Manual
User Manual:
Open the PDF directly: View PDF ![]() .
.
Page Count: 4
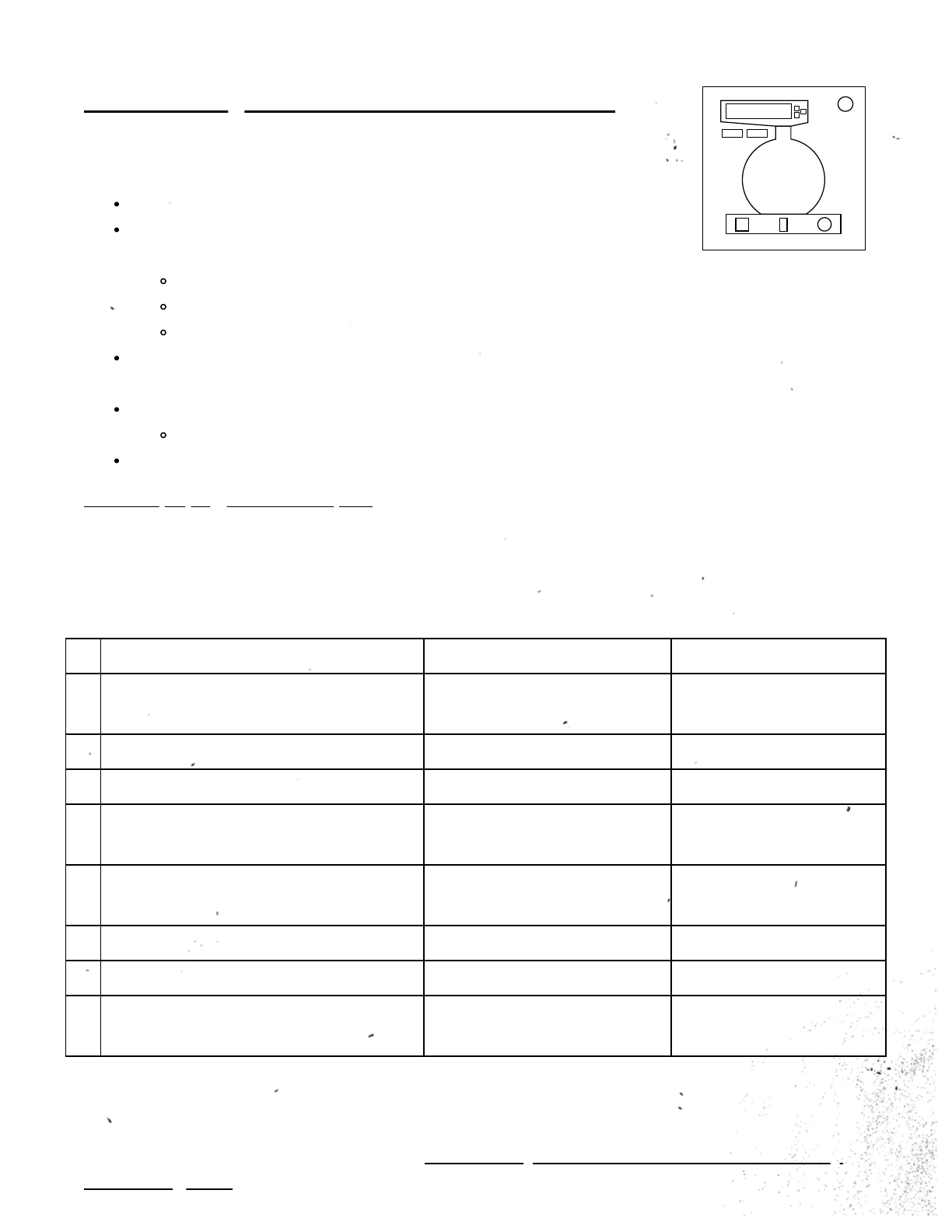
On the Subject of Chemical Reactions
Ok, now I just have to add... *BOOM*
This module contains:
A reaction vessel placed on a magnetic stirrer
Three addition consoles above the vessel, only one of
which is visible at a time:
Red: solids
Blue: liquids
Yellow: gases
Two buttons to switch between the addition consoles, labeled with the
corresponding colors
A reset button on the right of the magnetic stirrer
Press at any time to discard vessel contents
And a colored letter display on the left of the magnetic stirrer.
Determining the reaction
To determine the reaction, take the alphabetic position of the displayed letter.
If it is greater than the sum of serial number digits, subtract that sum from it.
Now repeatedly add or subtract 8 to your number until it is in the range 1-8. Use
the following table to determine your reaction:
# Reagents Product Solvent
1C H (COOH) NO + H NNH +
Na S O Luminol Propane-1,2,3-
triol
2 C H (OH) + 3 HNO + 3 H SO Nitroglycerin None
3 C H (CO) O + 2 C H OH + H SO Phenolphthalein Water
4C H (NH )(COOH) + HCl +
NaNO + C H N(CH ) Methyl red Ethanol
5U O + 6 NH + 15 H + 12 HF +
3 F
3 Uranium
hexafluoride Nitric acid
6 C H (OH)COOH + (CH CO) O Aspirin Sulfuric acid
7 C H Cl OH + (COCl) + N(CH CH ) TCPO Toluene
8KIO + 2 H O + CH (COOH) +
MnCl None* Sulfuric acid
* The "Briggs-Rauscher reaction" is cyclical and has no real products. When a
product is required for calculation, use potassium iodate.
If the reaction requires a solvent, add exactly 100ml of the solvent before any
other reagents. Doing otherwise will cause a strike.
Keep Talking and Nobody Explodes Mod Chemical Reactions
6 3 2 2 2 2
2 2 4
3 5 3 3 2 4
6 4 2 6 5 2 4
6 4 2
2 6 5 3 2
3 8 3 2
2
6 4 3 2
6 2 3 2 2 3 3
3 2 2 2 2
2
Determining product mass
Start with the value given by the color of the displayed letter:
Red Blue Green Yellow Cyan Orange Magenta White
14 22 3 48 37 19 25 50
Now do these steps in order:
Add the number of unlit indicators.
If there are two or more ports, multiply by the number of ports.
Repeatedly add or subtract 50 until your number is in the range 1-50.
Multiply your result by 10.
Finally, subtract the number of lit indicators.
This number is the mass of the product that you should produce, in g.
Calculating reagent amounts
First, calculate the molar mass of your product. Divide your product mass by
this molar mass. If the product has a factor in front of it in the reaction table,
also divide by that factor. Round to 3 digits after the decimal point. This
result is your base reagent amount, in mol.
Now, for each reagent in your given reaction:
For solids:
Calculate the molar mass of the reagent.
Multiply the molar mass by the base reagent amount.
If the reagent has a factor in front of it, multiply by that factor.
You now have the mass of this reagent that needs to be added, in g.
For liquids:
Calculate the molar volume of the reagent.
Multiply the molar volume by the base reagent amount.
If the reagent has a factor in front of it, multiply by that factor.
You now have the volume of this reagent that needs to be added, in ml.
For gases:
Take the standard molar volume for gases.
Multiply the molar volume by the base reagent amount.
If the reagent has a factor in front of it, multiply by that factor.
You now have the volume of this reagent that needs to be added, in l.
Round all resulting amounts to 3 digits after the decimal point.
Add the correct amounts of all reagents listed in the reaction, in any order.
The module will list all reagents either by chemical formula or by name.
Adding a reagent that is not part of the given reaction will cause a strike.
Note 1: It is always allowed to add a zero amount of any reagent. This is
equivalent to cancelling the input.
Note 2: If more than six digits are entered for the amount, the most significant
digit will be discarded, e.g. inputting six zeroes will reset the amount to zero.
Press the switch on the magnetic stirrer to start the reaction.
If not all reagents were added or any reagent amount was incorrect, a strike
will be issued and the vessel will be emptied.
Keep Talking and Nobody Explodes Mod Chemical Reactions
[1]
[1]
Calculating molar mass and volume
Molar mass (for solids and liquids):
For each element that appears in the compound's chemical formula, count how
many times it appears. Note that, for example, (CH ) means "2 instances of CH ", so
2 carbon and 6 hydrogen atoms in total.
Multiply each element's count with the atomic weight of that element (bottom
number in the periodic table, see appendix A). Then add them all together to get
the molar mass of your compound, in g/mol.
Note: Keep all decimal digits during this calculation, do not round your
numbers.
Molar volume for liquids:
Divide the molar mass of the compound by the density of the liquid (see compound
table below). Round the result to 3 digits after the decimal point. This is the
liquid's molar volume, in ml/mol.
Molar volume for gases:
Gases all have the same standard molar volume of 24.5 l/mol.
Chemical compounds
Name Formula State Density Name Formula State Density
Benzene-3-nitro-
1,2-dicarboxylic
acid C H (COOH) NO solid - Hydrazine H NNH liquid 1.02
Luminol C H (CONH) (NH ) - - Sodium
dithionite Na S O solid -
Propane-1,2,3-triol C H (OH) liquid 1.26 Nitric acid HNO liquid 1.51
2-Benzofuran-
1,3-dione C H (CO) O solid - Water H O liquid 1.00
Nitroglycerin C H (ONO ) - - Sulfuric acid H SO liquid 1.83
Phenolphthalein C H (CO)C(C H OH) O - - Phenol C H OH solid -
2-Aminobenzoic acid C H (NH )(COOH) solid - Hydrogen
chloride HCl gas -
Dimethylaniline C H N(CH ) liquid 0.96 Sodium
nitrite NaNO solid -
Ethanol C H OH liquid 0.79 Urania U O solid -
Methyl red C H (COOH)
(N )C H N(CH ) - - Ammonia NH gas -
2-Hydroxybenzoic
acid C H (OH)COOH solid - Hydrogen H gas -
Acetic anhydride (CH CO) O liquid 1.08 Hydrogen
fluoride HF gas -
Aspirin C H (CH CO)(COOH)O - - Fluorine F gas -
2,4,6-
Trichlorophenol C H Cl OH solid - Uranium
hexafluoride UF - -
Triethylamine N(CH CH ) liquid 0.73 Oxalyl
dichloride (COCl) liquid 1.48
Toluene C H CH liquid 0.87 Potassium
iodate KIO solid -
TCPO (COOC H Cl ) - - Hydrogen
peroxide H O liquid 1.45
Propanedioic acid CH (COOH) solid - Manganese
dichloride MnCl solid -
Keep Talking and Nobody Explodes Mod Chemical Reactions
3 2 3
[1]
6 3 2 2 2 2
6 3 2 2 2 2 4
3 5 3 3
6 4 2 2
3 5 2 3 2 4
6 4 6 4 2 6 5
6 4 2
6 5 3 2 2
2 5 3 8
6 4
2 6 4 3 2 3
6 4 2
3 2
6 4 3 2
6 2 3 6
2 3 3 2
6 5 3 3
6 2 3 2 2 2
2 2 2

Appendix A: Periodic Table of Elements
1
H
1.01
2
He
4.00
3
Li
6.94
4
Be
9.01
5
B
10.8
6
C
12.0
7
N
14.0
8
O
16.0
9
F
19.0
10
Ne
20.2
11
Na
23.0
12
Mg
24.3
13
Al
27.0
14
Si
28.1
15
P
31.0
16
S
32.1
17
Cl
35.5
18
Ar
39.9
19
K
39.1
20
Ca
40.1
21
Sc
45.0
22
Ti
47.9
23
V
51.0
24
Cr
52.0
25
Mn
54.9
26
Fe
55.8
27
Co
58.9
28
Ni
58.7
29
Cu
63.5
30
Zn
65.4
31
Ga
69.7
32
Ge
72.6
33
As
74.9
34
Se
79.0
35
Br
79.9
36
Kr
83.8
37
Rb
85.5
38
Sr
87.6
39
Y
88.9
40
Zr
91.2
41
Nb
92.9
42
Mo
96.0
43
Tc
98.0
44
Ru
101
45
Rh
103
46
Pd
106
47
Ag
108
48
Cd
112
49
In
115
50
Sn
119
51
Sb
122
52
Te
128
53
I
127
54
Xe
131
55
Cs
133
56
Ba
137
(La)
▼
72
Hf
178
73
Ta
181
74
W
184
75
Re
186
76
Os
190
77
Ir
192
78
Pt
195
79
Au
197
80
Hg
201
81
Tl
204
82
Pb
207
83
Bi
209
84
Po
209
85
At
210
86
Rn
222
87
Fr
223
88
Ra
226
(Ac)
▼
104
Rf
267
105
Db
268
106
Sg
269
107
Bh
270
108
Hs
277
109
Mt
278
110
Ds
281
111
Rg
282
112
Cn
285
113
Nh
286
114
Fl
289
115
Mc
290
116
Lv
293
117
Ts
294
118
Og
294
(La)
►
57
La
139
58
Ce
140
59
Pr
141
60
Nd
144
61
Pm
145
62
Sm
150
63
Eu
152
64
Gd
157
65
Tb
159
66
Dy
163
67
Ho
165
68
Er
167
69
Tm
169
70
Yb
173
71
Lu
175
(Ac)
►
89
Ac
227
90
Th
232
91
Pa
231
92
U
238
93
Np
237
94
Pu
244
95
Am
243
96
Cm
247
97
Bk
247
98
Cf
251
99
Es
252
100
Fm
257
101
Md
258
102
No
259
103
Lr
266
1: The rounding method to be used is "away from zero", so _.___5 should always be
rounded up.
Disclaimer: This module is based on real chemistry, but a lot of simplifications and approximations were
applied to make it viable for a module. Therefore, these reactions would not work this way in reality, and
might blow up in your face.
Also, you should probably stop reading this, or the bomb may blow up in your defuser's face.
Keep Talking and Nobody Explodes Mod Chemical Reactions