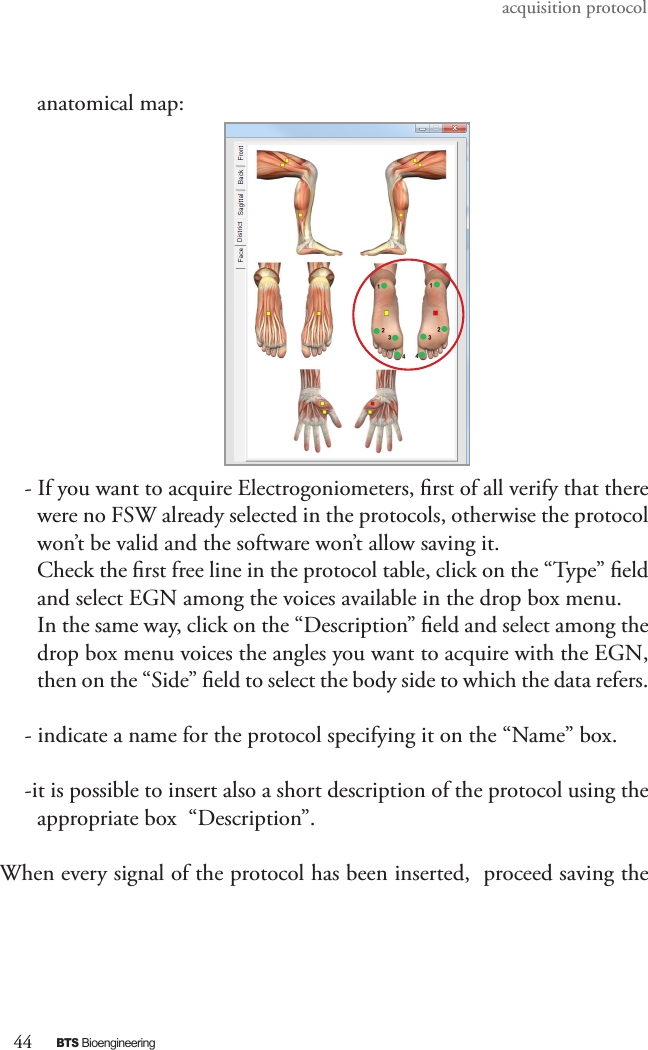
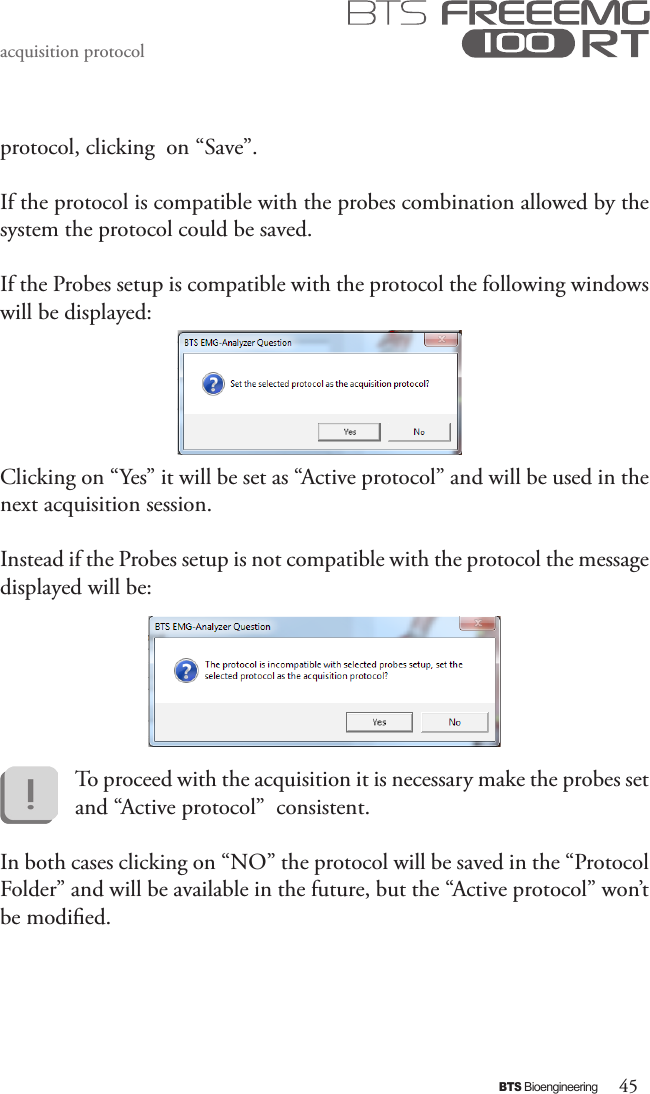
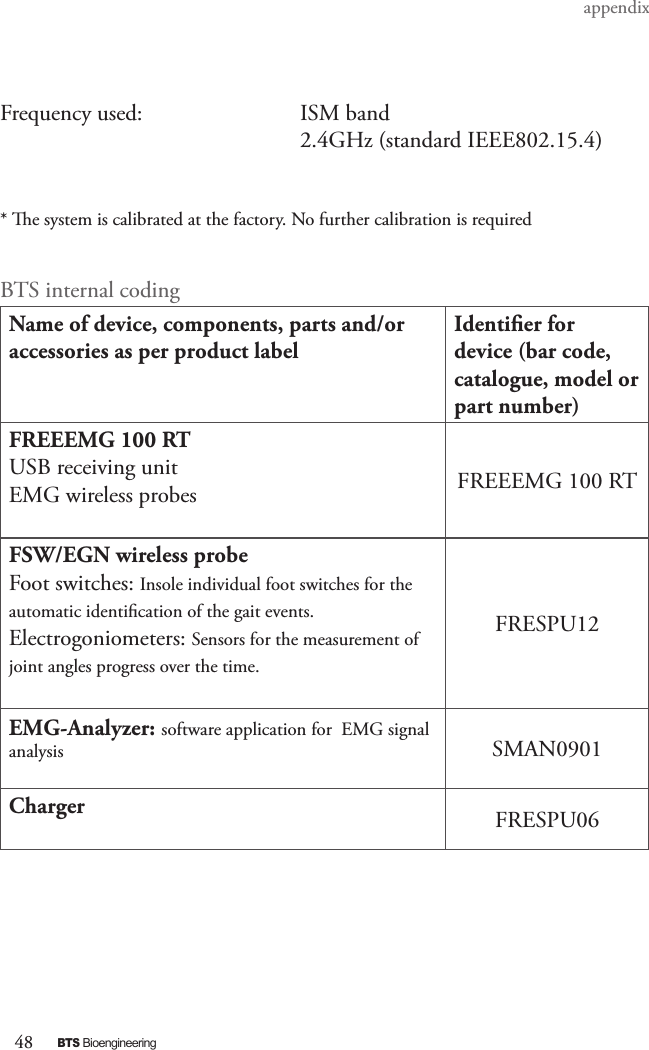
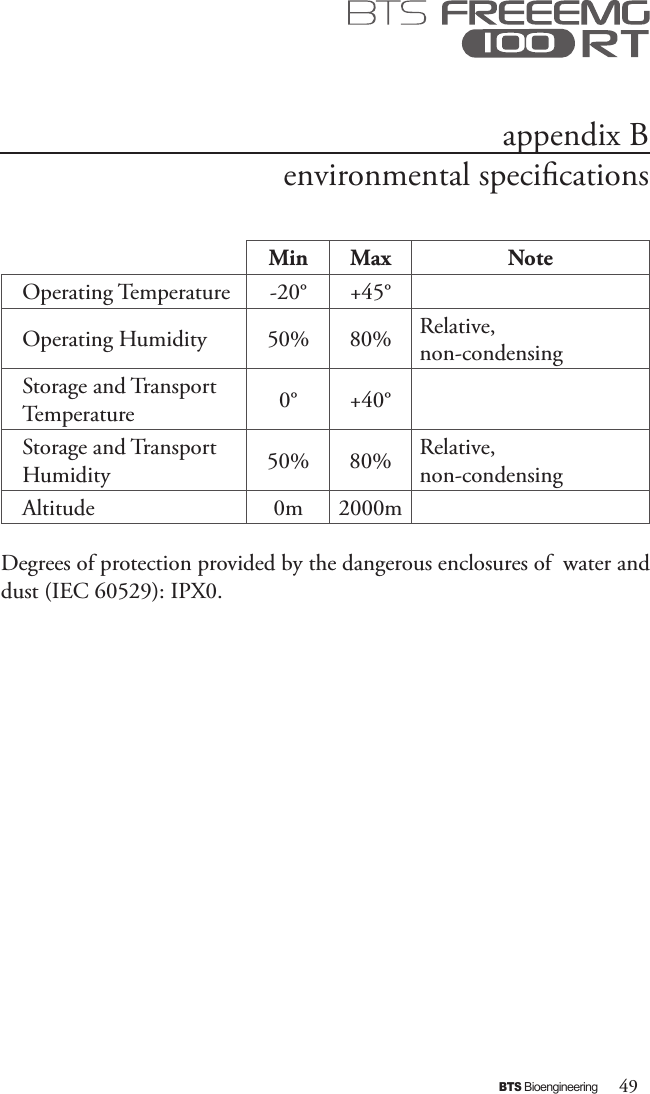
BTS BTSWEMG2 Wireless EMG sensor for EMG system User Manual EREMB 01091 06 FREEEMG 100 RT ENG v 2 0 3
BTS SpA Wireless EMG sensor for EMG system EREMB 01091 06 FREEEMG 100 RT ENG v 2 0 3
BTS >
Contents
- 1. EREMB-01091-06 FREEEMG 100 RT User Manual ENG v.2.0.3
- 2. ERFNC-00782-16 FREEEMG 300 User Manual ENG v.4.0.3
- 3. ERTMJ-00998-04 TMJOINT User Manual ENG v.3.0.0
EREMB-01091-06 FREEEMG 100 RT User Manual ENG v.2.0.3