122
–
1995 13 423 9 476 3 364 280 303 303 74
2000 25 821 17 423 6 613 1 069 716 1 640 2 356 72
2005 63 990 35 048 22 705 2 836 0 2 009 2 858 4 867 1 392 61
2010 84 121 45 416 32 616 3 422 0 2 667 6 326 8 993 0 58
2011 86 778 47 436 33 034 3 793 0 2 515 6 272 8 787 0 59
• 21 55 • 2012 92 818 52 901 32 972 4 432 2 513 5 035 7 548 62
Rwanda 1990 6 387
–
1995 3 054 1 840 676 338 200 200 73
2000 6 093 3 681 845 1 289 278 96 374 81
2005 7 220 4 166 859 1 727 97 371 460 831 83
2010 6 703 3 785 1 072 1 577 269 362 631 78
2011 6 623 3 811 1 017 1 300 242 253 161 414 0 79
• 89 53 • 2012 6 091 3 571 858 1 247 203 212 117 329 0 81
Sao Tome and 1990 17
–
Princi
p
e 1995
–
2000 97 30 56 7 4 4 35
2005 136 49 75 1 0 11 16 27 40
2010 121 47 63 10 0 1 1 2 0 43
2011 136 53 49 28 0 6 10 16 0 52
• 14 61 • 2012 115 59 37 16 0 3 12 15 0 61
Sene
g
al 1990 4 977
–
1995 7 561 5 421 1 073 504 563 563 83
2000 8 508 5 823 1 370 800 0 515 541 1 056 81
2005 9 765 6 722 1 557 921 565 355 920 81
2010 11 061 7 688 1 470 1 404 0 499 530 1 029 0 84
2011 11 022 7 765 1 389 1 315 0 553 566 1 119 0 85
• 66 89 • 2012 12 265 8 448 1 755 1 524 0 538 554 1 092 0 83
Se
y
chelles 1990 41
–
1995 8 6 2 1 0 0 75
2000 20 11 7 2 0 0 61
2005 14 8 3 1 0 2 0 2 73
2010 17 9 8 0 0 0 0 0 0 53
2011 21 2 13 6 0 0 0 0 0 13
• 59 22 • 2012 20 9 8 2 0 1 1 2 0 53
Sierra Leone 1990 632
–
1995 1 955 1 454 339 121 41 41 81
2000 3 760 2 472 821 400 67 374 441 75
2005 6 737 4 370 1 679 551 137 193 330 72
2010 12 859 6 898 4 919 831 211 336 547 58
2011 12 734 7 435 4 358 775 166 209 375 63
• 16 219 • 2012 13 074 8 031 4 241 570 232 280 512 65
South Africa 1990 80 400
–
1995 73 917 23 112 74 399 10 636 179 179 24
2000 151 239 75 967 16 392 17 486 56 202 56 202 82
2005 270 178 125 460 76 680 39 739 0 28 299 32 289 60 588 0 62
2010 354 786 132 107 151 772 52 095 0 18 812 41 768 60 580 0 47
2011 362 453 129 770 148 266 47 285 0 18 394 27 521 45 915 18 738 47
• 219 618 • 2012 323 664 119 898 134 631 42 467 0 26 668 25 918 52 586 0 47
Swaziland 1990
–
1995 2 050 660 687 219 489 489 49
2000 5 877 1 823 3 198 583 273 976 1 249 36
2005 8 705 2 187 4 106 1 458 311 159 470 643 35
2010 10 101 3 011 5 064 1 631 395 1 045 1 440 37
2011 8 337 2 408 4 228 1 395 0 306 843 1 149 0 36
• 0 582 • 2012 7 165 2 548 3 111 1 209 0 297 574 871 0 45
To
g
o 1990 1 324
–
1995 1 520 887 304 236 93 93 74
2000 1 409 984 91 287 47 86 133 92
2005 2 541 1 798 170 484 85 94 179 4 91
2010 2 791 2 096 164 397 0 134 106 240 0 93
2011 2 888 2 087 205 475 0 121 92 213 0 91
• 35 43 • 2012 2 843 2 112 168 444 0 119 69 188 0 93
U
g
anda 1990 14 740
–
1995 25 316 13 631 5 912 2 070 955 955 70
2000 30 372 17 246 9 003 2 618 1 505 0 1 505 66
2005 41 040 20 559 15 040 3 780 0 1 661 769 2 430 58
2010 42 885 23 456 13 567 4 571 0 1 291 2 661 3 952 0 63
2011 46 306 25 614 14 389 5 001 1 302 2 712 4 014 64
• 84 123 • 2012 44 663 24 916 13 270 5 143 0 1 334 2 548 3 882 0 65
United Re
p
ublic 1990 22 249 11 553 100
of Tanzania 1995 39 847 19 955 12 362 6 195 1 335 1 335 62
2000 54 442 24 049 17 624 10 997 1 772 1 772 58
2005 61 022 25 264 20 810 13 094 1 854 3 178 5 032 55
2010 61 098 24 769 21 184 13 715 1 430 2 355 3 785 54
2011 59 357 24 115 20 438 13 725 0 1 079 1 791 2 870 0 54
• 87 130 • 2012 62 178 25 138 21 393 14 595 0 1 052 1 714 2 766 0 54
Zambia 1990 16 863
–
1995 35 958 10 038 3 268 656 243 243 75
2000 49 806 12 927 25 222 10 202 1 455 1 455 34
2005 49 576 14 857 24 327 8 587 1 805 3 691 5 496 38
2010 44 154 12 639 20 412 9 255 0 1 848 4 462 6 310 0 38
2011 43 583 12 046 20 004 9 908 1 625 5 011 6 636 38
• 215 289 • 2012 40 726 12 645 17 050 9 174 1 857 4 551 6 408 43
Zimbabwe 1990 9 132
–
1995 30 831 8 965 10 934 5 040 737 737 45
2000 50 855 14 392 27 626 8 837 34
2005 50 454 13 155 29 074 6 721 0 1 504 4 437 5 941 31
2010 44 209 11 654 25 157 6 061 0 1 337 3 348 4 685 0 32
2011 38 404 12 596 19 172 5 192 0 1 444 2 901 4 345 0 40
• 87 261 • 2012 35 760 12 163 17 316 4 912 0 1 369 2 960 4 329 0 41
a
Rates are per 100 000 population.
b
NEW AND RELAPSE includes cases for which the treatment history is unknown.

166 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($7UHDWPHQWRXWFRPHVQHZVPHDUSRVLWLYHFDVHV±
% OF COHORT
TREATMENT SUCCESS (%)
a
1995–2011 YEAR NUMBER
NOTIFIED
SIZE OF
COHORT
COHORT AS
% NOTIFIED CURED COMPLETED DIED FAILED DEFAULTED NOT
EVALUATED
Algeria 1995 5 735 –
2000 8 328 8 328 100 80 7 1 2 5 5
2005 8 654 8 379 97 74 13 2 0 3 8
2009 8 402 8 438 100 81 10 2 1 3 3
2010 8 299 7 894 95 79 10 2 1 4 4
• 0 92 • 2011 7 790 7 364 95 81 11 2 0 4 2
Angola 1995 3 804 –
2000 9 053 6 392 71 68 3 2 26 2
2005 20 410 20 113 99 45 28 3 3 19 3
2009 22 488 21 627 96 47 25 4 2 18 5
2010 21 146 21 145 100 30 18 8 1 8 35
• 0 55 • 2011 21 703 21 703 100 36 19 7 2 13 23
Benin 1995 1 839 1 839 100 50 21 6 1 17 5
2000 2 277 2 277 100 57 20 6 2 11 3
2005 2 739 2 766 101 74 13 7 2 3 1
2009 2 960 2 963 100 8295210
2010 2 973 2 987 100 8475211
• 71 90 • 2011 3 331 3 324 100 8466310
Botswana 1995 1 903 2 060 108 13 54 5 1 12 15
2000 3 091 3 991 129 22 55 6 0 7 10
2005 3 170 3 335 105 37 33 7 1 8 15
2009 3 144 3 492 111 57 22 5349
2010 3 295 3 314 101 50 32 5238
• 67 81 • 2011 2 669 3 107 116 36 46 6239
Burkina Faso 1995 1 028 1 200 117 22 2 5 1 3 67
2000 1 545 1 574 102 53 7 13 2 16 9
2005 2 290 2 290 100 66 5 14 7 6 1
2009 3 061 3 061 100 72 4 10 9 4 2
2010 3 041 3 057 101 74 3 9 7 6 1
• 25 78 • 2011 3 450 3 442 100 74 4 9 6 6 1
Burundi 1995 1 121 1 798 160 25 20 3 0 14 38
2000 3 159 3 465 110 42 39 4 0 13 1
2005 3 262 3 424 105 52 27 4 0 17 1
2009 3 974 3 974 100 8373150
2010 4 590 4 590 100 8744130
• 45 92 • 2011 4 060 4 060 100 8844130
Cameroon 1995 2 896 2 740 95 45 8 7 1 35 4
2000 3 960 3 164 80 67 10 7 2 13 1
2005 13 001 13 169 101 66761145
2009 14 635 14 428 99 65 13 6 1 10 5
2010 14 464 14 464 100 64 14 6 1 10 5
• 53 80 • 2011 14 927 14 927 100 67 13 6 1 9 4
Cape Verde 1995 111 –
2000 14 – 64070029
2005 135 135 100 56 8 3 2 19 12
2009 172 –
2010 186 –
• 0 77 • 2011 182 182 100 55 23 4 0 9 10
Central African 1995 1 794 692 39 16 21 7 0 53 3
Republic 2000 1 366 – 36 21 0 3 34 5
2005 2 153 3 217 149 38 28 6 2 8 19
2009 5 132 5 132 100 33 20 3 1 13 30
2010 3 638 3 569 98 45 23 6 1 19 7
• 37 68 • 2011 3 479 3 205 92 44 23 4 1 18 10
Chad 1995 2 002 529 26 17 30 6 1 43 3
2000 –
2005 2 516 –
2009 3 820 3 820 100 55 22 4 2 15 3
2010 3 833 3 780 99 39 28 4 2 21 5
• 47 68 • 2011 4 434 4 430 100 45 23 4 1 19 8
Comoros 1995 103 113 110 9004060
2000 87 85 98 9124400
2005 79 70 89 9103401
2009 76 –
2010 87 – 9103212
• 90 25 • 2011 62 4 6 25 75 0
Congo 1995 2 013 –
2000 4 218 3 114 74 57 12 4 0 22 5
2005 3 640 4 121 113 244011358
2009 3 433 3 634 106 66 12 1 0 13 7
2010 3 568 3 447 97 63 13 2 1 12 8
• 0 71 • 2011 3 716 3 716 100 59 12 2 2 11 14
Côte d'Ivoire 1995 8 254 7 221 87 63641179
2000 10 276 10 631 103 47 10 5 2 16 20
2005 12 496 12 496 100 62 11 8 2 10 6
2009 14 300 14 300 100 69 10 8 3 7 4
2010 14 131 14 131 100 66 12 7 2 8 5
• 68 78 • 2011 14 416 14 416 100 6997293
Democratic 1995 20 914 16 247 78 55 20 5 1 10 9
Republic 2000 36 513 36 123 99 6986187
of the Congo 2005 65 040 65 066 100 8056144
2009 73 078 72 367 99 8534134
2010 73 653 73 448 100 8644133
• 74 87 • 2011 71 321 71 321 100 8254145
Equatorial 1995 219 219 100 8903080
Guinea 2000 –
2005 –
2009 490 490 100 47 19 3 1 16 14
2010 579 590 102 50 20 5 1 17 7
• 89 0 • 2011 611 –
Eritrea 1995 –
2000 590 765 130 64 12 8196
2005 687 688 100 8357121
2009 802 804 100 8325325
2010 832 804 97 8147315
• 0 87 • 2011 835 835 100 8344415
Ethiopia 1995 9 040 5 087 56 565521319
2000 30 510 29 662 97 63 17 6194
2005 38 525 39 430 102 64 14 5 1 4 12
2009 44 396 44 807 101 65 19 3 1 3 10
2010 46 634 46 634 100 66 17 3 1 3 10
• 61 90 • 2011 49 594 41 351 83 70 19 3 1 2 4
Gabon 1995 486 249 51 63 22 1 2 9 2
2000 –
2005 1 042 1 165 112 35 12 10 1 42 1
2009 1 244 1 163 93 37 18 1 1 25 18
2010 1 560 1 671 107 34 29 2 3 26 6
• 86 51 • 2011 1 740 1 654 95 26 25 2 1 36 11
a
TREATMENT SUCCESS = percent cured + percent completed then rounded to the nearest digit.

167 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
AFRICAN REGION
7$%/($7UHDWPHQWRXWFRPHVQHZVPHDUSRVLWLYHFDVHV±
% OF COHORT
TREATMENT SUCCESS (%)
a
1995–2011 YEAR NUMBER
NOTIFIED
SIZE OF
COHORT
COHORT AS
% NOTIFIED CURED COMPLETED DIED FAILED DEFAULTED NOT
EVALUATED
Gambia 1995 778 686 88 69751135
2000 919 –
2005 1 127 1 127 100 81 6 7132
2009 1 313 1 296 99 88 1 6121
2010 1 344 1 344 100 86 2 5133
• 76 88 • 2011 1 375 1 375 100 86 2 6222
Ghana 1995 2 638 361 14 41 13 11 2 11 22
2000 7 316 7 316 100 455631427
2005 7 505 7 584 101 68 5 9 2 11 5
2009 8 255 8 255 100 79 8 7133
2010 7 656 7 656 100 76 10 7133
• 54 86 • 2011 7 616 7 623 100 75 11 8132
Guinea 1995 2 263 2 263 100 62 17 6 2 9 5
2000 3 920 3 920 100 59971159
2005 5 479 5 811 106 657621010
2009 5 377 5 597 104 7265278
2010 7 041 7 250 103 7644269
• 78 82 • 2011 6 934 5 152 74 7664275
Guinea-Bissau 1995 956 959 100 42 23 6 0 23 6
2000 526 –
2005 1 132 1 167 103 51 18 12 1 11 7
2009 1 310 1 498 114 51 17 6 1 21 5
2010 1 409 1 271 90 54 18 6 0 14 7
• 65 73 • 2011 1 230 1 308 106 60 14 6 0 13 7
Kenya 1995 13 934 6 470 46 60 14 9 1 9 7
2000 28 773 28 376 99 66 14 5 0 9 6
2005 40 389 40 436 100 71 11 5 0 8 5
2009 37 402 37 402 100 7884164
2010 36 260 36 260 100 8163154
• 75 88 • 2011 37 085 36 717 99 8353143
Lesotho 1995 1 361 1 788 131 32 14 7 0 9 36
2000 3 041 –
2005 4 280 5 542 129 73 8 1 4 14
2009 3 976 4 070 102 59 11 11 2 5 12
2010 3 600 3 852 107 58 10 10 2 8 12
• 47 74 • 2011 3 666 3 666 100 63 11 11 2 6 7
Liberia 1995 1 154 1 595 138 79 5 5 12 0
2000 1 021 924 90 71926103
2005 2 167 2 167 100 60 16 3 0 12 8
2009 3 796 3 796 100 57 26 5193
2010 3 750 –
• 79 86 • 2011 4 261 3 853 90 64 22 4164
Madagascar 1995 8 026 9 101 113 478621620
2000 10 506 – 61971175
2005 13 056 15 298 117 67 7 6 1 13 5
2009 15 729 15 709 100 7834195
2010 16 795 16 789 100 7844194
• 55 83 • 2011 17 927 17 602 98 7944184
Malawi 1995 6 285 6 293 100 65 6 19 1 0 10
2000 8 260 8 296 100 70 3 19 1 4 3
2005 8 443 8 443 100 72 2 15 1 3 7
2009 7 623 7 624 100 8727121
2010 7 240 7 240 100 8627122
• 71 85 • 2011 7 003 7 012 100 8147224
Mali 1995 1 866 1 290 69 41 18 5 0 22 14
2000 2 527 –
2005 3 530 3 530 100 69 6 11 4 7 3
2009 5 163 4 454 86 66 12 10 4 7 2
2010 3 686 3 778 102 7608394
• 59 68 • 2011 3 777 3 777 100 55 13 7 3 7 15
Mauritania 1995 2 074 –
2000 1 583 –
2005 1 155 1 761 152 44 11 2 1 19 24
2009 1 555 1 563 101 51 12 3 1 10 23
2010 1 422 1 422 100 55 14 2 1 13 15
• 0 73 • 2011 1 009 1 450 144 57 16 2 0 16 9
Mauritius 1995 113 –
2000 115 160 139 0 92 2230
2005 110 110 100 86 3 6 5
2009 98 98 100 8804044
2010 105 105 100 9004150
• 0 90 • 2011 100 100 100 9005050
Mozambique 1995 10 566 10 566 100 34 5 3 1 9 48
2000 13 257 13 296 100 73 2 10 1 11 3
2005 17 877 17 877 100 78 1 12 1 5 2
2009 19 579 19 579 100 84 1 9 1 3 2
2010 20 097 20 097 100 8328141
• 39 0 • 2011 19 537 –
Namibia 1995 697 –
2000 4 012 4 012 100 41 15 6 2 15 21
2005 5 222 5 222 100 59 16 7 2 10 7
2009 4 608 4 702 102 74 11 5 4 4 2
2010 4 464 4 538 102 74 11 5 4 3 2
• 0 84 • 2011 4 503 4 502 100 74 10 5 5 5 0
Niger 1995 1 492 –
2000 3 045 3 193 105 42 22 8 4 12 11
2005 5 050 5 050 100 49 25 5 2 14 5
2009 6 347 6 313 99 66 13 7 2 7 5
2010 6 283 6 266 100 69 13 7 2 6 3
• 0 80 • 2011 6 604 6 604 100 66 15 5 3 9 3
Nigeria 1995 9 476 9 476 100 34 15 5 2 9 35
2000 17 423 16 372 94 65 14 6 2 11 2
2005 35 048 35 080 100 50 25 9 4 11 1
2009 44 863 44 863 100 73 10 5 1 8 4
2010 45 416 45 416 100 73 10 5 1 8 2
• 49 85 • 2011 47 436 47 436 100 77 9 5 1 7 2
Rwanda 1995 1 840 –
2000 3 681 3 776 103 52961428
2005 4 166 4 175 100 73 10 6236
2009 4 184 4 165 100 7785433
2010 3 785 3 806 101 8085421
• 0 89 • 2011 3 811 3 811 100 8455421
Sao Tome and 1995 –
Principe 2000 30 97 323 52 27 9570
2005 49 49 100 9802000
2009 52 50 96 9800200
2010 47 45 96 20 58 9 0 13 0
• 0 72 • 2011 53 53 100 45 26 4 19 6 0
a
TREATMENT SUCCESS = percent cured + percent completed then rounded to the nearest digit.

GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($7UHDWPHQWRXWFRPHVQHZVPHDUSRVLWLYHFDVHV±
% OF COHORT
TREATMENT SUCCESS (%)
a
1995–2011 YEAR NUMBER
NOTIFIED
SIZE OF
COHORT
COHORT AS
% NOTIFIED CURED COMPLETED DIED FAILED DEFAULTED NOT
EVALUATED
Senegal 1995 5 421 5 421 100 359461631
2000 5 823 5 823 100 439312122
2005 6 722 6 722 100 70 6 4 2 11 8
2009 7 883 7 883 100 81 3 4 2 5 5
2010 7 688 7 855 102 81 4 4 2 6 4
• 44 85 • 2011 7 765 7 898 102 82 3 3 2 7 3
Seychelles 1995 6 9 150 89 0 11 0 0 0
2000 11 11 100 82 0 0 0 9 9
2005 8 –
2009 11 11 100 55 9 18 0 0 18
2010 9 7 78 100 0 0 0 0 0
• 89 67 • 2011 2 9 450 56 11 0 0 11 22
Sierra Leone 1995 1 454 1 315 90 55 15 5 7 16 2
2000 2 472 2 296 93 70 7 6 2 13 2
2005 4 370 4 370 100 77 8 6 1 6 2
2009 6 092 6 083 100 68 10 6 1 11 4
2010 6 898 6 897 100 77 9 4 1 6 3
• 69 88 • 2011 7 435 7 351 99 79 9 3 1 6 2
South Africa 1995 23 112 28 209 122 40 18 4 4 15 19
2000 75 967 86 276 114 54 9 6 1 13 17
2005 125 460 134 782 107 58 13 7 2 10 10
2009 139 468 139 458 100 67 6 7 2 7 12
2010 132 107 134 250 102 7366276
• 58 79 • 2011 129 770 132 867 102 7456267
Swaziland 1995 660 –
2000 1 823 –
2005 2 187 2 187 100 22 20 6 2 5 45
2009 3 498 3 498 100 51 19 10 7 7 7
2010 3 011 3 011 100 51 22 11 9 6 2
• 0 73 • 2011 2 408 2 499 104 48 25 8855
Togo 1995 887 856 97 42 18 9 3 17 11
2000 984 –
2005 1 798 1 796 100 66 5 12 4 11 2
2009 2 267 2 267 100 77 4 10 4 3 2
2010 2 096 2 096 100 8138341
• 60 85 • 2011 2 087 2 075 99 8137251
Uganda 1995 13 631 15 301 112 26 18 7 1 13 36
2000 17 246 13 874 80 33 30 7 0 17 12
2005 20 559 20 559 100 32 41 6 0 16 5
2009 23 113 23 113 100 30 38 5 1 12 16
2010 23 456 23 456 100 35 36 5 1 11 13
• 44 77 • 2011 25 614 25 614 100 39 38 5 1 12 5
United Republic 1995 19 955 19 955 100 69 5 9 1 6 11
of Tanzania 2000 24 049 23 923 99 72 6 10 0 6 5
2005 25 264 25 324 100 7949044
2009 24 895 24 895 100 8265025
2010 24 769 24 373 98 8465023
• 73 88 • 2011 24 115 24 218 100 8074026
Zambia 1995 10 038 5 957 59 47 23 7 2 14 8
2000 12 927 7 014 54 48 19 7 6 6 14
2005 14 857 14 857 100 7688125
2009 12 995 12 995 100 85 66130
2010 12 639 12 639 100 83 6 6131
• 70 88 • 2011 12 046 12 711 106 82 5 4134
Zimbabwe 1995 8 965 9 702 108 32 21 10 0 10 26
2000 14 392 14 392 100 61 8 12 0 7 13
2005 13 155 12 860 98 59 9 12 2 7 12
2009 10 195 10 195 100 70 9 8176
2010 11 654 11 654 100 72 10 8 1 5 5
• 53 81 • 2011 12 596 12 596 100 73 8 8 1 4 6
a TREATMENT SUCCESS = percent cured + percent completed then rounded to the nearest digit.

169 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
AFRICAN REGION
7$%/($7UHDWPHQWRXWFRPHVUHWUHDWPHQWFDVHV±
% OF COHORT
TREATMENT SUCCESS (%)
a
1995–2011 YEAR NUMBER
NOTIFIED
SIZE OF
COHORT
COHORT AS
% NOTIFIED CURED COMPLETED DIED FAILED DEFAULTED NOT
EVALUATED
Algeria 1995 451 –
2000 547 512 94 61 16 5 4 5 10
2005 713 713 100 48 24 2 1 6 19
2009 612 553 90 72 12 4 2 5 5
2010 691 598 87 69 14 4 2 5 6
• 0 80 • 2011 610 588 96 65 15 3 4 11 3
Angola 1995 134 –
2000 540 –
2005 2 871 1 613 56 23 24 5 17 26 4
2009 3 863 3 044 79 45 21 5 4 21 3
2010 7 776 2 272 29 42 23 8 4 16 7
• 0 0 • 2011 4 444 4 444 100 0 0 0 0 100
Benin 1995 68 139 204 48 19 9 4 19 1
2000 280 282 101 61 21 5 1 11 0
2005 337 341 101 60 21 10 3 6 1
2009 271 270 100 70 11 11 6 1 1
2010 205 203 99 76 9 6 6 1 1
• 67 84 • 2011 262 262 100 80 4 8 5 1 1
Botswana 1995 147 –
2000 1 239 395 32 21 54 8 1 11 6
2005 548 219 40 33 28 11 5 12 11
2009 1 122 1 126 100 22 43 13 4 8 10
2010 1 072 1 027 96 20 46 14 3 7 10
• 0 70 • 2011 868 998 115 15 55 11 3 6 10
Burkina Faso 1995 45 26 58 65 12 8 12 0 4
2000 178 166 93 57 4 13 5 15 7
2005 327 272 83 71 4 6 10 6 4
2009 608 509 84 70 5 9 8 5 3
2010 552 475 86 72 4 9 8 6 1
• 77 75 • 2011 484 481 99 70 4 10 8 6 1
Burundi 1995 181 265 146 25 21 6 2 28 18
2000 225 92 41 50 13 15 3 17 1
2005 116 –
2009 238 238 100 81 3 6 3 4 2
2010 332 332 100 78 4 7 5 6 0
• 46 85 • 2011 315 315 100 80 4 6 5 4 0
Cameroon 1995 236 –
2000 251 347 138 50 10 9 5 26 2
2005 1 590 1 611 101 49 7 6 3 16 19
2009 1 569 1 516 97 51 18 9 2 13 7
2010 1 494 1 489 100 55 16 9 3 12 6
• 0 70 • 2011 1 661 1 661 100 54 16 9 3 12 6
Cape Verde 1995 30 –
2000 –
2005 34 34 100 41 15 0 0 24 21
2009 33 –
2010 27 –
• 0 37 • 2011 27 27 100 22 15 4 4 11 44
Central African 1995 188 –
Republic 2000 353 – 33 16 1 4 39 8
2005 291 291 100 53 30 9 0 8 1
2009 629 629 100 19 12 5 2 8 53
2010 421 284 67 35 24 7 4 25 6
• 0 55 • 2011 345 275 80 33 21 11 4 20 11
Chad 1995 203 92 45 29 18 5 2 40 4
2000 –
2005 515 –
2009 676 676 100 49 21 4 3 15 8
2010 708 704 99 38 35 4 2 18 3
• 48 60 • 2011 847 847 100 29 31 4 1 27 7
Comoros 1995 7 7 100 43 0 29 0 29 0
2000 5 5 100 100 0 0 0 0 0
2005 3 5 167 100 0 0 0 0 0
2009 6 –
2010 5 – 80 0 0 20 0 0
• 43 0 • 2011 11 –
Congo 1995 78 –
2000 819 187 23 49 13 3 3 28 4
2005 407 477 117 12 2 0 0 3 83
2009 451 418 93 59 22 2 1 14 2
2010 516 235 46 40 17 3 2 21 18
• 0 51 • 2011 507 528 104 51 0 5 4 10 31
Côte d'Ivoire 1995 649 –
2000 893 507 57 45 10 8 9 21 7
2005 980 980 100 43 14 8 7 13 15
2009 1 436 1 436 100 50 14 13 11 9 3
2010 1 519 1 519 100 51 14 12 8 11 3
• 0 67 • 2011 1 459 1 459 100 56 11 10 8 12 3
Democratic 1995 2 891 1 202 42 56 16 8 2 12 6
Republic 2000 2 637 –
of the Congo 2005 6 065 5 448 90 71 4 10 4 6 5
2009 8 666 7 193 83 54 23 8 2 4 8
2010 8 604 5 583 65 72 5 7 3 6 8
• 72 74 • 2011 7 919 4 572 58 68 5 8 2 5 12
Equatorial 1995 1 6 600 83 0 0 17 0 0
Guinea 2000 –
2005 –
2009 44 44 100 36 14 14 2 16 18
2010 67 41 61 32 15 22 0 27 5
• 83 0 • 2011 53 –
Eritrea 1995 –
2000 67 –
2005 124 –
2009 207 157 76 70 12 7 6 2 3
2010 208 120 58 81 8 9 2 1 0
• 0 69 • 2011 147 147 100 67 3 7 10 1 13
Ethiopia 1995 343 193 56 71 8 3 5 8 5
2000 2 777 1 556 56 60 11 10 4 8 7
2005 3 119 3 116 100 41 15 9 2 5 28
2009 3 544 2 942 83 47 21 5 2 3 23
2010 4 898 3 934 80 56 27 4 3 5 6
• 79 78 • 2011 4 621 1 796 39 57 21 4 0 3 15
Gabon 1995 44 –
2000 –
2005 257 150 58 18 12 5 3 60 3
2009 655 611 93 12 67 2 1 17 1
2010 558 147 26 32 33 3 3 26 2
• 0 40 • 2011 833 200 24 18 21 2 2 30 26
a
TREATMENT SUCCESS = percent cured + percent completed then rounded to the nearest digit.

170 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($7UHDWPHQWRXWFRPHVUHWUHDWPHQWFDVHV±
% OF COHORT
TREATMENT SUCCESS (%)
a
1995–2011 YEAR NUMBER
NOTIFIED
SIZE OF
COHORT
COHORT AS
% NOTIFIED CURED COMPLETED DIED FAILED DEFAULTED NOT
EVALUATED
Gambia 1995 6 45 750 69 0 11 2 11 7
2000 53 –
2005 166 –
2009 99 100 101 67 5 17 2 7 2
2010 81 81 100 30 6 6 1 0 57
• 69 78 • 2011 82 86 105 74 3 13 3 3 2
Ghana 1995 159 47 30 68 6 6 9 9 2
2000 502 –
2005 532 540 102 40 8 6 3 11 32
2009 860 717 83 50 26 10 2 3 10
2010 1 021 1 021 100 38 39 12 2 2 7
• 74 77 • 2011 878 878 100 40 38 12 3 4 5
Guinea 1995 55 112 204 44 23 3 9 13 8
2000 446 299 67 63 8 5 3 8 13
2005 458 458 100 45 16 10 7 13 11
2009 589 –
2010 648 111 17 55 14 8 5 13 6
• 67 64 • 2011 669 121 18 56 7 8 3 16 9
Guinea-Bissau 1995 59 –
2000 90 –
2005 138 146 106 44 34 8 0 8 7
2009 76 89 117 30 34 2 0 29 4
2010 192 140 73 23 31 10 0 27 9
• 0 68 • 2011 133 47 35 47 21 13 2 9 9
Kenya 1995 1 064 879 83 61 11 9 1 10 8
2000 2 477 1 964 79 65 11 2 8 10 4
2005 8 975 3 794 42 68 9 10 1 7 5
2009 10 711 4 859 45 70 8 8 4 7 4
2010 10 479 4 333 41 73 6 6 3 8 4
• 72 82 • 2011 10 017 7 235 72 77 5 4 4 7 3
Lesotho 1995 147 –
2000 1 481 –
2005 1 041 597 57 71 11 2 2 14
2009 1 970 1 931 98 20 42 17 2 4 15
2010 1 985 2 091 105 16 42 16 2 8 16
• 0 58 • 2011 1 728 1 728 100 17 41 18 2 10 12
Liberia 1995 –
2000 32 41 128 39 22 12 7 20 0
2005 57 57 100 75 9 2 9 5
2009 123 123 100 70 15 8 4 2 0
2010 170 –
• 0 82 • 2011 125 125 100 72 10 4 12 2 0
Madagascar 1995 596 –
2000 –
2005 1 498 1 825 122 65 7 7 2 12 6
2009 2 089 2 073 99 62 11 7 2 8 10
2010 2 109 1 800 85 71 3 8 2 9 8
• 0 80 • 2011 2 218 1 843 83 75 4 7 1 8 4
Malawi 1995 551 492 89 65 4 22 2 1 6
2000 764 797 104 61 5 23 1 6 3
2005 3 212 1 093 34 74 1 19 1 3 3
2009 2 470 788 32 83 2 9 2 2 1
2010 2 194 750 34 77 1 10 3 1 9
• 69 82 • 2011 2 163 670 31 79 3 10 1 3 5
Mali 1995 153 –
2000 239 –
2005 380 379 100 67 6 10 5 10 3
2009 425 390 92 67 8 9 6 7 3
2010 355 345 97 87 12 1 0 0 0
• 0 69 • 2011 321 321 100 64 5 7 4 4 15
Mauritania 1995 520 –
2000 938 –
2005 206 –
2009 182 182 100 48 13 3 1 20 14
2010 153 153 100 46 13 5 2 15 20
• 0 53 • 2011 103 133 129 43 10 10 2 17 19
Mauritius 1995 2 –
2000 12 2 17 0 0 50 50 0 0
2005 5 5 100 60 20 20 0
2009 5 5 100 60 0 20 0 20 0
2010 7 7 100 86 0 0 0 14 0
• 0 80 • 2011 5 5 100 80 0 0 0 20 0
Mozambique 1995 899 –
2000 1 463 1 594 109 69 3 11 4 11 2
2005 1 886 1 855 98 69 1 15 2 10 3
2009 3 630 –
2010 4 048 –
• 0 0 • 2011 4 252 –
Namibia 1995 88 –
2000 1 534 604 39 41 14 8 6 13 17
2005 1 823 2 009 110 24 29 11 3 13 22
2009 2 558 1 546 60 58 15 9 9 6 3
2010 2 522 2 548 101 63 15 6 10 5 2
• 0 80 • 2011 2 362 2 361 100 67 13 5 9 5 0
Niger 1995 –
2000 255 –
2005 754 –
2009 690 667 97 64 12 9 4 5 6
2010 667 661 99 64 11 10 3 5 7
• 0 76 • 2011 580 580 100 62 14 6 5 10 4
Nigeria 1995 303 –
2000 2 356 1 848 78 58 13 7 7 11 4
2005 4 867 3 662 75 48 18 2 11 20 1
2009 8 151 8 151 100 48 33 6 2 7 4
2010 8 993 8 993 100 43 39 4 4 7 3
• 0 82 • 2011 8 787 8 787 100 42 40 4 4 6 4
Rwanda 1995 200 –
2000 374 296 79 49 5 14 1 5 25
2005 831 506 61 56 9 15 3 4 13
2009 475 448 94 62 10 11 7 4 6
2010 631 446 71 65 9 9 6 4 6
• 0 80 • 2011 414 415 100 72 8 10 7 2 1
Sao Tome and 1995 –
Principe 2000 4 –
2005 27 0 0
2009 3 3 100 33 33 0 33 0 0
2010 2 12 600 0 50 8 17 8 17
• 0 31 • 2011 16 16 100 0 31 6 38 25 0
a
TREATMENT SUCCESS = percent cured + percent completed then rounded to the nearest digit.

171 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
AFRICAN REGION
7$%/($7UHDWPHQWRXWFRPHVUHWUHDWPHQWFDVHV±
% OF COHORT
TREATMENT SUCCESS (%)
a
1995–2011 YEAR NUMBER
NOTIFIED
SIZE OF
COHORT
COHORT AS
% NOTIFIED CURED COMPLETED DIED FAILED DEFAULTED NOT
EVALUATED
Senegal 1995 563 634 113 45 11 5 10 25 4
2000 1 056 931 88 40 8 4 3 23 23
2005 920 920 100 58 5 8 5 13 11
2009 1 112 889 80 67475108
2010 1 029 1 029 100 56463724
• 56 68 • 2011 1 119 914 82 64453149
Seychelles 1995 0 –
2000 0 –
2005 2 –
2009 0 0 –
2010 0 0 –
• 0 0 • 2011 0 0 –
Sierra Leone 1995 41 69 168 72 14 3441
2000 441 –
2005 330 328 99 68 7 6 3 15 1
2009 467 466 100 56 13 10 3 15 4
2010 547 543 99 65 11 5 2 15 2
• 87 70 • 2011 375 362 97 63 7 6 3 15 5
South Africa 1995 179 –
2000 56 202 24 847 44 438831919
2005 60 588 64 923 107 29 29 11 2 16 13
2009 65 916 34 122 52 53 8 10 3 12 15
2010 60 580 60 580 100 31 4 5 2 7 52
• 0 66 • 2011 45 915 31 168 68 59 7 9 3 12 10
Swaziland 1995 489 –
2000 1 249 –
2005 470 1 113 237 7 21 11 3 5 54
2009 1 474 1 474 100 14 41 17 9 10 8
2010 1 440 446 31 32 18 17 21 7 6
• 0 59 • 2011 1 149 1 151 100 12 46 15 8 5 13
Togo 1995 93 93 100 16 17 5 4 19 38
2000 133 –
2005 179 128 72 73 2 14 4 7 0
2009 214 237 111 68 3 18 3 4 5
2010 240 240 100 78 4 6 4 8 1
• 33 78 • 2011 213 210 99 75 3 8 4 8 1
Uganda 1995 955 –
2000 1 505 1 209 80 34 30 13 0 13 10
2005 2 430 –
2009 4 014 2 856 71 31 39 7 1 15 7
2010 3 952 2 764 70 31 34 8 1 12 13
• 0 71 • 2011 4 014 2 814 70 38 33 8 2 14 5
United Republic 1995 1 335 1 455 109 66 10 11 1 8 4
of Tanzania 2000 1 772 3 356 189 49 24 14 1 6 6
2005 5 032 5 067 101 37 39 13 1 4 6
2009 4 217 4 217 100 34 49 8 1 3 5
2010 3 785 3 714 98 37 47 9 1 3 4
• 76 82 • 2011 2 870 2 936 102 38 43 7 1 3 7
Zambia 1995 243 –
2000 1 455 894 61 52 15 11 4 5 12
2005 5 496 5 496 100 24 60 9134
2009 2 485 5 444 219 33 53 9140
2010 6 310 –
• 0 0 • 2011 6 636 –
Zimbabwe 1995 737 –
2000 1 063 – 51 14 17 1 8 9
2005 5 941 4 667 79 13 46 16 0 13 11
2009 4 685 1 203 26 72 8 11 0 5 4
2010 4 685 1 629 35 63 11 13 3 5 5
• 0 78 • 2011 4 345 1 772 41 63 15 11 4 4 3
a TREATMENT SUCCESS = percent cured + percent completed then rounded to the nearest digit.
172 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($+,9WHVWLQJDQGSURYLVLRQRI&37$57DQG,37–
% OF TB PATIENTS WITH
KNOWN HIV STATUS
2005–2012
YEAR
% OF TB
PATIENTS WITH
KNOWN HIV
STATUS
NUMBER OF TB
PATIENTS WITH
KNOWN HIV
STATUS
PATIENTS
NOTIFIED
(NEW AND
RETREAT)
NUMBER OF
HIV-POSITIVE
TB PATIENTS
% OF TESTED
TB PATIENTS
HIV-POSITIVE
% OF HIV-
POSITIVE TB
PATIENTS ON
CPT
% OF HIV-
POSITIVE TB
PATIENTS ON
ART
NUMBER OF
HIV-POSITIVE
PEOPLE
PROVIDED IPT
Algeria 2005 21 501
2010 22 530
2011 21 597
– – 2012 22 082
Angola 2005 38 317
2010 4.9 2 434 49 987 1 620 67 43 43
2011 10 5 107 48 926 789 15 100 100
– 23 • 2012 23 12 022 53 426 1 149 9.6 100 100 1 100
Benin 2005 15 503 3 457 57 11
2010 98 3 774 3 841 592 16 97 57
2011 99 4 259 4 320 727 17 98 74 339
• 15 98 • 2012 98 4 006 4 075 637 16
Botswana 2005 23 2 291 10 104 1 829 80 18 762
2010 81 6 147 7 632 4 018 65 79 43 738
2011 97 6 545 6 733 4 129 63 62 53
• 23 95 • 2012 95 5 940 6 223 3 759 63 90 65
Burkina Faso 2005 33 1 213 3 645 559 46 68 32
2010 93 4 761 5 135 839 18 98 60
2011 89 4 944 5 543 829 17 97 70
• 33 84 • 2012 84 4 567 5 405 671 15 96 75
Burundi 2005 6 627 0
2010 71 5 511 7 719 1 260 23 95 40
2011 71 4 817 6 828 1 036 22 95 48 0
– 82 • 2012 82 5 734 7 016 1 076 19 94 55
Cameroon 2005 0 0 22 073 0 0
2010 78 19 117 24 552 8 314 43 81 51
2011 81 20 280 25 126 7 731 38 87 62 1 373
• 0 82 • 2012 82 20 810 25 360 7 747 37 83 55
Cape Verde 2005 98 298 305 14 4.7 100
2010 365
2011 90 352 390 47 13
• 98 89 • 2012 89 378 425 45 12 44 98 123
Central African 2005 3 338
Republic 2010 39 2 638 6 760 862 33 0 62
2011 33 1 890 5 724 733 39 12 9.3
– 46 • 2012 46 3 839 8 283 1 483 39 28 20
Chad 2005 6 505
2010 39 3 801 9 697 663 17 53 45
2011 38 4 124 10 774 959 23 39 43
– 44 • 2012 44 4 766 10 800 960 20 65
Comoros 2005 100 112 112 2 1.8 100 100
2010 119 0 0 0
2011 3.4 4 119 4 100 100 100
• 100 3 • 2012 3.3 4 122 4 100 100 100 2
Congo 2005 9 961
2010 40 4 106 10 321 757 18 2.9 2.9
2011 20 2 247 11 143 687 31 24 26
– 17 • 2012 17 1 979 11 512 653 33 20 23
Côte d'Ivoire 2005 20 4 079 20 026 1 551 38 38 14
2010 73 16 991 23 210 4 112 24 80 27
2011 80 18 297 22 920 4 820 26 80 36
• 20 85 • 2012 85 20 663 24 222 5 482 27 75 44
Democratic 2005 1.9 1 885 99 558 386 20 74 0.78
Republic 2010 24 28 997 118 636 5 273 18 24 9.3
of the Congo 2011 27 30 636 114 290 4 942 16 54 23
• 2 31 • 2012 31 35 097 112 499 5 748 16 61 40
Equatorial 2005
Guinea 2010 92 786 853 225 29 85 31
2011 100 911 913 234 26 21
– – 2012
Eritrea 2005 3 612
2010 2 991
2011 3 093 0
– 59 • 2012 59 1 913 3 254 164 8.6
Ethiopia 2005 2.6 3 211 125 135 1 321 41 88 29 1 983
2010 43 66 955 156 928 9 809 15 69 39 6 636
2011 41 65 140 159 017 5 442 8.4 62 39 30 816
• 3 65 • 2012 65 96 245 147 592 9 819 10 37 82 30 395
Gabon 2005 7.1 185 2 611 185 100 100 0
2010 27 1 130 4 180 667 59 52 52
2011 46 2 252 4 916 578 26
• 7 100 • 2012 100 5 415 5 415 852 16 66
Gambia 2005 2 120
2010 97 1 962 2 030 224 11 93 46
2011 74 1 726 2 333
– 78 • 2012 78 1 859 2 387 302 16 97 48
Ghana 2005 7 844 12 124 340 40 100 37
2010 67 10 147 15 145 2 676 26 77 18
2011 79 12 587 15 840 2 907 23 72 28
• 7 78 • 2012 78 11 825 15 207 2 812 24 72 37
Guinea 2005 7 090
2010 51 5 776 11 324 1 483 26 87 41
2011 56 6 548 11 606 1 670 26 72 49
– 65 • 2012 65 7 575 11 641 1 859 25 83 49
Guinea-Bissau 2005 11 200 1 816 110 55 100 30 0
2010 46 1 046 2 259 396 38
2011 50 1 037 2 070 431 42 0 0
• 11 68 • 2012 68 1 322 1 950 517 39 0 0 0
Kenya 2005 14 15 658 108 401 8 954 57 44 17
2010 91 96 930 106 083 40 069 41 100 48
2011 93 97 136 103 981 38 175 39 97 64
• 14 94 • 2012 94 92 890 99 149 35 837 39 98 74
Lesotho 2005 1.4 156 11 404 127 81 79
2010 84 11 005 13 138 8 459 77 96 27
2011 89 11 413 12 785 8 519 75 95 68
• 1 88 • 2012 88 10 476 11 971 7 878 75 97 53 16 403
Liberia 2005 3.3 114 3 456 14 12 0
2010 53 3 533 6 668 283 8 8.5 0
2011 55 4 355 7 965 454 10 26 9.3
• 3 70 • 2012 70 5 661 8 132 772 14 90 15
Madagascar 2005 9 1 759 19 475 16 0.91
2010 65 16 439 25 106 39 0.24 36
2011 58 15 532 26 722 40 0.26
• 9 54 • 2012 54 14 146 26 209 19 0.13 95

173 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
AFRICAN REGION
7$%/($+,9WHVWLQJDQGSURYLVLRQRI&37$57DQG,37–
% OF TB PATIENTS WITH
KNOWN HIV STATUS
2005–2012
YEAR
% OF TB
PATIENTS WITH
KNOWN HIV
STATUS
NUMBER OF TB
PATIENTS WITH
KNOWN HIV
STATUS
PATIENTS
NOTIFIED
(NEW AND
RETREAT)
NUMBER OF
HIV-POSITIVE
TB PATIENTS
% OF TESTED
TB PATIENTS
HIV-POSITIVE
% OF HIV-
POSITIVE TB
PATIENTS ON
CPT
% OF HIV-
POSITIVE TB
PATIENTS ON
ART
NUMBER OF
HIV-POSITIVE
PEOPLE
PROVIDED IPT
Malawi 2005 44 12 243 27 610 8 447 69 92 49
2010 88 19 855 22 536 12 476 63 94 46
2011 83 17 334 20 854 10 341 60 89 60
• 44 93 • 2012 93 19 009 20 463 11 296 59 88 81 20 542
Mali 2005 4 884
2010 42 2 303 5 448 416 18 75 52 0
2011 35 1 963 5 573 404 21 72 69
– 28 • 2012 28 1 544 5 602 425 28 42 100
Mauritania 2005 0.45 10 2 218 0 0
2010 24 608 2 489 90 15 61 0
2011 0.66 12 1 820 12 100 100
• 0 – 2012 2 636
Mauritius 2005 91 115 127 2 1.7 100 50
2010 95 117 123 8 6.8 100 75
2011 93 108 116 8 7.4 100 62
• 91 96 • 2012 96 125 130 10 8 100 90
Mozambique 2005 33 718
2010 88 40 554 46 174 24 574 61 97 25 13 164
2011 91 43 096 47 452 26 538 62 91 29 17 064
– 94 • 2012 94 47 960 50 827 27 979 58 98 55 17 317
Namibia 2005 16 2 547 15 894 1 465 58
2010 76 9 534 12 625 5 227 55 93 44 13 989
2011 84 10 042 11 938 4 990 50 98 54 14 428
• 16 89 • 2012 89 9 927 11 145 4 688 47 99 72 11 906
Niger 2005 8 224 152 43 34
2010 48 4 925 10 345 405 8.2 37 0
2011 44 4 710 10 714 334 7.1 6.6 4.8
– 46 • 2012 46 5 166 11 207 431 8.3 31 16
Nigeria 2005 10 6 897 66 848 1 241 18
2010 79 71 844 90 447 17 736 25 59 33 1 750
2011 81 75 772 93 050 19 553 26 68 43 1 107
• 10 84 • 2012 84 82 641 97 853 19 342 23 80 56 2 257
Rwanda 2005 65 5 003 7 680 2 276 45 15 13
2010 98 6 914 7 065 2 199 32 97 72
2011 97 6 560 6 784 1 855 28 97 75
• 65 99 • 2012 99 6 131 6 208 1 601 26 99
Sao Tome and 2005 100 152 152 5 3.3 0 0
Principe 2010 92 112 122 13 12 92 54 0
2011 100 146 146 15 10 100 100 0
• 100 99 • 2012 99 126 127 18 14 100 100 0
Senegal 2005 10 120
2010 69 8 018 11 591 776 9.7 85 37
2011 76 8 757 11 588 877 10 85 48
– 78 • 2012 78 10 048 12 819 882 8.8 90 64 426
Seychelles 2005 14 2 100 100
2010 100 17 17 1 5.9 100 100 0
2011 100 21 21 4 19 75 100
– 100 • 2012 100 21 21 3 14 67 100 0
Sierra Leone 2005 6 930
2010 74 9 718 13 195 976 10 6.4 19
2011 78 10 159 12 943 902 8.9 25 28
– 87 • 2012 87 11 655 13 354 1 343 12 26 69 1 062
South Africa 2005 22 67 988 302 467 35 299 52 100 33 1 466
2010 54 213 006 396 554 128 457 60 74 54 146 247
2011 83 322 732 389 974 211 128 65 77 46 372 994
• 22 84 • 2012 84 294 196 349 582 190 093 65 74 54 369 747
Swaziland 2005 8 864
2010 86 9 536 11 146 7 788 82 93 35
2011 92 8 419 9 180 6 480 77 95 51
– 95 • 2012 95 7 363 7 739 5 666 77 98 66 1 934
Togo 2005 0 0 2 635 0 0
2010 77 2 242 2 897 632 28 72 49
2011 84 2 513 2 980 667 27 77 67
• 0 91 • 2012 91 2 657 2 912 625 24 87 76
Uganda 2005 25 10 555 41 809 7 523 71 25 10
2010 81 36 742 45 546 19 836 54 90 24
2011 80 39 394 49 018 20 725 53 93 32
• 25 86 • 2012 86 40 581 47 211 20 376 50 94 49
United Republic 2005 2.5 1 613 64 200 841 52 61 22
of Tanzania 2010 90 56 849 63 453 21 662 38 92 35
2011 88 53 842 61 148 20 632 38 95 38
• 3 82 • 2012 82 52 499 63 892 20 269 39 96 54
Zambia 2005 2 1 082 53 267 614 57 68
2010 84 40 704 48 616 26 571 65 75 48
2011 100 48 594 48 594 26 737 55 87 53
• 2 100 • 2012 100 45 269 45 277 24 309 54 93 60
Zimbabwe 2005 0 0 54 891 0
2010 86 41 062 47 557 31 849 78 88 45
2011 90 37 029 41 305 27 562 74 94 60 0
• 0 88 • 2012 88 34 212 38 720 23 957 70 26 18

174 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($7HVWLQJIRU0'57%DQGQXPEHURIFRQILUPHGFDVHVRI0'57%±
NEW PULMONARY CASES PREVIOUSLY TREATED CASES
YEAR
ESTIMATED CASES
OF MDR-TB AMONG
NOTIFIED
NUMBER OF
BACT+VE
b
TESTED FOR
MDR-TB
% OF
BACT+VE
b
TESTED FOR
MDR-TB
ESTIMATED CASES
OF MDR-TB AMONG
NOTIFIED
NUMBER OF
NOTIFIED
TESTED FOR
MDR-TB
% OF NOT IFIED
TESTED FOR
MDR-TB
Algeria 2005 74 809 9.1 164 23
2010 56 ––
2011 ––
2012 180 (69–290) 130 (56–250) – 52 (6.5–170) –
Angola 2005 ––
2010 3 ––
2011 40 29 0.13 –
2012 45 1 700 (780–2 500) 800 (44–1 500) – 860 (330–1 400) 45 1.0
Benin 2005 28 31 1.1 107 32
2010 15 103 3.5 6 2.9
2011 20 0 0 152 58
2012 25 54 (26–83) 17 (2.1–70) 26 0.78 37 (23–55) 110 39
Botswana 2005 ––
2010 106 488 11 286 27
2011 46 151 4.5 90 10
2012 53 140 (94–190) 120 (70–160) 349 14 29 (11–47) 149 34
Burkina Faso 2005 3 – 126 39
2010 31 1 <0.1 117 21
2011 42 1 <0.1 68 14
2012 38 150 (71–240) 79 (4.4–150) 7 0.20 75 (29–120) 72 19
Burundi 2005 ––
2010 24 22 0.48 2 0.60
2011 6 0 0 6 1.9
2012 24 150 (27–280) 120 (0.48–240) 1 <0.1 31 (11–52) 23 7.5
Cameroon 2005 ––
2010 35 0 0 35 2.3
2011 63 ––
2012 153 670 (140–1 200) 510 (2.0–1 000) 0 0 160 (57–270) 80 5.0
Cape Verde 2005 ––
2010 0 ––
2011 0 0 0 0 0
2012 0 9.8 (4.0–16) 6.1 (0.34–12) 0 0 3.6 (1.4–5.9) 0 0
Central African 2005 ––
Republic 2010 9 9 0.25 0 0
2011 15 0 0 56 16
2012 28 130 (36–220) 28 (0.72–160) – 97 (37–190) –
Chad 2005 ––
2010 3 0 0 0 0
2011 0 0 0 0 0
2012 0 320 (150–490) 160 (8.8–300) 0 0 160 (63–260) 0 0
Comoros 2005 ––
2010 ––
2011 0 ––
2012 0 2.5 (0.92–4.0) 1.7 (0.10–3.2) – 0.77 (0.30–1.2) –
Congo 2005 ––
2010 ––
2011 ––
2012 250 (43–450) 200 (0.79–400) – 49 (17–81) –
Côte d'Ivoire 2005 47 0 0 0 0
2010 50 0 0 72 4.7
2011 30 1 <0.1 29 2.0
2012 221 580 (270–890) 440 (190–850) 0 0 140 (49–240) 365 26
Democratic 2005 ––
Republic 2010 87 – 100 1.2
of the Congo 2011 121 22 <0.1 160 2.0
2012 81 2 900 (670–5 100) 2 100 (8.4–4 200) 12 <0.1 760 (260–1 300) 95 1.3
Equatorial 2005 ––
Guinea 2010 0 0 0 0 0
2011 3 ––
2012 – – – – –
Eritrea 2005 ––
2010 ––
2011 11 ––
2012 0 79 (38–120) 35 (1.9–66) – 44 (17–71) –
Ethiopia 2005 ––
2010 140 42 <0.1 510 10
2011 212 73 0.15 139 3.0
2012 284 2 000 (1 200–2 900) 1 600 (830–2 700) 469 0.99 480 (230–870) 180 4.4
Gabon 2005 ––
2010 0 ––
2011 ––
2012 0 170 (57–280) 100 (0.41–200) – 67 (23–110) –
Gambia 2005 ––
2010 0 ––
2011 0 ––
2012 0 9.9 (0–29) 9.9 (0.25–54) 0 0 0 (0–26) 0 0
Ghana 2005 1 50 0.62 2 0.38
2010 4 –212.1
2011 7 0 0 61 6.9
2012 20 390 (170–620) 240 (13–440) 0 0 160 (61–260) 44 5.3
Guinea 2005 20 215 3.9 34 7.4
2010 31 5 <0.1 26 4.0
2011 78 8 0.12 26 3.9
2012 69 250 (130–380) 47 (9.8–140) – 200 (100–340) –
Guinea-Bissau 2005 ––
2010 ––
2011 2 ––
2012 6 45 (15–75) 33 (1.8–63) – 12 (4.6–19) –
Kenya 2005 44 0 0 1829 20
2010 112 – 706 6.7
2011 166 92 0.25 1195 12
2012 225 2 800 (840–4 800) 1 800 (7.4–3 700) 78 0.21 980 (340–1 600) 1183 12
Lesotho 2005 ––
2010 117 ––
2011 64 ––
2012 46 170 (36–300) 77 (16–220) 5 0.15 94 (20–260) 28 1.7
Liberia 2005 ––
2010 0 0 0 0 0
2011 ––
2012 6 130 (32–230) 110 (6.3–210) – 18 (7.0–29) –
Madagascar 2005 ––
2010 3 60 0.36 24 1.1
2011 9 9 <0.1 64 2.9
2012 10 170 (32–310) 94 (26–240) 7 <0.1 76 (9.3–260) 63 3.2
TOTAL
CONFIRMED
CASES OF
MDR-TB
a
ESTIMATED CASES
OF MDR-TB AMONG
NOTIFIED
a
TOTAL CONFIRMED CASES OF MDR-TB includes cases with unknown previous treatment history (i.e. not included under NEW CASES or PREVIOUSLY TREATED CASES).
b
BACT+VE = bacteriologically positive cases.

175 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
AFRICAN REGION
7$%/($7HVWLQJIRU0'57%DQGQXPEHURIFRQILUPHGFDVHVRI0'57%±
NEW PULMONARY CASES PREVIOUSLY TREATED CASES
YEAR
ESTIMATED CASES
OF MDR-TB AMONG
NOTIFIED
NUMBER OF
BACT+VE
b
TESTED FOR
MDR-TB
% OF
BACT+VE
b
TESTED FOR
MDR-TB
ESTIMATED CASES
OF MDR-TB AMONG
NOTIFIED
NUMBER OF
NOTIFIED
TESTED FOR
MDR-TB
% OF NOT IFIED
TESTED FOR
MDR-TB
TOTAL
CONFIRMED
CASES OF
MDR-TB
a
ESTIMATED CASES
OF MDR-TB AMONG
NOTIFIED
Malawi 2005 9 – 917 29
2010 40 871 10 449 20
2011 26 102 1.5 552 26
2012 27 96 (45–150) 56 (18–130) 0 0 40 (27–57) 27 3.3
Mali 2005 2 0 0 0 0
2010 12 0 0 12 3.4
2011 10 ––
2012 12 140 (60–210) 76 (4.2–140) 23 0.62 60 (23–96) 39 13
Mauritania 2005 11 161 12 30 15
2010 35 ––
2011 8 3 0.30 4 3.9
2012 1 59 (26–92) 34 (1.9–64) 1 <0.1 25 (9.8–41) –
Mauritius 2005 0 114 100 3 60
2010 2 105 100 7 100
2011 1 100 100 5 100
2012 0 0 (0–0) 0 (0–3.6) 121 100 0 (0–2.4) 4 100
Mozambique 2005 115 113 0.63 305 16
2010 165 80 0.39 251 6.2
2011 283 206 1.1 443 10
2012 266 2 000 (1 300–2 700) 1 400 (900–2 000) 205 0.98 540 (0–1 100) 243 5.4
Namibia 2005 ––
2010 214 ––
2011 192 ––
2012 210 630 (510–750) 260 (190–350) – 370 (290–470) –
Niger 2005 ––
2010 39 0 0 47 7.0
2011 18 1 <0.1 21 3.6
2012 35 270 (110–420) 160 (9.0–300) 0 0 110 (42–180) 35 6.2
Nigeria 2005 ––
2010 21 27 <0.1 19 0.21
2011 95 12 <0.1 76 0.86
2012 107 3 600 (2 700–4 500) 2 500 (1 800–3 400) 11 <0.1 1 100 (770–1 500) 94 1.2
Rwanda 2005 35 57 1.4 0 0
2010 90 171 4.0 431 68
2011 76 ––
2012 58 240 (170–310) 180 (120–270) – 63 (51–76) –
Sao Tome and 2005 ––
Principe 2010 0 ––
2011 4 2 1.9 2 12
2012 8 15 (11–19) 1.7 (0.10–3.3) 16 27 13 (7.1–15) 8 53
Senegal 2005 ––
2010 38 41 0.53 66 6.4
2011 50 14 0.18 97 8.7
2012 27 400 (170–620) 220 (70–500) 25 0.30 180 (76–340) 113 10
Seychelles 2005 ––
2010 0 ––
2011 0 0 0 1 –
2012 0 0 (0–0) 0 (0–3.9) 14 82 0 (0–1.7) 2 100
Sierra Leone 2005 ––
2010 ––
2011 8 ––
2012 220 (0–460) 100 (2.7–570) – 120 (26–280) –
South Africa 2005 2000 ––
2010 7386 ––
2011 10085 ––
2012 15419 8 100 (6 900–9 400) 4 600 (3 700–5 800) – 3 500 (2 800–4 300) –
Swaziland 2005 ––
2010 326 148 2.9 505 35
2011 332 ––
2012 280 730 (560–890) 430 (270–590) – 290 (250–340) –
Togo 2005 ––
2010 2 ––
2011 4 86 4.1 83 39
2012 2 77 (35–120) 41 (2.3–78) 0 0 36 (14–58) 2 1.1
Uganda 2005 46 ––
2010 93 358 1.5 356 9.0
2011 71 316 1.2 360 9.0
2012 89 1 000 (660–1 300) 540 (230–860) 196 0.79 470 (260–750) 748 19
United Republic 2005 10 276 0.60 405 8.0
of Tanzania 2010 34 201 0.44 246 6.5
2011 68 83 0.34 17 0.59
2012 42 500 (13–1 000) 500 (140–1 300) 639 2.5 0 (0–160) 108 3.9
Zambia 2005 ––
2010 ––
2011 ––
2012 80 620 (290–940) 98 (12–350) – 520 (260–900) –
Zimbabwe 2005 ––
2010 17 ––
2011 118 0 0 0 0
2012 149 930 (430–1 400) 570 (300–960) 360 3.0 360 (76–970) 258 6.0
a
TOTAL CONFIRMED CASES OF MDR-TB includes cases with unknown previous treatment history (i.e. not included under NEW CASES or PREVIOUSLY TREATED CASES).
b
BACT+VE = bacteriologically positive cases.

176 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($1HZVPHDUSRVLWLYHFDVHQRWLILFDWLRQE\DJHDQGVH[±
MALE FEMALE
YEAR 0–14 15–24 25–34 35–44 45–54 55–64 65+
UN-
KNOWN
0–14 15–24 25–34 35–44 45–54 55–64 65+
UN-
KNOWN
MALE:FEMALE
RATIO
Al
g
eria 1995
–
2000 59 927 1 516 610 491 234 299 36 1 005 1 293 746 314 208 312 1.1
2005 53 1 309 1 841 919 473 314 426 102 1 044 820 389 270 229 465 1.6
2010 52 1 203 1 669 825 513 392 397 79 1 086 826 417 251 222 367 1.6
2011 42 1 147 1 513 881 483 345 347 58 1 050 787 383 211 202 341 1.6
2012 29 1 102 1 467 857 464 354 349 0 60 917 773 382 198 229 329 0 1.6
An
g
ola 1995 386 724 562 346 224 155 14 371 707 443 264 248 130 18 1.1
2000 186 999 1 003 912 482 312 194 247 1 142 1 091 844 417 200 120 1.0
2005 520 2 549 2 797 1 918 1 255 665 461 704 2 926 2 682 1 797 1 138 581 417 0.99
2010 448 2 900 3 584 2 415 1 424 691 355 558 2 763 2 594 1 688 958 482 286 1.3
2011 501 3 000 3 792 2 386 1 395 680 455 0 708 2 731 2 563 1 683 1 006 457 346 0 1.3
2012 390 2 804 3 627 2 529 1 427 732 424 592 2 501 2 540 1 617 1 028 529 384 1.3
Benin 1995 14 186 352 306 176 101 92 26 148 197 118 69 32 22 2.0
2000 19 277 428 327 213 103 74 36 239 275 149 76 45 25 1.7
2005 21 306 595 396 270 135 87 25 249 331 145 89 51 39 1.9
2010 18 314 631 443 267 164 85 0 29 265 382 200 98 42 35 0 1.8
2011 21 320 650 497 353 210 107 0 41 288 385 246 119 42 52 0 1.8
2012 23 314 595 524 329 179 121 0 39 264 346 221 105 65 46 0 1.9
Botswana 1995
–
2000 25 185 605 488 267 135 96 37 335 469 262 98 57 36 1.4
2005 27 260 563 506 272 135 97 45 321 491 253 97 55 48 1.4
2010 45 256 590 477 239 137 107 0 68 338 509 301 119 56 53 0 1.3
2011 36 220 464 354 206 110 94 0 65 286 421 211 105 48 49 0 1.3
2012 40 207 394 333 190 79 75 0 63 267 402 193 109 43 31 0 1.2
Burkina Faso 1995 4 67 133 124 62 48 29 7 76 53 39 26 11 10 2.1
2000 12 91 274 252 133 68 65 7 59 128 101 45 38 14 2.3
2005 18 181 430 370 273 144 113 15 125 248 174 109 54 40 2.0
2010 20 231 620 493 328 224 173 0 33 158 259 198 124 97 83 0 2.2
2011 22 265 708 582 375 262 196 0 31 163 277 221 146 110 92 0 2.3
2012 25 277 769 631 423 250 198 0 27 160 288 191 156 106 82 0 2.5
Burundi 1995 5 128 238 224 73 32 19 19 109 124 89 33 12 4 1.8
2000
–
2005 34 352 591 525 372 111 55 46 298 399 288 122 36 33 1.7
2010 56 481 773 651 570 270 157 0 78 390 421 332 225 99 87 0 1.8
2011 37 484 743 620 504 235 98 0 56 345 374 263 180 81 40 0 2.0
2012 45 447 801 667 461 233 103 0 74 338 367 283 162 64 30 0 2.1
Cameroon 1995 20 208 569 323 287 204 164 9 185 313 223 153 106 93 1.6
2000 41 518 842 584 284 130 75 63 368 530 293 139 60 33 1.7
2005 134 1 472 2 482 1 766 1 035 463 289 226 1 467 1 788 1 028 503 205 143 1.4
2010 106 1 497 2 750 1 996 1 314 559 329 172 1 474 2 031 1 121 642 290 183 1.4
2011 114 1 580 2 931 2 139 1 283 625 361 178 1 461 2 022 1 177 581 281 194 1.5
2012 108 1 597 2 900 2 182 1 304 658 375 184 1 417 2 053 1 177 579 295 187 1.5
Ca
p
e Verde 1995
–
2000
–
2005 0 22 23 26 9 2 8 2 9 16 4536 2.0
2010
–
2011 0 17 43 35 31 3 3 0 4 14 15 46340 2.6
2012 0 29 36 34 24 8 2 0 0 19 13 98340 2.4
Central African 1995 38 162 356 206 120 40 18 39 233 350 145 57 21 9 1.1
Re
p
ublic 2000
–
2005 29 40 1 136 160 26 35 15 30 32 420 145 30 40 15 2.0
2010 78 379 633 468 251 135 63 1 88 367 576 319 155 73 44 8 1.2
2011 70 362 576 467 269 119 59 4 96 382 530 289 162 62 26 6 1.2
2012 73 502 799 660 360 158 92 101 511 689 370 191 96 39 1.3
Chad 1995
–
2000
–
2005 25 194 535 409 229 123 82 28 148 298 211 148 59 27 1.7
2010 76 382 850 666 379 173 99 0 59 274 413 263 158 79 44 0 2.0
2011 92 469 951 764 418 184 121 0 84 296 438 298 166 109 44 0 2.1
2012 68 405 842 634 376 210 88 0 51 273 403 227 135 91 46 0 2.1
Comoros 1995 0 18 13 9784 11398652 1.3
2000 0 18 7 14 9 3 4 1 9 6 12 1 2 1 1.7
2005 0 12 96424 21074838 0.88
2010
–
2011 0 10 13 95250 28421010 2.4
2012 9 15 8466 1575113 2.1
Con
g
o 1995 16 265 409 221 73 44 15 17 296 353 167 61 38 11 1.1
2000
–
2005
–
2010 41 435 672 424 203 77 55 49 409 510 296 152 70 56 1.2
2011 58 453 705 462 222 80 76 0 72 408 463 332 200 88 97 0 1.2
2012 46 563 716 519 276 113 72 63 438 482 349 171 68 108 1.4
Côte d'Ivoire 1995 41 989 2 092 1 344 759 283 130 99 810 813 497 273 105 19 2.2
2000
–
2005 128 1 346 2 449 1 606 888 422 385 193 1 280 1 756 989 528 232 201 1.4
2010 159 1 751 2 858 1 882 1 010 505 375 246 1 431 1 819 1 051 531 304 209 1.5
2011 189 1 743 3 043 1 852 1 072 601 348 0 244 1 358 1 838 1 044 560 301 223 0 1.6
2012 163 1 743 3 087 2 017 1 032 552 430 204 1 306 1 870 1 120 536 337 263 1.6
Democratic 1995 373 1 572 2 382 1 890 1 184 634 289 331 1 223 1 532 1 232 863 427 137 1.4
Re
p
ublic 2000 485 4 048 5 833 4 151 2 549 1 295 602 718 4 422 5 146 3 309 1 724 855 351 1.1
of the Con
g
o 2005 1 321 6 675 9 808 7 577 5 022 2 637 1 499 1 695 7 570 8 501 5 832 3 898 2 054 951 1.1
2010 1 707 6 859 10 412 9 134 6 464 3 641 1 907 1 987 7 199 9 120 6 721 4 579 2 612 1 311 1.2
2011 1 579 6 640 9 872 8 932 6 415 3 584 1 911 1 800 6 802 8 742 6 541 4 537 2 671 1 295 1.2
2012 1 439 6 612 10 274 9 361 6 612 3 698 1 941 1 699 6 598 8 406 6 471 4 131 2 625 1 257 1.3
E
q
uatorial 1995 8 15 45 37 15 11 7 2 18 28 20 4 7 1 1.7
Guinea 2000
–
2005
–
2010 10 71 80 59 35 16 10 0 13 80 57 45 26 9 6 0 1.2
2011 11 77 90 89 59 22 12 0 15 76 81 46 21 9 3 0 1.4
2012
–
Eritrea 1995
–
2000 9 70 75 57 32 25 20 10 100 87 71 21 12 8 0.93
2005 9 68 73 50 45 51 39 8 67 127 72 39 21 18 0.95
2010 10 93 109 81 51 37 60 0 3 88 111 79 43 31 36 0 1.1
2011 0
–
2012 2 84 105 90 62 39 51 0 4 86 98 74 45 19 20 0 1.3
Ethio
p
ia 1995 247 1 221 1 017 541 276 142 51 283 908 781 382 152 64 15 1.4
2000 915 5 095 5 187 3 082 1 495 610 397 1 037 4 699 4 424 2 105 976 366 122 1.2
2005 1 109 6 726 6 181 3 454 1 985 1 027 475 1 326 5 885 5 663 2 730 1 296 513 155 1.2
2010 1 582 7 400 7 785 4 451 2 746 1 473 822 1 608 5 708 6 480 3 439 1 950 855 335 1.3
2011 1 847 7 835 9 246 3 881 2 771 1 218 771 1 983 6 570 7 917 3 069 1 564 719 303 1.2
2012
–
Gabon 1995 3 45 74 80 54 30 15 9 47 54 28 25 19 3 1.6
2000
–
2005 13 123 199 140 70 38 25 19 128 123 88 29 29 18 1.4
2010 15 145 223 208 130 89 91 0 13 110 164 122 100 86 64 0 1.4
2011 34 240 269 229 144 86 66 0 25 177 188 125 74 44 39 0 1.6
2012 42 236 286 228 166 101 41 0 29 185 165 109 78 45 34 0 1.7
Gambia 1995 3 68 181 88 72 29 24 4 39 61 44 25 12 8 2.4
2000
–
2005 13 133 292 206 62 53 44 2 84 87 64 38 22 27 2.5
2010 9 194 314 184 141 68 39 0 6 104 121 71 35 40 18 0 2.4
2011 14 183 271 181 136 87 56 0 16 103 112 88 63 32 33 0 2.1
2012 7 210 331 191 107 80 54 0 16 123 106 89 41 32 42 0 2.2

177 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
AFRICAN REGION
7$%/($1HZVPHDUSRVLWLYHFDVHQRWLILFDWLRQE\DJHDQGVH[±
MALE FEMALE
YEAR 0–14 15–24 25–34 35–44 45–54 55–64 65+
UN-
KNOWN
0–14 15–24 25–34 35–44 45–54 55–64 65+
UN-
KNOWN
MALE:FEMALE
RATIO
Ghana 1995 42 223 397 398 302 190 112 40 199 272 205 122 88 48 1.7
2000 73 550 1 266 1 115 811 495 426 74 456 791 566 338 179 176 1.8
2005 49 592 1 201 1 311 944 462 414 68 450 693 527 366 207 221 2.0
2010 63 570 1 146 1 301 1 030 540 447 64 446 667 560 369 204 249 2.0
2011 50 550 1 127 1 328 955 491 456 52 470 699 614 390 174 260 1.9
2012 30 559 1 051 1 271 921 512 462 51 418 563 468 332 188 271 2.1
Guinea 1995 18 244 538 357 189 98 61 28 202 255 153 64 37 19 2.0
2000 39 551 860 570 282 203 103 66 314 446 245 114 82 45 2.0
2005 51 749 1 165 778 463 195 130 65 594 583 354 203 94 55 1.8
2010 61 679 877 982 876 565 289 51 549 739 751 405 145 72 1.6
2011 45 1 051 1 537 955 541 293 197 85 709 688 432 219 109 73 2.0
2012 28 761 1 104 791 383 190 120 0 49 505 509 323 134 61 57 0 2.1
Guinea-Bissau 1995
–
2000 2 52 92 80 64 39 19 4 30 46 47 24 15 12 2.0
2005 14 116 167 153 130 72 42 13 78 110 92 82 44 19 1.6
2010 18 164 219 183 141 80 43 0 30 100 161 133 80 38 19 0 1.5
2011 6 140 230 181 104 65 36 0 12 119 122 90 56 44 25 0 1.6
2012 7 145 262 183 115 63 38 10 7 121 157 98 56 33 25 4 1.6
Ken
y
a 1995 154 2 072 3 073 1 675 920 485 296 187 1 802 1 759 741 411 242 117 1.6
2000 264 3 739 6 653 3 548 1 630 630 414 416 3 916 4 363 1 874 831 347 148 1.4
2005 359 4 790 8 832 5 069 2 521 1 031 590 577 5 144 6 521 2 781 1 266 593 315 1.3
2010 357 4 698 7 945 5 077 2 509 994 658 549 4 044 5 112 2 372 1 056 544 345 1.6
2011 356 4 773 8 376 5 201 2 660 1 045 665 0 629 4 183 4 917 2 434 1 025 477 344 0 1.6
2012 393 4 893 8 149 5 302 2 493 1 099 669 603 4 097 4 975 2 363 993 529 379 1.6
Lesotho 1995 9 108 214 256 189 96 88 14 106 125 71 49 17 19 2.4
2000 8 165 458 517 395 198 76 11 222 336 195 83 36 29 2.0
2005 32 395 695 397 148 82 37 19 226 721 616 494 297 121 0.72
2010 16 222 607 497 364 244 133 27 283 597 329 169 64 48 1.4
2011 19 179 584 493 329 245 121 23 311 572 307 185 84 58 1.3
2012 15 204 580 427 295 196 114 0 30 309 571 296 143 71 47 0 1.2
Liberia 1995
–
2000 12 133 196 127 52 17 26 21 140 149 88 28 16 16 1.2
2005 26 240 352 333 155 74 65 37 232 297 171 108 52 25 1.4
2010 90 338 621 510 295 114 21 0 254 339 488 259 171 151 99 0 1.1
2011 67 382 595 727 440 194 87 0 67 329 433 517 285 88 50 0 1.4
2012 65 382 627 667 406 129 83 0 61 354 535 605 292 79 57 0 1.2
Mada
g
ascar 1995 79 791 1 289 1 173 630 423 242 100 799 1 108 744 340 230 78 1.4
2000
–
2005 98 1 159 1 867 1 732 1 349 582 333 150 1 012 1 451 1 047 614 248 129 1.5
2010 204 1 721 1 621 2 525 1 782 960 485 0 323 1 621 1 943 1 376 946 397 192 0 1.4
2011 146 1 807 2 764 2 495 1 938 1 044 522 0 252 1 726 2 031 1 503 978 462 188 0 1.5
2012 177 1 725 2 474 2 460 1 927 1 059 490 0 242 1 720 1 848 1 420 914 474 199 0 1.5
Malawi 1995 25 493 1 195 833 519 215 89 65 802 1 028 573 294 108 45 1.2
2000 50 653 1 476 1 113 585 245 114 66 1 038 1 481 831 401 148 64 1.1
2005 58 622 1 653 1 031 549 279 157 84 913 1 598 859 386 180 74 1.1
2010 50 565 1 509 985 485 275 187 0 103 610 1 196 661 314 198 102 0 1.3
2011 70 519 1 486 1 050 440 238 201 79 601 1 119 660 283 161 96 1.3
2012 52 495 1 537 1 051 471 292 204 0 71 538 1 057 609 298 156 120 0 1.4
Mali 1995 27 72 357 294 181 138 102 31 132 184 128 107 61 52 1.7
2000 23 206 430 396 297 235 144 14 174 232 152 106 75 43 2.2
2005 26 350 628 539 365 263 193 33 208 348 245 152 101 72 2.0
2010 94 381 707 526 354 227 207 31 265 337 247 144 96 70 2.1
2011 25 370 772 515 352 267 230 42 255 393 223 147 118 68 2.0
2012 25 405 731 547 377 257 211 0 34 253 344 239 137 89 77 0 2.2
Mauritania 1995
–
2000
–
2005
–
2010 17 192 295 206 137 99 76 14 90 104 82 52 29 29 2.6
2011 36 165 185 131 106 58 55 28 68 72 47 36 19 20 2.5
2012 22 204 302 195 139 114 114 25 112 81 88 73 46 28 2.4
Mauritius 1995 2 17 13 22 27 13 8 2 4 12 10 8 4 4 2.3
2000 2 6 9 18 19 14 8 1588674 1.9
2005 10 15 21 20 10 6 4 5 5 11 2 1 2.9
2010 0 9 9 13 23 15 7 0 07944320 2.6
2011 0 10 13 9 17 10 8 0 0 7 12 2 3 6 3 0 2.0
2012 2 11 14 16 17 11 7 0 0 11 782840 2.0
Mozambi
q
ue 1995 187 1 136 1 475 1 338 1 022 664 320 226 994 1 314 1 016 551 234 89 1.4
2000
–
2005
–
2010
–
2011
–
2012
–
Namibia 1995 0 68 235 113 55 21 6 5 49 78 50 16 1 0 2.5
2000 18 269 874 665 300 147 81 16 352 654 348 161 76 52 1.4
2005 98 355 1 027 874 365 146 120 105 399 809 525 213 95 91 1.3
2010 36 359 852 680 287 146 126 67 429 685 382 206 122 87 1.3
2011 48 337 844 660 361 152 138 78 427 653 410 185 100 110 1.3
2012 61 358 810 686 292 157 137 81 394 582 396 198 84 97 1.4
Ni
g
er 1995
–
2000 29 270 174 441 252 151 78 31 123 206 168 151 63 9 1.9
2005 35 557 1 204 819 497 350 198 34 214 388 330 223 131 70 2.6
2010 44 669 1 587 988 615 415 342 39 272 418 347 238 174 135 2.9
2011 50 709 1 673 1 025 646 436 347 0 50 285 449 323 278 189 147 0 2.8
2012 40 702 1 752 1 133 747 444 360 0 48 260 485 302 237 214 124 0 3.1
Ni
g
eria 1995 450 845 921 937 557 611 515 404 842 795 770 724 654 451 1.0
2000 157 2 173 3 164 1 836 1 091 566 463 239 2 934 2 434 1 110 676 344 231 1.2
2005 325 3 824 6 758 4 544 2 863 1 464 950 482 3 996 4 884 2 448 1 350 745 415 1.4
2010 521 4 457 9 186 6 218 3 804 1 974 1 363 0 595 4 182 6 117 3 431 1 846 1 040 682 0 1.5
2011 529 4 549 9 520 6 550 4 230 2 248 1 443 0 578 4 198 6 168 3 574 2 014 1 112 724 0 1.6
2012 538 5 026 10 382 7 684 4 589 2 449 1 686 0 649 4 652 6 762 4 084 2 243 1 290 867 0 1.6
Rwanda 1995
–
2000 155 466 974 824 393 129 56 105 396 473 309 109 52 14 2.1
2005 45 494 713 592 408 142 71 73 483 442 262 157 60 29 1.6
2010 48 430 741 526 325 202 126 48 399 448 261 128 65 38 1.7
2011 42 423 795 500 376 210 124 0 50 358 398 235 146 87 67 0 1.8
2012 22 375 768 519 341 214 123 40 327 393 208 116 66 59 2.0
Sao Tome and 1995
–
Princi
p
e 2000 1 5 11 4 7 3 10 3 7 15 5 7 4 15 0.73
2005 2576452 1453230 1.7
2010 0 10 14 71010 05432000 2.4
2011 05987140 221041000 1.8
2012 1 6 11 88200 061060010 1.6
Sene
g
al 1995 94 717 1 219 813 408 300 213 84 428 461 283 203 126 72 2.3
2000 60 772 1 297 857 470 279 189 77 521 540 376 217 107 61 2.1
2005 71 1 050 1 561 904 533 274 236 83 709 568 351 185 116 81 2.2
2010 81 1 351 1 793 972 590 329 221 0 81 835 643 332 217 136 105 0 2.3
2011 75 1 264 1 835 981 582 335 214 89 88 807 664 362 208 144 74 43 2.2
2012 84 1 454 2 036 1 121 597 365 224 0 125 836 715 383 263 155 90 0 2.3
Se
y
chelles 1995 0201121 0010001 3.5
2000 2411 1011 2.7
2005 0212100 0011000 3.0
2010 00060010 02000000 3.5
2011 00100000 01000000 1.0
2012 00122000 00111010 1.2

GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($1HZVPHDUSRVLWLYHFDVHQRWLILFDWLRQE\DJHDQGVH[±
MALE FEMALE
YEAR 0–14 15–24 25–34 35–44 45–54 55–64 65+
UN-
KNOWN
0–14 15–24 25–34 35–44 45–54 55–64 65+
UN-
KNOWN
MALE:FEMALE
RATIO
Sierra Leone 1995 10 184 305 201 99 47 22 18 165 193 110 65 24 11 1.5
2000 18 287 486 361 190 113 47 27 249 298 225 92 49 30 1.5
2005 45 490 792 651 397 226 124 54 393 518 312 207 114 47 1.7
2010 64 718 1 176 1 076 663 320 254 77 648 742 556 293 180 131 1.6
2011 75 825 1 224 1 099 781 334 287 0 115 678 796 543 343 219 116 0 1.6
2012 70 858 1 324 1 213 841 416 274 80 703 861 667 391 201 132 1.6
South Africa 1995
–
2000 116 723 1 999 2 135 1 146 435 212 122 1 283 1 716 933 423 167 80 1.4
2005 2 035 10 422 20 576 19 465 11 143 4 124 1 705 2 561 13 632 19 343 11 338 5 416 2 352 1 348 1.2
2010 1 496 9 925 20 855 19 842 12 386 5 155 2 211 0 1 933 13 023 20 205 12 910 6 873 3 165 2 128 0 1.2
2011 1 472 9 772 20 487 19 360 12 111 5 220 2 164 0 1 932 12 751 19 250 12 807 6 955 3 266 2 223 0 1.2
2012 1 132 9 074 19 894 18 510 11 331 5 054 2 085 0 1 545 11 547 17 452 11 430 5 939 2 846 2 059 0 1.3
Swaziland 1995 4 59 117 130 98 40 16 5 52 57 39 29 8 6 2.4
2000 11 130 352 249 138 37 17 10 198 298 62 62 24 5 1.4
2005 9 162 406 285 139 57 27 14 318 453 207 73 21 8 0.99
2010 30 207 537 369 192 109 50 0 51 354 662 276 104 54 16 0 0.98
2011 16 161 459 318 158 69 46 35 281 495 220 86 40 24 1.0
2012 18 163 479 332 168 84 38 0 39 284 535 242 88 51 27 0 1.0
To
g
o 1995 7 95 151 123 82 64 49 9 80 96 45 38 23 15 1.9
2000 4 101 168 144 109 48 39 13 107 124 50 36 24 15 1.7
2005 11 177 320 283 125 79 69 23 157 236 146 67 41 32 1.5
2010 21 150 350 358 217 116 80 39 163 285 148 78 62 29 1.6
2011 15 169 340 350 234 123 85 0 11 167 277 146 89 50 38 0 1.7
2012 9 171 338 341 237 121 87 17 165 287 154 109 48 28 1.6
U
g
anda 1995 370 1 193 2 491 1 797 1 115 602 323 402 1 376 1 845 1 104 635 312 113 1.4
2000 283 1 511 3 497 2 479 1 279 607 395 400 1 649 2 782 1 510 671 316 163 1.3
2005 257 1 598 4 075 3 209 1 576 725 539 371 1 811 3 099 1 800 818 389 257 1.4
2010 268 2 055 4 735 4 133 2 214 905 613 16 401 1 964 2 923 1 691 924 365 248 1 1.8
2011 295 2 075 5 044 4 613 2 466 994 604 423 400 2 092 2 853 1 809 973 409 313 252 1.8
2012 272 2 174 5 029 4 493 2 479 1 015 633 21 364 2 194 2 912 1 733 864 419 281 33 1.8
United Re
p
ublic 1995 183 2 108 4 091 2 916 1 754 1 007 640 201 1 904 2 532 1 324 735 380 179 1.8
of Tanzania 2000 200 2 357 4 836 3 430 2 022 1 202 834 257 2 106 3 426 1 738 868 494 269 1.6
2005 190 2 062 4 939 4 025 2 310 1 279 1 054 271 1 852 3 521 1 892 968 547 354 1.7
2010 232 1 975 4 493 4 141 2 427 1 309 1 161 0 248 1 689 2 988 2 013 1 044 578 471 0 1.7
2011 190 1 975 4 405 4 073 2 402 1 211 1 127 221 1 660 2 896 2 140 944 490 381 1.8
2012 208 2 086 4 707 4 397 2 435 1 293 1 114 282 1 651 2 906 2 108 1 022 507 422 1.8
Zambia 1995 91 659 1 668 1 124 487 231 130 129 1 125 1 779 717 257 117 63 1.0
2000 349 2 175 2 610 3 045 435 261 174 150 932 1 118 1 305 186 112 75 2.3
2005 135 1 240 3 166 2 160 917 358 321 168 1 507 2 463 1 433 569 235 185 1.3
2010
–
2011 105 1 033 2 897 2 194 810 280 207 151 940 1 683 1 063 422 162 99 1.7
2012 141 1 003 3 088 2 412 846 319 220 180 1 024 1 646 1 077 376 189 124 1.7
Zimbabwe 1995
–
2000
–
2005 210 837 2 264 1 855 762 295 656 269 1 136 2 242 1 255 578 193 603 1.1
2010 150 710 2 208 1 682 761 350 252 0 173 974 2 185 1 283 490 265 171 0 1.1
2011 152 784 2 467 2 071 780 377 278 0 174 1 084 2 161 1 386 448 274 160 0 1.2
2012 120 783 2 421 2 086 796 360 271 0 173 939 2 053 1 286 483 231 161 0 1.3

179 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
AFRICAN REGION
a LED = Light emitting diode microscopes
b DST = Drug susceptibility testing
c LPA = Line probe assay
d NRL = National Reference Laboratory
7$%/($/DERUDWRULHV173VHUYLFHVGUXJPDQDJHPHQWDQGLQIHFWLRQFRQWURO
LABORATORIES FREE THROUGH NTP
SMEAR LABS
PER 100K
POPULATION
% OF SMEAR
LABS USING
LED
a
CULTURE
LABS PER 5M
POPULATION
DST
b
LABS
PER 5M
POPULATION
LPA
c
LABS
PER 5M
POPULATION
NUMBER OF
LABS USING
XPERT MTB/RIF
SECOND-
LINE DST
AVAILABLE
NRL
d
TB DIAGNOSIS
FIRST-
LINE
DRUGS
Algeria 0.6 0 3.8 0.3 0.1 1 In country Yes Yes (all suspects) Yes Yes
Angola 0.6 – 0.5 0.5 Yes Yes (all suspects) Yes No 1 870
Benin 0.8 9 0.5 0.5 0.5 1 In and out
of countr y Yes Yes
(
if TB is
confirmed) Yes Yes
Botswana 2.6 21 2.5 2.5 2.5 5 Out of
country Yes Yes (all suspects) Yes Yes
Burkina Faso 0.7 0 0.3 0.3 0.9 0 Out of
country Yes Yes (all suspects) Yes Yes
Burundi 1.7 9 0.5 0 0 0 Out of
country Yes Yes (all suspects) Yes Yes
Cameroon 1.1 4 0.9 0.5 0.5 1 In country Yes No Yes Yes
Cape Verde 3.2 0 0 0 0 1 Out of
country Yes Yes (all suspects) Yes Yes
Central African Republic 1.6 0 1.1 1.1 0 1 No Yes Yes (all suspects) Yes Yes
Chad 0.6 0 0 0 0 0 No Yes Yes (all suspects) Yes Yes 815
Comoros – Yes Yes (all suspects) Yes Yes
Congo 0.8 3 No Y es Yes
(
if TB is
confirmed) Yes Yes
Côte d'Ivoire 0.6 0 0.5 0.5 0 0 No Yes Yes (all suspects) Yes Yes
Democratic Republic
of the Congo 2.3 0 0.3 0.2 <0.1 26 In and out
of countr y Yes Yes
(
if TB is
confirmed) Yes Yes
Equatorial Guinea –
Eritrea 1.3 0 0 0 0 0 No Yes Yes (for smear-
positive TB) Yes Yes
Ethiopia 2.8 0 0.3 <0.1 0.3 7 No Yes Yes (all suspects) Yes Yes
Gabon 0.9 0 3.1 3.1 0 0 No Yes No Yes Yes 0
Gambia 1.8 31 2.8 2.8 0 0 No Yes Yes (all suspects) Yes Yes
Ghana 1.1 1 0.6 0.6 0.6 0 No Yes Yes (all suspects) Yes Yes
Guinea 0.5 6 0.4 0.4 0 1 No Yes Yes
(
if TB is
confirmed) Yes Yes
Guinea-Bissau 1.3 0 3.0 0 0 1 Out of
country Yes Yes (all suspects) Yes Yes
Kenya 4.2 8 0.2 0.2 0.2 15 In and out
of countr y Yes Yes (all suspects) Yes Yes 199
Lesotho 0.9 17 2.4 2.4 2.4 5 Out of
country Yes Yes (all suspects) Yes Yes
Liberia 3.9 0 0 0 0 0 Out of
country No Yes
(
if TB is
confirmed) Yes Yes 28
Madagascar 1.0 6 0.2 0.2 0.2 5 No Yes Yes (all suspects) Yes Yes
Malawi 1.4 19 1.3 0.6 0.3 19 No Yes Yes (all suspects) Yes Yes
Mali 0.4 0 1.0 0.3 0.3 0 No Yes Yes (all suspects) Yes Yes
Mauritania 1.4 – 1.3 1.3 No Yes Yes (all suspects) Yes Yes
Mauritius – Out of
country Yes Yes (all suspects) Yes Yes
Mozambique 1.2 9 0.6 0.4 0 12 Out of
country Yes Yes (all suspects) Yes Yes
Namibia 1.4 100 2.2 2.2 2.2 1 Out of
country Yes Yes (all suspects) Yes Yes
Niger 1.1 1 0.3 0.3 0 0 Out of
country Yes Yes (all suspects) Yes Yes
Nigeria 0.8 2 0.1 <0.1 0.1 32 Out of
country Yes Yes (all suspects) Yes Yes
Rwanda 1.7 13 0.9 0.9 0.9 6 Yes Yes (all suspects) Yes Yes
Sao Tome and Principe 4.3 0 0 0 0 0 No Yes Yes (all suspects) Yes Yes 104
Senegal 0.8 0 1.1 0.7 0.7 3 In country Yes Yes
(
if TB is
confirmed) Yes Yes
Seychelles – Out of
country Yes Yes (all suspects) Yes Yes
Sierra Leone 2.7 0 0 0 0 0 No Yes Yes (all suspects) Yes Yes
South Africa 0.4 97 1.4 1.4 1.4 100 In country Yes Yes (all suspects) Yes Yes
Swaziland 1.5 21 4.1 4.1 4.1 19 Yes Yes (all suspects) Yes Yes
Togo 1.7 0 0.8 0.8 0 1 No Yes Yes
(
if TB is
confirmed) Yes Yes
Uganda 3.2 8 0.6 0.6 0.6 25 Yes Yes (all suspects) Yes Yes
United Republic of Tanzania 2.0 17 0.4 0.1 0.3 13 Yes Yes (all suspects) Yes Yes
Zambia 1.5 1 1.1 In country Yes Yes (all suspects) Yes Yes
Zimbabwe 1.3 1 0.7 0.7 0 17 No Yes Yes (all suspects) Yes Yes
TB NOTIF.
RATE PER
100 000
HEALTH-CARE
WORKERS
RIFAMPICIN
USED
THROUGHOUT
TREATMENT
GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($0HDVXUHGSHUFHQWDJHRI7%FDVHVZLWK0'57%
D
PRVWUHFHQW\HDUDYDLODEOH
New TB cases Previously treated TB cases
Year Source Coverage Percentage Year Source Coverage Percentage
Algeria 2002 Survey National 1.4 (0.60–2.7) 2002 Survey National 9.1 (1.1–29)
Angola
Benin 2010 Survey National 0.5 (<0.1–2.0) 2011 Surveillance National 13 (8.2–20)
Botswana 2008 Survey National 2.5 (1.5–3.5) 2008 Survey National 6.6 (2.4–11)
Burkina Faso
Burundi
Cameroon
Cape Verde
Central African Republic 2009 Survey Sub-national 0.44 (<0.1–2.5) 1998 Survey Sub-national 18 (7.0–35)
Chad
Comoros
Congo
Côte d'Ivoire 2006 Survey National 2.5 (1.1–4.9)
Democratic Republic
of the Congo
Equatorial Guinea
Eritrea
Ethiopia 2005 Survey National 1.6 (0.86–2.8) 2005 Survey National 12 (5.6–21)
Gabon
Gambia 2000 Survey National 0.48 (<0.1–2.6) 2000 Survey National 0 (0–18)
Ghana
Guinea 1998 Survey Sub-national 0.56 (0.11–1.6) 1998 Survey Sub-national 28 (14–47)
Guinea-Bissau
Kenya
Lesotho 1995 Survey National 0.91 (0.19–2.6) 1995 Survey National 5.7 (1.2–16)
Liberia
Madagascar 2007 Survey National 0.49 (0.13–1.3) 2007 Survey National 3.9 (0.48–13)
Malawi 2011 Survey National 0.42 (0.14–0.97) 2011 Survey National 4.8 (3.2–6.9)
Mali
Mauritania
Mauritius 2012 Surveillance National 0 (0–3.0) 2012 Surveillance National 0 (0–60)
Mozambique 2007 Survey National 3.5 (2.2–4.8) 2007 Survey National 12 (0–25)
Namibia 2008 Survey National 3.8 (2.7–5.1) 2008 Survey National 16 (13–21)
Niger
Nigeria 2010 Survey National 2.9 (2.1–4.0) 2010 Survey National 14 (10–19)
Rwanda 2005 Survey National 3.9 (2.5–5.8) 2010 Surveillance National 19 (15–23)
Sao Tome and Principe 2012 Surveillance National 88 (47–100)
Senegal 2006 Survey National 2.1 (0.69–4.9) 2006 Survey National 17 (7.0–31)
Seychelles 2012 Surveillance National 0 (0–23) 2012 Surveillance National 0 (0–84)
Sierra Leone 1997 Survey National 0.85 (<0.1–4.7) 1997 Survey National 23 (5.0–54)
South Africa 2002 Survey National 1.8 (1.4–2.3) 2002 Survey National 6.7 (5.4–8.2)
Swaziland 2009 Survey National 7.7 (4.8–11) 2009 Survey National 34 (28–39)
Togo
Uganda 2011 Survey National 1.4 (0.60–2.2) 2011 Survey National 12 (6.8–19)
United Republic of Tanzania 2007 Survey National 1.1 (0.30–2.8) 2007 Survey National 0 (0–5.9)
Zambia 2008 Survey National 0.33 (<0.1–1.2) 2008 Survey National 8.1 (4.1–14)
Zimbabwe 1995 Survey National 1.9 (1.0–3.3) 1995 Survey National 8.3 (1.8–22)
a Empty rows indicate an absence of high-quality survey or surveillance data. In the absence of high-quality national data, high-quality sub-national data are used.

4')+101(6*'#/'4+%#5
6CDNG# 'UVKOCVGUQHVJGDWTFGPQHFKUGCUGECWUGFD[6$¿
6CDNG# +PEKFGPEGPQVKßECVKQPCPFECUGFGVGEVKQPTCVGUCNNHQTOU¿
6CDNG# %CUGPQVKßECVKQPU¿
Table A4.4 Treatment outcomes, new smear-positive cases, 1995–2011 192
Table A4.5 Treatment outcomes, retreatment cases, 1995–2011 195
6CDNG# *+8VGUVKPICPFRTQXKUKQPQH%26#46CPF+26¿
6CDNG# 6GUVKPIHQT/&46$CPFPWODGTQHEQPßTOGFECUGUQH/&46$¿
6CDNG# 0GYUOGCTRQUKVKXGECUGPQVKßECVKQPD[CIGCPFUGZ¿
Table A4.9 Laboratories, NTP services, drug management and infection control, 2012 205
6CDNG# /GCUWTGFRGTEGPVCIGQH6$ECUGUYKVJ/&46$OQUVTGEGPV[GCTCXCKNCDNG

GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
Estimates of mortality, prevalence and incidence
Estimated values are shown as best estimates followed by lower and upper bounds. e lower and upper bounds are dened
as the 2.5th and 97.5th centiles of outcome distributions produced in simulations. See ANNEX 1 for further details.
Estimated numbers are shown rounded to two signicant gures. Estimated rates are shown rounded to three signicant
gures unless the value is under 100, in which case rates are shown rounded to two signicant gures.
Estimates for all years are recalculated as new information becomes available and techniques are rened, so
they may dier from those published in previous reports in this series. e main updates implemented in this
report are explained in Box 2.1 of Chapter 2. Estimates published in previous global TB control reports should no
longer be used.
Data source
Data shown in this annex are taken from the WHO global TB database on 1 October 2013. Data shown in the main part of
the report were taken from the database in July 2013. As a result, data in this annex may dier slightly from those in the
main part of the report.
Data for all years can be downloaded from www.who.int/tb/data.
Country notes
Caribbean Islands
Data collection from Caribbean Islands that are not Member States of WHO was resumed in 2011 after a break of a few
years. is includes Aruba, Curaçao, Puerto Rico and Sint Maarten, which are Associate Members of the Pan American
Health Organization, plus the territories of Anguilla, Bermuda, Bonaire, Saint Eustatius and Saba, British Virgin Islands,
Cayman Islands, Montserrat and Turks and Caicos Islands. Data are not currently independently collected from the US
Virgin Islands
USA
In addition to the 51 reporting areas, the USA includes territories that report separately to WHO. e data for these terri-
tories are not included in the data reported by the USA. Denitions of case types and outcomes do not exactly match those
used by WHO.
Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
REGION OF THE AMERICAS
7$%/($(VWLPDWHVRIWKHEXUGHQRIGLVHDVHFDXVHGE\7%±
YEAR POPULATION
(MILLIONS)
NUMBER
(THOUSANDS) RATEaNUMBER
(THOUSANDS) RATEaNUMBER
(THOUSANDS) RATEa
MORTALITY (EXCLUDING HIV) PREVALENCE (INCLUDING HIV) INCIDENCE (INCLUDING HIV)
Anguilla 1990 < 1 0 (0–0) 0 (0–0) <0.01 (<0.01–<0.01) 60 (22–118) <0.01 (<0.01–<0.01) 24 (15–35)
1995 < 1 0 (0–0) 0 (0–0) <0.01 (<0.01–<0.01) 47 (23–81) <0.01 (<0.01–<0.01) 23 (20–27)
2000 < 1 0 (0–0) 0 (0–0) <0.01 (<0.01–0.011) 56 (24–102) <0.01 (<0.01–<0.01) 23 (18–27)
2005 < 1 0 (0–0) 0 (0–0) <0.01 (<0.01–0.012) 54 (23–99) <0.01 (<0.01–<0.01) 22 (18–26)
2010 < 1 0 (0–0) 0 (0–0) <0.01 (<0.01–0.012) 50 (22–89) <0.01 (<0.01–<0.01) 21 (18–25)
2011 < 1 0 (0–14) 0 (0–100 000) <0.01 (<0.01–0.012) 50 (22–89) <0.01 (<0.01–<0.01) 21 (17–25)
2012 < 1 0 (0–0) 0 (0–0) <0.01 (<0.01–0.012) 49 (22–88) <0.01 (<0.01–<0.01) 21 (17–25)
Antigua and 1990 < 1 <0.01 (<0.01–<0.01) 3.9 (3.5–4.3) <0.01 (<0.01–<0.01) 2.8 (0.83–6.0) <0.01 (<0.01–<0.01) 1.9 (1.6–2.1)
Barbuda 1995 < 1 0 (0–0) 0 (0–0) <0.01 (<0.01–<0.01) 4.8 (1.4–10) 0 (0–0) 0 (0–0)
2000 < 1 <0.01 (<0.01–<0.01) 1.8 (1.5–2.2) <0.01 (<0.01–0.012) 9.3 (4.6–16) <0.01 (<0.01–<0.01) 5.9 (5.2–6.7)
2005 < 1 <0.01 (<0.01–<0.01) 1.4 (1.3–1.4) <0.01 (<0.01–0.018) 9.4 (2.2–22) <0.01 (<0.01–<0.01) 8.4 (7.3–9.5)
2010 < 1 <0.01 (<0.01–<0.01) 1.4 (1.2–1.5) <0.01 (<0.01–0.016) 5.3 (0.24–18) <0.01 (<0.01–<0.01) 9.2 (8.1–10)
2011 < 1 <0.01 (<0.01–<0.01) 1.4 (1.2–1.5) <0.01 (<0.01–0.015) 6.8 (1.1–18) <0.01 (<0.01–<0.01) 7.8 (6.9–8.9)
2012 < 1 <0.01 (<0.01–<0.01) 1.4 (1.2–1.5) <0.01 (<0.01–<0.01) 4.8 (1.6–9.9) <0.01 (<0.01–<0.01) 3.9 (3.4–4.4)
Argentina 1990 33 1.4 (1.3–1.4) 4.2 (4.1–4.2) 33 (12–64) 102 (38–198) 19 (13–28) 60 (39–85)
1995 35 1.2 (1.1–1.2) 3.3 (3.3–3.4) 26 (12–46) 74 (33–131) 17 (14–21) 49 (40–59)
2000 37 0.84 (0.810–0.870) 2.3 (2.2–2.3) 22 (9.5–39) 59 (26–107) 15 (12–18) 40 (33–49)
2005 39 0.73 (0.700–0.760) 1.9 (1.8–2.0) 18 (7.8–34) 48 (20–87) 13 (11–15) 33 (27–40)
2010 40 0.54 (0.520–0.570) 1.3 (1.3–1.4) 16 (7.1–29) 40 (18–71) 11 (9.1–13) 27 (23–32)
2011 41 0.55 (0.520–0.570) 1.3 (1.3–1.4) 16 (6.8–28) 38 (17–68) 11 (8.9–13) 26 (22–31)
2012 41 0.55 (0.530–0.580) 1.3 (1.3–1.4) 15 (6.5–27) 36 (16–65) 10 (8.6–12) 25 (21–30)
Aruba 1990 < 1 <0.01 (<0.01–<0.01) 0.78 (<0.1–2.5) 0.013 (<0.01–0.024) 20 (7.9–38) <0.01 (<0.01–0.011) 16 (14–18)
1995 < 1 <0.01 (<0.01–<0.01) 0.78 (<0.1–2.5) 0.016 (<0.01–0.031) 20 (7.9–38) 0.013 (0.011–0.014) 16 (14–18)
2000 < 1 <0.01 (<0.01–<0.01) 0.78 (<0.1–2.5) 0.018 (<0.01–0.035) 20 (7.9–38) 0.014 (0.013–0.016) 16 (14–18)
2005 < 1 <0.01 (<0.01–<0.01) 0.78 (<0.1–2.5) 0.02 (<0.01–0.038) 20 (7.9–38) 0.016 (0.014–0.018) 16 (14–18)
2010 < 1 <0.01 (<0.01–<0.01) 0.78 (<0.1–2.5) 0.021 (<0.01–0.039) 20 (7.9–38) 0.016 (0.014–0.018) 16 (14–18)
2011 < 1 <0.01 (<0.01–<0.01) 0.78 (<0.1–2.5) 0.021 (<0.01–0.039) 20 (7.9–38) 0.016 (0.014–0.018) 16 (14–18)
2012 < 1 <0.01 (<0.01–<0.01) 0.78 (<0.1–2.5) 0.021 (<0.01–0.039) 20 (7.9–38) 0.016 (0.014–0.018) 16 (14–18)
Bahamas 1990 < 1 0.043 (0.043–0.043) 17 (17–17) 0.056 (0.024–0.100) 22 (9.5–39) 0.053 (0.046–0.060) 21 (18–23)
1995 < 1 0.012 (0.012–0.012) 4.3 (4.3–4.3) 0.064 (0.027–0.120) 23 (9.7–41) 0.066 (0.057–0.074) 23 (21–26)
2000 < 1 <0.01 (<0.01–<0.01) 2.2 (2.1–2.3) 0.1 (0.050–0.170) 34 (17–57) 0.094 (0.083–0.110) 32 (28–36)
2005 < 1 <0.01 (<0.01–<0.01) 1.1 (1.1–1.1) 0.056 (0.025–0.100) 17 (7.6–30) 0.055 (0.048–0.062) 17 (15–19)
2010 < 1 <0.01 (<0.01–<0.01) 0.51 (0.49–0.53) 0.037 (0.015–0.068) 10 (4.2–19) 0.036 (0.031–0.040) 9.9 (8.7–11)
2011 < 1 <0.01 (<0.01–<0.01) 0.43 (0.42–0.45) 0.058 (0.028–0.098) 16 (7.6–27) 0.047 (0.041–0.053) 13 (11–15)
2012 < 1 <0.01 (<0.01–<0.01) 0.37 (0.36–0.38) 0.04 (0.016–0.076) 11 (4.2–21) 0.037 (0.032–0.042) 9.9 (8.7–11)
Barbados 1990 < 1 0 (0–0) 0 (0–0) <0.01 (<0.01–0.014) 2.6 (0.93–5.2) <0.01 (<0.01–<0.01) 2.2 (1.9–2.5)
1995 < 1 <0.01 (<0.01–<0.01) 0.63 (0.62–0.64) <0.01 (<0.01–<0.01) 1.5 (0.56–3.0) <0.01 (<0.01–<0.01) 1.3 (1.1–1.5)
2000 < 1 <0.01 (<0.01–<0.01) 0.71 (0.70–0.73) <0.01 (<0.01–0.011) 2.1 (0.79–4.2) <0.01 (<0.01–<0.01) 1.3 (1.1–1.5)
2005 < 1 <0.01 (<0.01–<0.01) 0.62 (0.61–0.63) 0.016 (<0.01–0.027) 5.7 (2.7–9.9) 0.014 (0.012–0.016) 5.1 (4.4–5.7)
2010 < 1 <0.01 (<0.01–<0.01) 0.69 (0.67–0.70) <0.01 (<0.01–0.013) 2.8 (1.4–4.7) <0.01 (<0.01–<0.01) 2.5 (2.2–2.8)
2011 < 1 <0.01 (<0.01–<0.01) 0.69 (0.67–0.70) <0.01 (<0.01–<0.01) 0.79 (0.30–1.5) 0 (0–0) 0 (0–0)
2012 < 1 <0.01 (<0.01–<0.01) 0.69 (0.67–0.70) <0.01 (<0.01–<0.01) 1.8 (0.77–3.1) <0.01 (<0.01–<0.01) 1.6 (1.4–1.8)
Belize 1990 < 1 <0.01 (<0.01–<0.01) 2.5 (1.9–3.1) 0.1 (0.033–0.210) 55 (17–113) 0.075 (0.052–0.100) 40 (28–54)
1995 < 1 0.014 (0.013–0.016) 7 (6.3–7.7) 0.1 (0.037–0.200) 49 (18–95) 0.083 (0.068–0.099) 40 (33–48)
2000 < 1 <0.01 (<0.01–<0.01) 3.3 (3.2–3.3) 0.13 (0.047–0.240) 52 (20–101) 0.095 (0.078–0.110) 40 (33–48)
2005 < 1 <0.01 (<0.01–<0.01) 2.2 (2.1–2.2) 0.13 (0.055–0.230) 48 (20–86) 0.11 (0.094–0.120) 40 (34–46)
2010 < 1 0.013 (0.013–0.013) 4.3 (4.3–4.3) 0.16 (0.073–0.290) 53 (24–93) 0.12 (0.100–0.150) 40 (33–48)
2011 < 1 0.014 (0.014–0.014) 4.3 (4.3–4.3) 0.17 (0.073–0.290) 52 (23–93) 0.13 (0.100–0.150) 40 (33–48)
2012 < 1 0.014 (0.014–0.014) 4.3 (4.3–4.3) 0.17 (0.071–0.300) 51 (22–92) 0.13 (0.110–0.160) 40 (33–48)
Bermuda 1990 < 1 0 (0–0) 0 (0–0) <0.01 (<0.01–<0.01) 3.9 (1.2–8.3) 0 (0–0) 0 (0–0)
1995 < 1 0 (0–0) 0 (0–0) <0.01 (<0.01–0.013) 13 (6.6–22) <0.01 (<0.01–<0.01) 7.5 (6.6–8.5)
2000 < 1 0 (0–0) 0 (0–0) <0.01 (<0.01–<0.01) 1.2 (0.35–2.5) 0 (0–0) 0 (0–0)
2005 < 1 0 (0–0) 0 (0–0) <0.01 (<0.01–0.014) 13 (6.4–22) <0.01 (<0.01–<0.01) 8.1 (7.1–9.1)
2010 < 1 <0.01 (<0.01–<0.01) 0.18 (0.18–0.18) <0.01 (<0.01–<0.01) 3.8 (0.97–8.6) <0.01 (<0.01–<0.01) 1.8 (1.6–2.0)
2011 < 1 <0.01 (<0.01–<0.01) 0.18 (0.18–0.18) <0.01 (<0.01–<0.01) 3 (0.78–6.7) <0.01 (<0.01–<0.01) 1.8 (1.5–2.0)
2012 < 1 <0.01 (<0.01–<0.01) 0.18 (0.18–0.18) <0.01 (<0.01–0.012) 11 (5.3–19) <0.01 (<0.01–<0.01) 5.3 (4.6–6.0)
Bolivia 1990 7 2.7 (0.710–6.0) 40 (10–89) 28 (11–55) 419 (156–810) 17 (11–24) 251 (166–354)
(Plurinational 1995 8 2.5 (1.2–4.4) 33 (15–58) 27 (14–44) 352 (180–580) 16 (14–19) 215 (185–248)
State of) 2000 8 2.4 (1.0–4.3) 28 (12–51) 25 (12–43) 299 (145–506) 16 (13–19) 184 (151–221)
2005 9 2.3 (0.950–4.1) 24 (10–44) 24 (12–41) 258 (126–436) 15 (12–18) 158 (129–190)
2010 10 2.2 (0.940–4.0) 22 (9.3–39) 23 (11–39) 227 (113–381) 14 (11–16) 135 (111–161)
2011 10 2.2 (0.930–4.0) 21 (9.1–38) 23 (11–38) 221 (110–370) 14 (11–16) 131 (108–156)
2012 10 2.2 (0.930–3.9) 21 (8.8–37) 23 (11–38) 215 (107–360) 13 (11–16) 127 (105–151)
Bonaire, Saint 2010 < 1 0 (0–0) 0 (0–0) 0 (0–0) 0 (0–0)
Eustatius and Sab
a
2011 < 1 0 (0–0) 0 (0–0) <0.01 (<0.01–<0.01) 8.1 (3.2–15) <0.01 (<0.01–<0.01) 6.3 (5.5–7.2)
2012 < 1 0 (0–0) 0 (0–0) 0 (0–0) 0 (0–0)
Brazil 1990 150 10 (7.8–13) 7 (5.2–9.0) 210 (73–420) 140 (49–278) 130 (79–180) 84 (53–121)
1995 162 8.6 (6.8–11) 5.3 (4.2–6.6) 170 (76–290) 103 (47–180) 120 (94–140) 71 (58–85)
2000 175 7.7 (6.3–9.2) 4.4 (3.6–5.3) 150 (65–260) 84 (37–149) 110 (86–130) 60 (49–72)
2005 186 5.8 (5.2–6.5) 3.1 (2.8–3.5) 120 (52–220) 66 (28–119) 95 (80–110) 51 (43–60)
2010 195 5.4 (5.0–5.8) 2.7 (2.5–3.0) 110 (47–210) 58 (24–106) 91 (75–110) 46 (38–55)
2011 197 5.1 (4.8–5.4) 2.6 (2.4–2.8) 120 (54–220) 62 (27–112) 95 (78–110) 48 (40–57)
2012 199 4.9 (4.6–5.2) 2.5 (2.3–2.6) 120 (51–210) 59 (25–107) 92 (76–110) 46 (38–55)
British Virgin 1990 < 1 0 (0–0) 0 (0–0) <0.01 (<0.01–<0.01) 23 (9.3–43) <0.01 (<0.01–<0.01) 17 (15–20)
Islands 1995 < 1 <0.01 (<0.01–<0.01) 5.5 (5.5–5.5) <0.01 (<0.01–<0.01) 23 (9.5–43) <0.01 (<0.01–<0.01) 17 (15–20)
2000 < 1 <0.01 (<0.01–<0.01) 5.3 (5.1–5.5) <0.01 (<0.01–<0.01) 8.4 (2.5–18) <0.01 (<0.01–<0.01) 5.6 (4.9–6.3)
2005 < 1 <0.01 (<0.01–<0.01) 4.6 (4.4–4.7) <0.01 (<0.01–<0.01) 2.7 (0.82–5.8) 0 (0–0) 0 (0–0)
2010 < 1 <0.01 (<0.01–<0.01) 4.1 (4.0–4.3) <0.01 (<0.01–<0.01) 9.2 (4.3–16) <0.01 (<0.01–<0.01) 4.2 (3.7–4.8)
2011 < 1 <0.01 (<0.01–<0.01) 4.1 (4.0–4.3) <0.01 (<0.01–<0.01) 0.97 (0.29–2.0) 0 (0–0) 0 (0–0)
2012 < 1 <0.01 (<0.01–<0.01) 4.1 (4.0–4.3) <0.01 (<0.01–<0.01) 1 (0.30–2.1) 0 (0–0) 0 (0–0)
Canada 1990 28 0.12 (0.110–0.120) 0.42 (0.41–0.44) 3 (1.3–5.6) 11 (4.6–20) 2.3 (2.0–2.6) 8.3 (7.3–9.4)
1995 29 0.12 (0.120–0.120) 0.4 (0.40–0.40) 3 (1.3–5.4) 10 (4.3–18) 2.3 (2.0–2.6) 7.7 (6.8–8.7)
2000 31 0.082 (0.081–0.082) 0.27 (0.26–0.27) 2.6 (1.1–4.8) 8.5 (3.5–16) 2 (1.7–2.2) 6.5 (5.7–7.3)
2005 32 0.086 (0.086–0.086) 0.27 (0.27–0.27) 2.4 (1.0–4.3) 7.3 (3.1–13) 1.8 (1.6–2.0) 5.5 (4.8–6.3)
2010 34 0.074 (0.074–0.074) 0.22 (0.22–0.22) 2 (0.770–3.7) 5.7 (2.2–11) 1.6 (1.4–1.8) 4.6 (4.0–5.2)
2011 34 0.071 (0.070–0.071) 0.21 (0.20–0.21) 2.2 (0.950–3.9) 6.3 (2.7–11) 1.6 (1.4–1.9) 4.8 (4.2–5.4)
2012 35 0.067 (0.067–0.068) 0.19 (0.19–0.19) 2.1 (0.900–3.8) 6.1 (2.6–11) 1.6 (1.4–1.8) 4.6 (4.0–5.2)
Cayman Islands 1990 < 1 <0.01 (<0.01–<0.01) 4 (4.0–4.1) <0.01 (<0.01–<0.01) 12 (3.5–25) <0.01 (<0.01–<0.01) 9.2 (8.1–10)
1995 < 1 0 (0–0) 0 (0–0) <0.01 (<0.01–<0.01) 10 (4.4–18) <0.01 (<0.01–<0.01) 7.3 (6.4–8.2)
2000 < 1 0 (0–0) 0 (0–0) 0.012 (<0.01–0.020) 28 (14–48) <0.01 (<0.01–<0.01) 14 (12–16)
2005 < 1 0 (0–0) 0 (0–0) <0.01 (<0.01–<0.01) 2.2 (1.1–3.7) <0.01 (<0.01–<0.01) 1.2 (1.1–1.4)
2010 < 1 0 (0–0) 0 (0–0) <0.01 (<0.01–0.016) 15 (5.5–28) <0.01 (<0.01–<0.01) 8.3 (7.3–9.4)
2011 < 1 0 (0–0) 0 (0–0) <0.01 (<0.01–0.010) 8 (2.2–18) <0.01 (<0.01–<0.01) 4.1 (3.6–4.6)
2012 < 1 0 (0–0) 0 (0–0) 0.011 (<0.01–0.019) 18 (8.2–33) <0.01 (<0.01–<0.01) 12 (11–14)
Chile 1990 13 0.76 (0.710–0.820) 5.8 (5.3–6.2) 10 (4.4–18) 76 (34–136) 7.1 (6.2–8.0) 54 (47–61)
1995 14 0.5 (0.460–0.540) 3.5 (3.2–3.7) 6.4 (2.6–12) 44 (18–82) 4.8 (4.2–5.4) 33 (29–37)
2000 15 0.3 (0.290–0.310) 1.9 (1.8–2.0) 4.6 (1.9–8.6) 30 (12–55) 3.5 (3.0–3.9) 22 (20–25)
2005 16 0.24 (0.240–0.240) 1.5 (1.4–1.5) 4 (1.8–7.1) 25 (11–44) 2.9 (2.5–3.3) 18 (15–20)
2010 17 0.23 (0.220–0.230) 1.3 (1.3–1.4) 3.7 (1.5–6.7) 21 (8.9–39) 2.7 (2.4–3.1) 16 (14–18)
2011 17 0.22 (0.220–0.230) 1.3 (1.3–1.3) 3.8 (1.6–6.9) 22 (9.5–40) 2.8 (2.5–3.2) 16 (14–18)
2012 17 0.21 (0.210–0.220) 1.2 (1.2–1.2) 3.6 (1.4–6.7) 21 (8.3–38) 2.8 (2.4–3.1) 16 (14–18)
Colombia 1990 33 1.7 (1.4–2.0) 5 (4.2–5.9) 28 (10–55) 85 (31–165) 18 (12–25) 54 (36–75)
1995 37 2 (1.8–2.2) 5.3 (4.8–5.9) 30 (15–51) 83 (41–140) 18 (14–21) 48 (39–58)
2000 40 1.3 (1.2–1.4) 3.2 (3.0–3.4) 27 (13–46) 68 (33–116) 17 (14–21) 43 (35–52)
2005 43 1 (1.0–1.0) 2.4 (2.3–2.4) 25 (12–43) 58 (28–100) 17 (14–20) 38 (31–46)
2010 46 0.9 (0.890–0.910) 1.9 (1.9–2.0) 24 (11–41) 51 (24–87) 16 (13–19) 34 (28–41)
2011 47 0.84 (0.830–0.850) 1.8 (1.8–1.8) 23 (11–40) 49 (23–85) 16 (13–19) 34 (28–40)
2012 48 0.77 (0.760–0.790) 1.6 (1.6–1.6) 23 (11–39) 48 (22–83) 16 (13–19) 33 (27–39)
a Rates are per 100 000 population.

GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($(VWLPDWHVRIWKHEXUGHQRIGLVHDVHFDXVHGE\7%±
YEAR POPULATION
(MILLIONS)
NUMBER
(THOUSANDS) RATE
a
NUMBER
(THOUSANDS) RATE
a
NUMBER
(THOUSANDS) RATE
a
MORTALITY (EXCLUDING HIV) PREVALENCE (INCLUDING HIV) INCIDENCE (INCLUDING HIV)
Costa Rica 1990 3 0.078 (0.072–0.083) 2.5 (2.3–2.7) 3.6 (1.7–6.3) 118 (54–205) 1.5 (1.3–1.7) 48 (42–54)
1995 3 0.11 (0.100–0.110) 3.1 (3.0–3.2) 3 (1.5–5.0) 87 (44–143) 1.5 (1.3–1.7) 43 (37–48)
2000 4 0.07 (0.067–0.072) 1.8 (1.7–1.8) 2.5 (1.3–4.0) 63 (33–101) 1.4 (1.2–1.5) 35 (31–39)
2005 4 0.06 (0.058–0.061) 1.4 (1.3–1.4) 1.7 (0.920–2.8) 40 (21–65) 1 (0.880–1.1) 23 (20–26)
2010 5 0.043 (0.038–0.047) 0.92 (0.82–1.0) 0.93 (0.460–1.6) 20 (9.8–33) 0.65 (0.570–0.740) 14 (12–16)
2011 5 0.04 (0.036–0.045) 0.85 (0.76–0.95) 0.78 (0.360–1.4) 16 (7.6–29) 0.59 (0.510–0.660) 12 (11–14)
2012 5 0.038 (0.034–0.043) 0.8 (0.70–0.89) 0.6 (0.220–1.2) 12 (4.7–24) 0.51 (0.440–0.580) 11 (9.3–12)
Cuba 1990 11 0.062 (0.059–0.065) 0.58 (0.55–0.62) 6.4 (2.4–12) 60 (23–115) 2.6 (1.6–3.9) 25 (15–37)
1995 11 0.096 (0.095–0.098) 0.88 (0.87–0.89) 3.5 (1.7–5.9) 32 (15–54) 2 (1.7–2.5) 19 (15–23)
2000 11 0.046 (0.045–0.047) 0.41 (0.40–0.42) 2.2 (0.940–3.9) 19 (8.4–35) 1.4 (1.1–1.8) 13 (10–16)
2005 11 0.033 (0.033–0.034) 0.3 (0.30–0.30) 1.6 (0.730–2.8) 14 (6.4–25) 1 (0.850–1.3) 9.2 (7.5–11)
2010 11 0.039 (0.039–0.040) 0.35 (0.35–0.35) 1.6 (0.730–2.8) 14 (6.4–25) 1 (0.840–1.3) 9.3 (7.4–11)
2011 11 0.038 (0.038–0.038) 0.33 (0.33–0.34) 1.6 (0.700–2.8) 14 (6.2–25) 1 (0.830–1.3) 9.3 (7.4–11)
2012 11 0.038 (0.038–0.038) 0.33 (0.33–0.34) 1.6 (0.670–2.8) 14 (5.9–25) 1 (0.840–1.3) 9.3 (7.4–11)
Curaçao 2010 < 1 <0.01 (<0.01–<0.01) 0.19 (<0.1–0.63) <0.01 (<0.01–0.014) 5 (2.0–9.4) <0.01 (<0.01–<0.01) 3.9 (3.4–4.4)
2011 < 1 <0.01 (0–<0.01) <0.1 (0–0.19) <0.01 (<0.01–<0.01) 0.97 (0.38–1.8) <0.01 (<0.01–<0.01) 0.76 (0.67–0.86)
2012 < 1 <0.01 (0–<0.01) <0.1 (0–0.18) <0.01 (<0.01–<0.01) 0.95 (0.37–1.8) <0.01 (<0.01–<0.01) 0.74 (0.65–0.84)
Dominica 1990 < 1 <0.01 (<0.01–<0.01) 7.6 (7.3–7.9) 0.012 (<0.01–0.031) 17 (2.7–43) 0.01 (<0.01–0.015) 15 (9.3–21)
1995 < 1 <0.01 (<0.01–<0.01) 2.4 (2.2–2.5) 0.014 (<0.01–0.026) 19 (7.3–36) 0.01 (<0.01–0.012) 14 (12–17)
2000 < 1 <0.01 (<0.01–<0.01) 3.4 (3.1–3.7) 0.02 (<0.01–0.034) 28 (13–49) <0.01 (<0.01–0.012) 14 (11–17)
2005 < 1 <0.01 (<0.01–<0.01) 1.3 (1.3–1.3) 0.012 (<0.01–0.024) 17 (6.3–34) <0.01 (<0.01–0.011) 13 (11–16)
2010 < 1 <0.01 (<0.01–<0.01) 3.2 (3.2–3.3) 0.013 (<0.01–0.024) 19 (7.9–34) <0.01 (<0.01–0.011) 13 (11–16)
2011 < 1 <0.01 (<0.01–<0.01) 2.1 (2.1–2.1) 0.016 (<0.01–0.033) 23 (7.2–46) <0.01 (<0.01–0.011) 13 (11–16)
2012 < 1 <0.01 (<0.01–<0.01) 2 (2.0–2.0) 0.018 (<0.01–0.030) 25 (12–42) <0.01 (<0.01–0.011) 13 (11–15)
Dominican 1990 7 1 (0.550–1.6) 14 (7.6–22) 25 (9.4–47) 339 (130–646) 11 (6.6–16) 148 (91–218)
Republic 1995 8 1 (0.510–1.7) 13 (6.4–21) 17 (8.6–29) 215 (108–360) 9.7 (7.9–12) 121 (99–146)
2000 9 0.76 (0.400–1.2) 8.7 (4.6–14) 14 (6.8–23) 159 (78–268) 8.6 (7.1–10) 100 (82–120)
2005 9 0.59 (0.380–0.850) 6.3 (4.0–9.1) 12 (6.0–21) 131 (65–220) 7.7 (6.3–9.2) 82 (67–98)
2010 10 0.53 (0.390–0.680) 5.3 (3.9–6.8) 11 (5.3–18) 107 (53–181) 6.7 (5.6–8.0) 67 (55–80)
2011 10 0.49 (0.380–0.620) 4.9 (3.8–6.1) 10 (5.1–18) 103 (50–173) 6.6 (5.4–7.8) 65 (53–77)
2012 10 0.46 (0.380–0.550) 4.4 (3.7–5.3) 10 (4.9–17) 98 (48–165) 6.4 (5.3–7.6) 62 (51–74)
Ecuador 1990 10 2 (1.4–2.6) 19 (14–26) 34 (13–66) 340 (127–655) 18 (11–26) 174 (108–257)
1995 11 2 (1.4–2.7) 17 (12–24) 27 (14–46) 242 (121–403) 15 (13–19) 136 (111–164)
2000 13 1.8 (1.3–2.3) 14 (10–19) 23 (12–39) 187 (93–313) 13 (11–16) 107 (87–128)
2005 14 1.1 (0.910–1.2) 7.7 (6.6–9.0) 20 (10–34) 148 (74–248) 11 (9.4–14) 83 (68–100)
2010 15 0.69 (0.610–0.790) 4.6 (4.0–5.2) 17 (8.5–28) 113 (57–187) 9.7 (8.0–12) 65 (54–77)
2011 15 0.53 (0.460–0.610) 3.5 (3.0–4.0) 16 (8.1–27) 106 (53–176) 9.4 (7.8–11) 62 (51–74)
2012 15 0.41 (0.350–0.480) 2.7 (2.3–3.1) 15 (7.6–25) 98 (49–163) 9.1 (7.5–11) 59 (48–70)
El Salvador 1990 5 0.26 (0.150–0.390) 4.8 (2.8–7.4) 5.1 (1.7–10) 95 (32–191) 3.4 (2.3–4.7) 63 (43–88)
1995 6 0.22 (0.150–0.300) 3.8 (2.6–5.2) 3.1 (1.2–6.1) 55 (21–105) 2.6 (2.3–2.9) 45 (39–50)
2000 6 0.17 (0.120–0.210) 2.8 (2.1–3.6) 3.3 (1.6–5.7) 56 (27–96) 2.2 (1.8–2.6) 37 (30–44)
2005 6 0.11 (0.087–0.140) 1.8 (1.4–2.3) 3.7 (1.8–6.2) 60 (29–103) 2.4 (1.9–2.9) 39 (32–47)
2010 6 0.076 (0.056–0.100) 1.2 (0.90–1.6) 2.1 (0.740–4.0) 33 (12–65) 1.8 (1.5–2.0) 28 (24–33)
2011 6 0.071 (0.051–0.094) 1.1 (0.81–1.5) 2.1 (0.760–4.2) 34 (12–67) 1.7 (1.4–1.9) 27 (23–31)
2012 6 0.065 (0.046–0.088) 1 (0.73–1.4) 2.2 (0.770–4.3) 34 (12–68) 1.6 (1.4–1.8) 25 (22–29)
Grenada 1990 < 1 0 (0–0) 0 (0–0) 0.01 (<0.01–0.020) 11 (3.9–21) <0.01 (<0.01–<0.01) 4.6 (2.9–6.8)
1995 < 1 0 (0–0) 0 (0–0) <0.01 (<0.01–0.014) 8.2 (4.1–14) <0.01 (<0.01–<0.01) 4.5 (3.8–5.2)
2000 < 1 0 (0–0) 0 (0–0) <0.01 (<0.01–0.015) 8.6 (4.1–15) <0.01 (<0.01–<0.01) 4.4 (3.6–5.2)
2005 < 1 <0.01 (<0.01–<0.01) 1.6 (1.6–1.6) <0.01 (<0.01–0.014) 8.1 (3.9–14) <0.01 (<0.01–<0.01) 4.2 (3.5–5.1)
2010 < 1 <0.01 (<0.01–<0.01) 0.76 (0.75–0.76) <0.01 (<0.01–0.013) 5.5 (1.3–13) <0.01 (<0.01–<0.01) 4.1 (3.4–4.9)
2011 < 1 <0.01 (<0.01–<0.01) 0.99 (0.95–1.0) <0.01 (<0.01–0.015) 7.1 (2.2–15) <0.01 (<0.01–<0.01) 4.1 (3.4–4.9)
2012 < 1 <0.01 (<0.01–<0.01) 0.99 (0.95–1.0) <0.01 (<0.01–0.015) 6.8 (2.1–14) <0.01 (<0.01–<0.01) 4.1 (3.4–4.9)
Guatemala 1990 9 0.86 (0.800–0.930) 9.7 (9.0–10) 13 (4.8–24) 142 (53–274) 6.6 (4.1–9.7) 74 (47–109)
1995 10 0.63 (0.570–0.700) 6.3 (5.7–7.1) 14 (6.8–23) 139 (69–233) 7.1 (5.8–8.5) 71 (58–85)
2000 11 0.57 (0.510–0.640) 5.1 (4.6–5.7) 14 (7.1–24) 129 (63–217) 7.6 (6.2–9.1) 68 (55–81)
2005 13 0.41 (0.370–0.460) 3.3 (2.9–3.6) 15 (7.4–25) 119 (59–200) 8.2 (6.7–9.9) 65 (53–78)
2010 14 0.34 (0.310–0.370) 2.3 (2.1–2.6) 16 (7.9–27) 112 (55–189) 8.8 (7.3–11) 62 (51–73)
2011 15 0.32 (0.290–0.350) 2.2 (2.0–2.4) 16 (8.0–28) 111 (55–188) 9 (7.4–11) 61 (50–73)
2012 15 0.31 (0.280–0.340) 2.1 (1.9–2.2) 17 (8.2–28) 110 (54–187) 9.1 (7.5–11) 60 (50–72)
Guyana 1990 < 1 0.054 (0.036–0.075) 7.5 (5.0–10) 1.4 (0.520–2.7) 193 (72–372) 0.65 (0.400–0.960) 89 (55–132)
1995 < 1 0.067 (0.049–0.087) 9.1 (6.7–12) 1.1 (0.550–1.9) 153 (75–258) 0.65 (0.530–0.780) 89 (73–107)
2000 < 1 0.099 (0.061–0.150) 13 (8.2–20) 1 (0.490–1.8) 139 (66–239) 0.78 (0.630–0.930) 104 (85–125)
2005 < 1 0.12 (0.110–0.140) 16 (14–19) 1.1 (0.470–1.9) 139 (62–248) 0.88 (0.720–1.1) 115 (94–138)
2010 < 1 0.12 (0.098–0.130) 15 (12–17) 1 (0.430–1.9) 132 (55–241) 0.87 (0.720–1.0) 111 (91–132)
2011 < 1 0.12 (0.099–0.140) 15 (12–17) 1 (0.430–1.9) 131 (54–241) 0.87 (0.720–1.0) 110 (91–131)
2012 < 1 0.12 (0.099–0.140) 15 (12–17) 1 (0.430–1.9) 131 (54–242) 0.87 (0.710–1.0) 109 (90–130)
Haiti 1990 7 2.5 (0.450–6.4) 36 (6.3–90) 27 (8.2–56) 376 (115–787) 18 (11–26) 247 (153–365)
1995 8 3 (1.1–6.0) 39 (14–77) 30 (14–51) 378 (180–648) 19 (16–23) 247 (202–297)
2000 9 3.4 (1.2–6.7) 40 (14–78) 34 (17–58) 400 (193–681) 23 (19–28) 271 (221–325)
2005 9 3.4 (1.2–6.6) 37 (13–71) 36 (17–61) 388 (187–659) 25 (21–30) 272 (222–326)
2010 10 2.9 (1.1–5.4) 29 (11–55) 32 (15–55) 326 (156–556) 23 (19–27) 230 (190–275)
2011 10 2.7 (1.1–5.1) 27 (11–51) 31 (15–53) 309 (148–528) 22 (18–27) 222 (183–265)
2012 10 2.6 (1.0–4.9) 25 (10–48) 30 (14–52) 296 (140–509) 22 (18–26) 213 (176–254)
Honduras 1990 5 0.31 (0.096–0.650) 6.4 (2.0–13) 8.7 (3.0–18) 178 (60–358) 5.6 (3.6–7.9) 113 (73–162)
1995 6 0.35 (0.110–0.730) 6.2 (1.9–13) 9.5 (3.0–19) 169 (54–348) 6.4 (4.1–9.2) 115 (74–164)
2000 6 0.31 (0.060–0.760) 5 (0.96–12) 11 (3.4–22) 169 (54–347) 7.1 (4.6–10) 114 (74–163)
2005 7 0.26 (0.025–0.780) 3.8 (0.36–11) 7.7 (2.5–16) 112 (36–228) 5 (3.2–7.2) 73 (47–104)
2010 8 0.24 (<0.01–0.840) 3.1 (0.12–11) 6.4 (2.2–13) 84 (29–170) 4.1 (2.7–5.9) 54 (35–77)
2011 8 0.24 (<0.01–0.850) 3 (<0.1–11) 6.5 (2.2–13) 84 (28–169) 4.2 (2.7–6.0) 54 (35–77)
2012 8 0.23 (<0.01–0.850) 2.9 (<0.1–11) 6.5 (2.1–13) 82 (27–168) 4.3 (2.8–6.1) 54 (35–77)
Jamaica 1990 2 0.021 (0.016–0.026) 0.87 (0.68–1.1) 0.23 (0.081–0.440) 9.5 (3.4–19) 0.15 (0.110–0.210) 6.5 (4.7–8.8)
1995 2 0.025 (0.020–0.031) 1 (0.81–1.2) 0.22 (0.100–0.380) 8.9 (4.1–15) 0.16 (0.130–0.190) 6.5 (5.4–7.9)
2000 3 0.016 (0.013–0.020) 0.63 (0.51–0.77) 0.21 (0.095–0.370) 8.1 (3.7–14) 0.17 (0.140–0.200) 6.5 (5.4–7.9)
2005 3 0.011 (<0.01–0.013) 0.4 (0.32–0.48) 0.23 (0.110–0.400) 8.7 (4.1–15) 0.18 (0.140–0.210) 6.5 (5.4–7.9)
2010 3 <0.01 (<0.01–<0.01) 0.26 (0.21–0.31) 0.26 (0.130–0.430) 9.4 (4.6–16) 0.18 (0.150–0.210) 6.6 (5.4–7.8)
2011 3 <0.01 (<0.01–<0.01) 0.24 (0.20–0.28) 0.26 (0.130–0.440) 9.4 (4.6–16) 0.18 (0.150–0.220) 6.6 (5.4–7.8)
2012 3 <0.01 (<0.01–<0.01) 0.22 (0.18–0.26) 0.26 (0.130–0.440) 9.5 (4.7–16) 0.18 (0.150–0.220) 6.6 (5.4–7.8)
Mexico 1990 86 6.7 (6.4–7.0) 7.8 (7.5–8.1) 130 (61–210) 145 (71–246) 57 (49–66) 67 (57–77)
1995 95 5.3 (4.9–5.7) 5.5 (5.2–5.9) 85 (43–140) 89 (46–146) 44 (38–51) 46 (40–53)
2000 104 3.5 (3.3–3.6) 3.3 (3.2–3.5) 53 (27–87) 51 (26–84) 32 (27–37) 31 (26–36)
2005 111 2.7 (2.6–2.9) 2.5 (2.4–2.6) 38 (19–63) 34 (17–57) 25 (21–28) 22 (19–26)
2010 118 2.6 (2.5–2.7) 2.2 (2.1–2.3) 40 (19–67) 34 (16–57) 26 (23–30) 22 (19–26)
2011 119 2.2 (2.1–2.4) 1.9 (1.8–2.0) 41 (20–69) 34 (16–58) 27 (23–31) 23 (19–26)
2012 121 2.2 (2.1–2.3) 1.8 (1.7–1.9) 40 (19–69) 33 (16–57) 27 (23–32) 23 (19–26)
Montserrat 1990 < 1 0 (0–0) 0 (0–0) <0.01 (<0.01–<0.01) 20 (10–33) <0.01 (<0.01–<0.01) 11 (9.4–12)
1995 < 1 <0.01 (<0.01–<0.01) 11 (10–11) <0.01 (<0.01–<0.01) 8.2 (2.5–17) <0.01 (<0.01–<0.01) 4.1 (3.6–4.7)
2000 < 1 <0.01 (<0.01–<0.01) 21 (21–22) <0.01 (<0.01–<0.01) 11 (3.3–23) 0 (0–0) 0 (0–0)
2005 < 1 <0.01 (<0.01–<0.01) 22 (21–22) <0.01 (<0.01–<0.01) 42 (21–69) <0.01 (<0.01–<0.01) 24 (21–27)
2010 < 1 <0.01 (<0.01–<0.01) 21 (21–22) <0.01 (<0.01–<0.01) 9.9 (2.9–21) 0 (0–0) 0 (0–0)
2011 < 1 <0.01 (<0.01–<0.01) 24 (24–25) <0.01 (<0.01–<0.01) 4.7 (1.4–10) 0 (0–0) 0 (0–0)
2012 < 1 <0.01 (<0.01–<0.01) 24 (24–25) 0 (0–0) 0 (0–0)
Netherlands 1990 < 1 <0.01 (<0.01–<0.01) 0.59 (0.56–0.62) 0.013 (<0.01–0.024) 6.7 (2.5–13) <0.01 (<0.01–0.011) 5.3 (4.6–6.0)
Antilles 1995 < 1 <0.01 (<0.01–<0.01) 0.57 (0.54–0.60) 0.014 (<0.01–0.025) 7.2 (3.1–13) 0.01 (<0.01–0.012) 5.3 (4.6–6.0)
2000 < 1 <0.01 (<0.01–<0.01) 0.59 (0.58–0.61) <0.01 (<0.01–0.019) 5 (1.5–11) <0.01 (<0.01–<0.01) 3.2 (2.8–3.7)
2005 < 1 <0.01 (<0.01–<0.01) 0.59 (0.57–0.61) 0.01 (<0.01–0.021) 5.6 (1.7–12) <0.01 (<0.01–<0.01) 4.7 (4.1–5.3)
Nicaragua 1990 4 0.45 (0.260–0.680) 11 (6.4–16) 7.6 (2.8–15) 183 (68–354) 4.5 (2.9–6.3) 108 (71–152)
1995 5 0.41 (0.260–0.600) 8.9 (5.6–13) 6.4 (3.0–11) 137 (65–236) 4 (3.2–4.8) 85 (70–102)
2000 5 0.33 (0.230–0.440) 6.4 (4.5–8.7) 5.5 (2.6–9.5) 108 (51–186) 3.4 (2.8–4.1) 68 (55–81)
2005 5 0.3 (0.220–0.380) 5.4 (4.1–6.9) 4.6 (2.2–8.0) 85 (39–146) 2.9 (2.4–3.5) 53 (44–64)
2010 6 0.26 (0.200–0.330) 4.5 (3.5–5.6) 2.9 (1.0–5.9) 50 (17–101) 2.5 (2.1–2.8) 42 (36–49)
2011 6 0.19 (0.150–0.240) 3.3 (2.6–4.1) 3.1 (1.1–6.3) 53 (18–106) 2.4 (2.0–2.7) 40 (35–46)
2012 6 0.19 (0.150–0.230) 3.1 (2.4–3.8) 3.3 (1.1–6.7) 55 (19–112) 2.3 (2.0–2.7) 38 (33–44)
a
Rates are per 100 000 population.

Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
REGION OF THE AMERICAS
7$%/($(VWLPDWHVRIWKHEXUGHQRIGLVHDVHFDXVHGE\7%±
YEAR POPULATION
(MILLIONS)
NUMBER
(THOUSANDS) RATEaNUMBER
(THOUSANDS) RATEaNUMBER
(THOUSANDS) RATEa
MORTALITY (EXCLUDING HIV) PREVALENCE (INCLUDING HIV) INCIDENCE (INCLUDING HIV)
Panama 1990 2 0.2 (0.130–0.290) 8.1 (5.2–12) 1.9 (0.740–3.6) 77 (30–146) 1.2 (0.810–1.6) 47 (33–65)
1995 3 0.2 (0.160–0.250) 7.4 (5.8–9.2) 1.6 (0.600–3.0) 57 (22–110) 1.3 (1.1–1.6) 47 (39–57)
2000 3 0.2 (0.180–0.230) 6.6 (5.9–7.4) 1.6 (0.600–3.0) 52 (20–99) 1.4 (1.2–1.7) 47 (39–56)
2005 3 0.22 (0.210–0.230) 6.6 (6.3–6.9) 1.8 (0.670–3.4) 53 (20–101) 1.6 (1.3–1.9) 47 (39–57)
2010 4 0.19 (0.190–0.200) 5.2 (5.1–5.4) 2.2 (0.920–3.9) 59 (25–107) 1.8 (1.5–2.0) 48 (42–54)
2011 4 0.19 (0.190–0.190) 5.1 (5.0–5.2) 2.3 (1.0–4.0) 61 (28–108) 1.8 (1.6–2.0) 48 (42–54)
2012 4 0.19 (0.180–0.190) 4.9 (4.8–5.0) 2.4 (1.2–4.2) 64 (30–110) 1.8 (1.6–2.0) 48 (42–54)
Paraguay 1990 4 0.2 (0.150–0.250) 4.6 (3.5–5.9) 4.2 (2.0–7.0) 98 (48–165) 2.8 (2.6–3.0) 66 (61–72)
1995 5 0.23 (0.170–0.290) 4.8 (3.6–6.1) 3.7 (1.9–6.2) 78 (39–129) 2.5 (2.3–2.7) 52 (48–56)
2000 5 0.23 (0.160–0.310) 4.3 (3.1–5.8) 3.8 (1.9–6.4) 72 (36–120) 2.6 (2.4–2.8) 49 (45–53)
2005 6 0.28 (0.220–0.350) 4.7 (3.7–5.9) 4.1 (2.1–7.0) 70 (35–118) 2.9 (2.7–3.1) 49 (45–53)
2010 6 0.19 (0.160–0.230) 3 (2.5–3.6) 4.2 (2.0–7.1) 65 (32–110) 3 (2.7–3.2) 46 (42–50)
2011 7 0.2 (0.160–0.230) 3 (2.5–3.6) 4.2 (2.1–7.1) 64 (32–108) 3 (2.8–3.2) 45 (42–49)
2012 7 0.2 (0.160–0.240) 3 (2.5–3.6) 4.2 (2.1–7.1) 63 (31–106) 3 (2.8–3.2) 45 (41–48)
Peru 1990 22 7.5 (2.5–15) 34 (11–70) 120 (42–240) 554 (191–1 100) 69 (43–100) 317 (196–468)
1995 24 6.2 (3.3–9.9) 26 (14–41) 85 (37–150) 355 (156–634) 58 (47–70) 242 (198–290)
2000 26 3.7 (2.3–5.4) 14 (8.9–21) 70 (30–130) 268 (116–481) 48 (39–57) 184 (151–221)
2005 28 2.7 (2.1–3.4) 9.7 (7.4–12) 54 (23–99) 195 (82–357) 39 (33–46) 140 (118–164)
2010 29 1.8 (1.3–2.3) 6.1 (4.6–7.8) 37 (12–77) 127 (40–263) 31 (27–35) 106 (93–120)
2011 30 1.7 (1.2–2.1) 5.6 (4.1–7.2) 37 (12–74) 124 (42–248) 30 (26–34) 101 (88–114)
2012 30 1.5 (1.1–2.0) 5.1 (3.8–6.7) 36 (12–73) 121 (41–243) 29 (25–32) 95 (83–108)
Puerto Rico 1990 4 0.069 (0.069–0.070) 2 (2.0–2.0) 0.22 (0.064–0.460) 6.1 (1.8–13) 0.18 (0.160–0.210) 5.2 (4.6–5.9)
1995 4 0.081 (0.080–0.081) 2.2 (2.2–2.2) 0.4 (0.170–0.740) 11 (4.5–20) 0.3 (0.260–0.340) 8.2 (7.2–9.2)
2000 4 0.017 (0.017–0.017) 0.45 (0.45–0.45) 0.27 (0.110–0.490) 7 (2.9–13) 0.2 (0.180–0.230) 5.3 (4.6–6.0)
2005 4 0.017 (0.017–0.017) 0.45 (0.45–0.46) 0.15 (0.066–0.270) 4 (1.7–7.1) 0.13 (0.110–0.150) 3.5 (3.0–3.9)
2010 4 0.01 (0.010–0.010) 0.28 (0.27–0.28) 0.12 (0.055–0.210) 3.2 (1.5–5.6) 0.092 (0.081–0.100) 2.5 (2.2–2.8)
2011 4 <0.01 (<0.01–<0.01) 0.25 (0.25–0.25) 0.07 (0.026–0.140) 1.9 (0.69–3.7) 0.058 (0.050–0.065) 1.6 (1.4–1.8)
2012 4 <0.01 (<0.01–<0.01) 0.23 (0.23–0.23) 0.11 (0.053–0.200) 3 (1.4–5.3) 0.082 (0.072–0.092) 2.2 (1.9–2.5)
Saint Kitts and 1990 < 1 0 (0–0) 0 (0–0) <0.01 (<0.01–<0.01) 0.56 (0.17–1.2) 0 (0–0) 0 (0–0)
Nevis 1995 < 1 <0.01 (<0.01–<0.01) 2.1 (2.1–2.2) <0.01 (<0.01–0.014) 17 (6.2–32) <0.01 (<0.01–<0.01) 13 (12–15)
2000 < 1 <0.01 (<0.01–<0.01) 2.4 (2.3–2.5) <0.01 (<0.01–<0.01) 7.3 (2.2–15) 0 (0–0) 0 (0–0)
2005 < 1 <0.01 (<0.01–<0.01) 2.2 (2.1–2.3) <0.01 (<0.01–<0.01) 3.7 (1.1–7.9) 0 (0–0) 0 (0–0)
2010 < 1 <0.01 (<0.01–<0.01) 2.5 (2.3–2.6) <0.01 (<0.01–<0.01) 5.6 (1.6–12) <0.01 (<0.01–<0.01) 4.4 (3.8–5.0)
2011 < 1 <0.01 (<0.01–<0.01) 2.5 (2.3–2.6) <0.01 (<0.01–<0.01) 5.1 (1.3–12) <0.01 (<0.01–<0.01) 2.2 (1.9–2.5)
2012 < 1 <0.01 (<0.01–<0.01) 2.5 (2.3–2.6) <0.01 (<0.01–<0.01) 5.1 (1.7–10) <0.01 (<0.01–<0.01) 4.3 (3.8–4.9)
Saint Lucia 1990 < 1 <0.01 (<0.01–<0.01) 4 (3.8–4.2) 0.027 (<0.01–0.051) 19 (7.2–37) 0.021 (0.019–0.024) 15 (13–17)
1995 < 1 0.012 (<0.01–0.016) 8.3 (6.3–11) 0.035 (0.014–0.065) 24 (9.6–44) 0.027 (0.023–0.030) 18 (16–21)
2000 < 1 <0.01 (<0.01–<0.01) 0.81 (0.71–0.92) 0.024 (<0.01–0.044) 15 (5.9–28) 0.018 (0.016–0.021) 12 (10–13)
2005 < 1 <0.01 (<0.01–<0.01) 3.5 (3.3–3.6) 0.023 (<0.01–0.044) 14 (5.4–26) 0.018 (0.016–0.020) 11 (9.5–12)
2010 < 1 <0.01 (<0.01–<0.01) 1.4 (1.3–1.5) 0.015 (<0.01–0.033) 8.6 (2.5–18) 0.012 (0.011–0.014) 6.9 (6.1–7.8)
2011 < 1 <0.01 (<0.01–<0.01) 1.3 (1.2–1.4) 0.011 (<0.01–0.022) 6.1 (2.0–13) <0.01 (<0.01–0.010) 5.1 (4.5–5.8)
2012 < 1 <0.01 (<0.01–<0.01) 1.2 (1.1–1.4) <0.01 (<0.01–0.018) 4.8 (1.5–9.9) <0.01 (<0.01–<0.01) 3.3 (2.9–3.7)
Saint Vincent and 1990 < 1 <0.01 (<0.01–<0.01) 1 (0.95–1.1) 0.071 (0.025–0.140) 66 (24–129) 0.029 (0.018–0.043) 27 (17–40)
the Grenadines 1995 < 1 <0.01 (<0.01–<0.01) 3.7 (3.6–3.8) 0.061 (0.028–0.110) 57 (26–99) 0.029 (0.023–0.035) 27 (22–32)
2000 < 1 <0.01 (<0.01–<0.01) 3.3 (3.0–3.6) 0.055 (0.026–0.094) 51 (24–87) 0.028 (0.023–0.033) 26 (21–31)
2005 < 1 <0.01 (<0.01–<0.01) 0.86 (0.86–0.87) 0.049 (0.020–0.092) 45 (18–84) 0.027 (0.022–0.033) 25 (20–30)
2010 < 1 <0.01 (<0.01–<0.01) 4.8 (4.7–4.8) 0.035 (0.011–0.071) 32 (10–65) 0.027 (0.022–0.032) 24 (20–29)
2011 < 1 <0.01 (<0.01–<0.01) 2.6 (2.5–2.6) 0.031 (0.010–0.064) 29 (9.1–59) 0.026 (0.022–0.032) 24 (20–29)
2012 < 1 <0.01 (<0.01–<0.01) 2.6 (2.5–2.6) 0.027 (<0.01–0.061) 24 (5.6–56) 0.026 (0.022–0.031) 24 (20–29)
Sint Maarten 2010 < 1 <0.01 (0–<0.01) 0.4 (0–2.0) <0.01 (<0.01–0.011) 11 (3.0–25) <0.01 (<0.01–<0.01) 8.1 (7.1–9.2)
(Dutch part) 2011 < 1 <0.01 (0–<0.01) 0.26 (0–1.4) <0.01 (<0.01–<0.01) 7.4 (1.6–17) <0.01 (<0.01–<0.01) 5.3 (4.6–6.0)
2012 < 1 <0.01 (0–<0.01) 0.13 (<0.1–0.43) <0.01 (<0.01–<0.01) 3.3 (1.3–6.3) <0.01 (<0.01–<0.01) 2.6 (2.3–2.9)
Suriname 1990 < 1 0.027 (0.019–0.037) 6.7 (4.6–9.2) 0.53 (0.200–1.0) 129 (50–245) 0.26 (0.170–0.360) 63 (41–90)
1995 < 1 0.016 (<0.01–0.027) 3.6 (1.7–6.2) 0.69 (0.230–1.4) 157 (52–320) 0.4 (0.260–0.570) 92 (59–131)
2000 < 1 <0.01 (<0.01–<0.01) 1.2 (0.98–1.5) 0.6 (0.180–1.3) 128 (38–273) 0.4 (0.260–0.580) 86 (56–124)
2005 < 1 <0.01 (<0.01–<0.01) 1.7 (1.5–1.9) 0.47 (0.150–0.980) 94 (30–195) 0.31 (0.200–0.450) 63 (41–90)
2010 < 1 0.014 (0.012–0.015) 2.6 (2.3–2.9) 0.35 (0.120–0.700) 66 (22–133) 0.24 (0.160–0.340) 46 (31–65)
2011 < 1 0.014 (0.012–0.015) 2.6 (2.3–2.9) 0.33 (0.130–0.620) 62 (24–117) 0.23 (0.170–0.310) 44 (32–58)
2012 < 1 0.014 (0.012–0.016) 2.6 (2.3–2.9) 0.31 (0.120–0.590) 58 (22–110) 0.22 (0.160–0.290) 41 (30–55)
Trinidad and 1990 1 0.032 (0.031–0.033) 2.6 (2.5–2.7) 0.21 (0.099–0.350) 17 (8.1–29) 0.14 (0.120–0.160) 11 (9.9–13)
Tobago 1995 1 0.034 (0.033–0.034) 2.7 (2.7–2.7) 0.23 (0.090–0.440) 19 (7.2–35) 0.19 (0.170–0.220) 15 (13–17)
2000 1 0.025 (0.024–0.025) 1.9 (1.9–2.0) 0.29 (0.140–0.490) 23 (11–39) 0.23 (0.200–0.260) 18 (16–20)
2005 1 0.018 (0.018–0.018) 1.4 (1.4–1.4) 0.2 (0.073–0.390) 15 (5.7–30) 0.19 (0.170–0.220) 15 (13–17)
2010 1 0.028 (0.028–0.028) 2.1 (2.1–2.1) 0.26 (0.096–0.500) 20 (7.2–38) 0.25 (0.220–0.290) 19 (17–21)
2011 1 0.028 (0.028–0.028) 2.1 (2.1–2.1) 0.26 (0.096–0.520) 20 (7.2–39) 0.26 (0.230–0.290) 19 (17–22)
2012 1 0.028 (0.028–0.028) 2.1 (2.1–2.1) 0.37 (0.170–0.650) 28 (13–48) 0.32 (0.280–0.360) 24 (21–27)
Turks and Caicos 1990 < 1 0 (0–0) 0 (0–0) 0 (0–0) 0 (0–0)
Islands 1995 < 1 0 (0–0) 0 (0–0) <0.01 (<0.01–0.015) 47 (14–100) <0.01 (<0.01–<0.01) 36 (32–41)
2000 < 1 <0.01 (<0.01–<0.01) 6.1 (5.7–6.6) 0.016 (<0.01–0.029) 86 (37–156) 0.012 (0.010–0.014) 63 (55–72)
2005 < 1 <0.01 (<0.01–<0.01) 3.9 (3.9–4.0) 0.013 (<0.01–0.021) 47 (24–79) <0.01 (<0.01–<0.01) 29 (26–33)
2010 < 1 <0.01 (<0.01–<0.01) 3.5 (3.4–3.7) <0.01 (<0.01–0.015) 24 (7.5–50) <0.01 (<0.01–<0.01) 22 (20–25)
2011 < 1 <0.01 (<0.01–<0.01) 3.5 (3.4–3.7) 0.015 (<0.01–0.026) 47 (21–82) 0.01 (<0.01–0.012) 33 (29–37)
2012 < 1 <0.01 (<0.01–<0.01) 3.5 (3.4–3.7) 0.01 (<0.01–0.022) 32 (9.1–68) <0.01 (<0.01–0.010) 28 (25–32)
United States 1990 255 2.6 (2.5–2.6) 1 (0.99–1.0) 38 (15–71) 15 (5.9–28) 30 (26–33) 12 (10–13)
of America 1995 268 1.4 (1.4–1.4) 0.51 (0.50–0.52) 35 (15–62) 13 (5.7–23) 26 (23–30) 9.8 (8.5–11)
2000 285 0.81 (0.790–0.820) 0.28 (0.28–0.29) 24 (10–45) 8.6 (3.6–16) 19 (16–21) 6.6 (5.8–7.5)
2005 298 0.64 (0.640–0.650) 0.22 (0.21–0.22) 21 (9.3–38) 7.2 (3.1–13) 16 (14–18) 5.4 (4.8–6.1)
2010 312 0.61 (0.590–0.630) 0.2 (0.19–0.20) 17 (7.1–30) 5.3 (2.3–9.6) 13 (11–15) 4.1 (3.6–4.7)
2011 315 0.47 (0.440–0.500) 0.15 (0.14–0.16) 16 (6.7–28) 5 (2.1–9.0) 12 (11–14) 3.8 (3.4–4.3)
2012 318 0.44 (0.390–0.480) 0.14 (0.12–0.15) 15 (6.5–27) 4.7 (2.0–8.4) 11 (10–13) 3.6 (3.2–4.1)
Uruguay 1990 3 0.085 (0.078–0.092) 2.7 (2.5–3.0) 1.5 (0.690–2.6) 48 (22–83) 1 (0.890–1.2) 33 (29–37)
1995 3 0.076 (0.073–0.078) 2.3 (2.3–2.4) 0.91 (0.340–1.8) 28 (11–54) 0.72 (0.630–0.810) 22 (20–25)
2000 3 0.069 (0.066–0.072) 2.1 (2.0–2.2) 0.96 (0.410–1.7) 29 (12–53) 0.74 (0.650–0.840) 22 (20–25)
2005 3 0.067 (0.064–0.070) 2 (1.9–2.1) 0.89 (0.390–1.6) 27 (12–48) 0.72 (0.630–0.810) 22 (19–24)
2010 3 0.054 (0.051–0.057) 1.6 (1.5–1.7) 0.96 (0.400–1.8) 29 (12–53) 0.8 (0.700–0.910) 24 (21–27)
2011 3 0.053 (0.050–0.056) 1.6 (1.5–1.6) 1.2 (0.560–2.1) 36 (17–62) 0.94 (0.820–1.1) 28 (24–31)
2012 3 0.051 (0.048–0.054) 1.5 (1.4–1.6) 1.1 (0.490–2.1) 34 (14–61) 0.93 (0.810–1.1) 27 (24–31)
US Virgin Islands 1990 < 1 <0.01 (<0.01–<0.01) 3 (3.0–3.1) <0.01 (<0.01–0.011) 5.6 (2.1–11) <0.01 (<0.01–<0.01) 4.5 (3.9–5.0)
1995 < 1 <0.01 (<0.01–<0.01) 2.4 (2.4–2.4) <0.01 (<0.01–0.016) 7.3 (2.2–15) <0.01 (<0.01–<0.01) 4.3 (3.8–4.9)
2000 < 1 <0.01 (<0.01–<0.01) 2.8 (2.8–2.8) 0.011 (<0.01–0.020) 9.9 (3.8–19) <0.01 (<0.01–<0.01) 7.7 (6.8–8.7)
2005 < 1 <0.01 (<0.01–<0.01) 0.94 (0.94–0.95) 0.011 (<0.01–0.020) 9.9 (3.9–19) <0.01 (<0.01–<0.01) 7.7 (6.8–8.7)
2010 < 1 <0.01 (<0.01–<0.01) 0.96 (0.94–0.97) 0.011 (<0.01–0.020) 9.9 (3.9–19) <0.01 (<0.01–<0.01) 7.7 (6.8–8.7)
2011 < 1 <0.01 (<0.01–<0.01) 0.96 (0.94–0.97) 0.011 (<0.01–0.020) 9.9 (3.8–19) <0.01 (<0.01–<0.01) 7.7 (6.8–8.7)
2012 < 1 <0.01 (<0.01–<0.01) 0.96 (0.94–0.97) 0.011 (<0.01–0.020) 9.9 (3.9–19) <0.01 (<0.01–<0.01) 7.7 (6.8–8.7)
Venezuela 1990 20 0.85 (0.830–0.870) 4.3 (4.2–4.4) 11 (3.9–20) 53 (20–103) 7 (4.9–9.4) 35 (25–47)
(Bolivarian 1995 22 0.81 (0.790–0.830) 3.7 (3.6–3.8) 12 (5.4–20) 53 (25–92) 7.7 (6.3–9.2) 35 (28–42)
Republic of) 2000 24 0.67 (0.650–0.690) 2.7 (2.7–2.8) 12 (5.5–21) 50 (22–88) 8.4 (6.8–10) 34 (28–41)
2005 27 0.63 (0.630–0.630) 2.4 (2.3–2.4) 13 (6.2–23) 50 (23–87) 9 (7.4–11) 34 (28–41)
2010 29 0.71 (0.410–1.1) 2.4 (1.4–3.8) 15 (7.3–25) 52 (25–87) 9.7 (8.0–12) 33 (27–40)
2011 30 0.72 (0.410–1.1) 2.4 (1.4–3.8) 15 (7.3–25) 50 (25–85) 9.8 (8.1–12) 33 (27–40)
2012 30 0.73 (0.420–1.1) 2.4 (1.4–3.8) 15 (7.6–26) 52 (25–87) 9.9 (8.2–12) 33 (27–39)
a Rates are per 100 000 population.

GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($,QFLGHQFHQRWLILFDWLRQDQGFDVHGHWHFWLRQUDWHVDOOIRUPV±
INCIDENCE (INCLUDING HIV) INCIDENCE HIV-POSITIVE NOTIFIED NEW AND RELAPSE
b
CASE DETECTION
YEAR POPULATION
(MILLIONS)
NUMBER
(THOUSANDS) RATE
a
NUMBER
(THOUSANDS) RATE
a
NUMBER RATE
a
PERCENT
Anguilla 1990 < 1 <0.01 (<0.01–<0.01) 24 (15–35) 0 0 0
1995 < 1 <0.01 (<0.01–<0.01) 23 (20–27) 2 20 88 (75–100)
2000 < 1 <0.01 (<0.01–<0.01) 23 (18–27)
2005 < 1 <0.01 (<0.01–<0.01) 22 (18–26)
2010 < 1 <0.01 (<0.01–<0.01) 21 (18–25) 1 7.3 34 (29–41)
2011 < 1 <0.01 (<0.01–<0.01) 21 (17–25) 0 0 0
2012 < 1 <0.01 (<0.01–<0.01) 21 (17–25) 0 0 0
Antigua and 1990 < 1 <0.01 (<0.01–<0.01) 1.9 (1.6–2.1) 1 1.6 87 (77–99)
Barbuda 1995 < 1 0 (0–0) 0 (0–0) 0 0
2000 < 1 <0.01 (<0.01–<0.01) 5.9 (5.2–6.7) 4 5.2 87 (77–99)
2005 < 1 <0.01 (<0.01–<0.01) 8.4 (7.3–9.5) <0.01 (<0.01–<0.01) 4.2 (1.6–7.9) 6 7.3 87 (77–99)
2010 < 1 <0.01 (<0.01–<0.01) 9.2 (8.1–10) <0.01 (<0.01–<0.01) 7.7 (4.9–11) 7 8 87 (77–99)
2011 < 1 <0.01 (<0.01–<0.01) 7.8 (6.9–8.9) <0.01 (<0.01–<0.01) 4.9 (2.6–7.9) 6 6.8 87 (77–99)
2012 < 1 <0.01 (<0.01–<0.01) 3.9 (3.4–4.4) <0.01 (<0.01–<0.01) 1.5 (0.50–3.0) 3 3.4 87 (77–99)
Argentina 1990 33 19 (13–28) 60 (39–85) 0.16 (0.11–0.23) 0.5 (0.33–0.71) 12 309 38 63 (44–97)
1995 35 17 (14–21) 49 (40–59) 0.27 (0.22–0.32) 0.8 (0.62–0.91) 13 450 39 79 (65–96)
2000 37 15 (12–18) 40 (33–49) 0.29 (0.24–0.35) 0.8 (0.64–0.95) 11 767 32 79 (66–96)
2005 39 13 (11–15) 33 (27–40) 0.28 (0.23–0.34) 0.7 (0.59–0.87) 10 576 27 82 (68–100)
2010 40 11 (9.1–13) 27 (23–32) 0.27 (0.23–0.32) 0.7 (0.56–0.80) 7 336 18 67 (56–80)
2011 41 11 (8.9–13) 26 (22–31) 0.27 (0.23–0.32) 0.7 (0.55–0.80) 9 733 24 91 (76–110)
2012 41 10 (8.6–12) 25 (21–30) 0.27 (0.22–0.32) 0.7 (0.55–0.78) 8 758 21 84 (71–100)
Aruba 1990 < 1 <0.01 (<0.01–0.011) 16 (14–18)
1995 < 1 0.013 (0.011–0.014) 16 (14–18)
2000 < 1 0.014 (0.013–0.016) 16 (14–18)
2005 < 1 0.016 (0.014–0.018) 16 (14–18)
2010 < 1 0.016 (0.014–0.018) 16 (14–18) 6 5.9 37 (33–43)
2011 < 1 0.016 (0.014–0.018) 16 (14–18) 8 7.8 50 (44–57)
2012 < 1 0.016 (0.014–0.018) 16 (14–18) 28 27 170 (150–200)
Bahamas 1990 < 1 0.053 (0.046–0.060) 21 (18–23) 0.019 (0.017–0.021) 7.4 (6.5–8.3) 46 18 87 (77–99)
1995 < 1 0.066 (0.057–0.074) 23 (21–26) 0.029 (0.026–0.033) 10 (9.1–12) 57 20 87 (77–99)
2000 < 1 0.094 (0.083–0.110) 32 (28–36) 0.041 (0.036–0.047) 14 (12–16) 82 28 87 (77–99)
2005 < 1 0.055 (0.048–0.062) 17 (15–19) 0.022 (0.020–0.025) 6.8 (5.9–7.7) 48 15 87 (77–99)
2010 < 1 0.036 (0.031–0.040) 9.9 (8.7–11) 0.013 (0.012–0.015) 3.7 (3.2–4.2) 31 8.6 87 (77–99)
2011 < 1 0.047 (0.041–0.053) 13 (11–15) 0.012 (0.011–0.014) 3.3 (2.9–3.7) 41 11 87 (77–99)
2012 < 1 0.037 (0.032–0.042) 9.9 (8.7–11) <0.01 (<0.01–<0.01) 2.3 (2.0–2.6) 32 8.6 87 (77–99)
Barbados 1990 < 1 <0.01 (<0.01–<0.01) 2.2 (1.9–2.5) <0.01 (<0.01–<0.01) 0.1 (0.12–0.15) 5 1.9 87 (77–99)
1995 < 1 <0.01 (<0.01–<0.01) 1.3 (1.1–1.5) <0.01 (<0.01–<0.01) 0.2 (0.14–0.18) 3 1.1 87 (77–99)
2000 < 1 <0.01 (<0.01–<0.01) 1.3 (1.1–1.5) <0.01 (<0.01–<0.01) 0.3 (0.24–0.31) 3 1.1 87 (77–99)
2005 < 1 0.014 (0.012–0.016) 5.1 (4.4–5.7) <0.01 (<0.01–<0.01) 1.6 (1.4–1.8)
2010 < 1 <0.01 (<0.01–<0.01) 2.5 (2.2–2.8) <0.01 (<0.01–<0.01) 1 (0.83–1.1) 6 2.1 87 (77–99)
2011 < 1 0 (0–0) 0 (0–0) 0 0
2012 < 1 <0.01 (<0.01–<0.01) 1.6 (1.4–1.8) <0.01 (<0.01–<0.01) 0.5 (0.47–0.60) 4 1.4 87 (77–99)
Belize 1990 < 1 0.075 (0.052–0.100) 40 (28–54) <0.01 (<0.01–<0.01) 2.8 (1.9–3.8) 57 30 76 (56–110)
1995 < 1 0.083 (0.068–0.099) 40 (33–48) 0.011 (<0.01–0.013) 5.3 (4.3–6.4) 95 46 110 (96–140)
2000 < 1 0.095 (0.078–0.110) 40 (33–48) 0.018 (0.015–0.022) 7.5 (6.2–9.1) 106 44 110 (93–140)
2005 < 1 0.11 (0.094–0.120) 40 (34–46) 0.022 (0.019–0.026) 8.2 (7.1–9.4) 102 38 94 (82–110)
2010 < 1 0.12 (0.100–0.150) 40 (33–48) 0.025 (0.021–0.030) 8.2 (6.7–9.8) 145 47 120 (98–140)
2011 < 1 0.13 (0.100–0.150) 40 (33–48) 0.026 (0.021–0.031) 8.1 (6.6–9.7) 74 23 59 (49–72)
2012 < 1 0.13 (0.110–0.160) 40 (33–48) 0.026 (0.021–0.032) 8.1 (6.6–9.7) 84 26 65 (54–79)
Bermuda 1990 < 1 0 (0–0) 0 (0–0) 0 0
1995 < 1 <0.01 (<0.01–<0.01) 7.5 (6.6–8.5) 4 6.5 87 (77–99)
2000 < 1 0 (0–0) 0 (0–0) 0 0
2005 < 1 <0.01 (<0.01–<0.01) 8.1 (7.1–9.1)
2010 < 1 <0.01 (<0.01–<0.01) 1.8 (1.6–2.0) 1 1.5 87 (77–99)
2011 < 1 <0.01 (<0.01–<0.01) 1.8 (1.5–2.0) 1 1.5 87 (77–99)
2012 < 1 <0.01 (<0.01–<0.01) 5.3 (4.6–6.0) 3 4.6 87 (77–99)
Bolivia 1990 7 17 (11–24) 251 (166–354) 0.86 (0.57–1.2) 13 (8.4–18) 11 166 164 65 (46–99)
(Plurinational 1995 8 16 (14–19) 215 (185–248) 0.94 (0.80–1.1) 12 (11–14) 14 422 189 88 (76–100)
State of) 2000 8 16 (13–19) 184 (151–221) 0.86 (0.71–1.0) 10 (8.3–12) 10 127 119 65 (54–79)
2005 9 15 (12–18) 158 (129–190) 0.77 (0.63–0.92) 8.2 (6.7–9.8) 9 748 104 66 (55–81)
2010 10 14 (11–16) 135 (111–161) 0.52 (0.43–0.62) 5.1 (4.2–6.1) 8 363 82 61 (51–74)
2011 10 14 (11–16) 131 (108–156) 0.48 (0.40–0.57) 4.7 (3.8–5.5) 8 521 83 63 (53–76)
2012 10 13 (11–16) 127 (105–151) 0.43 (0.36–0.52) 4.1 (3.4–4.9) 8 257 79 62 (52–75)
Bonaire, Saint 2010 < 1 0 (0–0) 0 (0–0) 0 0
Eustatius and Sab
a
2011 < 1 <0.01 (<0.01–<0.01) 6.3 (5.5–7.2) 1 5.5 87 (77–99)
2012 < 1 0 (0–0) 0 (0–0) 0 0
Brazil 1990 150 130 (79–180) 84 (53–121) 5.5 (3.5–8.0) 3.7 (2.3–5.3) 74 570 50 60 (41–94)
1995 162 120 (94–140) 71 (58–85) 12 (9.9–15) 7.5 (6.1–9.0) 91 013 56 79 (66–97)
2000 175 110 (86–130) 60 (49–72) 12 (9.8–14) 6.9 (5.6–8.3) 77 899 45 74 (62–91)
2005 186 95 (80–110) 51 (43–60) 15 (12–18) 8 (6.7–9.4) 80 675 43 85 (72–100)
2010 195 91 (75–110) 46 (38–55) 17 (14–20) 8.5 (7.1–10) 74 395 38 82 (69–99)
2011 197 95 (78–110) 48 (40–57) 17 (14–20) 8.6 (7.1–10) 77 647 39 82 (69–99)
2012 199 92 (76–110) 46 (38–55) 16 (13–19) 8 (6.6–9.5) 75 122 38 82 (69–99)
British Virgin 1990 < 1 <0.01 (<0.01–<0.01) 17 (15–20)
Islands 1995 < 1 <0.01 (<0.01–<0.01) 17 (15–20)
2000 < 1 <0.01 (<0.01–<0.01) 5.6 (4.9–6.3) 1 4.8 87 (77–99)
2005 < 1 0 (0–0) 0 (0–0) 0 0
2010 < 1 <0.01 (<0.01–<0.01) 4.2 (3.7–4.8) 1 3.7 87 (77–99)
2011 < 1 0 (0–0) 0 (0–0) 0 0
2012 < 1 0 (0–0) 0 (0–0) 0 0
Canada 1990 28 2.3 (2.0–2.6) 8.3 (7.3–9.4) 0.11 (0.095–0.12) 0.4 (0.34–0.44) 1 997 7.2 87 (77–99)
1995 29 2.3 (2.0–2.6) 7.7 (6.8–8.7) 0.17 (0.15–0.20) 0.6 (0.52–0.67) 1 965 6.7 87 (77–99)
2000 31 2 (1.7–2.2) 6.5 (5.7–7.3) 0.098 (0.086–0.11) 0.3 (0.28–0.36) 1 723 5.6 87 (77–99)
2005 32 1.8 (1.6–2.0) 5.5 (4.8–6.3) 0.11 (0.094–0.12) 0.3 (0.29–0.38) 1 552 4.8 87 (77–99)
2010 34 1.6 (1.4–1.8) 4.6 (4.0–5.2) 0.11 (0.095–0.12) 0.3 (0.28–0.36) 1 361 4 87 (77–99)
2011 34 1.6 (1.4–1.9) 4.8 (4.2–5.4) 0.11 (0.098–0.13) 0.3 (0.28–0.37) 1 430 4.1 87 (77–99)
2012 35 1.6 (1.4–1.8) 4.6 (4.0–5.2) 0.11 (0.096–0.12) 0.3 (0.28–0.36) 1 653 4.7 100 (91–120)
Cayman Islands 1990 < 1 <0.01 (<0.01–<0.01) 9.2 (8.1–10) 2 8 87 (77–99)
1995 < 1 <0.01 (<0.01–<0.01) 7.3 (6.4–8.2) 2 6.3 87 (77–99)
2000 < 1 <0.01 (<0.01–<0.01) 14 (12–16) 5 12 87 (77–99)
2005 < 1 <0.01 (<0.01–<0.01) 1.2 (1.1–1.4)
2010 < 1 <0.01 (<0.01–<0.01) 8.3 (7.3–9.4) 4 7.2 87 (77–99)
2011 < 1 <0.01 (<0.01–<0.01) 4.1 (3.6–4.6) 2 3.5 87 (77–99)
2012 < 1 <0.01 (<0.01–<0.01) 12 (11–14) 6 10 87 (77–99)
Chile 1990 13 7.1 (6.2–8.0) 54 (47–61) 0.021 (0.019–0.024) 0.2 (0.14–0.18) 6 151 47 87 (77–99)
1995 14 4.8 (4.2–5.4) 33 (29–37) 0.043 (0.038–0.049) 0.3 (0.26–0.34) 4 150 29 87 (77–99)
2000 15 3.5 (3.0–3.9) 22 (20–25) 0.072 (0.063–0.081) 0.5 (0.41–0.53) 3 021 20 87 (77–99)
2005 16 2.9 (2.5–3.3) 18 (15–20) 0.084 (0.074–0.095) 0.5 (0.45–0.58) 2 505 15 87 (77–99)
2010 17 2.7 (2.4–3.1) 16 (14–18) 0.086 (0.076–0.098) 0.5 (0.44–0.57) 2 376 14 87 (77–99)
2011 17 2.8 (2.5–3.2) 16 (14–18) 0.088 (0.077–0.099) 0.5 (0.44–0.57) 2 450 14 87 (77–99)
2012 17 2.8 (2.4–3.1) 16 (14–18) 0.084 (0.074–0.095) 0.5 (0.42–0.54) 2 394 14 87 (77–99)
Colombia 1990 33 18 (12–25) 54 (36–75) 0.36 (0.24–0.50) 1.1 (0.73–1.5) 12 447 37 70 (50–100)
1995 37 18 (14–21) 48 (39–58) 1.2 (0.95–1.4) 3.2 (2.6–3.8) 9 912 27 56 (47–69)
2000 40 17 (14–21) 43 (35–52) 1.7 (1.4–2.0) 4.3 (3.5–5.1) 11 630 29 68 (56–83)
2005 43 17 (14–20) 38 (31–46) 1.8 (1.4–2.1) 4.1 (3.3–4.9) 10 360 24 62 (52–76)
2010 46 16 (13–19) 34 (28–41) 1.8 (1.5–2.1) 3.8 (3.1–4.6) 11 420 25 71 (60–86)
2011 47 16 (13–19) 34 (28–40) 1.7 (1.4–2.0) 3.5 (2.9–4.2) 11 884 25 75 (63–91)
2012 48 16 (13–19) 33 (27–39) 1.6 (1.3–1.9) 3.3 (2.7–4.0) 11 424 24 73 (61–88)
a Rates are per 100 000 population.
b NOTIFIED NEW AND RELAPSE includes cases for which the treatment history is unknown.

Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
REGION OF THE AMERICAS
7$%/($,QFLGHQFHQRWLILFDWLRQDQGFDVHGHWHFWLRQUDWHVDOOIRUPV±
INCIDENCE (INCLUDING HIV) INCIDENCE HIV-POSITIVE NOTIFIED NEW AND RELAPSE
b
CASE DETECTION
YEAR POPULATION
(MILLIONS)
NUMBER
(THOUSANDS) RATE
a
NUMBER
(THOUSANDS) RATE
a
NUMBER RATE
a
PERCENT
Costa Rica 1990 3 1.5 (1.3–1.7) 48 (42–54) 0.031 (0.027–0.035) 1 (0.87–1.1) 230 7.5 16 (14–18)
1995 3 1.5 (1.3–1.7) 43 (37–48) 0.066 (0.058–0.075) 1.9 (1.7–2.2) 586 17 40 (35–45)
2000 4 1.4 (1.2–1.5) 35 (31–39) 0.092 (0.080–0.10) 2.3 (2.0–2.7) 585 15 43 (38–49)
2005 4 1 (0.880–1.1) 23 (20–26) 0.074 (0.065–0.084) 1.7 (1.5–1.9) 534 12 53 (47–61)
2010 5 0.65 (0.570–0.740) 14 (12–16) 0.074 (0.065–0.084) 1.6 (1.4–1.8) 492 11 75 (67–86)
2011 5 0.59 (0.510–0.660) 12 (11–14) 0.071 (0.062–0.080) 1.5 (1.3–1.7) 514 11 88 (78–100)
2012 5 0.51 (0.440–0.580) 11 (9.3–12) 0.064 (0.056–0.072) 1.3 (1.2–1.5) 475 9.9 93 (82–110)
Cuba 1990 11 2.6 (1.6–3.9) 25 (15–37) 0.023 (0.014–0.034) 0.2 (0.13–0.32) 546 5.2 21 (14–34)
1995 11 2 (1.7–2.5) 19 (15–23) 0.028 (0.023–0.034) 0.3 (0.21–0.31) 1 553 14 76 (63–94)
2000 11 1.4 (1.1–1.8) 13 (10–16) 0.033 (0.026–0.040) 0.3 (0.23–0.36) 1 183 11 82 (67–100)
2005 11 1 (0.850–1.3) 9.2 (7.5–11) 0.023 (0.019–0.028) 0.2 (0.16–0.24) 772 6.8 74 (61–91)
2010 11 1 (0.840–1.3) 9.3 (7.4–11) 0.035 (0.028–0.043) 0.3 (0.25–0.38) 827 7.3 79 (65–99)
2011 11 1 (0.830–1.3) 9.3 (7.4–11) 0.037 (0.030–0.045) 0.3 (0.26–0.40) 805 7.1 77 (63–97)
2012 11 1 (0.840–1.3) 9.3 (7.4–11) 0.037 (0.030–0.046) 0.3 (0.26–0.41) 734 6.5 70 (57–88)
Curaçao 2010 < 1 <0.01 (<0.01–<0.01) 3.9 (3.4–4.4) 5 3.4 87 (77–99)
2011 < 1 <0.01 (<0.01–<0.01) 0.76 (0.67–0.86) 1 0.66 87 (77–99)
2012 < 1 <0.01 (<0.01–<0.01) 0.74 (0.65–0.84) 1 0.64 87 (77–99)
Dominica 1990 < 1 0.01 (<0.01–0.015) 15 (9.3–21) 6 8.5 57 (40–91)
1995 < 1 0.01 (<0.01–0.012) 14 (12–17) 8 11 78 (66–95)
2000 < 1 <0.01 (<0.01–0.012) 14 (11–17)
2005 < 1 <0.01 (<0.01–0.011) 13 (11–16)
2010 < 1 <0.01 (<0.01–0.011) 13 (11–16) 8 11 86 (72–100)
2011 < 1 <0.01 (<0.01–0.011) 13 (11–16) 2 2.8 22 (18–26)
2012 < 1 <0.01 (<0.01–0.011) 13 (11–15) 7 9.8 75 (63–92)
Dominican 1990 7 11 (6.6–16) 148 (91–218) 0.25 (0.15–0.37) 3.5 (2.1–5.1) 2 597 36 24 (16–39)
Republic 1995 8 9.7 (7.9–12) 121 (99–146) 0.81 (0.66–0.98) 10 (8.3–12) 4 053 51 42 (35–51)
2000 9 8.6 (7.1–10) 100 (82–120) 1.1 (0.90–1.3) 13 (10–15) 5 291 61 61 (51–75)
2005 9 7.7 (6.3–9.2) 82 (67–98) 0.98 (0.80–1.2) 11 (8.6–13) 5 003 54 65 (54–80)
2010 10 6.7 (5.6–8.0) 67 (55–80) 0.64 (0.53–0.76) 6.4 (5.2–7.6) 3 964 40 59 (49–71)
2011 10 6.6 (5.4–7.8) 65 (53–77) 0.59 (0.49–0.71) 5.8 (4.8–7.0) 4 309 42 66 (55–80)
2012 10 6.4 (5.3–7.6) 62 (51–74) 0.54 (0.44–0.64) 5.2 (4.3–6.3) 4 262 41 67 (56–81)
Ecuador 1990 10 18 (11–26) 174 (108–257) 0.71 (0.44–1.0) 7 (4.3–10) 8 243 81 47 (32–76)
1995 11 15 (13–19) 136 (111–164) 0.96 (0.78–1.2) 8.5 (6.9–10) 7 893 70 51 (43–63)
2000 13 13 (11–16) 107 (87–128) 1.2 (0.96–1.4) 9.4 (7.7–11) 6 908 55 52 (43–63)
2005 14 11 (9.4–14) 83 (68–100) 1.2 (0.99–1.5) 8.8 (7.2–11) 4 416 32 38 (32–47)
2010 15 9.7 (8.0–12) 65 (54–77) 0.97 (0.80–1.2) 6.5 (5.3–7.7) 4 832 32 50 (42–60)
2011 15 9.4 (7.8–11) 62 (51–74) 0.92 (0.76–1.1) 6 (5.0–7.2) 5 106 33 54 (45–66)
2012 15 9.1 (7.5–11) 59 (48–70) 0.84 (0.70–1.0) 5.4 (4.5–6.5) 5 456 35 60 (50–73)
El Salvador 1990 5 3.4 (2.3–4.7) 63 (43–88) 0.082 (0.055–0.11) 1.5 (1.0–2.1) 2 367 44 70 (50–100)
1995 6 2.6 (2.3–2.9) 45 (39–50) 0.16 (0.14–0.18) 2.7 (2.4–3.1) 2 422 42 95 (84–110)
2000 6 2.2 (1.8–2.6) 37 (30–44) 0.23 (0.19–0.28) 3.9 (3.2–4.7) 1 485 25 68 (56–83)
2005 6 2.4 (1.9–2.9) 39 (32–47) 0.27 (0.22–0.32) 4.4 (3.6–5.2) 1 794 30 75 (63–92)
2010 6 1.8 (1.5–2.0) 28 (24–33) 0.19 (0.16–0.21) 3 (2.5–3.4) 1 700 27 96 (83–110)
2011 6 1.7 (1.4–1.9) 27 (23–31) 0.18 (0.15–0.20) 2.8 (2.4–3.2) 1 896 30 110 (98–130)
2012 6 1.6 (1.4–1.8) 25 (22–29) 0.2 (0.17–0.23) 3.2 (2.7–3.7) 2 053 33 130 (110–150)
Grenada 1990 < 1 <0.01 (<0.01–<0.01) 4.6 (2.9–6.8) 0 0 0
1995 < 1 <0.01 (<0.01–<0.01) 4.5 (3.8–5.2) 4 4 89 (77–100)
2000 < 1 <0.01 (<0.01–<0.01) 4.4 (3.6–5.2) 0 0 0
2005 < 1 <0.01 (<0.01–<0.01) 4.2 (3.5–5.1)
2010 < 1 <0.01 (<0.01–<0.01) 4.1 (3.4–4.9) <0.01 (<0.01–<0.01) 1 (<0.1–3.2) 4 3.8 93 (78–110)
2011 < 1 <0.01 (<0.01–<0.01) 4.1 (3.4–4.9) 2 1.9 46 (39–56)
2012 < 1 <0.01 (<0.01–<0.01) 4.1 (3.4–4.9) <0.01 (0–<0.01) 0.5 (0–3.0) 1 0.95 23 (19–28)
Guatemala 1990 9 6.6 (4.1–9.7) 74 (47–109) 0.14 (0.086–0.20) 1.5 (0.97–2.3) 3 813 43 58 (39–92)
1995 10 7.1 (5.8–8.5) 71 (58–85) 0.38 (0.31–0.46) 3.8 (3.1–4.6) 3 119 31 44 (37–54)
2000 11 7.6 (6.2–9.1) 68 (55–81) 0.78 (0.63–0.93) 6.9 (5.7–8.3) 2 913 26 38 (32–47)
2005 13 8.2 (6.7–9.9) 65 (53–78) 0.99 (0.81–1.2) 7.8 (6.4–9.4) 3 803 30 46 (39–57)
2010 14 8.8 (7.3–11) 62 (51–73) 1.3 (1.1–1.5) 9 (7.4–11) 3 322 23 38 (32–46)
2011 15 9 (7.4–11) 61 (50–73) 1.4 (1.1–1.7) 9.5 (7.8–11) 3 040 21 34 (28–41)
2012 15 9.1 (7.5–11) 60 (50–72) 1.5 (1.2–1.8) 10 (8.2–12) 3 442 23 38 (32–46)
Guyana 1990 < 1 0.65 (0.400–0.960) 89 (55–132) 0.05 (0.031–0.074) 6.9 (4.3–10) 168 23 26 (18–42)
1995 < 1 0.65 (0.530–0.780) 89 (73–107) 0.12 (0.098–0.14) 16 (13–20) 296 41 45 (38–56)
2000 < 1 0.78 (0.630–0.930) 104 (85–125) 0.24 (0.20–0.29) 33 (27–39) 422 57 54 (45–67)
2005 < 1 0.88 (0.720–1.1) 115 (94–138) 0.27 (0.22–0.33) 36 (29–43) 639 84 73 (61–89)
2010 < 1 0.87 (0.720–1.0) 111 (91–132) 0.22 (0.18–0.27) 28 (23–34) 712 91 82 (68–99)
2011 < 1 0.87 (0.720–1.0) 110 (91–131) 0.22 (0.18–0.26) 27 (22–33) 710 90 82 (68–99)
2012 < 1 0.87 (0.710–1.0) 109 (90–130) 0.2 (0.16–0.23) 25 (20–29) 748 94 86 (72–100)
Haiti 1990 7 18 (11–26) 247 (153–365) 3.2 (2.0–4.8) 45 (28–67)
1995 8 19 (16–23) 247 (202–297) 5.6 (4.6–6.8) 72 (59–87) 6 212 79 32 (27–39)
2000 9 23 (19–28) 271 (221–325) 6.8 (5.6–8.2) 79 (65–95) 10 420 121 45 (37–55)
2005 9 25 (21–30) 272 (222–326) 6.5 (5.3–7.9) 71 (58–85) 14 311 155 57 (47–70)
2010 10 23 (19–27) 230 (190–275) 4.6 (3.8–5.5) 47 (39–56) 14 222 144 62 (52–76)
2011 10 22 (18–27) 222 (183–265) 4.5 (3.7–5.4) 45 (37–54) 14 315 143 64 (54–78)
2012 10 22 (18–26) 213 (176–254) 4.3 (3.5–5.1) 42 (35–50) 16 568 163 76 (64–93)
Honduras 1990 5 5.6 (3.6–7.9) 113 (73–162) 0.24 (0.16–0.35) 4.9 (3.2–7.1) 3 647 74 66 (46–100)
1995 6 6.4 (4.1–9.2) 115 (74–164) 0.77 (0.49–1.1) 14 (8.7–20) 4 984 89 78 (54–120)
2000 6 7.1 (4.6–10) 114 (74–163) 0.84 (0.52–1.2) 14 (8.4–20) 6 406 103 90 (63–140)
2005 7 5 (3.2–7.2) 73 (47–104) 0.37 (0.23–0.54) 5.4 (3.3–7.9) 3 333 48 66 (46–100)
2010 8 4.1 (2.7–5.9) 54 (35–77) 0.19 (0.12–0.28) 2.5 (1.5–3.7) 2 876 38 70 (49–110)
2011 8 4.2 (2.7–6.0) 54 (35–77) 0.18 (0.11–0.26) 2.3 (1.4–3.4) 3 233 42 77 (54–120)
2012 8 4.3 (2.8–6.1) 54 (35–77) 0.17 (0.10–0.25) 2.1 (1.3–3.2) 3 014 38 70 (49–110)
Jamaica 1990 2 0.15 (0.110–0.210) 6.5 (4.7–8.8) 0.01 (<0.01–0.013) 0.4 (0.30–0.57) 123 5.2 79 (59–110)
1995 2 0.16 (0.130–0.190) 6.5 (5.4–7.9) 0.033 (0.027–0.040) 1.4 (1.1–1.6) 109 4.4 68 (56–83)
2000 3 0.17 (0.140–0.200) 6.5 (5.4–7.9) 0.052 (0.042–0.062) 2 (1.6–2.4) 127 4.9 75 (63–92)
2005 3 0.18 (0.140–0.210) 6.5 (5.4–7.9) 0.053 (0.043–0.064) 2 (1.6–2.4) 90 3.4 51 (43–63)
2010 3 0.18 (0.150–0.210) 6.6 (5.4–7.8) 0.042 (0.034–0.050) 1.5 (1.3–1.8) 130 4.7 72 (61–88)
2011 3 0.18 (0.150–0.220) 6.6 (5.4–7.8) 0.041 (0.034–0.049) 1.5 (1.2–1.8) 105 3.8 58 (49–70)
2012 3 0.18 (0.150–0.220) 6.6 (5.4–7.8) 0.04 (0.033–0.048) 1.5 (1.2–1.7) 91 3.3 50 (42–61)
Mexico 1990 86 57 (49–66) 67 (57–77) 2.6 (2.2–3.0) 3 (2.6–3.5) 14 437 17 25 (22–29)
1995 95 44 (38–51) 46 (40–53) 2.6 (2.2–3.0) 2.7 (2.3–3.1) 11 329 12 26 (22–30)
2000 104 32 (27–37) 31 (26–36) 2 (1.7–2.3) 1.9 (1.6–2.2) 18 434 18 58 (50–67)
2005 111 25 (21–28) 22 (19–26) 1.5 (1.3–1.7) 1.3 (1.1–1.5) 18 524 17 75 (66–87)
2010 118 26 (23–30) 22 (19–26) 1.5 (1.3–1.7) 1.3 (1.1–1.5) 20 155 17 77 (66–89)
2011 119 27 (23–31) 23 (19–26) 1.6 (1.3–1.8) 1.3 (1.1–1.5) 19 857 17 73 (63–86)
2012 121 27 (23–32) 23 (19–26) 1.6 (1.4–1.8) 1.3 (1.1–1.5) 20 470 17 75 (64–87)
Montserrat 1990 < 1 <0.01 (<0.01–<0.01) 11 (9.4–12) 1 9.3 87 (77–99)
1995 < 1 <0.01 (<0.01–<0.01) 4.1 (3.6–4.7)
2000 < 1 0 (0–0) 0 (0–0) 0 0
2005 < 1 <0.01 (<0.01–<0.01) 24 (21–27) 1 21 87 (77–99)
2010 < 1 0 (0–0) 0 (0–0) 0 0
2011 < 1 0 (0–0) 0 (0–0) 0 0
2012 < 1 0 (0–0) 0 (0–0) 0 0
Netherlands 1990 < 1 <0.01 (<0.01–0.011) 5.3 (4.6–6.0)
Antilles 1995 < 1 0.01 (<0.01–0.012) 5.3 (4.6–6.0)
2000 < 1 <0.01 (<0.01–<0.01) 3.2 (2.8–3.7) 5 2.8 87 (77–99)
2005 < 1 <0.01 (<0.01–<0.01) 4.7 (4.1–5.3)
Nicaragua 1990 4 4.5 (2.9–6.3) 108 (71–152) 0.015 (<0.01–0.025) 0.4 (0.18–0.61) 2 944 71 66 (47–100)
1995 5 4 (3.2–4.8) 85 (70–102) 0.02 (0.011–0.031) 0.4 (0.24–0.66) 2 842 61 72 (60–88)
2000 5 3.4 (2.8–4.1) 68 (55–81) 0.027 (0.015–0.041) 0.5 (0.30–0.81) 2 402 47 70 (58–85)
2005 5 2.9 (2.4–3.5) 53 (44–64) 0.034 (0.020–0.053) 0.6 (0.36–0.97) 1 907 35 65 (54–80)
2010 6 2.5 (2.1–2.8) 42 (36–49) 0.042 (0.025–0.062) 0.7 (0.43–1.1) 2 448 42 100 (86–120)
2011 6 2.4 (2.0–2.7) 40 (35–46) 0.044 (0.027–0.066) 0.8 (0.45–1.1) 2 693 46 110 (98–130)
2012 6 2.3 (2.0–2.7) 38 (33–44) 0.047 (0.028–0.070) 0.8 (0.47–1.2) 2 790 47 120 (110–140)
a Rates are per 100 000 population.
b NOTIFIED NEW AND RELAPSE includes cases for which the treatment history is unknown.

GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($,QFLGHQFHQRWLILFDWLRQDQGFDVHGHWHFWLRQUDWHVDOOIRUPV±
INCIDENCE (INCLUDING HIV) INCIDENCE HIV-POSITIVE NOTIFIED NEW AND RELAPSE
b
CASE DETECTION
YEAR POPULATION
(MILLIONS)
NUMBER
(THOUSANDS) RATEaNUMBER
(THOUSANDS) RATEaNUMBER RATEaPERCENT
Panama 1990 2 1.2 (0.810–1.6) 47 (33–65) 0.072 (0.050–0.099) 2.9 (2.0–4.0) 846 34 72 (52–100)
1995 3 1.3 (1.1–1.6) 47 (39–57) 0.21 (0.17–0.25) 7.5 (6.1–9.0) 1 300 47 99 (83–120)
2000 3 1.4 (1.2–1.7) 47 (39–56) 0.29 (0.24–0.34) 9.5 (7.9–11) 1 169 38 81 (68–97)
2005 3 1.6 (1.3–1.9) 47 (39–57) 0.3 (0.24–0.36) 8.8 (7.2–11) 1 637 49 100 (85–130)
2010 4 1.8 (1.5–2.0) 48 (42–54) 0.25 (0.22–0.28) 6.7 (5.9–7.6) 1 496 41 85 (76–97)
2011 4 1.8 (1.6–2.0) 48 (42–54) 0.24 (0.21–0.27) 6.5 (5.7–7.3) 1 571 42 88 (78–100)
2012 4 1.8 (1.6–2.0) 48 (42–54) 0.23 (0.20–0.26) 6.1 (5.3–6.8) 1 520 40 84 (75–95)
Paragua
y
1990 4 2.8 (2.6–3.0) 66 (61–72) 0.05 (0.046–0.055) 1.2 (1.1–1.3) 2 167 51 77 (71–84)
1995 5 2.5 (2.3–2.7) 52 (48–56) 0.1 (0.093–0.11) 2.1 (1.9–2.3) 1 745 36 70 (65–75)
2000 5 2.6 (2.4–2.8) 49 (45–53) 0.15 (0.13–0.16) 2.7 (2.5–2.9) 1 950 36 74 (69–81)
2005 6 2.9 (2.7–3.1) 49 (45–53) 0.2 (0.18–0.21) 3.3 (3.1–3.6) 2 075 35 71 (66–77)
2010 6 3 (2.7–3.2) 46 (42–50) 0.2 (0.19–0.22) 3.2 (2.9–3.4) 2 352 36 79 (73–86)
2011 7 3 (2.8–3.2) 45 (42–49) 0.22 (0.20–0.24) 3.4 (3.1–3.6) 2 372 36 79 (73–86)
2012 7 3 (2.8–3.2) 45 (41–48) 0.24 (0.22–0.26) 3.5 (3.3–3.8) 2 416 36 81 (75–88)
Peru 1990 22 69 (43–100) 317 (196–468) 0.58 (0.36–0.86) 2.7 (1.6–3.9) 37 905 174 55 (37–89)
1995 24 58 (47–70) 242 (198–290) 1.2 (0.98–1.4) 5 (4.1–6.0) 45 310 189 78 (65–96)
2000 26 48 (39–57) 184 (151–221) 1.3 (1.0–1.5) 4.9 (4.0–5.8) 38 661 149 81 (67–98)
2005 28 39 (33–46) 140 (118–164) 0.89 (0.75–1.0) 3.2 (2.7–3.8) 33 747 122 87 (74–100)
2010 29 31 (27–35) 106 (93–120) 0.53 (0.46–0.60) 1.8 (1.6–2.0) 31 073 106 100 (88–110)
2011 30 30 (26–34) 101 (88–114) 0.5 (0.44–0.57) 1.7 (1.5–1.9) 31 241 105 100 (93–120)
2012 30 29 (25–32) 95 (83–108) 0.49 (0.43–0.55) 1.6 (1.4–1.8) 29 760 99 100 (92–120)
Puerto Rico 1990 4 0.18 (0.160–0.210) 5.2 (4.6–5.9) 159 4.5 87 (77–99)
1995 4 0.3 (0.260–0.340) 8.2 (7.2–9.2) 262 7.1 87 (77–99)
2000 4 0.2 (0.180–0.230) 5.3 (4.6–6.0) 174 4.6 87 (77–99)
2005 4 0.13 (0.110–0.150) 3.5 (3.0–3.9) 0.039 (0.027–0.054) 1 (0.72–1.4) 113 3 87 (77–99)
2010 4 0.092 (0.081–0.100) 2.5 (2.2–2.8) 0.017 (<0.01–0.027) 0.5 (0.25–0.72) 80 2.2 87 (77–99)
2011 4 0.058 (0.050–0.065) 1.6 (1.4–1.8) 0.012 (<0.01–0.020) 0.3 (0.15–0.53) 50 1.4 87 (77–99)
2012 4 0.082 (0.072–0.092) 2.2 (1.9–2.5) 0.013 (<0.01–0.024) 0.4 (0.14–0.65) 71 1.9 87 (77–99)
Saint Kitts and 1990 < 1 0 (0–0) 0 (0–0) 0 0
Nevis 1995 < 1 <0.01 (<0.01–<0.01) 13 (12–15) 5 12 87 (77–99)
2000 < 1 0 (0–0) 0 (0–0) 0 0
2005 < 1 0 (0–0) 0 (0–0) 0 0
2010 < 1 <0.01 (<0.01–<0.01) 4.4 (3.8–5.0) 2 3.8 87 (77–99)
2011 < 1 <0.01 (<0.01–<0.01) 2.2 (1.9–2.5) 1 1.9 87 (77–99)
2012 < 1 <0.01 (<0.01–<0.01) 4.3 (3.8–4.9) 2 3.7 87 (77–99)
Saint Lucia 1990 < 1 0.021 (0.019–0.024) 15 (13–17) 13 9.4 61 (54–70)
1995 < 1 0.027 (0.023–0.030) 18 (16–21) 11 7.5 41 (36–47)
2000 < 1 0.018 (0.016–0.021) 12 (10–13) 9 5.7 49 (43–56)
2005 < 1 0.018 (0.016–0.020) 11 (9.5–12) 14 8.5 78 (69–89)
2010 < 1 0.012 (0.011–0.014) 6.9 (6.1–7.8) 9 5.1 73 (65–84)
2011 < 1 <0.01 (<0.01–0.010) 5.1 (4.5–5.8) <0.01 (<0.01–<0.01) 0.7 (<0.1–2.7) 7 3.9 76 (67–87)
2012 < 1 <0.01 (<0.01–<0.01) 3.3 (2.9–3.7) <0.01 (<0.01–<0.01) 0.4 (0–1.7) 11 6.1 180 (160–210)
Saint Vincent and 1990 < 1 0.029 (0.018–0.043) 27 (17–40) 2 1.9 6.8 (4.6–11)
the Grenadines 1995 < 1 0.029 (0.023–0.035) 27 (22–32) 13 12 45 (38–55)
2000 < 1 0.028 (0.023–0.033) 26 (21–31) 16 15 57 (48–70)
2005 < 1 0.027 (0.022–0.033) 25 (20–30) <0.01 (<0.01–0.015) 3.6 (<0.1–13) 7 6.4 26 (21–31)
2010 < 1 0.027 (0.022–0.032) 24 (20–29) <0.01 (<0.01–0.017) 7.3 (2.0–16) 15 14 56 (47–68)
2011 < 1 0.026 (0.022–0.032) 24 (20–29) <0.01 (<0.01–0.016) 7.6 (2.9–14) 17 16 64 (54–78)
2012 < 1 0.026 (0.022–0.031) 24 (20–29) <0.01 (<0.01–0.017) 8.7 (3.9–15) 30 27 110 (96–140)
Sint Maarten 2010 < 1 <0.01 (<0.01–<0.01) 8.1 (7.1–9.2) 3 7.1 87 (77–99)
(Dutch part) 2011 < 1 <0.01 (<0.01–<0.01) 5.3 (4.6–6.0) 2 4.6 87 (77–99)
2012 < 1 <0.01 (<0.01–<0.01) 2.6 (2.3–2.9) 1 2.3 87 (77–99)
Suriname 1990 < 1 0.26 (0.170–0.360) 63 (41–90) 0.023 (0.015–0.033) 5.7 (3.7–8.2) 82 20 32 (22–50)
1995 < 1 0.4 (0.260–0.570) 92 (59–131) 0.12 (0.077–0.17) 28 (18–39)
2000 < 1 0.4 (0.260–0.580) 86 (56–124) 0.16 (0.10–0.22) 34 (22–48) 89 19 22 (15–34)
2005 < 1 0.31 (0.200–0.450) 63 (41–90) 0.1 (0.066–0.15) 21 (13–29) 117 23 37 (26–58)
2010 < 1 0.24 (0.160–0.340) 46 (31–65) 0.06 (0.040–0.085) 12 (7.7–16) 194 37 79 (57–120)
2011 < 1 0.23 (0.170–0.310) 44 (32–58) 0.053 (0.038–0.071) 10 (7.2–13) 125 24 54 (41–74)
2012 < 1 0.22 (0.160–0.290) 41 (30–55) 0.047 (0.033–0.062) 8.7 (6.2–12) 128 24 58 (44–80)
Trinidad and 1990 1 0.14 (0.120–0.160) 11 (9.9–13) <0.01 (<0.01–<0.01) 0.2 (0.16–0.21) 120 9.8 87 (77–99)
Tobago 1995 1 0.19 (0.170–0.220) 15 (13–17) 0.018 (0.016–0.021) 1.5 (1.3–1.7) 166 13 87 (77–99)
2000 1 0.23 (0.200–0.260) 18 (16–20) 0.049 (0.043–0.055) 3.9 (3.4–4.4) 198 16 87 (77–99)
2005 1 0.19 (0.170–0.220) 15 (13–17) 0.048 (0.042–0.054) 3.7 (3.2–4.2) 166 13 87 (77–99)
2010 1 0.25 (0.220–0.290) 19 (17–21) 0.07 (0.061–0.079) 5.2 (4.6–5.9) 219 16 87 (77–99)
2011 1 0.26 (0.230–0.290) 19 (17–22) 0.069 (0.060–0.078) 5.1 (4.5–5.8) 224 17 87 (77–99)
2012 1 0.32 (0.280–0.360) 24 (21–27) 0.083 (0.073–0.094) 6.2 (5.5–7.0) 274 20 87 (77–99)
Turks and Caicos 1990 < 1 0 (0–0) 0 (0–0) 0 0
Islands 1995 < 1 <0.01 (<0.01–<0.01) 36 (32–41)
2000 < 1 0.012 (0.010–0.014) 63 (55–72)
2005 < 1 <0.01 (<0.01–<0.01) 29 (26–33)
2010 < 1 <0.01 (<0.01–<0.01) 22 (20–25) <0.01 (<0.01–<0.01) 4.5 (0.21–15) 6 19 87 (77–99)
2011 < 1 0.01 (<0.01–0.012) 33 (29–37) 9 28 87 (77–99)
2012 < 1 <0.01 (<0.01–0.010) 28 (25–32) 8 25 87 (77–99)
United States 1990 255 30 (26–33) 12 (10–13) 1.6 (1.4–1.8) 0.6 (0.54–0.69) 25 701 10 87 (77–99)
of America 1995 268 26 (23–30) 9.8 (8.5–11) 2.2 (1.9–2.5) 0.8 (0.71–0.91) 22 728 8.5 87 (77–99)
2000 285 19 (16–21) 6.6 (5.8–7.5) 1.2 (1.1–1.4) 0.4 (0.38–0.49) 16 310 5.7 87 (77–99)
2005 298 16 (14–18) 5.4 (4.8–6.1) 1.3 (1.2–1.5) 0.5 (0.39–0.50) 14 080 4.7 87 (77–99)
2010 312 13 (11–15) 4.1 (3.6–4.7) 1.2 (1.0–1.4) 0.4 (0.34–0.43) 11 181 3.6 87 (77–99)
2011 315 12 (11–14) 3.8 (3.4–4.3) 1.1 (1.0–1.3) 0.4 (0.32–0.41) 10 521 3.3 87 (77–99)
2012 318 11 (10–13) 3.6 (3.2–4.1) 1.1 (0.96–1.2) 0.4 (0.30–0.39) 9 945 3.1 87 (77–99)
Urugua
y
1990 3 1 (0.890–1.2) 33 (29–37) 0.013 (0.011–0.014) 0.4 (0.35–0.46) 886 28 87 (77–99)
1995 3 0.72 (0.630–0.810) 22 (20–25) 0.022 (0.019–0.025) 0.7 (0.60–0.78) 625 19 87 (77–99)
2000 3 0.74 (0.650–0.840) 22 (20–25) 0.067 (0.059–0.076) 2 (1.8–2.3) 645 19 87 (77–99)
2005 3 0.72 (0.630–0.810) 22 (19–24) 0.11 (0.093–0.12) 3.2 (2.8–3.6) 622 19 87 (77–99)
2010 3 0.8 (0.700–0.910) 24 (21–27) 0.13 (0.11–0.14) 3.8 (3.3–4.3) 699 21 87 (77–99)
2011 3 0.94 (0.820–1.1) 28 (24–31) 0.15 (0.13–0.17) 4.4 (3.9–5.0) 817 24 87 (77–99)
2012 3 0.93 (0.810–1.1) 27 (24–31) 0.14 (0.12–0.16) 4.2 (3.7–4.8) 808 24 87 (77–99)
US Virgin Islands 1990 < 1 <0.01 (<0.01–<0.01) 4.5 (3.9–5.0) 4 3.9 87 (77–99)
1995 < 1 <0.01 (<0.01–<0.01) 4.3 (3.8–4.9) 4 3.7 87 (77–99)
2000 < 1 <0.01 (<0.01–<0.01) 7.7 (6.8–8.7)
2005 < 1 <0.01 (<0.01–<0.01) 7.7 (6.8–8.7)
2010 < 1 <0.01 (<0.01–<0.01) 7.7 (6.8–8.7)
2011 < 1 <0.01 (<0.01–<0.01) 7.7 (6.8–8.7)
2012 < 1 <0.01 (<0.01–<0.01) 7.7 (6.8–8.7)
Venezuela 1990 20 7 (4.9–9.4) 35 (25–47) 0.24 (0.17–0.33) 1.2 (0.88–1.7) 5 457 28 78 (58–110)
(Bolivarian 1995 22 7.7 (6.3–9.2) 35 (28–42) 0.42 (0.30–0.56) 1.9 (1.4–2.5) 5 578 25 73 (60–89)
Republic of) 2000 24 8.4 (6.8–10) 34 (28–41) 0.59 (0.44–0.78) 2.4 (1.8–3.2) 6 466 26 77 (64–95)
2005 27 9 (7.4–11) 34 (28–41) 0.76 (0.56–0.98) 2.8 (2.1–3.7) 6 847 26 76 (63–93)
2010 29 9.7 (8.0–12) 33 (27–40) 0.89 (0.72–1.1) 3.1 (2.5–3.7) 6 451 22 67 (56–81)
2011 30 9.8 (8.1–12) 33 (27–40) 1.3 (1.0–1.5) 4.3 (3.5–5.2) 6 282 21 64 (54–78)
2012 30 9.9 (8.2–12) 33 (27–39) 1.2 (0.94–1.4) 3.9 (3.1–4.7) 6 495 22 65 (55–79)
a Rates are per 100 000 population.
b NOTIFIED NEW AND RELAPSE includes cases for which the treatment history is unknown.

Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
REGION OF THE AMERICAS
7$%/($&DVHQRWLILFDWLRQV±
NEW CASES
YEAR SMEAR-
POSITIVE
SMEAR-NEGATIVE/
UNKNOWN
EXTRA-
PULMONARY OTHER RELAPSE RE-TREAT EXCL.
RELAPSE
TOTAL
RETREAT
HISTORY
UNKNOWN
NEW AND
RELAPSE
b
% SMEAR-
POS AMONG
NEW PULM
NEW AND RELAPSE
NOTIFICATION RATE
a
1990–2012
Anguilla 1990 0 –
1995 2 0 2 0 0 0 0
2000 –
2005 –
2010 1 0 1 0 0 0 0 0 0 0
2011 0 0 0 0 0 0 0 0 0
• 0 0 • 2012 0 0 0 0 0 0 0 0 0
Antigua and 1990 1 –
Barbuda 1995 0 –
2000 4 3 1 0 0 0 75
2005 6 6 0 0 0 0 0 0 0 100
2010 7 6 0 0 0 0 0 0 1 100
2011 6 6 0 0 0 0 2 2 0 100
• 2 3 • 2012 3 1 1 1 0 0 1 1 50
Argentina 1990 12 309 –
1995 13 450 5 698 4 668 3 067 55
2000 11 767 4 749 4 110 1 773 104 1 724 1 828 54
2005 10 576 4 709 3 357 1 561 0 143 666 809 806 58
2010 7 336 3 973 2 011 854 159 290 426 716 49 66
2011 9 733 5 150 2 705 1 426 138 314 758 1 072 66
• 38 21 • 2012 8 758 4 661 2 341 1 291 143 322 848 1 170 67
Aruba 1990 –
1995 –
2000 –
2005 –
2010 6 4 2 67
2011 8 7 0 1 100
• 0 27 • 2012 28 6 20 1 0 1 1 2 0 23
Bahamas 1990 46 –
1995 57 38 11 8 1 1 78
2000 82 56 23 4 0 0 0 71
2005 48 30 8 7 1 2 2 4 0 79
2010 31 19 3 7 1 1 1 2 0 86
2011 41 23 12 5 0 1 1 2 0 66
• 18 9 • 2012 32 21 11 0 0 0 0 0 0 66
Barbados 1990 5 –
1995 3 3 100
2000 3 3 0 0 0 0 0 100
2005 –
2010 6 6 0 0 0 0 0 0 0 100
2011 0 0 0 0 0 0 0 0 0
• 2 1 • 2012 4 4 0 0 0 0 0 0 0 100
Belize 1990 57 –
1995 95 36 34 1 4 4 51
2000 106 44 55 1 6 0 6 44
2005 102 59 29 3 0 11 4 15 0 67
2010 145 97 47 0 0 1 0 1 0 67
2011 74 64 0 0 0 10 2 12 0 100
• 30 26 • 2012 84 36 36 5 0 7 0 7 0 50
Bermuda 1990 0 –
1995 4 2 2 50
2000 0 0 0 0 0 0 0
2005 –
2010 1 1 0 0 0 0 0 0 0 100
2011 1 0 1 0 0 0 0 0 0 0
• 0 5 • 2012 3 1 1 1 0 0 0 0 0 50
Bolivia 1990 11 166 –
(Plurinational 1995 14 422 7 010 1 408 1 133 63 63 83
State of) 2000 10 127 6 458 1 565 1 288 451 1 630 2 081 80
2005 9 748 6 278 1 250 1 673 547 225 772 83
2010 8 363 5 613 630 1 694 0 408 257 665 18 90
2011 8 521 5 746 643 1 721 411 226 637 90
• 164 79 • 2012 8 257 5 568 571 1 672 0 446 227 673 0 91
Bonaire, Saint 2010 0 0 0 0 0
Eustatius and Saba 2011 1 0 0 0 0 1 0 1 0
2012 0 0 0 0 0 0 0 0 0
Brazil 1990 74 570 –
1995 91 013 45 650 29 291 13 814 61
2000 77 899 41 186 23 622 10 457 2 634 8 700 11 334 64
2005 80 675 42 093 23 990 11 037 3 089 6 548 9 637 466 64
2010 74 395 37 932 23 030 10 017 18 3 398 7 551 10 949 0 62
2011 77 647 40 294 20 961 10 067 15 3 555 6 490 10 045 2 755 66
• 50 38 • 2012 75 122 40 152 20 770 10 297 11 3 867 7 633 11 500 25 66
British Virgin 1990 –
Islands 1995 –
2000 1 1 100
2005 0 0 0 0 0 0 0 0 0
2010 1 1 0 0 0 0 0 0 0 100
2011 0 0 0 0 0 0 0 0 0
• 0 0 • 2012 0 0 0 0 0 0 0 0 0
Canada 1990 1 997 549 516 723 0 180 180 29 52
1995 1 965 436 656 634 0 195 195 44 40
2000 1 723 492 528 482 20 145 145 56 48
2005 1 552 433 446 562 4 39 64 103 68 49
2010 1 361 358 472 444 0 48 24 72 39 43
2011 1 430 407 456 469 0 59 22 81 39 47
• 7 5 • 2012 1 653 478 574 519 0 58 33 91 24 45
Cayman Islands 1990 2 –
1995 2 0 2 1 0 0 0
2000 5 5 0 0 0 0 0 100
2005 –
2010 4 2 2 0 0 0 0 0 0 50
2011 2 1 1 0 0 0 0 0 0 50
• 8 10 • 2012 6 5 1 0 0 0 0 0 0 83
Chile 1990 6 151 –
1995 4 150 1 561 1 284 1 017 225 225 55
2000 3 021 1 290 879 694 158 158 59
2005 2 505 1 186 502 631 186 128 314 70
2010 2 376 1 154 502 553 0 167 96 263 0 70
2011 2 450 1 196 473 594 0 187 85 272 0 72
• 47 14 • 2012 2 394 1 173 538 518 0 165 66 231 0 69
Colombia 1990 12 447 –
1995 9 912 7 530 1 380 1 002 85
2000 11 630 8 358 1 446 1 487 339 339 85
2005 10 360 6 870 1 429 1 618 443 443 83
2010 11 420 7 028 1 696 1 985 311 400 469 869 0 81
2011 11 884 6 807 2 355 2 275 0 447 554 1 001 0 74
• 37 24 • 2012 11 424 6 523 2 279 2 264 0 358 405 763 0 74
a
Rates are per 100 000 population.
b
NEW AND RELAPSE includes cases for w hich the treatment history is unknown.

190 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($&DVHQRWLILFDWLRQV±
NEW CASES
YEAR SMEAR-
POSITIVE
SMEAR-NEGATIVE/
UNKNOWN
EXTRA-
PULMONARY OTHER RELAPSE RE-TREAT EXCL.
RELAPSE
TOTAL
RETREAT
HISTORY
UNKNOWN
NEW AND
RELAPSE
b
% SMEAR-
POS AMONG
NEW PULM
NEW AND RELAPSE
NOTIFICATION RATE
a
1990–2012
Costa Rica 1990 230 –
1995 586 245 71 31 0 0 78
2000 585 349 184 98 35 35 65
2005 534 330 81 104 19 26 45 80
2010 492 267 89 108 1 25 7 32 2 75
2011 514 285 128 85 0 16 10 26 0 69
• 7 10 • 2012 475 257 99 102 0 17 5 22 0 72
Cuba 1990 546 –
1995 1 553 834 520 199 54 54 62
2000 1 183 675 257 201 50 122 172 72
2005 772 467 160 103 40 9 49 2 74
2010 827 462 212 98 10 45 11 56 0 69
2011 805 437 219 86 6 57 16 73 0 67
• 5 7 • 2012 734 374 200 112 2 46 14 60 0 65
Curaçao 2010 5 5 0 0 0 100
2011 1 0 1 0 0 0 0 0 0 0
2012 1 1 0 0 0 0 0 0 0 100
Dominica 1990 6 –
1995 8 5 3 3 100
2000 –
2005 –
2010 8 8 0 0 0 0 0 0 0 100
2011 2 2 0 0 0 0 1 1 0 100
• 8 10 • 2012 7 5 1 0 0 1 1 2 0 83
Dominican 1990 2 597 –
Republic 1995 4 053 2 787 1 418 244 204 204 66
2000 5 291 2 907 1 234 540 610 610 70
2005 5 003 2 949 1 032 602 420 309 729 74
2010 3 964 2 159 803 578 100 324 196 520 0 73
2011 4 309 2 454 809 655 49 342 163 505 0 75
• 36 41 • 2012 4 262 2 483 817 544 44 374 178 552 0 75
Ecuador 1990 8 243 –
1995 7 893 5 890 2 237 420 72
2000 6 908 5 064 1 338 400 106 280 386 79
2005 4 416 3 048 635 330 403 392 795 83
2010 4 832 3 373 404 655 0 400 263 663 0 89
2011 5 106 3 521 380 808 0 397 244 641 0 90
• 81 35 • 2012 5 456 3 856 285 856 111 348 315 663 93
El Salvador 1990 2 367 –
1995 2 422 2 241 181 –
2000 1 485 1 008 278 108 91 180 271 78
2005 1 794 1 059 402 255 78 36 114 0 72
2010 1 700 972 338 328 0 62 30 92 0 74
2011 1 896 1 079 371 384 0 62 21 83 0 74
• 44 33 • 2012 2 053 1 237 313 415 0 88 10 98 0 80
Grenada 1990 0 –
1995 4 2 100
2000 0 0 0 0 0 0 0
2005 –
2010 4 4 0 0 0 0 0 0 0 100
2011 2 1 1 0 0 0 0 0 0 50
• 0 1 • 2012 1 1 0 0 0 0 0 0 0 100
Guatemala 1990 3 813 –
1995 3 119 2 368 546 205 249 249 81
2000 2 913 2 052 518 202 141 141 80
2005 3 803 2 420 588 256 101 58 159 438 80
2010 3 322 2 121 265 348 436 152 29 181 0 89
2011 3 040 1 961 309 243 415 112 48 160 0 86
• 43 23 • 2012 3 442 2 212 382 311 393 144 57 201 85
Guyana 1990 168 –
1995 296 85 187 22 2 2 31
2000 422 119 231 34 38 46 84 34
2005 639 240 352 33 6 8 17 25 0 41
2010 712 325 274 75 0 38 124 162 0 54
2011 710 323 282 78 0 27 206 233 0 53
• 23 94 • 2012 748 309 339 77 0 23 221 244 0 48
Haiti 1990 –
1995 6 212 –
2000 10 420 5 887 2 930 1 367 236 110 346 67
2005 14 311 7 340 5 292 1 484 195 33 228 58
2010 14 222 8 242 4 335 1 307 0 338 43 381 0 66
2011 14 315 8 011 4 553 1 374 0 377 46 423 0 64
• 0 163 • 2012 16 568 9 254 4 956 1 914 0 444 155 599 65
Honduras 1990 3 647 –
1995 4 984 2 306 2 214 232 100 100 51
2000 6 406 3 404 2 396 370 236 236 59
2005 3 333 2 069 721 362 181 181 74
2010 2 876 1 842 482 382 0 170 25 195 0 79
2011 3 233 2 060 616 377 0 180 10 190 77
• 74 38 • 2012 3 014 1 945 509 362 0 198 32 230 0 79
Jamaica 1990 123 –
1995 109 93 14 2 2 2 87
2000 127 90 20 4 13 13 82
2005 90 53 31 6 0 0 5 5 0 63
2010 130 76 46 6 0 2 17 19 0 62
2011 105 35 39 6 24 1 3 4 0 47
• 5 3 • 2012 91 46 33 9 0 3 3 6 0 58
Mexico 1990 14 437 –
1995 11 329 9 220 1 807 302 84
2000 18 434 11 676 1 675 2 081 421 914 1 335 87
2005 18 524 11 997 421 2 657 2 831 618 1 408 2 026 97
2010 20 155 12 572 2 812 3 464 0 722 544 1 266 585 82
2011 19 857 12 960 2 497 3 529 0 871 671 1 542 0 84
• 17 17 • 2012 20 470 13 038 2 681 3 839 139 773 878 1 651 0 83
Montserrat 1990 1 –
1995 –
2000 0 0 0 0 0 0 0
2005 1 1 0 0 0 0 0 0 0 100
2010 0 0 0 0 0 0 0 0
2011 0 0 0 0 0 0 0 0 0
• 9 0 • 2012 0 0 0 0 0 0 0 0 0
Netherlands 1990 –
Antilles 1995 –
2000 5 2 3 0 0 0 0 0 0 40
• 0 0 • 2005 –
a
Rates are per 100 000 population.
b
NEW AND RELAPSE includes cases for which the treatment history is unknown.

191 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
REGION OF THE AMERICAS
7$%/($&DVHQRWLILFDWLRQV±
NEW CASES
YEAR SMEAR-
POSITIVE
SMEAR-NEGATIVE/
UNKNOWN
EXTRA-
PULMONARY OTHER RELAPSE RE-TREAT EXCL.
RELAPSE
TOTAL
RETREAT
HISTORY
UNKNOWN
NEW AND
RELAPSE
b
% SMEAR-
POS AMONG
NEW PULM
NEW AND RELAPSE
NOTIFICATION RATE
a
1990–2012
Nicaragua 1990 2 944 –
1995 2 842 1 568 854 253 167 167 65
2000 2 402 1 471 541 231 159 159 73
2005 1 907 1 253 395 160 99 169 268 0 76
2010 2 448 1 440 575 274 0 159 127 286 0 71
2011 2 693 1 552 653 335 0 153 129 282 0 70
• 71 47 • 2012 2 790 1 484 817 339 0 150 144 294 0 64
Panama 1990 846 –
1995 1 300 1 066 114 28 108 108 90
2000 1 169 460 589 74 5 41 93 134 44
2005 1 637 860 505 216 56 191 247 63
2010 1 496 707 425 287 0 77 134 211 0 62
2011 1 571 830 433 235 0 55 124 179 18 66
• 34 40 • 2012 1 520 778 434 248 0 60 155 215 0 64
Paraguay 1990 2 167 993 100
1995 1 745 748 870 127 28 28 46
2000 1 950 900 791 170 14 516 530 53
2005 2 075 1 260 665 150 273 273 65
2010 2 352 1 318 499 269 86 105 109 214 75 73
2011 2 372 1 371 515 251 108 127 177 304 0 73
• 51 36 • 2012 2 416 1 391 494 221 187 123 207 330 0 74
Peru 1990 37 905 –
1995 45 310 32 096 7 803 5 411 80
2000 38 661 22 580 6 018 5 682 4 381 4 381 79
2005 33 747 18 490 5 592 5 335 809 3 195 1 794 4 989 326 77
2010 31 073 17 264 5 201 5 185 647 2 776 1 404 4 180 77
2011 31 241 17 754 5 164 5 564 712 2 047 1 603 3 650 0 77
• 174 99 • 2012 29 760 17 653 4 556 5 233 583 1 735 1 945 3 680 0 79
Puerto Rico 1990 159 –
1995 262 128 111 23 54
2000 174 81 69 24 54
2005 113 60 37 16 0 0 0 0 0 62
2010 80 37 35 4 0 4 0 4 0 51
2011 50 29 13 8 0 0 0 0 0 69
• 5 2 • 2012 71 41 17 10 0 3 0 3 0 71
Saint Kitts and 1990 0 –
Nevis 1995 5 4 100
2000 0 0 0 0 0 0 0
2005 0 0 0 0 0 0 2 2
2010 2 2 0 0 0 0 0 0 0 100
2011 1 1 0 0 0 0 0 0 0 100
• 0 4 • 2012 2 2 0 0 0 0 0 0 0 100
Saint Lucia 1990 13 –
1995 11 11 100
2000 9 7 1 0 1 2 3 88
2005 14 11 1 0 0 2 0 2 92
2010 9 9 0 0 0 0 0 0 0 100
2011 7 7 0 0 0 0 0 0 0 100
• 9 6 • 2012 11 11 0 0 0 0 0 0 0 100
Saint Vincent and 1990 2 –
the Grenadines 1995 13 5 7 0 4 4 42
2000 16 9 4 0 3 0 3 69
2005 7 6 1 0 0 0 0 0 0 86
2010 15 8 7 0 0 0 2 2 0 53
2011 17 8 9 0 0 0 0 0 0 47
• 2 27 • 2012 30 27 3 0 0 0 4 4 0 90
Sint Maarten 2010 3 3 0 0 0 0 0 0 100
(Dutch part) 2011 2 2 0 0 0 0 0 100
2012 1 0 1 0 0 0 0 0 0 0
Suriname 1990 82 –
1995 –
2000 89 37 40 12 0 1 1 48
2005 117 49 54 6 2 6 2 8 0 48
2010 194 130 42 14 2 6 10 16 0 76
2011 125 64 34 20 1 5 6 11 1 65
• 20 24 • 2012 128 83 28 13 2 2 5 7 0 75
Trinidad and 1990 120 –
Tobago 1995 166 7 68 12 22 22 9
2000 198 115 61 17 5 26 31 65
2005 166 95 50 12 0 9 13 22 66
2010 219 136 58 20 0 5 39 44 0 70
2011 224 121 77 19 0 7 42 49 0 61
• 10 20 • 2012 274 167 81 19 0 7 47 54 0 67
Turks and Caicos 1990 0 –
Islands 1995 –
2000 –
2005 –
2010 6 3 1 1 0 1 1 2 0 75
2011 9 8 1 0 0 0 1 1 0 89
• 0 25 • 2012 8 5 2 1 0 0 0 0 0 71
United States 1990 25 701 –
of America 1995 22 728 8 093 10 795 3 835 5 43
2000 16 310 5 883 7 204 3 211 12 45
2005 14 080 5 111 6 030 2 939 0 46
2010 11 181 3 695 4 990 2 134 362 43
2011 10 521 3 742 4 556 2 189 34 45
• 10 3 • 2012 9 945 3 563 4 261 2 100 21 46
Uruguay 1990 886 –
1995 625 349 178 78 20 20 66
2000 645 348 165 77 39 39 68
2005 622 355 147 73 32 15 4 19 71
2010 699 368 218 72 0 41 0 41 0 63
2011 817 467 249 48 0 53 0 53 0 65
• 28 24 • 2012 808 432 269 59 0 48 7 55 0 62
US Virgin Islands 1990 4 –
1995 4 2 2 0 50
2000 –
2005 –
2010 –
2011 –
• 4 0 • 2012 –
Venezuela 1990 5 457 –
(Bolivarian 1995 5 578 3 056 1 517 709 272 272 67
Republic of) 2000 6 466 3 525 1 616 948 377 377 69
2005 6 847 3 653 1 853 1 094 247 103 350 66
2010 6 451 3 252 1 758 1 077 0 248 194 442 116 65
2011 6 282 3 224 1 649 1 196 0 213 195 408 0 66
• 28 22 • 2012 6 495 3 446 1 617 1 143 0 289 282 571 0 68
a
Rates are per 100 000 population.
b
NEW AND RELAPSE includes cases for which the treatment history is unknown.

192 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($7UHDWPHQWRXWFRPHVQHZVPHDUSRVLWLYHFDVHV±
% OF COHORT
TREATMENT SUCCESS (%)
a
1995–2011 YEAR NUMBER
NOTIFIED
SIZE OF
COHORT
COHORT AS
% NOTIFIED CURED COMPLETED DIED FAILED DEFAULTED NOT
EVALUATED
Anguilla 1995 0 –
2000 –
2005 –
2009 0 –
2010 0 0 –
• 0 0 • 2011 0 0 –
Antigua and 1995 –
Barbuda 2000 3 4 133 100 0 0 0 0 0
2005 6 6 100 50 33 17
2009 1 3 300 67 0 33 0 0 0
2010 6 6 100 0 33 33 0 33 0
• 0 17 • 2011 6 6 100 17 33 50 0
Argentina 1995 5 698 5 707 100 5 7 1 0 3 84
2000 4 749 5 177 109 26 20 5 0 6 43
2005 4 709 4 709 100 19 34 5 0 5 37
2009 4 044 5 062 125 19 26 4 0 7 43
2010 3 973 5 088 128 20 27 4 0 8 40
• 12 52 • 2011 5 150 5 600 109 18 33 5 0 8 36
Aruba 1995 –
2000 –
2005 –
2009 6 –
2010 4 –
• 0 92 • 2011 7 13 186 92 8 0
Bahamas 1995 38 –
2000 56 –
2005 30 30 100 17 40 17 7 20 0
2009 26 26 100 12 69 8 0 12 0
2010 19 19 100 16 53 16 5 11 0
• 0 70 • 2011 23 23 100 4 65 26 0 4 0
Barbados 1995 3 –
2000 3 –
2005 11 – 45 45 9 0
2009 2 2 100 100 0 0 0 0 0
2010 6 6 100 100 0 0 0 0 0
• 0 0 • 2011 0 0 –
Belize 1995 36 29 81 52 0 10 3 28 7
2000 44 45 102 78 0 9 0 2 11
2005 59 59 100 56 19 12 2 12 0
2009 82 –
2010 97 142 146
• 52 0 • 2011 64 –
Bermuda 1995 2 –
2000 0 –
2005 –
2009 1 – 0 0 0 0 0 100
2010 1 1 100 0 0 0 0 0 100
• 0 0 • 2011 0 1 – 0 0 0 0 0 100
Bolivia 1995 7 010 7 010 100 53 9 4 1 9 24
(Plurinational 2000 6 458 6 212 96 73 6 4 1 9 7
State of) 2005 6 278 6 278 100 76 2 3 1 5 12
2009 5 937 5 897 99 84 1 4 1 5 4
2010 5 613 5 571 99 86 2 4 1 5 3
• 62 86 • 2011 5 746 5 770 100 84 2 3 1 5 5
Bonaire, Saint 2009 0 –
Eustatius and Saba 2010 0 0 –
2011 0 0 –
Brazil 1995 45 650 45 650 100 17 0 1 1 3 79
2000 41 186 34 007 83 49 22 4 0 9 16
2005 42 093 42 093 100 31 44 5 1 9 9
2009 39 267 40 818 104 31 41 5 1 10 11
2010 37 932 41 840 110 37 37 5 0 11 10
• 17 76 • 2011 40 294 42 764 106 37 38 5 0 10 9
British Virgin 1995 –
Islands 2000 1 1 100 100 0
2005 0 –
2009 1 – 0 100 0 0 0 0
2010 1 0 0
• 0 0 • 2011 0 0 –
Canada 1995 436 –
2000 492 492 100 22 13 5 1 59
2005 433 459 106 8 59 9 0 1 22
2009 462 850 184 10 65 7 0 0 17
2010 358 854 239 12 65 8 0 0 15
• 0 62 • 2011 407 858 211 8 54 9 0 0 29
Cayman Islands 1995 0 –
2000 5 5 100 0 40 0 0 0 60
2005 1 – 0 0 0 0 100 0
2009 1 2 200 50 0 0 0 0 50
2010 2 2 100 50 0 0 0 0 50
• 0 100 • 2011 1 1 100 100 0 0 0 0 0
Chile 1995 1 561 1 111 71 79 7 0 8 5
2000 1 290 1 360 105 82 9 0 6 2
2005 1 186 1 147 97 83 0 9 0 6 2
2009 1 152 1 365 118 61 11 9 0 7 12
2010 1 154 1 437 125 51 20 9 0 6 14
• 79 71 • 2011 1 196 1 462 122 50 22 7 0 6 15
Colombia 1995 7 530 –
2000 8 358 1 634 20 70 10 5 1 8 6
2005 6 870 7 778 113 63 9 6 1 7 14
2009 7 319 6 899 94 68 9 6 2 9 6
2010 7 028 7 364 105 69 10 7 1 9 4
• 0 77 • 2011 6 807 6 805 100 66 11 7 1 10 5
Costa Rica 1995 245 –
2000 349 349 100 43 14 10 1 12 19
2005 330 306 93 85 4 5 2 3 1
2009 271 166 61 49 4 5 1 1 39
2010 267 297 111 75 12 7 2 2 2
• 0 88 • 2011 285 282 99 85 3 7 1 2 2
Cuba 1995 834 834 100 90 0 4 3 2 2
2000 675 673 100 91 2 4 1 1 1
2005 467 466 100 90 2 6 1 1 1
2009 418 415 99 87 3 7 2 1 0
2010 462 458 99 89 1 7 1 2 0
• 90 88 • 2011 437 443 101 83 5 8 2 3 0
a
TREATMENT SUCCESS = percent cured + percent completed then rounded to the nearest digit.

193 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
REGION OF THE AMERICAS
7$%/($7UHDWPHQWRXWFRPHVQHZVPHDUSRVLWLYHFDVHV±
% OF COHORT
TREATMENT SUCCESS (%)
a
1995–2011 YEAR NUMBER
NOTIFIED
SIZE OF
COHORT
COHORT AS
% NOTIFIED CURED COMPLETED DIED FAILED DEFAULTED NOT
EVALUATED
Curaçao 2009 5 –
2010 5 –
2011 0 1 –
Dominica 1995 5 –
2000 –
2005 –
2009 4 4 100 100 0 0 0 0 0
2010 8 3 38 67 33 0 0 0 0
• 0 100 • 2011 2 2 100 100 0 0 0 0 0
Dominican 1995 2 787 2 007 72 43 21 5 2 13 16
Republic 2000 2 907 2 760 95 37 34 5 2 19 4
2005 2 949 2 697 91 80 5 4 2 7 3
2009 2 441 2 441 100 79 6 4 2 7 2
2010 2 159 2 194 102 73 7 5 1 7 6
• 64 83 • 2011 2 454 2 454 100 76 7 4 2 8 3
Ecuador 1995 5 890 5 236 89 39 2 8 14 37
2000 5 064 –
2005 3 048 2 150 71 81 3 3 3 6 5
2009 3 317 3 330 100 71 4 4 3 8 11
2010 3 373 3 373 100 75 4 3 3 7 8
• 39 78 • 2011 3 521 3 441 98 73 4 3 3 7 10
El Salvador 1995 –
2000 1 008 1 008 100 78 1 7 1 5 8
2005 1 059 1 059 100 91 0 4 1 2 1
2009 930 930 100 88 1 5 4 2 0
2010 972 972 100 91 1 4 2 2 0
• 0 93 • 2011 1 079 1 079 100 93 0 4 1 2 0
Grenada 1995 2 –
2000 0 –
2005 6 – 67 33 0
2009 4 4 100 50 50 0
2010 4 4 100 75 0 25 0 0 0
• 0 100 • 2011 1 1 100 100 0
Guatemala 1995 2 368 2 368 100 56 5 3 1 4 31
2000 2 052 1 908 93 75 11 5 1 7 1
2005 2 420 –
2009 1 609 2 121 132 77 6 6 1 9 1
2010 2 121 2 121 100 77 6 6 1 9 1
• 61 86 • 2011 1 961 2 056 105 81 5 5 1 8 0
Guyana 1995 85 296 348 10 34 11 1 38 6
2000 119 119 100 43 13 12 5 24 3
2005 240 257 107 2 57 7 26 9
2009 328 328 100 13 57 8 1 19 2
2010 325 325 100 30 41 6 1 18 4
• 44 72 • 2011 323 323 100 22 50 7 1 16 4
Haiti 1995 3 081 – 70 4 1 21 3
2000 5 887 5 887 100 57 14 5 1 13 10
2005 7 340 7 340 100 72 8 6 1 7 6
2009 8 435 – 67 12 5 1 8 7
2010 8 242 8 242 100 72 10 5 1 7 5
• 70 84 • 2011 8 011 8 390 105 74 10 4 1 6 5
Honduras 1995 2 306 2 226 97 39 25 7 0 4 25
2000 3 404 2 362 69 81 5 6 1 5 3
2005 2 069 1 905 92 81 7 5 0 4 3
2009 1 881 1 881 100 79 6 6 1 6 2
2010 1 842 1 918 104 79 6 6 1 6 2
• 64 88 • 2011 2 060 2 004 97 82 6 5 0 5 2
Jamaica 1995 93 93 100 2 65 10 1 17 5
2000 90 99 110 5 40 23 0 11 20
2005 53 53 100 4 53 13 0 26 4
2009 77 76 99 55 14 14 0 11 5
2010 76 76 100 13 34 9 0 5 38
• 67 47 • 2011 35 59 169 25 22 7 0 5 41
Mexico 1995 9 220 9 220 100 69 6 4 3 12 6
2000 11 676 11 538 99 64 12 6 1 9 8
2005 11 997 12 172 101 71 6 5 1 6 11
2009 11 862 11 821 100 82 4 6 1 5 2
2010 12 572 12 304 98 82 4 6 1 5 1
• 75 86 • 2011 12 960 12 622 97 72 15 5 1 4 2
Montserrat 1995 –
2000 0 –
2005 1 –
2009 0 –
2010 0 0 –
• 0 0 • 2011 0 –
Netherlands 1995 –
Antilles 2000 2 5 250 20 80
• 0 2005 –
Nicaragua 1995 1 568 1 536 98 66 14 4 2 10 4
2000 1 471 1 437 98 70 13 5 1 9 2
2005 1 253 1 496 119 73 12 5 2 6 3
2009 1 329 1 552 117 69 16 4 1 7 3
2010 1 440 1 704 118 66 18 5 2 6 3
• 80 86 • 2011 1 552 1 565 101 68 18 3 2 7 3
Panama 1995 1 066 1 388 130 10 60 14 1 13 3
2000 460 460 100 27 33 7 2 22 10
2005 860 873 102 68 12 8 0 10 1
2009 755 768 102 65 16 7 1 12 0
2010 707 717 101 64 16 7 1 12 0
• 69 84 • 2011 830 861 104 68 16 5 1 10 0
Paraguay 1995 748 748 100 8 43 3 0 17 29
2000 900 900 100 21 45 5 0 22 7
2005 1 260 1 452 115 46 33 5 8 7
2009 1 498 1 467 98 75 5 7 0 5 7
2010 1 318 1 317 100 69 9 8 0 5 8
• 51 78 • 2011 1 371 1 367 100 70 8 7 1 5 9
Peru 1995 32 096 28 185 88 75 9 3 2 6 6
2000 22 580 22 230 98 90 0 2 2 3 4
2005 18 490 14 793 80 91 2 2 4 1
2009 17 391 14 212 82 70 11 3 1 6 9
2010 17 264 17 264 100 57 12 2 5 5 20
• 83 74 • 2011 17 754 16 694 94 68 6 3 7 5 11
Puerto Rico 1995 128 128 100 68 23 8 2
2000 81 81 100 64 31 5 0
2005 60 60 100 75 0 22 0 3 0
2009 30 37 123 81 0 16 0 0 3
2010 37 37 100 78 0 14 3 5 0
• 68 73 • 2011 29 40 138 0 72 25 2 0 0
a
TREATMENT SUCCESS = percent cured + percent completed then rounded to the nearest digit.

194 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($7UHDWPHQWRXWFRPHVQHZVPHDUSRVLWLYHFDVHV±
% OF COHORT
TREATMENT SUCCESS (%)
a
1995–2011 YEAR NUMBER
NOTIFIED
SIZE OF
COHORT
COHORT AS
% NOTIFIED CURED COMPLETED DIED FAILED DEFAULTED NOT
EVALUATED
Saint Kitts and 1995 4 5 125 20 40 20 0 20 0
Nevis 2000 0 –
2005 0 –
2009 4 5 125 80 0 0 0 0 20
2010 2 2 100 100 0 0000
• 60 100 • 2011 1 1 100 100 0 0000
Saint Lucia 1995 11 –
2000 7 8 114 88 12 0000
2005 11 13 118 15 54 31 0 0 0
2009 7 7 100 57 29 0 14 0
2010 9 9 100 22 67 0 0 0 11
• 0 57 • 2011 7 7 100 43 14 14 29 0 0
Saint Vincent and 1995 5 –
the Grenadines 2000 9 13 144 100 0 0 0 0 0
2005 6 –
2009 3 1 33 0 0 0 0 0 100
2010 8 8 100 0 0 0 0 0 100
• 0 56 • 2011 8 9 112 44 11 11 0 33 0
Sint Maarten 2009 –
(Dutch part) 2010 3 3 100 100 0000
2011 2 2 100 100 0 0000
Suriname 1995 51 – 10 4 12 8 67
2000 37 37 100 49 19 16 0 14 3
2005 49 –
2009 149 143 96 64 3 11 1 16 5
2010 130 73 56 60 0 12 0 4 23
• 14 76 • 2011 64 75 117 71 5 13 0 4 7
Trinidad and 1995 7 78 1 114 49 21 19 1 10 0
Tobago 2000 115 194 169 22 46 11 2 6 13
2005 95 106 112 68 4 12 16 0
2009 154 154 100 61 8 14 1 14 1
2010 136 136 100 72 4 9 3 11 1
• 69 72 • 2011 121 123 102 69 3 11 1 15 1
Turks and Caicos 1995 –
Islands 2000 2 – 0 0 0 0 100 0
2005 3 – 33 33 0 0 0 33
2009 –
2010 3 4 133 75 25
• 0 22 • 2011 8 9 112 22 67 11
United States 1995 8 093 8 116 100 76 15 4 6
of America 2000 5 883 5 901 100 83 11 3 3
2005 5 111 5 136 100 84 8 2 6
2009 4 014 7 460 186 60 6 1 32
2010 3 695 7 034 190 64 6 1 29
• 76 78 • 2011 3 742 5 955 159 78 6 1 15
Uruguay 1995 349 370 106 41 27 10 1 4 17
2000 348 344 99 85 0 13 1 1 0
2005 355 345 97 80 4 11 0 4 1
2009 409 406 99 73 7 12 0 6 2
2010 368 368 100 80 5 10 0 5 0
• 68 85 • 2011 467 467 100 8148070
US Virgin Islands 1995 2 2 100 50 0 0 0 50
2000 –
2005 –
2009 –
2010 –
• 50 0 • 2011 –
Venezuela 1995 3 056 3 056 100 68641813
(Bolivarian 2000 3 525 3 390 96 76040136
Republic of) 2005 3 653 3 581 98 83 5 0 10 2
2009 3 436 3 433 100 84040111
2010 3 252 3 157 97 83050110
• 74 80 • 2011 3 224 3 224 100 80050123
a TREATMENT SUCCESS = percent cured + percent completed then rounded to the nearest digit.

195 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
REGION OF THE AMERICAS
7$%/($7UHDWPHQWRXWFRPHVUHWUHDWPHQWFDVHV±
% OF COHORT
TREATMENT SUCCESS (%)
a
1995–2011 YEAR NUMBER
NOTIFIED
SIZE OF
COHORT
COHORT AS
% NOTIFIED CURED COMPLETED DIED FAILED DEFAULTED NOT
EVALUATED
Anguilla 1995 0 –
2000 –
2005 –
2009 0 –
2010 0 0 –
• 0 0 • 2011 0 0 –
Antigua and 1995 –
Barbuda 2000 0 –
2005 0 –
2009 2 1 50 100 0 0 0 0 0
2010 0 0 –
• 0 50 • 2011 2 2 100 50 0 50 0 0 0
Argentina 1995 –
2000 1 828 –
2005 809 1 615 200 7 26 5 0 9 53
2009 827 893 108 10 20 4 1 13 52
2010 716 1 114 156 9 23 4 1 15 49
• 0 41 • 2011 1 072 1 492 139 9 33 5 0 16 38
Aruba 1995 –
2000 –
2005 –
2009 –
2010 –
• 0 0 • 2011 –
Bahamas 1995 1 –
2000 0 –
2005 4 4 100 25 50 0 0 25 0
2009 5 5 100 20 60 20 0 0 0
2010 2 2 100 0 100 0 0 0 0
• 0 100 • 2011 2 2 100 0 100 0 0 0 0
Barbados 1995 –
2000 0 –
2005 –
2009 0 0 –
2010 0 0 –
• 0 0 • 2011 0 0 –
Belize 1995 4 13 325 23 0 23 8 38 8
2000 6 –
2005 15 14 93 57 29 14 0 0 0
2009 12 –
2010 1 1 100
• 23 0 • 2011 12 –
Bermuda 1995 –
2000 0 –
2005 –
2009 0 –
2010 0 0 –
• 0 0 • 2011 0 0 –
Bolivia 1995 63 462 733 57 9 7 5 15 7
(Plurinational 2000 2 081 804 39 49 11 12 2 8 16
State of) 2005 772 772 100 63 3 5 3 7 19
2009 732 598 82 73 5 7 2 7 7
2010 665 589 89 72 5 5 3 10 5
• 66 73 • 2011 637 560 88 71 2 6 3 10 7
Bonaire, Saint 2009 0 –
Eustatius and Saba 2010 0 –
2011 1 1 100
Brazil 1995 –
2000 11 334 7 859 69 30 10 4 0 14 41
2005 9 637 9 479 98 26 22 7 2 19 25
2009 9 818 10 664 109 15 28 8 2 23 24
2010 10 949 10 721 98 18 28 8 2 25 19
• 0 49 • 2011 10 045 12 083 120 19 30 9 2 23 17
British Virgin 1995 –
Islands 2000 –
2005 0 –
2009 0 –
2010 0 0 –
• 0 0 • 2011 0 0 –
Canada 1995 195 –
2000 145 145 100 16 16 6 1 2 60
2005 103 106 103 8 59 7 0 3 23
2009 94 95 101 4 60 7 0 1 27
2010 72 94 131 15 56 9 0 0 20
• 0 56 • 2011 81 101 125 8 49 9 0 0 35
Cayman Islands 1995 0 –
2000 0 –
2005 0 –
2009 0 0 –
2010 0 0 –
• 0 0 • 2011 0 0 –
Chile 1995 225 –
2000 158 150 95 32 26 8 1 18 15
2005 314 140 45 69 3 14 1 9 3
2009 306 219 72 15 9 7 2 7 60
2010 263 336 128 14 12 6 2 9 58
• 0 43 • 2011 272 281 103 24 19 7 1 15 33
Colombia 1995 –
2000 339 –
2005 443 0 0
2009 616 –
2010 869 920 106 11 5 3 1 7 73
• 0 45 • 2011 1 001 1 001 100 32 13 7 4 23 22
Costa Rica 1995 0 –
2000 35 69 197 23 9 10 3 25 30
2005 45 49 109 55 12 4 2 24 2
2009 31 2 6 0 0 50 0 0 50
2010 32 35 109 37 43 11 0 9 0
• 0 81 • 2011 26 26 100 54 27 12 4 4 0
Cuba 1995 54 55 102 82 0 7 5 5 0
2000 172 58 34 78 7 10 3 2 0
2005 49 48 98 67 6 4 2 21
2009 51 61 120 69 5 15 5 7 0
2010 56 55 98 67 15 4 4 11 0
• 82 68 • 2011 73 72 99 53 15 19 3 10 0
a
TREATMENT SUCCESS = percent cured + percent completed then rounded to the nearest digit.

196 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($7UHDWPHQWRXWFRPHVUHWUHDWPHQWFDVHV±
% OF COHORT
TREATMENT SUCCESS (%)
a
1995–2011 YEAR NUMBER
NOTIFIED
SIZE OF
COHORT
COHORT AS
% NOTIFIED CURED COMPLETED DIED FAILED DEFAULTED NOT
EVALUATED
Curaçao 2009 –
2010 –
2011 0 0 –
Dominica 1995 3 –
2000 –
2005 –
2009 1 0 0
2010 0 1 – 0 100 0 0 0 0
• 0 0 • 2011 1 1 100 0 0 0 100 0 0
Dominican 1995 204 –
Republic 2000 610 498 82 29 26 3 4 27 11
2005 729 530 73 56 5 7 8 19 6
2009 452 434 96 47 6 13 5 29 0
2010 520 384 74 51 13 9 5 18 4
• 0 61 • 2011 505 415 82 46 15 7 5 20 6
Ecuador 1995 –
2000 386 –
2005 795 554 70 56 8 5 10 12 9
2009 756 756 100 46 9 6 8 16 16
2010 663 –
• 0 35 • 2011 641 641 100 29 6 3 5 10 46
El Salvador 1995 –
2000 271 181 67 63 3 9 3 18 3
2005 114 114 100 68 0 6 4 13 8
2009 113 113 100 85 3 3 1 8 1
2010 92 92 100 88 2 2 3 1 3
• 0 90 • 2011 83 83 100 90 0 2 0 5 2
Grenada 1995 –
2000 0 –
2005 –
2009 0 0 –
2010 0 0 –
• 0 0 • 2011 0 0 –
Guatemala 1995 249 254 102 59 15 4 2 4 17
2000 141 164 116 63 16 4 4 10 2
2005 159 –
2009 128 181 141 55 8 5 7 20 4
2010 181 181 100 55 8 5 7 20 4
• 73 64 • 2011 160 182 114 51 14 11 4 20 1
Guyana 1995 2 –
2000 84 38 45 24 29 13 5 26 3
2005 25 23 92 22 35 9 9 13 13
2009 205 205 100 0 51 14 0 18 17
2010 162 162 100 0 52 14 1 28 5
• 0 49 • 2011 233 233 100 6 43 9 1 33 8
Haiti 1995 –
2000 346 55 16 42 15 5 7 22 9
2005 228 228 100 63 7 3 0 13 14
2009 381 – 49 20 7 3 10 11
2010 381 381 100 60 14 5 2 10 8
• 0 72 • 2011 423 453 107 61 11 4 6 10 9
Honduras 1995 100 –
2000 236 180 76 44 10 8 2 6 29
2005 181 169 93 59 9 6 2 17 7
2009 225 192 85 50 7 10 1 10 22
2010 195 164 84 66 9 7 2 15 1
• 0 68 • 2011 190 165 87 64 5 9 2 16 4
Jamaica 1995 2 6 300 0 67 17 0 17 0
2000 13 –
2005 5 5 100 20 80 0
2009 20 19 95 16 58 5 0 21 0
2010 19 19 100 5 26 26 0 21 21
• 67 25 • 2011 4 4 100 0 25 0 0 25 50
Mexico 1995 –
2000 1 335 138 10 33 4 8 7 12 36
2005 2 026 1 456 72 48 7 7 4 14 20
2009 1 535 1 229 80 56 5 9 6 10 14
2010 1 266 1 272 100 55 7 9 6 11 10
• 0 61 • 2011 1 542 1 352 88 47 14 10 5 10 14
Montserrat 1995 –
2000 0 –
2005 0 –
2009 0 –
2010 0 0 –
• 0 0 • 2011 0 –
Netherlands 1995 –
Antilles 2000 0 –
• 0 2005 –
Nicaragua 1995 167 289 173 69 10 4 3 11 3
2000 159 230 145 65 10 6 2 15 2
2005 268 181 68 71 12 7 2 7 2
2009 282 178 63 70 6 3 6 11 3
2010 286 204 71 60 16 8 4 9 2
• 78 69 • 2011 282 134 48 58 10 10 2 14 4
Panama 1995 108 –
2000 134 42 31 19 24 2 0 48 7
2005 247 237 96 23 35 9 4 22 7
2009 235 203 86 18 30 10 0 37 4
2010 211 208 99 23 34 11 3 30 0
• 0 59 • 2011 179 203 113 24 34 11 2 28 0
Paraguay 1995 28 –
2000 530 144 27 19 40 6 1 25 9
2005 273 164 60 44 26 4 10 16
2009 177 188 106 47 9 9 4 11 20
2010 214 216 101 54 6 8 2 9 20
• 0 67 • 2011 304 228 75 60 7 4 4 9 16
Peru 1995 –
2000 4 381 4 521 103 78 0 4 7 6 4
2005 4 989 2 299 46 78 5 5 11 1
2009 4 324 2 163 50 49 21 4 2 12 12
2010 4 180 –
• 0 0 • 2011 3 650 –
Puerto Rico 1995 –
2000 –
2005 0 113 – 73 23 0 4 1
2009 0 0 –
2010 4 4 100 50 0 25 25 0 0
• 0 0 • 2011 0 0 –
a
TREATMENT SUCCESS = percent cured + percent completed then rounded to the nearest digit.

197 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
REGION OF THE AMERICAS
7$%/($7UHDWPHQWRXWFRPHVUHWUHDWPHQWFDVHV±
% OF COHORT
TREATMENT SUCCESS (%)
a
1995–2011 YEAR NUMBER
NOTIFIED
SIZE OF
COHORT
COHORT AS
% NOTIFIED CURED COMPLETED DIED FAILED DEFAULTED NOT
EVALUATED
Saint Kitts and 1995 –
Nevis 2000 0 –
2005 2 2 100 50 50
2009 0 0 –
2010 0 0 –
• 0 0 • 2011 0 0 –
Saint Lucia 1995 –
2000 3 1 33 100 0 0 0 0 0
2005 2 –
2009 3 3 100 33 67 0 0 0
2010 0 0 –
• 0 0 • 2011 0 0 –
Saint Vincent and 1995 4 –
the Grenadines 2000 3 3 100 100 00000
2005 0 –
2009 2 1 50 00001000
2010 2 0 0
• 0 0 • 2011 0 0 –
Sint Maarten 2009 –
(Dutch part) 2010 0 –
2011 0 0 –
Suriname 1995 –
2000 1 –
2005 8 –
2009 15 12 80 50080420
2010 16 11 69 45 9 27 0 0 18
• 0 64 • 2011 11 11 100 45 18 36 0 0 0
Trinidad and 1995 22 –
Tobago 2000 31 22 71 23 45 14 9 9 0
2005 22 21 95 19 38 29 14 0
2009 60 60 100 48 20 15 0 17 0
2010 44 44 100 43 20 14 0 23 0
• 0 35 • 2011 49 49 100 22 12 12 0 51 2
Turks and Caicos 1995 –
Islands 2000 –
2005 3 – 33 33 33 0 0 0
2009 –
2010 2 0 0
• 0 0 • 2011 1 0 0
United States 1995 –
of America 2000 –
2005 –
2009 –
2010 –
• 0 0 • 2011 –
Uruguay 1995 20 25 125 56 20 16 0 8 0
2000 39 –
2005 19 30 158 57 17 13 3 7 3
2009 37 41 111 46 10 34 0 7 2
2010 41 41 100 56 20 15 0 5 5
• 76 79 • 2011 53 53 100 74 6 11 0 9 0
US Virgin Islands 1995 –
2000 –
2005 –
2009 –
2010 –
• 0 0 • 2011 –
Venezuela 1995 272 –
(Bolivarian 2000 377 –
Republic of) 2005 350 247 71 80 4 2 12 2
2009 428 261 61 80042132
2010 442 248 56 83061100
• 0 80 • 2011 408 400 98 80090100
a TREATMENT SUCCESS = percent cured + percent completed then rounded to the nearest digit.
GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($+,9WHVWLQJDQGSURYLVLRQRI&37$57DQG,37–
% OF TB PATIENTS WITH
KNOWN HIV STATUS
2005–2012
YEAR
% OF TB
PATIENTS WITH
KNOWN HIV
STATUS
NUMBER OF TB
PATIENTS WITH
KNOWN HIV
STATUS
PATIENTS
NOTIFIED
(NEW AND
RETREAT)
NUMBER OF
HIV-POSITIVE
TB PATIENTS
% OF TESTED
TB PATIENTS
HIV-POSITIVE
% OF HIV-
POSITIVE TB
PATIENTS ON
CPT
% OF HIV-
POSITIVE TB
PATIENTS ON
ART
NUMBER OF
HIV-POSITIVE
PEOPLE
PROVIDED IPT
Anguilla 2005
2010 0 0 1 0
2011 0 0 0
– – 2012 0 0 0
Antigua and 2005 100 6 6 3 50 100 100 0
Barbuda 2010 86 6 7 5 83 40 100 0
2011 75 6 8 4 67 50 100 5
• 100 100 • 2012 100 4 4 2 50 50 100 1
Argentina 2005 11 242
2010 14 1 121 7 762 672 60
2011 13 1 313 10 491 735 56
– 15 • 2012 15 1 434 9 606 685 48
Aruba 2005
2010 6
2011 8
– 3 • 2012 3.4 1 29 1 100 100
Bahamas 2005 50
2010 100 33 32 16 48 31 75
2011 100 42 42 12 29 42 67
– 100 • 2012 100 32 32 8 25 38 62
Barbados 2005 8 2 25 0
2010 100 6 6 2 33 0 100
2011 0 0 0
– 100 • 2012 100 4 4 1 25 0 100
Belize 2005 100 106 106 25 24 68 68 409
2010 98 142 145 29 20 100 100
2011 84 64 76 24 38 100
• 100 81 • 2012 81 68 84 19 28 100
Bermuda 2005 1 0 0
2010 100 1 1 0 0
2011 100 1 1 0 0
– 100 • 2012 100 3 3 0 0
Bolivia 2005 0 0 9 973 0 50
(Plurinational 2010 22 1 897 8 620 130 6.9 0 87
State of) 2011 45 3 928 8 747 333 8.5 36
• 0 60 • 2012 60 5 049 8 484 164 3.2 100
Bonaire, Saint 2010 0 0 0
Eustatius and Saba 2011 0 0 1 0
2012 0 0 0
Brazil 2005 59 51 552 87 223 8 249 16 0 85 674
2010 63 51 764 81 946 9 338 18 92
2011 64 53 455 84 137 9 088 17 0 100
• 59 55 • 2012 55 45 733 82 755 9 049 20 0 100
British Virgin 2005 0 0 0 27
Islands 2010 0 0 1 0
2011 0 0 0
– – 2012 0 0 0
Canada 2005 26 414 1 616 63 15
2010 48 658 1 385 53 8.1
2011 35 513 1 452 61 12
• 26 42 • 2012 42 716 1 686 57 8
Cayman Islands 2005 1 0 0
2010 75 3 4 0 0
2011 100 2 2 0 0
– 100 • 2012 100 6 6 0 0
Chile 2005 2 633
2010 2 472
2011 11 286 2 535 148 52
– 16 • 2012 16 392 2 460 140 36
Colombia 2005 53 5 537 10 360 353 6.4
2010 43 5 079 11 889 1 231 24 35
2011 53 6 579 12 438 1 292 20 36
• 53 66 • 2012 66 7 791 11 829 1 400 18 34
Costa Rica 2005 67 374 560 50 13 84
2010 99 494 499 54 11 0 0
2011 96 505 524 36 7.1 0 0
• 67 94 • 2012 94 453 480 49 11 0
Cuba 2005 93 729 781 0 0
2010 100 862 838 56 6.5 0 62 1 366
2011 95 780 821 62 7.9 34 89 1 429
• 93 83 • 2012 83 618 748 54 8.7 81 94 1 339
Curaçao 2010 0 0 5 0
2011 0 0 1 0
2012 100 1 1 1 100
Dominica 2005 0
2010 38 3 8 1 33 100 100
2011 67 2 3 0 0
– 75 • 2012 75 6 8 0 0
Dominican 2005 1.5 78 5 312 3 3.8 0 0 953
Republic 2010 60 2 489 4 160 547 22 7.9 3.8 5 041
2011 57 2 540 4 472 460 18 58 93
• 1 61 • 2012 61 2 721 4 440 557 20 69 48
Ecuador 2005 0.21 10 4 808 3 30 0
2010 100 5 183 5 095 427 8.2 100
2011 68 3 640 5 350 576 16
• 0 86 • 2012 86 4 974 5 771 669 13
El Salvador 2005 84 1 544 1 830 188 12 20 38
2010 96 1 667 1 730 180 11 82 63 455
2011 98 1 878 1 917 194 10 85 77
• 84 99 • 2012 99 2 036 2 063 214 11 66 83
Grenada 2005 0 0
2010 100 4 4 1 25 0 0 0
2011 100 2 2 0 0
– 100 • 2012 100 1 1 0 0 0
Guatemala 2005 16 600 3 861 478 80 240
2010 63 2 121 3 351 255 12 100 100
2011 72 2 223 3 088 285 13 16 30
• 16 85 • 2012 85 2 982 3 499 293 9.8 0 95
Guyana 2005 70 456 656 80 18
2010 88 734 836 209 28 77 59 144
2011 93 852 916 199 23 94 83 119
• 70 94 • 2012 94 914 969 284 31 71 59 154
Haiti 2005 0 0 14 344 1 797
2010 67 9 518 14 265 1 892 20 13 9.8 4 112
2011 78 11 213 14 361 2 320 21 11 6.9
• 0 81 • 2012 81 13 518 16 723 2 705 20 59 46 15 283

199 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
REGION OF THE AMERICAS
7$%/($+,9WHVWLQJDQGSURYLVLRQRI&37$57DQG,37–
% OF TB PATIENTS WITH
KNOWN HIV STATUS
2005–2012
YEAR
% OF TB
PATIENTS WITH
KNOWN HIV
STATUS
NUMBER OF TB
PATIENTS WITH
KNOWN HIV
STATUS
PATIENTS
NOTIFIED
(NEW AND
RETREAT)
NUMBER OF
HIV-POSITIVE
TB PATIENTS
% OF TESTED
TB PATIENTS
HIV-POSITIVE
% OF HIV-
POSITIVE TB
PATIENTS ON
CPT
% OF HIV-
POSITIVE TB
PATIENTS ON
ART
NUMBER OF
HIV-POSITIVE
PEOPLE
PROVIDED IPT
Honduras 2005 44 1 455 3 333 200 14 0 0 0
2010 54 1 557 2 901 201 13 90 90 27
2011 75 2 443 3 243 261 11 50 72
• 44 76 • 2012 76 2 312 3 046 259 11 52 74 286
Jamaica 2005 83 79 95 28 35 43 54
2010 87 128 147 30 23 100
2011 85 92 108 17 18 82
• 83 69 • 2012 69 65 94 13 20
Mexico 2005 6.9 1 382 19 932 217 16
2010 43 8 915 20 699 1 645 18 100 26
2011 56 11 416 20 528 1 520 13 70 25
• 7 70 • 2012 70 15 005 21 348 1 233 8.2 70 24
Montserrat 2005 100 1 1 0 0
2010 0 0 0
2011 0 0 0
• 100 – 2012 0
Netherlands Antilles 2005 2 2 100
Nicaragua 2005 0 0 2 076 30 0
2010 56 1 440 2 575 60 4.2 67 67 465
2011 55 1 552 2 822 60 3.9 67 67 152
• 0 72 • 2012 72 2 117 2 934 105 5 78 74 230
Panama 2005 86 1 569 1 828 200 13 10 400
2010 96 1 558 1 630 240 15 63 84
2011 95 1 608 1 695 241 15 94 93
• 86 96 • 2012 96 1 600 1 675 224 14 89 65
Paraguay 2005 2 348
2010 33 817 2 461 144 18 0 67
2011 60 1 533 2 549 174 11 25 56
– 73 • 2012 73 1 906 2 623 154 8.1 60 79
Peru 2005 1.9 668 35 541 668 100 1 214
2010 29 9 539 32 477 853 8.9 1.2 1 183
2011 21 7 052 32 844 960 14 68
• 2 18 • 2012 18 5 836 31 705 979 17 87 1 416
Puerto Rico 2005 82 93 113 28 30
2010 95 76 80 14 18 43 50
2011 92 46 50 10 22 50 50
• 82 86 • 2012 86 61 71 11 18 82 36
Saint Kitts and 2005 2
Nevis 2010 100 2 2 0 0
2011 100 1 1 0 0
– 100 • 2012 100 2 2 0 0
Saint Lucia 2005 7.1 1 14 0 0
2010 100 9 9 0 0
2011 100 7 7 1 14 100 100
• 7 100 • 2012 100 11 11 1 9.1 100 100 1
Saint Vincent and 2005 100 7 7 1 14 0 0
the Grenadines 2010 59 10 17 3 30 100
2011 76 13 17 5 38 80 80
• 100 91 • 2012 91 31 34 9 29 67 67
Sint Maarten 2010 100 3 3 0 0
(Dutch part) 2011 100 2 2 0 0
2012 100 1 1 0 0
Suriname 2005 73 87 119 20 23 10
2010 85 173 204 58 34 10 38
2011 89 117 131 38 32 18 55
• 73 91 • 2012 91 121 133 36 30 69
Trinidad and 2005 69 124 179 42 34 29 36 0
Tobago 2010 98 254 258 58 23 19 34 11
2011 95 252 266 84 33 24 19
• 69 97 • 2012 97 311 321 82 26 28 29
Turks and Caicos 2005 5 1 20 0 0
Islands 2010 71 5 7 1 20 100 100
2011 10 1 10 0 0
– 0 • 2012 0 0 8 0
United States 2005 59 8 273 14 080 1 035 13
of America 2010 66 7 404 11 181 627 8.5
2011 83 8 752 10 521 668 7.6
• 59 84 • 2012 84 8 376 9 945 625 7.5
Uruguay 2005 92 574 626 74 13 0
2010 92 646 699 104 16 0 34
2011 94 769 817 110 14 0 31
• 92 95 • 2012 95 775 815 134 17 0 24
US Virgin Islands 2005
2010
2011
– – 2012
Venezuela 2005 39 2 678 6 950 392 15 0 39
(Bolivarian 2010 78 5 213 6 645 479 9.2 33 102
Republic of) 2011 64 4 133 6 477 519 13 32
• 39 73 • 2012 73 4 956 6 777 581 12 89

200 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($7HVWLQJIRU0'57%DQGQXPEHURIFRQILUPHGFDVHVRI0'57%±
NEW PULMONARY CASES PREVIOUSLY TREATED CASES
YEAR
ESTIMATED CASES
OF MDR-TB AMONG
NOTIFIED
NUMBER OF
BACT+VE
b
TESTED FOR
MDR-TB
% OF
BACT+VE
b
TESTED FOR
MDR-TB
ESTIMATED CASES
OF MDR-TB AMONG
NOTIFIED
NUMBER OF
NOTIFIED
TESTED FOR
MDR-TB
% OF NOT IFIED
TESTED FOR
MDR-TB
TOTAL
CONFIRMED
CASES OF
MDR-TB
a
ESTIMATED CASES
OF MDR-TB AMONG
NOTIFIED
Anguilla 2005 ––
2010 0 0 – 0 –
2011 0 0 – 0 –
2012 0 0 (0–0) 0 (0–0) 0 – 0 (0–0) 0 –
Antigua and 2005 ––
Barbuda 2010 0 0 0 0 –
2011 0 0 0 0 0
2012 0 0.18 (<0.1–0.27) <0.1 (<0.1–<0.1) 0 0 0.14 (<0.1–0.23) 0 0
Argentina 2005 276 2369 46 1290 160
2010 109 ––
2011 103 ––
2012 63 340 (230–440) 160 (88–260) – 180 (110–260) –
Aruba 2005 ––
2010 ––
2011 0 5 71 –
2012 0.85 (0.57–1.1) 0.57 (0.36–0.81) – 0.27 (<0.1–0.46) –
Bahamas 2005 ––
2010 0 21 95 2 100
2011 1 31 97 1 50
2012 1 1.2 (<0.1–6.1) 1.2 (<0.1–6.1) 27 84 0 (0–0) 0 –
Barbados 2005 ––
2010 0 0 0 0 –
2011 0 0 – 0 –
2012 0 <0.1 (<0.1–0.10) <0.1 (<0.1–0.10) 0 0 0 (0–0) 0 –
Belize 2005 0 0 0 3 20
2010 0 ––
2011 0 ––
2012 0 2.5 (1.7–3.4) 1.6 (1.0–2.2) 0 0 0.96 (0.32–1.6) 0 0
Bermuda 2005 ––
2010 0 1 100 0 –
2011 0 1 100 0 –
2012 0 0 (0–1.7) 0 (0–1.7) 2 200 0 (0–0) 0 –
Bolivia 2005 63 ––
(Plurinational 2010 106 0 0 664 100
State of) 2011 83 98 1.7 597 94
2012 117 150 (88–210) 74 (27–160) 1376 22 75 (60–94) 634 94
Bonaire, Saint 2010 0 0 – 0 –
Eustatius and Sab
a
2011 1 – 1 100
2012 0 0 (0–0) 0 (0–0) 0 – 0 (0–0) 0 –
Brazil 2005 373 – 5917 61
2010 573 22 <0.1 643 5.9
2011 566 21 <0.1 604 6.0
2012 684 1 700 (1 400–2 000) 850 (620–1 100) 700 1.6 860 (660–1 100) 198 1.7
British Virgin 2005 ––
Islands 2010 0 0 0 0 –
2011 0 0 – 0 –
2012 0 0 (0–0) 0 (0–0) 0 – 0 (0–0) 0 –
Canada 2005 22 1130 150 –
2010 15 987 130 51 71
2011 19 ––
2012 9 7.4 (2.2–13) 6.0 (2.4–12) 1244 140 1.4 (<0.1–7.8) 63 69
Cayman Islands 2005 ––
2010 0 1 50 0 –
2011 0 1 100 0 –
2012 0 0 (0–3.1) 0 (0–3.1) 5 100 0 (0–0) 0 –
Chile 2005 6 49 3.2 226 72
2010 10 65 4.4 276 100
2011 9 71 4.8 277 100
2012 18 19 (7.5–30) 12 (4.4–26) 125 8.4 6.7 (2.2–15) 172 74
Colombia 2005 ––
2010 131 1240 17 495 57
2011 108 2620 36 568 57
2012 105 310 (220–400) 210 (140–320) 2378 33 98 (74–130) 391 51
Costa Rica 2005 3 2 0.49 1 2.2
2010 3 203 64 –
2011 0 32 9.6 16 62
2012 1 6.4 (0.81–12) 5.4 (1.5–14) 273 95 1.0 (<0.1–5.0) 22 100
Cuba 2005 1 169 32 19 39
2010 7 174 36 31 55
2011 10 313 60 76 100
2012 8 11 (3.4–19) 4.3 (0.52–15) 269 61 7.1 (2.7–14) 51 85
Curaçao 2010 0 5 100 –
2011 0 1 100 0 –
2012 0 0 (0–0.98) 0 (0–0.98) 1 100 0 (0–0) 0 –
Dominica 2005 ––
2010 0 1 12 1 –
2011 0 1 50 1 100
2012 0 0 (0–0) 0 (0–5.9) 2 40 0 (0–2.0) 1 50
Dominican 2005 ––
Republic 2010 108 32 1.4 106 20
2011 117 12 0.42 77 15
2012 92 330 (230–430) 220 (140–330) 79 3.1 110 (71–150) 193 35
Ecuador 2005 253 117 3.2 502 63
2010 176 363 10 584 88
2011 354 239 6.3 284 44
2012 223 380 (320–450) 210 (150–280) 529 13 170 (150–190) 827 120
El Salvador 2005 14 12 1.1 14 12
2010 2 0 0 2 2.2
2011 4 238 22 69 83
2012 8 16 (5.9–26) 5.1 (0.61–18) 252 20 11 (4.8–20) 73 74
Grenada 2005 ––
2010 0 ––
2011 0 ––
2012 0 <0.1 (<0.1–<0.1) <0.1 (<0.1–<0.1) – 0 (0–0) –
Guatemala 2005 40 20 0.83 40 25
2010 18 –189.9
2011 27 0 0 27 17
2012 69 140 (100–180) 89 (55–140) 37 1.4 53 (40–69) 74 37
Guyana 2005 ––
2010 5 0 0 0 0
2011 3 2 0.62 55 24
2012 0 48 (25–70) 14 (9.1–20) 3 0.97 33 (11–56) 1 0.41
a
TOTAL CONFIRMED CASES OF MDR-TB includes cases with unknown previous treatment history (i.e. not included under NEW CASES or PREVIOUSLY TREATED CASES).
b
BACT+VE = bacteriologically positive cases.

201 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
REGION OF THE AMERICAS
7$%/($7HVWLQJIRU0'57%DQGQXPEHURIFRQILUPHGFDVHVRI0'57%±
NEW PULMONARY CASES PREVIOUSLY TREATED CASES
YEAR
ESTIMATED CASES
OF MDR-TB AMONG
NOTIFIED
NUMBER OF
BACT+VE
b
TESTED FOR
MDR-TB
% OF
BACT+VE
b
TESTED FOR
MDR-TB
ESTIMATED CASES
OF MDR-TB AMONG
NOTIFIED
NUMBER OF
NOTIFIED
TESTED FOR
MDR-TB
% OF NOT IFIED
TESTED FOR
MDR-TB
TOTAL
CONFIRMED
CASES OF
MDR-TB
a
ESTIMATED CASES
OF MDR-TB AMONG
NOTIFIED
Haiti 2005 53 0.72 –
2010 41 2 <0.1 39 10
2011 86 ––
2012 81 390 (270–520) 310 (200–440) – 82 (28–140) 81 14
Honduras 2005 3 3 0.13 0 0
2010 9 57 3.1 62 32
2011 5 30 1.5 65 34
2012 6 71 (37–110) 43 (19–84) 41 2.1 28 (13–51) 96 42
Jamaica 2005 0 11 19 2 40
2010 1 40 31 5 26
2011 1 28 64 1 25
2012 0 2.6 (1.7–3.4) 1.7 (1.1–2.4) 16 28 0.82 (0.28–1.4) 0 0
Mexico 2005 394 314 2.1 74 3.7
2010 140 21 0.16 505 40
2011 140 6 <0.1 180 12
2012 114 480 (350–620) 380 (330–440) 13 <0.1 100 (84–130) 148 9.0
Montserrat 2005 1 0 0 0 –
2010 0 0 – 0 –
2011 0 0 – 0 –
2012 0 0 (0–0) 0 (0–0) – 0 (0–0) –
Netherlands Antille
s
2005 ––
Nicaragua 2005 50 8 0.64 8 3.0
2010 18 50 3.5 150 52
2011 13 200 13 67 24
2012 21 46 (21–70) 14 (1.7–52) – 31 (18–49) –
Panama 2005 5 29 3.3 48 19
2010 10 58 8.2 17 8.1
2011 7 25 2.3 40 22
2012 11 56 (35–78) 27 (17–38) 2 0.26 29 (9.9–49) 7 3.3
Paraguay 2005 13 ––
2010 1 115 8.2 52 24
2011 6 227 15 93 31
2012 7 55 (19–90) 6.5 (0.16–36) 235 15 48 (20–92) 89 27
Peru 2005 2748 – 2336 47
2010 1048 ––
2011 1663 1199 6.5 598 16
2012 1225 2 200 (2 100–2 300) 890 (820–960) 14484 79 1 300 (1 200–1 400) 1902 52
Puerto Rico 2005 0 ––
2010 0 69 100 4 100
2011 3 44 110 0 –
2012 1 1.0 (0–2.6) 0 (0–3.8) 52 98 1.0 (<0.1–2.7) 3 100
Saint Kitts and 2005 ––
Nevis 2010 0 0 0 0 –
2011 0 0 0 0 –
2012 0 <0.1 (<0.1–<0.1) <0.1 (<0.1–<0.1) 0 0 0 (0–0) 0 –
Saint Lucia 2005 ––
2010 0 0 0 0 –
2011 0 2 29 0 –
2012 0 0.24 (0.15–0.34) 0.24 (0.15–0.34) 0 0 0 (0–0) 0 –
Saint Vincent and 2005 6 6 86 0 –
the Grenadines 2010 0 2 22 –
2011 0 1 12 0 –
2012 0 1.2 (0.78–1.6) 0.66 (0.42–0.93) 2 7.4 0.55 (0.18–0.92) 0 0
Sint Maarten 2010 0 ––
(Dutch part) 2011 0 0 0 –
2012 0 <0.1 (<0.1–<0.1) <0.1 (<0.1–<0.1) – 0 (0–0) –
Suriname 2005 1 49 44 0 0
2010 0 1 0.70 –
2011 0 0 0 0 0
2012 0 3.4 (2.4–4.5) 2.5 (1.6–3.5) – 0.96 (0.32–1.6) –
Trinidad and 2005 3 0 0 3 14
Tobago 2010 0 ––
2011 ––
2012 0 11 (8.4–13) 4.5 (2.2–6.4) 6 2.4 6.4 (5.0–7.9) 10 19
Turks and Caicos 2005 ––
Islands 2010 1 ––
2011 ––
2012 0.13 (<0.1–0.18) 0.13 (<0.1–0.18) – 0 (0–0) –
United States 2005 124 10064 110 505 –
of America 2010 107 7593 110 345 –
2011 119 6899 99 304 –
2012 81 81 (63–100) 81 (63–100) 6790 100 – 339 –
Uruguay 2005 ––
2010 1 160 36 22 54
2011 1 422 75 38 72
2012 1 1.3 (0–3.8) 0 (0–5.5) 466 88 1.3 (<0.1–6.9) 42 76
US Virgin Islands 2005 ––
2010 ––
2011 ––
2012 – – – – –
Venezuela 2005 28 163 4.3 15 4.3
(Bolivarian 2010 21 26 0.78 160 36
Republic of) 2011 25 565 17 195 48
2012 21 100 (58–150) 26 (7.2–67) 460 13 77 (43–120) 148 26
a
TOTAL CONFIRMED CASES OF MDR-TB includes cases with unknown previous treatment history (i.e. not included under NEW CASES or PREVIOUSLY TREATED CASES).
b
BACT+VE = bacteriologically positive cases.

202 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($1HZVPHDUSRVLWLYHFDVHQRWLILFDWLRQE\DJHDQGVH[±
MALE FEMALE
YEAR 0–14 15–24 25–34 35–44 45–54 55–64 65+
UN-
KNOWN
0–14 15–24 25–34 35–44 45–54 55–64 65+
UN-
KNOWN
MALE:FEMALE
RATIO
An
g
uilla 1995
–
2000
–
2005
–
2010 0000001 0000000
–
2011 00000000 00000000
–
2012 00000000 00000000
–
Anti
g
ua and 1995
–
Barbuda 2000 0000001 1110000 0.33
2005 1 1 2 2 0.50
2010 00202100 00100000 5.0
2011 00113110 01000000 7.0
2012 00001000 00000000
–
Ar
g
entina 1995
–
2000 97 278 594 402 419 368 330 121 544 479 262 230 179 216 1.2
2005 64 621 530 358 384 340 348 90 530 474 290 198 169 240 1.3
2010 56 536 491 309 302 340 282 2 59 421 426 233 184 153 176 1 1.4
2011 143 664 657 434 397 358 289 9 142 587 470 279 192 169 213 4 1.4
2012 59 533 484 299 180 182 181 15 67 652 614 375 364 321 322 13 0.71
Aruba 1995
–
2000
–
2005
–
2010
–
2011 4 1 1 1 2.5
2012 2 3 1 4 2 1 1 0.56
Bahamas 1995 3357422 0172001 2.4
2000 1279432 2578231 1.0
2005
–
2010 02350200 05110000 1.7
2011 02362210 01330000 2.3
2012 01162020 02511000 1.3
Barbados 1995
–
2000 0002000 0010000 2.0
2005
–
2010 00012000 01002000 1.0
2011 00000000 00000000
–
2012 00000000 00000000
–
Belize 1995 1124011 0620112 0.83
2000 2572635 0212414 2.1
2005 0886853 0444324 1.8
2010 2 9 16 22 24 11 18 4577945 2.5
2011 0 8 14 9 16 2 0 0208410 3.3
2012 12754320 04344100 1.5
Bermuda 1995
–
2000
–
2005
–
2010 00010000 00000000
–
2011 00000000 00000000
–
2012 00000000 00100000
–
Bolivia 1995
–
(
Plurinational 2000 166 1 182 797 518 466 340 366 191 831 588 334 254 192 233 1.5
State of
)
2005 157 1 320 725 439 391 346 415 160 846 533 276 226 182 262 1.5
2010 95 1 150 622 415 395 338 409 119 744 471 238 191 162 264 1.6
2011 100 1 231 685 372 371 302 457 146 778 459 235 183 155 272 1.6
2012 99 1 096 672 368 358 353 380 101 792 480 223 204 193 249 1.5
Bonaire, Saint 2010 0000000 0000000
–
Eustatius and Saba 2011 00000000 00000000
–
2012 00000000 00000000
–
Brazil 1995
–
2000 1 894 7 268 11 568 11 906 8 623 5 085 4 494 1 859 6 719 7 215 5 395 3 582 2 384 2 496 1.7
2005 317 5 074 6 119 6 128 5 259 2 803 2 140 355 3 496 3 663 2 626 1 897 1 112 1 104 2.0
2010 298 4 405 6 381 5 293 4 762 2 875 1 947 43 280 2 677 3 008 2 211 1 720 1 038 979 15 2.2
2011 336 4 877 6 755 5 462 5 054 3 083 2 142 41 356 2 815 3 131 2 230 1 779 1 164 1 069 0 2.2
2012 277 5 027 6 811 5 387 5 128 3 103 2 160 38 303 2 798 3 013 2 173 1 785 1 113 1 030 6 2.3
British Vir
g
in 1995
–
Islands 2000 1
–
2005
–
2010 00000100 00000000
–
2011 00000000 00000000
–
2012 00000000 00000000
–
Canada 1995 1 28 31 60 34 41 70 7 33 28 22 12 18 51 1.5
2000 5 34 45 46 41 32 79 4 33 40 30 25 12 66 1.3
2005 3 37 45 44 40 20 68 6 28 40 27 24 13 37 1.5
2010 3 30 28 36 32 25 62 0 1 28 24 16 10 19 44 0 1.5
2011 2 34 36 31 40 33 70 0 3 23 29 28 14 9 55 0 1.5
2012 1 33 32 53 51 35 97 0 6 32 34 29 19 11 45 0 1.7
Ca
y
man Islands 1995
–
2000 0031010
–
2005
–
2010 00010000 00001000 1.0
2011 00000100 00000000
–
2012 00221000 00000000
–
Chile 1995 24 148 182 204 155 141 163 24 100 120 108 75 73 107 1.7
2000 6 81 160 198 150 132 126 10 66 96 70 54 58 83 2.0
2005 3 74 128 179 162 115 133 4 55 78 60 56 36 93 2.1
2010 2 90 115 144 159 122 157 0 6 56 76 59 56 40 72 0 2.2
2011 4 88 139 143 164 127 134 0 6 62 75 66 69 48 71 0 2.0
2012 4 91 122 135 170 117 149 5 4 59 69 53 56 60 76 3 2.1
Colombia 1995
–
2000 246 763 1 030 963 743 610 746 194 587 758 523 381 304 510 1.6
2005 178 623 685 666 687 510 695 179 581 533 457 389 292 395 1.4
2010 148 602 765 540 710 610 814 0 146 560 576 428 374 284 471 0 1.5
2011 105 663 714 558 702 594 753 0 98 461 535 324 337 278 390 0 1.7
2012 92 613 744 497 653 616 740 0 79 519 555 376 355 252 432 0 1.5
Costa Rica 1995 1 17 38 24 19 23 22 2 17 15 11 7 9 14 1.9
2000 14 31 53 62 39 28 49 13 21 33 24 20 23 24 1.7
2005 1 43 38 53 34 20 34 1 21 31 18 16 6 14 2.1
2010 2 18 48 33 27 22 28 1 0 18 20 12 14 15 8 1 2.0
2011 0 23 24 29 33 22 36 0 2 18 27 23 19 12 17 0 1.4
2012 2 18 33 28 34 41 23 2 2 11 24 11 12 8 5 3 2.4
Cuba 1995 2 59 118 83 75 75 156 1 17 52 29 39 48 80 2.1
2000 0 71 167 90 74 55 75 2 9 22 26 22 23 39 3.7
2005 2 20 73 90 50 58 51 2 14 17 26 13 22 29 2.8
2010 3 17 61 89 78 53 57 0 1 15 15 14 16 17 26 0 3.4
2011 2 14 51 83 86 50 48 0 1 6 18 18 17 17 26 0 3.2
2012 1 15 45 83 70 45 36 0 0 13 12 16 12 13 13 0 3.7
Cura
ç
ao 2010 00021000 01100000 1.5
2011 0000000 0000000
–
2012 00000000 01000000
–
Dominica 1995
–
2000
–
2005
–
2010 00000310 00012100 1.0
2011 0 0 20000 00000000
–
2012 02001200 00000000
–

203 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
REGION OF THE AMERICAS
7$%/($1HZVPHDUSRVLWLYHFDVHQRWLILFDWLRQE\DJHDQGVH[±
MALE FEMALE
YEAR 0–14 15–24 25–34 35–44 45–54 55–64 65+
UN-
KNOWN
0–14 15–24 25–34 35–44 45–54 55–64 65+
UN-
KNOWN
MALE:FEMALE
RATIO
Dominican 1995
–
Re
p
ublic 2000 73 410 481 344 173 125 113 65 317 325 212 115 79 75 1.4
2005 43 399 483 386 228 123 105 57 339 332 209 119 72 54 1.5
2010 29 276 346 292 170 112 85 0 43 239 207 142 102 54 62 0 1.5
2011 20 333 406 318 200 133 112 0 30 242 274 159 103 66 58 0 1.6
2012 15 317 489 315 197 126 111 0 26 230 260 148 119 62 68 0 1.7
Ecuador 1995
–
2000
–
2005 48 446 468 308 237 150 159 48 329 305 199 139 85 127 1.5
2010 32 499 529 314 309 227 246 52 298 308 178 158 113 110 1.8
2011 45 481 547 364 323 272 232 49 340 311 177 141 118 121 1.8
2012 37 506 567 387 359 291 333 59 333 337 184 164 146 153 1.8
El Salvador 1995
–
2000 13 99 124 114 92 62 107 28 81 76 63 63 39 47 1.5
2005 5 97 140 128 104 74 117 6 85 82 59 50 42 70 1.7
2010 5 101 170 96 77 62 101 0 6 63 65 49 58 51 68 0 1.7
2011 3 114 183 106 96 77 115 0 6 61 61 44 52 69 92 0 1.8
2012 5 131 194 122 100 87 115 0 5 81 73 80 90 64 90 0 1.6
Grenada 1995
–
2000
–
2005
–
2010 1 1 1 1 3.0
2011 00001000 00000000
–
2012 1
–
Guatemala 1995 51 235 280 236 165 142 139 51 224 255 221 146 129 94 1.1
2000 36 220 236 216 177 112 140 41 199 167 175 135 87 111 1.2
2005 39 251 258 185 187 127 115 38 339 245 277 176 88 95 0.92
2010 60 187 245 207 172 143 165 29 194 190 179 139 108 103 1.3
2011 18 197 205 172 162 136 152 25 186 192 154 154 102 106 1.1
2012
–
Gu
y
ana 1995 7856967 3576524 1.5
2000 4 20 19 14 7 6 9 1 11 87553 2.0
2005 12 48 130 116 81 41 20 14 41 62 41 30 11 9 2.2
2010 2 32 38 65 49 22 13 0 2 22 25 19 20 10 6 0 2.1
2011 8 26 54 61 54 19 13 0 2 17 19 17 17 7 9 0 2.7
2012 5 30 39 68 64 23 8 0 4 17 10 17 12 7 5 0 3.3
Haiti 1995
–
2000 67 836 898 613 350 147 118 96 914 857 513 275 132 71 1.1
2005 69 1 045 1 035 701 451 222 156 116 1 097 1 099 633 414 170 132 1.0
2010 98 1 225 1 357 718 469 259 160 0 158 1 268 1 223 608 358 207 134 0 1.1
2011 102 1 155 1 342 670 442 206 132 0 148 1 282 1 250 595 363 196 128 0 1.0
2012 126 1 359 1 563 758 473 271 164 160 1 476 1 415 698 416 219 156 1.0
Honduras 1995 42 280 540 204 130 236 58 54 208 292 134 76 136 48 1.6
2000 30 123 371 246 277 214 43 25 21 269 258 270 160 38 1.3
2005 13 238 280 215 152 134 152 27 219 222 125 107 81 104 1.3
2010 15 177 246 207 165 113 157 0 28 186 163 106 103 69 107 0 1.4
2011 17 194 291 227 184 120 184 0 19 181 194 138 99 98 126 0 1.4
2012 18 247 285 192 184 129 146 0 15 180 157 115 88 75 114 0 1.6
Jamaica 1995 2 9 14 9 11 8 9 2765522 2.1
2000 0 6 13 13 15 6 5 1887251 1.8
2005 04661067 0154013 2.8
2010 1 7 15 15 8670 05451020 3.5
2011 02634430 13403110 1.7
2012 1 10 835510 11542000 2.5
Mexico 1995
–
2000 214 1 079 1 387 1 162 1 235 972 1 126 176 663 828 698 832 595 709 1.6
2005 100 1 095 1 376 1 314 1 238 1 042 1 288 125 771 733 710 784 637 784 1.6
2010 125 1 081 1 375 1 380 1 392 1 119 1 303 0 112 791 763 730 852 713 836 0 1.6
2011 128 1 124 1 440 1 503 1 532 1 112 1 299 0 136 776 765 698 889 734 824 0 1.7
2012 133 1 153 1 480 1 522 1 484 1 153 1 284 0 134 778 743 686 840 824 824 0 1.7
Montserrat 1995
–
2000
–
2005 1
–
2010 00000000 00000000
–
2011 00000000 00000000
–
2012 00000000 00000000
–
Netherlands 1995
–
Antilles 2000 0012000 0010010 1.5
2005
–
Nic ar a
g
ua 1995 23 178 172 175 126 96 92 24 176 215 98 83 64 46 1.2
2000 18 194 174 147 108 64 90 34 188 173 98 76 46 61 1.2
2005 17 163 159 116 106 61 79 23 135 122 103 61 54 47 1.3
2010 22 157 189 141 115 82 108 0 27 154 149 92 75 50 79 0 1.3
2011 10 273 235 156 108 61 94 0 4 61 145 161 108 64 72 0 1.5
2012 00000000 00000000
–
Panama 1995 86 155 193 112 126 42 83 72 120 111 75 57 16 40 1.6
2000 3 44 78 61 37 27 26 6 43 34 35 19 12 16 1.7
2005 5 76 129 129 84 57 49 11 73 81 62 33 30 41 1.6
2010 6 69 127 80 62 61 49 0 7 51 52 46 45 23 29 0 1.8
2011 10 96 104 91 99 63 47 0 11 55 64 58 44 40 48 0 1.6
2012 19 88 103 104 67 51 61 0 9 62 57 45 46 22 44 0 1.7
Para
g
ua
y
1995 18 64 71 96 74 57 61 13 65 49 46 35 34 53 1.5
2000 16 112 103 105 86 80 71 12 69 86 41 41 30 46 1.8
2005 23 168 185 136 117 87 99 31 89 98 69 52 29 71 1.9
2010 18 163 244 129 143 103 99 11 18 106 99 39 50 46 45 5 2.2
2011 9 182 238 135 151 124 103 6 14 110 103 55 39 36 62 4 2.2
2012 4 180 230 158 143 116 129 7 16 95 98 60 55 38 60 2 2.3
Peru 1995 147 1 311 849 454 322 200 216 149 1 005 660 373 259 162 152 1.3
2000 552 5 290 2 875 1 546 1 041 801 796 633 3 686 2 472 1 156 609 499 624 1.3
2005 371 3 802 2 670 1 513 1 075 641 708 375 2 674 2 111 1 046 699 333 472 1.4
2010
–
2011
–
2012
–
Puerto Rico 1995 4 3 12 20 15 9 19 1265749 2.4
2000 0 1 4 19 9 10 14 1453713 2.4
2005 0447977 0325417 1.7
2010 00324580 01026240 1.5
2011 01436620 01110310 3.1
2012 015168100 00312310 3.1
Saint Kitts and 1995
–
Nevis 2000
–
2005
–
2010 00001000 00010000 1.0
2011 00000100 00000000
–
2012 00000010 00100000 1.0
Saint Lucia 1995
–
2000 0001012 0101010 1.3
2005 0000212 1101102 0.83
2010 00120120 00010110 2.0
2011 00110320 00000000
–
2012 02014020 01000100 4.5

204 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($1HZVPHDUSRVLWLYHFDVHQRWLILFDWLRQE\DJHDQGVH[±
MALE FEMALE
YEAR 0–14 15–24 25–34 35–44 45–54 55–64 65+
UN-
KNOWN
0–14 15–24 25–34 35–44 45–54 55–64 65+
UN-
KNOWN
MALE:FEMALE
RATIO
Saint Vincent and 1995
–
the Grenadines 2000 0104201 1000000 8.0
2005 0002102 0010100 2.5
2010 00103020 00100100 3.0
2011 00222000 00101000 3.0
2012 01513540 01313000 2.4
Sint Maarten 2010 1 20.50
(
Dutch
p
art
)
2011 1 11.0
2012 00000000 01000000
–
Suriname 1995
–
2000 1663204 2363011 1.4
2005 0 7 8 12 6 3 4 0321212 3.6
2010 0 5 21 35 19 5 10 0 1 4 6 10 6 2 8 0 2.6
2011 0 4 7 15 18 3 5 0 01152210 4.3
2012 0 6 7 15 14 9 7 0 21757102 2.3
Trinidad and 1995 2 6 15 10 12 7 4 0642530 2.8
Toba
g
o 2000 0 7 18 27 17 7 7 0579524 2.6
2005 0 10 11 13 21 10 3 0493543 2.4
2010 0 11 21 17 32 20 8 0 04775220 4.0
2011 1 14 27 13 15 16 7 0 16734250 3.3
2012 0 7 31 22 28 12 11 0 2 9 11 10 8 4 12 0 2.0
Turks and Caicos 1995
–
Islands 2000
–
2005
–
2010 00010000 00011000 0.50
2011 02320100 00000000
–
2012 0 0 20000 00201000 0.67
United States 1995 19 355 876 1 417 1 121 742 1 099 26 280 579 499 285 202 591 2.3
of America 2000 6 365 602 906 904 577 738 14 246 376 349 253 152 396 2.3
2005 14 383 535 666 767 499 624 11 241 348 276 242 161 322 2.2
2010 5 246 360 371 505 403 466 2 9 195 265 183 165 130 223 0 2.0
2011 12 235 403 374 557 434 486 0 15 160 254 199 150 138 269 1 2.1
2012 10 239 322 333 502 455 529 0 14 161 262 169 175 148 243 1 2.0
Uru
g
ua
y
1995 4 28 40 35 49 38 50 2 21 26 18 12 9 17 2.3
2000 0 36 48 45 41 30 34 2 28 22 21 13 12 16 2.1
2005 1 42 48 39 45 34 36 1 33 30 17 9 8 12 2.2
2010 1 46 70 35 46 33 31 0 3 24 36 12 10 5 16 0 2.5
2011 0 58 93 55 45 36 37 0 1 29 55 19 12 11 16 0 2.3
2012 3 38 98 56 52 39 29 0 2 25 26 21 15 13 15 0 2.7
US Vir
g
in Islands 1995 0001100
–
2000
–
2005
–
2010
–
2011
–
2012
–
Venezuela 1995
–
(
Bolivarian 2000
–
Re
p
ublic of
)
2005 35 312 395 413 402 265 332 37 351 299 267 183 146 216 1.4
2010 22 320 376 333 391 253 288 26 269 306 188 145 147 188 1.6
2011 28 340 353 303 363 307 241 0 25 252 316 178 178 150 190 0 1.5
2012 23 379 405 353 375 319 273 0 32 276 281 203 167 161 199 0 1.6

205 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
REGION OF THE AMERICAS
a LED = Light emitting diode microscopes
b DST = Drug susceptibility testing
c LPA = Line probe assay
d NRL = National Reference Laboratory
7$%/($/DERUDWRULHV173VHUYLFHVGUXJPDQDJHPHQWDQGLQIHFWLRQFRQWURO
LABORATORIES FREE THROUGH NTP
SMEAR LABS
PER 100K
POPULATION
% OF SMEAR
LABS USING
LED
a
CULTURE
LABS PER 5M
POPULATION
DST
b
LABS
PER 5M
POPULATION
LPA
c
LABS
PER 5M
POPULATION
NUMBER OF
LABS USING
XPERT MTB/RIF
SECOND-
LINE DST
AVAILABLE
NRL
d
TB DIAGNOSIS
FIRST-
LINE
DRUGS
TB NOTIF.
RATE PER
100 000
HEALTH-CARE
WORKERS
RIFAMPICIN
USED
THROUGHOUT
TREATMENT
Anguilla – No No No No No
Antigua and Barbuda – Out of
country Yes Yes (all suspects) Yes Yes
Argentina 1.7 0 12.5 1.9 0 0 In country Yes Yes (all suspects) Yes Yes
Aruba – Out of
country Yes No No Yes
Bahamas – Out of
country Yes Yes (other criteria) Yes Yes
Barbados – Out of
country Yes Yes (all suspects) Yes Yes
Belize 0.9 0 0 0 0 0 Out of
country No Yes (all suspects) Yes Yes
Bermuda – Out of
country Yes Yes (all suspects) Yes Yes
Bolivia
(Plurinational State of) 5.1 0 24.8 0.5 0 0 Out of
country Yes Yes (all suspects) Yes Yes
Bonaire, Saint Eustatius
and Saba –Out of
country Yes Yes (all suspects) Don't
know Yes
Brazil 2.0 – 5.5 0.9 0.2 13 In country Yes Yes (all suspects) Yes Yes
British Virgin Islands – Out of
country Yes Yes (all suspects) No Yes
Canada – In country Yes Yes (all suspects) Yes Yes
Cayman Islands – Out of
country Yes Yes (all suspects) Yes Yes
Chile 1.0 0 11.2 0.3 0.3 1 In country Yes Yes (all suspects) Yes Yes
Colombia 5.6 0 124.6 0.4 0.5 4 In country Yes Yes (all suspects) Yes Yes
Costa Rica 2.2 0 14.6 1 0 4 No Yes Yes (all suspects) Yes Yes
Cuba – No Yes Yes (all suspects) Yes Yes
Curaçao – Out of
country Yes Yes (all suspects) Yes Yes
Dominica – Out of
country Yes Yes (all suspects) Yes Yes
Dominican Republic 2.0 2 5.8 1 0 0 In country Yes Yes (all suspects) Yes Yes
Ecuador 2.3 0 5.8 0.3 5 In country Yes Yes (all suspects) Yes Yes
El Salvador 3.3 0 8.7 0.8 0 1 Out of
country Yes Yes (all suspects) Yes Yes 83
Grenada – Yes (all suspects) Yes Yes
Guatemala 1.9 18 3.3 1 0 0 Out of
country Yes Yes (all suspects) Yes Yes 14
Guyana 2.5 100 6.3 6.3 6.3 0 In and out
of countr y Yes Yes (all suspects) Yes Yes
Haiti 2.5 6 1.0 1 1 7 Yes Yes (all suspects) Yes Yes
Honduras 2.1 0 3.2 0.6 0 0 Out of
country Yes Yes (all suspects) Yes Yes 109
Jamaica 0.1 100 0 0 0 0 Out of
country Yes Yes (all suspects) Yes Yes 21
Mexico 1.0 0 2.7 0.6 <0.1 6 In and out
of countr y Yes Yes (all suspects) Yes Yes 33
Montserrat – Out of
country No Yes (all suspects) No Yes
Nicaragua 3.2 100 1.7 0.8 0 Out of
country Yes Yes (all suspects) Yes Yes
Panama 1.4 0 14.5 1.3 1.3 3 No Yes Yes
(
if TB is
confirmed) Yes Yes
Paraguay 1.8 23 8.2 0.7 0 0 In country Yes Yes (all suspects) Yes Yes 7
Peru 4.8 0 11.0 1.8 0.2 0 In country Yes Yes (all suspects) Yes Yes
Puerto Rico – Out of
country Yes Yes (all suspects) Yes Yes
Saint Kitts and Nevis – No No Yes (all suspects) Yes Yes
Saint Lucia – Out of
country Yes Yes (all suspects) Yes Yes
Saint Vincent and
the Grenadines –Out of
country Yes Yes (all suspects) Yes Yes
Sint Maarten (Dutch part) – Out of
country No Yes (other criteria) Yes Yes
Suriname 0.6 0 9.4 0 0 2 Out of
country Yes Yes (all suspects) Yes Yes
Trinidad and Tobago – In and out
of countr y Yes Yes (all suspects) Yes Yes
Turks and Caicos Islands –
United States of America – In country Yes Yes (all suspects) Yes Yes
Uruguay <0.1 100 1.5 1.5 1.5 0 Out of
country Yes Yes (all suspects) Yes Yes 31
US Virgin Islands –
Venezuela
(Bolivarian Republic of) 0.8 0 3.5 0.2 0.2 0 In country Yes Yes (all suspects) Yes Yes 2 242

206 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($0HDVXUHGSHUFHQWDJHRI7%FDVHVZLWK0'57%
D
PRVWUHFHQW\HDUDYDLODEOH
New TB cases Previously treated TB cases
Year Source Coverage Percentage Year Source Coverage Percentage
Anguilla
Antigua and Barbuda
Argentina 2005 Survey National 2.2 (1.2–3.6) 2005 Survey National 15 (9.8–23)
Aruba
Bahamas 2012 Surveillance National 3.7 (<0.1–19) 2012 Surveillance National 0 (0–98)
Barbados
Belize
Bermuda 2012 Surveillance National 0 (0–84) 2012 Surveillance National 0 (0–98)
Bolivia
(Plurinational State of) 1996 Survey National 1.2 (0.44–2.6) 2012 Surveillance National 11 (8.9–14)
Bonaire, Saint Eustatius
and Saba 2011 Surveillance National 50 (1.3–99) 2011 Surveillance National 100 (2.5–100)
Brazil 2008 Survey Sub-national 1.4 (1.0–1.8) 2008 Survey Sub-national 7.5 (5.7–9.9)
British Virgin Islands
Canada 2012 Surveillance National 0.57 (0.23–1.2) 2012 Surveillance National 1.6 (<0.1–8.5)
Cayman Islands 2012 Surveillance National 0 (0–52) 2012 Surveillance National 0 (0–98)
Chile 2001 Survey National 0.69 (0.25–1.5) 2012 Surveillance National 2.9 (0.95–6.7)
Colombia 2005 Survey National 2.4 (1.6–3.6) 2012 Surveillance National 13 (9.6–17)
Costa Rica 2006 Survey National 1.5 (0.42–3.9) 2012 Surveillance National 4.5 (0.12–23)
Cuba 2012 Surveillance National 0.74 (<0.1–2.7) 2012 Surveillance National 12 (4.4–24)
Curaçao 2012 Surveillance National 0 (0–98) 2012 Surveillance National 0 (0–98)
Dominica 2011 Surveillance National 0 (0–98) 2012 Surveillance National 0 (0–98)
Dominican Republic 1995 Survey National 6.6 (4.1–10) 1995 Survey National 20 (13–28)
Ecuador 2002 Survey National 4.9 (3.5–6.7) 2012 Surveillance National 26 (23–29)
El Salvador 2001 Survey National 0.33 (<0.1–1.2) 2012 Surveillance National 11 (4.9–20)
Grenada
Guatemala 2002 Survey National 3 (1.8–4.6) 2002 Survey National 26 (20–34)
Guyana
Haiti
Honduras 2004 Survey National 1.8 (0.76–3.4) 2004 Survey National 12 (5.8–22)
Jamaica
Mexico 2009 Survey National 2.4 (2.1–2.8) 2009 Survey National 6.3 (5.1–7.8)
Montserrat
Nicaragua 2006 Survey National 0.63 (<0.1–2.2) 2010 Surveillance National 11 (6.2–17)
Panama
Paraguay 2008 Survey National 0.31 (<0.1–1.7) 2008 Survey National 15 (6.1–28)
Peru 2012 Surveillance National 3.9 (3.6–4.2) 2012 Surveillance National 35 (33–37)
Puerto Rico 2012 Surveillance National 0 (0–6.5) 2012 Surveillance National 33 (0.84–91)
Saint Kitts and Nevis
Saint Lucia
Saint Vincent and
the Grenadines
Sint Maarten (Dutch part)
Suriname
Trinidad and Tobago
Turks and Caicos Islands
United States of America 2012 Surveillance National 1 (0.80–1.3) 2012 Surveillance National 2.9 (1.4–5.4)
Uruguay 2012 Surveillance National 0 (0–0.79) 2012 Surveillance National 2.4 (<0.1–13)
Venezuela
(Bolivarian Republic of) 1999 Survey National 0.52 (0.14–1.3) 1999 Survey National 13 (7.6–22)
a Empty rows indicate an absence of high-quality survey or surveillance data. In the absence of high-quality national data, high-quality sub-national data are used.

'#56'40/'&+6'44#0'#04')+10
Table A4.1 Estimates of the burden of disease caused by TB, 1990–2012 209
6CDNG# +PEKFGPEGPQVKßECVKQPCPFECUGFGVGEVKQPTCVGUCNNHQTOU¿
6CDNG# %CUGPQVKßECVKQPU¿
Table A4.4 Treatment outcomes, new smear-positive cases, 1995–2011 215
Table A4.5 Treatment outcomes, retreatment cases, 1995–2011 217
6CDNG# *+8VGUVKPICPFRTQXKUKQPQH%26#46CPF+26¿
6CDNG# 6GUVKPIHQT/&46$CPFPWODGTQHEQPßTOGFECUGUQH/&46$¿
6CDNG# 0GYUOGCTRQUKVKXGECUGPQVKßECVKQPD[CIGCPFUGZ¿
Table A4.9 Laboratories, NTP services, drug management and infection control, 2012 223
6CDNG# /GCUWTGFRGTEGPVCIGQH6$ECUGUYKVJ/&46$OQUVTGEGPV[GCTCXCKNCDNG

GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
Estimates of mortality, prevalence and incidence
Estimated values are shown as best estimates followed by lower and upper bounds. e lower and upper bounds are dened
as the 2.5th and 97.5th centiles of outcome distributions produced in simulations. See ANNEX 1 for further details.
Estimated numbers are shown rounded to two signicant gures. Estimated rates are shown rounded to three signicant
gures unless the value is under 100, in which case rates are shown rounded to two signicant gures.
Estimates for all years are recalculated as new information becomes available and techniques are rened, so
they may dier from those published in previous reports in this series. e main updates implemented in this
report are explained in Box 2.1 of Chapter 2. Estimates published in previous global TB control reports should no
longer be used.
Data source
Data shown in this annex are taken from the WHO global TB database on 1 October 2013. Data shown in the main part of
the report were taken from the database in July 2013. As a result, data in this annex may dier slightly from those in the
main part of the report.
Data for all years can be downloaded from www.who.int/tb/data.

209 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
EASTERN MEDITERRANEAN REGION
7$%/($(VWLPDWHVRIWKHEXUGHQRIGLVHDVHFDXVHGE\7%±
YEAR POPULATION
(MILLIONS)
NUMBER
(THOUSANDS) RATE
a
NUMBER
(THOUSANDS) RATE
a
NUMBER
(THOUSANDS) RATE
a
MORTALITY (EXCLUDING HIV) PREVALENCE (INCLUDING HIV) INCIDENCE (INCLUDING HIV)
Afghanistan 1990 12 3.7 (0.860–8.5) 31 (7.3–72) 38 (13–77) 327 (112–655) 22 (14–33) 189 (117–279)
1995 18 8.7 (2.9–18) 49 (17–100) 79 (37–140) 447 (208–775) 33 (27–40) 189 (155–227)
2000 21 11 (4.0–21) 53 (19–102) 92 (43–160) 449 (210–775) 39 (32–47) 189 (155–227)
2005 25 9.7 (3.9–18) 39 (16–73) 92 (46–150) 369 (185–617) 47 (38–56) 189 (155–227)
2010 28 10 (4.2–18) 35 (15–65) 99 (50–160) 350 (177–580) 54 (44–64) 189 (156–225)
2011 29 10 (4.4–19) 36 (15–66) 100 (52–170) 352 (177–585) 55 (45–66) 189 (156–225)
2012 30 11 (4.6–20) 37 (15–68) 110 (54–180) 358 (181–595) 56 (47–67) 189 (156–226)
Bahrain 1990 < 1 0.034 (0.032–0.037) 7 (6.5–7.4) 0.16 (0.049–0.350) 33 (9.9–70) 0.13 (0.120–0.150) 27 (24–31)
1995 < 1 0.02 (0.018–0.022) 3.5 (3.2–3.8) 0.081 (0.024–0.170) 14 (4.3–30) 0.049 (0.043–0.056) 8.8 (7.7–9.9)
2000 < 1 0.017 (0.015–0.020) 2.5 (2.2–3.0) 0.37 (0.180–0.630) 56 (27–94) 0.24 (0.210–0.270) 36 (31–40)
2005 < 1 <0.01 (<0.01–<0.01) 0.85 (0.78–0.93) 0.42 (0.170–0.790) 48 (19–89) 0.32 (0.280–0.360) 37 (32–41)
2010 1 <0.01 (<0.01–<0.01) 0.44 (0.38–0.51) 0.34 (0.110–0.690) 27 (9.0–55) 0.28 (0.250–0.320) 23 (20–26)
2011 1 <0.01 (<0.01–<0.01) 0.39 (0.32–0.46) 0.32 (0.120–0.630) 25 (9.0–49) 0.26 (0.230–0.290) 20 (18–23)
2012 1 <0.01 (<0.01–<0.01) 0.34 (0.28–0.41) 0.38 (0.180–0.650) 29 (14–49) 0.26 (0.230–0.290) 20 (17–22)
Djibouti 1990 < 1 0.59 (0.140–1.3) 99 (24–226) 6.2 (2.2–12) 1 050 (368–2 070) 3.7 (2.3–5.3) 619 (395–893)
1995 < 1 0.4 (0.160–0.750) 60 (23–114) 5.4 (2.2–10) 809 (326–1 510) 4.1 (3.4–4.9) 619 (506–744)
2000 < 1 0.41 (0.180–0.740) 57 (25–102) 5.6 (2.4–10) 775 (333–1 400) 4.5 (3.8–5.2) 619 (528–718)
2005 < 1 0.65 (0.260–1.2) 83 (33–156) 7.1 (3.4–12) 920 (444–1 570) 4.8 (3.9–5.8) 619 (506–744)
2010 < 1 0.68 (0.290–1.2) 82 (34–149) 7.7 (3.7–13) 922 (440–1 580) 5.2 (4.3–6.2) 620 (512–738)
2011 < 1 0.67 (0.280–1.2) 79 (34–144) 7.7 (3.6–13) 911 (430–1 570) 5.2 (4.3–6.2) 620 (512–738)
2012 < 1 0.66 (0.280–1.2) 76 (33–139) 7.7 (3.6–13) 897 (418–1 560) 5.3 (4.4–6.3) 620 (512–738)
Egypt 1990 56 1.8 (1.4–2.2) 3.2 (2.5–3.9) 48 (22–84) 85 (39–149) 19 (16–23) 34 (29–40)
1995 61 1.5 (1.2–1.9) 2.5 (1.9–3.2) 37 (19–61) 60 (31–99) 19 (16–23) 32 (27–37)
2000 66 1.1 (0.840–1.4) 1.7 (1.3–2.2) 28 (14–46) 42 (20–70) 17 (14–20) 26 (22–30)
2005 72 0.76 (0.700–0.830) 1.1 (0.97–1.2) 24 (12–41) 34 (17–57) 15 (13–18) 21 (18–25)
2010 78 0.45 (0.420–0.490) 0.58 (0.54–0.62) 23 (12–39) 30 (15–50) 14 (12–16) 18 (15–21)
2011 79 0.56 (0.530–0.600) 0.71 (0.66–0.76) 23 (12–39) 29 (15–49) 14 (12–16) 17 (15–20)
2012 81 0.38 (0.350–0.400) 0.46 (0.43–0.50) 23 (12–39) 29 (15–48) 14 (12–16) 17 (14–19)
Iran (Islamic 1990 56 2.6 (0.870–5.3) 4.6 (1.5–9.4) 29 (12–53) 51 (21–93) 18 (13–23) 31 (23–41)
Republic of) 1995 60 3.2 (1.1–6.5) 5.4 (1.8–11) 35 (15–64) 58 (24–106) 21 (16–28) 35 (26–46)
2000 66 2.5 (0.830–5.1) 3.8 (1.3–7.7) 27 (11–51) 41 (17–77) 17 (12–22) 26 (19–34)
2005 70 2.1 (0.680–4.2) 3 (0.97–6.0) 23 (9.4–42) 32 (13–60) 14 (10–19) 20 (15–27)
2010 74 2.2 (0.730–4.5) 3 (0.98–6.0) 24 (9.9–44) 32 (13–59) 15 (11–19) 20 (14–26)
2011 75 2.3 (0.780–4.7) 3.1 (1.0–6.3) 25 (11–47) 34 (14–62) 16 (11–21) 21 (15–27)
2012 76 2.2 (0.700–4.7) 2.9 (0.92–6.1) 25 (10–47) 33 (13–61) 16 (11–21) 21 (15–28)
Iraq 1990 18 1.2 (0.410–2.4) 6.9 (2.3–14) 17 (4.9–35) 94 (28–200) 9.5 (8.3–11) 54 (47–62)
1995 20 1.2 (0.310–2.6) 5.7 (1.5–13) 16 (4.9–34) 79 (24–167) 11 (9.4–12) 53 (46–60)
2000 24 1.1 (0.180–2.9) 4.7 (0.77–12) 14 (5.0–29) 61 (21–121) 12 (10–14) 50 (44–57)
2005 27 1.1 (0.100–3.1) 3.9 (0.38–11) 19 (8.8–33) 70 (32–122) 13 (11–15) 48 (42–54)
2010 31 0.98 (0.039–3.4) 3.2 (0.13–11) 24 (13–40) 78 (41–128) 14 (12–16) 45 (40–52)
2011 32 0.97 (0.030–3.5) 3 (0.10–11) 23 (12–39) 73 (37–123) 14 (13–16) 45 (39–51)
2012 33 0.96 (0.025–3.5) 2.9 (<0.1–11) 24 (12–40) 73 (36–122) 15 (13–17) 45 (39–51)
Jordan 1990 3 0.041 (0–0.330) 1.2 (0–9.9) 0.61 (0.230–1.2) 18 (6.8–35) 0.48 (0.420–0.550) 14 (13–16)
1995 4 0.04 (0–0.390) 0.93 (0–9.0) 0.65 (0.250–1.2) 15 (5.8–29) 0.51 (0.450–0.580) 12 (10–13)
2000 5 0.039 (0–0.410) 0.81 (0–8.6) 0.48 (0.180–0.930) 10 (3.8–20) 0.38 (0.340–0.440) 8.1 (7.1–9.1)
2005 5 0.036 (0–0.410) 0.7 (0–7.8) 0.47 (0.170–0.910) 9 (3.3–17) 0.38 (0.330–0.430) 7.2 (6.3–8.1)
2010 6 0.037 (0–0.420) 0.57 (0–6.5) 0.57 (0.240–1.0) 8.8 (3.8–16) 0.41 (0.360–0.460) 6.3 (5.5–7.1)
2011 7 0.037 (0–0.420) 0.55 (0–6.3) 0.57 (0.250–1.0) 8.5 (3.7–15) 0.4 (0.350–0.460) 6 (5.2–6.8)
2012 7 0.037 (0–0.420) 0.53 (0–6.0) 0.6 (0.280–1.0) 8.5 (3.9–15) 0.4 (0.360–0.460) 5.8 (5.1–6.5)
Kuwait 1990 2 0.019 (0.017–0.022) 0.94 (0.81–1.1) 0.48 (0.230–0.830) 23 (11–40) 0.32 (0.280–0.360) 15 (14–18)
1995 2 0.023 (0.021–0.024) 1.4 (1.3–1.5) 0.46 (0.160–0.930) 29 (9.9–59) 0.39 (0.340–0.440) 24 (21–28)
2000 2 0.015 (0.014–0.015) 0.76 (0.75–0.78) 0.77 (0.300–1.5) 41 (16–77) 0.59 (0.520–0.670) 31 (27–35)
2005 2 0.023 (0.022–0.023) 0.99 (0.97–1.0) 0.71 (0.240–1.4) 31 (11–62) 0.59 (0.520–0.670) 26 (23–29)
2010 3 0.033 (0.033–0.033) 1.1 (1.1–1.1) 1.7 (0.860–2.9) 58 (29–98) 1.1 (0.960–1.2) 37 (32–42)
2011 3 0.018 (0.018–0.018) 0.58 (0.57–0.58) 0.98 (0.330–2.0) 31 (11–63) 0.77 (0.680–0.870) 25 (22–28)
2012 3 0.031 (0.030–0.031) 0.94 (0.93–0.95) 1.1 (0.360–2.1) 33 (11–65) 0.85 (0.740–0.960) 26 (23–30)
Lebanon 1990 3 0.085 (0.046–0.130) 3.1 (1.7–5.0) 1.2 (0.460–2.3) 45 (17–87) 0.94 (0.820–1.1) 35 (31–39)
1995 3 0.067 (0.034–0.110) 2.2 (1.1–3.7) 1.1 (0.340–2.2) 35 (11–72) 0.88 (0.770–1.0) 29 (26–33)
2000 3 0.04 (0.020–0.069) 1.2 (0.61–2.1) 0.66 (0.220–1.3) 20 (6.9–41) 0.56 (0.490–0.630) 17 (15–20)
2005 4 0.046 (0.025–0.074) 1.2 (0.62–1.8) 0.61 (0.260–1.1) 15 (6.5–28) 0.45 (0.400–0.510) 11 (10–13)
2010 4 0.065 (0.035–0.110) 1.5 (0.80–2.4) 0.83 (0.370–1.5) 19 (8.6–34) 0.6 (0.530–0.680) 14 (12–16)
2011 4 0.073 (0.039–0.120) 1.6 (0.86–2.6) 0.91 (0.410–1.6) 20 (9.1–36) 0.67 (0.590–0.760) 15 (13–17)
2012 5 0.072 (0.038–0.120) 1.5 (0.81–2.5) 0.95 (0.390–1.7) 20 (8.5–37) 0.73 (0.640–0.830) 16 (14–18)
Libyan Arab 1990 4 0.44 (0.170–0.840) 10 (3.9–20) 3.6 (1.7–6.4) 86 (39–150) 1.7 (1.4–2.0) 40 (33–48)
Jamahiriya 1995 5 0.28 (0.120–0.500) 5.9 (2.6–11) 2.9 (1.3–5.1) 61 (27–108) 1.9 (1.5–2.3) 40 (33–48)
2000 5 0.27 (0.120–0.490) 5.3 (2.3–9.4) 3 (1.3–5.4) 57 (24–104) 2.1 (1.7–2.5) 40 (33–48)
2005 6 0.23 (0.120–0.390) 4.2 (2.1–6.9) 2.8 (1.1–5.5) 51 (19–98) 2.2 (1.9–2.6) 40 (34–46)
2010 6 0.32 (0.140–0.570) 5.4 (2.4–9.5) 3.5 (1.5–6.3) 58 (25–104) 2.4 (2.0–2.9) 40 (33–48)
2011 6 0.34 (0.140–0.610) 5.5 (2.3–10) 3.7 (1.8–6.4) 61 (29–105) 2.4 (2.0–2.9) 40 (33–48)
2012 6 0.42 (0.180–0.760) 6.8 (2.9–12) 4.1 (1.9–7.0) 66 (31–113) 2.5 (2.0–2.9) 40 (33–48)
Morocco 1990 25 6.2 (4.8–7.7) 25 (19–31) 57 (24–110) 232 (97–426) 36 (27–47) 147 (110–189)
1995 27 5.2 (3.7–6.8) 19 (14–25) 64 (30–110) 240 (112–415) 41 (33–49) 152 (124–182)
2000 29 4.3 (2.8–6.1) 15 (9.7–21) 46 (20–84) 161 (68–292) 33 (29–38) 117 (102–132)
2005 30 3.5 (2.0–5.5) 12 (6.8–18) 41 (17–75) 137 (57–251) 30 (26–34) 100 (88–113)
2010 32 3.1 (1.5–5.2) 9.8 (4.9–16) 44 (19–79) 138 (59–251) 32 (28–36) 100 (88–114)
2011 32 3 (1.5–5.2) 9.5 (4.6–16) 46 (20–82) 143 (62–257) 33 (29–37) 103 (90–117)
2012 33 3 (1.4–5.1) 9.2 (4.4–16) 46 (19–83) 140 (58–257) 33 (29–38) 103 (90–117)
Oman 1990 2 0.059 (<0.01–0.200) 3.2 (0.14–11) 0.8 (0.360–1.4) 44 (20–78) 0.55 (0.490–0.630) 31 (27–35)
1995 2 0.05 (<0.01–0.230) 2.3 (<0.1–10) 0.4 (0.140–0.790) 18 (6.5–37) 0.32 (0.280–0.360) 15 (13–17)
2000 2 0.041 (<0.01–0.230) 1.8 (0–10) 0.57 (0.280–0.960) 26 (13–44) 0.37 (0.320–0.420) 17 (15–19)
2005 3 0.035 (0–0.250) 1.4 (0–10) 0.36 (0.120–0.730) 14 (4.9–29) 0.3 (0.260–0.340) 12 (10–13)
2010 3 0.028 (0–0.260) 1 (0–9.4) 0.45 (0.170–0.870) 16 (6.1–31) 0.35 (0.310–0.400) 13 (11–14)
2011 3 0.029 (0–0.280) 0.97 (0–9.2) 0.51 (0.200–0.950) 17 (6.7–31) 0.39 (0.340–0.440) 13 (11–15)
2012 3 0.031 (0–0.300) 0.92 (0–9.0) 0.6 (0.260–1.1) 18 (7.8–33) 0.44 (0.380–0.500) 13 (12–15)
Pakistan 1990 111 80 (24–170) 72 (22–152) 650 (250–1 300) 589 (222–1 130) 260 (160–380) 231 (143–341)
1995 127 90 (32–180) 71 (25–139) 740 (330–1 300) 584 (262–1 030) 290 (240–350) 231 (189–278)
2000 144 99 (36–190) 69 (25–135) 820 (370–1 400) 573 (260–1 010) 330 (270–400) 231 (189–278)
2005 158 84 (33–160) 53 (21–101) 760 (380–1 300) 483 (239–810) 370 (300–440) 231 (189–278)
2010 173 64 (28–110) 37 (16–66) 670 (330–1 100) 389 (191–657) 400 (330–480) 231 (190–276)
2011 176 62 (27–110) 35 (15–63) 670 (330–1 100) 381 (185–647) 410 (340–490) 231 (190–276)
2012 179 62 (27–110) 34 (15–61) 670 (320–1 100) 376 (181–641) 410 (340–490) 231 (190–276)
Qatar 1990 < 1 0.031 (0.030–0.032) 6.5 (6.3–6.6) 0.28 (0.110–0.520) 59 (24–108) 0.21 (0.190–0.240) 44 (39–50)
1995 < 1 0.016 (0.016–0.017) 3.3 (3.1–3.4) 0.54 (0.260–0.910) 107 (52–182) 0.35 (0.310–0.400) 70 (61–79)
2000 < 1 <0.01 (<0.01–<0.01) 0.7 (0.62–0.78) 0.43 (0.180–0.780) 72 (30–132) 0.32 (0.280–0.360) 54 (47–61)
2005 < 1 <0.01 (<0.01–<0.01) 0.15 (0.12–0.17) 0.53 (0.230–0.950) 64 (28–115) 0.37 (0.330–0.420) 46 (40–52)
2010 2 <0.01 (<0.01–<0.01) 0.22 (0.15–0.30) 0.82 (0.290–1.6) 47 (17–92) 0.67 (0.580–0.750) 38 (33–43)
2011 2 <0.01 (<0.01–<0.01) 0.19 (0.12–0.27) 0.78 (0.260–1.6) 41 (14–82) 0.64 (0.560–0.720) 33 (29–38)
2012 2 <0.01 (<0.01–<0.01) 0.17 (0.10–0.25) 1.2 (0.560–2.1) 60 (27–105) 0.84 (0.730–0.950) 41 (36–46)
Saudi Arabia 1990 16 0.63 (0.061–1.9) 3.9 (0.37–11) 4 (1.8–7.0) 25 (11–43) 2.8 (2.4–3.1) 17 (15–19)
1995 19 0.71 (0.068–2.1) 3.8 (0.37–11) 3.9 (1.4–7.6) 21 (7.7–41) 3.1 (2.7–3.5) 17 (15–19)
2000 20 0.79 (0.075–2.3) 3.9 (0.37–11) 5.3 (2.2–9.7) 26 (11–48) 4 (3.5–4.5) 20 (17–22)
2005 25 0.95 (0.091–2.8) 3.9 (0.37–11) 5.1 (1.9–9.8) 21 (7.8–40) 4.1 (3.6–4.6) 16 (14–19)
2010 27 1.1 (0.100–3.1) 3.9 (0.37–11) 7.7 (3.7–13) 28 (13–48) 5.1 (4.5–5.8) 19 (17–21)
2011 28 1.1 (0.100–3.2) 3.9 (0.37–11) 6.1 (2.5–11) 22 (9.1–40) 4.5 (4.0–5.1) 16 (14–18)
2012 28 1.1 (0.110–3.2) 3.9 (0.39–11) 4.9 (1.6–10) 17 (5.5–36) 4.2 (3.7–4.8) 15 (13–17)
Somalia 1990 6 5.7 (1.7–12) 90 (27–190) 46 (17–89) 732 (272–1 410) 18 (11–27) 285 (176–421)
1995 6 5 (1.8–9.7) 79 (29–153) 42 (19–73) 663 (305–1 160) 18 (15–22) 285 (233–343)
2000 7 5 (1.9–9.7) 68 (26–131) 45 (21–76) 604 (291–1 030) 21 (17–25) 285 (233–343)
2005 8 4.8 (1.9–8.9) 56 (23–105) 45 (23–76) 537 (267–900) 24 (20–29) 285 (233–343)
2010 10 5.7 (2.3–11) 59 (24–111) 53 (27–89) 555 (279–925) 28 (23–33) 286 (236–340)
2011 10 6.1 (2.4–11) 61 (24–115) 56 (28–94) 566 (283–947) 28 (23–34) 286 (236–340)
2012 10 6.5 (2.5–12) 64 (25–120) 59 (29–99) 581 (287–975) 29 (24–35) 286 (236–340)
a
Rates are per 100 000 population.

210 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($(VWLPDWHVRIWKHEXUGHQRIGLVHDVHFDXVHGE\7%±
YEAR POPULATION
(MILLIONS)
NUMBER
(THOUSANDS) RATEaNUMBER
(THOUSANDS) RATEaNUMBER
(THOUSANDS) RATEa
MORTALITY (EXCLUDING HIV) PREVALENCE (INCLUDING HIV) INCIDENCE (INCLUDING HIV)
South Sudan 2011 10 3.1 (1.3–5.6) 30 (13–54) 28 (13–47) 268 (129–456) 15 (13–18) 146 (121–174)
2012 11 3.2 (1.4–5.8) 30 (13–54) 28 (13–47) 257 (124–437) 16 (13–19) 146 (121–174)
Sudan 1990 26 11 (4.4–22) 44 (17–84) 99 (48–170) 386 (185–659) 44 (36–52) 170 (140–203)
1995 30 9 (3.8–16) 30 (13–55) 89 (45–150) 296 (149–491) 47 (39–56) 158 (130–188)
2000 34 9.3 (4.0–17) 27 (12–49) 90 (45–150) 262 (132–436) 50 (41–59) 144 (119–172)
2005 40 9.3 (4.0–17) 24 (10–42) 90 (45–150) 226 (113–378) 53 (43–63) 133 (110–158)
2010 46 10 (4.3–18) 22 (9.4–40) 96 (48–160) 210 (105–350) 54 (45–65) 119 (98–142)
2011 36 8 (3.4–15) 22 (9.3–40) 76 (38–130) 209 (105–347) 42 (35–51) 117 (96–139)
2012 37 8 (3.3–15) 22 (9.0–40) 77 (39–130) 207 (104–345) 42 (35–51) 114 (94–136)
Syrian Arab 1990 12 0.97 (0.270–2.1) 7.8 (2.2–17) 11 (3.6–22) 86 (29–174) 7.5 (5.3–10) 61 (43–82)
Republic 1995 14 0.85 (0.370–1.5) 5.9 (2.6–11) 9.3 (3.9–17) 65 (27–119) 6.6 (5.4–7.9) 46 (38–55)
2000 16 0.56 (0.280–0.930) 3.4 (1.7–5.7) 7 (2.5–14) 43 (15–85) 5.7 (4.9–6.6) 35 (30–40)
2005 18 0.47 (0.220–0.810) 2.6 (1.2–4.4) 5.9 (2.1–12) 33 (11–65) 4.8 (4.0–5.6) 26 (22–31)
2010 22 0.47 (0.210–0.830) 2.2 (0.99–3.9) 5.6 (2.1–11) 26 (9.8–50) 4.3 (3.5–5.1) 20 (16–24)
2011 22 0.47 (0.210–0.830) 2.2 (0.98–3.8) 5.5 (2.1–10) 25 (9.8–47) 4.1 (3.4–4.9) 19 (16–22)
2012 22 0.46 (0.210–0.820) 2.1 (0.96–3.7) 5.3 (2.1–9.9) 24 (9.7–45) 3.9 (3.2–4.6) 18 (15–21)
Tunisia 1990 8 0.24 (0.130–0.370) 2.9 (1.6–4.6) 3.2 (1.3–5.8) 39 (16–72) 2.3 (2.0–2.6) 29 (25–32)
1995 9 0.3 (0.160–0.470) 3.3 (1.8–5.3) 3.9 (1.7–6.9) 43 (18–77) 2.7 (2.4–3.1) 31 (27–35)
2000 10 0.26 (0.140–0.410) 2.7 (1.5–4.3) 3.3 (1.4–6.0) 35 (15–63) 2.4 (2.1–2.7) 25 (22–28)
2005 10 0.25 (0.140–0.400) 2.5 (1.4–4.0) 3.3 (1.4–5.9) 33 (14–59) 2.4 (2.1–2.7) 23 (21–27)
2010 11 0.31 (0.170–0.500) 2.9 (1.6–4.7) 4.1 (1.7–7.6) 39 (16–71) 3 (2.6–3.4) 28 (25–32)
2011 11 0.33 (0.180–0.520) 3.1 (1.7–4.8) 4.4 (1.8–8.1) 41 (17–75) 3.2 (2.8–3.6) 30 (26–34)
2012 11 0.32 (0.170–0.500) 2.9 (1.6–4.6) 4.5 (1.7–8.5) 41 (16–78) 3.4 (3.0–3.8) 31 (27–35)
United Arab 1990 2 0.017 (0–0.110) 0.95 (0–6.1) 0.39 (0.170–0.710) 22 (9.2–39) 0.22 (0.160–0.280) 12 (8.7–16)
Emirates 1995 2 0.022 (0–0.140) 0.95 (0–6.1) 0.51 (0.220–0.910) 22 (9.2–39) 0.28 (0.200–0.370) 12 (8.7–16)
2000 3 0.029 (0–0.190) 0.95 (0–6.1) 0.65 (0.280–1.2) 22 (9.3–39) 0.36 (0.260–0.480) 12 (8.7–16)
2005 4 0.02 (0–0.150) 0.49 (0–3.6) 0.44 (0.190–0.800) 11 (4.5–19) 0.21 (0.150–0.270) 5 (3.6–6.5)
2010 8 0.022 (0–0.150) 0.26 (0–1.8) 0.52 (0.230–0.930) 6.2 (2.7–11) 0.26 (0.190–0.340) 3.1 (2.3–4.1)
2011 9 0.015 (0–0.097) 0.17 (0–1.1) 0.37 (0.160–0.660) 4.2 (1.8–7.4) 0.21 (0.150–0.270) 2.3 (1.7–3.0)
2012 9 <0.01 (<0.01–0.045) 0.1 (0–0.49) 0.22 (0.077–0.440) 2.4 (0.84–4.8) 0.16 (0.120–0.210) 1.7 (1.2–2.3)
West Bank 1990 2 <0.01 (<0.01–<0.01) 0.45 (0.43–0.46) 0.18 (0.091–0.320) 8.6 (4.4–15) 0.12 (0.110–0.140) 6 (5.2–6.8)
and Gaza Strip 1995 3 0.035 (0.034–0.036) 1.3 (1.3–1.4) 0.27 (0.230–0.800) 10 (8.7–31) 0.22 (0.200–0.250) 8.6 (7.5–9.7)
2000 3 0.018 (0.018–0.019) 0.57 (0.56–0.58) 0.45 (0.340–1.3) 14 (11–41) 0.33 (0.290–0.370) 10 (9.0–12)
2005 4 0.012 (0.012–0.012) 0.34 (0.33–0.35) 0.29 (0.240–0.910) 8.1 (6.8–26) 0.23 (0.200–0.260) 6.5 (5.7–7.3)
2010 4 <0.01 (<0.01–<0.01) 0.23 (0.23–0.24) 0.25 (0.240–0.870) 6.1 (6.0–22) 0.21 (0.190–0.240) 5.3 (4.6–6.0)
2011 4 <0.01 (<0.01–<0.01) 0.23 (0.22–0.23) 0.34 (0.290–1.1) 8.3 (7.1–26) 0.26 (0.230–0.290) 6.3 (5.5–7.1)
2012 4 <0.01 (<0.01–<0.01) 0.23 (0.22–0.23) 0.47 (0.370–1.4) 11 (8.7–32) 0.32 (0.280–0.360) 7.6 (6.7–8.6)
Yemen 1990 12 3.8 (1.1–8.2) 32 (9.3–70) 35 (13–66) 293 (112–558) 16 (10–24) 137 (85–202)
1995 15 3.5 (1.5–6.3) 23 (9.8–42) 36 (18–60) 239 (118–401) 21 (17–25) 137 (112–165)
2000 18 3.3 (1.4–6.0) 19 (8.1–34) 35 (17–59) 198 (97–335) 20 (17–24) 116 (94–139)
2005 20 2.8 (1.2–5.2) 14 (5.9–26) 29 (14–48) 142 (71–239) 16 (13–19) 81 (66–97)
2010 23 1.4 (0.640–2.5) 6.2 (2.8–11) 17 (7.5–29) 73 (33–129) 11 (9.2–13) 49 (40–58)
2011 23 1.4 (0.630–2.5) 6 (2.7–11) 17 (7.4–30) 72 (32–129) 11 (9.4–14) 49 (40–58)
2012 24 1.3 (0.600–2.4) 5.6 (2.5–9.9) 17 (7.1–30) 70 (30–127) 12 (9.6–14) 49 (40–58)
a Rates are per 100 000 population.
211 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
EASTERN MEDITERRANEAN REGION
7$%/($,QFLGHQFHQRWLILFDWLRQDQGFDVHGHWHFWLRQUDWHVDOOIRUPV±
INCIDENCE (INCLUDING HIV) INCIDENCE HIV-POSITIVE NOTIFIED NEW AND RELAPSE
b
CASE DETECTION
YEAR POPULATION
(MILLIONS)
NUMBER
(THOUSANDS) RATE
a
NUMBER
(THOUSANDS) RATE
a
NUMBER RATE
a
PERCENT
Afghanistan 1990 12 22 (14–33) 189 (117–279) 0.041 (0.025–0.060) 0.4 (0.22–0.52) 4 332 37 20 (13–32)
1995 18 33 (27–40) 189 (155–227) 0.072 (0.040–0.11) 0.4 (0.23–0.65)
2000 21 39 (32–47) 189 (155–227) 0.1 (0.058–0.16) 0.5 (0.28–0.77) 7 107 35 18 (15–22)
2005 25 47 (38–56) 189 (155–227) 0.16 (0.090–0.24) 0.6 (0.36–0.96) 21 844 88 46 (39–57)
2010 28 54 (44–64) 189 (156–225) 0.25 (0.15–0.38) 0.9 (0.54–1.3) 28 029 99 52 (44–63)
2011 29 55 (45–66) 189 (156–225) 0.28 (0.17–0.41) 1 (0.58–1.4) 27 983 96 51 (43–62)
2012 30 56 (47–67) 189 (156–226) 0.31 (0.19–0.46) 1 (0.63–1.5) 29 381 99 52 (44–63)
Bahrain 1990 < 1 0.13 (0.120–0.150) 27 (24–31) 117 24 87 (77–99)
1995 < 1 0.049 (0.043–0.056) 8.8 (7.7–9.9) 43 7.6 87 (77–99)
2000 < 1 0.24 (0.210–0.270) 36 (31–40) 207 31 87 (77–99)
2005 < 1 0.32 (0.280–0.360) 37 (32–41) 280 32 87 (77–99)
2010 1 0.28 (0.250–0.320) 23 (20–26) 0.011 (<0.01–0.022) 0.8 (0.28–1.7) 246 20 87 (77–99)
2011 1 0.26 (0.230–0.290) 20 (18–23) 0.012 (<0.01–0.023) 0.9 (0.33–1.8) 225 17 87 (77–99)
2012 1 0.26 (0.230–0.290) 20 (17–22) 0.012 (<0.01–0.025) 0.9 (0.25–1.9) 225 17 87 (77–99)
Djibouti 1990 < 1 3.7 (2.3–5.3) 619 (395–893) 0.082 (0.052–0.12) 14 (8.9–20) 2 100 356 57 (40–90)
1995 < 1 4.1 (3.4–4.9) 619 (506–744) 0.43 (0.35–0.52) 65 (53–78)
2000 < 1 4.5 (3.8–5.2) 619 (528–718) 0.74 (0.63–0.86) 102 (87–118) 3 971 549 89 (76–100)
2005 < 1 4.8 (3.9–5.8) 619 (506–744) 0.74 (0.61–0.89) 96 (78–115) 3 109 400 65 (54–79)
2010 < 1 5.2 (4.3–6.2) 620 (512–738) 0.6 (0.49–0.71) 72 (59–85) 4 172 500 81 (68–98)
2011 < 1 5.2 (4.3–6.2) 620 (512–738) 0.57 (0.47–0.68) 68 (56–80) 3 686 435 70 (59–85)
2012 < 1 5.3 (4.4–6.3) 620 (512–738) 0.54 (0.45–0.64) 63 (52–75) 3 474 404 65 (55–79)
Egypt 1990 56 19 (16–23) 34 (29–40) <0.01 (<0.01–0.012) <0.1 (<0.1–<0.1) 2 142 3.8 11 (9.4–13)
1995 61 19 (16–23) 32 (27–37) 0.029 (0.024–0.034) <0.1 (<0.1–<0.1) 11 145 18 58 (49–68)
2000 66 17 (14–20) 26 (22–30) 0.1 (0.085–0.12) 0.2 (0.13–0.18) 10 762 16 63 (54–75)
2005 72 15 (13–18) 21 (18–25) 0.18 (0.15–0.21) 0.3 (0.21–0.29) 11 446 16 75 (64–89)
2010 78 14 (12–16) 18 (15–21) 0.14 (0.12–0.17) 0.2 (0.16–0.21) 9 260 12 66 (57–78)
2011 79 14 (12–16) 17 (15–20) 0.14 (0.12–0.16) 0.2 (0.15–0.20) 8 974 11 65 (56–76)
2012 81 14 (12–16) 17 (14–19) 0.13 (0.11–0.16) 0.2 (0.14–0.19) 8 453 10 62 (54–73)
Iran (Islamic 1990 56 18 (13–23) 31 (23–41) 0.011 (<0.01–0.014) <0.1 (<0.1–<0.1) 9 255 16 53 (40–72)
Republic of) 1995 60 21 (16–28) 35 (26–46) 0.051 (0.037–0.067) <0.1 (<0.1–0.11) 15 936 26 75 (57–100)
2000 66 17 (12–22) 26 (19–34) 0.15 (0.11–0.20) 0.2 (0.16–0.30) 11 850 18 70 (53–96)
2005 70 14 (10–19) 20 (15–27) 0.21 (0.15–0.27) 0.3 (0.21–0.39) 9 212 13 65 (49–89)
2010 74 15 (11–19) 20 (14–26) 0.26 (0.19–0.34) 0.4 (0.25–0.46) 10 362 14 70 (53–96)
2011 75 16 (11–21) 21 (15–27) 0.28 (0.20–0.37) 0.4 (0.27–0.49) 10 980 15 70 (53–96)
2012 76 16 (11–21) 21 (15–28) 0.29 (0.21–0.39) 0.4 (0.28–0.51) 11 042 14 70 (52–97)
Iraq 1990 18 9.5 (8.3–11) 54 (47–62) 0 (0–0) 0 (0–0) 14 735 84 160 (140–180)
1995 20 11 (9.4–12) 53 (46–60) 0 (0–0) 0 (0–0) 9 697 48 90 (80–100)
2000 24 12 (10–14) 50 (44–57) 0 (0–0) 0 (0–0) 9 697 41 81 (71–93)
2005 27 13 (11–15) 48 (42–54) 0 (0–0) 0 (0–0) 9 454 35 72 (64–82)
2010 31 14 (12–16) 45 (40–52) <0.01 (0–0.010) <0.1 (0–<0.1) 9 707 31 69 (61–79)
2011 32 14 (13–16) 45 (39–51) 0 (0–0) 0 (0–0) 8 837 28 62 (54–71)
2012 33 15 (13–17) 45 (39–51) 0 (0–0) 0 (0–0) 8 664 26 59 (52–68)
Jordan 1990 3 0.48 (0.420–0.550) 14 (13–16) 439 13 91 (80–100)
1995 4 0.51 (0.450–0.580) 12 (10–13) 498 12 97 (86–110)
2000 5 0.38 (0.340–0.440) 8.1 (7.1–9.1) 306 6.4 80 (70–91)
2005 5 0.38 (0.330–0.430) 7.2 (6.3–8.1) 367 7 98 (86–110)
2010 6 0.41 (0.360–0.460) 6.3 (5.5–7.1) 338 5.2 83 (74–95)
2011 7 0.4 (0.350–0.460) 6 (5.2–6.8) 328 4.9 81 (72–93)
2012 7 0.4 (0.360–0.460) 5.8 (5.1–6.5) <0.01 (0–<0.01) 0 (0–0) 331 4.7 82 (72–93)
Kuwait 1990 2 0.32 (0.280–0.360) 15 (14–18) 277 13 87 (77–99)
1995 2 0.39 (0.340–0.440) 24 (21–28) 336 21 87 (77–99)
2000 2 0.59 (0.520–0.670) 31 (27–35) <0.01 (<0.01–<0.01) 0.2 (<0.1–0.49) 513 27 87 (77–99)
2005 2 0.59 (0.520–0.670) 26 (23–29) <0.01 (<0.01–<0.01) 0.2 (<0.1–0.41) 517 23 87 (77–99)
2010 3 1.1 (0.960–1.2) 37 (32–42) <0.01 (<0.01–<0.01) 0.1 (<0.1–0.31) 957 32 87 (77–99)
2011 3 0.77 (0.680–0.870) 25 (22–28) 672 22 87 (77–99)
2012 3 0.85 (0.740–0.960) 26 (23–30) <0.01 (<0.01–<0.01) <0.1 (0–<0.1) 737 23 87 (77–99)
Lebanon 1990 3 0.94 (0.820–1.1) 35 (31–39) <0.01 (<0.01–<0.01) 0.3 (0.23–0.30)
1995 3 0.88 (0.770–1.0) 29 (26–33) 0.012 (0.011–0.014) 0.4 (0.35–0.45) 983 32 110 (98–130)
2000 3 0.56 (0.490–0.630) 17 (15–20) 0.014 (0.012–0.015) 0.4 (0.37–0.48) 571 18 100 (90–120)
2005 4 0.45 (0.400–0.510) 11 (10–13) 0.017 (0.015–0.019) 0.4 (0.37–0.47) 391 9.8 86 (76–98)
2010 4 0.6 (0.530–0.680) 14 (12–16) 0.031 (0.027–0.035) 0.7 (0.62–0.80) 513 12 85 (75–97)
2011 4 0.67 (0.590–0.760) 15 (13–17) 0.036 (0.032–0.041) 0.8 (0.71–0.92) 496 11 74 (65–84)
2012 5 0.73 (0.640–0.830) 16 (14–18) 0.041 (0.036–0.047) 0.9 (0.77–1.0) 630 14 86 (76–99)
Libyan Arab 1990 4 1.7 (1.4–2.0) 40 (33–48) 442 10 26 (22–32)
Jamahiriya 1995 5 1.9 (1.5–2.3) 40 (33–48) 1 440 30 76 (63–93)
2000 5 2.1 (1.7–2.5) 40 (33–48) 1 341 26 65 (54–79)
2005 6 2.2 (1.9–2.6) 40 (34–46) 2 098 38 94 (81–110)
2010 6 2.4 (2.0–2.9) 40 (33–48)
2011 6 2.4 (2.0–2.9) 40 (33–48) 0.21 (0.16–0.26) 3.4 (2.6–4.3) 1 518 25 62 (52–76)
2012 6 2.5 (2.0–2.9) 40 (33–48) 1 549 25 63 (53–77)
Morocco 1990 25 36 (27–47) 147 (110–189) 0.025 (0.019–0.033) 0.1 (<0.1–0.13) 27 658 112 76 (59–100)
1995 27 41 (33–49) 152 (124–182) 0.094 (0.076–0.11) 0.4 (0.28–0.42) 29 829 111 73 (61–90)
2000 29 33 (29–38) 117 (102–132) 0.19 (0.16–0.21) 0.7 (0.57–0.74) 28 852 100 86 (76–98)
2005 30 30 (26–34) 100 (88–113) 0.29 (0.26–0.33) 1 (0.85–1.1) 26 269 87 87 (77–99)
2010 32 32 (28–36) 100 (88–114) 0.5 (0.43–0.56) 1.6 (1.4–1.8) 28 359 90 89 (79–100)
2011 32 33 (29–37) 103 (90–117) 0.55 (0.48–0.62) 1.7 (1.5–1.9) 28 640 89 87 (77–99)
2012 33 33 (29–38) 103 (90–117) 0.59 (0.51–0.67) 1.8 (1.6–2.0) 28 635 88 86 (75–98)
Oman 1990 2 0.55 (0.490–0.630) 31 (27–35) <0.01 (<0.01–<0.01) 0.1 (0.10–0.14) 482 27 87 (77–99)
1995 2 0.32 (0.280–0.360) 15 (13–17) <0.01 (<0.01–<0.01) 0.1 (<0.1–0.12) 276 13 87 (77–99)
2000 2 0.37 (0.320–0.420) 17 (15–19) <0.01 (<0.01–<0.01) 0.1 (<0.1–0.11) 321 15 87 (77–99)
2005 3 0.3 (0.260–0.340) 12 (10–13) <0.01 (<0.01–<0.01) 0.1 (<0.1–0.12) 261 10 87 (77–99)
2010 3 0.35 (0.310–0.400) 13 (11–14) <0.01 (<0.01–<0.01) 0.3 (0.26–0.34) 308 11 87 (77–99)
2011 3 0.39 (0.340–0.440) 13 (11–15) 0.011 (<0.01–0.012) 0.4 (0.31–0.41) 337 11 87 (77–99)
2012 3 0.44 (0.380–0.500) 13 (12–15) 0.014 (0.013–0.016) 0.4 (0.38–0.49) 382 12 87 (77–99)
Pakistan 1990 111 260 (160–380) 231 (143–341) 0.026 (0.016–0.038) <0.1 (<0.1–<0.1) 156 759 141 61 (41–99)
1995 127 290 (240–350) 231 (189–278) 0.059 (0.048–0.070) <0.1 (<0.1–<0.1) 13 142 10 4.5 (3.7–5.5)
2000 144 330 (270–400) 231 (189–278) 0.23 (0.19–0.28) 0.2 (0.13–0.19) 11 050 7.7 3.3 (2.8–4.1)
2005 158 370 (300–440) 231 (189–278) 0.8 (0.65–0.98) 0.5 (0.41–0.62) 142 017 90 39 (32–48)
2010 173 400 (330–480) 231 (190–276) 2.4 (2.0–2.9) 1.4 (1.1–1.7) 264 235 153 66 (55–80)
2011 176 410 (340–490) 231 (190–276) 3.1 (2.5–3.7) 1.7 (1.4–2.1) 264 934 150 65 (54–79)
2012 179 410 (340–490) 231 (190–276) 3.8 (3.1–4.6) 2.1 (1.7–2.6) 267 475 149 65 (54–78)
Qatar 1990 < 1 0.21 (0.190–0.240) 44 (39–50) 184 39 87 (77–99)
1995 < 1 0.35 (0.310–0.400) 70 (61–79) 304 61 87 (77–99)
2000 < 1 0.32 (0.280–0.360) 54 (47–61) 279 47 87 (77–99)
2005 < 1 0.37 (0.330–0.420) 46 (40–52) 325 40 87 (77–99)
2010 2 0.67 (0.580–0.750) 38 (33–43) 580 33 87 (77–99)
2011 2 0.64 (0.560–0.720) 33 (29–38) <0.01 (<0.01–<0.01) <0.1 (<0.1–0.12) 553 29 87 (77–99)
2012 2 0.84 (0.730–0.950) 41 (36–46) <0.01 (<0.01–<0.01) <0.1 (0–0.12) 728 36 87 (77–99)
Saudi Arabia 1990 16 2.8 (2.4–3.1) 17 (15–19) 2 415 15 87 (77–99)
1995 19 3.1 (2.7–3.5) 17 (15–19)
2000 20 4 (3.5–4.5) 20 (17–22) 3 452 17 87 (77–99)
2005 25 4.1 (3.6–4.6) 16 (14–19) 3 539 14 87 (77–99)
2010 27 5.1 (4.5–5.8) 19 (17–21) 0.12 (0.092–0.15) 0.4 (0.34–0.56) 4 465 16 87 (77–99)
2011 28 4.5 (4.0–5.1) 16 (14–18) 0.1 (0.077–0.13) 0.4 (0.28–0.46) 3 932 14 87 (77–99)
2012 28 4.2 (3.7–4.8) 15 (13–17) 3 690 13 87 (77–99)
Somalia 1990 6 18 (11–27) 285 (176–421) 0.3 (0.19–0.44) 4.8 (2.9–7.0)
1995 6 18 (15–22) 285 (233–343) 0.56 (0.46–0.68) 8.9 (7.3–11) 2 504 39 14 (12–17)
2000 7 21 (17–25) 285 (233–343) 0.77 (0.63–0.93) 10 (8.6–13) 5 686 77 27 (22–33)
2005 8 24 (20–29) 285 (233–343) 0.85 (0.70–1.0) 10 (8.2–12) 12 904 152 53 (44–65)
2010 10 28 (23–33) 286 (236–340) 0.85 (0.70–1.0) 8.9 (7.3–11) 10 139 105 37 (31–45)
2011 10 28 (23–34) 286 (236–340) 0.85 (0.70–1.0) 8.6 (7.1–10) 11 653 118 41 (35–50)
2012 10 29 (24–35) 286 (236–340) 0.85 (0.70–1.0) 8.3 (6.9–9.9) 11 975 117 41 (34–50)
a
Rates are per 100 000 population.
b
NOTIFIED NEW AND RELAPSE includes cases for which the treatment history is unknown.

212 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($,QFLGHQFHQRWLILFDWLRQDQGFDVHGHWHFWLRQUDWHVDOOIRUPV±
INCIDENCE (INCLUDING HIV) INCIDENCE HIV-POSITIVE NOTIFIED NEW AND RELAPSE
b
CASE DETECTION
YEAR POPULATION
(MILLIONS)
NUMBER
(THOUSANDS) RATEaNUMBER
(THOUSANDS) RATEaNUMBER RATEaPERCENT
South Sudan 2011 10 15 (13–18) 146 (121–174) 7 217 70 48 (40–58)
2012 11 16 (13–19) 146 (121–174) 8 403 78 53 (45–64)
Sudan 1990 26 44 (36–52) 170 (140–203) 0.4 (0.33–0.48) 1.6 (1.3–1.9) 212 0.82 0.48 (0.41–0.59)
1995 30 47 (39–56) 158 (130–188) 1.5 (1.2–1.8) 5 (4.1–6.0) 14 320 48 30 (25–37)
2000 34 50 (41–59) 144 (119–172) 3.5 (2.9–4.2) 10 (8.5–12) 24 807 72 50 (42–61)
2005 40 53 (43–63) 133 (110–158) 5.2 (4.3–6.2) 13 (11–16) 27 562 70 52 (44–64)
2010 46 54 (45–65) 119 (98–142) 5.6 (4.6–6.7) 12 (10–15) 26 131 57 48 (40–58)
2011 36 42 (35–51) 117 (96–139) 4.4 (3.6–5.3) 12 (10–14) 19 348 53 46 (38–55)
2012 37 42 (35–51) 114 (94–136) 4.3 (3.5–5.1) 12 (9.5–14) 18 775 50 44 (37–54)
Syrian Arab 1990 12 7.5 (5.3–10) 61 (43–82) 6 018 48 80 (59–110)
Republic 1995 14 6.6 (5.4–7.9) 46 (38–55) 4 404 31 67 (56–82)
2000 16 5.7 (4.9–6.6) 35 (30–40) 5 090 31 89 (77–100)
2005 18 4.8 (4.0–5.6) 26 (22–31) 4 310 24 90 (77–110)
2010 22 4.3 (3.5–5.1) 20 (16–24) 3 666 17 86 (72–100)
2011 22 4.1 (3.4–4.9) 19 (16–22) 3 620 17 88 (74–110)
2012 22 3.9 (3.2–4.6) 18 (15–21) 3 003 14 77 (65–93)
Tunisia 1990 8 2.3 (2.0–2.6) 29 (25–32) <0.01 (<0.01–<0.01) 0 (0–0) 2 054 25 89 (78–100)
1995 9 2.7 (2.4–3.1) 31 (27–35) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 2 383 27 87 (77–99)
2000 10 2.4 (2.1–2.7) 25 (22–28) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 2 038 21 86 (76–98)
2005 10 2.4 (2.1–2.7) 23 (21–27) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 2 079 21 88 (78–100)
2010 11 3 (2.6–3.4) 28 (25–32) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 2 368 22 79 (70–90)
2011 11 3.2 (2.8–3.6) 30 (26–34) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 3 015 28 94 (83–110)
2012 11 3.4 (3.0–3.8) 31 (27–35) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 3 239 30 96 (84–110)
United Arab 1990 2 0.22 (0.160–0.280) 12 (8.7–16) 285 16 130 (100–180)
Emirates 1995 2 0.28 (0.200–0.370) 12 (8.7–16)
2000 3 0.36 (0.260–0.480) 12 (8.7–16) 115 3.8 32 (24–44)
2005 4 0.21 (0.150–0.270) 5 (3.6–6.5) 103 2.5 50 (38–69)
2010 8 0.26 (0.190–0.340) 3.1 (2.3–4.1) 0.012 (<0.01–0.030) 0.2 (<0.1–0.35) 131 1.6 50 (38–69)
2011 9 0.21 (0.150–0.270) 2.3 (1.7–3.0) <0.01 (<0.01–0.021) <0.1 (<0.1–0.23) 103 1.2 50 (38–69)
2012 9 0.16 (0.120–0.210) 1.7 (1.2–2.3) 79 0.86 50 (38–69)
West Bank 1990 2 0.12 (0.110–0.140) 6 (5.2–6.8) 64 3.1 51 (45–59)
and Gaza Strip 1995 3 0.22 (0.200–0.250) 8.6 (7.5–9.7) 77 3 35 (30–39)
2000 3 0.33 (0.290–0.370) 10 (9.0–12) 82 2.6 25 (22–28)
2005 4 0.23 (0.200–0.260) 6.5 (5.7–7.3) 28 0.79 12 (11–14)
2010 4 0.21 (0.190–0.240) 5.3 (4.6–6.0) 31 0.77 15 (13–17)
2011 4 0.26 (0.230–0.290) 6.3 (5.5–7.1) 32 0.78 12 (11–14)
2012 4 0.32 (0.280–0.360) 7.6 (6.7–8.6) 32 0.76 10 (8.8–11)
Yemen 1990 12 16 (10–24) 137 (85–202) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 4 650 39 29 (20–47)
1995 15 21 (17–25) 137 (112–165) 0.031 (0.022–0.042) 0.2 (0.14–0.28) 14 428 96 70 (58–86)
2000 18 20 (17–24) 116 (94–139) 0.11 (0.074–0.14) 0.6 (0.42–0.81) 13 651 78 67 (56–83)
2005 20 16 (13–19) 81 (66–97) 0.18 (0.12–0.25) 0.9 (0.58–1.3) 9 063 45 56 (46–68)
2010 23 11 (9.2–13) 49 (40–58) 0.15 (0.093–0.21) 0.7 (0.41–0.93) 8 916 39 80 (67–97)
2011 23 11 (9.4–14) 49 (40–58) 0.15 (0.096–0.22) 0.7 (0.41–0.94) 8 636 37 76 (64–92)
2012 24 12 (9.6–14) 49 (40–58) 0.16 (0.098–0.23) 0.7 (0.41–0.95) 9 867 41 85 (71–100)
a Rates are per 100 000 population.
b NOTIFIED NEW AND RELAPSE includes cases for which the treatment history is unknown.

213 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
EASTERN MEDITERRANEAN REGION
7$%/($&DVHQRWLILFDWLRQV±
NEW CASES
YEAR SMEAR-
POSITIVE
SMEAR-NEGATIVE/
UNKNOWN
EXTRA-
PULMONARY OTHER RELAPSE RE-TREAT EXCL.
RELAPSE
TOTAL
RETREAT
HISTORY
UNKNOWN
NEW AND
RELAPSE
b
% SMEAR-
POS AMONG
NEW PULM
NEW AND RELAPSE
NOTIFICATION RATE
a
1990–2012
Afghanistan 1990 4 332 –
1995 –
2000 7 107 2 892 2 358 1 620 237 237 55
2005 21 844 9 949 6 085 4 954 856 856 62
2010 28 029 12 947 7 085 6 248 633 1 116 209 1 325 65
2011 27 983 13 789 6 155 6 286 623 1 130 184 1 314 69
• 37 99 • 2012 29 381 13 319 7 405 6 906 702 1 049 197 1 246 64
Bahrain 1990 117 –
1995 43 17 14 85 0 0 55
2000 207 23 16 8 59
2005 280 101 72 107 0 0 0 0 0 58
2010 246 90 58 98 0 0 0 0 0 61
2011 225 89 47 89 0 0 0 0 0 65
• 24 17 • 2012 225 101 47 77 0 0 0 0 0 68
Djibouti 1990 2 100 –
1995 –
2000 3 971 1 391 518 1 875 184 184 73
2005 3 109 1 120 739 1 058 0 192 61 253 0 60
2010 4 172 1 181 538 2 253 0 200 19 219 0 69
2011 3 686 1 336 569 1 587 0 194 37 231 0 70
• 356 404 • 2012 3 474 1 170 547 1 567 190 72 262 68
Egypt 1990 2 142 –
1995 11 145 4 229 9 204 4 684 753 753 31
2000 10 762 4 606 2 693 2 843 620 620 63
2005 11 446 5 217 2 617 3 163 0 449 289 738 0 67
2010 9 260 4 679 1 158 3 048 0 375 328 703 0 80
2011 8 974 4 508 1 055 3 074 0 337 333 670 0 81
• 4 10 • 2012 8 453 4 295 937 2 915 0 306 300 606 0 82
Iran (Islamic 1990 9 255 –
Republic of) 1995 15 936 5 347 6 432 3 779 477 477 45
2000 11 850 5 361 2 642 3 442 405 405 67
2005 9 212 4 581 1 807 2 530 274 154 428 20 72
2010 10 362 5 188 1 985 2 869 0 320 440 760 0 72
2011 10 980 5 539 1 980 3 076 0 385 515 900 0 74
• 16 14 • 2012 11 042 5 409 2 191 3 105 0 337 441 778 0 71
Iraq 1990 14 735 1 587 12 394 754 11
1995 9 697 3 194 13 962 1 367 68 68 19
2000 9 697 3 194 3 188 2 753 562 562 50
2005 9 454 3 096 2 887 2 703 768 768 52
2010 9 707 3 618 2 693 3 009 0 387 390 777 0 57
2011 8 837 3 059 2 463 2 957 0 358 411 769 0 55
• 84 26 • 2012 8 664 2 760 2 315 3 261 0 328 435 763 0 54
Jordan 1990 439 –
1995 498 187 210 101 6 6 47
2000 306 89 69 145 3 3 56
2005 367 86 76 187 12 6 4 10 0 53
2010 338 117 69 150 0 2 16 18 0 63
2011 328 103 81 128 0 2 16 18 14 56
• 13 5 • 2012 331 85 73 172 0 1 18 19 0 54
Kuwait 1990 277 –
1995 336 175 42 115 0 4 0 4 0 81
2000 513 180 89 244 0 0 0 0 0 67
2005 517 187 95 234 0 1 0 1 0 66
2010 957 385 163 407 0 2 0 2 0 70
2011 672 222 141 309 0 0 0 0 0 61
• 13 23 • 2012 737 328 140 269 0 0 0 0 0 70
Lebanon 1990 –
1995 983 197 528 255 3 3 27
2000 571 202 149 214 6 6 58
2005 391 131 75 181 0 4 0 4 0 64
2010 513 194 99 210 0 10 2 12 0 66
2011 496 188 101 206 1 1 65
• 0 14 • 2012 630 240 131 250 0 9 0 9 0 65
Libyan Arab 1990 442 –
Jamahiriya 1995 1 440 626 814 –
2000 1 341 607 82 652 88
2005 2 098 860 474 762 2 269 271 64
2010 –
2011 1 518 731 305 462 0 20 27 47 71
• 10 25 • 2012 1 549 644 372 533 0 63
Morocco 1990 27 658 –
1995 29 829 14 171 4 095 11 563 78
2000 28 852 12 872 2 934 13 046 81
2005 26 269 12 757 2 142 11 370 0 86
2010 28 359 12 239 2 174 12 730 0 1 216 429 1 645 0 85
2011 28 640 11 822 2 272 13 331 0 1 215 1 130 2 345 0 84
• 112 88 • 2012 28 635 11 572 2 343 13 522 0 1 198 764 1 962 0 83
Oman 1990 482 –
1995 276 135 60 81 0 0 69
2000 321 164 37 112 8 8 82
2005 261 131 37 89 4 4 78
2010 308 152 28 124 0 4 5 9 0 84
2011 337 180 32 122 0 3 0 3 0 85
• 27 12 • 2012 382 205 39 131 0 7 1 8 0 84
Pakistan 1990 156 759 –
1995 13 142 2 578 3 806 3 037 184 184 40
2000 11 050 3 285 5 578 1 846 341 341 37
2005 142 017 48 220 68 337 22 789 2 671 2 754 5 425 41
2010 264 235 104 263 105 623 45 443 0 5 870 5 055 10 925 3 036 50
2011 264 934 105 733 103 824 45 537 0 5 947 5 460 11 407 3 893 50
• 141 149 • 2012 267 475 110 545 109 425 41 410 0 6 095 5 622 11 717 0 50
Qatar 1990 184 –
1995 304 60 135 109 1 1 31
2000 279 53 98 128 0 0 35
2005 325 96 73 156 0 0 0 57
2010 580 223 101 256 0 0 0 0 0 69
2011 553 197 120 236 0 62
• 39 36 • 2012 728 180 331 217 0 0 0 0 0 35
Saudi Arabia 1990 2 415 –
1995 –
2000 3 452 1 595 722 1 023 112 112 69
2005 3 539 1 722 545 1 067 0 205 205 76
2010 4 465 2 302 687 1 311 0 122 84 206 43 77
2011 3 932 2 055 586 1 227 64 83 147 78
• 15 13 • 2012 3 690 2 028 549 1 022 91 143 234 79
a Rates are per 100 000 population.
b NEW AND RELAPSE includes cases for which the treatment history is unknown.

214 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($&DVHQRWLILFDWLRQV±
NEW CASES
YEAR SMEAR-
POSITIVE
SMEAR-NEGATIVE/
UNKNOWN
EXTRA-
PULMONARY OTHER RELAPSE RE-TREAT EXCL.
RELAPSE
TOTAL
RETREAT
HISTORY
UNKNOWN
NEW AND
RELAPSE
b
% SMEAR-
POS AMONG
NEW PULM
NEW AND RELAPSE
NOTIFICATION RATE
a
1990–2012
Somalia 1990 –
1995 2 504 1 572 692 318 134 134 69
2000 5 686 3 776 837 722 351 351 82
2005 12 904 7 068 3 168 2 258 0 410 102 512 0 69
2010 10 139 5 225 2 654 1 885 0 375 330 705 0 66
2011 11 653 5 884 3 159 2 261 0 349 368 717 0 65
• 0 117 • 2012 11 975 6 127 3 188 2 271 0 389 310 699 0 66
South Sudan 2011 7 217 2 797 2 610 1 639 171 366 537 52
2012 8 403 3 120 3 413 1 685 185 521 706 0 48
Sudan 1990 212 –
1995 14 320 8 761 2 655 1 675 474 474 77
2000 24 807 12 311 6 512 3 843 2 141 2 141 65
2005 27 562 12 730 9 212 5 434 0 186 1 616 1 802 58
2010 26 131 9 958 9 144 6 217 812 1 110 1 922 52
2011 19 348 7 266 6 746 4 624 0 712 1 037 1 749 0 52
• 1 50 • 2012 18 775 6 587 6 948 4 561 0 679 1 056 1 735 0 49
Syrian Arab 1990 6 018 –
Republic 1995 4 404 1 295 1 507 1 574 28 28 46
2000 5 090 1 584 1 409 2 000 97 97 53
2005 4 310 1 350 796 2 103 0 61 83 144 63
2010 3 666 1 122 544 1 948 0 52 161 213 0 67
2011 3 620 1 027 393 1 915 0 60 55 115 225 72
• 48 14 • 2012 3 003 809 364 1 702 0 44 32 76 84 69
Tunisia 1990 2 054 –
1995 2 383 1 243 407 733 75
2000 2 038 1 099 179 727 61 61 86
2005 2 079 915 239 874 51 51 79
2010 2 368 1 091 151 1 090 36 36 88
2011 3 015 1 031 317 1 616 51 51 76
• 25 30 • 2012 3 239 1 059 282 1 853 0 45 19 64 0 79
United Arab 1990 285 –
Emirates 1995 –
2000 115 73 3 41 0 0 96
2005 103 62 12 25 0 4 2 6 0 84
2010 131 56 28 47 0 0 1 1 0 67
2011 103 46 27 30 0 0 3 3 0 63
• 16 1 • 2012 79 42 15 20 0 2 6 8 0 74
West Bank 1990 64 –
and Gaza Strip 1995 77 9 58 10 13
2000 82 37 100
2005 28 7 6 15 54
2010 31 13 6 12 0 0 0 0 0 68
2011 32 11 5 13 0 3 0 3 0 69
• 3 1 • 2012 32 17 6 8 0 1 0 1 0 74
Yemen 1990 4 650 –
1995 14 428 3 681 7 390 3 082 275 275 33
2000 13 651 5 565 4 176 3 470 440 440 57
2005 9 063 3 379 2 780 2 553 351 351 55
2010 8 916 3 584 2 313 2 715 0 304 134 438 0 61
2011 8 636 3 135 2 400 2 880 0 221 77 298 0 57
• 39 41 • 2012 9 867 3 321 2 808 3 486 0 252 83 335 54
a Rates are per 100 000 population.
b NEW AND RELAPSE includes cases for which the treatment history is unknown.

215 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
EASTERN MEDITERRANEAN REGION
7$%/($7UHDWPHQWRXWFRPHVQHZVPHDUSRVLWLYHFDVHV±
% OF COHORT
TREATMENT SUCCESS (%)
a
1995–2011 YEAR NUMBER
NOTIFIED
SIZE OF
COHORT
COHORT AS
% NOTIFIED CURED COMPLETED DIED FAILED DEFAULTED NOT
EVALUATED
Afghanistan 1995 –
2000 2 892 3 136 108 76 9 3 3 6 2
2005 9 949 10 013 101 83 72125
2009 12 497 12 497 100 8342129
2010 12 947 12 947 100 86 3 2125
• 0 91 • 2011 13 789 13 789 100 88 4 2125
Bahrain 1995 17 –
2000 23 22 96 73 0 27 0 0 0
2005 101 15 15 93 0 7000
2009 131 192 147 98 0 2000
2010 90 162 180 96 0 4 0 0 0
• 0 34 • 2011 89 124 139 34 0 1 0 0 65
Djibouti 1995 1 751 – 60 16 3 1 20 1
2000 1 391 1 391 100 48 14 2 1 21 14
2005 1 120 1 120 100 71911162
2009 1 377 1 277 93 72711173
2010 1 181 1 177 100 68 12 1 1 16 2
• 75 82 • 2011 1 336 1 334 100 65 17 1 1 13 3
Egypt 1995 4 229 2 118 50 38 24 2 3 19 14
2000 4 606 4 611 100 75 12 3 2 5 3
2005 5 217 5 154 99 66 13 3 2 3 13
2009 5 201 5 201 100 72 16 3 2 4 3
2010 4 679 4 682 100 59 27 3 3 4 4
• 62 88 • 2011 4 508 4 508 100 66 21 3 2 3 5
Iran (Islamic 1995 5 347 –
Republic of) 2000 5 361 5 866 109 81 4 6233
2005 4 581 4 581 100 78 5 7334
2009 5 152 5 201 101 77 6 7325
2010 5 188 5 269 102 77 6 7433
• 0 85 • 2011 5 539 5 532 100 79 6 8431
Iraq 1995 3 194 11 553 362 60 20 0 5 10 5
2000 3 194 3 194 100 86 5 3 2 3 1
2005 3 096 3 096 100 76 10 3 2 7 3
2009 3 347 3 347 100 80 10 2 1 6 1
2010 3 618 3 618 100 80 9 3 1 6 1
• 80 89 • 2011 3 059 3 059 100 83 6 3 2 5 1
Jordan 1995 187 193 103 91 1 3123
2000 89 89 100 89 1 2 1 4 2
2005 86 86 100 71 12 5 7 6 0
2009 109 109 100 54 21 6 7 11 0
2010 117 117 100 57 30 1 3 6 3
• 92 92 • 2011 103 103 100 46 47 3 0 5 0
Kuwait 1995 175 175 100 40 31 3 0 1 25
2000 180 180 100 54 15 1 0 9 21
2005 187 187 100 53 10 1 0 7 29
2009 386 386 100 41 44 0 0 4 11
2010 385 385 100 63 24 0 0 3 9
• 71 93 • 2011 222 222 100 84 9 0 0 3 4
Lebanon 1995 197 200 102 35 56 0 0 10 0
2000 202 190 94 89 3 4131
2005 131 131 100 81 11 2160
2009 179 179 100 65 17 6 1 2 10
2010 194 192 99 68 12 2 1 18 0
• 91 80 • 2011 188 188 100 65 15 2 2 16
Libyan Arab 1995 626 – 65 0 1 1 33 0
Jamahiriya 2000 607 –
2005 860 860 100 40 29 2 0 27 2
2009 936 –
2010 792 – 43 21 2 0 31 3
• 65 59 • 2011 731 731 100 42 17 1 0 37 3
Morocco 1995 14 171 14 171 100 75 14 2 1 7 1
2000 12 872 12 872 100 8273171
2005 12 757 12 683 99 7652197
2009 11 907 11 935 100 77 82292
2010 12 239 12 492 102 77 8 2192
• 90 80 • 2011 11 822 11 822 100 73 7 2189
Oman 1995 135 93 69 84 09115
2000 164 112 68 93 04300
2005 131 104 79 90 10 0
2009 164 334 204 49 49 2000
2010 152 152 100 97 03000
• 84 97 • 2011 180 212 118 95 23000
Pakistan 1995 2 578 802 31 51 20 4 1 20 4
2000 3 285 4 074 124 58 16 4 1 17 4
2005 48 220 48 205 100 71 13 3194
2009 101 887 101 809 100 74 17 2142
2010 104 263 104 434 100 75 16 2 1 4 2
• 70 92 • 2011 105 733 105 733 100 75 16 2 1 4 2
Qatar 1995 60 43 72 81 0 5 0 0 14
2000 53 53 100 66 0 8 0 0 26
2005 96 96 100 74 9 1 0 0 16
2009 220 5 2 80 0 0 0 20 0
2010 223 219 98 63 3 0 0 0 33
• 81 49 • 2011 197 294 149 46 2 0 0 32 19
Saudi Arabia 1995 –
2000 1 595 1 285 81 62 11 7 0 13 6
2005 1 722 1 722 100 60 5 7 1 10 17
2009 2 201 2 201 100 54 11 6 1 10 18
2010 2 302 2 302 100 52 10 5 1 14 18
• 0 61 • 2011 2 055 2 055 100 53 9 6 1 16 17
Somalia 1995 1 572 1 278 81 82 4 4 5 5 0
2000 3 776 3 776 100 81 2 4 2 3 9
2005 7 068 7 059 100 85 4 4 1 4 2
2009 6 047 6 047 100 83 2 4 2 3 7
2010 5 225 5 225 100 87 2 3 2 3 4
• 86 86 • 2011 5 884 5 884 100 84 2 4 2 3 6
South Sudan 2010 2 114 – 67 8 5 1 15 3
2011 2 797 2 767 99 62 11 4 1 18 4
Sudan 1995 8 761 8 326 95 44 35 2 7 11 1
2000 12 311 14 599 119 50 25 4 2 9 11
2005 12 730 12 730 100 64 18 3 1 9 5
2009 10 541 10 883 103 62 19 3 1 10 6
2010 9 958 7 729 78 56 24 2 1 12 5
• 79 70 • 2011 7 266 7 266 100 47 23 2 1 13 14
a TREATMENT SUCCESS = percent cured + percent completed then rounded to the nearest digit.

216 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($7UHDWPHQWRXWFRPHVQHZVPHDUSRVLWLYHFDVHV±
% OF COHORT
TREATMENT SUCCESS (%)
a
1995–2011 YEAR NUMBER
NOTIFIED
SIZE OF
COHORT
COHORT AS
% NOTIFIED CURED COMPLETED DIED FAILED DEFAULTED NOT
EVALUATED
Syrian Arab 1995 1 295 1 295 100 45 16 2 9 24 5
Republic 2000 1 584 1 562 99 69 10 4 3 11 4
2005 1 350 1 350 100 76 13 3 2 6 1
2009 1 143 1 144 100 76 12 4 1 4 3
2010 1 122 1 122 100 75 14 3 2 4 2
• 61 84 • 2011 1 027 1 009 98 65 19 3 2 10 1
Tunisia 1995 1 243 –
2000 1 099 1 099 100 8743222
2005 915 910 99 8372124
2009 931 931 100 72 11 3239
2010 1 091 1 091 100 62 24 3146
• 0 87 • 2011 1 031 1 026 100 63 24 3155
United Arab 1995 –
Emirates 2000 73 73 100 56 18 7 4 5 10
2005 62 62 100 42 31 6 0 15 6
2009 71 71 100 21 52 11 1 14 0
2010 56 55 98 24 45 7 0 24 0
• 0 73 • 2011 46 60 130 2 72 3 0 23 0
West Bank 1995 9 13 144 100 0
and Gaza Strip 2000 37 –
2005 7 12 171 58 42 0 0 0 0
2009 10 11 110 18 64 9090
2010 13 12 92 8 75 0 17 0 0
• 100 100 • 2011 11 11 100 18 82 0 0 0 0
Yemen 1995 3 681 3 681 100 43 9 1 1 35 11
2000 5 565 5 565 100 59 13 3 1 14 10
2005 3 379 3 566 106 69 11 3 1 6 10
2009 3 576 3 557 99 7993144
2010 3 584 3 584 100 7793147
• 52 88 • 2011 3 135 3 174 101 7992153
a TREATMENT SUCCESS = percent cured + percent completed then rounded to the nearest digit.

217 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
EASTERN MEDITERRANEAN REGION
7$%/($7UHDWPHQWRXWFRPHVUHWUHDWPHQWFDVHV±
% OF COHORT
TREATMENT SUCCESS (%)
a
1995–2011 YEAR NUMBER
NOTIFIED
SIZE OF
COHORT
COHORT AS
% NOTIFIED CURED COMPLETED DIED FAILED DEFAULTED NOT
EVALUATED
Afghanistan 1995 –
2000 237 304 128 73 5 4 4 11 3
2005 856 856 100 87 2 3 1 2 5
2009 1 290 –
2010 1 325 1 325 100 73 6 3 3 1 14
• 0 77 • 2011 1 314 1 937 147 58 19 2 2 2 17
Bahrain 1995 0 –
2000 –
2005 0 –
2009 0 0 –
2010 0 0 –
• 0 0 • 2011 0 0 –
Djibouti 1995 –
2000 184 268 146 27 9 0 3 22 37
2005 253 253 100 58 10 3 2 24 2
2009 210 194 92 67 8 3 2 18 3
2010 219 213 97 53 17 6 2 19 3
• 0 63 • 2011 231 227 98 47 16 4 5 22 6
Egypt 1995 753 –
2000 620 956 154 52 11 7 12 13 5
2005 738 738 100 41 17 10 12 8 12
2009 748 748 100 39 39 6 5 7 4
2010 703 703 100 38 34 6 8 8 6
• 0 72 • 2011 670 599 89 35 36 6 6 10 7
Iran (Islamic 1995 477 –
Republic of) 2000 405 606 150 63 13 6 5 6 7
2005 428 448 105 68 8 9 3 4 8
2009 773 708 92 48 25 8 3 5 11
2010 760 781 103 49 20 8 5 4 15
• 0 72 • 2011 900 892 99 49 22 9 4 4 12
Iraq 1995 68 –
2000 562 –
2005 768 953 124 60 12 4 8 12 4
2009 751 751 100 57 27 3 3 9 1
2010 777 777 100 36 40 4 5 13 3
• 0 75 • 2011 769 769 100 39 36 5 7 12 1
Jordan 1995 6 –
2000 3 6 200 83 17 0 0 0 0
2005 10 –
2009 20 24 120 17 62 4 0 17 0
2010 18 5 28 0 60 0 20 0 20
• 0 80 • 2011 18 15 83 13 67 0 0 20 0
Kuwait 1995 4 –
2000 0 –
2005 1 1 100 0 100 0 0 0 0
2009 1 1 100 0 100 0 0 0 0
2010 2 2 100 0 100 0 0 0 0
• 0 0 • 2011 0 0 –
Lebanon 1995 3 –
2000 6 5 83 80 20
2005 4 4 100 75 25 0 0 0 0
2009 10 10 100 60 20 0 0 0 20
2010 12 12 100 58 17 8 0 17 0
• 0 100 • 2011 1 1 100 100 0
Libyan Arab 1995 –
Jamahiriya 2000 –
2005 271 –
2009 23 –
2010 85 – 11 22 2 0 45 20
• 0 0 • 2011 47 –
Morocco 1995 1 469 – 65 12 4 4 10 7
2000 –
2005 1 650 – 55 17 4 5 14 5
2009 1 605 1 668 104 60 9 4 3 16 8
2010 1 645 2 899 176 40 24 4 3 21 9
• 76 66 • 2011 2 345 2 623 112 38 28 3 3 21 8
Oman 1995 0 –
2000 8 7 88 86 0 0 14 0 0
2005 4 –
2009 7 7 100 57 43 0 0 0 0
2010 9 9 100 44 56 0 0 0 0
• 0 67 • 2011 3 3 100 67 0 33 0 0 0
Pakistan 1995 184 374 203 48 22 2 5 24 0
2000 341 907 266 37 17 6 3 29 8
2005 5 425 5 009 92 61 15 5 3 11 5
2009 9 200 8 801 96 63 18 4 3 8 3
2010 10 925 8 394 77 68 16 3 3 6 3
• 70 80 • 2011 11 407 11 407 100 63 17 4 3 8 4
Qatar 1995 1 3 300 67 0 0 0 0 33
2000 0 –
2005 0 –
2009 0 0 –
2010 0 0 –
• 67 0 • 2011 0 –
Saudi Arabia 1995 –
2000 112 139 124 43 15 7 3 13 19
2005 205 96 47 409951819
2009 144 151 105 45 15 8 1 17 14
2010 206 249 121 31 19 8 2 22 17
• 0 63 • 2011 147 147 100 41 22 9 3 10 15
Somalia 1995 134 –
2000 351 351 100 53155334
2005 512 524 102 7656256
2009 655 655 100 50 10 6 4 3 27
2010 705 705 100 48 14 6 5 4 23
• 0 72 • 2011 717 717 100 43 29 7 6 4 11
South Sudan 2010 434 – 23 34 5 9 23 5
2011 537 527 98 20 38 7 2 28 5
Sudan 1995 474 –
2000 2 141 –
2005 1 802 1 828 101 53 29 3196
2009 1 993 2 147 108 33 38 3 1 15 10
2010 1 922 1 517 79 28 40 2 1 14 16
• 0 55 • 2011 1 749 1 749 100 22 33 3 1 13 27
a TREATMENT SUCCESS = percent cured + percent completed then rounded to the nearest digit.

GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($7UHDWPHQWRXWFRPHVUHWUHDWPHQWFDVHV±
% OF COHORT
TREATMENT SUCCESS (%)
a
1995–2011 YEAR NUMBER
NOTIFIED
SIZE OF
COHORT
COHORT AS
% NOTIFIED CURED COMPLETED DIED FAILED DEFAULTED NOT
EVALUATED
Syrian Arab 1995 28 –
Republic 2000 97 189 195 44 10 4 20 15 7
2005 144 144 100 53 14 5 9 19 0
2009 176 176 100 48 22 9 4 15 3
2010 213 213 100 23 58 4 3 11 1
• 0 70 • 2011 115 225 196 20 49 5 5 20 1
Tunisia 1995 –
2000 61 42 69 740521010
2005 51 –
2009 42 –
2010 36 –
• 0 79 • 2011 51 52 102 54 25 2 8 10 2
United Arab 1995 –
Emirates 2000 0 –
2005 6 5 83 80 0 0 0 20 0
2009 0 0 –
2010 1 3 300 0 67 33 0 0 0
• 0 33 • 2011 3 3 100 0 33 0 0 67 0
West Bank 1995 –
and Gaza Strip 2000 –
2005 0 –
2009 2 0 0
2010 0 –
• 0 0 • 2011 3 –
Yemen 1995 275 14 5 29 14 21 14 14 7
2000 440 437 99 64 8 7 6 11 4
2005 351 351 100 48 9 2 3 7 30
2009 314 291 93 70 7 3 4 7 9
2010 438 –
• 43 67 • 2011 298 298 100 62 5 5 3 6 19
a TREATMENT SUCCESS = percent cured + percent completed then rounded to the nearest digit.
219 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
EASTERN MEDITERRANEAN REGION
7$%/($+,9WHVWLQJDQGSURYLVLRQRI&37$57DQG,37–
% OF TB PATIENTS WITH
KNOWN HIV STATUS
2005–2012
YEAR
% OF TB
PATIENTS WITH
KNOWN HIV
STATUS
NUMBER OF TB
PATIENTS WITH
KNOWN HIV
STATUS
PATIENTS
NOTIFIED
(NEW AND
RETREAT)
NUMBER OF
HIV-POSITIVE
TB PATIENTS
% OF TESTED
TB PATIENTS
HIV-POSITIVE
% OF HIV-
POSITIVE TB
PATIENTS ON
CPT
% OF HIV-
POSITIVE TB
PATIENTS ON
ART
NUMBER OF
HIV-POSITIVE
PEOPLE
PROVIDED IPT
Afghanistan 2005 21 844
2010 18 5 170 28 238 2 <0.1 100 100
2011 23 6 445 28 167 5 <0.1 80 80
– 25 • 2012 25 7 275 29 578 5 <0.1 100 100 25
Bahrain 2005 46 128 280 6 4.7 0 0
2010 65 161 246 6 3.7 0 0
2011 66 148 225 7 4.7 0 43
• 46 82 • 2012 82 184 225 1 0.54 0 100
Djibouti 2005 7.1 224 3 170 135 60 15 15 0
2010 52 2 163 4 191 248 11 0 11
2011 19 718 3 723 177 25 22
• 7 36 • 2012 36 1 289 3 546 130 10 64 0
Egypt 2005 11 735
2010 47 4 483 9 588 7 0.16 100 100 0
2011 37 3 441 9 307 12 0.35 100 100
– 17 • 2012 17 1 514 8 753 17 1.1 100 100
Iran (Islamic 2005 9 366
Republic of) 2010 8.4 904 10 802 254 28 16 28 161
2011 12 1 343 11 495 291 22 20 37
– 14 • 2012 14 1 574 11 483 283 18 27 41 155
Iraq 2005 9 454
2010 66 6 711 10 097 1 <0.1 100 0 0
2011 84 7 754 9 248 2 <0.1 100 50
– 86 • 2012 86 7 821 9 099 2 <0.1 50 50
Jordan 2005 23 86 371 0 0
2010 99 352 354 0 0 0
2011 78 267 344 1 0.37 100 100
• 23 51 • 2012 51 177 349 0 0
Kuwait 2005 100 517 517 3 0.58 100 100
2010 100 957 957 3 0.31 100 100
2011 100 672 672 0 0
• 100 100 • 2012 100 737 737 3 0.41 100 100
Lebanon 2005 0.77 3 391 3 100 0
2010 52 269 515 7 2.6 100 100 68
2011 48 236 496 9 3.8 100 100
• 1 67 • 2012 67 424 630 3 0.71 100 100 9
Libyan Arab 2005 2 367
Jamahiriya 2010 2 128 212 10 1.4
2011 97 1 498 1 545 128 8.5 0
– 100 • 2012 100 1 549 1 549 105 6.8
Morocco 2005 26 269
2010 0.75 215 28 788 17 7.9 100 100
2011 6.2 1 856 29 770 41 2.2 100 68
– 20 • 2012 20 5 827 29 399 357 6.1 100 100
Oman 2005 98 257 261 10 3.9 100 100
2010 100 313 313 4 1.3 100 100 0
2011 100 337 337 8 2.4 88 88
• 98 100 • 2012 100 383 383 14 3.7 100 100
Pakistan 2005 0 0 144 771 0
2010 2.3 6 283 269 290 28 0.45 39 43
2011 3.1 8 264 270 394 34 0.41 100 56
• 0 4 • 2012 3.8 10 419 273 097 30 0.29 100 73
Qatar 2005 100 325 325 0 0
2010 0 0 580 0
2011 0 0 553 0
• 100 0 • 2012 0.14 1 728 1 100 100 100
Saudi Arabia 2005 3 539
2010 72 3 278 4 549 77 2.3
2011 86 3 469 4 015 77 2.2 14
– 89 • 2012 89 3 420 3 833 79 2.3
Somalia 2005 0 0 13 006 21 38 0
2010 26 2 741 10 469 231 8.4 68 26
2011 34 4 140 12 021 206 5 85 20 0
• 0 44 • 2012 44 5 359 12 285 192 3.6 79 27
South Sudan 2011 47 3 542 7 583 428 12 82 27
2012 51 4 584 8 924 534 12 62 28
Sudan 2005 0.62 180 29 178 150 83 10 10
2010 28 7 532 27 241 247 3.3 160 100
2011 15 3 082 20 385 292 9.5 0 25
• 1 15 • 2012 15 3 070 19 831 231 7.5 0 17
Syrian Arab 2005 7.9 345 4 393 0 0
Republic 2010 2.2 85 3 827 5 5.9 100 0 0
2011 16 586 3 675 7 1.2 100 100
• 8 53 • 2012 53 1 601 3 035 5 0.31 100 100
Tunisia 2005 6.2 129 2 079 2 1.6 100 100
2010 6.6 156 2 368 7 4.5 100 100 24
2011 12 360 3 015 10 2.8 100 100 38
• 6 18 • 2012 18 593 3 258 14 2.4 100 100 54
United Arab 2005 105
Emirates 2010 64 84 132 4 4.8 100 100
2011 76 81 106 3 3.7 100 100
– 62 • 2012 62 53 85 4 7.5
West Bank 2005 0 0 28 0
and Gaza Strip 2010 100 31 31 0 0
2011 100 32 32 0 0
• 0 100 • 2012 100 32 32 0 0
Yemen 2005 0 0 9 063 0
2010 0 0 9 050 0 0
2011 0 0 8 713 0 0
• 0 6 • 2012 6.2 612 9 950 26 4.2 62 0

220 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($7HVWLQJIRU0'57%DQGQXPEHURIFRQILUPHGFDVHVRI0'57%±
NEW PULMONARY CASES PREVIOUSLY TREATED CASES
YEAR
ESTIMATED CASES
OF MDR-TB AMONG
NOTIFIED
NUMBER OF
BACT+VE
b
TESTED FOR
MDR-TB
% OF
BACT+VE
b
TESTED FOR
MDR-TB
ESTIMATED CASES
OF MDR-TB AMONG
NOTIFIED
NUMBER OF
NOTIFIED
TESTED FOR
MDR-TB
% OF NOT IFIED
TESTED FOR
MDR-TB
TOTAL
CONFIRMED
CASES OF
MDR-TB
a
ESTIMATED CASES
OF MDR-TB AMONG
NOTIFIED
Afghanistan 2005 ––
2010 19 238 1.8 34 2.6
2011 19 ––
2012 31 1 100 (0–2 900) 750 (21–2 600) – 400 (93–700) 38 3.0
Bahrain 2005 4 2 2.0 0 –
2010 0 162 70 0 –
2011 9 154 99 0 –
2012 4 2.8 (0.57–8.0) 2.8 (0.57–8.0) 160 110 0 (0–0) 1 –
Djibouti 2005 39 0 0 0 0
2010 ––
2011 0 ––
2012 96 81 (40–120) 31 (1.7–58) – 50 (19–81) –
Egypt 2005 ––
2010 ––
2011 134 39 0.70 497 74
2012 116 330 (270–390) 180 (99–260) 31 0.59 150 (130–180) 438 72
Iran (Islamic 2005 27 205 4.5 41 9.6
Republic of) 2010 58 271 4.7 169 22
2011 43 717 13 322 36
2012 50 750 (590–910) 380 (260–530) 411 6.8 380 (270–480) 207 27
Iraq 2005 ––
2010 110 0 0 185 24
2011 84 – 224 29
2012 62 420 (0–870) 180 (5.1–610) 69 2.5 240 (57–430) 159 21
Jordan 2005 19 98 97 33 330
2010 10 74 63 7 39
2011 4 55 30 6 33
2012 13 15 (5.4–25) 10 (3.7–21) 77 91 5.4 (0.70–13) 6 32
Kuwait 2005 6 516 280 1 100
2010 5 437 100 0 0
2011 0 282 100 0 –
2012 4 0 (0–6.1) 0 (0–6.1) – 0 (0–0) –
Lebanon 2005 3 48 37 4 100
2010 7 4 2.1 14 120
2011 3 18 9.6 1 100
2012 6 9.9 (3.5–16) 3.9 (0.47–14) 10 4.2 6.0 (2.0–8.6) 6 67
Libyan Arab 2005 8 4 0.47 –
Jamahiriya 2010 ––
2011 1 ––
2012 36 (1.0–120) 36 (1.0–120) – – –
Morocco 2005 180 180 1.4 –
2010 54 47 0.38 403 24
2011 45 61 0.50 229 9.8
2012 80 300 (190–410) 66 (22–150) 103 0.85 240 (150–350) 416 21
Oman 2005 5 125 95 11 280
2010 1 185 59 8 89
2011 4 219 100 3 100
2012 6 5.9 (1.2–11) 5.9 (2.2–13) 248 100 0 (0–3.0) 8 100
Pakistan 2005 ––
2010 444 9 <0.1 306 2.8
2011 344 ––
2012 1602 11 000 (0–29 000) 7 700 (220–27 000) 461 0.42 3 700 (880–6 600) 154 1.3
Qatar 2005 2 264 190 0 –
2010 4 324 100 0 –
2011 4 9 1.6 0 –
2012 2 6.3 (1.7–16) 6.3 (1.7–16) 10 2.0 0 (0–0) –
Saudi Arabia 2005 ––
2010 14 ––
2011 22 ––
2012 20 84 (64–100) 46 (36–62) – 37 (28–48) –
Somalia 2005 ––
2010 57 488 9.3 79 11
2011 20 261 4.4 14 2.0
2012 0 770 (600–930) 480 (250–720) 0 0 280 (160–410) 0 0
South Sudan 2011 6 –81.5
2012 3 250 (120–390) 120 (6.5–220) – 140 (52–220) –
Sudan 2005 45 – 4 0.22
2010 49 ––
2011 62 36 0.29 82 4.7
2012 116 580 (280–870) 240 (14–460) 43 0.65 330 (130–540) 129 7.4
Syrian Arab 2005 7 0 0 0 0
Republic 2010 25 63 1.7 12 5.6
2011 24 408 12 70 61
2012 13 97 (65–130) 73 (46–110) 155 13 24 (16–33) 23 30
Tunisia 2005 ––
2010 12 6 0.55 6 17
2011 12 2 0.19 10 20
2012 15 19 (7.0–30) 11 (0–23) 3 0.28 7.6 (2.9–12) 12 19
United Arab 2005 4 ––
Emirates 2010 0 ––
2011 1 3 5.0 0 0
2012 2 2.0 (1.5–2.5) 1.0 (0.51–1.5) 26 52 0.95 (0.74–1.2) 3 38
West Bank 2005 ––
and Gaza Strip 2010 0 0 0 0 –
2011 0 0 0 0 0
2012 0 1.1 (0–3.0) 0.81 (<0.1–2.8) 0 0 0.32 (<0.1–0.56) 0 0
Yemen 2005 1 ––
2010 4 89 1.5 34 7.8
2011
–
–
2012 8 150
(
100–210
)
110
(
31–180
)
183 5.5 49
(
27–73
)
17 5.1
a TOTAL CONFIRMED CASES OF MDR-TB includes cases with unknown previous treatment history (i.e. not included under NEW CASES or PREVIOUSLY TREATED
CASES).
b BACT+VE = bacteriologically positive cases.

221 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
EASTERN MEDITERRANEAN REGION
7$%/($1HZVPHDUSRVLWLYHFDVHQRWLILFDWLRQE\DJHDQGVH[±
MALE FEMALE
YEAR 0–14 15–24 25–34 35–44 45–54 55–64 65+
UN-
KNOWN
0–14 15–24 25–34 35–44 45–54 55–64 65+
UN-
KNOWN
MALE:FEMALE
RATIO
Af
g
hanistan 1995
–
2000 52 228 183 149 129 94 80 93 414 565 339 205 99 36 0.52
2005 151 606 560 472 453 470 419 320 1 651 1 959 1 302 869 471 246 0.46
2010 197 986 819 491 490 641 622 0 445 2 107 2 263 1 455 1 112 831 488 0 0.49
2011 204 1 010 895 613 570 700 692 465 2 167 2 325 1 564 1 146 903 535 0.51
2012 188 1 116 801 586 521 585 651 0 400 2 280 2 204 1 482 1 150 850 505 0 0.50
Bahrain 1995 0012313 0112011 1.7
2000 0032534 0120111 2.8
2005 0002304 1103100 1.5
2010 0 10 16 11 12 4 4 0 0 8 15 7111 1.7
2011 1 5 19 13 14 8 2 0 09566010 2.3
2012 0 9 28 16 11 8 2 0 1 2 11 84100 2.7
D
j
ibouti 1995
–
2000 17 302 347 139 67 60 42 12 147 156 47 31 17 10 2.3
2005 18 220 252 119 62 47 29 23 123 117 66 23 13 8 2.0
2010 28 211 243 151 67 49 20 0 20 104 120 89 36 24 19 0 1.9
2011 35 212 265 149 97 45 33 0 31 139 118 104 57 30 21 0 1.7
2012 22 208 240 147 81 47 26 20 132 94 73 36 26 18 1.9
Egypt 1995 223 542 665 460 408 463 160 134 288 367 274 256 160 75 1.9
2000 21 641 827 667 476 307 158 55 457 343 257 211 112 48 2.1
2005 25 524 606 421 414 243 123 48 431 298 205 218 132 42 1.7
2010 9 358 617 783 725 407 217 0 8 199 352 423 292 192 97 0 2.0
2011 23 382 611 596 715 387 168 0 7 192 355 387 280 198 94 0 1.9
2012 23 373 597 582 698 379 164 0 8 187 346 379 274 193 92 0 1.9
Iran (Islamic 1995 118 751 754 636 494 737 921 234 1 039 890 664 613 685 788 0.90
Republic of) 2000 29 438 467 387 295 344 642 77 593 410 322 320 407 647 0.94
2005 16 352 531 338 281 260 630 45 394 205 186 260 382 701 1.1
2010 18 292 487 354 296 310 760 0 54 433 288 208 276 398 1 014 0 0.94
2011 13 289 543 398 315 351 877 0 37 473 313 184 296 441 1 009 0 1.0
2012 16 288 601 442 303 317 850 0 43 434 318 206 252 374 965 0 1.1
Iraq 1995 1 125 862 1 409 1 085 863 900 271 725 304 1 208 915 800 886 200 1.3
2000 21 627 317 297 205 135 101 37 338 241 136 134 103 87 1.6
2005 13 424 644 261 245 189 148 44 305 260 151 197 135 80 1.6
2010 42 370 482 384 276 286 228 73 394 294 198 205 220 166 1.3
2011 35 304 395 313 237 223 183 0 66 368 258 164 159 201 153 0 1.2
2012 27 283 317 263 203 203 180 36 340 225 154 186 174 169 1.1
Jordan 1995 0 19 37 17 20 26 11 1 15 4 10 14 12 7 2.1
2000 0816139142 0891225 2.3
2005 08179465 1666585 1.3
2010 2 5 14 10 12 12 6 0 3 14 24 4 3 5 3 0 1.1
2011 09101381350 081184860 1.3
2012 081285770 191271350 1.2
Kuwait 1995 0 15 51 32 17 9 0 0 8 24 9 4 4 2 2.4
2000 0 10 44 32 21 11 5 1 11 24 12 5 3 1 2.2
2005 0 12 45 29 26 8 3 0 13 31 11 3 1 5 1.9
2010 1 16 67 50 48 10 11 0 4 41 78 30 10 11 8 0 1.1
2011 0 13 41 36 35 11 5 0 0 23 30 15 9 2 2 0 1.7
2012 0 14 59 49 35 15 3 0 3 40 73 15 12 6 4 0 1.1
Lebanon 1995 3 26 32 30 16 16 10 1 16 18 13 8 5 3 2.1
2000 5 16 28 20 15 17 14 4 31 26 9 7 4 6 1.3
2005 0 12 19 15 10 12 8 1 25 14 8 3 3 1 1.4
2010 1 8 21 15 12 12 10 0 0 36 48 17 7 4 3 0 0.69
2011 1 14 18 13 15 6 8 0 37 51 12 9 1 3 0.66
2012 2 18 21 13 14 12 6 2 48 72 16 9 4 3 0.56
Libyan Arab 1995 2 112 212 78 46 22 21 5 34 31 19 20 13 11 3.7
Jamahiriya 2000 5 101 239 86 36 29 32 6 43 35 24 24 16 22 3.1
2005 2 114 293 168 52 19 35 8 36 36 35 21 21 20 3.9
2010 –
2011 5 85 173 148 54 18 21 0 8 59 47 37 22 25 29 0 2.2
2012 2 86 136 136 63 31 22 0 10 47 37 19 24 18 13 0 2.8
Morocco 1995 142 2 508 2 872 1 737 819 573 553 191 1 708 1 288 703 461 317 299 1.9
2000 99 2 061 2 423 1 705 855 485 595 170 1 530 1 121 672 398 406 352 1.8
2005 79 2 222 2 515 1 583 1 057 580 591 167 1 330 943 546 403 343 398 2.1
2010 51 1 982 2 553 1 611 1 273 712 515 0 117 1 098 841 426 386 310 364 0 2.5
2011 79 1 929 2 450 1 479 1 175 682 518 0 100 1 153 794 433 371 324 335 0 2.4
2012 54 1 840 2 426 1 423 1 183 672 561 0 77 1 162 832 408 306 286 342 0 2.4
Oman 1995 1 7 12 7 7 10 11 2 18 13 5 5 6 3 1.1
2000 1 8 9 11 12 9 11 2 17 5 7 5 11 6 1.2
2005 1 21 11 24 15 19 5 2 13 5 3 4 5 3 2.7
2010 2 12 27 15 16 8 10 0 3 18 22 6 4 4 5 0 1.5
2011 1 17 25 12 23 10 11 0 5 20 21 9 13 7 6 0 1.2
2012 0 18 33 23 12 8 19 0 0 20 37 10 10 9 6 0 1.2
Pakistan 1995 29 274 230 178 140 124 95 85 375 381 267 178 143 79 0.71
2000 55 498 387 256 232 153 130 130 591 416 274 163 103 56 0.99
2005 621 5 278 4 759 4 263 3 834 3 332 2 453 1 447 6 463 5 611 3 987 2 866 2 060 1 338 1.0
2010 1 548 11 860 10 462 8 320 7 969 6 934 6 066 3 212 14 481 10 513 7 749 6 410 4 879 4 338 1.0
2011 1 216 12 143 10 515 8 435 8 608 7 320 6 323 2 679 14 652 10 684 7 880 6 590 4 977 3 711 1.1
2012 1 317 12 605 10 838 8 848 9 026 7 753 6 492 0 2 630 15 445 10 902 8 263 6 876 5 494 4 056 0 1.1
Qatar 1995 0 8 12 11 13 4 4 1 2 3 1 0 0 1 6.5
2000 07199721 0043100 5.6
2005 19 15 17 19 5 1 5 10 2 1 2 0 3.8
2010 0 59 72 38 22 5 0 0 0 7 16 2 1 1 0 0 7.3
2011 0 36 64 36 14 10 3 0 9 15 6 1 2 1 4.8
2012 0 34 52 45 21 8 0 160 2 6 9 1 1 0 1 20 8.0
Saudi Arabia 1995 –
2000 0 131 268 213 158 86 107 28 172 182 79 51 50 70 1.5
2005 8 182 276 201 175 70 107 31 205 184 98 73 51 61 1.4
2010 14 335 458 242 210 116 102 0 33 239 271 105 70 49 58 0 1.8
2011 4 227 406 225 225 113 106 0 35 200 245 110 64 49 46 0 1.7
2012 13 228 394 214 210 133 96 0 28 207 236 107 50 49 63 0 1.7
Somalia 1995 46 334 730 201 127 278 109 38 158 139 97 40 25 16 3.6
2000 113 740 724 408 254 195 142 85 354 319 219 110 72 41 2.1
2005 125 1 343 1 114 725 458 330 319 169 752 636 436 292 212 157 1.7
2010 109 1 036 886 496 355 266 277 0 91 467 444 341 188 137 132 0 1.9
2011 113 1 147 1 047 587 398 330 277 0 114 495 465 348 260 168 135 0 2.0
2012 129 1 147 1 014 560 449 296 307 0 121 553 554 396 267 165 169 0 1.8
South Sudan 2011 39 251 599 402 259 135 57 0 60 181 318 239 172 59 26 0 1.7
2012 42 356 753 462 267 135 87 58 212 302 221 139 62 24 2.1
Sudan 1995 250 604 796 634 486 362 337 359 490 613 299 403 342 305 1.2
2000 785 1 028 1 511 1 351 1 119 638 677 817 925 1 134 905 771 327 323 1.4
2005 425 1 358 1 990 1 541 1 151 724 493 381 1 102 1 203 978 729 411 244 1.5
2010 209 1 185 1 781 1 335 863 497 391 195 761 979 772 520 279 191 1.7
2011 107 899 1 359 981 689 386 372 0 113 512 620 513 352 188 175 0 1.9
2012 117 869 1 274 802 466 404 331 0 115 536 562 470 299 170 172 0 1.8

222 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($1HZVPHDUSRVLWLYHFDVHQRWLILFDWLRQE\DJHDQGVH[±
MALE FEMALE
YEAR 0–14 15–24 25–34 35–44 45–54 55–64 65+
UN-
KNOWN
0–14 15–24 25–34 35–44 45–54 55–64 65+
UN-
KNOWN
MALE:FEMALE
RATIO
Syrian Arab 1995 13 332 255 111 70 59 50 22 158 97 53 44 37 20 2.1
Republic 2000 8 359 289 125 86 76 55 23 195 101 53 46 38 28 2.1
2005 9 266 237 111 112 62 63 27 182 108 59 59 32 23 1.8
2010 7 170 212 101 80 65 49 0 16 164 105 47 41 38 27 0 1.6
2011 8 139 195 116 81 49 45 0 20 113 97 56 35 36 37 0 1.6
2012 7 91 146 90 85 46 41 5 104 75 35 33 32 19 1.7
Tunisia 1995 –
2000 16 139 208 156 109 65 101 7 68 59 43 21 21 58 2.9
2005 5 103 172 133 115 53 81 7 66 61 39 36 16 28 2.6
2010 9 115 194 170 125 93 88 4 64 64 39 34 40 52 2.7
2011 6 110 194 118 126 108 63 0 10 60 60 50 44 35 47 0 2.4
2012 10 88 191 149 114 93 88 0 7 51 56 46 48 46 72 0 2.2
United Arab 1995 –
Emirates 2000 2 4 4 6 5 12 10 3 16 1 3 0 0 4 1.6
2005 –
2010 171373440 12415130 2.3
2011 03735130 46632120 0.92
2012 02445520 05223440 1.1
West Bank 1995 1200103 0100100 3.5
and Gaza Strip 2000 –
2005 1 1 3 1 1 2.5
2010 02021130 00101200 2.2
2011 10111030 00110200 1.8
2012 02212420 01100110 3.2
Yemen 1995 57 400 605 256 201 148 45 83 420 720 348 200 106 92 0.87
2000 110 789 689 493 314 255 127 161 799 627 517 345 247 92 1.0
2005 48 493 553 366 242 149 78 44 426 410 265 181 85 39 1.3
2010 68 507 569 322 231 164 138 0 98 471 409 264 174 106 63 0 1.3
2011 33 406 471 297 193 143 96 0 85 446 375 251 168 113 58 0 1.1
2012 30 436 472 315 232 172 122 75 437 381 246 207 115 81 1.2
223 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
EASTERN MEDITERRANEAN REGION
a LED = Light emitting diode microscopes
b DST = Drug susceptibility testing
c LPA = Line probe assay
d NRL = National Reference Laboratory
7$%/($/DERUDWRULHV173VHUYLFHVGUXJPDQDJHPHQWDQGLQIHFWLRQFRQWURO
LABORATORIES FREE THROUGH NTP
SMEAR LABS
PER 100K
POPULATION
% OF SMEAR
LABS USING
LED
a
CULTURE
LABS PER 5M
POPULATION
DST
b
LABS
PER 5M
POPULATION
LPA
c
LABS
PER 5M
POPULATION
NUMBER OF
LABS USING
XPERT MTB/RIF
SECOND-
LINE DST
AVAILABLE
NRL
d
TB DIAGNOSIS
FIRST-
LINE
DRUGS
TB NOTIF.
RATE PER
100 000
HEALTH-CARE
WORKERS
RIFAMPICIN
USED
THROUGHOUT
TREATMENT
Afghanistan 2.0 2 0.3 0 0 1 Out of
country Yes Yes (all suspects) Yes No
Bahrain 1.4 11 7.6 3.8 7.6 1 Out of
country No Yes (all suspects) Yes Yes
Djibouti 2.1 0 5.8 5.8 5.8 1 In country Yes Yes (all suspects) Yes Yes
Egypt 0.2 0 1.1 <0.1 0 0 In country Yes Yes (all suspects) Yes Yes 0
Iran (Islamic Republic of) 0.5 0 3.6 0.5 0 0 In country Yes Yes (all suspects) Yes Yes
Iraq 0.8 0 1.5 0.2 0 5 Out of
country Yes Yes (all suspects) Yes Yes 1
Jordan 0.2 0 0.7 0.7 0 1 No Yes Yes (all suspects) Yes Yes 0
Kuwait 0.4 0 1.5 1.5 0 0 No Yes Yes (all suspects) Yes Yes 0
Lebanon 6.0 0 3.2 1.1 2.2 3 Out of
country Yes Yes (all suspects) Yes Yes 0
Libya 0.4 – 6.5 1.6 No Yes Yes (all suspects) Yes Yes
Morocco 0.5 13 2.2 0.3 0 0 No Yes Yes (all suspects) Yes Yes
Oman 7.5 0 13.6 1.5 1.5 0 In country Yes Yes (all suspects) Yes Yes 8
Pakistan 0.8 0 0.2 0.1 <0.1 15 Yes Yes (all suspects) Yes Yes
Qatar <0.1 0 2.4 2.4 2.4 1 Yes Yes (all suspects) Yes Yes 0
Saudi Arabia 0.3 1 2.1 2.1 0.4 8 No Yes Yes (all suspects) Yes Yes
Somalia 0.6 0 0 0 0 3 No No Yes (all suspects) Yes Yes
South Sudan 0.6 – Out of
country No Yes (all suspects) Yes Yes
Sudan 0.8 0 0.1 0.1 0 0 No Yes Yes
(
if TB is
confirmed) Yes Yes
Syrian Arab Republic 1.4 – 0.2 0.2 0.2 0 No Yes Yes (all suspects) Yes Yes 30 508
Tunisia 0.7 0 5.1 2.3 0.5 2 In country Yes Yes (all suspects) Yes Yes
United Arab Emirates – No Yes (all suspects) Yes Yes
West Bank and
Gaza Strip 1.5 0 1.2 0 0 0 No Yes Yes (all suspects) Yes Yes 204
Yemen 1.0 – 0.8 0.4 No Yes Yes
(
if TB is
confirmed) Yes Yes 36

224 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($0HDVXUHGSHUFHQWDJHRI7%FDVHVZLWK0'57%
D
PRVWUHFHQW\HDUDYDLODEOH
New TB cases Previously treated TB cases
Year Source Coverage Percentage Year Source Coverage Percentage
Afghanistan
Bahrain 2012 Surveillance National 1.9 (0.39–5.4) 2012 Surveillance National 100 (2.5–100)
Djibouti
Egypt 2011 Survey National 3.4 (1.9–4.9) 2012 Surveillance National 25 (21–29)
Iran (Islamic Republic of) 1998 Survey National 5 (3.4–7.0) 1998 Survey National 48 (35–62)
Iraq
Jordan 2009 Surveillance National 6.3 (2.4–13) 2009 Surveillance National 29 (3.7–71)
Kuwait 2011 Surveillance National 0 (0–1.3) 2011 Surveillance National 0 (0–98)
Lebanon 2003 Survey National 1.1 (0.13–3.8) 2012 Surveillance National 67 (22–96)
Libya
Morocco 2006 Survey National 0.48 (0.15–1.1) 2006 Survey National 12 (7.8–18)
Oman 2012 Surveillance National 2.4 (0.89–5.2) 2012 Surveillance National 0 (0–37)
Pakistan
Qatar 2010 Surveillance National 1.2 (0.34–3.1) 2010 Surveillance National 0 (0–98)
Saudi Arabia 2010 Survey National 1.8 (1.4–2.4) 2010 Survey National 16 (12–21)
Somalia 2011 Survey National 5.2 (2.7–7.7) 2011 Survey National 41 (23–58)
South Sudan
Sudan
Syrian Arab Republic 2003 Survey National 6.2 (3.9–9.3) 2011 Surveillance National 31 (21–44)
Tunisia 2012 Survey National 0.82 (0–1.7) 2012 Survey National 12 (4.5–19)
United Arab Emirates
West Bank and
Gaza Strip
Yemen 2011 Survey National 1.7 (0.50–3.0) 2011 Survey National 15 (8.1–22)
a Empty rows indicate an absence of high-quality survey or surveillance data. In the absence of high-quality national data, high-quality sub-national data are used.

'7412'#04')+10
Table A4.1 Estimates of the burden of disease caused by TB, 1990–2012 227
6CDNG# +PEKFGPEGPQVKßECVKQPCPFECUGFGVGEVKQPTCVGUCNNHQTOU¿
6CDNG# %CUGPQVKßECVKQPU¿
Table A4.4 Treatment outcomes, new smear-positive cases, 1995–2011 239
Table A4.5 Treatment outcomes, retreatment cases, 1995–2011 242
6CDNG# *+8VGUVKPICPFRTQXKUKQPQH%26#46CPF+26¿
6CDNG# 6GUVKPIHQT/&46$CPFPWODGTQHEQPßTOGFECUGUQH/&46$¿
6CDNG# 0GYUOGCTRQUKVKXGECUGPQVKßECVKQPD[CIGCPFUGZ¿
Table A4.9 Laboratories, NTP services, drug management and infection control, 2012 252
6CDNG# /GCUWTGFRGTEGPVCIGQH6$ECUGUYKVJ/&46$OQUVTGEGPV[GCTCXCKNCDNG

226 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
Estimates of mortality, prevalence and incidence
Estimated values are shown as best estimates followed by lower and upper bounds. e lower and upper bounds are dened
as the 2.5th and 97.5th centiles of outcome distributions produced in simulations. See ANNEX 1 for further details.
Estimated numbers are shown rounded to two signicant gures. Estimated rates are shown rounded to three signicant
gures unless the value is under 100, in which case rates are shown rounded to two signicant gures.
Estimates for all years are recalculated as new information becomes available and techniques are rened, so
they may dier from those published in previous reports in this series. e main updates implemented in this
report are explained in Box 2.1 of Chapter 2. Estimates published in previous global TB control reports should no
longer be used.
Data source
Data shown in this annex are taken from the WHO global TB database on 1 October 2013. Data shown in the main part of
the report were taken from the database in July 2013. As a result, data in this annex may dier slightly from those in the
main part of the report.
Data for all years can be downloaded from www.who.int/tb/data.
Country notes
EU/EEA countries
Notication and treatment outcome data for European Union and European Economic Area countries are provisional.
Denmark
Data for Denmark exclude Greenland.
France
Data from France include data from 5 overseas departments (French Guiana, Guadeloupe, Martinique, Mayotte and
Réunion).
Russian Federation
e reported number of TB patients with known HIV status in 2010–2012 (Table A4.6) is for new TB patients in the civilian
sector only. It was not possible to calculate the percentage of all TB patients with known HIV status.

227 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
EUROPEAN REGION
7$%/($(VWLPDWHVRIWKHEXUGHQRIGLVHDVHFDXVHGE\7%±
YEAR POPULATION
(MILLIONS)
NUMBER
(THOUSANDS) RATE
a
NUMBER
(THOUSANDS) RATE
a
NUMBER
(THOUSANDS) RATE
a
MORTALITY (EXCLUDING HIV) PREVALENCE (INCLUDING HIV) INCIDENCE (INCLUDING HIV)
Albania 1990 3 0.11 (0.081–0.130) 3.1 (2.3–3.9) 1.2 (0.440–2.4) 36 (13–70) 0.84 (0.600–1.1) 24 (18–32)
1995 3 0.023 (0.018–0.028) 0.68 (0.55–0.83) 1.1 (0.420–2.1) 32 (12–61) 0.82 (0.680–0.970) 24 (20–29)
2000 3 0.027 (0.019–0.037) 0.82 (0.57–1.1) 0.98 (0.380–1.8) 30 (12–56) 0.75 (0.630–0.870) 23 (19–26)
2005 3 0.018 (0.013–0.025) 0.57 (0.39–0.79) 0.87 (0.370–1.6) 27 (11–49) 0.63 (0.530–0.730) 20 (17–23)
2010 3 0.012 (<0.01–0.018) 0.38 (0.23–0.58) 0.74 (0.320–1.3) 24 (10–43) 0.53 (0.450–0.620) 17 (14–20)
2011 3 0.011 (<0.01–0.017) 0.34 (0.20–0.53) 0.72 (0.310–1.3) 23 (9.7–42) 0.52 (0.440–0.610) 17 (14–19)
2012 3 <0.01 (<0.01–0.016) 0.31 (0.16–0.49) 0.68 (0.280–1.3) 22 (8.9–40) 0.51 (0.430–0.590) 16 (14–19)
Andorra 1990 < 1 <0.01 (<0.01–<0.01) 2.4 (0.16–7.6) 0.034 (0.013–0.064) 62 (24–118) 0.026 (0.023–0.030) 49 (43–55)
1995 < 1 <0.01 (<0.01–<0.01) 2.1 (<0.1–8.5) 0.033 (0.015–0.058) 51 (23–91) 0.023 (0.020–0.026) 37 (32–41)
2000 < 1 <0.01 (0–<0.01) 1.3 (0–6.2) 0.021 (<0.01–0.035) 31 (15–54) 0.014 (0.012–0.016) 21 (18–24)
2005 < 1 <0.01 (0–<0.01) 0.86 (0–3.9) 0.017 (<0.01–0.029) 21 (9.8–36) 0.012 (0.010–0.013) 14 (12–16)
2010 < 1 <0.01 (<0.01–<0.01) 0.58 (<0.1–2.3) 0.011 (<0.01–0.020) 14 (6.3–26) <0.01 (<0.01–<0.01) 10 (9.1–12)
2011 < 1 <0.01 (<0.01–<0.01) 0.32 (0.16–0.54) <0.01 (<0.01–0.015) 9 (2.7–19) <0.01 (<0.01–<0.01) 4.4 (3.9–5.0)
2012 < 1 <0.01 (0–<0.01) 0.91 (0–4.9) 0.017 (<0.01–0.028) 21 (11–36) 0.01 (<0.01–0.012) 13 (12–15)
Armenia 1990 4 0.16 (0.110–0.200) 4.4 (3.2–5.8) 1 (0.420–1.8) 28 (12–52) 0.63 (0.470–0.810) 18 (13–23)
1995 3 0.19 (0.160–0.230) 6 (4.9–7.2) 1.9 (0.890–3.3) 59 (28–101) 1.2 (1.0–1.4) 38 (32–44)
2000 3 0.19 (0.170–0.220) 6.3 (5.6–7.0) 2.9 (1.4–4.8) 93 (46–158) 1.9 (1.6–2.1) 61 (53–68)
2005 3 0.26 (0.190–0.320) 8.5 (6.5–11) 3.5 (1.7–6.0) 118 (58–198) 2.3 (2.1–2.6) 77 (68–87)
2010 3 0.23 (0.180–0.280) 7.7 (6.1–9.5) 2.7 (1.2–4.8) 92 (42–161) 1.8 (1.6–2.2) 62 (53–73)
2011 3 0.17 (0.130–0.210) 5.6 (4.5–6.9) 2.3 (1.0–4.2) 79 (34–142) 1.6 (1.3–1.9) 55 (45–65)
2012 3 0.19 (0.150–0.230) 6.3 (5.1–7.6) 2.4 (1.1–4.1) 79 (37–137) 1.5 (1.3–1.8) 52 (43–61)
Austria 1990 8 0.14 (0.140–0.140) 1.8 (1.8–1.9) 2.5 (1.1–4.5) 33 (15–58) 1.7 (1.5–2.0) 23 (20–26)
1995 8 0.074 (0.074–0.074) 0.93 (0.92–0.93) 2.5 (1.2–4.4) 32 (15–55) 1.7 (1.5–1.9) 21 (19–24)
2000 8 0.069 (0.069–0.069) 0.86 (0.86–0.86) 2 (0.890–3.5) 25 (11–43) 1.4 (1.2–1.5) 17 (15–19)
2005 8 0.05 (0.050–0.051) 0.61 (0.61–0.61) 1.5 (0.690–2.7) 19 (8.4–33) 1.1 (0.940–1.2) 13 (11–15)
2010 8 0.032 (0.032–0.033) 0.38 (0.38–0.39) 1 (0.420–1.9) 12 (5.0–23) 0.76 (0.660–0.860) 9 (7.9–10)
2011 8 0.04 (0.040–0.041) 0.48 (0.47–0.49) 1.1 (0.510–2.0) 13 (6.0–23) 0.77 (0.680–0.870) 9.2 (8.0–10)
2012 8 0.035 (0.035–0.036) 0.42 (0.41–0.42) 0.91 (0.370–1.7) 11 (4.4–20) 0.67 (0.590–0.760) 7.9 (6.9–8.9)
Azerbaijan 1990 7 0.82 (0.610–1.1) 11 (8.5–15) 54 (25–94) 744 (343–1 300) 22 (18–26) 305 (252–363)
1995 8 1.8 (1.3–2.3) 23 (17–29) 120 (56–220) 1 600 (717–2 820) 49 (41–59) 637 (526–759)
2000 8 1.8 (1.4–2.2) 22 (17–27) 140 (62–240) 1 690 (768–2 970) 55 (46–66) 682 (563–813)
2005 9 0.82 (0.660–1.0) 9.6 (7.7–12) 66 (31–110) 776 (366–1 340) 29 (24–34) 335 (276–398)
2010 9 0.39 (0.330–0.440) 4.2 (3.7–4.9) 20 (9.9–34) 221 (109–371) 12 (9.8–14) 131 (108–156)
2011 9 0.39 (0.340–0.450) 4.2 (3.7–4.9) 16 (7.3–28) 172 (79–302) 10 (8.6–12) 113 (93–135)
2012 9 0.39 (0.340–0.450) 4.2 (3.7–4.9) 12 (4.1–23) 124 (44–245) 8.9 (7.3–11) 95 (78–114)
Belarus 1990 10 0.5 (0.470–0.540) 4.9 (4.6–5.2) 5.2 (2.2–9.5) 51 (22–93) 3.5 (2.8–4.3) 34 (27–42)
1995 10 0.76 (0.700–0.830) 7.5 (6.9–8.1) 11 (5.1–19) 106 (51–182) 6.9 (5.9–8.1) 68 (58–80)
2000 10 0.8 (0.760–0.850) 8.1 (7.6–8.5) 13 (6.0–23) 130 (60–225) 8.4 (6.9–9.9) 84 (69–100)
2005 10 1.1 (0.990–1.1) 11 (10–12) 11 (4.4–19) 109 (46–199) 6.9 (5.3–8.8) 72 (55–91)
2010 9 0.76 (0.700–0.820) 8 (7.3–8.6) 10 (4.6–18) 107 (48–189) 6.7 (5.3–8.1) 70 (56–86)
2011 9 0.66 (0.600–0.720) 7 (6.3–7.7) 10 (4.6–18) 107 (49–188) 6.6 (5.4–8.0) 70 (57–85)
2012 9 0.57 (0.510–0.630) 6 (5.4–6.7) 10 (4.7–18) 108 (50–188) 6.6 (5.4–8.0) 70 (57–85)
Belgium 1990 10 0.1 (0.097–0.100) 1 (0.97–1.0) 2.6 (1.1–4.6) 26 (11–46) 1.8 (1.6–2.1) 18 (16–21)
1995 10 0.13 (0.130–0.130) 1.3 (1.3–1.3) 2.2 (0.930–3.9) 21 (9.1–39) 1.6 (1.4–1.8) 16 (14–18)
2000 10 0.081 (0.080–0.083) 0.79 (0.78–0.81) 2.1 (0.920–3.7) 20 (9.0–36) 1.5 (1.3–1.7) 14 (13–16)
2005 11 0.062 (0.062–0.063) 0.59 (0.59–0.60) 1.7 (0.690–3.0) 16 (6.6–29) 1.2 (1.1–1.4) 12 (10–13)
2010 11 0.043 (0.043–0.044) 0.4 (0.39–0.40) 1.7 (0.740–2.9) 15 (6.8–27) 1.2 (1.0–1.3) 11 (9.5–12)
2011 11 0.041 (0.041–0.042) 0.38 (0.37–0.38) 1.5 (0.660–2.8) 14 (6.0–25) 1.1 (0.990–1.3) 10 (9.0–12)
2012 11 0.04 (0.039–0.040) 0.36 (0.35–0.36) 1.4 (0.560–2.6) 13 (5.1–24) 1.1 (0.940–1.2) 9.7 (8.5–11)
Bosnia and 1990 5 0.46 (0.440–0.480) 10 (9.7–11) 6.5 (1.9–14) 145 (43–307) 4.2 (2.6–6.2) 94 (58–138)
Herzegovina 1995 4 0.22 (0.210–0.230) 6.3 (5.9–6.6) 4.6 (2.1–8.1) 131 (59–229) 3 (2.4–3.6) 84 (69–101)
2000 4 0.23 (0.210–0.240) 5.9 (5.5–6.3) 2.8 (0.830–5.9) 73 (22–154) 2.4 (2.0–2.9) 63 (51–75)
2005 4 0.21 (0.200–0.230) 5.5 (5.0–6.0) 2.3 (0.640–5.0) 59 (17–129) 2 (1.7–2.4) 52 (43–63)
2010 4 0.2 (0.180–0.220) 5.2 (4.6–5.7) 2.6 (1.1–4.7) 67 (28–123) 1.9 (1.6–2.2) 50 (43–57)
2011 4 0.2 (0.170–0.220) 5.1 (4.6–5.7) 2.7 (1.2–4.8) 70 (31–124) 1.9 (1.6–2.2) 49 (42–56)
2012 4 0.2 (0.180–0.220) 5.2 (4.6–5.8) 2.8 (1.3–4.8) 73 (35–126) 1.9 (1.6–2.1) 49 (42–56)
Bulgaria 1990 9 0.22 (0.210–0.220) 2.4 (2.4–2.5) 4.2 (1.9–7.5) 48 (21–85) 2.9 (2.5–3.3) 33 (29–37)
1995 8 0.34 (0.340–0.350) 4.1 (4.0–4.2) 8.4 (4.2–14) 101 (51–169) 5.2 (4.5–5.9) 62 (54–71)
2000 8 0.59 (0.570–0.600) 7.3 (7.2–7.5) 7 (3.3–12) 88 (42–151) 4.6 (4.0–5.3) 58 (50–66)
2005 8 0.26 (0.260–0.270) 3.4 (3.4–3.5) 6.2 (3.0–11) 81 (39–139) 4.1 (3.6–4.6) 53 (46–61)
2010 7 0.19 (0.190–0.190) 2.6 (2.5–2.6) 3.9 (1.6–7.1) 53 (22–97) 2.8 (2.5–3.2) 38 (33–43)
2011 7 0.16 (0.160–0.160) 2.2 (2.2–2.2) 3.5 (1.4–6.4) 48 (20–88) 2.6 (2.2–2.9) 35 (30–40)
2012 7 0.15 (0.150–0.150) 2 (2.0–2.1) 3.1 (1.3–5.8) 43 (17–80) 2.3 (2.0–2.6) 32 (28–36)
Croatia 1990 5 0.39 (0.380–0.410) 8.2 (8.0–8.5) 4.4 (2.1–7.7) 92 (43–160) 3 (2.6–3.4) 62 (54–70)
1995 5 0.25 (0.240–0.270) 5.4 (5.0–5.7) 3.3 (1.3–6.1) 70 (28–130) 2.4 (2.1–2.8) 52 (45–59)
2000 4 0.19 (0.180–0.200) 4.2 (4.0–4.4) 2.5 (1.0–4.7) 57 (23–105) 1.9 (1.6–2.1) 42 (37–47)
2005 4 0.11 (0.110–0.110) 2.5 (2.5–2.5) 1.6 (0.620–3.0) 36 (14–69) 1.2 (1.1–1.4) 28 (24–31)
2010 4 0.082 (0.082–0.083) 1.9 (1.9–1.9) 1.1 (0.420–2.0) 24 (9.7–46) 0.79 (0.690–0.900) 18 (16–21)
2011 4 0.066 (0.066–0.067) 1.5 (1.5–1.5) 0.96 (0.390–1.8) 22 (9.1–41) 0.71 (0.620–0.810) 16 (14–19)
2012 4 0.061 (0.060–0.062) 1.4 (1.4–1.4) 0.84 (0.340–1.6) 20 (7.9–36) 0.62 (0.540–0.700) 14 (13–16)
Cyprus 1990 < 1 <0.01 (<0.01–<0.01) 0.2 (0.16–0.25) 0.038 (0.011–0.080) 5 (1.5–10) 0.033 (0.029–0.038) 4.4 (3.8–4.9)
1995 < 1 <0.01 (<0.01–<0.01) 0.2 (0.16–0.25) 0.05 (0.017–0.100) 5.8 (2.0–12) 0.041 (0.036–0.047) 4.8 (4.2–5.5)
2000 < 1 0 (0–0) 0 (0–0) 0.045 (0.015–0.091) 4.8 (1.6–9.6) 0.038 (0.033–0.043) 4 (3.5–4.6)
2005 1 <0.01 (<0.01–<0.01) 0.37 (0.32–0.41) 0.051 (0.020–0.095) 4.9 (2.0–9.2) 0.039 (0.034–0.044) 3.8 (3.3–4.3)
2010 1 <0.01 (<0.01–<0.01) 0.11 (0.10–0.13) 0.1 (0.049–0.180) 9.4 (4.4–16) 0.07 (0.061–0.079) 6.4 (5.6–7.2)
2011 1 <0.01 (<0.01–<0.01) 0.21 (0.19–0.23) 0.069 (0.023–0.140) 6.2 (2.1–13) 0.059 (0.051–0.066) 5.3 (4.6–5.9)
2012 1 <0.01 (<0.01–<0.01) 0.2 (0.16–0.25) 0.069 (0.021–0.150) 6.1 (1.8–13) 0.061 (0.053–0.069) 5.4 (4.7–6.1)
Czech Republic 1990 10 0.19 (0.190–0.190) 1.8 (1.8–1.8) 3.1 (1.3–5.6) 30 (12–54) 2.2 (2.0–2.5) 22 (19–24)
1995 10 0.092 (0.091–0.092) 0.89 (0.88–0.89) 2.8 (1.1–5.3) 27 (11–51) 2.1 (1.8–2.4) 20 (18–23)
2000 10 0.12 (0.120–0.120) 1.2 (1.2–1.2) 2.2 (0.880–4.1) 21 (8.6–40) 1.6 (1.4–1.8) 16 (14–18)
2005 10 0.065 (0.064–0.065) 0.63 (0.63–0.63) 1.6 (0.690–2.8) 15 (6.7–28) 1.1 (0.980–1.3) 11 (9.6–12)
2010 11 0.035 (0.035–0.035) 0.33 (0.33–0.34) 0.99 (0.420–1.8) 9.4 (4.0–17) 0.72 (0.630–0.820) 6.8 (6.0–7.7)
2011 11 0.051 (0.050–0.051) 0.48 (0.47–0.48) 0.9 (0.380–1.6) 8.5 (3.6–16) 0.65 (0.570–0.740) 6.2 (5.4–7.0)
2012 11 0.037 (0.037–0.037) 0.35 (0.35–0.35) 0.77 (0.310–1.4) 7.2 (2.9–13) 0.57 (0.500–0.640) 5.3 (4.7–6.0)
Denmark 1990 5 0.054 (0.053–0.056) 1.1 (1.0–1.1) 0.61 (0.300–1.0) 12 (5.8–20) 0.4 (0.350–0.460) 7.8 (6.9–8.9)
1995 5 0.024 (0.023–0.025) 0.47 (0.45–0.48) 0.64 (0.220–1.3) 12 (4.3–24) 0.52 (0.450–0.580) 9.8 (8.6–11)
2000 5 0.021 (0.020–0.021) 0.38 (0.38–0.39) 1 (0.490–1.7) 19 (9.1–33) 0.68 (0.590–0.760) 13 (11–14)
2005 5 0.019 (0.019–0.020) 0.36 (0.35–0.36) 0.67 (0.310–1.2) 12 (5.7–21) 0.45 (0.400–0.510) 8.4 (7.3–9.5)
2010 6 0.035 (0.034–0.036) 0.63 (0.61–0.66) 0.46 (0.170–0.880) 8.3 (3.1–16) 0.36 (0.320–0.410) 6.5 (5.7–7.3)
2011 6 0.017 (0.016–0.018) 0.3 (0.28–0.32) 0.58 (0.260–1.0) 10 (4.6–19) 0.41 (0.360–0.470) 7.4 (6.5–8.4)
2012 6 0.022 (0.021–0.023) 0.4 (0.38–0.42) 0.56 (0.240–1.0) 10 (4.2–18) 0.41 (0.360–0.470) 7.4 (6.5–8.4)
Estonia 1990 2 0.071 (0.070–0.072) 4.5 (4.5–4.6) 0.81 (0.410–1.3) 52 (26–85) 0.49 (0.430–0.550) 31 (27–35)
1995 1 0.15 (0.140–0.150) 10 (9.9–10) 0.93 (0.350–1.8) 65 (24–125) 0.72 (0.630–0.810) 50 (44–57)
2000 1 0.11 (0.110–0.110) 8 (7.9–8.2) 1.3 (0.610–2.3) 97 (45–168) 0.91 (0.800–1.0) 67 (58–75)
2005 1 0.049 (0.048–0.050) 3.7 (3.6–3.8) 0.7 (0.280–1.3) 53 (21–98) 0.55 (0.480–0.620) 42 (36–47)
2010 1 0.036 (0.035–0.036) 2.7 (2.7–2.8) 0.37 (0.130–0.740) 29 (10–57) 0.33 (0.290–0.370) 25 (22–28)
2011 1 0.036 (0.036–0.037) 2.8 (2.8–2.8) 0.44 (0.200–0.780) 34 (15–60) 0.34 (0.300–0.390) 26 (23–30)
2012 1 0.036 (0.036–0.036) 2.8 (2.8–2.8) 0.38 (0.160–0.680) 29 (13–52) 0.3 (0.260–0.340) 23 (20–26)
Finland 1990 5 0.13 (0.130–0.130) 2.5 (2.5–2.5) 1.3 (0.550–2.3) 25 (11–45) 0.89 (0.780–1.0) 18 (16–20)
1995 5 0.092 (0.092–0.092) 1.8 (1.8–1.8) 1.1 (0.500–1.9) 22 (9.8–38) 0.76 (0.670–0.860) 15 (13–17)
2000 5 0.083 (0.083–0.084) 1.6 (1.6–1.6) 0.85 (0.370–1.5) 16 (7.2–30) 0.61 (0.530–0.690) 12 (10–13)
2005 5 0.037 (0.037–0.037) 0.71 (0.71–0.71) 0.55 (0.240–0.980) 10 (4.5–19) 0.39 (0.340–0.440) 7.4 (6.5–8.4)
2010 5 0.016 (0.016–0.016) 0.3 (0.30–0.30) 0.48 (0.190–0.900) 9 (3.6–17) 0.36 (0.310–0.410) 6.7 (5.9–7.6)
2011 5 0.021 (0.021–0.021) 0.39 (0.39–0.39) 0.48 (0.190–0.900) 8.9 (3.6–17) 0.36 (0.310–0.410) 6.7 (5.8–7.5)
2012 5 0.015 (0.015–0.015) 0.29 (0.28–0.29) 0.39 (0.130–0.790) 7.2 (2.4–15) 0.3 (0.260–0.340) 5.5 (4.9–6.3)
France 1990 57 1 (0.980–1.0) 1.8 (1.7–1.8) 16 (7.7–26) 28 (14–47) 11 (11–12) 20 (19–21)
1995 58 0.79 (0.760–0.810) 1.4 (1.3–1.4) 16 (8.5–27) 28 (15–46) 11 (10–12) 19 (18–20)
2000 59 0.65 (0.630–0.670) 1.1 (1.1–1.1) 11 (5.6–19) 19 (9.4–31) 7.7 (7.2–8.1) 13 (12–14)
2005 61 0.43 (0.410–0.440) 0.7 (0.68–0.72) 8.5 (4.0–15) 14 (6.4–24) 6.3 (5.9–6.7) 10 (9.5–11)
2010 63 0.33 (0.320–0.350) 0.53 (0.51–0.55) 8.9 (4.6–15) 14 (7.3–23) 6 (5.6–6.4) 9.5 (8.9–10)
2011 64 0.31 (0.300–0.330) 0.5 (0.47–0.52) 8.7 (4.5–14) 14 (7.1–22) 5.9 (5.5–6.2) 9.2 (8.6–9.8)
2012 64 0.3 (0.280–0.310) 0.46 (0.44–0.49) 7.4 (3.7–12) 12 (5.7–20) 5.3 (4.9–5.6) 8.2 (7.7–8.7)
a
Rates are per 100 000 population.

GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($(VWLPDWHVRIWKHEXUGHQRIGLVHDVHFDXVHGE\7%±
YEAR POPULATION
(MILLIONS)
NUMBER
(THOUSANDS) RATE
a
NUMBER
(THOUSANDS) RATE
a
NUMBER
(THOUSANDS) RATE
a
MORTALITY (EXCLUDING HIV) PREVALENCE (INCLUDING HIV) INCIDENCE (INCLUDING HIV)
Georgia 1990 5 0.48 (0.430–0.550) 8.9 (7.8–10) 38 (18–67) 704 (326–1 220) 15 (14–17) 280 (250–312)
1995 5 0.42 (0.360–0.470) 8.2 (7.2–9.3) 29 (15–48) 571 (290–944) 13 (12–15) 263 (234–293)
2000 5 0.37 (0.320–0.420) 7.7 (6.7–8.8) 24 (13–40) 516 (270–840) 12 (11–14) 256 (228–285)
2005 4 0.17 (0.150–0.200) 3.8 (3.3–4.5) 14 (7.5–23) 315 (168–508) 7.8 (7.0–8.7) 175 (156–195)
2010 4 0.67 (0.370–1.1) 15 (8.4–24) 8.2 (3.8–14) 186 (87–323) 5.6 (5.0–6.2) 128 (114–142)
2011 4 0.2 (0.160–0.240) 4.5 (3.7–5.5) 7.9 (3.7–14) 182 (84–316) 5.5 (4.9–6.1) 125 (112–140)
2012 4 0.2 (0.160–0.240) 4.5 (3.7–5.5) 6.9 (2.9–13) 158 (67–288) 5 (4.5–5.6) 116 (103–130)
Germany 1990 80 1.1 (1.0–1.1) 1.3 (1.3–1.3) 23 (10–42) 29 (13–53) 17 (15–19) 21 (18–24)
1995 83 1.2 (1.2–1.2) 1.4 (1.4–1.5) 19 (7.8–35) 23 (9.3–42) 14 (12–16) 17 (15–19)
2000 84 0.49 (0.490–0.500) 0.59 (0.58–0.60) 15 (6.6–26) 18 (8.0–32) 10 (9.1–12) 12 (11–14)
2005 84 0.32 (0.320–0.330) 0.39 (0.38–0.39) 9.3 (4.1–17) 11 (4.9–20) 6.6 (5.7–7.4) 7.8 (6.9–8.8)
2010 83 0.3 (0.290–0.300) 0.36 (0.35–0.36) 6.4 (2.7–12) 7.7 (3.2–14) 4.7 (4.1–5.3) 5.6 (4.9–6.4)
2011 83 0.29 (0.280–0.290) 0.35 (0.34–0.35) 6.6 (2.9–12) 7.9 (3.5–14) 4.7 (4.1–5.3) 5.7 (5.0–6.4)
2012 83 0.29 (0.280–0.290) 0.35 (0.34–0.35) 6.4 (2.8–12) 7.8 (3.3–14) 4.6 (4.1–5.3) 5.6 (4.9–6.4)
Greece 1990 10 0.16 (0.160–0.170) 1.6 (1.5–1.7) 1.5 (0.690–2.6) 15 (6.8–25) 1 (0.880–1.1) 9.9 (8.7–11)
1995 11 0.16 (0.150–0.170) 1.5 (1.4–1.6) 1.5 (0.620–2.7) 14 (5.8–25) 1.1 (0.950–1.2) 10 (8.9–11)
2000 11 0.089 (0.085–0.092) 0.81 (0.77–0.84) 1 (0.390–2.0) 9.5 (3.6–18) 0.81 (0.710–0.920) 7.4 (6.4–8.3)
2005 11 0.094 (0.090–0.098) 0.85 (0.81–0.89) 1.2 (0.590–2.1) 11 (5.3–19) 0.8 (0.700–0.900) 7.2 (6.3–8.2)
2010 11 0.074 (0.070–0.078) 0.66 (0.63–0.70) 0.62 (0.200–1.3) 5.6 (1.8–11) 0.51 (0.450–0.580) 4.6 (4.0–5.2)
2011 11 0.078 (0.075–0.082) 0.7 (0.67–0.74) 0.7 (0.280–1.3) 6.3 (2.6–12) 0.52 (0.460–0.590) 4.7 (4.1–5.3)
2012 11 0.076 (0.073–0.080) 0.69 (0.65–0.72) 0.71 (0.310–1.3) 6.3 (2.8–11) 0.5 (0.440–0.570) 4.5 (3.9–5.1)
Greenland 1990 < 1 <0.01 (<0.01–0.017) 9.5 (0.61–30) 0.14 (0.053–0.260) 245 (96–462) 0.11 (0.093–0.120) 191 (167–216)
1995 < 1 <0.01 (<0.01–0.017) 9.5 (0.61–30) 0.14 (0.054–0.260) 245 (96–463) 0.11 (0.093–0.120) 191 (167–216)
2000 < 1 <0.01 (<0.01–0.017) 9.5 (0.61–30) 0.14 (0.053–0.260) 245 (95–464) 0.11 (0.094–0.120) 191 (167–216)
2005 < 1 <0.01 (<0.01–0.018) 9.6 (0.54–31) 0.14 (0.056–0.260) 247 (98–463) 0.11 (0.095–0.120) 191 (167–216)
2010 < 1 <0.01 (<0.01–0.038) 14 (<0.1–66) 0.2 (0.092–0.340) 345 (163–595) 0.13 (0.110–0.150) 232 (203–262)
2011 < 1 <0.01 (<0.01–0.038) 14 (<0.1–66) 0.2 (0.092–0.340) 346 (163–596) 0.13 (0.120–0.150) 234 (205–264)
2012 < 1 <0.01 (<0.01–<0.01) 6.7 (1.8–15) 0.11 (0.032–0.230) 190 (57–401) 0.097 (0.085–0.110) 170 (149–193)
Hungary 1990 10 0.55 (0.550–0.550) 5.3 (5.3–5.3) 5.5 (2.3–10) 53 (22–96) 4 (3.5–4.5) 39 (34–44)
1995 10 0.57 (0.570–0.580) 5.5 (5.5–5.6) 6.9 (3.0–12) 67 (29–120) 4.9 (4.3–5.6) 48 (42–54)
2000 10 0.36 (0.350–0.360) 3.5 (3.5–3.5) 5.2 (2.2–9.6) 51 (22–94) 3.8 (3.3–4.3) 37 (33–42)
2005 10 0.18 (0.180–0.180) 1.8 (1.8–1.8) 3 (1.2–5.5) 29 (12–55) 2.2 (1.9–2.5) 22 (19–25)
2010 10 0.1 (0.100–0.100) 1 (1.0–1.0) 2.6 (1.3–4.5) 26 (13–45) 1.7 (1.5–2.0) 17 (15–19)
2011 10 0.075 (0.075–0.075) 0.75 (0.75–0.75) 2.9 (1.4–4.8) 29 (14–48) 1.8 (1.6–2.0) 18 (16–20)
2012 10 0.073 (0.073–0.073) 0.73 (0.73–0.73) 2.9 (1.4–4.8) 29 (14–48) 1.8 (1.6–2.0) 18 (16–20)
Iceland 1990 < 1 <0.01 (<0.01–<0.01) 0.4 (0.40–0.40) 0.03 (0.014–0.053) 12 (5.5–21) 0.021 (0.018–0.023) 8.1 (7.1–9.2)
1995 < 1 <0.01 (<0.01–<0.01) 0.71 (0.71–0.71) 0.017 (<0.01–0.034) 6.3 (2.2–13) 0.014 (0.012–0.016) 5.2 (4.5–5.8)
2000 < 1 <0.01 (<0.01–<0.01) 0.36 (0.36–0.37) 0.023 (0.011–0.038) 8.1 (4.0–14) 0.015 (0.013–0.017) 5.3 (4.7–6.0)
2005 < 1 <0.01 (<0.01–<0.01) 0.33 (0.33–0.33) 0.016 (<0.01–0.028) 5.3 (2.4–9.3) 0.012 (0.010–0.013) 3.9 (3.4–4.4)
2010 < 1 <0.01 (<0.01–<0.01) 0.29 (0.29–0.29) 0.046 (0.024–0.075) 14 (7.5–24) 0.025 (0.022–0.029) 8 (7.0–9.0)
2011 < 1 <0.01 (<0.01–<0.01) 0.28 (0.28–0.28) 0.013 (<0.01–0.027) 4.1 (1.4–8.4) <0.01 (<0.01–0.010) 2.9 (2.5–3.2)
2012 < 1 <0.01 (<0.01–<0.01) 0.27 (0.27–0.27) 0.014 (<0.01–0.028) 4.3 (1.5–8.5) 0.012 (0.010–0.013) 3.5 (3.1–4.0)
Ireland 1990 4 0.051 (0.051–0.052) 1.5 (1.4–1.5) 0.94 (0.360–1.8) 27 (10–51) 0.72 (0.630–0.810) 20 (18–23)
1995 4 0.036 (0.036–0.036) 1 (1.0–1.0) 0.68 (0.260–1.3) 19 (7.1–36) 0.53 (0.460–0.600) 15 (13–17)
2000 4 0.059 (0.058–0.059) 1.5 (1.5–1.5) 0.59 (0.240–1.1) 16 (6.2–29) 0.44 (0.390–0.500) 12 (10–13)
2005 4 0.015 (0.015–0.015) 0.37 (0.37–0.37) 0.68 (0.290–1.2) 16 (7.1–29) 0.49 (0.430–0.550) 12 (10–13)
2010 4 0.027 (0.027–0.027) 0.61 (0.61–0.61) 0.61 (0.250–1.1) 14 (5.5–25) 0.46 (0.400–0.520) 10 (8.9–12)
2011 5 0.02 (0.019–0.020) 0.43 (0.43–0.43) 0.64 (0.280–1.1) 14 (6.2–25) 0.46 (0.400–0.520) 10 (8.9–11)
2012 5 0.018 (0.018–0.018) 0.39 (0.38–0.39) 0.5 (0.180–0.970) 11 (4.0–21) 0.39 (0.340–0.440) 8.6 (7.5–9.7)
Israel 1990 4 0.02 (0.020–0.021) 0.45 (0.43–0.46) 0.39 (0.130–0.780) 8.6 (2.9–17) 0.27 (0.240–0.300) 6 (5.2–6.8)
1995 5 0.072 (0.069–0.074) 1.3 (1.3–1.4) 0.56 (0.190–1.1) 10 (3.6–21) 0.46 (0.400–0.520) 8.6 (7.5–9.7)
2000 6 0.034 (0.034–0.035) 0.57 (0.56–0.58) 0.85 (0.370–1.5) 14 (6.1–25) 0.62 (0.540–0.700) 10 (9.0–12)
2005 7 0.022 (0.022–0.023) 0.34 (0.33–0.35) 0.54 (0.200–1.0) 8.1 (3.1–16) 0.43 (0.370–0.480) 6.5 (5.7–7.3)
2010 7 0.017 (0.017–0.018) 0.23 (0.23–0.24) 0.46 (0.150–0.940) 6.1 (2.0–13) 0.39 (0.340–0.440) 5.3 (4.6–6.0)
2011 8 0.017 (0.017–0.018) 0.23 (0.22–0.23) 0.63 (0.260–1.2) 8.3 (3.5–15) 0.47 (0.420–0.540) 6.3 (5.5–7.1)
2012 8 0.017 (0.017–0.018) 0.23 (0.22–0.23) 0.85 (0.400–1.5) 11 (5.2–19) 0.58 (0.510–0.660) 7.6 (6.7–8.6)
Italy 1990 57 0.61 (0.590–0.630) 1.1 (1.0–1.1) 7 (3.2–12) 12 (5.6–22) 4.9 (4.3–5.5) 8.6 (7.5–9.7)
1995 57 0.68 (0.660–0.690) 1.2 (1.2–1.2) 9.7 (4.6–17) 17 (8.1–29) 6.5 (5.7–7.3) 11 (10–13)
2000 57 0.5 (0.480–0.520) 0.87 (0.84–0.91) 5.1 (1.7–10) 9 (3.0–18) 4 (3.5–4.6) 7.1 (6.2–8.0)
2005 59 0.37 (0.370–0.370) 0.63 (0.63–0.64) 6.1 (2.6–11) 10 (4.4–19) 4.4 (3.9–5.0) 7.5 (6.6–8.5)
2010 61 0.3 (0.300–0.300) 0.49 (0.49–0.50) 4.5 (1.6–9.0) 7.5 (2.6–15) 3.7 (3.2–4.1) 6 (5.3–6.8)
2011 61 0.28 (0.280–0.280) 0.46 (0.46–0.47) 5.3 (2.2–9.8) 8.8 (3.6–16) 3.9 (3.4–4.5) 6.5 (5.7–7.3)
2012 61 0.26 (0.260–0.270) 0.43 (0.43–0.44) 5.7 (2.5–10) 9.4 (4.1–17) 4.1 (3.6–4.6) 6.7 (5.8–7.5)
Kazakhstan 1990 16 2.1 (1.9–2.3) 13 (11–14) 19 (8.3–33) 116 (51–207) 13 (11–15) 79 (66–92)
1995 16 5.2 (4.8–5.6) 33 (31–36) 110 (54–180) 706 (347–1 190) 50 (42–58) 318 (269–372)
2000 15 4.8 (4.3–5.3) 33 (29–37) 97 (50–160) 668 (344–1 100) 51 (43–60) 351 (297–411)
2005 15 4.2 (3.8–4.5) 28 (25–30) 51 (23–92) 340 (149–608) 35 (30–41) 235 (199–275)
2010 16 2.1 (1.9–2.4) 13 (12–15) 42 (19–75) 266 (118–472) 29 (24–34) 182 (154–213)
2011 16 1.7 (1.5–1.9) 11 (9.1–12) 50 (25–85) 312 (154–526) 31 (26–36) 193 (163–225)
2012 16 1.3 (1.0–1.5) 7.8 (6.3–9.3) 31 (12–57) 189 (77–350) 22 (19–26) 137 (116–160)
Kyrgyzstan 1990 4 0.4 (0.340–0.470) 9.1 (7.6–11) 7.5 (3.8–12) 170 (86–283) 4 (3.3–4.8) 92 (76–109)
1995 5 0.72 (0.620–0.830) 16 (13–18) 15 (7.5–25) 326 (164–542) 7.7 (6.4–9.2) 168 (138–200)
2000 5 1.3 (1.1–1.4) 25 (23–28) 22 (11–37) 449 (227–747) 12 (10–15) 249 (205–296)
2005 5 0.82 (0.810–0.830) 16 (16–16) 17 (8.0–29) 334 (159–571) 10 (8.6–12) 208 (171–248)
2010 5 0.61 (0.610–0.610) 11 (11–12) 11 (4.7–20) 204 (89–367) 7.5 (6.2–9.0) 141 (116–168)
2011 5 0.57 (0.560–0.570) 10 (10–11) 11 (5.2–20) 211 (96–370) 7.6 (6.3–9.1) 141 (116–168)
2012 5 0.52 (0.510–0.530) 9.5 (9.3–9.8) 12 (5.5–21) 217 (101–376) 7.7 (6.4–9.2) 141 (116–168)
Latvia 1990 3 0.19 (0.190–0.190) 7.2 (7.1–7.3) 3 (1.6–4.9) 114 (60–186) 1.5 (1.3–1.7) 57 (50–65)
1995 2 0.34 (0.340–0.350) 14 (14–14) 5.9 (3.1–9.6) 237 (125–385) 3.1 (2.7–3.5) 126 (111–142)
2000 2 0.3 (0.290–0.310) 13 (12–13) 4.6 (2.3–7.6) 194 (99–321) 2.9 (2.5–3.2) 121 (106–137)
2005 2 0.18 (0.180–0.190) 8.1 (7.9–8.3) 2.3 (0.990–4.1) 102 (45–183) 1.7 (1.5–1.9) 75 (66–85)
2010 2 0.083 (0.080–0.086) 4 (3.8–4.1) 1.3 (0.540–2.4) 63 (26–117) 1 (0.930–1.2) 50 (45–56)
2011 2 0.068 (0.066–0.070) 3.3 (3.2–3.4) 1.2 (0.520–2.3) 60 (25–111) 0.99 (0.890–1.1) 48 (43–53)
2012 2 0.053 (0.052–0.054) 2.6 (2.5–2.6) 1.6 (0.780–2.6) 76 (38–127) 1.1 (1.0–1.2) 53 (49–58)
Lithuania 1990 4 0.26 (0.260–0.260) 7 (7.0–7.0) 2.5 (1.2–4.4) 69 (32–118) 1.6 (1.4–1.9) 44 (37–52)
1995 4 0.49 (0.490–0.500) 14 (13–14) 4.9 (2.4–8.5) 136 (65–234) 3.2 (2.8–3.7) 89 (77–102)
2000 3 0.37 (0.360–0.370) 11 (10–11) 5.5 (2.7–9.3) 157 (78–265) 3.6 (3.2–4.0) 103 (92–114)
2005 3 0.36 (0.360–0.360) 11 (11–11) 4.2 (2.0–7.2) 127 (60–219) 2.8 (2.5–3.2) 87 (76–97)
2010 3 0.21 (0.210–0.210) 6.9 (6.8–6.9) 3.1 (1.4–5.6) 103 (46–181) 2.2 (1.9–2.5) 73 (63–82)
2011 3 0.15 (0.150–0.150) 5 (4.9–5.0) 3 (1.3–5.2) 97 (44–171) 2.1 (1.8–2.4) 69 (61–78)
2012 3 0.09 (0.088–0.091) 3 (2.9–3.0) 2.8 (1.3–5.0) 93 (42–164) 2 (1.8–2.3) 66 (58–75)
Luxembourg 1990 < 1 <0.01 (<0.01–<0.01) 0.55 (0.54–0.56) 0.083 (0.039–0.140) 22 (10–37) 0.055 (0.048–0.062) 14 (13–16)
1995 < 1 0 (0–0) 0 (0–0) 0.045 (0.015–0.091) 11 (3.7–22) 0.037 (0.032–0.042) 9 (7.9–10)
2000 < 1 <0.01 (<0.01–<0.01) 0.24 (0.23–0.24) 0.076 (0.036–0.130) 17 (8.4–30) 0.051 (0.044–0.057) 12 (10–13)
2005 < 1 <0.01 (<0.01–<0.01) 0.22 (0.21–0.22) 0.06 (0.026–0.110) 13 (5.7–23) 0.043 (0.037–0.048) 9.3 (8.1–11)
2010 < 1 <0.01 (<0.01–<0.01) 0.19 (0.19–0.20) 0.045 (0.019–0.083) 8.9 (3.8–16) 0.033 (0.029–0.038) 6.6 (5.8–7.4)
2011 < 1 <0.01 (<0.01–<0.01) 0.19 (0.19–0.19) 0.036 (0.013–0.071) 7 (2.5–14) 0.029 (0.025–0.033) 5.6 (4.9–6.3)
2012 < 1 <0.01 (<0.01–<0.01) 0.42 (0.41–0.43) 0.053 (0.026–0.089) 10 (4.9–17) 0.034 (0.030–0.039) 6.5 (5.7–7.4)
Malta 1990 < 1 <0.01 (<0.01–<0.01) 0.28 (0.27–0.29) 0.025 (<0.01–0.052) 6.8 (2.2–14) 0.015 (0.013–0.017) 4 (3.5–4.5)
1995 < 1 <0.01 (<0.01–<0.01) 0.27 (0.26–0.28) 0.024 (<0.01–0.049) 6.1 (2.0–12) 0.013 (0.011–0.014) 3.2 (2.8–3.6)
2000 < 1 <0.01 (<0.01–<0.01) 0.25 (0.25–0.25) 0.025 (0.011–0.046) 6.2 (2.7–11) 0.018 (0.016–0.021) 4.5 (4.0–5.1)
2005 < 1 <0.01 (<0.01–<0.01) 0.23 (0.23–0.23) 0.031 (0.011–0.061) 7.5 (2.6–15) 0.025 (0.022–0.029) 6.1 (5.3–6.9)
2010 < 1 <0.01 (<0.01–<0.01) 0.25 (0.24–0.25) 0.043 (0.014–0.087) 10 (3.4–20) 0.033 (0.029–0.038) 7.9 (6.9–8.9)
2011 < 1 <0.01 (<0.01–<0.01) 0.69 (0.69–0.69) 0.044 (0.015–0.089) 10 (3.4–21) 0.035 (0.030–0.039) 8.1 (7.1–9.2)
2012 < 1 <0.01 (<0.01–<0.01) 0.37 (0.36–0.37) 0.069 (0.032–0.120) 16 (7.5–28) 0.048 (0.042–0.055) 11 (9.9–13)
Monaco 1990 < 1 <0.01 (0–<0.01) 0.27 (0–1.5) <0.01 (<0.01–<0.01) 6.4 (3.2–11) <0.01 (<0.01–<0.01) 3.9 (3.4–4.4)
1995 < 1 <0.01 (0–<0.01) 0.26 (0–1.4) <0.01 (<0.01–<0.01) 6 (3.0–10) <0.01 (<0.01–<0.01) 3.7 (3.3–4.2)
2000 < 1 <0.01 (0–<0.01) <0.1 (<0.1–0.17) <0.01 (<0.01–<0.01) 1.7 (0.50–3.6) 0 (0–0) 0 (0–0)
2005 < 1 <0.01 (0–<0.01) <0.1 (0–0.54) <0.01 (<0.01–<0.01) 2 (1.0–3.3) <0.01 (<0.01–<0.01) 1.2 (1.0–1.3)
2010 < 1 <0.01 (0–<0.01) 0.21 (0–1.1) <0.01 (<0.01–<0.01) 5 (2.5–8.4) <0.01 (<0.01–<0.01) 3.1 (2.7–3.5)
2011 < 1 <0.01 (0–<0.01) <0.1 (<0.1–0.24) <0.01 (<0.01–<0.01) 2.4 (0.73–5.2) <0.01 (<0.01–<0.01) 2.1 (1.8–2.4)
2012 < 1 <0.01 (<0.01–<0.01) 0.1 (<0.1–0.24) <0.01 (<0.01–<0.01) 2.7 (0.81–5.7) <0.01 (<0.01–<0.01) 2.1 (1.8–2.4)
a
Rates are per 100 000 population.
229 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
EUROPEAN REGION
7$%/($(VWLPDWHVRIWKHEXUGHQRIGLVHDVHFDXVHGE\7%±
YEAR POPULATION
(MILLIONS)
NUMBER
(THOUSANDS) RATE
a
NUMBER
(THOUSANDS) RATE
a
NUMBER
(THOUSANDS) RATE
a
MORTALITY (EXCLUDING HIV) PREVALENCE (INCLUDING HIV) INCIDENCE (INCLUDING HIV)
Montenegro 2005 < 1 <0.01 (<0.01–<0.01) 0.57 (0.52–0.62) 0.23 (0.086–0.430) 37 (14–70) 0.18 (0.160–0.200) 29 (26–33)
2010 < 1 <0.01 (<0.01–<0.01) 0.19 (0.13–0.25) 0.17 (0.073–0.320) 28 (12–51) 0.13 (0.110–0.140) 20 (18–23)
2011 < 1 <0.01 (<0.01–<0.01) 0.19 (0.13–0.25) 0.18 (0.082–0.320) 29 (13–51) 0.13 (0.110–0.140) 20 (18–23)
2012 < 1 <0.01 (<0.01–<0.01) 0.19 (0.13–0.25) 0.15 (0.065–0.280) 25 (10–45) 0.11 (0.100–0.130) 18 (16–20)
Netherlands 1990 15 0.034 (0.033–0.035) 0.23 (0.22–0.23) 2.2 (0.920–3.9) 15 (6.2–26) 1.6 (1.4–1.8) 11 (9.3–12)
1995 15 0.044 (0.043–0.045) 0.29 (0.28–0.29) 2.6 (1.1–4.6) 17 (7.4–30) 1.9 (1.6–2.1) 12 (11–14)
2000 16 0.034 (0.033–0.035) 0.21 (0.21–0.22) 1.8 (0.620–3.5) 11 (3.9–22) 1.4 (1.3–1.6) 9 (7.9–10)
2005 16 0.033 (0.032–0.034) 0.2 (0.20–0.21) 1.7 (0.700–3.2) 10 (4.3–19) 1.3 (1.1–1.5) 8 (7.0–9.0)
2010 17 0.032 (0.031–0.032) 0.19 (0.19–0.20) 1.7 (0.780–3.0) 10 (4.7–18) 1.2 (1.1–1.4) 7.2 (6.3–8.2)
2011 17 0.019 (0.019–0.019) 0.11 (0.11–0.12) 1.5 (0.670–2.8) 9.2 (4.0–17) 1.1 (0.990–1.3) 6.8 (5.9–7.7)
2012 17 0.028 (0.028–0.029) 0.17 (0.17–0.17) 1.4 (0.550–2.6) 8.2 (3.3–15) 1.1 (0.930–1.2) 6.3 (5.5–7.2)
Norway 1990 4 0.026 (0.025–0.026) 0.6 (0.59–0.62) 0.43 (0.160–0.820) 10 (3.8–19) 0.33 (0.290–0.370) 7.7 (6.8–8.7)
1995 4 0.019 (0.019–0.020) 0.44 (0.43–0.45) 0.38 (0.170–0.690) 8.8 (3.8–16) 0.27 (0.240–0.310) 6.2 (5.5–7.0)
2000 4 0.01 (0.010–0.011) 0.23 (0.22–0.23) 0.32 (0.110–0.630) 7.1 (2.5–14) 0.25 (0.220–0.290) 5.7 (5.0–6.4)
2005 5 <0.01 (<0.01–0.010) 0.21 (0.21–0.22) 0.42 (0.160–0.790) 9 (3.5–17) 0.32 (0.280–0.360) 6.9 (6.0–7.8)
2010 5 0.01 (<0.01–0.010) 0.21 (0.20–0.21) 0.44 (0.170–0.850) 9.1 (3.4–17) 0.34 (0.300–0.390) 7 (6.1–7.9)
2011 5 <0.01 (<0.01–<0.01) 0.12 (0.12–0.13) 0.52 (0.220–0.940) 10 (4.4–19) 0.37 (0.330–0.420) 7.5 (6.6–8.5)
2012 5 <0.01 (<0.01–<0.01) 0.14 (0.14–0.14) 0.51 (0.220–0.940) 10 (4.3–19) 0.37 (0.330–0.420) 7.5 (6.6–8.5)
Poland 1990 38 1.4 (1.4–1.5) 3.8 (3.6–3.9) 25 (10–47) 66 (27–122) 19 (16–21) 49 (43–55)
1995 38 1.2 (1.2–1.3) 3.2 (3.0–3.3) 26 (11–46) 67 (29–121) 18 (16–21) 48 (42–54)
2000 38 1.1 (1.1–1.2) 2.9 (2.8–3.0) 17 (6.6–31) 44 (17–82) 13 (11–14) 33 (29–37)
2005 38 0.85 (0.820–0.880) 2.2 (2.2–2.3) 13 (5.1–24) 33 (13–62) 9.4 (8.3–11) 25 (22–28)
2010 38 0.61 (0.580–0.630) 1.6 (1.5–1.6) 11 (4.1–20) 28 (11–53) 8.1 (7.1–9.1) 21 (18–24)
2011 38 0.7 (0.670–0.730) 1.8 (1.7–1.9) 13 (6.2–23) 35 (16–61) 9.1 (8.0–10) 24 (21–27)
2012 38 0.67 (0.640–0.700) 1.8 (1.7–1.8) 11 (4.2–20) 28 (11–53) 8.1 (7.1–9.2) 21 (19–24)
Portugal 1990 10 0.31 (0.290–0.330) 3.1 (2.9–3.3) 9.8 (4.2–18) 99 (43–180) 7.1 (6.3–8.1) 72 (63–82)
1995 10 0.35 (0.330–0.370) 3.5 (3.2–3.7) 8.6 (3.7–15) 85 (37–152) 6.4 (5.6–7.3) 64 (56–72)
2000 10 0.29 (0.270–0.310) 2.8 (2.6–3.0) 5.9 (2.2–11) 57 (21–110) 4.9 (4.3–5.5) 47 (41–53)
2005 11 0.18 (0.170–0.190) 1.7 (1.6–1.8) 4.6 (1.8–8.7) 44 (17–83) 3.8 (3.3–4.3) 36 (32–41)
2010 11 0.13 (0.120–0.130) 1.2 (1.1–1.3) 3.4 (1.4–6.4) 32 (13–61) 2.9 (2.5–3.2) 27 (24–31)
2011 11 0.15 (0.140–0.160) 1.4 (1.3–1.5) 3.4 (1.4–6.2) 32 (13–59) 2.8 (2.4–3.1) 26 (23–30)
2012 11 0.14 (0.130–0.140) 1.3 (1.2–1.4) 3.6 (1.6–6.4) 34 (15–61) 2.8 (2.4–3.1) 26 (23–30)
Republic of 1990 4 0.25 (0.230–0.260) 5.6 (5.2–6.1) 3.5 (1.5–6.2) 79 (34–142) 2.3 (1.9–2.8) 54 (44–64)
Moldova 1995 4 0.55 (0.510–0.580) 13 (12–13) 8.9 (4.5–15) 206 (104–342) 4.7 (3.9–5.6) 109 (90–130)
2000 4 0.72 (0.660–0.780) 17 (16–19) 10 (5.2–17) 254 (126–425) 6 (5.0–7.2) 147 (121–175)
2005 4 0.75 (0.700–0.790) 20 (19–21) 9.5 (4.1–17) 252 (108–454) 6.6 (5.4–7.9) 175 (144–209)
2010 4 0.57 (0.550–0.590) 16 (15–16) 8.8 (4.0–15) 245 (112–430) 5.9 (4.9–7.1) 166 (137–198)
2011 4 0.48 (0.470–0.500) 14 (13–14) 8.6 (4.0–15) 242 (113–419) 5.7 (4.7–6.8) 161 (133–192)
2012 4 0.63 (0.620–0.640) 18 (18–18) 8.8 (4.2–15) 249 (120–424) 5.6 (4.6–6.7) 160 (132–190)
Romania 1990 23 1.6 (1.6–1.6) 6.9 (6.9–6.9) 67 (34–110) 287 (145–478) 34 (28–41) 146 (120–174)
1995 23 2.6 (2.6–2.6) 11 (11–11) 81 (41–130) 351 (177–583) 43 (36–52) 189 (155–226)
2000 22 2.1 (2.1–2.1) 9.5 (9.5–9.5) 66 (32–110) 295 (142–504) 41 (33–48) 181 (149–216)
2005 22 1.7 (1.7–1.7) 7.8 (7.8–7.8) 46 (20–84) 209 (88–380) 32 (27–39) 147 (121–175)
2010 22 1.4 (1.4–1.4) 6.5 (6.5–6.5) 34 (15–62) 158 (69–282) 24 (20–28) 109 (89–130)
2011 22 1.3 (1.3–1.3) 5.9 (5.9–6.0) 33 (15–58) 151 (68–266) 22 (18–26) 101 (83–121)
2012 22 1.2 (1.2–1.2) 5.6 (5.5–5.6) 31 (15–55) 144 (67–251) 20 (17–24) 94 (77–112)
Russian 1990 148 12 (12–12) 8.2 (8.1–8.3) 120 (59–200) 81 (40–136) 70 (59–81) 47 (40–55)
Federation 1995 149 24 (24–25) 16 (16–17) 240 (120–400) 163 (82–271) 140 (120–170) 96 (81–112)
2000 147 31 (31–32) 21 (21–22) 300 (150–510) 206 (101–348) 190 (160–220) 127 (108–149)
2005 144 32 (31–33) 22 (22–23) 320 (160–540) 223 (112–372) 190 (160–230) 135 (114–158)
2010 144 23 (22–24) 16 (16–16) 220 (100–380) 152 (69–266) 150 (130–180) 106 (89–123)
2011 143 21 (21–22) 15 (14–15) 190 (85–340) 135 (59–240) 140 (120–160) 97 (82–114)
2012 143 19 (18–20) 13 (13–14) 170 (73–320) 121 (51–221) 130 (110–150) 91 (77–106)
San Marino 1990 < 1 0 (0–0) 0 (0–0) <0.01 (<0.01–<0.01) 7 (2.1–15) <0.01 (<0.01–<0.01) 4.8 (4.2–5.4)
1995 < 1 0 (0–0) 0 (0–0) <0.01 (<0.01–<0.01) 15 (7.7–25) <0.01 (<0.01–<0.01) 9 (7.8–10)
2000 < 1 0 (0–0) 0 (0–0) <0.01 (<0.01–<0.01) 8.5 (4.2–14) <0.01 (<0.01–<0.01) 4.3 (3.7–4.8)
2005 < 1 0 (0–0) 0 (0–0) <0.01 (<0.01–<0.01) 1.8 (0.55–3.9) <0.01 (<0.01–<0.01) 1.5 (1.3–1.7)
2010 < 1 0 (0–0) 0 (0–0) <0.01 (<0.01–<0.01) 2 (0.82–3.7) <0.01 (<0.01–<0.01) 1.5 (1.3–1.7)
2011 < 1 0 (0–0) 0 (0–0) <0.01 (<0.01–<0.01) 2 (0.83–3.7) <0.01 (<0.01–<0.01) 1.5 (1.3–1.7)
2012 < 1 0 (0–0) 0 (0–0) <0.01 (<0.01–<0.01) 2 (0.79–3.7) <0.01 (<0.01–<0.01) 1.5 (1.3–1.7)
Serbia 2005 10 0.28 (0.250–0.300) 2.8 (2.5–3.1) 5.1 (2.1–9.3) 51 (21–93) 3.7 (3.2–4.2) 37 (32–42)
2010 10 0.16 (0.150–0.180) 1.7 (1.5–1.9) 3.7 (1.5–6.8) 38 (16–70) 2.7 (2.3–3.0) 28 (24–32)
2011 10 0.16 (0.140–0.170) 1.6 (1.4–1.8) 3.5 (1.5–6.3) 36 (16–66) 2.5 (2.2–2.8) 26 (23–30)
2012 10 0.14 (0.120–0.160) 1.5 (1.3–1.6) 3 (1.2–5.4) 31 (13–57) 2.2 (1.9–2.4) 23 (20–26)
Serbia & 1990 10 0.6 (0.580–0.620) 5.8 (5.6–5.9) 11 (4.5–22) 111 (43–209) 7 (5.0–9.4) 68 (48–91)
Montenegro 1995 11 0.5 (0.480–0.520) 4.5 (4.4–4.7) 12 (5.8–20) 108 (53–183) 6.7 (5.7–7.9) 61 (52–72)
2000 11 0.41 (0.400–0.430) 3.8 (3.6–4.0) 7.7 (3.6–13) 71 (33–123) 5 (4.2–5.8) 46 (38–54)
Slovakia 1990 5 0.11 (0.110–0.110) 2.1 (2.1–2.1) 1.9 (0.600–4.0) 36 (11–75) 1.7 (1.5–1.9) 32 (28–36)
1995 5 0.084 (0.084–0.085) 1.6 (1.6–1.6) 2.4 (1.0–4.5) 46 (19–83) 1.8 (1.6–2.0) 33 (29–37)
2000 5 0.054 (0.053–0.054) 0.99 (0.99–1.0) 1.5 (0.590–2.9) 28 (11–54) 1.2 (1.0–1.3) 22 (19–24)
2005 5 0.046 (0.045–0.046) 0.85 (0.84–0.85) 1.1 (0.450–2.1) 21 (8.3–38) 0.82 (0.720–0.920) 15 (13–17)
2010 5 0.033 (0.033–0.033) 0.61 (0.61–0.62) 0.62 (0.250–1.2) 11 (4.5–22) 0.47 (0.410–0.530) 8.7 (7.6–9.8)
2011 5 0.034 (0.034–0.035) 0.63 (0.63–0.63) 0.61 (0.260–1.1) 11 (4.9–20) 0.43 (0.380–0.490) 8 (7.0–9.0)
2012 5 0.034 (0.034–0.035) 0.63 (0.63–0.63) 0.52 (0.220–0.930) 9.5 (4.1–17) 0.37 (0.320–0.420) 6.8 (5.9–7.7)
Slovenia 1990 2 0.05 (0.049–0.051) 2.5 (2.5–2.5) 1.2 (0.550–2.1) 60 (27–106) 0.83 (0.730–0.940) 41 (36–47)
1995 2 0.032 (0.032–0.033) 1.6 (1.6–1.6) 0.81 (0.330–1.5) 41 (16–76) 0.6 (0.530–0.680) 30 (27–34)
2000 2 0.017 (0.017–0.017) 0.86 (0.85–0.88) 0.56 (0.220–1.1) 28 (11–53) 0.42 (0.370–0.480) 21 (19–24)
2005 2 0.017 (0.016–0.017) 0.83 (0.82–0.84) 0.46 (0.210–0.790) 23 (11–40) 0.31 (0.270–0.350) 15 (14–18)
2010 2 0.019 (0.018–0.019) 0.9 (0.90–0.91) 0.27 (0.110–0.490) 13 (5.4–24) 0.19 (0.170–0.220) 9.5 (8.3–11)
2011 2 0.02 (0.020–0.020) 0.97 (0.96–0.97) 0.31 (0.150–0.540) 15 (7.3–26) 0.21 (0.180–0.240) 10 (8.8–11)
2012 2 0.02 (0.020–0.020) 0.97 (0.96–0.97) 0.19 (0.061–0.380) 9 (3.0–18) 0.15 (0.140–0.170) 7.5 (6.5–8.4)
Spain 1990 39 0.89 (0.870–0.900) 2.3 (2.2–2.3) 11 (3.8–21) 28 (9.8–54) 8.7 (7.7–9.9) 22 (20–25)
1995 39 0.62 (0.610–0.620) 1.6 (1.5–1.6) 13 (5.3–24) 33 (13–60) 10 (8.8–11) 26 (22–29)
2000 40 0.4 (0.400–0.410) 1 (0.99–1.0) 12 (5.1–22) 30 (13–54) 9.2 (8.1–10) 23 (20–26)
2005 43 0.35 (0.350–0.360) 0.81 (0.80–0.82) 11 (4.4–20) 25 (10–45) 8.4 (7.3–9.5) 19 (17–22)
2010 46 0.3 (0.300–0.310) 0.66 (0.65–0.67) 10 (4.4–18) 22 (9.5–40) 7.8 (6.8–8.8) 17 (15–19)
2011 47 0.23 (0.230–0.230) 0.49 (0.49–0.50) 9.6 (4.1–17) 21 (8.8–37) 7.4 (6.4–8.3) 16 (14–18)
2012 47 0.27 (0.260–0.270) 0.57 (0.57–0.58) 8.1 (3.2–15) 17 (6.8–33) 6.5 (5.7–7.4) 14 (12–16)
Sweden 1990 9 0.06 (0.060–0.061) 0.7 (0.70–0.71) 0.87 (0.350–1.6) 10 (4.1–19) 0.64 (0.560–0.720) 7.5 (6.6–8.5)
1995 9 0.024 (0.024–0.024) 0.27 (0.27–0.28) 0.94 (0.430–1.7) 11 (4.9–19) 0.65 (0.570–0.730) 7.3 (6.4–8.3)
2000 9 0.018 (0.018–0.018) 0.2 (0.20–0.21) 0.66 (0.280–1.2) 7.5 (3.2–14) 0.48 (0.420–0.540) 5.4 (4.7–6.1)
2005 9 0.015 (0.015–0.015) 0.17 (0.16–0.17) 0.97 (0.480–1.6) 11 (5.3–18) 0.62 (0.540–0.700) 6.9 (6.0–7.8)
2010 9 0.014 (0.014–0.014) 0.15 (0.14–0.15) 1.1 (0.490–1.8) 11 (5.2–20) 0.72 (0.630–0.810) 7.6 (6.7–8.6)
2011 9 0.014 (0.013–0.014) 0.14 (0.14–0.15) 0.79 (0.280–1.6) 8.4 (3.0–16) 0.63 (0.550–0.710) 6.6 (5.8–7.5)
2012 10 0.013 (0.013–0.013) 0.14 (0.13–0.14) 0.92 (0.370–1.7) 9.6 (3.9–18) 0.68 (0.600–0.770) 7.2 (6.3–8.1)
Switzerland 1990 7 0.086 (0.085–0.087) 1.3 (1.3–1.3) 2.1 (0.940–3.7) 31 (14–56) 1.5 (1.3–1.7) 22 (19–25)
1995 7 0.047 (0.046–0.047) 0.67 (0.66–0.68) 1.3 (0.530–2.4) 18 (7.5–34) 0.95 (0.840–1.1) 14 (12–15)
2000 7 0.034 (0.034–0.035) 0.48 (0.47–0.49) 0.83 (0.290–1.6) 12 (4.1–23) 0.66 (0.580–0.750) 9.3 (8.1–10)
2005 7 0.022 (0.021–0.022) 0.29 (0.29–0.30) 0.82 (0.360–1.5) 11 (4.8–20) 0.59 (0.520–0.670) 8 (7.0–9.0)
2010 8 0.019 (0.019–0.020) 0.25 (0.24–0.25) 0.85 (0.390–1.5) 11 (5.0–19) 0.58 (0.510–0.660) 7.5 (6.5–8.4)
2011 8 0.018 (0.017–0.018) 0.22 (0.22–0.23) 0.9 (0.420–1.5) 11 (5.4–19) 0.6 (0.530–0.680) 7.6 (6.7–8.6)
2012 8 0.017 (0.017–0.018) 0.22 (0.21–0.22) 0.57 (0.190–1.2) 7.1 (2.3–15) 0.48 (0.420–0.540) 6 (5.2–6.8)
Tajikistan 1990 5 0.34 (0.240–0.460) 6.4 (4.5–8.6) 6.4 (3.2–11) 121 (60–203) 3.7 (3.0–4.4) 70 (58–83)
1995 6 0.69 (0.490–0.930) 12 (8.4–16) 20 (9.5–35) 350 (164–604) 8.6 (7.1–10) 148 (122–176)
2000 6 1.2 (0.690–1.9) 20 (11–31) 30 (15–52) 493 (238–839) 14 (11–16) 220 (182–263)
2005 7 0.96 (0.720–1.2) 14 (11–18) 27 (13–44) 393 (198–654) 14 (11–16) 200 (165–238)
2010 8 0.69 (0.520–0.880) 9 (6.8–12) 16 (7.6–27) 206 (99–352) 9.8 (8.1–12) 129 (106–153)
2011 8 0.65 (0.490–0.830) 8.3 (6.2–11) 14 (6.6–25) 181 (85–315) 9.2 (7.6–11) 118 (97–140)
2012 8 0.61 (0.460–0.780) 7.6 (5.7–9.7) 13 (5.8–23) 160 (72–283) 8.6 (7.1–10) 108 (89–128)
a
Rates are per 100 000 population.
230 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($(VWLPDWHVRIWKHEXUGHQRIGLVHDVHFDXVHGE\7%±
YEAR POPULATION
(MILLIONS)
NUMBER
(THOUSANDS) RATE
a
NUMBER
(THOUSANDS) RATE
a
NUMBER
(THOUSANDS) RATE
a
MORTALITY (EXCLUDING HIV) PREVALENCE (INCLUDING HIV) INCIDENCE (INCLUDING HIV)
The Former 1990 2 0.17 (0.160–0.180) 8.4 (7.9–9.0) 3.3 (0.990–7.1) 167 (49–354) 1.6 (1.0–2.4) 81 (50–119)
Yugoslav Republic 1995 2 0.1 (0.094–0.110) 5.1 (4.8–5.4) 1.6 (0.650–2.9) 81 (33–150) 1.1 (0.930–1.4) 58 (47–69)
of Macedonia 2000 2 0.21 (0.200–0.230) 10 (9.6–11) 1.2 (0.540–2.2) 61 (26–109) 0.85 (0.690–1.0) 41 (34–50)
2005 2 0.066 (0.064–0.069) 3.2 (3.1–3.3) 0.69 (0.200–1.5) 33 (9.8–70) 0.62 (0.560–0.680) 30 (27–33)
2010 2 0.034 (0.033–0.035) 1.6 (1.6–1.7) 0.54 (0.190–1.1) 26 (9.0–51) 0.44 (0.380–0.510) 21 (18–24)
2011 2 0.024 (0.023–0.025) 1.1 (1.1–1.2) 0.54 (0.210–1.0) 25 (10–48) 0.41 (0.360–0.480) 20 (17–23)
2012 2 0.017 (0.016–0.018) 0.82 (0.77–0.87) 0.54 (0.240–0.970) 26 (11–46) 0.39 (0.330–0.450) 18 (16–21)
Turkey 1990 54 3.4 (0.780–7.8) 6.2 (1.4–14) 27 (11–51) 51 (20–95) 28 (25–32) 52 (46–59)
1995 59 2.4 (0.860–4.6) 4 (1.5–7.8) 34 (16–57) 58 (28–98) 26 (23–30) 45 (40–51)
2000 63 2 (0.840–3.7) 3.2 (1.3–5.8) 28 (14–48) 45 (22–76) 21 (18–23) 33 (29–37)
2005 68 0.99 (0.590–1.5) 1.5 (0.86–2.2) 19 (8.6–33) 28 (13–48) 23 (20–26) 34 (29–38)
2010 72 0.55 (0.390–0.740) 0.76 (0.53–1.0) 17 (8.1–30) 24 (11–42) 18 (16–21) 25 (22–29)
2011 73 0.47 (0.340–0.610) 0.64 (0.47–0.83) 17 (8.0–30) 24 (11–41) 17 (15–20) 24 (21–27)
2012 74 0.39 (0.300–0.480) 0.52 (0.41–0.65) 17 (7.9–30) 23 (11–40) 16 (14–18) 22 (19–25)
Turkmenistan 1990 4 0.49 (0.400–0.590) 13 (11–16) 5.6 (2.5–9.7) 152 (69–265) 3.5 (2.8–4.2) 95 (76–115)
1995 4 0.83 (0.720–0.950) 20 (17–23) 13 (6.1–22) 302 (145–515) 6.6 (5.4–7.8) 157 (129–187)
2000 5 1.3 (0.820–1.8) 28 (18–40) 18 (8.6–31) 400 (191–685) 9.4 (7.6–11) 209 (170–252)
2005 5 1.1 (0.700–1.6) 23 (15–33) 16 (7.6–27) 333 (160–569) 8.3 (6.8–10) 175 (144–210)
2010 5 0.6 (0.390–0.860) 12 (7.7–17) 8.4 (4.0–14) 166 (79–283) 5.2 (4.3–6.1) 103 (86–121)
2011 5 0.5 (0.330–0.720) 9.9 (6.4–14) 6.8 (3.0–12) 133 (58–238) 4.5 (3.7–5.5) 89 (73–107)
2012 5 0.43 (0.260–0.660) 8.4 (5.0–13) 5.1 (1.8–10) 99 (35–196) 3.9 (3.1–4.8) 75 (59–94)
Ukraine 1990 52 5 (4.7–5.2) 9.6 (9.2–10) 33 (15–60) 65 (28–116) 23 (19–27) 45 (37–53)
1995 51 7.8 (7.5–8.0) 15 (15–16) 69 (35–110) 135 (68–223) 38 (31–45) 74 (62–88)
2000 49 11 (11–11) 23 (23–23) 81 (38–140) 164 (77–284) 53 (44–63) 108 (90–129)
2005 47 12 (12–12) 25 (25–26) 75 (31–140) 159 (65–293) 57 (48–68) 121 (101–144)
2010 46 7.4 (7.3–7.5) 16 (16–16) 68 (32–120) 149 (70–257) 48 (41–57) 105 (88–123)
2011 46 7.1 (7.0–7.2) 16 (15–16) 66 (31–110) 144 (68–248) 46 (38–54) 99 (83–118)
2012 46 6.1 (6.0–6.2) 13 (13–14) 62 (29–110) 137 (65–236) 42 (35–51) 93 (77–112)
United Kingdom of 1990 57 0.44 (0.440–0.450) 0.78 (0.77–0.78) 8.6 (3.5–16) 15 (6.0–28) 6.6 (6.2–7.1) 12 (11–12)
Great Britain and 1995 58 0.51 (0.510–0.520) 0.88 (0.87–0.89) 9.3 (3.9–17) 16 (6.8–29) 6.9 (6.5–7.4) 12 (11–13)
Northern Ireland 2000 59 0.43 (0.420–0.440) 0.73 (0.72–0.74) 9.2 (3.9–17) 16 (6.6–29) 7 (6.5–7.4) 12 (11–13)
2005 60 0.39 (0.380–0.390) 0.64 (0.63–0.65) 13 (6.0–22) 21 (10–37) 9.2 (8.6–9.8) 15 (14–16)
2010 62 0.32 (0.320–0.330) 0.52 (0.52–0.53) 11 (4.2–21) 18 (6.8–34) 8.9 (8.3–9.4) 14 (13–15)
2011 62 0.34 (0.340–0.340) 0.54 (0.54–0.55) 13 (5.4–23) 20 (8.6–36) 9.5 (8.8–10) 15 (14–16)
2012 63 0.34 (0.340–0.340) 0.54 (0.54–0.55) 13 (5.4–23) 20 (8.7–36) 9.4 (8.8–10) 15 (14–16)
Uzbekistan 1990 21 1.7 (1.5–1.9) 8.3 (7.2–9.4) 54 (27–90) 262 (130–438) 26 (21–31) 125 (103–149)
1995 23 2.7 (2.4–3.1) 12 (10–13) 100 (50–170) 447 (216–760) 46 (38–55) 200 (165–238)
2000 25 4.3 (3.7–5.0) 17 (15–20) 160 (77–270) 647 (310–1 100) 71 (59–85) 287 (237–342)
2005 26 3.5 (3.1–4.0) 14 (12–15) 130 (63–210) 485 (240–814) 61 (50–72) 233 (193–278)
2010 28 1.5 (0.850–2.3) 5.3 (3.1–8.3) 63 (32–100) 227 (115–376) 34 (28–40) 122 (101–146)
2011 28 1.1 (0.600–1.6) 3.7 (2.1–5.8) 52 (26–86) 183 (92–304) 29 (24–34) 101 (84–121)
2012 29 0.6 (0.350–0.930) 2.1 (1.2–3.3) 39 (19–65) 135 (67–227) 22 (18–27) 78 (65–93)
a Rates are per 100 000 population.

231 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
EUROPEAN REGION
7$%/($,QFLGHQFHQRWLILFDWLRQDQGFDVHGHWHFWLRQUDWHVDOOIRUPV±
INCIDENCE (INCLUDING HIV) INCIDENCE HIV-POSITIVE NOTIFIED NEW AND RELAPSE
b
CASE DETECTION
YEAR POPULATION
(MILLIONS)
NUMBER
(THOUSANDS) RATE
a
NUMBER
(THOUSANDS) RATE
a
NUMBER RATE
a
PERCENT
Albania 1990 3 0.84 (0.600–1.1) 24 (18–32) 653 19 78 (59–110)
1995 3 0.82 (0.680–0.970) 24 (20–29) 641 19 78 (66–94)
2000 3 0.75 (0.630–0.870) 23 (19–26) 604 18 81 (69–95)
2005 3 0.63 (0.530–0.730) 20 (17–23) 506 16 81 (69–95)
2010 3 0.53 (0.450–0.620) 17 (14–20) 431 14 81 (69–95)
2011 3 0.52 (0.440–0.610) 17 (14–19) 422 13 81 (69–95)
2012 3 0.51 (0.430–0.590) 16 (14–19) 408 13 81 (69–95)
Andorra 1990 < 1 0.026 (0.023–0.030) 49 (43–55) 23 42 87 (77–99)
1995 < 1 0.023 (0.020–0.026) 37 (32–41)
2000 < 1 0.014 (0.012–0.016) 21 (18–24) 12 18 87 (77–99)
2005 < 1 0.012 (0.010–0.013) 14 (12–16) 10 12 87 (77–99)
2010 < 1 <0.01 (<0.01–<0.01) 10 (9.1–12) 7 9 87 (77–99)
2011 < 1 <0.01 (<0.01–<0.01) 4.4 (3.9–5.0) 3 3.9 87 (77–99)
2012 < 1 0.01 (<0.01–0.012) 13 (12–15) 9 11 87 (77–99)
Armenia 1990 4 0.63 (0.470–0.810) 18 (13–23) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 590 17 94 (73–130)
1995 3 1.2 (1.0–1.4) 38 (32–44) <0.01 (<0.01–<0.01) 0.2 (0.17–0.24) 1 000 31 82 (70–98)
2000 3 1.9 (1.6–2.1) 61 (53–68) 0.028 (0.025–0.032) 0.9 (0.81–1.0) 1 333 43 71 (63–81)
2005 3 2.3 (2.1–2.6) 77 (68–87) 0.06 (0.053–0.068) 2 (1.8–2.2) 2 206 73 95 (84–110)
2010 3 1.8 (1.6–2.2) 62 (53–73) 0.048 (0.040–0.056) 1.6 (1.4–1.9) 1 410 48 76 (65–90)
2011 3 1.6 (1.3–1.9) 55 (45–65) 0.041 (0.034–0.049) 1.4 (1.1–1.6) 1 261 43 78 (65–94)
2012 3 1.5 (1.3–1.8) 52 (43–61) 0.038 (0.032–0.045) 1.3 (1.1–1.5) 1 213 41 79 (67–95)
Austria 1990 8 1.7 (1.5–2.0) 23 (20–26) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 1 521 20 87 (77–99)
1995 8 1.7 (1.5–1.9) 21 (19–24) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 1 481 19 87 (77–99)
2000 8 1.4 (1.2–1.5) 17 (15–19) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 1 185 15 87 (77–99)
2005 8 1.1 (0.940–1.2) 13 (11–15) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 928 11 87 (77–99)
2010 8 0.76 (0.660–0.860) 9 (7.9–10) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 659 7.8 87 (77–99)
2011 8 0.77 (0.680–0.870) 9.2 (8.0–10) <0.01 (<0.01–<0.01) <0.1 (<0.1–0.10) 671 8 87 (77–99)
2012 8 0.67 (0.590–0.760) 7.9 (6.9–8.9) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 620 7.3 93 (82–110)
Azerbaijan 1990 7 22 (18–26) 305 (252–363) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 2 620 36 12 (10–14)
1995 8 49 (41–59) 637 (526–759) 0.035 (0.029–0.041) 0.5 (0.37–0.53) 1 630 21 3.3 (2.8–4.0)
2000 8 55 (46–66) 682 (563–813) 0.19 (0.16–0.22) 2.3 (1.9–2.8) 5 187 64 9.4 (7.9–11)
2005 9 29 (24–34) 335 (276–398) 0.22 (0.18–0.26) 2.5 (2.1–3.0) 6 034 70 21 (18–26)
2010 9 12 (9.8–14) 131 (108–156) 0.12 (0.10–0.14) 1.3 (1.1–1.6) 7 550 83 64 (53–77)
2011 9 10 (8.6–12) 113 (93–135) 0.11 (0.089–0.13) 1.2 (0.97–1.4) 9 146 99 88 (74–110)
2012 9 8.9 (7.3–11) 95 (78–114) 0.094 (0.077–0.11) 1 (0.83–1.2) 6 363 68 72 (60–87)
Belarus 1990 10 3.5 (2.8–4.3) 34 (27–42) 3 039 30 86 (70–110)
1995 10 6.9 (5.9–8.1) 68 (58–80) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 4 854 48 70 (60–82)
2000 10 8.4 (6.9–9.9) 84 (69–100) 0.017 (0.014–0.020) 0.2 (0.14–0.20) 6 799 68 81 (68–98)
2005 10 6.9 (5.3–8.8) 72 (55–91) 0.13 (0.096–0.16) 1.3 (1.0–1.6) 5 308 55 76 (61–100)
2010 9 6.7 (5.3–8.1) 70 (56–86) 0.26 (0.20–0.31) 2.7 (2.2–3.3) 5 098 54 76 (63–96)
2011 9 6.6 (5.4–8.0) 70 (57–85) 0.27 (0.22–0.32) 2.8 (2.3–3.4) 4 697 50 71 (59–87)
2012 9 6.6 (5.4–8.0) 70 (57–85) 0.28 (0.23–0.34) 3 (2.4–3.6) 4 783 51 72 (60–89)
Belgium 1990 10 1.8 (1.6–2.1) 18 (16–21) 0.01 (<0.01–0.011) 0.1 (<0.1–0.12) 1 577 16 87 (77–99)
1995 10 1.6 (1.4–1.8) 16 (14–18) 0.038 (0.033–0.043) 0.4 (0.33–0.42) 1 380 14 87 (77–99)
2000 10 1.5 (1.3–1.7) 14 (13–16) 0.041 (0.036–0.046) 0.4 (0.35–0.45) 1 278 12 87 (77–99)
2005 11 1.2 (1.1–1.4) 12 (10–13) 0.041 (0.036–0.047) 0.4 (0.34–0.45) 1 076 10 87 (77–99)
2010 11 1.2 (1.0–1.3) 11 (9.5–12) 0.044 (0.038–0.050) 0.4 (0.35–0.45) 1 028 9.4 87 (77–99)
2011 11 1.1 (0.990–1.3) 10 (9.0–12) 0.042 (0.037–0.048) 0.4 (0.34–0.44) 985 8.9 87 (77–99)
2012 11 1.1 (0.940–1.2) 9.7 (8.5–11) 0.041 (0.036–0.046) 0.4 (0.32–0.42) 909 8.2 85 (75–97)
Bosnia and 1990 5 4.2 (2.6–6.2) 94 (58–138) 4 073 90 96 (65–160)
Herzegovina 1995 4 3 (2.4–3.6) 84 (69–101) 2 132 61 72 (60–88)
2000 4 2.4 (2.0–2.9) 63 (51–75) 2 476 65 100 (86–130)
2005 4 2 (1.7–2.4) 52 (43–63) 2 111 54 100 (87–130)
2010 4 1.9 (1.6–2.2) 50 (43–57) 1 321 34 69 (60–81)
2011 4 1.9 (1.6–2.2) 49 (42–56) 1 360 35 72 (63–84)
2012 4 1.9 (1.6–2.1) 49 (42–56) 1 409 37 76 (66–88)
Bulgaria 1990 9 2.9 (2.5–3.3) 33 (29–37) <0.01 (<0.01–<0.01) 0 (0–0) 2 256 26 78 (68–89)
1995 8 5.2 (4.5–5.9) 62 (54–71) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 3 245 39 62 (55–71)
2000 8 4.6 (4.0–5.3) 58 (50–66) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 3 349 42 72 (64–83)
2005 8 4.1 (3.6–4.6) 53 (46–61) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 3 225 42 79 (69–91)
2010 7 2.8 (2.5–3.2) 38 (33–43) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 2 412 33 86 (75–98)
2011 7 2.6 (2.2–2.9) 35 (30–40) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 2 172 30 85 (74–97)
2012 7 2.3 (2.0–2.6) 32 (28–36) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 2 081 29 90 (79–100)
Croatia 1990 5 3 (2.6–3.4) 62 (54–70) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 2 576 54 87 (77–99)
1995 5 2.4 (2.1–2.8) 52 (45–59) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 2 114 45 87 (77–99)
2000 4 1.9 (1.6–2.1) 42 (37–47) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 1 630 36 87 (77–99)
2005 4 1.2 (1.1–1.4) 28 (24–31) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 1 050 24 87 (77–99)
2010 4 0.79 (0.690–0.900) 18 (16–21) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 688 16 87 (77–99)
2011 4 0.71 (0.620–0.810) 16 (14–19) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 619 14 87 (77–99)
2012 4 0.62 (0.540–0.700) 14 (13–16) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1)
Cyprus 1990 < 1 0.033 (0.029–0.038) 4.4 (3.8–4.9) 29 3.8 87 (77–99)
1995 < 1 0.041 (0.036–0.047) 4.8 (4.2–5.5) 36 4.2 87 (77–99)
2000 < 1 0.038 (0.033–0.043) 4 (3.5–4.6) 33 3.5 87 (77–99)
2005 1 0.039 (0.034–0.044) 3.8 (3.3–4.3) 34 3.3 87 (77–99)
2010 1 0.07 (0.061–0.079) 6.4 (5.6–7.2) 61 5.5 87 (77–99)
2011 1 0.059 (0.051–0.066) 5.3 (4.6–5.9) 51 4.6 87 (77–99)
2012 1 0.061 (0.053–0.069) 5.4 (4.7–6.1) 63 5.6 100 (92–120)
Czech Republic 1990 10 2.2 (2.0–2.5) 22 (19–24) <0.01 (<0.01–<0.01) 0 (0–0) 1 937 19 87 (77–99)
1995 10 2.1 (1.8–2.4) 20 (18–23) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 1 834 18 87 (77–99)
2000 10 1.6 (1.4–1.8) 16 (14–18) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 1 414 14 87 (77–99)
2005 10 1.1 (0.980–1.3) 11 (9.6–12) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 973 9.5 87 (77–99)
2010 11 0.72 (0.630–0.820) 6.8 (6.0–7.7) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 627 5.9 87 (77–99)
2011 11 0.65 (0.570–0.740) 6.2 (5.4–7.0) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 569 5.4 87 (77–99)
2012 11 0.57 (0.500–0.640) 5.3 (4.7–6.0) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 565 5.3 99 (88–110)
Denmark 1990 5 0.4 (0.350–0.460) 7.8 (6.9–8.9) <0.01 (<0.01–<0.01) 0.1 (<0.1–0.12) 350 6.8 87 (77–99)
1995 5 0.52 (0.450–0.580) 9.8 (8.6–11) 0.011 (<0.01–0.013) 0.2 (0.19–0.24) 448 8.6 87 (77–99)
2000 5 0.68 (0.590–0.760) 13 (11–14) <0.01 (<0.01–0.010) 0.2 (0.15–0.20) 587 11 87 (77–99)
2005 5 0.45 (0.400–0.510) 8.4 (7.3–9.5) <0.01 (<0.01–<0.01) 0.1 (0.11–0.14) 395 7.3 87 (77–99)
2010 6 0.36 (0.320–0.410) 6.5 (5.7–7.3) <0.01 (<0.01–<0.01) 0.1 (<0.1–0.12) 313 5.6 87 (77–99)
2011 6 0.41 (0.360–0.470) 7.4 (6.5–8.4) <0.01 (<0.01–<0.01) 0.1 (0.11–0.14) 359 6.4 87 (77–99)
2012 6 0.41 (0.360–0.470) 7.4 (6.5–8.4) <0.01 (<0.01–<0.01) 0.1 (0.11–0.14)
Estonia 1990 2 0.49 (0.430–0.550) 31 (27–35) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 423 27 87 (77–99)
1995 1 0.72 (0.630–0.810) 50 (44–57) <0.01 (<0.01–<0.01) 0.2 (0.16–0.21) 624 44 87 (77–99)
2000 1 0.91 (0.800–1.0) 67 (58–75) 0.021 (0.018–0.024) 1.5 (1.4–1.7) 791 58 87 (77–99)
2005 1 0.55 (0.480–0.620) 42 (36–47) 0.044 (0.038–0.050) 3.3 (2.9–3.7) 479 36 87 (77–99)
2010 1 0.33 (0.290–0.370) 25 (22–28) 0.04 (0.035–0.045) 3.1 (2.7–3.5) 283 22 87 (77–99)
2011 1 0.34 (0.300–0.390) 26 (23–30) 0.043 (0.038–0.048) 3.3 (2.9–3.7) 296 23 87 (77–99)
2012 1 0.3 (0.260–0.340) 23 (20–26) 0.039 (0.034–0.044) 3 (2.7–3.4) 259 20 87 (77–99)
Finland 1990 5 0.89 (0.780–1.0) 18 (16–20) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 772 15 87 (77–99)
1995 5 0.76 (0.670–0.860) 15 (13–17) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 661 13 87 (77–99)
2000 5 0.61 (0.530–0.690) 12 (10–13) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 527 10 87 (77–99)
2005 5 0.39 (0.340–0.440) 7.4 (6.5–8.4) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 339 6.5 87 (77–99)
2010 5 0.36 (0.310–0.410) 6.7 (5.9–7.6) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 312 5.8 87 (77–99)
2011 5 0.36 (0.310–0.410) 6.7 (5.8–7.5) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 312 5.8 87 (77–99)
2012 5 0.3 (0.260–0.340) 5.5 (4.9–6.3) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 261 4.8 87 (77–99)
France 1990 57 11 (11–12) 20 (19–21) 0.87 (0.82–0.93) 1.5 (1.4–1.6) 9 030 16 80 (75–85)
1995 58 11 (10–12) 19 (18–20) 0.76 (0.72–0.81) 1.3 (1.2–1.4) 8 723 15 80 (75–85)
2000 59 7.7 (7.2–8.1) 13 (12–14) 0.41 (0.38–0.43) 0.7 (0.64–0.73) 6 122 10 80 (75–85)
2005 61 6.3 (5.9–6.7) 10 (9.5–11) 0.42 (0.39–0.44) 0.7 (0.63–0.72) 5 003 8.1 80 (75–85)
2010 63 6 (5.6–6.4) 9.5 (8.9–10) 0.42 (0.40–0.45) 0.7 (0.63–0.71) 4 801 7.6 80 (75–85)
2011 64 5.9 (5.5–6.2) 9.2 (8.6–9.8) 0.4 (0.38–0.43) 0.6 (0.60–0.68) 4 681 7.4 80 (75–85)
2012 64 5.3 (4.9–5.6) 8.2 (7.7–8.7) 0.37 (0.34–0.39) 0.6 (0.54–0.61)
a
Rates are per 100 000 population.
b
NOTIFIED NEW AND RELAPSE includes cases for which the treatment history is unknown.

232 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($,QFLGHQFHQRWLILFDWLRQDQGFDVHGHWHFWLRQUDWHVDOOIRUPV±
INCIDENCE (INCLUDING HIV) INCIDENCE HIV-POSITIVE NOTIFIED NEW AND RELAPSE
b
CASE DETECTION
YEAR POPULATION
(MILLIONS)
NUMBER
(THOUSANDS) RATE
a
NUMBER
(THOUSANDS) RATE
a
NUMBER RATE
a
PERCENT
Georgia 1990 5 15 (14–17) 280 (250–312) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 1 537 28 10 (9.0–11)
1995 5 13 (12–15) 263 (234–293) 0.012 (0.011–0.013) 0.2 (0.21–0.26) 1 625 32 12 (11–14)
2000 5 12 (11–14) 256 (228–285) 0.024 (0.022–0.027) 0.5 (0.46–0.57) 4 397 93 36 (32–41)
2005 4 7.8 (7.0–8.7) 175 (156–195) 0.031 (0.028–0.035) 0.7 (0.62–0.78) 4 503 101 58 (52–65)
2010 4 5.6 (5.0–6.2) 128 (114–142) 0.043 (0.038–0.048) 1 (0.88–1.1) 4 678 107 83 (75–94)
2011 4 5.5 (4.9–6.1) 125 (112–140) 0.048 (0.043–0.054) 1.1 (0.98–1.2) 4 547 104 83 (74–93)
2012 4 5 (4.5–5.6) 116 (103–130) 0.05 (0.045–0.056) 1.2 (1.0–1.3) 3 940 90 78 (70–88)
Germany 1990 80 17 (15–19) 21 (18–24) 0.081 (0.071–0.092) 0.1 (<0.1–0.11) 14 653 18 87 (77–99)
1995 83 14 (12–16) 17 (15–19) 0.083 (0.073–0.094) 0.1 (<0.1–0.11) 12 198 15 87 (77–99)
2000 84 10 (9.1–12) 12 (11–14) 0.054 (0.047–0.061) <0.1 (<0.1–<0.1) 9 064 11 87 (77–99)
2005 84 6.6 (5.7–7.4) 7.8 (6.9–8.8) 0.043 (0.037–0.048) <0.1 (<0.1–<0.1) 5 700 6.8 87 (77–99)
2010 83 4.7 (4.1–5.3) 5.6 (4.9–6.4) 0.034 (0.029–0.038) <0.1 (<0.1–<0.1) 4 059 4.9 87 (77–99)
2011 83 4.7 (4.1–5.3) 5.7 (5.0–6.4) 0.034 (0.030–0.038) <0.1 (<0.1–<0.1) 4 089 4.9 87 (77–99)
2012 83 4.6 (4.1–5.3) 5.6 (4.9–6.4) 0.034 (0.030–0.039) <0.1 (<0.1–<0.1) 4 043 4.9 87 (77–99)
Greece 1990 10 1 (0.880–1.1) 9.9 (8.7–11) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 877 8.6 87 (77–99)
1995 11 1.1 (0.950–1.2) 10 (8.9–11) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 939 8.8 87 (77–99)
2000 11 0.81 (0.710–0.920) 7.4 (6.4–8.3) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 703 6.4 87 (77–99)
2005 11 0.8 (0.700–0.900) 7.2 (6.3–8.2) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 693 6.3 87 (77–99)
2010 11 0.51 (0.450–0.580) 4.6 (4.0–5.2) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 445 4 87 (77–99)
2011 11 0.52 (0.460–0.590) 4.7 (4.1–5.3) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 454 4.1 87 (77–99)
2012 11 0.5 (0.440–0.570) 4.5 (3.9–5.1) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1)
Greenland 1990 < 1 0.11 (0.093–0.120) 191 (167–216)
1995 < 1 0.11 (0.093–0.120) 191 (167–216)
2000 < 1 0.11 (0.094–0.120) 191 (167–216)
2005 < 1 0.11 (0.095–0.120) 191 (167–216)
2010 < 1 0.13 (0.110–0.150) 232 (203–262) 114 202 87 (77–99)
2011 < 1 0.13 (0.120–0.150) 234 (205–264) 115 203 87 (77–99)
2012 < 1 0.097 (0.085–0.110) 170 (149–193) 84 148 87 (77–99)
Hungary 1990 10 4 (3.5–4.5) 39 (34–44) 0.024 (0.021–0.027) 0.2 (0.20–0.26) 3 588 35 90 (79–100)
1995 10 4.9 (4.3–5.6) 48 (42–54) 0.028 (0.024–0.031) 0.3 (0.23–0.30) 4 339 42 88 (78–100)
2000 10 3.8 (3.3–4.3) 37 (33–42) 0.015 (0.013–0.017) 0.2 (0.13–0.17) 3 073 30 81 (71–92)
2005 10 2.2 (1.9–2.5) 22 (19–25) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 1 808 18 82 (72–93)
2010 10 1.7 (1.5–2.0) 17 (15–19) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 1 543 15 90 (79–100)
2011 10 1.8 (1.6–2.0) 18 (16–20) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 1 279 13 72 (63–82)
2012 10 1.8 (1.6–2.0) 18 (16–20) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 1 159 12 65 (57–75)
Iceland 1990 < 1 0.021 (0.018–0.023) 8.1 (7.1–9.2) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 18 7.1 87 (77–99)
1995 < 1 0.014 (0.012–0.016) 5.2 (4.5–5.8) <0.01 (<0.01–<0.01) 0.1 (<0.1–0.12) 12 4.5 87 (77–99)
2000 < 1 0.015 (0.013–0.017) 5.3 (4.7–6.0) <0.01 (<0.01–<0.01) 0.2 (0.16–0.21) 13 4.6 87 (77–99)
2005 < 1 0.012 (0.010–0.013) 3.9 (3.4–4.4) <0.01 (<0.01–<0.01) 0.2 (0.21–0.27) 10 3.4 87 (77–99)
2010 < 1 0.025 (0.022–0.029) 8 (7.0–9.0) <0.01 (<0.01–<0.01) 0.6 (0.49–0.63) 22 6.9 87 (77–99)
2011 < 1 <0.01 (<0.01–0.010) 2.9 (2.5–3.2) <0.01 (<0.01–<0.01) 0.2 (0.17–0.22) 8 2.5 87 (77–99)
2012 < 1 0.012 (0.010–0.013) 3.5 (3.1–4.0) <0.01 (<0.01–<0.01) 0.2 (0.21–0.28) 10 3.1 87 (77–99)
Ireland 1990 4 0.72 (0.630–0.810) 20 (18–23) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 624 18 87 (77–99)
1995 4 0.53 (0.460–0.600) 15 (13–17) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 458 13 87 (77–99)
2000 4 0.44 (0.390–0.500) 12 (10–13) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 386 10 87 (77–99)
2005 4 0.49 (0.430–0.550) 12 (10–13) <0.01 (<0.01–<0.01) 0.1 (<0.1–0.11) 423 10 87 (77–99)
2010 4 0.46 (0.400–0.520) 10 (8.9–12) <0.01 (<0.01–<0.01) 0.1 (<0.1–0.11) 396 8.9 87 (77–99)
2011 5 0.46 (0.400–0.520) 10 (8.9–11) <0.01 (<0.01–<0.01) 0.1 (<0.1–0.11) 398 8.8 87 (77–99)
2012 5 0.39 (0.340–0.440) 8.6 (7.5–9.7) <0.01 (<0.01–<0.01) <0.1 (<0.1–0.10) 341 7.5 87 (77–99)
Israel 1990 4 0.27 (0.240–0.300) 6 (5.2–6.8) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 234 5.2 87 (77–99)
1995 5 0.46 (0.400–0.520) 8.6 (7.5–9.7) 0.012 (0.010–0.013) 0.2 (0.19–0.25) 398 7.5 87 (77–99)
2000 6 0.62 (0.540–0.700) 10 (9.0–12) 0.019 (0.017–0.022) 0.3 (0.28–0.37) 537 8.9 87 (77–99)
2005 7 0.43 (0.370–0.480) 6.5 (5.7–7.3) 0.014 (0.012–0.016) 0.2 (0.19–0.24) 371 5.6 87 (77–99)
2010 7 0.39 (0.340–0.440) 5.3 (4.6–6.0) 0.014 (0.013–0.016) 0.2 (0.17–0.22) 340 4.6 87 (77–99)
2011 8 0.47 (0.420–0.540) 6.3 (5.5–7.1) 0.018 (0.016–0.020) 0.2 (0.21–0.27) 412 5.5 87 (77–99)
2012 8 0.58 (0.510–0.660) 7.6 (6.7–8.6) 0.022 (0.019–0.025) 0.3 (0.25–0.33) 506 6.6 87 (77–99)
Italy 1990 57 4.9 (4.3–5.5) 8.6 (7.5–9.7) 0.052 (0.046–0.059) <0.1 (<0.1–0.10) 4 246 7.5 87 (77–99)
1995 57 6.5 (5.7–7.3) 11 (10–13) 0.13 (0.11–0.15) 0.2 (0.20–0.26) 5 627 9.9 87 (77–99)
2000 57 4 (3.5–4.6) 7.1 (6.2–8.0) 0.056 (0.049–0.064) 0.1 (<0.1–0.11) 3 501 6.1 87 (77–99)
2005 59 4.4 (3.9–5.0) 7.5 (6.6–8.5) 0.065 (0.057–0.073) 0.1 (0.10–0.12) 3 844 6.6 87 (77–99)
2010 61 3.7 (3.2–4.1) 6 (5.3–6.8) 0.055 (0.049–0.063) <0.1 (<0.1–0.10) 3 175 5.2 87 (77–99)
2011 61 3.9 (3.4–4.5) 6.5 (5.7–7.3) 0.06 (0.053–0.068) 0.1 (<0.1–0.11) 3 421 5.6 87 (77–99)
2012 61 4.1 (3.6–4.6) 6.7 (5.8–7.5) 0.062 (0.055–0.071) 0.1 (<0.1–0.12)
Kazakhstan 1990 16 13 (11–15) 79 (66–92) <0.01 (<0.01–<0.01) <0.1 (0–<0.1) 10 969 68 86 (74–100)
1995 16 50 (42–58) 318 (269–372) 0.03 (0.025–0.035) 0.2 (0.16–0.22) 11 310 73 23 (20–27)
2000 15 51 (43–60) 351 (297–411) 0.23 (0.19–0.27) 1.6 (1.3–1.9) 25 843 177 50 (43–60)
2005 15 35 (30–41) 235 (199–275) 0.28 (0.24–0.33) 1.9 (1.6–2.2) 28 629 190 81 (69–96)
2010 16 29 (24–34) 182 (154–213) 0.3 (0.26–0.36) 1.9 (1.6–2.2) 23 399 147 81 (69–96)
2011 16 31 (26–36) 193 (163–225) 0.35 (0.29–0.41) 2.2 (1.8–2.5) 25 074 156 81 (69–96)
2012 16 22 (19–26) 137 (116–160) 0.26 (0.22–0.31) 1.6 (1.4–1.9) 18 006 111 81 (69–96)
Kyrgyzstan 1990 4 4 (3.3–4.8) 92 (76–109) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 2 306 52 57 (48–69)
1995 5 7.7 (6.4–9.2) 168 (138–200) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 3 393 74 44 (37–53)
2000 5 12 (10–15) 249 (205–296) 0.016 (0.013–0.019) 0.3 (0.27–0.38) 6 205 125 50 (42–61)
2005 5 10 (8.6–12) 208 (171–248) 0.059 (0.048–0.070) 1.2 (0.96–1.4) 6 329 126 60 (51–73)
2010 5 7.5 (6.2–9.0) 141 (116–168) 0.17 (0.14–0.21) 3.2 (2.7–3.8) 5 652 106 75 (63–91)
2011 5 7.6 (6.3–9.1) 141 (116–168) 0.22 (0.19–0.27) 4.2 (3.4–5.0) 5 980 111 78 (66–95)
2012 5 7.7 (6.4–9.2) 141 (116–168) 0.29 (0.24–0.34) 5.3 (4.4–6.3) 6 195 113 80 (67–97)
Latvia 1990 3 1.5 (1.3–1.7) 57 (50–65) <0.01 (<0.01–<0.01) 0.1 (<0.1–0.12) 906 34 59 (52–68)
1995 2 3.1 (2.7–3.5) 126 (111–142) 0.02 (0.018–0.023) 0.8 (0.71–0.91) 1 541 62 49 (44–56)
2000 2 2.9 (2.5–3.2) 121 (106–137) 0.06 (0.053–0.068) 2.6 (2.2–2.9) 1 982 84 69 (61–79)
2005 2 1.7 (1.5–1.9) 75 (66–85) 0.089 (0.078–0.10) 4 (3.5–4.5) 1 409 63 84 (74–96)
2010 2 1 (0.930–1.2) 50 (45–56) 0.089 (0.079–0.099) 4.3 (3.8–4.8) 913 44 87 (78–98)
2011 2 0.99 (0.890–1.1) 48 (43–53) 0.089 (0.079–0.099) 4.3 (3.8–4.8) 864 42 87 (78–97)
2012 2 1.1 (1.0–1.2) 53 (49–58) 0.1 (0.093–0.11) 5 (4.5–5.4) 959 47 87 (80–95)
Lithuania 1990 4 1.6 (1.4–1.9) 44 (37–52) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 1 471 40 90 (77–110)
1995 4 3.2 (2.8–3.7) 89 (77–102) 0.013 (0.011–0.015) 0.4 (0.31–0.41) 2 362 65 73 (64–85)
2000 3 3.6 (3.2–4.0) 103 (92–114) 0.037 (0.033–0.041) 1.1 (0.95–1.2) 2 657 76 74 (66–83)
2005 3 2.8 (2.5–3.2) 87 (76–97) 0.058 (0.051–0.065) 1.8 (1.6–2.0) 2 114 64 74 (66–84)
2010 3 2.2 (1.9–2.5) 73 (63–82) 0.067 (0.058–0.076) 2.2 (1.9–2.5) 1 751 57 79 (69–90)
2011 3 2.1 (1.8–2.4) 69 (61–78) 0.07 (0.061–0.079) 2.3 (2.0–2.6) 1 748 57 83 (73–95)
2012 3 2 (1.8–2.3) 66 (58–75) 0.071 (0.062–0.080) 2.3 (2.0–2.6) 1 635 54 82 (72–93)
Luxembourg 1990 < 1 0.055 (0.048–0.062) 14 (13–16) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 48 13 87 (77–99)
1995 < 1 0.037 (0.032–0.042) 9 (7.9–10) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 32 7.8 87 (77–99)
2000 < 1 0.051 (0.044–0.057) 12 (10–13) <0.01 (<0.01–<0.01) 0.1 (<0.1–0.17) 44 10 87 (77–99)
2005 < 1 0.043 (0.037–0.048) 9.3 (8.1–11) <0.01 (<0.01–<0.01) 0.2 (0.10–0.21) 37 8.1 87 (77–99)
2010 < 1 0.033 (0.029–0.038) 6.6 (5.8–7.4) <0.01 (<0.01–<0.01) 0.1 (<0.1–0.15) 29 5.7 87 (77–99)
2011 < 1 0.029 (0.025–0.033) 5.6 (4.9–6.3) <0.01 (<0.01–<0.01) <0.1 (<0.1–0.13) 25 4.8 87 (77–99)
2012 < 1 0.034 (0.030–0.039) 6.5 (5.7–7.4) <0.01 (<0.01–<0.01) 0.1 (<0.1–0.15) 45 8.6 130 (120–150)
Malta 1990 < 1 0.015 (0.013–0.017) 4 (3.5–4.5) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 13 3.5 87 (77–99)
1995 < 1 0.013 (0.011–0.014) 3.2 (2.8–3.6) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 11 2.8 87 (77–99)
2000 < 1 0.018 (0.016–0.021) 4.5 (4.0–5.1) <0.01 (<0.01–<0.01) <0.1 (<0.1–0.10) 16 3.9 87 (77–99)
2005 < 1 0.025 (0.022–0.029) 6.1 (5.3–6.9) <0.01 (<0.01–<0.01) 0.2 (0.16–0.20) 22 5.3 87 (77–99)
2010 < 1 0.033 (0.029–0.038) 7.9 (6.9–8.9) <0.01 (<0.01–<0.01) 0.3 (0.22–0.28) 29 6.8 87 (77–99)
2011 < 1 0.035 (0.030–0.039) 8.1 (7.1–9.2) <0.01 (<0.01–<0.01) 0.3 (0.22–0.28) 30 7 87 (77–99)
2012 < 1 0.048 (0.042–0.055) 11 (9.9–13) <0.01 (<0.01–<0.01) 0.3 (0.30–0.39) 42 9.8 87 (77–99)
Monaco 1990 < 1 <0.01 (<0.01–<0.01) 3.9 (3.4–4.4) 1 3.4 87 (77–99)
1995 < 1 <0.01 (<0.01–<0.01) 3.7 (3.3–4.2) 1 3.3 87 (77–99)
2000 < 1 0 (0–0) 0 (0–0) 0 0
2005 < 1 <0.01 (<0.01–<0.01) 1.2 (1.0–1.3)
2010 < 1 <0.01 (<0.01–<0.01) 3.1 (2.7–3.5) 1 2.7 87 (77–99)
2011 < 1 <0.01 (<0.01–<0.01) 2.1 (1.8–2.4)
2012 < 1 <0.01 (<0.01–<0.01) 2.1 (1.8–2.4)
a
Rates are per 100 000 population.
b
NOTIFIED NEW AND RELAPSE includes cases for which the treatment history is unknown.
233 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
EUROPEAN REGION
7$%/($,QFLGHQFHQRWLILFDWLRQDQGFDVHGHWHFWLRQUDWHVDOOIRUPV±
INCIDENCE (INCLUDING HIV) INCIDENCE HIV-POSITIVE NOTIFIED NEW AND RELAPSE
b
CASE DETECTION
YEAR POPULATION
(MILLIONS)
NUMBER
(THOUSANDS) RATE
a
NUMBER
(THOUSANDS) RATE
a
NUMBER RATE
a
PERCENT
Montenegro 2005 < 1 0.18 (0.160–0.200) 29 (26–33) 156 25 87 (77–99)
2010 < 1 0.13 (0.110–0.140) 20 (18–23) <0.01 (0–<0.01) 0.2 (0–1.2) 110 18 87 (78–98)
2011 < 1 0.13 (0.110–0.140) 20 (18–23) 110 18 87 (78–97)
2012 < 1 0.11 (0.100–0.130) 18 (16–20) <0.01 (0–<0.01) <0.1 (0–0.54) 98 16 87 (78–97)
Netherlands 1990 15 1.6 (1.4–1.8) 11 (9.3–12) 0.013 (0.012–0.015) <0.1 (<0.1–0.10) 1 369 9.2 87 (77–99)
1995 15 1.9 (1.6–2.1) 12 (11–14) 0.058 (0.051–0.065) 0.4 (0.33–0.42) 1 619 10 87 (77–99)
2000 16 1.4 (1.3–1.6) 9 (7.9–10) 0.05 (0.044–0.057) 0.3 (0.28–0.36) 1 244 7.8 87 (77–99)
2005 16 1.3 (1.1–1.5) 8 (7.0–9.0) 0.046 (0.040–0.052) 0.3 (0.25–0.32) 1 127 6.9 87 (77–99)
2010 17 1.2 (1.1–1.4) 7.2 (6.3–8.2) 0.048 (0.042–0.054) 0.3 (0.25–0.33) 1 046 6.3 87 (77–99)
2011 17 1.1 (0.990–1.3) 6.8 (5.9–7.7) 0.046 (0.040–0.052) 0.3 (0.24–0.31) 981 5.9 87 (77–99)
2012 17 1.1 (0.930–1.2) 6.3 (5.5–7.2) 0.043 (0.038–0.049) 0.3 (0.23–0.29) 920 5.5 87 (77–99)
Norway 1990 4 0.33 (0.290–0.370) 7.7 (6.8–8.7) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 285 6.7 87 (77–99)
1995 4 0.27 (0.240–0.310) 6.2 (5.5–7.0) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 236 5.4 87 (77–99)
2000 4 0.25 (0.220–0.290) 5.7 (5.0–6.4) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 221 4.9 87 (77–99)
2005 5 0.32 (0.280–0.360) 6.9 (6.0–7.8) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 276 6 87 (77–99)
2010 5 0.34 (0.300–0.390) 7 (6.1–7.9) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 297 6.1 87 (77–99)
2011 5 0.37 (0.330–0.420) 7.5 (6.6–8.5) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 324 6.6 87 (77–99)
2012 5 0.37 (0.330–0.420) 7.5 (6.6–8.5) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1)
Poland 1990 38 19 (16–21) 49 (43–55) 0.019 (0.016–0.021) <0.1 (<0.1–<0.1) 16 136 42 87 (77–99)
1995 38 18 (16–21) 48 (42–54) 0.051 (0.045–0.058) 0.1 (0.12–0.15) 15 958 41 87 (77–99)
2000 38 13 (11–14) 33 (29–37) 0.039 (0.034–0.044) 0.1 (<0.1–0.12) 10 931 29 87 (77–99)
2005 38 9.4 (8.3–11) 25 (22–28) 0.034 (0.030–0.038) <0.1 (<0.1–0.10) 8 203 21 87 (77–99)
2010 38 8.1 (7.1–9.1) 21 (18–24) 0.031 (0.028–0.036) <0.1 (<0.1–<0.1) 7 002 18 87 (77–99)
2011 38 9.1 (8.0–10) 24 (21–27) 0.037 (0.032–0.041) 0.1 (<0.1–0.11) 7 946 21 87 (77–99)
2012 38 8.1 (7.1–9.2) 21 (19–24) 0.032 (0.028–0.037) <0.1 (<0.1–0.10) 7 054 18 87 (77–99)
Portugal 1990 10 7.1 (6.3–8.1) 72 (63–82) 0.11 (0.094–0.12) 1.1 (0.95–1.2) 6 214 63 87 (77–99)
1995 10 6.4 (5.6–7.3) 64 (56–72) 0.37 (0.33–0.42) 3.7 (3.2–4.2) 5 577 55 87 (77–99)
2000 10 4.9 (4.3–5.5) 47 (41–53) 0.35 (0.31–0.40) 3.4 (3.0–3.9) 4 227 41 87 (77–99)
2005 11 3.8 (3.3–4.3) 36 (32–41) 0.38 (0.33–0.43) 3.6 (3.1–4.1) 3 308 31 87 (77–99)
2010 11 2.9 (2.5–3.2) 27 (24–31) 0.33 (0.29–0.37) 3.1 (2.7–3.5) 2 487 23 87 (77–99)
2011 11 2.8 (2.4–3.1) 26 (23–30) 0.32 (0.28–0.36) 3 (2.7–3.4) 2 406 23 87 (77–99)
2012 11 2.8 (2.4–3.1) 26 (23–30) 0.33 (0.29–0.37) 3.1 (2.7–3.5) 2 490 23 89 (79–100)
Republic of 1990 4 2.3 (1.9–2.8) 54 (44–64) <0.01 (<0.01–<0.01) <0.1 (<0.1–0.11) 1 728 40 74 (62–89)
Moldova 1995 4 4.7 (3.9–5.6) 109 (90–130) 0.018 (0.015–0.022) 0.4 (0.35–0.51) 2 925 67 62 (52–75)
2000 4 6 (5.0–7.2) 147 (121–175) 0.061 (0.051–0.073) 1.5 (1.2–1.8) 2 935 71 49 (41–59)
2005 4 6.6 (5.4–7.9) 175 (144–209) 0.18 (0.14–0.21) 4.7 (3.8–5.6) 5 141 136 78 (65–95)
2010 4 5.9 (4.9–7.1) 166 (137–198) 0.31 (0.26–0.37) 8.7 (7.1–10) 4 135 116 70 (59–85)
2011 4 5.7 (4.7–6.8) 161 (133–192) 0.33 (0.27–0.40) 9.4 (7.7–11) 4 233 119 74 (62–90)
2012 4 5.6 (4.6–6.7) 160 (132–190) 0.34 (0.28–0.40) 9.6 (7.9–11) 4 409 125 79 (66–95)
Romania 1990 23 34 (28–41) 146 (120–174) 0.075 (0.062–0.089) 0.3 (0.26–0.38) 16 256 70 48 (40–58)
1995 23 43 (36–52) 189 (155–226) 0.36 (0.30–0.43) 1.6 (1.3–1.9) 23 271 101 54 (45–65)
2000 22 41 (33–48) 181 (149–216) 0.47 (0.38–0.56) 2.1 (1.7–2.5) 27 470 123 68 (57–82)
2005 22 32 (27–39) 147 (121–175) 0.66 (0.55–0.79) 3 (2.5–3.6) 26 106 118 81 (67–98)
2010 22 24 (20–28) 109 (89–130) 0.76 (0.63–0.91) 3.5 (2.9–4.2) 18 379 84 77 (65–94)
2011 22 22 (18–26) 101 (83–121) 0.67 (0.55–0.80) 3.1 (2.5–3.7) 16 992 78 77 (64–93)
2012 22 20 (17–24) 94 (77–112) 0.6 (0.49–0.72) 2.8 (2.3–3.3) 16 036 74 79 (66–95)
Russian 1990 148 70 (59–81) 47 (40–55) 50 641 34 73 (62–86)
Federation 1995 149 140 (120–170) 96 (81–112) 0.014 (0.012–0.017) <0.1 (<0.1–<0.1) 84 980 57 60 (51–70)
2000 147 190 (160–220) 127 (108–149) 0.41 (0.35–0.48) 0.3 (0.24–0.33) 140 677 96 75 (65–89)
2005 144 190 (160–230) 135 (114–158) 5.8 (4.9–6.8) 4 (3.4–4.7) 127 930 89 66 (56–78)
2010 144 150 (130–180) 106 (89–123) 8.7 (7.4–10) 6.1 (5.1–7.1) 125 310 87 83 (71–98)
2011 143 140 (120–160) 97 (82–114) 9.1 (7.7–11) 6.3 (5.3–7.4) 112 910 79 81 (69–95)
2012 143 130 (110–150) 91 (77–106) 9.3 (7.9–11) 6.5 (5.5–7.5) 105 753 74 81 (70–96)
San Marino 1990 < 1 <0.01 (<0.01–<0.01) 4.8 (4.2–5.4) 1 4.1 87 (77–99)
1995 < 1 <0.01 (<0.01–<0.01) 9 (7.8–10) 2 7.8 87 (77–99)
2000 < 1 <0.01 (<0.01–<0.01) 4.3 (3.7–4.8) 1 3.7 87 (77–99)
2005 < 1 <0.01 (<0.01–<0.01) 1.5 (1.3–1.7)
2010 < 1 <0.01 (<0.01–<0.01) 1.5 (1.3–1.7)
2011 < 1 <0.01 (<0.01–<0.01) 1.5 (1.3–1.7)
2012 < 1 <0.01 (<0.01–<0.01) 1.5 (1.3–1.7)
Serbia 2005 10 3.7 (3.2–4.2) 37 (32–42) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 3 208 32 87 (77–100)
2010 10 2.7 (2.3–3.0) 28 (24–32) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 2 333 24 87 (77–100)
2011 10 2.5 (2.2–2.8) 26 (23–30) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 2 174 23 87 (77–100)
2012 10 2.2 (1.9–2.4) 23 (20–26) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 1 872 20 87 (77–100)
Serbia & 1990 10 7 (5.0–9.4) 68 (48–91) 4 194 41 60 (45–84)
Montenegro 1995 11 6.7 (5.7–7.9) 61 (52–72) 2 798 25 41 (35–49)
2000 11 5 (4.2–5.8) 46 (38–54) 2 864 26 58 (49–69)
Slovakia 1990 5 1.7 (1.5–1.9) 32 (28–36) 1 448 27 87 (77–99)
1995 5 1.8 (1.6–2.0) 33 (29–37) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 1 540 29 87 (77–99)
2000 5 1.2 (1.0–1.3) 22 (19–24) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 1 010 19 87 (77–99)
2005 5 0.82 (0.720–0.920) 15 (13–17) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 710 13 87 (77–99)
2010 5 0.47 (0.410–0.530) 8.7 (7.6–9.8) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 409 7.5 87 (77–99)
2011 5 0.43 (0.380–0.490) 8 (7.0–9.0) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 378 6.9 87 (77–99)
2012 5 0.37 (0.320–0.420) 6.8 (5.9–7.7) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 321 5.9 87 (77–99)
Slovenia 1990 2 0.83 (0.730–0.940) 41 (36–47) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 722 36 87 (77–99)
1995 2 0.6 (0.530–0.680) 30 (27–34) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 525 26 87 (77–99)
2000 2 0.42 (0.370–0.480) 21 (19–24) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 368 18 87 (77–99)
2005 2 0.31 (0.270–0.350) 15 (14–18) <0.01 (<0.01–<0.01) <0.1 (<0.1–0.10) 269 13 87 (77–99)
2010 2 0.19 (0.170–0.220) 9.5 (8.3–11) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 169 8.2 87 (77–99)
2011 2 0.21 (0.180–0.240) 10 (8.8–11) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 181 8.8 87 (77–99)
2012 2 0.15 (0.140–0.170) 7.5 (6.5–8.4) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 134 6.5 87 (77–99)
Spain 1990 39 8.7 (7.7–9.9) 22 (20–25) 0.4 (0.35–0.45) 1 (0.90–1.2) 7 600 20 87 (77–99)
1995 39 10 (8.8–11) 26 (22–29) 0.84 (0.73–0.95) 2.1 (1.9–2.4) 8 764 22 87 (77–99)
2000 40 9.2 (8.1–10) 23 (20–26) 0.71 (0.62–0.81) 1.8 (1.5–2.0) 7 993 20 87 (77–99)
2005 43 8.4 (7.3–9.5) 19 (17–22) 0.77 (0.67–0.87) 1.8 (1.6–2.0) 7 281 17 87 (77–99)
2010 46 7.8 (6.8–8.8) 17 (15–19) 0.7 (0.61–0.79) 1.5 (1.3–1.7) 6 765 15 87 (77–99)
2011 47 7.4 (6.4–8.3) 16 (14–18) 0.66 (0.58–0.75) 1.4 (1.2–1.6) 6 392 14 87 (77–99)
2012 47 6.5 (5.7–7.4) 14 (12–16) 0.58 (0.51–0.66) 1.3 (1.1–1.4) 5 677 12 87 (77–99)
Sweden 1990 9 0.64 (0.560–0.720) 7.5 (6.6–8.5) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 557 6.5 87 (77–99)
1995 9 0.65 (0.570–0.730) 7.3 (6.4–8.3) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 564 6.4 87 (77–99)
2000 9 0.48 (0.420–0.540) 5.4 (4.7–6.1) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 417 4.7 87 (77–99)
2005 9 0.62 (0.540–0.700) 6.9 (6.0–7.8) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 539 6 87 (77–99)
2010 9 0.72 (0.630–0.810) 7.6 (6.7–8.6) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 623 6.6 87 (77–99)
2011 9 0.63 (0.550–0.710) 6.6 (5.8–7.5) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 544 5.8 87 (77–99)
2012 10 0.68 (0.600–0.770) 7.2 (6.3–8.1) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 593 6.2 87 (77–99)
Switzerland 1990 7 1.5 (1.3–1.7) 22 (19–25) 0.011 (<0.01–0.013) 0.2 (0.15–0.19) 1 278 19 87 (77–99)
1995 7 0.95 (0.840–1.1) 14 (12–15) 0.015 (0.013–0.017) 0.2 (0.18–0.24) 830 12 87 (77–99)
2000 7 0.66 (0.580–0.750) 9.3 (8.1–10) <0.01 (<0.01–<0.01) 0.1 (0.11–0.14) 577 8.1 87 (77–99)
2005 7 0.59 (0.520–0.670) 8 (7.0–9.0) <0.01 (<0.01–<0.01) 0.1 (<0.1–0.12) 514 6.9 87 (77–99)
2010 8 0.58 (0.510–0.660) 7.5 (6.5–8.4) <0.01 (<0.01–0.010) 0.1 (0.10–0.13) 508 6.5 87 (77–99)
2011 8 0.6 (0.530–0.680) 7.6 (6.7–8.6) <0.01 (<0.01–0.011) 0.1 (0.10–0.13) 524 6.6 87 (77–99)
2012 8 0.48 (0.420–0.540) 6 (5.2–6.8) <0.01 (<0.01–<0.01) <0.1 (<0.1–0.11) 416 5.2 87 (77–99)
Tajikistan 1990 5 3.7 (3.0–4.4) 70 (58–83) <0.01 (<0.01–<0.01) 0.1 (<0.1–0.12) 2 460 46 67 (56–81)
1995 6 8.6 (7.1–10) 148 (122–176) 0.03 (0.025–0.036) 0.5 (0.42–0.62) 2 029 35 24 (20–29)
2000 6 14 (11–16) 220 (182–263) 0.14 (0.11–0.16) 2.2 (1.8–2.7) 2 779 45 20 (17–25)
2005 7 14 (11–16) 200 (165–238) 0.25 (0.20–0.30) 3.6 (3.0–4.3) 5 460 80 40 (34–49)
2010 8 9.8 (8.1–12) 129 (106–153) 0.22 (0.18–0.26) 2.8 (2.3–3.4) 6 994 92 71 (60–86)
2011 8 9.2 (7.6–11) 118 (97–140) 0.21 (0.17–0.25) 2.7 (2.2–3.2) 7 035 90 76 (64–93)
2012 8 8.6 (7.1–10) 108 (89–128) 0.2 (0.16–0.24) 2.5 (2.0–3.0) 6 508 81 75 (63–91)
a
Rates are per 100 000 population.
b
NOTIFIED NEW AND RELAPSE includes cases for which the treatment history is unknown.
234 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($,QFLGHQFHQRWLILFDWLRQDQGFDVHGHWHFWLRQUDWHVDOOIRUPV±
INCIDENCE (INCLUDING HIV) INCIDENCE HIV-POSITIVE NOTIFIED NEW AND RELAPSE
b
CASE DETECTION
YEAR POPULATION
(MILLIONS)
NUMBER
(THOUSANDS) RATEaNUMBER
(THOUSANDS) RATEaNUMBER RATEaPERCENT
The Former 1990 2 1.6 (1.0–2.4) 81 (50–119)
Yugoslav Republic 1995 2 1.1 (0.930–1.4) 58 (47–69) 786 40 69 (58–85)
of Macedonia 2000 2 0.85 (0.690–1.0) 41 (34–50) 641 31 75 (63–92)
2005 2 0.62 (0.560–0.680) 30 (27–33) 598 29 97 (88–110)
2010 2 0.44 (0.380–0.510) 21 (18–24) 384 18 87 (75–100)
2011 2 0.41 (0.360–0.480) 20 (17–23) 335 16 81 (70–94)
2012 2 0.39 (0.330–0.450) 18 (16–21) 346 16 89 (78–100)
Turkey 1990 54 28 (25–32) 52 (46–59) 24 468 45 87 (77–99)
1995 59 26 (23–30) 45 (40–51) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 22 981 39 87 (77–99)
2000 63 21 (18–23) 33 (29–37) 0.019 (0.016–0.021) <0.1 (<0.1–<0.1) 18 038 29 87 (77–99)
2005 68 23 (20–26) 34 (29–38) 0.05 (0.044–0.057) <0.1 (<0.1–<0.1) 19 744 29 87 (77–99)
2010 72 18 (16–21) 25 (22–29) 0.033 (0.029–0.037) <0.1 (<0.1–<0.1) 15 879 22 87 (77–99)
2011 73 17 (15–20) 24 (21–27) 0.033 (0.029–0.037) <0.1 (<0.1–<0.1) 15 054 21 87 (77–99)
2012 74 16 (14–18) 22 (19–25) 0.033 (0.028–0.037) <0.1 (<0.1–<0.1) 14 139 19 87 (77–99)
Turkmenistan 1990 4 3.5 (2.8–4.2) 95 (76–115) 2 325 63 67 (55–83)
1995 4 6.6 (5.4–7.8) 157 (129–187) 1 939 46 30 (25–36)
2000 5 9.4 (7.6–11) 209 (170–252) 4 038 90 43 (36–53)
2005 5 8.3 (6.8–10) 175 (144–210) 3 191 67 38 (32–47)
2010 5 5.2 (4.3–6.1) 103 (86–121) 3 230 64 62 (53–74)
2011 5 4.5 (3.7–5.5) 89 (73–107)
2012 5 3.9 (3.1–4.8) 75 (59–94)
Ukraine 1990 52 23 (19–27) 45 (37–53) 16 465 32 71 (60–86)
1995 51 38 (31–45) 74 (62–88) 0.15 (0.12–0.18) 0.3 (0.24–0.34) 21 459 42 57 (48–68)
2000 49 53 (44–63) 108 (90–129) 2.5 (2.0–2.9) 5 (4.1–5.9) 32 945 67 62 (52–75)
2005 47 57 (48–68) 121 (101–144) 5.8 (4.8–6.9) 12 (10–15) 39 608 84 69 (58–83)
2010 46 48 (41–57) 105 (88–123) 5.7 (4.8–6.7) 12 (10–15) 33 857 74 70 (60–83)
2011 46 46 (38–54) 99 (83–118) 5.3 (4.4–6.3) 12 (9.6–14) 34 237 75 75 (63–90)
2012 46 42 (35–51) 93 (77–112) 4.8 (3.9–5.7) 10 (8.6–13) 40 990 90 96 (81–120)
United Kingdom of 1990 57 6.6 (6.2–7.1) 12 (11–12) 0.071 (0.066–0.077) 0.1 (0.11–0.13) 5 908 10 89 (84–95)
Great Britain and 1995 58 6.9 (6.5–7.4) 12 (11–13) 0.087 (0.061–0.12) 0.2 (0.10–0.20) 6 176 11 89 (84–95)
Northern Ireland 2000 59 7 (6.5–7.4) 12 (11–13) 0.12 (0.088–0.16) 0.2 (0.15–0.27) 6 220 11 89 (84–95)
2005 60 9.2 (8.6–9.8) 15 (14–16) 0.25 (0.19–0.32) 0.4 (0.31–0.53) 8 173 14 89 (84–95)
2010 62 8.9 (8.3–9.4) 14 (13–15) 0.3 (0.23–0.38) 0.5 (0.37–0.62) 7 907 13 89 (84–95)
2011 62 9.5 (8.8–10) 15 (14–16) 0.32 (0.25–0.41) 0.5 (0.40–0.66) 8 439 14 89 (84–95)
2012 63 9.4 (8.8–10) 15 (14–16) 0.33 (0.25–0.41) 0.5 (0.40–0.66) 8 269 13 88 (82–94)
Uzbekistan 1990 21 26 (21–31) 125 (103–149) 0.057 (0.047–0.067) 0.3 (0.23–0.33) 9 414 46 37 (31–44)
1995 23 46 (38–55) 200 (165–238) 0.22 (0.18–0.26) 1 (0.79–1.1) 9 866 43 22 (18–26)
2000 25 71 (59–85) 287 (237–342) 0.57 (0.47–0.68) 2.3 (1.9–2.7) 15 750 63 22 (19–27)
2005 26 61 (50–72) 233 (193–278) 0.69 (0.57–0.82) 2.7 (2.2–3.2) 21 513 83 35 (30–43)
2010 28 34 (28–40) 122 (101–146) 0.56 (0.46–0.67) 2 (1.7–2.4) 16 883 61 50 (42–60)
2011 28 29 (24–34) 101 (84–121) 0.51 (0.42–0.61) 1.8 (1.5–2.2) 15 345 55 54 (45–65)
2012 29 22 (18–27) 78 (65–93) 0.44 (0.37–0.53) 1.6 (1.3–1.9) 14 832 52 66 (56–80)
a Rates are per 100 000 population.
b NOTIFIED NEW AND RELAPSE includes cases for which the treatment history is unknown.

235 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
EUROPEAN REGION
7$%/($&DVHQRWLILFDWLRQV±
NEW CASES
YEAR SMEAR-
POSITIVE
SMEAR-NEGATIVE/
UNKNOWN
EXTRA-
PULMONARY OTHER RELAPSE RE-TREAT EXCL.
RELAPSE
TOTAL
RETREAT
HISTORY
UNKNOWN
NEW AND
RELAPSE
b
% SMEAR-
POS AMONG
NEW PULM
NEW AND RELAPSE
NOTIFICATION RATE
a
1990–2012
Albania 1990 653 –
1995 641 139 223 226 53 53 38
2000 604 171 188 234 11 8 19 48
2005 506 196 134 167 9 34 43 59
2010 431 145 105 165 0 16 14 30 0 58
2011 422 180 105 128 0 9 9 18 0 63
• 19 13 • 2012 408 185 100 106 0 17 12 29 0 65
Andorra 1990 23 –
1995 –
2000 12 1 9 2 0 0 0 10
2005 10 5 1 4 0 0 0 83
2010 7 0 4 3 0 0 0 0 0 0
2011 3 1 2 0 0 0 1 1 0 33
• 42 11 • 2012 9 2 3 3 0 0 0 0 1 40
Armenia 1990 590 –
1995 1 000 436 451 75 38 38 49
2000 1 333 621 505 153 54 22 76 55
2005 2 206 581 1 049 365 211 116 327 36
2010 1 410 339 639 351 0 81 370 451 0 35
2011 1 261 329 582 289 0 61 321 382 0 36
• 17 41 • 2012 1 213 315 553 255 0 90 305 395 0 36
Austria 1990 1 521 –
1995 1 481 467 765 249 38
2000 1 185 324 652 209 0 30 30 33
2005 928 234 519 175 0 26 26 31
2010 659 76 213 69 0 0 29 29 301 26
2011 671 94 217 85 0 4 16 20 271 30
• 20 7 • 2012 620 95 218 97 0 12 28 40 198 30
Azerbaijan 1990 2 620 –
1995 1 630 669 620 93 47 47 52
2000 5 187 890 3 978 245 74 0 74 18
2005 6 034 1 561 2 508 651 1 314 1 886 3 200 38
2010 7 550 1 997 2 275 965 0 1 153 844 1 997 1 160 47
2011 9 146 1 426 2 740 1 130 0 1 201 954 2 155 2 649 34
• 36 68 • 2012 6 363 1 301 2 313 1 002 1 747 1 777 3 524 36
Belarus 1990 3 039 –
1995 4 854 1 845 2 148 518 343 343 46
2000 6 799 2 547 2 985 442 825 0 825 46
2005 5 308 1 235 3 710 363 1 049 1 049 25
2010 5 098 1 269 2 647 429 0 658 456 1 114 95 32
2011 4 697 1 217 2 439 387 654 421 1 075 33
• 30 51 • 2012 4 783 1 277 2 184 381 941 463 1 404 37
Belgium 1990 1 577 –
1995 1 380 400 534 366 80 80 43
2000 1 278 409 454 326 89 0 89 47
2005 1 076 380 406 290 68 68 48
2010 1 028 244 340 230 0 87 87 214 42
2011 985 240 273 192 0 59 59 280 47
• 16 8 • 2012 909 235 237 179 0 78 78 258 50
Bosnia and 1990 4 073 –
Herzegovina 1995 2 132 865 997 140 130 130 46
2000 2 476 759 1 287 261 169 24 193 37
2005 2 111 640 1 106 258 107 49 156 0 37
2010 1 321 441 529 161 158 32 69 101 0 45
2011 1 360 547 611 162 0 40 25 65 0 47
• 90 37 • 2012 1 409 569 554 176 2 108 11 119 0 51
Bulgaria 1990 2 256 –
1995 3 245 1 087 1 709 449 39
2000 3 349 2 524 0 442 383 0 383 100
2005 3 225 1 214 1 511 376 124 77 201 45
2010 2 412 806 748 747 0 111 237 348 0 52
2011 2 172 716 708 628 0 120 235 355 0 50
• 26 29 • 2012 2 081 741 618 606 1 115 199 314 0 55
Croatia 1990 2 576 –
1995 2 114 1 204 703 165 42 42 63
2000 1 630 –
2005 1 050 372 575 103 0 94 94 39
2010 688 183 382 87 36 7 43 32
2011 619 201 343 75 37
• 54 0 • 2012 –
Cyprus 1990 29 –
1995 36 6 11 13 0 0 35
2000 33 4 10 17 0 0 29
2005 34 9 13 12 0 3 3 41
2010 61 8 12 13 0 0 0 0 28 40
2011 51 11 14 5 1 0 3 3 20 44
• 4 6 • 2012 63 15 28 11 0 0 6 6 9 35
Czech Republic 1990 1 937 –
1995 1 834 487 1 026 300 21 21 32
2000 1 414 420 679 290 25 0 25 38
2005 973 308 461 204 0 34 34 40
2010 627 200 333 94 0 0 51 51 0 38
2011 569 188 307 74 0 0 31 31 0 38
• 19 5 • 2012 565 208 268 89 0 0 40 40 0 44
Denmark 1990 350 –
1995 448 128 186 128 6 6 41
2000 587 171 244 144 28 0 28 41
2005 395 129 145 121 0 29 29 47
2010 313 115 102 39 57 46 46 0 53
2011 359 124 100 45 90 22 22 0 55
• 7 0 • 2012 –
Estonia 1990 423 –
1995 624 369 124 60 71 71 75
2000 791 255 320 67 116 0 116 44
2005 479 162 217 46 54 40 94 43
2010 283 99 134 17 0 33 46 79 0 42
2011 296 123 124 18 0 31 45 76 0 50
• 27 20 • 2012 259 105 110 19 0 25 31 56 0 49
Finland 1990 772 –
1995 661 244 193 224 56
2000 527 205 136 157 29 0 29 60
2005 339 130 114 95 0 22 22 0 53
2010 312 82 146 84 0 0 15 15 0 36
2011 312 82 143 87 0 0 13 13 0 36
• 15 5 • 2012 261 78 104 70 0 5 13 18 4 43
a Rates are per 100 000 population.
b NEW AND RELAPSE includes cases for which the treatment history is unknown.

236 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($&DVHQRWLILFDWLRQV±
NEW CASES
YEAR SMEAR-
POSITIVE
SMEAR-NEGATIVE/
UNKNOWN
EXTRA-
PULMONARY OTHER RELAPSE RE-TREAT EXCL.
RELAPSE
TOTAL
RETREAT
HISTORY
UNKNOWN
NEW AND
RELAPSE
b
% SMEAR-
POS AMONG
NEW PULM
NEW AND RELAPSE
NOTIFICATION RATE
a
1990–2012
France 1990 9 030 –
1995 8 723 3 449 2 969 2 305 54
2000 6 122 1 815 1 364 1 665 0 0 57
2005 5 003 1 941 1 557 1 389 0 371 371 116 55
2010 4 801 960 1 015 765 12 0 315 315 2 049 49
2011 4 681 906 1 016 710 7 0 261 261 2 042 47
• 16 0 • 2012 –
Georgia 1990 1 537 –
1995 1 625 221 1 087 121 196 196 17
2000 4 397 601 2 213 1 324 259 422 681 21
2005 4 503 1 509 1 524 1 261 207 1 945 2 152 2 50
2010 4 678 2 140 1 088 1 155 0 291 1 118 1 409 4 66
2011 4 547 2 026 1 141 1 056 0 324 986 1 310 0 64
• 28 90 • 2012 3 940 1 648 1 186 944 0 161 1 034 1 195 1 58
Germany 1990 14 653 –
1995 12 198 3 852 6 473 1 873 37
2000 9 064 –
2005 5 700 1 379 2 801 1 211 148 345 493 161 33
2010 4 059 910 1 713 789 16 96 271 367 535 35
2011 4 089 951 1 787 735 17 73 227 300 526 35
• 18 5 • 2012 4 043 928 1 580 812 10 52 195 247 661 37
Greece 1990 877 –
1995 939 –
2000 703 235 339 81 48 48 41
2005 693 197 322 107 0 74 74 67 38
2010 445 178 129 49 0 0 44 44 89 58
2011 454 236 156 57 3 0 35 35 2 60
• 9 0 • 2012 –
Greenland 1990 –
1995 –
2000 –
2005 –
2010 114 38 59 7 10 2 12 39
2011 115 34 73 5 3 3 32
• 0 148 • 2012 84 33 44 5 2 2 43
Hungary 1990 3 588 –
1995 4 339 796 3 292 251 19
2000 3 073 412 2 361 221 79 292 371 15
2005 1 808 423 1 137 117 131 216 347 27
2010 1 543 270 1 147 70 0 56 198 254 0 19
2011 1 279 260 910 53 0 55 166 221 1 22
• 35 12 • 2012 1 159 273 831 35 0 20 64 84 0 25
Iceland 1990 18 –
1995 12 2 3 7 0 0 40
2000 13 1 7 4 1 0 1 12
2005 10 2 3 5 0 1 1 40
2010 22 6 12 4 0 0 0 0 0 33
2011 8 1 2 5 0 0 1 1 0 33
• 7 3 • 2012 10 2 5 3 0 0 1 1 0 29
Ireland 1990 624 –
1995 458 –
2000 386 138 150 96 2 20 22 48
2005 423 130 156 99 2 38 40 36 45
2010 396 84 122 112 1 31 31 77 41
2011 398 85 110 82 3 27 27 118 44
• 18 7 • 2012 341 77 97 75 1 25 25 91 44
Israel 1990 234 –
1995 398 –
2000 537 216 213 100 0 8 0 8 0 50
2005 371 142 168 55 0 6 1 7 0 46
2010 340 103 162 74 0 1 3 4 0 39
2011 412 135 207 66 0 4 6 10 0 39
• 5 7 • 2012 506 142 254 102 0 8 3 11 0 36
Italy 1990 4 246 –
1995 5 627 1 413 2 700 1 514 34
2000 3 501 687 891 522 269 356 625 44
2005 3 844 1 275 1 506 1 047 0 293 293 16 46
2010 3 175 586 779 328 0 0 74 74 1 482 43
2011 3 421 587 790 641 0 0 100 100 1 403 43
• 7 0 • 2012 –
Kazakhstan 1990 10 969 –
1995 11 310 3 022 5 966 1 002 1 320 1 320 34
2000 25 843 8 903 11 324 2 555 3 061 2 032 5 093 44
2005 28 629 6 911 14 472 920 3 209 11 800 15 009 3 117 32
2010 23 399 4 769 8 745 2 127 0 4 062 5 151 9 213 3 696 35
2011 25 074 4 157 8 242 1 997 0 4 739 1 230 5 969 5 939 34
• 68 111 • 2012 18 006 3 884 7 892 1 844 9 4 377 3 517 7 894 0 33
Kyrgyzstan 1990 2 306 –
1995 3 393 832 1 685 749 127 127 33
2000 6 205 1 296 2 929 1 683 297 258 555 31
2005 6 329 1 972 2 141 1 805 411 436 847 0 48
2010 5 652 1 645 2 028 1 635 0 344 643 987 45
2011 5 980 1 537 2 125 1 518 0 349 686 1 035 451 42
• 52 113 • 2012 6 195 1 594 2 448 1 809 344 721 1 065 0 39
Latvia 1990 906 –
1995 1 541 504 693 226 118 118 42
2000 1 982 637 793 285 267 108 375 45
2005 1 409 536 554 148 171 34 205 49
2010 913 339 400 86 0 88 21 109 0 46
2011 864 293 410 85 0 76 21 97 0 42
• 34 47 • 2012 959 342 438 100 0 79 34 113 0 44
Lithuania 1990 1 471 –
1995 2 362 979 1 049 206 128 128 48
2000 2 657 776 1 051 503 327 182 509 42
2005 2 114 964 793 357 0 460 460 55
2010 1 751 719 633 221 0 177 187 364 1 53
2011 1 748 681 664 187 0 213 156 369 3 51
• 40 54 • 2012 1 635 726 548 156 0 204 146 350 1 57
Luxembourg 1990 48 –
1995 32 –
2000 44 21 19 0 4 4 52
2005 37 14 20 3 0 0 0 41
2010 29 0 18 6 0 0 0 0 5 0
2011 25 4 4 3 0 0 1 1 14 50
• 13 9 • 2012 45 0 0 0 0 1 0 1 44
a Rates are per 100 000 population.
b NEW AND RELAPSE includes cases for which the treatment history is unknown.

237 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
EUROPEAN REGION
7$%/($&DVHQRWLILFDWLRQV±
NEW CASES
YEAR SMEAR-
POSITIVE
SMEAR-NEGATIVE/
UNKNOWN
EXTRA-
PULMONARY OTHER RELAPSE RE-TREAT EXCL.
RELAPSE
TOTAL
RETREAT
HISTORY
UNKNOWN
NEW AND
RELAPSE
b
% SMEAR-
POS AMONG
NEW PULM
NEW AND RELAPSE
NOTIFICATION RATE
a
1990–2012
Malta 1990 13 –
1995 11 5 4 2 0 0 56
2000 16 5 9 2 0 0 0 36
2005 22 5 10 6 0 1 1 1 33
2010 29 4 6 10 0 0 3 3 9 40
2011 30 7 8 7 0 0 3 3 8 47
• 3 10 • 2012 42 9 20 12 0 0 1 1 1 31
Monaco 1990 1 –
1995 1 –
2000 0 0 0 0 0 0 0
2005 –
2010 1 1 –
2011 –
• 3 0 • 2012 –
Montenegro 2005 156 64 66 13 13 14 27 49
2010 110 39 49 14 0 8 4 12 0 44
2011 110 48 40 12 0 10 2 12 0 55
16 • 2012 98 45 36 13 0 4 9 13 0 56
Netherlands 1990 1 369 –
1995 1 619 575 1 522 513 27
2000 1 244 289 528 427 0 70 70 35
2005 1 127 237 491 385 14 30 44 33
2010 1 046 176 370 463 4 16 27 43 17 32
2011 981 177 353 425 3 12 26 38 11 33
• 9 6 • 2012 920 163 300 444 0 11 38 49 2 35
Norway 1990 285 –
1995 236 62 57 89 28 28 52
2000 221 37 103 79 2 10 12 26
2005 276 48 119 102 0 14 14 7 29
2010 297 49 110 115 0 42 42 23 31
2011 324 40 134 139 1 37 37 10 23
• 7 0 • 2012 –
Poland 1990 16 136 –
1995 15 958 6 955 7 285 647 1 071 1 071 49
2000 10 931 3 180 6 392 477 882 0 882 33
2005 8 203 2 823 4 591 789 0 1 077 1 077 38
2010 7 002 2 484 3 625 501 0 392 507 899 0 41
2011 7 946 2 587 4 344 584 0 431 532 963 0 37
• 42 18 • 2012 7 054 2 433 3 729 503 0 389 488 877 0 39
Portugal 1990 6 214 –
1995 5 577 2 019 1 531 1 759 268 268 57
2000 4 227 1 863 1 005 1 178 177 304 481 65
2005 3 308 1 302 974 905 122 228 350 5 57
2010 2 487 912 791 679 16 89 139 228 0 54
2011 2 406 876 813 629 7 81 134 215 0 52
• 63 23 • 2012 2 490 920 805 670 7 88 100 188 0 53
Republic of 1990 1 728 –
Moldova 1995 2 925 665 1 958 154 148 148 25
2000 2 935 651 1 788 122 374 0 374 27
2005 5 141 1 696 2 237 568 640 1 137 1 777 43
2010 4 135 1 267 2 073 405 0 377 1 312 1 689 13 38
2011 4 233 1 272 2 140 424 0 372 1 108 1 480 25 37
• 40 125 • 2012 4 409 1 346 2 062 396 0 559 932 1 491 46 39
Romania 1990 16 256 –
1995 23 271 10 469 8 303 3 422 1 077 1 077 56
2000 27 470 10 202 10 180 3 474 3 614 156 3 770 50
2005 26 106 10 801 8 038 3 568 3 697 3 241 6 938 2 57
2010 18 379 7 951 5 113 2 899 0 2 416 2 699 5 115 0 61
2011 16 992 7 386 4 528 2 629 0 2 449 2 220 4 669 0 62
• 70 74 • 2012 16 036 7 077 4 342 2 481 0 2 136 2 188 4 324 0 62
Russian 1990 50 641 –
Federation 1995 84 980 37 512 42 241 5 227 47
2000 140 677 27 467 102 228 5 313 5 669 12 478 18 147 21
2005 127 930 32 605 74 301 12 320 8 704 26 449 35 153 30
2010 125 310 31 416 67 894 3 513 7 081 8 737 37 243 45 980 6 669 32
2011 112 910 29 191 65 106 10 023 8 590 46 569 55 159 31
• 34 74 • 2012 105 753 27 467 60 058 10 017 0 8 211 44 168 52 379 0 31
San Marino 1990 1 –
1995 2 –
2000 1 1 0 0 0 0 0 100
2005 –
2010 –
2011 –
• 4 0 • 2012 –
Serbia 2005 3 208 1 105 1 584 479 40 260 300 0 41
2010 2 333 977 700 501 0 148 52 200 7 58
2011 2 174 905 745 401 0 120 42 162 3 55
20 • 2012 1 872 819 787 130 0 134 45 179 2 51
Serbia (without Kosovo) 2005 2 146 873 714 245 55
2010 1 449 690 431 202 119 119 62
2011 1 299 654 372 155 91 91 64
2012 1 170 569 369 86 86 61
Kosovo 2005 1 062 232 596 234 28
2010 884 287 269 299 29 29 52
2011 875 251 349 246 29 29 42
2012 702 250 404 48 48 38
Serbia & 1990 4 194 –
Montenegro 1995 2 798 1 497 930 173 198 198 62
• 0 2000 2 864 0 2 486 175 203 0 203 0
Slovakia 1990 1 448 –
1995 1 540 788 555 177 20 20 59
2000 1 010 236 469 203 102 18 120 33
2005 710 162 356 134 58 50 108 31
2010 409 112 190 59 0 25 30 55 23 37
2011 378 96 170 57 0 29 21 50 26 36
• 27 6 • 2012 321 96 144 39 0 25 24 49 17 40
Slovenia 1990 722 –
1995 525 303 83 109 30 30 78
2000 368 145 133 59 31 16 47 52
2005 269 109 110 30 20 9 29 50
2010 169 64 67 30 0 8 3 11 0 49
2011 181 82 73 26 0 11 11 0 53
• 36 6 • 2012 134 47 64 13 0 10 4 14 0 42
a Rates are per 100 000 population.
b NEW AND RELAPSE includes cases for which the treatment history is unknown.

GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($&DVHQRWLILFDWLRQV±
NEW CASES
YEAR SMEAR-
POSITIVE
SMEAR-NEGATIVE/
UNKNOWN
EXTRA-
PULMONARY OTHER RELAPSE RE-TREAT EXCL.
RELAPSE
TOTAL
RETREAT
HISTORY
UNKNOWN
NEW AND
RELAPSE
b
% SMEAR-
POS AMONG
NEW PULM
NEW AND RELAPSE
NOTIFICATION RATE
a
1990–2012
Spain 1990 7 600 –
1995 8 764 2 605 6 159 30
2000 7 993 3 423 4 446 124 0 0 0 43
2005 7 281 2 511 3 880 890 0 1 078 1 078 39
2010 6 765 2 076 2 621 1 680 0 0 324 324 388 44
2011 6 392 2 186 2 242 1 616 0 0 370 370 348 49
• 20 12 • 2012 5 677 1 984 1 855 1 508 0 0 314 314 330 52
Sweden 1990 557 –
1995 564 102 235 216 11 11 30
2000 417 118 147 152 0 40 40 45
2005 539 134 208 197 0 30 30 39
2010 623 117 226 209 0 0 52 52 71 34
2011 544 99 182 173 0 3 42 45 87 35
• 7 6 • 2012 593 101 233 229 0 0 39 39 30 30
Switzerland 1990 1 278 –
1995 830 185 515 126 5 5 26
2000 577 86 216 102 63 63 173 28
2005 514 84 187 110 49 49 133 31
2010 508 82 149 91 40 40 186 35
2011 524 90 170 119 54 54 145 35
• 19 5 • 2012 416 87 124 84 47 47 121 41
Tajikistan 1990 2 460 –
1995 2 029 1 042 617 370 370 63
2000 2 779 434 1 918 427 18
2005 5 460 1 745 2 175 1 417 123 2 066 2 189 45
2010 6 994 2 290 2 038 1 631 0 338 647 985 697 53
2011 7 035 2 174 2 148 1 613 0 355 574 929 745 50
• 46 81 • 2012 6 508 2 041 1 911 1 532 0 327 421 748 697 52
The Former 1990 –
Yu
g
oslav Re
p
ubli
c
1995 786 319 376 66 25 25 46
of Macedonia 2000 641 167 308 150 16 0 16 35
2005 598 178 236 141 43 60 103 43
2010 384 141 135 92 0 16 36 52 0 51
2011 335 132 99 76 0 28 27 55 0 57
• 0 16 • 2012 346 147 95 78 0 22 9 31 4 61
Turkey 1990 24 468 –
1995 22 981 4 383 17 534 1 064 20
2000 18 038 4 315 8 544 4 371 808 808 34
2005 19 744 7 450 5 944 5 359 991 1 559 2 550 56
2010 15 879 5 375 4 191 5 617 0 696 672 1 368 0 56
2011 15 054 4 927 3 925 5 565 0 637 625 1 262 0 56
• 45 19 • 2012 14 139 4 585 3 829 5 121 0 604 552 1 156 0 54
Turkmenistan 1990 2 325 –
1995 1 939 544 1 327 1 67 67 29
2000 4 038 1 017 2 709 241 71 1 894 1 965 27
2005 3 191 995 1 498 656 42 100 142 40
2010 3 230 1 153 1 248 473 274 82 82 48
2011 –
• 63 0 • 2012 –
Ukraine 1990 16 465 –
1995 21 459 8 263 9 793 1 514 1 889 1 889 46
2000 32 945 10 738 17 258 1 739 3 210 0 3 210 38
2005 39 608 –
2010 33 857 9 976 17 599 3 355 365 2 562 2 552 5 114 36
2011 34 237 10 502 14 106 3 367 3 213 3 049 8 439 11 488 43
• 32 90 • 2012 40 990 11 030 17 398 3 344 3 650 4 579 8 229 5 568 39
United Kingdom of 1990 5 908 –
Great Britain and 1995 6 176 4 162 2 014 –
Northern Ireland 2000 6 220 1 204 2 037 2 478 0 0 0 37
2005 8 173 1 821 2 752 3 600 0 460 460 40
2010 7 907 1 201 2 551 3 443 24 576 576 688 32
2011 8 439 1 204 2 827 3 783 36 524 524 589 30
• 10 13 • 2012 8 269 1 251 2 751 3 676 53 482 482 538 31
Uzbekistan 1990 9 414 –
1995 9 866 2 735 5 798 1 333 32
2000 15 750 3 825 10 142 1 760 23 324 347 27
2005 21 513 5 695 7 857 6 324 1 637 7 378 9 015 42
2010 16 883 4 711 6 735 4 288 0 1 149 3 447 4 596 0 41
2011 15 345 4 198 5 958 3 839 0 506 568 1 074 844 41
• 46 52 • 2012 14 832 4 030 6 137 3 965 0 655 1 978 2 633 45 40
a Rates are per 100 000 population.
b NEW AND RELAPSE includes cases for which the treatment history is unknown.

239 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
EUROPEAN REGION
7$%/($7UHDWPHQWRXWFRPHVQHZVPHDUSRVLWLYHFDVHV±
% OF COHORT
TREATMENT SUCCESS (%)
a
1995–2011 YEAR NUMBER
NOTIFIED
SIZE OF
COHORT
COHORT AS
% NOTIFIED CURED COMPLETED DIED FAILED DEFAULTED NOT
EVALUATED
Albania 1995 139 –
2000 171 –
2005 196 196 100 43 35 4 2 5 11
2009 171 171 100 64 25 2 1 4 4
2010 145 145 100 49 42 3 0 3 3
• 0 93 • 2011 180 180 100 65 28 2 0 4 1
Andorra 1995 –
2000 1 2 200 50 50 0
2005 5 5 100 80 0 0 0 0 20
2009 2 3 150 33 67 0 0 0 0
2010 0 0 –
• 0 100 • 2011 1 1 100 0 100 0 0 0 0
Armenia 1995 436 507 116 52 2 8 36 1 0
2000 621 447 72 81 6 4 3 7 0
2005 581 581 100 59 13 3 5 14 4
2009 440 440 100 60 12 7 3 8 10
2010 339 339 100 55 16 4 15 8 1
• 55 63 • 2011 329 329 100 44 19 6 25 6 0
Austria 1995 467 383 82 2 81 10 0 7 1
2000 324 298 92 0 73 9 0 6 11
2005 234 230 98 17 58 7 0 7 11
2009 90 226 251 8 59 9 0 8 16
2010 76 206 271 6 59 6 0 6 23
• 82 71 • 2011 94 221 235 7 64 6 0 7 15
Azerbaijan 1995 669 538 80 58 7 12 19 4
2000 890 890 100 89 0 1 2 3 4
2005 1 561 1 561 100 48 11 4 4 12 22
2009 1 487 1 480 100 47 15 3 7 16 12
2010 1 997 1 919 96 47 30 3 4 10 6
• 65 78 • 2011 1 426 2 208 155 33 44 3 6 10 4
Belarus 1995 1 845 –
2000 2 547 –
2005 1 235 –
2009 1 201 2 160 180 64 0 10 4 1 20
2010 1 269 2 184 172 66 0 8 22 1 2
• 0 60 • 2011 1 217 2 169 178 59 1 6 31 1 1
Belgium 1995 400 –
2000 409 358 88 25 41 10 1 17 6
2005 380 304 80 21 45 10 0 0 24
2009 280 485 173 14 62 8 0 11 4
2010 244 473 194 15 61 7 0 11 7
• 0 77 • 2011 240 405 169 28 50 7 0 10 6
Bosnia and 1995 865 865 100 97 1 0 1 1 1
Herzegovina 2000 759 756 100 77 18 1 1 2 1
2005 640 1 035 162 93 3 1 0 0 2
2009 609 852 140 97 2 0 0 0 0
2010 441 441 100 91 7 1 0 0 0
• 97 70 • 2011 547 693 127 43 27 5 1 1 24
Bulgaria 1995 1 087 –
2000 2 524 –
2005 1 214 1 342 111 82 3 4 2 7 1
2009 894 1 055 118 78 7 9 2 4 1
2010 806 946 117 84 2 8 2 3 1
• 0 86 • 2011 716 853 119 84 2 8 1 3 2
Croatia 1995 1 204 –
2000 –
2005 372 391 105 40 7 7 0 1 45
2009 302 234 77 48 15 26 0 3 7
2010 183 181 99 58 17 14 0 4 7
• 0 0 • 2011 201 –
Cyprus 1995 6 6 100 100 0 0 0 0 0
2000 4 –
2005 9 8 89 38 25 12 0 0 25
2009 14 28 200 29 0 0 0 0 71
2010 8 20 250 25 0 0 0 0 75
• 100 64 • 2011 11 22 200 55 9 14 0 0 23
Czech Republic 1995 487 487 100 57 3 0 3 2 35
2000 420 396 94 59 11 17 1 1 11
2005 308 315 102 62 10 6 0 2 20
2009 218 402 184 66 2 21 0 7 4
2010 200 361 180 66 3 17 0 7 7
• 60 69 • 2011 188 377 201 66 3 17 0 9 5
Denmark 1995 128 –
2000 171 110 64 37 49 5 0 0 9
2005 129 128 99 44 39 6 1 2 8
2009 101 175 173 22 31 4 1 1 42
2010 115 217 189 31 33 11 2 0 22
• 0 0 • 2011 124 –
Estonia 1995 369 –
2000 255 257 101 67 2 11 1 6 12
2005 162 162 100 70 2 8 1 10 10
2009 135 240 178 57 1 15 2 6 18
2010 99 191 193 65 3 10 2 4 17
• 0 59 • 2011 123 202 164 57 2 11 1 5 23
Finland 1995 244 –
2000 205 –
2005 130 –
2009 93 227 244 33 34 17 0 1 14
2010 82 184 224 48 27 9 0 2 15
• 0 67 • 2011 82 181 221 39 29 18 1 0 14
France 1995 3 449 –
2000 1 815 –
2005 1 941 –
2009 1 019 –
2010 960 –
• 0 0 • 2011 906 –
Georgia 1995 221 221 100 41 18 8 3 29 2
2000 601 807 134 38 25 3 9 25 0
2005 1 509 1 489 99 60 13 3 5 13 7
2009 2 055 2 352 114 57 19 3 12 7 3
2010 2 140 2 500 117 59 17 3 12 7 2
• 58 76 • 2011 2 026 2 513 124 57 19 2 15 5 2
Germany 1995 3 852 –
2000 454 – 61 16 16 1 2 4
2005 1 379 1 199 87 39 32 9 0 2 18
2009 1 025 2 220 217 33 44 12 0 1 9
2010 910 2 064 227 32 44 12 0 2 9
• 0 70 • 2011 951 2 113 222 29 42 11 0 2 17
a
TREATMENT SUCCESS = percent cured + percent completed then rounded to the nearest digit.

240 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($7UHDWPHQWRXWFRPHVQHZVPHDUSRVLWLYHFDVHV±
% OF COHORT
TREATMENT SUCCESS (%)
a
1995–2011 YEAR NUMBER
NOTIFIED
SIZE OF
COHORT
COHORT AS
% NOTIFIED CURED COMPLETED DIED FAILED DEFAULTED NOT
EVALUATED
Greece 1995 –
2000 235 –
2005 197 –
2009 198 –
2010 178 –
• 0 0 • 2011 236 –
Greenland 1995 –
2000 –
2005 –
2009 24 –
2010 38 –
• 0 0 • 2011 34 –
Hungary 1995 796 –
2000 412 651 158 28 36 10 3 12 11
2005 423 412 97 32 13 13 12 9 20
2009 363 597 164 45 12 10 19 7 7
2010 270 515 191 64 5 12 0 10 9
• 0 0 • 2011 260 0 0
Iceland 1995 2 2 100 0 100 0 0 0 0
2000 1 2 200 0 100 0 0 0 0
2005 2 2 100 0 100 0 0 0 0
2009 3 4 133 0 75 0 0 0 25
2010 6 16 267 0 88 6 0 0 6
• 100 0 • 2011 1 0 0
Ireland 1995 –
2000 138 73 53 33 51 12 0 4 0
2005 130 107 82 3 62 9 3 1 22
2009 95 188 198 5 62 9 0 1 23
2010 84 164 195 0 73 7 0 1 19
• 0 64 • 2011 85 153 180 54 10 5 0 3 29
Israel 1995 –
2000 216 336 156 65 18 15 0 0 1
2005 142 227 160 69 15 11 0 3 2
2009 119 202 170 72 14 10 0 1 3
2010 103 99 96 69 7 11 0 0 13
• 0 77 • 2011 135 242 179 69 9 10 0 2 10
Italy 1995 1 413 295 21 73 6 3 2 11 4
2000 687 223 32 37 36 1 0 9 16
2005 1 275 –
2009 885 –
2010 586 –
• 80 0 • 2011 587 –
Kazakhstan 1995 3 022 –
2000 8 903 8 781 99 76 3 5 10 3 3
2005 6 911 6 884 100 70 1 5 12 5 8
2009 5 213 5 355 103 62 0 4 30 3 2
2010 4 769 4 919 103 61 0 3 7 2 27
• 0 61 • 2011 4 157 4 306 104 61 0 4 6 2 27
Kyrgyzstan 1995 832 –
2000 1 296 1 233 95 73 9 3 4 5 6
2005 1 972 1 897 96 81 4 3 5 5 2
2009 1 609 1 543 96 79 4 3 4 6 4
2010 1 645 –
• 0 78 • 2011 1 537 1 537 100 75 3 3 11 5 3
Latvia 1995 504 475 94 61 0 9 3 21 7
2000 637 637 100 68 4 12 3 7 7
2005 536 536 100 72 1 11 1 7 8
2009 367 592 161 72 3 9 1 5 11
2010 339 596 176 72 3 8 1 6 10
• 61 73 • 2011 293 559 191 72 1 10 0 5 11
Lithuania 1995 979 –
2000 776 776 100 73 10 4 12 2
2005 964 958 99 70 0 11 3 11 6
2009 742 1 033 139 73 0 10 2 9 6
2010 719 959 133 68 0 11 1 11 8
• 0 73 • 2011 681 1 000 147 73 0 11 1 8 7
Luxembourg 1995 37 – 100 0 0 0 0 0
2000 21 –
2005 14 0 0
2009 –
2010 0 14 – 0 0 7 0 0 93
• 100 0 • 2011 4 6 150 0 0 17 0 0 83
Malta 1995 5 5 100 80 20 0 0 0 0
2000 5 4 80 0 100 0 0 0 0
2005 5 5 100 0 100 0 0 0 0
2009 12 10 83 0 80 0 0 0 20
2010 4 5 125 0 80 0 0 0 20
• 100 58 • 2011 7 12 171 0 58 0 0 8 33
Monaco 1995 –
2000 0 –
2005 –
2009 –
2010 –
• 0 0 • 2011 –
Montenegro 2005 64 63 98 10 21 70
2009 53 78 147 49 37 8 0 4 3
2010 39 39 100 46 41 5 0 3 5
86 • 2011 48 56 117 25 61 12 0 0 2
Netherlands 1995 575 715 124 17 55 8 5 15
2000 289 301 104 23 53 6 0 3 15
2005 237 208 88 9 75 7 0 1 8
2009 203 454 224 11 69 9 0 3 8
2010 176 469 266 1 76 7 0 4 12
• 72 81 • 2011 177 437 247 1 81 5 0 3 11
Norway 1995 62 87 140 43 34 14 1 8 0
2000 37 37 100 49 22 14 3 3 11
2005 48 47 98 62 30 2 0 4 2
2009 42 146 348 45 37 4 1 0 13
2010 49 139 284 68 24 3 1 0 5
• 77 0 • 2011 40 –
Poland 1995 6 955 –
2000 3 180 214 7 50 22 11 6 6 5
2005 2 823 2 823 100 65 12 5 1 9 8
2009 2 658 4 391 165 48 19 5 0 10 17
2010 2 484 3 998 161 47 19 6 0 9 19
• 0 60 • 2011 2 587 4 699 182 43 17 9 0 9 22
a
TREATMENT SUCCESS = percent cured + percent completed then rounded to the nearest digit.

241 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
EUROPEAN REGION
7$%/($7UHDWPHQWRXWFRPHVQHZVPHDUSRVLWLYHFDVHV±
% OF COHORT
TREATMENT SUCCESS (%)
a
1995–2011 YEAR NUMBER
NOTIFIED
SIZE OF
COHORT
COHORT AS
% NOTIFIED CURED COMPLETED DIED FAILED DEFAULTED NOT
EVALUATED
Portugal 1995 2 019 1 240 61 45 23 4 4 4 19
2000 1 863 1 924 103 9 71 6 0 5 9
2005 1 302 1 393 107 13 76 6 0 4 2
2009 1 043 1 565 150 9 75 6 0 3 7
2010 912 –
• 69 80 • 2011 876 1 387 158 9 72 5 0 3 12
Republic of 1995 665 –
Moldova 2000 651 651 100 1 62 0 0 0 37
2005 1 696 1 690 100 60 2 9 11 11 7
2009 1 318 1 318 100 49 5 10 17 14 5
2010 1 267 1 267 100 52 5 11 5 13 13
• 0 62 • 2011 1 272 1 272 100 57 5 9 18 10 1
Romania 1995 10 469 11 597 111 38 13 6 7 6 31
2000 10 202 10 158 100 28 42 4 8 8 9
2005 10 801 10 929 101 71 11 5 4 6 4
2009 8 987 10 737 119 72 14 4 4 6 1
2010 7 951 9 445 119 70 14 5 4 6 2
• 51 85 • 2011 7 386 8 886 120 71 14 5 3 5 1
Russian 1995 37 512 54 0 54 11 15 6 11 4
Federation 2000 27 467 3 616 13 64 4 6 13 9 4
2005 32 605 25 692 79 55 3 13 14 11 4
2009 33 351 32 316 97 52 3 11 20 8 5
2010 31 416 30 123 96 50 3 12 23 7 5
• 65 54 • 2011 29 191 36 747 126 48 5 9 10 7 20
San Marino 1995 –
2000 1 1 100 0 0 100 0 0 0
2005 –
2009 –
2010 –
• 0 0 • 2011 –
Serbia 2005 1 105 1 154 104 72 13 5 1 5 4
2009 1 055 1 392 132 80 6 6 1 4 2
2010 977 988 101 79 8 6 1 4 2
87 • 2011 905 894 99 80 7 7 0 4 2
Serbia & 1995 1 497 1 956 131 34 18 2 3 10 33
Montenegro 2000 0 267 – 82 7 4 0 6 1
Slovakia 1995 788 807 102 64 16 4 16
2000 236 238 101 81 0 14 1 2 1
2005 162 158 98 66 26 6 0 1 1
2009 121 174 144 82 0 14 0 2 2
2010 112 177 158 84 0 12 0 3 1
• 64 91 • 2011 96 138 144 91 1 7 0 0 2
Slovenia 1995 303 270 89 64 26 4 2 1 3
2000 145 145 100 33 51 8 0 5 3
2005 109 109 100 47 38 12 0 1 3
2009 85 149 175 24 63 9 1 1 3
2010 64 123 192 28 57 11 0 1 3
• 90 81 • 2011 82 151 184 37 44 18 0 1 1
Spain 1995 2 605 –
2000 3 423 –
2005 2 511 –
2009 2 236 –
2010 2 076 3 574 172 39 32 6 0 1 23
• 0 73 • 2011 2 186 3 335 153 42 31 7 0 1 19
Sweden 1995 102 –
2000 118 112 95 0 79 11 0 2 8
2005 134 133 99 0 74 6 1 1 18
2009 107 255 238 0 85 6 0 1 8
2010 117 289 247 70 15 5 0 1 9
• 0 83 • 2011 99 247 249 51 32 5 1 2 9
Switzerland 1995 185 –
2000 86 –
2005 84 –
2009 74 –
2010 82 –
• 0 0 • 2011 90 –
Tajikistan 1995 1 042 348 33 69 18 7 3 2 0
2000 434 665 153 74 3 15 8 0 0
2005 1 745 1 729 99 74 9 4 6 7 0
2009 1 972 1 972 100 75 6 4 8 5 1
2010 2 290 2 290 100 76 4 5 11 3 1
• 88 80 • 2011 2 174 2 174 100 74 6 5 11 3 1
The Former 1995 319 222 70 61 9 13 9 9 0
Yugoslav Republic 2000 167 152 91 51 35 4 2 7 1
of Macedonia 2005 178 179 101 62 22 2 0 14 0
2009 198 199 101 85 5 4 2 5 0
2010 141 143 101 83 7 4 3 2 1
• 70 95 • 2011 132 130 98 78 16 3 0 2 0
Turkey 1995 4 383 –
2000 4 315 3 461 80 0 73 3 0 6 19
2005 7 450 7 450 100 45 44 2 0 5 3
2009 6 007 6 007 100 61 30 3 1 2 3
2010 5 375 5 375 100 63 29 3 1 3 2
• 0 90 • 2011 4 927 4 927 100 60 31 3 1 3 3
Turkmenistan 1995 544 544 100 55 18 11 7 2 7
2000 1 017 1 017 100 79 2 9 6 3 1
2005 995 995 100 70 14 6 4 5 1
2009 1 370 1 375 100 83 1 5 6 5 1
2010 1 153 –
• 73 0 • 2011 –
Ukraine 1995 8 263 9 564 116 83 6 7 4
2000 10 738 –
2005 –
2009 13 632 13 111 96 52 7 13 16 8 3
2010 9 976 13 279 133 51 9 13 17 8 3
• 83 58 • 2011 10 502 13 714 131 48 10 13 18 7 4
United Kingdom of 1995 –
Great Britain and 2000 1 204 –
Northern Ireland 2005 1 821 1 348 74 0 68 7 0 1 24
2009 1 256 2 569 205 0 82 6 0 5 7
2010 1 201 2 602 217 0 81 5 0 6 8
• 0 80 • 2011 1 204 2 952 245 0 80 6 0 6 8
Uzbekistan 1995 2 735 2 598 95 78 0 9 7 4 3
2000 3 825 1 030 27 27 53 3 6 5 6
2005 5 695 5 336 94 72 9 6 6 7 1
2009 4 959 4 959 100 77 5 6 5 5 3
2010 4 711 4 711 100 76 5 6 6 5 3
• 78 78 • 2011 4 198 4 198 100 73 5 6 6 6 4
a
TREATMENT SUCCESS = percent cured + percent completed then rounded to the nearest digit.

242 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($7UHDWPHQWRXWFRPHVUHWUHDWPHQWFDVHV±
% OF COHORT
TREATMENT SUCCESS (%)
a
1995–2011 YEAR NUMBER
NOTIFIED
SIZE OF
COHORT
COHORT AS
% NOTIFIED CURED COMPLETED DIED FAILED DEFAULTED NOT
EVALUATED
Albania 1995 53 –
2000 19 –
2005 43 30 70 37 37 3 0 10 13
2009 21 21 100 38 38 10 0 10 5
2010 30 30 100 43 47 3 0 7 0
• 0 83 • 2011 18 18 100 28 56 6 0 6 6
Andorra 1995 –
2000 0 –
2005 0 –
2009 2 2 100 0 100 0 0 0 0
2010 0 0 –
• 0 0 • 2011 1 1 100 0 0 0 0 0 100
Armenia 1995 38 6 16 50 0 0 17 33 0
2000 76 54 71 52 15 7 7 19 0
2005 327 327 100 13 28 7 12 37 4
2009 542 542 100 9 54 8 4 15 10
2010 451 451 100 5 62 6 10 13 4
• 50 68 • 2011 382 382 100 5 63 4 9 15 3
Austria 1995 –
2000 30 10 33 0 80 0 0 0 20
2005 26 27 104 11 56 11 0 11 11
2009 25 37 148 3 38 5 0 30 24
2010 29 29 100 14 45 0 0 0 41
• 0 43 • 2011 20 21 105 0 43 14 5 5 33
Azerbaijan 1995 47 –
2000 74 74 100 59 7 5 11 14 4
2005 3 200 1 314 41 28 9 6 6 13 38
2009 2 384 1 687 71 39 14 6 9 19 13
2010 1 997 4 194 210 14 49 3 4 15 15
• 0 71 • 2011 2 155 4 005 186 8 63 3 5 12 8
Belarus 1995 343 –
2000 825 –
2005 1 049 –
2009 878 616 70 38 4 13 7 1 37
2010 1 114 792 71 20 28 10 36 1 5
• 0 29 • 2011 1 075 1 020 95 21 8 7 59 3 3
Belgium 1995 80 –
2000 89 55 62 16 45 13 0 15 11
2005 68 47 69 17 21 19 0 0 43
2009 76 – 11 57 9 0 12 12
2010 87 85 98 8 55 6 0 12 19
• 0 61 • 2011 59 56 95 16 45 9 0 16 14
Bosnia and 1995 130 –
Herzegovina 2000 193 122 63 79 15 3 1 2 0
2005 156 106 68 85 8 4 1 2 1
2009 113 116 103 52 32 5 3 3 5
2010 101 101 100 83 12 2 1 1 1
• 0 63 • 2011 65 104 160 19 43 7 0 3 28
Bulgaria 1995 –
2000 383 –
2005 201 198 99 57 10 7 11 14 2
2009 372 384 103 32 38 12 5 8 5
2010 348 348 100 32 31 13 6 12 5
• 0 66 • 2011 355 355 100 30 36 9 5 11 8
Croatia 1995 42 –
2000 –
2005 94 92 98 20 13 9 1 1 57
2009 62 22 35 27 23 36 5 5 5
2010 43 37 86 59 16 14 3 8
• 0 0 • 2011 –
Cyprus 1995 0 –
2000 0 –
2005 3 2 67 0 100 0 0 0 0
2009 3 6 200 17 0 0 0 0 83
2010 0 0 –
• 0 100 • 2011 3 3 100 67 33 0 0 0 0
Czech Republic 1995 21 –
2000 25 38 152 53 11 8 3 0 26
2005 34 31 91 16 39 3 0 3 39
2009 62 – 34 34 18 0 2 13
2010 51 49 96 41 33 16 0 0 10
• 0 75 • 2011 31 32 103 44 31 12 0 9 3
Denmark 1995 6 –
2000 28 15 54 27 60 7 0 0 7
2005 29 22 76 27 64 5 0 5 0
2009 10 42 420 12 40 2 2 0 43
2010 46 35 76 20 40 11 3 0 26
• 0 0 • 2011 22 –
Estonia 1995 71 –
2000 116 59 51 54 2 3 0 3 37
2005 94 89 95 21 20 3 4 26 25
2009 80 82 102 34 17 15 6 9 20
2010 79 81 103 28 11 11 2 15 32
• 0 31 • 2011 76 75 99 15 16 21 1 11 36
Finland 1995 –
2000 29 –
2005 22 –
2009 14 – 29 7 0 0 0 64
2010 15 13 87 38 8 0 0 0 54
• 0 25 • 2011 13 12 92 25 0 8 0 0 67
France 1995 –
2000 0 –
2005 371 –
2009 –
2010 315 –
• 0 0 • 2011 261 –
Georgia 1995 196 298 152 8 24 12 9 45 2
2000 681 470 69 23 31 10 8 29 0
2005 2 152 2 037 95 19 35 7 10 23 6
2009 566 1 521 269 26 34 5 17 15 3
2010 1 409 1 421 101 26 35 5 17 11 4
• 32 61 • 2011 1 310 1 321 101 27 34 4 23 8 4
Germany 1995 –
2000 63 – 51 21 16 3 5 5
2005 493 432 88 30 36 9 0 7 18
2009 252 344 137 21 44 12 0 5 17
2010 367 364 99 25 47 12 1 6 10
• 0 58 • 2011 300 289 96 17 41 10 0 6 27
a
TREATMENT SUCCESS = percent cured + percent completed then rounded to the nearest digit.

243 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
EUROPEAN REGION
7$%/($7UHDWPHQWRXWFRPHVUHWUHDWPHQWFDVHV±
% OF COHORT
TREATMENT SUCCESS (%)
a
1995–2011 YEAR NUMBER
NOTIFIED
SIZE OF
COHORT
COHORT AS
% NOTIFIED CURED COMPLETED DIED FAILED DEFAULTED NOT
EVALUATED
Greece 1995 –
2000 48 –
2005 74 –
2009 3 –
2010 44 –
• 0 0 • 2011 35 –
Greenland 1995 –
2000 –
2005 –
2009 6 –
2010 12 –
• 0 0 • 2011 3 –
Hungary 1995 –
2000 371 122 33 16 20 15 9 11 30
2005 347 333 96 12 37 13 8 11 18
2009 211 208 99 35 26 13 12 6 8
2010 254 254 100 13 49 11 0 17 9
• 0 0 • 2011 221 0 0
Iceland 1995 0 –
2000 1 1 100 0 100 0 0 0 0
2005 1 –
2009 1 1 100 0 100 0 0 0 0
2010 0 0 –
• 0 100 • 2011 1 1 100 0 100 0 0 0 0
Ireland 1995 –
2000 22 10 45 40 0 10 10 40 0
2005 40 14 35 7 57 7 0 0 29
2009 16 52 325 4 58 8 0 0 31
2010 31 33 106 0 55 15 0 3 27
• 0 54 • 2011 27 26 96 50 4 15 0 0 31
Israel 1995 –
2000 8 8 100 12 25 62 0 0 0
2005 7 7 100 71 14 14 0 0 0
2009 9 9 100 56 11 11 0 0 22
2010 4 5 125 80 0 20 0 0 0
• 0 50 • 2011 10 10 100 40 10 10 0 20 20
Italy 1995 31 – 42 6 26 10 13 3
2000 625 26 4 31 15 4 12 8 31
2005 293 –
2009 –
2010 74 –
• 48 0 • 2011 100 –
Kazakhstan 1995 1 320 –
2000 5 093 2 901 57 62 4 10 14 5 5
2005 15 009 4 085 27 46 1 13 14 6 19
2009 9 371 9 392 100 22 27 9 34 6 3
2010 9 213 8 734 95 23 24 9 4 5 35
• 0 36 • 2011 5 969 5 026 84 36 0 11 4 5 44
Kyrgyzstan 1995 127 –
2000 555 278 50 59 15 8 8 6 4
2005 847 845 100 40 31 8 9 11 1
2009 758 924 122 28 43 7 6 7 9
2010 987 –
• 0 56 • 2011 1 035 523 51 49 6 9 22 8 5
Latvia 1995 118 –
2000 375 205 55 39 2 19 3 8 29
2005 205 205 100 50 1 10 1 9 29
2009 147 148 101 43 1 14 0 14 28
2010 109 110 101 60 2 6 0 12 20
• 0 51 • 2011 97 97 100 45 5 10 1 12 26
Lithuania 1995 128 –
2000 509 282 55 45 0 21 8 22 5
2005 460 455 99 27 2 25 4 22 19
2009 404 404 100 30 0 24 5 22 20
2010 364 364 100 31 1 18 4 22 25
• 0 33 • 2011 369 369 100 33 0 16 2 23 25
Luxembourg 1995 –
2000 4 –
2005 0 –
2009 –
2010 0 0 –
• 0 0 • 2011 1 0 0
Malta 1995 0 –
2000 0 1 – 0 100 0 0 0 0
2005 1 1 100 0 100 0 0 0 0
2009 2 2 100 0 50 0 0 0 50
2010 3 3 100 0 67 0 0 0 33
• 0 100 • 2011 3 3 100 0 100 0 0 0 0
Monaco 1995 –
2000 0 –
2005 –
2009 –
2010 –
• 0 0 • 2011 –
Montenegro 2005 27 10 37 20 20 60
2009 11 11 100 45 27 9 0 0 18
2010 12 14 117 50 36 0 0 0 14
83 • 2011 12 12 100 67 17 8 0 0 8
Netherlands 1995 –
2000 70 18 26 28 22 6 0 6 39
2005 44 28 64 11 68 4 0 7 11
2009 46 49 107 4 67 2 0 4 22
2010 43 44 102 5 61 9 0 7 18
• 0 80 • 2011 38 46 121 0 80 0 0 2 17
Norway 1995 28 –
2000 12 3 25 33 0 67 0 0 0
2005 14 9 64 44 33 22 0 0 0
2009 30 – 33 47 13 0 0 7
2010 42 40 95 20 52 15 5 0 8
• 0 0 • 2011 37 –
Poland 1995 1 071 –
2000 882 56 6 64 12 14 0 4 5
2005 1 077 985 91 22 31 6 0 32 9
2009 688 942 137 30 32 5 0 14 18
2010 899 899 100 28 33 8 0 10 21
• 0 53 • 2011 963 963 100 25 28 10 0 12 24
a
TREATMENT SUCCESS = percent cured + percent completed then rounded to the nearest digit.

244 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($7UHDWPHQWRXWFRPHVUHWUHDWPHQWFDVHV±
% OF COHORT
TREATMENT SUCCESS (%)
a
1995–2011 YEAR NUMBER
NOTIFIED
SIZE OF
COHORT
COHORT AS
% NOTIFIED CURED COMPLETED DIED FAILED DEFAULTED NOT
EVALUATED
Portugal 1995 268 133 50 38 17 6 6 9 24
2000 481 209 43 10 66 4 0 7 14
2005 350 293 84 8 66 10 1 9 6
2009 271 265 98 7 62 7 0 8 16
2010 228 –
• 55 61 • 2011 215 204 95 3 58 4 0 2 32
Republic of 1995 148 –
Moldova 2000 374 1 0 0 0 0 100 0 0
2005 1 777 1 713 96 22 19 13 16 17 13
2009 1 663 1 663 100 15 20 15 26 20 4
2010 1 689 1 702 101 15 17 14 5 17 32
• 0 38 • 2011 1 480 1 500 101 18 20 13 28 17 4
Romania 1995 1 077 –
2000 3 770 2 605 69 24 20 9 20 17 11
2005 6 938 6 737 97 39 13 10 10 14 14
2009 5 401 5 391 100 38 19 10 12 16 4
2010 5 115 5 118 100 37 18 11 12 17 6
• 0 58 • 2011 4 669 4 667 100 39 19 11 11 15 5
Russian 1995 12 – 42 17 25 8 8 0
Federation 2000 18 147 1 694 9 25 24 10 21 9 11
2005 35 153 10 855 31 33 4 16 26 16 5
2009 32 569 16 726 51 31 3 13 32 12 9
2010 45 980 14 609 32 31 4 12 33 12 9
• 58 42 • 2011 55 159 26 062 47 20 22 10 15 10 23
San Marino 1995 –
2000 0 –
2005 –
2009 –
2010 –
• 0 0 • 2011 –
Serbia 2005 300 284 95 46 26 10 2 12 3
2009 203 244 120 61 13 9 0 12 5
2010 200 203 101 55 21 9 1 10 3
78 • 2011 162 164 101 60 18 5 1 8 9
Serbia & 1995 198 –
Montenegro 2000 203 21 10 67 10 10 0 14 0
Slovakia 1995 20 –
2000 120 46 38 78 0 11 2 4 4
2005 108 101 94 50 38 7 0 3 3
2009 79 79 100 34 48 14 1 0 3
2010 55 55 100 44 40 15 0 0 2
• 0 88 • 2011 50 50 100 48 40 2 4 2 4
Slovenia 1995 30 –
2000 47 24 51 29 46 4 0 12 8
2005 29 27 93 44 41 4 0 4 7
2009 8 8 100 12 75 0 0 0 12
2010 11 11 100 18 45 36 0 0 0
• 0 100 • 2011 11 11 100 27 73 0 0 0 0
Spain 1995 –
2000 0 –
2005 1 078 –
2009 –
2010 324 351 108 25 31 9 0 2 33
• 0 56 • 2011 370 388 105 26 30 13 0 2 28
Sweden 1995 11 –
2000 40 9 22 0 78 0 0 11 11
2005 30 16 53 0 75 0 0 0 25
2009 45 – 0 69 13 0 7 11
2010 52 52 100 21 54 2 0 0 23
• 0 78 • 2011 45 45 100 22 56 2 0 4 16
Switzerland 1995 5 –
2000 63 –
2005 49 –
2009 51 –
2010 40 –
• 0 0 • 2011 54 –
Tajikistan 1995 370 –
2000 –
2005 2 189 1 762 80 29 47 9 8 6 1
2009 533 1 618 304 29 43 11 10 6 1
2010 985 1 732 176 33 38 11 11 4 1
• 0 71 • 2011 929 1 674 180 29 41 10 13 5 1
The Former 1995 25 –
Yugoslav Republic 2000 16 –
of Macedonia 2005 103 97 94 24 33 7 2 32 2
2009 56 56 100 39 39 7 2 11 2
2010 52 52 100 29 37 17 4 12 2
• 0 78 • 2011 55 55 100 38 40 9 4 7 2
Turkey 1995 –
2000 808 –
2005 2 550 1 593 62 24 46 5 2 12 11
2009 1 445 1 459 101 29 44 3 2 9 13
2010 1 368 1 368 100 25 43 5 2 7 17
• 0 68 • 2011 1 262 1 262 100 22 46 4 2 10 16
Turkmenistan 1995 67 –
2000 1 965 495 25 66 9 7 11 6 1
2005 142 142 100 42 26 13 10 9 0
2009 –
2010 82 –
• 0 0 • 2011 –
Ukraine 1995 1 889 –
2000 3 210 –
2005 –
2009 5 477 10 424 190 18 29 14 22 12 5
2010 5 114 9 812 192 17 29 14 23 10 7
• 0 34 • 2011 11 488 6 413 56 26 8 16 33 9 7
United Kingdom of 1995 –
Great Britain and 2000 0 –
Northern Ireland 2005 460 147 32 0 57 4 0 3 36
2009 791 – 0 79 7 0 5 9
2010 576 576 100 0 74 7 0 7 12
• 0 80 • 2011 524 492 94 0 80 6 0 6 8
Uzbekistan 1995 –
2000 347 764 220 20 55 8 8 9 0
2005 9 015 3 999 44 28 41 9 7 14 1
2009 2 451 2 451 100 30 39 11 7 9 5
2010 4 596 4 527 98 25 48 10 5 9 4
• 0 72 • 2011 1 074 1 074 100 40 32 9 10 8 1
a
TREATMENT SUCCESS = percent cured + percent completed then rounded to the nearest digit.

245 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
EUROPEAN REGION
7$%/($+,9WHVWLQJDQGSURYLVLRQRI&37$57DQG,37–
% OF TB PATIENTS WITH
KNOWN HIV STATUS
2005–2012
YEAR
% OF TB
PATIENTS WITH
KNOWN HIV
STATUS
NUMBER OF TB
PATIENTS WITH
KNOWN HIV
STATUS
PATIENTS
NOTIFIED
(NEW AND
RETREAT)
NUMBER OF
HIV-POSITIVE
TB PATIENTS
% OF TESTED
TB PATIENTS
HIV-POSITIVE
% OF HIV-
POSITIVE TB
PATIENTS ON
CPT
% OF HIV-
POSITIVE TB
PATIENTS ON
ART
NUMBER OF
HIV-POSITIVE
PEOPLE
PROVIDED IPT
Albania 2005 15 81 540 1 1.2
2010 42 186 445 0 0 5
2011 39 170 431 2 1.2 100 100
• 15 55 • 2012 55 233 420 7 3 100 100 2
Andorra 2005 10
2010 0 0 7 0
2011 0 0 4 0
–
11 • 2012 11 1 9 0 0
Armenia 2005 12 270 2 322 6 2.2 83 33
2010 70 1 242 1 780 17 1.4 47 41
2011 95 1 499 1 582 49 3.3 80 80
• 12 100 • 2012 100 1 518 1 518 79 5.2 70 70 0
Austria 2005 954
2010 688
2011 687
–
–
2012 648
Azerbai
j
an 2005 7 920
2010 75 6 290 8 394 48 0.76 62
2011 74 7 448 10 100 36 0.48 61 41
–
96 • 2012 96 7 849 8 140 129 1.6 49 21
Belarus 2005 6 357 139
2010 93 5 153 5 554 190 3.7 257
2011 100 5 118 5 118 217 4.2 29 32
–
100 • 2012 100 5 246 5 246 229 4.4 67 258
Bel
g
ium 2005 82 937 1 144 52 5.5
2010 87 969 1 115 66 6.8
2011 81 845 1 044 44 5.2
• 82 56 • 2012 56 556 987 43 7.7
Bosnia and 2005 2 160
Herze
g
ovina 2010 0 0 1 390 0
2011 4.7 65 1 385 0 0 0
–
4 • 2012 3.9 56 1 420 0 0
Bul
g
aria 2005 0.7 23 3 302
2010 67 1 773 2 649 2 0.11 0 100
2011 71 1 698 2 407 5 0.29 0 100
• 1 66 • 2012 66 1 513 2 280 3 0.2 0 100
Croatia 2005 1 144
2010 695 1 1
2011 619 4 3
–
–
2012
C
yp
rus 2005 0 0 37 0
2010 61
2011 54 1
• 0
–
2012 69
Czech Re
p
ublic 2005 19 189 1 007 2 1.1
2010 26 177 678 5 2.8
2011 26 153 600 4 2.6
• 19 22 • 2012 22 136 605 6 4.4
Denmar
k
2005 424 8
2010 0 0 359 0
2011 73 277 381 10 3.6
–
–
2012
Estonia 2005 94 490 519 33 6.7 0
2010 91 298 329 34 11 47
2011 92 315 341 46 15 61
• 94 93 • 2012 93 271 290 45 17 62
Finland 2005 0.83 3 361 3 100
2010 0.92 3 327 3 100
2011 0.92 3 325 3 100
• 1
–
2012 274
France 2005 5 374
2010 24 1 233 5 116 121 9.8
2011 27 1 354 4 942 95 7
–
–
2012
Geor
g
ia 2005 10 674 6 448 13 1.9 54 100
2010 32 1 841 5 796 35 1.9 63 77
2011 46 2 550 5 533 50 2 56 76 61
• 10 38 • 2012 38 1 881 4 974 33 1.8 79 79 97
German
y
2005 6 045
2010 4 330
2011 4 316
–
–
2012 4 238
Greece 2005 767
2010 489
2011 489
–
–
2012
Greenland 2005
2010 116
2011 115
–
–
2012 84
Hun
g
ar
y
2005 2 024
2010 <0.1 1 1 741 1 100 100
2011 <0.1 1 1 445 1 100 100
–
–
2012 1 223
Iceland 2005 91 10 11 1 10 100 100
2010 95 21 22 1 4.8 0 0
2011 100 9 9 0 0
• 91 100 • 2012 100 11 11 0 0
Ireland 2005 6.1 28 461 11 39
2010 23 98 427 15 15
2011 30 128 425 21 16
• 6 27 • 2012 27 97 366 14 14
Israel 2005 85 316 372 17 5.4
2010 90 308 343 13 4.2
2011 92 384 418 24 6.2
• 85 99 • 2012 99 503 509 16 3.2
Ital
y
2005 4 137
2010 3 249
2011 3 521
–
–
2012
Kazakhstan 2005 77 31 187 40 429 183 0.59 41 7.7
2010 84 23 854 28 550 333 1.4 26 7.5 1 063
2011 85 22 480 26 304 352 1.6 20 9.1 1 329
• 77 98 • 2012 98 21 184 21 523 441 2.1 16 58 862
K
y
r
gy
zstan 2005 6 765
2010 2.9 183 6 295 183 100 68 37
2011 100 6 666 6 666 153 2.3 60 86 4
–
100 • 2012 100 6 916 6 916 151 2.2 67 78 5
246 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($+,9WHVWLQJDQGSURYLVLRQRI&37$57DQG,37–
% OF TB PATIENTS WITH
KNOWN HIV STATUS
2005–2012
YEAR
% OF TB
PATIENTS WITH
KNOWN HIV
STATUS
NUMBER OF TB
PATIENTS WITH
KNOWN HIV
STATUS
PATIENTS
NOTIFIED
(NEW AND
RETREAT)
NUMBER OF
HIV-POSITIVE
TB PATIENTS
% OF TESTED
TB PATIENTS
HIV-POSITIVE
% OF HIV-
POSITIVE TB
PATIENTS ON
CPT
% OF HIV-
POSITIVE TB
PATIENTS ON
ART
NUMBER OF
HIV-POSITIVE
PEOPLE
PROVIDED IPT
Latvia 2005 85 1 226 1 443 53 4.3 55
2010 85 794 934 71 8.9 76
2011 85 752 885 71 9.4 41 66
• 85 85 • 2012 85 844 993 114 14 39 57
Lithuania 2005 2 574 7
2010 1 938 19
2011 1 904
–
–
2012 1 781
Luxembour
g
2005 37
2010 29
2011 26
–
–
2012 45
Malta 2005 4.3 1 23 0 0
2010 81 26 32 3 12 0
2011 91 30 33 5 17 4
• 4 98 • 2012 98 42 43 4 9.5
Monaco 2005
2010 1
2011
–
–
2012
Montene
g
ro 2005 4.7 8 170 0 0
2010 74 84 114 1 1.2 0 100
2011 82 92 112 0 0
• 5 77 • 2012 77 82 107 0 0
Netherlands 2005 22 252 1 157 61 24
2010 38 413 1 073 48 12 21
2011 49 490 1 007 31 6.3
• 22 42 • 2012 42 407 958 28 6.9
Norw a
y
2005 0 0 290
2010 339
2011 361
• 0
–
2012 3
Poland 2005 9 280
2010 0.29 22 7 509 22 100
2011 0.31 26 8 478 26 100
–
0 • 2012 0.34 26 7 542
Portu
g
al 2005 70 2 485 3 536 571 23
2010 65 1 720 2 626 303 18 100 100
2011 86 2 185 2 540 315 14
• 70 65 • 2012 65 1 672 2 590 291 17
Re
p
ublic of 2005 100 6 469 6 278 9 0.14
Moldova 2010 95 5 192 5 447 308 5.9 9.7 31 0
2011 94 5 017 5 341 285 5.7
• 103 100 • 2012 100 5 348 5 341 303 5.7 34
Romania 2005 37 10 860 29 347 160 1.5
2010 37 7 833 21 078 241 3.1 41 89 133
2011 50 9 608 19 212 244 2.5 59 90 145
• 37 53 • 2012 53 9 699 18 224 229 2.4 76 90 174
Russian 2005 55 85 537 154 379 3 533 4.1
Federation 2010 84 669 162 553 3 633 200
2011 79 494 159 479 4 104
• 55
–
2012 75 995 149 921 4 880
San Marino 2005
2010
2011
–
–
2012
Serbia 2005 <0.1 3 3 468 3 100 430 400
2010 0.67 16 2 385 12 75 0 100 4
2011 3.2 72 2 216 6 8.3 0 100
• 0 2 • 2012 2 39 1 917 6 15 0 100
Slovakia 2005 95 720 760 1 0.14 0 100
2010 100 439 439 1 0.23 100 100 0
2011 99 395 399 0 0
• 95 93 • 2012 93 322 345 0 0 0
Slovenia 2005 38 107 278 0 0
2010 76 130 172 1 0.77
2011 77 147 192 0 0
• 38 75 • 2012 75 104 138 0 0
S
p
ain 2005 8 359
2010 69 4 909 7 089 456 9.3
2011 68 4 569 6 762 414 9.1
–
70 • 2012 70 4 179 5 991 370 8.9
Sweden 2005 0 0 569
2010 675
2011 586
• 0
–
2012 632
Switzerland 2005 563
2010 548
2011 578
–
–
2012 463
Ta
j
ikistan 2005 8.9 670 7 526 1 0.15 0 0
2010 53 4 049 7 641 100 2.5 73 54 0
2011 82 6 241 7 609 115 1.8 70 57 315
• 9 92 • 2012 92 6 375 6 929 88 1.4 80 89 157
The Former 2005 0.3 2 658 2 100 0 100
Yu
g
oslav Re
p
ublic 2010 9.3 39 420 0 0 0
of Macedonia 2011 12 45 362 0 0 0
• 0 41 • 2012 41 145 355 0 0 0
Turke
y
2005 0 0 21 303 0
2010 3.5 581 16 551 14 2.4 36 64
2011 46 7 241 15 679 29 0.4 48 93
• 0 59 • 2012 59 8 646 14 691 45 0.52 49 78
Turkmenistan 2005 3 291
2010 100 3 230 3 230 0 0
2011
–
–
2012
Ukraine 2005 39 608 1 526 0
2010 95 34 621 36 409 5 752 17 39 5 029
2011 74 31 776 42 676 4 157 13 63
–
75 • 2012 75 34 181 45 569 4 726 14 72 71 14 352
United Kin
g
dom of 2005 8 633
Great Britain and 2010 8 483 378
Northern Ireland 2011 8 963 326
–
–
2012 8 751
Uzbekistan 2005 120 35 801 28 891 147 0.41 0 0
2010 100 20 330 20 330 427 2.1 92 37
2011 100 15 913 15 913 546 3.4 96 32 2 630
• 124 100 • 2012 100 16 810 16 810 820 4.9 95 13 2 010

247 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
EUROPEAN REGION
7$%/($7HVWLQJIRU0'57%DQGQXPEHURIFRQILUPHGFDVHVRI0'57%±
NEW PULMONARY CASES PREVIOUSLY TREATED CASES
YEAR
ESTIMATED CASES
OF MDR-TB AMONG
NOTIFIED
NUMBER OF
BACT+VE
b
TESTED FOR
MDR-TB
% OF
BACT+VE
b
TESTED FOR
MDR-TB
ESTIMATED CASES
OF MDR-TB AMONG
NOTIFIED
NUMBER OF
NOTIFIED
TESTED FOR
MDR-TB
% OF NOT IFIED
TESTED FOR
MDR-TB
TOTAL
CONFIRMED
CASES OF
MDR-TB
a
ESTIMATED CASES
OF MDR-TB AMONG
NOTIFIED
Albania 2005 1 161 75 12 28
2010 2 186 76 19 63
2011 5 194 87 11 61
2012 1 1.7
(
0–4.9
)
1.7
(
<0.1–9.1
)
172 76 0
(
0–6.3
)
15 52
Andorra 2005 0 9 150
–
2010 0 4 100 0
–
2011 0 1 100 1 100
2012 0 0
(
0–4.9
)
0
(
0–4.9
)
4 100 0
(
0–0
)
0
–
Armenia 2005 162 576 99 182 56
2010 177 361 87 99 22
2011 79 439 96 90 24
2012 92 250
(
220–280
)
82
(
61–110
)
420 94 170
(
150–190
)
91 23
Austria 2005 13 570 110 16 62
2010 15 203 99 15 52
2011 19 257 95 11 55
2012 27 18
(
6.7–30
)
11
(
5.1–20
)
254 93 7.3
(
0.91–21
)
25 62
Azerbai
j
an 2005 800 453 29 366 11
2010 552 801 19 960 48
2011 811 569 25 151 7.0
2012 596 2 800
(
2 600–3 000
)
810
(
670–960
)
–
2 000
(
1 800–2 200
)
–
Belarus 2005
–
–
2010 1576 1972 90 1697 150
2011 1594 2084 94 948 88
2012 1604 2 200
(
2 100–2 200
)
1 200
(
1 100–1 300
)
2164 90 960
(
920–1 000
)
1183 84
Bel
g
ium 2005 11 588 89 41 60
2010 19 466 97 52 60
2011 15 524 94 35 59
2012 20 15
(
5.8–25
)
6.3
(
2.5–13
)
503 95 8.9
(
2.5–21
)
53 68
Bosnia and 2005 11 1035 100 106 68
Herze
g
ovina 2010 2 600 100 47 47
2011 7 704 99 41 63
2012 7 13
(
2.0–24
)
1.6
(
<0.1–8.9
)
724 97 12
(
3.2–28
)
66 55
Bul
g
aria 2005 47 482 40 691 340
2010 56 801 85 165 47
2011 55 588 62 145 41
2012 49 100
(
78–130
)
32
(
18–51
)
687 71 73
(
52–98
)
142 45
Croatia 2005 6 586 100 61 65
2010 0
–
–
2011 8 353 96 40
–
2012
–
–
–
–
–
C
yp
rus 2005 1 16 84 0 0
2010 0 14 70 0
–
2011 1 25 96 2 67
2012 0 1.7
(
0–5.0
)
1.7
(
<0.1–8.8
)
40 93 0
(
0–5.1
)
233
Czech Re
p
ublic 2005 13 562 100 20 59
2010 9 352 97 28 55
2011 7 392 96 16 52
2012 4 9.8
(
2.3–17
)
7.3
(
2.7–16
)
371 93 2.5
(
<0.1–12
)
26 65
Denmar
k
2005 5 307 140 18 62
2010 2 209 98 30 65
2011 3 257 100 14 64
2012 1
–
–
–
–
–
Estonia 2005 79 316 110 71 76
2010 63 197 100 61 77
2011 78 210 100 52 68
2012 62 70
(
56–85
)
42
(
31–56
)
193 100 28
(
20–36
)
46 82
Finland 2005 3 198 85 22 100
2010 6 184 96 7 47
2011 5 237 97 8 62
2012 3 2.7
(
0–5.6
)
2.7
(
0.55–7.6
)
206 99 0
(
0–4.2
)
14 78
France 2005 24 1291 47 112 30
2010 23 1187 120 91 29
2011 40 1232 73 110 42
2012 39
–
–
–
–
–
Geor
g
ia 2005 195 799 53 515 24
2010 359 1987 80 558 40
2011 475 2197 83 675 52
2012 346 630
(
570–690
)
260
(
220–300
)
1931 84 370
(
330–420
)
541 45
German
y
2005 105 3094 98 251 51
2010 48 2215 110 184 50
2011 56 2382 91 148 49
2012 64 62
(
44–81
)
37
(
25–52
)
2198 89 26
(
13–43
)
116 47
Greece 2005 12 497 170 0 0
2010 2 115 37 15 34
2011 5 148 44 11 31
2012
–
–
–
–
–
Greenland 2005
–
–
2010 1
–
–
2011
–
–
2012 1.6
(
1.0–2.2
)
1.4
(
0.69–2.0
)
–
0.24
(
0.18–0.29
)
–
Hun
g
ar
y
2005 26 442 62 88 25
2010 19 474 92 80 31
2011 30 411 73 68 31
2012 12 31
(
15–46
)
23
(
11–42
)
411 79 7.3
(
3.0–14
)
31 37
Iceland 2005 0 7 140 1 100
2010 0 19 120 0
–
2011 0 4 80 0 0
2012 1 1.0
(
1.0–1.0
)
0
(
0–4.2
)
4 100 1.0
(
<0.1–1.0
)
1 100
Ireland 2005 3 200 110 10 25
2010 2 200 130 22 71
2011 3 176 85 15 56
2012 5 1.8
(
0–4.4
)
1.8
(
0.22–6.6
)
190 97 0
(
0–4.9
)
17 68
Israel 2005 16 259 110 6 86
2010 12 245 120 2 50
2011 11 275 99 9 90
2012 17 22
(
12–32
)
19
(
11–30
)
318 98 3.7
(
0.48–8.5
)
655
Ital
y
2005
–
–
2010
–
–
2011
–
–
2012
–
–
–
–
–
Kazakhstan 2005
–
–
2010 7387 5214 100 4655 51
2011 7408 5293 83 4790 80
2012 7608 7 000
(
6 900–7 200
)
2 700
(
2 600–2 800
)
8154 140 4 300
(
4 300–4 400
)
10443 130
K
y
r
gy
zstan 2005 989 837 20 152 18
2010 566 225 14 264 27
2011 806
–
–
2012 958 1 800
(
1 600–2 000
)
1 100
(
910–1 200
)
1659 99 730
(
690–770
)
831 78
a
TOTAL CONFIRMED CASES OF MDR-TB includes cases with unknown previous treatment history (i.e. not included under NEW CASES or PREVIOUSLY TREATED CASES).
b
BACT+VE = bacteriologically positive cases.

GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($7HVWLQJIRU0'57%DQGQXPEHURIFRQILUPHGFDVHVRI0'57%±
NEW PULMONARY CASES PREVIOUSLY TREATED CASES
YEAR
ESTIMATED CASES
OF MDR-TB AMONG
NOTIFIED
NUMBER OF
BACT+VE
b
TESTED FOR
MDR-TB
% OF
BACT+VE
b
TESTED FOR
MDR-TB
ESTIMATED CASES
OF MDR-TB AMONG
NOTIFIED
NUMBER OF
NOTIFIED
TESTED FOR
MDR-TB
% OF NOT IFIED
TESTED FOR
MDR-TB
TOTAL
CONFIRMED
CASES OF
MDR-TB
a
ESTIMATED CASES
OF MDR-TB AMONG
NOTIFIED
Latvia 2005 160 873 100 182 89
2010 87 613 100 102 94
2011 105 562 96 82 85
2012 110 120
(
100–140
)
87
(
69–110
)
666 97 36
(
26–48
)
100 88
Lithuania 2005 338 1293 100 440 96
2010 310 959 100 360 99
2011 296 1031 100 369 100
2012 271 300
(
270–330
)
150
(
120–170
)
1017 100 150
(
140–170
)
350 100
Luxembour
g
2005 0 36 110
–
2010 0 17 120 0
–
2011 2 7 100 1 100
2012 0 0
(
0–0.98
)
0
(
0–0
)
0
–
0
(
0–0.98
)
1 100
Malta 2005 0 11 140
–
2010 1 11 220 2 67
2011 0 17 89 0 0
2012 0 0
(
0–0
)
0
(
0–7.2
)
13 81 0
(
0–0.98
)
1 100
Monaco 2005
–
–
2010 1
–
–
2011
–
–
2012
–
–
–
–
–
Montene
g
ro 2005 2 82 88 14 52
2010 0 61 100 12 100
2011 1 57 100 13 110
2012 0 0
(
0–0
)
0
(
0–5.0
)
58 98 0
(
0–6.8
)
538
Netherlands 2005 7 709 130 30 68
2010 11 741 160 29 67
2011 15 695 99 22 58
2012 11 9.1
(
3.5–15
)
7.4
(
3.5–13
)
628 99 1.8
(
<0.1–9.0
)
28 57
Norw a
y
2005 3 193 150 8 57
2010 8 139 100 21 50
2011 4 229 97 22 59
2012 6
–
–
–
–
–
Poland 2005 72 5409 120
–
2010 30 3238 81 468 52
2011 41 4416 88 577 60
2012 31 48
(
31–65
)
30
(
18–47
)
4073 90 18
(
9.0–32
)
535 61
Portu
g
al 2005 28 1407 77 172 49
2010 19 982 77 94 41
2011 22 1155 73 97 45
2012 17 35
(
21–50
)
25
(
15–41
)
1219 72 9.7
(
3.2–22
)
102 54
Re
p
ublic of 2005 338 536 32 652 37
Moldova 2010 1082 1381 49 1140 67
2011 1001 1379 74 1006 68
2012 894 1 700
(
1 600–1 800
)
810
(
730–890
)
1264 67 930
(
880–980
)
933 63
Romania 2005 530 1594 13 1300 19
2010 502 3338 39 2011 39
2011 530 3855 41 2171 46
2012 500 800
(
610–980
)
320
(
210–480
)
3645 40 480
(
350–630
)
1864 43
Russian 2005
–
–
Federation 2010 13692 35862 72 13405 29
2011 13785 34007 78 13620 25
2012 13612 46 000
(
43 000–49 000
)
20 000
(
18 000–22 000
)
32647 79 25 000
(
23 000–28 000
)
12324 24
San Marino 2005
–
–
2010
–
–
2011
–
–
2012
–
–
–
–
–
Serbia 2005 9 1112 76 121 40
2010 12 811 67 113 56
2011 9 863 91 100 62
2012 9 20
(
7.0–33
)
13
(
4.9–29
)
716 84 6.5
(
1.3–18
)
83 46
Slovakia 2005 8 248 82 56 52
2010 1 185 100 32 58
2011 5 147 92 29 58
2012 4 1.8
(
0–5.3
)
0
(
0–6.2
)
142 95 1.8
(
<0.1–9.3
)
27 55
Slovenia 2005 1 217 110 28 97
2010 0 123 100 9 82
2011 0 171 100 11 100
2012 0 0
(
0–0
)
0
(
0–3.5
)
114 100 0
(
0–3.7
)
12 86
S
p
ain 2005
–
–
2010 49 1009 34 110 34
2011 41 1013 24 96 26
2012 37 31
(
13–49
)
8.5
(
1.0–31
)
802 21 22
(
10–41
)
69 22
Sweden 2005 4 425 150 17 57
2010 18 288 100 24 46
2011 17 375 100 31 69
2012 14 11
(
5.0–18
)
8.1
(
4.1–14
)
453 100 3.2
(
0.40–11
)
24 62
Switzerland 2005 5 326 150 30 61
2010 9 270 130 33 82
2011 8 304 98 40 74
2012 8 8.6
(
2.4–15
)
2.6
(
0.53–7.4
)
246 98 6.1
(
1.7–14
)
31 66
Ta
j
ikistan 2005
–
–
2010 333 160 7.0 223 23
2011 604 161 7.4 415 45
2012 694 910
(
800–1 000
)
490
(
390–620
)
919 45 420
(
390–450
)
496 66
The Former 2005 4 106 51 19 18
Yu
g
oslav Re
p
ublic 2010 7 153 110 28 54
of Macedonia 2011 1 130 72 25 45
2012 4 4.8
(
0.47–9.1
)
0
(
0–5.7
)
155 81 4.8
(
1.4–11
)
26 84
Turke
y
2005 191 3237 38 508 20
2010 250 4342 64 615 45
2011 262 4221 63 602 48
2012 291 520
(
460–580
)
270
(
230–310
)
4742 71 250
(
220–290
)
641 55
Turkmenistan 2005
–
–
2010 38 81 7.0 63 77
2011 158 306
–
156
–
2012
–
–
–
–
–
Ukraine 2005
–
–
2010 5336 9194 66 4840 95
2011 4305 10352 61 4413 38
2012 6934 6 800
(
6 500–7 000
)
4 100
(
3 900–4 300
)
11185 77 2 600
(
2 600–2 700
)
5925 72
United Kin
g
dom of 2005 39 3428 100 271 59
Great Britain and 2010 60 3970 150 247 43
Northern Ireland 2011 81 4549 95 234 45
2012 81 69
(
54–85
)
54
(
42–70
)
4570 97 15
(
8.1–25
)
244 51
Uzbekistan 2005 86 0 0 435 4.8
2010 1023 2845 60 1180 26
2011 1385 484 9.5 123 11
2012 1728 4 000 (3 700–4 300) 2 400 (1 800–3 000) 2703 56 1 600 (1 400–1 900) 798 30
a
TOTAL CONFIRMED CASES OF MDR-TB includes cases with unknown previous treatment history (i.e. not included under NEW CASES or PREVIOUSLY TREATED CASES).
b
BACT+VE = bacteriologically positive cases.
249 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
EUROPEAN REGION
7$%/($1HZVPHDUSRVLWLYHFDVHQRWLILFDWLRQE\DJHDQGVH[±
MALE FEMALE
YEAR 0–14 15–24 25–34 35–44 45–54 55–64 65+
UN-
KNOWN
0–14 15–24 25–34 35–44 45–54 55–64 65+
UN-
KNOWN
MALE:FEMALE
RATIO
Albania 1995 0 0 0 0 19 40 30 0 1 0 0 13 20 16 1.8
2000 2 19 21 14 24 19 16 3 11 10 8 8 5 11 2.1
2005 0 26 21 16 31 20 37 0 0 3 9 5 5 5 18 0 3.4
2010 0 28 17 14 16 16 15 0 2 11 7 6 3 2 8 0 2.7
2011 0 29 26 18 30 9 22 0 1 14 10 6 2 1 12 0 2.9
2012 0 33 34 16 15 11 23 0 0 17 9 6 3 6 12 0 2.5
Andorra 1995 –
2000 0010000 –
2005 00110000 01110000 0.67
2010 00000000 00000000 –
2011 00001000 00000000 –
2012 00000110 00000000 –
Armenia 1995 1 18 16 11 10 8 1 1 1 7 2 1 1 5.0
2000 2 152 130 131 63 26 21 1 24 27 24 8 8 4 5.5
2005 3 170 104 83 84 30 24 0 3 27 21 10 11 4 7 0 6.0
2010 0 36 75 49 68 27 15 0 1 24 17 4 7 8 8 0 3.9
2011 0 28 65 52 71 42 8 0 0 19 16 9 7 7 5 0 4.2
2012 1 23 67 60 56 34 18 0 0 13 19 12 2 5 5 0 4.6
Austria 1995 4 37 95 82 89 71 73 6 22 52 32 21 18 59 2.1
2000 1 17 30 59 42 23 41 1 11 22 12 11 6 22 2.5
2005 1 32 23 22 41 24 30 0 0 13 11 8 3 5 10 0 3.5
2010 04412138100 15422560 2.0
2011 0 8 11 9 13 11 13 0 0 11 6 4 1 3 4 0 2.2
2012 1587199130 110844150 1.9
Azerbaijan 1995 0 13 29 14 6 4 1 0 5 18 0 0 0 0 2.9
2000 0 9 24 33 42 30 0 0 3 3 6 3 0 0 9.2
2005 77 109 297 215 209 187 88 0 90 64 98 47 32 24 24 0 3.1
2010 0 328 371 267 280 30 27 3 141 100 57 73 9 18 3.2
2011 –
2012 4 230 223 170 176 95 48 0 8 115 89 35 50 35 23 0 2.7
Belarus 1995 –
2000 –
2005 71 180 273 287 118 62 25 53 50 43 11 62 4.1
2010 0 65 173 224 293 163 58 0 1 28 52 56 37 28 91 0 3.3
2011 1 53 156 228 290 138 48 3 37 67 47 39 27 83 3.0
2012 0 44 174 250 266 158 73 0 1 34 64 47 45 28 93 0 3.1
Belgium 1995 3 23 49 63 52 54 102 3 12 24 32 17 10 34 2.6
2000 3 20 57 39 55 32 56 6 15 15 19 4 13 27 2.6
2005 1 26 50 32 27 15 47 0 2 27 31 15 12 4 23 0 1.7
2010 4 20 39 30 29 21 19 0 6 13 18 19 11 5 10 0 2.0
2011 8 25 50 33 25 18 27 0 3 13 14 9 3 5 7 0 3.4
2012 3 25 33 18 27 22 18 0 5 23 23 17 9 7 5 0 1.6
Bosnia and 1995 0 15 61 90 140 139 100 0 40 67 64 49 77 23 1.7
Herzegovina 2000 4 56 82 99 66 58 77 4 30 46 29 29 48 124 1.4
2005 1 22 58 61 78 44 80 1 2 35 39 33 28 28 130 0 1.2
2010 1 27 37 34 61 46 51 0 0 27 19 16 10 18 94 0 1.4
2011 2 33 32 52 75 61 62 0 3 17 27 17 13 25 128 0 1.4
2012 1 23 32 58 74 62 92 1 0 33 26 21 10 25 116 1 1.5
Bulgaria 1995 –
2000 0 13 16 20 3 9 10 0 11 14 7 3 4 6 1.6
2005 9 98 150 195 195 150 136 0 9 90 111 59 29 37 70 0 2.3
2010 1 40 115 143 133 90 65 0 3 42 59 43 23 15 34 0 2.7
2011 2 38 100 110 122 92 61 0 2 41 40 36 28 14 30 0 2.7
2012 0 46 89 130 131 82 57 0 0 37 50 44 24 16 35 0 2.6
Croatia 1995 6 38 97 210 132 178 141 10 50 57 57 38 60 130 2.0
2000 –
2005 1 24 27 48 72 47 34 0 1 12 18 15 11 6 56 0 2.1
2010 0 10 19 18 38 25 24 1 3 8 4 2 1 30 2.7
2011 0 12 5 20 31 31 21 0 0 12 14 14 8 7 26 0 1.5
2012 –
Cyprus 1995 0110112 0111201 1.0
2000 –
2005 03111010 01000000 7.0
2010 02100000 00310001 0.60
2011 00340010 01020000 2.7
2012 00421100 03210010 1.1
Czech Republic 1995 2 10 22 83 88 53 90 0 9 11 20 13 19 88 2.2
2000 0 7 31 52 89 61 59 0 15 13 9 10 7 57 2.7
2005 0 8 24 57 55 45 46 0 0 3 14 16 7 5 28 0 3.2
2010 0 12 19 36 29 29 19 0 0 6 10 11 7 2 20 0 2.6
2011 0 10 29 20 38 28 24 0 0 4 9 4 4 3 15 0 3.8
2012 0 7 21 24 42 33 22 0 1 3 11 8 3 7 26 0 2.5
Denmark 1995 0 7 16 28 18 9 11 2 7 13 8 4 3 2 2.3
2000 5 10 20 24 16 11 14 5 16 15 14 6 7 8 1.4
2005 0 12 12 18 23 9 7 0 2 11 5 13 9 3 5 0 1.7
2010 0 8 22 10 13 16 2 0 0 4 5 15 8 8 4 0 1.6
2011 051418321640 05597270 2.5
2012 –
Estonia 1995 –
2000 0 6 31 53 56 35 15 0 9 11 14 11 4 10 3.3
2005 0 9 25 19 40 12 7 0 0 6 11 8 11 6 8 0 2.2
2010 03721251280 03533630 3.3
2011 0 4 22 16 14 18 13 0 0 4 8 12 3 3 6 0 2.4
2012 061513211790 05724150 3.4
Finland 1995 1 1 10 25 28 24 61 1 1 6 7 4 10 65 1.6
2000 0 3 8 22 19 28 53 0 1 5 3 4 6 49 2.0
2005 15431411250 034106200 1.9
2010 0106898180 032412110 2.6
2011 0144711270 123531130 1.9
2012 029759210 140423110 2.1
France 1995 30 156 431 502 414 297 496 36 138 226 176 90 92 365 2.1
2000 10 136 248 247 211 125 244 18 108 127 89 46 43 155 2.1
2005 12 127 212 222 196 134 205 0 16 104 134 82 56 38 180 0 1.8
2010 10 60 139 114 99 76 110 0 10 47 76 49 45 25 97 0 1.7
2011 12 88 112 116 94 73 101 0 7 58 67 48 36 23 65 0 2.0
2012 –
Georgia 1995 2 20 30 25 40 18 12 2 8 17 17 18 7 5 2.0
2000 4 76 111 113 63 45 28 1 49 37 33 17 10 5 2.9
2005 0 226 272 268 207 76 60 4 109 105 58 46 17 47 2.9
2010 5 340 529 341 264 143 77 0 5 135 118 62 52 28 41 0 3.9
2011 5 271 478 333 251 139 93 0 8 136 132 59 32 35 54 0 3.4
2012 4 200 314 248 235 150 81 0 5 101 116 72 43 32 47 0 3.0
Germany 1995 14 179 453 539 460 442 625 17 115 251 167 89 104 397 2.4
2000 –
2005 6 59 113 171 167 92 167 0 4 51 104 73 43 37 103 0 1.9
2010 1 43 92 97 141 87 136 0 3 44 63 61 38 26 76 0 1.9
2011 1 43 96 106 141 69 131 0 2 44 92 59 54 26 86 0 1.6
2012 4 43 99 113 147 105 99 0 5 49 99 37 47 24 55 1 1.9

250 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($1HZVPHDUSRVLWLYHFDVHQRWLILFDWLRQE\DJHDQGVH[±
MALE FEMALE
YEAR 0–14 15–24 25–34 35–44 45–54 55–64 65+
UN-
KNOWN
0–14 15–24 25–34 35–44 45–54 55–64 65+
UN-
KNOWN
MALE:FEMALE
RATIO
Greece 1995 –
2000 1 10 22 32 24 19 46 0 2 9 10 5 6 25 2.7
2005 1 14 25 22 14 12 23 5 0 13 18 8 7 2 17 0 1.8
2010 1 19 27 20 18 19 22 3 3 2 13 4 4 4 15 1 2.8
2011 2 30 30 26 24 19 38 2 1 9 14 9 3 5 20 1 2.8
2012 –
Greenland 1995 –
2000 –
2005 –
2010 05755220 15004200 2.2
2011 01020331 0811230 1.3
2012 0635151 2303103 1.8
Hungary 1995 –
2000 0 8 24 85 104 58 27 1 7 17 19 22 10 30 2.9
2005 0 6 24 67 117 67 39 0 1 5 13 11 22 15 33 0 3.2
2010 1 9 15 36 51 52 23 0 0 9 16 14 9 15 20 0 2.3
2011 0 11 18 34 46 53 28 0 0 3 9 8 9 12 29 0 2.7
2012 2 7 15 29 64 41 25 0 0 8 14 15 11 14 28 0 2.0
Iceland 1995 0000001 0000001 1.0
2000 01 –
2005 00010010 00000000 –
2010 00103000 00110000 2.0
2011 00000000 00000010 –
2012 00001000 00100000 1.0
Ireland 1995 –
2000 0 10 7 7 6 4 12 0 13 8 13 6 7 15 0.74
2005 16102110760 091033080 1.8
2010 08184115110 07823250 2.1
2011 0791011780 08778210 1.6
2012 179991240 15664220 2.0
Israel 1995 –
2000 0 20 26 23 23 13 38 0 3 10 16 6 3 3 32 0 2.0
2005 1 4 15 18 15 5 26 0 0 6 14 7 7 5 19 0 1.4
2010 1 13 28 12 8 4 6 0 0 1 8 10 2 0 10 0 2.3
2011 0 29 30 11 5 9 9 0 0 10 10 7 4 4 7 0 2.2
2012 0 9 33 20 3 6 13 0 0 4 20 11 4 2 17 0 1.4
Italy 1995 9 59 202 157 94 124 289 7 52 93 57 40 51 168 2.0
2000 12 63 96 75 58 54 112 6 38 58 33 13 19 39 2.3
2005 8 93 191 137 101 61 115 24 3 80 145 56 25 19 70 9 1.8
2010 14 40 75 66 32 31 58 2 25 41 57 41 22 22 54 6 1.2
2011 0 51 88 81 52 24 59 0 5 41 73 37 18 14 43 1 1.5
2012 –
Kazakhstan 1995 –
2000 36 1 057 1 409 1 379 923 439 218 84 999 1 079 599 275 202 204 1.6
2005 31 917 1 142 983 795 274 175 0 46 751 767 436 286 121 187 0 1.7
2010 15 675 754 595 511 251 127 0 33 566 520 263 205 122 132 0 1.6
2011 6 602 716 516 515 235 91 0 15 439 495 260 190 109 117 0 1.6
2012 9 508 586 514 479 233 98 0 16 415 411 241 177 97 100 0 1.7
Kyrgyzstan 1995 3 109 171 165 65 38 30 1 70 94 34 18 15 19 2.3
2000 4 128 227 205 115 52 46 6 128 146 100 41 30 29 1.6
2005 1 247 303 269 194 66 84 0 15 215 236 141 70 33 98 0 1.4
2010 5 261 260 188 141 64 48 0 5 223 199 98 71 40 42 0 1.4
2011 6 225 204 179 168 77 41 0 13 200 191 84 60 50 39 0 1.4
2012 4 210 255 207 184 86 30 0 8 195 173 108 55 42 37 0 1.6
Latvia 1995 0 20 44 71 70 40 30 0 22 49 55 47 27 29 1.2
2000 0 53 106 124 111 64 34 2 25 41 27 28 7 15 3.4
2005 1 22 71 104 117 55 34 0 0 17 31 31 23 18 12 0 3.1
2010 0 20 44 65 71 39 15 0 0 6 19 25 12 10 13 0 3.0
2011 0 11 42 58 50 33 18 0 0 7 16 19 14 12 13 0 2.6
2012 0 19 62 67 59 36 15 0 1 14 15 14 16 15 9 0 3.1
Lithuania 1995 4 46 132 225 176 90 77 5 6 53 45 32 16 42 3.8
2000 1 38 97 145 155 74 68 0 20 37 39 32 22 48 2.9
2005 0 42 118 186 187 108 67 0 1 25 41 57 49 23 54 0 2.8
2010 1 34 75 128 157 89 54 0 1 20 36 31 43 18 32 0 3.0
2011 1 25 52 126 158 77 55 0 0 20 31 37 38 16 45 0 2.6
2012 0 35 73 143 148 91 60 0 1 8 28 55 36 20 28 0 3.1
Luxembourg 1995 –
2000 –
2005 00221120 00211100 1.6
2010 00000000 00000000 –
2011 01001100 01000000 3.0
2012 00000000 00000000 –
Malta 1995 0001000 0010012 0.25
2000 0101101 –
2005 01111100 00000000 –
2010 00300000 00100000 3.0
2011 02400000 01000000 6.0
2012 12220000 01100000 3.5
Monaco 1995 –
2000 –
2005 –
2010 –
2011 –
2012 –
Montenegro 2005 035715480 00734080 1.9
2010 01144710 13323180 0.86
2011 012811730 14243110 2.0
2012 034510340 04512130 1.8
Netherlands 1995 22 79 119 75 28 9 10 24 56 50 13 10 8 7 2.0
2000 0 34 63 41 25 10 21 4 29 22 16 9 5 10 2.0
2005 0 23 42 23 26 14 19 0 3 14 19 11 9 1 4 0 2.4
2010 0 22 29 22 20 9 17 0 1 9 14 13 5 4 11 0 2.1
2011 2 22 35 19 23 14 13 0 2 13 13 7 7 4 3 0 2.6
2012 1 15 31 14 18 9 15 0 4 7 18 15 4 6 6 0 1.7
Norway 1995 04863512 0472038 1.6
2000 0193624 131 25 2.1
2005 09464430 04721030 1.8
2010 09971420 05732000 1.9
2011 03733110 014601010 0.82
2012 –
Poland 1995 3 122 295 795 565 369 377 4 129 163 225 111 107 414 2.2
2000 1 99 303 812 782 361 434 1 99 158 211 170 82 421 2.4
2005 3 109 199 389 639 292 310 0 3 95 142 112 151 63 316 0 2.2
2010 3 70 205 310 574 393 237 0 2 59 118 82 104 82 245 0 2.6
2011 5 69 187 314 560 439 275 0 1 67 96 90 130 99 255 0 2.5
2012 1 82 183 306 471 438 267 0 1 54 96 106 102 100 226 0 2.6

251 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
EUROPEAN REGION
7$%/($1HZVPHDUSRVLWLYHFDVHQRWLILFDWLRQE\DJHDQGVH[±
MALE FEMALE
YEAR 0–14 15–24 25–34 35–44 45–54 55–64 65+
UN-
KNOWN
0–14 15–24 25–34 35–44 45–54 55–64 65+
UN-
KNOWN
MALE:FEMALE
RATIO
Portugal 1995 11 215 363 328 200 173 164 7 139 172 87 33 42 85 2.6
2000 8 147 375 349 208 140 140 5 114 154 87 41 25 64 2.8
2005 5 85 227 284 181 90 93 5 7 67 109 66 29 11 42 1 2.9
2010 3 55 110 199 152 70 76 0 3 54 62 54 36 10 28 0 2.7
2011 3 56 87 177 172 75 74 3 4 43 58 56 30 12 25 1 2.8
2012 1 56 103 187 153 79 75 1 6 52 62 66 28 19 32 0 2.5
Republic of 1995 0 55 115 166 95 65 15 2 42 38 31 19 10 12 3.3
Moldova 2000 2 52 31 36 13 13 6 1 16 32 45 23 14 6 1.1
2005 2 211 337 345 313 106 31 0 3 97 92 57 61 23 18 0 3.8
2010 0 119 243 244 248 113 21 0 6 47 90 46 47 23 20 0 3.5
2011 2 94 257 250 267 107 21 0 3 66 79 51 41 20 14 0 3.6
2012 0 99 234 256 284 131 31 0 3 58 95 48 56 26 25 0 3.3
Romania 1995 387 1 662 2 322 3 608 2 587 1 751 784 355 1 352 1 240 871 479 396 417 2.6
2000 46 832 1 508 1 799 1 684 916 533 53 701 766 484 341 207 321 2.5
2005 36 752 1 511 1 786 1 999 952 638 4 55 758 780 493 374 219 442 2 2.5
2010 21 669 865 1 336 1 293 895 567 0 40 503 477 400 275 172 438 0 2.4
2011 19 623 813 1 192 1 104 837 541 0 26 475 513 407 214 196 426 0 2.3
2012 17 556 764 1 297 1 053 831 495 0 22 431 433 371 188 184 435 0 2.4
Russian 1995 –
Federation 2000 1 295 526 596 402 151 54 1 43 73 74 38 31 44 6.7
2005 –
2010 8 2 228 6 276 5 571 5 361 2 787 920 0 28 1 247 2 554 1 719 1 182 745 790 0 2.8
2011 15 1 826 5 726 5 338 4 928 2 664 845 36 1 139 2 394 1 643 1 166 719 752 2.7
2012 17 1 568 5 472 5 115 4 446 2 629 839 0 31 997 2 292 1 595 1 081 637 748 0 2.7
San Marino 1995 –
2000 1 –
2005 –
2010 –
2011 –
2012 –
Serbia 2005 3 62 96 118 156 112 132 0 6 69 76 55 49 22 149 0 1.6
2010 2 76 70 93 116 83 109 0 5 66 74 46 39 34 164 0 1.3
2011 2 60 73 74 122 112 101 0 5 46 59 43 30 20 129 0 1.6
2012 1 70 72 77 98 86 77 0 4 46 66 38 31 35 118 0 1.4
Serbia & 1995 10 108 204 317 296 350 386 11 127 167 133 83 158 275 1.8
Montenegro 2000 –
Slovakia 1995 4 18 44 123 108 63 152 5 16 17 22 24 33 159 1.9
2000 2 6 15 31 50 16 32 0 5 9 7 5 4 54 1.8
2005 0 3 13 16 25 25 20 0 0 1 8 9 5 6 27 0 1.8
2010 1 7 7 18 17 17 15 0 0 1 6 7 2 3 11 0 2.7
2011 06862016130 023461110 2.6
2012 02917201270 023461130 2.3
Slovenia 1995 1 13 39 63 36 26 27 0 7 24 11 9 5 42 2.1
2000 0 3 11 36 22 14 17 0 3 9 3 4 3 20 2.5
2005 0 4 10 16 15 11 14 0 0 4 4 6 5 4 16 0 1.8
2010 0471096120 01524130 3.0
2011 0391612850 005421170 1.8
2012 02648650 02301190 1.9
Spain 1995 22 132 337 242 150 112 228 23 90 129 64 39 34 98 2.6
2000 –
2005 13 166 394 367 230 140 230 2 10 142 252 151 63 24 108 2 2.1
2010 6 139 306 291 286 146 184 1 14 130 251 151 54 23 76 0 1.9
2011 15 135 325 292 277 162 197 2 15 142 249 161 75 30 100 0 1.8
2012 10 112 259 299 276 156 220 3 15 101 202 161 70 24 74 1 2.1
Sweden 1995 1 5 12 8 5 4 27 0 10 13 5 5 4 14 1.2
2000 0 9 10 12 11 4 25 1 9 8 10 2 2 15 1.5
2005 0 7 21 16 10 5 16 0 1 10 15 12 5 3 13 0 1.3
2010 1 10 28 8 5 5 13 0 2 9 16 11 4 2 3 0 1.5
2011 1 14 15 12 8 3 8 0 0 12 9 10 2 2 3 0 1.6
2012 0816898130 0111037260 1.6
Switzerland 1995 0 12 23 26 23 13 27 1 13 20 9 1 2 15 2.0
2000 0517107660 181172150 1.5
2005 28101111270 061183140 1.5
2010 061296580 071564130 1.3
2011 28161013730 261324220 1.9
2012 0318865110 071571240 1.4
Tajikistan 1995 –
2000 –
2005 8 308 279 164 104 54 48 0 26 225 185 151 89 43 53 0 1.2
2010 12 398 366 214 129 93 74 0 23 320 272 111 109 87 82 0 1.3
2011 8 343 365 181 128 75 77 0 31 314 229 104 100 105 114 0 1.2
2012 8 346 320 169 124 75 72 0 16 243 243 105 99 94 127 0 1.2
The Former 1995 2 15 42 45 33 29 24 2 32 30 20 11 17 17 1.5
Yugoslav Republic 2000 5 8 14 20 19 20 14 1 15 14 17 5 5 10 1.5
of Macedonia 2005 2 14 20 23 20 18 13 1 2 17 13 10 7 5 13 0 1.7
2010 0 6 19 24 24 12 11 0 0 9 12 7 7 4 6 0 2.1
2011 3 17 11 19 21 10 6 0 1 14 9 6 3 1 11 0 1.9
2012 0 16 14 12 19 15 13 0 0 12 14 9 6 10 7 0 1.5
Turkey 1995 –
2000 –
2005 33 1 148 1 295 1 028 963 534 429 0 50 699 474 243 175 166 213 0 2.7
2010 23 631 779 703 778 514 407 0 33 485 384 193 141 101 203 0 2.5
2011 22 550 693 608 696 482 412 0 25 409 385 195 117 121 212 0 2.4
2012 20 507 655 575 650 476 398 0 30 369 308 168 97 105 227 0 2.5
Turkmenistan 1995 1 11 188 0 79 30 0 2 15 146 0 47 25 0 1.3
2000 16 103 185 144 127 31 21 19 73 140 76 31 34 17 1.6
2005 2 148 181 146 97 51 13 0 3 100 101 72 46 27 8 0 1.8
2010 1 130 212 183 141 51 26 2 112 112 74 46 38 25 1.8
2011 –
2012 –
Ukraine 1995 10 385 1 076 2 064 1 515 1 087 437 21 314 380 327 182 185 280 3.9
2000 21 693 1 552 2 385 2 007 1 062 532 41 487 590 447 298 218 405 3.3
2005 –
2010 7 417 2 559 2.9
2011 8 539 1 991 2 209 1 796 881 377 0 11 348 741 603 388 230 380 0 2.9
2012 9 546 2 028 2 393 1 926 965 389 10 334 771 609 401 218 431 3.0
United Kingdom of 1995 –
Great Britain and 2000 8 86 130 96 87 75 138 9 95 114 60 31 31 67 1.5
Northern Ireland 2005 9 135 200 166 95 95 124 0 14 115 163 80 39 28 83 1 1.6
2010 7 132 169 135 108 60 108 0 15 110 131 81 42 40 58 0 1.5
2011 3 137 193 137 97 69 100 0 19 120 129 75 45 26 49 0 1.6
2012 8 156 184 137 118 88 88 0 17 109 141 81 55 17 52 0 1.7
Uzbekistan 1995 –
2000 6 351 749 510 346 213 107 11 261 547 288 213 112 111 1.5
2005 25 596 831 723 522 263 313 40 538 597 375 288 217 367 1.4
2010 8 487 574 529 479 293 297 0 22 365 512 308 248 239 350 0 1.3
2011 8 378 493 453 440 306 253 0 11 335 418 233 245 293 332 0 1.2
2012 10 360 506 403 449 313 273 0 9 319 367 237 201 261 322 0 1.3

252 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
a LED = Light emitting diode microscopes
b DST = Drug susceptibility testing
c LPA = Line probe assay
d NRL = National Reference Laboratory
7$%/($/DERUDWRULHV173VHUYLFHVGUXJPDQDJHPHQWDQGLQIHFWLRQFRQWURO
LABORATORIES FREE THROUGH NTP
SMEAR LABS
PER 100K
POPULATION
% OF SMEAR
LABS USING
LED
a
CULTURE
LABS PER 5M
POPULATION
DST
b
LABS
PER 5M
POPULATION
LPA
c
LABS
PER 5M
POPULATION
NUMBER OF
LABS USING
XPERT MTB/RIF
SECOND-
LINE DST
AVAILABLE
NRL
d
TB DIAGNOSIS
FIRST-
LINE
DRUGS
TB NOTIF.
RATE PER
100 000
HEALTH-CARE
WORKERS
RIFAMPICIN
USED
THROUGHOUT
TREATMENT
Albania 0.5 0 1.6 1.6 1.6 0 No Yes Yes (all suspects) Yes Yes
Andorra 10.2 0 510.5 510.5 0 0 In and out
of countr y Yes Yes (all suspects) Yes Yes
Armenia 1.0 0 1.7 1.7 1.7 0 In country Yes Yes (all suspects) Yes Yes
Austria –
Azerbaijan 0.8 4 3.8 1.6 0.5 7 Yes Yes (all suspects) Yes Yes
Belarus 2.1 2 15.4 4.3 4.3 8 In country Yes Yes (all suspects) Yes Yes 25
Belgium 1.0 – 51.5 6.3 3.6 19 Yes Yes (other criteria) Yes Yes
Bosnia and Herzegovina 0.4 100 17.0 3.9 0 0 Out of
country Yes Yes (all suspects) Yes Yes
Bulgaria 0.5 0 21.3 9.6 2.7 0 In country Yes Yes (all suspects) Yes Yes
Croatia –
Cyprus –
Czech Republic 0.4 – 21.6 8.9 8.9 In country Yes Yes (all suspects) Yes Yes
Denmark 0.2 – 0.9 0.9 0.9 In country Yes Yes (all suspects) Yes Yes
Estonia 0.4 100 7.7 7.7 7.7 2 In country Yes Yes (all suspects) Yes Yes 12
Finland 0.2 100 10.2 0.9 2.8 1 In country Yes Yes (all suspects) Yes Yes
France 0.4 – 18.0 5.5 1.6 20 In country Yes Yes (all suspects) Yes Yes
Georgia 0.3 9 2.3 1.1 2.3 1 In country Yes Yes (all suspects) Yes Yes 107
Germany 0.3 – 11.4 5.1 4.5 141 Yes Yes (all suspects) Yes Yes
Greece –
Greenland –
Hungary 0.1 0 6.0 3.5 1 3 Yes Yes (all suspects) Yes Yes
Iceland 0.3 100 15.3 15.3 15.3 0 Out of
country Yes Yes
(
if TB is
confirmed) Yes Yes
Ireland 0.2 27 10.9 3.3 2.2 3 Out of
country Yes Yes (all suspects) No Yes
Israel 0.2 – 12.4 1.3 0.7 1 In country Yes Yes (all suspects) Yes Yes
Italy –
Kazakhstan 2.9 0 6.8 6.8 3.4 4 In country Yes Yes (all suspects) Yes Yes 204
Kyrgyzstan 2.2 0 10.0 2.7 1.8 7 In and out
of countr y Yes Yes (all suspects) Yes Yes 34
Latvia 0.8 0 9.7 2.4 2.4 2 In country Yes Yes (all suspects) Yes Yes
Lithuania 0.4 8 9.9 9.9 3.3 7 In and out
of countr y Yes Yes
(
if TB is
confirmed) Yes Yes
Luxembourg 0.2 100 9.5 9.5 9.5 0 Out of
country Yes Yes (all suspects) Yes Yes
Malta 0.2 0 11.7 0 0 0 Out of
country No Yes (all suspects) Yes Yes
Monaco –
Montenegro 0.2 0 8.1 8.1 0 0 Out of
country Yes Yes
(
if TB is
confirmed) Yes Yes 56
Netherlands 0.3 – 11.1 1.5 1.2 2 In country Yes Yes (all suspects) No Yes
Norway 0.3 0 9.0 3 4 3 In and out
of countr y Yes Yes (all suspects) Yes Yes
Poland 0.2 0 10.6 6 1.4 3 Yes Yes (all suspects) Yes Yes
Portugal 0.5 – 22.2 10.4 Yes Yes (all suspects) Yes Yes
Republic of Moldova 1.7 0 5.7 5.7 4.3 24 Yes Yes (all suspects) Yes Yes
Romania 0.5 1 20.9 9.9 0.9 0 In and out
of countr y Yes Yes (all suspects) Yes Yes 51
Russian Federation 0.7 – 4.1 3.8 In country No Yes (all suspects) Yes Yes
San Marino –
Serbia 0.3 0 15.2 2.1 0.5 0 Out of
country Yes Yes
(
if TB is
confirmed) Yes Yes 8
Slovakia 0.1 14 6.4 1.8 1.8 2 In and out
of countr y Yes Yes (all suspects) Yes Yes
Slovenia 0.1 67 7.3 2.4 2.4 1 In and out
of countr y Yes Yes (all suspects) Yes Yes
Spain – Yes Yes (all suspects) Yes Yes
Sweden <0.1 – 2.6 2.6 2.6 0 In country Yes Yes (all suspects) Yes Yes
Switzerland 0.5 – 14.4 6.3 14 In country Yes Yes (all suspects) No Yes
Tajikistan 1.1 4 1.9 0.6 0.6 3 In country Yes Yes
(
if TB is
confirmed) Yes Yes 34
The Former Yugoslav
Republic of Macedonia 0.3 0 7.1 2.4 0 0 Out of
country Yes Yes
(
if TB is
confirmed) Yes Yes 22
Turkey 0.5 – 10.7 5.1 0.6 18 In country Yes Yes (all suspects) Yes Yes 25
Turkmenistan –
Ukraine 1.8 5 9.4 4.5 0 15 Yes Yes (all suspects) Yes Yes 61
United Kingdom of Great
Britain and Northern
Ireland
–Yes Yes (all suspects) Yes Yes
Uzbekistan 1.0 1 1.2 0.5 0.5 7 In country Yes Yes (all suspects) Yes Yes 29

253 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
EUROPEAN REGION
7$%/($0HDVXUHGSHUFHQWDJHRI7%FDVHVZLWK0'57%
D
PRVWUHFHQW\HDUDYDLODEOH
New TB cases Previously treated TB cases
Year Source Coverage Percentage Year Source Coverage Percentage
Albania 2012 Surveillance National 0.58 (<0.1–3.2) 2012 Surveillance National 0 (0–22)
Andorra 2011 Surveillance National 0 (0–98) 2011 Surveillance National 0 (0–98)
Armenia 2007 Survey National 9.4 (7.0–12) 2007 Survey National 43 (38–49)
Austria 2011 Surveillance National 3.5 (1.6–6.5) 2011 Surveillance National 18 (2.3–52)
Azerbaijan 2007 Survey Sub-national 22 (19–27) 2007 Survey Sub-national 56 (50–62)
Belarus 2012 Surveillance National 35 (33–37) 2012 Surveillance National 69 (66–71)
Belgium 2011 Surveillance National 1.3 (0.54–2.7) 2011 Surveillance National 11 (3.2–27)
Bosnia and Herzegovina 2011 Surveillance National 0.14 (0–0.79) 2011 Surveillance National 9.8 (2.7–23)
Bulgaria 2012 Surveillance National 2.3 (1.3–3.8) 2012 Surveillance National 23 (17–31)
Croatia 2011 Surveillance National 0.28 (<0.1–1.6) 2011 Surveillance National 2.5 (<0.1–13)
Cyprus 2011 Surveillance National 4 (0.10–20) 2011 Surveillance National 0 (0–84)
Czech Republic 2011 Surveillance National 1.5 (0.56–3.3) 2011 Surveillance National 6.3 (0.16–30)
Denmark 2011 Surveillance National 1.2 (0.24–3.4) 2011 Surveillance National 0 (0–23)
Estonia 2012 Surveillance National 20 (14–26) 2012 Surveillance National 50 (35–65)
Finland 2012 Surveillance National 1.5 (0.30–4.2) 2012 Surveillance National 0 (0–23)
France 2009 Surveillance National 0.45 (0.24–0.77) 2009 Surveillance National 13 (7.4–21)
Georgia 2012 Surveillance National 9.2 (7.9–11) 2012 Surveillance National 31 (27–35)
Germany 2012 Surveillance National 1.5 (1.0–2.0) 2012 Surveillance National 10 (5.5–17)
Greece 2010 Surveillance National 0.87 (<0.1–4.7) 2010 Surveillance National 6.7 (0.17–32)
Greenland
Hungary 2010 Surveillance National 2.1 (1.0–3.8) 2010 Surveillance National 8.8 (3.6–17)
Iceland 2012 Surveillance National 0 (0–60) 2012 Surveillance National 100 (2.5–100)
Ireland 2012 Surveillance National 1.1 (0.13–3.8) 2012 Surveillance National 0 (0–20)
Israel 2012 Surveillance National 4.7 (2.7–7.7) 2012 Surveillance National 33 (4.3–78)
Italy 2011 Surveillance Sub-national 3.9 (2.7–5.6) 2011 Surveillance Sub-national 5.4 (3.5–8.0)
Kazakhstan 2012 Surveillance National 23 (22–24) 2012 Surveillance National 55 (54–56)
Kyrgyzstan 2011 Survey National 26 (23–30) 2012 Surveillance National 68 (65–72)
Latvia 2012 Surveillance National 11 (8.8–14) 2012 Surveillance National 32 (23–42)
Lithuania 2012 Surveillance National 11 (9.5–14) 2012 Surveillance National 44 (39–49)
Luxembourg 2011 Surveillance National 0 (0–41) 2011 Surveillance National 0 (0–98)
Malta 2012 Surveillance National 0 (0–25) 2012 Surveillance National 0 (0–98)
Monaco
Montenegro 2012 Surveillance National 0 (0–6.2) 2012 Surveillance National 0 (0–52)
Netherlands 2012 Surveillance National 1.6 (0.77–2.9) 2012 Surveillance National 3.6 (<0.1–18)
Norway 2011 Surveillance National 1.3 (0.27–3.8) 2011 Surveillance National 0 (0–15)
Poland 2012 Surveillance National 0.49 (0.30–0.76) 2012 Surveillance National 2.1 (1.0–3.6)
Portugal 2011 Surveillance National 1.5 (0.86–2.3) 2011 Surveillance National 5.2 (1.7–12)
Republic of Moldova 2012 Surveillance National 24 (21–26) 2012 Surveillance National 62 (59–65)
Romania 2004 Survey National 2.8 (1.8–4.2) 2004 Survey National 11 (8.0–15)
Russian Federation 2011 Surveillance Sub-national 23 (21–25) 2011 Surveillance Sub-national 49 (44–53)
San Marino
Serbia 2012 Surveillance National 0.84 (0.31–1.8) 2012 Surveillance National 3.6 (0.75–10)
Slovakia 2012 Surveillance National 0 (0–2.6) 2012 Surveillance National 3.7 (<0.1–19)
Slovenia 2012 Surveillance National 0 (0–3.2) 2012 Surveillance National 0 (0–26)
Spain 2001, 2005 Surveillance Sub-national 0.22 (<0.1–0.80) 2001, 2005 Surveillance Sub-national 7.1 (3.3–13)
Sweden 2012 Surveillance National 2.4 (1.2–4.3) 2012 Surveillance National 8.3 (1.0–27)
Switzerland 2012 Surveillance National 1.2 (0.25–3.5) 2012 Surveillance National 13 (3.6–30)
Tajikistan 2011 Survey National 13 (9.8–16) 2012 Surveillance National 56 (52–60)
The Former Yugoslav
Republic of Macedonia 2012 Surveillance National 0 (0–2.4) 2012 Surveillance National 15 (4.4–35)
Turkey 2012 Surveillance National 3.2 (2.7–3.7) 2012 Surveillance National 22 (19–25)
Turkmenistan 2002 Survey Sub-national 3.8 (1.1–9.5) 2002 Survey Sub-national 18 (11–27)
Ukraine 2012 Surveillance National 14 (14–15) 2012 Surveillance National 32 (31–33)
United Kingdom of Great
Britain and Northern
Ireland
2011 Surveillance National 1.3 (1.0–1.7) 2011 Surveillance National 5.6 (3.0–9.3)
Uzbekistan 2011 Survey National 23 (18–29) 2011 Survey National 62 (52–71)
a Empty rows indicate an absence of high-quality survey or surveillance data. In the absence of high-quality national data, high-quality sub-national data are used.

5176*'#56#5+#4')+10
Table A4.1 Estimates of the burden of disease caused by TB, 1990–2012 257
6CDNG# +PEKFGPEGPQVKßECVKQPCPFECUGFGVGEVKQPTCVGUCNNHQTOU¿
6CDNG# %CUGPQVKßECVKQPU¿
Table A4.4 Treatment outcomes, new smear-positive cases, 1995–2011 260
Table A4.5 Treatment outcomes, retreatment cases, 1995–2011 261
6CDNG# *+8VGUVKPICPFRTQXKUKQPQH%26#46CPF+26¿
6CDNG# 6GUVKPIHQT/&46$CPFPWODGTQHEQPßTOGFECUGUQH/&46$¿
6CDNG# 0GYUOGCTRQUKVKXGECUGPQVKßECVKQPD[CIGCPFUGZ¿
Table A4.9 Laboratories, NTP services, drug management and infection control, 2012 265
6CDNG# /GCUWTGFRGTEGPVCIGQH6$ECUGUYKVJ/&46$OQUVTGEGPV[GCTCXCKNCDNG

256 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
Estimates of mortality, prevalence and incidence
Estimated values are shown as best estimates followed by lower and upper bounds. e lower and upper bounds are dened
as the 2.5th and 97.5th centiles of outcome distributions produced in simulations. See ANNEX 1 for further details.
Estimated numbers are shown rounded to two signicant gures. Estimated rates are shown rounded to three signicant
gures unless the value is under 100, in which case rates are shown rounded to two signicant gures.
Estimates for all years are recalculated as new information becomes available and techniques are rened, so
they may dier from those published in previous reports in this series. e main updates implemented in this
report are explained in Box 2.1 of Chapter 2. Estimates published in previous global TB control reports should no
longer be used.
Data source
Data shown in this annex are taken from the WHO global TB database on 1 October 2013. Data shown in the main part of
the report were taken from the database in July 2013. As a result, data in this annex may dier slightly from those in the
main part of the report.
Data for all years can be downloaded from www.who.int/tb/data.
Country notes
Bangladesh
Estimates of TB disease burden have not been ocially approved by the national TB programme (NTP) in Bangladesh. A
joint reassessment by WHO and the NTP will be undertaken following the completion of the prevalence survey planned
for 2014.
India
Estimates of TB disease burden for India have not yet been ocially approved by the Ministry of Health & Family Welfare,
Government of India and should therefore be considered provisional.

257 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
SOUTH-EAST ASIA REGION
7$%/($(VWLPDWHVRIWKHEXUGHQRIGLVHDVHFDXVHGE\7%±
YEAR POPULATION
(MILLIONS)
NUMBER
(THOUSANDS) RATEaNUMBER
(THOUSANDS) RATEaNUMBER
(THOUSANDS) RATEa
MORTALITY (EXCLUDING HIV) PREVALENCE (INCLUDING HIV) INCIDENCE (INCLUDING HIV)
Bangladesh 1990 107 66 (20–140) 61 (18–130) 560 (220–1 100) 525 (202–998) 240 (150–360) 225 (139–331)
1995 120 72 (27–140) 60 (22–116) 620 (290–1 100) 518 (244–893) 270 (220–320) 225 (184–270)
2000 132 77 (29–150) 58 (22–111) 670 (320–1 100) 507 (243–866) 300 (240–360) 225 (184–270)
2005 143 74 (29–140) 52 (20–98) 670 (330–1 100) 469 (231–790) 320 (260–390) 225 (184–270)
2010 151 69 (28–130) 46 (19–85) 660 (330–1 100) 437 (220–727) 340 (280–410) 225 (185–268)
2011 153 70 (28–130) 46 (19–84) 660 (340–1 100) 435 (220–722) 340 (280–410) 225 (185–268)
2012 155 70 (29–130) 45 (19–84) 670 (340–1 100) 434 (218–721) 350 (290–410) 225 (185–268)
Bhutan 1990 < 1 1 (0.410–2.0) 194 (77–365) 10 (4.7–17) 1 860 (881–3 190) 4.2 (3.6–4.8) 784 (673–903)
1995 < 1 0.55 (0.230–1.0) 109 (45–200) 6 (3.0–10) 1 180 (599–1 960) 2.9 (2.5–3.3) 561 (482–646)
2000 < 1 0.41 (0.180–0.740) 73 (31–132) 4.3 (2.2–6.9) 754 (392–1 230) 2.3 (1.9–2.6) 402 (345–463)
2005 < 1 0.35 (0.160–0.600) 53 (25–92) 3.5 (1.8–5.7) 536 (279–875) 1.9 (1.6–2.2) 287 (247–331)
2010 < 1 0.11 (0.068–0.160) 15 (9.5–22) 2.2 (1.1–3.8) 313 (149–536) 1.5 (1.3–1.7) 206 (177–237)
2011 < 1 0.11 (0.068–0.160) 15 (9.4–22) 2 (0.840–3.5) 269 (115–486) 1.4 (1.2–1.6) 192 (165–222)
2012 < 1 0.1 (0.062–0.150) 14 (8.4–21) 1.7 (0.580–3.3) 225 (79–446) 1.3 (1.1–1.5) 180 (154–207)
Democratic 1990 20 4.7 (4.3–5.0) 23 (21–25) 97 (26–210) 479 (130–1 050) 77 (44–120) 383 (219–592)
People's Republic 1995 22 4.6 (4.2–5.0) 21 (19–23) 100 (28–230) 479 (130–1 050) 83 (48–130) 383 (219–592)
of Korea 2000 23 4 (3.7–4.3) 17 (16–19) 110 (30–240) 479 (130–1 050) 87 (50–140) 383 (219–592)
2005 24 3 (2.7–3.2) 12 (12–13) 110 (31–250) 479 (130–1 050) 91 (52–140) 383 (219–592)
2010 25 2.5 (2.3–2.6) 10 (9.5–11) 120 (33–270) 494 (134–1 080) 97 (85–110) 395 (348–445)
2011 25 2.5 (2.4–2.6) 10 (9.6–11) 120 (34–270) 505 (137–1 110) 100 (92–110) 404 (372–437)
2012 25 2.2 (2.1–2.4) 9 (8.6–9.5) 130 (34–280) 511 (139–1 120) 100 (92–110) 409 (373–447)
India 1990 869 330 (220–480) 38 (25–55) 4 000 (3 600–4 500) 465 (415–518) 1 900 (1 600–2 200) 216 (182–254)
1995 956 370 (240–520) 38 (25–55) 4 400 (4 000–5 000) 465 (414–519) 2 100 (1 800–2 300) 216 (189–245)
2000 1 042 400 (260–570) 39 (25–55) 4 600 (4 000–5 200) 438 (382–498) 2 300 (2 000–2 500) 216 (195–239)
2005 1 127 400 (290–530) 36 (26–47) 4 100 (3 300–5 000) 365 (295–443) 2 400 (2 100–2 600) 209 (188–231)
2010 1 206 320 (210–460) 27 (17–38) 3 200 (2 200–4 500) 269 (181–374) 2 200 (2 000–2 500) 185 (167–204)
2011 1 221 300 (190–420) 24 (16–35) 3 000 (2 100–4 200) 249 (168–346) 2 200 (2 000–2 400) 181 (163–199)
2012 1 237 270 (170–390) 22 (14–32) 2 800 (1 900–3 900) 230 (155–319) 2 200 (2 000–2 400) 176 (159–193)
Indonesia 1990 179 95 (33–190) 53 (18–106) 790 (330–1 400) 442 (186–806) 370 (270–480) 206 (149–271)
1995 194 120 (42–230) 61 (21–120) 940 (400–1 700) 483 (205–878) 400 (310–500) 205 (159–256)
2000 209 120 (42–220) 55 (20–107) 990 (460–1 700) 474 (222–821) 430 (340–520) 204 (164–249)
2005 224 84 (34–160) 38 (15–70) 830 (410–1 400) 369 (183–621) 450 (360–540) 199 (160–242)
2010 241 67 (30–120) 28 (12–50) 740 (360–1 300) 306 (148–521) 450 (380–540) 189 (156–224)
2011 244 67 (30–120) 27 (12–49) 730 (350–1 200) 301 (145–512) 460 (380–540) 187 (155–222)
2012 247 67 (30–120) 27 (12–48) 730 (350–1 200) 297 (144–506) 460 (380–540) 185 (153–220)
Maldives 1990 < 1 0.059 (0.052–0.067) 27 (24–31) 0.67 (0.260–1.3) 311 (119–593) 0.32 (0.200–0.480) 150 (92–221)
1995 < 1 0.033 (0.027–0.040) 14 (11–17) 0.48 (0.230–0.820) 197 (95–336) 0.29 (0.230–0.350) 118 (96–142)
2000 < 1 0.015 (0.010–0.019) 5.4 (3.8–7.1) 0.22 (0.082–0.430) 81 (30–157) 0.17 (0.130–0.200) 60 (49–73)
2005 < 1 <0.01 (<0.01–<0.01) 2 (1.6–2.5) 0.23 (0.100–0.410) 78 (34–138) 0.15 (0.120–0.180) 51 (42–62)
2010 < 1 <0.01 (<0.01–<0.01) 2.3 (2.0–2.5) 0.17 (0.065–0.310) 51 (20–96) 0.12 (0.097–0.140) 36 (30–44)
2011 < 1 <0.01 (<0.01–<0.01) 1.9 (1.7–2.1) 0.14 (0.051–0.280) 43 (15–85) 0.11 (0.088–0.130) 33 (27–39)
2012 < 1 <0.01 (<0.01–<0.01) 2 (1.8–2.2) 0.22 (0.100–0.380) 65 (30–113) 0.14 (0.110–0.170) 41 (33–49)
Myanmar 1990 42 48 (17–97) 115 (39–230) 380 (170–650) 894 (414–1 550) 170 (120–220) 393 (290–512)
1995 45 53 (19–110) 118 (41–234) 400 (190–680) 881 (421–1 500) 180 (140–230) 404 (314–505)
2000 48 51 (19–100) 106 (39–207) 400 (200–670) 831 (415–1 390) 200 (160–240) 412 (333–498)
2005 50 35 (15–65) 70 (29–129) 320 (170–530) 647 (333–1 060) 200 (170–240) 403 (340–472)
2010 52 26 (12–46) 51 (23–89) 270 (210–340) 525 (404–661) 200 (170–230) 384 (329–444)
2011 52 26 (12–45) 49 (23–86) 260 (200–330) 506 (390–637) 200 (170–230) 381 (326–439)
2012 53 25 (12–44) 48 (23–84) 260 (200–320) 489 (377–616) 200 (170–230) 377 (322–435)
Nepal 1990 18 7.5 (2.2–16) 41 (12–88) 66 (25–130) 364 (140–692) 30 (18–44) 163 (101–241)
1995 21 6.1 (2.5–11) 29 (12–54) 61 (30–100) 295 (147–493) 34 (27–40) 163 (133–196)
2000 23 5 (2.2–8.9) 21 (9.4–38) 58 (26–100) 248 (113–436) 38 (31–45) 163 (133–196)
2005 25 4.9 (2.1–8.9) 20 (8.4–35) 59 (26–110) 235 (101–424) 41 (34–50) 163 (133–196)
2010 27 5.3 (2.4–9.4) 20 (8.8–35) 64 (28–110) 238 (105–425) 44 (36–52) 163 (135–194)
2011 27 5.4 (2.4–9.6) 20 (8.8–35) 64 (28–110) 236 (103–423) 44 (37–53) 163 (135–194)
2012 27 5.5 (2.5–9.8) 20 (9.0–36) 66 (29–120) 241 (106–429) 45 (37–53) 163 (135–195)
Sri Lanka 1990 17 1.3 (0.750–2.0) 7.5 (4.3–12) 20 (7.4–40) 118 (43–231) 11 (7.2–17) 66 (42–96)
1995 18 1.6 (0.970–2.5) 9 (5.3–14) 23 (11–38) 125 (63–207) 12 (9.9–14) 66 (54–79)
2000 19 1.9 (1.1–2.8) 10 (6.0–15) 22 (11–36) 115 (57–192) 12 (10–15) 66 (54–79)
2005 20 1.4 (1.0–1.8) 6.9 (5.2–8.8) 22 (10–37) 108 (52–185) 13 (11–16) 66 (54–79)
2010 21 0.59 (0.480–0.710) 2.8 (2.3–3.4) 22 (11–38) 108 (52–184) 14 (11–16) 66 (55–79)
2011 21 0.41 (0.330–0.500) 2 (1.6–2.4) 23 (11–39) 108 (52–184) 14 (11–17) 66 (55–79)
2012 21 0.24 (0.180–0.310) 1.1 (0.84–1.4) 23 (11–39) 109 (52–185) 14 (12–17) 66 (55–79)
Thailand 1990 57 11 (4.9–20) 20 (8.6–35) 130 (63–220) 227 (111–383) 78 (65–93) 138 (114–164)
1995 59 11 (4.7–20) 19 (8.0–34) 130 (64–210) 217 (109–362) 77 (63–91) 130 (107–154)
2000 62 20 (7.9–37) 31 (13–59) 180 (86–300) 286 (139–487) 110 (88–130) 171 (141–203)
2005 66 15 (6.6–27) 23 (10–42) 150 (77–260) 236 (117–395) 100 (84–120) 154 (127–184)
2010 66 10 (4.5–18) 16 (6.8–28) 120 (55–210) 179 (83–309) 85 (70–100) 128 (106–153)
2011 67 9.5 (4.1–17) 14 (6.2–26) 110 (51–200) 168 (76–296) 82 (68–98) 124 (102–147)
2012 67 9.2 (3.8–17) 14 (5.8–25) 110 (47–190) 159 (71–282) 80 (66–95) 119 (98–142)
Timor-Leste 2005 < 1 0.67 (0.290–1.2) 67 (29–121) 7.2 (3.1–13) 722 (306–1 310) 5 (4.0–6.0) 498 (406–601)
2010 1 0.62 (0.280–1.1) 57 (26–102) 7.2 (2.8–14) 666 (259–1 260) 5.4 (4.4–6.4) 498 (409–596)
2011 1 0.67 (0.300–1.2) 62 (28–109) 7.6 (3.1–14) 689 (279–1 280) 5.5 (4.5–6.5) 498 (409–596)
2012 1 0.82 (0.360–1.5) 74 (33–132) 8.4 (3.8–15) 758 (342–1 340) 5.6 (4.6–6.6) 498 (409–596)
a Rates are per 100 000 population.

GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($,QFLGHQFHQRWLILFDWLRQDQGFDVHGHWHFWLRQUDWHVDOOIRUPV±
INCIDENCE (INCLUDING HIV) INCIDENCE HIV-POSITIVE NOTIFIED NEW AND RELAPSE
b
CASE DETECTION
YEAR POPULATION
(MILLIONS)
NUMBER
(THOUSANDS) RATEaNUMBER
(THOUSANDS) RATEaNUMBER RATEaPERCENT
Bangladesh 1990 107 240 (150–360) 225 (139–331) 0.048 (0.030–0.071) <0.1 (<0.1–<0.1) 48 673 45 20 (14–33)
1995 120 270 (220–320) 225 (184–270) 0.054 (0.044–0.065) <0.1 (<0.1–<0.1) 56 437 47 21 (17–26)
2000 132 300 (240–360) 225 (184–270) 0.089 (0.073–0.11) <0.1 (<0.1–<0.1) 75 557 57 25 (21–31)
2005 143 320 (260–390) 225 (184–270) 0.19 (0.16–0.23) 0.1 (0.11–0.16) 123 118 86 38 (32–47)
2010 151 340 (280–410) 225 (185–268) 0.31 (0.25–0.36) 0.2 (0.17–0.24) 153 892 102 45 (38–55)
2011 153 340 (280–410) 225 (185–268) 0.34 (0.28–0.41) 0.2 (0.18–0.27) 154 358 101 45 (38–55)
2012 155 350 (290–410) 225 (185–268) 0.24 (0.20–0.29) 0.2 (0.13–0.19) 168 683 109 49 (41–59)
Bhutan 1990 < 1 4.2 (3.6–4.8) 784 (673–903) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 1 154 215 27 (24–32)
1995 < 1 2.9 (2.5–3.3) 561 (482–646) <0.01 (<0.01–<0.01) 0.2 (0.14–0.19) 1 299 255 45 (39–53)
2000 < 1 2.3 (1.9–2.6) 402 (345–463) <0.01 (<0.01–<0.01) 0.4 (0.38–0.51) 1 140 202 50 (44–59)
2005 < 1 1.9 (1.6–2.2) 287 (247–331) <0.01 (<0.01–<0.01) 1.2 (1.0–1.4) 1 007 155 54 (47–63)
2010 < 1 1.5 (1.3–1.7) 206 (177–237) 0.019 (0.016–0.022) 2.6 (2.3–3.0) 1 311 183 89 (77–100)
2011 < 1 1.4 (1.2–1.6) 192 (165–222) 0.021 (0.018–0.025) 2.9 (2.5–3.4) 1 235 169 88 (76–100)
2012 < 1 1.3 (1.1–1.5) 180 (154–207) 0.024 (0.021–0.028) 3.3 (2.8–3.8) 1 130 152 85 (73–99)
Democratic 1990 20 77 (44–120) 383 (219–592)
People's Republic 1995 22 83 (48–130) 383 (219–592) 0.033 (0.018–0.054) 0.2 (<0.1–0.25)
of Korea 2000 23 87 (50–140) 383 (219–592) 0.087 (0.043–0.15) 0.4 (0.19–0.65) 34 131 149 39 (25–68)
2005 24 91 (52–140) 383 (219–592) 0.11 (0.054–0.18) 0.5 (0.23–0.77) 42 722 179 47 (30–82)
2010 25 97 (85–110) 395 (348–445) 0.13 (0.085–0.18) 0.5 (0.34–0.72) 84 648 345 87 (78–99)
2011 25 100 (92–110) 404 (372–437) 0.13 (0.084–0.18) 0.5 (0.34–0.75) 91 433 371 92 (85–100)
2012 25 100 (92–110) 409 (373–447) 0.13 (0.086–0.19) 0.5 (0.35–0.76) 91 885 371 91 (83–100)
India 1990 869 1 900 (1 600–2 200) 216 (182–254) 19 (16–22) 2.2 (1.8–2.6) 1 519 182 175 81 (69–96)
1995 956 2 100 (1 800–2 300) 216 (189–245) 90 (78–100) 9.4 (8.2–11) 1 218 183 127 59 (52–67)
2000 1 042 2 300 (2 000–2 500) 216 (195–239) 170 (150–190) 16 (14–18) 1 115 718 107 49 (45–55)
2005 1 127 2 400 (2 100–2 600) 209 (188–231) 170 (160–190) 16 (14–17) 1 156 248 103 49 (44–55)
2010 1 206 2 200 (2 000–2 500) 185 (167–204) 130 (120–150) 11 (10–12) 1 339 866 111 60 (54–66)
2011 1 221 2 200 (2 000–2 400) 181 (163–199) 130 (120–140) 11 (9.6–12) 1 323 949 108 60 (54–66)
2012 1 237 2 200 (2 000–2 400) 176 (159–193) 130 (120–140) 10 (9.4–12) 1 289 836 104 59 (54–66)
Indonesia 1990 179 370 (270–480) 206 (149–271) 74 470 42 20 (15–28)
1995 194 400 (310–500) 205 (159–256) 35 529 18 8.9 (7.1–12)
2000 209 430 (340–520) 204 (164–249) 0.085 (0.068–0.10) <0.1 (<0.1–<0.1) 84 591 40 20 (16–25)
2005 224 450 (360–540) 199 (160–242) 1.7 (1.3–2.1) 0.8 (0.59–0.94) 254 601 113 57 (47–71)
2010 241 450 (380–540) 189 (156–224) 5.7 (4.3–7.3) 2.4 (1.8–3.0) 300 659 125 66 (56–80)
2011 244 460 (380–540) 187 (155–222) 6.7 (5.0–8.5) 2.7 (2.1–3.5) 318 949 131 70 (59–85)
2012 247 460 (380–540) 185 (153–220) 7.5 (5.6–9.7) 3.1 (2.3–3.9) 328 824 133 72 (61–87)
Maldives 1990 < 1 0.32 (0.200–0.480) 150 (92–221) <0.01 (<0.01–<0.01) 0.2 (0.10–0.43) 152 70 47 (32–76)
1995 < 1 0.29 (0.230–0.350) 118 (96–142) <0.01 (<0.01–<0.01) 0.3 (0.13–0.40) 231 94 80 (66–98)
2000 < 1 0.17 (0.130–0.200) 60 (49–73) <0.01 (<0.01–<0.01) 0.1 (<0.1–0.21) 132 48 80 (66–98)
2005 < 1 0.15 (0.120–0.180) 51 (42–62) <0.01 (<0.01–<0.01) <0.1 (<0.1–0.15) 122 41 80 (66–98)
2010 < 1 0.12 (0.097–0.140) 36 (30–44) <0.01 (<0.01–<0.01) <0.1 (<0.1–0.10) 95 29 80 (66–98)
2011 < 1 0.11 (0.088–0.130) 33 (27–39) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 87 26 80 (66–98)
2012 < 1 0.14 (0.110–0.170) 41 (33–49) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 110 33 80 (66–98)
Myanmar 1990 42 170 (120–220) 393 (290–512) 0.9 (0.66–1.2) 2.1 (1.6–2.8) 12 416 29 7.5 (5.8–10)
1995 45 180 (140–230) 404 (314–505) 6.2 (4.8–7.7) 14 (11–17) 18 229 40 10 (8.0–13)
2000 48 200 (160–240) 412 (333–498) 15 (12–18) 30 (24–36) 30 840 64 15 (13–19)
2005 50 200 (170–240) 403 (340–472) 22 (18–25) 43 (36–50) 107 009 213 53 (45–63)
2010 52 200 (170–230) 384 (329–444) 21 (18–24) 40 (34–46) 131 590 253 66 (57–77)
2011 52 200 (170–230) 381 (326–439) 20 (17–23) 38 (32–43) 136 737 261 69 (59–80)
2012 53 200 (170–230) 377 (322–435) 19 (16–21) 35 (30–41) 141 170 267 71 (62–83)
Nepal 1990 18 30 (18–44) 163 (101–241) <0.01 (<0.01–0.013) <0.1 (<0.1–<0.1) 10 142 56 34 (23–56)
1995 21 34 (27–40) 163 (133–196) 0.081 (0.066–0.097) 0.4 (0.32–0.47) 19 804 96 59 (49–72)
2000 23 38 (31–45) 163 (133–196) 0.52 (0.42–0.62) 2.2 (1.8–2.7) 29 519 127 78 (65–95)
2005 25 41 (34–50) 163 (133–196) 1.4 (1.1–1.6) 5.4 (4.4–6.5) 33 448 132 81 (67–99)
2010 27 44 (36–52) 163 (135–194) 1.5 (1.2–1.7) 5.4 (4.5–6.5) 35 114 131 80 (67–97)
2011 27 44 (37–53) 163 (135–194) 1.4 (1.1–1.7) 5.1 (4.2–6.1) 35 434 130 80 (67–97)
2012 27 45 (37–53) 163 (135–195) 1.1 (0.94–1.4) 4.2 (3.4–5.0) 35 195 128 78 (66–95)
Sri Lanka 1990 17 11 (7.2–17) 66 (42–96) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 6 666 38 58 (40–92)
1995 18 12 (9.9–14) 66 (54–79) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 5 956 33 49 (41–60)
2000 19 12 (10–15) 66 (54–79) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 8 413 45 67 (56–83)
2005 20 13 (11–16) 66 (54–79) 0.011 (<0.01–0.013) <0.1 (<0.1–<0.1) 9 451 47 72 (60–88)
2010 21 14 (11–16) 66 (55–79) 0.014 (0.011–0.016) <0.1 (<0.1–<0.1) 9 934 48 72 (61–88)
2011 21 14 (11–17) 66 (55–79) 0.015 (0.012–0.019) <0.1 (<0.1–<0.1) 10 181 49 73 (62–89)
2012 21 14 (12–17) 66 (55–79) 0.017 (0.014–0.020) <0.1 (<0.1–0.10) 9 155 43 66 (55–80)
Thailand 1990 57 78 (65–93) 138 (114–164) 2.4 (2.0–2.9) 4.3 (3.5–5.1) 46 510 82 60 (50–72)
1995 59 77 (63–91) 130 (107–154) 12 (9.7–14) 20 (16–24) 45 428 77 59 (50–72)
2000 62 110 (88–130) 171 (141–203) 25 (21–30) 40 (33–48) 34 187 55 32 (27–39)
2005 66 100 (84–120) 154 (127–184) 19 (16–23) 29 (24–35) 57 895 88 57 (48–69)
2010 66 85 (70–100) 128 (106–153) 13 (11–16) 20 (17–24) 67 128 101 79 (66–95)
2011 67 82 (68–98) 124 (102–147) 13 (11–15) 19 (16–23) 65 824 99 80 (67–97)
2012 67 80 (66–95) 119 (98–142) 12 (10–14) 18 (15–22) 60 304 90 76 (64–92)
Timor-Leste 2005 < 1 5 (4.0–6.0) 498 (406–601) 3 767 378 76 (63–93)
2010 1 5.4 (4.4–6.4) 498 (409–596)
2011 1 5.5 (4.5–6.5) 498 (409–596) 4 386 400 80 (67–98)
2012 1 5.6 (4.6–6.6) 498 (409–596) 3 828 344 69 (58–84)
a Rates are per 100 000 population.
b NOTIFIED NEW AND RELAPSE includes cases for which the treatment history is unknown.

259 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
SOUTH-EAST ASIA REGION
7$%/($&DVHQRWLILFDWLRQV±
NEW CASES
YEAR SMEAR-
POSITIVE
SMEAR-NEGATIVE/
UNKNOWN
EXTRA-
PULMONARY OTHER RELAPSE RE-TREAT EXCL.
RELAPSE
TOTAL
RETREAT
HISTORY
UNKNOWN
NEW AND
RELAPSE
b
% SMEAR-
POS AMONG
NEW PULM
NEW AND RELAPSE
NOTIFICATION RATE
a
1990–2012
Bangladesh 1990 48 673 –
1995 56 437 20 524 19 297 2 060 729 729 52
2000 75 557 38 484 29 396 5 914 1 763 1 763 57
2005 123 118 84 848 23 076 11 318 3 876 3 876 79
2010 153 892 105 772 21 625 23 506 0 2 989 4 806 7 795 0 83
2011 154 358 98 948 21 921 27 329 0 2 701 4 665 7 366 3 459 82
• 45 109 • 2012 168 683 106 790 24 451 30 549 0 3 065 4 936 8 001 3 828 81
Bhutan 1990 1 154 –
1995 1 299 367 657 265 10 10 36
2000 1 140 347 430 363 36 36 45
2005 1 007 308 272 387 40 11 51 53
2010 1 311 457 275 518 0 61 21 82 0 62
2011 1 235 382 225 573 0 55 15 70 0 63
• 215 152 • 2012 1 130 420 127 519 64 15 79 77
Democratic 1990 –
Peo
p
le's Re
p
ubli
c
1995 –
of Korea 2000 34 131 16 440 13 801 3 787 103 103 54
2005 42 722 17 796 18 123 5 381 58 1 364 7 752 9 116 50
2010 84 648 31 240 36 285 13 715 3 408 11 650 15 058 46
2011 91 433 31 279 37 457 16 828 5 869 7 638 13 507 46
• 0 371 • 2012 91 885 31 904 35 959 17 321 6 701 7 514 14 215 47
India 1990 1 519 182 –
1995 1 218 183 264 515 880 589 68 979 690 690 23
2000 1 115 718 349 374 650 345 98 006 17 993 80 072 98 065 35
2005 1 156 248 508 890 399 066 171 838 1 381 75 073 148 580 223 653 0 56
2010 1 339 866 630 165 366 381 231 121 1 508 110 691 182 281 292 972 63
2011 1 323 949 642 321 340 203 226 965 1 952 112 508 191 923 304 431 65
• 175 104 • 2012 1 289 836 629 589 317 616 234 029 2 139 106 463 177 749 284 212 66
Indonesia 1990 74 470 –
1995 35 529 31 768 34 0 106 106 100
2000 84 591 52 338 15 035 833 1 448 1 448 78
2005 254 601 158 640 85 373 6 142 4 446 4 446 65
2010 300 659 183 366 101 247 11 659 0 4 387 2 202 6 589 0 64
2011 318 949 197 797 101 750 14 054 5 348 2 359 7 707 66
• 42 133 • 2012 328 824 202 319 104 866 15 697 5 942 2 600 8 542 66
Maldives 1990 152 –
1995 231 114 89 18 10 10 56
2000 132 65 31 32 4 0 4 68
2005 122 66 23 29 0 4 1 5 0 74
2010 95 41 20 33 0 1 2 3 0 67
2011 87 47 12 28 0 0 1 1 0 80
• 70 33 • 2012 110 52 17 41 0 0 1 1 0 75
Myanmar 1990 12 416 –
1995 18 229 8 681 7 058 653 1 837 1 837 55
2000 30 840 17 254 8 659 2 304 2 623 2 623 67
2005 107 009 36 541 35 601 30 252 4 615 982 5 597 51
2010 131 590 42 318 56 840 27 976 4 456 5 813 10 269 43
2011 136 737 42 324 62 038 27 769 4 606 6 403 11 009 41
• 29 267 • 2012 141 170 42 909 73 042 20 661 0 4 558 6 979 11 537 0 37
Nepal 1990 10 142 –
1995 19 804 8 591 7 938 2 489 786 786 52
2000 29 519 13 683 9 074 4 955 1 807 1 807 60
2005 33 448 14 617 9 474 7 013 0 2 344 629 2 973 61
2010 35 114 15 569 9 718 7 210 0 2 617 495 3 112 0 62
2011 35 434 15 000 9 662 7 484 926 2 362 520 2 882 0 61
• 56 128 • 2012 35 195 15 057 9 128 7 865 865 2 280 440 2 720 0 62
Sri Lanka 1990 6 666 2 769 3 241 656 46
1995 5 956 3 049 1 677 982 248 248 65
2000 8 413 4 314 2 261 1 561 277 372 649 66
2005 9 451 4 868 2 198 1 917 0 266 244 510 202 69
2010 9 934 4 635 2 145 2 548 0 219 161 380 387 68
2011 10 181 4 490 2 405 2 612 0 248 147 395 426 65
• 38 43 • 2012 9 155 4 269 1 889 2 349 0 245 188 433 403 69
Thailand 1990 46 510 –
1995 45 428 20 273 22 606 1 419 1 130 1 130 47
2000 34 187 17 754 12 439 2 953 1 041 1 041 59
2005 57 895 29 762 18 837 7 501 1 795 1 795 61
2010 67 128 33 450 20 927 10 135 0 1 885 1 111 2 996 731 62
2011 65 824 33 169 20 726 10 014 0 1 915 1 852 3 767 0 62
• 82 90 • 2012 60 304 30 998 17 537 8 852 1 887 904 2 791 1 030 64
Timor-Leste 2005 3 767 1 035 2 142 554 36 16 52 33
2010 –
2011 4 386 1 610 2 401 337 0 38 31 69 0 40
344 • 2012 3 828 1 545 1 823 420 0 40 9 49 46
a Rates are per 100 000 population.
b NEW AND RELAPSE includes cases for which the treatment history is unknown.

260 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($7UHDWPHQWRXWFRPHVQHZVPHDUSRVLWLYHFDVHV±
% OF COHORT
TREATMENT SUCCESS (%)
a
1995–2011 YEAR NUMBER
NOTIFIED
SIZE OF
COHORT
COHORT AS
% NOTIFIED CURED COMPLETED DIED FAILED DEFAULTED NOT
EVALUATED
Bangladesh 1995 20 524 10 867 53 66 5 5 2 10 12
2000 38 484 38 484 100 77 4 4 1 9 5
2005 84 848 84 848 100 9114122
2009 109 402 109 075 100 9114122
2010 105 772 105 659 100 9014122
• 71 92 • 2011 98 948 98 932 100 9114122
Bhutan 1995 367 433 118 78 20 0 0 1 1
2000 347 347 100 75 15 4 3 3 0
2005 308 340 110 8475310
2009 434 434 100 8663320
2010 457 454 99 8733312
• 97 91 • 2011 382 381 100 8833511
Democratic 1995 –
People's Republic 2000 16 440 14 571 89 73 9 3 7 5 3
of Korea 2005 17 796 17 796 100 8452422
2009 29 366 29 366 100 8552422
2010 31 240 31 240 100 8643421
• 0 90 • 2011 31 279 31 279 100 8733421
India 1995 264 515 264 722 100 1 25 0 0 0 75
2000 349 374 349 328 100 31 4 1 1 7 57
2005 508 890 507 204 100 83 2 5 2 7 1
2009 624 617 624 617 100 85 2 4 2 6 1
2010 630 165 630 165 100 8534261
• 25 88 • 2011 642 321 642 321 100 8534251
Indonesia 1995 31 768 3 018 10 73 18 2 0 6 1
2000 52 338 52 338 100 70 17 2 1 4 5
2005 158 640 158 640 100 83 82142
2009 169 213 169 213 100 84 7 2142
2010 183 366 183 366 100 84 7 2 1 4 3
• 91 90 • 2011 197 797 197 797 100 84 6 2 1 4 3
Maldives 1995 114 114 100 96 2 3000
2000 65 59 91 97 0 2002
2005 66 70 106 86 0 6 0 3 6
2009 45 45 100 47022444
2010 41 44 107 82 0 9 2 0 7
• 97 81 • 2011 47 48 102 81 0 2 0 0 17
Myanmar 1995 8 681 7 872 91 53 14 4 4 18 7
2000 17 254 16 792 97 7395292
2005 36 541 36 652 100 77 7 6 3 5 2
2009 41 357 41 811 101 77 86352
2010 42 318 42 200 100 77 8 5342
• 67 86 • 2011 42 324 42 310 100 77 9 5342
Nepal 1995 8 591 8 053 94 56 17 3 2 18 6
2000 13 683 12 992 95 79 55172
2005 14 617 14 617 100 87 1 5132
2009 15 442 15 468 100 87 3 4 1 3 2
2010 15 569 15 569 100 8823133
• 73 90 • 2011 15 000 15 000 100 8824132
Sri Lanka 1995 3 049 3 058 100 75 4 3 0 13 4
2000 4 314 4 314 100 75 4 4 1 15 2
2005 4 868 4 841 99 83 3 5 1 6 1
2009 4 764 4 754 100 83 3 6 2 4 3
2010 4 635 4 635 100 83 4 7 1 4 1
• 79 87 • 2011 4 490 4 490 100 83 3 5 1 5 2
Thailand 1995 20 273 20 273 100 36 28 2 0 9 24
2000 17 754 23 061 130 65382715
2005 29 762 29 919 101 7058279
2009 32 810 27 597 84 8157132
2010 33 450 30 317 91 7967232
• 64 85 • 2011 33 169 30 711 93 7967133
Timor-Leste 2005 1 035 1 035 100 61 21 5 1 11 2
2009 1 206 –
2010 1 530 – 80 8 4 1 4 4
91 • 2011 1 610 1 610 100 8653032
a TREATMENT SUCCESS = percent cured + percent completed then rounded to the nearest digit.

261 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
SOUTH-EAST ASIA REGION
7$%/($7UHDWPHQWRXWFRPHVUHWUHDWPHQWFDVHV±
% OF COHORT
TREATMENT SUCCESS (%)
a
1995–2011 YEAR NUMBER
NOTIFIED
SIZE OF
COHORT
COHORT AS
% NOTIFIED CURED COMPLETED DIED FAILED DEFAULTED NOT
EVALUATED
Bangladesh 1995 729 1 179 162 71 3 5 8 11 2
2000 1 763 1 815 103 70 2 4 2 7 14
2005 3 876 3 876 100 73 6 4 2 5 9
2009 4 099 6 637 162 66 16 6 2 5 6
2010 7 795 7 814 100 47 33 5 2 5 8
• 75 82 • 2011 7 366 7 369 100 46 36 5 2 4 7
Bhutan 1995 10 22 220 50 9 0 23 14 5
2000 36 –
2005 51 52 102 65 10 6 8 2 10
2009 76 76 100 70 12 8 7 3 1
2010 82 81 99 78 6 1 7 5 2
• 59 76 • 2011 70 67 96 70 6 7 12 1 3
Democratic 1995 –
People's Republic 2000 103 1 285 1 248 75 11 2 4 2 5
of Korea 2005 9 116 9 116 100 70 6 3 12 5 4
2009 14 576 14 576 100 74 9 2 11 2 2
2010 15 058 15 058 100 76 8 4 8 3 2
• 0 84 • 2011 13 507 13 507 100 77 8 5 7 2 1
India 1995 690 551 80 64 6 4 3 13 9
2000 98 065 48 133 49 55 15 7 5 16 2
2005 223 653 224 143 100 47 24 7 4 16 1
2009 289 756 289 756 100 45 29 7 4 13 1
2010 292 972 292 972 100 45 30 7 4 13 2
• 70 75 • 2011 304 431 304 431 100 43 31 7 4 12 3
Indonesia 1995 106 76 72 22 9 0 0 1 67
2000 1 448 2 530 175 50 22 3 3 7 15
2005 4 446 4 812 108 63 15 3 4 8 7
2009 5 688 5 687 100 53 20 4 3 12 8
2010 6 589 6 589 100 53 20 5 3 11 8
• 32 71 • 2011 7 707 7 707 100 53 18 5 3 11 9
Maldives 1995 10 –
2000 4 5 125 100 0
2005 5 5 100 80 20 0 0 0 0
2009 5 1 20 0 0 0 0 0 100
2010 3 0 0
• 0 0 • 2011 1 0 0
Myanmar 1995 1 837 1 443 79 55 8 4 4 19 9
2000 2 623 3 001 114 65 9 7 4 12 3
2005 5 597 6 556 117 58 14 10 6 7 5
2009 9 717 9 540 98 44 28 11 5 7 4
2010 10 269 10 106 98 41 32 11 5 7 3
• 64 72 • 2011 11 009 11 087 101 38 34 12 6 8 3
Nepal 1995 786 –
2000 1 807 2 047 113 73 3 4 8 7 4
2005 2 973 2 973 100 81 2 4 6 4 3
2009 3 117 3 063 98 82 3 6 3 4 3
2010 3 112 3 112 100 82 3 5 3 4 4
• 0 85 • 2011 2 882 2 882 100 83 2 5 4 3 3
Sri Lanka 1995 248 –
2000 649 521 80 44 20 6 1 26 3
2005 510 504 99 67552183
2009 409 408 100 66781135
2010 380 380 100 7167294
• 0 75 • 2011 395 395 100 6968395
Thailand 1995 1 130 –
2000 1 041 –
2005 1 795 2 285 127 52 6 12 5 7 18
2009 3 929 2 542 65 58 10 11 5 7 9
2010 2 996 2 580 86 55 11 12 5 7 10
• 0 69 • 2011 3 767 2 737 73 57 12 11 5 7 8
Timor-Leste 2005 52 56 108 9602020
2009 52 –
2010 56 – 7792472
77 • 2011 69 69 100 71 6 4 6 13
a TREATMENT SUCCESS = percent cured + percent completed then rounded to the nearest digit.

262 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($+,9WHVWLQJDQGSURYLVLRQRI&37$57DQG,37–
% OF TB PATIENTS WITH
KNOWN HIV STATUS
2005–2012
YEAR
% OF TB
PATIENTS WITH
KNOWN HIV
STATUS
NUMBER OF TB
PATIENTS WITH
KNOWN HIV
STATUS
PATIENTS
NOTIFIED
(NEW AND
RETREAT)
NUMBER OF
HIV-POSITIVE
TB PATIENTS
% OF TESTED
TB PATIENTS
HIV-POSITIVE
% OF HIV-
POSITIVE TB
PATIENTS ON
CPT
% OF HIV-
POSITIVE TB
PATIENTS ON
ART
NUMBER OF
HIV-POSITIVE
PEOPLE
PROVIDED IPT
Bangladesh 2005 0 0 123 118
2010 1.1 1 778 158 698 4 0.22 100 100 64
2011 1.2 1 900 159 023 53 2.8 100 100 0
• 0 1 • 2012 1.2 2 086 173 619 63 3 100 100 0
Bhutan 2005 0 0 1 018 1 0 0
2010 1 332
2011 1 250
• 0 – 2012 1 145
Democratic 2005 50 474
People's Republic 2010 0 0 96 298 0
of Korea 2011 0 0 99 071 0
– – 2012 99 399
India 2005 2.3 29 488 1 304 828 6 411 22
2010 32 480 752 1 522 147 41 476 8.6 90 57
2011 45 688 530 1 515 872 44 702 6.5 91 59
• 2 56 • 2012 56 821 807 1 467 585 44 063 5.4 92 59
Indonesia 2005 254 601
2010 0.91 2 751 302 861 1 106 40 63 29
2011 1.9 6 003 321 308 2 547 42 67 39
– 1 • 2012 0.81 2 676 331 424 754 28 18 29
Maldives 2005 123
2010 0 0 97 0 0
2011 6.8 6 88 0 0
– 1 • 2012 0.9 1 111 1 100 0 0 0
Myanmar 2005 2 2 109 107 991 611 29 50 31 0
2010 3.2 4 362 137 403 961 22 100 94 514
2011 3.1 4 496 143 140 900 20 100 80 361
• 2 13 • 2012 13 19 219 148 149 5 161 27 83
Nepal 2005 0 0 34 077
2010 0 0 35 609 0
2011 42 15 000 35 954 55 0.37 100 100
• 0 42 • 2012 42 15 057 35 635 217 1.4 100 100
Sri Lanka 2005 9 695 2 0 0
2010 10 1 015 10 095 13 1.3 100 54 3
2011 18 1 832 10 328 21 1.1 71 100 7
– 36 • 2012 36 3 379 9 343 23 0.68 22 48 8
Thailand 2005 57 895
2010 82 55 692 68 239 8 959 16 71 54
2011 74 49 770 67 676 7 326 15 75 59
– 72 • 2012 72 44 035 61 208 5 807 13 77 62
Timor-Leste 2005 0 0 3 783
2010
2011 6.2 276 4 417 4 1.4 100
• 0 20 • 2012 20 766 3 837 4 0.52 100

263 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
SOUTH-EAST ASIA REGION
7$%/($7HVWLQJIRU0'57%DQGQXPEHURIFRQILUPHGFDVHVRI0'57%±
NEW PULMONARY CASES PREVIOUSLY TREATED CASES
YEAR
ESTIMATED CASES
OF MDR-TB AMONG
NOTIFIED
NUMBER OF
BACT+VE
b
TESTED FOR
MDR-TB
% OF
BACT+VE
b
TESTED FOR
MDR-TB
ESTIMATED CASES
OF MDR-TB AMONG
NOTIFIED
NUMBER OF
NOTIFIED
TESTED FOR
MDR-TB
% OF NOT IFIED
TESTED FOR
MDR-TB
TOTAL
CONFIRMED
CASES OF
MDR-TB
a
ESTIMATED CASES
OF MDR-TB AMONG
NOTIFIED
Bangladesh 2005 ––
2010 339 – 339 4.3
2011 509 71 <0.1 761 10
2012 513 4 200 (3 100–5 200) 1 900 (920–3 300) 41 <0.1 2 300 (1 900–2 700) 557 7.0
Bhutan 2005 2 2 0.65 3 5.9
2010 17 108 24 30 37
2011 21 48 13 26 37
2012 16 25 (20–30) 12 (8.8–15) 52 12 13 (8.8–17) 2 2.5
Democratic 2005 ––
People's Republic 2010 ––
of Korea 2011 37 –430.32
2012 25 3 800 (3 000–4 600) 1 500 (1 100–1 900) – 2 300 (1 600–3 000) 31 0.22
India 2005 34 ––
2010 2967 ––
2011 4237 ––
2012 16588 64 000 (49 000–79 000) 21 000 (18 000–25 000) – 43 000 (32 000–54 000) –
Indonesia 2005 ––
2010 182 0 0 324 4.9
2011 383 5 <0.1 695 9.0
2012 428 6 900 (5 200–8 500) 5 800 (4 300–7 700) 2 <0.1 1 000 (690–1 500) 821 9.6
Maldives 2005 ––
2010 0 0 0 0 0
2011 0 0 0 0 0
2012 0 1.7 (1.3–2.1) 1.5 (1.1–1.9) 0 0 0.16 (0.11–0.21) 0 0
Myanmar 2005 ––
2010 192 ––
2011 690 ––
2012 778 6 000 (4 600–7 500) 4 900 (3 600–6 500) – 1 200 (790–1 600) –
Nepal 2005 ––
2010 229 126 0.81 193 6.2
2011 213 0 0 0 0
2012 354 990 (660–1 300) 570 (320–950) 188 1.2 420 (270–620) 640 24
Sri Lanka 2005 32 659 12 417 82
2010 11 839 18 378 99
2011 13 1080 24 408 100
2012 5 21 (0–43) 11 (0.28–61) 1069 23 9.6 (4.4–18) 238 55
Thailand 2005 ––
2010 ––
2011 510 ––
2012 492 1 800 (1 400–2 200) 800 (480–1 200) – 960 (780–1 200) –
Timor-Leste 2005 ––
2010 5 ––
2011 2 0 0 2 2.9
2012 3 82 (62–100) 74 (54–94) – 7.9 (5.4–10) 3 6.1
a TOTAL CONFIRMED CASES OF MDR-TB includes cases with unknown previous treatment history (i.e. not included under NEW CASES or PREVIOUSLY TREATED
CASES).
b BACT+VE = bacteriologically positive cases.

264 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($1HZVPHDUSRVLWLYHFDVHQRWLILFDWLRQE\DJHDQGVH[±
MALE FEMALE
YEAR 0–14 15–24 25–34 35–44 45–54 55–64 65+
UN-
KNOWN
0–14 15–24 25–34 35–44 45–54 55–64 65+
UN-
KNOWN
MALE:FEMALE
RATIO
Bangladesh 1995 29 505 983 1 001 748 648 424 64 309 546 360 236 132 38 2.6
2000 256 3 640 5 643 5 750 4 718 3 667 2 837 495 3 029 3 238 2 247 1 315 778 370 2.3
2005 524 8 170 10 443 11 423 11 038 8 476 7 453 751 6 776 6 785 5 538 3 960 2 281 1 230 2.1
2010 365 10 460 12 535 11 409 12 758 11 176 11 536 0 653 9 221 8 279 6 185 5 458 3 484 2 250 0 2.0
2011 309 9 606 11 616 10 152 11 728 10 746 11 301 0 623 8 849 7 679 5 683 4 946 3 457 2 253 0 2.0
2012 316 9 479 12 021 10 837 12 744 11 843 12 236 0 650 9 355 8 175 6 342 6 044 4 043 2 705 0 1.9
Bhutan 1995 2 42 65 36 35 24 11 12 43 44 25 12 9 8 1.4
2000 6 65 41 30 24 12 2 7 57 34 31 23 3 2 1.1
2005 1 47 58 26 23 14 12 9 45 38 13 11 9 2 1.4
2010 108 50 25 12 26 13 0 17 104 45 18 18 10 9 0 1.1
2011 2 88 39 26 14 20 19 2 92 40 19 12 4 5 1.2
2012 6 82 56 30 11 17 11 6 92 58 14 18 9 10 1.0
Democratic 1995 –
People's Republic 2000 293 928 1 508 2 927 2 519 1 167 651 167 683 1 121 2 004 1 524 591 357 1.6
of Korea 2005 167 1 409 2 422 2 688 2 040 1 185 485 166 1 127 1 756 1 890 1 381 764 336 1.4
2010 447 2 524 4 046 4 849 4 061 2 629 1 153 407 1 493 2 461 2 910 2 276 1 347 637 1.7
2011 314 2 218 4 066 5 493 4 542 2 474 1 024 227 1 390 2 264 3 093 2 409 1 271 494 1.8
2012 293 2 439 4 015 5 055 4 373 2 699 1 150 227 1 447 2 475 3 005 2 623 1 527 576 1.7
India 1995 16 334 391 287 216 123 68 32 179 169 80 49 30 11 2.6
2000 1 588 20 963 31 090 30 829 24 230 15 308 8 534 2 250 14 495 17 287 11 768 7 516 4 594 2 697 2.2
2005 3 185 62 620 74 678 76 870 64 843 43 038 24 726 6 292 45 136 45 629 28 577 17 042 10 513 5 408 2.2
2010 4 871 78 278 82 757 90 440 81 210 60 766 38 442 8 544 53 415 49 425 34 035 22 719 15 527 9 735 2.3
2011 4 649 78 096 82 762 89 706 82 921 63 625 42 443 8 336 53 958 49 227 34 698 23 977 17 182 10 731 2.2
2012 4 697 75 502 79 594 88 111 82 356 63 814 41 322 8 260 53 975 47 511 33 378 23 267 17 300 10 502 2.2
Indonesia 1995 6 203 297 306 302 228 109 16 160 244 282 192 90 33 1.4
2000 –
2005 846 15 215 20 906 18 401 17 847 13 509 6 390 946 13 916 16 393 13 022 10 927 7 539 2 783 1.4
2010 714 16 501 24 645 21 090 20 977 17 329 7 910 0 816 14 800 17 838 14 629 13 142 9 524 3 451 0 1.5
2011 787 17 406 25 429 22 353 22 885 19 404 9 089 927 15 840 18 703 15 900 14 533 10 556 3 985 1.5
2012 824 17 304 25 460 23 057 23 751 20 204 9 554 879 15 875 18 484 16 146 15 215 11 321 4 245 1.5
Maldives 1995 1 28 11 10 8 10 6 1 13 8 4 6 6 2 1.8
2000 09102553 01145452 1.1
2005 0985665 11071224 1.4
2010 08604560 12341010 2.4
2011 012738130 04312120 2.6
2012 08624540 07633220 1.3
Myanmar 1995 42 713 1 423 1 401 977 677 298 58 535 729 729 450 343 154 1.8
2000 88 1 459 2 636 2 781 2 161 1 235 836 72 1 040 1 592 1 397 987 592 378 1.8
2005 132 3 401 5 877 5 888 4 585 2 557 1 764 147 2 376 3 047 2 563 2 101 1 218 885 2.0
2010 106 3 043 6 578 6 688 5 607 3 632 2 308 196 2 452 3 454 2 752 2 525 1 838 1 139 1.9
2011 120 2 923 6 182 6 319 5 680 3 954 2 500 187 2 401 3 317 2 760 2 554 2 010 1 407 1.9
2012 146 2 898 6 263 6 469 5 837 3 945 2 626 0 192 2 357 3 368 2 721 2 600 2 023 1 464 0 1.9
Nepal 1995 –
2000 170 1 904 1 763 1 713 1 491 1 294 772 176 1 267 1 078 833 575 419 228 2.0
2005 148 1 946 1 685 1 722 1 806 1 759 820 195 1 208 1 111 797 658 532 230 2.1
2010 165 2 110 1 832 1 724 1 856 1 857 1 126 0 192 1 177 1 036 819 681 642 352 0 2.2
2011 245 1 914 1 755 1 723 1 732 1 710 1 180 0 247 1 182 978 752 624 604 354 0 2.2
2012 250 1 906 1 756 1 644 1 708 1 773 1 203 210 1 227 1 036 666 638 643 397 2.1
Sri Lanka 1995 10 163 361 519 521 365 261 15 207 206 142 122 81 56 2.7
2000 25 266 459 695 793 484 360 23 312 264 176 202 144 113 2.5
2005 9 341 520 724 918 657 424 19 295 261 189 200 154 130 2.9
2010 14 268 539 602 884 683 448 15 255 233 171 183 186 154 2.9
2011 12 246 459 585 828 653 479 0 13 270 217 191 192 191 154 0 2.7
2012 7 243 420 504 799 672 456 0 17 242 200 162 211 200 136 0 2.7
Thailand 1995 59 1 191 2 936 2 948 2 434 2 607 2 346 52 741 888 782 936 1 175 1 178 2.5
2000 27 859 2 570 2 380 2 117 1 908 2 213 32 624 1 035 780 873 1 016 1 321 2.1
2005 44 1 344 3 814 4 393 4 003 2 831 3 407 57 907 1 662 1 334 1 367 1 259 1 938 2.3
2010 55 1 506 3 695 5 253 5 042 3 625 4 189 82 1 087 1 930 1 749 1 467 1 494 2 276 2.3
2011 38 1 546 3 650 5 139 5 140 3 734 4 080 76 1 214 1 773 1 658 1 586 1 402 2 133 2.4
2012 35 1 444 3 277 4 705 4 867 3 780 3 863 82 995 1 491 1 613 1 424 1 364 2 058 2.4
Timor-Leste 2005 8 136 149 116 119 52 47 8 127 90 76 60 18 29 1.5
2010 –
2011 14 199 177 137 114 99 146 0 16 176 182 113 85 77 75 0 1.2
2012 7 196 172 128 119 114 129 0 12 154 143 120 75 84 92 0 1.3

265 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
SOUTH-EAST ASIA REGION
a LED = Light emitting diode microscopes
b DST = Drug susceptibility testing
c LPA = Line probe assay
d NRL = National Reference Laboratory
7$%/($/DERUDWRULHV173VHUYLFHVGUXJPDQDJHPHQWDQGLQIHFWLRQFRQWURO
LABORATORIES FREE THROUGH NTP
SMEAR LABS
PER 100K
POPULATION
% OF SMEAR
LABS USING
LED
a
CULTURE
LABS PER 5M
POPULATION
DST
b
LABS
PER 5M
POPULATION
LPA
c
LABS
PER 5M
POPULATION
NUMBER OF
LABS USING
XPERT MTB/RIF
SECOND-
LINE DST
AVAILABLE
NRL
d
TB DIAGNOSIS
FIRST-
LINE
DRUGS
TB NOTIF.
RATE PER
100 000
HEALTH-CARE
WORKERS
RIFAMPICIN
USED
THROUGHOUT
TREATMENT
Bangladesh 0.7 2 <0.1 <0.1 <0.1 12 Out of
country Yes Yes (all suspects) Yes Yes
Bhutan 4.7 0 6.7 6.7 Out of
country Yes Yes
(
if TB is
confirmed) Yes Yes
Democratic People's
Republic of Korea 1.3 0 0.2 0.2 0 0 Out of
country Yes Yes (all suspects) Yes Yes
India 1.1 2 0.3 0.2 0.1 32 Yes Yes (all suspects) Yes Yes
Indonesia 2.3 0 0.9 0.1 <0.1 9 In country Yes Yes (other criteria) Yes Yes
Maldives 20.7 0 14.8 0 0 0 Out of
country Yes Yes (all suspects) Yes Yes
Myanmar 0.9 14 0.2 0.2 0.2 3 In and out
of countr y Yes Yes (all suspects) Yes Yes
Nepal 1.9 2 0.4 0.4 9 In country Yes Yes (all suspects) Yes Yes
Sri Lanka 1.0 0 0.7 0.2 0.2 1 Out of
country Yes Yes (all suspects) Yes Yes 53
Thailand 1.6 6 4.9 1.3 0.9 14 In country Yes Yes (all suspects) Yes Yes
Timor-Leste 1.6 – 1 No Yes Yes (all suspects) Yes No

266 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($0HDVXUHGSHUFHQWDJHRI7%FDVHVZLWK0'57%
D
PRVWUHFHQW\HDUDYDLODEOH
New TB cases Previously treated TB cases
Year Source Coverage Percentage Year Source Coverage Percentage
Bangladesh 2011 Survey National 1.4 (0.70–2.5) 2011 Survey National 29 (24–34)
Bhutan
Democratic People's
Republic of Korea
India 2001, 2004, 2006, 2009 Survey Sub-national 2.2 (1.9–2.6) 2006, 2009 Survey Sub-national 15 (11–19)
Indonesia 2004, 2006, 2010 Survey Sub-national 1.9 (1.4–2.5) 2006, 2010 Survey Sub-national 12 (8.1–17)
Maldives
Myanmar 2008 Survey National 4.2 (3.1–5.6) 2008 Survey National 10 (6.9–14)
Nepal 2011 Survey National 2.3 (1.3–3.8) 2011 Survey National 15 (10–23)
Sri Lanka 2006 Survey National 0.18 (0–0.99) 2011 Surveillance National 2.2 (1.0–4.1)
Thailand 2006 Survey National 1.7 (1.0–2.6) 2006 Survey National 35 (28–42)
Timor-Leste
a Empty rows indicate an absence of high-quality survey or surveillance data. In the absence of high-quality national data, high-quality sub-national data are used.

9'56'402#%+(+%4')+10
Table A4.1 Estimates of the burden of disease caused by TB, 1990–2012 269
6CDNG# +PEKFGPEGPQVKßECVKQPCPFECUGFGVGEVKQPTCVGUCNNHQTOU¿
6CDNG# %CUGPQVKßECVKQPU¿
6CDNG# 6TGCVOGPVQWVEQOGUPGYUOGCTRQUKVKXGECUGU¿
6CDNG# 6TGCVOGPVQWVEQOGUTGVTGCVOGPVECUGU¿
6CDNG# *+8VGUVKPICPFRTQXKUKQPQH%26#46CPF+26¿
6CDNG# 6GUVKPIHQT/&46$CPFPWODGTQHEQPßTOGFECUGUQH/&46$¿
6CDNG# 0GYUOGCTRQUKVKXGECUGPQVKßECVKQPD[CIGCPFUGZ¿
6CDNG# .CDQTCVQTKGU062UGTXKEGUFTWIOCPCIGOGPVCPFKPHGEVKQPEQPVTQN
6CDNG# /GCUWTGFRGTEGPVCIGQH6$ECUGUYKVJ/&46$OQUVTGEGPV[GCTCXCKNCDNG

GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
Estimates of mortality, prevalence and incidence
Estimated values are shown as best estimates followed by lower and upper bounds. e lower and upper bounds are dened
as the 2.5th and 97.5th centiles of outcome distributions produced in simulations. See ANNEX 1 for further details.
Estimated numbers are shown rounded to two signicant gures. Estimated rates are shown rounded to three signicant
gures unless the value is under 100, in which case rates are shown rounded to two signicant gures.
Estimates for all years are recalculated as new information becomes available and techniques are rened, so
they may dier from those published in previous reports in this series. e main updates implemented in this
report are explained in Box 2.1 of Chapter 2. Estimates published in previous global TB control reports should no
longer be used.
Data source
Data shown in this annex are taken from the WHO global TB database on 1 October 2013. Data shown in the main part of
the report were taken from the database in July 2013. As a result, data in this annex may dier slightly from those in the
main part of the report.
Data for all years can be downloaded from www.who.int/tb/data.
269 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
WESTERN PACIFIC REGION
7$%/($(VWLPDWHVRIWKHEXUGHQRIGLVHDVHFDXVHGE\7%±
YEAR POPULATION
(MILLIONS)
NUMBER
(THOUSANDS) RATE
a
NUMBER
(THOUSANDS) RATE
a
NUMBER
(THOUSANDS) RATE
a
MORTALITY (EXCLUDING HIV) PREVALENCE (INCLUDING HIV) INCIDENCE (INCLUDING HIV)
American Samoa 1990 < 1 <0.01 (<0.01–<0.01) 5.1 (2.1–9.5) 0.022 (0.010–0.037) 46 (22–79) 0.012 (<0.01–0.015) 26 (21–31)
1995 < 1 <0.01 (<0.01–<0.01) 2.4 (0.95–4.4) 0.011 (<0.01–0.019) 21 (10–37) <0.01 (<0.01–<0.01) 12 (9.4–14)
2000 < 1 <0.01 (<0.01–<0.01) 0.82 (0.35–1.5) <0.01 (<0.01–0.010) 9.4 (3.6–18) <0.01 (<0.01–<0.01) 6.9 (5.6–8.4)
2005 < 1 <0.01 (<0.01–<0.01) 2.4 (0.99–4.5) 0.013 (<0.01–0.023) 23 (11–38) <0.01 (<0.01–<0.01) 13 (10–15)
2010 < 1 <0.01 (<0.01–<0.01) 0.9 (0.14–2.3) <0.01 (<0.01–0.014) 11 (3.0–25) <0.01 (<0.01–<0.01) 7.8 (6.3–9.4)
2011 < 1 <0.01 (<0.01–<0.01) 0.95 (0.17–2.4) <0.01 (<0.01–0.014) 12 (3.2–25) <0.01 (<0.01–<0.01) 7.8 (6.3–9.4)
2012 < 1 <0.01 (<0.01–<0.01) 0.88 (0.23–2.0) <0.01 (<0.01–0.012) 11 (3.6–22) <0.01 (<0.01–<0.01) 7.3 (5.9–8.9)
Australia 1990 17 0.061 (0.061–0.062) 0.36 (0.35–0.36) 1.7 (0.750–2.9) 9.7 (4.4–17) 1.2 (1.0–1.3) 6.8 (6.0–7.7)
1995 18 0.027 (0.027–0.028) 0.15 (0.15–0.16) 1.7 (0.740–3.0) 9.4 (4.1–17) 1.2 (1.1–1.4) 6.8 (6.0–7.7)
2000 19 0.036 (0.035–0.036) 0.19 (0.18–0.19) 1.7 (0.740–3.0) 8.7 (3.9–16) 1.2 (1.1–1.4) 6.2 (5.5–7.0)
2005 21 0.041 (0.041–0.042) 0.2 (0.20–0.20) 1.6 (0.650–3.0) 7.8 (3.2–14) 1.2 (1.1–1.4) 5.9 (5.1–6.6)
2010 22 0.051 (0.050–0.051) 0.23 (0.23–0.23) 2 (0.830–3.6) 8.8 (3.7–16) 1.4 (1.3–1.6) 6.5 (5.7–7.3)
2011 23 0.04 (0.040–0.041) 0.18 (0.18–0.18) 1.9 (0.740–3.5) 8.2 (3.3–15) 1.4 (1.2–1.6) 6.3 (5.5–7.1)
2012 23 0.045 (0.044–0.045) 0.19 (0.19–0.19) 2 (0.860–3.7) 8.8 (3.7–16) 1.5 (1.3–1.7) 6.5 (5.7–7.4)
Brunei 1990 < 1 <0.01 (<0.01–<0.01) 3 (2.9–3.2) 0.2 (0.070–0.400) 78 (27–154) 0.16 (0.140–0.190) 64 (56–72)
Darussalam 1995 < 1 <0.01 (<0.01–<0.01) 3 (2.9–3.2) 0.21 (0.064–0.440) 71 (22–150) 0.18 (0.160–0.210) 63 (55–71)
2000 < 1 0.014 (0.014–0.015) 4.3 (4.2–4.5) 0.55 (0.270–0.930) 165 (81–280) 0.35 (0.310–0.400) 106 (93–120)
2005 < 1 0.011 (0.010–0.011) 2.9 (2.8–3.0) 0.23 (0.080–0.470) 64 (22–128) 0.19 (0.160–0.210) 51 (45–58)
2010 < 1 0.012 (0.012–0.013) 3 (2.9–3.2) 0.4 (0.180–0.700) 99 (45–174) 0.27 (0.240–0.310) 68 (60–77)
2011 < 1 0.012 (0.012–0.013) 3 (2.9–3.2) 0.36 (0.140–0.660) 87 (36–162) 0.26 (0.230–0.300) 65 (57–74)
2012 < 1 0.013 (0.012–0.013) 3 (2.9–3.2) 0.37 (0.140–0.700) 90 (35–169) 0.28 (0.240–0.320) 68 (59–77)
Cambodia 1990 9 14 (4.9–28) 157 (54–314) 150 (96–220) 1 670 (1 060–2 410) 53 (38–69) 580 (423–761)
1995 11 15 (5.3–29) 139 (49–274) 180 (130–230) 1 670 (1 220–2 180) 62 (48–78) 578 (448–724)
2000 12 16 (5.7–31) 128 (47–251) 200 (160–240) 1 620 (1 310–1 960) 71 (56–87) 577 (458–710)
2005 13 13 (5.1–23) 94 (38–175) 160 (140–190) 1 230 (1 020–1 460) 68 (57–81) 510 (424–604)
2010 14 9.8 (4.5–17) 68 (31–120) 130 (110–150) 875 (737–1 020) 63 (54–72) 437 (376–503)
2011 15 9.5 (4.4–17) 65 (30–114) 120 (100–140) 817 (690–954) 62 (53–71) 424 (364–489)
2012 15 9.3 (4.3–16) 63 (29–110) 110 (96–130) 764 (645–892) 61 (52–70) 411 (353–474)
China 1990 1 165 220 (190–240) 19 (17–21) 2 500 (2 300–2 700) 215 (201–230) 1 800 (1 400–2 200) 153 (121–189)
1995 1 238 170 (140–200) 13 (11–16) 2 400 (2 200–2 700) 195 (176–216) 1 600 (1 300–1 900) 129 (106–154)
2000 1 280 110 (84–140) 8.7 (6.5–11) 2 200 (1 900–2 500) 170 (146–196) 1 400 (1 200–1 600) 109 (92–126)
2005 1 318 75 (72–77) 5.7 (5.5–5.9) 1 800 (1 600–2 100) 140 (121–160) 1 200 (1 100–1 400) 92 (80–105)
2010 1 360 52 (50–53) 3.8 (3.7–3.9) 1 500 (1 300–1 700) 108 (94–123) 1 100 (930–1 200) 78 (68–88)
2011 1 368 48 (46–50) 3.5 (3.4–3.6) 1 400 (1 200–1 600) 104 (91–119) 1 000 (900–1 200) 75 (66–85)
2012 1 377 44 (43–46) 3.2 (3.1–3.3) 1 400 (1 200–1 600) 99 (86–113) 1 000 (880–1 100) 73 (64–82)
China, Hong Kong 1990 6 0.37 (0.360–0.370) 6.3 (6.2–6.4) 9.8 (4.0–18) 169 (69–314) 7.5 (6.6–8.5) 129 (113–146)
SAR 1995 6 0.38 (0.380–0.380) 6.2 (6.1–6.2) 8.7 (3.1–17) 142 (50–280) 7.1 (6.3–8.1) 116 (102–132)
2000 7 0.27 (0.270–0.280) 4 (4.0–4.0) 8.2 (2.8–17) 120 (40–243) 6.9 (6.1–7.8) 101 (89–115)
2005 7 0.24 (0.240–0.250) 3.5 (3.5–3.6) 9 (3.8–16) 130 (55–237) 6.5 (5.7–7.4) 94 (83–107)
2010 7 0.19 (0.180–0.190) 2.6 (2.6–2.7) 7.7 (3.2–14) 110 (46–202) 5.7 (5.0–6.4) 81 (71–91)
2011 7 0.19 (0.180–0.190) 2.6 (2.6–2.7) 7.3 (3.0–14) 103 (42–191) 5.4 (4.8–6.2) 77 (67–87)
2012 7 0.19 (0.190–0.190) 2.6 (2.6–2.7) 7.7 (3.4–14) 108 (47–195) 5.5 (4.8–6.3) 77 (68–88)
China, Macao 1990 < 1 0.036 (0.018–0.060) 10 (5.1–17) 0.6 (0.290–1.0) 167 (81–285) 0.39 (0.350–0.450) 110 (96–124)
SAR 1995 < 1 0.022 (<0.01–0.050) 5.4 (1.3–12) 0.55 (0.180–1.1) 137 (45–278) 0.46 (0.410–0.520) 116 (102–131)
2000 < 1 0.02 (<0.01–0.052) 4.6 (0.74–12) 0.65 (0.250–1.2) 151 (57–289) 0.52 (0.450–0.580) 120 (105–135)
2005 < 1 0.015 (<0.01–0.051) 3.3 (0.16–11) 0.66 (0.300–1.2) 141 (64–249) 0.46 (0.400–0.520) 98 (86–111)
2010 < 1 0.015 (<0.01–0.058) 2.8 (<0.1–11) 0.64 (0.280–1.1) 119 (52–214) 0.45 (0.400–0.510) 85 (74–96)
2011 < 1 0.015 (<0.01–0.059) 2.8 (<0.1–11) 0.59 (0.240–1.1) 108 (44–200) 0.44 (0.380–0.490) 80 (70–91)
2012 < 1 0.015 (<0.01–0.059) 2.8 (<0.1–11) 0.65 (0.280–1.2) 117 (50–211) 0.46 (0.410–0.530) 83 (73–94)
Cook Islands 1990 < 1 <0.01 (<0.01–<0.01) 0.79 (0.73–0.85) <0.01 (<0.01–<0.01) 12 (3.4–25) 0 (0–0) 0 (0–0)
1995 < 1 <0.01 (<0.01–<0.01) 1.1 (0.63–1.7) <0.01 (<0.01–<0.01) 17 (5.0–35) <0.01 (<0.01–<0.01) 13 (11–14)
2000 < 1 <0.01 (<0.01–<0.01) 0.51 (0.26–0.84) <0.01 (<0.01–<0.01) 7.6 (2.3–16) <0.01 (<0.01–<0.01) 6.5 (5.7–7.3)
2005 < 1 <0.01 (<0.01–<0.01) 0.62 (0.34–0.98) <0.01 (<0.01–<0.01) 7.5 (2.9–14) <0.01 (<0.01–<0.01) 5.9 (5.2–6.7)
2010 < 1 <0.01 (<0.01–<0.01) 0.4 (0.34–0.46) <0.01 (<0.01–<0.01) 6 (1.8–13) 0 (0–0) 0 (0–0)
2011 < 1 <0.01 (0–<0.01) 0.53 (<0.1–1.9) <0.01 (<0.01–<0.01) 7.4 (1.1–20) <0.01 (<0.01–<0.01) 5.6 (4.9–6.4)
2012 < 1 <0.01 (<0.01–<0.01) 0.6 (0.33–0.97) <0.01 (<0.01–<0.01) 7.2 (2.9–14) <0.01 (<0.01–<0.01) 5.6 (4.9–6.3)
Fiji 1990 < 1 0.051 (0.020–0.097) 7 (2.7–13) 1.8 (0.890–3.0) 244 (123–407) 0.81 (0.710–0.920) 112 (98–126)
1995 < 1 0.039 (0.018–0.069) 5.1 (2.3–8.9) 1.3 (0.650–2.1) 165 (84–273) 0.6 (0.530–0.680) 77 (68–87)
2000 < 1 0.03 (0.021–0.040) 3.7 (2.6–4.9) 0.91 (0.470–1.5) 112 (58–184) 0.44 (0.390–0.500) 54 (48–62)
2005 < 1 0.022 (0.020–0.024) 2.7 (2.4–2.9) 0.66 (0.340–1.1) 80 (42–131) 0.33 (0.290–0.370) 40 (35–45)
2010 < 1 0.016 (0.016–0.017) 1.9 (1.9–2.0) 0.39 (0.200–0.640) 45 (23–75) 0.24 (0.210–0.270) 28 (24–32)
2011 < 1 0.015 (0.015–0.016) 1.8 (1.7–1.8) 0.32 (0.140–0.570) 37 (16–66) 0.23 (0.200–0.260) 26 (23–29)
2012 < 1 0.015 (0.014–0.015) 1.7 (1.6–1.7) 0.26 (0.088–0.530) 30 (10–61) 0.21 (0.190–0.240) 24 (21–27)
French Polynesia 1990 < 1 <0.01 (<0.01–0.016) 1.9 (<0.1–7.8) 0.095 (0.042–0.170) 48 (21–85) 0.068 (0.059–0.077) 34 (30–39)
1995 < 1 <0.01 (<0.01–0.015) 2.3 (0.19–6.8) 0.13 (0.048–0.240) 59 (22–113) 0.1 (0.088–0.110) 47 (41–53)
2000 < 1 <0.01 (<0.01–<0.01) 1.2 (0.36–2.7) 0.083 (0.025–0.180) 35 (10–74) 0.071 (0.062–0.081) 30 (26–34)
2005 < 1 <0.01 (<0.01–0.015) 1.6 (<0.1–6.1) 0.099 (0.043–0.180) 39 (17–70) 0.072 (0.063–0.082) 28 (25–32)
2010 < 1 <0.01 (<0.01–<0.01) 0.78 (0.25–1.6) 0.059 (0.018–0.120) 22 (6.6–46) 0.047 (0.041–0.053) 18 (15–20)
2011 < 1 <0.01 (<0.01–0.022) 1.7 (0–8.0) 0.11 (0.052–0.190) 41 (19–70) 0.074 (0.064–0.083) 27 (24–31)
2012 < 1 <0.01 (<0.01–<0.01) 0.98 (0.12–2.7) 0.071 (0.026–0.140) 26 (9.4–51) 0.058 (0.050–0.065) 21 (18–24)
Guam 1990 < 1 <0.01 (<0.01–0.012) 2.7 (<0.1–9.5) 0.088 (0.037–0.160) 67 (28–124) 0.066 (0.058–0.075) 50 (44–57)
1995 < 1 <0.01 (<0.01–0.023) 3.9 (<0.1–16) 0.14 (0.062–0.250) 96 (43–170) 0.099 (0.087–0.110) 68 (60–77)
2000 < 1 <0.01 (<0.01–<0.01) 1.9 (0.22–5.3) 0.077 (0.028–0.150) 49 (18–96) 0.062 (0.054–0.070) 40 (35–45)
2005 < 1 <0.01 (<0.01–0.022) 2.9 (0–14) 0.12 (0.059–0.200) 74 (37–124) 0.072 (0.063–0.082) 46 (40–52)
2010 < 1 <0.01 (<0.01–0.036) 4.6 (<0.1–23) 0.19 (0.095–0.310) 118 (59–196) 0.12 (0.100–0.130) 73 (64–82)
2011 < 1 <0.01 (<0.01–0.012) 2.7 (0.33–7.6) 0.13 (0.049–0.240) 78 (31–148) 0.094 (0.083–0.110) 59 (51–66)
2012 < 1 <0.01 (<0.01–<0.01) 2.2 (0.68–4.5) 0.11 (0.036–0.220) 66 (22–134) 0.078 (0.069–0.089) 48 (42–54)
Japan 1990 122 3.8 (3.7–3.9) 3.1 (3.0–3.2) 83 (35–150) 68 (29–123) 60 (52–67) 49 (43–55)
1995 124 3.3 (3.2–3.3) 2.6 (2.6–2.7) 66 (26–120) 53 (21–99) 50 (43–56) 40 (35–45)
2000 126 2.8 (2.7–2.8) 2.2 (2.2–2.2) 64 (28–110) 51 (23–91) 45 (40–51) 36 (32–41)
2005 127 2.3 (2.3–2.4) 1.8 (1.8–1.9) 43 (18–79) 34 (14–62) 31 (27–35) 25 (22–28)
2010 127 2.2 (2.1–2.3) 1.7 (1.7–1.8) 37 (16–66) 29 (12–52) 26 (23–30) 20 (18–23)
2011 127 2.3 (2.2–2.3) 1.8 (1.7–1.8) 35 (15–64) 28 (12–50) 25 (22–29) 20 (18–23)
2012 127 2.1 (2.0–2.2) 1.7 (1.6–1.7) 33 (13–61) 26 (11–48) 24 (21–28) 19 (17–22)
Kiribati 1990 < 1 0.039 (0.029–0.051) 55 (41–72) 0.18 (0.080–0.310) 249 (113–437) 0.083 (0.066–0.100) 116 (93–143)
1995 < 1 0.044 (0.031–0.058) 57 (41–76) 0.59 (0.260–1.0) 770 (347–1 360) 0.39 (0.310–0.460) 505 (410–609)
2000 < 1 0.013 (<0.01–0.016) 15 (11–20) 0.4 (0.140–0.790) 487 (174–957) 0.31 (0.250–0.380) 372 (296–456)
2005 < 1 0.015 (0.015–0.016) 17 (16–17) 0.68 (0.300–1.2) 747 (335–1 320) 0.44 (0.360–0.530) 488 (396–588)
2010 < 1 0.016 (<0.01–0.025) 17 (9.5–26) 0.47 (0.160–0.930) 477 (166–949) 0.36 (0.290–0.430) 366 (298–441)
2011 < 1 0.017 (<0.01–0.026) 17 (9.5–26) 0.66 (0.300–1.2) 664 (298–1 170) 0.43 (0.350–0.520) 432 (351–521)
2012 < 1 0.017 (<0.01–0.026) 17 (9.5–26) 0.63 (0.270–1.1) 628 (270–1 130) 0.43 (0.350–0.520) 429 (349–517)
Lao People's 1990 4 1.7 (1.1–2.6) 41 (25–60) 63 (32–110) 1 490 (746–2 490) 21 (13–31) 492 (304–725)
Democratic 1995 5 1.4 (0.860–2.0) 29 (18–42) 60 (32–95) 1 220 (664–1 950) 20 (12–29) 403 (249–593)
Republic 2000 5 1.1 (0.700–1.7) 21 (13–31) 52 (30–79) 961 (557–1 470) 18 (11–26) 330 (204–486)
2005 6 0.91 (0.560–1.3) 16 (9.7–23) 43 (26–63) 737 (453–1 090) 16 (9.7–23) 270 (167–398)
2010 6 0.76 (0.470–1.1) 12 (7.3–17) 36 (23–52) 565 (366–807) 14 (8.8–21) 221 (137–326)
2011 7 0.73 (0.450–1.1) 11 (6.9–17) 35 (23–50) 540 (353–767) 14 (8.6–20) 213 (131–313)
2012 7 0.72 (0.430–1.1) 11 (6.5–16) 34 (22–48) 514 (335–729) 14 (8.4–20) 204 (126–301)
Malaysia 1990 18 1.2 (0.370–2.5) 6.6 (2.0–14) 44 (23–71) 242 (128–392) 23 (21–26) 127 (113–142)
1995 21 1.4 (0.480–2.7) 6.6 (2.3–13) 39 (21–63) 189 (102–303) 22 (20–25) 108 (97–120)
2000 23 1.6 (0.710–2.9) 6.9 (3.0–12) 35 (18–56) 148 (79–239) 22 (20–24) 95 (86–103)
2005 26 1.5 (0.810–2.4) 5.8 (3.2–9.1) 33 (18–54) 129 (68–210) 22 (20–24) 86 (79–94)
2010 28 1.5 (1.0–2.2) 5.4 (3.6–7.6) 32 (15–54) 112 (54–190) 23 (21–25) 82 (75–89)
2011 29 1.6 (1.1–2.1) 5.4 (3.8–7.3) 31 (14–54) 107 (49–186) 23 (21–25) 81 (74–88)
2012 29 1.6 (1.2–2.1) 5.4 (4.1–7.0) 29 (13–53) 101 (43–183) 24 (22–26) 80 (74–87)
Marshall Islands 1990 < 1 0.013 (<0.01–0.062) 28 (<0.1–130) 0.12 (<0.01–0.470) 251 (3.3–1 000) 0.065 (<0.01–0.190) 137 (14–396)
1995 < 1 0.018 (<0.01–0.069) 35 (0.72–134) 0.17 (<0.01–0.550) 332 (19–1 080) 0.097 (0.024–0.220) 190 (46–432)
2000 < 1 0.033 (<0.01–0.070) 62 (18–135) 0.28 (0.099–0.540) 532 (190–1 040) 0.14 (0.084–0.200) 263 (161–389)
2005 < 1 0.033 (<0.01–0.130) 64 (1.3–245) 0.34 (0.027–1.0) 651 (53–1 980) 0.19 (0.050–0.420) 363 (96–803)
2010 < 1 0.047 (<0.01–0.200) 89 (0.54–385) 0.47 (0.019–1.6) 903 (36–3 100) 0.26 (0.051–0.650) 502 (97–1 230)
2011 < 1 0.051 (<0.01–0.220) 98 (0.78–414) 0.51 (0.022–1.7) 973 (43–3 290) 0.28 (0.054–0.690) 536 (103–1 320)
2012 < 1 0.058 (<0.01–0.240) 111 (1.4–448) 0.57 (0.028–1.9) 1 080 (54–3 560) 0.3 (0.058–0.740) 572 (110–1 400)
a
Rates are per 100 000 population.

270 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($(VWLPDWHVRIWKHEXUGHQRIGLVHDVHFDXVHGE\7%±
YEAR POPULATION
(MILLIONS)
NUMBER
(THOUSANDS) RATE
a
NUMBER
(THOUSANDS) RATE
a
NUMBER
(THOUSANDS) RATE
a
MORTALITY (EXCLUDING HIV) PREVALENCE (INCLUDING HIV) INCIDENCE (INCLUDING HIV)
Micronesia 1990 < 1 0.035 (0–0.220) 36 (0–227) 0.45 (<0.01–1.9) 464 (2.8–2 010) 0.36 (0.100–0.800) 379 (104–827)
(Federated 1995 < 1 0.077 (0.019–0.180) 72 (18–163) 0.68 (0.220–1.4) 629 (203–1 290) 0.35 (0.200–0.540) 325 (185–505)
States of) 2000 < 1 0.07 (0.024–0.140) 65 (22–130) 0.6 (0.250–1.1) 560 (237–1 020) 0.3 (0.210–0.400) 279 (200–371)
2005 < 1 0.053 (0.016–0.110) 50 (15–104) 0.47 (0.180–0.900) 446 (172–848) 0.25 (0.170–0.360) 240 (158–338)
2010 < 1 0.032 (<0.01–0.100) 31 (1.8–100) 0.33 (0.045–0.870) 314 (44–844) 0.21 (0.092–0.380) 206 (89–371)
2011 < 1 0.028 (<0.01–0.098) 27 (1.1–95) 0.32 (0.046–0.870) 313 (44–837) 0.21 (0.089–0.370) 200 (86–360)
2012 < 1 0.025 (<0.01–0.093) 24 (0.62–90) 0.28 (0.028–0.810) 270 (27–782) 0.2 (0.086–0.360) 194 (83–349)
Mongolia 1990 2 0.52 (0.400–0.650) 24 (18–30) 20 (9.3–36) 938 (425–1 650) 8.8 (7.5–10) 405 (345–470)
1995 2 0.42 (0.300–0.560) 18 (13–24) 14 (7.1–24) 625 (308–1 050) 7.2 (6.3–8.2) 314 (274–356)
2000 2 0.32 (0.190–0.470) 13 (8.1–20) 10 (5.3–17) 431 (221–710) 6.1 (5.5–6.7) 254 (228–281)
2005 3 0.24 (0.120–0.400) 9.4 (4.6–16) 8.5 (4.1–14) 335 (162–569) 5.7 (5.2–6.1) 225 (207–243)
2010 3 0.2 (0.079–0.390) 7.5 (2.9–14) 9.6 (4.9–16) 353 (181–580) 6.1 (5.7–6.5) 224 (209–240)
2011 3 0.2 (0.077–0.390) 7.4 (2.8–14) 10 (5.3–16) 364 (191–591) 6.1 (5.7–6.6) 223 (208–239)
2012 3 0.2 (0.075–0.390) 7.2 (2.7–14) 11 (5.7–17) 380 (204–608) 6.2 (5.8–6.7) 223 (208–239)
Nauru 1990 < 1 <0.01 (<0.01–<0.01) 9.1 (5.0–14) 0.01 (<0.01–0.019) 111 (43–213) <0.01 (<0.01–<0.01) 88 (77–99)
1995 < 1 <0.01 (<0.01–<0.01) 4.7 (2.5–7.5) <0.01 (<0.01–<0.01) 54 (22–99) <0.01 (<0.01–<0.01) 40 (35–46)
2000 < 1 <0.01 (<0.01–<0.01) 7.2 (3.5–12) <0.01 (<0.01–0.012) 72 (35–122) <0.01 (<0.01–<0.01) 46 (40–52)
2005 < 1 <0.01 (<0.01–<0.01) 23 (10–41) 0.022 (0.011–0.036) 216 (109–359) 0.013 (0.011–0.014) 125 (110–142)
2010 < 1 <0.01 (<0.01–<0.01) 3.7 (2.3–5.4) <0.01 (<0.01–0.012) 55 (17–116) <0.01 (<0.01–<0.01) 34 (30–39)
2011 < 1 <0.01 (<0.01–<0.01) 8.1 (4.1–14) <0.01 (<0.01–0.015) 86 (41–147) <0.01 (<0.01–<0.01) 57 (50–65)
2012 < 1 <0.01 (<0.01–<0.01) 9.5 (4.4–17) <0.01 (<0.01–0.015) 91 (46–151) <0.01 (<0.01–<0.01) 54 (47–61)
New Caledonia 1990 < 1 <0.01 (<0.01–0.024) 4.7 (0.40–14) 0.21 (0.079–0.400) 123 (47–236) 0.16 (0.140–0.190) 98 (85–110)
1995 < 1 <0.01 (<0.01–<0.01) 2.1 (0.56–4.6) 0.11 (0.033–0.240) 59 (18–125) 0.1 (0.088–0.110) 53 (46–60)
2000 < 1 <0.01 (<0.01–0.034) 3.3 (0–16) 0.17 (0.080–0.280) 79 (38–134) 0.11 (0.095–0.120) 51 (45–58)
2005 < 1 <0.01 (<0.01–<0.01) 1.1 (0.13–3.1) 0.067 (0.024–0.130) 29 (11–57) 0.054 (0.047–0.061) 24 (21–27)
2010 < 1 <0.01 (<0.01–0.011) 1.2 (<0.1–4.5) 0.076 (0.032–0.140) 31 (13–56) 0.056 (0.049–0.064) 23 (20–26)
2011 < 1 <0.01 (<0.01–0.014) 1.3 (<0.1–5.5) 0.084 (0.037–0.150) 33 (15–60) 0.06 (0.052–0.068) 24 (21–27)
2012 < 1 <0.01 (<0.01–<0.01) 0.74 (0.23–1.6) 0.053 (0.016–0.110) 21 (6.2–44) 0.044 (0.038–0.049) 17 (15–20)
New Zealand 1990 3 0.019 (0.018–0.019) 0.55 (0.54–0.55) 0.58 (0.270–1.0) 17 (7.9–30) 0.4 (0.350–0.450) 12 (10–13)
1995 4 0.021 (0.021–0.021) 0.58 (0.57–0.58) 0.67 (0.320–1.2) 18 (8.7–31) 0.45 (0.390–0.510) 12 (11–14)
2000 4 0.012 (0.012–0.012) 0.32 (0.31–0.32) 0.49 (0.170–0.980) 13 (4.5–25) 0.4 (0.350–0.450) 10 (9.0–12)
2005 4 <0.01 (<0.01–<0.01) 0.15 (0.15–0.15) 0.51 (0.200–0.950) 12 (4.9–23) 0.38 (0.330–0.430) 9.2 (8.1–10)
2010 4 <0.01 (<0.01–<0.01) 0.12 (0.12–0.12) 0.48 (0.210–0.870) 11 (4.8–20) 0.35 (0.300–0.390) 7.9 (6.9–9.0)
2011 4 <0.01 (<0.01–<0.01) 0.1 (0.10–0.10) 0.5 (0.220–0.880) 11 (5.1–20) 0.35 (0.310–0.400) 7.9 (7.0–9.0)
2012 4 <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 0.47 (0.200–0.840) 10 (4.5–19) 0.34 (0.300–0.380) 7.6 (6.6–8.6)
Niue 1990 < 1 <0.01 (<0.01–<0.01) 2.9 (2.8–3.0) <0.01 (<0.01–<0.01) 43 (13–91) 0 (0–0) 0 (0–0)
1995 < 1 <0.01 (<0.01–<0.01) 3 (3.0–3.1) <0.01 (<0.01–<0.01) 45 (13–96) 0 (0–0) 0 (0–0)
2000 < 1 <0.01 (<0.01–<0.01) 3.1 (3.1–3.2) <0.01 (<0.01–<0.01) 47 (14–99) 0 (0–0) 0 (0–0)
2005 < 1 <0.01 (<0.01–<0.01) 1.7 (1.7–1.8) <0.01 (<0.01–<0.01) 26 (7.6–54) 0 (0–0) 0 (0–0)
2010 < 1 <0.01 (<0.01–<0.01) 1.4 (1.3–1.4) <0.01 (<0.01–<0.01) 20 (6.1–43) 0 (0–0) 0 (0–0)
2011 < 1 <0.01 (<0.01–<0.01) 19 (4.7–42) <0.01 (<0.01–<0.01) 170 (59–341) <0.01 (<0.01–<0.01) 81 (71–91)
2012 < 1 <0.01 (<0.01–<0.01) 3.1 (1.7–4.9) <0.01 (<0.01–<0.01) 46 (14–97) <0.01 (<0.01–<0.01) 37 (32–42)
Northern Mariana 1990 < 1 <0.01 (<0.01–<0.01) 3.1 (0.91–6.5) 0.038 (0.011–0.081) 86 (26–183) 0.032 (0.028–0.036) 73 (64–83)
Islands 1995 < 1 <0.01 (<0.01–<0.01) 4.4 (1.5–8.8) 0.071 (0.021–0.150) 123 (36–261) 0.055 (0.048–0.062) 96 (84–109)
2000 < 1 <0.01 (<0.01–0.018) 6.9 (0.13–26) 0.12 (0.050–0.210) 172 (74–312) 0.086 (0.076–0.098) 126 (110–143)
2005 < 1 <0.01 (<0.01–0.019) 6.3 (<0.1–30) 0.1 (0.052–0.180) 163 (81–273) 0.066 (0.057–0.074) 102 (89–115)
2010 < 1 <0.01 (<0.01–<0.01) 3.2 (0.37–8.9) 0.049 (0.019–0.093) 91 (35–172) 0.037 (0.032–0.042) 68 (60–77)
2011 < 1 <0.01 (<0.01–<0.01) 3.6 (0.15–13) 0.054 (0.023–0.097) 101 (43–182) 0.038 (0.033–0.043) 71 (62–81)
2012 < 1 <0.01 (<0.01–<0.01) 3.5 (0.17–12) 0.052 (0.022–0.094) 97 (42–175) 0.037 (0.032–0.042) 69 (60–78)
Palau 1990 < 1 <0.01 (<0.01–<0.01) 3.4 (1.0–7.1) <0.01 (<0.01–0.017) 50 (12–112) <0.01 (<0.01–<0.01) 45 (36–54)
1995 < 1 <0.01 (<0.01–<0.01) 17 (7.3–31) 0.034 (0.013–0.065) 197 (76–376) 0.025 (0.021–0.031) 147 (119–178)
2000 < 1 <0.01 (<0.01–<0.01) 26 (11–48) 0.049 (0.023–0.085) 256 (119–444) 0.03 (0.024–0.036) 156 (127–189)
2005 < 1 <0.01 (<0.01–<0.01) 10 (3.8–20) 0.022 (<0.01–0.039) 110 (48–198) 0.013 (0.011–0.016) 67 (54–81)
2010 < 1 <0.01 (<0.01–<0.01) 23 (9.0–43) 0.045 (0.022–0.076) 221 (108–372) 0.024 (0.019–0.029) 116 (94–140)
2011 < 1 <0.01 (<0.01–<0.01) 8.8 (3.8–16) 0.021 (<0.01–0.039) 100 (40–187) 0.015 (0.012–0.018) 73 (59–88)
2012 < 1 <0.01 (<0.01–<0.01) 4.4 (2.9–6.2) 0.014 (<0.01–0.029) 65 (19–138) <0.01 (<0.01–<0.01) 24 (20–29)
Papua New 1990 4 3.4 (1.2–6.9) 82 (28–165) 30 (12–55) 715 (289–1 330) 13 (8.5–18) 308 (203–435)
Guinea 1995 5 3 (1.0–5.9) 63 (22–125) 29 (12–54) 620 (250–1 160) 15 (10–21) 322 (212–453)
2000 5 2.8 (0.910–5.8) 52 (17–107) 32 (12–61) 586 (219–1 130) 19 (12–26) 349 (230–492)
2005 6 3.4 (1.1–6.9) 55 (18–112) 37 (14–71) 607 (230–1 160) 22 (14–31) 358 (236–505)
2010 7 3.7 (1.2–7.5) 54 (18–110) 39 (14–76) 568 (208–1 100) 24 (16–34) 348 (229–491)
2011 7 3.7 (1.2–7.6) 53 (17–109) 38 (14–76) 549 (194–1 080) 24 (16–34) 346 (228–488)
2012 7 3.9 (1.3–7.8) 54 (18–109) 39 (13–77) 541 (187–1 080) 25 (16–35) 348 (230–490)
Philippines 1990 62 34 (26–44) 55 (42–70) 620 (480–790) 1 000 (768–1 270) 240 (150–360) 393 (243–580)
1995 70 35 (30–40) 50 (43–58) 630 (480–800) 904 (692–1 140) 250 (200–300) 360 (294–432)
2000 78 31 (29–34) 40 (38–43) 600 (480–740) 775 (616–953) 260 (210–310) 329 (269–395)
2005 86 30 (28–32) 35 (32–37) 540 (470–630) 633 (544–729) 260 (210–310) 301 (246–361)
2010 93 25 (24–27) 27 (25–29) 470 (410–530) 502 (441–566) 260 (210–310) 275 (227–328)
2011 95 24 (23–26) 26 (24–28) 460 (400–520) 484 (425–546) 260 (210–310) 270 (223–322)
2012 97 23 (22–25) 24 (22–26) 450 (390–500) 461 (405–520) 260 (210–310) 265 (219–316)
Republic of Korea 1990 43 3.7 (0.170–13) 8.7 (0.40–29) 96 (78–110) 223 (182–267) 73 (64–83) 171 (150–194)
1995 45 2.7 (0.044–10) 5.9 (0.10–23) 90 (74–110) 202 (166–243) 48 (42–55) 108 (95–123)
2000 46 1.2 (0.460–2.4) 2.7 (1.0–5.2) 85 (69–100) 184 (150–221) 25 (22–28) 54 (48–62)
2005 47 2.7 (0.040–11) 5.8 (<0.1–23) 79 (64–94) 167 (136–201) 49 (43–56) 105 (92–119)
2010 48 2.5 (0.190–7.5) 5.1 (0.40–16) 73 (60–88) 152 (124–182) 51 (44–57) 105 (92–118)
2011 49 2.7 (0.120–9.1) 5.6 (0.25–19) 72 (59–87) 149 (121–179) 53 (47–60) 109 (96–124)
2012 49 2.6 (0.160–8.5) 5.4 (0.32–17) 71 (58–86) 146 (119–175) 53 (46–60) 108 (95–122)
Samoa 1990 < 1 <0.01 (<0.01–0.015) 5 (2.1–9.0) 0.086 (0.037–0.160) 53 (23–96) 0.059 (0.047–0.071) 36 (29–44)
1995 < 1 <0.01 (<0.01–0.013) 4.2 (1.6–7.9) 0.075 (0.030–0.140) 44 (18–81) 0.051 (0.039–0.063) 30 (23–37)
2000 < 1 <0.01 (<0.01–0.011) 3.1 (1.1–6.3) 0.059 (0.022–0.110) 34 (12–65) 0.041 (0.030–0.053) 23 (17–30)
2005 < 1 <0.01 (<0.01–<0.01) 2.3 (0.98–4.1) 0.045 (0.018–0.083) 25 (10–46) 0.032 (0.026–0.039) 18 (14–22)
2010 < 1 <0.01 (<0.01–0.010) 3 (1.2–5.6) 0.053 (0.025–0.092) 29 (13–50) 0.031 (0.025–0.038) 17 (13–21)
2011 < 1 <0.01 (<0.01–0.011) 3.1 (1.3–5.8) 0.055 (0.026–0.095) 29 (14–51) 0.032 (0.026–0.039) 17 (14–21)
2012 < 1 <0.01 (<0.01–0.011) 3.2 (1.3–6.0) 0.057 (0.027–0.099) 30 (14–52) 0.033 (0.027–0.040) 18 (14–21)
Singapore 1990 3 0.12 (0.120–0.120) 4 (3.8–4.1) 2.5 (1.0–4.6) 82 (33–152) 1.8 (1.6–2.1) 61 (53–69)
1995 3 0.13 (0.120–0.130) 3.6 (3.4–3.8) 2.9 (1.2–5.4) 84 (35–154) 2.2 (1.9–2.5) 62 (55–71)
2000 4 0.12 (0.110–0.140) 3.2 (2.8–3.6) 2.7 (1.1–4.9) 68 (29–125) 2 (1.7–2.2) 51 (44–57)
2005 4 0.082 (0.071–0.094) 1.8 (1.6–2.1) 2.1 (0.890–3.9) 47 (20–86) 1.6 (1.4–1.8) 35 (31–40)
2010 5 0.097 (0.082–0.110) 1.9 (1.6–2.2) 2.3 (0.850–4.4) 45 (17–86) 1.8 (1.6–2.0) 35 (31–40)
2011 5 0.087 (0.073–0.100) 1.7 (1.4–1.9) 2.3 (0.770–4.6) 44 (15–88) 1.9 (1.7–2.1) 36 (32–41)
2012 5 0.089 (0.075–0.110) 1.7 (1.4–2.0) 3.9 (1.9–6.6) 73 (35–125) 2.6 (2.3–3.0) 50 (44–56)
Solomon Islands 1990 < 1 0.22 (0.063–0.480) 71 (20–154) 1.9 (0.690–3.8) 619 (222–1 210) 0.97 (0.600–1.4) 312 (193–460)
1995 < 1 0.2 (0.077–0.370) 54 (21–103) 1.7 (0.810–2.9) 473 (225–810) 0.86 (0.710–1.0) 240 (196–288)
2000 < 1 0.17 (0.068–0.330) 42 (16–79) 1.5 (0.710–2.6) 364 (173–624) 0.76 (0.620–0.910) 185 (151–222)
2005 < 1 0.13 (0.053–0.240) 27 (11–50) 1.2 (0.560–2.0) 251 (120–429) 0.67 (0.540–0.800) 142 (116–171)
2010 < 1 0.09 (0.039–0.160) 17 (7.5–30) 0.9 (0.420–1.6) 171 (79–297) 0.57 (0.470–0.680) 108 (89–129)
2011 < 1 0.085 (0.038–0.150) 16 (7.0–28) 0.86 (0.400–1.5) 160 (74–279) 0.55 (0.460–0.660) 103 (85–123)
2012 < 1 0.082 (0.036–0.150) 15 (6.6–27) 0.83 (0.380–1.5) 151 (70–264) 0.54 (0.440–0.640) 97 (80–116)
Tokelau 1990 < 1 <0.01 (<0.01–<0.01) 6.4 (2.0–13) <0.01 (<0.01–<0.01) 85 (24–185) <0.01 (<0.01–<0.01) 72 (57–90)
1995 < 1 <0.01 (0–<0.01) 4.9 (<0.1–22) <0.01 (<0.01–<0.01) 54 (2.4–185) <0.01 (<0.01–<0.01) 39 (13–80)
2000 < 1 <0.01 (<0.01–<0.01) 4.1 (0.57–11) <0.01 (<0.01–<0.01) 32 (5.3–82) <0.01 (<0.01–<0.01) 13 (3.5–28)
2005 < 1 0 (0–0) <0.1 (0–0.10) 0 (0–<0.01) 0.26 (<0.1–0.54) 0 (0–0) 0 (0–0)
2010 < 1 0 (0–0) 0 (0–<0.1) 0 (0–0) 0 (0–0)
2011 < 1 0 (0–0) 0 (0–<0.1) 0 (0–0) 0 (0–0)
2012 < 1 0 (0–0) 0 (0–<0.1) 0 (0–0) 0 (0–0)
Tonga 1990 < 1 <0.01 (<0.01–0.010) 5.9 (2.7–10) 0.056 (0.027–0.097) 59 (28–102) 0.036 (0.030–0.042) 38 (32–45)
1995 < 1 <0.01 (<0.01–<0.01) 4.2 (2.0–7.3) 0.045 (0.019–0.081) 46 (20–84) 0.032 (0.027–0.037) 33 (28–39)
2000 < 1 <0.01 (<0.01–<0.01) 3.6 (1.4–6.8) 0.038 (0.015–0.073) 39 (15–74) 0.027 (0.021–0.034) 28 (22–35)
2005 < 1 <0.01 (<0.01–<0.01) 3 (1.3–5.5) 0.033 (0.013–0.060) 32 (13–60) 0.023 (0.018–0.028) 22 (18–27)
2010 < 1 <0.01 (<0.01–<0.01) 2.6 (1.0–5.0) 0.029 (0.013–0.052) 28 (13–50) 0.017 (0.015–0.020) 17 (14–20)
2011 < 1 <0.01 (<0.01–<0.01) 2.6 (1.0–4.9) 0.028 (0.013–0.049) 27 (12–47) 0.016 (0.014–0.019) 16 (13–18)
2012 < 1 <0.01 (<0.01–<0.01) 2.5 (1.1–4.6) 0.027 (0.014–0.045) 26 (13–43) 0.015 (0.013–0.018) 14 (12–17)
a
Rates are per 100 000 population.
271 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
WESTERN PACIFIC REGION
7$%/($(VWLPDWHVRIWKHEXUGHQRIGLVHDVHFDXVHGE\7%±
YEAR POPULATION
(MILLIONS)
NUMBER
(THOUSANDS) RATEaNUMBER
(THOUSANDS) RATEaNUMBER
(THOUSANDS) RATEa
MORTALITY (EXCLUDING HIV) PREVALENCE (INCLUDING HIV) INCIDENCE (INCLUDING HIV)
Tuvalu 1990 < 1 <0.01 (<0.01–0.019) 98 (27–212) 0.083 (0.029–0.160) 921 (327–1 820) 0.048 (0.031–0.069) 536 (347–766)
1995 < 1 <0.01 (<0.01–0.021) 73 (5.1–227) 0.066 (<0.01–0.170) 711 (101–1 900) 0.04 (0.017–0.074) 437 (181–805)
2000 < 1 <0.01 (<0.01–0.014) 68 (19–146) 0.059 (0.021–0.120) 626 (226–1 230) 0.034 (0.022–0.048) 357 (231–510)
2005 < 1 <0.01 (<0.01–0.010) 50 (15–105) 0.047 (0.017–0.089) 480 (180–923) 0.028 (0.019–0.039) 291 (198–402)
2010 < 1 <0.01 (<0.01–<0.01) 18 (7.3–33) 0.022 (<0.01–0.044) 222 (75–448) 0.018 (0.014–0.021) 178 (145–215)
2011 < 1 <0.01 (<0.01–<0.01) 12 (3.8–24) 0.017 (<0.01–0.036) 176 (53–371) 0.015 (0.012–0.018) 152 (124–184)
2012 < 1 <0.01 (<0.01–<0.01) 37 (16–68) 0.037 (0.017–0.065) 377 (172–658) 0.024 (0.019–0.029) 241 (196–290)
Vanuatu 1990 < 1 0.016 (<0.01–0.032) 11 (3.9–22) 0.22 (0.062–0.470) 148 (43–319) 0.19 (0.150–0.230) 127 (103–154)
1995 < 1 0.011 (<0.01–0.019) 6.5 (2.9–11) 0.16 (0.049–0.340) 97 (29–204) 0.11 (0.085–0.130) 63 (51–76)
2000 < 1 0.03 (0.013–0.054) 16 (6.9–29) 0.31 (0.140–0.550) 166 (74–295) 0.2 (0.160–0.250) 110 (89–132)
2005 < 1 0.029 (0.012–0.052) 14 (5.9–25) 0.28 (0.130–0.490) 134 (63–232) 0.17 (0.140–0.210) 83 (68–99)
2010 < 1 0.024 (0.011–0.043) 10 (4.5–18) 0.25 (0.110–0.440) 105 (47–185) 0.16 (0.130–0.200) 69 (57–83)
2011 < 1 0.022 (<0.01–0.039) 9.1 (4.1–16) 0.24 (0.100–0.420) 97 (42–175) 0.16 (0.130–0.190) 67 (55–80)
2012 < 1 0.02 (<0.01–0.035) 7.9 (3.6–14) 0.22 (0.090–0.410) 89 (36–165) 0.16 (0.130–0.190) 65 (53–77)
Viet Nam 1990 69 36 (21–55) 52 (30–79) 360 (150–670) 525 (212–976) 170 (120–240) 251 (172–344)
1995 76 32 (20–47) 42 (26–61) 340 (150–610) 451 (198–805) 170 (120–220) 220 (155–295)
2000 81 27 (18–38) 33 (22–47) 290 (130–510) 353 (156–629) 160 (120–210) 197 (142–260)
2005 85 23 (16–31) 27 (19–37) 240 (110–440) 288 (125–517) 150 (110–190) 176 (131–229)
2010 89 19 (13–26) 22 (15–29) 210 (87–390) 238 (97–440) 140 (100–180) 155 (115–201)
2011 90 19 (13–25) 21 (14–28) 200 (82–380) 227 (91–424) 140 (100–180) 151 (112–197)
2012 91 18 (12–25) 20 (13–27) 200 (78–370) 218 (86–410) 130 (99–170) 147 (109–192)
Wallis and Futuna 1990 < 1 <0.01 (<0.01–<0.01) 17 (9.2–27) 0.028 (0.011–0.052) 201 (80–378) 0.022 (0.019–0.024) 156 (137–176)
Islands 1995 < 1 <0.01 (<0.01–<0.01) 4.2 (2.3–6.6) <0.01 (<0.01–0.019) 62 (19–132) <0.01 (<0.01–<0.01) 49 (43–55)
2000 < 1 <0.01 (<0.01–<0.01) 4.2 (3.6–4.9) <0.01 (<0.01–0.019) 63 (19–132) <0.01 (<0.01–<0.01) 15 (13–17)
2005 < 1 <0.01 (<0.01–<0.01) 4.7 (2.6–7.5) 0.01 (<0.01–0.021) 70 (21–148) <0.01 (<0.01–<0.01) 57 (50–64)
2010 < 1 <0.01 (<0.01–<0.01) 2.8 (1.4–4.6) <0.01 (<0.01–0.012) 42 (13–88) <0.01 (<0.01–<0.01) 36 (31–41)
2011 < 1 <0.01 (<0.01–<0.01) 2.6 (1.5–4.1) <0.01 (<0.01–0.011) 41 (13–85) <0.01 (<0.01–<0.01) 17 (15–19)
2012 < 1 <0.01 (<0.01–<0.01) 13 (5.6–23) 0.016 (<0.01–0.026) 117 (59–193) <0.01 (<0.01–<0.01) 65 (57–74)
a Rates are per 100 000 population.
272 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($,QFLGHQFHQRWLILFDWLRQDQGFDVHGHWHFWLRQUDWHVDOOIRUPV±
INCIDENCE (INCLUDING HIV) INCIDENCE HIV-POSITIVE NOTIFIED NEW AND RELAPSE
b
CASE DETECTION
YEAR POPULATION
(MILLIONS)
NUMBER
(THOUSANDS) RATE
a
NUMBER
(THOUSANDS) RATE
a
NUMBER RATE
a
PERCENT
American Samoa 1990 < 1 0.012 (<0.01–0.015) 26 (21–31) 9 19 75 (62–93)
1995 < 1 <0.01 (<0.01–<0.01) 12 (9.4–14)
2000 < 1 <0.01 (<0.01–<0.01) 6.9 (5.6–8.4) 3 5.2 75 (62–93)
2005 < 1 <0.01 (<0.01–<0.01) 13 (10–15) 6 10 80 (66–99)
2010 < 1 <0.01 (<0.01–<0.01) 7.8 (6.3–9.4) 4 7.2 92 (76–110)
2011 < 1 <0.01 (<0.01–<0.01) 7.8 (6.3–9.4) 3 5.4 70 (58–86)
2012 < 1 <0.01 (<0.01–<0.01) 7.3 (5.9–8.9)
Australia 1990 17 1.2 (1.0–1.3) 6.8 (6.0–7.7) 0.028 (0.024–0.031) 0.2 (0.14–0.18) 1 016 5.9 87 (77–99)
1995 18 1.2 (1.1–1.4) 6.8 (6.0–7.7) 0.047 (0.041–0.053) 0.3 (0.23–0.29) 1 073 5.9 87 (77–99)
2000 19 1.2 (1.1–1.4) 6.2 (5.5–7.0) 0.029 (0.026–0.033) 0.2 (0.13–0.17) 1 043 5.4 87 (77–99)
2005 21 1.2 (1.1–1.4) 5.9 (5.1–6.6) 0.028 (0.025–0.032) 0.1 (0.12–0.16) 1 046 5.1 87 (77–99)
2010 22 1.4 (1.3–1.6) 6.5 (5.7–7.3) 0.036 (0.031–0.041) 0.2 (0.14–0.18) 1 257 5.6 87 (77–99)
2011 23 1.4 (1.2–1.6) 6.3 (5.5–7.1) 0.036 (0.031–0.040) 0.2 (0.14–0.18) 1 239 5.4 87 (77–99)
2012 23 1.5 (1.3–1.7) 6.5 (5.7–7.4) 0.038 (0.033–0.043) 0.2 (0.14–0.19) 1 305 5.7 87 (77–99)
Brunei 1990 < 1 0.16 (0.140–0.190) 64 (56–72) 143 56 87 (77–99)
Darussalam 1995 < 1 0.18 (0.160–0.210) 63 (55–71)
2000 < 1 0.35 (0.310–0.400) 106 (93–120) 307 93 87 (77–99)
2005 < 1 0.19 (0.160–0.210) 51 (45–58) <0.01 (<0.01–<0.01) 0.6 (<0.1–2.0) 163 44 87 (77–99)
2010 < 1 0.27 (0.240–0.310) 68 (60–77) <0.01 (0–<0.01) 0.3 (0–1.4) 237 59 87 (77–99)
2011 < 1 0.26 (0.230–0.300) 65 (57–74) <0.01 (<0.01–<0.01) 0.9 (0.12–2.3) 230 57 87 (77–99)
2012 < 1 0.28 (0.240–0.320) 68 (59–77) <0.01 (0–<0.01) 0.4 (0–2.1) 243 59 87 (77–99)
Cambodia 1990 9 53 (38–69) 580 (423–761) 0.99 (0.72–1.3) 11 (8.0–14) 6 501 72 12 (9.4–17)
1995 11 62 (48–78) 578 (448–724) 5.1 (4.0–6.4) 48 (37–60) 14 603 136 23 (19–30)
2000 12 71 (56–87) 577 (458–710) 7.9 (6.3–9.7) 65 (51–80) 18 891 155 27 (22–34)
2005 13 68 (57–81) 510 (424–604) 5.8 (4.8–6.8) 43 (36–51) 35 535 266 52 (44–63)
2010 14 63 (54–72) 437 (376–503) 3.1 (2.7–3.6) 22 (19–25) 40 460 282 64 (56–75)
2011 15 62 (53–71) 424 (364–489) 3.1 (2.6–3.5) 21 (18–24) 38 555 264 62 (54–73)
2012 15 61 (52–70) 411 (353–474) 2.7 (2.3–3.1) 18 (15–21) 40 185 270 66 (57–77)
China 1990 1 165 1 800 (1 400–2 200) 153 (121–189) 0.18 (0.14–0.22) <0.1 (<0.1–<0.1) 375 481 32 21 (17–27)
1995 1 238 1 600 (1 300–1 900) 129 (106–154) 1.4 (1.2–1.7) 0.1 (0.10–0.14) 515 764 42 32 (27–39)
2000 1 280 1 400 (1 200–1 600) 109 (92–126) 4.2 (3.6–4.9) 0.3 (0.28–0.38) 454 372 35 33 (28–38)
2005 1 318 1 200 (1 100–1 400) 92 (80–105) 6.3 (5.5–7.2) 0.5 (0.42–0.54) 899 729 68 74 (65–85)
2010 1 360 1 100 (930–1 200) 78 (68–88) 7.6 (6.7–8.6) 0.6 (0.49–0.63) 908 399 67 86 (76–98)
2011 1 368 1 000 (900–1 200) 75 (66–85) 7.6 (6.7–8.6) 0.6 (0.49–0.63) 899 669 66 88 (78–100)
2012 1 377 1 000 (880–1 100) 73 (64–82) 7.3 (6.4–8.2) 0.5 (0.47–0.60) 890 645 65 89 (79–100)
China, Hong Kong 1990 6 7.5 (6.6–8.5) 129 (113–146) 6 510 112 87 (77–99)
SAR 1995 6 7.1 (6.3–8.1) 116 (102–132) 6 212 101 87 (77–99)
2000 7 6.9 (6.1–7.8) 101 (89–115) 6 015 88 87 (77–99)
2005 7 6.5 (5.7–7.4) 94 (83–107) 0.054 (0.036–0.075) 0.8 (0.53–1.1) 5 660 82 87 (77–99)
2010 7 5.7 (5.0–6.4) 81 (71–91) 0.036 (0.022–0.053) 0.5 (0.31–0.75) 4 935 70 87 (77–99)
2011 7 5.4 (4.8–6.2) 77 (67–87) 0.049 (0.033–0.069) 0.7 (0.46–0.97) 4 739 67 87 (77–99)
2012 7 5.5 (4.8–6.3) 77 (68–88) 0.044 (0.026–0.067) 0.6 (0.37–0.94) 4 809 67 87 (77–99)
China, Macao 1990 < 1 0.39 (0.350–0.450) 110 (96–124) 343 95 87 (77–99)
SAR 1995 < 1 0.46 (0.410–0.520) 116 (102–131) 402 101 87 (77–99)
2000 < 1 0.52 (0.450–0.580) 120 (105–135) 449 104 87 (77–99)
2005 < 1 0.46 (0.400–0.520) 98 (86–111) <0.01 (0–<0.01) 0.3 (0–1.3) 398 85 87 (77–99)
2010 < 1 0.45 (0.400–0.510) 85 (74–96) <0.01 (<0.01–<0.01) 0.6 (<0.1–1.7) 394 74 87 (77–99)
2011 < 1 0.44 (0.380–0.490) 80 (70–91) <0.01 (<0.01–<0.01) 0.5 (<0.1–1.5) 380 70 87 (77–99)
2012 < 1 0.46 (0.410–0.530) 83 (73–94) <0.01 (<0.01–<0.01) 0.4 (<0.1–1.5) 404 73 87 (77–99)
Cook Islands 1990 < 1 0 (0–0) 0 (0–0) 0 0
1995 < 1 <0.01 (<0.01–<0.01) 13 (11–14) 2 11 87 (77–99)
2000 < 1 <0.01 (<0.01–<0.01) 6.5 (5.7–7.3) 1 5.6 87 (77–99)
2005 < 1 <0.01 (<0.01–<0.01) 5.9 (5.2–6.7) 1 5.2 87 (77–99)
2010 < 1 0 (0–0) 0 (0–0) 0 0
2011 < 1 <0.01 (<0.01–<0.01) 5.6 (4.9–6.4) 1 4.9 87 (77–99)
2012 < 1 <0.01 (<0.01–<0.01) 5.6 (4.9–6.3) 1 4.9 87 (77–99)
Fiji 1990 < 1 0.81 (0.710–0.920) 112 (98–126) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 226 31 28 (25–32)
1995 < 1 0.6 (0.530–0.680) 77 (68–87) <0.01 (<0.01–<0.01) <0.1 (<0.1–0.10) 203 26 34 (30–39)
2000 < 1 0.44 (0.390–0.500) 54 (48–62) <0.01 (<0.01–<0.01) 0.2 (0.14–0.18) 144 18 33 (29–37)
2005 < 1 0.33 (0.290–0.370) 40 (35–45) <0.01 (<0.01–<0.01) 0.2 (0.21–0.27) 132 16 41 (36–46)
2010 < 1 0.24 (0.210–0.270) 28 (24–32) <0.01 (<0.01–<0.01) 0.2 (0.13–0.17) 189 22 79 (70–90)
2011 < 1 0.23 (0.200–0.260) 26 (23–29) <0.01 (<0.01–<0.01) 0.2 (0.13–0.17) 215 25 95 (84–110)
2012 < 1 0.21 (0.190–0.240) 24 (21–27) <0.01 (<0.01–<0.01) 0.2 (0.13–0.17) 210 24 99 (87–110)
French Polynesia 1990 < 1 0.068 (0.059–0.077) 34 (30–39) 59 30 87 (77–99)
1995 < 1 0.1 (0.088–0.110) 47 (41–53)
2000 < 1 0.071 (0.062–0.081) 30 (26–34) 62 26 87 (77–99)
2005 < 1 0.072 (0.063–0.082) 28 (25–32) 63 25 87 (77–99)
2010 < 1 0.047 (0.041–0.053) 18 (15–20) 41 15 87 (77–99)
2011 < 1 0.074 (0.064–0.083) 27 (24–31) 64 24 87 (77–99)
2012 < 1 0.058 (0.050–0.065) 21 (18–24) 50 18 87 (77–99)
Guam 1990 < 1 0.066 (0.058–0.075) 50 (44–57)
1995 < 1 0.099 (0.087–0.110) 68 (60–77)
2000 < 1 0.062 (0.054–0.070) 40 (35–45) 54 35 87 (77–99)
2005 < 1 0.072 (0.063–0.082) 46 (40–52) 63 40 87 (77–99)
2010 < 1 0.12 (0.100–0.130) 73 (64–82) <0.01 (0–<0.01) 1.2 (0–5.5) 101 63 87 (77–99)
2011 < 1 0.094 (0.083–0.110) 59 (51–66) 82 51 87 (77–99)
2012 < 1 0.078 (0.069–0.089) 48 (42–54) <0.01 (0–<0.01) 0.3 (0–2.9) 68 42 87 (77–99)
Japan 1990 122 60 (52–67) 49 (43–55) 0.24 (0.21–0.28) 0.2 (0.18–0.23) 51 821 42 87 (77–99)
1995 124 50 (43–56) 40 (35–45) 0.22 (0.20–0.25) 0.2 (0.16–0.20) 43 078 35 87 (77–99)
2000 126 45 (40–51) 36 (32–41) 0.15 (0.13–0.17) 0.1 (0.11–0.14) 39 384 31 87 (77–99)
2005 127 31 (27–35) 25 (22–28) 0.13 (0.11–0.15) 0.1 (<0.1–0.11) 27 194 21 87 (77–99)
2010 127 26 (23–30) 20 (18–23) 0.1 (0.091–0.12) <0.1 (<0.1–<0.1) 22 693 18 87 (77–99)
2011 127 25 (22–29) 20 (18–23) 0.1 (0.089–0.12) <0.1 (<0.1–<0.1) 22 119 17 87 (77–99)
2012 127 24 (21–28) 19 (17–22) 0.098 (0.085–0.11) <0.1 (<0.1–<0.1) 20 857 16 86 (76–98)
Kiribati 1990 < 1 0.083 (0.066–0.100) 116 (93–143) 68 96 82 (67–100)
1995 < 1 0.39 (0.310–0.460) 505 (410–609)
2000 < 1 0.31 (0.250–0.380) 372 (296–456) 252 304 82 (67–100)
2005 < 1 0.44 (0.360–0.530) 488 (396–588) 332 367 75 (62–93)
2010 < 1 0.36 (0.290–0.430) 366 (298–441) <0.01 (<0.01–<0.01) 2.5 (1.7–3.4) 286 293 80 (66–98)
2011 < 1 0.43 (0.350–0.520) 432 (351–521) 343 346 80 (66–98)
2012 < 1 0.43 (0.350–0.520) 429 (349–517) 346 343 80 (66–98)
Lao People's 1990 4 21 (13–31) 492 (304–725) <0.01 (<0.01–<0.01) 0.1 (<0.1–0.15) 1 826 43 8.7 (5.9–14)
Democratic 1995 5 20 (12–29) 403 (249–593) 0.026 (0.015–0.039) 0.5 (0.30–0.81) 830 17 4.2 (2.9–6.8)
Republic 2000 5 18 (11–26) 330 (204–486) 0.092 (0.051–0.15) 1.7 (0.95–2.7) 2 227 41 13 (8.5–20)
2005 6 16 (9.7–23) 270 (167–398) 0.17 (0.093–0.28) 3 (1.6–4.8) 3 766 65 24 (16–39)
2010 6 14 (8.8–21) 221 (137–326) 0.23 (0.12–0.37) 3.6 (1.9–5.8) 4 061 63 29 (19–46)
2011 7 14 (8.6–20) 213 (131–313) 0.24 (0.13–0.39) 3.7 (2.0–6.0) 4 360 67 31 (21–51)
2012 7 14 (8.4–20) 204 (126–301) 0.25 (0.13–0.41) 3.8 (2.0–6.2) 4 118 62 30 (21–49)
Malaysia 1990 18 23 (21–26) 127 (113–142) 0.18 (0.16–0.20) 1 (0.88–1.1) 11 702 64 51 (45–57)
1995 21 22 (20–25) 108 (97–120) 1.1 (0.96–1.2) 5.2 (4.6–5.7) 11 778 57 53 (47–58)
2000 23 22 (20–24) 95 (86–103) 1.9 (1.7–2.1) 8 (7.3–8.8) 15 057 64 68 (62–75)
2005 26 22 (20–24) 86 (79–94) 2.2 (2.0–2.3) 8.3 (7.6–9.1) 15 415 60 69 (64–76)
2010 28 23 (21–25) 82 (75–89) 2.3 (2.1–2.5) 8.1 (7.4–8.8) 18 517 65 80 (74–87)
2011 29 23 (21–25) 81 (74–88) 2.3 (2.1–2.5) 7.8 (7.2–8.5) 19 808 69 85 (78–93)
2012 29 24 (22–26) 80 (74–87) 2.3 (2.2–2.6) 8 (7.4–8.7) 21 851 75 93 (85–100)
Marshall Islands 1990 < 1 0.065 (<0.01–0.190) 137 (14–396)
1995 < 1 0.097 (0.024–0.220) 190 (46–432)
2000 < 1 0.14 (0.084–0.200) 263 (161–389) 34 65 25 (17–41)
2005 < 1 0.19 (0.050–0.420) 363 (96–803) 111 213 59 (27–220)
2010 < 1 0.26 (0.051–0.650) 502 (97–1 230) 193 368 73 (30–380)
2011 < 1 0.28 (0.054–0.690) 536 (103–1 320) <0.01 (0–0.019) 6.1 (0–36) 139 265 49 (20–260)
2012 < 1 0.3 (0.058–0.740) 572 (110–1 400) <0.01 (0–0.015) 2.6 (0–29) 145 276 48 (20–250)
a
Rates are per 100 000 population.
b
NOTIFIED NEW AND RELAPSE includes cases for which the treatment history is unknown.

273 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
WESTERN PACIFIC REGION
7$%/($,QFLGHQFHQRWLILFDWLRQDQGFDVHGHWHFWLRQUDWHVDOOIRUPV±
INCIDENCE (INCLUDING HIV) INCIDENCE HIV-POSITIVE NOTIFIED NEW AND RELAPSE
b
CASE DETECTION
YEAR POPULATION
(MILLIONS)
NUMBER
(THOUSANDS) RATE
a
NUMBER
(THOUSANDS) RATE
a
NUMBER RATE
a
PERCENT
Micronesia 1990 < 1 0.36 (0.100–0.800) 379 (104–827) 367 381 100 (46–370)
(Federated 1995 < 1 0.35 (0.200–0.540) 325 (185–505) 172 160 49 (32–87)
States of) 2000 < 1 0.3 (0.210–0.400) 279 (200–371) 91 85 30 (23–42)
2005 < 1 0.25 (0.170–0.360) 240 (158–338) 98 92 38 (27–58)
2010 < 1 0.21 (0.092–0.380) 206 (89–371) 164 158 77 (43–180)
2011 < 1 0.21 (0.089–0.370) 200 (86–360) 148 143 72 (40–170)
2012 < 1 0.2 (0.086–0.360) 194 (83–349) 144 139 72 (40–170)
Mongolia 1990 2 8.8 (7.5–10) 405 (345–470) 1 659 76 19 (16–22)
1995 2 7.2 (6.3–8.2) 314 (274–356) 2 780 121 39 (34–44)
2000 2 6.1 (5.5–6.7) 254 (228–281) 3 109 130 51 (46–57)
2005 3 5.7 (5.2–6.1) 225 (207–243) <0.01 (<0.01–<0.01) <0.1 (<0.1–<0.1) 4 601 182 81 (75–88)
2010 3 6.1 (5.7–6.5) 224 (209–240) <0.01 (<0.01–<0.01) 0.2 (0.21–0.24) 4 458 164 73 (68–79)
2011 3 6.1 (5.7–6.6) 223 (208–239) <0.01 (<0.01–<0.01) 0.3 (0.27–0.31) 4 217 153 69 (64–74)
2012 3 6.2 (5.8–6.7) 223 (208–239) 0.011 (<0.01–0.011) 0.4 (0.35–0.41) 4 128 148 66 (62–71)
Nauru 1990 < 1 <0.01 (<0.01–<0.01) 88 (77–99) 7 76 87 (77–99)
1995 < 1 <0.01 (<0.01–<0.01) 40 (35–46)
2000 < 1 <0.01 (<0.01–<0.01) 46 (40–52) 4 40 87 (77–99)
2005 < 1 0.013 (0.011–0.014) 125 (110–142) 11 109 87 (77–99)
2010 < 1 <0.01 (<0.01–<0.01) 34 (30–39) 3 30 87 (77–99)
2011 < 1 <0.01 (<0.01–<0.01) 57 (50–65) 5 50 87 (77–99)
2012 < 1 <0.01 (<0.01–<0.01) 54 (47–61)
New Caledonia 1990 < 1 0.16 (0.140–0.190) 98 (85–110) 143 85 87 (77–99)
1995 < 1 0.1 (0.088–0.110) 53 (46–60) 87 46 87 (77–99)
2000 < 1 0.11 (0.095–0.120) 51 (45–58) 94 45 87 (77–99)
2005 < 1 0.054 (0.047–0.061) 24 (21–27) 47 21 87 (77–99)
2010 < 1 0.056 (0.049–0.064) 23 (20–26) 49 20 87 (77–99)
2011 < 1 0.06 (0.052–0.068) 24 (21–27) 52 21 87 (77–99)
2012 < 1 0.044 (0.038–0.049) 17 (15–20) 38 15 87 (77–99)
New Zealand 1990 3 0.4 (0.350–0.450) 12 (10–13) <0.01 (<0.01–<0.01) 0.1 (<0.1–0.11) 348 10 87 (77–99)
1995 4 0.45 (0.390–0.510) 12 (11–14) <0.01 (<0.01–<0.01) 0.2 (0.17–0.22) 391 11 87 (77–99)
2000 4 0.4 (0.350–0.450) 10 (9.0–12) <0.01 (<0.01–<0.01) 0.2 (0.13–0.17) 344 8.9 87 (77–99)
2005 4 0.38 (0.330–0.430) 9.2 (8.1–10) <0.01 (<0.01–<0.01) 0.1 (0.12–0.16) 332 8 87 (77–99)
2010 4 0.35 (0.300–0.390) 7.9 (6.9–9.0) <0.01 (<0.01–<0.01) 0.1 (0.11–0.15) 301 6.9 87 (77–99)
2011 4 0.35 (0.310–0.400) 7.9 (7.0–9.0) <0.01 (<0.01–<0.01) 0.1 (0.11–0.15) 305 6.9 87 (77–99)
2012 4 0.34 (0.300–0.380) 7.6 (6.6–8.6) <0.01 (<0.01–<0.01) 0.1 (0.11–0.14) 293 6.6 87 (77–99)
Niue 1990 < 1 0 (0–0) 0 (0–0) 0 0
1995 < 1 0 (0–0) 0 (0–0) 0 0
2000 < 1 0 (0–0) 0 (0–0) 0 0
2005 < 1 0 (0–0) 0 (0–0) 0 0
2010 < 1 0 (0–0) 0 (0–0) 0 0
2011 < 1 <0.01 (<0.01–<0.01) 81 (71–91) 1 70 87 (77–99)
2012 < 1 <0.01 (<0.01–<0.01) 37 (32–42) 0 0 0
Northern Mariana 1990 < 1 0.032 (0.028–0.036) 73 (64–83) 28 64 87 (77–99)
Islands 1995 < 1 0.055 (0.048–0.062) 96 (84–109) 48 83 87 (77–99)
2000 < 1 0.086 (0.076–0.098) 126 (110–143) 75 110 87 (77–99)
2005 < 1 0.066 (0.057–0.074) 102 (89–115) 57 89 87 (77–99)
2010 < 1 0.037 (0.032–0.042) 68 (60–77) 32 59 87 (77–99)
2011 < 1 0.038 (0.033–0.043) 71 (62–81) 33 62 87 (77–99)
2012 < 1 0.037 (0.032–0.042) 69 (60–78) 32 60 87 (77–99)
Palau 1990 < 1 <0.01 (<0.01–<0.01) 45 (36–54)
1995 < 1 0.025 (0.021–0.031) 147 (119–178) 19 110 75 (62–93)
2000 < 1 0.03 (0.024–0.036) 156 (127–189)
2005 < 1 0.013 (0.011–0.016) 67 (54–81) 10 50 75 (62–93)
2010 < 1 0.024 (0.019–0.029) 116 (94–140) 19 93 80 (66–98)
2011 < 1 0.015 (0.012–0.018) 73 (59–88) 12 58 80 (66–98)
2012 < 1 <0.01 (<0.01–<0.01) 24 (20–29) 4 19 80 (66–98)
Papua New 1990 4 13 (8.5–18) 308 (203–435) 0.1 (0.067–0.14) 2.4 (1.6–3.4) 2 497 60 19 (14–30)
Guinea 1995 5 15 (10–21) 322 (212–453) 0.46 (0.30–0.65) 9.7 (6.4–14) 8 041 171 53 (38–80)
2000 5 19 (12–26) 349 (230–492) 1 (0.68–1.4) 19 (13–27) 10 520 196 56 (40–85)
2005 6 22 (14–31) 358 (236–505) 1.4 (0.92–2.0) 23 (15–32) 12 564 206 58 (41–87)
2010 7 24 (16–34) 348 (229–491) 1.1 (0.75–1.6) 17 (11–23) 14 531 212 61 (43–92)
2011 7 24 (16–34) 346 (228–488) 1.2 (0.76–1.6) 16 (11–23) 14 893 212 61 (44–93)
2012 7 25 (16–35) 348 (230–490) 1.1 (0.71–1.5) 15 (9.9–21) 20 557 287 82 (59–120)
Philippines 1990 62 240 (150–360) 393 (243–580) 0.024 (0.015–0.036) <0.1 (<0.1–<0.1) 317 008 512 130 (88–210)
1995 70 250 (200–300) 360 (294–432) 0.025 (0.020–0.030) <0.1 (<0.1–<0.1) 119 186 171 48 (40–58)
2000 78 260 (210–310) 329 (269–395) 0.077 (0.063–0.092) 0.1 (<0.1–0.12) 119 914 154 47 (39–57)
2005 86 260 (210–310) 301 (246–361) 0.18 (0.15–0.22) 0.2 (0.17–0.25) 137 100 160 53 (44–65)
2010 93 260 (210–310) 275 (227–328) 0.39 (0.32–0.46) 0.4 (0.34–0.49) 166 323 178 65 (54–79)
2011 95 260 (210–310) 270 (223–322) 0.46 (0.38–0.55) 0.5 (0.40–0.58) 195 560 206 76 (64–92)
2012 97 260 (210–310) 265 (219–316) 0.46 (0.38–0.55) 0.5 (0.39–0.57) 216 627 224 84 (71–100)
Republic of Korea 1990 43 73 (64–83) 171 (150–194) 0.051 (0.045–0.058) 0.1 (0.10–0.14) 63 904 149 87 (77–99)
1995 45 48 (42–55) 108 (95–123) 0.044 (0.038–0.049) 0.1 (<0.1–0.11) 42 117 94 87 (77–99)
2000 46 25 (22–28) 54 (48–62) 0.023 (0.020–0.026) <0.1 (<0.1–<0.1) 21 782 47 87 (77–99)
2005 47 49 (43–56) 105 (92–119) 0.089 (0.078–0.10) 0.2 (0.17–0.21) 42 892 91 87 (77–99)
2010 48 51 (44–57) 105 (92–118) 0.13 (0.12–0.15) 0.3 (0.24–0.31) 44 063 91 87 (77–99)
2011 49 53 (47–60) 109 (96–124) 0.14 (0.13–0.16) 0.3 (0.26–0.33) 46 253 95 87 (77–99)
2012 49 53 (46–60) 108 (95–122) 0.15 (0.13–0.17) 0.3 (0.27–0.35) 43 702 89 82 (73–94)
Samoa 1990 < 1 0.059 (0.047–0.071) 36 (29–44) 44 27 75 (62–93)
1995 < 1 0.051 (0.039–0.063) 30 (23–37) 45 26 89 (71–110)
2000 < 1 0.041 (0.030–0.053) 23 (17–30) 43 25 110 (82–140)
2005 < 1 0.032 (0.026–0.039) 18 (14–22) 24 13 75 (62–92)
2010 < 1 0.031 (0.025–0.038) 17 (13–21) 14 7.5 45 (37–56)
2011 < 1 0.032 (0.026–0.039) 17 (14–21) 20 11 62 (51–78)
2012 < 1 0.033 (0.027–0.040) 18 (14–21) 22 12 66 (55–82)
Singapore 1990 3 1.8 (1.6–2.1) 61 (53–69) <0.01 (<0.01–<0.01) 0.2 (0.21–0.27) 1 591 53 87 (77–99)
1995 3 2.2 (1.9–2.5) 62 (55–71) 0.044 (0.038–0.049) 1.3 (1.1–1.4) 1 889 54 87 (77–99)
2000 4 2 (1.7–2.2) 51 (44–57) 0.06 (0.053–0.068) 1.5 (1.3–1.7) 1 728 44 87 (77–99)
2005 4 1.6 (1.4–1.8) 35 (31–40) 0.06 (0.052–0.068) 1.3 (1.2–1.5) 1 376 31 87 (77–99)
2010 5 1.8 (1.6–2.0) 35 (31–40) 0.07 (0.061–0.079) 1.4 (1.2–1.6) 1 560 31 87 (77–99)
2011 5 1.9 (1.7–2.1) 36 (32–41) 0.072 (0.063–0.081) 1.4 (1.2–1.6) 1 641 32 87 (77–99)
2012 5 2.6 (2.3–3.0) 50 (44–56) 0.098 (0.086–0.11) 1.9 (1.6–2.1) 2 301 43 87 (77–99)
Solomon Islands 1990 < 1 0.97 (0.600–1.4) 312 (193–460) 382 122 39 (27–64)
1995 < 1 0.86 (0.710–1.0) 240 (196–288) 352 98 41 (34–50)
2000 < 1 0.76 (0.620–0.910) 185 (151–222) 302 73 40 (33–49)
2005 < 1 0.67 (0.540–0.800) 142 (116–171) 397 85 60 (50–73)
2010 < 1 0.57 (0.470–0.680) 108 (89–129) 338 64 59 (50–72)
2011 < 1 0.55 (0.460–0.660) 103 (85–123) 398 74 72 (60–87)
2012 < 1 0.54 (0.440–0.640) 97 (80–116) 361 66 67 (57–82)
Tokelau 1990 < 1 <0.01 (<0.01–<0.01) 72 (57–90) 1 62 86 (69–110)
1995 < 1 <0.01 (<0.01–<0.01) 39 (13–80) 2 132 340 (160–1 000)
2000 < 1 <0.01 (<0.01–<0.01) 13 (3.5–28) 0 0 0
2005 < 1 0 (0–0) 0 (0–0) 0 0
2010 < 1 0 (0–0) 0 (0–0) 0 0
2011 < 1 0 (0–0) 0 (0–0) 0 0
2012 < 1 0 (0–0) 0 (0–0)
Tonga 1990 < 1 0.036 (0.030–0.042) 38 (32–45) 23 24 64 (54–76)
1995 < 1 0.032 (0.027–0.037) 33 (28–39) 20 21 63 (54–75)
2000 < 1 0.027 (0.021–0.034) 28 (22–35) 24 24 88 (70–110)
2005 < 1 0.023 (0.018–0.028) 22 (18–27) 18 18 79 (65–99)
2010 < 1 0.017 (0.015–0.020) 17 (14–20) 11 11 63 (54–75)
2011 < 1 0.016 (0.014–0.019) 16 (13–18) 9 8.6 55 (47–66)
2012 < 1 0.015 (0.013–0.018) 14 (12–17) 11 10 73 (62–87)
a
Rates are per 100 000 population.
b
NOTIFIED NEW AND RELAPSE includes cases for which the treatment history is unknown.
274 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($,QFLGHQFHQRWLILFDWLRQDQGFDVHGHWHFWLRQUDWHVDOOIRUPV±
INCIDENCE (INCLUDING HIV) INCIDENCE HIV-POSITIVE NOTIFIED NEW AND RELAPSE
b
CASE DETECTION
YEAR POPULATION
(MILLIONS)
NUMBER
(THOUSANDS) RATEaNUMBER
(THOUSANDS) RATEaNUMBER RATEaPERCENT
Tuvalu 1990 < 1 0.048 (0.031–0.069) 536 (347–766) 23 255 48 (33–74)
1995 < 1 0.04 (0.017–0.074) 437 (181–805) 36 390 89 (48–220)
2000 < 1 0.034 (0.022–0.048) 357 (231–510) 16 170 48 (33–74)
2005 < 1 0.028 (0.019–0.039) 291 (198–402) 12 124 43 (31–63)
2010 < 1 0.018 (0.014–0.021) 178 (145–215) 14 142 80 (66–98)
2011 < 1 0.015 (0.012–0.018) 152 (124–184) 12 122 80 (66–98)
2012 < 1 0.024 (0.019–0.029) 241 (196–290) 19 193 80 (66–98)
Vanuatu 1990 < 1 0.19 (0.150–0.230) 127 (103–154) 140 95 75 (62–93)
1995 < 1 0.11 (0.085–0.130) 63 (51–76) 79 47 75 (62–93)
2000 < 1 0.2 (0.160–0.250) 110 (89–132) 152 82 75 (62–93)
2005 < 1 0.17 (0.140–0.210) 83 (68–99) 76 36 44 (37–54)
2010 < 1 0.16 (0.130–0.200) 69 (57–83) 116 49 71 (59–86)
2011 < 1 0.16 (0.130–0.190) 67 (55–80) 110 45 68 (57–83)
2012 < 1 0.16 (0.130–0.190) 65 (53–77) 125 51 78 (66–95)
Viet Nam 1990 69 170 (120–240) 251 (172–344) 50 203 73 29 (21–42)
1995 76 170 (120–220) 220 (155–295) 0.083 (0.059–0.11) 0.1 (<0.1–0.15) 55 739 73 33 (25–47)
2000 81 160 (120–210) 197 (142–260) 1.7 (1.3–2.3) 2.2 (1.6–2.9) 89 792 111 56 (43–78)
2005 85 150 (110–190) 176 (131–229) 7.6 (5.6–9.9) 8.9 (6.6–12) 94 916 112 63 (49–86)
2010 89 140 (100–180) 155 (115–201) 9.2 (6.8–12) 10 (7.6–13) 97 448 109 70 (54–95)
2011 90 140 (100–180) 151 (112–197) 9.2 (6.8–12) 10 (7.6–13) 98 804 110 73 (56–98)
2012 91 130 (99–170) 147 (109–192) 9.3 (6.9–12) 10 (7.6–13) 102 112 112 76 (59–100)
Wallis and Futuna 1990 < 1 0.022 (0.019–0.024) 156 (137–176)
Islands 1995 < 1 <0.01 (<0.01–<0.01) 49 (43–55) 6 42 87 (77–99)
2000 < 1 <0.01 (<0.01–<0.01) 15 (13–17)
2005 < 1 <0.01 (<0.01–<0.01) 57 (50–64) 7 49 87 (77–99)
2010 < 1 <0.01 (<0.01–<0.01) 36 (31–41)
2011 < 1 <0.01 (<0.01–<0.01) 17 (15–19) 2 15 87 (77–99)
2012 < 1 <0.01 (<0.01–<0.01) 65 (57–74)
a Rates are per 100 000 population.
b NOTIFIED NEW AND RELAPSE includes cases for which the treatment history is unknown.

275 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
WESTERN PACIFIC REGION
7$%/($&DVHQRWLILFDWLRQV±
NEW CASES
YEAR SMEAR-
POSITIVE
SMEAR-NEGATIVE/
UNKNOWN
EXTRA-
PULMONARY OTHER RELAPSE RE-TREAT EXCL.
RELAPSE
TOTAL
RETREAT
HISTORY
UNKNOWN
NEW AND
RELAPSE
b
% SMEAR-
POS AMONG
NEW PULM
NEW AND RELAPSE
NOTIFICATION RATE
a
1990–2012
American Samoa 1990 9 –
1995 –
2000 3 2 0 1 0 0 100
2005 6 3 2 0 0 1 0 1 0 60
2010 4 0 3 1 0 0 0 0 0 0
2011 3 0 3 0 0 0 0 0 0 0
• 19 0 • 2012 –
Australia 1990 1 016 –
1995 1 073 –
2000 1 043 251 362 369 17 17 41
2005 1 046 241 339 450 16 27 43 42
2010 1 257 274 410 457 5 41 24 65 70 40
2011 1 239 301 436 463 2 20 29 49 17 41
• 6 6 • 2012 1 305 290 408 498 63 26 20 46 20 42
Brunei 1990 143 –
Darussalam 1995 –
2000 307 84 166 42 15 15 34
2005 163 101 30 27 5 0 5 0 77
2010 237 146 30 43 13 5 0 5 0 83
2011 230 109 52 48 13 8 0 8 0 68
• 56 59 • 2012 243 119 79 31 0 14 0 14 0 60
Cambodia 1990 6 501 –
1995 14 603 11 101 1 465 1 428 605 605 88
2000 18 891 14 822 1 108 2 147 814 814 93
2005 35 535 21 001 7 057 6 759 718 588 1 306 75
2010 40 460 17 454 8 301 14 239 0 466 1 168 1 634 0 68
2011 38 555 15 812 7 686 14 690 0 367 1 115 1 482 0 67
• 72 270 • 2012 40 185 14 838 8 509 15 290 0 446 73 519 1 102 64
China 1990 375 481 –
1995 515 764 134 488 203 088 1 560 18 693 18 693 40
2000 454 372 204 765 229 943 19 664 53 480 73 144 47
2005 899 729 472 719 329 157 42 845 49 707 90 780 140 487 5 301 59
2010 908 399 429 899 432 868 6 325 0 39 307 14 909 54 216 0 50
2011 899 669 377 005 481 514 6 540 0 34 610 12 215 46 825 0 44
• 32 65 • 2012 890 645 316 332 536 050 6 479 0 31 784 10 033 41 817 0 37
China, Hong Kong 1990 6 510 –
SAR 1995 6 212 –
2000 6 015 1 940 3 115 772 188 594 782 38
2005 5 660 1 561 3 179 701 0 219 500 719 0 33
2010 4 935 1 475 2 352 792 0 316 197 513 0 39
2011 4 739 1 380 2 244 815 0 300 187 487 0 38
• 112 67 • 2012 4 809 1 463 2 206 817 0 323 160 483 0 40
China, Macao 1990 343 –
SAR 1995 402 141 94 70 49 49 60
2000 449 160 180 50 12 12 47
2005 398 136 162 43 0 14 17 31 43 46
2010 394 123 175 49 0 21 39 60 26 41
2011 380 148 126 46 0 21 2 23 39 54
• 95 73 • 2012 404 156 139 31 0 26 2 28 52 53
Cook Islands 1990 0 0 0 0 0 0 0 0 0
1995 2 2 0 0 0 0 0 0 0 100
2000 1 0 1 0 0 0 0 0 0 0
2005 1 1 0 0 0 0 0 0 0 100
2010 0 0 0 0 0 0 0 0 0
2011 1 1 0 0 0 0 0 0 0 100
• 0 5 • 2012 1 0 0 0 0 1 0 1 0
Fiji 1990 226 84 105 37 44
1995 203 68 99 34 2 0 2 41
2000 144 62 42 40 0 0 0 60
2005 132 63 29 40 68
2010 189 89 45 45 0 10 2 12 0 66
2011 215 107 62 44 0 2 5 7 0 63
• 31 24 • 2012 210 111 54 40 0 5 8 13 0 67
French Polynesia 1990 59 –
1995 –
2000 62 29 19 10 1 1 60
2005 63 21 25 14 3 0 3 0 46
2010 41 13 18 6 0 4 0 4 0 42
2011 64 22 27 13 0 2 0 2 0 45
• 30 18 • 2012 50 26 10 8 0 6 0 6 0 72
Guam 1990 –
1995 –
2000 54 43 5 6 1 1 90
2005 63 27 26 9 0 1 1 2 0 51
2010 101 39 51 9 0 2 0 2 0 43
2011 82 28 39 11 0 3 0 3 1 42
• 0 42 • 2012 68 23 37 8 0 0 0 0 0 38
Japan 1990 51 821 –
1995 43 078 14 367 25 172 2 803 736 736 36
2000 39 384 11 853 19 118 7 046 1 367 1 367 38
2005 27 194 10 931 10 056 5 340 867 1 125 1 992 52
2010 22 693 8 237 8 630 4 632 0 1 194 568 1 762 49
2011 22 119 7 937 8 231 4 826 0 1 125 562 1 687 0 49
• 42 16 • 2012 20 857 7 663 7 675 4 609 0 910 426 1 336 50
Kiribati 1990 68 –
1995 –
2000 252 54 47 106 3 3 53
2005 332 124 79 126 3 7 10 61
2010 286 118 91 71 0 6 8 14 0 56
2011 343 140 109 87 0 7 11 18 0 56
• 96 343 • 2012 346 134 122 73 9 8 2 10 0 52
Lao People's 1990 1 826 –
Democratic 1995 830 478 404 95 2 2 54
Republic 2000 2 227 1 526 457 180 64 64 77
2005 3 766 2 801 484 275 139 41 180 67 85
2010 4 061 3 119 394 323 163 22 185 62 89
2011 4 360 3 271 516 349 170 27 197 54 86
• 43 62 • 2012 4 118 3 062 484 351 168 38 206 53 86
Malaysia 1990 11 702 –
1995 11 778 6 688 4 021 1 069 210 210 62
2000 15 057 8 156 5 517 1 384 0 0 60
2005 15 415 8 446 4 862 1 702 0 332 651 983 73 63
2010 18 517 11 135 4 338 2 545 0 499 820 1 319 0 72
2011 19 808 11 862 4 501 2 888 0 557 858 1 415 0 72
• 64 75 • 2012 21 851 13 311 4 993 2 945 0 602 859 1 461 0 73
a Rates are per 100 000 population.
b NEW AND RELAPSE includes cases for which the treatment history is unknown.

276 GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($&DVHQRWLILFDWLRQV±
NEW CASES
YEAR SMEAR-
POSITIVE
SMEAR-NEGATIVE/
UNKNOWN
EXTRA-
PULMONARY OTHER RELAPSE RE-TREAT EXCL.
RELAPSE
TOTAL
RETREAT
HISTORY
UNKNOWN
NEW AND
RELAPSE
b
% SMEAR-
POS AMONG
NEW PULM
NEW AND RELAPSE
NOTIFICATION RATE
a
1990–2012
Marshall Islands 1990 –
1995 –
2000 34 11 25 9 0 0 31
2005 111 48 31 28 4 1 5 0 61
2010 193 59 64 65 0 2 8 10 3 48
2011 139 44 30 57 0 8 12 20 0 59
• 0 276 • 2012 145 54 53 29 0 4 2 6 5 50
Micronesia 1990 367 –
(Federated 1995 172 9 79 18 2 2 10
States of) 2000 91 15 69 4 3 3 18
2005 98 32 35 19 5 7 14 21 48
2010 164 53 79 25 0 3 10 13 4 40
2011 148 45 73 28 0 2 2 4 0 38
• 381 139 • 2012 144 43 77 22 0 2 2 4 0 36
Mongolia 1990 1 659 –
1995 2 780 455 1 330 976 82 82 25
2000 3 109 1 389 732 862 126 126 65
2005 4 601 1 868 897 1 620 0 216 125 341 0 68
2010 4 458 1 837 701 1 675 0 245 343 588 0 72
2011 4 217 1 723 684 1 578 0 232 316 548 0 72
• 76 148 • 2012 4 128 1 716 617 1 611 0 184 325 509 0 74
Nauru 1990 7 –
1995 –
2000 4 4 0 0 0 0 100
2005 11 0 11 0
2010 3 1 1 1 0 0 0 0 0 50
2011 5 3 1 1 0 0 0 0 0 75
• 76 0 • 2012 –
New Caledonia 1990 143 –
1995 87 21 81 9 4 4 21
2000 94 20 15 29 4 4 57
2005 47 16 15 15 1 6 7 0 52
2010 49 20 16 13 0 8 8 0 56
2011 52 13 18 19 0 2 0 2 0 42
• 85 15 • 2012 38 13 11 12 1 1 0 1 0 54
New Zealand 1990 348 –
1995 391 78 222 34 4 4 26
2000 344 74 133 130 7 0 7 36
2005 332 83 114 95 29 11 8 19 42
2010 301 86 68 134 6 7 4 11 56
2011 305 88 81 121 13 2 4 6 52
• 10 7 • 2012 293 68 99 112 3 11 4 15 0 41
Niue 1990 0 –
1995 0 0 1 0 0 0
2000 0 0 0
2005 0 0
2010 0 0 0 0 0 0 0 0 0
2011 1 0 0 0 1 0 0 0 0
• 0 0 • 2012 0 0 0 0 0 0 0 0 0
Northern Mariana 1990 28 –
Islands 1995 48 14 26 8 0 0 35
2000 75 27 37 11 0 0 42
2005 57 15 35 7 0 0 0 0 0 30
2010 32 17 13 2 0 0 0 0 0 57
2011 33 15 16 2 0 0 0 0 0 48
• 64 60 • 2012 32 10 17 4 1 0 2 2 0 37
Palau 1990 –
1995 19 9 6 4 0 0 60
2000 –
2005 10 3 6 1 0 0 0 33
2010 19 9 10 0 0 0 0 0 0 47
2011 12 4 6 1 0 1 0 1 0 40
• 0 19 • 2012 4 3 1 0 0 0 0 0 0 75
Papua New 1990 2 497 –
Guinea 1995 8 041 1 652 3 767 2 349 273 273 30
2000 10 520 1 933 4 405 3 227 955 955 30
2005 12 564 1 805 5 105 4 198 1 456 1 456 26
2010 14 531 2 584 5 907 5 798 242 1 582 1 824 30
2011 14 893 1 882 6 494 6 373 0 144 1 431 1 575 0 22
• 60 287 • 2012 20 557 2 862 9 195 8 277 0 223 1 931 2 154 0 24
Philippines 1990 317 008 –
1995 119 186 94 768 140 712 8 8 8 40
2000 119 914 67 056 52 858 56
2005 137 100 81 647 50 347 1 149 0 3 957 3 957 62
2010 166 323 89 198 72 440 1 610 0 3 075 8 066 11 141 0 55
2011 195 560 93 580 96 529 2 234 0 3 217 10 528 13 745 0 49
• 512 224 • 2012 216 627 94 006 115 263 3 274 0 4 084 13 535 17 619 0 45
Republic of Korea 1990 63 904 –
1995 42 117 11 754 19 360 2 082 2 082 38
2000 21 782 8 216 11 304 2 262 2 262 42
2005 42 892 11 638 18 460 5 171 0 3 021 4 077 7 098 4 602 39
2010 44 063 11 596 18 660 8 795 0 2 838 4 038 6 876 2 174 38
2011 46 253 11 714 18 386 9 457 0 3 032 4 238 7 270 3 664 39
• 149 89 • 2012 43 702 12 137 18 938 8 470 0 4 157 5 830 9 987 0 39
Samoa 1990 44 –
1995 45 15 30 6 0 0 33
2000 43 13 18 12 0 0 42
2005 24 11 8 5 0 0 0 0 0 58
2010 14 6 5 3 0 0 0 0 0 55
2011 20 6 12 2 0 0 0 0 33
• 27 12 • 2012 22 15 4 3 0 0 0 0 0 79
Singapore 1990 1 591 –
1995 1 889 455 1 187 127 120 120 28
2000 1 728 248 869 165 55 55 22
2005 1 376 552 570 174 0 60 93 153 20 49
2010 1 560 530 735 213 0 82 48 130 0 42
2011 1 641 592 717 224 0 108 54 162 0 45
• 53 43 • 2012 2 301 678 1 219 306 0 98 63 161 0 36
Solomon Islands 1990 382 –
1995 352 109 133 97 13 13 45
2000 302 109 128 65 0 0 46
2005 397 169 161 62 0 5 0 5 0 51
2010 338 133 98 105 0 2 3 5 0 58
2011 398 159 108 127 0 4 7 11 0 60
• 122 66 • 2012 361 157 87 112 0 5 11 16 0 64
a Rates are per 100 000 population.
b NEW AND RELAPSE includes cases for which the treatment history is unknown.
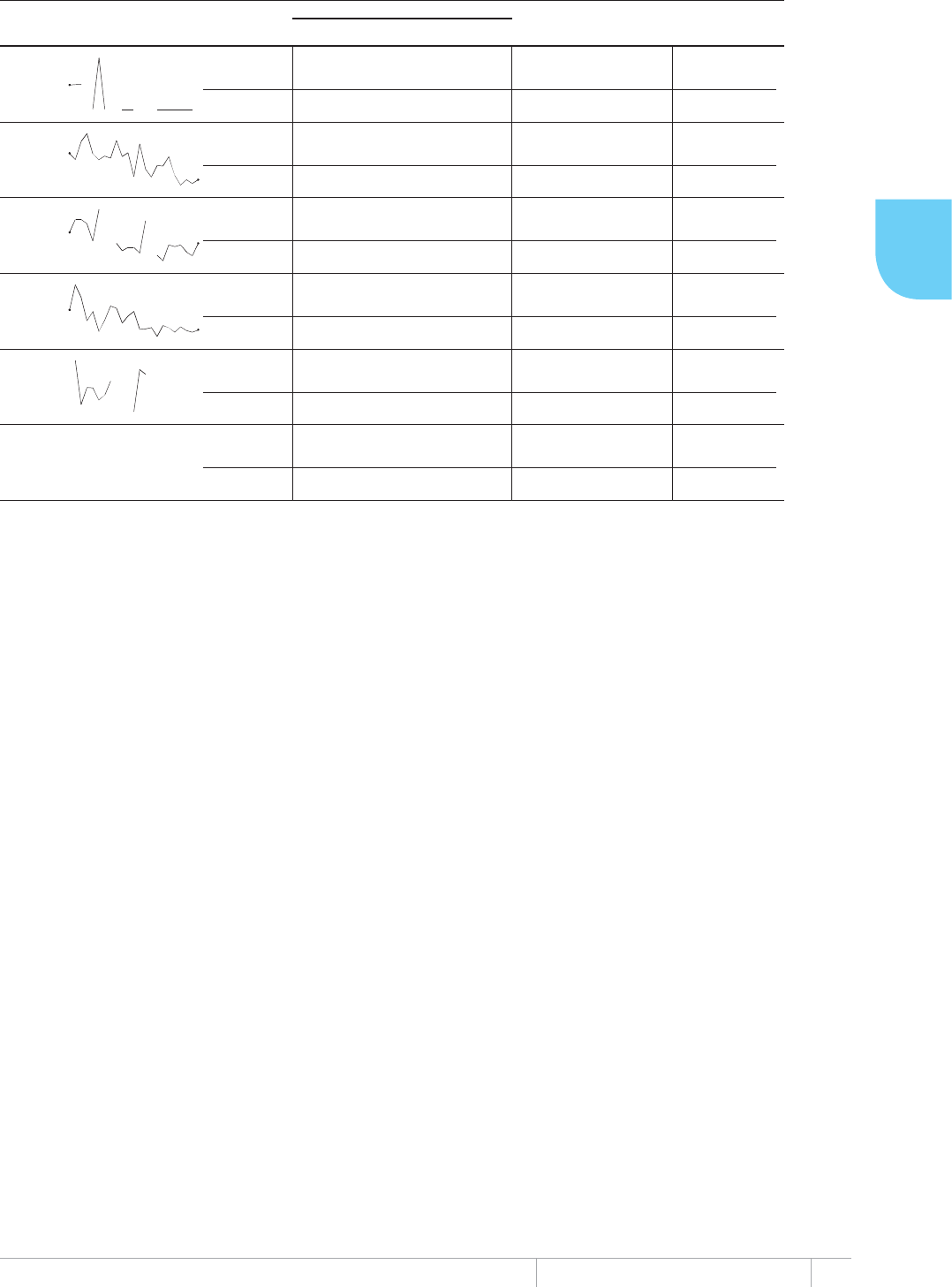
277 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
WESTERN PACIFIC REGION
7$%/($&DVHQRWLILFDWLRQV±
NEW CASES
YEAR SMEAR-
POSITIVE
SMEAR-NEGATIVE/
UNKNOWN
EXTRA-
PULMONARY OTHER RELAPSE RE-TREAT EXCL.
RELAPSE
TOTAL
RETREAT
HISTORY
UNKNOWN
NEW AND
RELAPSE
b
% SMEAR-
POS AMONG
NEW PULM
NEW AND RELAPSE
NOTIFICATION RATE
a
1990–2012
Tokelau 1990 1 –
1995 2 1 1 0 0 0 50
2000 0 0 0 0 0 0
2005 0 0 0 0 0 0 0 0 0
2010 0 0 0 0 0 0 0 0 0
2011 0 0 0 0 0 0 0 0 0
• 62 0 • 2012 –
Tonga 1990 23 –
1995 20 9 2 9 0 0 82
2000 24 15 5 3 1 1 75
2005 18 11 3 4 79
2010 11 6 3 2 0 0 0 0 0 67
2011 9 6 3 0 0 0 0 0 0 67
• 24 10 • 2012 11 9 1 1 0 0 0 0 0 90
Tuvalu 1990 23 –
1995 36 6 13 16 1 1 32
2000 16 0 7 7 0
2005 12 5 3 4 3 3 62
2010 14 5 2 7 0 0 0 0 0 71
2011 12 4 4 4 0 0 1 1 0 50
• 255 193 • 2012 19 8 2 9 0 0 1 1 0 80
Vanuatu 1990 140 –
1995 79 30 27 21 1 1 53
2000 152 63 56 28 5 5 53
2005 76 35 21 17 0 3 5 8 0 62
2010 116 44 33 35 3 1 0 1 0 57
2011 110 49 14 46 0 1 2 3 0 78
• 95 51 • 2012 125 51 22 51 0 1 1 2 0 70
Viet Nam 1990 50 203 –
1995 55 739 37 550 8 379 6 194 3 616 3 616 82
2000 89 792 53 169 17 993 13 137 5 493 5 493 75
2005 94 916 55 492 16 429 16 670 0 6 325 976 7 301 0 77
2010 97 448 52 145 18 237 17 651 0 6 834 1 574 8 408 2 581 74
2011 98 804 50 751 20 373 18 077 2 678 6 925 1 714 8 639 71
• 0 0 • 2012 102 112 51 033 21 706 18 904 3 210 7 259 1 794 9 053 70
Wallis and Futuna 1990 –
Islands 1995 6 3 2 0 1 1 60
2000 –
2005 7 1 6 14
2010 –
2011 2 2 0 0 0 0 0 0 0 100
• 0 0 • 2012 –
a Rates are per 100 000 population.
b NEW AND RELAPSE includes cases for which the treatment history is unknown.

GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($7UHDWPHQWRXWFRPHVQHZVPHDUSRVLWLYHFDVHV±
% OF COHORT
TREATMENT SUCCESS (%)
a
1995–2011 YEAR NUMBER
NOTIFIED
SIZE OF
COHORT
COHORT AS
% NOTIFIED CURED COMPLETED DIED FAILED DEFAULTED NOT
EVALUATED
American Samoa 1995 4 – 10000000
2000 2 2 100 0 100 0 0 0 0
2005 3 4 133 75 25
2009 0 3 – 0 100 0 0 0 0
2010 0 0 –
• 100 0 • 2011 0 –
Australia 1995 –
2000 251 238 95 27 45 9 0 3 16
2005 241 241 100 12 68 10 2 8
2009 267 606 227 6 73 3 0 1 16
2010 274 629 230 8 72 3 0 2 15
• 0 77 • 2011 301 527 175 7 70 6 0 1 16
Brunei 1995 –
Darussalam 2000 84 84 100 42 21 17 0 4 17
2005 101 101 100 66 5 7 0 2 20
2009 140 164 117 63 8 9 0 0 20
2010 146 176 121 61 20 7 0 0 12
• 0 66 • 2011 109 109 100 66 0 9 0 0 25
Cambodia 1995 11 101 4 363 39 83 8 2 1 4 2
2000 14 822 14 775 100 88 4 4 0 4 1
2005 21 001 21 001 100 89 4 3 0 2 2
2009 17 863 17 863 100 92 3 2 0 1 1
2010 17 454 17 454 100 91 3 2 0 1 2
• 91 93 • 2011 15 812 15 884 100 90 4 2 0 1 3
China 1995 134 488 131 413 98 72 22 2 1 1 3
2000 204 765 213 766 104 93 1 2 1 3
2005 472 719 472 719 100 92 2 2 1 1 3
2009 449 152 449 039 100 93 2 1 1 1 2
2010 429 899 429 790 100 94 2 1 1 0 2
• 93 95 • 2011 377 005 377 005 100 94 2 1 1 1 2
China, Hong Kong 1995 –
SAR 2000 1 940 1 940 100 55 5 5 6 4 24
2005 1 561 1 561 100 60 3 5 9 3 20
2009 1 444 1 441 100 59 11 15 0 3 12
2010 1 475 1 487 101 57 11 15 0 4 13
• 0 69 • 2011 1 380 1 378 100 59 10 14 0 4 13
China, Macao 1995 141 –
SAR 2000 160 160 100 81 8 6 0 4 1
2005 136 136 100 93 0 4 0 1 3
2009 116 115 99 86 2 3 0 2 7
2010 123 219 178 93 0 3 0 1 3
• 0 86 • 2011 148 147 99 86 0 5 0 1 7
Cook Islands 1995 2 2 100 100 0 0 0 0 0
2000 0 –
2005 1 1 100 100 0 0 0 0 0
2009 1 0 0
2010 0 0 –
• 100 0 • 2011 1 1 100 0 0 0 0 0 100
Fiji 1995 68 73 107 78 8 7 0 3 4
2000 62 62 100 81 5 5 0 8 2
2005 63 68 108 71 0 10 0 10 9
2009 83 79 95 89 5 4 0 1 1
2010 89 89 100 65 2 6 0 24 3
• 86 93 • 2011 107 107 100 81 12 1 0 3 3
French Polynesia 1995 33 – 67030219
2000 29 62 214 0 97 2 2 0 0
2005 21 18 86 89 11 0 0 0
2009 17 18 106 89 6 0 6 0
2010 13 13 100 92 0 8 0 0 0
• 67 80 • 2011 22 20 91 80 0 5 0 15 0
Guam 1995 –
2000 43 43 100 93 0 7 0 0 0
2005 27 27 100 85 0 11 0 0 4
2009 31 47 152 96 0 2 0 0 2
2010 39 51 131 84 0 16 0 0 0
• 0 79 • 2011 28 28 100 79 0 14 0 0 7
Japan 1995 14 367 –
2000 11 853 10 348 87 30 15 5 4 1 44
2005 10 931 10 931 100 38 22 11 3 1 26
2009 8 853 8 772 99 21 31 19 1 4 24
2010 8 237 8 242 100 20 32 21 1 3 24
• 0 0 • 2011 7 937 –
Kiribati 1995 31 – 45 42 13 0
2000 54 54 100 83 7 7 2 0 0
2005 124 123 99 62 31 7 0 1 0
2009 145 144 99 84 13 3 0 0 0
2010 118 117 99 88 5 5 2 0 0
• 87 94 • 2011 140 140 100 74 21 4 1 1 0
Lao People's 1995 478 343 72 62 8 6 2 19 4
Democratic 2000 1 526 1 588 104 68 9 7 0 9 7
Republic 2005 2 801 2 802 100 85 5 5 1 3 1
2009 3 034 3 034 100 91 2 4 1 2 1
2010 3 119 3 119 100 89 3 6 0 2 0
• 70 92 • 2011 3 271 3 271 100 87 5 0 5 2 1
Malaysia 1995 6 688 13 398 200 69 0 6 2 8 14
2000 8 156 7 915 97 0 78 8 0 10 4
2005 8 446 8 446 100 69 1 9 0 5 16
2009 9 981 9 981 100 78 1 9 0 4 9
2010 11 135 11 135 100 79 1 9 0 4 7
• 69 79 • 2011 11 862 11 862 100 78 1 9 0 4 8
Marshall Islands 1995 163 – 3 21 7 0 67 1
2000 11 11 100 64 27 0 0 9 0
2005 48 47 98 85 2 2 2 9
2009 52 58 112 71 14 9 0 3 3
2010 59 71 120 63 17 8 0 1 10
• 25 88 • 2011 44 50 114 86 2 6 0 6
Micronesia 1995 9 10 111 80 0 10 0 10 0
(Federated 2000 15 14 93 93 0 7 0 0 0
States of) 2005 32 20 62 75 5 10 5 0 5
2009 61 60 98 65 23 3 2 0 7
2010 53 59 111 97 0 3 0 0 0
• 80 96 • 2011 45 51 113 80 16 4 0 0 0
Mongolia 1995 455 455 100 66 7 8 6 10 2
2000 1 389 1 389 100 83 4 3 3 4 3
2005 1 868 1 868 100 82 6 3 5 3 2
2009 1 809 1 809 100 84 4 2 7 2 0
2010 1 837 1 837 100 83 3 2 8 3 0
• 74 86 • 2011 1 723 1 723 100 82 3 2 7 4 1
a
TREATMENT SUCCESS = percent cured + percent completed then rounded to the nearest digit.

279 Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
WESTERN PACIFIC REGION
7$%/($7UHDWPHQWRXWFRPHVQHZVPHDUSRVLWLYHFDVHV±
% OF COHORT
TREATMENT SUCCESS (%)
a
1995–2011 YEAR NUMBER
NOTIFIED
SIZE OF
COHORT
COHORT AS
% NOTIFIED CURED COMPLETED DIED FAILED DEFAULTED NOT
EVALUATED
Nauru 1995 –
2000 4 4 100 25 75
2005 0 3 – 0 67 33 0 0 0
2009 1 0 0
2010 1 3 300 0 67 0 0 0 33
• 0 0 • 2011 3 –
New Caledonia 1995 21 32 152 75 12 3 9
2000 20 45 225 33 56 9 2 0
2005 16 16 100 88 6 6 0 0 0
2009 15 15 100 0 93 0 0 7 0
2010 20 21 105 0 76 19 0 5 0
• 75 35 • 2011 13 23 177 0 35 9 0 0 57
New Zealand 1995 78 –
2000 74 73 99 5 25 23 47
2005 83 84 101 0 60 6 0 1 33
2009 90 92 102 76 7 1 16
2010 86 86 100 74 17 8
• 0 56 • 2011 88 141 160 0 56 7 0 1 36
Niue 1995 0 –
2000 0 –
2005 0 0 –
2009 0 –
2010 0 0 –
• 0 0 • 2011 0 –
Northern Mariana 1995 14 –
Islands 2000 27 27 100 81 0 0 0 0 19
2005 15 15 100 73 0 0 0 0 27
2009 16 16 100 0 81 0 0 0 19
2010 17 17 100 0 82 0 0 0 18
• 0 89 • 2011 15 19 127 89 11 0 0 0
Palau 1995 9 9 100 56 11 0 0 11 22
2000 –
2005 3 3 100 100 0 0 0 0 0
2009 6 8 133 62 12 25 0 0 0
2010 9 16 178 75 12 12 0 0 0
• 67 57 • 2011 4 7 175 29 29 14 0 14 14
Papua New 1995 1 652 4 904 297 56 4 0 15 25
Guinea 2000 1 933 422 22 39 24 2 0 26 9
2005 1 805 1 292 72 57 14 4 1 19 5
2009 2 238 2 584 115 58 13 4 2 16 6
2010 2 584 2 530 98 48 10 3 2 14 23
• 56 69 • 2011 1 882 2 322 123 53 16 4 3 19 5
Philippines 1995 94 768 90 297 95 54 6 1 1 5 34
2000 67 056 50 196 75 73 15 2 1 6 3
2005 81 647 81 125 99 82 7 2 1 4 3
2009 88 806 88 806 100 82 7 2 1 4 4
2010 89 198 89 198 100 85 7 2 1 4 2
• 60 90 • 2011 93 580 93 580 100 83 7 2 1 4 3
Republic of Korea 1995 11 754 11 675 99 74 2 2 3 5 14
2000 8 216 3 231 39 81 2 2 1 3 12
2005 11 638 3 752 32 81 2 1 1 4 11
2009 11 285 3 813 34 81 2 1 1 3 12
2010 11 596 2 828 24 85 4 1 0 3 6
• 76 80 • 2011 11 714 2 288 20 78 2 1 0 3 16
Samoa 1995 15 15 100 13 67 20 0 0 0
2000 13 13 100 85 8 8 0 0 0
2005 11 11 100 91 0 9 0 0 0
2009 8 10 125 90 0 10 0 0 0
2010 6 6 100 100 0 0 0 0 0
• 80 83 • 2011 6 6 100 83 0 17 0 0 0
Singapore 1995 455 122 27 71 15 2 0 11 0
2000 248 242 98 71 14 0 14 0
2005 552 548 99 83 14 0 2 1
2009 552 937 170 65 17 15 0 1 2
2010 530 948 179 62 17 17 0 1 3
• 86 83 • 2011 592 979 165 69 14 15 0 1 1
Solomon Islands 1995 109 368 338 65 6 0 4 26
2000 109 109 100 73 7 5 0 4 11
2005 169 169 100 56 30 8 0 4 2
2009 138 138 100 67 22 4 1 3 3
2010 133 133 100 57 30 1 3 5 4
• 65 90 • 2011 159 156 98 54 36 3 3 1 4
Tokelau 1995 1 –
2000 0 –
2005 0 –
2009 0 0 –
2010 0 –
• 0 0 • 2011 0 –
Tonga 1995 9 20 222 75 0 10 5 0 10
2000 15 15 100 93 0 0 7 0 0
2005 11 11 100 73 0 18 0 0 9
2009 6 6 100 83 0 17 0 0 0
2010 6 6 100 83 0 17 0 0 0
• 75 100 • 2011 6 6 100 100 0 0 0 0 0
Tuvalu 1995 6 –
2000 0 7 – 86 14 0
2005 5 6120 10000000
2009 8 8 100 88 0 0 0 0 12
2010 5 5 100 100 0
• 0 75 • 2011 4 4 100 75 25 0
Vanuatu 1995 30 13 43 38 46 15 0 0 0
2000 63 26 41 77 12 8 0 4 0
2005 35 42 120 64 17 10 7 2 0
2009 47 47 100 81 15 4 0 0 0
2010 44 44 100 66 14 16 0 2 2
• 85 82 • 2011 49 49 100 53 29 10 4 2 2
Viet Nam 1995 37 550 38 189 102 84 5 3 2 4 2
2000 53 169 53 169 100 90 2 3 1 2 2
2005 55 492 55 492 100 90 2 3 1 1 2
2009 51 291 51 387 100 90 2 3 1 2 2
2010 52 145 52 147 100 91 2 3 1 2 2
• 89 93 • 2011 50 751 50 751 100 91 2 3 1 2 2
Wallis and Futuna 1995 3 –
Islands 2000 –
2005 1 –
2009 2 –
2010 2 – 0 100 0 0 0 0
• 0 0 • 2011 2 –
a
TREATMENT SUCCESS = percent cured + percent completed then rounded to the nearest digit.

GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($7UHDWPHQWRXWFRPHVUHWUHDWPHQWFDVHV±
% OF COHORT
TREATMENT SUCCESS (%)
a
1995–2011 YEAR NUMBER
NOTIFIED
SIZE OF
COHORT
COHORT AS
% NOTIFIED CURED COMPLETED DIED FAILED DEFAULTED NOT
EVALUATED
American Samoa 1995 –
2000 0 –
2005 1 1 100 100 0
2009 0 0 –
2010 0 0 –
• 0 0 • 2011 0 –
Australia 1995 –
2000 17 11 65 9 73 9 0 0 9
2005 43 43 100 16 56 5 5 19
2009 61 65 107 6 60 3 2 8 22
2010 65 58 89 5 64 5 0 3 22
• 0 66 • 2011 49 67 137 1 64 6 0 3 25
Brunei 1995 –
Darussalam 2000 15 –
2005 5 5 100 40 40 20 0 0 0
2009 0 0 –
2010 5 5100 10000000
• 0 63 • 2011 8 8 100 62 0 25 0 0 12
Cambodia 1995 605 436 72 59 26 5 3 3 4
2000 814 827 102 85 5 6 1 4 0
2005 1 306 1 306 100 49 27 9 2 3 11
2009 1 429 1 429 100 34 45 3 1 1 15
2010 1 634 1 524 93 30 44 4 1 1 20
• 85 74 • 2011 1 482 409 28 66 8 7 5 5 10
China 1995 18 693 54 052 289 90 2 2 3 1 1
2000 73 144 43 252 59 86 2 1 1 1 8
2005 140 487 89 239 64 85 5 3 3 1 4
2009 59 583 59 853 100 86 4 2 2 1 4
2010 54 216 54 469 100 86 4 2 2 1 5
• 92 90 • 2011 46 825 46 825 100 87 4 2 3 1 4
China, Hong Kong 1995 –
SAR 2000 782 218 28 27 26 4 17 18 8
2005 719 716 100 40 18 4 9 7 22
2009 509 481 94 26 38 15 0 6 14
2010 513 512 100 34 34 12 0 4 16
• 0 62 • 2011 487 453 93 27 35 15 0 7 16
China, Macao 1995 49 –
SAR 2000 12 37 308 68 16 11 0 5 0
2005 31 37 119 51 24 11 0 0 14
2009 45 46 102 43 35 11 0 7 4
2010 60 35 58 51 14 14 0 11 9
• 0 96 • 2011 23 28 122 79 18 4 0 0 0
Cook Islands 1995 0 –
2000 0 –
2005 0 0 –
2009 0 0 –
2010 0 0 –
• 0 0 • 2011 0 0 –
Fiji 1995 2 –
2000 0 –
2005 0 –
2009 2 5 250 40 40 20 0 0 0
2010 12 12 100 50 17 17 0 17 0
• 0 57 • 2011 7 7 100 29 29 14 14 14 0
French Polynesia 1995 2 – 50 0 50 0 0 0
2000 1 –
2005 3 4 133 75 25 0
2009 5 5 100 0 100 0 0 0 0
2010 4 4 100 0 75 25 0 0 0
• 50 100 • 2011 2 4 200 75 25 0 0 0 0
Guam 1995 –
2000 1 –
2005 2 2 100 50 0 0 0 50 0
2009 1 1 100 100 0 0 0 0 0
2010 2 2 100 100 0 0 0 0 0
• 0 100 • 2011 3 3 100 67 33 0 0 0 0
Japan 1995 736 –
2000 1 367 1 169 86 31 15 5 6 1 41
2005 1 992 1 992 100 29 16 8 2 2 43
2009 1 751 1 452 83 15 32 15 1 6 31
2010 1 762 1 466 83 14 32 17 1 5 31
• 0 0 • 2011 1 687 –
Kiribati 1995 –
2000 3 9 300 89 0 11 0 0 0
2005 10 3 30 100 0
2009 4 6 150 83 17 0 0 0 0
2010 14 20 143 25 45 30 0 0 0
• 0 74 • 2011 18 19 106 21 53 5 0 21 0
Lao People's 1995 2 1 50 100 0 0 0 0 0
Democratic 2000 64 64 100 41 8 11 8 11 22
Republic 2005 180 181 101 75 12 6 2 5 1
2009 184 184 100 85 3 8 2 1 0
2010 185 184 99 76 7 12 3 3 0
• 100 81 • 2011 197 170 86 72 9 2 8 3 6
Malaysia 1995 210 –
2000 0 –
2005 983 1 056 107 46 9 8 1 9 27
2009 1 181 1 181 100 33 27 9 1 6 23
2010 1 319 1 319 100 35 24 12 1 12 17
• 0 54 • 2011 1 415 1 415 100 34 20 9 1 8 28
Marshall Islands 1995 –
2000 0 –
2005 5 20 400 60 10 30
2009 2 8 400 12 75 0 0 12 0
2010 10 4 40 25 25 50 0 0 0
• 0 100 • 2011 20 20 100 30 70 0 0 0 0
Micronesia 1995 2 9450 10000000
(Federated 2000 3 20 667 25 60 5 10 0 0
States of) 2005 21 9 43 11 89 0
2009 9 16 178 0 19 75 0 0 6
2010 13 10 77 20 10 10 0 20 40
• 100 100 • 2011 4 1 25 0 100 0 0 0 0
Mongolia 1995 82 23 28 61 0 9 13 13 4
2000 126 126 100 57 14 8 8 7 6
2005 341 443 130 39 34 9 11 4 3
2009 569 380 67 60 13 4 17 4 2
2010 588 234 40 19 61 9 6 2 4
• 61 74 • 2011 548 548 100 39 35 5 15 4 2
a
TREATMENT SUCCESS = percent cured + percent completed then rounded to the nearest digit.

Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
WESTERN PACIFIC REGION
7$%/($7UHDWPHQWRXWFRPHVUHWUHDWPHQWFDVHV±
% OF COHORT
TREATMENT SUCCESS (%)
a
1995–2011 YEAR NUMBER
NOTIFIED
SIZE OF
COHORT
COHORT AS
% NOTIFIED CURED COMPLETED DIED FAILED DEFAULTED NOT
EVALUATED
Nauru 1995 –
2000 0 –
2005 0 –
2009 0 1 – 100 0
2010 0 0 –
• 0 0 • 2011 0 –
New Caledonia 1995 4 4 100 100 0
2000 4 –
2005 7 7 100 86 0 14 0 0 0
2009 9 9 100 0 89 0 0 0 11
2010 8 8 100 0 88 12 0 0 0
• 100 0 • 2011 2 1 50 0 0 100 0 0 0
New Zealand 1995 4 –
2000 7 23 329 0 30 4 65
2005 19 18 95 0 67 0 0 0 33
2009 9 9 100 67 11 22
2010 11 11 100 73 18 9
• 0 50 • 2011 6 6 100 0 50 0 0 0 50
Niue 1995 0 –
2000 –
2005 0 –
2009 –
2010 0 0 –
• 0 0 • 2011 0 –
Northern Mariana 1995 0 –
Islands 2000 0 –
2005 0 0 –
2009 0 0 –
2010 0 0 –
• 0 0 • 2011 0 0 –
Palau 1995 0 –
2000 –
2005 0 0 –
2009 0 0 –
2010 0 0 –
• 0 0 • 2011 1 0 0
Papua New 1995 273 –
Guinea 2000 955 68 7 29 35 4 1 21 9
2005 1 456 65 4 42 14 15 6 20 3
2009 1 388 530 38 36 22 5 5 29 3
2010 1 824 444 24 35 11 5 5 18 27
• 0 52 • 2011 1 575 398 25 32 20 7 5 22 14
Philippines 1995 8 –
2000 –
2005 3 957 –
2009 9 575 4 362 46 48 13 4 4 5 26
2010 11 141 4 554 41 53 15 5 5 6 16
• 0 65 • 2011 13 745 4 583 33 47 18 5 4 6 20
Republic of Korea 1995 2 082 2 004 96 39 1 1 2 3 53
2000 2 262 131 6 59 2 3 3 12 21
2005 7 098 3 331 47 72 3 2 0 6 18
2009 6 880 2 420 35 69 3 2 1 5 21
2010 6 876 1 813 26 76 4 2 0 6 12
• 40 74 • 2011 7 270 1 346 19 70 3 1 0 5 19
Samoa 1995 0 –
2000 0 –
2005 0 0 –
2009 0 0 –
2010 0 0 –
• 0 0 • 2011 0 0 –
Singapore 1995 120 –
2000 55 –
2005 153 149 97 79 15 0 5 1
2009 132 130 98 37 39 20 0 1 3
2010 130 127 98 47 31 17 0 2 3
• 0 76 • 2011 162 160 99 43 33 22 0 2 0
Solomon Islands 1995 13 –
2000 0 –
2005 5 5 100 20 40 20 20 0 0
2009 2 2 100 50 50 0 0 0 0
2010 5 5 100 80 0 20 0 0 0
• 0 100 • 2011 11 10 91 30 70 0 0 0 0
Tokelau 1995 0 –
2000 0 –
2005 0 –
2009 0 0 –
2010 0 –
• 0 0 • 2011 0 –
Tonga 1995 0 9 – 10000000
2000 1 1 100 100 0
2005 0 –
2009 0 0 –
2010 0 0 –
• 100 0 • 2011 0 0 –
Tuvalu 1995 1 –
2000 –
2005 3 0 0
2009 0 0 –
2010 0 0 –
• 0 0 • 2011 1 0 0
Vanuatu 1995 1 –
2000 5 5 100 100 0 0 0 0 0
2005 8 0 0
2009 3 3 100 100 0 0 0 0 0
2010 1 1 100 100 0 0 0 0 0
• 0 100 • 2011 3 3 100 67 33 0 0 0 0
Viet Nam 1995 3 616 2 384 66 80 2 5 8 2 4
2000 5 493 8 806 160 74 5 6 5 3 7
2005 7 301 7 374 101 79 4 5 6 3 3
2009 8 131 357 4 67 6 8 2 10 7
2010 8 408 398 5 61 8 8 4 12 6
• 81 82 • 2011 8 639 8 641 100 79 3 5 5 3 5
Wallis and Futuna 1995 1 –
Islands 2000 –
2005 –
2009 0 –
2010 0 –
• 0 0 • 2011 0 –
a
TREATMENT SUCCESS = percent cured + percent completed then rounded to the nearest digit.

GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($+,9WHVWLQJDQGSURYLVLRQRI&37$57DQG,37–
% OF TB PATIENTS WITH
KNOWN HIV STATUS
2005–2012
YEAR
% OF TB
PATIENTS WITH
KNOWN HIV
STATUS
NUMBER OF TB
PATIENTS WITH
KNOWN HIV
STATUS
PATIENTS
NOTIFIED
(NEW AND
RETREAT)
NUMBER OF
HIV-POSITIVE
TB PATIENTS
% OF TESTED
TB PATIENTS
HIV-POSITIVE
% OF HIV-
POSITIVE TB
PATIENTS ON
CPT
% OF HIV-
POSITIVE TB
PATIENTS ON
ART
NUMBER OF
HIV-POSITIVE
PEOPLE
PROVIDED IPT
American Samoa 2005 0 0 6
2010 75 3 4 0 0
2011 100 3 3 0 0
• 0 – 2012
Australia 2005 42 448 1 073 22 4.9 9.1 0
2010 54 686 1 281 24 3.5
2011 59 750 1 268 19 2.5
• 42 56 • 2012 56 740 1 325 8 1.1
Brunei 2005 100 163 163 2 1.2 0 0
Darussalam 2010 100 237 237 1 0.42 100 100 2
2011 100 230 230 3 1.3 100 100 0
• 100 100 • 2012 100 243 243 2 0.82 100 100 0
Cambodia 2005 2.9 1 044 36 123 86 8.2
2010 77 32 236 41 628 2 112 6.6 65 45 491
2011 82 32 544 39 670 1 656 5.1 88 79 1 305
• 3 80 • 2012 80 32 359 40 258 1 433 4.4 98 88 1 145
China 2005 990 509
2010 16 145 919 923 308 4 542 3.1 45
2011 23 208 681 911 884 4 715 2.3 36
– 34 • 2012 34 309 385 900 678 5 866 1.9 59
China, Hong Kong 2005 68 4 209 6 160 35 0.83 49 54
SAR 2010 75 3 833 5 132 24 0.63 17 29
2011 74 3 656 4 926 28 0.77
• 68 75 • 2012 75 3 707 4 969 22 0.59
China, Macao 2005 91 378 415 1 0.26 0 100
SAR 2010 92 399 433 3 0.75 33 33
2011 94 360 382 2 0.56 50 50
• 91 89 • 2012 89 360 406 4 1.1 0 25
Cook Islands 2005 0 0 1 0
2010 0 0 0
2011 100 1 1 0 0
• 0 100 • 2012 100 1 1 0 0
Fiji 2005 100 132 132 1 0.76 0 0
2010 82 157 191 3 1.9 100 100 0
2011 73 160 220 3 1.9 100 100
• 100 58 • 2012 58 127 218 5 3.9 100 60 1
French Polynesia 2005 48 30 63 0 0
2010 27 11 41 0 0
2011 27 17 64 1 5.9 100 100
• 48 44 • 2012 44 22 50 0 0
Guam 2005 72 46 64 0 0
2010 62 63 101 1 1.6 100 100
2011 65 53 82 0 0
• 72 68 • 2012 68 46 68 0 0
Japan 2005 28 319
2010 52 12 098 23 261 53 0.44
2011 49 11 221 22 681 75 0.67
– 16 • 2012 16 3 328 21 283 62 1.9
Kiribati 2005 13 44 339 2 4.5 0 0
2010 54 159 294 0 0 2
2011 77 274 354 0 0
• 13 43 • 2012 43 150 348 0 0
Lao People's 2005 3 807
Democratic 2010 38 1 533 4 083 182 12 100
Republic 2011 46 2 012 4 387 222 11 76
– 48 • 2012 48 1 999 4 156 234 12 78 303
Malaysia 2005 73 11 661 16 066 1 468 13
2010 91 17 577 19 337 1 628 9.3 22 22
2011 89 18 472 20 666 1 629 8.8 48 48
• 73 97 • 2012 97 22 124 22 710 1 347 6.1 32 1 120
Marshall Islands 2005 77 86 112 0 0
2010 68 137 201 0 0
2011 91 137 151 1 0.73 0 100
• 77 60 • 2012 60 88 147 0 0
Micronesia 2005 6.2 7 112 0 0
(Federated 2010 49 85 174 0 0
States of) 2011 97 145 150 0 0
• 6 100 • 2012 100 146 146 0 0 0
Mongolia 2005 <0.1 1 4 726 1 100 100 100
2010 89 4 256 4 801 2 <0.1 100 100 0
2011 80 3 612 4 533 3 <0.1 100 100 0
• 0 78 • 2012 78 3 465 4 453 4 0.12 75 75 0
Nauru 2005 0 0 11 0
2010 0 0 3 0
2011 0 0 5 0
• 0 – 2012
New Caledonia 2005 40 21 53 0 0
2010 0 0 57 0
2011 0 0 52
• 40 – 2012 38
New Zealand 2005 41 140 340 8 5.7
2010 60 183 305 3 1.6
2011 57 175 309 3 1.7
• 41 58 • 2012 58 171 297 3 1.8
Niue 2005 0 0 0
2010 0 0 0
2011 100 1 1 0 0
– – 2012 0
Northern Mariana 2005 98 56 57 0 0
Islands 2010 100 32 32 0 0
2011 94 31 33 0 0
• 98 79 • 2012 79 27 34 1 3.7 0 0
Palau 2005 90 9 10 0 0
2010 95 18 19 0 0
2011 83 10 12 1 10 0 0
• 90 100 • 2012 100 4 4 0 0
Papua New 2005 12 564
Guinea 2010 13 2 122 16 113 222 10 135
2011 29 4 671 16 324 531 11 256
– 17 • 2012 17 3 713 22 488 364 9.8 89 325
Philippines 2005 137 100
2010 0.94 1 634 174 389 2 0.12 0 0 16
2011 1.9 3 917 206 088 9 0.23 226
– 1 • 2012 0.89 2 040 230 162 4 0.2

Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
WESTERN PACIFIC REGION
7$%/($+,9WHVWLQJDQGSURYLVLRQRI&37$57DQG,37–
% OF TB PATIENTS WITH
KNOWN HIV STATUS
2005–2012
YEAR
% OF TB
PATIENTS WITH
KNOWN HIV
STATUS
NUMBER OF TB
PATIENTS WITH
KNOWN HIV
STATUS
PATIENTS
NOTIFIED
(NEW AND
RETREAT)
NUMBER OF
HIV-POSITIVE
TB PATIENTS
% OF TESTED
TB PATIENTS
HIV-POSITIVE
% OF HIV-
POSITIVE TB
PATIENTS ON
CPT
% OF HIV-
POSITIVE TB
PATIENTS ON
ART
NUMBER OF
HIV-POSITIVE
PEOPLE
PROVIDED IPT
Republic of Korea 2005 46 969
2010 48 101 135
2011 50 491 129
– – 2012 49 532
Samoa 2005 0 0 24 0
2010 21 3 14 0 0 0
2011 0 0 20 0
• 0 0 • 2012 0 0 22 0
Singapore 2005 1 469
2010 74 1 184 1 608 50 4.2
2011 79 1 332 1 695 61 4.6
– 84 • 2012 84 1 978 2 364 47 2.4
Solomon Islands 2005 0 0 397 0
2010 11 39 341 0 0 0
2011 17 70 405 0 0
• 0 12 • 2012 12 45 372 0 0
Tokelau 2005 0 0 0
2010 0 0 0
2011 0 0 0
– – 2012
Tonga 2005 18
2010 73 8 11 0 0
2011 100 9 9 0 0
– 100 • 2012 100 11 11 0 0
Tuvalu 2005 0 0 15 0
2010 0 0 14 0
2011 31 4 13 0 0
• 0 45 • 2012 45 9 20 0 0
Vanuatu 2005 0 0 81 0
2010 7.8 9 116 0 0
2011 45 50 112 0 0
• 0 52 • 2012 52 65 126 0 0
Viet Nam 2005 15 14 128 95 892 595 4.2
2010 43 42 356 99 022 3 515 8.3 62 43 1 317
2011 59 59 176 100 518 4 703 7.9 72 48
• 15 66 • 2012 66 68 259 103 906 4 775 7 73 47 5 663
Wallis and Futuna 2005 7 0
2010 10 0 0
2011 400 8 2 0 0
– – 2012

GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($7HVWLQJIRU0'57%DQGQXPEHURIFRQILUPHGFDVHVRI0'57%±
NEW PULMONARY CASES PREVIOUSLY TREATED CASES
YEAR
ESTIMATED CASES
OF MDR-TB AMONG
NOTIFIED
NUMBER OF
BACT+VE
b
TESTED FOR
MDR-TB
% OF
BACT+VE
b
TESTED FOR
MDR-TB
ESTIMATED CASES
OF MDR-TB AMONG
NOTIFIED
NUMBER OF
NOTIFIED
TESTED FOR
MDR-TB
% OF NOT IFIED
TESTED FOR
MDR-TB
TOTAL
CONFIRMED
CASES OF
MDR-TB
a
ESTIMATED CASES
OF MDR-TB AMONG
NOTIFIED
American Samoa 2005 ––
2010 0 0 – 0 –
2011 0 1 – 0 –
2012 – – – – –
Australia 2005 12 ––
2010 33 868 160 48 74
2011 28 652 99 26 53
2012 18 17 (9.2–25) 14 (8.1–23) 861 130 3.0 (0.36–9.9) 31 67
Brunei 2005 ––
Darussalam 2010 0 181 100 5 100
2011 0 205 130 8 100
2012 0 0 (0–0) 0 (0–4.4) 166 100 0 (0–3.2) 14 100
Cambodia 2005 ––
2010 31 5 <0.1 93 5.7
2011 56 18 <0.1 190 13
2012 75 380 (190–580) 330 (160–590) 16 0.11 56 (21–110) 86 17
China 2005 ––
2010 2792 ––
2011 1601 9940 2.6 –
2012 3007 59 000 (52 000–66 000) 49 000 (38 000–60 000) 11472 3.6 11 000 (9 000–12 000) 4861 12
China, Hong Kong 2005 41 3271 96 163 23
SAR 2010 28 1897 61 211 41
2011 23 1992 79 207 43
2012 26 48 (30–66) 36 (22–55) 2061 76 12 (4.6–27) 232 48
China, Macao 2005 9 265 190 19 61
SAR 2010 6 221 89 39 65
2011 5 258 110 24 100
2012 8 8.3 (3.0–14) 2.3 (0.27–8.1) 261 110 6.0 (2.3–11) 28 100
Cook Islands 2005 ––
2010 0 0 – 0 –
2011 0 0 0 0 –
2012 1 1.0 (<0.1–1.0) 0 (0–0) 0 – 1.0 (<0.1–1.0) 1 100
Fiji 2005 ––
2010 0 4 4.5 4 33
2011 0 18 17 0 0
2012 0 0 (0–0) 0 (0–14) 15 9.1 0 (0–13) 1 7.7
French Polynesia 2005 0 – 3 100
2010 0 27 87 4 100
2011 0 47 110 1 50
2012 0 0 (0–0) 0 (0–4.2) 30 91 0 (0–3.6) 4 67
Guam 2005 1 39 110 0 0
2010 2 56 110 2 100
2011 0 43 110 2 67
2012 0 0 (0–6.7) 0 (0–6.7) 31 100 0 (0–0) 0 –
Japan 2005 ––
2010 68 7684 54 694 39
2011 60 7400 51 670 40
2012 64 240 (180–300) 110 (65–170) 8564 66 130 (96–180) 583 44
Kiribati 2005 1 1 0.81 –
2010 0 0 0 0 0
2011 0 ––
2012 0 15 (12–18) 13 (9.5–16) 0 0 2.3 (1.9–2.7) 0 0
Lao People's 2005 ––
Democratic 2010 2 ––
Republic 2011 4 ––
2012 10 220 (180–260) 170 (130–220) 14 0.46 48 (40–56) 48 23
Malaysia 2005 1 15010 180 1056 110
2010 64 ––
2011 141 ––
2012 74 18 (0–54) 18 (0.46–100) – 0 (0–250) –
Marshall Islands 2005 2 52 110 3 60
2010 1 68 96 3 30
2011 1 50 100 4 20
2012 3 4.4 (0–9.3) 4.4 (0.92–12) 73 140 0 (0–5.9) 0 0
Micronesia 2005 1 35 110 21 100
(Federated 2010 1 50 70 3 23
States of) 2011 1 44 98 0 0
2012 3 6.8 (5.4–8.2) 5.9 (4.3–7.3) 5 8.6 0.93 (0.78–1.1) 0 0
Mongolia 2005 0 0 0 16 4.7
2010 187 40 2.2 561 95
2011 185 157 9.1 602 110
2012 210 170 (140–190) 33 (15–58) 196 11 130 (120–150) 681 130
Nauru 2005 ––
2010 ––
2011 0 0 0 –
2012 – – – – –
New Caledonia 2005 ––
2010 0 20 62 0 0
2011 0 24 140 0 0
2012 0 0 (0–0) 0 (0–3.1) 28 120 0 (0–0.98) 0 0
New Zealand 2005 4 247 150 14 74
2010 4 243 180 10 91
2011 2 229 160 5 83
2012 4 3.7 (0–9.2) 0.75 (<0.1–4.1) 221 150 3.0 (<0.1–11) 12 80
Niue 2005 ––
2010 0 ––
2011 0 ––
2012 0 0 (0–0) 0 (0–0) – 0 (0–0) –
Northern Mariana 2005 2 24 100 1 –
Islands 2010 0 17 100 0 –
2011 0 19 100 0 –
2012 0 0 (0–0) 0 (0–6.1) 15 100 0 (0–2.0) 0 0
Palau 2005 0 3 100 0 –
2010 0 11 58 0 –
2011 1 8 100 0 0
2012 0 0 (0–2.8) 0 (0–2.8) 3 100 0 (0–0) 0 –
Papua New 2005 ––
Guinea 2010 ––
2011 15 ––
2012 58 1 100 (930–1 300) 590 (430–740) – 500 (420–590) –
Philippines 2005 274 4 <0.1 138 3.5
2010 522 3 <0.1 297 2.7
2011 1148 25 <0.1 2325 17
2012 679 12 000 (9 300–15 000) 8 500 (6 000–11 000) 35 <0.1 3 700 (2 500–5 100) 2038 12
a TOTAL CONFIRMED CASES OF MDR-TB includes cases with unknown previous treatment history (i.e. not included under NEW CASES or PREVIOUSLY TREATED
CASES).
b BACT+VE = bacteriologically positive cases.

Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
WESTERN PACIFIC REGION
7$%/($7HVWLQJIRU0'57%DQGQXPEHURIFRQILUPHGFDVHVRI0'57%±
NEW PULMONARY CASES PREVIOUSLY TREATED CASES
YEAR
ESTIMATED CASES
OF MDR-TB AMONG
NOTIFIED
NUMBER OF
BACT+VE
b
TESTED FOR
MDR-TB
% OF
BACT+VE
b
TESTED FOR
MDR-TB
ESTIMATED CASES
OF MDR-TB AMONG
NOTIFIED
NUMBER OF
NOTIFIED
TESTED FOR
MDR-TB
% OF NOT IFIED
TESTED FOR
MDR-TB
TOTAL
CONFIRMED
CASES OF
MDR-TB
a
ESTIMATED CASES
OF MDR-TB AMONG
NOTIFIED
Republic of Korea 2005 ––
2010 450 ––
2011 516 3431 17 968 13
2012 1212 2 200 (1 800–2 700) 840 (660–1 100) – 1 400 (1 000–1 900) –
Samoa 2005 ––
2010 0 0 0 0 –
2011 0 ––
2012 0 0 (0–4.1) 0 (0–4.1) 15 79 0 (0–0) –
Singapore 2005 3 895 96 105 69
2010 3 923 97 79 61
2011 6 952 97 104 64
2012 22 36 (21–51) 31 (18–48) 1178 98 5.2 (1.1–15) 93 58
Solomon Islands 2005 ––
2010 0 1 0.75 1 20
2011 0 0 0 0 0
2012 0 12 (9.1–15) 12 (8.8–15) 9 5.7 0 (0–3.3) 16 100
Tokelau 2005 ––
2010 0 0 – 0 –
2011 ––
2012 – – – – –
Tonga 2005 ––
2010 0 0 0 0 –
2011 0 0 0 0 –
2012 0 0.49 (0.36–0.61) 0.49 (0.36–0.61) 0 0 0 (0–0) 0 –
Tuvalu 2005 ––
2010 0 0 0 0 –
2011 0 ––
2012 2 0.72 (0.60–0.85) 0.49 (0.36–0.61) 1 11 0.23 (0.19–0.27) –
Vanuatu 2005 ––
2010 0 ––
2011 0 0 0 0 0
2012 0 0.47 (0.39–0.54) 0 (0–8.7) 0 0 0.47 (0.39–0.54) 0 0
Viet Nam 2005 ––
2010 101 ––
2011 601 ––
2012 273 3 800 (3 000–4 600) 2 100 (1 500–2 800) – 1 700 (1 300–2 300) –
Wallis and Futuna 2005 ––
Islands 2010 ––
2011 0 0 0 0 –
2012 – – – – –
a TOTAL CONFIRMED CASES OF MDR-TB includes cases with unknown previous treatment history (i.e. not included under NEW CASES or PREVIOUSLY TREATED
CASES).
b BACT+VE = bacteriologically positive cases.

GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
7$%/($1HZVPHDUSRVLWLYHFDVHQRWLILFDWLRQE\DJHDQGVH[±
MALE FEMALE
YEAR 0–14 15–24 25–34 35–44 45–54 55–64 65+
UN-
KNOWN
0–14 15–24 25–34 35–44 45–54 55–64 65+
UN-
KNOWN
MALE:FEMALE
RATIO
American Samoa 1995 –
2000 1 1 1 2.0
2005 12 –
2010 00000000 00000000 –
2011 –
2012 –
Australia 1995 –
2000 3 16 35 25 24 19 49 0 15 19 12 15 5 14 2.1
2005 0 32 27 23 11 12 30 2 18 26 11 10 6 14 1.6
2010 2 42 33 22 25 9 27 0 4 36 43 12 2 5 12 0 1.4
2011 2 38 44 26 19 12 37 0 3 26 40 23 7 7 17 0 1.4
2012 3 26 40 17 25 16 37 0 1 27 48 15 11 9 15 0 1.3
Brunei 1995 –
Darussalam 2000 064155715 0469634 1.6
2005 0 9 19 19 12 9 0 0 9 11 8 3 2 0 2.1
2010 0 17 15 13 18 7 18 0 2 7 15 12 8 4 10 0 1.5
2011 0 11 11 11 10 11 13 0 2 5 9 6 7 3 10 0 1.6
2012 0 10 13 15 13 8 19 0 0 5 6 9 10 6 5 0 1.9
Cambodia 1995 161 453 1 244 1 147 1 253 1 257 707 123 388 1 133 1 435 1 426 1 180 578 0.99
2000 26 519 1 323 1 618 1 456 1 373 1 058 38 457 1 157 1 649 1 798 1 459 892 0.99
2005 49 894 1 600 2 349 2 043 1 964 1 811 45 790 1 413 2 089 2 323 2 058 1 573 1.0
2010 39 750 1 564 1 760 2 105 1 531 1 599 60 752 1 321 1 303 1 732 1 607 1 331 1.2
2011 34 791 1 469 1 557 1 972 1 439 1 339 39 690 1 211 1 092 1 528 1 473 1 242 1.2
2012 31 673 1 256 1 414 1 904 1 434 1 526 22 612 1 088 957 1 424 1 302 1 198 1.2
China 1995 1 102 12 791 18 306 15 487 13 105 13 489 10 130 1 169 10 890 13 250 8 376 5 679 4 579 2 841 1.8
2000 1 131 19 111 29 399 25 206 25 593 21 429 21 771 1 420 14 536 18 496 12 377 9 899 7 102 6 296 2.0
2005 1 416 43 005 49 558 55 400 54 872 53 822 69 779 1 864 31 180 27 759 24 728 19 889 18 203 21 244 2.3
2010 759 42 851 38 880 50 246 52 925 56 754 64 514 0 926 27 064 21 022 20 422 16 075 17 441 20 020 0 2.5
2011 645 37 514 34 597 43 087 47 949 51 315 55 881 0 733 22 859 18 347 17 119 14 103 15 218 17 638 0 2.6
2012 511 29 018 28 324 34 505 40 428 44 821 49 413 0 580 17 786 15 549 13 485 11 981 13 384 16 547 0 2.5
China, Hong Kong 1995 –
SAR 2000 4 78 102 160 211 236 578 5 65 115 86 44 45 211 2.4
2005 3 76 84 108 200 168 453 3 67 81 92 57 34 135 2.3
2010 2 52 84 99 184 166 413 0 3 49 101 76 64 49 133 0 2.1
2011 2 72 52 63 172 189 384 0 3 56 89 69 60 53 116 0 2.1
2012 4 63 67 95 174 178 430 0 1 45 110 76 51 54 115 0 2.2
China, Macao 1995 0 7 19 20 13 12 16 0 9 18 12 4 5 6 1.6
SAR 2000 0 10 8 25 22 9 17 0 10 4 6 6 3 13 2.2
2005 3 6 9 21 23 17 22 0 5 9 7 8 1 5 2.9
2010 0 17 5 7 22 20 11 0 0 7 6 10 5 7 6 0 2.0
2011 0 20 22 22 47 39 24 0 0 28 25 17 18 6 6 0 1.7
2012 0 10 12 13 22 32 17 0 1 12 11 13 3 7 3 0 2.1
Cook Islands 1995 0000010 0000100 1.0
2000 0000000 0000000 –
2005 0100000 0000000 –
2010 00000000 00000000 –
2011 00000000 01000000 –
2012 00000000 00000000 –
Fiji 1995 08109423 11092343 1.1
2000 08613542 0757140 1.6
2005 7 9 18 18 14 16 6 7 7 9 6 4 6 5 2.0
2010 1715116240 1111251850 1.1
2011 0121689940 1131775230 1.2
2012 2 14 12 9 12 5 7 0 2 11 10 7 6 7 7 0 1.2
French Polynesia 1995 –
2000 1334443 1410100 3.1
2005 0222042 0230113 1.2
2010 03101110 01103010 1.2
2011 03115130 03301010 1.8
2012 01223330 02303400 1.2
Guam 1995 –
2000 2166969 0312522 2.6
2005 0244224 0311202 2.0
2010 02355730 10433030 1.8
2011 01027440 01110340 1.8
2012 01045260 00002120 3.6
Japan 1995 15 342 627 995 1 847 2 059 4 089 14 258 476 298 476 637 2 234 2.3
2000 2 246 572 676 1 494 1 509 3 816 5 222 464 213 292 384 1 958 2.4
2005 9 197 488 605 868 1 418 3 867 5 187 428 249 224 309 2 077 2.1
2010 1 128 252 382 469 911 3 326 6 89 232 194 155 183 1 909 2.0
2011 0 96 215 367 465 812 3 256 0 5 94 213 203 148 223 1 840 0 1.9
2012 2 94 209 309 415 741 3 230 0 2 79 180 169 111 175 1 947 0 1.9
Kiribati 1995 –
2000 2933382 2563413 1.2
2005 3 15 15 12 17 4 1 5 22 12 7 7 3 1 1.2
2010 32713109620 515748540 1.5
2011 4 17 9 3 10 9 3 0 6 26 12 9 16 12 4 0 0.65
2012 4 19 12 16 17 11 5 0 4 15 11 10 7 2 1 0 1.7
Lao People's 1995 6 56 71 68 78 90 55 3 49 49 69 54 52 26 1.4
Democratic 2000 7 92 128 166 201 177 176 10 59 95 131 122 91 71 1.6
Republic 2005 13 136 223 296 373 300 352 7 101 186 205 244 192 178 1.5
2010 8 157 254 287 416 385 380 13 133 152 215 269 225 225 1.5
2011 8 145 275 323 474 416 375 14 141 204 208 267 215 206 1.6
2012 10 144 236 326 424 381 365 11 119 197 192 246 210 201 1.6
Malaysia 1995 59 640 879 775 788 374 1 072 58 446 448 345 316 149 339 2.2
2000 32 694 1 138 1 177 908 814 891 41 464 564 424 367 356 286 2.3
2005 244 1 179 2 218 2 277 1 980 1 427 1 507 208 1 044 1 061 947 816 586 572 2.1
2010 129 884 1 438 1 599 1 453 967 981 0 152 704 881 592 542 425 388 0 2.0
2011 63 948 1 564 1 559 1 594 1 245 1 054 0 77 837 876 584 599 459 403 0 2.1
2012 74 1 060 1 575 1 677 1 762 1 409 1 260 0 105 903 1 010 710 693 590 483 0 2.0
Marshall Islands 1995 –
2000 3541353 7730220 1.1
2005 2445611 1924342 0.92
2010 010146620 59224800 0.97
2011 17233310 15825210 0.83
2012 03869220 05725320 1.2
Micronesia 1995 0103100 0010001 2.5
(Federated 2000 0201001 4311011 0.36
States of) 2005 –
2010 38124400 58934200 0.71
2011 48562010 55231210 1.4
2012 38542010 56231300 1.2
Mongolia 1995 37 99 111 68 19 13 15 30 70 78 33 15 9 25 1.4
2000 6 181 260 171 68 38 23 32 200 213 113 41 26 17 1.2
2005 7 271 253 232 147 52 36 15 320 270 145 63 32 25 1.1
2010 3 285 255 231 154 50 40 0 12 296 246 112 83 42 28 0 1.2
2011 2 246 289 205 170 71 41 0 10 250 192 121 61 40 25 0 1.5
2012 7 257 268 191 184 63 37 0 11 250 208 97 82 28 33 0 1.4

Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
WESTERN PACIFIC REGION
7$%/($1HZVPHDUSRVLWLYHFDVHQRWLILFDWLRQE\DJHDQGVH[±
MALE FEMALE
YEAR 0–14 15–24 25–34 35–44 45–54 55–64 65+
UN-
KNOWN
0–14 15–24 25–34 35–44 45–54 55–64 65+
UN-
KNOWN
MALE:FEMALE
RATIO
Nauru 1995 –
2000 1 1 1 0.50
2005 –
2010 00000000 00010000 –
2011 00011000 00000010 2.0
2012 –
New Caledonia1995 3234223 2113301 1.7
2000 1134234 1811324 0.90
2005 0210030 0121204 0.60
2010 01231430 01010130 2.3
2011 00031230 00110110 2.2
2012 2 3 2 1 1 1 1 2.7
New Zealand 1995 0433577 1234224 1.6
2000 06568107 16650410 1.3
2005 461066510 11196612 1.3
2010 0613465110 212765360 1.1
2011 1125577110 48845380 1.2
2012 079246140 34823150 1.6
Niue 1995 –
2000 –
2005 0000000 0000000 –
2010 –
2011 –
2012 00000000 00000000 –
Northern Mariana 1995 11351033 0026411 1.9
Islands 2000 1489932 010177311 0.92
2005 0013412 0001111 2.8
2010 02003300 02013210 0.89
2011 00001530 00102300 1.5
2012 00031100 00310000 1.2
Palau 1995 0230210 0000100 8.0
2000 –
2005 2 1 –
2010 01211110 10110000 2.3
2011 00010200 00100000 3.0
2012 00000000 00011010 –
Papua New 1995 –
Guinea 2000 8 87 70 30 21 12 5 6 77 45 21 15 5 1 1.4
2005 28 183 205 108 94 48 12 38 200 204 124 65 35 2 1.0
2010 37 279 260 196 135 87 27 64 313 292 191 97 52 9 1.0
2011 50 278 265 152 122 71 18 53 302 272 146 97 55 15 1.0
2012 54 415 387 250 182 121 37 0 55 398 395 208 156 95 29 80 1.0
Philippines 1995 2 43 56 61 46 47 26 1 20 32 26 20 19 11 2.2
2000 –
2005 482 7 358 11 275 13 253 12 531 7 646 4 279 374 3 710 5 268 5 565 4 603 3 274 2 029 2.3
2010 511 9 320 12 224 13 716 13 651 8 923 4 742 0 454 4 825 5 489 5 301 4 643 3 329 2 070 0 2.4
2011 573 9 725 12 804 14 474 14 002 9 568 4 845 448 5 155 5 848 5 521 4 880 3 501 2 236 2.4
2012 583 9 754 12 576 14 140 13 996 9 676 5 097 466 5 104 5 954 5 584 5 068 3 605 2 380 2.3
Republic of Korea 1995 27 1 131 1 613 1 425 1 207 1 307 1 225 46 908 863 431 296 408 867 2.1
2000 19 821 1 085 988 853 731 901 25 546 544 393 220 295 795 1.9
2005 22 687 1 171 1 326 1 336 1 005 1 669 27 590 842 491 370 373 1 729 1.6
2010 22 537 705 1 049 1 496 1 029 1 997 0 23 472 686 509 487 368 2 216 0 1.4
2011 13 491 712 1 019 1 414 1 145 2 132 0 37 446 688 520 432 421 2 244 0 1.4
2012 11 500 699 956 1 562 1 238 2 255 5 22 436 664 444 377 397 2 569 2 1.5
Samoa 1995 0111032 1220011 1.1
2000 0311121 0211000 2.2
2005 0401100 0202010 1.2
2010 1 1 1 3 2 1 3 1.0
2011 01000000 02101010 0.20
2012 04311100 11011010 2.0
Singapore 1995 0 9 40 60 62 70 94 1 8 18 21 22 19 31 2.8
2000 1 8 9 34 51 26 64 1 9 8 7 9 5 16 3.5
2005 0 8 25 61 94 96 118 0 5 20 33 29 20 43 2.7
2010 0 11 21 38 105 86 120 0 1 15 21 26 21 21 44 0 2.6
2011 0 21 21 44 108 119 126 0 0 11 25 23 23 20 51 0 2.9
2012 1 31 36 54 106 124 143 0 0 26 46 27 26 19 39 0 2.7
Solomon Islands 1995 2 14 6 5 7 9 3 3 17 11 7 12 13 0 0.73
2000 3 13 4 8 8 10 6 8 15 13 7 7 5 2 0.91
2005 4 14 18 9 15 12 11 9 23 21 12 11 9 1 0.97
2010 4 16 18 16 8 3 3 0 4 19 17 11 5 4 5 0 1.0
2011 3 15 22 12 7 8 6 0 3 13 27 15 10 16 2 0 0.85
2012 3 20 19 10 12 8 6 0 5 20 18 11 8 12 5 0 0.99
Tokelau 1995 –
2000 –
2005 –
2010 00000000 00000000 –
2011 –
2012 –
Tonga 1995 0100012 0011021 0.80
2000 211 15 111 11 2.0
2005 0210210 0210020 1.2
2010 00010130 00001000 5.0
2011 00100010 20110000 0.50
2012 00002020 02001110 0.80
Tuvalu 1995 1010010 0110010 1.0
2000 –
2005 1 1 1 2 0.67
2010 0100120 0010000 4.0
2011 1 1 1 1 3.0
2012 1 1 1 2 1 2 0.60
Vanuatu 1995 0625340 0502300 2.0
2000 27511052 53157331 0.86
2005 1455041 0512412 1.3
2010 46315200 35335310 0.91
2011 23465420 05754200 1.1
2012 04342220 312554230 0.50
Viet Nam 1995 –
2000 51 2 367 6 147 8 209 6 713 5 150 7 712 64 1 334 2 320 2 754 2 594 2 847 4 907 2.2
2005 54 3 408 7 105 8 738 8 606 4 958 7 573 47 1 747 2 293 2 116 2 298 2 023 4 604 2.7
2010 59 3 205 7 036 7 851 8 564 5 790 6 248 0 53 1 870 2 454 1 681 1 864 1 863 3 751 0 2.9
2011 61 3 099 6 677 7 763 8 474 6 107 5 821 0 64 1 863 2 325 1 681 1 814 1 878 3 124 0 3.0
2012 58 2 993 6 689 7 680 8 481 6 315 5 920 0 84 1 841 2 481 1 626 1 683 1 884 3 298 0 3.0
Wallis and Futuna 1995 –
Islands 2000 –
2005 –
2010 –
2011 2 –
2012 –

GLOBAL TUBERCULOSIS REPORT 2013 Data for all years can be downloaded from www.who.int/tb/data
a LED = Light emitting diode microscopes
b DST = Drug susceptibility testing
c LPA = Line probe assay
d NRL = National Reference Laboratory
7$%/($/DERUDWRULHV173VHUYLFHVGUXJPDQDJHPHQWDQGLQIHFWLRQFRQWURO
LABORATORIES FREE THROUGH NTP
SMEAR LABS
PER 100K
POPULATION
% OF SMEAR
LABS USING
LED
a
CULTURE
LABS PER 5M
POPULATION
DST
b
LABS
PER 5M
POPULATION
LPA
c
LABS
PER 5M
POPULATION
NUMBER OF
LABS USING
XPERT MTB/RIF
SECOND-
LINE DST
AVAILABLE
NRL
d
TB DIAGNOSIS
FIRST-
LINE
DRUGS
TB NOTIF.
RATE PER
100 000
HEALTH-CARE
WORKERS
RIFAMPICIN
USED
THROUGHOUT
TREATMENT
American Samoa –
Australia – In country Yes Yes (all suspects) Yes Yes
Brunei Darussalam 0.2 0 12.1 12.1 12.1 0 Out of
country Yes Yes (all suspects) Yes Yes 26
Cambodia 1.4 10 1.0 0.3 0 6 No Yes Yes (all suspects) Yes Yes
China 0.2 2 3.7 0.7 <0.1 16 In country Yes Yes (all suspects) Yes Yes
China, Hong Kong SAR 0.4 3 9.1 1.4 1.4 9 In country Yes Yes (all suspects) Yes Yes
China, Macao SAR 0.4 0 9.0 9 0 0 In country No Yes (all suspects) Yes Yes
Cook Islands – Out of
country Yes Yes (other criteria) No Yes
Fiji 0.5 0 5.7 0 0 3 No Yes Yes (all suspects) Yes Yes 41
French Polynesia – Out of
country Yes Yes
(
if TB is
confirmed) Yes Yes
Guam – Out of
country Yes Yes (all suspects) Yes Yes
Japan – In and out
of countr y Yes No No Yes
Kiribati 2.0 0 49.6 0 0 0 Out of
country Yes Yes (all suspects) Yes Yes 93
Lao People's Democratic
Republic 2.4 0 2.3 0.8 0.8 0 No Yes Yes (all suspects) Yes No
Malaysia 2.6 4 6.2 0.2 0.3 0 In country Yes Yes (all suspects) Yes Yes 106
Marshall Islands 5.7 33 95.1 95.1 95.1 1 Out of
country No Yes (all suspects) Yes Yes
Micronesia
(Federated States of) 3.9 0 0 0 0 0 In and out
of countr y Yes Yes (all suspects) Yes Yes
Mongolia 1.4 8 3.6 1.8 1.8 0 In country Yes Yes (all suspects) Yes Yes 53
Nauru –
New Caledonia – Out of
country Yes Yes (all suspects) Yes Yes
New Zealand – In country Yes Yes (all suspects) Yes Yes
Niue –
Northern Mariana Islands – Out of
country No Yes (all suspects) Yes Yes
Palau 9.6 0 240.9 240.9 240.9 1 Out of
country Yes Yes (all suspects) Yes Yes 0
Papua New Guinea 1.6 0 0 0 0 6 Yes Yes
(
if TB is
confirmed) Yes Yes
Philippines 2.7 0 0.7 0.2 <0.1 17 In country Yes Yes (all suspects) Yes Yes
Republic of Korea 1.0 – 51.0 0.7 2 2 Yes Yes (all suspects) Yes Yes 204
Samoa – Out of
country Yes Yes (all suspects) Yes Yes
Singapore – In country Yes No No Yes
Solomon Islands 1.5 0 0 0 0 0 Out of
country Yes Yes (all suspects) Yes Yes 0
Tokelau –
Tonga – Out of
country Yes Yes (all suspects) Yes Yes
Tuvalu – Out of
country Yes Yes (all suspects) Yes Yes
Vanuatu 4.0 100 0 0 0 0 Out of
country Yes Yes (all suspects) Yes Yes 111
Viet Nam 0.9 0 1.4 0.1 0.1 22 In country Yes Yes (for smear-
positive TB) Yes No
Wallis and Futuna Islands –

Data for all years can be downloaded from www.who.int/tb/data GLOBAL TUBERCULOSIS REPORT 2013
WESTERN PACIFIC REGION
7$%/($0HDVXUHGSHUFHQWDJHRI7%FDVHVZLWK0'57%
D
PRVWUHFHQW\HDUDYDLODEOH
New TB cases Previously treated TB cases
Year Source Coverage Percentage Year Source Coverage Percentage
American Samoa
Australia 2012 Surveillance National 1.9 (1.1–3.0) 2012 Surveillance National 6.5 (0.79–21)
Brunei Darussalam 2012 Surveillance National 0 (0–2.2) 2012 Surveillance National 0 (0–23)
Cambodia 2007 Survey National 1.4 (0.71–2.5) 2007 Survey National 11 (4.0–22)
China 2007 Survey National 5.7 (4.5–7.0) 2007 Survey National 26 (22–30)
China, Hong Kong SAR 2012 Surveillance National 0.97 (0.59–1.5) 2012 Surveillance National 2.6 (0.95–5.5)
China, Macao SAR 2012 Surveillance National 0.77 (<0.1–2.7) 2012 Surveillance National 21 (8.3–41)
Cook Islands 2012 Surveillance National 0 (0–98) 2012 Surveillance National 100 (2.5–100)
Fiji 2006 Surveillance National 0 (0–8.2) 2006 Surveillance National 0 (0–98)
French Polynesia 2012 Surveillance National 0 (0–12) 2012 Surveillance National 0 (0–60)
Guam 2012 Surveillance National 0 (0–11) 2012 Surveillance National 12 (9.2–15)
Japan 2002 Surveillance National 0.7 (0.42–1.1) 2002 Surveillance National 9.8 (7.1–13)
Kiribati
Lao People's Democratic
Republic
Malaysia 1997 Survey Sub-national 0.1 (0–0.56) 1997 Survey Sub-national 0 (0–17)
Marshall Islands 2012 Surveillance National 4.1 (0.86–12) 2012 Surveillance National 0 (0–98)
Micronesia
(Federated States of)
Mongolia 2007 Survey National 1.4 (0.66–2.5) 2012 Surveillance National 26 (23–30)
Nauru
New Caledonia 2012 Surveillance National 0 (0–12) 2012 Surveillance National 0 (0–98)
New Zealand 2011 Surveillance National 0.44 (<0.1–2.4) 2011 Surveillance National 20 (0.51–72)
Niue
Northern Mariana Islands 2012 Surveillance National 0 (0–22) 2012 Surveillance National 0 (0–98)
Palau 2012 Surveillance National 0 (0–71) 2012 Surveillance National 23 (20–27)
Papua New Guinea
Philippines 2004 Survey National 4 (2.9–5.5) 2004 Survey National 21 (14–29)
Republic of Korea 2004 Survey National 2.7 (2.1–3.4) 2004 Survey National 14 (10–19)
Samoa 2012 Surveillance National 0 (0–22) 2012 Surveillance National 0 (0–98)
Singapore 2012 Surveillance National 1.6 (0.97–2.5) 2012 Surveillance National 3.2 (0.67–9.1)
Solomon Islands 2012 Surveillance National 0 (0–21)
Tokelau
Tonga
Tuvalu
Vanuatu 2006 Surveillance National 0 (0–12)
Viet Nam 2006 Survey National 2.7 (2.0–3.7) 2006 Survey National 19 (14–25)
Wallis and Futuna Islands
a Empty rows indicate an absence of high-quality survey or surveillance data. In the absence of high-quality national data, high-quality sub-national data are used.

For further information about tuberculosis contact:
Information Resource Centre HTM/GTB
World Health Organization
20 Avenue Appia, 1211–Geneva–27, Switzerland
Email: tbdocs@who.int
Web site: www.who.int/tb
ISBN 978 92 4 1564656
The World Health Organization monitors the
global tuberculosis epidemic in support of
national TB control programmes.