Spacelabs Healthcare 010-0914-00 User Manual sl logo fh7
Spacelabs Healthcare, Inc. sl logo fh7
Contents
- 1. User Manual 1 of 3
- 2. User Manual 2 0f 3
- 3. User Manual 3 of 3
User Manual 2 0f 3
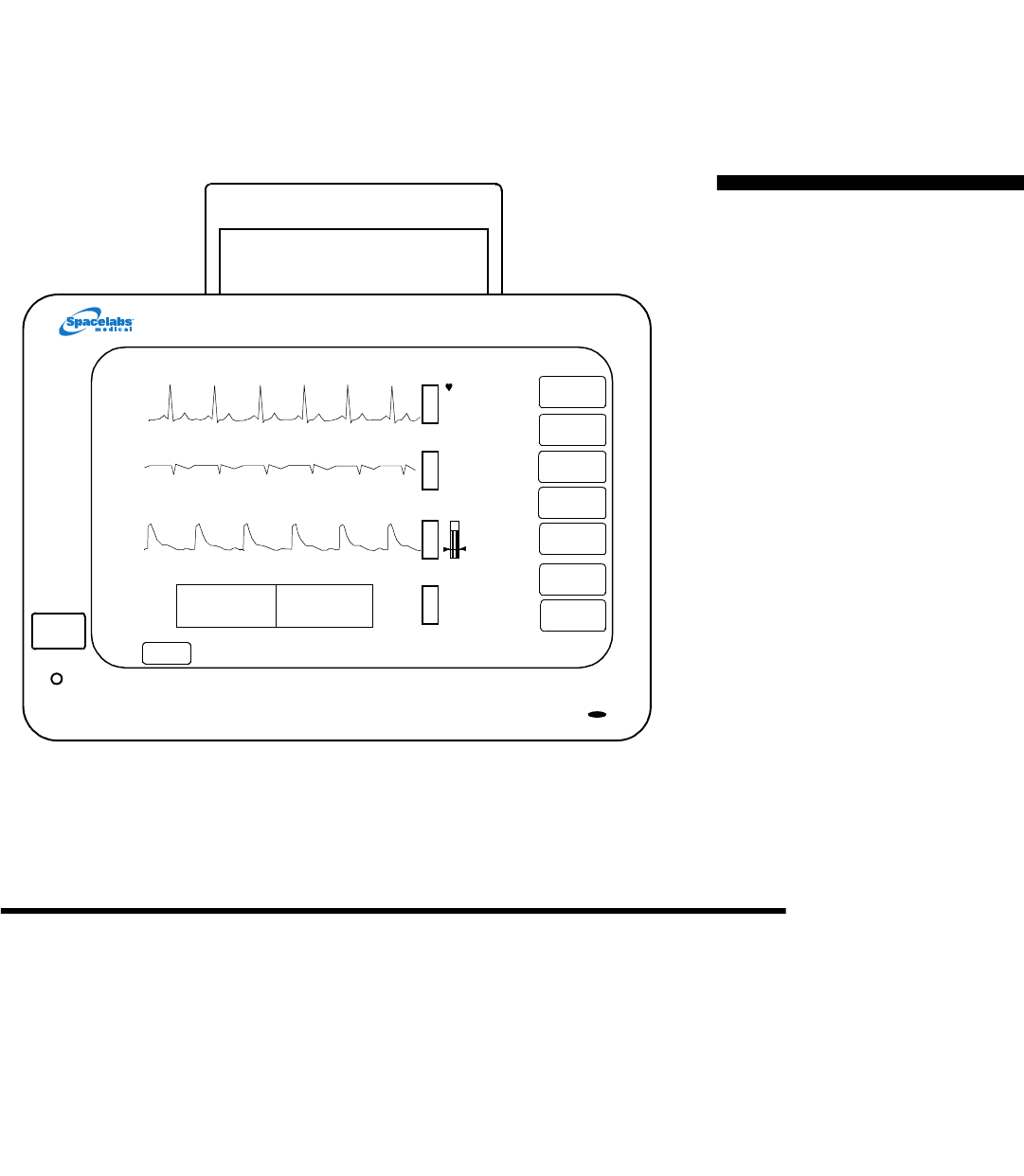
SPECIFICATIONS
Page 1of 4
Touchscreen —With the exception of power
(ON/OFF), all controls are on-screen
touchkeys; touch is sensed by infrared optical
devices; optional controls include mouse and
keyboard
Waveform Capacity —four, five, or six
waveforms
Module Capacity —90369 accepts one
Ultraview or PCMS module internally and will
support up to two additional modules using the
90499 module housing
Parameter Capacity —18 parameters utilizing
Ultraview and PCMS modules as well as
Flexport®interfaces
Trends —24 hours of trend data can be
displayed
Graphic —1-, 2-, 6-,12- or 24-hour segments;
data is stored in 1-minute resolutions (6-hour
segment is the default)
Ultraview
1050
Monitor
90369
■Lightweight, compact, highly portable
■Shares same touchscreen controls as
other Ultraview Care Network™and
PCMS™monitors of appropriate levels
■Compatible with the full line of
Ultraview Care Network and PCMS
modules
■Large, 10.4-inch (26.42 cm), TFT color
display with 140° viewing angle
■Data Shuttle®option allows up to 24
hours of patient data to be transferred
to other Ultraview Care Network and
PCMS monitors
■Advanced power management system
maximizesbatteryperformanceduring
transport; includes battery
“fuel-gauge”
■Optional interactive bed-to-bed
Ethernet communication
■Instant “Quicknet” 10BaseT Ethernet
connection
■Optional wireless Ethernet withone to
five outbound waveformsand numeric
vital signs transmitted at
2.4 GHz
■Optional built-in 2-channel recorder
■Optional mainstream EtCO2, O2,and
Min CO2
■Support for up to 250 nodes on a
network with the Expanded Network
feature
PRINTER
CONTROLS
PREVIOUS
MENU
NORMAL
SCREEN
SPECIAL
FUNCTIONS
ALM
MONITOR
SETUP
RECORD
HELP
97 100
S
P
O
2
E
C
G
85
69 bpm
(80 )
E
C
G
N
I
B
P
II MON PACED
ST =0.16
A = 0 ROW 5
120
40
VI
ST =0.00
%
116
64
69
LAST BP =
11:40
03/30
m
m
H
g150
100
S
11:32
11:33
11:34
11:35
11:36
110/ 69( 84)
113/ 73( 88)
113/ 69( 84)
82/ 51( 64)
108/ 67( 83)
11:37
11:37
11:38
11:39
11:40
110/ 73( 82)
104/ 67( 79)
114/ 73( 85)
103/ 70( 78)
116/ 64( 80)
SUSPEND
TM

SPECIFICATIONS
Ultraview
1050
Monitor
90369
Page 2of 4
Tabular —Time increments of 1, 5, 10, 15, or
30 minutes; 1, 1.5 or 3hours (1 hour is the
default)
Remote View/Alarm Watch —When equipped
with the Ethernet option, the 1050 provides a
waveform display from aremote bedside or
telemetry patient on the Ultraview network
either on request (Remote View) and/or in
response to an alarm (Alarm Watch); the 1050
provides awaveform display from up to 32
selected beds; an Ultraview bedside monitor
can be remotely viewed by up to 16 network
devices simultaneously (e.g., monitors,
workstations)
Display —
Trace Height: 1.65 in (4.2 cm)
2.36 in (6.0 cm)
Sweep Speed: Avariety of speeds are
available under module
control
Ethernet Communication —10BaseT
telephone-style modular connector (RJ45)
provided
Wireless Ethernet Capability —Provides
2.4 GHz frequency-hopping spread-spectrum
communication; the wireless Ethernet will send
one to five waveforms and numeric vital signs
to the Ultraview Care Network
Color TFT Display (TFT) —
Resolution —640 by 480 pixels
Size —8.31 in (21.12 cm) wide
6.24 in (15.84 cm) high
Software Updates —Software updates including
new features and capabilities are easily
updated over the network
Options —
F — Ethernet interface, SDLC, audio I/O, video,
alarm, serial; provides noninteractive
bed-to-bed communication
H—Capnography, Ethernet interface, SDLC,
audio I/O, video, alarm, serial
N — Vital Signs Calculations
O — Drug Dose Calculations
P—Interactive Network Functions —Adds
interactive remote view and alarm watch
capabilities for parameters displayed from
remote bedsides and remote functionality
for all trends (requires
option For H)
Q—Data Shuttle to transfer patient information
to another monitor
R — Patient Data Logger
U — Dual Channel Internal Recorder
Z — Wireless Ethernet Communication
04 — Four waveforms
05 — Five waveforms
06 — Six waveforms
ELECTRICAL SPECIFICATIONS
Mains Power —Line voltage: 100-240 VAC;
Frequency: 50-60 Hz
Batteries —Both sealed-lead acid (SLA) and
Spacelabs Medical nickel metal hydride
(NiMH) are supported. One or two 12 V(2.30
Ah SLA or 2.45 Ah NiMH) batteries may be
used, providing up to 2.0 hours of operation
(TFT display) for up to 300 charge/discharge
cycles with SLA and up to 2.5 hours of
operation (TFT display) for up to 600 charge/
discharge cycles with NiMH; operating time is
dependent on configuration and usage; 1.5
hours are required to charge batteries to 100%
of capacity with AC connected to rear panel
and mains switch in the OFF position, 3-hour
charge time required with the mains switch in
the ON position
Power Fail Backup —Will maintain patient data
for aminimum of three minutes
Isolation —Chassis leakage current not greater
than 300 µA(meets AAMI, UL2601-1,
CSA #601.1 and IEC 601-1 standards)
PHYSICAL DIMENSIONS
Height: 8.3 in (21.1 cm)
Depth: 6.2 in (15.8 cm)
Width: 11.7 in (29.7 cm)
Weight: 10.0 Ibs (4.6 kg)
DUAL CHANNEL INTERNAL RECORDER
—Option U
Printing Method —Thermal array print head
Resolution —Eight dots per mm vertical and
32 dots per mm horizontal at 25mm per
second sweep speed
Paper —Heat-sensitive paper, 50mm wide x
30m long, available in aroll
Traces —All monitored parameters, including
waveforms and graphic trends, full annotation
included
Frequency Response —Determined by the
parameter recorded
Chart Speed —1.56, 3.12, 6.25, 12.5, 25 and
50mm per second (depending on the monitor
sweep speed selected)
Alarm Record —Records any parameters in an
alarm state when “Record on Alarm” is active
Auto Run —20 seconds or duration of alarm
violation (whichever is longer)
SPECIFICATIONS Ultraview
1050
Monitor
90369
Page 3of 4
Controls —Continue, Slow, Stop, Unit Off
Indicators —Paper Out, Unit Off
Record —Allows selection of up to two active
monitor channels plus trends
ENVIRONMENTAL REQUIREMENTS
Storage —
Temperature: -13° to 140°F (-25° to 60°C)
Humidity: 95% (non-condensing)
Altitude: 0 to 40,000 ft (0 to 12,192 m)
Operating —
Temperature: 32° to 122°F (0° to 50°C)
Humidity: 95% (non-condensing)
Altitude: 0 to 15,000 ft (0 to 4,572 m)
ELECTROMAGNETIC COMPATIBILITY
EN60601-1-2, 1993-04
Emissions — (CISPR 11) EN55011, Class B
EN61000-3-2: Harmonics
EN6100-3-3: Flicker
Mil-Std-461D: RE101
Immunity —
IEC 1000-4-2: ESD, 8kV contact/15 kV air
IEC 1000-4-3: RF Fields, 20 volts/meter,
26 MHz to 1GHz
IEC 1000-4-4: Burst, 1 kV data and
I/O ports/2 kV power ports
IEC 1000-4-5: Surge, 1kV differential/
2kV common mode
IEC 1000-4-6: Conducted RF, 3volt r.m.s.
150 kHz to 80 MHz
IEC1000-4-8: Magnetic Field (50/60 Hz),
60 Amps/meter
IEC 1000-4-11: Power quality, voltage and
frequency variations
Mil-Std 461D: CS101, 12 volt r.m.s.
10 kHz to 150 kHz
Mil-Std 461D: CS114, 120 dBµA
10 kHz to 400 MHz
REGULATORY APPROVALS
ETL listed and meets standard UL2601-1 for
electrical safety; approved by CSA; CE
marked in accordance with the Medical Device
Directive, 93/42/EEC
ACCESSORIES
119-0251-01 100-240 volt AC converter
Note: This AC converter is
mandatory and is included at
no charge
146-0018-00 Rechargeable sealed-lead
acid (SLA) battery
146-0055-00 Rechargeable nickel metal
hydride (NiMH) battery
Note: NiMH batteries require
units with a minimum serial
number of 369-1XXXXX. SLA
batteries may be used with all
serial number units.
010-1114-00 External sealed-lead acid
battery charger (not
compatible with NiMH battery
packs); recharges 1to 4
removable SLA batteries;
100-240 V
016-0347-00 Wall mount
016-0369-00 Bed rail mount (for 90369
without capnography option)
016-0369-01 Bed rail mount (for 90369 with
capnography option)
016-0447-00 Roll Stand
040-0992-00 Conversion Kit 10BaseT to
AUI
010-0609-00 Mouse
For information about required supplies,
please refer to the Spacelabs Medical
Supplies Products Catalog

Ultraview
1050
Monitor
90369
Spacelabs Medical, Inc.
15220 N.E. 40th Street
P.O . Box 97013
Redmond, WA 98073-9713
Telephone: (425) 882-3700
Fax: (425) 885-4877
Telex: 4740085 SPL UI
Ultraview, Ultraview Care
Network, PCMS, Data Shuttle
and Flexport are trademarks of
Spacelabs Medical, Inc.
Other brands and product
names are trademarks of their
respective owners.
All specifications are subject to
change without notice.
www.spacelabs.com
©Spacelabs Medical, Inc. 2001
061-0750-00 Rev. M05/2001
Page 4of 4