RESEARCH INSTRUMENTS 670808 RI WITNESS ADMIN & CARD READER User Manual
RESEARCH INSTRUMENTS LTD RI WITNESS ADMIN & CARD READER
User Manual
Document 6-70-808UM, Issue 2,DRF: 3646, 28 July 2016
RESEARCH INSTRUMENTS LTD
Bickland Industrial Park, Falmouth, Cornwall TR11 4TA, UK
t: +44 (0) 1326 372 753 | f: +44 (0) 1326 378 783 | e: sales@research-instruments.com
www.research-instruments.com
RI WITNESS™
Admin & Card Reader
User Manual

CONTENTS
SECTION 1 - PREFACE 1
SECTION 2 - INTRODUCTION TO RI WITNESS 2
Intended Use 2
Contraindicaons 2
Applicable Part Numbers 2
OponalPartNumbers 2
Related Documents 3
Compability 3
Installaon 3
SECTION 3 - SAFETY WARNINGS 4
GuidanceandManufacturer’sDeclaraon(Part15ofFCC)
—ElectromagnecEmissions 4
Safety/InformaonSymbols 5
Safety and Reliability 6
RFID Reader Environment 6
SECTION 4 - PRODUCT OVERVIEW 7
Admin & Card Reader 7
Contents Research Instruments Ltd

Admin&CardReaderSpecicaonTable 8
SECTION 5 - RI WITNESS BASIC OPERATION 9
ConnecontotheSoware 9
CardReaderFuncon 9
AdminReaderFuncon 10
IUIFuncon 10
SECTION 6 - TROUBLESHOOTING 11
SECTION 7- CARE AND MAINTENANCE 12
Cleaning 12
SECTION 8 - REPAIRS AND RETURNS 13
Reuse Statement 13
RI Repairs System 13
RI Returns System 13
Product Disposal (European Union) 14
Contact Details 14
Feedback 14
Contents
Research Instruments Ltd

Secon 1
1
1
Preface
Research Instruments Ltd
SECTION 1 - PREFACE
Thank you for choosing RI Witness.
This manual provides all the necessary informaon to use RI Witness
Admin&CardReaderandshouldbereadinconjunconwithanymanuals
provided with other RI Witness hardware or soware components that
you are using. The system should be operated by trained personnel only.
Allseconsofthismanualshouldbereadandunderstoodfullybeforeany
operaon of the system. Please see the Intended Use secon for more
informaon.
Iftheoperatorisunsureofanyoftheinformaoncontainedinthismanual
theyshouldcontactResearchInstrumentsoranappointedrepresentave
beforeaempngtousethisequipment.
In no event does Research Instruments Ltd (RI) assume the liability for any
technical or editorial errors of commission, or omission; nor is RI liable for
direct,indirect,incidental,orconsequenaldamagesarisingoutoftheuse
or inability to use this manual.
The informaon in this manual is current at the me of publicaon.
Our commitment to product improvement requires that we reserve
the right to change equipment, procedures and specicaons at any
me. The latest version of the User Manual can be downloaded from
soware.research-instruments.com.TheRIWitnessmanualbelongswith
the RI Witness system and should be passed on with the system if relocated
to another clinic.
The use of ™ in this manual indicates a trademark of Research Instruments
Ltd. Any other brand names, referred to in this manual, are trademarks of
theirrespecveowners.
© This manual is protected by copyright, all rights reserved, and no part
maybephotocopiedorreproducedinanyformwithoutthepriorwrien
consent of RI.

Secon 2
Introducon
2
Research Instruments Ltd
2
SECTION 2 - INTRODUCTION TO RI WITNESS
Intended Use
Toidenfyandtrackhumansamples,usingRFIDtechnology,throughthe
assistedreproducon(AR)cycle,includingcryopreservaon.
Contraindicaons
This device is not intended to be exposed to known sources of
electromagnecInterference(EMI)withmedicaldevicessuchasdiathermy,
andelectromagnecsecuritysystems,egmetaldetectorsandelectronic
arclesurveillancesystems.
Applicable indicaons for use are subject to the regulaons of the country
into which the device is sold. Availability of RI Witness for clinical use is
dependent on the regulatory approval status of RI Witness within the
country the device is intended to be sold into.
Applicable Part Numbers
Part Number Descripon
6-70-808 RI Witness Admin & Card Reader
Oponal Part Numbers
Part Number Descripon
6-70-811 RI Witness Admin & Card Reader Wall Mount Kit
6-70-812 RI Witness Admin & Card Reader Stand

Secon 2
3
Introducon
Research Instruments Ltd
2
Related Documents
6-70-121UM RIWitnessSowareManual
Compability
RIWitnessisusedinconjunconwiththefollowing:
• Essenalmedicaldevices,egdishesandtubes,maybeARornon-AR
specic.
• Non-essenal medical devices, eg safety cabinets, incubators,
micromanipulators, lasers.
• Nonmedicaldevices(generallaboratoryequipment),egworkbenches,
microscopes, PCs.
This device has RFID reader capability. If it is the intenon that it be
employed in a clinical lab, we recommend its use alongside other medical
devices and that the performance of these medical devices be monitored
forpotenaleectsofEMIdisturbances,andreportedwhenappropriate.
Installaon
Installaons of the RI Witness Admin & Card Reader plate should be
carried out by an RI technician or other RI authorised personnel. Incorrect
installaoncouldresultinoverallpoorperformance.
Secon 2
Introducon
4
Research Instruments Ltd
2
SECTION 3 - SAFETY WARNINGS
DO NOT disassemble or modify any part of the RI Witness
Admin&CardReader,orsubstuteanycomponent.Doing
so may result in damage to samples. This voids the warranty
and/or service contract.
The system should be operated by qualied and trained
personnel only.
Thissymbolindicatescauonarytextwhichshouldbe
followed to avoid injury to users or damage to samples.
Note: This equipmenthas been tested and found to complies with the
limits for a Class A digital device, pursuant to part 15 of the Federal
Communicaons Commission (FCC) Rules. These limits are designed to
provide reasonable protecon against harmful interference when the
equipment is operated in a commercial environment. This equipment
generates,uses,andcanradiateradiofrequencyenergyand,ifnotinstalled
andusedinaccordancewiththeinstruconmanual,maycauseharmful
interferencetoradiocommunicaons.Operaonofthisequipmentina
residenalareaislikelytocauseharmfulinterferenceinwhichcasethe
userwillberequiredtocorrecttheinterferenceattheirownexpense.
Note: This device complies with Industry Canada’s licence-exempt RSSs.
Operaonissubjecttothefollowingtwocondions:
1. This device may not cause interference.
2. This device must accept any interference, including interference that
maycauseundesiredoperaonofthedevice.
Guidance and Manufacturer’s Declaraon (Part 15 of FCC)
— Electromagnec Emissions

Secon 3
5
Safety Warnings
Research Instruments Ltd
3
Safety/Informaon Symbols
Symbol Meaning
Indicatesinstruconfordisposalofgoods.
In accordance with the European Direcve for
R&TTEDirecve99-5-EC.
Indicates the medical device manufacturer.
Indicates the date of manufacture.
Indicates the need for the user to consult the
instrucons for use for important cauonary
informaonsuchaswarningsandprecauonsthat
cannot, for a variety of reasons, be presented on
the device itself.
Consultinstruconsforuse.
The ve digit number is a unique idener
assigned to the product.
Indicates the RI part number.
S
N
REF
Secon 3
Safety Warnings
6
Research Instruments Ltd
3
Safety and Reliability
Pleasereadthismanualcarefullyandfollowtheinstruconstoensurethat
the system will work safely and reliably.
RFID Reader Environment
AnRIWitnesssystemusesRadioFrequencyIdencaon(RFID)readers
to monitor a work area. Readers detect RFID tagged containers that are
placed in the work area.
The performance of RFID tag detecon may be compromised by the
proximityofmetalobjectsorelectricalequipment.
Forcleaning,thereadermaybeliedandreturnedtothesameposion.
See “Cleaning” on page 12 for more details.
Do not place metal objects near reader.
Donotplaceelectricalequipmentnearreader.

Secon 4
7
Product Overview
Research Instruments Ltd
4
SECTION 4 - PRODUCT OVERVIEW
RI Witness is a system which operates within an assisted reproducon
(AR) clinic seng and provides a method of idenfying human samples
throughoutanARcycle(fromeggandspermcollecontoembryotransfer).
Thesystemisintendedtominimisetherisksassociatedwithtradional/
manualdouble-checkingandprovidestheessenalcontrolsnecessaryto
ensure eggs, sperm and embryos are correctly matched and treated during
the AR process.
The RI Witness system comprises hardware, rmware and soware
components, which can be congured depending on the treatment
acvies,numberofARcyclesconducted,sizeandlayoutoftheARclinic.
RFID (radio frequency idencaon) technology provides the means of
idenfyingthecontainers(dishes,tubes)inwhicheggs,spermandembryos
are transferred and stored. The containers are labelled by a clinician with
aspecialRFIDtagwhichhasbeenassignedauniqueidener.Theunique
idenerislinkedtoapaent/couple(specicparentage).
As samples are processed as part of an AR cycle, RFID readers (both heated
andnon-heated)readthetagsonthecontainerandtheiridentyandstatus
is conrmed on-screen. If containers holding samples of incompable
origincomeintocontactatanystageofthisprocess,thesystemacvates
an alarm and prompts the clinician to respond.
This manual refers only to the Admin & Card Reader.
Other devices in the RI Witness range have their own manuals, as does the
soware.
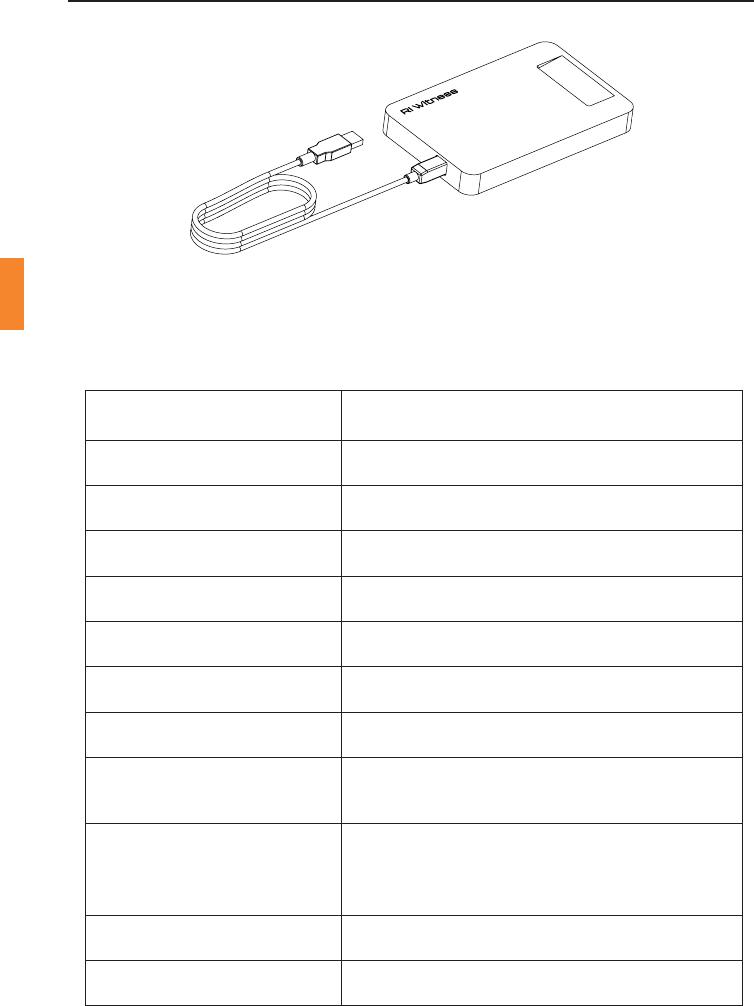
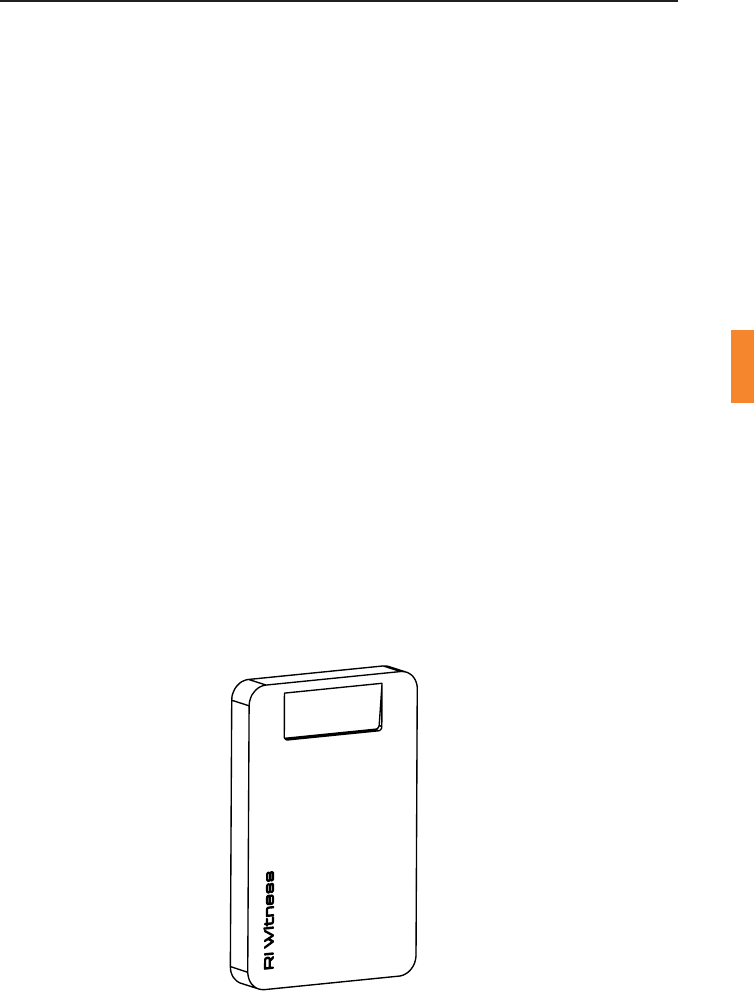
Admin & Card Reader
TheAdmin&Cardreaderisamulfunconaldevicethatcanbeusedto
idenfy RFID tags contained in paent cards or axed to sperm tubes,
spermpotsorevenpaentles.Itcanbeusedonadeskintherecepon
oroce,orinthetheatremountedonawall.Acardslotisprovidedto
securelyholdapaentcard.Allothertagscanbereadbyplacingthem
against the surface of the device.
Secon 4
Product Overview
8
Research Instruments Ltd
4
Figure 4-1 Admin & Card Reader
Admin & Card Reader Specicaon Table
RFID Reader
Frequency 13.56MHz
Power Output 0.5 W
Read Range >3cm
Power Supply
Input 5VDC Max 0.5A
Output 2.5W Max
USB USB 2.0 Socket Type B
Material(s) Corian (Housing)
ABS (Lower cover)
Operang Temperature
Temperature:-25˚C(-13˚F)to60˚C
(140˚F)
Humidity:5%to95%RH(NonCondensing)
Dimensions 160x100x18 mm
Mass 0.4 kg
Secon 5
9
RI Witness Basic Operaon
Research Instruments Ltd
5
SECTION 5 - RI WITNESS BASIC OPERATION
Connecon to the Soware
Plug the device into the tablet or PC (or powered USB hub) using the USB
cableprovidedwiththedevice.OncetheWindowsoperangsystemhas
recognisedthedeviceopentheRIWitnessWorkAreasoware.
To verify that the RI Witness WorkArea soware can communicate
successfully, navigate to the WorkArea Status window (click the yellow
triangle or press the (i) icon). This will bring up the WorkArea Status window
in which the Admin & Card Reader should be listed in the Connected
Devicesseconwithagreencknexttoit.
For more detailed set up informaon, refer to the RI Witness soware
manual (6-70-121UM).
Card Reader Funcon
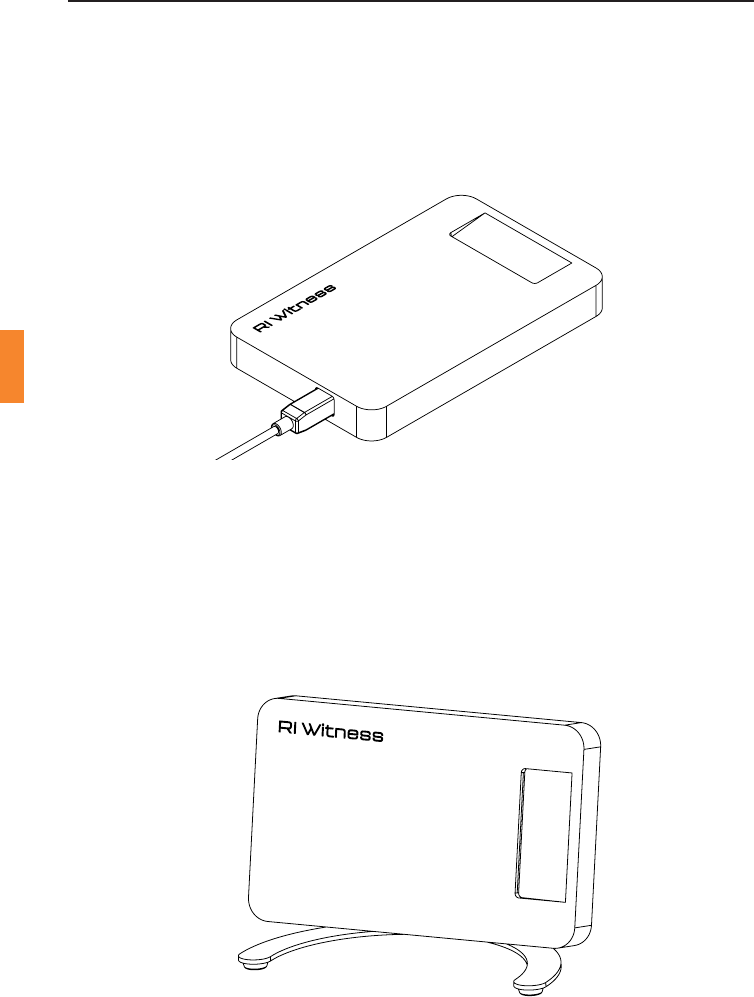
Thedevicecanbemountedinthetheatreonthewalloronthesideofaow
hood.InthisorientaonthepaentIDcardisintendedtobeinsertedinto
theslotandkepttherefortheduraonoftheprocedure.Instruconsand
allnecessaryhardwareformounngthedeviceonthewallareincludedin
the wall mount kit.
Figure 5-1 Card Reader Funcon
Secon 5
RI Witness Basic Operaon
10
Research Instruments Ltd
5
Figure 5-2 Admin Reader Funcon
Admin Reader Funcon
ThedevicecanalsobeusedintheReceponorManager’soce.Thelarge
at surface allows plenty of room to place paent ID cards and various
other tag types against the surface of the device.
Figure 5-3 IUI Funcon
IUI Funcon
TheoponalstandallowstheusertoinsertapaentIDcardintothecard
slot whilst simultaneously reading a sperm tube during an IUI procedure.
Instruconsand allnecessaryhardwareformounngthecardontothe
stand are included with the stand.

Secon 6
11
Troubleshoong
Research Instruments Ltd
6
SECTION 6 - TROUBLESHOOTING
Problem Possible Cause Soluon
Tags Reading
Intermiently
or Only in
Certain Areas
Loose
connecon
Check the USB cable is fully inserted at both
the PC and device socket. Replace cable if
necessary to ensure it is not the cable itself
at fault.
RF Noise/
interference
Many devices, especially large metallic
surfaces can cause RF Noise, interference
oraectthetuningoftheantenna.
Move the reader away from metallic
surfaces if possible. Contact an RI service
representave.
PCB faulty ContactanRIServicerepresentave
Tags Not
Reading
Broken tag Check the tag on another device
Tag not
encrypted
NavigatetotheWorkAreaSengsscreen,
then click Connected Devices, then Admin &
Card Reader, then click the down arrow next
to Tags. Non encrypted tags are shown as
Not Valid.
RF noise
Many devices, especially large metallic
surfaces can cause RF Noise, interference or
aect the tuning of the antenna. Move the
reader away from metallic surfaces if possible.
ContactanRIservicerepresentave.
Loose
connecon
Check the USB cable is fully inserted at both
the PC and device socket. Replace cable if
necessary to ensure it is not the cable itself
at fault.
WorkArea
conguraon
Ensure that the device is correctly being
idenedbythePC,Admin&CardReader
should be displayed in the connected device
display.
PCB faulty ContactanRIServicerepresentave.
Secon 7
Care and Maintenance
12
Research Instruments Ltd
7
SECTION 7- CARE AND MAINTENANCE
Cleaning
Thereadermaybecleanedwithasoclothandmilddetergent.Ensure
that no liquid is spilled down the card slot, permanent damage can be
caused.
Do not use solvents for cleaning.
Do not disconnect readers.

Secon 8
13
Repairs and Returns
Research Instruments Ltd
8
SECTION 8 - REPAIRS AND RETURNS
Reuse Statement
Assuming RI Witness is regularly maintained and rounely serviced, it
shouldperformasrequiredforaminimumof7yearsconnualuse,aer
whichmewerecommendyouconsideritsreplacement.Shouldyounoce
impaired performance and/or any issues where safety is compromised, or
have any other concerns during the use of RI Witness, seek the advice of
RIortheirauthorisedrepresentavepromptly.
RI Repairs System
In the event that you have a problem with an RI instrument, please follow
the procedure below.
1. Readthe‘Troubleshoong’secon.
2. IfyourequireanyfurtherhelpcontactyourdistributororRIdirectly.
RIwilltrytoresolvetheproblemasquicklyaspossible.
RI Returns System
1. ContactRItoobtainaReturnedMaterialsAuthorisaon(RMA)
number.
Note: Goods will not be replaced or refunded without prior
agreementandclearlystangtheRMAnumber.
2. Pack the item carefully in its original packaging. RI will not accept
responsibility for damage due to incorrect packaging. Replacement
itemsoraddionalrepairswillbeinvoiced.
3. Clearly label the package with the RMA number, mark the package
“Urgent - Returned Items For Repair”, and ship to the address on
the next page. Goods should be insured for their full value during
shipping.
Do not use solvents for cleaning.
Do not disconnect readers.
Secon 8
Repairs and Returns
14
Research Instruments Ltd
8
Product Disposal (European Union)
If the product is no longer serviceable it must be sent back to RI
to be destroyed in an environmentally safe way. Do not dispose
of RI Witness products with ‘normal’ waste.
Contact Details
Research Instruments Ltd, Bickland Industrial Park,
Falmouth, Cornwall, TR11 4TA, UK
Tel: +44 (0) 1326 372 753 Fax: +44 (0) 1326 378 783
E-mail: service@research-instruments.com
Website: www.research-instruments.com
Feedback
Thank you for purchasing an RI product. To help RI develop the best tools
forART,werelyoncustomerfeedback.Ifyouhaveanysuggesonsforhow
wecanimproveourproductsortheinformaonweprovidewiththem,
please send to feedback@research-instruments.com. Your comments
willhelpusdeveloptheproductandsupporngmaterialstomeetfuture
needs.
Thank you.